Abstract
Pancreatic cancer is a major cause of cancer-related death, but despondently, the outlook and prognosis for this resistant type of tumor have remained grim for a long time. Currently, it is extremely challenging to prevent or detect it early enough for effective treatment because patients rarely exhibit symptoms and there are no reliable indicators for detection. Most patients have advanced or spreading cancer that is difficult to treat, and treatments like chemotherapy and radiotherapy can only slightly prolong their life by a few months. Immunotherapy has revolutionized the treatment of pancreatic cancer, yet its effectiveness is limited by the tumor's immunosuppressive and hard-to-reach microenvironment. First, this article explains the immunosuppressive microenvironment of pancreatic cancer and highlights a wide range of immunotherapy options, including therapies involving oncolytic viruses, modified T cells (T-cell receptor [TCR]-engineered and chimeric antigen receptor [CAR] T-cell therapy), CAR natural killer cell therapy, cytokine-induced killer cells, immune checkpoint inhibitors, immunomodulators, cancer vaccines, and strategies targeting myeloid cells in the context of contemporary knowledge and future trends. Lastly, it discusses the main challenges ahead of pancreatic cancer immunotherapy.
Keywords: Pancreatic cancer immunotherapy, Pancreatic ductal adenocarcinoma, Adoptive cell therapy, CAR NK cell therapy, CAR T-cell therapy, Immune checkpoint blockade, Immune checkpoint inhibitor, Oncolytic virus therapy, Cancer vaccine
Introduction
Pancreatic cancer comprises mostly pancreatic ductal adenocarcinoma (PDAC), a persistent and recalcitrant disease [1], and is responsible for an estimated 50,550 deaths in the United States of America in 2023 [2]. Diagnosis in the early stages of metastasis or late-stage is common since symptoms are often vague. The current approach for treating PDAC is standard cytotoxic chemotherapy, but it only extends overall survival (OS) by a few months [3–5].
PDAC carcinogenesis like all the solid tumors is mediated by the gradual build-up of driver mutations, such as the oncogene KRAS (G12D mutation) [6–9] and the tumor suppressor gene TP53 [10, 11]. These molecular modifications are accompanied by corresponding histological alterations during different stages of PDAC development [12]. The morphological progression initiates with the formation of precursor lesions known as pancreatic intraepithelial neoplasia (PanIN) [13], which then advance to invasive adenocarcinoma. Changes in the surrounding tissue stroma occur as cancer continues to advance. The non-transformed tissue stroma, composed of components such as immunological, vascular, and connective tissue, plays a vital role in maintaining homeostasis in response to damage. However, cancer exploits these physiological responses to create a favorable tumor microenvironment (TME) for its efficient growth [12, 14]. Indeed, cancer resembles "persistent wounds", and alterations in the stroma are the outcome of "abnormal wound healing" [15].
Immunotherapeutic strategies possess a significant capability in inducing strong immune responses against tumors. Immunomodulators, immune checkpoint blockade (ICB), and adoptive cell transfer therapy could potentially offer hopeful strategies [16–18]. Remarkable outcomes have been achieved from 2010 to the present through clinical research that utilizes various immunotherapeutic approaches to treat patients with different types of cancer [19–22]. The immune responses specifically targeting cancer cells, triggered by immunotherapy, differ from those stimulated by tumor-directed therapies. Furthermore, these responses can endure for a prolonged period even after the treatment is discontinued [23, 24]. However, the application of immunotherapy yields insufficient results for the vast majority of PDACs. This is predominantly attributed to the characteristics of its TME, which is deficient in effector T cells that have previously been exposed to antigens [25].
Tumor immunotherapy has revolutionized the treatment of various solid tumors. Nevertheless, current immunotherapies have had limited success in improving survival for patients with PDAC [26, 27]. The immunological resistance of PDAC to immunotherapies can be attributed to its low mutational burden and the hostile TME characterized by fibrosis, hypoxia, and immunosuppression [28–30]. However, a meta-analysis suggested that targeted immunotherapy is more effective than standard treatments in increasing survival and enhancing immune responses in pancreatic cancer patients [31]. Moreover, combining chemotherapy and surgery with other immunotherapies may synergistically improve outcomes. Various cytotoxic drugs and adjuvant therapies have been shown to sensitize the TME to immunotherapy by inducing immunogenic cell death, modifying evasive immune processes, and reducing immune suppression [32, 33].
Immunotherapy is presently emerging as a focal point in the treatment of pancreatic cancer. This persistent tumor primarily escapes immune detection through various means, including the secretion of immunosuppressive factors like transforming growth factor-beta (TGF-β), the creation of an immunosuppressive environment lacking T lymphocytes, and the expression of immune checkpoints such as programmed death-ligand 1 (PD-L1) and PD-L2 [4, 34]. Furthermore, research is being conducted on ICB to activate T-cell function in pancreatic cancer [35–37]. The pancreatic cancer microenvironment is characterized by extensive desmoplasia, a scarcity of effector T lymphocytes, and an immunophenotype dominated by T helper 2 (TH2) cells, all of which facilitate the evasion of cancer cells from immune surveillance [38–40]. Consequently, monoclonal antibodies (mAbs) targeting programmed cell death protein 1 (PD-1) and PD-L1 have shown limited efficacy [4]. Moreover, immunotherapies like PD-1 inhibition may benefit only a small percentage of cancer patients (3%) who have hyper-mutation and microsatellite instability [41].
This article delves headfirst into a comprehensive analysis of the immunosuppressive microenvironment in pancreatic cancer. In the context of contemporary knowledge and future trends, the article elaborates on a wide range of immunotherapies, such as oncolytic virus therapy (OVT), adoptive cell transfer therapy including T-cell receptor (TCR)-engineered T cells therapy, chimeric antigen receptor (CAR) T-cell therapy, CAR natural killer (NK) cell therapy, and cytokine-induced killer cells. Additionally, it examines immune checkpoint inhibitors (ICIs) and immunomodulators, cancer vaccines, and immunotherapeutic approaches that target myeloid cells. Lastly, the article highlights the effects of the gut microbiome in modulating response to ICIs and the emerging role of CRISPR/Cas9 gene-editing technology in pancreatic cancer immunotherapy. Finally, it discusses the main challenges ahead of pancreatic cancer immunotherapy.
Exploring the tumor microenvironment (TME) of pancreatic cancer
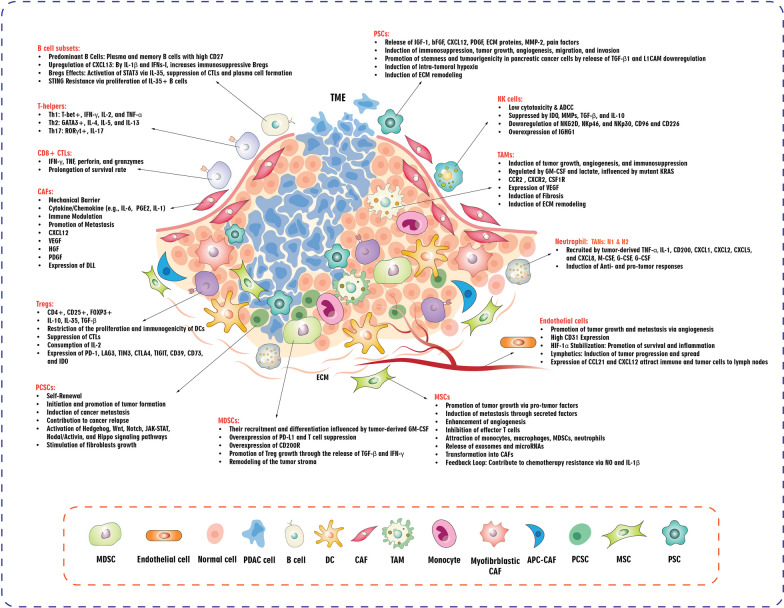
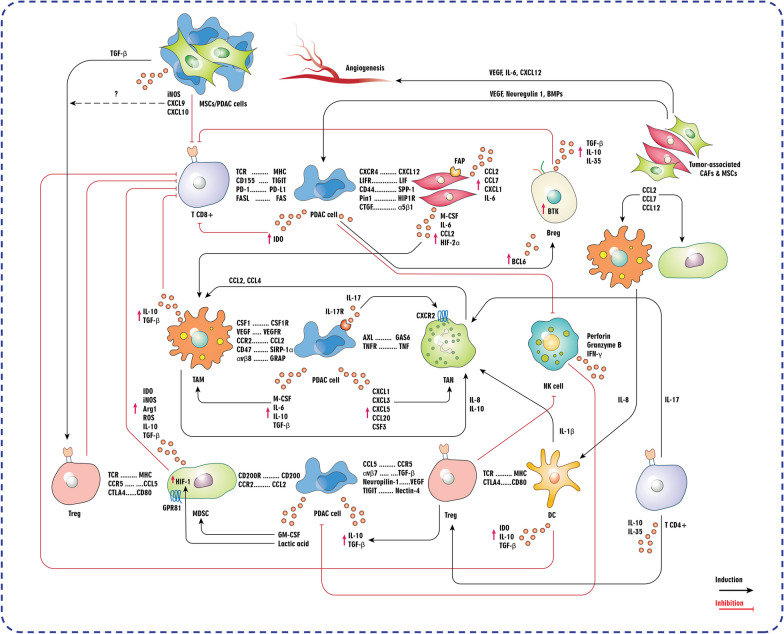
The complicated interaction between tumor cells and their adjacent microenvironment significantly impacts the development of solid tumors. Determining the outcome of cancer, whether it progresses or regresses, heavily relies on the immune environment present in tumors. This environment is made up of various cell types such as adaptive immune cells, macrophages, dendritic cells (DCs), NK cells, and other innate immune cells [42]. PDAC serves as a prime example of the various types of communication that can occur between tumors and surrounding tissue. PDAC demonstrates strong resistance to new immunotherapies due to the exclusive collaboration between different immune cells, resulting in the creation of a highly immunosuppressive setting that aids tumor advancement [12, 43–46]. The "cold" TME is a distinct feature of a pancreatic tumor wherein a considerable infiltration of myeloid cells is observed, and CD8+ T cells are usually absent, resulting in immunological characteristics [47]. Given the heterogeneous nature of pancreatic TME, components may have dual, contradicting roles (Table 1). In this section, we outline the involvement of immune cells and non-immune cells in the TME of pancreatic cancer and cross-talk between these cells (Figs. 1 and 2).
Table 1.
Dual role of key components of pancreatic tumor microenvironment
| Component | Pro-tumor effects | Anti-tumor/limiting effects |
|---|---|---|
| T lymphocytes | CD4+ T cell supported cancer progression by secreting IL-17 and IL-27 [551, 552] | Cytotoxic TILs induced tumor regression [553, 554] |
| Regulatory T lymphocytes (Tregs) | Treg suppressed immunity against early stage pancreatic intraepithelial neoplasms [555] | Treg depletion led to accelerated tumor progression [104] |
| B lymphocytes | B cells supported tumor progression/proliferation by secreting IL-35 and activating immunosuppressive TAMs [556, 557] | Insufficient data |
| Myeloid cells | CD11b+ myeloid cells are required for oncogenic Kras-driven PanIN formation [165, 166, 558] | Reinvigorating dysregulated myeloid cells in therapeutic settings (e.g., using CD40 agonist) [130, 407, 431, 559] |
| CAF | Regulating tumor metabolism for cancer cell proliferation and suppressing anti-tumor immunity [35, 560–563] | Increased matrix deposition and forming a dense and stiff matrix around early PDAC cells [242, 564, 565] |
| ECM | Supporting cancer cell proliferation and migration [566, 567] | Cancer-cell-derived fibrillar collagen and type I collagen restrains tumor growth [568, 569] |
CAF: Cancer-associated fibroblast, ECM: Extracellular matrix, PanIN: Pancreatic intraepithelial neoplasia, PDAC: Pancreatic ductal adenocarcinoma, TAMs: Tumor-associated macrophages, TILs: Tumor-infiltrating lymphocytes
Fig. 1.
Tumor microenvironment (TME) in pancreatic cancer. ADCC: Antibody-dependent cellular cytotoxicity; APC: Antigen-presenting cell; CAF: Cancer-associated fibroblast; CTL: Cytotoxic T lymphocyte; DC: Dendritic cell; DLL: Delta like canonical notch ligand; ECM: Extracellular matrix; GM-CSF: Granulocyte–macrophage colony-stimulating factor; HGF: Hepatocyte growth factor; IDO: Indoleamine 2,3-dioxygenase; IFNs-I: Type I interferons; IFN-γ: Interferon-gamma; IL-2: Interleukin 2; MDSC: Myeloid-derived suppressor cell; MMP: Matrix metalloproteinase; MQ: Macrophage; MSC: Mesenchymal stromal cell; NK: Natural killer; NO: Nitric oxide; PCSC: Pancreatic cancer stem cell; PDAC: Pancreatic ductal adenocarcinoma; PDGF: Platelet-derived growth factor; PSC: Pancreatic stellate cell; STING: Stimulator of interferon genes; TAM: Tumor-associated macrophage; TAN: Tumor-associated neutrophil; TGF-β: Transforming growth factor beta; Th1: Type 1 T helper; TNF-α: Tumor necrosis factor alpha; Treg: Regulatory T cell; VEGF: Vascular endothelial growth factor
Fig. 2.
Crosstalk between pancreatic ductal adenocarcinoma (PDAC) cells and key components of tumor microenvironment (TME). Arg1: Arginase 1; BMPs: Bone morphogenetic proteins; Breg: Regulatory B cell; BTK: Bruton's tyrosine kinase; CAFs: Cancer-associated fibroblast; CSF1: Colony stimulating factor 1; CTGF: Connective tissue growth factor; DC: Dendritic cell; FAP: Fibroblast activation protein; HIF: Hypoxia-inducible factor; IDO: Indoleamine 2,3-dioxygenase; iNOS: Inducible nitric oxide synthase; LIF: Leukemia inhibitory factor; M-CSF: Macrophage colony-stimulating factor; MDSC: Myeloid-derived suppressor cell; MHC: Major histocompatibility complex; MSCs: Mesenchymal stem/stromal cells; NK: Natural killer; Pin1: Peptidylpropyl isomerase; ROS: Reactive oxygen species; SPP-1: Osteopontin/secreted phosphoprotein 1; TAM: Tumor-associated macrophage; TAN: Tumor-associated neutrophil; TCR: T cell receptor; TGF-β: Transforming growth factor beta; TIGIT: T cell immunoreceptor with Ig and ITIM domains; TNF: Tumor necrosis factor; Treg: Regulatory T cell; VEGF: Vascular endothelial growth factor
The role of immune cells
The TME comprises various immune cells, each with distinct roles and significance. This section will elucidate the functions of these immune cells within the TME.
Role of T lymphocytes in TME
The immunological diversity among tumors in patients with PDAC is wide-ranging, characterized by varying densities of infiltrating T-cells and the composition of T-cell subpopulations [48–51]. The presence of desmoplastic elements might not influence the accumulation of T cells, thus revealing a separate spatial arrangement of T cells in PDAC [50]. This challenges the idea that the inhibitory environment shaped by fibroblasts and desmoplastic stroma suppresses the infiltration of T cells [52, 53]. In pancreatic tumors, the extravasation of T cells is constrained by the desmoplastic stroma [54], leading to immune exclusion, the induction of immunosuppression, and the inefficacy of anti-cancer therapies [55].
The presence of more CD8+ cytotoxic T lymphocytes (CTLs) encircling cancerous cells is associated with a boost in the survival rates of patients [50]. According to the study, in patients who had a better survival, tumor samples exhibited a greater percentage of CD8+ T cells, but a lesser percentage of CD4+ T cells compared to tumor samples from patients with a short survival [51]. These results highlight the complexity of the immune response in PDAC and raise questions about the role of the TME in shaping immune profiles. Further investigation is needed to fully understand these findings and their implications for future treatments. In the subsequent discourse, we explicate the pivotal contribution of T cells in the TME according to distinct T cell phenotypes.
Cytotoxic T lymphocytes (CTLs)
The principal participants in the battle against cancer cells are the CTLs that produce IFN-γ, TNF, perforin, and granzymes. These CTLs are responsible for generating durable memory cells that grant protection against cancer cells in the times to come. CTLs can recognize and kill tumor cells that express cognate tumor antigens. This specific recognition is achieved through the interaction between the TCR on CTLs and the peptide-major histocompatibility complex (MHC) on the tumor cell surface. Once the recognition occurs, CTLs induce the death of the target cell through apoptosis [56].
Previous research has demonstrated that the prognosis of individuals diagnosed with pancreatic cancer is influenced by the distribution of CD8+ TILs [57]. Increased survival in pancreatic cancer is associated with an elevation in the quantity of CD8+ T lymphocytes found within the tumor tissue [35, 50, 51]. Furthermore, in prior investigations involving surgically removed samples from pancreatic cancer cases, it has been observed that the quantity of CD8+ T cells located in the TME exhibited a positive association with the survival rate of patients [57–60]. Early mortality related to pancreatic cancer was correlated with the percentage of CD8+ T cells in the peripheral region [61].
The dysfunction and exhaustion of CD8+ CTLs within tumors is characterized by both a decline in their ability to perform their intended functions and the presence of inhibitory receptors like PD-1, T-cell immunoglobulin and mucin domain 3 (TIM-3), and lymphocyte-activation gene 3 (LAG-3), which hinder their activity. Additionally, there are changes to their gene expression patterns. According to a model studying pancreatic cancer, the signaling of the IL-18 receptor is responsible for regulating the exhaustion of tumor-targeting CD8+ T lymphocytes. This occurs by activating the IL-2/STAT5/mTOR pathway [62]. Neo-adjuvant chemotherapy exhibits a reduction in the population of CD8+ T cells with functional exhaustion in patients affected by PDAC [63].
T helper (TH) cells: TH1, TH2, and TH17
Type 1 T helper (TH1) TH1 cells, designated as a subgroup among TH cells, emerge from the activation of naïve CD4+ T cells by antigen-presenting cells (APCs) under the influence of IL-12. TH1 cells strengthen the immune response of type I immune cells by promoting the activation, proliferation, and mobilization of CTLs, M1 macrophages, and NK cells. This immune reaction aids in defending the body against intracellular infections and tumor cells. These cells express the T-box transcription factor TBX21 (T-bet) and are responsible for generating anti-cancer elements such as IFN-γ, IL-2, and TNF-α [64]. Nonetheless, in the case of PDAC patients, the impact of TH1 cells remains uncertain due to the possibility that IFN-γ could induce pro-tumorigenic consequences [65]. This is because IFN-γ has the potential to elevate the expression of PD-L1 in cancer cells, thereby hindering the effectiveness of anti-tumor immunity [66]. Murine models of PDAC demonstrate that TH1 cells play a crucial role in providing defense against tumors, while in human cases, these cells are linked with extended survival [67].
Microbial dysbiosis and the disruption of epithelial barrier function are considered inducing factors in the neoplastic transformation [68, 69]. In this regard, the contribution of the microbiome to the development of pancreatic cancer and drug resistance of PDAC has been recognized [70, 71]. Bacterial ablation is associated with immunogenic reprogramming of the TME, promoting TH1 differentiation of CD4+ T cells [70].
Type 2 T helper (TH2) GATA binding protein 3 (GATA3) is responsible for defining specialized TH2 cells, known for their proficiency in combating helminths and their involvement in allergies and asthma. These differentiated cells secrete interleukin IL-4, IL-5, and IL-13. Interestingly, the differentiation of TH1 cells is hindered by TH2 cells, and vice versa. There has been an association made between the activation of DCs and the induction of TH2 responses, and it is specifically linked to the thymic stromal lymphopoietin (TSLP), which is classified as a cytokine similar to IL-7 [64]. The prevalence of GATA3+ TH2 cell infiltration surpasses the occurrence of T-bet+ TH1 cell infiltration in pancreatic cancer. The development of the disease is associated with a higher ratio of GATA3+/T-bet+ tumor-infiltrating lymphocytes (TILs) [72, 73]. IL-4 enhances the growth of pancreatic cancer cells in humans [74]. Additionally, a worse OS rate is observed in patients suffering from PDAC characterized by a higher concentration of TH2 cytokines in their bloodstream [74]. Likewise, poor survival is linked with TH2-induced inflammation in individuals suffering from pancreatic cancer [75]. However, a study reported that the inhibition of pancreas cancer growth occurs when TH2 cells enhance the anti-tumorigenic responses of macrophages and eosinophils [76].
Given the fact that ligation of Toll-like receptor 4 (TLR4) could potentially heighten inflammation in the pancreas, it can be postulated that the activation of TLR4 may play a pivotal role in the onset of pancreatic cancer. An investigation demonstrated that DCs evoke CD4+ TH2 cells for pancreatic antigens, thereby advancing the transition from pancreatitis to cancer. Moreover, the restraint of MyD88 is accountable for inducing these outcomes [77].
Type 17 T helper (TH17) The commitment to the TH17 cell lineage begins with the action of TGF-β and IL-6, and this lineage is sustained by IL-23 while being strengthened by the autocrine production of IL-21. The crucial factors RORγt and STAT3 are necessary for the development of TH17 cells and the expression of IL-17 cytokines. TH17 cells play an important role in maintaining mucosal barriers and contributing to pathogen clearance at mucosal surfaces [64]. Elevated quantities of TH17 lymphocytes have been observed in multiple types of human malignancies, such as ovarian, pancreatic, kidney, and gastric cancer [78–80]. According to several investigations, the existence of augmented levels of TH17 cells in tumor tissues or peripheral blood is linked to the progression of cancer [81, 82]. The aggressive form of the disease was found to be associated with a significant increase in the quantity of IL-17 produced by CD4+ TILs [83]. Conversely, alternative studies propose contrasting results and indicate that TH17 cells might possess a strong anti-tumor impact, as they are present in individuals with restricted disease or those who have survived for an extensive period of time [84, 85]. Indeed, there is an ongoing debate regarding the involvement of CD4+ TH17 cells in cancer [86].
