Abstract
Hypertension is a globally prevalent condition, and low adherence to antihypertensive therapy is considered one of the main causes of poor blood pressure (BP) control. Non-adherence to antihypertensive treatment is a complex issue that can arise from various factors; however, gaining an understanding of this provides key targets for intervention strategies. This study aimed to provide an overview of the current status and recent developments regarding our understanding of the determinants of patients' adherence to antihypertensives. A systematic review was performed using the electronic databases MEDLINE/PubMed, Web of Science, Scientific Electronic Library Online (SciELO), and “Índex das Revistas Médicas Portuguesas”, which included studies published between 2017 and 2021 following the PICOS model: (P) Adult patients with the diagnosis of primary hypertension, using at least one antihypertensive agent; (I) all interventions on both pharmacological and non-pharmacological level; (C) patient’s adherence against their non-adherence; (O) changes in adherence to the therapeutic plan; and (S) any study design (except review articles) written in English, French, Spanish or Portuguese.
Articles were reviewed by two researchers and their quality was assessed. Subsequently, determinants were classified according to their consistent or inconsistent association with adherence or non-adherence. Only 45 of the 635 reports identified met the inclusion criteria. Adherence was consistently associated with patient satisfaction with communication, patient-provider relationship, their treatment, and use of eHealth and mHealth strategies; a patient’s mental and physical health, including depression, cognitive impairment, frailty, and disability, previous hospitalization, occurrence of vital events; drug treatment type and appearance; and unwillingness due to health literacy, self-efficacy, and both implicit and explicit attitudes towards treatment. There were discrepancies regarding the association of other factors to adherence, but these inconsistent factors should also be taken into account. In conclusion, the barriers to adherence are varied and often interconnected between socioeconomic, patient, therapy, condition, and healthcare system levels. Healthcare teams should invest in studying patients’ non-adherence motives and tailoring interventions to individual levels, by using a multifaceted approach to assess adherence. Further research is needed to analyze the impact of implicit attitudes, the use of new technological approaches, and the influence of factors that are inconsistently associated with non-adherence, to understand their potential in implementing adherence strategies.
Keywords: telemedical technology, mobile apps (mhealth), preventive health, antihypertensive agents, primary health care, medication adherence strategies, antihypertensive therapy, hypertension
Introduction and background
Cardiovascular diseases (CVD) are considered a global public health problem and remain the leading cause of death and incapacity worldwide; they account for 32% of all global deaths, of which 85% are attributed to stroke and heart attacks [1]. In 2019, across OECD (Organization for Economic Co-operation and Development) countries, CVD emerged as the number one treatable cause of mortality, representing 36% of premature deaths amenable to treatment, and the third disease with most preventable deaths through effective public health measures and primary care intervention (19% of all preventable deaths) [2]. Despite the significant improvements in CVD mortality rates, evidence from some countries has shown a slowdown in the reduction of this rate, even before the coronavirus disease 2019 (COVID-19) pandemic, due to certain challenges including the rapid ageing of the population and the difficulty in promoting practices to deal with some risk factors, such as obesity and diabetes [3].
In line with worldwide trends, CVD represents the main cause of mortality in Portugal (29.9% of all deaths in 2019) [4,5], with arterial hypertension emerging as the most important risk factor for several complications such as ischaemic heart disease, stroke, chronic kidney disease and dementia [6-8]. Additionally, in light of the current epidemiological scenario we are living in, it is important to note that high blood pressure (HBP) increases the risk of severe COVID-19 infection [9,10]. In 2014, 25.3% (2.2 million people) reported having HBP in a Portuguese national survey, with the majority of them being females [11].
The diagnosis of hypertension is established based on the following parameters: systolic blood pressure (SBP) ≥140 mmHg and/or diastolic blood pressure (DBP) ≥90 mmHg [12]. Achieving good control of blood pressure (BP) is possible by using both non-pharmacological and pharmacological treatment, and studies have proven the efficacy of these interventions [13]. The impact of lowering BP can be so relevant that even modest improvements in SBP (reduction of 10 mmHg) or DBP (reduction of 5 mmHg) can have an appreciable effect on health outcomes, reducing the risk of cardiovascular events [14]. The approach to good BP control depends on multiple factors, including pathophysiological and pharmacological aspects, levels of adherence, and therapeutic inertia [15]. However, despite substantial efforts in promoting healthy lifestyles and the major progress made in pharmacological treatment, low adherence to preventive measures and prescribed medication is considered one of the main causes of the lack of BP control in many individuals [16]. A systematic review and meta-analyses published in 2017 estimated that 45.2% of hypertensive patients fail to adhere to prescribed regimens, especially among patients with uncontrolled BP (83.7%), highlighting the importance of addressing low adherence to the therapeutic plan in BP management [17].
The World Health Organization defines adherence to long-term therapy as “the extent to which a person’s behaviour - taking medication, following a diet, and/or executing lifestyle changes, corresponds with agreed recommendations from a healthcare provider”. The reported rates of adherence to pharmacotherapy oscillate between 50 and 70%, depending on the group being studied, the follow-up duration, the methods applied for adherence evaluation, and the different drug classes involved [18]. In the process of adherence, both patient and provider understand and agree with the recommendations. However, it is not unusual that this term is used as a synonym for compliance. This last concept brings a different connotation since it implies passive behaviour from the patient who is willing to follow the provider’s recommendations, despite not necessarily agreeing with them [19].
The complexity surrounding a patient’s behaviour towards the prescribed therapy cannot easily be captured in a single word, as it tends to amplify the clinician’s control over the process of taking medications and underrate the patient’s beliefs, personal circumstances, and available resources [20]. Adherence often emerges as a multidimensional phenomenon, which goes beyond the misleading idea that only the patient is responsible for taking their treatment, and can be classified into five dimensions that might interact with each other: patient-related factors, condition-related factors, therapy-related factors, social/economic-related factors, and health system/healthcare team-related factors [18]. The acknowledgement of the complexity regarding the determinants of patients’ adherence allows us to avoid exclusively blaming the patients for their non-adherence and guide health systems and healthcare teams to identify effective solutions, which is critical in reducing the burden related to hypertension and its complications.
Given the importance of medication adherence (MA) in the control of BP and the critical role of different factors in patient adherence, this study aims to provide an overview, via a systematic approach, of the current status and recent developments regarding the determinants of patient adherence to treatment in the adult population, by assessing and comparing different predictors of adherence to both pharmacological and non-pharmacological treatment for hypertension.
Review
Materials and methods
Design and Registration
We conducted a systematic review of all scientific articles available (according to the search strategy defined below) and followed the Cochrane Handbook, a methodology guide for systematic reviews. The protocol has been registered in the International Prospective Register of Systematic Reviews (PROSPERO) under the number CRD42022301595, and the study was conducted based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.
Search Strategy
A search for relevant articles published between January 1, 2017, and December 31, 2021, was performed on electronic databases such as Medline (via PubMed), Web of Science, Scientific Electronic Library Online (SciELO), and other national/regional databases (Índex - Revistas Médicas Portuguesas).
The general search terms used on Pubmed were ((("Patient Compliance"[Title/Abstract] OR "Medication Adherence"[Title/Abstract]) OR ("Patient Compliance"[Majr] OR "Medication Adherence"[Majr])) AND ((high blood pressure [Title/Abstract] OR hypertension [Title/Abstract]) OR ("Hypertension"[Majr]))) AND ("Antihypertensive Agents"[Majr]). The search was restricted to articles written in English, French, Spanish, and Portuguese, whose abstracts were available online and involved the adult population. A similar search strategy was used in other databases, but the MeSH terms and text words were modified/adapted as per the specific requirements of each database.
The research question was elaborated using the PICOS model: (P) Adult patients with the diagnosis of primary hypertension, using at least one antihypertensive agent; (I) all interventions on both pharmacological and non-pharmacological levels; (C) patient’s adherence against their non-adherence; (O) changes in adherence to the therapeutic plan; and (S) any study design (except review articles) written in English, French, Spanish or Portuguese: “What influences patient’s adherence to antihypertensive therapies?” The reviewing team’s decision to include adherence to both pharmacological and non-pharmacological treatment was based on the fact that the approach in previous studies frequently focuses on determinants of both types of treatment. Thus, the conclusions would be more reliable since treatment adherence carries these two dimensions together.
Eligibility Criteria
Studies meeting the following criteria were included: (i) type of study: any study design conducted in English, French, Spanish or Portuguese, preferring those with a higher level of evidence [i.e., randomized controlled trials (RCT)], except for all types of reviews; (ii) type of population: adult patients aged 18 years or over, of any gender or ethnicity, with a diagnosis of primary hypertension, as well as participants with other comorbidities (e.g., diabetes); (iii) type of intervention: adherence to both pharmacological and non-pharmacological treatment using direct or indirect measures of adherence and/or based on patient’s perspective of the impact on their quality of life, changes in the pharmacological treatment, combination of both pharmacological and non-pharmacological treatment, adherence aids (e.g., reminders/alarms), and use of educational measures (e.g., communication between the patient and the healthcare professional); articles examining adherence to multiple pharmacological therapies were deemed eligible for inclusion if antihypertensive therapy was among the different pharmacotherapeutic agents being studied; (iv) type of comparator: patient’s adherence against the non-adherence; (v) type of outcome: primary outcome: the adherence to the antihypertensive therapy measured using the different tools available; secondar outcomes: the reduction and/or control of the systolic/diastolic blood pressure and patient-reported information on quality of life or symptoms.
The exclusion criteria were as follows: (i) studies in languages other than the ones in the inclusion criteria, duplicate articles from different databases, and articles whose titles and/or abstracts did not meet the inclusion criteria. Articles that required payment for access were analyzed on a case-by-case basis and excluded if they were not deemed exceptionally relevant to this study. Review articles were excluded, to avoid a high risk of duplicating the results; (ii) studies involving patients under 18 years of age, patients with secondary hypertension (e.g., pre-eclampsia, hyperaldosteronism), and pregnant or breast-feeding women; (iii) studies involving other interventions besides pharmacological and non-pharmacological treatments (i.e., surgical).
Study Selection
All of the retrieved articles were organized and screened using the online software Covidence and the Endnote Web reference management software. After removing duplicates, the articles’ titles and abstracts were screened by two independent reviewers who were blinded to each other’s decisions. The articles and abstracts that matched the objective of this review were eligible for a full-text review. The two authors assessed the eligibility criteria and applied them to the final articles and those that met the inclusion criteria were retrieved. Finally, studies that did not meet any of the inclusion criteria were excluded. Disagreements were always solved by reaching a consensus and, therefore, there was no need to involve a third author in the selection process. Finally, after browsing the reference lists, the data were extracted from each study to synthesize their findings.
Data Extraction and Management
Data extraction was performed by one investigator and included the following items: authors, date of the study, study design, research aim, sample size, mean age, tool used in the measurement of adherence, and definition of adherence. To summarize the major findings and the direction of the associations with MA, data regarding the different determinants affecting adherence were organized into five domains: patient characteristics, socioeconomic status, comorbidities-related, therapy-related, and healthcare/health system issues. For each domain, subcategories were introduced according to the determinants present in the included studies. In addition, to systematize the information, factors were organized into three columns to signify whether they had a statistically significant association with adherence, a statistically significant association with non-adherence, or a non-significant association at all.
