Abstract
Cellular senescence has a complex role in lymphocyte carcinogenesis and drug resistance of lymphomas. Senescent lymphoma cells combine with immunocytes to create an ageing environment that can be reprogrammed with a senescence-associated secretory phenotype, which gradually promotes therapeutic resistance. Certain signalling pathways, such as the NF-κB, Wnt and PI3K/AKT/mTOR pathways, regulate the tumour ageing microenvironment and induce the proliferation and progression of lymphoma cells. Therefore, targeting senescence-related enzymes or their signal transduction pathways may overcome radiotherapy or chemotherapy resistance and enhance the efficacy of relapsed/refractory lymphoma treatments. Mechanisms underlying drug resistance in lymphomas are complex. The ageing microenvironment is a novel factor that contributes to drug resistance in lymphomas. In terms of clinical translation, some senolytics have been used in clinical trials on patients with relapsed or refractory lymphoma. Combining immunotherapy with epigenetic drugs may achieve better therapeutic effects; however, senescent cells exhibit considerable heterogeneity and lymphoma has several subtypes. Extensive research is necessary to achieve the practical application of senolytics in relapsed or refractory lymphomas. This review summarises the mechanisms of senescence-associated drug resistance in lymphoma, as well as emerging strategies using senolytics, to overcome therapeutic resistance in lymphoma.
Key words: ageing microenvironment, cellular senescence, drug resistance, lymphoma, senescence-associated secretory phenotype, senolytics
1. Introduction
Cell senescence is a process that permanently arrests cellular proliferation, which not only triggers tissue remodelling during organismal development and after body injury but also leads to inflammation, tumourigenesis and decreased regenerative function of tissues (1-3). Cellular senescence is classified into the following types: i) Replicative senescence; ii) stress-induced premature senescence (SIPS), including DNA damage- and oxidative stress-induced senescence; iii) oncogene-induced senescence (OIS); iv) paracrine senescence; v) therapy-induced senescence (TIS); vi) mitochondrial dysfunction-associated senescence; and vii) epigenetically induced senescence (3,4).
Although cell senescence is generally considered beneficial, malignancy is also associated with senescence, both of which are caused by accumulation of time-dependent cellular damage (5). Senescent cells can secrete cytokines [including interleukin (IL)-6, IL-8, tumour necrosis factor (TNF)α and interferon-(IFN)γ], chemokines [C-C motif ligand 2 and C-X-C motif chemokine ligand (CXCL)1], growth factors (granulocyte-macrophage colony-stimulating factor) and matrix metalloprotein (matrix metalloproteinase 9), into the ageing environment and gradually induce a senescent-associated secretory phenotype (SASP) (6,7). In tissues, the short-term accumulation of aged cells is beneficial for renewing the in vivo environment; however, the long-term accumulation of aged tumour cells may induce disequilibrium in an organism or local organs and gradually result in the formation of an aged tumour microenvironment (TME) (8). Among lymphatic system malignancies, lymphoma development, including histopathology, clinical treatment and prognosis, differs between young and old adults (9). In Hodgkin's lymphoma (HL), Reed-Sternberg (RS) cells retain the characteristics of senescent cells, staining specifically for senescence-associated β-galactosidase (SA-β-gal) and exhibiting abnormal expression of cell cycle inhibitors p21Cip1/p16INK4a/p53, and negative Ki-67 expression. In addition, RS-like cells exhibit a SASP in their microenvironments (10).
Drug resistance is one of the main challenges encountered by clinicians in lymphoma treatment. When treated with a single drug, tumours can resist multiple chemically unrelated drugs, resulting in multidrug resistance (MDR) (11). Common factors involved in MDR include: i) Expression of efflux transporters, such as P-glycoprotein [P-gp; also called ATP binding cassette subfamily B member 1 (ABCB1)], ATP binding cassette subfamily G member 2 [ABCG2, breast cancer resistance (BCR) protein], and multidrug resistance-associated protein 1. P-gp, encoded by MDR1, has a crucial role in lymphoma multidrug resistance (12). Anthracycline resistance is mediated by the overexpression of P-gp, a transmembrane protein that functions as an efflux pump, actively transporting anthracyclines out of cancer cells, thereby reducing their intracellular concentration and efficacy (13). ii) Tumour stem-like cells (also known as side populations) (14). However, the additional mechanisms underlying lymphoma resistance remain to be explored. Hanahan (15) reported that tumour cells undergo transient ageing in cases of therapeutic resistance. These senescent tumour cells appear dormant and evade targeted drug attacks (16). In such cases, SASP have a crucial role in the development of lymphoma resistance (17). In murine models of Burkitt's lymphoma, DNA damage has been found to induce the release of paracrine factors [such as IL-6 and metalloproteinase tissue inhibitor 1 (Timp-1)], which contribute to lymphoma cell survival in the TME after chemotherapy and create a 'chemoresistant niche' (18). These studies suggest that the ageing microenvironment, in which the SASP is involved, may have a critical role in the mechanisms of chemotherapy resistance in lymphoma.
The present review, from the perspective of preclinical research and clinical applications, focuses on the interactions between the ageing TME and lymphocyte carcinogenesis and discusses how the ageing microenvironment is reprogrammed and promotes the development of lymphoma resistance. Finally, opportunities and challenges in solving the problem of drug resistance in lymphoma were discussed from the perspective of an ageing microenvironment.
2. The ageing microenvironment mediates lymphocyte carcinogenesis
B-cell-derived lymphoma
The expression of SASP markers, such as cytokines TNF-α/IL-6/IL-8 and microRNA-155, -16 and -93, have been detected in memory B cells (19). Compared to young mice, older mice showed considerable differences in B-cell composition in lymphatic tissues. This part of the B lymphocyte lineage is termed age-associated B cells, which secrete TNF-α and suppress pro-B-cell survival and B lymphopoiesis (20,21).
T cell-derived lymphoma
Changes in the ageing host and senescent cell metabolism can seriously affect T-cell development, maintenance and function (22). For instance, T cells expressing T cell immunoglobulin and mucin domain-containing protein 3 include a subset of senescent cells arrested in the G1/S phase (23). In old mouse models, memory CD8+ T cells with CD44lowCD62Lhigh express high levels of stem cell antigen-1 (24,25). Furthermore, γδ T-cells exert the function of killing lymphoma cells in the TME. However, in an ageing microenvironment, a large number of changes in the composition of the γδ T-cell pool in peripheral lymph nodes (pLNs) may lead to an imbalance of γδ T-cell responses in tumours and accelerate tumour growth. Ageing alters the function of the T cell receptor δ chain and the clonal structure of γδ T-cell subsets. In old mice, increased IL-7 expression mediates the expansion of the γδ17 T-cell compartment, which is related to lymphoma growth (26). Furthermore, human type 17 T-helper (Th17) cells possess stem cell-like properties and intervention with Th17 stemness may help address Th17-associated drug resistance, microenvironmental disturbances and organ disabilities (27). Failure of the T-cell metabolism leads to the accumulation of circulating cytokines that act as systemic inducers of ageing. Immune dysregulation ultimately leads to T cell-derived lymphoid tissue neoplasms (28).
Natural killer (NK) cell-derived lymphoma
There is a close relationship between abnormal NK-cell function, carcinogenesis and the ageing microenvironment. A key feature of NK cells is the expression of human leukocyte antigen class I-specific receptors, which manifests as the downregulation of NK group 2A (NKG2A) receptor and upregulation of killer cell immunoglobulin-like receptor (KIR) family members. However, the ageing microenvironment affects the function and phenotype of KIR/NKG2A repertoires, which cause abnormal or cancerous functions in NK cells (29). In addition, NK cells from patients with germline gain-of function mutations in PIK3CD (encoding the PI3K p110δ catalytic subunit) exhibit abnormal differentiation, which is associated with the overexpression of immunosenescence/depletion-related markers. Immunosenescence may lead to a decline in the ability of the immune system to monitor and clear abnormal cells, thus providing a more favourable environment for NK cells to become cancerous (30). In summary, the complex mechanism between NK-cell cancer and the immunosenescent microenvironment needs to be further explored, as it is crucial for the treatment and prevention of NK cell-derived lymphoma.
3. Biomarkers of senescence-mediated lymphoma resistance
Senescent cells are heterogeneous at different stages of cancer (31). Researchers have often combined senescent cells, drug resistance and tumour stem cell markers to study ageing-induced drug resistance. Biomarkers of senescent cells in lymphoma require further investigation. In this chapter, the biomarkers of senescent lymphoma cells that have been reported to date are discussed.
SA-β-gal
SA-β-gal is widely recognised as a critical biomarker for senescent cells, including lymphoma. Milanovic et al (32) selected SA-β-gal and 5-bromo-2'-deoxyuridine/propidium iodide (BrdU/PI) as markers to analyse senescent cells with stemness in B-cell lymphoma in both human cell lines and mouse models. Senescent lymphoma cells were detected by SA-β-gal staining in the ageing environment of relapsed or refractory (R/R) diffuse large B-cell lymphoma with elevated expression of the SENEX gene (33). SA-β-gal expression is important in the study of drug resistance mechanisms, as it allows researchers to identify whether cellular senescence has been induced and understand how senescent lymphoma cells resist treatment, which may potentially be applied in lymphoma treatment.
SASP
The SASP has a critical role in lymphoma progression, enhances pro-cancerous effects on surrounding lymphoma cells and tissues via paracrine and autocrine pathways, and may affect the immune response in human cell lines and mouse models (34). SASPs act as both promoters and inhibitors of lymphoma development. Certain SASP components promote tumour-cell formation and development; however, other components may have a role in the immune response, helping to fight cancer cells. All these functions of the SASP components depend on the antitumour drug action time, drug intensity and immune microenvironment of the organisms involved. The relationship between SASP and lymphoma is a complex area of research and scientists are working to develop new treatments or intervention strategies to improve treatment outcomes in lymphoma cases involving drug resistance (35).
Suppressor of variegation 3-9 homolog 1 (SUV39H1)
Trimethylation of histone H3 lysine 9 (H3K9me3) and the H3K9me3-specific histone methyltransferase SUV39H1 have been reported as biomarkers for senescent lymphoma cells in human cell lines and mouse models (36). H3K9me-mediated ageing blocks the response to the oncogene Ras. Loss of SUV39H1 activates Ras expression and enhances lymphoma aggressiveness (36). In addition, TIS relies on SUV39H1, which promotes lymphoma (37). Overexpressed MYC is a well-known lymphoma oncogene that can induce self-renewal of stem cells and prevent cell differentiation (38-40). The function of MYC is limited by TGF-β regulation and the development of lymphoma is inhibited by SUV39H1-dependent cellular senescence (41).
RAS
RAS represents a class of signalling proteins belonging to the small GTPase family and includes various subtypes, such as H-RAS, K-RAS and N-RAS (42). RAS proteins have key regulatory roles in normal cell growth and development; however, aberrant RAS signalling pathway activity has been implicated in various tumours and other diseases. RAS is also involved in the regulation of cellular senescence in human cell lines and mouse models (43-45).
Patients with high expression of the RAS oncogene (also termed K-RAS) have a poor prognosis and often exhibit MDR (46). RAS is the most common marker used in studies of lymphoma OIS. It is regulated by another important senescence-associated biomarker, SUV39H1, and drives cell-cycle arrest, which exacerbates the promalignant paracrine activity of the SASP, confirming the core feature of genotoxic stress-induced senescence (36). In general, the RAS signalling pathway is involved in the regulation of cellular senescence, which is essential for understanding the occurrence and development of drug resistance in lymphoma (47).
ABC protein family
The ABC protein family is closely associated with drug resistance in tumours. Lymphoma stem-like cells (CD34+ and CD44+) with high ABCG2 expression exhibited a senescent phenotype in mouse models (37). Multidrug resistance protein 4 (ABCC4) is overexpressed in human NK/T-cell lymphoma (NKTCL) cells, which modifies the sensitivity of chemotherapy to epirubicin and cisplatin and may be a functional therapeutic target (48). In senescent cells, ATP-binding box subfamily B member 4 mediates the synthesis and release of small extracellular vesicles and enhances drug resistance in cancer cells (49). ABCA1 participates in the export of free cholesterol by specific cells, such as macrophages and foam cells, into the lymphoma microenvironment, which in turn promotes lymphoma cell proliferation (50). ABCG2 enhances the multidrug resistance profile of human NKTCL side-population cells (51). Thus, ABC protein family members combined with senescence-associated biomarkers (e.g. SA-β-gal), may be used as indicators to evaluate senescence-related drug-resistant traits.