IL-17A plays a significant role in PDAC by assisting in the early stages of cancer development [87, 88], controlling the characteristics of PDAC cancer stem cells (CSCs) [89], advancing tumor growth [83, 88, 90], and causing resistance to checkpoint inhibitors through the formation of NETs [91]. Additionally, recent studies have revealed that IL-17A affects the transcriptome of cancer-associated fibroblasts (CAFs) [92]. Prominently, the induction of CAFs that are inflammatory is promoted by T cells that produce IL-17A, thus contributing to the progression of PDAC [93]. The promotion of tumorigenesis is facilitated by the upregulation of B7-H4 through IL-17/IL-17 receptor signaling in the pancreatic epithelium [94]. These findings accentuate the role of TH17 cells in favor of pancreatic cancer progression.
Contrary to the aforementioned findings, there exist findings demonstrating that TH17 cells act against tumor cells. Enhancing survival in a murine model of pancreatic cancer is observed through the promotion of TH17 cell development within the TME [95]. All in all, the role of TH17 and IL-17A in pancreatic cancer is not yet fully understood, with evidence suggesting both pro-tumorigenic and anti-tumorigenic effects. Further research is needed to elucidate the mechanisms through which IL-17A influences pancreatic cancer progression and to determine the potential therapeutic implications of targeting IL-17A in this disease.
Regulatory T cells (Tregs)
Tregs express CD4, CD25, and a chief transcription factor, called forkhead box P3 (FOXP3). The prevention of autoimmune disorders, the limitation of chronic inflammatory diseases, and the maintenance of peripheral tolerance all hinge upon Tregs. Furthermore, Tregs play a crucial role in the tumor environment, influencing cancer progression and immune responses [96]. Tregs can exert their suppressive effects through various mechanisms, whether by direct contact or independently. These mechanisms include: The production of suppressive cytokines such as TGF-β, IL-10, and IL-35. The engagement of inhibitory immune checkpoints and enzymes, such as cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), PD-1, LAG-3, TIM-3, T cell immunoreceptor with Ig and ITIM domains (TIGIT), CD39, CD73, and IDO. The induction of direct cytotoxicity through the release of perforin/granzyme. The disruption of T effector cell activity through metabolic alterations, specifically IL-2 consumption. The initiation of a tolerogenic environment by inducing tolerogenic DCs, which then facilitates T cell exhaustion [97–99].
In the peripheral blood and TME, individuals suffering from pancreatic cancer exhibit an increased frequency of Tregs [100, 101]. Tregs play a part in controlling the immune response as PDAC advances from a premalignant state to a cancerous stage. The presence of elevated Tregs is linked to a more unfavorable prognosis in PDAC [102]. Tregs possess the ability to restrict the proliferation and immunogenicity of DCs in pancreatic cancer. Additionally, the stimulation of anti-tumor immunity in pancreatic cancer is achieved by diminishing Tregs in a manner that relies on CD8+ -activated T-cells [103]. Contrariwise, the depletion of Tregs shapes the TME, leading to an acceleration of pancreatic carcinogenesis [104]. There is an expansion of pro-inflammatory and immunosuppressive Tregs which simultaneously express RORγt and FOXP3 [105]. The underlying rationale for this dual functionality can be elucidated as follows: the presence of plasticity within the pancreatic cancer microenvironment enables the Tregs to exhibit the characteristic phenotype of TH17 cells.
Role of NK cells in TME
NK cells, which are a distinct type of immune cell found in the innate immune system, are believed to play a role in monitoring and controlling tumor growth and tumor immunosurveillance [106, 107]. Both preclinical and clinical studies have demonstrated a link between decreased NK cell activity and an increased susceptibility to cancer as well as a higher chance of cancer spread and metastasis [108–110]. Researchers have identified several mediators, including indoleamine 2, 3-dioxygenase (IDO), matrix metalloproteinases (MMPs), TGF-β, and IL-10, that contribute to immune suppression in pancreatic cancer, impeding the ability of NK cells to recognize and eliminate tumor cells [28].
The survival of individuals with PDAC was found to be positively correlated with the relative frequency of NK cells in their blood. However, PDAC-associated NK cells demonstrated lower cytotoxicity compared to those of healthy participants [111]. Patients with PDAC were observed to have diminished expression of NKG2D, NKp46, and NKp30 on their peripheral NK cells, which was connected to the patient’s stage and histological grade [112]. Furthermore, the decreased expression of CD96 and CD226 (key regulators of NK cell function) on NK cells was linked to the development of cancer in PDAC patients [113]. Additionally, the evasion of NK cells in human pancreatic cancer is associated with the expression of Igγ-1 chain C region (IGHG1). Mechanistically, the presence of IGHG1 suppressed the cytotoxic activity of NK cells by inhibiting antibody-dependent cellular cytotoxicity (ADCC) [114]. Moreover, impaired localization resulting from the absence of CXCR2 and impaired tumor cytotoxicity contributed to NK cell immune evasion in patients with pancreatic cancer [115]. The function of NK cells is inhibited in the microenvironment of human PDAC by activated pancreatic stellate cells [116]. In pancreatic cancer, the orchestration of anti-tumor immune responses through CXCL8 (IL-8) by radiotherapy is reliant on NK cells. In xenografted mice, the use of high-dose radiotherapy in conjunction with adoptive NK cell transfer resulted in enhanced tumor control compared to using either treatment alone, indicating that combining NK cells with radiotherapy is a logical approach for cancer therapy [117]. Inhibiting the protein growth arrest specific 6 (Gas6), which is generated by tumor-associated macrophages (TAMs) and CAFs within the TME of PDAC, reverses the process of epithelial-mesenchymal transition (EMT) and enhances the activation of NK cells [118].
Role of DCs in TME
DCs, which are crucial for effective anti-tumor T cell responses, are scarce in the pancreatic tumor environment and are usually found at the tumor edges [119]. An increased presence of type-1 conventional DCs (cDC1s) within the entire tumor area and the tumor stroma was notably linked to improved disease-free survival (DFS). Furthermore, a rise in the number of cDC2s infiltrating the tumor’s epithelial layer was associated with enhanced DFS and OS [120]. Furthermore, patients with pancreatic cancer have been shown to have lower levels of DCs in their blood [121]. Interestingly, higher levels of circulating DCs are linked to better survival rates in these patients [121, 122]. Additionally, the surgical removal of the pancreatic tumor has been found to enhance the function of blood DCs, suggesting that the tumor itself may influence immune function [123, 124].
Cytokines originating from tumors, including TGF-β, IL-10, and IL-6, have been identified as factors that inhibit the survival and growth of DCs [125]. MDSCs generate nitric oxide (NO) and obstruct the activation of DCs [126]. In pancreatic tumors, T-cell dysfunction is common, and improving DC-mediated T-cell activation could be key for treatment. Dysfunction of cDC1s in PDACs leads to unresponsiveness to checkpoint immunotherapy. A study of 106 samples from PDAC patients showed decreased levels of circulating cDC2s, which was linked to poor prognosis. Elevated levels of IL-6 in PDAC patients were found to negatively impact DC numbers and differentiation. This suggests that inflammatory cytokines suppress DCs, impairing antitumor immunity [127].
DCs control T cells via cross-priming (cross-presentation). It is an open question in PDAC whether boosting the cross-priming capacity of DCs can enhance the T cells’ anti-tumor activity and remodel the TME. In the process of cross-priming, foreign antigens are absorbed by APCs, processed, and then displayed on MHC-I. This sequence of events ultimately triggers the activation of CD8+ T-cell responses [128]. Research has shown that the cross-priming of cDC1 is not only necessary for starting CD8+ T-cell responses as tumors progress, but it also has a pivotal role in the reactivation of tumor-specific CD8+ T cells through immunotherapy, leading to tumor shrinkage [129]. However, during the development of pancreatic cancer, the maturation of cDC1 is increasingly and universally hindered [130], impairing cross-presentation machinery. As a first proof of concept, a study tested whether cross-presentation by DCs could activate pancreatic tumor-specific CD8+ T cells in vaccinated pancreatic cancer patients. The process of in vivo cross-priming leads to the activation of mesothelin (MSLN)-specific CD8+ T cells in patients who received a vaccine for allogeneic pancreatic tumors. Also, the vaccine recruits DCs that cross-prime and generate MSLN-specific CD8+ T cells, which are capable of destroying tumor cells expressing MSLN [131]. All in all, the immunosuppressive pancreatic TME leads to the disruption of the cross-priming ability of DCs. Thus, finding solutions to reinvigorate the DCs to cross-prime tumor antigens paves the way for developing novel therapies that boost the anti-tumor immune response mediated by CD8+ T cells.
Role of macrophages in TME
Monocytes in circulation are drawn towards the TME and transform into macrophages, called TAMs, when exposed to cytokines, chemokines, and various stimuli, including high levels of concentration of hypoxia and lactic acid [132–134]. Several studies revealed that the CCL2/CCR2 and CXCL17/CXCR8 axes are involved in recruiting monocytes into the site of inflammation and tumor [135, 136]. TAMs display diverse polarization states called functional states. A wide range of TAM subpopulations has been discovered and is continuously growing. They are commonly classified as “M1” and “M2” macrophages. M1 macrophages, as typically described, generate pro-inflammatory cytokines with mainly anti-neoplastic impacts, whereas M2 macrophages produce anti-inflammatory signals that potentially accelerate tumor development [137–140]. The presence of tissue-resident macrophages in PDAC is a result of their origin from embryonic hematopoiesis, and these macrophages play a crucial role in advancing the progression of tumors [141].
A range of scientific investigations on various tumor types, including pancreatic cancer, have demonstrated a contrary association between the invasion of TAMs and the prognosis of patients [133, 142–144]. Multiple research groups have confirmed that TAMs are responsible for fostering immunosuppression, angiogenesis, and the growth of tumors in mouse models of PDAC. Their mechanism involves the release of growth factors like vascular endothelial growth factor (VEGF), cytokines, and proteases [145–149]. Within the PDAC microenvironment, the presence of granulocyte–macrophage colony-stimulating factor (GM-CSF) and lactate plays a crucial function in the polarization of TAMs, which are molecules discharged from cancer cells in a manner reliant on a mutant KRAS. A study has shown that TAM gene expression and metabolism are adversely affected by GM-CSF, disrupting their regulation through PI3K-AKT pathway signaling [150]. Collagen turnover in pancreatic cancer causes metabolic reprogramming of TAMs, leading to the promotion of fibrosis and extracellular matrix (ECM) remodeling [151].
The effectiveness of treatment in PDAC can be significantly reduced by TAMs. TAMs impact the function of cytidine deaminase, which is a critical enzyme in the metabolism of gemcitabine. This, in turn, leads to resistance to gemcitabine-based treatments in animal models of PDAC [152]. In mice models of PDAC, the suppression of C–C chemokine receptor type 2 (CCR2) promotes T-cell infiltration, enhances the efficacy of radiotherapy and chemotherapy, and diminishes metastasis by preventing the migration of monocytes to the TME [153–155]. Also, the combination of CCR2 and CXCR2 inhibitors can interrupt the accumulation of CCR2+ TAMs and CXCR2+ tumor-associated neutrophils (TANs) in the TME and enhance the effectiveness of chemotherapy in treating PDAC [147]. Moreover, the expression of CXCR2 is also reported on TAMs [156, 157]. For example, in Pten-null prostate tumors, CXCR2+ TAMs are abundant. Activating CXCR2 shifts these macrophages to an anti-inflammatory state, but blocking CXCR2 with a selective antagonist reprograms them to a pro-inflammatory state [156]. Also, in pancreatic cancer mouse models, CXCR2+CD68+ macrophages (M2 phenotype) are recruited to the TME by tumor-derived CXCL8, where they contribute to local immunosuppression, thereby reducing the effectiveness of PD-1 blockade therapy [157]. Thus, blocking the CXCR2 pathway offers a therapeutic option for enhancing cancer immunotherapy in PDAC. In a study, the tumor burden, M2 macrophage polarization, and migration are reduced, and the response to immunotherapy with anti-PD-1 is enhanced by ladarixin, a CXCR1/2 dual-inhibitor [158]. In pancreatic cancer models, the reprogramming of TAMs through colony-stimulating factor 1 (CSF1)/colony-stimulating factor 1 receptor (CSF-1R) blockade enhances the response to T-cell checkpoint immunotherapy [159].
Role of myeloid-derived suppressor cells (MDSCs) in TME
MDSCs, a diverse group of immature myeloid cells, are commonly categorized into two types: monocytic (M-MDSC) and granulocytic (polymorphonuclear [PMN]-MDSC). M-MDSCs closely resemble monocytes in terms of their phenotype and physical characteristics, while PMN-MDSCs are equivalent to neutrophils. MDSCs play a paramount role in cancer progression by promoting immunosuppression, shaping the TME, and facilitating the formation of pre-metastatic niches. Within the microenvironment of human tumors, MDSCs are abundant, and typically, PMN-MDSCs make up more than 80% of all MDSCs associated with tumors [160, 161]. Furthermore, in the circulation of the portal vein, the survival and immunoresistance of PDAC circulating tumor cells are supported by influencing the differentiation of MDSCs [162].
The levels of MDSCs in human PDAC are associated with the stage of cancer [143, 163, 164]. GM-CSF, produced by tumor cells at the early stages of cancer, plays a crucial role in the recruitment and differentiation of MDSCs, as confirmed by studies on genetically modified mice [165, 166]. CD73 causes the acceleration of pancreatic cancer pathogenesis by inducing T cell suppression through GM-CSF/MDSC [167]. Additionally, the receptor for advanced glycation end products (RAGE) facilitates the accumulation of MDSCs and promotes pancreatic carcinogenesis [168]. High expression levels of Yes-associated protein (YAP) or MDSC-associated genes indicate poor survival in PDAC patients. YAP expression levels are significantly correlated with a gene signature associated with MDSCs in primary human PDAC [169]. Following the mutation of KRAS, the transcription regulator YAP, as a downstream molecule of the oncogenic KRAS, plays a crucial role in the neoplastic development leading to PDAC [170]. The interaction between YAP/TAZ (downstream effectors of the Hippo pathway) and TEAD proteins facilitates the cancer-promoting functions of YAP. Thus, small-molecule inhibitors like GNE-7883 and IAG933, which block the interactions between YAP/TAZ and TEAD, can disrupt oncogenic YAP/TAZ signaling in RAS-altered tumors like PDAC [171, 172]. Within the PDAC microenvironment, CD200, a regulator of myeloid cell function, is upregulated. Moreover, MDSCs from PDAC patients show increased expression of the CD200 receptor. CD200 expression may regulate the development of MDSCs in the microenvironment of PDAC [173].
MDSCs control the inhibition of tumor activity in CD4+ and CD8+ T lymphocytes. T-cell activation is repressed by PD-L1, which is upregulated by MDSCs through the PD-L1/PD-1 interaction [174]. Furthermore, in an interleukin-10 (IL-10)-dependent manner, MDSCs can limit T-cell activity by promoting the growth of immune-suppressive regulatory T cells (Tregs) through the release of TGF-β and interferon-gamma (IFN-γ) [175, 176]. MDSCs play a significant role in both primary and acquired resistance to cancer immunotherapy [177]. In PDAC, reducing MDSCs enhances the accumulation of stimulated CD8+ T lymphocytes within the tumor, leading to cell death in tumor epithelial cells and remodeling of the tumor stroma [178]. Strategic MDSC targeting has been observed to effectively revitalize cytotoxic anti-tumor responses in PDAC cases. This mechanism induces the repolarization of TAMs and instigates the activation of the inflammasome machinery, thereby leading to the production of IL-18. The subsequent upregulation of IL-18 notably amplifies the functional capabilities of T-cells and NK cells within the TME [179]. In conclusion, targeting MDSCs presents a promising approach to the treatment of PDAC, and it has shown positive effects in revitalizing cytotoxic anti-tumor responses and enhancing the functional capabilities of T cells and NK cells. Therefore, further research into MDSC targeting could potentially lead to more effective therapeutic strategies for PDAC.
Role of neutrophils in TME
Neutrophils act as the first line of protection in the body against infection and respond to a broad range of pro-inflammatory signals and alarmins, such as cancer cells. These cells possess adaptability or plasticity, allowing them to adjust their actions when faced with different inflammatory triggers [180]. Because of the inflammatory state of the TME in PDAC, tumor cells secrete pro-inflammatory substances like tumor necrosis factor-alpha (TNF-α) and IL-12, causing the recruitment of neutrophils to the location of the tumor [181]. Factors secreted by tumor cells can attract neutrophils. Neutrophils can be drawn in by IL-1, CD200, CXCR2 ligands (like CXCL1 [in human and mouse], CXCL2 [in human and mouse], CXCL5 [in human], and CXCL8 [in human]) [182], GM-CSF (in human), granulocyte colony-stimulating factor (G-CSF; in human and mouse), and various other substances. These factors are released by tumor cells to attract neutrophils [182–184]. There exists a notable correlation between shortened survival and worse prognosis in patients with PDAC and increased quantities of neutrophils infiltrating the TME [60, 185].
The roles of neutrophils in the TME vary depending on their polarization states, either promoting or suppressing cancer growth. TME attracts TANs through the action of cytokines and chemokines. TANs can be categorized based on their activation and cytokine profile, which determines their impact on the growth of tumor cells. N1 TANs exhibit a beneficial effect on tumor suppression either through direct cytotoxicity or indirect means. N2 TANs, on the other hand, promote immunosuppression, tumor expansion, angiogenesis, and metastasis by causing DNA instability and releasing cytokines and chemokines [186]. Recently, a new type of TANs called T3 neutrophils has been discovered. These T3 neutrophils stimulate angiogenesis, thus improving the ability of pancreatic tumors to survive in low-oxygen and nutrient-deficient environments [187]. Identifying the plasticity of N1/N2 neutrophils has been deemed a critical prognostic marker, potentially demonstrating TME and immune evasion in PDAC patients [188]. Neutrophils with anti-tumor properties can directly eliminate tumor cells through the production of reactive oxygen and nitrogen species. Additionally, they have the ability to activate T cells and attract pro-inflammatory M1 macrophages. Conversely, neutrophils that aid tumor development secrete MMP-9, facilitating the growth of new blood vessels and the dissemination of tumor cells. These neutrophils can also hinder the function of NK cells while recruiting anti-inflammatory M2 macrophages and Tregs. Further, suppressor neutrophils, referred to as PMN-MDSCs, as well as other pro-tumoral neutrophils, impede the activity of CD8+ T cells [24, 180]. The growth of pancreatic cancer is reduced and the effectiveness of ICB treatment with anti-PD-1 is enhanced through the inhibition of TANs by lorlatinib [189]. The metastasis of pancreatic cancer is facilitated by neutrophils that infiltrate as a result of chemotherapy. This is achieved through the activation of the Gas6/AXL signaling pathway [184].
Neutrophils differentiate themselves from other immune cells by producing neutrophil extracellular traps (NETs), consisting of DNA fibers and proteolytic enzymes released to counteract infections [190]. Nevertheless, recent studies have suggested that NETs might contribute to cancer metastasis. By examining a PDAC mouse model, researchers investigated the effects of DNase I, a NET inhibitor, and observed a reduction in liver metastasis [191]. In the PDAC milieu, neutrophil recruitment and NETosis are triggered by IL-17 [91]. The activation of the IL-1β/epidermal growth factor receptor (EGFR)/extracellular-signal-regulated kinase (ERK) pathway is prompted by NETs, resulting in the promotion of migration, invasion, and EMT of pancreatic cancer cells [192].
Role of B lymphocytes in TME
A study found that a high density of B cells within tertiary lymphoid tissues of human PDAC is associated with longer survival rates, germinal center immune signature, and CD8+ TILs infiltration [193]. In the TME of PDAC, the predominant B cells are plasma cells and memory B cells, which exhibit high levels of CD27 expression. However, numerous studies have discovered that the upregulation of CXCL13, triggered by IL-1β and type I interferons (IFNs-I), leads to an increased influx of regulatory B cells (Bregs) that perform immunosuppressive activities [194–196]. Bregs can activate STAT3 signaling within themselves and CD8+ T cells via IL-35. This activation leads to two distinct effects: firstly, the transcriptional regulator BCL-6 experiences an increase in naive B cells, which interferes with the transformation of B cells into plasma cells; secondly, the operational capacity of CTLs is suppressed [197, 198]. Recent research discovered that the resistance to the stimulator of interferon genes (STING) agonists in PDAC is attributed to the induction of IL-35+ B cell proliferation. The systemic application of anti-IL-35 and STING agonist (cyclic guanosine monophosphate-adenosine monophosphate [cGAMP]) can work together to suppress the amplification of Bregs and boost the effectiveness of NK cells [199]. A clinical trial showed that ibrutinib (a Bruton tyrosine kinase inhibitor) plus nab-paclitaxel/gemcitabine did not improve OS or progression-free survival (PFS) for patients with PDAC [200].
The role of non-immune cells
Within the microenvironment of pancreatic tumors, there exists a variety of non-immune cells. This section delves into a discussion about the most significant among them.
