Abstract
Somatosensory neurons provide the nervous system with information about mechanical forces originating inside and outside the body. Here, we use connectomics to reconstruct and analyze neural circuits downstream of the largest somatosensory organ in the Drosophila leg, the femoral chordotonal organ (FeCO). The FeCO has been proposed to support both proprioceptive sensing of the fly’s femur-tibia joint and exteroceptive sensing of substrate vibrations, but it remains unknown which sensory neurons and central circuits contribute to each of these functions. We found that different subtypes of FeCO sensory neurons feed into distinct proprioceptive and exteroceptive pathways. Position- and movement-encoding FeCO neurons connect to local leg motor control circuits in the ventral nerve cord (VNC), indicating a proprioceptive function. In contrast, signals from the vibration-encoding FeCO neurons are integrated across legs and transmitted to auditory regions in the brain, indicating an exteroceptive function. Overall, our analyses reveal the structure of specialized circuits for processing proprioceptive and exteroceptive signals from the fly leg. They also demonstrate how analyzing patterns of synaptic connectivity can distill organizing principles from complex sensorimotor circuits.
Introduction
To coordinate complex behaviors, circuits in the central nervous system (CNS) require continuous information about the body and the environment. An important source of feedback are somatosensory neurons, which provide the nervous system with information about mechanical forces acting on an animal’s body1,2. Somatosensory neurons are typically described as either exteroceptive, detecting mechanical forces generated in the external world, or proprioceptive, detecting the position or movement of body parts. However, because they are embedded within the body, many somatosensory neurons can detect both externally- and self-generated forces, making it difficult to determine whether specific somatosensory neurons are exteroceptive, proprioceptive, or both. Recording from primary somatosensory neurons in behaving animals can resolve the types of mechanical stimuli they encode3, but such experiments are technically difficult and often not feasible. An alternative approach is to map the connectivity of sensory neurons with downstream circuits, which can provide clues to about putative function. For example, some proprioceptor axons synapse directly onto motor neurons to generate rapid reflexes4 (Figure 1A). Somatosensory signals can also be integrated with other sensory cues and internal states to inform action selection in response to external perturbations5,6.
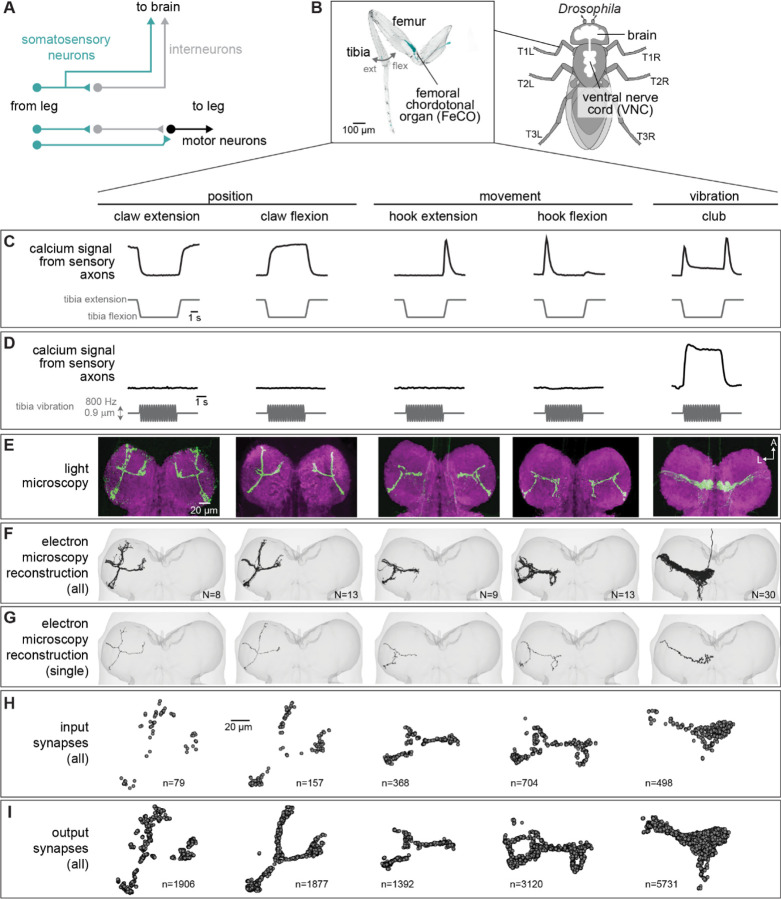
Figure 1. Connectomic reconstruction of axonal projections from somatosensory neurons in the femoral chordotonal organ (FeCO) of a female Drosophila.
(A) Schematic of local and ascending VNC circuits for leg somatosensation and motor control. (B) Left: Confocal image of a Drosophila front leg showing the location of FeCO cell bodies and dendrites. Green: GFP; gray: cuticle auto-fluorescence. Right: Schematic showing the fly brain and ventral nerve cord (VNC) (C-I) Anatomical and functional subtypes of somatosensory neurons in the Drosophila FeCO. (C) Calcium signals from FeCO axons of each subtype (GCaMP, black traces) in response to a controlled movement of the femur-tibia joint (gray traces). Adapted from Mamiya et al., (2023). (D) Calcium signals from FeCO axons of each subtype (GCaMP, black traces) in response to an 800 Hz vibration of the femur-tibia joint (gray traces). Adapted from Mamiya et al., (2023). (E) Confocal images of the axons of each FeCO subtype in the fly ventral nerve cord (VNC). Green: GFP; magenta: neuropil stain (nc82). Adapted from Agrawal et al., (2020) A: anterior; L: lateral. (F) Reconstructed FeCO axons from each subtype in the front left leg neuromere of the FANC connectome (from left to right, N=8, 13, 9, 13, 35 neurons). (G) Single reconstructed axons from each FeCO subtype in the front left leg neuromere of the FANC connectome. (H) Locations of all input synapses received by each FeCO subtype (i.e. postsynaptic sites). n indicates the number of synapses. (I) Locations of all output synapses made by each FeCO subtype (i.e. presynaptic sites). n indicates the number of synapses.
Mapping the flow of sensory signals into the nervous system has recently become feasible in small organisms thanks to advances in serial-section electron microscopy (EM) and computational image segmentation, which enable the reconstruction of whole synaptic wiring diagrams, or connectomes. Some of the most comprehensive connectomes reconstructed to date include the brain and ventral nerve cord (VNC) of the adult fruit fly, Drosophila melanogaster7–11. The fly brain connectomes have already revealed new insight into the organization and function of sensory organs on the head. For example, mapping the projections of mechanosensory neurons from the fly’s antenna into the brain revealed the organization of circuits that support song detection, antennal grooming, and escape12,13. Volumetric EM datasets of the fly VNC8,9, which is analogous to the vertebrate spinal cord, now make it possible to reconstruct and analyze the function of somatosensory signals from other parts of the fly’s body, including the legs and wings.
In this study, we use connectomic analyses of brain and VNC circuits to investigate the largest somatosensory organ in the Drosophila leg, the femoral chordotonal organ (FeCO) (Figure 1B). The Drosophila FeCO is comprised of ~150 excitatory (cholinergic) sensory neurons that can be separated into five functionally and anatomically distinct subtypes: (1) extension- and (2) flexion-encoding claw neurons encode tibia position, (3) extension- and (4) flexion-encoding hook neurons sense tibia movement, and (5) club neurons encode bidirectional tibia movement and low-amplitude (<1 µm), high frequency vibration (Figure 1C–D)14,15. The cell bodies and dendrites of the FeCO are located in the femur of each leg and their axons project into the VNC (Figure 1B)14–17.
The FeCO is typically described as a proprioceptive organ that monitors the movement and position of the femur-tibia joint15,18,19. However, behavioral evidence suggests that the FeCO may also detect externally-generated substrate vibrations, perhaps to aid in social communication, predator detection, and courtship20–23. It is currently unknown to what degree the five subtypes of FeCO sensory neurons are specialized to support specific proprioceptive or exteroceptive functions. The club neurons are the only FeCO subtype that respond to tibia vibration (Figure 1D), suggesting that they may support exteroceptive vibration sensing14,15. However, club neurons also respond to larger movements of the tibia like those that occur during walking, suggesting that they could also be proprioceptive (Figure 1C). Intracellular recordings from second-order neurons have identified distinct pathways for proprioceptive and vibration sensing, but in some cases also revealed complex pooling of signals from multiple FeCO subtypes24,25.
Here, we use the FANC26 and Flywire7 connectome datasets to reconstruct and analyze neural circuits downstream of the FeCO of the fly’s front left (T1L) leg. We find that position- and movement-encoding claw and hook neurons are primarily proprioceptive and connect to local circuits within the VNC for leg motor control. In contrast, vibration-encoding club neurons connect to intersegmental and ascending circuits that integrate mechanosensory information from the legs, wings, and neck, and relay it to the brain. Then, by identifying these ascending projections within the brain, we confirm that this leg vibration information is relayed to auditory circuits downstream of the antenna. We also identify sparse pathways that mediate interactions between proprioceptive and exteroceptive circuits, revealing how vibration signals may directly influence motor output. Overall, our analyses suggest that the FeCO supports both proprioceptive and exteroceptive functions, which are achieved via specialized somatosensory neurons connected to specialized downstream circuits.
Results
Reconstruction and identification of FeCO axons in the FANC connectome
Using software for collaborative proofreading and visualization of the segmented FANC EM dataset (see Methods), we reconstructed the anatomy and synaptic connectivity of roughly half the FeCO axons from the front left leg (78 total axons, Figure 1F–I). We focused our reconstruction efforts on these FeCO axons because they project to the front left neuromere of the VNC (also referred to as left T1 or T1L), the region of the Drosophila VNC with the most complete information about leg sensorimotor circuits. All of the motor neurons controlling the muscles of the front left leg and their presynaptic partners have been previously identified and reconstructed in FANC26,27, and prior neurophysiological recordings of FeCO axons and their downstream targets were made in the same region14,15,24,25,28,29. Unfortunately, leg sensory axons are among the most difficult neurons to reconstruct in the available VNC connectomes, likely due to rapid cell-death that begins when the legs are separated from the VNC during sample preparation. Although our dataset is missing some FeCO axons, we found that the number of novel postsynaptic partners decreased as we added more axons to the dataset (Figure S1), suggesting that our reconstruction covers the major components of the postsynaptic circuitry.