4. Signal transduction pathways in senescence-mediated lymphoma resistance
The relationship between senescence and lymphoma resistance involves complex signalling pathways. It should be noted that lymphomas are a group of highly heterogeneous diseases; thus, different molecular subtypes and individual idiosyncrasies may have different regulatory mechanisms involving their signalling pathways. Therefore, the mechanisms underlying the signalling pathways involved in cellular senescence in lymphoma must be studied in depth according to specific lymphoma subtypes. Certain key signalling pathways that may have critical roles in senescence-related lymphoma resistance were discussed in the following sections.
P53/NF-κB signalling
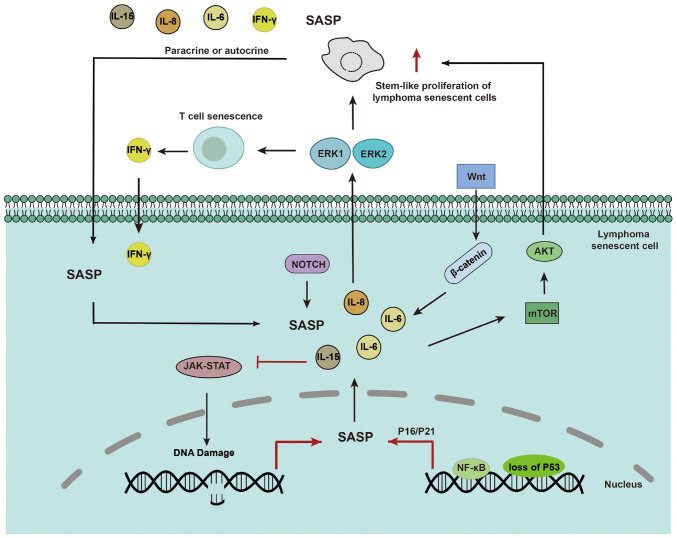
The SASP depends on paracrine mechanisms involving interleukins IL-6 and IL-8 to induce epithelial-mesenchymal transition and aggressive malignancy. Loss of p53 exacerbates the paracrine activity of the SASP (47). Proteomic analyses have identified NF-κB as a critical regulator of SASP in senescent chromatin. Subunit p65 of NF-κB is a major transcription factor and accumulates on senescent chromatin. NF-κB inhibition triggers escape from immune recognition by NK cells and bypasses the senescence mechanism by collaborating with inactivated p53. In mouse lymphoma models, NF-κB inhibition alters the microenvironment of treatment-induced senescence by controlling SASP, promoting drug resistance and early recurrence of lymphoma, and shortening survival time (17). In HL, RS cellular senescence with abnormal expression of SA-β-gal and p21Cip1/p16INK4a/p53 may also be the origin of the pro-inflammatory microenvironment and gradual development of SASP and promote HL drug resistance. NF-κB inhibitors (such as JSH-23 and curcumin) decrease the secretion of IL-6 from RS-like cells in HL (10).
Wnt signalling
The Wnt signalling pathway has a critical role in cell fate determination and differentiation. In senescent lymphoma cells, the aberrant activation of Wnt signalling may lead to the maintenance of a stem cell-like state, inhibiting normal cell differentiation. Abnormal Wnt/β-catenin signalling promotes tumour stem-like cell proliferation and thus has a critical role in tumour resistance and treatment responses (52). Wnt signalling has been reported to participate in the SASP-mediated proliferation of stem-like cells in the B-cell lymphoma SASP (32). The application of Wnt protein pathway inhibitors can potentially enable more precise and personalised treatment of drug-resistant lymphoma.
MAPK signalling
Extracellular signal-regulated protein kinases (ERK), such as ERK1 and ERK2, act on cytotoxic T cells (CTLs) and participate in IFN-mediated resistance of T lymphoma cells (53). Furthermore, numerous aneuploid senescent cells often exist as double-hit or double-expressing diffuse large B-cell lymphomas (DH/DE-DLBCLs) after treatment. This mechanism of mitotic escape leads to drug resistance. These aneuploid senescent cells increase ERK/MAPK activity via Bruton's tyrosine kinase (BTK) signalling and the chronically active BCR pathway, resulting in elevated metabolism (54).
PI3K/AKT/mTOR signalling
The PI3K/AKT/mTOR pathway is a potent cell proliferation and growth signalling pathway that is also involved in the regulation of the cell cycle and apoptosis. In lymphoma, overactivation of this pathway is frequently observed under repeated stress conditions, leading to abnormal cell proliferation (55,56). Senescent cells escape the treated DH/DE-DLBCL microenvironment and acquire improved metabolic properties via the AKT/mTOR pathway, resulting in drug resistance (54). Downregulation of this pathway may help to restore the apoptotic capacity of cells and regulate the cell cycle, thereby inhibiting lymphoma-cell proliferation.
Notch signalling
In lymphoma, the aberrant activation of the Notch signalling pathway is closely associated with disease onset and progression. In senescent lymphoma cells, Notch signalling cascades with fibroblast growth factor and Wnt signalling to maintain self-renewal of tumour stem cells and remodel the TME (57). Currently, research on the mechanism of action of Notch in lymphoma-cell senescence is expanding, and future studies will help to reveal the underlying molecular mechanisms in greater detail.
JAK-STAT signalling
The JAK-STAT signalling pathway is a key pathway involved in cell proliferation, differentiation and immune responses. Senescent lymphoma cells may evade detection and attack by the immune system by manipulating the JAK-STAT signalling pathway (58). The JAK/3 signalling pathway is required for lymphoma cell survival after doxorubicin (DOX) treatment in vivo and in vitro (18). Certain cytokines have a role in JAK-STAT signalling. IL-15 stimulates JAK3-STAT5 signalling and inhibits the expression of DNA damage response genes, thus delaying CD8+ T-cell senescence (59). IL-6 reverses DOX resistance in nasal NKTCL by downregulating ABCC4 and inactivating the JAK2/STAT3/NF-κB/P65 pathway (Fig. 1) (60). Therefore, inhibition of the JAK-STAT pathway by interfering with the expression of SASP to kill drug-resistant lymphoma cells with a senescent phenotype is a promising direction for future research.
Figure 1.
Main signalling pathway mechanisms of SASP in lymphoma cellular resistance. The loss of p53 and expression of NF-κB exacerbate the paracrine activity of SASP, which promotes cytokine secretion, as well as signal transduction pathways, including mTOR/NOTCH/ERK/JAK-STAT, mediate the stem-like proliferation of lymphoma senescent cells. All of these facilitate the drug resistant characteristics of lymphoma cells. SASP, senescent-associated secretory phenotype.
5. Research model applications in senescence-associated lymphoma resistance
Cell and animal models have revealed several mechanisms through which senescent cells develop drug resistance, facilitating the development of additional senolytic drugs based on these models. Different types of senescence have been observed in various cellular and animal models. In this chapter, various research models for lymphoma senescence-associated resistance are discussed.
Cell models
For TIS, the induction time has a critical role for constructing cell models of lymphoma senescence-associated resistance. In clinical patients, after completion of a cancer treatment regimen, TIS in tumours and normal tissues can gradually develop within 10 days to 6 weeks (31). In an in vitro assay, tumour cell lines treated with various chemotherapeutic drugs were used as cell models to study senescence-associated resistance. The senescent phenotype can be identified via SA-β-gal assays and SASP-related cytokine detection (such as IL-4, IL6, IL-1β) (61). One study reported that β-gal-positive cells could be induced in non-small cell lung cancer cell lines after 2-6 days of treatment with cisplatin (62). To date, limited research has been conducted on TIS-cell lymphoma models.
Animal models
For SIPS, excision repair cross complement 1 knockout/deletion mice have been used as a DNA-damaging model to study senolytic drugs (63,64). In OIS, BUB1 mitotic checkpoint serine/threonine kinase B (BubR1) encodes a vital component of mitotic spindle assembly. The BubR1H/H mouse is an ideal model of cellular senescence (63,65). Mdr2-/- mice treated with navitoclax (ABT263) are good models for primary lymphoma resistance research (66). Telomerase knockout mice are ideal models for studying senescence-associated mechanisms of immune escape (67). In the case of TIS, primary Eμ-Myc transgenic Bcl2-overexpressing lymphomas treated with adriamycin are a recognised mouse model for the study of TIS of B-cell lymphoma. SUV39H1 inhibits the conversion of normal cells into induced pluripotent stem cells. SUV39H1-deficient Eμ-Myc mice have been demonstrated to be a suitable model of senescence-associated resistance in B-cell lymphoma (32,68).
In one study, a patient-derived DLBCL xenograft model (PDX) was successfully established to screen for drug sensitivity and explore resistance mechanisms (69). Cell-derived mouse xenograft models have been frequently used to study the mechanisms underlying drug-resistant lymphoma (70).
Three-dimensional (3D) organoid models
Multiple lymphoid organs have been designed in the field of immune engineering, such as the engineering of bone marrow and thymus tissue (71). The lack of understanding of the factors that regulate lymphoma resistance and the establishment of predictors is mainly due to the need for in vitro lymphoma models to accurately study the lymphoma microenvironment. In contrast to other tumour types, the development of lymphoma organoids is slow (72).
Tian et al (72) established a synthetic hydrogel 3D organoid microenvironment for Band T-cell lymphomas using biomaterials such as maleimide-functionalised 4-arm polyethylene glycol and thiolated cross-linkers, which could effectively detect drug sensitivity and resistance. Of note, B-cell lymphoma cell lines (HBL-1, DOHH2, WSU-DLCL-2, SUDHL-4, SUDHL-6, OCI-Ly10 and OCI-Ly12) and a T-cell lymphoma cell line (HuT-78) were used to successfully mimic the 3D organoid microenvironment (72). To examine the efficacy of novel antitumour drugs, Vidal-Crespo et al (69) used patient lymph nodes to establish 3D spheroids using the hanging drop method, whereas Ceccato et al (73) used human femoral bones to construct a TME of DLBCL-bone marrow. Patient-derived tumour xenografts and NOD. Cg-rkdcscid Il2rgtm1Wjl/SzJ mice were used to establish the PDX mouse model. Subsequently, tumours were removed and grown in bioadhesive polyethene glycol hydrogels to establish DLBCL 3D organoids (70,74). Similarly, to further explore whether senolytics are effective in humans, the investigators used human tissue explants (including tumour tissues) by in vitro culture to demonstrate the efficacy of senolytics (63).
6. Drug mechanisms in senescence-related lymphoma resistance
Chemotherapy resistance associated with an ageing microenvironment
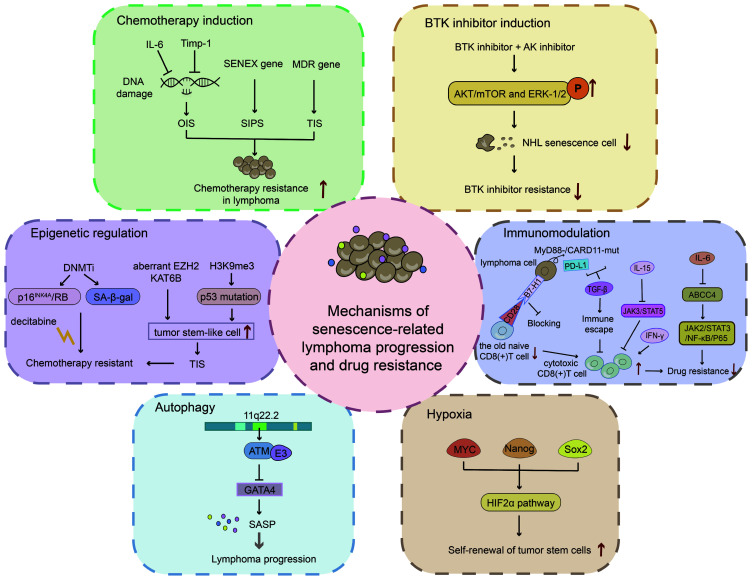
Different chemotherapeutic drugs have different mechanisms of action. Moderate doses of chemotherapy increase the likelihood of SASP and higher doses can directly lead to cell death (75). The autocrine IL-6 signalling loop is induced when the oncogene RAS is activated, and this autocrine loop enhances OIS (36,47). Gilbert and Hemann (18) used mature Burkitt's lymphoma mouse models to demonstrate that the paracrine factors recombinant IL-6 and Timp-1 in the ageing microenvironment are released into the thymus to resist tumour cell DNA damage, resulting in minimal residual tumour lesions. Researchers have suggested that patients with extranodal NKTCL exhibit primary resistance to anthracycline regimens, mainly because of the overexpression of the MDR gene (76). Increasing evidence has confirmed that DOX can induce an ageing microenvironment, leading to lymphoma resistance. For instance, in DLBCL, SIPS can activate SENEX and mediate DOX resistance (33,77). Additional ageing microenvironment-related drug resistance mechanisms in various types of lymphomas need to be further explored.