Pancreatic cancer stem cells (PCSCs)
PCSCs are a subset of cancer cells that exhibit stem cell-like characteristics, including the ability to self-renew and initiate tumorigenesis. They are believed to contribute to the initiation, metastasis, and recurrence of PDAC, and are also responsible for resistance to chemotherapy and radiation. PCSCs express several markers, including CD133, CD24, CD44, microtubule-associated doublecortin-like kinase 1 (DCLK1), CXCR4, epithelial-specific antigen (ESA), OCT4, nestin, and ABCB1 [201, 202]. In PDAC, stem cells display unusual activation of multiple signaling pathways that are generally active in embryonic growth. This irregular signaling via mechanisms such as Hedgehog, Wnt, Notch, JAK-STAT, Nodal/Activin, and Hippo enables PCSCs to preserve their self-renewal ability, develop resistance to chemotherapy and radiation, enhance their capacity to induce tumors, and spread to other parts of the body [202]. A specific subpopulation of CSCs, identified by CD133 and CXCR4 markers, is crucial for tumor metastasis in human pancreatic cancer. Depleting this subpopulation can significantly reduce metastasis. Modulating the CXCL12/CXCR4 axis could be a potential strategy to inhibit CSC metastasis [203]. The E2F1/4-pRb/RBL2 axis, which undergoes deregulation following a KRAS mutation, is instrumental in maintaining equilibrium among signaling pathways controlling stem cell-like characteristics of CSCs. This axis governs the production of Wnt ligands, thereby managing the self-renewal, resistance to chemotherapy, and invasive nature of PCSCs, along with the proliferation of fibroblasts [204]. This axis might be a therapeutic target for eradicating PCSCs.
Mesenchymal stem/stromal cells (MSCs)
MSCs are a heterogeneous group of progenitor cells that transform into tumor-associated mesenchymal stem cells (TA-MSCs) within TME, influencing tumor growth, metastasis, angiogenesis, and treatment responses through the secretion of various factors, and their immunosuppressive properties could be targeted to enhance anti-tumor immunity [205]. First of all, TA-MSCs can release CCL2, CCl7, and CCL12 to recruit monocytes, macrophages, MDSCs, and neutrophils [206]. They also produce CXCL9 [207], CXCL10 [207], CXCL11 [207], inducible nitric oxide synthase (iNOS) [207], and IDO [208], resulting in the inhibition of effector T cells. Mechanistically, TA-MSCs produce large amounts of pro-metastatic and pro-tumor factors such as neuregulin-1 [209], VEGF [210], bone morphogenetic proteins [211], TGF-β [212], CCL5 [213], CXCL10 [214], CXCL12 [215], CD81-positive exosomes [216], and MMPs [217]. Also, they can adjust tumor cell’s response to chemotherapy by generating factors like polyunsaturated fatty acids [218], PDGF [219], hepatocyte growth factor [220], NO [221], and exosomes carrying these factors and microRNAs [222, 223]. In patients with pancreatic cancer, the presence of MSCs in the peripheral blood is notable as they are thought to migrate to the tumor mass [224]. Evidence suggests that a significant portion of CAFs may originate from MSCs, which can differentiate and express CAF markers, such as vimentin and FAP when exposed to conditioned media from various human cancer cell cultures like pancreatic cancer [225]. In a pancreatic cancer tumor model, VEGF is secreted by bone marrow mesenchymal stem cells (BM-MSCs) that are co-injected with tumor cells, which aids in the promotion of tumor angiogenesis [210]. TA-MSCs can produce NO, which induces resistance to etoposide in pancreatic tumor cells and forms a positive feedback loop with IL-1β, contributing to chemotherapy resistance [221].
Cancer-associated fibroblasts (CAFs)
CAFs are a hodgepodge and heterogeneous group of stromal cells that produce ECM proteins. These cells, typically spindle-shaped, express activated fibroblast markers like fibroblast activation protein (FAP) and α-smooth muscle actin. They are associated with various tumor-promoting activities, including tumorigenesis, angiogenesis, immunosuppression, and metastasis [226, 227]. CAFs in PDAC can originate from diverse cells like adipocytes, pericytes, bone marrow-derived macrophages, endothelial/epithelial cells, mesothelial cells, MSCs, resident tissue fibroblasts, and pancreatic stellate cells (PSCs) [228]. In PDAC stroma, CAFs interact with cancer cells through both direct cell-to-cell and paracrine mechanisms. CAFs are heterogeneous and include three subtypes: myofibroblastic, inflammatory, and antigen-presenting. Myofibroblastic CAFs are induced by cancer cells through TGF-β, and they create a mechanical barrier that can both promote and inhibit tumor growth. Inflammatory CAFs, located away from the tumor cells, are reprogrammed by IL-1 to generate cytokines and chemokines (like IL-6), which further stimulate cancer growth. Lastly, antigen-presenting CAFs express MHC class II molecules and modulate the immune cells in the stroma. These diverse interactions contribute to the complex dynamics of the PDAC stroma [12]. In the pancreatic environment, CAFs play a significant role in creating an immune-suppressive milieu by releasing substances like prostaglandin E2 (PGE2), IL-1, IL-6, CXCL2, CXCL12, and CXCL8 [35, 229–231]. Not only do these fibroblasts attract and control immune-suppressing cells, but they also hinder the anti-cancer activities of CD8+ T cells by increasing the expression of inhibitory immune checkpoints [230]. Recently, a study identified three distinct metastasis-associated fibroblasts (MAFs) populations, with the generation of pro-metastatic myofibroblastic-MAFs (myMAFs) being critically dependent on macrophages. These myMAFs are induced through a STAT3-dependent mechanism and in turn promote an immunosuppressive macrophage phenotype, inhibiting cytotoxic T-cell functions. Blocking STAT3 pharmacologically or depleting it in myMAFs restores an anti-tumor immune response and reduces metastasis, providing potential targets to inhibit PDAC liver metastasis [232].
Pancreatic stellate cells (PSCs)
Approximately 7% of pancreatic cells are made up of PSCs, which are located in both the exocrine and endocrine regions of the pancreatic tissue. The interaction between PSCs and pancreatic cancer cells promotes tumor progression. Mechanistically, PSCs release several growth factors/mediators (such as insulin-like growth factor 1 [IGF-1], basal fibroblast growth factor [bFGF], platelet-derived growth factor [PDGF], stromal cell-derived factor 1 [SDF-1], and ECM proteins) and MMPs, which provoke the proliferation, migration, and invasion of pancreatic tumor cells. In response, pancreatic cancer cells produce TGF-β1, PDGF, and VEGF, which in turn stimulate PSCs to increase the migration and proliferation of CAFs and the production of ECM [233, 234]. Indeed, a key characteristic of PDAC is a desmoplastic reaction, seen in both primary and metastatic tumors. This reaction is caused by the activation of PSCs, by cancer cells, leading to fibrosis around the tumor [235, 236]. This fibrosis (also known as desmoplasia) forms a mechanical barrier around the tumor cells, hindering proper vascularization, limiting the effectiveness of chemotherapy, and resulting in poor immune cell infiltration [237]. PSCs serve as a significant source of MMP-2 and they hasten the advancement of the tumor in a murine xenograft model [238]. Also, TGF-β1 secreted by PSCs promotes stemness and tumourigenicity in pancreatic cancer cells through L1CAM downregulation [239]. Overall, PSCs are linked to ECM production and remodeling, intra-tumoral hypoxia, resistance/barrier to chemotherapy, proliferation, invasion, migration, reduced apoptosis, angiogenesis, immune suppression, and pain factors [234].
Endothelial cells
PDAC often has abnormal blood and lymphatic vessels, leading to a hostile microenvironment characterized by high acidity, hypoxia, aberrant metabolism, and immune evasion. In response, tumors stimulate angiogenesis, promoting tumor growth and metastasis [46, 240]. Studies reveal that high expression of the endothelial cell marker CD31 and genes involved in vascular stability correlate with better prognosis and improved survival in PDAC [241, 242]. This suggests that a subset of patients with highly vascular PDAC may benefit from antiangiogenic therapies [242].
Inadequate vasculature in tumors restricts nutrient, oxygen, and leukocyte delivery, leading to hypoxia in PDAC. Hypoxia-inducible factor 1α (HIF-1α) is stabilized in poorly vascularized PDAC tumors [243], activating genes crucial for metabolism, angiogenesis, cell survival, and inflammation [244]. Elevated HIF-1α levels are linked to poor prognosis in many cancers [244]. However, in PDAC, HIF-1α deletion accelerates tumor growth, facilitated by infiltrating B cells, demonstrating PDAC’s resilience and complex redundancies that support disease progression [245].
Lymphatics, in addition to blood vessels, play a crucial role in the progression of PDAC. They serve as a major pathway for leukocytes to transport tumor antigens to lymph nodes and for cancer cells to spread, often resulting in worse survival outcomes [46, 246, 247]. Chemokines play a role in lymphangiogenesis and cell migration, with lymphatic endothelial cells secreting CCL21 to attract DCs and tumor cells expressing CCR7 potentially using this mechanism for dissemination [248]. Likewise, CXCL12 produced in lymph nodes may attract cancer cells or leukocytes expressing CXCR4 [249].
Immunotherapeutic approaches in pancreatic cancer treatment
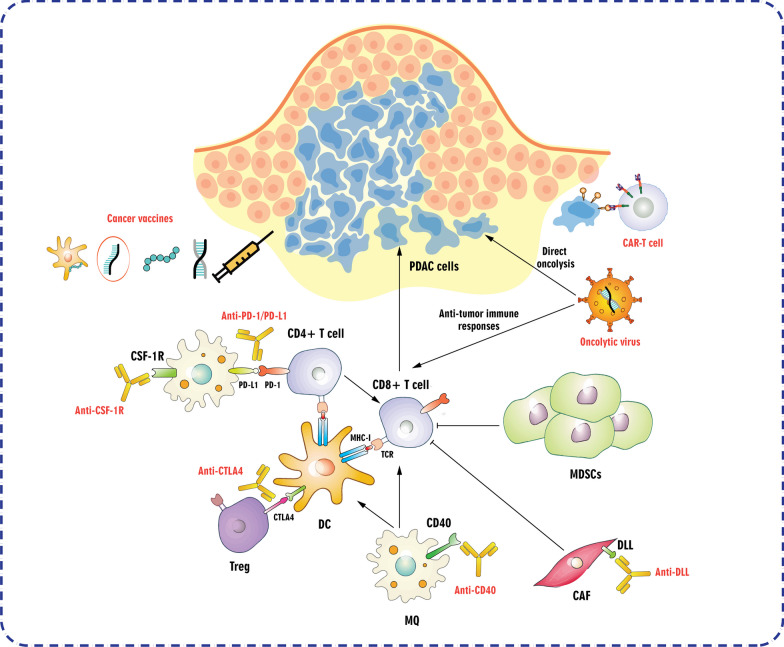
Pancreatic cancer is classified as non-immunogenic and immunologically cold since it does not effectively react to commonly employed ICIs such as anti-PD-1 and anti-CTLA-4. This resistance is partly caused by the immunosuppressive circumstances within the TME. In other words, although ICB has achieved explosive success, PDAC has shown limited response to ICB treatment alone. Research on using ICB alone or in combination with anti-PD-1 and anti-CTLA-4 antibodies has yielded overall response rates (ORRs) of 0% and 3%, respectively [250]. In this part, we will delineate immunotherapeutic strategies such as OVT, adoptive cell transfer therapy, ICB, cancer vaccine, and immunotherapies targeting myeloid cells (Fig. 3).
Fig. 3.
Immunotherapeutic strategies in pancreatic cancer treatment. The immune response to pancreatic ductal adenocarcinoma (PDAC) is guided by antigen-presenting machinery involving dendritic cells (DCs), inflammatory macrophages, and CD4+ helper T cells, leading to the activation of CD8+ cytotoxic T cells to eliminate the cancer. However, regulatory T cells (Tregs) and suppressor cells can inhibit this response, creating an immunosuppressive tumor microenvironment. Various strategies have been suggested to counteract these inhibitory pathways. CAF: Cancer-associated fibroblast; CAR: Chimeric antigen receptor; CSF-1R: Colony-stimulating factor 1 receptor; CTLA4: Cytotoxic T-lymphocyte associated protein 4; DLL: Delta-like ligand; MDSC: Myeloid-derived suppressor cell; MHC: Major histocompatibility complex; MQ: Macrophage; PD-1: Programmed cell death protein 1; PD-L1: Programmed death-ligand 1; TCR: T cell receptor
Oncolytic virus therapy (OVT)
OVT represents an innovative form of immunotherapy where an oncolytic virus, upon infiltrating and lysing a cancerous cell, initiates an immune reaction within the patient by discharging tumor antigens into the circulatory system [251]. Oncolytic viruses possess desirable qualities and specificity that make them an attractive strategy for treatment. Research is currently underway, exploring and utilizing diverse oncolytic DNA and RNA viruses for the treatment of different cancer forms. Their ability to invade cancer cells is made possible by the genetic composition of these viruses [252].
Talimogene laherparepvec (T-VEC or OncoVEXGM-CSF), a Herpes simplex virus (HSV), has become the inaugural oncolytic virus approved by the US Food and Drug Administration (FDA) for the treatment of melanoma. The T-VEC virus harbors the genetic integration of the GM-CSF gene. T-VEC exhibited remarkable lytic properties when tested against various tumor cell lines, encompassing pancreatic cancer cells [253, 254]. Furthermore, both NV1020 (r7020) and G207, two distinct herpes simplex oncolytic viruses, effectively invade and annihilate human pancreatic cancer cells in vitro and in vivo [255]. HF10 is a virus that has originated from HSV-1 and has experienced an unexpected mutation. This particular virus has the ability to substantially combat tumors without causing any damage to healthy tissue. The treatment of locally advanced pancreatic cancer involves the secure administration of HF10 through direct injection, alongside erlotinib and gemcitabine [256]. The anti-tumor response and apoptosis are enhanced in pancreatic cancer when an H-1 oncolytic parvovirus is combined with a hypoxia-inducible factor (HIF)-1α inhibitor, resulting in increased effectiveness [257].
VCN-01, a type of oncolytic adenovirus, has been specifically designed to reproduce within cancer cells that possess a faulty RB1 pathway. Moreover, it has the ability to generate hyaluronidase, which serves to expedite the spread of the virus within the tumor. Additionally, it facilitates the migration of both chemotherapy medications and immune cells into the tumor. VCN-01 exhibited augmented anti-cancer properties when administered in conjunction with chemotherapy to animals with PDAC. Remarkably, the hyaluronidase produced by VCN-01 effectively obliterated the tumor stroma, thereby bolstering the transport of various therapeutic drugs such as chemotherapy and therapeutic antibodies [258]. A clinical experiment exhibited that it is feasible to administer VCN-01 through an intravenous route for the treatment of patients suffering from PDAC and this administration method is associated with adverse events (AEs) that can be predicted and controlled. Intravenous VCN-01 has exhibited a positive tolerability profile [259]. These results establish a helpful bedrock for the future use of OVT in pancreatic cancer immunotherapy. Furthermore, several clinical trials are underway to evaluate the efficacy of various oncolytic virus-oriented therapies in pancreatic cancer. A phase I/II trial demonstrated that the combination of intratumoral injections of LOAd703, an oncolytic adenovirus with transgenes encoding trimerized, membrane-bound (TMZ)-CD40L and 4-1BB ligand, with standard nab-paclitaxel/gemcitabine chemotherapy was both safe and feasible for patients with unresectable or metastatic PDAC. The treatment met the target response rate at the highest dose level, with an ORR of 44% and a disease control rate of 94% (NCT02705196) [260]. Moreover, a study found that the combination of pelareorep and pembrolizumab showed modest efficacy in unselected patients, with a clinical benefit rate of 42% among the 12 patients. Notably, the treatment led to significant immunological changes, including a decrease in VDAC1 expression in peripheral CD8+ T cells and on-treatment peripheral CD4+ Treg levels in patients who responded to the treatment (NCT03723915) [261]. The efficacy of talimogene laherparepvec (T-VEC), administered endoscopically, will be assessed in a clinical trial for the treatment of locally advanced or metastatic pancreatic cancer that is refractory to at least one chemotherapy regimen (NCT03086642).
A study demonstrates promising findings for a new technology called ONCOTECH, which combines oncolytic adenoviruses (OAs) with T cells to enhance the delivery of viruses to tumors. The engineered OAs target the immune checkpoint protein PD-L1. In mouse models of PDAC, ONCOTECH displayed a notable increase in OAs within tumor cells, resulting in a significant decrease in PD-L1 expression and better survival rates. In summary, ONCOTECH has the potential to be a successful approach in combining virotherapy and cell therapy for cancer treatment [262].
Adoptive cell transfer therapy
Adoptive cellular therapy, which is a type of immunotherapy, holds promise for cancer patients. By utilizing the patient's immune cells, such as T cells, this technique endeavors to combat the disease. These immune cells are frequently obtained, replicated, and altered to augment their efficiency in directing their focus on cancer. The progress made by the FDA in granting approval to CAR T-cell therapy for certain blood cancers has greatly propelled this area of medical research. Modified T cells possess the ability to discern tumor cells through their unique molecular features [263]. In the subsequent discussion, we shall elucidate and analyze these various immunotherapeutic approaches.
Tumor-infiltrating lymphocyte (TIL) therapy
TILs, which are mononuclear cells naturally infiltrating the TME, can also be known as immune cells present at the tumor site. TIL therapy remains a hopeful treatment approach whereby the patient's TILs are utilized following the surgical extraction of the cancerous growth, followed by the cultivation of these cells outside the body and subsequent reinfusion back into the patient [264–266]. Successful techniques for increasing the production and reactivity of TILs encompass inhibiting the PD-1 receptor, stimulating the CD137 receptor (4-1BB), and augmenting CD8+ T cell levels [267]. According to a study, it was found that functional expanded TILs from tumors in the pancreas possess the capability to identify antigens associated with pancreatic cancer [267]. Based on a meta-analysis, the long-term oncological prognosis of patients with PDAC is significantly associated with specific categories of TILs, specifically CD8+ T cells [57]. At the present moment, two ongoing clinical trials are currently in the process of recruiting participants. These trials will aim to implement TIL therapy on individuals who are affected by metastatic PDAC (NCT03935893 and NCT01174121). The former trial will assess the efficacy of the adoptive transfer of autologous TILs in combination with fludarabine and cyclophosphamide, while the latter trial will investigate the efficacy of young TILs in combination with aldesleukin (a recombinant analog of IL-2), pembrolizumab, cyclophosphamide, and fludarabine. To further explain, the young-TIL approach involves minimal in vitro culturing of TILs and does not select for tumor recognition before they are rapidly expanded and infused into the patients. This method has achieved objective response rates similar to those of used TILs screened for tumor recognition, without introducing any additional toxicities [268].
Genetically modified T cells therapy
TCR-engineered T-cell therapy
The production of TCR-engineered T cells involves modifying T cells outside the body to express TCRs that recognize tumor antigens. TCRs have the capacity to detect peptides displayed by both MHC class I and II [269]. Investigating the safety and effectiveness of autologous MSLN-specific TCR T cells in patients with stage IV pancreatic cancer is the objective of a phase I clinical trial (NCT04809766). In this trial, autologous MSLN-specific TCR-T Cells were used in combination with bendamustine, cyclophosphamide, and fludarabine. Patients received three infusions of TCR-TMSLN cells every 21 days following leukapheresis. The main focus was on safety and dose-limiting toxicities, but the study also looked at ORR, PFS, and OS. The goal is to achieve a significant ORR of 20% among the 15 participants [270].
The patient with metastatic PDAC received autologous TCR-engineered T cells as treatment. These modified T cells express two allogeneic human leukocyte antigen (HLA)-C*08:02-restricted KRAS G12D in a clonal manner. Remarkably, the patient's visceral metastasis showed regression, with an overall partial response of 72%. Furthermore, the therapeutic effect persisted for a duration of 6 months. Moreover, after six months of the T-cell transfer, the modified T cells accounted for more than 2% of all circulating T cells in the peripheral circulation [271].
CAR T-cell therapy
CAR T cells can be compared to the administration of a living drug to patients. At, the CAR T-cell therapies that are accessible are tailored according to each patient's needs. These therapies are created by gathering T cells from the patient and modifying them in the lab to generate CARs on the cell surface. The specific CARs possess the ability to detect and attach themselves to particular proteins, known as tumor antigens, located on the outer surface of cancer cells. Despite its impressive clinical outcomes in the treatment of specific subgroups of B-cell leukemia or lymphoma, CAR T-cell therapy encounters numerous impediments that impede its widespread application in the treatment of solid tumors and hematological malignancies. Impediments such as life-threatening toxicities, cytokine release syndrome (CRS), inadequate anti-tumor efficacy, antigen escape, and limited trafficking all pose obstacles to the successful implementation of CAR T-cell treatment [272, 273]. Tables 2 and 3 provide a comprehensive overview of data regarding CAR T-cell therapy in both preclinical and clinical trial settings.
Table 2.
Evidence from clinical studies supporting the use of CAR T-cell therapy for pancreatic cancer treatment
| Target | CAR’s molecular structure | Combination therapy | Clinical outcomes (= number of patients) | References |
|---|---|---|---|---|
| CD133 | Anti-CD133 ScFv + Human CD137 + CD3ζ | Nab-paclitaxel + Cyclophosphamide | PR = 2, SD = 3, PD = 2 | [307]/NCT02541370 |
| EGFR | Anti-EGFR ScFv + CD8α + CD137 + CD3ζ | Nab-paclitaxel + Cyclophosphamide | PR = 4, SD = 8, PD = 2 | [290]/NCT01869166 |
| HER2 | Anti-HER2 ScFv + CD8α + CD137 + CD3ζ | Nab-paclitaxel + Cyclophosphamide | PR = 0, SD = 0, PD = 2 | [286]/NCT01935843 |
| MSLN | Anti-MSLN ScFv + 4- 1BB + CD3ζ | N/A | SD = 2, PD = 1 | [301]/NCT01897415 |
EGFR: Epidermal growth factor receptor, HER2: Human epidermal growth factor receptor 2, MSLN: Mesothelin, N/A: Not applicable, ScFv: Single-chain variable fragment, PR: Partial response, SD: Stable disease, PD: Progressive disease
Table 3.