The FeCO consists of five functional subtypes that encode tibia position (extension/flexion), directional movement (extension/flexion), and bidirectional movement/vibration (Figure 1C–D)19. We sorted the reconstructed FeCO axons in the connectome into these functional subtypes based on axon morphology and comparison with light microscopy images (Figure 1E–G; see Methods). Based on an X-ray reconstruction of the peripheral cell bodies14, we estimate that we reconstructed ~50% of the T1L axons of each subtype: 8 claw extension, 13 claw flexion, and 9 hook extension axons (of ~58 axons), 13 hook flexion axons (of ~28 axons), and 35 club axons (of ~66 axons). Overall, EM reconstructions of axons from each subtype resemble light-level images of FeCO axons15, including 5 club axons from the T1L leg that send an ascending projection to the brain. A few FeCO axons, however, demonstrate unexpected within-type diversity, including axons with shortened or doubled branches (Supplemental Table 1). As expected for sensory neurons, all FeCO axons have more presynaptic sites (i.e., output synapses) than postsynaptic sites (i.e. input synapses) (Figure 1H–I). Generally, the locations of pre- and postsynaptic sites are intermingled; FeCO axons do not have distinct pre- and postsynaptic zones.
Claw and hook (but not club) axons provide feedback to local leg motor circuits
To investigate pathways downstream of the different FeCO subtypes, we reconstructed the anatomy and synaptic connectivity of all postsynaptic partners that receive at least 4 synapses from a FeCO axon, a threshold used by previous studies7,30,31. We classified all postsynaptic VNC neurons into six morphological classes (see Methods): local neurons located entirely in the T1L neuromere: (1) interneurons, (2) motor neurons, (3) sensory neurons, (4) descending and (5) ascending neurons that connect the brain and VNC, and (6) intersegmental neurons, which span multiple VNC neuromeres (Figure 2A). We interpret connectivity of FeCO axons with local interneurons or leg motor neurons as suggesting a role in local, rapid feedback control of leg motor output. In contrast, we interpret connectivity with ascending neurons as suggesting a role in mediating sensation and behavior on longer timescales, such as sensory perception and action selection.
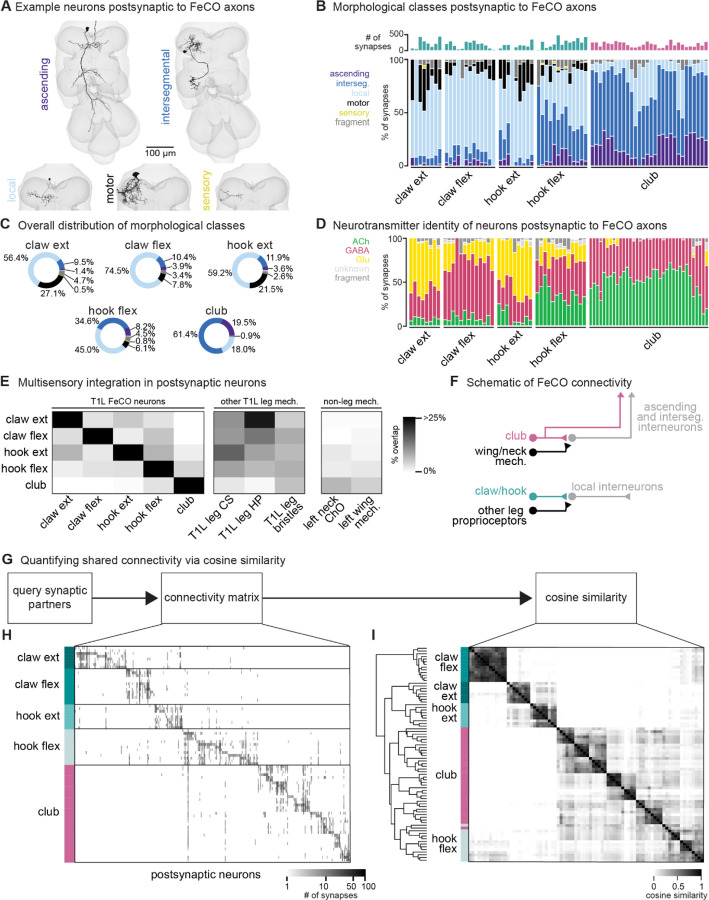
Figure 2. FeCO neurons exhibit subtype-specific postsynaptic connectivity.
(A) We reconstructed all VNC neurons postsynaptic to FeCO axons from the front left leg (T1L) and classified them into morphological classes. Example provided from each class. (B) Percent of synapses from each FeCO axon that are made onto VNC neurons of each morphological class. Top bar plot shows the total number of output synapses made by each FeCO axon. (C) Per FeCO subtype, the total fraction of output synapses made onto each morphological class. (D) Proportion of total synapses made by each FeCO neuron onto cholinergic (green), glutamatergic (yellow), GABAergic (pink), and unidentified (light gray) hemilineages. (E) Heatmap shows the percent of neurons postsynaptic to a particular T1L FeCO subtype (as indicated along the rows) that also receive synaptic input from an alternate somatosensory population: T1L FeCO neurons, including claw extension axons, claw flexion axons, hook extension axons, hook flexion axons, or club axons, other T1L leg mechanosensory neurons, including campaniform sensilla axons (CS), hairplate axons (HP), or bristle axons, and non-leg mechanosensory neurons, including left neck chordotonal organ axons or left wing somatosensory axons. We found that neurons postsynaptic to claw and hook axons also integrate information from other leg proprioceptors such as HP axons and CS axons. In contrast, neurons postsynaptic to club axons do not integrate information from other leg proprioceptors, but they do integrate information from wing and neck somatosensory axons. (F) Schematic of FeCO connectivity. Club information is conveyed primarily to ascending and intersegmental neurons, who also receive information from wing and neck somatosensory neurons. Information from claw and hook axons is primarily relayed to local interneurons, which also receive information from other leg proprioceptive neurons. (G) By querying the connectivity of each postsynaptic partner of each reconstructed FeCO neuron, we obtained H) a connectivity matrix and I) a cosine similarity matrix. (H) Connectivity matrix between FeCO axons and postsynaptic VNC neurons. The shading of each tick indicates the number of synapses from each FeCO axon (row) onto each postsynaptic VNC neuron (column). Colored bars along the left indicate the presynaptic FeCO subtype for that row. FeCO axons are organized by morphological subtype and then by their cosine similarity scores. VNC neurons are organized by their cosine similarity scores. (I) Clustered pairwise cosine similarity matrices of all FeCO axons based on their postsynaptic connectivity. The cosine similarity between two neurons is the dot product of the normalized (unit) column weight vectors. If two FeCO neurons synapse with similar synaptic weights onto the same postsynaptic neuron, relative to the FeCO’s total output, the pairwise cosine similarity is 1. FeCO neurons with similar postsynaptic connectivity patterns cluster together, forming connectivity clusters.
We found that the majority of synapses from claw and hook extension axons are onto local VNC interneurons and leg motor neurons (Figure 2B–C). In contrast, more than half of all synapses from club axons are onto intersegmental neurons. Hook flexion axons are somewhere in between, making roughly similar proportions of their synapses onto local and intersegmental postsynaptic partners. Club and hook flexion axons also make a notably high number of synapses onto ascending neurons that convey leg somatosensory information to the brain.
We next used anatomical criteria to identify the developmental origins of all pre- and postsynaptic partners of FeCO axons (see Methods). About 95% of adult neurons in the Drosophila VNC arise from 30 segmentally repeated neuroblasts (neural stem cells), each of which divides to form an ‘A’ and ‘B’ hemilineage32. Developmental hemilineages are an effective means to classify VNC cell types: neurons of the same hemilineage release the same primary neurotransmitter33 and express similar transcription factors34,35. Previous research also suggests that neurons within a hemilineage are functionally related: thermogenetic activation of single hemilineages drove coordinated movements of legs or wings36, and connectome analyses of larval VNC neurons demonstrated that neurons within a hemilineage share common synaptic partners37.
We found that club axons target neurons from different hemilineages than claw and hook axons (Figure S2). Neurons that are primarily postsynaptic to club axons come from hemilineages 8B, 10B, 23B, 0A/0B, 1B, and 9A. Of those, only 1B and 9A neurons receive any synaptic input from claw axons, but the connectivity is weak (~6.34% and 0.46% of total FeCO input, respectively). Club and hook flexion axons target some shared hemilineages, including 23B, 1B, and 9A. Neurons from all other identified hemilineages are predominantly postsynaptic to claw or hook axons and do not receive any synaptic input from club axons. We further used the hemilineage designations to infer a neuron’s likely primary neurotransmitter (Figure 2D). The majority of Drosophila neurons release one of three primary neurotransmitters: acetylcholine, GABA, or glutamate34,38. In the fly, acetylcholine is typically excitatory, while GABA is typically inhibitory34,39,40. Glutamate is excitatory at the fly neuromuscular junction, acting on ionotropic glutamate receptors (GluRs), but is frequently inhibitory in the CNS, acting on the glutamate-gated chloride channel, GluCl41. Club axons synapse onto very few putative glutamatergic neurons (Figure 2D) compared to claw and hook axons.
We conducted similar analyses examining the presynaptic inputs to FeCO axons (Figure S3). Generally, hook axons receive the most input synapses and have the most presynaptic partners, which include local, ascending, and intersegmental neurons (Figure S3A–B). The majority of input synapses to FeCO axons are GABAergic (Figure S3D). The strongest input comes from 9A neurons, which are primarily presynaptic to hook axons (Figure S3C–D). Recent work found that a subset of 9A neurons suppress expected proprioceptive feedback during voluntary movement such as walking or grooming29.
Together, these differences in postsynaptic connectivity suggest that claw and hook axons are connected to postsynaptic partners that are distinct from those downstream of club axons. These downstream partners differ in their morphology as well as their developmental stem-cell lineage. Hook and claw axon connectivity with local and motor neurons suggests that they play a role in fast feedback control of leg motor output. In contrast, club axons connect to intersegmental and ascending pathways that could relay leg vibration information to the brain to support detection of external mechanosensory signals. (Figure 2F). In support of this conclusion, we found that the neurons that receive input from claw and hook axons also receive input from other leg proprioceptors, such as hair plate and campaniform sensilla neurons, whereas the VNC neurons that receive input from club axons receive input from somatosensory neurons on the neck and wing, not on the leg (Figure 2E–F).