BTK inhibitor resistance is associated with the ageing microenvironment
Specific inhibitors targeting B-cell receptor-associated kinases (BAKs), such as BTK and PI3K, have revolutionised the treatment of B lymphoid malignancies. Evidence suggests that BAK inhibitors are a class of drugs that modulate the cancer (haematopoietic) microenvironment (78,79). However, at times, ibrutinib alone has a poor effect in the treatment of DH/DE-DLBCL. This may be related to the formation of drug-resistant senescent cells in the TME. Alisertib is a senolytic aurora kinase (AK) inhibitor that functions as a senolytic (80). This can disrupt the escape mechanism of the immune microenvironment and block senescent cells. Thus, a BTK inhibitor combined with an AK inhibitor may overcome drug resistance in DH/DE-DLBCL (54).
Asparaginase inhibits lymphoma progression by promoting cell senescence
In the treatment of certain peripheral T-cell lymphomas (PTCL), such as NKTCL, drug resistance often develops to cyclophosphamide, DOX, vincristine and prednisone (CHOP) regimens containing DOX. Furthermore, asparaginase-containing regimens, such as cisplatin, dexamethasone, gemcitabine and pegaspargase (DDGP) or pegaspargase, gemcitabine and oxaliplatin (P-Gemox) are often chosen by clinicians (81).
This may be because asparaginase does not depend on classical resistance mechanisms; for example, MDR-associated ABC family members lead to drug resistance. Asparagine has long been considered a tumour metabolite (82). It is related to mTORC1 activity and intracellular asparagine can be exchanged with extracellular amino acids, thereby regulating the uptake of amino acids, particularly serine, arginine, and histidine, which are important regulators of amino acid homeostasis, anabolism and the proliferation of cancer cells (83). Because asparaginase, the cornerstone of extranodal NKTCL and acute lymphoblastic leukaemia (ALL) therapy, is irreplaceable, exploring the mechanisms involved in its action is important (84).
Researchers isolated Rhodospirillum rubrum L-asparaginase (RrA), a telomerase inhibitor that promotes the senescence of Jurkat cells (a type of T-cell lymphoma cell). RrA cannot inhibit telomerase directly but can downregulate hTERT expression and induce replicative senescence in lymphoma cells (85,86). The association between cellular senescence and amino acid metabolism involving L-asparaginase is currently unknown. This may be key to future research on L-asparaginase resistance in R/R lymphomas.
Epigenetic drugs reverse senescence-related drug resistance in lymphoma
Decitabine (5-aza-CdR), a DNA methyltransferase inhibitor, was reported to inhibit the progression of anaplastic large cell lymphoma (ALCL) and promote the expression of p16INK4A in the retinoblastoma protein and SA-β-gal pathways (87). This may be a crucial mechanism in decitabine treatment-resistant ALCL (88).
Zeste homologue 2 is a histone methyltransferase enhancer whose aberrant expression directs cells towards the cancer stem cell state by regulating the balance between self-renewal and cell differentiation (89).
Histone acetyltransferases (KATs) have essential roles in various organisms. KAT6A (MOZ or MYST3) and KAT6B (MORF or QKF) belong to the KAT family of oncogenes that inhibit cellular senescence. Allele deletion of KAT6A prolongs the median survival of Myc transgenic mice with lymphoma. Selective inhibitors of KAT6B, such as WM-1119, suppress lymphoma development in mice. These results suggest that KAT6A and KAT6B inhibitors may be effective therapeutic agents for lymphoma (90,91).
However, various drugs are still in preclinical stages. Epigenetic modifications involving H3K9me3 were indicated to drive stem cell-like functions in B-cell lymphomas in a senescent model employing Eμ-Myc transgenic mice. Specifically, H3K9me3 modifications or p53 mutations interfere with cellular senescence in lymphoma and cause cells to re-enter the cell cycle, which manifests as a stem cell-like function in lymphomas with the failure of chemotherapy-induced senescence (32). In addition, senescence-associated epigenetic genes mediated by H3K9me3 in mice helped successfully predict the prognosis of patients with lymphoma (92). Furthermore, RNA modifications may also influence stem cell function. For instance, increased m6A reader insulin-like growth factor 2 mRNA binding protein 2 expression determines the transcriptional level and function of hematopoietic stem cells (HSCs) (93).
Immunotherapeutic drugs regulate senescence-related lymphoma resistance
T-cell immune responses are compromised in ageing environments. B7-H1, a member of the B7-family, is highly expressed in older naïve CD8+ T cells and is considered to negatively regulate CD8+ T cells. In lymphoma immunotherapy, CD8+ CTLs are the main immune cells that attack tumours. The downregulation of CD8+ T cells in ageing environments is a critical reason for unsuccessful treatment (94). Adaptive T-cell immunity controls senescence-prone myeloid differentiation factor 88 (MyD88) or caspase recruitment domain-containing protein 11 (CARD11)-mutant B-cell lymphomas (95).
Programmed cell death protein 1 (PD-1) inhibitors are effective immunotherapeutic drugs that improve the prognosis of patients with relapsed/refractory lymphoma (96). Senescent cells express the immune checkpoint protein PD-1 ligand 1 (PD-L1). PD-L1-containing cells are sensitive to T-cell surveillance and exhibit resistance even in the presence of SASPs. Blocking PD-L1 and eliminating senescent cells using immune checkpoints represent promising therapeutic strategies (97). Furthermore, PD-1 is highly expressed in the effector memory T cells of older mice, exhibiting proliferative hyporesponsiveness (98). In aged mice, blocking the PD-1/PD-L1 pathway may restore T-cell function (99). Furthermore, a study investigating the reduction in immune-related adverse events (irAEs) caused by immune checkpoint blockade (ICB) reported that anti-PD-1 therapy induces irAE-like multiple organ dysfunction in older mice with tumours, but not in young mice. However, ICB-induced irAEs are mitigated by blocking CD4+ T cell-dependent IL-21 or CXCL13 activity and by B-cell depletion (100).
Chemotherapy and radiation therapy induce tumour-cell senescence and contribute to the generation of SASPs, which affect the immune response. Numerous senescent cells secrete excessive amounts of SASP, leading to drug resistance. Chimeric antigen receptor T-cell therapy is a new treatment option to overcome this resistance (101). However, the underlying mechanism remains to be fully elucidated.
Cytokines in the ageing microenvironment are a double-edged sword in lymphoma treatment. These are also regarded as part of the SASP. Specific CTLs lead to the emergence of drug-resistant cancer in the presence of IFN-γ in the TME. In E.G7-OVA murine T-cell lymphoma models, ERK-specific CTLs combine to release IFN-γ into the TME, resulting in the clonal proliferation of resistant cells. Based on these findings, scientists hypothesised that IFN-γ mediates CTL-dependent immunoediting. ERK-specific CTLs combined with IFN-γ alter the copy number of DNA damage response (53). IL-15 increases CD8+ T-cell function by delaying JAK3-STAT5-associated senescence (59). IL-6 reverses DOX resistance in nasal NKTCL by downregulating ABCC4 and inactivating the JAK2/STAT3/NF-κB/P65 pathway (60). Increased expression of IL-7 in aged mouse T-cell regions mediates the expansion of γδ 17 T-cell compartments, alters the cellular composition of the γδ T-cell pool in pLNs and leads to accelerated tumour growth (26).
Other senescence-related mechanisms in lymphoma resistance Autophagy
Ataxia-telangiectasia mutated (ATM) kinase has a pivotal role in DNA damage response and is a critical molecule in regulating the connection between autophagy and senescence. ATM promotes SASP formation by suppressing selective autophagy of GATA binding protein 4 (102,103). ATM has been identified in mantle cell lymphoma (MCL) as a mutated gene in the 11q22.2 region and engages in crucial interactions with E3 ubiquitin ligase in B cell lymphomas (104,105). ATM deletion induces cell malignancy (106).
Hypoxia-mediated molecules
Hypoxia induces tumour tolerance to radiotherapy and chemotherapy, and promotes malignant biological behaviours, such as tumour-cell growth, invasion and metastasis. The hypoxia-inducible factor 2α stemness pathway is regulated by MYC and mediated by Nanog and Sox2 to maintain a state of self-renewal in tumour stem cells. The stemness pathway also plays a crucial role in overcoming drug resistance (Fig. 2) (107).
Figure 2.
Mechanisms of senescence-associated lymphoma progression and drug resistance. Multiple mechanisms of lymphomagenesis and drug resistance participate in the ageing environment, including DNA damage caused by chemotherapy drugs, BTK inhibitor resistance mediated by signalling pathways, epigenetic regulation inducing TIS, immune regulation restraining drug resistance in lymphoma, autophagy mediating lymphoma progression, and hypoxia promoting lymphoma stem cell self-renewal. SASP, senescent-associated secretory phenotype; SA-β-gal, senescence-associated β-galactosidase; TIS, therapy-induced senescence; SIPS, stress-induced premature senescence; OIS, oncogene-induced senescence; Timp-1, tissue inhibitor of metalloproteinases-1; BTK, Bruton's tyrosine kinase; AK, aurora kinase; NHL, non-Hodgkin lymphoma; DNMTi, DNA methyltransferase inhibitors; EZH2, enhancer of zeste homolog 2; H3K9me3, trimethylation of histone H3 lysine 9; PD-L1, programmed cell death 1 ligand 1; ABCC4, ATP-binding cassette transporter 4; ATM, ataxia-telangiectasia mutated; E3, ubiquitin ligase; GATA4, GATA binding protein 4.
7. Treatment strategies for senescence-mediated lymphoma resistance
Treatment resistance often develops after multi-line chemotherapy. Senotherapeutics may represent a promising therapeutic strategy, as certain senescent cells are reprogrammed and exhibit resistance to treatment. The National Cancer Institute has proposed the 'one-two punch cancer therapy' strategy to first induce senescent cells and then clear such cells in the ageing environment of tumours. For individualised resistant lymphoma, cellular senescence drugs may be chosen in combination with other treatments. For instance, immunotherapy or epigenetic drugs combined with senolytics are more effective in clearing senescent cells (31,32). From the perspective of an ageing environment to overcome drug resistance in lymphoma, it may be concluded that these drugs provide more treatment options for patients with drug-resistant lymphoma.
Hsp90 inhibitors
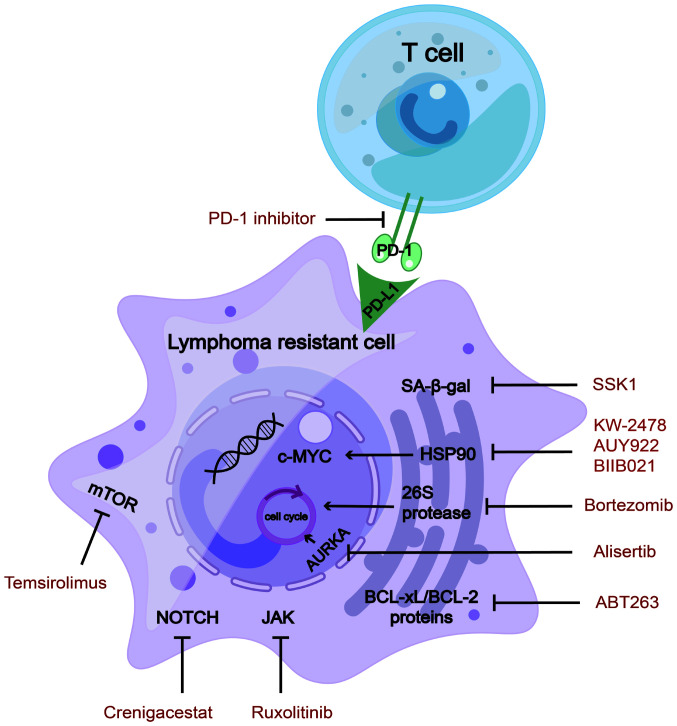
Heat shock protein 90 (Hsp90) is an oncogene that promotes c-MYC mRNA stability (108), regulates tumour immunology (109) and participates in aberrant metabolism of lymphoma cells (110). Hsp90 inhibitors act as novel senolytic agents (111) that have rapidly developed in preclinical research on lymphomas. They downregulate DNA replication in MCL, indicating a senolytic function (112). Hsp90 inhibitors sensitise lymphoma cells to the chemotherapeutic drugs cisplatin and the BTK inhibitor ibrutinib (113,114). BIIB021, a classic Hsp90 inhibitor, functions as a senolytic agent and overcame multidrug resistance in a preclinical study on lymphomas (115).