Clinical trials in pancreatic cancer investigating the effectiveness of CAR T-cell therapy
| Target | Agent/intervention | Combination therapy | Participants with pancreatic cancer | Phase | Outcomes* | Status | Reference/NCT Identifier |
|---|---|---|---|---|---|---|---|
| ROR2 | CCT301-59 | N/A | 18 | I | Safety, efficacy, and kinetics of CCT301-59 | Unknown | NCT03960060 |
| HER2 | CCT303-406 | N/A | 15 | I | Safety, tolerability, DLTs, and MTD | Recruiting | NCT04511871 |
| CD22 | CAR-T/CAR-TILs cells contains anti-CD22 CAR and a ScFv fragment of anti-PD-L1 | N/A | 30 | I | ORR, PFS, OS, and AEs | Recruiting | NCT04556669 |
| CD70 | Anti-hCD70 CAR transduced peripheral blood lymphocytes | Cyclophosphamide + Fludarabine + Aldesleukin | 124 | I, II | Safety and regression of CD70 expressing tumors | Recruiting | NCT02830724 |
| CEA | Anti-CEA CAR-T cells | N/A | 5 | I |
No on-target/off-tumor serious AEs above grade 3 Complete metabolic response within the liver Normalization of serum tumor markers and an abundance of CAR+ cells in tumor specimens |
Completed | [570]/NCT02850536 |
| Claudin18.2 | CT041 | N/A | 110 | I, II |
No DLTs, treatment-related deaths, and severe CRS One patient received tocilizumab 2 patients had partial response, 2 had stable disease, and 3 had progression of disease |
Active, not recruiting | [571]/NCT04404595 |
| Anti-PD-1 + Paclitaxel or Irinotecan or Apatinib | 192 | I, II | Two patients received the therapy. First patient and second patient had partial and complete response, respectively | Recruting | [312]/NCT04581473 | ||
| EpCAM | Anti-EpCAM CAR T cells | N/A | 60 | I, II | Determining toxicity profile, persistence of CAR T cells, and efficacy | Unknown | NCT03013712 |
| EpCAM/TM4SF1 | TM4SF1 and EpCAM-positive CAR T-cell therapy | N/A | 72 | N/A | Determining safety, persistence of CAR T cells, and ORR | Unknown | NCT04151186 |
| MSLN | huCART-meso cells | N/A | 18 | I | Determining AEs, ORR, PFS, and OS | Recruiting | NCT03323944 |
*In cases where the outcomes of the clinical trial have not yet been released, the goals (primary/secondary outcome measures) of the study are mentioned
AEs: Adverse events, BCMA: B-cell maturation antigen, CAR: Chimeric antigen receptor, CD: Cluster of differentiation 19, CEA: Carcinoembryonic antigen, CRS: Cytokine release syndrome, DLTs: Dose-limiting toxicities, EGFR: Epidermal growth factor receptor, EpCAM: Epithelial cell adhesion molecule, GD2: Disialoganglioside, GP3: Glypican 3, HER2: Human epidermal growth factor receptor 2, MSLN: Mesothelin, MTD: Maximum tolerated dose, MUC-1: Mucin-1, N/A: Not applicable, ORR: Overall response rate, OS: Overall survival, PSCA: Prostate stem cell antigen, PD-1: Programmed cell death protein 1, PD-L1: Programmed death-ligand 1, PFS: Progression-free survival, ROR2: Receptor tyrosine kinase-like orphan receptor 2, TM4SF1: Transmembrane 4 L Six Family Member 1
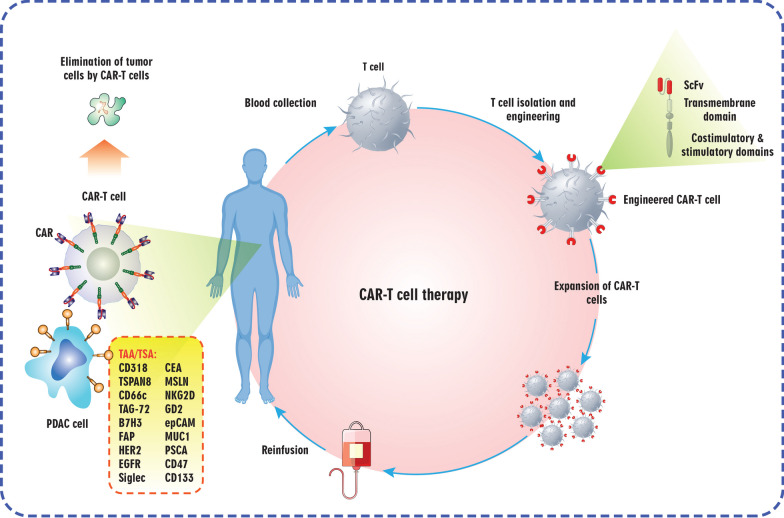
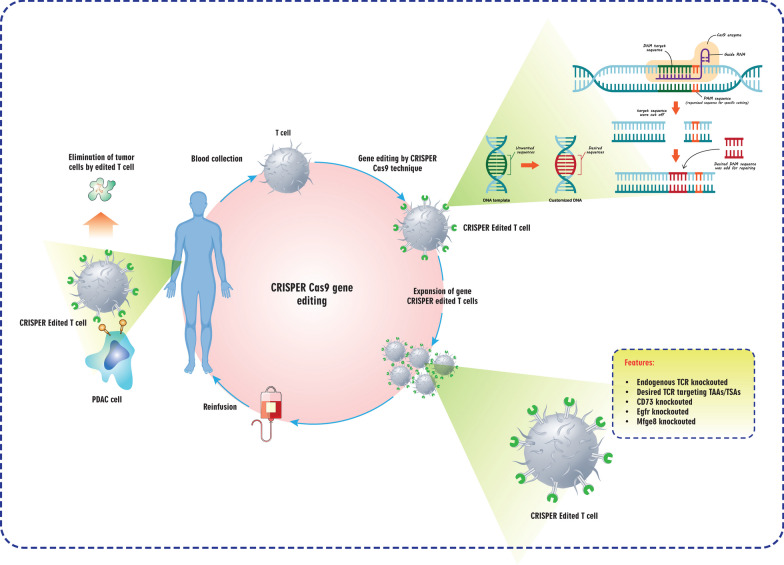
A crucial obstacle to the effective use of cellular immunotherapy for treating PDAC, specifically CAR T-cell therapy, is the lack of suitable tumor-specific antigens. In their research, Schäfer et al. pinpointed CD318, TSPAN8, and CD66c as potential target molecules for CAR T-cell-based immunotherapy in PDAC, among a pool of 371 antigens [274]. Highlighted in the subsequent text are the appropriate therapeutic targets for the CAR T-cell therapy of pancreatic cancer (Fig. 4).
Fig. 4.
An overview of chimeric antigen receptor (CAR) T cell therapy concept. CAR T cell therapy is a treatment approach, whereby T cells from an individual are modified in a laboratory setting to possess the ability to identify specific antigens found on cancer cells, leading to their elimination. (1) This process involves removing autologous T cells from the patient's blood. (2) Subsequently, the T cells are manipulated by introducing a gene encoding a specialized receptor, known as a CAR, into their genetic makeup through viral vectors. (3) This genetic alteration results in the expression of the CAR protein on the surface of the patient's T cells, thereby creating CAR T cells. These CAR T cells are then multiplied and expanded in laboratory conditions, producing millions of them. (4) Eventually, these CAR T cells are administered to the patient through intravenous infusion. (5) The CAR T cells attach themselves to the cancer cells by binding to the antigens present on their surface and proceed to eradicate the cancer cells. EGFR: Epidermal growth factor receptor; FAP: Fibroblast activation protein; MSLN: Mesothelin; PDAC: Pancreatic ductal adenocarcinoma; ScFv: Single-chain variable fragment; TAA: Tumor-associated antigen; TSA: Tumor-specific antigen
B7H3 (CD276) B7H3, a molecule found on the surface of cells, acts as an immune checkpoint and hinders the activation of T-cells and the ability of NK cells to kill. The promise of targeting B7H3 for CAR T-cell therapy arises from its high expression in numerous cancer types while being minimally expressed in healthy tissues [275]. Survival was achieved in mice following treatment with B7H3 CAR T cells, and there were no observed AEs [276]. The outcome of studies conducted in vitro revealed that these cells exhibited a potent ability to suppress the growth of cancer cells in the pancreas [276, 277].
Fibroblast activation protein (FAP) FAP is a type-II transmembrane serine protease expressed almost exclusively on CAFs. In mouse models of solid tumors, the growth of tumors can be effectively suppressed by FAP-expressing stromal cells being targeted by CAR T cells designed specifically for FAP [278, 279]. When FAP-specific CAR T cells are administered along with anti-PD-1 treatment, the combination leads to a synergistic reduction in pancreatic tumor growth and significantly elongated survival in mouse models compared to alternative treatment combinations [54].
Human epidermal growth factor receptor 2 (HER2) HER2, a glycoprotein located on the cell membrane, performs a function in promoting cell division and distinction during various stages, including embryonic and adult periods. HER2 contributes to tumor progression, growth, and spread by obstructing cell death, triggering the formation of new blood vessels, and boosting cell movement [275]. The expression of the HER2 in pancreatic cancer is controversial [280]. However, it has been detected in 20–60% of PDACs according to certain research studies [281, 282]. Also, HER2 might be a potential target in immunotherapy for a small subset of patients with pancreatic cancer, since a report explains nearly 50% of PDAC cases have a total HER2 expression of 2 + or above [283, 284].
The combination treatment of oncolytic adeno-immunotherapy and HER2-specific CAR-T cells shows promising results in eradicating metastatic PDAC. This combinational therapy enhances the migration of CAR-T cells to the tumor site, while also stimulating systemic host immune responses that improve the overall anti-tumor activity [285]. The clinical effect of anti-HER2 CAR T cells was assessed in a study involving 11 patients, two of whom had metastatic pancreatic cancer. The optimal overall outcome for both patients was disease stability, with a PFS of 5.3 and 8.3 months, respectively [286]. The potential clinical outcomes of the treatment should be proven in clinical trials with a larger sample size including patients with PDAC.
All in all, in different studies, the expression of HER2 in pancreatic cancer is controversial and varies from high-level expression [287] to low-level expression [288], making it a potential target for personalized immunotherapy of PDAC. Thus, it is reasonable that HER2 should not be ignored in such a heterogeneous disease with limited treatment options.
Epidermal growth factor receptor (EGFR) The EGFR protein, which spans across the membrane, has the capability of binding to various proteins from the EGF family that are located outside the cell. Around 90% of patients diagnosed with PDAC exhibit an identifiable amount of EGFR [289]. For individuals diagnosed with metastatic pancreatic cancer, the safety and effectiveness of the treatment were demonstrated by a median overall survival (mOS) of 4.9 months among the entire group of 14 patients who received anti-EGFR CAR T cells [290].
Sialic acid-binding immunoglobulin-type lectin (Siglec) Cell-surface proteins known as Siglecs exhibit the ability to attach themselves to sialic acid. These proteins are predominantly present in immune cells, belonging to a specific group within the I-type lectins. Targeting sialic acids on tumor cells can be accomplished through direct means as well. A new advancement comprises the development of CAR T cells based on Siglec-7/9, which specifically target tumor cells that express sialic acid, causing a delay in the growth of tumors within a melanoma model [291]. According to a study, Siglec-7 and Siglec-9 ligands are specifically expressed by PDAC cells, indicating the potential effectiveness of CAR T cells in combating PDAC [292]. Furthermore, enhancing the effectiveness of solid tumor cellular immunotherapy is greatly facilitated by the cancer cell desialylation approach that reverses the state of immune evasion. By eliminating the Siglec-5 and Siglec-10 genes, it became possible to make a CAR macrophage that exhibits enhanced anti-cancer activity as a result of blocking the glycoimmune checkpoint [293].
Carcinoembryonic antigen (CEA) In order to evaluate the efficacy of CEA-specific CAR T cells in combination with recombinant human IL-12 for the treatment of various solid tumors, an experiment was conducted. The findings illustrated that the incorporation of rhIL-12 alongside anti-CEA CAR T cells notably augmented their capacity to suppress the proliferation of pancreatic tumor cells when compared to solely utilizing CEA CAR T-cell treatment [294]. Regarding the central role of IL-12 in CAR T cells, a study has proven that membrane-bound IL-12 in CAR T cells targeting TAG72 promotes anti-tumor responses against human ovarian cancer xenograft models [295]. However, there is a need to apply this approach in PDAC that has not been met.
Additionally, CEACAM7 (CGM2), which is a part of the CEA protein family, may serve as a potential target for PDAC and is specifically present exclusively in the colon and the pancreas. The remission of xenograft tumors occurs as a result of the targeted destruction of pancreatic cancer cells expressing the specific antigen by CAR T cells designed to recognize CEACAM7 [296].
Mesothelin (MSLN) CAR T cells have the ability to be altered in a way that enables them to identify a cell surface antigen called MSLN. This antigen is associated with the invasion of tumors and is present in mesothelial tissues, albeit in small amounts. However, it is highly expressed in PDAC [275]. A potent anti-MSLN hYP218 CAR T cells possess improved abilities to infiltrate and remain in tumors, enhancing their effectiveness in combating pancreatic cancer in vitro and in vivo [297]. CAR T cell therapy, which targets both MSLN and CD19 simultaneously, proved to be a safe and well-tolerated approach in treating individuals suffering from metastatic PDAC [298]. In orthotopic animal models of human pancreatic cancer, it was demonstrated that MSLN-specific CAR T cells are efficient [299]. Mice with extremely aggressive PDAC experience tumor shrinkage when subjected to a mixture of MSLN-redirected CAR T cells and TNF-α/IL-2-armed oncolytic adenoviruses [300]. In a phase 1 trial, T cells engineered to express a CAR specific for MSLN were tested in six patients with chemotherapy-refractory metastatic PDAC. The treatment was well tolerated, with no serious toxicities. Disease stabilized in two patients, and one patient showed a significant reduction in tumor metabolic activity, providing evidence of the potential anti-tumor activity of these engineered T cells [301].
Disialoganglioside (GD2) GD2, present on the external cellular membrane, is integral to the immunological characteristics of mammalian cells; however, it rarely elicits an immune reaction. Due to the prevalence of GD2 in embryonal malignancies such as brain tumors and its infrequent manifestation in healthy cells, it is viable to target GD2 molecules using CAR T cells specific to this molecule [275].
Natural killer group 2D (NKG2D) The NKG2D receptor shows potential as a target for immunotherapy of malignant neoplasms. CAR T cells specific to NKG2D have been employed in the treatment of patients with hematologic and solid tumors. An evaluation was conducted by researchers to determine the practicality and safety of NKG2D-specific CAR T cells, resulting in the discovery that their capacity to multiply and endure within the body was restricted. Gao and colleagues have successfully suppressed the 4.1R gene in NKG2D-specific CAR T cells, thereby augmenting the efficacy of CAR T cells in combatting pancreatic carcinoma [302].
Epithelial cell adhesion molecule (EpCAM) EpCAM is a transmembrane glycoprotein of type I that is excessively expressed in various carcinomas, for instance, colon, stomach, PDAC, and endometrial malignancies. Its connection to the Wnt/β-catenin signaling pathway has been observed, which is believed to trigger inadequate infiltration of T-cells in different human malignancies [275]. A number of clinical trials have been registered to utilize EpCAM-specific CAR T cells in individuals suffering from pancreatic cancer (NCT04151186 and NCT03013712). In these trials, outcomes like toxicity profile, survival time and persistence of CAR T cells in vivo, and anti-tumor efficacy will be measured.
Mucin-1 (MUC-1; CD227) At the apical surface of epithelial cells, the transmembrane mucin glycoprotein MUC-1 shows a high level of expression. In more than 80% of human PDAC, MUC-1 is excessively expressed. This excessive expression of MUC-1 is associated with a grim prognosis and increased metastasis. Moreover, through the upregulation of multidrug resistance genes, MUC-1 boosts chemo-resistance in pancreatic cancer cells [303]. In xenograft models of pancreatic cancer, there was successful inhibition of tumor growth by anti-MUC-1 CAR T cells, which exhibited effective targeting capabilities and induced cytotoxicity [304]. To investigate the effectiveness of MUC-1-specific CAR T cells on individuals suffering from pancreatic cancer, two clinical trials have been officially registered under the codes NCT05239143 and NCT04025216. The former clinical trials demonstrated three patients with different types of cancer have been treated with anti-MUC-1 CAR T cells, showing good tolerance and no observed toxicities [305]. Latter clinical trial showed safety and preliminary efficacy in treating various solid tumors, with no dose-limiting toxicities observed in the six treated patients and preliminary results indicating stable disease in all patients [306].
CD133 Both hematopoietic cells and epithelial cells exhibit the pentaspan transmembrane glycoprotein CD133. CD133 has been identified not only in pancreatic cancer CSCs but also in different tumors like hepatocellular and gastric carcinomas, emphasizing its widespread presence in malignancies. In 50% of cases involving pancreatic cancer, the expression of CD133 was observed to be significantly high [289]. CD133-specific CAR T cells were administered to 7 individuals who suffer from PDAC during a phase 1 clinical trial. Before the infusion of CAR T cells, the patients were treated with cyclophosphamide and nab-paclitaxel. The outcomes of the trial exhibited 3 instances of disease stabilization, 2 instances of partial remission, and 2 instances of disease progression [307].
Prostate stem cell antigen (PSCA) Initially, PSCA was identified as a surface glycoprotein that consists of 123 amino acids and is linked to glycophosphatidylinositol. Its function remained unknown, although it showed significant presence in prostate cancers while exhibiting minimal levels in the prostate epithelium, urinary bladder, kidney, esophagus, stomach, and placenta. Further investigations confirmed its amplified expression in various human cancers, such as pancreatic cancer, while being absent in a healthy pancreas. By employing a humanized mouse model for pancreatic cancer, it was observed that CAR T cells specifically targeting PSCA were able to prompt the eradication of tumors [308].
CD47 CD47, an immunoglobulin superfamily member, frequently exhibits heightened expression in various hematological and solid cancer tumors. Its crucial function involves the inhibition of phagocytosis, leading to enhanced tumor survival, metastasis, and angiogenesis. CD47 is recognized as a "don’t eat me" since it bonds with signaling regulatory protein alpha (SIRP-α) and obstructs the phagocytosis of cancer cells [309]. The blocking of pancreatic xenograft tumor growth is efficiently accomplished and cancer cells are effectively killed by CD47-specific CAR-T cells [310]. A significant challenge in using CD47-CAR-T cells could be the potential detrimental effects on red blood cells and platelets due to the expression of CD47 on these cells. However, in this study, CD47-CAR-T cells were administered intratumorally, which may prevent the induction of toxic effects on other cells. Therefore, the clearing of blood cells during systemic injection of this treatment should be considered as a potential AE.
Claudin18.2 (CLDN18.2) The protein known as CLDN18.2 is an isoform-specific to the stomach of the CLDN18 tight junction protein. This protein is found in high levels in various types of cancer, particularly those affecting the digestive system like pancreatic cancer. Therefore, it could be a promising candidate for cancer treatment strategies [311–313]. Studies indicated a positive response rate for CT041, which are autologous T cells that have been genetically modified to express a CAR targeting CLDN18.2, in cases of digestive system malignancies [311, 314–316]. Two patients with metastatic pancreatic cancer were treated with anti-CLDN18.2 CAR T cell therapy (CT041) after standard treatments failed. Both patients experienced CRS, which was managed with tocilizumab. The first patient showed a partial response with a significant reduction in lung metastasis, while the second patient achieved a complete response. Both cases experienced an increase in CD8+ T cells and Treg cells, a decrease in CD4+ T cells and B cells, an increase in IL-8, and a decrease in TGF-β1. The tumors were well-controlled at the last follow-up [312].
CAR-NK cell therapy
CAR NK cell therapy is a promising strategy in cancer treatment that seeks to enhance the cancer-fighting power of NK cells. CAR-NK cells are engineered to express CARs that recognize specific antigens in cancer cells, which allows them to target and kill cancer cells more effectively [106, 317]. Table 4 provides a comprehensive comparison between CAR T cells and CAR NK cells. Compared to CAR T cells, CAR NK cells possess multiple benefits. Their limited lifespan implies a decreased likelihood of unintended harm to healthy cells (referred to as on-target/off-tumor toxicity). The unique set of cytokines they release signifies a reduced potential for CRS and neurotoxicity. Furthermore, their lower propensity for alloreactivity facilitates the production of off-the-shelf allogeneic CAR NK cells derived from NK cell lines [318]. However, they may have several negative points, which restrict their broad application in clinical contexts. First of all, difficulties in proper antigen selection, antigen heterogeneity, donor selection, challenges in designing an effective CAR, and difficulties in producing and storing CAR NK cells are fundamental hurdles in this type of treatment modality [106, 319]. Secondly, issues such as NK cell infiltration into tumor sites and the short half-life of NK cells must be considered [319]. This short lifespan can necessitate repeated administrations to achieve a durable response. Furthermore, the need for continuous immune surveillance and prevention of cancer recurrence requires the reprogramming of CAR NK cells with memory cell properties and long-term survival in vivo [320, 321]. Lastly, NK cells have several inhibitory killer-cell immunoglobulin-like receptors (KIRs) on their surface, which are cognate with their ligands, HLA molecules. Thus, the universally expressed HLA molecules on nucleated cells can inhibit CAR NK cell function [318]. Thus, the translation of CAR NK cell therapy from bench to bedside requires addressing the aforementioned challenges properly.
Table 4.