FeCO axons demonstrate subtype-specific downstream connectivity
We next investigated the specific postsynaptic partners targeted by claw, hook, and club axons and the degree to which FeCO axons synapse onto distinct or overlapping circuits. First, we constructed a connectivity matrix to look at the postsynaptic connectivity of each FeCO neuron, organizing the rows of the matrix by FeCO subtype (Figure 2G–H). Generally, postsynaptic connectivity is sparse, with each FeCO neuron contacting only about 21.1 ± 1.1 (mean ± s.e.m.) distinct postsynaptic partners. To quantify this connectivity structure, we calculated the cosine similarity score for pairs of FeCO axons based on their synaptic outputs (Figure 2I; see Methods). Two FeCO axons have a high cosine similarity score if they make the same relative number of synapses onto the same postsynaptic neurons. Low similarity scores indicate either that two FeCO axons share few postsynaptic partners or that the relative number of synapses onto common postsynaptic partners are different.
Hierarchical clustering of cosine similarity scores confirmed that FeCO axons of the same subtype provide similar synaptic output to the same postsynaptic partners (Figure 2I). FeCO axons tuned to different tibia positions (claw flexion vs. claw extension axons) or movement directions (hook flexion vs. hook extension axons) demonstrate very low (almost zero) cosine similarity scores, indicating that their postsynaptic connectivity is very different. Instead, hook and claw axons that share directional selectivity (claw and hook flexion or claw and hook extension axons) demonstrate some shared connectivity, as suggested by cosine similarity scores above zero. Unexpectedly, we found that hook flexion axons and club axons share some postsynaptic connectivity, as demonstrated by their relatively high cosine similarity scores and co-clustering. For example, one specific VNC interneuron received synaptic input from almost all club and hook flexion axons. We also calculated and clustered the similarity scores for FeCO axons based on their synaptic inputs (Figure S3E–F). However, because FeCO axons have far fewer (and in some cases zero) presynaptic partners (2.9 ± 0.3 neurons, mean ± s.e.m.), these similarity scores are dominated by the shared connectivity of just a few presynaptic neurons. Claw extension and claw flexion axons receive little shared synaptic input. In contrast, hook flexion and hook extension axons all receive very similar synaptic input. Only a small number of club axons receive presynaptic input, but those that do exhibit high similarity to one another, except for two club axons whose upstream connectivity is more similar to that of hook axons.
In summary, FeCO axons demonstrate subtype-specific pre- and postsynaptic connectivity. FeCO axons within a subtype are generally more similar in their connectivity than FeCO axons of different subtypes, suggesting that information from each subtype is conveyed in parallel to different downstream neurons.
Claw and hook axons connect directly and indirectly to leg motor neurons
Thus far, we have found that club axons synapse on VNC neurons from different morphological classes and developmental hemilineages than the claw and hook axons. This segregated connectivity suggests that signals from club neurons are relayed to distinct downstream circuits with different functions than claw and hook neurons. Given that club neurons are the only subtype that respond to low amplitude, high frequency vibration, we hypothesized that this distinct connectivity could reflect an exteroceptive function of club neurons compared to the proprioceptive function of claw and hook neurons. To explore this hypothesis, we next examined how each FeCO subtype connects to leg motor circuits.
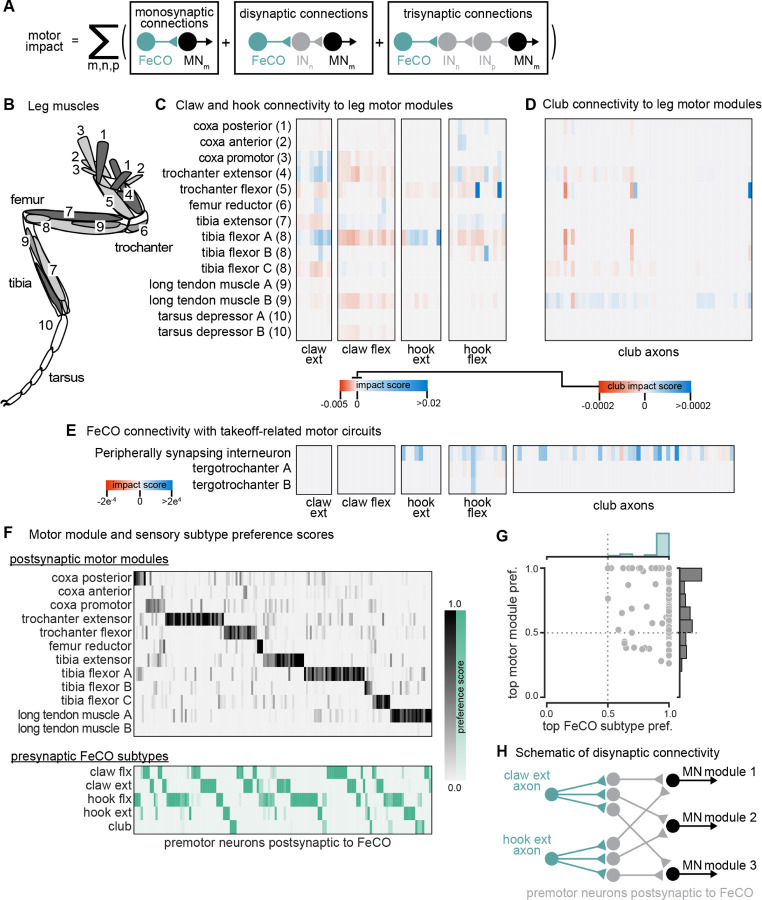
Some FeCO axons synapse directly onto motor neurons, but they also indirectly excite or inhibit motor neurons via intervening interneurons. We developed an impact score metric (Figure 3A) that takes into account both direct and indirect connections between FeCO axons and motor neurons, as well as the putative neurotransmitter predicted by hemilineage assignment (see Methods). We then calculated the motor impact score between each FeCO axon and functional pools of leg motor neurons, called motor modules27 (Figure 3B–C). Motor modules contain varying numbers of motor neurons that, based on their presynaptic connectivity patterns, comprise a functional motor pool driving a similar movement (e.g., tibia extension).
Figure 3. The connectivity of hook and claw neurons is structured to impact activity of leg motor neurons.
(A) We developed an impact score that takes into account monosynaptic, disynaptic, and trisynaptic connections between FeCO axons and leg motor neurons (see methods). (B) Schematic of the 18 muscles controlling the fly’s front leg. Numbers correspond to motor module labels in panel C. (C) Motor impact scores of claw and hook axons on leg motor modules. Motor modules are functional groupings of motor neurons that receive common synaptic input and act on the same joint27. The target muscles of each motor module are indicated in panel B. (D) Motor impact scores of club axons on leg motor modules. Note the scale bar change from panel C. (E) Motor impact scores of FeCO axons onto take-off related motor circuits. The peripherally synapsing interneuron is a premotor neuron involved in takeoff. The tergotrochanter is a leg muscle that is not active during walking but is instead involved in jumping and takeoff 26,43,44. (F) Motor module preference scores (gray, top) and FeCO subtype preference scores (green, bottom) for each premotor VNC neuron that receives input from FeCO axons (columns). Premotor neurons are arranged according to their preferred motor module followed by their preferred FeCO subtype. (G) Motor module preference (y-axis) plotted against FeCO subtype preference (x-axis) for each premotor VNC neuron that receives input from FeCO axons. (H) Schematic representation of the predominant connectivity pattern seen between FeCO neurons and motor modules. Premotor neurons postsynaptic to the FeCO are primarily dedicated to relaying information from a particular FeCO subtype to a particular motor module.
Claw and hook neurons make direct and indirect connections with many leg motor neurons (Figure 3C). The pattern of their connectivity is consistent with previous recordings of motor neuron activity and optogenetic manipulations in Drosophila24,28. Claw and hook flexion axons provide strong excitatory feedback to motor neurons that extend the tibia and inhibitory feedback to motor neurons that flex the tibia. Claw and hook extension axons provide excitatory feedback to motor neurons that flex the tibia and strong inhibitory feedback to motor neurons that extend the tibia. Claw extension axons also provide excitatory feedback to other motor modules, such as the motor neurons that move the coxa forward (coxa promotor) and extend the trochanter. This connectivity suggests that FeCO feedback supports leg motor synergies that span multiple leg joints.
Consistent with our hypothesis that club neurons do not support local leg motor control, club axon connectivity with leg motor neurons is weak, demonstrated by a low impact score (Figure 3D, note different scale bar). Club axons form no direct synapses onto leg motor neurons. However, they do indirectly and weakly connect to leg motor neurons innervating the long tendon muscle (LTM) (Figure 3D), which has been shown to control substrate grip42. Club axons also indirectly connect to the premotor peripherally synapsing interneuron (PSI) (Figure 3E), which is associated with control of the wing during takeoff26,43,44. This connectivity suggests a pathway by which activation of club neurons could lead to startle or escape behaviors, such as freezing and take-off.
Finally, we analyzed the overall organization of FeCO axons’ premotor connectivity. We found that all postsynaptic premotor interneurons have a strong preference for a single FeCO sensory subtype and a single MN module (Figure 3F–G). This organization suggests that fly leg motor circuits are organized according to a “labeled line” structure, with dedicated interneurons connecting a single FeCO subtype with a single motor module (Figure 3H).