In 2015, a phase I clinical trial of KW-2478, a novel Hsp90 inhibitor, reported that 96% of patients with R/R B-cell malignancy temporarily achieved stable disease. Long-term survival outcomes were not assessed. The most common AEs were diarrhoea, fatigue, headache and hypertension. Most patients showed generally good tolerance (116). The phase II study of AUY922 enrolled 14 patients with DLBCL and 6 patients with PTCL. Across the whole group, the response rate stood at 10%, encompassing complete response (CR) in 7% of DLBCL cases and partial response (PR) in 17% of patients with PTCL. In the majority of cases, the toxicity profile appeared to be satisfactory (117).
Hsp90 inhibitors have demonstrated favorable efficacy and tolerable side effects in a subset of patients with R/R lymphoma, with significant interindividual variability. However, the majority of patients experience disease progression after administration, indicating that monotherapy with Hsp90 inhibitors may not promptly control disease progression, necessitating combination with other drugs to enhance therapeutic efficacy. However, as they are agents targeting cellular senescence, further investigation is needed to elucidate the therapeutic biomarkers and long-term survival rates in patients treated with Hsp90 inhibitors.
AK inhibitors
AK inhibitors are promising inducers of aneuploidy and senescence in patients with haematological malignancies. Aurora kinase A (AURKA) is a key enzyme involved in cell-cycle regulation (118), which may be applied to the first step in 'one-two punch therapy' to induce cellular senescence. In preclinical studies, AURKA was found to be overexpressed in various haematological malignancies, including DH/DE-DLBCL (118), multiple myeloma (MM) (119) and NKTCL (120). MK-8745 (a small-molecule AURKA inhibitor) significantly increased apoptosis of NKTCL cells and induced cell-cycle arrest (120).
Currently, AK inhibitors are in the clinical trial stage for relapsed/refractory lymphomas. In 2014, monotherapy with the AURKA inhibitor alisertib achieved a clinical response in a phase II clinical trial for a total of 48 patients, including those with aggressive B- and T-cell lymphomas. The objective response rate (ORR) of 48 patients in this study was 27%. Most common AEs were neutropenia and leukopenia (121). In a study from 2018, an AURKA inhibitor combined with rituximab or rituximab/vincristine showed synergistic effects in a phase I clinical trial of 45 patients in total with aggressive B-cell lymphoma. It is worth noting that among the 45 patients, the regimen only demonstrated activity in 20 patients with non-germinal center B-cell (non-GCB) DLBCL. Out of 20 patients, 9 (45%) showed a response (4 CRs, 5 PRs). The most common AE was neutropenia. Of note, all of the patients with non-GCB DLBCL showed good tolerance (122).
Overall, AK inhibitors are promising senolytics for the development of new therapies for patients with lymphoma. However, there is currently limited data on AK inhibitors specifically targeting subtypes of lymphoma, with numerous studies being descriptive clinical investigations lacking statistical and evidence-based medicine support (121,122). Further research is warranted to provide more comprehensive insight into this area.
Proteasome inhibitors
Bortezomib is a proteasome inhibitor and a senolytic agent. The primary mechanism of action of bortezomib is to suppress chymotrypsin-like sites in 26S protease, which then promotes cell-cycle arrest and, ultimately, apoptosis (123). Because endoplasmic reticulum (ER) homeostasis is strictly controlled, the level of ER stress beyond a certain point can change from a pro-survival pathway to a pro-apoptotic pathway, ultimately leading to ER stress-induced apoptosis. Bortezomib is a clinically available senolytic drug that targets ER homeostasis (62) and has been approved as a second-line treatment for MCL and MM (123,124).
Furthermore, in 2018, phase III clinical trials investigating bortezomib in 487 patients newly diagnosed with stage II-IV MCL reported a significantly prolonged median overall survival (OS) in the bortezomib treatment arms compared to those without bortezomib (90.7 vs. 55.7 months). AEs occurred in 42 and 57% of patients who died in the bortezomib and non-bortezomib groups, respectively, with progressive disease emerging as the predominant cause of mortality (125). In 2022, in a phase III clinical study, 209 patients with T-cell lymphoblastic lymphoma (T-LL) were included, comparing those not on bortezomib to those on bortezomib. The four-year OS rate of patients with T-LL stood at 89.5% when treated with bortezomib, in contrast to 78.3% in the absence of bortezomib. The general toxicity rates of grade 3 and above were comparable in both groups (126).
In summary, the safety profile of bortezomib was found to be well-managed and predictable. These findings provided evidence for the efficacy of bortezomib as an active therapeutic agent in MCL and T-LL.
Pathway inhibitors
Senescence-associated pathway inhibitors for the treatment of R/R lymphoma are currently undergoing clinical trials. Below, the pathway inhibitors currently under study in this field are summarised.
mTOR inhibitors
mTOR inhibitors have been comprehensively studied to extend the lifespan. mTOR affects T- and B-lymphocyte differentiation, function and cell death (127). In a phase I/II clinical trial investigating the mTOR inhibitor temsirolimus in combination with lenalidomide for R/R lymphomas, an ORR of 26% (13% CR) was achieved for patients with DLBCL. Among patients with classical HL (cHL), many of whom had relapsed following both brentuximab vedotin and autologous stem cell transplantation, the ORR was 80% (35% CR). Grade 3/4 hematologic AEs were frequent and included anemia, lymphopenia, neutropenia, thrombocytopenia and leucocytosis (128). The combination regimen involving temsirolimus and lenalidomide proved to be viable and showed promising efficacy in the treatment of lymphoma.
Notch pathway inhibitors
Advanced T-cell leukaemia and B-cell lymphoma may be accompanied by NOTCH1 mutations. Notch signalling cascades crosstalk with Wnt signalling and maintain stem-cell characteristics in tumour stem cells. Small molecule γ secretase inhibitors (such as AL101, MRK-560 and nirogacestat) and antibody-based biologics targeting Notch ligands or receptors (such as ABT-165, AMG 119, rovalpituzumab and tesirine) have been developed into drugs (57). The Notch pathway inhibitor crenigacestat was designed for use in a phase I clinical trial of R/R T-cell acute lymphoblastic leukemia (T-ALL) and T-cell lymphoblastic lymphoma. A total of 36 patients were enrolled, with a median event-free survival of 1.18 months. The most common AEs occurring in ≥30% of patients at any grade were diarrhea, vomiting and nausea (129). In summary, findings from this phase 1 trial suggest that, while there is some indication of clinical efficacy, the advancement of crenigacestat for T-ALL/T-LBL treatment may be constrained by gastrointestinal toxicities.
JAK/STAT inhibitors
The JAK/STAT signalling pathway is closely associated with senescence-related resistant lymphoma. SB1518 is a selective inhibitor of JAK2, a member of the JAK family, demonstrating its function as an antitumour agent in mutant JAK2-driven lymphocyte lines (130). A phase II clinical trial with the JAK inhibitor ruxolitinib was performed in 45 patients with PTCL and 7 patients with mycosis fungoides. The ORR and clinical benefit rate were 25 and 35%, respectively. AEs predominantly encompassed cytopenias, within the controllable range (131). In addition, a phase II clinical trial involving ruxolitinib was conducted for R/R cHL. The median progression-free survival was 3.6 months and the estimated 1-year OS rate was 50.6% (median OS not reached). A single grade IV AE (7.1%) indicated the toxicity was tolerated (132). In short, ruxolitinib monotherapy appears to have limited efficacy. Focusing on combined treatments is warranted to improve the efficiency and durability of responses.
Bcl-2 family protein inhibitors
The BCL-2 family of protein inhibitors has achieved success in the treatment of lymphoma. The use of these inhibitors has also been associated with senescence-associated drug resistance in lymphomas, particularly relapsed or refractory lymphomas. Bcl-2 family inhibitor navitoclax (ABT263, a BCL-xL/BCL-2 inhibitor) has senolytic functions in R/R ALL or LBL, and senescent bone marrow HSCs (133,134). In a phase I study of the selective BCL-2 inhibitor, the efficacy of venetoclax in combination with low-dose navitoclax and chemotherapy was evaluated in pediatric and adult patients with R/R ALL/LBL. The ORR was 60% and the treatment was well tolerated by patients (131). In conclusion, dual inhibition of BCL-2 and BCL-XL by venetoclax and narvesone in combination with chemotherapy is associated with significant response rates and a good safety profile, which is a suitable treatment option for R/R ALL or LBL.
Immunoadjuvant therapy
PD-1/PD-L1 inhibitors restore T-cell function in aged mice. From the perspective of the ageing microenvironment, PD-1 inhibitors used to treat relapsed and refractory T-cell lymphomas have achieved good treatment results in preclinical studies (99). In MyD88- or CARD11-mutant B-cell lymphomas, senescent tumour cells express high levels of PD-L1, thus mediating age-related immune evasion (95). In recent years, PD-1 inhibitors have been used to treat various relapsed or refractory lymphomas, such as R/R cHL (135) and R/R follicular lymphoma (136). However, PD-1 inhibitors targeting senescent lymphoma cells have not been studied in clinical trials.
Reversing T-cell senescence is another means of treating senescence-related drug-resistant cells. Studies have shown that T cells with mitochondrial dysfunction, owing to mitochondrial transcription factor A (TFAM) deficiency, can act as senescence accelerators. Blocking the TNF-α signalling pathway with nicotinamide adenine dinucleotide precursors may partially prevent premature senescence in TFAM-deficient mice (28). From the perspective of clinical transformation, the NAD+ precursor nicotinamide riboside is a promising drug to reverse immune-cell or lymphoma-cell senescence and may represent an emerging treatment for drug resistance in lymphoma.
Other promising senolytics
Alternative medicines, particularly Traditional Chinese Medicines (TCM), have an important role in lymphoma ageing-related drug resistance by regulating immune function, improving the TME and regulating antioxidant and anti-inflammatory pathways. However, TCM therapy requires an individualised regimen and should be combined with modern medical therapy regimens in clinical treatment to achieve the best therapeutic effect (137,138). For instance, dasatinib combined with quercetin as a classic senolytic agent is effective in the senescence-associated treatment of solid tumours (75,139,140). Dasatinib is a vital tyrosine kinase inhibitor used for targeted leukaemia and lymphoma therapy (141,142). Quercetin, a TCM drug, also exhibits anti-lymphoma effects (143). When combined with dasatinib, it may serve as an efficacious senolytic agent in patients with clinically resistant lymphoma. Further research on the use of TCM for the treatment of senescence-related drug resistance in lymphomas is necessary.
The β-gal-targeting drug SSK1 can selectively clear senescent cells and may represent a novel drug to intervene in senescence-related drug resistance in lymphoma cases (144).
Bromodomain and extra-terminal inhibitors (JQ1/RVX2135) have been found to synergise with ATR inhibitors (AZ20 and VE-821) to induce DNA damage, apoptosis, SASP pathways and ER stress in Myc-induced lymphoma cells in preclinical research (28) and represent promising senolytic agents for ageing-associated drug resistance in lymphoma treatment (Table I, Fig. 3).
Table I.
Applications of treatments in senescence-related lymphoma resistance.
| Classification | Drug name | Target | Phase | NCT number | Diseases | (Refs.) |
|---|---|---|---|---|---|---|
| Senolytics | KW-2478 AUY922 | Hsp90 inhibitor | Phase I/Phase II | NCT00457782/NCT01485536 | B cell malignancies/NHL | (116,117) |
| Bortezomib | Proteasome inhibitor | Phase III/Phase III | NCT00722137/NCT02112916 | MCL/T-LL | (124,125) | |
| Navitoclax (ABT263) | Bcl-2/Bcl-xL inhibitors | Phase I | NCT03181126 | R/R ALL or LL | (132) | |
| SSK1 | β-galactosidase | Preclinical | - | Targeting senescent cells | (143) | |
| JQ1/RVX2135 | BETi | Preclinical | - | Myc-induced lymphoma mice | (144) | |
| Alisertib | Aurora kinase A inhibitor | Phase I/II | NCT01397825/NCT00807495 | R/R aggressive B-cell and T-cell lymphoma | (121,122) | |
| Pathway inhibitors | Temsirolimus | mTOR inhibitor | Phase I/Phase II | NCT01076543 | R/R lymphoma (including DLBCL/FL/cHL) | (127) |
| Crenigacestat | NOTCH inhibitor | Phase I | NCT02518113 | R/R T-ALL/T-LBL | (128) | |
| Ruxolitinib | JAK inhibitor | Phase II/Phase II | NCT02974647/NCT02164500 | R/R PTCL/cHL | (130,131) | |
| Immunoadjuvant therapy | PD-1 inhibitor | PD-1 inhibitor | Preclinical | - | T-cell lymphomas/B-cell lymphomas | (95,99) |
| NAD+ precursor nicotinamide riboside | NAD+ precursor nicotinamide riboside | Preclinical | - | Targeting senescent T-cells | (28) |
R/R, relapsed or refractory; T-ALL, T-cell acute lymphoblastic leukemia; T-LL, T-cell lymphoblastic lymphoma; MCL, mantle cell lymphoma; DLBCL, diffuse large B-cell lymphoma; FL, follicular lymphoma; cHL, classical Hodgkin lymphoma; NHL, non-Hodgkin lymphoma; PTCL, peripheral T-cell lymphoma; HSP, heat shock protein; PD-1, programmed cell death 1; BETi, bromodomain and extra-terminal inhibitors; NCT, National Clinical Trials.