A comprehensive comparison between CAR T cell and CAR NK cell therapies
| Difference | CAR T cell | CAR NK cell | References |
|---|---|---|---|
| Key markers | TCR, CD3 | CD16, CD56 | [572] |
| Receptor activated | NKG2D, NKG2C, NKp44, KIR | [572] | |
| CAR generations | Five generations | Four generations | [106, 573–576] |
| Intracellular and co-stimulatory signaling domains | CD3ζ, CD28, 4-1BB (CD137), CD27, CD40, OX40 (CD134) | CD3ζ, DAP10, DAP12, 2B4 (CD244), 4-1BB, CD28 | [318, 576–578] |
| Production of memory cells | + + + | + | [106, 579, 580] |
| Off-the-shelf products | + (HLA-matched allogeneic CAR T cells) | + (non-HLA-matched allogeneic and NK cell lines like NK-92 cells) | [318, 581] |
| Time for manufacturing |
1 to 2 weeks Rapid manufacturing in 24 h is also reported |
Exact timeline can vary (typically 2 to 4 weeks) | [582, 583] |
| Redosing |
Not limited by cell number Risk of alloimmunization |
Not limited by cell number | [319] |
| In vitro expansion during manufacturing | + (Autologous or allogeneic T cells can be expanded after CAR transduction.) | + (autologous NK cell, iPSCs, and NK-92 cells can be pre-expanded before CAR transduction.) | [318, 584] |
| In vivo persistence |
Relative long-term persistence of functional CAR T cells (armored CAR T cells) Intermediate (weeks to months) In some patients with leukemia, CAR-T cells can be identified several years after being infused |
Low and limited persistence in the absence of cytokine Short-term lifespan without IL-15 Cord blood-derived CAR NK cells can persist for at least 12 months (Liu et al.) |
[572, 585, 586] |
| Immune cell sources |
Autologous PBMCs PBMCs from well-matched donor |
Peripheral blood Umbilical cord blood and cord blood HPSCs Differentiated pluripotent stem cells (e.g., iPSCs) NK-92 cell line (an immortalized NK lymphoma cell line) |
[106, 572, 584] |
| Cytotoxicity mechanisms |
In a CAR-dependent manner Perforin and granzyme Inducing apoptotic signaling pathways in tumor cells |
In both CAR-dependent and -independent manners Perforin and granzyme ADCC through CD16 Inducing apoptosis |
[318, 572] |
| Risks and toxicities |
+ + + (CRS, neurotoxicity, and GVHD) Risk of malignancy after treatment (low risk) |
+ (less common) A protocol for freezing and thawing needs to be developed and clinically evaluated for a ready-to-use product |
[319, 572, 587, 588] |
| Infiltration to TME | Poor (particularly in cold tumors) | Usually poor | [318, 589] |
| Combination therapies |
Chemotherapy (like cyclophosphamide) Radiotherapy Immune checkpoint inhibitors (like anti-PD-1) Oncolytic viruses Cancer vaccines Immunomodulatory agents Allogeneic hematopoietic cell transplantation Metabolic inhibitors |
Immune checkpoint inhibitors (anti-NKG2A antibody, monalizumab, lirilumab, and so on) Immunomodulatory drugs (lenalidomide) Epigenetic modulators (vorinostat) Oncolytic viruses (adenoviruses) Small-molecule inhibitors (GSK3i) |
[106, 590] |
| Clinical trials |
Extensive clinical trials Proven effectiveness (at least 6 FDA-approved CAR T cell therapies) |
Limited clinical trials No FDA-approved CAR NK cell therapies yet Clinical efficacy reported in some studies |
[106, 318, 576] |
ADCC: Antibody-dependent cellular cytotoxicity, CAR: Chimeric antigen receptor, CRS: Cytokine release syndrome, FDA: The U.S. food and Drug Administration, GSK3i: Glycogen synthase kinase-3 inhibitor, GVHD: Graft-versus-host disease, HLA: Human leukocyte antigen, HPSCs: Hematopoietic stem and progenitor cells, iPSCs: Induced pluripotent stem cells, KIR: Killer-cell immunoglobulin-like receptor, NK: Natural killer, PBMC: Peripheral blood mononuclear cell, PD-1: Programmed cell death protein 1, TCR: T cell receptor, TME: Tumor microenvironment
When ROBO1 is targeted, CAR-NK immunotherapy accompanied by radiation therapy proves to be more effective in treating human PDAC in an orthotopic mouse model [322]. A study demonstrates the effectiveness of a novel human NK cell-based immunotherapy targeting PSCA. It found that these cells effectively suppressed PSCA+ pancreatic cancer in vitro and in vivo. The therapy showed promising results without causing systemic toxicity [323]. Furthermore, the inhibition of tumor growth and enhancement of survival were observed in a mouse model of pancreatic cancer when utilizing a fusion of CAR-NK cells that targeted MSLN, along with cGAMP, an agonist for STING [324]. There are two clinical trials that have been registered for the implementation of ROBO1 and MUC-1-specific CAR NK cells in the existing clinical scenario of immunotherapeutic methods, specifically for patients diagnosed with pancreatic cancer (NCT03941457 and NCT02839954). Outcome measurements include an examination of the safety profile and ORR.
In the realm of immunotherapy for pancreatic cancer, PSCA has recently gained acclaim as a promising contender. Research findings highlight that CAR-NK cells designed to target PSCA demonstrate notable efficacy in combating advanced PDAC in humans, all while ensuring the absence of any harmful effects at a systemic level [323]. These positive outcomes provide a rigorous rationale for the future progression of clinical trials.
Induced pluripotent stem cells (iPSCs) provide a convenient supply of lymphocytes for immunotherapy. These NK cells, derived from iPSCs, express essential NK-defining markers such as CD56 and CD16. They demonstrate cytotoxicity through cytokine secretion and ADCC, showing potential for cancer treatment [325, 326]. The first-in-class, off-the-shelf iPSC-derived NK cell therapy called FT500 is currently being evaluated in a phase I clinical trial. This trial aims to treat advanced solid tumors, including pancreatic cancer. FT500 is administered both as a monotherapy and in combination with checkpoint inhibitor therapy (nivolumab, pembrolizumab, Atezolizumab), IL-2, cyclophosphamide, and fludarabine (NCT03841110).
Cytokine-induced killer (CIK) cell therapy
CIK cells form a diverse group of CD8+ T cells that were produced from lymphocytes extracted from human peripheral blood and simply expanded ex vivo through incubation with an anti-CD3 antibody, IFN-γ, and IL-2. Through FasL and perforin, they have the ability to eliminate cancer cells. Depending on the existence of the cell surface molecule CD56, CIK cells are additionally categorized into two primary subsets: T cells that are positive for CD3 and CD56, and T cells that are positive for CD3 but negative for CD56 [327]. Adopting CIK cells and transferring them has proven to be highly effective and safe in cancer treatment, as demonstrated by the increased survival of individuals affected by different types of tumors. When utilized alongside chemotherapy, CIK cell therapy exhibits enhanced efficiency in thwarting cancer relapse and enhancing patients' prognosis [24].
Researchers have investigated the application of CIK cells as a potential second-line treatment for advanced pancreatic cancer, which has yielded encouraging outcomes in both standalone usage and when combined with other therapeutic methods. In a phase II clinical investigation, the inclusion of CIK cells alongside gemcitabine-refractory advanced pancreatic cancer demonstrated a mOS of 6.2 months among patients [328]. In advanced pancreatic cancer, gemcitabine-resistant patients who underwent CIK cell therapy in combination with S-1, an oral fluoropyrimidine derivative, demonstrated a mOS of 6.6 months, surpassing the mOS of patients solely treated with S-1 alone (6.1 months) [329]. Following CIK cell therapy, individuals diagnosed with advanced pancreatic cancer exhibit notable enhancements in the OS [330].
Immune checkpoint-oriented immunotherapy
Immunotherapy has become a leading pillar of cancer treatment, thanks to the triumph of an effective ICB method, mainly exemplified by the approval of ipilimumab in 2011. By inhibiting specific inhibitory immune checkpoints like CTLA-4, PD-1, and PD-L1, ICB actively halts or reverses the development of acquired peripheral tolerance to cancer antigens, consequently restoring T-cell activation [331]. Table 5 presents a comprehensive overview of clinical trials investigating the potential of ICIs and immunomodulatory agents in the treatment of pancreatic cancer. In the subsequent discourse, we elucidate the pivotal significance of immune checkpoints in the therapeutic intervention of pancreatic cancer.
Table 5.
Clinical trials in pancreatic cancer investigating the effectiveness of immune checkpoint inhibitors
| Agent | Target | Combination therapy | Participants | Phase | Outcomes* | Status | NCT identifier/References |
|---|---|---|---|---|---|---|---|
| Atezolizumab + Selicrelumab (CD40 agonist) + Bevacizumab + Tiragolumab + Tocilizumab | PD-L1, CD40, VEGF, TIGIT, IL-6R | Nab-Paclitaxel + Gemcitabine + Oxaliplatin + Leucovorin + Cobimetinib + Fluorouracil + PEGPH20 + BL-8040 (CXCR4 antagonist) + RO6874281 + AB928 + LSTA1 | 340 | I, II |
ORR = 6.1% (n = 66) in atezolizumab plus PEGPH20 group (arm A) and ORR = 2.4% (n = 42) in chemotherapy group (arm B) 65.2% (arm A) and 61.9% (arm B) had grade 3/4 AEs 4.5% (arm A) and 2.4% (arm B) had grade 5 AEs Limited clinical activity |
Active, not recruiting | NCT03193190/[591] |
| Pembrolizumab | PD-1 | Onivyde (chemotherapy) + BL-8040 (Motixafortide) | 80 | II |
BL-8040 increased CD8+ effector T cells and decreased MDSCs and circulating regulatory T cells DCR = 34.5% (nine patients with stable disease and one patient with partial response) and mOS = 3.3 months (n = 29) in cohort 1 ORR = 32%, DCR = 77%, and mDOR = 7.8 months in cohort 2 (n = 22) |
Completed | NCT02826486/[506] |
| Epacadostat (INCB24360) + CRS207 + GVAX cancer vaccine | 40 | II |
Determining the recommended dose of epacadostat Assessing survival |
Active, not recruiting | NCT03006302 | ||
| Cyclophosphamide + GVAX cancer vaccine + Radiotherapy | 58 | II |
mOS = 15.8 months and distant metastasis free survival = 9.7 months Grade 3 AEs included 1 case each of dermatitis, colitis, DKA, nephritis, and pneumonitis Increased densities of granzyme B+CD8+ T cells, TH1, and TH17 in TME Increased M2-like tumor-associated macrophages in TME |
Completed | NCT02648282/[592, 593] | ||
| Olaptesed pegol (NOX-A12) | 20 | I, II |
25% of patients achieved stable disease OS = 42% at 6 months and 19% at 12 months mPFS = 1.87 months No objective responses NOX-A12 monotherapy stimulated TH1 cytokines (IFN-γ and IL-2) |
Completed | NCT03168139/[594] | ||
| Defactinib | 59 | I, II |
Determining safety and tolerability Determining early indications of improved anti-cancer immunotherapy |
Recruiting | NCT02758587 | ||
| Acalabrutinib (ACP-196) | 77 | II |
14.3% (monotherapy arm; n = 37) and 15.8% (combination therapy arm; n = 40) had grade 3–4 AEs ORR = 0% and DCR = 14.3% in monotherapy arm Vs. ORR = 7.9% and DCR = 21.1% in combination therapy arm mPFS = 1.4 months |
Completed | NCT02362048/[595] | ||
| Gemcitabine hydrochloride + Nab-paclitaxel + Paricalcitol | 9 | I |
Determining the safety Determining number of tumor infiltrating lymphocytes in resected pancreatic cancer subjects Evaluating OS, DFS, PD-L1 expression, CD8+CD45RO+ cells, and immune signature in TME |
Completed | NCT02930902 | ||
| Defactinib + Gemcitabine | 43 | I |
DCR = 58.8% (n = 17) with one partial response and nine stable disease mPFS = 4.2 months and OS = 9.1 months |
Completed | NCT02546531/[596] | ||
| Cabiralizumab (FPA008) + Nivolumab (BMS-936558) | CSF-1R, PD-1 | N/A | 313 | I | Determining mPFS, OS, ORR, mDOR, pharmacokinetics, and safety | Completed | NCT02526017 |
| AMG820 + Pembrolizumab | CSF-1R, PD-1 | N/A | 117 | I, II |
87.9% with grade ≥ 3 AEs Most common AEs: increased aspartate aminotransferase (59.5%), fatigue (48.3%), periorbital/face edema (48.3%), and rash/maculopapular rash (37.1%) Immune-related partial response in 3 patients (3%; DOR 9.2, 10.0, 12.5 months) Immune-related stable disease in 39 patients (34%) |
Completed | NCT02713529/[597] |
| Pembrolizumab + IMC-CS4 | PD-1 + CSF-1R | Cyclophosphamide + GVAX cancer vaccine | 12 | I |
Grade 3/4 AEs: rash and diarrhea mDFS/mOS = 12.6/20.4 month and pathologic response rate = 78% 75% of patients (6/8) had a > 80% increase of CD8+ T cells |
Completed | NCT03153410/[415] |
| Pembrolizumab | PD-1 | ARRY-382 (PF-07265804; CSF-1R inhibitor) | 82 | I, II |
Limited clinical benefits AEs: increased transaminases and creatine phosphokinase 3.7% of patients (1/27) had a partial response lasting 2.4 months (n = 27) |
Terminated due to insufficient efficacy | NCT02880371/[598] |
| Durvalumab (MEDI4736) | PD-L1 | Pexidartinib (PLX3397; a CSF-1R-directed tyrosine kinase inhibitor) | 48 | I |
No unexpected toxicities 15% patients had stable disease (n = 47) Increased CSF-1 and FLT3-L concentrations and decreased CD14lowCD16high monocytes and DCs in blood Impaired IFN-λ/IL-29 production by type 1 conventional DCs Limited antitumor clinical activity |
Completed | NCT02777710/[599] |
| Ipilimumab | CTLA-4 | Gemcitabine hydrochloride | 21 | I |
Common grade 3 or 4 AEs: anemia (48%), leukopenia (48%), and neutropenia (43%) ORR = 14% (3/21) and seven patients had stable disease mOS = 6.90 months and mPFS = 2.78 months Median response duration = 11 months |
Completed | NCT01473940/[600] |
| Botensilimab (AGEN1181) | CTLA-4 | Gemcitabine + Nab-paclitaxel | 78 | II | Determining PFS, complete response, ORR, DOR, change from baseline in carbohydrate antigen 19–9, and potential AEs | Recruiting | NCT05630183 |
| Nivolumab | PD-1 | Nab-paclitaxel + Gemcitabine + Carboplatin | 114 | I |
Dose-limiting toxicity = One hepatitis among 6 patients 96% (48/50) had grade 3/4 AEs (n = 50) 2% (1/50) had grade 5 AEs mPFS = 5.5 months, mOS = 9.9 months, and ORR = 18% mPFS/mOS = 5.5/9.7 months (PD-L1 < 5%) and 6.8/11.6 months (PD-L1 ≥ 5%) Significant increased Ki67+ CD8+/CD4+ cells |
Completed | NCT02309177/[601] |
| Cyclophosphamide + GVAX cancer vaccine + Radiotherapy | 30 | II |
Progressive disease = 5.5% (1/18), occult intra-operative metastatic disease = 16.6% (3/18) and definitive surgical resection = 77.7% (14/18; n = 18) mOS = 20.4 months (median follow-up of 20.5 months) and pathologic response rate = 35% Grade ≥ 3 AEs: two cases (nivolumab-related autoimmune hepatitis and SBRT-related pancytopenia) |
Completed | NCT03161379/[602] | ||
| Nab-Paclitaxel + Gemcitabine + APX005M (sotigalimab; CD40 agonist) | 129 | I, II |
Arm A (nivo/chemo; n = 34): 1-year OS = 57.7% (P = 0.006), mOS = 16.7 months, mPFS = 6.4 months, ORR = 50%, DCR = 74%, and mDOR = 7.4 months Arm B (sotiga/chemo; n = 36): 1-year OS = 48.1% (P = 0.062), mOS = 11.4 months, mPFS = 7.3 months, ORR = 33%, DCR = 78%, and mDOR = 5.6 months Arm C (sotiga/nivo/chemo; n = 35): 1-year OS = 41.3% (P = 0.233), mOS = 10.1 months, mPFS = 6.7 months, ORR = 31%, DCR = 69%, and mDOR = 7.9 months Common AEs: CRS (0%, arm A; 24%, arm B; 34%, arm C), thrombocytopenia (50%, arm A; 57%, arm B; 63%, arm C), elevated liver function tests (67%, arm A; 81%, arm B; 74%, arm C) |
Completed | NCT03214250/[603] | ||
| CRS-207 vaccine + GVAX + Cyclophosphamide | 93 | II |
mOS = 5.9 months in nivolumab group (n = 51; arm A) and 6.1 months in group without nivolumab (n = 42; arm B) ORRs = 4% in arm A and 2% in arm B grade ≥ 3 AEs = 35.3% in arm A and 11.9% in arm B Changes in TME include an increase in CD8+ T cells and a decrease in CD68+ myeloid cells |
Completed | NCT02243371/[604] | ||
| Cabiralizumab + Nivolumab | CSF-1R + PD-1 | Nab-paclitaxel + Onivyde + Fluorouracil + Gemcitabine + Oxaliplatin + Leucovorin + Irinotecan hydrochloride | 202 | II | Determining PFS, ORR, DOR, OS, and AEs | Completed | NCT03336216 |
| Durvalumab + Tremelimumab | PD-L1, CTLA-4 | Radiotherapy | 65 | I, II |
No dose limiting toxicities Grade 3–4 AEs: lymphopenia and anemia 3 patients with partial response and 7 patients with stable disease (n = 31) mPFS/OS = 1.7/3.4 months (cohort 1), mPFS/OS = 2.6/9.1 months (cohort 2), mPFS/OS = 1.6/3 months (cohort 3), and mPFS/OS = 3.2/6.4 months (cohort 4) |
Completed | NCT02311361/[605] |
| Nivolumab + Ipilimumab | PD-1, CTLA-4 | Radiotherapy | 30 | II |
ORR = 4% (1/28), DCR = 11% (3/28), PFS = 2.3 months, and OS = 2.9 months 1 patient had a complete response at 13 months Grade 3–4 AEs: lymphopenia (grade 4 in 1 patient), neutropenia, fatigue, muscle weakness, ALT/AST increase, hepatobiliary dysfunction, acute kidney injury, hyperglycemia, and hypokalemia |
Active, not recruiting | NCT04361162/[606] |
| Anti-PD-1 monoclonal antibody | PD-1 | Radiotherapy | 21 | II | Determining ORR, OS, and AEs | Unknown | NCT03374293 |
| Nivolumab (BMS-936558) + Urelumab (BMS-663513) + BMS-986253 | PD-1, CD137 (4-1BB), IL-8 | Cyclophosphamide + GVAX cancer vaccine | 76 | II |
Arm A (GVAX/cyclopho; n = 16): mDFS/mOS = 13.90/23.59 months Arm B (nivo/GVAX/cyclopho; n = 14): mDFS/mOS = 14.98/27.01 months Arm C (urelu/nivo/GVAX/cyclopho; n = 10): mDFS/mOS = 33.51/35.55 months Increased intratumoral CD8+CD137+ cells in arm C compared t arm B (P = 0.003) Improved DFS (P = 0.242 and P = 0.173) and OS (P = 0.377 and P = 0.279) in arm C compared to arms A and B |
Recruiting | NCT02451982/[381] |
| BGB-A425 + Tislelizumab (BGB-A317) + LBL-007 | TIM-3, PD-1, LAG-3 | N/A | 358 | I, II | Determining AEs, maximum tolerated dose, ORR, DOR, DCR, PFS, and pharmacokinetics | Recruiting | NCT03744468 |
*In cases where the outcomes of the clinical trial have not yet been released, the goals (primary/secondary outcome measures) of the study are mentioned
AEs: Adverse events, CSF-1R: Colony stimulating factor 1 receptor, CTLA-4: Cytotoxic T lymphocyte antigen-4, DC: Dendritic cell, DCR: Disease control rate, ICB: Immune checkpoint blockade, IL-6R: Interleukin 6 receptor, LAG-3: Lymphocyte activation gene-3, mDFS: Median disease-free survival, mDOR: Median duration of response, mOS: Median overall survival, N/A: Not applicable, ORR: Objective response rate, PD-1: Programmed cell death protein 1, PD-L1: Programmed death-ligand 1, mPFS: Median progression-free survival, TH1: Type 1 T helper, TIGIT: T-cell immunoreceptor with Ig and ITIM domains, TIM-3: T-cell immunoglobulin and mucin domain-containing protein 3, TME: Tumor microenvironment, VEGF: Vascular endothelial growth factor
Inhibitory immune checkpoints
PD-1/PD-L1 axis
The PD-1/PD-L1 axis has been studied in relation to immune checkpoint molecules in pancreatic cancer following the successful use of anti-PD-1/PD-L1 treatment in melanoma. PD-1 is a member of the B7-CD28 protein family and its expression is associated with T-cell exhaustion. PD-1 ligands (PD-L1 and PD-L2) are expressed by tumor cells, MDSCs, TAMs, and tumor-infiltrating DCs. Engagement between PD-1 and PD-L1 leads to T-cell exhaustion by blocking T-cell activation [332, 333]. Certain malignancies have demonstrated promising results when treated solely with PD-1/PD-L1 inhibitors [334, 335]. PD-L1 inhibitors elicit different reactions in individuals, as evidenced by some PD-L1 positive patients exhibiting unfavorable responses while some PD-L1 negative patients responding favorably. This implies the potential involvement of other PD-1 ligands, like PD-L2, in impacting the efficacy of PD-1 axis immunotherapy in specific cancers. A body of research accentuates the notion that PD-L2 influences the anti-PD-1 axis immunotherapy, particularly in PDAC [336, 337]. Chemotherapy-induced senescent cancer cells modify the TME, promoting immunosuppression and pancreatic tumor growth. PD-L2 is highly upregulated in senescent cancer cells, helping them evade the immune system and persist within tumors. Blocking PD-L2 in combination with chemotherapy leads to tumor regression and remission in mice [338], offering a promising therapeutic strategy targeting senescence-induced vulnerabilities.