Club connectivity is consistent with a putative tonotopic map of tibia vibration frequency
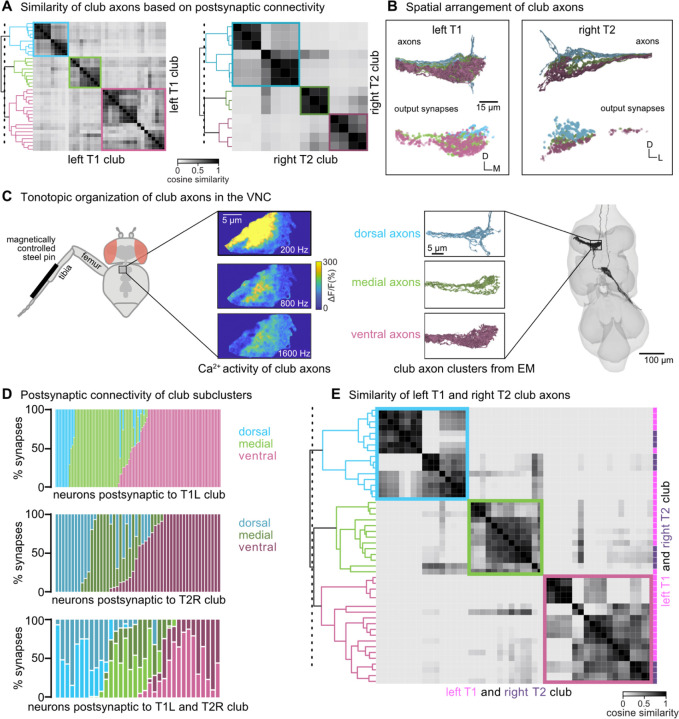
We next sought to understand how club neurons are functionally organized by further investigating their anatomical projection patterns and postsynaptic connectivity. Among the five subtypes, club axons stood out as having the most variable postsynaptic connectivity patterns. Specifically, club axons separated into subclusters that were more similar to one another than other club axons (Figure 2I). Past recordings of calcium activity from FeCO neurons in response to vibration of the tibia revealed that club axons are organized tonotopically14,15. We therefore wondered whether the connectivity clusters we found in the VNC connectome could represent functional groupings of club axons tuned to similar vibration frequencies.
In support of this hypothesis, we found that the spatial organization of the connectivity subclusters of club axons reflects the tonotopy observed in prior experimental recordings. Club axons cluster into 3 groups based on the cosine similarity of their postsynaptic connectivity (Figure 4A). These connectivity clusters spatially tile the dorsal-ventral axis (Figure 4B), similar to the tonotopic organization seen in calcium imaging of club axons in response to tibia vibration (Figure 4C)15. Accordingly, we hypothesize that the most ventral populations are sensitive to lower frequency vibrations, whereas the most dorsal populations (including ascending club axons) are sensitive to higher frequency vibrations. To determine whether this spatial organization is replicated in other leg neuromeres, we reconstructed 14 club axons from the middle right leg (right T2). Club axons from this leg also separate into three spatially distinct subclusters that span the dorsal-ventral axis (Figure 4A–B). We also found that VNC neurons postsynaptic to club axons receive most of their input from club axons from the same connectivity cluster (dorsal, medial, or ventral) across multiple legs (Figure 4D). Club axons in the most dorsal clusters of the left T1 and right T2 legs connect to overlapping downstream partners regardless of their leg of origin. Similarly, axons in the ventral-most cluster connect to overlapping downstream partners. Thus, irrespective of the leg of origin, club axons cluster based on their dorsal-ventral organization (Figure 4E).
Figure 4. Club neurons cluster into spatial groups that reflect the putative tonotopic map of tibia vibration frequency.
(A) Clustered pairwise cosine similarity matrices of the club axons based on postsynaptic connectivity. Sensory neurons with similar postsynaptic connectivity patterns cluster together, forming connectivity clusters. Left: Matrices for club axons in front left leg with connectivity clusters highlighted in blue (n = 10), green (n = 9), pink (n = 18). Right: Matrices for club axons in the middle right leg (T2R) with connectivity clusters highlighted in blue (n = 7), green (n = 3), pink (n = 4). (B) Top, Club axons within each connectivity cluster form 3 groups that span the dorsal-ventral axis: dorsal (blue), medial (green), and ventral (pink). Bottom, the spatial location of the output synapses for each of the club neurons color-coded by the corresponding connectivity cluster. (C) The dorsal-ventral organization of connectivity clusters is consistent with tonotopic mapping of tibia vibration frequency recorded from club axons with calcium imaging15. Left, Schematic of the experimental set-up. Calcium data from Mamiya et al., (2018) depicting calcium responses from club axons to vibration frequencies (200 Hz, 800 Hz, 1600 Hz) applied to the tibia. Right, reconstructed club axons in the FANC dataset separated by connectivity clusters. (D) Fraction of input synapses from club neurons onto downstream partners. Club neurons are grouped based on connectivity cluster (dorsal: blue, medial: green, ventral: pink). (E) Clustered pairwise similarity matrices of the T1L (pink) and T2R (gray) club axons based on shared postsynaptic connectivity. Sensory neurons with similar postsynaptic connectivity patterns cluster together regardless of their leg of origin.
In summary, we found that club axons form subclusters that tile the dorsal-ventral axis and share postsynaptic partners. We propose that individual club neurons are spatially clustered along the dorsal-ventral axis based on similarities in vibration frequency tuning. The putative tonotopic structure observed in club axons is preserved in postsynaptic neurons.
Interneurons postsynaptic to club axons integrate information across legs
Club axons primarily synapse onto ascending and intersegmental interneurons (Figure 2). These downstream neurons are positioned to integrate vibration information across multiple legs, perhaps to enable the fly to detect and localize external vibrations (Figure 5A). We next investigated the structure of this putative downstream integration by analyzing the circuitry postsynaptic to club axons. In addition to reconstructing the interneurons directly downstream of club neurons, we also reconstructed a subset of third order interneurons.
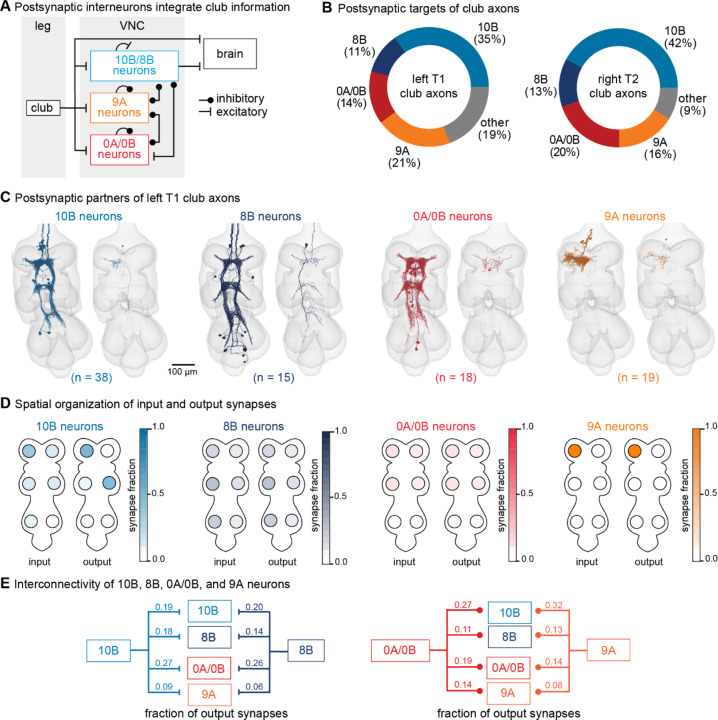
Figure 5. VNC neurons integrate vibration signals from club neurons across multiple legs.
(A) Schematic of the multi-layered connectivity downstream of club axons. (B) Fraction of total output synapses from club neurons onto downstream interneurons separated by hemilineage class. 10B (light blue, n = 38), 8B (dark blue, n = 15), 0A/0B (red, n = 18). 9A (orange, n = 19) (C) Reconstructed interneurons downstream of left T1 club neurons from the FANC dataset. Left image shows all reconstructed neurons of a given hemilineage that are downstream of T1L club neurons. Right image shows a single example neuron from that hemilineage. (D) Heatmap depicting the spatial locations of input (left) and output (right) synapses for 10B, 8B, 0A/0B, and 9A interneurons that are downstream of T1L club neurons. (E) Circuit diagram depicting recurrent connections between 10B, 8B, 0A/0B, and 9A interneurons. Each line indicates an excitatory or inhibitory connection and is labeled with the fraction of total output synapses each interneuron class makes with another interneuron class.
The major downstream partners of club neurons include 8B, 10B, 0A/0B, and 9A neurons (Figure 5B). These neurons express different primary neurotransmitters – 8B and 10B are cholinergic, whereas 0A/0B and 9A are GABAergic. They also possess distinct morphologies that imply specialized roles in transforming club information (Figure 5C–D). Individual 10B neurons primarily receive input from one leg and project to the contralateral and adjacent legs, whereas 8B neurons arborize broadly and have mixed input and output synapses in all six neuromeres. 0A/0B neurons project bilaterally and have pre- and post-synaptic sites on both the right and left side of each VNC segment. 9A neurons are the most localized, with their input and output synapses contained within a single neuromere. The diversity of these interneuron morphologies suggests that club information is broadly relayed across the CNS through parallel pathways that integrate club information locally within a leg and globally across multiple legs. Integration of club signals within a leg could be important for amplification while integration across legs could be important for spatial localization of vibration signals. Lateral and disinhibitory circuits may sculpt vibration information, for example via normalization or gain control across the population.
Additionally, 10B, 8B, 0A/0B, and 9A neurons downstream of club neurons exhibit high levels of recurrent connectivity among interneurons from different hemilineages and different legs (Figure 5E). Overall, the circuitry downstream of club axons is complex, interconnected, and multi-layered. We hypothesize that this highly interconnected circuit architecture supports the fly’s capacity to localize substrate vibrations in the external environment.
Leg vibration information integrates with auditory circuits in the brain
Vibration signals from club neurons are relayed to the brain by ascending club sensory axons and ascending 8B and 10B neurons (Figure 6A). Since the FeCO has been implicated in sensing substrate vibrations for courtship and escape, we hypothesized that leg vibration information carried by the ascending projections is integrated in the brain with other sensory information from the antennae. The fly antenna also contains a chordotonal organ known as the Johnston’s Organ, which detects antennal displacements and local air vibrations45–47. To this end, we next identified where these ascending neurons project to in the brain and analyzed their downstream connectivity.