Figure 3.
Inhibitors that target senescence-related drug-resistant lymphoma cells. Manipulating the target inhibitors of SA-β-gal/HSP90/26S protease/AURKA/Bcl-2/Bcl-xL and mTOR/NOTCH/JAK pathway inhibitors has been shown to be effective in treating lymphoma-resistant cells. In the ageing environment of lymphoma, PD-1 inhibitors restrain the combination of T-cells and drug-resistant cells. SA-β-gal, senescence-associated β-galactosidase; HSP, heat shock protein; PD-1, programmed cell death 1; PD-L1, PD-1 ligand 1; AURKA, aurora kinase A.
8. Conclusion and perspectives
In the application of the 'one-two punch cancer therapy', it is necessary to distinguish the differentiation outcome of senescent cells at different periods. In the early stages, cells gradually become senescent and move towards cell death (through processes such as apoptosis, ferroptosis and cuproptosis). In later stages, a large number of senescent cells form an ageing microenvironment and move towards treatment resistance through SASP reprogramming. The senescent messaging secretome has an important role in the mechanisms underlying chemotherapy/radiotherapy, targeted therapy and immunotherapy resistance in lymphomas. In the early stages, senescent cells can be treated with pro-cellular senescence drugs, and in the later stages, drug-resistant senescent cells can be removed using senolytics or SASP inhibitors in conjunction with SASP reprogramming. Furthermore, senolytics combined with immunotherapy and epigenetic drugs may be more effective for patients with relapsed or refractory lymphoma.
It should be noted that the type of lymphoma and specific circumstances of the patient may influence the choice of treatment. Therefore, the treatment of lymphoma often requires individualised and condition-based decision-making, including the consideration of factors such as senescence-related drug resistance. Currently, the development of several senolytic drugs is limited to preclinical studies and clinical trials. The heterogeneity of senescent cells is a major obstacle to their clinical transformation. However, the development of senescence-associated targeted drugs may lead to important breakthroughs in overcoming lymphoma drug resistance.
Acknowledgments
Not applicable.
Funding Statement
This study was supported by the Henan Provincial Science and Technology Research Project (grant no. SBGJ202001008), the National Natural Science Foundation of China (grant no. 82070210) and the Outstanding Youth Item of the Henan Health-Related Technological and Innovative Talents Project (grant no. 11459).
Availability of data and materials
Not applicable.
Authors' contributions
Investigation: YZ, JC, QH and SQ. Software: YG and MM. Supervision: MZ, XZ and QC. Writing-original draft: YZ and JC. Writing-review and editing: QH, SQ, ZW, QY, WS, LD, ZS, XZ and QC. All of the authors have read and approved the final version of the manuscript. Data authentication is not applicable.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Di Micco R, Krizhanovsky V, Baker D, d'Adda di Fagagna F. Cellular senescence in ageing: From mechanisms to therapeutic opportunities. Nat Rev Mol Cell Biol. 2021;22:75–95. doi: 10.1038/s41580-020-00314-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Munoz-Espin D, Serrano M. Cellular senescence: From physiology to pathology. Nat Rev Mol Cell Biol. 2014;15:482–496. doi: 10.1038/nrm3823. [DOI] [PubMed] [Google Scholar]
- 3.Hernandez-Segura A, Nehme J, Demaria M. Hallmarks of cellular senescence. Trends Cell Biol. 2018;28:436–453. doi: 10.1016/j.tcb.2018.02.001. [DOI] [PubMed] [Google Scholar]
- 4.Hu D, Yuan S, Zhong J, Liu Z, Wang Y, Liu L, Li J, Wen F, Liu J, Zhang J. Cellular senescence and hematological malignancies: From pathogenesis to therapeutics. Pharmacol Ther. 2021;223:107817. doi: 10.1016/j.pharmthera.2021.107817. [DOI] [PubMed] [Google Scholar]
- 5.Fane M, Weeraratna AT. How the ageing microenvironment influences tumour progression. Nat Rev Cancer. 2020;20:89–106. doi: 10.1038/s41568-019-0222-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Davalos AR, Coppe JP, Campisi J, Desprez PY. Senescent cells as a source of inflammatory factors for tumor progression. Cancer Metastasis Rev. 2010;29:273–283. doi: 10.1007/s10555-010-9220-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hao X, Wang C, Zhang R. Chromatin basis of the senescence-associated secretory phenotype. Trends Cell Biol. 2022;32:513–526. doi: 10.1016/j.tcb.2021.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yin Y, Chen H, Wang Y, Zhang L, Wang X. Roles of extracellular vesicles in the aging microenvironment and age-related diseases. J Extracell Vesicles. 2021;10:e12154. doi: 10.1002/jev2.12154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Newell GR, Spitz MR, Sider JG. Cancer and age. Semin Oncol. 1989;16:3–9. [PubMed] [Google Scholar]
- 10.Gopas J, Stern E, Zurgil U, Ozer J, Ben-Ari A, Shubinsky G, Braiman A, Sinay R, Ezratty J, Dronov V, et al. Reed-Sternberg cells in Hodgkin's lymphoma present features of cellular senescence. Cell Death Dis. 2016;7:e2457. doi: 10.1038/cddis.2016.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Aouali N, Eddabra L, Macadre J, Morjani H. Immunosuppressors and reversion of multidrug-resistance. Crit Rev Oncol Hematol. 2005;56:61–70. doi: 10.1016/j.critrevonc.2004.12.010. [DOI] [PubMed] [Google Scholar]
- 12.Kang YK, Zhan Z, Regis J, Alvarez M, Robey R, Meadows B, Dickstein B, Lee JS, Otsuki T, Stetler-Stevenson M, et al. Expression of mdr-1 in refractory lymphoma: quantitation by polymerase chain reaction and validation of the assay. Blood. 1995;86:1515–1524. [PubMed] [Google Scholar]
- 13.Karai E, Szebenyi K, Windt T, Feher S, Szendi E, Dekay V, Vajdovich P, Szakacs G, Furedi A. Celecoxib prevents doxorubicin-induced multidrug resistance in canine and mouse lymphoma cell lines. Cancers (Basel) 2020;12:1117. doi: 10.3390/cancers12051117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang X, Fu X, Dong M, Yang Z, Wu S, Ma M, Li Z, Wang X, Li L, Li X, et al. Conserved cell populations in doxorubicin-resistant human nasal natural killer/T cell lymphoma cell line: Super multidrug resistant cells? Cancer Cell Int. 2018;18:150. doi: 10.1186/s12935-018-0644-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.De Blander H, Morel AP, Senaratne AP, Ouzounova M, Puisieux A. Cellular plasticity: A route to senescence exit and tumorigenesis. Cancers (Basel) 2021;13:4561. doi: 10.3390/cancers13184561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hanahan D. Hallmarks of cancer: New dimensions. Cancer Discov. 2022;12:31–46. doi: 10.1158/2159-8290.CD-21-1059. [DOI] [PubMed] [Google Scholar]
- 17.Chien Y, Scuoppo C, Wang X, Fang X, Balgley B, Bolden JE, Premsrirut P, Luo W, Chicas A, Lee CS, et al. Control of the senescence-associated secretory phenotype by NF-κB promotes senescence and enhances chemosensitivity. Genes Dev. 2011;25:2125–2136. doi: 10.1101/gad.17276711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gilbert LA ands Hemann MT. DNA damage-mediated induction of a chemoresistant niche. Cell. 2010;143:355–366. doi: 10.1016/j.cell.2010.09.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Frasca D, Diaz A, Romero M, Blomberg BB. Human peripheral late/exhausted memory B cells express a senescent-associated secretory phenotype and preferentially utilize metabolic signaling pathways. Exp Gerontol. 2017;87(Pt A):113–120. doi: 10.1016/j.exger.2016.12.001. [DOI] [PubMed] [Google Scholar]
- 20.Riley RL, Khomtchouk K, Blomberg BB. Age-associated B cells (ABC) inhibit B lymphopoiesis and alter antibody repertoires in old age. Cell Immunol. 2017;321:61–67. doi: 10.1016/j.cellimm.2017.04.008. [DOI] [PubMed] [Google Scholar]
- 21.Jia X, Bene J, Balazs N, Szabo K, Berta G, Herczeg R, Gyenesei A, Balogh P. Age-Associated B cell features of the murine high-grade B Cell Lymphoma Bc.DLFL1 and its extranodal expansion in abdominal adipose tissues. J Immunol. 2022;208:2866–2876. doi: 10.4049/jimmunol.2100956. [DOI] [PubMed] [Google Scholar]
- 22.Han S, Georgiev P, Ringel AE, Sharpe AH, Haigis MC. Age-associated remodeling of T cell immunity and metabolism. Cell Metab. 2023;35:36–55. doi: 10.1016/j.cmet.2022.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Huang X, Bai X, Cao Y, Wu J, Huang M, Tang D, Tao S, Zhu T, Liu Y, Yang Y, et al. Lymphoma endothelium preferentially expresses Tim-3 and facilitates the progression of lymphoma by mediating immune evasion. J Exp Med. 2010;207:505–520. doi: 10.1084/jem.20090397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhang Y, Joe G, Hexner E, Zhu J, Emerson SG. Host-reactive CD8+ memory stem cells in graft-versus-host disease. Nat Med. 2005;11:1299–1305. doi: 10.1038/nm1326. [DOI] [PubMed] [Google Scholar]
- 25.Zhou B, Zhao Z, Zhang X, Deng W, Li Y. Effect of allogenic bone marrow mesenchymal stem cell transplantation on t cells of old mice. Cell Reprogram. 2020;22:30–35. doi: 10.1089/cell.2019.0055. [DOI] [PubMed] [Google Scholar]
- 26.Chen HC, Eling N, Martinez-Jimenez CP, O'Brien LM, Carbonaro V, Marioni JC, Odom DT, de la Roche M. IL-7-dependent compositional changes within the γδ T cell pool in lymph nodes during ageing lead to an unbalanced anti-tumour response. EMBO Rep. 2019;20:e47379. doi: 10.15252/embr.201847379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Crespo J, Sun H, Welling TH, Tian Z, Zou W. T cell anergy, exhaustion, senescence, and stemness in the tumor microenvironment. Curr Opin Immunol. 2013;25:214–221. doi: 10.1016/j.coi.2012.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Desdin-Mico G, Soto-Heredero G, Aranda JF, Oller J, Carrasco E, Gabande-Rodriguez E, Blanco EM, Alfranca A, Cusso L, Desco M, et al. T cells with dysfunctional mitochondria induce multimorbidity and premature senescence. Science. 2020;368:1371–1376. doi: 10.1126/science.aax0860. [DOI] [PubMed] [Google Scholar]
- 29.Manser AR, Uhrberg M. Age-related changes in natural killer cell repertoires: Impact on NK cell function and immune surveillance. Cancer Immunol Immunother. 2016;65:417–426. doi: 10.1007/s00262-015-1750-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Edwards ESJ, Bier J, Cole TS, Wong M, Hsu P, Berglund LJ, Boztug K, Lau A, Gostick E, Price DA, et al. Activating PIK3CD mutations impair human cytotoxic lymphocyte differentiation and function and EBV immunity. J Allergy Clin Immunol. 2019;143:276–291.e6. doi: 10.1016/j.jaci.2018.04.030. [DOI] [PubMed] [Google Scholar]
- 31.Prasanna PG, Citrin DE, Hildesheim J, Ahmed MM, Venkatachalam S, Riscuta G, Xi D, Zheng G, Deursen JV, Goronzy J, et al. Therapy-Induced Senescence: Opportunities to improve anticancer therapy. J Natl Cancer Inst. 2021;113:1285–1298. doi: 10.1093/jnci/djab064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Milanovic M, Fan DNY, Belenki D, Dabritz JHM, Zhao Z, Yu Y, Dorr JR, Dimitrova L, Lenze D, Monteiro Barbosa IA, et al. Senescence-associated reprogramming promotes cancer stemness. Nature. 2018;553:96–100. doi: 10.1038/nature25167. [DOI] [PubMed] [Google Scholar]