Combination immunotherapy targeting PD-L1 and CCL5 has shown benefits in PDAC by decreasing Treg and TAM infiltration, inducing CD8+ T-cell activation, promoting tumor regression, and improving OS [339]. Tumor regression, improved OS, and the generation of anti-tumor memory cells were achieved by the joint action of anti-tumor necrosis factor receptor 2 (TNFR2) and PD-L1 monoclonal antibodies, by reducing the infiltration of Tregs and TAMs while activating CD8+ T-cells in PDAC microenvironment [340]. ADH-503, an agonist of CD11b, exerts an agonistic influence on innate immune responses, leading to a reprogramming effect. This reprogramming enhances the response of innate immune responses towards immunotherapies, specifically anti-PD-L1 antagonists and anti-4-1BB agonists, thereby facilitating a more effective therapeutic outcome in the treatment of pancreatic cancer [341]. A bispecific immunocytokine (PD-1/IL-2 complex) targeting of PD-1 and IL-2Rβγ enhances tumor-antigen-specific T-cell activation while reducing Treg-mediated suppression. The use of this immunocytokine, combined with radiotherapy, attenuates the progression of pancreatic cancer and impedes its metastatic potential [342].
CTLA-4 (CD152)
CTLA-4 is predominantly found in Tregs and its expression increases when T-cells are activated. CTLA-4 works intrinsically by suppressing the co-stimulatory signal within the cell, inhibiting T-cell activation. It also acts externally by removing CD80 and CD86 from APCs, which reduces the response of effector T cells [332]. Controlling the pathway of CTLA-4/CD80 regulates the entry of T cells into the microenvironment of pancreatic cancer. By interrupting the interaction between CTLA-4 and CD80, one can induce the infiltration of CD4+ and CD8+ T-cells into the microenvironment of PDAC [343]. Pancreatic tumors can be regressed by inhibiting both IL-6 and CTLA-4, and this regression occurs through a T cell and CXCR3-dependent mechanism [344].
LAG-3
Cancer cells utilize the LAG-3 signaling pathway to escape the immune system's detection. Through interaction with Galectin-3, activated T cells experience decreased functionality. Moreover, the activity of plasmacytoid DCs, responsible for initiating the growth of naïve T cells, is hindered by LAG-3. Additionally, LAG-3 has the capacity to regulate T-cell proliferation, reduce memory and effector T-cell immune responses, and heighten immunosuppression through the suppression mediated by Tregs [332]. Pancreatic cancer patients with TILs that express LAG-3 exhibit lower rates of DFS [345]. Anti-tumor immunity and enduring response in pancreatic cancer can be achieved by directing attention towards T cell checkpoints 4-1BB and LAG-3, alongside myeloid cell CXCR1/CXCR2 [346].
TIGIT
The expression of TIGIT occurs on the surface of immune cells and results in the inhibition of T-cell stimulation. By attaching to CD155 and CD112, TIGIT generates signals that suppress the activation of T-cells. Additionally, TIGIT can competitively bind to CD226 or CD96 along with CD155 and CD112 in order to suppress the active signal received by T-cells [347]. Increased PD-1 and TIGIT expression were evident in intratumoral T cells [348]. Thus, to optimize the responses of CD8+ T cells against tumors, it is necessary to co-block the TIGIT and PD-1 inhibitory pathways due to their mechanistic convergence [349]. The co-blockade of PD-1 and TIGIT on tissue-resident memory T cells in PDAC revitalizes them [350]. In pancreatic cancer, the CD155/TIGIT axis plays a significant role in boosting and sustaining immune evasion [351]. Combining TIGIT and PD-1 blockade enhances the efficacy of vaccinations in a model of pancreatic cancer [352]. The reinvigoration of T lymphocytes specific to pancreatic tumor cells occurred as a result of the co-blockade of TIGIT/PD-1 and the stimulation of CD40 agonist [351]. A research study uncovered that interactions in samples of human PDAC decrease following chemotherapy, specifically between TIGIT on CD8+ T cells and its receptor on cancer cells. TIGIT was identified as the primary inhibitory checkpoint molecule of CD8+ T cells, revealing that chemotherapy greatly affects the PDAC TME and potentially enhances resistance to immunotherapy [353].
V-domain Ig-containing suppressor of T-cell activation (VISTA)
VISTA is an original member of the B7 family checkpoint molecules. It exerts a distinctive influence on cancer immune evasion through its distinct expression patterns and functions. In contrast to checkpoints that mainly control T-cell effector function and exhaustion, VISTA has various roles. It aids in the functioning of MDSCs, governs the activation of NK cells, promotes the survival of Tregs, restricts antigen presentation on APCs, and also maintains T cells in a state of rest [354–356]. The expression of VISTA is associated with a more favorable prognosis in cases of pancreatic cancer [357]. Pancreatic cancer exhibits an increased expression of the immunological checkpoint VISTA. It has been shown that the activation of VISTA hinders the production of cytokines by T cells that are obtained from metastatic pancreatic cancers [358]. Given this, monoclonal antibodies against VISTA could potentially function as a beneficial immunotherapeutic approach for individuals diagnosed with pancreatic cancer [357, 358].
CD39/CD73 axis
Extracellular adenosine is a metabolite that suppresses the immune system and affects adversely both innate and adaptive immune responses. It is accumulated through the actions of two ectonucleotidases, CD39 and CD73. Adenosine exerts its immunosuppressive effects by binding to A2A receptors on lymphoid and myeloid cells, as well as A2B receptors on myeloid cells. These A2B receptors are frequently overexpressed in cancer cells and have been found to promote tumor growth, spread, and resistance to chemotherapy [359, 360]. In PDAC, the levels of CD73 are notably elevated compared to other types of cancer. This correlation is associated with negative clinical results [361]. The findings of a study highlight the significant role of CD39 and CD73 in promoting PDAC progression. The expression of these ectonucleotidases was associated with worse survival outcomes in human PDAC samples and disrupted the positive impact of tumor-infiltrating CD8+ T cells. Furthermore, targeting both CD39 and CD73 demonstrated superior anti-tumor activity compared to individual inhibition, emphasizing the potential of these molecules as therapeutic targets in PDAC [362]. Several anti-CD73/CD39 antibody-oriented clinical trials are underway, which will assess the effectiveness of agents like oleclumab or MEDI9447 (anti-CD73; NCT02503774), TTX-030 (anti-CD39; NCT03884556), and CPI-006 or mupadolimab (anti-CD73; NCT03454451) [363] alone or in combination with other ICIs. Regarding NCT02503774, the study involved the treatment of 192 patients with oleclumab and durvalumab (anti-PD-L1), with no instances of dose-limiting toxicities during the escalation phase. The most frequently observed side effects were fatigue, diarrhea, and rash. While the escalation phase showed no objective response, the expansion cohorts demonstrated some positive response rates [364].
Bispecific antibodies (BsAbs)
BsAbs have been engineered to effectively engage two specific antigens at the same time. These specialized antibodies effectively modulate the immune response by redirecting and stimulating immune cells, blocking the co-inhibitory receptors on these cells, activating molecules that enhance the immune response, interfering with specific signaling pathways, and employing a strategy of simultaneously targeting multiple cancer antigens [365]. BsAbs have been developed for pancreatic cancer treatment, with examples such as anti-EGFR × HER2 [366], anti-CD3 × CEA [367], MCLA-128 (anti-HER2 × HER3 BsAb; zenocutuzumab) [368], anti-CD3 × EGFR BsAb [369], anti-CD3 (Vγ9TCR) × HER2/Neu [370], XmAb22841 (anti-LAG-3 × CTLA-4; NCT03849469), XmAb23104 (anti-PD-1 × inducible co-stimulatory molecule [ICOS]) [371], ATOR-1015 (anti-CTLA-4 × OX40) [372], and KN046 (anti-CTLA-4 × PD-L1) [373]. BsAb targeting CD3 and EGFR-armed activated T cells have the ability to target and kill drug-resistant pancreatic cancer cells. Furthermore, the "priming" of these resistant cells with BsAb-armed activated T cells enhances their responsiveness to chemotherapeutic drugs through modulation of ABC transporter expression [369]. These findings provide insight into the use of BsAbs for immunotherapy against PDAC. A trial tests XmAb23104’s efficacy and safety in treating advanced solid tumors, both alone and with ipilimumab (NCT03752398). The study showed that XmAb23104 was generally well tolerated at doses up to 15 mg/kg in subjects with advanced solid tumors. Clinical activity was observed, including partial responses in three subjects and stable disease for over 12 months in two subjects [371].
Cancer vaccines
There are several types of cancer vaccines, including whole tumor cell vaccines, DC vaccines, peptide vaccines, DNA vaccines, and mRNA vaccines. While conventional immunotherapies may demonstrate efficacy against cancers featuring identifiable surface antigens specific to tumors, cancer vaccines possess the capability to also encompass a wider range of intracellular antigens for targeting purposes. Up to this point, the FDA has granted approval to a solitary therapeutic vaccine for cancer treatment, namely sipuleucel-T (PROVENGE). This particular vaccine solely enhances patient survival in prostate cancer cases by a mere 4 months [374].
Vaccination is being examined to activate or enhance pre-existing immune responses using agents like GVAX (pancreatic cell lines modified with GM-CSF) or CRS207 (live attenuated Listeria monocytogenes expressing MSLN), either alone or in combination with a mAb targeting the CD40 molecule to activate APCs [41]. In this section, we provide an overview of various types of cancer vaccines and underscore the significant studies conducted with these vaccines (Tables 6, 7).
Table 6.
Categorization of cancer vaccines in the context of pancreatic cancer therapy
| Type | Mechanism of action | Cancer vaccine | References |
|---|---|---|---|
| Whole tumor cell vaccine | Irradiated tumor cells elicit an immune response targeting TSAs or TAAs, which are proteins expressed by tumor cells | Algenpantucel-L (NLG0205) | [375] |
| Photothermal nanoparticle-loaded tumor cells | [607] | ||
| GVAX | [41, 381, 608] | ||
| DC vaccine | DCs are pulsed with TAAs or TSAs, presenting them to effector T cells, resulting in specific immune responses against tumor cells expressing corresponding tumor antigens | MUC1-pulsed DCs | [383] |
| WT1-pulsed DCs | [388] | ||
| α-Galactosylceramide-pulsed DCs | [392] | ||
| MAGE-A3-pulsed DCs | [609] | ||
| Mesothelioma lysate-pulsed DCs | [390] | ||
| DC vaccine plus LAK cells | [391] | ||
| DCs loaded with mRNA encoding CEA | [610] | ||
| Peptide vaccine | Epitope, peptide, or protein expressed by pancreatic tumor cells elicits robust immune responses | RAS oncogene-based vaccine: GI-4000, TG01 | [611–614] |
| HSP-peptide complex-based vaccines: HSPPC-96 | [615] | ||
| Survivin-based vaccine: AYACNTSTL | [616] | ||
| VEGFR-based vaccine: VXM01 | [404, 617, 618] | ||
| Gastrin-based vaccine: G17DT | [619, 620] | ||
| Telomerase-based vaccine: GV1001 | [393, 394] | ||
| DNA vaccine | Transferring a DNA into an organism's system with the aim of producing an antigen, which in turn triggers a safeguarding immune response | ENO1 DNA vaccine | [397, 398] |
| MUC1-VNTRn | [399] | ||
| Chimeric DNA encoding FAPα and survivin | [400] | ||
| mRNA vaccine | The mRNA encoding TSAs, TAAs, and tumor neo-antigens induce a robust immune response | RO7198457 (Autogene Cevumeran) | [401]/NCT04161755 |
| Viral/bacterial vector-based vaccine | This type of vaccine use an immunogenic viral or bacterial vectors to deliver the mRNA encoding TAAs or TSAs | Heat-killed whole cell vaccine of Mycobacterium Obuense: IMM101 | [621, 622] |
| Live-attenuated Listeria Monocytogenes encoding MSLN: CRS-207 | [41] | ||
| Intracellular delivering Salmonella | [403] | ||
| Stem cell-based vaccine | Induction of anti-tumor immunity by oncofetal antigens | iPSC-based cancer vaccine, comprised of autologous iPSCs and CpG | [406] |
CEA: Carcinoembryonic antigen, DC: Dendritic cell, ENO1: α-Enolase, FAPα: Fibroblast activation protein alpha, HSP: Heat-shock protein, iPSC: Induced pluripotent stem cell, LAK: Lymphokine-activated killer, MSLN: Mesothelin, MUC1: Mucin-1, TAA: Tumor-associated antigen, TSA: Tumor-specific antigen, VEGFR: Vascular endothelial growth factor receptor, VNTRn: Variable number tandem repeat, WT1: Wilms’ tumor 1
Table 7.
Key clinical trials of cancer vaccines in pancreatic cancer therapy
| Agent | Target(s) | Mechanism of action | Combination therapy | Participants | Phase | Outcomes* | Status | NCT identifier/Reference |
|---|---|---|---|---|---|---|---|---|
| mRNA-5671/V941 | KRAS-G12D, KRAS-G12V, KRAS-G13D, KRAS-G12C | A tetravalent mRNA-based cancer vaccine for targeting oncogenic KRAS mutations in PDAC | Pembrolizumab | 70 | I | Determining AEs, dose-limiting toxicities, ORR, and changes in the quantity of mutant KRAS specific T cells | Completed | NCT03948763 |
| RO7198457 (Autogene Cevumeran/BNT122) | TSAs and TAAs | A personalized mRNA cancer vaccine delivering mRNA containing specific pancreatic cancer neo-antigens | Atezolizumab + FOLFIRINOX | 29 | I |
Longer median recurrence-free survival in responders (receiving vaccine) compared with non-responders (not receiving vaccine; P = 0.003) at an 18-month median follow-up 6% (1/16) and 100% of patients had grade 3 and 1–2 AEs, respectively (n = 16) Vaccine-expanded T cells were up to 10% of all blood T cells |
Active, not recruiting | NCT04161755/[401] |
| ELI-002/ELI-002 2P | KRAS-G12D, KRAS-G12R | A peptide-based cancer vaccine that consists of amphiphile KRAS peptides combined with CpG adjuvant | N/A | 25 | I |
No dose-limiting toxicities Median relapse-free survival = 16.33 months 84% (21/25) patients showed mKRAS-specific T cell responses |
Active, not recruiting | NCT04853017/[396] |
| KRAS peptide vaccine + Poly-ICLC adjuvant | KRAS | A peptide-based cancer vaccine against mutant KRAS | N/A | 25 | I | Determining drug-related toxicities, changes of IFN-γ producing mutant-KRAS-specific CD8+ and CD4+ T cells at 5/13/17 weeks, and changes in T cell quality (e.g., memory, exhaustion, poly-functionality, and activation) | Recruiting | NCT05013216 |
| KRAS-G12C, KRAS-G12V, KRAS-G12D, KRAS-G12A, KRAS-G13D, KRAS-G12R | A cancer vaccine against oncogenic KRAS mutations | Nivolumab + Ipilimumab | 30 | I | Determining OS, DFS, PFS, ORR, changes of IFN-γ producing mutant-KRAS-specific CD8+ and CD4+ T cells, and drug-related toxicities | Recruiting | NCT04117087 | |
| Personalized synthetic peptide vaccine | TSAs and TAAs | Inducing TSA/TAA-specific effector T-cell immune responses | Imiquimod (TLR7 agonist) + Pembrolizumab + Sotigalimab (APX005M; a CD40 agonist) | 150 | I | Determining AEs, OS, PFS, RFS, response rate, and changes in neoantigen-specific T cell response | Recruiting | NCT02600949 |
*In cases where the outcomes of the clinical trial have not yet been released, the goals (primary/secondary outcome measures) of the study are mentioned
AEs: Adverse events, APC: Antigen-presenting cell, DFS: Disease-free survival, N/A: Not applicable, ORR: Objective response rate, OS: Overall survival, PFS: Progression-free survival, RFS: Recurrence-free survival, TAA: Tumor-associated antigen, TLR7: Toll-like receptor 7, TSA: Tumor-specific antigen
Whole tumor cell vaccines
Utilizing a tumor cell vaccination is a simple and straightforward approach to tumor immunotherapy. The tumor cell vaccination contains both CD4+ helper T-cell and CTL epitopes. Algenpantucel-L (NLG0205) is an example of such a vaccine. Results from a phase II study demonstrated that the combination of Algenpantucel-L with the adjuvants gemcitabine and 5-fluoruracil yielded an 86% survival rate at one year, a 51% survival rate at two years, and a 42% survival rate at three years [375]. Nevertheless, a study demonstrated that Algenpantucel immunotherapy did not yield advantages for patients suffering from advanced PDAC, despite following the standard of care, neo-adjuvant chemotherapy, and chemoradiation [376].
To elicit T-cell immune responses against various tumor antigens, scientists manufactured a pancreatic cancer vaccine called GVAX. This particular vaccine, classified as allogeneic, consists of human GM-CSF-secreting whole tumor cells [377–380]. A study's findings reveal that neo-adjuvant and adjuvant GVAX, with or without nivolumab (an anti-PD-1 monoclonal antibody) and urelumab (an anti-CD137 agonist), are safe. Furthermore, treating with GVAX alongside nivolumab and urelumab leads to a remarkable increase in tumor-infiltrating activated effector T cells. This combination also demonstrates efficacy by substantially enhancing DFS in comparison to GVAX with or without nivolumab [381].
DC vaccines
DCs that were isolated from the patient's peripheral blood were loaded with tumor-associated antigens (TAAs) or tumor-derived mRNA. After the administration of these vaccines, the modified DCs proceed to the lymph nodes, where they transmit antigens to T lymphocytes and concurrently induce co-stimulatory signals [382]. In a study, DCs were gathered from 7 patients who had stage III/IV pancreatic cancer through the employment of apheresis. Afterwards, these collected DCs underwent the process of being pulsed with MUC-1 peptide. The injection of MUC-1-pulsed DCs in these patients exhibited both safety and efficacy, successfully triggering an immune response towards the MUC-1 [383].
Many pancreatic cancer cells exhibit overexpression of the Wilms’ tumor 1 (WT1). Several studies assessed the effectiveness of using DCs pulsed with WT1 peptides and chemotherapy in treating advanced pancreatic cancer [384–388]. A retrospective multicenter analysis was conducted on 255 patients with pancreatic cancer. These patients were receiving standard chemotherapy and a DC vaccine. This study showed that in patients with pancreatic cancer who received a DC vaccine, a positive erythema reaction at the site of DC vaccine injection was linked to better survival [389].
The impact of using mesothelioma lysate-loaded DCs in combination with FGK45 (a CD40 agonist) was examined in PDAC mice models. This innovative technique provoked a remarkable alteration in the transcriptome of the tumor, involving the suppressive indicators on CD8+ T cells, and resulted in a considerable improvement in survival [390]. The administration of lymphokine-activated killer (LAK) cell therapy significantly extended the survival of patients with advanced pancreatic cancer who underwent DC vaccine-based immunotherapy along with gemcitabine. Nevertheless, the use of immunotherapy on its own enhanced the quantity of cancer antigen–targeting CTLs while decreasing the presence of Tregs [391]. In vivo, the induction of anti-tumor immunity against pancreatic cancer is achieved through DC vaccines that have been pulsed with alpha-galactosylceramide [392].
Peptide vaccines
The peptide vaccine candidate GV1001 possesses certain noteworthy cell-penetrating peptide characteristics. GV1001 is generated from a peptide derived from a reverse-transcriptase portion of telomerase, or hTERT. In a phase II study, a significant immune response was observed in a majority of patients with advanced pancreatic cancer who received GV1001, and these immune responders had a notably improved median survival compared to non-responders [393]. However, a subsequent phase III clinical trial combining chemotherapy with the GV1001 vaccine did not yield a significant improvement in OS [394]. Another peptide cancer vaccine, KIF20A-66, was also examined in a phase I/II trial and found to be well-tolerated. The mOS and median progression-free survival (mPFS) were reported as 142 days and 56 days, respectively [395]. Recently, the phase 1 study of ELI-002 2P in patients with KRAS-mutated pancreatic cancer demonstrated promising results. ELI-002 2P is a cancer vaccine that specifically targets lymph nodes and consists of three components. These components include modified G12D and G12R mKRAS long peptides, which have been modified with amphiphiles, and an amphiphile-modified TLR9 agonistic CpG-7909 DNA. The therapy was well-tolerated, induced significant T-cell responses, and resulted in biomarker clearance and improved relapse-free survival. These findings suggest that ELI-002 2P has the potential to be an effective treatment option for patients with immunotherapy-recalcitrant KRAS-mutated tumors [396].
DNA vaccines
Several studies have shown that DNA vaccines targeting TAAs can effectively prolong survival in mice with PDAC. Targeting α-Enolase (ENO1) with a DNA vaccine has been particularly effective [397]. In addition, combining this DNA vaccine with chemotherapy using gemcitabine has shown improved efficacy against multiple TAAs, including ENO1, glyceraldeheyde-3-phosphate dehydrogenase (G3P), keratin, type II cytoskeletal 8 (K2C8), and far upstream binding protein 1 (FUBP1) [398]. Another DNA vaccine targeting mucin 1-variable number tandem repeat (MUC1-VNTRn) has demonstrated strong cytotoxic effects in both in vivo and in vitro experiments [399]. Furthermore, a chimeric DNA vaccine that targets human fibroblast activation protein alpha (FAPα) and survivin has been shown to reduce immunosuppressive cells and increase TILs, thereby creating a more favorable TME for immune responses against pancreatic tumors [400].