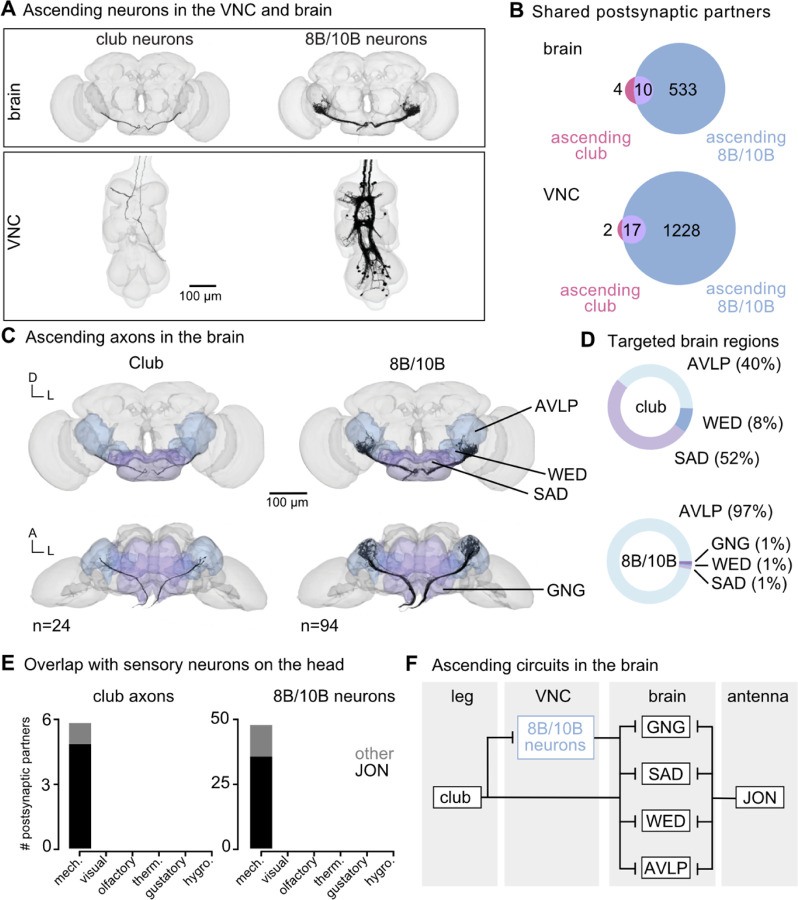
Figure 6. Vibration signals from club neurons are transmitted directly and indirectly to the brain and integrated with auditory signals from the antenna.
(A) Ascending club axons and ascending 8B and 10B interneurons that are reconstructed in the FANC (bottom, 8 club axons and 52 interneurons) and Flywire (top, 24 sensory axons and 94 interneuron axons) datasets. (B) Venn diagrams of shared postsynaptic partners between ascending club neurons and ascending 8B/10B neurons in the VNC (top) and brain (bottom). (C) Images of the ascending club axons (n = 24) and ascending 8B and 10B interneurons (n = 94) in the brain dataset with targeted brain regions highlighted (Flywire). Axons project to the anterior ventrolateral protocerebrum (AVLP), wedge (WED), saddle (SAD), and gnathal ganglion (GNG). (D) Percentage of synaptic outputs from ascending club axons (top) and ascending 8B/10Bs interneurons (bottom) in each brain region. The ascending club axons and ascending interneurons differ with respect to distribution of output synapse location. (E) Number of postsynaptic partners of ascending club axons (left) and ascending 8Bs/10Bs (right) that are shared with other sensory neurons in the brain. JON = Johnston’s Organ. (F) Circuit diagram depicting the projection patterns of ascending club and ascending 8B/10B interneurons in the brain which integrate with antennal auditory circuits.
Within the FANC connectome, we reconstructed 8 ascending club axons from two legs, 58 ascending 10B neurons, and 52 ascending 8B neurons (Figure 6A, bottom). We then used a connectome of the fly brain7,31 to identify ascending projections that matched light-level morphology of ascending projections from club axons, 8B neurons, and 10B neurons (Figure 6A, top). Within the brain connectome, we found 24 axons that matched the projections of ascending club axons and 94 axons that matched the ascending projections of 8B and 10B neurons. Due to the similarity of their ascending projections, we could not resolve which interneuron axons belonged to which hemilineage (8B or 10B) in the brain connectome. However, 8B/10B branching patterns in the brain are notably different from those of ascending club neurons. Club axons are smooth with few branches, while 10B/8B axons branch extensively.
To understand how the information in these two ascending pathways differs, we compared their connectivity in the VNC and the brain (Figure 6B). Ascending club axons and 8B/10B neurons are interconnected and share several downstream partners in the VNC. Approximately 30% of output synapses from the ascending club axons are onto ascending 8B/10B interneurons. Additionally, the majority of postsynaptic partners of ascending club axons in the VNC and the brain also receive input from ascending 8B/10B interneurons (Figure 6B). However, 8B/10B neurons have many more postsynaptic partners than club neurons. Thus, 8B/10B neurons target many of the same postsynaptic partners as ascending clubs, but also contact several non-overlapping downstream partners as well.
Consistent with our hypothesis that club neurons are exteroceptive, we found that ascending club and 8B/10B neurons target known auditory brain regions: the anterior ventrolateral protocerebrum (AVLP), wedge (WED), saddle (SAD), and gnathal ganglion (GNG) (Figure 6C–D). The AVLP, WED, SAD, and GNG receive integrated auditory information48 and are responsive to courtship song49. Neurons in the WED also respond to antennal vibrations and are tonotopically organized45. Across sensory modalities, the ascending club and 8B/10B neurons converge onto shared downstream partners with antennal mechanosensory neurons, namely Johnston’s Organ neurons (JONs) (Figure 6E). This shared connectivity suggests that, in the brain, leg vibration information is compared to mechanosensory information from the antennae. We speculate that this comparison could contribute to the detection and localization of mechanical vibrations in the external environment (Figure 6F).
Discussion
Here, we use connectomic reconstruction of neural circuits to infer the function of limb somatosensory neurons from patterns of synaptic connectivity. Our analyses suggest a dual function for the Drosophila femoral chordotonal organ (FeCO): position and movement-sensing claw and hook neurons are primarily proprioceptive, providing feedback to local leg motor circuits. In contrast, vibration-sensing club neurons are primarily exteroceptive and provide feedback to intersegmental circuits that integrate somatosensory information across multiple limbs and then convey that information to the brain. Prior experiments in Drosophila and other insects had suggested a dual function15,18–23, but it was previously unknown whether proprioception and exteroception were supported by distinct or overlapping subtypes of FeCO sensory neurons and downstream interneurons. These analyses demonstrate the power of connectomic mapping and analysis to identify putative functions of somatosensory neurons. They also motivate future work that tests the function of circuits for limb proprioception and exteroception in behaving flies.
Role of the FeCO in local leg motor control
We found that movement- and position-sensing hook and claw axons synapse directly and indirectly onto leg motor neurons (Figure 3). For example, extension-sensitive claw and hook axons provide excitatory input to the tibia and tarsus flexor motor neurons and inhibitory input to tibia extensor motor neurons. Flexion-sensitive claw and hook axons demonstrate the opposite pattern of connectivity, primarily exciting extensors and inhibiting flexors. This connectivity is consistent with prior evidence that the FeCO contributes to stabilization of leg posture18,24,28. Claw and hook axons also provide feedback to motor neurons controlling movement about other joints. Multi-joint, intra-leg feedback from the FeCO has also been found in locusts50 and wētās51.
Proprioceptive feedback needs to be flexibly tuned to reflect behavioral demands52. For example, during voluntary movement, proprioceptive pathways promoting stabilizing reflexes may be attenuated to avoid opposing the intended movement. One possible mechanism underlying this context-dependent tuning is presynaptic inhibition of sensory axons53,54. In support of this mechanism, we found several inhibitory upstream partners of claw and hook axons (Figure S3). We also showed in a recent study that hook axons are presynaptically inhibited during voluntary movement29. In addition to direct feedback onto somatosensory axons, proprioceptive feedback is also likely tuned via context-dependent inhibition of downstream pathways.
Finally, we found that claw and hook axons synapse onto a small number of intersegmental and ascending neurons (Figure 2). Intersegmental projections could relay proprioceptive information to the motor circuits of other legs. However, past work suggests that feedback from the FeCO of one leg does not strongly affect control of other legs – manipulating activity of FeCO neurons has little effect on inter-leg coordination55–57. One study described intersegmental neural pathways that receive input from the FeCO and mediate reflexes of adjacent legs, though they did not specifically test the effects of manipulating FeCO activity58. Ascending neurons that are postsynaptic to claw and hook neurons could relay leg proprioceptive information to the brain to inform motor planning. Calcium imaging experiments have shown that many ascending neurons are active during behaviors like walking59. Additionally, visual neurons and neurons within motor planning regions of the fly brain, such as the central complex, encode walking stride, speed, and turning behavior even in the absence of visual input, suggesting that they receive self-motion cues from the legs60–62.
Tonotopic organization of club axons
Individual club neurons are tuned to specific vibration frequencies, collectively forming a tonotopic map in the VNC19,63. We found that club axons are spatially organized into sub-clusters with shared postsynaptic connectivity that tile the dorsal-ventral axis of the VNC (Figure 4). By comparing this spatial organization to prior recordings of club axon activity in the VNC63, we hypothesize that the most dorsal club axons respond to higher frequencies while the most ventral club axons respond to lower frequencies. While all club neurons synapse onto ascending interneurons, only the most dorsal club axons ascend directly to the brain. Thus, we predict two pathways for club information to reach the brain: 1) a direct sensory pathway that carries high-frequency vibrations from the legs and 2) an indirect pathway that carries tonotopically organized and spatially localized vibration information. High-frequency information may be particularly salient, necessitating a more rapid behavioral response compared to broadband vibrations. Vibration sensing is an important yet understudied sensory modality in insects and other animals64. For example, recent evidence from mice shows that auditory and high-frequency tactile vibration signals converge in the inferior colliculus and are used to avoid mechanically vibrating environments65.
Putative exteroceptive function of club neurons
Our analysis of the connectome supports the hypothesis that club neurons primarily function as vibration-sensing exteroceptors. Insects utilize substrate vibrations for social communication, predator detection, and environmental sensing, such as wind66. Many insect species possess subgenual organs, specialized vibration sensors in the tibia, but flies and beetles lack these sensory structures66. Thus, club neurons may be the primary sensors for detecting substrate vibrations in the fly leg. Previous studies across multiple insect species have implicated the FeCO in escape and courtship responses20,22,23. Crustaceans also have leg chordotonal organs that can detect both joint movement and external substrate vibrations, with reported involvement in detecting vibration-based social communications67–69.