- 33.Wang J, Tao Q, Pan Y, Wanyan Z, Zhu F, Xu X, Wang H, Yi L, Zhou M, Zhai Z. Stress-induced premature senescence activated by the SENEX gene mediates apoptosis resistance of diffuse large B-cell lymphoma via promoting immunosuppressive cells and cytokines. Immun Inflamm Dis. 2020;8:672–683. doi: 10.1002/iid3.356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chen LS, Balakrishnan K, Gandhi V. Inflammation and survival pathways: Chronic lymphocytic leukemia as a model system. Biochem Pharmacol. 2010;80:1936–1945. doi: 10.1016/j.bcp.2010.07.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Birch J, Gil J. Senescence and the SASP: Many therapeutic avenues. Genes Dev. 2020;34:1565–1576. doi: 10.1101/gad.343129.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Braig M, Lee S, Loddenkemper C, Rudolph C, Peters AH, Schlegelberger B, Stein H, Dorken B, Jenuwein T, Schmitt CA. Oncogene-induced senescence as an initial barrier in lymphoma development. Nature. 2005;436:660–665. doi: 10.1038/nature03841. [DOI] [PubMed] [Google Scholar]
- 37.Dorr JR, Yu Y, Milanovic M, Beuster G, Zasada C, Dabritz JH, Lisec J, Lenze D, Gerhardt A, Schleicher K, et al. Synthetic lethal metabolic targeting of cellular senescence in cancer therapy. Nature. 2013;501:421–425. doi: 10.1038/nature12437. [DOI] [PubMed] [Google Scholar]
- 38.Scafuro M, Capasso L, Carafa V, Altucci L, Nebbioso A. Gene Transactivation and Transrepression in MYC-Driven Cancers. Int J Mol Sci. 2021;22:3458. doi: 10.3390/ijms22073458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ben-Porath I, Thomson MW, Carey VJ, Ge R, Bell GW, Regev A, Weinberg RA. An embryonic stem cell-like gene expression signature in poorly differentiated aggressive human tumors. Nat Genet. 2008;40:499–507. doi: 10.1038/ng.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cao J, Li L, Chen C, Lv C, Meng F, Zeng L, Li Z, Wu Q, Zhao K, Pan B, et al. RNA interference-mediated silencing of NANOG leads to reduced proliferation and self-renewal, cell cycle arrest and apoptosis in T-cell acute lymphoblastic leukemia cells via the p53 signaling pathway. Leuk Res. 2013;37:1170–1177. doi: 10.1016/j.leukres.2013.04.021. [DOI] [PubMed] [Google Scholar]
- 41.Reimann M, Lee S, Loddenkemper C, Dorr JR, Tabor V, Aichele P, Stein H, Dorken B, Jenuwein T, Schmitt CA. Tumor stroma-derived TGF-beta limits myc-driven lymphomagenesis via Suv39h1-dependent senescence. Cancer Cell. 2010;17:262–272. doi: 10.1016/j.ccr.2009.12.043. [DOI] [PubMed] [Google Scholar]
- 42.Moore AR, Rosenberg SC, McCormick F, Malek S. RAS-targeted therapies: Is the undruggable drugged? Nat Rev Drug Discov. 2020;19:533–552. doi: 10.1038/s41573-020-0068-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Harrell Stewart DR, Clark GJ. Pumping the brakes on RAS-negative regulators and death effectors of RAS. J Cell Sci. 2020;133:jcs238865. doi: 10.1242/jcs.238865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Moiseeva O, Guillon J, Ferbeyre G. Senescence: A program in the road to cell elimination and cancer. Semin Cancer Biol. 2022;81:48–53. doi: 10.1016/j.semcancer.2020.12.017. [DOI] [PubMed] [Google Scholar]
- 45.Caceres-Gutierrez RE, Alfaro-Mora Y, Andonegui MA, Diaz-Chavez J, Herrera LA. The influence of oncogenic RAS on chemotherapy and radiotherapy resistance through DNA repair pathways. Front Cell Dev Biol. 2022;10:751367. doi: 10.3389/fcell.2022.751367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Punekar SR, Velcheti V, Neel BG, Wong KK. The current state of the art and future trends in RAS-targeted cancer therapies. Nat Rev Clin Oncol. 2022;19:637–655. doi: 10.1038/s41571-022-00671-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Coppe JP, Patil CK, Rodier F, Sun Y, Munoz DP, Goldstein J, Nelson PS, Desprez PY, Campisi J. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008;6:2853–2868. doi: 10.1371/journal.pbio.0060301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zhang X, Zhao L, Li X, Wang X, Li L, Fu X, Sun Z, Li Z, Nan F, Chang Y, Zhang M. ATP-binding cassette sub-family C member 4 (ABCC4) is overexpressed in human NK/T-cell lymphoma and regulates chemotherapy sensitivity: Potential as a functional therapeutic target. Leuk Res. 2015;39:1448–1454. doi: 10.1016/j.leukres.2015.10.001. [DOI] [PubMed] [Google Scholar]
- 49.Han L, Long Q, Li S, Xu Q, Zhang B, Dou X, Qian M, Jiramongkol Y, Guo J, Cao L, et al. Senescent stromal cells promote cancer resistance through SIRT1 Loss-potentiated overproduction of small extracellular vesicles. Cancer Res. 2020;80:3383–3398. doi: 10.1158/0008-5472.CAN-20-0506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Yano H, Fujiwara Y, Horlad H, Pan C, Kai K, Niino D, Ohsawa K, Higashi M, Nosaka K, Okuno Y, et al. Blocking cholesterol efflux mechanism is a potential target for antilymphoma therapy. Cancer Sci. 2022;113:2129–2143. doi: 10.1111/cas.15349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wu S, Zhang X, Dong M, Yang Z, Zhang M, Chen Q. sATP-binding cassette subfamily G member 2 enhances the multidrug resistance properties of human nasal natural killer/T cell lymphoma side population cells. Oncol Rep. 2020;44:1467–1478. doi: 10.3892/or.2020.7722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhang Y, Wang X. Targeting the Wnt/β-catenin signaling pathway in cancer. J Hematol Oncol. 2020;13:165. doi: 10.1186/s13045-020-00990-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Takeda K, Nakayama M, Hayakawa Y, Kojima Y, Ikeda H, Imai N, Ogasawara K, Okumura K, Thomas DM, Smyth MJ. IFN-gamma is required for cytotoxic T cell-dependent cancer genome immunoediting. Nat Commun. 2017;8:14607. doi: 10.1038/ncomms14607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Islam S, Qi W, Morales C, Cooke L, Spier C, Weterings E, Mahadevan D. Disruption of aneuploidy and senescence induced by aurora inhibition promotes intrinsic apoptosis in double hit or double expressor diffuse large B-cell lymphomas. Mol Cancer Ther. 2017;16:2083–2093. doi: 10.1158/1535-7163.MCT-17-0089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Tewari D, Patni P, Bishayee A, Sah AN, Bishayee A. Natural products targeting the PI3K-Akt-mTOR signaling pathway in cancer: A novel therapeutic strategy. Semin Cancer Biol. 2022;80:1–17. doi: 10.1016/j.semcancer.2019.12.008. [DOI] [PubMed] [Google Scholar]
- 56.Pi M, Kuang H, Yue C, Yang Q, Wu A, Li Y, Assaraf YG, Yang DH, Wu S. Targeting metabolism to overcome cancer drug resistance: A promising therapeutic strategy for diffuse large B cell lymphoma. Drug Resist Updat. 2022;61:100822. doi: 10.1016/j.drup.2022.100822. [DOI] [PubMed] [Google Scholar]
- 57.Katoh M, Katoh M. Precision medicine for human cancers with Notch signaling dysregulation (Review) Int J Mol Med. 2020;45:279–297. doi: 10.3892/ijmm.2019.4418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Patil K, Sher G, Kuttikrishnan S, Moton S, Alam M, Buddenkotte J, Ahmad A, Steinhoff M, Uddin S. The cross-talk between miRNAs and JAK/STAT pathway in cutaneous T cell lymphoma: Emphasis on therapeutic opportunities. Semin Cell Dev Biol. 2024;154(Pt C):239–249. doi: 10.1016/j.semcdb.2022.09.015. [DOI] [PubMed] [Google Scholar]
- 59.Weng J, Moriarty KE, Baio FE, Chu F, Kim SD, He J, Jie Z, Xie X, Ma W, Qian J, et al. IL-15 enhances the antitumor effect of human antigen-specific CD8(+) T cells by cellular senescence delay. Oncoimmunology. 2016;5:e1237327. doi: 10.1080/2162402X.2016.1237327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wang L, Li LR, Zhang L, Wang JW. The landscape of new drugs in extranodal NK/T-cell lymphoma. Cancer Treat Rev. 2020;89:102065. doi: 10.1016/j.ctrv.2020.102065. [DOI] [PubMed] [Google Scholar]
- 61.Ge H, Ke J, Xu N, Li H, Gong J, Li X, Song Y, Zhu H, Bai C. Dexamethasone alleviates pemetrexed-induced senescence in non-small-cell lung cancer. Food Chem Toxicol. 2018;119:86–97. doi: 10.1016/j.fct.2018.05.025. [DOI] [PubMed] [Google Scholar]
- 62.Ei ZZ, Choochuay K, Tubsuwan A, Pinkaew D, Suksomtip M, Vinayanuwattikun C, Chanvorachote P, Chunhacha P. GRP78/BiP determines senescence evasion cell fate after cisplatin-based chemotherapy. Sci Rep. 2021;11:22448. doi: 10.1038/s41598-021-01540-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Robbins PD, Jurk D, Khosla S, Kirkland JL, LeBrasseur NK, Miller JD, Passos JF, Pignolo RJ, Tchkonia T, Niedernhofer LJ. Senolytic Drugs: Reducing senescent cell viability to extend health Span. Annu Rev Pharmacol Toxicol. 2021;61:779–803. doi: 10.1146/annurev-pharmtox-050120-105018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Azim HA, Jr, Pruneri G, Raviele PR, Steffanoni S, Martinelli G, Peccatori FA. ERCC1 Expression in Diffuse Large B-Cell lymphoma patients treated with a cisplatin-based regimen : A brief communication. J Egypt Natl Canc Inst. 2007;19:176–177. [PubMed] [Google Scholar]
- 65.Lee H. Impaired phosphorylation and mis-localization of Bub1 and BubR1 are responsible for the defective mitotic checkpoint function in Brca2-mutant thymic lymphomas. Exp Mol Med. 2003;35:448–453. doi: 10.1038/emm.2003.58. [DOI] [PubMed] [Google Scholar]
- 66.Moncsek A, Al-Suraih MS, Trussoni CE, O'Hara SP, Splinter PL, Zuber C, Patsenker E, Valli PV, Fingas CD, Weber A, et al. Targeting senescent cholangiocytes and activated fibroblasts with B-cell lymphoma-extra large inhibitors ameliorates fibrosis in multidrug resistance 2 gene knockout (Mdr2(-/-)) mice. Hepatology. 2018;67:247–259. doi: 10.1002/hep.29464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Matthe DM, Thoma OM, Sperka T, Neurath MF, Waldner MJ. Telomerase deficiency reflects age-associated changes in CD4+ T cells. Immun Ageing. 2022;19:16. doi: 10.1186/s12979-022-00273-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Reimann M, Loddenkemper C, Rudolph C, Schildhauer I, Teichmann B, Stein H, Schlegelberger B, Dorken B, Schmitt CA. The Myc-evoked DNA damage response accounts for treatment resistance in primary lymphomas in vivo. Blood. 2007;110:2996–3004. doi: 10.1182/blood-2007-02-075614. [DOI] [PubMed] [Google Scholar]