RNA vaccines
Personalized cancer vaccines made of mRNA include mRNA that encodes specific tumor-specific antigens (TSAs) and TAAs. Subsequently, APCs take in the mRNA and exhibit the matching peptide antigens, which prompts immune responses encompassing CTLs and memory T cells. RO7198457, also known as BNT122, represents an mRNA-based cancer vaccine that aims to elicit T-cell-triggered immune reactions against tumor neo-antigens. Various clinical studies are scheduled to be conducted among individuals diagnosed with diverse types of cancer, such as pancreatic cancer (NCT04161755 and NCT05968326), solid tumors (NCT03289962), melanoma (NCT03815058), and colon cancer (NCT04486378). However, many of these trials’ results have not been released yet. Next, the outcomes of the clinical trial NCT04161755 are explained.
An mRNA vaccine called autogene cevumeran was generated using uridine mRNA-lipoplex nanoparticles. After the surgical procedure, a combination therapy that included atezolizumab, the mRNA vaccine (with a maximum of 20 neo-antigens per patient), and chemotherapy was conducted. The results indicated that vaccine-enhanced T cells, which accounted for as much as 10% of the total T cells in the bloodstream, experienced re-expansion through a vaccine booster. These re-expanded cells consisted of durable, polyfunctional CD8+ T cells that targeted pancreatic cancer neo-antigens. After a median follow-up period of 18 months, patients who exhibited vaccine-enhanced T cells demonstrated a significantly prolonged median recurrence-free survival when compared to the control group [401].
Viral/bacterial vector-based vaccines
These cancer vaccines use modified viruses or bacteria as vectors to deliver genetic code for tumor antigens into human cells. The infected cells then produce tumor antigens, which trigger an immune response in the host. The bacterial vector can be used to treat castration-resistant prostate cancer, and the bacterial-based cancer vaccine has shown promising anti-tumor effects in clinical trials [402]. One of the widely recognized cancer vaccines of this kind is CRS207, which is a live attenuated strain of Listeria monocytogenes engineered to express MSLN. The utilization of the CRS207 vaccine in individuals diagnosed with metastatic pancreatic cancer has demonstrated promising outcomes in terms of prolonged survival rates while causing minimal detrimental effects to patients [41]. The utilization of an exogenous immunization antigen, administered via Salmonella bacteria acting as a vector, effectively redirects the attention of CD8+ T cells towards cancer cells within the cytoplasm of tumor cells. Consequently, this approach leads to the complete eradication of pancreatic tumors, the enhancement of anti-tumor immunity, and a significant extension in survival duration, as demonstrated in PDAC mouse models [403]. Moreover, VEGFR-2, a target for anti-angiogenic intervention, is expressed on tumor vasculature. VXM01, an oral tumor vaccine using attenuated Salmonella with a VEGFR-2 expression plasmid, was tested in a phase I trial with advanced pancreatic cancer patients. The study found that VXM01 was well tolerated, with no dose-limiting toxicities and significant increases in VEGFR2-specific T effector responses. Vaccinated patients showed reduced tumor perfusion and elevated serum biomarkers indicative of anti-angiogenic activity, which correlated with preexisting VEGFR2-specific T-cell levels [404].
Stem cell-based vaccines
The fact that cancer cells and embryonic tissues have several similar cellular and molecular characteristics suggests that we can potentially utilize iPSCs to stimulate anti-tumor responses within cancer vaccines. Indeed, iPSCs share gene expression profiles with tumor cells. The prevention of tumor growth in murine breast cancer, mesothelioma, and melanoma models is achieved by iPSC vaccines. Acting as an adjuvant, the iPSC vaccine effectively hinders the reoccurrence of melanoma and decreases the spread of tumors [405]. Research demonstrates that a cancer vaccine derived from iPSCs stimulates a defensive immune response in a PDAC mouse model. Furthermore, this immune response is linked to heightened CD8+ effector and memory T cell reactions against tumor cells, the generation of antibodies specifically targeting cancer cells, and a reduction in immunosuppressive Tregs composed of CD4+ T cells [406].
Strategies based on targeting myeloid cells and CAFs
In this section, our primary objective is to elucidate the therapeutic potential associated with specifically targeting myeloid cells in the context of pancreatic cancer (Table 8).
Table 8.
Targeted approach towards myeloid cells and cancer-associated fibroblasts in pancreatic cancer clinical trials
| Strategy | Target/agent | Cellular target | Combination therapy | Participants | Phase | Outcomes* | Status | NCT Identifier/reference |
|---|---|---|---|---|---|---|---|---|
| Targeting immunosuppressive myeloid cells | CCR2, 5/BMS-813160 | M-MDSCs | Nivolumab + Gemcitabine + Nab-paclitaxel | 40 | I, II | Detrmining OS, ORR, PFS, and AEs | Active, not recruiting | NCT03496662 |
| CXCR2/SX-682 | PMN-MDSCs | Nivolumab | 20 | I | Determining maximum tolerated dose, changes in TME composition, PFS, and OS | Recruiting | NCT04477343 | |
| Reprogramming DCs | FLT3L/CDX-301, CD40 agonists/CDX-1140 | DCs | Pembrolizumab + Chemotherapy | 132 | I | Determining safety, tolerability, ORR, clinical benefit rate, duration of response, PFS, OS, pharmacokinetics, and immunogenicity | Completed | NCT03329950 |
| Targeting CAFs | VDR/Paricalcitol | CAFs | Gemcitabine + Nabpaclitaxel | 36 | I, II | Determining AEs, OS, PFS, and response rate | Active, not recruiting | NCT03520790 |
| Nivolumab + Albuminbound paclitaxel + Cisplatin + Gemcitabine | 10 | II |
Grade 3–4 AEs (n = 24): thrombocytopenia (76%), anemia (44%), and colitis (12%) Partial response = 19, stable disease = 2, progressive disease = 2, ORR = 83%, and mPFS/mOS = 8.17/15.3 months |
Active, not recruiting | NCT02754726/[623] | |||
| Inhibition of TGF-β | ATR2/Losartan | CAFs | FOLFIRINOX + Nivolumab + Radiotherapy + Surgery | 168 | II | Determining R0 resection rate, PFS, OS, pathologic complete response, and AEs | Active, not recruiting | NCT03563248 |
| Inhibition of IL-1β | IL-1β/Canakinumab (ACZ885) | CAFs | Spartalizumab + Nabpaclitaxel + Gemcitabine | 10 | I |
No dose limiting toxicities Grade 3/4 AEs: neutropenia (60%) and anemia (50%), pneumonitis (10%), and no fatal AEs (n = 10) Partial response = 3, stable disease = 5, and progressive disease = 2 1-year OS = 60% Activation of CD8+ T cells and increased serum levels of CXCL9/10 in both responder and non-responder patients |
Active, not recruiting | NCT04581343/[423, 624, 625] |
*In cases where the outcomes of the clinical trial have not yet been released, the goals (primary/secondary outcome measures) of the study are mentioned
AEs: Adverse events, ATR2: Angiotensin II receptor, CAF: Cancer-associated fibroblast, DC: Dendritic cell, FLT3L: Fms-related receptor tyrosine kinase 3 ligand, IL-1β: Interleukin-1 beta, M-MDSC: Monocytic myeloid-derived suppressor cell, N/A: Not applicable, ORR: Objective response rate, OS: Overall survival, PFS: Progression-free survival, PMN-MDSC: Polymorphonuclear myeloid-derived suppressor cell, TGF-β: Transforming growth factor-beta, VDR: Vitamin D receptor
Targeting macrophages
Therapies that retrain macrophages to engulf and destroy tumor cells may provide a new approach to treating cancer. Antibodies that stimulate the phagocytic program in macrophages were initially found in pancreatic cancer patients treated with anti-CD40 agonistic antibodies [407]. It was believed that these antibodies only affected macrophages, but further research showed they also improved the function of DCs and T-cell priming [408, 409]. However, a phase II trial found that an anti-CD40 antibody called APX005M (sotigalimab) did not improve clinical outcomes in pancreatic cancer patients, suggesting that its mechanism of action may be different in humans [410]. As mentioned earlier, CD47 is a protein found on cancer cells that prevents them from being engulfed by macrophages. However, blocking CD47 alone does not have a significant effect on some types of solid tumors [411]. Macrophages can be stimulated to have anti-cancer properties, including engulfing cancer cells expressing CD47, by using a specific molecule, CpG oligodeoxynucleotide (an agonist for the TLR9) [412]. The utilization of a specific TLR9 ligand known as K3-SPG for in situ vaccination prompts a durable immune response and enhances the effects of both local and systemic immunotherapy in preclinical models [413], which may be associated with overcoming T-cell exhaustion [414]. Moreover, signal transduction by the CSF-1R in macrophages could be a useful target for improving the immune response in pancreatic tumors and enhancing the effectiveness of immunotherapy. Blocking the CSF1/CSF-1R pathway eliminates TAMs from tumors and reprograms remaining macrophages to enhance anti-tumor immunity. This blockage improves interferon responses, increases infiltration of CTLs, and prevents tumor growth [159]. In a trial investigating the safety and immunologic impact of GVAX in combination with cyclophosphamide, pembrolizumab, and IMC-CS4 (a CSF-1R inhibitor), nine patients were enrolled, with two experiencing severe immune-related side effects (diarrhea and rash). The study reported a median DFS of 12.6 months and OS of 20.4 months, with 78% achieving major pathological response post-surgery. The primary immunologic endpoint was met, with 75% of patients showing a significant increase in CD8+ T cells and granzyme B+ CD8+ T cells following triple therapy. No significant change in myeloid cell density was observed, suggesting macrophages were reprogrammed rather than depleted (NCT03153410) [415].
PDAC is a type of cancer that spreads to the liver with the help of macrophages. The process of macrophages engulfing dead cells, known as efferocytosis, promotes liver metastasis by changing the macrophages. A protein called progranulin in macrophages affects their ability to break down cells, leading to a change in the macrophages and an increase in arginase 1 levels. Blocking efferocytosis or reducing progranulin levels can decrease liver metastasis and enhance the function of CD8+ T cells [416]. Targeting these mechanisms may prevent the spread of PDAC to the liver.
Targeting CAFs
Although previous attempts to target CAFs in PDAC have failed, there is renewed interest in targeting subgroups of fibroblasts or their secreted products. Schwann cells provoke CAFs in the microenvironment of PDAC [417]. Suppressing stromal TGF-βR2 leads to a decrease in IL-6 production from CAFs, which in turn results in diminished STAT3 activation in tumor cells and a reversal of the immunosuppressive environment [418]. Also, In vivo neutralization of TGF-β remodels CAF dynamics, reducing myofibroblasts and promoting interferon-responsive fibroblasts. This enhances anti-tumor immunity and the effectiveness of PD-1 immunotherapy [419].
It is suggested that vitamin D might play a role in inducing a state of rest or inactivity in fibroblasts. A study indicates that the stroma of human pancreatic tumors contains the vitamin D receptor (VDR). Using calcipotriol, a ligand of VDR, as a treatment significantly reduces inflammation and fibrosis in both pancreatitis and tumor stroma. The study demonstrates that VDR plays a crucial role as a transcriptional regulator of PSCs, aiding them to revert to a dormant state. This results in stromal alterations, enhanced intratumoral delivery of the chemotherapy drug gemcitabine, a decrease in tumor size, and a survival rate increase of 57% compared to chemotherapy alone [420]. In light of this finding, two phase II clinical trials are currently in progress for patients with PDAC. The first trial (NCT03520790) aims to combine paricalcitol with gemcitabine and nab-paclitaxel, while the second trial (NCT02754726) seeks to combine nivolumab with gemcitabine, paclitaxel, and cisplatin. These trials are actively recruiting participants to further investigate the potential benefits of these treatment approaches.
IL-1β has multiple effects on the TME, including promoting the development of CAFs. These CAFs, in turn, produce IL-6, which creates an environment that helps tumors evade the immune system and allows tumor cells to survive for longer [231, 421]. Not only immune cells, but PDAC tumor cells themselves can also produce IL-1β. In preclinical studies, blocking IL-1β has shown promising results when combined with blocking PD-1 [422]. A phase I clinical trial (NCT04581343) evaluated the efficacy of combining gemcitabine and nab-paclitaxel with two antibodies—one that blocks IL-1β (canakinumab) and another that blocks PD-1 (spartalizumab). The most common severe AEs (Grade 3/4) included neutropenia (60%) and anemia (50%), with no fatalities. One patient discontinued spartalizumab due to grade 3 pneumonitis. There were 3 confirmed partial responses, 5 patients with stable disease, and 2 patients with disease progression as their best response (n = 10), and a 1-year OS rate of 60% was reported. Both patients who responded and those who did not showed CD8+ T cell activation in peripheral blood and increased serum levels of IFN-induced chemokines CXCL9/10 [423].
CAFs have elevated levels of the protein PIN1. PIN1 promotes several cancer-related pathways by affecting the structure of phosphorylated proteins. Blocking PIN1 with drugs has shown promise in treating cancer. Several small molecule drugs, such as all-trans retinoic acid (ATRA), have been identified as PIN1 inhibitors and have been used to study their functions in cancer development [424, 425]. Clinical trials have shown positive results when combining ATRA with chemotherapy in patients with advanced pancreatic cancer [426]. PIN1 inhibition also reduces the formation of fibrous tissue within tumors and increases sensitivity to chemotherapy drugs. In addition, PIN1 inhibition may enhance the effectiveness of immunotherapy [425, 427]. Animal models have demonstrated reduced tumor growth when treated with a combination of a PIN1 inhibitor AG17724, an antibody against FAPα, and DNA aptamers that recruit specific immune cells [424].
Placental growth factor (PlGF) is a protein expressed mainly in the placenta. Blocking PlGF in animal models of intrahepatic cholangiocarcinoma led to improved survival by decreasing desmoplasia and enriching quiescent CAFs [428]. PlGF also promotes liver fibrosis, tumor angiogenesis, and cancer cell metastasis [429]. In pancreatic cancer, PlGF is upregulated by chemotherapy, leading to the generation of extracellular matrix by CAFs. Combining atezolizumab (an anti-PD-L1 mAb) with PlGF/VEGF inhibition targeting CD141+ CAFs enhances the efficacy of chemotherapy [429].
Reprogramming DCs
The restoration of the expression of peptide-MHC complexes and co-stimulatory molecules is achieved through DC reprogramming. This reprogramming enabled the display of tumor antigens originating within the context of MHC-I, ultimately enhancing the targeted elimination by CD8+ CTLs [430]. Studies in mice have demonstrated that cDCs play a crucial role in initiating immune responses specific to tumors by CD8+ T lymphocytes. However, pancreatic cancer lacks an adequate presence of these cDCs [130, 431]. In comparison to lung adenocarcinoma mouse models, models of PDAC displayed a notable scarcity of CD103+ cDCs. Following treatment with FMS-like tyrosine kinase 3 ligand (FLT3L), which augments the number of intratumoral cDCs, mouse models of PDAC showed renewed sensitivity to CD40 agonist antibody and radiation therapy [431]. Currently, a trial is being conducted to investigate the potential of combining CDX-1140, a CD40 agonist antibody, with FLT3L (CDX-301) in patients with PDAC (NCT03329950).
The activation of DCs can be provoked by the death of tumor cells, and the subsequent ingestion of fragments from these tumor cells also triggers regulatory processes in DCs that hinder their interaction with T cells. By subjecting DCs to microbial products that stimulate TLR signaling, such as pIpC or CpG DNA, which imitate viral nucleic acid, this regulatory function can be bypassed. Hence, innate immune adjuvants are incorporated into vaccination strategies. Additionally, targeted medications that affect the pathways of DNA replication and repair can activate the STING pathway, which subsequently stimulates the production of IFN-I and enhances the activation of DCs [432, 433]. As a result, in animal models of pancreatic cancer, the administration of STING agonists boosts inflammation in the surrounding immune environment and reduces tumor load [434]. Moreover, using PtIV-MSA-2 conjugates containing cisplatin and a STING agonist is effective against pancreatic cancer, leading to increased immune cell infiltration and activation in tumor tissues [435].
The enforcement of expression of the transcription factors PU.1, IRF8, and BATF3 (PIB) is adequate to trigger the cDC1 phenotype. By this reprogramming through PIB, cancer cells are transformed into capable APCs, offering an approach to counteract the strategies employed by tumors to evade immune surveillance. These reprogrammed DCs were capable of presenting endogenous tumor antigens on MHC-I and facilitating targeted killing by CD8+ T cells [430].
Targeting immunosuppressive MDSCs
There is significant synergy between PD-1 blockade and the CD11b agonist as it substantially decreases the accumulation of the majority of myeloid cell types in PDAC mice models [436]. Furthermore, CD11b agonists like GB1275 cause reprogramming of the innate immune system, leading to an enhanced response of pancreatic cancer to immunotherapies [341]. Mice that were administered CCR2 inhibitors specifically aimed at circulating monocytes experienced a reduction in the PDAC tumor load [153]. A study found the ideal dose of the CCR2 inhibitor PF-04136309 to be used alongside chemotherapy in a clinical trial for pancreatic cancer patients. The combination therapy was found to be safe and well-tolerated by the patients. The study also discovered that the inhibitor caused monocytes to accumulate in the bone marrow of patients, leading to a decrease in circulating monocytes and M-MDSCs in the TME. This resulted in significant reductions in the size of the primary tumors [437]. Nevertheless, the combination of nab-paclitaxel/gemcitabine along with PF-04136309 resulted in notable pulmonary toxicity and failed to demonstrate a favorable indication [438]. The signaling of CXCR2 is found to be increased in myeloid cells. The absence of CXCR2 leads to a decrease in metastasis and its inhibition extends the period of survival without tumors in mice. Also, the suppression of CXCR2 improves the infiltration of T cells and makes them more responsive to anti-PD-1 therapy [439].
Gut microbiome in modulating immune checkpoint blockade
Preclinical research on mice with sarcoma, melanoma, and colon cancer revealed that the most effective responses to anti-CTLA-4 and anti-PD-L1 treatments were reliant on the existence of certain species of gut bacteria. This underscores the connection between the gut microbiome and the success of immune checkpoint therapy [440–444]. The connection between certain gut bacteria and the immune response has also been noted in individuals with cancer. These bacteria impact the operation and maturation of immune cells in lymph nodes or within the TME, thus dictating the success of ICB [445]. Specific gut bacteria have been identified to affect immune responses in cancer. For instance, Bacteroides fragilis can trigger TH1 responses and assist in the development of DCs in tumors, enhancing the effectiveness of anti-CTLA-4 treatment. Bifidobacterium can modify the activation of DCs and amplify the activity of CD8+ T cells that are specific to the tumor. Akkermansia muciniphila can augment the penetration of particular CD4+ T cells into tumors and elevate the proportion of effector to regulatory CCR9+CXCR3+CD4+ T cells [22, 440, 441, 446].
The fact that pancreatic cancer tissues contain a unique microbial fingerprint that aids in acquiring cancer characteristics and influences the long-term survival of patients has now been widely accepted and acknowledged [447–449]. Pancreatic cancer progression and the effect of particular treatments have been linked to separated alterations observable in the microbiome of the gut and tumor [442, 450, 451]. For instance, a tryptophan metabolite derived from tryptophan by gut microbiota, known as indole-3-acetic acid (3-IAA), is linked to improved responses to treatment. Alterations in diet or the administration of 3-IAA enhanced the effectiveness of chemotherapy in mouse models of PDAC. This effectiveness is associated with myeloperoxidase, which oxidizes 3-IAA, resulting in an increase in ROS and a decrease in autophagy in cancer cells, thereby inhibiting their proliferation [452]. In another study, the oncogenic mutation KRAS G12D boosts IL-33 production, promoting type 2 immunity in PDAC. The tumor’s mycobiome further increases IL-33 secretion. This IL-33 then recruits and activates TH2 cells and innate lymphoid cells 2 (ILC2s) in the PDAC TME. Remarkably, either deleting IL-33 genetically or administering anti-fungal treatment leads to PDAC tumor regression [453]. This highlights the crucial roles of IL-33 and the tumor’s mycobiome in PDAC progression and potential treatment strategies.
Multiple studies conducted on murine models of PDAC have demonstrated that eliminating the gut microbiome with antimicrobial agents could potentially amplify the susceptibility of tumors to ICIs and diminish the overall burden of tumors [70, 454, 455]. While the majority of the literature concentrates on the potential employment of microbiota-centered interventions together with chemotherapy and ICB, it emerges as plausible that microbiome modulation may also be employed concurrently with CAR T cells, antibody–drug conjugates, and immunotherapies that are yet to be established [456]. A multitude of current research and clinical trials are exploring the possibility of modifying the microbiome to boost the efficacy of ICB. It has been demonstrated that fecal microbial transplantation can enhance ICB outcomes and reduce associated AEs in patients [457–459]. Furthermore, altering the diet is a potential approach to adjust the gut microbiome, and an increasing number of preclinical studies indicate its potential to enhance the response to ICB [460].