In Drosophila, courtship song involves both airborne and substrate-borne vibrations. Males vibrate their wings, producing an airborne “song” that is detected by females via the antennae70–73. Males also tap their abdomens, producing substrate-borne vibrations that promote pausing in females74. Genetic silencing of FeCO neurons in female flies reduces their receptivity to male courtship song22, suggesting that the FeCO is involved in courtship. Consistent with this hypothesis, we found downstream neurons in the brain that integrate vibration information from both legs and antennae (Figure 6). Vibration-sensitive JONs and club neurons respond to overlapping vibration frequency ranges applied to the antenna or leg, respectively19,45–47,63, but the full sensitivity range for each group has not been carefully measured. We hypothesize that the integration of vibration information from the antennae and legs could inform courtship behavior by providing overlapping information regarding both airborne and substrate-borne courtship communication. Importantly, in this study, we reconstructed the FeCO circuits within a female VNC. If club neurons are involved in detecting courtship-related signals, the circuitry downstream of the club neurons could be sexually dimorphic and thus different in male flies.
Aside from courtship, vibration information from club neurons could be used for detecting movements of predators or other threats, such as wind or rain. Consistent with this hypothesis, we found that club axons are indirectly connected to escape-related neurons, namely the motor neurons innervating the long tendon muscle (LTM) and the premotor peripherally synapsing interneuron (PSI) (Figure 3). Activation of club neurons could promote leg freezing via activation of the LTM, or take-off via activation of the PSI.
Lack of convergence across FeCO subtypes in second-order neurons
We were surprised to find that the downstream connectivity of each FeCO subtype is quite distinct: very few VNC neurons receive synapses from more than one FeCO subtype (Figure 2). Past work had proposed a higher degree of convergence across FeCO subtypes. Using whole cell patch-clamp recordings and 2-photon calcium imaging, one study found multiple VNC interneuron cell types that encode combinations of femur-tibia joint movement, position, and vibration, suggesting that they receive input from multiple FeCO subtypes24. Another combined optogenetic activation and calcium imaging to directly map the functional connectivity between FeCO axons and their downstream partners25. That study also found examples of VNC interneurons that receive inputs from more than one FeCO subtype. One possible explanation for this discrepancy is that our connectomic analyses predominantly focused on direct connections between FeCO neurons and their synaptic partners. The integration of information from multiple FeCO subtypes could be via indirect connections involving multiple intervening interneurons. In addition, Agrawal et al., (2020) found strong evidence for gap junctions that connect FeCO sensory neurons with some downstream partners. The FANC EM dataset was not imaged at sufficient spatial resolution to resolve electrical synapses. Finally, we did find some weak shared connectivity between FeCO cell types that share directional selectivity, such as claw and hook flexion axons or claw and hook extension axons. Due to the adventitious nature of their physiology experiments, Agrawal et al., (2020) and Chen et al., (2021) may have, by chance, characterized the few interneurons that do indeed receive synaptic information from multiple FeCO subtypes.
We did find, however, substantial overlap in the downstream connectivity of FeCO neurons and other leg proprioceptive neurons, such as campaniform sensilla (CS) and hairplates (HP) (Figure 2E). In fact, claw and hook axons shared a larger number of downstream partners with CS or HP neurons than with other FeCO subtypes. Work from stick insects and other models suggest that such multimodal input is important for context-dependent control of proprioceptive reflexes75,76. For example, signals from load-sensing CS neurons can reduce the effect of FeCO activation on leg motor neurons.
Although there is no overlap in the downstream connectivity of claw axons or hook extension axons and club axons, we did find some overlap in the connectivity of hook flexion and club axons (Figure 2). In fact, some club axons share more downstream connectivity with hook flexion axons than with other club axons. This finding is consistent with Agrawal et al., (2020), who found one cell type, 9Aa neurons, that respond to both flexion and vibration of the femur-tibia joint. However, the implications of this overlap remain to be investigated. For example, such overlap could enable the fly to determine if the source of the vibration is due to movement of its leg. Alternatively, perhaps flies concurrently sense movement and vibration information from the leg to assess substrate texture.
Looking forward
Connectome analysis is a powerful tool to generate and falsify hypotheses about circuit function. Thanks to advances in serial-section electron microscopy and image segmentation, we are close to having multiple connectomes of small model organisms. As more neurons within these connectomes are connected to specific functions, such as motor neurons that control a particular joint or sensory neurons that detect specific signals, these maps become increasingly useful anatomical frameworks for generating hypotheses about the neural control of behavior. Though physiological and behavioral measurements are still necessary in order to determine how a circuit functions, our study illustrates how a global view of synaptic connectivity can reveal organizing principles that motivate future experiments.
Methods
Reconstruction of neurons in the FANC connectome
Neurons in the Female Adult Nerve Cord (FANC) electron microscopy dataset8 were previously segmented in an automated manner 26. To manually correct the automated segmentation of our neurons of interest, we used Google’s collaborative Neuroglancer interface77. Many of the FeCO axons in T1L were previously identified8, and most of the claw and hook axons were previously corrected29. Here, we identified and corrected additional claw axons as well as all club axons in T1L and T3R. Identification was guided by light-level images of FeCO subtype-specific genetic driver lines15,19.
To reconstruct pre- and postsynaptic partners of FeCO neurons, we identified all objects in the automated segmentation that received at least 4 synapses from an FeCO neuron or made at least 3 synapses onto an FeCO neuron. We then proofread those objects until associated with either a cell body, or an identified descending or sensory process. A small number of objects were categorized as fragment segments and could not be connected to a cell body or an identified descending or sensory process. We deemed a neuron as “proofread” once its cell body was attached, its full backbone reconstructed, and as many branches as could be confidently attached. Neuron annotations were managed by CAVE, the Connectome Annotation Versioning Engine78. We used custom Python scripts to interact with CAVE via CAVEclient78.
Novel partners analysis
To identify the number of new postsynaptic partners added to our dataset per each FeCO sensory neuron we reconstructed, we first found all postsynaptic partners of all reconstructed T1L FeCO axons. Then, we randomly sampled the FeCO neurons one at a time (without replacement) in a cumulative fashion, and calculated how many novel postsynaptic partners were connected to each additional FeCO neuron. We re-did this random sampling fifty times.
Cosine similarity scores
Cosine similarity (for example, Figure 2I) was calculated using the cosine similarity method from the scikit-learn python package. Cosine similarity scores were then hierarchically clustered using the agglomerative clustering methods from the scikit-learn python package.
Definition of cell classes
Neurons pre- and postsynaptic of FeCO axons were identified as motor, sensory, ascending, descending, intersegmental, or local neurons. Motor neurons have a cell body in the VNC and a process in the leg nerve. These neurons were recently identified in the FANC dataset for the front left leg26,27. Sensory neurons have a process in the leg nerve but no cell body in the VNC. Ascending neurons have a process in the neck connective and a cell body in the VNC. Descending neurons have a process in the neck connective but no cell body in the VNC. Intersegmental and local neurons have a cell body and all processes in the VNC. The processes of intersegmental neurons spanned multiple neuromeres, whereas those of local neurons were contained in a single neuromere. All pre- and postsynaptic neurons were manually checked to make sure they were in the correct categories.
Identification of hemilineages
In Drosophila, neurons that share a developmental origin (i.e., belong to the same hemilineage) possess common anatomical features36 and release the same fast-acting neurotransmitter (e.g. GABA, glutamate, or acetylcholine)33. We took advantage of this knowledge to identify the hemilineage of each neuron upstream and downstream of FeCO axons in the FANC connectome. We first identified and grouped together local, intersegmental, and ascending VNC neurons based on where their primary neurite entered into the neuropil. These groups of similar primary neurites were then identified as known hemilineages using light microscopy images of sparse GAL4 lines, cell body position along the dorsal-ventral axis33,36,79,80, and personal communication (James W. Truman, David Shepherd, Haluk Lacin, and Elizabeth Marin). Putative neurotransmitter was then assigned by referencing Lacin et al., (2019). Not all of the clues are available for all of the neurite bundles. See Supplemental Table 2 for links to view entire populations of each hemilineage in Neuroglancer, an online tool for viewing connectomics datasets77.
Motor impact score
A presynaptic neuron’s monosynaptic impact score onto a postsynaptic neuron is defined as the number of synapses made by the presynaptic neuron onto the postsynaptic neuron, divided by the total number of input synapses received by the postsynaptic neuron. Then, based on the presynaptic neurons’ putative neurotransmitter according to its hemilineage assignment, this impact score is either considered excitatory (or positive) or inhibitory (or negative). In the fly, acetylcholine is typically excitatory, while GABA is typically inhibitory34,39,40. Glutamate is excitatory at the fly neuromuscular junction, acting on ionotropic glutamate receptors (GluRs), but is frequently inhibitory in the CNS, acting on the glutamate-gated chloride channel, GluCl 41.
To compute the motor impact score of a given FeCO neuron onto a motor module (Figure 3), we summed together the calculated impact scores of direct, monosynaptic connections, disynaptic connections, and trisynaptic connections between the FeCO neuron and all MNs within a module. The impact score of monosynaptic connections between an FeCO neuron and a motor module is as described above but summed across all MNs within a module. We assume that FeCO input to MNs would be cholinergic, and thus excitatory.
For the impact score of a disynaptic connection, we first found all neurons with an identified hemilineage that were postsynaptic to the FeCO neuron and presynaptic to the MNs within the relevant module. We then multiplied the monosynaptic impact score from the FeCO neuron onto one of these postFeCO/preMN neurons by the impact score of the postFeCO/preMN neuron onto the MNs within a module. If the postFeCO/preMN neuron was identified as cholinergic, then this disynaptic impact score was considered to be excitatory/positive, and if it was identified as GABAergic or glutamatergic, then it was considered to be inhibitory/negative. We then summed together all disynaptic impact scores from the FeCO neuron to the MNs of a module.
For the impact score of a trisynaptic connection, we first found all neurons with an identified hemilineage that were postsynaptic to the FeCO neuron (postFeCOs), all neurons with an identified hemilineage that were presynaptic to the MNs within the relevant module (preMNs), and then only included postFeCO neurons that were presynaptic to a preMN neuron, and preMN neurons that were postsynaptic to a postFeCO neuron. We then multiplied the monosynaptic impact score from the FeCO neuron onto a postFeCO neuron by the impact score of the postFeCO neuron onto a preMN neuron, and this was multiplied by the impact score of the preMN neuron onto the MNs within a module. If both the postFeCO and preMN neurons were excitatory or both inhibitory, then this trisynaptic impact score was positive. If one neuron was inhibitory and one was excitatory, then this trisynaptic impact score was negative. We then summed together all trisynaptic impact scores from the FeCO neuron to the MNs of a module.