- 69.Vidal-Crespo A, Matas-Cespedes A, Rodriguez V, Rossi C, Valero JG, Serrat N, Sanjuan-Pla A, Menendez P, Roue G, Lopez-Guillermo A, et al. Daratumumab displays in vitro and in vivo anti-tumor activity in models of B-cell non-Hodgkin lymphoma and improves responses to standard chemo-immunotherapy regimens. Haematologica. 2020;105:1032–1041. doi: 10.3324/haematol.2018.211904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Fontan L, Goldstein R, Casalena G, Durant M, Teater MR, Wilson J, Phillip J, Xia M, Shah S, Us I, et al. Identification of MALT1 feedback mechanisms enables rational design of potent antilymphoma regimens for ABC-DLBCL. Blood. 2021;137:788–800. doi: 10.1182/blood.2019004713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kim S, Shah SB, Graney PL, Singh A. Multiscale engineering of immune cells and lymphoid organs. Nat Rev Mater. 2019;4:355–378. doi: 10.1038/s41578-019-0100-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Tian YF, Ahn H, Schneider RS, Yang SN, Roman-Gonzalez L, Melnick AM, Cerchietti L, Singh A. Integrin-specific hydrogels as adaptable tumor organoids for malignant B and T cells. Biomaterials. 2015;73:110–119. doi: 10.1016/j.biomaterials.2015.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ceccato J, Piazza M, Pizzi M, Manni S, Piazza F, Caputo I, Cinetto F, Pisoni L, Trojan D, Scarpa R, et al. A bone-based 3D scaffold as an in-vitro model of microenvironment-DLBCL lymphoma cell interaction. Front Oncol. 2022;12:947823. doi: 10.3389/fonc.2022.947823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Shah SB, Carlson CR, Lai K, Zhong Z, Marsico G, Lee KM, Felix Velez NE, Abeles EB, Allam M, Hu T, et al. Combinatorial treatment rescues tumour-microenvironment-mediated attenuation of MALT1 inhibitors in B-cell lymphomas. Nat Mater. 2023;22:511–523. doi: 10.1038/s41563-023-01495-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Wyld L, Bellantuono I, Tchkonia T, Morgan J, Turner O, Foss F, George J, Danson S, Kirkland JL. Senescence and cancer: A review of clinical implications of senescence and senotherapies. Cancers (Basel) 2020;12:2134. doi: 10.3390/cancers12082134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Wang B, Li XQ, Ma X, Hong X, Lu H, Guo Y. Immunohistochemical expression and clinical significance of P-glycoprotein in previously untreated extranodal NK/T-cell lymphoma, nasal type. Am J Hematol. 2008;83:795–799. doi: 10.1002/ajh.21256. [DOI] [PubMed] [Google Scholar]
- 77.Wang J, Wang Z, Wang H, Wanyan Z, Pan Y, Zhu F, Tao Q, Zhai Z. Stress-Induced premature senescence promotes proliferation by activating the SENEX and p16(INK4a)/Retinoblastoma (Rb) pathway in diffuse large B-Cell lymphoma. Turk J Haematol. 2019;36:247–254. doi: 10.4274/tjh.galenos.2019.2019.0117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Nguyen PH, Niesen E, Hallek M. New roles for B cell receptor associated kinases: when the B cell is not the target. Leukemia. 2019;33:576–587. doi: 10.1038/s41375-018-0366-8. [DOI] [PubMed] [Google Scholar]
- 79.Alu A, Lei H, Han X, Wei Y, Wei X. BTK inhibitors in the treatment of hematological malignancies and inflammatory diseases: Mechanisms and clinical studies. J Hematol Oncol. 2022;15:138. doi: 10.1186/s13045-022-01353-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Chibaya L, Snyder J, Ruscetti M. Senescence and the tumor-immune landscape: Implications for cancer immunotherapy. Semin Cancer Biol. 2022;86(Pt 3):827–845. doi: 10.1016/j.semcancer.2022.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Wang X, Zhang L, Liu X, Li X, Li L, Fu X, Sun Z, Wu J, Zhang X, Yan J, et al. Efficacy and safety of a pegasparaginase-based chemotherapy regimen vs an L-asparaginase-Based chemotherapy regimen for newly diagnosed advanced extranodal natural Killer/T-Cell lymphoma: A randomized clinical trial. JAMA Oncol. 2022;8:1035–1041. doi: 10.1001/jamaoncol.2022.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Kidd JG. Regression of transplanted lymphomas induced in vivo by means of normal guinea pig serum. II. Studies on the nature of the active serum constituent: histological mechanism of the regression: tests for effects of guinea pig serum on lymphoma cells in vitro: Discussion. J Exp Med. 1953;98:583–606. doi: 10.1084/jem.98.6.583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Krall AS, Xu S, Graeber TG, Braas D, Christofk HR. Asparagine promotes cancer cell proliferation through use as an amino acid exchange factor. Nat Commun. 2016;7:11457. doi: 10.1038/ncomms11457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Wang L, Yang J, Wang HN, Fu RY, Liu XD, Piao YS, Wei LQ, Wang JW, Zhang L. LncRNA BCYRN1-induced autophagy enhances asparaginase resistance in extranodal NK/T-cell lymphoma. Theranostics. 2021;11:925–940. doi: 10.7150/thno.46655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Zhdanov DD, Pokrovsky VS, Pokrovskaya MV, Alexandrova SS, Eldarov MA, Grishin DV, Basharov MM, Gladilina YA, Podobed OV, Sokolov NN. Inhibition of telomerase activity and induction of apoptosis by Rhodospirillum rubrum L-asparaginase in cancer Jurkat cell line and normal human CD4+ T lymphocytes. Cancer Med. 2017;6:2697–2712. doi: 10.1002/cam4.1218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Zhdanov DD, Pokrovsky VS, Pokrovskaya MV, Alexandrova SS, Eldarov MA, Grishin DV, Basharov MM, Gladilina YA, Podobed OV, Sokolov NN. Rhodospirillum rubruml-asparaginase targets tumor growth by a dual mechanism involving telomerase inhibition. Biochem Biophys Res Commun. 2017;492:282–288. doi: 10.1016/j.bbrc.2017.08.078. [DOI] [PubMed] [Google Scholar]
- 87.Hassler MR, Klisaroska A, Kollmann K, Steiner I, Bilban M, Schiefer AI, Sexl V, Egger G. Antineoplastic activity of the DNA methyltransferase inhibitor 5-aza-2'-deoxycytidine in anaplastic large cell lymphoma. Biochimie. 2012;94:2297–2307. doi: 10.1016/j.biochi.2012.05.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Arosio G, Sharma GG, Villa M, Mauri M, Crespiatico I, Fonta na D, Ma nf roni C, Mastini C, Zappa M, Magistroni V, et al. Synergistic drug combinations prevent resistance in ALK+ anaplastic large cell lymphoma. Cancers (Basel) 2021;13:4422. doi: 10.3390/cancers13174422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Lund K, Adams PD, Copland M. EZH2 in normal and malignant hematopoiesis. Leukemia. 2014;28:44–49. doi: 10.1038/leu.2013.288. [DOI] [PubMed] [Google Scholar]
- 90.Baell JB, Leaver DJ, Hermans SJ, Kelly GL, Brennan MS, Downer NL, Nguyen N, Wichmann J, McRae HM, Yang Y, et al. Inhibitors of histone acetyltransferases KAT6A/B induce senescence and arrest tumour growth. Nature. 2018;560:253–257. doi: 10.1038/s41586-018-0387-5. [DOI] [PubMed] [Google Scholar]
- 91.Huang F. New KAT6 inhibitors induce senescence and arrest cancer growth. Synth Syst Biotechnol. 2018;3:244–245. doi: 10.1016/j.synbio.2018.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Schleich K, Kase J, Dorr JR, Trescher S, Bhattacharya A, Yu Y, Wailes EM, Fan DNY, Lohneis P, Milanovic M, et al. H3K9me3-mediated epigenetic regulation of senescence in mice predicts outcome of lymphoma patients. Nat Commun. 2020;11:3651. doi: 10.1038/s41467-020-17467-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Yin R, Chang J, Li Y, Gao Z, Qiu Q, Wang Q, Han G, Chai J, Feng M, Wang P, et al. Differential m(6)A RNA landscapes across hematopoiesis reveal a role for IGF2BP2 in preserving hematopoietic stem cell function. Cell Stem Cell. 2022;29:149–159 e7. doi: 10.1016/j.stem.2021.09.014. [DOI] [PubMed] [Google Scholar]
- 94.Mirza N, Duque MA, Dominguez AL, Schrum AG, Dong H, Lustgarten J. B7-H1 expression on old CD8+ T cells negatively regulates the activation of immune responses in aged animals. J Immunol. 2010;184:5466–5474. doi: 10.4049/jimmunol.0903561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Reimann M, Schrezenmeier J, Richter-Pechanska P, Dolnik A, Hick TP, Schleich K, Cai X, Fan DNY, Lohneis P, Masswig S, et al. Adaptive T-cell immunity controls senescence-prone MyD88- or CARD11-mutant B-cell lymphomas. Blood. 2021;137:2785–2799. doi: 10.1182/blood.2020005244. [DOI] [PubMed] [Google Scholar]
- 96.Kline J, Godfrey J, Ansell SM. The immune landscape and response to immune checkpoint blockade therapy in lymphoma. Blood. 2020;135:523–533. doi: 10.1182/blood.2019000847. [DOI] [PubMed] [Google Scholar]
- 97.Wang TW, Johmura Y, Suzuki N, Omori S, Migita T, Yamaguchi K, Hatakeyama S, Yamazaki S, Shimizu E, Imoto S, et al. Blocking PD-L1-PD-1 improves senescence surveillance and ageing phenotypes. Nature. 2022;611:358–364. doi: 10.1038/s41586-022-05388-4. [DOI] [PubMed] [Google Scholar]
- 98.Shimada Y, Hayashi M, Nagasaka Y, Ohno-Iwashita Y, Inomata M. Age-associated up-regulation of a negative co-stimulatory receptor PD-1 in mouse CD4+ T cells. Exp Gerontol. 2009;44:517–522. doi: 10.1016/j.exger.2009.05.003. [DOI] [PubMed] [Google Scholar]
- 99.Lages CS, Lewkowich I, Sproles A, Wills-Karp M, Chougnet C. Partial restoration of T-cell function in aged mice by in vitro blockade of the PD-1/PD-L1 pathway. Aging Cell. 2010;9:785–798. doi: 10.1111/j.1474-9726.2010.00611.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Tsukamoto H, Komohara Y, Tomita Y, Miura Y, Motoshima T, Imamura K, Kimura T, Ikeda T, Fujiwara Y, Yano H, et al. Aging-associated and CD4 T-cell-dependent ectopic CXCL13 activation predisposes to anti-PD-1 therapy-induced adverse events. Proc Natl Acad Sci USA. 2022;119:e2205378119. doi: 10.1073/pnas.2205378119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Etxebeste-Mitxeltorena M, Del Rincon-Loza I, Martin-Antonio B. Tumor secretome to adoptive cellular immunotherapy: Reduce me before I make you my partner. Front Immunol. 2021;12:717850. doi: 10.3389/fimmu.2021.717850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Stagni V, Ferri A, Cirotti C, Barila D. ATM kinase-dependent regulation of autophagy: A key player in senescence? Front Cell Dev Biol. 2021;8:599048. doi: 10.3389/fcell.2020.599048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Kang C, Xu Q, Martin TD, Li MZ, Demaria M, Aron L, Lu T, Yankner BA, Campisi J, Elledge SJ. The DNA damage response induces inflammation and senescence by inhibiting autophagy of GATA4. Science. 2015;349:aaa5612. doi: 10.1126/science.aaa5612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Bea S, Valdes-Mas R, Navarro A, Salaverria I, Martin-Garcia D, Jares P, Gine E, Pinyol M, Royo C, Nadeu F, et al. Landscape of somatic mutations and clonal evolution in mantle cell lymphoma. Proc Natl Acad Sci USA. 2013;110:18250–18255. doi: 10.1073/pnas.1314608110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Sarkar A, Stellrecht CM, Vangapandu HV, Ayres M, Kaipparettu BA, Park JH, Balakrishnan K, Burks JK, Pandita TK, Hittelman WN, et al. Ataxia-telangiectasia mutated interacts with Parkin and induces mitophagy independent of kinase activity. Evidence from mantle cell lymphoma. Haematologica. 2021;106:495–512. doi: 10.3324/haematol.2019.234385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Milanovic M, Shao Z, Estes VM, Wang XS, Menolfi D, Lin X, Lee BJ, Xu J, Cupo OM, Wang D, Zha S. FATC domain deletion compromises ATM protein stability, blocks lymphocyte development, and promotes lymphomagenesis. J Immunol. 2021;206:1228–1239. doi: 10.4049/jimmunol.2000967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Das B, Pal B, Bhuyan R, Li H, Sarma A, Gayan S, Talukdar J, Sandhya S, Bhuyan S, Gogoi G, et al. MYC Regulates the HIF2α stemness pathway via Nanog and Sox2 to Maintain Self-Renewal in cancer stem cells versus non-stem cancer cells. Cancer Res. 2019;79:4015–4025. doi: 10.1158/0008-5472.CAN-18-2847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Zhou X, Wen Y, Tian Y, He M, Ke X, Huang Z, He Y, Liu L, Scharf A, Lu M, et al. Heat Shock Protein 90α-Dependent B-Cell-2-Associated Transcription Factor 1 promotes hepatocellular carcinoma proliferation by regulating MYC Proto-Oncogene c-MYC mRNA Stability. Hepatology. 2019;69:1564–1581. doi: 10.1002/hep.30172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Albakova Z, Mangasarova Y, Albakov A, Nikulina E, Kravchenko S, Sapozhnikov A. Aberrant HSP90 expression in lymphocytes and HSP90 Response to Anti-PD-1 therapy in lymphoma patients. Front Immunol. 2022;13:893137. doi: 10.3389/fimmu.2022.893137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Calvo-Vidal MN, Zamponi N, Krumsiek J, Stockslager MA, Revuelta MV, Phillip JM, Marullo R, Tikhonova E, Kotlov N, Patel J, et al. Oncogenic HSP90 facilitates metabolic alterations in aggressive B-cell lymphomas. Cancer Res. 2021;81:5202–5216. doi: 10.1158/0008-5472.CAN-21-2734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Fuhrmann-Stroissnigg H, Ling YY, Zhao J, McGowan SJ, Zhu Y, Brooks RW, Grassi D, Gregg SQ, Stripay JL, Dorronsoro A, et al. Identification of HSP90 inhibitors as a novel class of senolytics. Nat Commun. 2017;8:422. doi: 10.1038/s41467-017-00314-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Liu H, Lu Z, Shi X, Liu L, Zhang P, Golemis EA, Tu Z. HSP90 inhibition downregulates DNA replication and repair genes via E2F1 repression. J Biol Chem. 2021;297:100996. doi: 10.1016/j.jbc.2021.100996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Schmidt L, Issa II, Haraldsdottir H, Hald JL, Schmitz A, Due H, Dybkaer K. Hsp90 inhibition sensitizes DLBCL cells to cisplatin. Cancer Chemother Pharmacol. 2022;89:431–440. doi: 10.1007/s00280-022-04407-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Lu Z, Wang Z, Tu Z, Liu H. HSP90 inhibitor ganetespib enhances the sensitivity of mantle cell lymphoma to bruton's tyrosine kinase inhibitor ibrutinib. Front Pharmacol. 2022;13:864194. doi: 10.3389/fphar.2022.864194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.He W, Hu H. BIIB021, an Hsp90 inhibitor: A promising therapeutic strategy for blood malignancies (Review) Oncol Rep. 2018;40:3–15. doi: 10.3892/or.2018.6422. [DOI] [PubMed] [Google Scholar]