Quality of Evidence and Risk of Bias
To ensure the quality of the analyzed articles, all eligible studies were reviewed by two researchers to appraise their risk of bias, using different tools according to the study design of the article being evaluated. For RCTs, we applied the “Rob2: Cochrane risk-of-bias tool” that assessed the following five components: (i) randomization process, (ii) deviations from the intended interventions, (iii) missing outcome data, (iv) measurement of the outcome, and (v) selection of the reported results. The quality assessment for each component was classified into three possible categories (high risk of bias, some concerns, and low risk of bias) and an overall judgment was stated. For cohort studies, we applied the “ROBINS-I: Cochrane risk-of-bias tool for non-randomized studies of interventions”, which has seven components: (i) confounding, (ii) selection bias, (iii) classification of intervention, (iv) intended intervention, (v) missing data, (vi) measures of outcome, and (vii) reported results. The evaluation was expressed in five possible outcomes (low risk, moderate risk, serious risk, critical risk, and no information). Finally, cross-sectional studies were evaluated using the “JBI Critical Appraisal Checklist for Analytical cross-sectional studies”, which involved eight questions with the following possible answers: “Yes”, “No” and “Unclear”. The assessment of the risk of bias was based on the percentage of “yes” scores, considering the following three levels of classification: high quality (>70% of “yes” scores), moderate quality (from 50 to 69%), and low quality (<50%).
Results
Study Selection
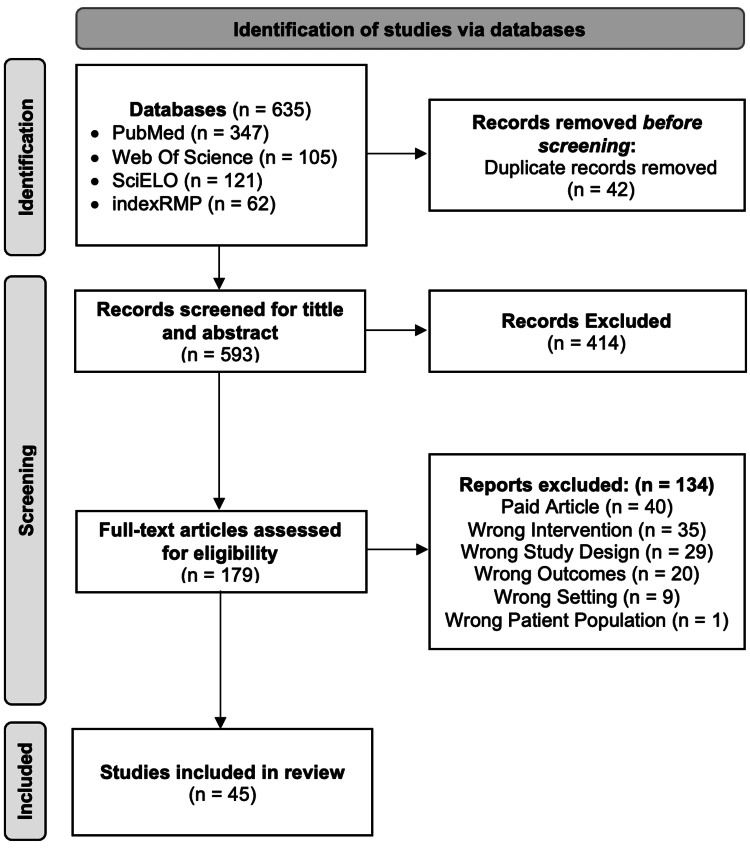
The literature search on the reported databases elicited 635 articles. After removing 42 duplicates, 593 articles qualified for a title and abstract screening, and according to the inclusion criteria, 179 underwent full-text review. A detailed assessment of the 179 full-text articles resulted in the elimination of an additional 134. Finally, 45 articles that fully met the eligibility were included in this review. Figure 1 shows the PRISMA flow chart illustrating the search and study selection.
Figure 1. PRISMA flow chart depicting the literature search and study selection.
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses
Study Characteristics
The characteristics of the included studies on adherence to antihypertensive therapy are presented in Table 1 [21-65]. Overall, we included 21 cross-sectional studies [22-24,28,30-32,35,36,40,41,45,47,48,50-52,56,57,62,64], 19 cohort studies [21,26,27,29,33,34,37-39,42-44,49,53-55,58,60,65], four RCTs [25,46,59,61], and one quasi-experimental study [63]. The total number of patients included for analysis was 1,533,220, with individual study sample sizes ranging from 32 to 484,493 participants. The mean age of the participants ranged from 46.5 to 76.6 years old, with most of the studies having a population with a mean age above 50 years.
Table 1. Characteristics of included studies on adherence to antihypertensive therapy.
CMA: cumulative medication adherence; K-Wood-MAS-4: Krousel-Wood Medication Adherence Scale; MA: medication adherence); MARS: medication adherence rate scale; MGL: Morisky Green Levine; MMAS-8: Morisky Medication Adherence Scale with 8 items; MPR: medication possession ratio; MRA: medication refill adherence; PDC: proportion of days covered; PDT: percentage of doses taken; PDTc: percentage of doses taken correctly; PMC: proportion of months covered; TDM: therapeutic drug monitoring
| Author | Year | Study design | Study objective | Sample size (N); mean age (A) | Measurement of adherence | Definition of adherence |
| Arancón-Monge et al. [21] | 2020 | Cohort study | To study if changes in appearance and name in bioequivalent drugs are associated with non-adherence to antihypertensive and lipid-lowering medications | N = 274 patients; A = 72 years | MGL (scores ranged from 0 to 4); direct count of the medication | MGL: high adherence (score of 4). Direct count: non-adherence if the patient forgets to take more than 2 doses/trimester |
| Avataneo et al. [22] | 2018 | Cross-sectional study | To assess adherence to treatment in patients with reported resistant hypertension using therapeutic drug monitoring and to study parameters that might predict non-adherence | N = 50 patients; A = 56 years | TDM specialist opinion. A homemade questionnaire with 10 items (scores ranged from 0 to 10) | TDM: fully adherent patients – detectable plasma concentrations of all prescribed drugs. Specialist opinion: comment on patient adherence based on their personal experience before knowing TDM results. Questionnaire: high adherence (scores ranged from 9 to 10) |
| Barbosa et al. [23] | 2019 | Cross-sectional study | To evaluate predictors of adherence to antihypertensive therapies in adults/elderly | N = 257 patients; A = Not reported | A questionnaire with 24 items, that accessed four dimensions: patient, disease/treatment, healthcare service, and environment (scores ranged from 24 to 120) | Adherent (score of 73–120) |
| Berhe et al. [24] | 2017 | Cross-sectional study | To evaluate the impact of adverse drug events and treatment satisfaction on antihypertensive medication adherence | N = 925 patients; A = 57 years | MMAS-8 (scores ranged from 0 to 8) | High adherence (score of 8) |
| Chandler et al. [25] | 2019 | Randomized controlled trial | To evaluate the efficacy of the SMASH (smartphone app) in establishing systolic blood pressure control via increased medication adherence | N = 54 patients; A = 46.5 ± 9.9 years | MMAS-8 (scores ranged from 0 to 8); medication tray: timestamps of openings of pill tray compartments (scores ranged from 0 to 1) | MMAS-8: high adherence (score of 8). Medication tray: adherent if doses are taken within 0-3 hours (score of 1) |
| Chang et al. [26] | 2021 | Cohort study | To study the association between the patient-clinician relationship and adherence to antihypertensive medications among black adults with hypertension | N = 2571 patients; A = 58 years | MRA | Adherent if MRA ≥80% |
| Cho et al. [27] | 2018 | Cohort study | To study the association between cognitive function and antihypertensive medication adherence in elderly patients without dementia | N = 42,132 patients; A = not reported | CMA using prescription data | Adherent if CMA ≥80% |
| Craig et al. [28] | 2021 | Cross-sectional study | To study the contributions of implicit and explicit attitudes in explaining differences between both objective and subjective antihypertensive medication adherence measures | N = 85 patients; A = 62.3 years | PDC K-Wood-MAS-4 (scores ranged from 0 to 4) | Non-adherent if PDC <0.8 or K-Wood-MAS-4 ≥1 |
| Del Pinto et al. [29] | 2021 | Cohort study | To assess antihypertensive treatment adherence and BP control rates in treated hypertensive elderly patients according to different treatment patterns | N = 13,196 patients; A = 73.2 ± 7.5 years | The ratio between the number of pills in boxes used and those expected in 6 months | Adherent if rate ≥80% |
| Durand et al. [30] | 2018 | Cross-sectional study | To study predictors of long-term medication adherence in patients who appear to have treatment-resistant hypertension using a composite adherence score | N = 204 patients; A = 69.9 years | Adherence composite of MMAS-8 (scores ranged from 0 to 8) and MARS (scores ranged from 0 to 10); prescription refill records; biochemical assay of urine (TDM) | Non-adherent if: MMAS-8 <6 and/or MARS <23. Prescription refill records <80%. Urine sample: undetectable concentrations of all the prescribed drugs |
| Fang et al. [31] | 2020 | Cross-sectional study | To study the association of cost-related medication non-adherence barriers and hypertension management among US adults with self-reported hypertension | N = 7498 patients; A = not reported | A questionnaire with 3 items to assess cost-related medication non-adherence | Non-adherent if answered “yes” to any of the 3 questions |
| Fernandez et al. [32] | 2017 | Cross-sectional study | To study factors related to non-adherence to pharmacological treatment and to design an educational program | N = 102 patients; A = not reported | MGL (scores ranged from 0 to 4) | High adherence (score of 4) |
| Fortuna et al. [33] | 2018 | Cohort study | To study the association between the patient experience with care and medication adherence | N = 2128 patients; A = 50.4 years | MMAS-8 (scores ranged from 0 to 8) | High adherence (score of 8) |
| Gao et al. [34] | 2020 | Cohort study | To study the determinants of antihypertensive medication adherence | N = 7638 patients; A = 67.6 ± 11.1 years | PDC | Good adherence if PDC >0.8 |
| Gewehr et al. [35] | 2018 | Cross-sectional study | To study the adherence to antihypertensive agents and the determinants associated with non-adherence in primary healthcare | N = 156 patients; A = 66.9 ± 8.3 years | Brief Medication Questionnaire (scores ranged from 0 to 3) | High adherence (score of 0–1) |
| Gniwa et al. [36] | 2019 | Cross-sectional study | To investigate therapeutic adherence and to recognize factors associated with poor adherence | N = 276 patients; A = 64.9 ± 10.2 years | Girerd test (6 items) (scores ranged from 0 to 6) | Good observers (total of yes = 0) |
| Gupta et al. [37] | 2017 | Cohort study | To study the therapeutic applications of biochemical screening for the presence of antihypertensive medications in bodily fluids | N = 331 patients; A = 54.3 years (UK patients)/51.4 years (Czech patients) | Blood and urine measure of metabolites using liquid chromatography-tandem mass spectrometry | Adherent patients - detectable concentrations of all prescribed drugs in serum/urine |
| Gupta et al. [38] | 2017 | Cohort study | To study the relationships between non-adherence and main demographic and BP-lowering therapy-related factors | N = 1348 patients; A = 55.8 years (UK patients)/54.4 years (Czech patients) | Blood and urine measure of metabolites using liquid chromatography-tandem mass spectrometry | Adherent patients - detectable concentrations of all prescribed drugs in serum/urine |
| Hargrove et al. [39] | 2017 | Cohort study | To evaluate whether individual factors predict adherence trajectories and to improve the classification of antihypertensive adherence using group-based trajectory models | N = 282,520 patients; A = 75 years | PMC, PDC | Adherent if PMC ≥80% days and/or PDC ≥80% |
| Heizomi et al. [40] | 2020 | Cross-sectional study | To study the characteristics related to health literacy among hypertensive patients, its relationship with adherence to antihypertensive medications and to determine the validity of the four-item MGL scale | N = 300 patients; A = 56.7 years | MGL (scores ranged from 0 to 4) | MGL: high adherence (score of 4) |
| Ishida et al. [41] | 2019 | Cohort study | To study treatment patterns and medication adherence to different antihypertensive drug classes and to evaluate whether fixed-dose combinations were being prescribed alone or simultaneously with other antihypertensive drugs | N = 47,891 patients; A = 70.1 years | PDC | High adherence if PDC ≥80% |
| Kim et al. [42] | 2019 | Cohort study | To study the effect of single-pill combination on adherence to antihypertensive medication in a real-world setting | N = 116,677 patients; A = not reported | MPR: dividing the total days supplied by the number of days between the first and last refills | No more information was given |