Preference score
To compute the preference score for a motor module (Figure 3), we summed the number of synapses onto each MN within a module (as defined by Lesser et al., 2023) and divided by the total synapses onto all MNs. To compute the sensory subtype preference score for a FeCO subtype (Figure 3), we summed the number of synapses received from all FeCO neurons of a given subtype and divided by the total synapses received from all FeCO neurons.
Circuit analysis in the FAFB/FlyWire connectome
To study connectivity in the brain, we used the Full Adult Fly Brain connectome (FAFB; (Zheng et al., 2018) reconstructed and proofread by the FlyWire community (Dorkenwald et al., 2023a; Schlegel et al., 2023; Zheng). All data are from public release version 630.
Identification of ascending neurons in FAFB/FlyWire connectome
First, we manually screened through the repository of Gen1 MCFO images on FlyLight80 for candidate images of VNCs that exhibit hallmark expression of the ascending club axons, ascending 8B interneurons, and ascending 10B interneurons in the VNC. To identify the anatomy of the ascending projections in the brain, we matched the ascending axons and interneurons in the VNC to the corresponding images in the brain. Next, we matched the anatomy of the ascending projections in the brain based on the light-level images to the FAFB dataset using flywire.ai82 and the Codex platform83. Specifically, we queried neurons classified as ascending and cholinergic84,85, then matched candidates to the light-level images of the target neurons. See Supplemental Table 3 for links to view the ascending neurons in Neuroglancer77.
Software and data availability
Data presented in the paper was analyzed from the CAVE materialization v604 timestamp 1684915801.222989. Annotated connectivity matrices (Figure 2) will be available as python Pandas data frames (https://pandas.pydata.org/) at the git-hub repository: https://github.com/sagrawal/Lee_2024. Also available at the repository are scripts to recreate the analyses and figures in the paper, as well as scripts to recreate the connectivity matrices for users authorized to interact with the CAVEclient. Links to public segmentations are available throughout the text, as well as in a document at the git-hub repository. All analysis was performed in Python 3.9 using custom code, making extensive use of CAVEclient (https://github.com/seung-lab/CAVEclient) and CloudVolume to interact with data infrastructure, and libraries Matplotlib, Numpy, Pandas, Scikit-learn, Scipy, stats-models and VTK for general computation, machine learning and data visualization. Additional code is available at https://github.com/htem/FANC_auto_recon, providing additional tutorials, code and documentation for interacting with FANC.
Supplementary Material
Key resources table
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Deposited data | FANC connectome | Azevedo et al. (2022) | https://fanc.community | |
| Deposited data | FAFB/FlyWire connectome | Dorkenwald et al. (2023, Schlegel et al. (2023) | https://flywire.ai | |
| Software, algorithm | CAVEclient | Dorkenwald et al. (2023) | https://github.com/seunglab/CAVEclient | |
| Software, algorithm | neuPrint | Plaza et al. (2022) | https://neuprint.janelia.org/ | |
| Software, algorithm | Neuroglancer | RRID:SCR_015631 | ||
| Software, algorithm | Python | RRID:SCR_008394 |
Acknowledgements
We thank members of the Tuthill laboratory for technical assistance and feedback on the manuscript. We also thank Jasper S. Phelps, Wei-Chung Allen Lee, and the FANC community for their contributions to the proofreading of the VNC connectome. We thank Leila Elabaddy, Ellen Lesser, Shirin Mohammadian, Gwendolyn Swannell, and Brandon Pratt for granting us permission to use their unpublished reconstructions of sensory neurons in FANC. We thank Jim Truman, David Shepherd, Haluk Lacin, and Elizabeth Marin for assistance with hemilineage identification. This work was supported by a Postdoctoral Research Fellowship from the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) project 432196121 to C.J.D, a Searle Scholar Award, a Klingenstein-Simons Fellowship, a Pew Biomedical Scholar Award, a McKnight Scholar Award, a Sloan Research Fellowship, the New York Stem Cell Foundation, and NIH grants R01NS102333 and U19NS104655 to J.C.T., NIH grants K99NS117657 and R00NS117657 to S.A, and NIH grant T32 NS 99578-3 to S.J.L. and J.C.T.
References
- 1.O’Connor D. H., Krubitzer L. & Bensmaia S. Of mice and monkeys: Somatosensory processing in two prominent animal models. Prog. Neurobiol. 201, 102008 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tuthill J. C. & Wilson R. I. Mechanosensation and Adaptive Motor Control in Insects. Curr. Biol. 26, R1022–R1038 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Turecek J. & Ginty D. D. Coding of self and environment by Pacinian neurons in freely moving animals. 2023.09.11.557225 Preprint at 10.1101/2023.09.11.557225 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Reschechtko S. & Pruszynski J. A. Stretch reflexes. Curr. Biol. CB 30, R1025–R1030 (2020). [DOI] [PubMed] [Google Scholar]
- 5.Abraira V. E. & Ginty D. D. The Sensory Neurons of Touch. Neuron 79, 618–639 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Macefield V. G. The roles of mechanoreceptors in muscle and skin in human proprioception. Curr. Opin. Physiol. 21, 48–56 (2021). [Google Scholar]
- 7.Dorkenwald S. et al. Neuronal wiring diagram of an adult brain. bioRxiv 2023.06.27.546656 (2023) doi: 10.1101/2023.06.27.546656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Phelps J. S. et al. Reconstruction of motor control circuits in adult Drosophila using automated transmission electron microscopy. Cell 184, 759–774.e18 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Takemura S. et al. A Connectome of the Male Drosophila Ventral Nerve Cord. 2023.06.05.543757 Preprint at 10.1101/2023.06.05.543757 (2023). [DOI] [Google Scholar]
- 10.Winding M. et al. The connectome of an insect brain. Science 379, eadd9330 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zheng Z. et al. A Complete Electron Microscopy Volume of the Brain of Adult Drosophila melanogaster. Cell 174, 730–743.e22 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hampel S. et al. Distinct subpopulations of mechanosensory chordotonal organ neurons elicit grooming of the fruit fly antennae. eLife 9, e59976 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kim H. et al. Wiring patterns from auditory sensory neurons to the escape and song-relay pathways in fruit flies. J. Comp. Neurol. 528, 2068–2098 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mamiya A., Gurung P. & Tuthill J. C. Neural coding of leg proprioception in Drosophila. Neuron 100, 636–650 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Phillis R., Statton D., Caruccio P. & Murphey R. K. Mutations in the 8 kDa dynein light chain gene disrupt sensory axon projections in the Drosophila imaginal CNS. Development 122, 2955–2963 (1996). [DOI] [PubMed] [Google Scholar]
- 17.Smith S. A. & Shepherd D. Central afferent projections of proprioceptive sensory neurons in Drosophila revealed with the enhancer-trap technique. J. Comp. Neurol. 364, 311–323 (1996). [DOI] [PubMed] [Google Scholar]
- 18.Field L. H. & Matheson T. Chordotonal Organs of Insects. in Advances in Insect Physiology (ed. Evans P. D.) vol. 27 1–228 (Academic Press, 1998). [Google Scholar]
- 19.Mamiya A. et al. Biomechanical origins of proprioceptor feature selectivity and topographic maps in the Drosophila leg. Neuron 111, 3230–3243.e14 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Eberhard M. J. B. et al. Structure and sensory physiology of the leg scolopidial organs in Mantophasmatodea and their role in vibrational communication. Arthropod Struct. Dev. 39, 230–241 (2010). [DOI] [PubMed] [Google Scholar]
- 21.Field L. H. & Pflüger H.-J. The femoral chordotonal organ: A bifunctional orthopteran (Locusta migratoria) sense organ? Comp. Biochem. Physiol. A Physiol. 93, 729–743 (1989). [Google Scholar]
- 22.McKelvey E. G. Z. et al. Drosophila females receive male substrate-borne signals through specific leg neurons during courtship. Curr. Biol. 31, 3894–3904.e5 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Takanashi T., Fukaya M., Nakamuta K., Skals N. & Nishino H. Substrate vibrations mediate behavioral responses via femoral chordotonal organs in a cerambycid beetle. Zool. Lett. 2, 18 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Agrawal S. et al. Central processing of leg proprioception in Drosophila. eLife 9, e60299 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chen C. et al. Functional architecture of neural circuits for leg proprioception in Drosophila. Curr Biol 31, 5163–5175.e7 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Azevedo A. et al. Tools for comprehensive reconstruction and analysis of Drosophila motor circuits. (2022) doi: 10.1101/2022.12.15.520299. [DOI] [Google Scholar]
- 27.Lesser E. et al. Synaptic architecture of leg and wing motor control networks in Drosophila. bioRxiv 2023.05.30.542725 (2023) doi: 10.1101/2023.05.30.542725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Azevedo A. W. et al. A size principle for recruitment of Drosophila leg motor neurons. Elife 9, e56754 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Dallmann C. J., Agrawal S., Cook A., Brunton B. W. & Tuthill J. C. Presynaptic inhibition selectively suppresses leg proprioception in behaving Drosophila. bioRxiv 2023.10.20.563322 (2023). [Google Scholar]
- 30.Scheffer L. K. et al. A connectome and analysis of the adult Drosophila central brain. eLife 9, e57443 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schlegel P. et al. Whole-brain annotation and multi-connectome cell typing quantifies circuit stereotypy in Drosophila. 2023.06.27.546055 Preprint at 10.1101/2023.06.27.546055 (2023). [DOI] [Google Scholar]