- 116.Yong K, Cavet J, Johnson P, Morgan G, Williams C, Nakashima D, Akinaga S, Oakervee H, Cavenagh J. Phase I study of KW-2478, a novel Hsp90 inhibitor, in patients with B-cell malignancies. Br J Cancer. 2016;114:7–13. doi: 10.1038/bjc.2015.422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Oki Y, Younes A, Knickerbocker J, Samaniego F, Nastoupil L, Hagemeister F, Romaguera J, Fowler N, Kwak L, Westin J. Experience with HSP90 inhibitor AUY922 in patients with relapsed or refractory non-Hodgkin lymphoma. Haematologica. 2015;100:e272–e274. doi: 10.3324/haematol.2015.126557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Marumoto T, Zhang D, Saya H. Aurora-A-a guardian of poles. Nat Rev Cancer. 2005;5:42–50. doi: 10.1038/nrc1526. [DOI] [PubMed] [Google Scholar]
- 119.Farag SS. The potential role of Aurora kinase inhibitors in haematological malignancies. Br J Haematol. 2011;155:561–579. doi: 10.1111/j.1365-2141.2011.08898.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Iqbal J, Weisenburger DD, Chowdhury A, Tsai MY, Srivastava G, Greiner TC, Kucuk C, Deffenbacher K, Vose J, Smith L, et al. Natural killer cell lymphoma shares strikingly similar molecular features with a group of non-hepatosplenic gammadelta T-cell lymphoma and is highly sensitive to a novel aurora kinase A inhibitor in vitro. Leukemia. 2011;25:348–358. doi: 10.1038/leu.2010.255. [DOI] [PubMed] [Google Scholar]
- 121.Friedberg JW, Mahadevan D, Cebula E, Persky D, Lossos I, Agarwal AB, Jung J, Burack R, Zhou X, Leonard EJ, et al. Phase II study of alisertib, a selective Aurora A kinase inhibitor, in relapsed and refractory aggressive B- and T-cell non-Hodgkin lymphomas. J Clin Oncol. 2014;32:44–50. doi: 10.1200/JCO.2012.46.8793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Kelly KR, Friedberg JW, Park SI, McDonagh K, Hayslip J, Persky D, Ruan J, Puvvada S, Rosen P, Iyer SP, et al. Phase I study of the investigational aurora a kinase inhibitor alisertib plus rituximab or rituximab/vincristine in relapsed/refractory aggressive B-cell lymphoma. Clin Cancer Res. 2018;24:6150–6159. doi: 10.1158/1078-0432.CCR-18-0286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Tan CRC, Abdul-Majeed S, Cael B, Barta SK. Clinical pharmacokinetics and pharmacodynamics of bortezomib. Clin Pharmacokinet. 2019;58:157–168. doi: 10.1007/s40262-018-0679-9. [DOI] [PubMed] [Google Scholar]
- 124.Robak T, Jin J, Pylypenko H, Verhoef G, Siritanaratkul N, Drach J, Raderer M, Mayer J, Pereira J, Tumyan G, et al. Frontline bortezomib, rituximab, cyclophosphamide, doxorubicin, and prednisone (VR-CAP) versus rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) in transplantation-ineligible patients with newly diagnosed mantle cell lymphoma: final overall survival results of a randomised, open-label, phase 3 study. Lancet Oncol. 2018;19:1449–1458. doi: 10.1016/S1470-2045(18)30685-5. [DOI] [PubMed] [Google Scholar]
- 125.Teachey DT, Devidas M, Wood BL, Chen Z, Hayashi RJ, Hermiston ML, Annett RD, Archer JH, Asselin BL, August KJ, et al. Children's Oncology Group Trial AALL1231: A Phase III clinical trial testing bortezomib in newly diagnosed T-Cell acute lymphoblastic leukemia and lymphoma. J Clin Oncol. 2022;40:2106–2118. doi: 10.1200/JCO.21.02678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Hurez V, Dao V, Liu A, Pandeswara S, Gelfond J, Sun L, Bergman M, Orihuela CJ, Galvan V, Padron A, et al. Chronic mTOR inhibition in mice with rapamycin alters T, B, myeloid, and innate lymphoid cells and gut flora and prolongs life of immune-deficient mice. Aging Cell. 2015;14:945–956. doi: 10.1111/acel.12380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Major A, Kline J, Karrison TG, Fishkin PAS, Kimball AS, Petrich AM, Nattam S, Rao K, Sleckman BG, Cohen K, et al. Phase I/II clinical trial of temsirolimus and lenalidomide in patients with relapsed and refractory lymphomas. Haematologica. 2022;107:1608–1618. doi: 10.3324/haematol.2021.278853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Borthakur G, Martinelli G, Raffoux E, Chevallier P, Chromik J, Lithio A, Smith CL, Yuen E, Oakley GJ, III, Benhadji KA, DeAngelo DJ. Phase 1 study to evaluate Crenigacestat (LY3039478) in combination with dexamethasone in patients with T-cell acute lymphoblastic leukemia and lymphoma. Cancer. 2021;127:372–380. doi: 10.1002/cncr.33188. [DOI] [PubMed] [Google Scholar]
- 129.Hart S, Goh KC, Novotny-Diermayr V, Hu CY, Hentze H, Tan YC, Madan B, Amalini C, Loh YK, Ong LC, et al. SB1518, a novel macrocyclic pyrimidine-based JAK2 inhibitor for the treatment of myeloid and lymphoid malignancies. Leukemia. 2011;25:1751–1759. doi: 10.1038/leu.2011.148. [DOI] [PubMed] [Google Scholar]
- 130.Moskowitz AJ, Ghione P, Jacobsen E, Ruan J, Schatz JH, Noor S, Myskowski P, Vardhana S, Ganesan N, Hancock H, et al. A phase 2 biomarker-driven study of ruxolitinib demonstrates effectiveness of JAK/STAT targeting in T-cell lymphomas. Blood. 2021;138:2828–2837. doi: 10.1182/blood.2021013379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Gillessen S, Pluetschow A, Vucinic V, Ostermann H, Kobe C, Brockelmann PJ, Boll B, Eichenauer DA, Heger JM, Borchmann S, et al. JAK inhibition with ruxolitinib in relapsed or refractory classical Hodgkin lymphoma: Final results of a phase II, open label, multicentre clinical trial (JeRiCHO) Eur J Haematol. 2022;109:728–735. doi: 10.1111/ejh.13859. [DOI] [PubMed] [Google Scholar]
- 132.Pullarkat VA, Lacayo NJ, Jabbour E, Rubnitz JE, Bajel A, Laetsch TW, Leonard J, Colace SI, Khaw SL, Fleming SA, et al. Venetoclax and navitoclax in combination with chemotherapy in patients with relapsed or refractory acute lymphoblastic leukemia and lymphoblastic lymphoma. Cancer Discov. 2021;11:1440–1453. doi: 10.1158/2159-8290.CD-20-1465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Chang J, Wang Y, Shao L, Laberge RM, Demaria M, Campisi J, Janakiraman K, Sharpless NE, Ding S, Feng W, et al. Clearance of senescent cells by ABT263 rejuvenates aged hematopoietic stem cells in mice. Nat Med. 2016;22:78–83. doi: 10.1038/nm.4010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Mei MG, Lee HJ, Palmer JM, Chen R, Tsai NC, Chen L, McBride K, Smith DL, Melgar I, Song JY, et al. Responseadapted anti-PD-1-based salvage therapy for Hodgkin lymphoma with nivolumab alone or in combination with ICE. Blood. 2022;139:3605–3616. doi: 10.1182/blood.2022015423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Nastoupil LJ, Chin CK, Westin JR, Fowler NH, Samaniego F, Cheng X, Ma MCJ, Wang Z, Chu F, Dsouza L, et al. Safety and activity of pembrolizumab in combination with rituximab in relapsed or refractory follicular lymphoma. Blood Adv. 2022;6:1143–1151. doi: 10.1182/bloodadvances.2021006240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Rybicka M, Zhao J, Piotrowicz K, Ptasnik S, Mitka K, Kocot-Kępska M, Hui KK. Promoting whole person health: Exploring the role of traditional Chinese medicine in Polish healthcare. J Integr Med. 2023;21:509–517. doi: 10.1016/j.joim.2023.10.001. [DOI] [PubMed] [Google Scholar]
- 137.Luo J, Shen S, Xia J, Wang J, Gu Z. Mitochondria as the Essence of Yang Qi in the human body. Phenomics. 2022;2:336–348. doi: 10.1007/s43657-022-00060-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Zhu Y, Tchkonia T, Pirtskhalava T, Gower AC, Ding H, Giorgadze N, Palmer AK, Ikeno Y, Hubbard GB, Lenburg M, et al. The Achilles' heel of senescent cells: From transcriptome to senolytic drugs. Aging Cell. 2015;14:644–658. doi: 10.1111/acel.12344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Zhu Y, Tchkonia T, Fuhrmann-Stroissnigg H, Dai HM, Ling YY, Stout MB, Pirtskhalava T, Giorgadze N, Johnson KO, Giles CB, et al. Identification of a novel senolytic agent, navitoclax, targeting the Bcl-2 family of anti-apoptotic factors. Aging Cell. 2016;15:428–435. doi: 10.1111/acel.12445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Yoshimura S, Panetta JC, Hu J, Li L, Gocho Y, Du G, Umezawa A, Karol SE, Pui CH, Mullighan CG, et al. Preclinical pharmacokinetic and pharmacodynamic evaluation of dasatinib and ponatinib for the treatment of T-cell acute lymphoblastic leukemia. Leukemia. 2023;37:1194–1203. doi: 10.1038/s41375-023-01900-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Senapati J, Sasaki K, Issa GC, Lipton JH, Radich JP, Jabbour E, Kantarjian HM. Management of chronic myeloid leukemia in 2023-common ground and common sense. Blood Cancer J. 2023;13:58. doi: 10.1038/s41408-023-00823-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Fujii K, Idogawa M, Suzuki N, Iwatsuki K, Kanekura T. Functional Depletion of HSP72 by siRNA and quercetin enhances vorinostat-induced apoptosis in an HSP72-overexpressing cutaneous T-Cell lymphoma cell line, Hut78. Int J Mol Sci. 2021;22:11258. doi: 10.3390/ijms222011258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Cai Y, Zhou H, Zhu Y, Sun Q, Ji Y, Xue A, Wang Y, Chen W, Yu X, Wang L, et al. Elimination of senescent cells by β-galactosidase-targeted prodrug attenuates inflammation and restores physical function in aged mice. Cell Res. 2020;30:574–589. doi: 10.1038/s41422-020-0314-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Muralidharan SV, Bhadury J, Nilsson LM, Green LC, McLure KG, Nilsson JA. BET bromodomain inhibitors synergize with ATR inhibitors to induce DNA damage, apoptosis, senescence-associated secretory pathway and ER stress in Myc-induced lymphoma cells. Oncogene. 2016;35:4689–4697. doi: 10.1038/onc.2015.521. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.