| Kulkarni et al. [43] | 2021 | Cohort study | To study pharmacotherapy non-adherence using urine screens among patients with uncontrolled HTN, with analyses of demographics, polypharmacy, medication type, and comorbidities | N = 174 patients; A = 56 years | Urine measure of metabolites using liquid chromatography-tandem mass spectrometry | Adherent patients - detectable concentrations of all prescribed drugs in urine |
| Lauffenburger et al. [44] | 2017 | Cohort Study | To evaluate patterns of antihypertensive therapy initiation, compare persistence and long-term adherence to treatments in patients initiating fixed-dose combinations and other antihypertensive therapies, and identify other determinants of adherence and persistence to antihypertensives | N = 484,493 patients; A = 47.2 years | PDC | Fully adherent if PDC >0.8 to at least one antihypertensive |
| Macquart de Terline et al. [45] | 2019 | Cross-sectional | To study adherence to medication and association with socioeconomics, clinical and treatment predictors of low adherence among hypertensive patients in 12 sub-Saharan African countries | N = 2198 patients; A = 57.7 ± 12.0 years (women) and 59.2 ± 11.4 years (men) | MMAS-8 (scores ranged from 0 to 8) | High adherence (score of 8) |
| Morawski et al. [46] | 2018 | Randomized controlled trial | To study the association between medication adherence and blood pressure control with a smartphone app among patients with uncontrolled hypertension | N = 411 patients; A = 52.0 years | MMAS-8 (scores ranged from 0 to 8) | High adherence (score of 8) |
| Nascimento et al. [47] | 2021 | Cross-sectional | To study the factors associated with adherence to the non-pharmacological treatment of hypertension in primary healthcare | N = 421 patients; A = 59.9 ± 11 years | Instrument to evaluate the adherence of participants to non-pharmacological therapies on the following parameters: weight (BMI) and abdominal circumference (AC) control, the practice of physical activity, and alcohol consumption | Patient considered adherent to each measure: weight control: when BMI <25 kg/m2 (below 65 years old) or <27 kg/m2 (above 65 years old); AC: when AC <80 cm (women) and <94 cm (men); physical activity: who practiced moderate physical activities, aerobic training, or resistance training; alcohol consumption: maximum 2 doses a day (men) and maximum 1 dose (women and low-weight individuals) |
| Nashilongo et al. [48] | 2017 | Cross-sectional | To evaluate the levels and predictors of compliance to antihypertensive therapy among patients in primary healthcare and to validate the Hill-Bone compliance scale | N = 120 patients; A = 47.3 ± 11.1 years | Percentage adherence level per patient calculated from the scores on the Hill-Bone Blood Pressure Scale | Perfect adherence (100%) and acceptable adherence (≥80%) |
| Nishimura et al. [49] | 2020 | Cohort study | To study the adherence pattern of new users of antihypertensive drugs and to evaluate possible patient and facility characteristics connected with low adherence | N = 31,592 patients; A = 51.7 years | PDC | High adherence if PDC ≥80% |
| Paczkowska et al. [50] | 2021 | Cross-sectional | To evaluate the impact of patient knowledge on adherence to medical recommendations/improving the efficacy of hypertension treatment and to analyze sociodemographic/clinical factors that affect the level of patient knowledge regarding hypertension | N = 488 patients; A = 63.7 ± 13 | Questionnaire (5 items) regarding treatment adherence | No more information was given |
| Pan et al. [51] | 2019 | Cross-sectional | To study the factors influencing adherence behaviors and determine the interventions that improve adherence in hypertensive patients | N = 488 patients; A = not reported | Therapeutic Adherence Scale for Hypertensive Patients (TASHP) with 25 items (scores ranged from 25 to 125) | Low adherence if TASHP <109 |
| Pluta et al. [52] | 2020 | Cross-sectional | To study the level of acceptance of illness and compliance with therapeutic recommendations in patients with hypertension | N = 200 patients; A = 49.1 ± 11.6 years | MMAS-8 (scores ranged from 0 to 8) | High adherence (score of 8) |
| Rea et al. [53] | 2021 | Cohort study | To study the relationship between initial antihypertensive treatment strategy and adherence in patients starting treatment with one drug or a dual single-pill combination | N = 63,448 patients; A = 59 years | PDC | High adherence if PDC >75% |
| Schoenthaler et al. [54] | 2017 | Cohort study | To study the effect of patient-provider communication on medication adherence among hypertensive patients in a primary care setting | N = 92 patients; A = 59.7 years | Adaptive statistical modeling (ASM) of electronic monitoring device: division into 5 clusters | Adaptively generated adherence calculated using the ASM methods: very high adherence if fits on cluster 1 (97%) or cluster 2 (93%) |
| Shani et al. [55] | 2019 | Cohort study | To study medication adherence to oral antihypertensive medications, to compare adherence rates to different medications, and to study determinants among patients with good adherence | N = 31,530 patients; A = not reported | Rate of pharmacy-filled prescription | Good adherence: purchasing at least 9 monthly prescriptions equals at least 75% adherence |
| Shi et al. [56] | 2019 | Cross-sectional | To study the impact of medication literacy among hypertensive patients on their medication adherence and to develop strategies to improve hypertensive patients’ skills in dealing with the disease | N = 420 patients; A = 60.6 years | MMAS-8 (scores ranged from 0 to 8) | High adherence (score of 8) |
| Shimels et al. [57] | 2021 | Cross-sectional | To evaluate the dimension and associated factors of poor medication adherence among diabetic and hypertensive patients during the COVID-19 pandemic | N = 409 patients; A = 56.5 years | MMAS-8 (scores ranged from 0 to 8) | High adherence (score of 8) |
| Singh et al. [58] | 2019 | Cohort study | To study and identify potential risk factors for medication non-adherence | N = 8,218 patients; A = 76.6 ± 5.4 years (derivation cohort) and 76.0 ± 5.4 years (validation cohort) | PDC | Non-adherent if PDC <0.8 |
| Sung et al. [59] | 2021 | Randomized controlled trial | To evaluate if a triple-component single-pill combination improved medication adherence over an equivalent two-pill combination therapy, using a Medication Event Monitoring System (MEMS) | N = 145 patients; A = 56.0 ± 15.3 years | Using MEMS data to calculate the percentage of doses taken (PDT) and the percentage of days on which the prescribed dose was taken correctly (PDTc) | High adherence if PDT ≥80% and PDTc ≥80% |
| Tajeu et al. [60] | 2019 | Cohort study | To evaluate factors in antihypertensive medication non-persistence and low adherence among adults aged <65 years | N = 379,658 patients; A = not reported | PDC | Low adherence if PDC <80% |
| Varleta et al. [61] | 2017 | Randomized controlled trial | To evaluate whether the effect of a mobile phone text messaging intervention messaging improves self-reported antihypertensive drug adherence in patients with hypertension, against a population with no text messaging | N = 314 patients; A = 60 ± 10 years | MGL (scores ranged from 0 to 4) | High adherence (score of 4) |
| Vázquez Machado et al. [62] | 2019 | Cross-sectional | To study the occurrence of depressive disorders and vital events in patients with HBP and their relationship with adherence to antihypertensive therapy | N = 222 patients; A = not reported | MMAS-8 (scores ranged from 0 to 8) | Low adherence (score of 0–5) |
| Vieira et al. [63] | 2021 | Quasi-experimental study | To study the impact of using a monthly electronic medication organizer device (Supermed) on medication adherence | N = 32 patients; A = 71.4 ± 5.6 years | MGL (scores ranged from 0 to 4) | High adherence (score of 3–4) |
| Wakai et al. [64] | 2021 | Cross-sectional | To study the importance related to the number of medications and complexity of medication regimens for medication adherence and blood pressure control in patients with hypertension | N = 1,057 patients; A = not reported | Pharmacists conducted the first interview for patients and assessed the patients’ medication adherence to brought medicines immediately upon admission | Poor adherence if the patients cannot manage medications by themselves or if they require their family caregivers and/or nurses to manage their medications |
| Wu et al. [65] | 2018 | Cohort study | To evaluate how a health-coaching intervention would affect medication adherence and blood pressure, and to study if variations in medication adherence itself would be associated with changes in blood pressure | N = 477 patients; A = 57.1 ± 12.0 years | MMAS-8 (scores ranged from 0 to 8) | High adherence (score of 6–8) |
Quality of evidence and risk of bias assessment revealed that most studies included had low to moderate risk of bias (see appendices). One cross-sectional study was identified as having a high risk of bias [32] since it did not clarify the inclusion criteria, the study setting, or possible cofounders of the study or strategies to deal with them and was not clear about the statistical approach applied. One RCT was deemed to have some concerns [25] in risk of bias because the control group compared to the intervention group did not use an electronic medication tray, even though this tool was used as part of a secondary outcome measure; this slight difference between groups might have influenced the real outcomes.
Adherence to antihypertensive therapy was assessed using several tools labeled as direct and indirect. Direct measures that study the patient’s medication-related behavior using the measurement of the drug or its metabolite concentration in different body fluids such as blood or urine were only used in five studies [22,30,37,38,43]. By contrast, indirect measures were the most prevalent tools applied. Clinical assessment using questionnaires and surveys was the most prevalent tool, with the Morisky Medication Adherence Scale with 8 items (MMAS-8) being the measure more frequently used in 11 studies [24,25,30,33,45,46,52,56,57,62,65] of the 45, followed by five studies [21,32,40,61,63] that used Morisky Green Levine (MGL) scale with four items. A considerable number of other questionnaires have been used, with both objective and subjective measures, and with numerous versions to accommodate various circumstances [23,31,35,36,47,48,50,51].
One study used pharmacist-conducted interviews with patients, assessing patients’ medication adherence to the medicines brought with them, immediately on admission [64]. Measures involving secondary data analyses curated from data systems such as electronic prescription records and insurance data were commonly used, with the proportion of days covered (PDC) being assessed in nine articles [28,34,39,41,44,49,53,58,60]. Other investigations included the use of medication possession ratio (MPR) [42], medication refill adherence (MRA) [26], cumulative medication adherence (CMA) [27], the proportion of months covered (PMC) [39], percentage of doses taken (PDT), and percentage of doses taken correctly (PDTc) [59], as well as other ratios specifically calculated [29, 55]. Other adherence methods employed to study adherence included the direct count of the medication [21], the use of electronic medication packaging devices [25,54], and specialist opinion [22].