- 32.Truman J. W., Moats W., Altman J., Marin E. C. & Williams D. W. Role of Notch signaling in establishing the hemilineages of secondary neurons in Drosophila melanogaster. Development 137, 53–61 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lacin H. et al. Neurotransmitter identity is acquired in a lineage-restricted manner in the Drosophila CNS. eLife 8, e43701 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Allen A. M. et al. A single-cell transcriptomic atlas of the adult Drosophila ventral nerve cord. bioRxiv 2019.12.20.883884 (2019) doi: 10.1101/2019.12.20.883884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lacin H. & Truman J. W. Lineage mapping identifies molecular and architectural similarities between the larval and adult Drosophila central nervous system. eLife 5, e13399 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Harris R. M., Pfeiffer B. D., Rubin G. M. & Truman J. W. Neuron hemilineages provide the functional ground plan for the Drosophila ventral nervous system. eLife 4, e04493 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mark B. et al. A developmental framework linking neurogenesis and circuit formation in the Drosophila CNS. eLife 10, e67510 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Li H. et al. Fly Cell Atlas: A single-nucleus transcriptomic atlas of the adult fruit fly. Science 375, eabk2432 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Gowda S. B. M. et al. GABAergic inhibition of leg motoneurons is required for normal walking behavior in freely moving Drosophila. Proc. Natl. Acad. Sci. 115, E2115–E2124 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lees K. et al. Actions of agonists, fipronil and ivermectin on the predominant in vivo splice and edit variant (RDLbd, I/V) of the Drosophila GABA receptor expressed in Xenopus laevis oocytes. PloS One 9, e97468 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Liu W. W. & Wilson R. I. Glutamate is an inhibitory neurotransmitter in the Drosophila olfactory system. Proc. Natl. Acad. Sci. U. S. A. 110, 10294–10299 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zill S. N., Chaudhry S., Büschges A. & Schmitz J. Force feedback reinforces muscle synergies in insect legs. Arthropod Struct. Dev. 44, 541–553 (2015). [DOI] [PubMed] [Google Scholar]
- 43.King D. G. & Wyman R. J. Anatomy of the giant fibre pathway in Drosophila. I. Three thoracic components of the pathway. J. Neurocytol. 9, 753–770 (1980). [DOI] [PubMed] [Google Scholar]
- 44.Tanouye M. A. & Wyman R. J. Motor outputs of giant nerve fiber in Drosophila. J. Neurophysiol. 44, 405–421 (1980). [DOI] [PubMed] [Google Scholar]
- 45.Patella P. & Wilson R. I. Functional Maps of Mechanosensory Features in the Drosophila Brain. Curr. Biol. CB 28, 1189–1203.e5 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ishikawa Y., Okamoto N., Nakamura M., Kim H. & Kamikouchi A. Anatomic and Physiologic Heterogeneity of Subgroup-A Auditory Sensory Neurons in Fruit Flies. Front. Neural Circuits 11, 46 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yorozu S. et al. Distinct sensory representations of wind and near-field sound in the Drosophila brain. Nature 458, 201–205 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Matsuo E. et al. Organization of projection neurons and local neurons of the primary auditory center in the fruit fly Drosophila melanogaster. J. Comp. Neurol. 524, 1099–1164 (2016). [DOI] [PubMed] [Google Scholar]
- 49.Baker C. A. et al. Neural network organization for courtship-song feature detection in Drosophila. Curr. Biol. CB 32, 3317–3333.e7 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Burrows M. & Horridge G. A. The organization of inputs to motoneurons of the locust metathoracic leg. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 269, 49–94 (1974). [DOI] [PubMed] [Google Scholar]
- 51.Field L. H. & Rind F. C. A single insect chordotonal organ mediates inter- and intra-segmental leg reflexes. Comp. Biochem. Physiol. A Physiol. 68, 99–102 (1981). [Google Scholar]
- 52.Azim E. & Seki K. Gain control in the sensorimotor system. Curr. Opin. Physiol. 8, 177–187 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Koch S. C. et al. RORβ Spinal Interneurons Gate Sensory Transmission during Locomotion to Secure a Fluid Walking Gait. Neuron 96, 1419–1431.e5 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.McComas A. J. Hypothesis: Hughlings Jackson and presynaptic inhibition: is there a big picture? J. Neurophysiol. 116, 41–50 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Chockley A. S. et al. Subsets of leg proprioceptors influence leg kinematics but not interleg coordination in Drosophila melanogaster walking. J. Exp. Biol. 225, jeb244245 (2022). [DOI] [PubMed] [Google Scholar]
- 56.Delcomyn F. Factors Regulating Insect Walking. Annu. Rev. Entomol. 30, 239–256 (1985). [Google Scholar]
- 57.Pratt B. G., Lee S.-Y. J., Chou G. M. & Tuthill J. C. Miniature linear and split-belt treadmills reveal mechanisms of adaptive motor control in walking Drosophila. 2024.02.23.581656 Preprint at 10.1101/2024.02.23.581656 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Laurent G. & Burrows M. Intersegmental interneurons can control the gain of reflexes in adjacent segments of the locust by their action on nonspiking local interneurons. J. Neurosci. Off. J. Soc. Neurosci. 9, 3030–3039 (1989). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Chen C.-L. et al. Ascending neurons convey behavioral state to integrative sensory and action selection brain regions. Nat. Neurosci. 26, 682–695 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Cruz T. L. & Chiappe M. E. Multilevel visuomotor control of locomotion in Drosophila. Curr. Opin. Neurobiol. 82, 102774 (2023). [DOI] [PubMed] [Google Scholar]
- 61.Fujiwara T., Brotas M. & Chiappe M. E. Walking strides direct rapid and flexible recruitment of visual circuits for course control in Drosophila. Neuron 110, 2124–2138.e8 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Pfeiffer K. & Homberg U. Organization and functional roles of the central complex in the insect brain. Annu. Rev. Entomol. 59, 165–184 (2014). [DOI] [PubMed] [Google Scholar]
- 64.Hill P. S. M. & Wessel A. Biotremology. Curr. Biol. 26, R187–R191 (2016). [DOI] [PubMed] [Google Scholar]
- 65.Huey E. L. et al. The auditory midbrain mediates tactile vibration sensing. bioRxiv 2024.03.08.584077 (2024) doi: 10.1101/2024.03.08.584077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Virant-Doberlet M., Stritih-Peljhan N., Žunič-Kosi A. & Polajnar J. Functional Diversity of Vibrational Signaling Systems in Insects. Annu. Rev. Entomol. 68, 191–210 (2023). [DOI] [PubMed] [Google Scholar]
- 67.Burke W. An Organ for Proprioception and Vibration Sense in Carcinus Maenas. J. Exp. Biol. 31, 127–138 (1954). [Google Scholar]
- 68.Cohen M. J. The crustacean myochordotonal organ as a proprioceptive system. Comp. Biochem. Physiol. 8, 223–243 (1963). [DOI] [PubMed] [Google Scholar]
- 69.Salmon M., Horch K. & Hyatt G. Barth’s myochordotonal organ as a receptor for auditory and vibrational stimuli in fiddler crabs (Uca pugilator and U. minax). Mar. Freshw. Behav. Phy 4, 187–194 (1977). [Google Scholar]
- 70.Ewing A. W. & Bennet-Clark H. C. The Courtship Songs of Drosophila. Behaviour 31, 288–301 (1968). [Google Scholar]
- 71.Kamikouchi A. et al. The neural basis of Drosophila gravity-sensing and hearing. Nature 458, 165–171 (2009). [DOI] [PubMed] [Google Scholar]
- 72.Murthy M. Unraveling the auditory system of Drosophila. Curr. Opin. Neurobiol. 20, 281–287 (2010). [DOI] [PubMed] [Google Scholar]
- 73.Shorey H. H. Nature of the Sound Produced by Drosophila melanogaster during Courtship. Science 137, 677–678 (1962). [DOI] [PubMed] [Google Scholar]
- 74.Fabre C. C. G. et al. Substrate-Borne Vibratory Communication during Courtship in Drosophila melanogaster. Curr. Biol. 22, 2180–2185 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Gebehart C., Hooper S. L. & Büschges A. Non-linear multimodal integration in a distributed premotor network controls proprioceptive reflex gain in the insect leg. Curr. Biol. CB 32, 3847–3854.e3 (2022). [DOI] [PubMed] [Google Scholar]
- 76.Gebehart C. & Büschges A. The processing of proprioceptive signals in distributed networks: insights from insect motor control. J. Exp. Biol. 227, jeb246182 (2024). [DOI] [PubMed] [Google Scholar]
- 77.Maitin-Shepard J. et al. google/neuroglancer: Zenodo 10.5281/zenodo.5573294 (2021). [DOI] [Google Scholar]
- 78.Dorkenwald S. et al. CAVE: Connectome Annotation Versioning Engine. bioRxiv 2023.07.26.550598 (2023) doi: 10.1101/2023.07.26.550598. [DOI] [Google Scholar]
- 79.Marin E. C. et al. Systematic annotation of a complete adult male Drosophila nerve cord connectome reveals principles of functional organisation. bioRxiv 2023.06.05.543407 (2023) doi: 10.1101/2023.06.05.543407. [DOI] [Google Scholar]
- 80.Meissner G. W. et al. A searchable image resource of Drosophila GAL4 driver expression patterns with single neuron resolution. eLife 12, e80660 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Buhmann J. et al. Automatic detection of synaptic partners in a whole-brain Drosophila electron microscopy data set. Nat. Methods 18, 771–774 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Dorkenwald S. et al. FlyWire: online community for whole-brain connectomics. Nat. Methods 19, 119–128 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Matsliah A. et al. Neuronal “parts list” and wiring diagram for a visual system. bioRxiv 2023.10.12.562119 (2023) doi: 10.1101/2023.10.12.562119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Eckstein N. et al. Neurotransmitter Classification from Electron Microscopy Images at Synaptic Sites in Drosophila Melanogaster. 2020.06.12.148775 Preprint at 10.1101/2020.06.12.148775 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Heinrich L., Funke J., Pape C., Nunez-Iglesias J. & Saalfeld S. Synaptic Cleft Segmentation in Non-isotropic Volume Electron Microscopy of the Complete Drosophila Brain. in Medical Image Computing and Computer Assisted Intervention – MICCAI 2018 (eds. Frangi A. F., Schnabel J. A., Davatzikos C., Alberola-López C. & Fichtinger G.) 317–325 (Springer International Publishing, Cham, 2018). doi: 10.1007/978-3-030-00934-2_36. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data presented in the paper was analyzed from the CAVE materialization v604 timestamp 1684915801.222989. Annotated connectivity matrices (Figure 2) will be available as python Pandas data frames (https://pandas.pydata.org/) at the git-hub repository: https://github.com/sagrawal/Lee_2024. Also available at the repository are scripts to recreate the analyses and figures in the paper, as well as scripts to recreate the connectivity matrices for users authorized to interact with the CAVEclient. Links to public segmentations are available throughout the text, as well as in a document at the git-hub repository. All analysis was performed in Python 3.9 using custom code, making extensive use of CAVEclient (https://github.com/seung-lab/CAVEclient) and CloudVolume to interact with data infrastructure, and libraries Matplotlib, Numpy, Pandas, Scikit-learn, Scipy, stats-models and VTK for general computation, machine learning and data visualization. Additional code is available at https://github.com/htem/FANC_auto_recon, providing additional tutorials, code and documentation for interacting with FANC.