Several studies opted for a multifeatured approach, selecting two or more of the medication adherence measures previously mentioned to increase the accuracy of the information needed to correctly evaluate patients' adherence levels; e.g., one study applied four tools [MMAS-8, medication adherence rate scale (MARS), prescription refill records, and therapeutic drug monitoring (TDM)], individually and to calculate a composite score [30]. Another study regarding patients’ adherence to non-pharmacological treatment used a composed instrument that included the following parameters: weight and abdominal circumference control, practice of physical activity, and alcohol consumption [47]. Across all of the examined domains, the following associations had a constant and statistically significant association with adherence: satisfaction with communication, patient-provider relationship, digital/electronic health (eHealth) and telehealth/telemedicine services using smartphones and other technological devices (mHealth), having depression/occurrence of vital events in the patient’s life, cognitive impairment, frailty and disability, history of previous hospitalization, the type of drug, drug appearance, satisfaction with their treatment, health literacy, self-efficacy, and implicit/explicit attitudes.
Higher adherence was observed among black patients with a high level of communication with their clinicians and with higher levels of involvement in shared decision-making [26]. A better patient-provider relationship, where clinicians always explained things clearly and always listened to their patients while showing concern, led to patients being more likely to be adherent, irrespective of their ethnic group [26,33]. On the other hand, lower adherence was more common when discussions with their clinicians were less patient-centered, less psychosocially focused, and characterized by a lack of detailed discussion or clear explanation about their antihypertensive medications [54]. These three studies used a cohort study design, and when assessing the methodological quality, two of them were rated as moderate risk [26,54], and one study as low risk [33].
Both eHealth and mHealth technological solutions have shown positive effects in enhancing adherence. The SMASH app proved to increase MA and lower SBP and DBP at each subsequent evaluation during a nine-month period, managing information from medication trays and BP monitors connected to the app, and sending alerts to patients to remind them to take their medicines and monitor BP [25]. However, as previously mentioned, this RCT showed some quality concerns. The Medisafe app, used in an RCT with a low risk of bias, demonstrated an improvement in self-reported adherence, mostly in patients with previously low levels of adherence, making them moderately adherent by the end of the follow-up period, but with no changes in SBP [46]. This app involved self-reporting of medication and BP measurements, providing alerts to remind patients to take their medicines, and allowing them to designate a “Medfriend” that could access patients’ reports and work as a peer support helping them to comply with the recommended therapy.
Another RCT with a low risk of bias suggested that using mobile phone text messaging containing educative information (about diet, medication schedule, and statements regarding the importance of medication intake) seemed to improve adherence, although the decreased BP values were not statistically significant [61]. Application of telemedicine tools in a literacy-sensitive and motivational coaching program, with monthly telephone encounters with rural patients, improved medication adherence over time and demonstrated effective drops in systolic and diastolic BP, predominantly in the low adherence group at the baseline compared to the higher adherence groups in a cohort study with low risk of bias [65]. One quasi-experimental study used an electronic medication organizer equipped with an alarm clock to help adults organize and remind the daily intake of their medicines (especially for those not familiar with modern technology such as mobile phone apps) and gave healthcare professionals access to patient’s reported adherence by the device [63]. In this study, the authors observed a significant change in adherence patterns (from less to moderate adherence) and a drop in the mean SBP and DBP of 18.5 mmHg and 4.3 mmHg, respectively; however, these results must be interpreted carefully since the study seems to carry a moderate risk of bias.
Some condition-related factors are important modifiers of adherence, with non-adherence being higher among those with newly diagnosed depression, with depression symptoms, the necessity of psychopharmacological treatment, or being affected by three or more vital events (defined as adverse circumstances that occur at any stage of the patient’s life and can induce discomfort and anxiety, like the death of close family members, interpersonal conflicts, and suffering from physical illnesses) [60, 62]. A concept-wide association study utilizing text from clinical notes to identify possible risk factors for medication non-adherence found that phrases on clinical notes including “mental illness”, “anxiety/depression”, “mood”, “SSRI”, “cognition”, “confusion”, “mental status” and “memory” worked as a predictor of poor adherence [58].
Medication adherence worsens with mild cognitive impairment, even in patients without dementia [27]. It is also lower for those with a high probability of being frail and who experienced serious fall injuries following the initiation of antihypertensive medication [39,60]. Hypertensive patients with a history of previous hospitalization were unlikely to be adherent [44,49]. In contrast, one study concluded that not having a history of previous hospitalization was associated with non-adherence [39]. In the first two studies, the mean age was 47.2 and 51.7 years respectively, while it was 75 years in the third, with all three of them having large sample sizes. Based on the methodological quality assessment of the studies in the domain of comorbidities-related factors, most of the studies showed a low risk of bias, with only three being rated as moderate risk [27,44,62].
Several therapy-related factors affect adherence, the most notable being the drug class consumed, with patients who initiated angiotensin receptor blockers (ARBs) [41,49,60], calcium channel blockers (CCB) [45,49], or angiotensin-converting-enzyme inhibitors (ACE-I) [60] monotherapy more likely to be adherent, whereas those who initiated beta blockers (BB) [47,49], thiazides diuretics [38,49,60], or loop diuretics [60] tended to show low adherence. Although one study associated CCB use with poor medication adherence in a cross-sectional setting with a low risk of bias, the authors affirmed that this finding was related to the limited availability of alternative antihypertensive agents in the study setting [24]. ACE-I showed a high association with adherence, but when compared to the use of ARBs, patients under ACE-I medication had lower adherence [49]. However, the low adherence in the study previously mentioned was attributed by the authors to the population in the study (east Asians) being rarely prescribed this class since they have higher rates of adverse effects to ACE-Is (i.e., cough).
Two articles showed adherence to be highly variable between individual drugs of the same class: bendroflumethiazide and chlortalidone were associated with both the highest and lowest adherence levels respectively, and with CCB (nifedipine, felodipine, and lercanidipine) showing higher adherence compared to other classes in a cohort study with low risk of bias; however, these discrepancies are likely due to pharmacokinetic properties and/or tolerability profiles for the different drugs [43]. Another cohort study with a moderate risk of bias reported amlodipine to be more commonly associated with higher adherence when compared to Disothiazide [55]. Two studies found that the appearance of drugs had a significant role in maintaining adherence to the recommended treatment. Changes in appearance and/or name in bioequivalent drugs could negatively affect patients' adherence and lead to errors in medication-taking behavior; also, the difficulty in reading the information on the medicine packages has been associated with non-adherence [21,35]. Overall treatment satisfaction (evaluated in the following three domains: effectiveness, side effects, and convenience) was reported to be an important predictor of good adherence [24].
Health literacy played an important role in three studies presenting with low risk of bias. A statistically significant association between low adherence and health literacy levels was found, with the main domains associated being communication and decision-making skills, independently of sociodemographic characteristics [40]. Medication literacy is positively related to adherence to therapy and involves three main domains: knowledge, attitude, and behaviors (but no association was found with skill literacy) though the correlations were weak [56]. Another study demonstrated that patient’s knowledge of hypertension was associated with better adherence to both non-pharmacological (regular physical activity, weight reduction diet, and dietary salt restriction) as well as pharmacological treatment, lower and better controlled SBP and DBP, and fewer hospitalizations when compared to those with low literacy levels [50]. Poor self-efficacy in managing hypertension, which refers to the individual’s confidence in their capacity to accomplish the therapeutic recommendations, is associated with low adherence [28]. Furthermore, implicit and explicit attitudes are related to adherence, with both of them explaining a considerable variance in low pharmacy refill and self-reported adherence. Implicit attitudes seem to affect medication-taking behavior, working as a subconscious or automatic response (both in favor and against taking medications as prescribed), that may not be noticed when using self-reported adherence questionnaires [28,36].
Other determinants evaluated in the included studies neither showed a constant relationship with adherence nor any statistically significant associations: patient’s gender, age, racial/ethnic minority, educational level, marital status, employment, financial status, social support/aids, living in metropolitan areas compared to rural areas, income level of the country, healthcare provider visits or access to specialized health care use, having health insurance or access to healthcare, having other comorbidities, diagnosis of diabetes, history of cardiovascular diseases, relation with the duration of the diagnosis of HBP, smoking or drug/alcohol abuse, side effects from the pharmacological therapy, complexity of the overall medication regimen, dose regimen with antihypertensives, patient’s body mass index, beliefs regarding the disease, and acceptance of illness.
Discussion
The purpose of this study was to summarize the evidence regarding the predictors of adherence to antihypertensive therapies in studies published in the last five years, describe the limitations of the existing research, and highlight important areas to address in the future. It involved a systematic review where the predictors of adherence were identified, extracted, and categorized into five domains. The overall quality assessment of the 45 included studies revealed small concerns, with the majority of the articles exhibiting a moderate to strong methodological quality, and statistically significant association with adherence, statistically significant association with non-adherence, or non-significant association, which are summarized in Table 2.
Table 2. Determinants of adherence to antihypertensive therapy.
| Determinants of adherence | Number of studies associated with adherence | Number of studies associated with non-adherence | Number of studies with nonsignificant association | |
| Socioeconomic | Gender | 2 [31,52] | 10 [21,36,38,40,43,44,47,49,51,60] | 11 [22,23,27,35,40,45,48,55,56,64,65] |
| Age | 10 [23,31,38,43,44,47-49,55,60] | 3 [21,34,64] | 8 [22,24,35,45,51,52,56,65] | |
| Racial/ethnic minority status | 1 [39] | 1 [65] | 3 [23,31,35] | |
| Education level | 1 [52] | 6 [23,24,31,48,51,57] | ||
| Marital status | 1 [23] | 2 [28,35] | 3 [23,24,35] | |
| Employment | 2 [51,52] | 1 [23] | 3 [47,48,56] | |
| Financial status | 2 [27,56] | 5 [35,36,45,47,57] | 3 [23,48,55] | |
| Social support/aid | 3 [22,23,48] | 1 [57] | ||
| Living in metropolitan vs. rural areas | 1 [27] | 1 [51] | 1 [52] | |
| Country income level | 1 [45] | 1 [45] | ||
| Healthcare system/team | Healthcare provider visits | 3 [34,48,57] | 2 [32,58] | 1 [33] |
| Specialized healthcare use | 2 [37,49] | 1 [44] | 2 [23,24] | |
| Satisfaction with communication | 1 [26] | |||
| Patient-provider relationship | 2 [26,33] | 1 [54] | ||
| Health insurance | 1 [60] | 1 [60] | 1 [44] | |
| Access to healthcare | 1 [27] | 1 [35] | ||
| eHealth/mHealth | 5 [25,46,61,63,65] | |||
| Condition-related | Comorbidities | 3 [27,39,49] | 4 [21,27,49,57] | 6 [24,45,48,51,52,56] |
| Having diabetes | 3 [34,47,49] | 3 [54,58,60] | 5 [22,24,55,57,64] | |
| Having depression/vital events | 2 [60,62] | |||
| History of CVD | 3 [27,39,44] | 2 [22,60] | 3 [24,55,64] | |
| Cognitive impairment | 2 [27,58] | |||
| Duration of HBP | 2 [23,51] | 1 [24] | 4 [35,45,52,56] | |
| Frailty and disability | 2 [39,60] | |||
| Drug/alcohol abuse | 4 [22,24,47,57] | 4 [23,24,55,64] | ||
| Previous hospitalization | 1 [39] | 3 [39,44,49] | ||
| Therapy-related | Side effects | 3 [24,58,65] | 2 [51,59] | |
| Medication regimen | 3 [29,55,59] | 6 [21,24,49,58,60,64] | 3 [35,43,49] | |
| Dose regimen | 8 [29,34,41,42,44,49,53,59] | 5 [35,36,38,41,43] | 3 [41,51,56] | |
| Type of drug | 6 [24,41,45,49,55,60] | 6 [24,38,43,47,49,60] | ||
| Drug appearance | 2 [21,35] | |||
| Treatment satisfaction | 1 [24] | |||
| Patient-related | Body mass index | 4 [22,45,55,64] | ||
| Beliefs | 1 [58] | 1 [30] | ||
| Health literacy | 2 [50,56] | 1 [40] | ||
| Acceptance of illness | 2 [30,52] | |||
| Self-efficacy | 1 [28] | |||
| Implicit and explicit attitudes | 1 [28] | 2 [28,36] | ||
The role of the patient-provider relationship has been shown to promote adherence to antihypertensive therapy, with the main contributor to this relationship being the quality of the explanations provided regarding the therapy, with a patient-centered speech that addresses psychological concerns, while promoting empowerment and involvement in the decision-making process. These findings are in line with previous review articles, which illustrated the increasing importance of understanding the patient’s perspective to promote adherence and how the quality of communication (showing concern and providing information) appears to have more influence than the absolute quantity of time spent with the clinical interview [66,67]. Practitioners should investigate adherence in a blame-free environment, offer individualized solutions, and promote patients' role in making decisions about their therapy plans.
Advances in electronic and digital health technology (eHealth and mHealth) could serve as supportive tools to improve adherence. In this review, two mobile phone apps were analyzed and showed positive effects on adherence to therapy using alerts to remind patients to take their medications, using measuring tools of adherence and blood pressure control (both wireless connected to the phone or introduced through self-reported systems), and elaborating reports about individuals that allow clinicians to study their behavior towards medication [25,46]. An RCT investigating the impact of text messaging containing educative information also showed improvement in adherence [61]. Loureiro et al. conducted a systematic review on the impact of the use of mobile phone technologies and reported positive results and better effectiveness in promoting adherence, although with moderate quality and some inconsistent findings [68]. A review article found a tendency of increased medication adherence levels and better blood pressure control while using mobile phone interventions (such as text messages providing drug intake/blood pressure monitoring reminders or smartphone applications with associated alerts according to the patient’s records on their adherence behavior) [69].
A systematic review published in the European Heart Journal reported similar results [70]. Monthly telephone encounters with patients engaged in a motivational coaching program seemed to increase medication adherence over time and demonstrated effective drop changes in systolic and diastolic BP, proving that telemedicine strategies, which have become a more common practice during the COVID-19 outbreak, can be useful [65]. For older adults and/or those not familiar with modern technologies, the use of electronic multicompartment medication devices with reminder systems might improve adherence and blood pressure control [63]. However, according to a systematic review, several studies suffer from methodological limitations, such as a study included in this review [71]. All these technological interventions showed a promising non-pharmacological strategy to consider as an adjunct to enhance adherence to antihypertensive medication and to follow-up adherence trajectories, but further investigations are required.
Psychological disorders such as depression, experiencing depressive symptoms or stressful life events (vital events) were predictors of low adherence, which is in line with other literature reviews and meta-analyses [72-74]; however, this association was not always consistent as some studies reported no link to adherence [67,75]. There was also a direct association with poor self-efficacy and low medication adherence, also confirmed in previous studies [73,76]. Furthermore, depression might lead to low adherence through the mechanism of low self-efficacy, as has been previously described [77,78]. Cognitive impairment was also associated with low adherence, even in patients without dementia [27]. Similarly, Luz et al. conducted a systematic review that confirmed the association between cognitive impairment and low adherence, but the mechanisms underlying it were still not clear [79].
Measures that promote the early detection of patients with possible diagnostic of depression, cognitive impairment and individuals with higher frailty and/or presenting with disabilities, will enable physicians to adjust care to adapt to the different constraints/conditions and optimize medication management to achieve a good blood pressure control. For example, one trial found that integrated management of hypertension and depression in patients with both conditions led to better outcomes in medication adherence [80]. Other factors negatively associated with adherence to antihypertensive therapy included being frail and having a history of serious fall injuries, with similar results found in another review [81].
History of previous hospitalization was not clearly demonstrated as a determinant of adherence, since two studies showed a significant association with low adherence [44,49]; however, another study showed that not having a history of hospitalization was linked to low adherence [39]. According to an integrative review of the literature, hospitalization was a predictor of higher medication adherence [81]. One possible explanation for higher adherence in patients with a history of hospitalization might be linked to the influence of stopping and reinitiating medication, observed previously in the use of statins, but this factor should be confirmed for antihypertensive medication [82].
Persistent low antihypertensive adherence can be partly explained by explicit (conscious or deliberative) behaviors, as well as by implicit attitudes underlying a subconscious or automatic response that influences medication-taking behavior, which might be expressed through forgetfulness [28,36]. Indeed, a study included in this review found significant data where implicit and explicit attitudes accounted for an additional variation in pharmacy refill adherence, but only explicit attitudes explained the difference in self-reported questionnaires [28]. These findings were consistent with prior data observed in patients with other chronic diseases [83,84]. Therefore, implicit attitudes toward medications may explain adherence variation that could not be explained when using self-reported questionnaires, which reinforced the importance of using both objective and subjective measures when investigating patients’ medication adherence. Adults with high health literacy related to both medication and hypertension knowledge not only improved medication adherence and systolic and diastolic BP levels but also had higher engagement in non-pharmacological therapy, such as regular physical activity, weight reduction diet, and dietary salt restriction. Similar results were found in three of the reviews [73,81,85].
The drug class of the medication prescribed was shown to be linked to strengthening patient adherence. ARBs, ACE-I, and CCB classes in monotherapy had a positive correlation with adherence. Moreover, low adherence was common in patients who were prescribed BB and diuretics, and the side effects of these drugs may explain this since they had been previously considered a major factor behind poor adherence [86]. The use of BB not only affected medication adherence but also non-pharmacological treatment (i.e., physical activity), because of the dizziness due to the hypotensive effect induced by this drug. On the other hand, diuretics can cause urinary frequency and erectile dysfunction, which might not be tolerated by patients. A meta-analysis by Kronish et al. has reported higher adherence to ACE-Is and ARBs, followed by CCBs, with the lowest adherence reported in patients under diuretics and β-blockers [87]. ACE-Is are widely prescribed in Western countries, being used as a primary treatment for hypertension [88,89]. When treatment was convenient to the patient, side effects were removed, and effectiveness was achieved, patient satisfaction was attained, and that satisfaction worked as a predictor of good adherence. Barbosa et al. found that treatment satisfaction was associated with higher levels of compliance and improved persistence with the proposed therapy [90]. Other studies have also mentioned the association between adherence and satisfaction with treatment [81,91].
Changing the appearance/name of bioequivalent drugs or having difficulty in reading the information in the respective packaging seemed to negatively affect patients’ adherence, which aligns with other studies [92,93]. Patients would probably benefit from prescriptions based on the active principle of the drugs and better regulation of the packaging. The name and appearance should be the same among bioequivalent drug classes. This challenge has been previously mentioned and concrete solutions presented [94]. Even though hypertension is a very common condition and adherence to therapy has been widely studied over the past years, findings from several of the included studies demonstrated some discrepancy, with a lot of possible determinants having an inconsistent association and lack of significance, despite having been previously mentioned by other reviews as having a statistically significant association with adherence to pharmacological and non-pharmacological therapy; this brings to fore something that is well known: antihypertensive therapy adherence behavior is multifactorial, with a complex interaction associated with different factors such as like socioeconomic, healthcare system/team-related, condition-related, therapy-related, and patient-related [95,96].
Since the absence of substantial evidence does not necessarily mean the absence of effectiveness, it is wise to highlight some other predictors that may negatively influence adherence but have not been explored and come across as relevant in other reviews. These include social support and financial aid (i.e., co-payment of healthcare access/medicines), marital status, racial/ethnic minorities, previous history of cardiovascular diseases, complexity of the prescribed regimen, and acceptance of illness (since hypertension frequently presents as an asymptomatic condition). Given the multifactorial nature of medication adherence, we can easily understand that no unique intervention is usually effective in isolation and efforts should be made to apply interventions at multiple levels [97]. Additionally, these interventions should be tailored at an individual level, ensuring patient-centered care, through active involvement of patients, their families, and healthcare providers.
Study Limitations
This systematic review has a few limitations. Primarily, it included studies that used different measurement instruments and methodologies, with different cut-off points to label patients’ behavior as adherent or non-adherent. Furthermore, there was a potential risk of overestimation and recall bias since a considerable number of studies used self-reported measures. Additionally, due to the considerable heterogeneity among the included studies (especially as a consequence of having different tools to measure adherence), the diverse populations being studied (age, gender, country, socioeconomic and cultural setting) and the duration of the follow-up period among different studies, it is difficult to conduct a meta-analysis, which is considered to provide the highest level of evidence and could lead to more significant conclusions.
Another limitation was that a large proportion of the included studies used a cross-sectional design, which constrained the establishment of causal relationships and, eventually, the generalization of the findings to the targeted populations. Several other limitations in this study are those inherent to systematic reviews in general. In this review, we included only those studies found via well-known electronic databases, excluding gray literature, books, and other dissertations/studies that could be obtained manually. Reporting bias and publication bias may be present given that positive findings have a higher chance of being published, while studies with no statistically significant findings are not likely to be published in peer-reviewed journals. Hence, our findings must be interpreted within the context of our study limitations. Future research should strive to establish acceptable parameters to measure adherence and define a gold standard method, preferably involving a combination of both direct and indirect measures of adherence. Studies should also employ longitudinal designs, with longer follow-up periods that enable investigators to assess long-term adherence behaviors and trajectories over time.
Conclusions
Adherence to both pharmacological and non-pharmacological therapy plays an important role in the control of BP in patients with hypertension, and ensuring it represents a key challenge in public health. The barriers to adherence are multiple, complex, and often interconnected between socioeconomic, patient, therapy, condition, and healthcare system levels. Hence, healthcare teams should endeavor to study patients’ non-adherence motives, using the most suitable tools to devise adherence interventions and tailor them to individual patient needs. Based on our findings, poor adherence is linked to factors such as an unsatisfactory patient-practitioner relationship and communication, a history of depression or occurrence of vital events, cognitive impairment, low health literacy and self-efficacy, presence of frailty and/or disabilities, or a history of previous hospitalization. Moreover, the antihypertensive agent chosen, with its possible side effects, and changes in medication appearance might also influence treatment satisfaction and, ultimately, explain low adherence patterns.
In this systematic review, besides traditional determinants of adherence, implicit attitudes towards medication emerged as a novel determinant that should be taken into account when determining future interventions to achieve better outcomes in patients’ therapy adherence. Furthermore, integrating new technological approaches with hybrid systems, combining eHealth and mHealth, has the potential to aid in better follow-up and improve therapy adherence, while empowering patients to deal with the condition. We strongly recommend conducting additional research with sturdier evidence and involving newer technologies.
Acknowledgments
I am deeply grateful to Dr. Sónia Marques Moreira for her invaluable guidance and support. Her mentorship has been instrumental in shaping my journey. Thank you for your wisdom and dedication.
Appendices
Table 3. Joanna Briggs Institute (JBI) critical appraisal checklist for analytical cross-sectional study.
Q1) Were the criteria for inclusion in the sample clearly defined? Q2) Were the study subjects and the setting described in detail? Q.3) Was the exposure measured in a valid and reliable way? Q.4) Were objective and standard criteria used for measuring the condition? Q.5) Were confounding factors identified? Q.6) Were strategies to deal with confounding factors stated? Q.7) Were the outcomes measured in a valid and reliable way? Q.8) Was appropriate statistical analysis used?
“✓” indicates yes; “✕” indicates no; “?” indicates unclear. Criteria: high quality (>70% of “yes” scores), moderate quality (50-69%), and low quality (<50%)
| Study | Q1 | Q2 | Q3 | Q4 | Q5 | Q6 | Q7 | Q8 | % yes/risk |
| Avataneo et al. [22] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | Low |
| Barbosa et al. [23] | ✓ | ✓ | ✕ | ✕ | ✕ | ✕ | ✓ | ✓ | Moderate |
| Berhe et al. [24] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | Low |
| Craig et al. [28] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | Low |
| Durand et al. [30] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | Low |
| Fang et al. [31] | ✓ | ✓ | ✕ | ✕ | ✓ | ✓ | ✕ | ✓ | Moderate |
| Fernandez et al. [32] | ✕ | ✕ | ✓ | ✓ | ✕ | ✕ | ✓ | ✕ | High |
| Gewehr et al. [35] | ✓ | ✓ | ✓ | ✓ | ✕ | ✕ | ✓ | ✓ | Low |
| Gniwa et al. [36] | ✓ | ✓ | ✓ | ✕ | ✕ | ✕ | ✓ | ✓ | Moderate |
| Heizomi et al. [40] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | Low |
| Ishida et al. [41] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | Low |
| Macquart de Terline et al. [45] | ✕ | ✕ | ✓ | ✓ | ✓ | ✕ | ✓ | ✓ | Moderate |
| Nascimento et al. [47] | ✓ | ✓ | ? | ✕ | ✕ | ✕ | ? | ✓ | Moderate |
| Nashilongo et al. [48] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | Low |
| Paczkowska et al. [50] | ✓ | ✓ | ✕ | ✓ | ✓ | ✕ | ✓ | ✓ | Low |
| Pan et al. [51] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | Low |
| Pluta et al. [52] | ✓ | ✓ | ✓ | ✓ | ✕ | ✕ | ✓ | ✓ | Low |
| Shi et al. [56] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | Low |
| Shimels et al. [57] | ✓ | ✓ | ✓ | ✓ | ✕ | ✕ | ✕ | ✓ | Moderate |
| Vázquez Machado et al. [62] | ✕ | ✕ | ✓ | ✓ | ✕ | ✕ | ✓ | ✓ | Moderate |
| Wakai et al. [64] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | Low |
Table 4. Risk Of Bias in Non-randomized Studies - of Interventions (ROBINS-I) assessment tool.
| Study | Confounding | Selection bias | Classification of intervention | Intended intervention | Missing data | Measures of outcome | Reported results | Overall |
| Arancón-Monge et al. [21] | Low | Low | Low | Low | Moderate | Moderate | Low | Moderate |
| Chang et al. [26] | Low | Low | Low | Low | Low | Moderate | Low | Moderate |
| Cho et al. [27] | Low | Moderate | Low | Low | Low | Low | Low | Moderate |
| Del Pinto et al. [29] | Low | Moderate | Low | Low | Low | Low | Low | Moderate |
| Fortuna et al. [33] | Low | Low | Low | Low | Low | Low | Low | Low |
| Gao et al. [34] | Low | Low | Low | Low | Low | Low | Low | Low |
| Gupta et al. [37] | Moderate | Moderate | Low | Low | Low | Moderate | Low | Moderate |
| Gupta et al. [38] | Moderate | Moderate | Moderate | Low | Low | Low | Low | Moderate |
| Hargrove et al. [39] | Low | Low | Low | Low | Low | Low | Low | Low |
| Kim et al. [42] | Low | Low | Low | Low | Low | Moderate | Low | Moderate |
| Kulkarni et al. [43] | Low | Low | Low | Low | Low | Low | Low | Low |
| Lauffenburger et al. [44] | Moderate | Moderate | Low | Low | Low | Moderate | Low | Moderate |
| Nishimura et al. [49] | Low | Low | Low | Low | Low | Low | Low | Low |
| Rea et al. [53] | Low | Low | Low | Low | Moderate | Low | Low | Moderate |
| Schoenthaler et al. [54] | Moderate | Moderate | Low | Low | Low | Low | Low | Moderate |
| Shani et al. [55] | Moderate | Low | Low | Low | Low | Low | Low | Moderate |
| Singh et al. [58] | Low | Low | Low | Low | Low | Low | Low | Low |
| Tajeu et al. [60] | Low | Low | Low | Low | Low | Low | Low | Low |
| Vieira et al. [63] | Moderate | Moderate | Low | Low | Low | Low | Low | Moderate |
| Wu et al. [65] | Low | Low | Low | Low | Low | Low | Low | Low |
Table 5. Revised Cochrane risk-of-bias tool for randomized trials (RoB 2) assessment tool.
| Study | Randomization process | Deviations from the intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported result | Overall |
| Chandler et al. [25] | Low | Low | Low | Some concerns | Low | Some concerns |
| Morawski et al. [46] | Low | Low | Low | Low | Low | Low |
| Sung et al. [59] | Low | Low | Low | Low | Low | Low |
| Varleta et al. [61] | Low | Low | Low | Low | Low | Low |
The authors have declared that no competing interests exist.
Author Contributions
Concept and design: Pedro D. Ferreira Jr., Jose A. Simoes Sr., Denise C. Velho Sr.
Acquisition, analysis, or interpretation of data: Pedro D. Ferreira Jr., Jose A. Simoes Sr., Denise C. Velho Sr.
Drafting of the manuscript: Pedro D. Ferreira Jr., Jose A. Simoes Sr., Denise C. Velho Sr.
Critical review of the manuscript for important intellectual content: Pedro D. Ferreira Jr., Jose A. Simoes Sr., Denise C. Velho Sr.
Supervision: Jose A. Simoes Sr.
References
- 1.World Health Organization. Cardiovascular diseases (CVDs) [ Dec; 2021 ]. 2021. https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)?gad_source=1&gclid=Cj0KCQjw2uiwBhCXARIsACMvIU3ullV7CSALBVRw3gbdjNwoER9ZsMQt1uBtiY497VODH9O6maCsyEIaAoV4EALw_wcB https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)?gad_source=1&gclid=Cj0KCQjw2uiwBhCXARIsACMvIU3ullV7CSALBVRw3gbdjNwoER9ZsMQt1uBtiY497VODH9O6maCsyEIaAoV4EALw_wcB
- 2.OECD. Health at a glance 2021: OECD indicators. [ Dec; 2021 ]. 2021. https://www.oecd-ilibrary.org/social-issues-migration-health/health-at-a-glance-2021_ae3016b9-en https://www.oecd-ilibrary.org/social-issues-migration-health/health-at-a-glance-2021_ae3016b9-en
- 3.Trends in life expectancy in EU and other OECD countries: why are improvements slowing? Raleigh V. https://www.oecd-ilibrary.org/social-issues-migration-health/trends-in-life-expectancy-in-eu-and-other-oecd-countries_223159ab-en OECD Health Work Pap. 2019;108:24–26. [Google Scholar]
- 4.Instituto Nacional de Estatística. National Statistics Institute (site in Portuguese) Estatísticas da Saúde. [ Dec; 2021 ]. Estatísticas da Saúde. https://www.ine.pt/ngt_server/attachfileu.jsp?look_parentBoui=492336067&att_display=n&att_download=y https://www.ine.pt/ngt_server/attachfileu.jsp?look_parentBoui=492336067&att_display=n&att_download=y
- 5.Directorate-General for Health. National program for cerebrovascular diseases (site in Portuguese) Lisboa: DGS. [ Dec; 2021 ]. 2017. https://comum.rcaap.pt/handle/10400.26/21114 https://comum.rcaap.pt/handle/10400.26/21114
- 6.Prevalence, awareness, treatment and control of hypertension and salt intake in Portugal: changes over a decade. The PHYSA study. Polonia J, Martins L, Pinto F, Nazare J. J Hypertens. 2014;32:1211–1221. doi: 10.1097/HJH.0000000000000162. [DOI] [PubMed] [Google Scholar]
- 7.Worldwide trends in hypertension prevalence and progress in treatment and control from 1990 to 2019: a pooled analysis of 1201 population-representative studies with 104 million participants. Lancet. 2021;398:957–980. doi: 10.1016/S0140-6736(21)01330-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.High blood pressure and cardiovascular disease. Fuchs FD, Whelton PK. Hypertension. 2020;75:285–292. doi: 10.1161/HYPERTENSIONAHA.119.14240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.World Health O. Hypertension and COVID- 19: scientific brief, 17 June 2021. WHO. Hypertension and COVID-19: scientific brief. [ Dec; 2021 ]. 2021. https://iris.who.int/bitstream/handle/10665/341848/WHO-2019-nCoV-Sci-Brief-Hypertension-2021.1-eng.pdf?sequence=1 https://iris.who.int/bitstream/handle/10665/341848/WHO-2019-nCoV-Sci-Brief-Hypertension-2021.1-eng.pdf?sequence=1
- 10.Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. Richardson S, Hirsch JS, Narasimhan M, et al. JAMA. 2020;323:2052–2059. doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Instituto Nacional de Estatística. Inquérito Nacional de Saúde 2014. National Statistics Institute. 2014 National Health Survey (site in Portuguese) Lisboa : INE. [ Dec; 2021 ]. 2014. https://www.ine.pt/xportal/xmain?xpid=INE&xpgid=ine_publicacoes&PUBLICACOESpub_boui=263714091&PUBLICACOESmodo=2 https://www.ine.pt/xportal/xmain?xpid=INE&xpgid=ine_publicacoes&PUBLICACOESpub_boui=263714091&PUBLICACOESmodo=2
- 12.2018 ESC/ESH guidelines for the management of arterial hypertension. Williams B, Mancia G, Spiering W, et al. Eur Heart J. 2018;39:3021–3104. doi: 10.1093/eurheartj/ehy339. [DOI] [PubMed] [Google Scholar]
- 13.Global epidemiology, health burden and effective interventions for elevated blood pressure and hypertension. Zhou B, Perel P, Mensah GA, Ezzati M. Nat Rev Cardiol. 2021;18:785–802. doi: 10.1038/s41569-021-00559-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Effects of blood pressure lowering on outcome incidence in hypertension. 1. Overview, meta-analyses, and meta-regression analyses of randomized trials. Thomopoulos C, Parati G, Zanchetti A. J Hypertens. 2014;32:2285–2295. doi: 10.1097/HJH.0000000000000378. [DOI] [PubMed] [Google Scholar]
- 15.Implications for medical activity of differences between individuals with controlled and uncontrolled hypertension. Martins RD, Santiago LM, Reis MT, Roque AC, Pinto M, Simões JA, Rosendo I. Rev Port Cardiol (Engl Ed) 2019;38:745–753. doi: 10.1016/j.repc.2019.05.009. [DOI] [PubMed] [Google Scholar]
- 16.Managing 'resistance': is adherence a target for treatment? Burnier M. Curr Opin Nephrol Hypertens. 2014;23:439–443. doi: 10.1097/MNH.0000000000000045. [DOI] [PubMed] [Google Scholar]
- 17.Nonadherence to antihypertensive drugs: a systematic review and meta-analysis. Abegaz TM, Shehab A, Gebreyohannes EA, Bhagavathula AS, Elnour AA. Medicine (Baltimore) 2017;96:0. doi: 10.1097/MD.0000000000005641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.De Geest S, Sabaté E. Eur J Cardiovasc Nurs. Vol. 2. 2003: Geneva, Switzerland: World Health Organization; 2003. Adherence to long-term therapies: evidence for action; p. 323. [DOI] [PubMed] [Google Scholar]
- 19.Compliance, concordance, adherence. Aronson JK. Br J Clin Pharmacol. 2007;63:383–384. doi: 10.1111/j.1365-2125.2007.02893.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Medication adherence: WHO cares? Brown MT, Bussell JK. Mayo Clin Proc. 2011;86:304–314. doi: 10.4065/mcp.2010.0575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Effects of changing the appearance of medications in safety and adherence in chronic patients over 65 years of age in primary care. CAMBIMED Study (Article in Spanish) Arancón-Monge JM, de Castro-Cuenca A, Serrano-Vázquez Á, Campos-Díaz L, Rodríguez Barrientos R, Del Cura-González I. Aten Primaria. 2020;52:77–85. doi: 10.1016/j.aprim.2019.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Therapeutic drug monitoring-guided definition of adherence profiles in resistant hypertension and identification of predictors of poor adherence. Avataneo V, De Nicolò A, Rabbia F, et al. Br J Clin Pharmacol. 2018;84:2535–2543. doi: 10.1111/bcp.13706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Factors associated with adherence of adults/elderly people to the treatment of arterial hypertension in primary care (Article in Portuguese) Barbosa MEM, Bertelli EVM, Aggio CdM, Scolari GAdS, Marcon SS, Carreira L. Rev Enferm UERJ. 2019;27:2–4. [Google Scholar]
- 24.Impact of adverse drug events and treatment satisfaction on patient adherence with antihypertensive medication - a study in ambulatory patients. Berhe DF, Taxis K, Haaijer-Ruskamp FM, Mulugeta A, Mengistu YT, Burgerhof JG, Mol PG. Br J Clin Pharmacol. 2017;83:2107–2117. doi: 10.1111/bcp.13312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Impact of a culturally tailored mHealth medication regimen self-management program upon blood pressure among hypertensive Hispanic adults. Chandler J, Sox L, Kellam K, Feder L, Nemeth L, Treiber F. Int J Environ Res Public Health. 2019;16:4–6. doi: 10.3390/ijerph16071226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Association between patient-clinician relationships and adherence to antihypertensive medications among black adults: an observational study design. Chang TJ, Bridges JF, Bynum M, et al. J Am Heart Assoc. 2021;10:0. doi: 10.1161/JAHA.120.019943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Association between cognitive impairment and poor antihypertensive medication adherence in elderly hypertensive patients without dementia. Cho MH, Shin DW, Chang SA, et al. Sci Rep. 2018;8:11688. doi: 10.1038/s41598-018-29974-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Implicit and explicit attitudes toward antihypertensive medications explain variation in pharmacy refill and self-reported adherence beyond traditional risk factors: potential novel mechanism underlying adherence. Craig LS, Peacock E, Mohundro BL, et al. J Am Heart Assoc. 2021;10:0. doi: 10.1161/JAHA.120.018986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Real-world antihypertensive treatment patterns, treatment adherence, and blood pressure control in the elderly: an Italian awareness-raising campaign on hypertension by senior Italia FederAnziani, the Italian Society of Hypertension and the Italian Federation of General Practitioners. Del Pinto R, Desideri G, Ferri C, Agabiti Rosei E. High Blood Press Cardiovasc Prev. 2021;28:457–466. doi: 10.1007/s40292-021-00465-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Medication adherence for resistant hypertension: assessing theoretical predictors of adherence using direct and indirect adherence measures. Durand H, Hayes P, Harhen B, et al. Br J Health Psychol. 2018;23:949–966. doi: 10.1111/bjhp.12332. [DOI] [PubMed] [Google Scholar]
- 31.Association between cost-related medication nonadherence and hypertension management among US adults. Fang J, Chang T, Wang G, Loustalot F. Am J Hypertens. 2020;33:879–886. doi: 10.1093/ajh/hpaa072. [DOI] [PubMed] [Google Scholar]
- 32.Factors related with non-adherence to pharmacological treatment in hypertensive patients of the "Policlinico XX Aniversario". An educational program design. Fernandez J, Mota CD, Tandron JMC, Ferrer YB, Esmores ZA. https://www.medigraphic.com/cgi-bin/new/resumenI.cgi?IDARTICULO=77344 Revista de Enferm Cardiovasc. 2017;9:18–20. [Google Scholar]
- 33.Patient experience with care and its association with adherence to hypertension medications. Fortuna RJ, Nagel AK, Rocco TA, Legette-Sobers S, Quigley DD. Am J Hypertens. 2018;31:340–345. doi: 10.1093/ajh/hpx200. [DOI] [PubMed] [Google Scholar]
- 34.A clinical prediction model of medication adherence in hypertensive patients in a Chinese community hospital in Beijing. Gao W, Liu H, Ge C, Liu X, Jia H, Wu H, Peng X. Am J Hypertens. 2020;33:1038–1046. doi: 10.1093/ajh/hpaa111. [DOI] [PubMed] [Google Scholar]
- 35.Adherence to pharmacological treatment of arterial hypertension in primary health care (Article in Portuguese) Gewehr DM, Bandeira VA, Gelatti GT, Colet C, Oliveira KR. Saúde Debate. 2018;42:179–190. [Google Scholar]
- 36.Predictors of poor adherence to hypertension treatment. Gniwa Omezzine R, Akkara A, Abdelkafi Koubaa A, Belguith Sriha A, Rdissi A, Amamou K. https://europepmc.org/article/med/31729707. Tunis Med. 2019;97:564–571. [PubMed] [Google Scholar]
- 37.Biochemical screening for nonadherence is associated with blood pressure reduction and improvement in adherence. Gupta P, Patel P, Štrauch B, et al. Hypertension. 2017;70:1042–1048. doi: 10.1161/HYPERTENSIONAHA.117.09631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Risk factors for nonadherence to antihypertensive treatment. Gupta P, Patel P, Štrauch B, et al. Hypertension. 2017;69:1113–1120. doi: 10.1161/HYPERTENSIONAHA.116.08729. [DOI] [PubMed] [Google Scholar]
- 39.Antihypertensive adherence trajectories among older adults in the first year after initiation of therapy. Hargrove JL, Pate V, Casteel CH, Golightly YM, Loehr LR, Marshall SW, Stürmer T. Am J Hypertens. 2017;30:1015–1023. doi: 10.1093/ajh/hpx086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gender differences in the associations between health literacy and medication adherence in hypertension: a population-based survey in Heris County, Iran. Heizomi H, Iraji Z, Vaezi R, Bhalla D, Morisky DE, Nadrian H. Vasc Health Risk Manag. 2020;16:157–166. doi: 10.2147/VHRM.S245052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Treatment patterns and adherence to antihypertensive combination therapies in Japan using a claims database. Ishida T, Oh A, Hiroi S, Shimasaki Y, Nishigaki N, Tsuchihashi T. Hypertens Res. 2019;42:249–256. doi: 10.1038/s41440-018-0127-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Effects of combination drugs on antihypertensive medication adherence in a real-world setting: a Korean nationwide study. Kim SJ, Kwon OD, Cho B, Oh SW, Lee CM, Choi HC. BMJ Open. 2019;9:0. doi: 10.1136/bmjopen-2019-029862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nonadherence to antihypertensive medications amongst patients with uncontrolled hypertension: A retrospective study. Kulkarni S, Rao R, Goodman JD, Connolly K, O'Shaughnessy KM. Medicine (Baltimore) 2021;100:0. doi: 10.1097/MD.0000000000024654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Effect of combination therapy on adherence among US patients initiating therapy for hypertension: a cohort study. Lauffenburger JC, Landon JE, Fischer MA. J Gen Intern Med. 2017;32:619–625. doi: 10.1007/s11606-016-3972-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Factors associated with poor adherence to medication among hypertensive patients in twelve low and middle-income Sub-Saharan countries. Macquart de Terline D, Kane A, Kramoh KE, et al. PLoS One. 2019;14:0. doi: 10.1371/journal.pone.0219266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Association of a smartphone application with medication adherence and blood pressure control: the MedISAFE-BP randomized clinical trial. Morawski K, Ghazinouri R, Krumme A, et al. JAMA Intern Med. 2018;178:802–809. doi: 10.1001/jamainternmed.2018.0447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Factors associated to the adherence to the non-pharmachological treatment of hypertension in primary health care. Nascimento MO, Belo RM, Araújo TL, Silva KG, Barros MD, Figueirêdo TR, Bezerra SM. Rev Bras Enferm. 2021;74:0. doi: 10.1590/0034-7167-2020-0173. [DOI] [PubMed] [Google Scholar]
- 48.Assessing adherence to antihypertensive therapy in primary health care in Namibia: findings and implications. Nashilongo MM, Singu B, Kalemeera F, et al. Cardiovasc Drugs Ther. 2017;31:565–578. doi: 10.1007/s10557-017-6756-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Adherence to antihypertensive medication and its predictors among non-elderly adults in Japan. Nishimura S, Kumamaru H, Shoji S, Sawano M, Kohsaka S, Miyata H. Hypertens Res. 2020;43:705–714. doi: 10.1038/s41440-020-0440-2. [DOI] [PubMed] [Google Scholar]
- 50.Impact of patient knowledge on hypertension treatment adherence and efficacy: a single-centre study in Poland. Paczkowska A, Hoffmann K, Kus K, et al. Int J Med Sci. 2021;18:852–860. doi: 10.7150/ijms.48139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Determinants of hypertension treatment adherence among a Chinese population using the therapeutic adherence scale for hypertensive patients. Pan J, Wu L, Wang H, Lei T, Hu B, Xue X, Li Q. Medicine (Baltimore) 2019;98:0. doi: 10.1097/MD.0000000000016116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Acceptance of illness and compliance with therapeutic recommendations in patients with hypertension. Pluta A, Sulikowska B, Manitius J, Posieczek Z, Marzec A, Morisky DE. Int J Environ Res Public Health. 2020;17:42–44. doi: 10.3390/ijerph17186789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Adherence to treatment by initial antihypertensive mono and combination therapies. Rea F, Savaré L, Franchi M, Corrao G, Mancia G. Am J Hypertens. 2021;34:1083–1091. doi: 10.1093/ajh/hpab083. [DOI] [PubMed] [Google Scholar]
- 54.Addressing the social needs of hypertensive patients: the role of patient-provider communication as a predictor of medication adherence. Schoenthaler A, Knafl GJ, Fiscella K, Ogedegbe G. Circ Cardiovasc Qual Outcomes. 2017;10:32–34. doi: 10.1161/CIRCOUTCOMES.117.003659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Adherence to oral antihypertensive medications, are all medications equal? Shani M, Lustman A, Vinker S. J Clin Hypertens (Greenwich) 2019;21:243–248. doi: 10.1111/jch.13475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Association between medication literacy and medication adherence among patients with hypertension. Shi S, Shen Z, Duan Y, Ding S, Zhong Z. Front Pharmacol. 2019;10:822. doi: 10.3389/fphar.2019.00822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Magnitude and associated factors of poor medication adherence among diabetic and hypertensive patients visiting public health facilities in Ethiopia during the COVID-19 pandemic. Shimels T, Asrat Kassu R, Bogale G, et al. PLoS One. 2021;16:0. doi: 10.1371/journal.pone.0249222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.A concept-wide association study to identify potential risk factors for nonadherence among prevalent users of antihypertensives. Singh K, Choudhry NK, Krumme AA, McKay C, McElwee NE, Kimura J, Franklin JM. Pharmacoepidemiol Drug Saf. 2019;28:1299–1308. doi: 10.1002/pds.4850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Adherence to triple-component antihypertensive regimens is higher with single-pill than equivalent two-pill regimens: a randomized controlled trial. Sung J, Ahn KT, Cho BR, et al. Clin Transl Sci. 2021;14:1185–1192. doi: 10.1111/cts.12979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Antihypertensive medication nonpersistence and low adherence for adults <65 years initiating treatment in 2007-2014. Tajeu GS, Kent ST, Huang L, et al. Hypertension. 2019;74:35–46. doi: 10.1161/HYPERTENSIONAHA.118.12495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Mobile phone text messaging improves antihypertensive drug adherence in the community. Varleta P, Acevedo M, Akel C, et al. J Clin Hypertens (Greenwich) 2017;19:1276–1284. doi: 10.1111/jch.13098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Depressive disorders and life events in patients with arterial hypertension and their relationship with therapeutic adherence (Article in Spanish) Vázquez Machado A, Mukamutara J, Meireles Ochoa MY, et al. http://scielo.sld.cu/scielo.php?pid=S1028-48182019000501079&script=sci_abstract&tlng=pt Multimed. 2019;23:1079–1094. [Google Scholar]
- 63.The use of an electronic medication organizer device with alarm to improve medication adherence of older adults with hypertension. Vieira LB, Reis AM, Ramos CÁ, Reis TM, Cassiani SH. Einstein (Sao Paulo) 2021;19:0. doi: 10.31744/einstein_journal/2021AO6011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Effect of number of medications and complexity of regimens on medication adherence and blood pressure management in hospitalized patients with hypertension. Wakai E, Ikemura K, Kato C, Okuda M. PLoS One. 2021;16:0. doi: 10.1371/journal.pone.0252944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.The effect of a practice-based multicomponent intervention that includes health coaching on medication adherence and blood pressure control in rural primary care. Wu JR, Cummings DM, Li Q, Hinderliter A, Bosworth HB, Tillman J, DeWalt D. J Clin Hypertens (Greenwich) 2018;20:757–764. doi: 10.1111/jch.13265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Medication adherence in hypertension. Poulter NR, Borghi C, Parati G, Pathak A, Toli D, Williams B, Schmieder RE. J Hypertens. 2020;38:579–587. doi: 10.1097/HJH.0000000000002294. [DOI] [PubMed] [Google Scholar]
- 67.Factors associated with antihypertensive medication non-adherence: a systematic review. van der Laan DM, Elders PJ, Boons CC, Beckeringh JJ, Nijpels G, Hugtenburg JG. J Hum Hypertens. 2017;31:687–694. doi: 10.1038/jhh.2017.48. [DOI] [PubMed] [Google Scholar]
- 68.Therapeutic adherence in hypertensive patients: review of the role of new platforms via cell phone or smartphone (Article in Portuguese) Loureiro RM, de Azevedo DA. Portuguese J Family Med and Gen Pract. 2021;37:330–341. [Google Scholar]
- 69.Mobile phone-based intervention in hypertension management. Andre N, Wibawanti R, Siswanto BB. Int J Hypertens. 2019;2019:9021017. doi: 10.1155/2019/9021017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.The role of mHealth for improving medication adherence in patients with cardiovascular disease: a systematic review. Gandapur Y, Kianoush S, Kelli HM, et al. Eur Heart J Qual Care Clin Outcomes. 2016;2:237–244. doi: 10.1093/ehjqcco/qcw018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.A systematic review of electronic multi-compartment medication devices with reminder systems for improving adherence to self-administered medications. Paterson M, Kinnear M, Bond C, McKinstry B. Int J Pharm Pract. 2017;25:185–194. doi: 10.1111/ijpp.12242. [DOI] [PubMed] [Google Scholar]
- 72.Depression and medication adherence in the treatment of chronic diseases in the United States: a meta-analysis. Grenard JL, Munjas BA, Adams JL, Suttorp M, Maglione M, McGlynn EA, Gellad WF. J Gen Intern Med. 2011;26:1175–1182. doi: 10.1007/s11606-011-1704-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Adherence to antihypertensive therapy. Peacock E, Krousel-Wood M. Med Clin North Am. 2017;101:229–245. doi: 10.1016/j.mcna.2016.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Life events, coping, and antihypertensive medication adherence among older adults: the cohort study of medication adherence among older adults. Holt EW, Muntner P, Joyce C, Morisky DE, Webber LS, Krousel-Wood M. Am J Epidemiol. 2012;176:0–71. doi: 10.1093/aje/kws233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Adherence to antihypertensive medication in Russia: a scoping review of studies on levels, determinants and intervention strategies published between 2000 and 2017. Bochkareva EV, Butina EK, Kim IV, Kontsevaya AV, Drapkina OM, Leon D, McKee M. Arch Public Health. 2019;77:43. doi: 10.1186/s13690-019-0366-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Application of the health belief model to improve the understanding of antihypertensive medication adherence among Chinese patients. Yue Z, Li C, Weilin Q, Bin W. Patient Educ Couns. 2015;98:669–673. doi: 10.1016/j.pec.2015.02.007. [DOI] [PubMed] [Google Scholar]
- 77.Self-efficacy mediates the relationship between depressive symptoms and medication adherence among hypertensive African Americans. Schoenthaler A, Ogedegbe G, Allegrante JP. Health Educ Behav. 2009;36:127–137. doi: 10.1177/1090198107309459. [DOI] [PubMed] [Google Scholar]
- 78.The mediating role of health beliefs in the relationship between depressive symptoms and medication adherence in persons with diabetes. Chao J, Nau DP, Aikens JE, Taylor SD. Res Social Adm Pharm. 2005;1:508–525. doi: 10.1016/j.sapharm.2005.09.002. [DOI] [PubMed] [Google Scholar]
- 79.Adherence to antihypertensive treatment in elderly people with cognitive impairment: systematic review (Article in Portuguese) Alencar Luz AL, Griep RH, Landim MB, Alencar DD, Macedo JB. Revista Cogitare Enferm. 2021;4:2–4. [Google Scholar]
- 80.Integration of depression and hypertension treatment: a pilot, randomized controlled trial. Bogner HR, de Vries HF. Ann Fam Med. 2008;6:295–301. doi: 10.1370/afm.843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Conceptual model of medication adherence in older adults with high blood pressure-an integrative review of the literature. Oori MJ, Mohammadi F, Norouzi K, Fallahi-Khoshknab M, Ebadi A. Curr Hypertens Rev. 2019;15:85–92. doi: 10.2174/1573402114666181022152313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Physician follow-up and provider continuity are associated with long-term medication adherence: a study of the dynamics of statin use. Brookhart MA, Patrick AR, Schneeweiss S, et al. Arch Intern Med. 2007;167:847–852. doi: 10.1001/archinte.167.8.847. [DOI] [PubMed] [Google Scholar]
- 83.Disentangling rheumatoid arthritis patients' implicit and explicit attitudes toward methotrexate. Linn AJ, Vandeberg L, Wennekers AM, Vervloet M, van Dijk L, van den Bemt BJ. Front Pharmacol. 2016;7:233. doi: 10.3389/fphar.2016.00233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Implicit versus explicit attitudes toward psychiatric medication: implications for insight and treatment adherence. Rüsch N, Todd AR, Bodenhausen GV, Weiden PJ, Corrigan PW. Schizophr Res. 2009;112:119–122. doi: 10.1016/j.schres.2009.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.US trends in prevalence, awareness, treatment, and control of hypertension, 1988-2008. Egan BM, Zhao Y, Axon RN. JAMA. 2010;303:2043–2050. doi: 10.1001/jama.2010.650. [DOI] [PubMed] [Google Scholar]
- 86.Adherence to medication. Osterberg L, Blaschke T. N Engl J Med. 2005;353:487–497. doi: 10.1056/NEJMra050100. [DOI] [PubMed] [Google Scholar]
- 87.Meta-analysis: impact of drug class on adherence to antihypertensives. Kronish IM, Woodward M, Sergie Z, Ogedegbe G, Falzon L, Mann DM. Circulation. 2011;123:1611–1621. doi: 10.1161/CIRCULATIONAHA.110.983874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Medication adherence and persistence according to different antihypertensive drug classes: a retrospective cohort study of 255,500 patients. Schulz M, Krueger K, Schuessel K, Friedland K, Laufs U, Mueller WE, Ude M. Int J Cardiol. 2016;220:668–676. doi: 10.1016/j.ijcard.2016.06.263. [DOI] [PubMed] [Google Scholar]
- 89.Exploring factors associated with patients' adherence to antihypertensive drugs among people with primary hypertension in the United Kingdom. Kurdi AI, Chen LC, Elliott RA. J Hypertens. 2017;35:1881–1890. doi: 10.1097/HJH.0000000000001382. [DOI] [PubMed] [Google Scholar]
- 90.A literature review to explore the link between treatment satisfaction and adherence, compliance, and persistence. Barbosa CD, Balp MM, Kulich K, Germain N, Rofail D. Patient Prefer Adherence. 2012;6:39–48. doi: 10.2147/PPA.S24752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.The association between social support and medication adherence in patients with hypertension: a systematic review. Shahin W, Kennedy GA, Stupans I. Pharm Pract (Granada) 2021;19:2300. doi: 10.18549/PharmPract.2021.2.2300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Generic substitution: additional challenge for adherence in hypertensive patients? Håkonsen H, Eilertsen M, Borge H, Toverud EL. Curr Med Res Opin. 2009;25:2515–2521. doi: 10.1185/03007990903192223. [DOI] [PubMed] [Google Scholar]
- 93.Why do the same drugs look different? Pills, trade dress, and public health. Greene JA, Kesselheim AS. https://njphasite-dev.s3.amazonaws.com/ckeditor_assets/pictures/181/original_nejmhle1101722.pdf. N Engl J Med. 2021;365:54–56. doi: 10.1056/NEJMhle1101722. [DOI] [PubMed] [Google Scholar]
- 94.Look alike/sound alike drugs: a literature review on causes and solutions. Ciociano N, Bagnasco L. Int J Clin Pharm. 2014;36:233–242. doi: 10.1007/s11096-013-9885-6. [DOI] [PubMed] [Google Scholar]
- 95.Predictors of decline in medication adherence: results from the cohort study of medication adherence among older adults. Krousel-Wood M, Joyce C, Holt E, et al. Hypertension. 2011;58:804–810. doi: 10.1161/HYPERTENSIONAHA.111.176859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Medication adherence: a key factor in achieving blood pressure control and good clinical outcomes in hypertensive patients. Krousel-Wood M, Thomas S, Muntner P, Morisky D. Curr Opin Cardiol. 2004;19:357–362. doi: 10.1097/01.hco.0000126978.03828.9e. [DOI] [PubMed] [Google Scholar]
- 97.How do we better translate adherence research into improvements in patient care? La Caze A, Gujral G, Cottrell WN. Int J Clin Pharm. 2014;36:10–14. doi: 10.1007/s11096-013-9869-6. [DOI] [PubMed] [Google Scholar]