A study suggests that the composition of the tumor microbiome in resected pancreatic adenocarcinoma patients plays a significant role in long-term survival. The higher alpha-diversity in the tumor microbiome of long-term survivors and the identified microbiome signature predictive of long-term survivorship highlight the importance of the tumor microbiota in influencing the natural history of the disease. Furthermore, the findings from fecal microbiota transplantation experiments demonstrate the ability to modulate the tumor microbiome and affect tumor growth and immune infiltration, indicating the potential for targeted interventions to improve patient outcomes [459]. Moreover, in individuals with pancreatic cancers, a higher presence of Megasphaera within the tumor has been linked to improved survival rates following anti-PD-1 therapy [461]. Additionally, bacterial elimination in PDAC leads to immune changes, reducing MDSCs, promoting CD8+ T-cell activation, and increasing differentiation of TH1 cells and M1 macrophages. This also enhances immunotherapy effectiveness by increasing PD-1 expression. The PDAC microbiome induces TLRs driven-T cell anergy, suggesting the microbiome’s role in immune suppression and its potential as a therapeutic target [70].
All in all, the association of pancreatic cancer with gut and tumor microbiome in the context of cancer immunotherapy is an interesting research area in treatment, prognosis, and predicting response to immunotherapy. Approaches like modulating the gut microbiome, fecal microbial transplantation, and dietary regimen-oriented interventions might improve the clinical outcome in patients undergoing cancer immunotherapy.
CRISPR/Cas9 and pancreatic cancer immunotherapy
CRISPR/Cas9, a precise gene editing tool, is revolutionizing cancer research and treatment. The combination of CRISPR/Cas9 and cancer immunotherapy may further broaden the application of immunotherapy to more cancer patients, and ongoing clinical trials are using the CRISPR/Cas9 system in immune cells to modify genomes in a target-specific manner. The CRISPR/Cas9 system's ability to create site-specific, highly efficient gene knockout makes it a desirable tool to address long-standing challenges in cancer treatment, such as T cell exhaustion and TME immunosuppression [462, 463]. In the context of pancreatic cancer, there are numerous studies utilizing CRISPR/Cas9 for gene knockout (Fig. 5) [464–468]. The utilization of CRISPR/Cas9 methodology to disrupt the CD73 gene in both human and murine cellular models of pancreatic cancer demonstrated that CD73 inactivation impeded cellular proliferation and motility, leading to a halt in the G1 phase of the cell cycle. Additionally, it was observed that deletion of CD73 hindered the ERK/STAT3 signaling pathway while stimulating the E-cadherin pathway [469]. A study found that mesenchymal-like pancreatic cancer cells are more resistant to immune cell-mediated killing than the parental epithelial-like cells. In this study, the researchers used CRISPR-Cas9 knockout screens to identify the genes involved in this resistance. They discovered several mesenchymal-specific regulators, such as Egfr and Mfge8, that were responsible for inhibiting immune cell function [470]. The application of CRISPR/Cas9 technology to introduce targeted BRCA1/2 mutations enables the reinstatement of olaparib responsiveness in pancreatic cancer cells [471]. Apart from potential challenges and limitations of CRISPR/Cas9 such as off-target toxicity, Cas9-related immunogenicity, and off-target mutations, these studies highlight the useful application of this genome editing tool in the context of pancreatic cancer immunotherapy.
Fig. 5.
The emerging role of genome editing technology CRISPR/Cas9 in pancreatic cancer treatment. Utilizing CRISPR/Cas9 technology, autologous T cells are genetically modified to eliminate or alter genes that contribute to T cell exhaustion or resistance to immunotherapy. Once modified, these cells are reinfused into the patient, effectively improving the eradication of pancreatic ductal adenocarcinoma (PDAC) cells. TAA: Tumor-associated antigen; TCR: T cell receptor; TSA: Tumor-specific antigen
Potential strategies improving efficacy of pancreatic cancer immunotherapies
Combination therapy
Pancreatic cancer presents a range of mechanisms that resist immunotherapy. Therapies that target only one mechanism have not yielded successful results. The research proposes the optimization of the benefits of current agents through logical combinations. The suggested approach for advanced immunotherapy for PDAC involves combinations that amplify immune activation, inhibit immune checkpoints, improve the TME, and are compatible with conventional cytotoxic therapy [472, 473]. There are a plethora of combination therapies for cancer immunotherapy of PDAC (Tables 5, 7, 8, 9). For example, BMS-687681, which acts as a dual antagonist for CCR2/5, was used in conjunction with anti-PD-1 and radiotherapy. The results indicated an increase in the infiltration of intratumoral effector and memory T cells, while simultaneously observing a decrease in the infiltration of Tregs, M2 TAMs, and MDSCs [474]. Overall, combination therapies could improve the efficacy of immunotherapies due to providing a potential for synergistic effects.
Table 9.
Key combination therapies of PDAC
| Immunotherapeutic strategy | Immunotherapeutic agents | Combination therapy | Study design | Results | References |
|---|---|---|---|---|---|
| Cancer vaccine and ICB | GVAX cancer vaccine and nivolumab | Urelumab (anti-CD137 [4-1BB] agonist) | Murine syngeneic model of metastatic PDAC |
Activated, effector memory T cells ↑ Conversion of T cells from an exhausted status to an activated status ↑ Expression of costimulatory molecules CD137 and OX40 on T cells ↑ Expression of IFN-γ in EOMES+ exhausted TILs Survival ↑ |
[626] |
| ICB | Anti-PD-1 antibody | Az (a histamine receptor H1 antagonist) |
Pancreatic cancer cell specimens from PDAC patients Orthotopic models |
Upregulation of MHC-I expression in tumor cells via cholesterol biosynthesis signaling ↑ CD8+ CTLs cell penetration and efficacy ↑ Resistance to ICB ↓ |
[627] |
| ICB | Anti-PD-1 antibody | Anti-IL-8 antibody | Humanized mouse model of PDAC |
Anti-tumor activity of anti-PD-1 ↑ Number of infiltrating granulocytic myeloid cells in TME ↑ Type I interferon production by infiltrating granulocytic myeloid cells ↑ |
[628] |
| ICB | Anti-PD-1 antibody | Focal adhesion kinase inhibitor (FAKi), PEGylated recombinant human hyaluronidase (PEGPH20), and anti-CXCR4 antibody | Murine liver metastasis syngeneic model of PDAC |
Survival ↑ T-cell infiltration ↑ Number of effector memory T cells ↑ Number of MDSCs ↓ Number of CXCR4-expressing myeloid cells (granulocytes) ↓ Liver metastasis ↓ |
[484] |
| ICB | Nivolumab | GVAX cancer vaccine, radiotherapy, and BMS-687681 (dual antagonist of CCR2 and CCR5) | PDAC mouse models |
GVAX and nivolumab-induced CCR2/CCR5 expression Intratumoral effector and memory T cell infiltration ↑ Expression of CCL17/CCL22 chemokines ↑ Infiltration of Tregs, M2-like TAMs, and monocytic-MDSCs ↓ Expression of immunosuppressive CCL2/CCL5 chemokines ↓ Anti-tumor efficacy and survival ↑ No improvement of anti-tumor activity by adding GVAX |
[474] |
| ICB | Anti-PD-1 antibody (clone RMP1-14) | Gemcitabine-conjugated polymer (PGEM; a STING agonist) loaded with PF-6309 (a CCR2 antagonist) | Pancreatic tumor spheroid model and orthotopic tumor model |
Pancreatic tumor burden ↓ Sensitization of PDAC tumors to anti-PD-1 therapy ↑ Providing anti-tumor immunity through reversing the CCL2/CCL7-mediated immunosuppression |
[629] |
| ICB | Anti-PD-1 and anti-CTLA-4 | IACS-8803 (a STING agonist) | Orthotopic Kras+/G12D TP53+/R172H Pdx1-Cre (KPC) derived models PDAC |
Inflammatory remodeling of the PDAC stroma ↑ Survival ↑ Sensitivity to ICB ↑ |
[495] |
CTL: Cytotoxic T lymphocyte, CTLA-4: Cytotoxic T-lymphocyte associated protein 4, ICB: Immune checkpoint blockade, MDSC: Myeloid-derived suppressor cell, PD-1: Programmed cell death protein 1, PDAC: Pancreatic ductal adenocarcinoma, STING: Stimulator of interferon genes, TAM: Tumor-associated macrophage, TIL: Tumor-infiltrating lymphocyte, Treg: Regulatory T cell, ↑: Increase, ↓: Decrease
Costimulatory molecule agonists
CD40 activation and using CD40 agonists are a novel clinical opportunity for cancer immunotherapy [407, 475–478]. There are several agonistic anti-CD40 antibodies, such as SGN-40, SEA-CD40, selicrelumab, APX005M, CDX-1140, and ADC1013, applicable in clinical trials [477]. In a phase I clinical trial, the combination of an agonistic anti-CD40 antibody and gemcitabine for treating PDAC was tested. The treatment showed only a slight effect, but its safety was confirmed [479]. The combination of the CD40 agonist and gemcitabine could potentially overcome resistance to anti-PD-1/CTLA-4 therapy by increasing the accumulation of CD8+ T cells that fight against tumors in PDAC [480]. A study found that selicrelumab (an agonist CD40 antibody) significantly altered the TME in PDAC patients. Selicrelumab-treated tumors were enriched with T cells (82%) compared to untreated (37%) and chemotherapy/chemoradiation-treated tumors (23%). Additionally, selicrelumab reduced tumor fibrosis, decreased M2-like tumor-associated macrophages, and matured intratumoral DCs. The treatment had an acceptable toxicity profile and resulted in an overall survival of 23.4 months [475]. Moreover, a study demonstrates that using a nanofluidic drug-eluting seed (NDES) for sustained, low-dose intratumoral delivery of CD40 monoclonal antibody can alter the TME and reduce tumor size in mouse models of PDAC [478]. These findings elucidate the therapeutic mechanisms of CD40 targeting and modification of the TME in pancreatic cancer, aiming to enhance the effectiveness of immunotherapies against cold tumors like PDAC.
Neutralizing tumor acidity
Acidosis plays a significant role as an immunosuppressive mechanism that contributes to the proliferation of PDAC and immune escape [481]. A study investigates the application of L-DOS47, a urease immuno-conjugate, for the purpose of neutralizing the acidity of tumors and enhancing the response to immunotherapy. L-DOS47 attaches to CEACAM6, a protein that is predominantly present in gastrointestinal cancers, and increases the local pH by breaking down urea into two NH4 + and one CO2. This was experimented on a model of pancreatic tumors in mice, and it was observed that L-DOS47 elevated the extracellular pH of the tumor. When L-DOS47 was used in conjunction with anti-PD-1, it significantly boosted the effectiveness of the monotherapy, leading to a reduction in tumor growth for a duration of up to 4 weeks [482]. This study paves the way for using L-DOS47 in future clinical trials.
Targeting desmoplastic barriers of TME
A significant obstacle to the effectiveness of cancer immunotherapies in PDAC is the presence of desmoplastic barriers within the stromal ECM, such as hyaluronan. These mechanical barriers encapsulate the tumor cells, thereby restricting their exposure to immunotherapeutic agents. The targeted removal of hyaluronan in a mouse model of PDAC resulted in better vascular permeability and enhanced drug delivery. This led to increased effectiveness of chemotherapy when combined with the cytotoxic chemotherapy drug, gemcitabine [483]. A study found that combining PEGPH20 (a PEGylated recombinant human hyaluronidase), focal adhesion kinase inhibitor, and anti-PD-1 antibody treatments improved survival in PDAC-bearing mice, increased T-cell infiltration, altered T-cell phenotype and metabolism, reduced granulocytes, and decreased CXCR4-expressing myeloid cells. Additionally, adding an anti-CXCR4 antibody significantly reduced metastatic rates in a PDAC liver metastasis model [484].
Innate immune activation
A useful strategy in combating pancreatic cancer involves the stimulation of the body’s innate immune system to bolster its anti-cancer defenses. This is accomplished by, for instance, utilizing a genetically altered version of Listeria monocytogenes, which is engineered to produce MSLN. Alongside this, a vaccine named GVAX is used. The synergistic effect of this combination has been shown to enhance the survival rates of patients [41, 378, 485]. This therapeutic approach transforms PDACs into a state that is more receptive to immune responses. This change is marked by a rise in T cell infiltration and the formation of tertiary lymphoid clusters within the tumors [380], likely converting the cold tumor into the hot tumor. Innate immune cells use cGAS to trigger inflammatory signals when they bind to the pathogen or damage-related molecular patterns (PAMPs/DAMPs). This process leads to the production of cGAMP and the activation of STING, a protein in the endoplasmic reticulum, which then promotes cellular gene programs resulting in the production of IFN-I [486, 487]. IFNs-I (IFN-α and IFN-β) are essential for the development of CD8+ T cells that fight against tumors. Tumors that are inflamed with T cells, often referred to as “hot” tumors, have been linked to a transcriptional signature of type 1 interferon [488, 489]. The activation of STING, either systemically or within the tumor, through STING agonists, has been shown to reverse immune-suppression and cause tumor shrinkage in various preclinical cancer studies [434, 490–494]. IACS-8803 (a STING agonist) increases sensitivity to anti-PD-1 and anti-CTLA-4 immunotherapy in the orthotopic PDAC models [495]. Furthermore, a STING agonist called IMSA101 boosts CAR T cell function in a mouse pancreatic tumor model, which is facilitated through STING agonist-induced IL-18 secretion [496]. All in all, the STING innate immune sensing pathway, when activated, could potentially transform tumors lacking T cell infiltrates into tumors with infiltrating T cells, and thus offers a promising target in PDAC immunotherapy.
TME-modulating agents
A successful immunotherapy for PDAC usually requires combining different treatments to help T cells infiltrate and stay activated in the hostile TME. Current research is focused on developing strategies to improve the PDAC TME, boost the immune response, and enhance the effectiveness of T cell therapy [44, 497].
ADH-503 is a small molecule that binds to CD11b and enhances the adhesion of myeloid cells, inhibiting their migration into tissues [498]. It also shifts TAM polarization to an anti-tumor phenotype, improves survival in PDAC-bearing mice, and sensitizes PDAC tumors to anti-PD-1/PD-L1 immunotherapy [341]. In clinical trials, ADH-503 was well tolerated with common side effects, but no clinical responses were observed in pancreatic cancer patients (NCT04060342) [499].
In PDAC, it is expected that targeting the CCL2/CCR2 pathway would help to reduce the accumulation of TAMs in the TME. A CCR2 inhibitor, PF-04136309, was tested in combination with chemotherapy in patients with pancreatic cancer. A manageable level of safety was observed. Early correlational studies indicated a decrease in TAMs and an increase in TILs (NCT01413022) [437]. Another trial combining PF-04136309 with chemotherapy in pancreatic cancer patients showed high rates of lung toxicity and no significant improvement in efficacy (NCT02732938) [438]. A study tested the CCR2 antagonist CCX872-B in combination with FOLFIRINOX for patients with advanced pancreatic cancer. Analysis showed an OS rate of 29% at 18 months with no safety concerns (NCT02345408) [500]. BMS-813160 is a dual antagonist for CCR2 and CCR5 that is being tested in combination with chemotherapy or immunotherapy in patients with advanced pancreatic or colorectal cancer, but no results from the study have been reported yet (NCT03184870) [501].
Several studies have shown that by inhibiting the CXCR4/CXCL12 axis, the PDAC TME can be modified. For example, when CXCR4 was knocked down, the invasion potential of pancreatic cancer cells in vitro was decreased. Treating fresh human PDAC slices with a combination of PD-1 and CXCR4 blockade resulted in enhanced tumor cell death and lymphocyte expansion into the juxtatumoral compartment [502]. In a mouse model of PDAC, administering the CXCR4 inhibitor AMD3100 (plerixafor) led to the accumulation of T cells among cancer cells, resulting in a synergistic tumoricidal effect when combined with anti-PD-L1 immunotherapy [35]. A trial tested AMD3100 in patients with colorectal and pancreatic cancer, resulting in decreased tumor markers (circulating tumor DNA and IL-8) and changes in immune cells (reduced number of CAFs and increased number of effector TILs/NK cells) [503–505]. Motixafortide (BL-8040; CXCR4 antagonist) is a synthetic peptide that is administered subcutaneously and has shown promising results in combination with pembrolizumab in treating metastatic PDAC, increasing CD8+ T cell infiltration and decreasing MDSCs and circulating Tregs (NCT02826486) [506]. NOX-A12 (olaptesed pegol) is a PEGylated drug that inhibits CXCL12 and enhances the activity of anti-PD-1 therapy in pre-clinical models [507]. In a phase 1/2 study with advanced PDAC patients, NOX-A12 in combination with pembrolizumab led to induced TH1 cytokines, prolonged stable disease, and increased effector immune cells in tumor biopsy tissue (NCT03168139) [508].
Pharmacologic inhibition of the A2A adenosine receptor enhances the effectiveness of anti-PD-1 therapy [509]. Several anti-CD73 therapeutics and adenosine receptor inhibitors have been developed [510]. Oleclumab (MEDI9447), a monoclonal antibody that targets CD73, demonstrated positive outcomes in inhibiting tumor progression and promoting immune cell infiltration in colon cancer models. When used in conjunction with anti-PD-1 treatment, it resulted in the elimination of tumors in 60% of animal subjects [511]. Clinical studies of oleclumab, either alone or in combination with durvalumab, in patients who did not respond to anti-PD-L1 therapies like advanced pancreatic cancer revealed good tolerability and some partial responses (22 and 28 months) in a small subset of patients (2/73; NCT02503774) [512]. A study evaluated the safety of quemliclustat (a small molecule inhibitor of CD73) in combination with standard treatment and zimberelimab in patients with metastatic PDAC. The safety profile is similar to single agents, with no new toxicities. Some patients showed partial responses with long-lasting effects (NCT04104672) [513].
Main challenges ahead of pancreatic cancer immunotherapy
Immunotherapy for pancreatic cancer is a significant therapeutic strategy. However, despite comprehensive studies, there are obstacles in translating research outcomes and determining the best therapeutic combinations. These challenges necessitate a joint effort from scientists and medical practitioners to deepen our comprehension of the interactions between cancer and the immune system and to enhance the treatment choices available to patients. In this part, we will explore in greater detail the chief hurdles facing immunotherapeutic strategies for pancreatic cancer.
Low antigenic strength and number of neo-antigens
During the process of tumor development, non-synonymous gene mutations occur, leading to the generation of neo-antigens that are exclusively expressed by tumor cells. Pancreatic cancers carry a moderate load of these non-synonymous neo-antigenic mutations [497]. In essence, PDACs show a low load of neo-epitopes; therefore, the tumors are more likely to adapt to immune pressure and escape T cell-mediated killing through cancer immunoediting. In a study, T cell immunity was assessed in a mouse model of pancreatic cancer, revealing a low level of mutations, no anticipated neo-epitopes resulting from these mutations, and resistance to respond to checkpoint immunotherapy [514]. Also, pancreatic tumors that have the greatest quantity of neo-antigens and the highest concentration of CD8+ T cell infiltrates are linked to the longest survival rates in patients. Moreover, enrichment of neo-antigens in the tumor antigen MUC16 (CA125) was observed in long-term survivors of pancreatic cancer [53]. Thus, the quality of neo-antigens as a biomarker for PDAC could potentially steer the use of immunotherapies [53, 515]. Additionally, the BCL2A1 neo-epitope is presented as a potential target for personalized immunotherapy, which stimulates CTLs to combat pancreatic cancer cells [516]. All in all, neo-antigens might broaden the horizon towards personalized immunotherapy of pancreatic cancer. A major challenge restricting their applicability, however, is that a low number of neo-antigens is rarely shared among patients [497], making the use of relevant treatment approaches cumbersome and costly.
Primary, adaptive, and acquired resistance
In primary resistance, there can be instances where cancer does not respond to immunotherapy, potentially due to adaptive immune resistance mechanisms. Adaptive resistance pertains to a resistance strategy where the cancer, even though identified by the immune system, shields itself by adjusting to the immune attack. Lastly, acquired resistance refers to a situation where a cancer initially shows a response to immunotherapy, but after a certain period, it experiences a relapse and advances [517]. From a biological perspective, resistance to immunotherapy can be linked to both intrinsic factors in tumor cells and extrinsic factors associated with TME like ECM and stroma-derived factors, immune cells/factors, and intratumoral microbiota. Tumor cell-intrinsic factors include genetic/epigenetic defects, IFN-γ signaling, lack of neo-antigens, oncogenic signaling pathways, and epigenetic reprogramming [22]. FAP+ CAFs hinder the anti-tumor activity of T cells in pancreatic cancer. However, directing therapies towards these FAP+ subtypes improves the tumor’s response to anti-PD-L1 [35]. T-cell exclusion is a process that resists immune checkpoint therapy, and it’s particularly noticeable in ‘cold’ tumors like pancreatic cancer, which have a low presence of T cells in TME. Some cancer-causing pathways might allow tumors to use this method to avoid the immune system. For instance, the activation of Wnt/β-catenin within the tumor cells has been demonstrated to result in the exclusion of T cells from the TME [518, 519]. Moreover, PTEN deficiency is linked to provoke PI3K-AKT pathway signaling and is connected to decreased presence of CD8+ T cells and unfavorable clinical outcomes from immunotherapy [520]. A recent study found that the loss of interferon regulatory factor 6 (Irf6) leads to resistance to immunotherapy, and its re-expression improves immunotherapy responses to PDAC [521]. All in all, the resistance to immunotherapy in pancreatic cancer is complex and influenced by both internal and external factors within the tumor. A sophisticated strategy of basic and translational/clinical research is needed to understand these mechanisms and identify tumor-specific resistance patterns.
Immune-related adverse events (irAEs)
The irAEs are diverse and can affect any organ. Different immunotherapy regimens have unique toxicity patterns like CRS and neurologic toxicities, making understanding their mechanisms crucial [522–525]. However, a positive association exists between the occurrence of non-lethal irAEs and the response to ICB [526]. Mild (Grade 1–2) effects are observed in over 90% of patients, whereas severe (Grades 3–5) effects can occur in 20–60% of patients [527]. Studies highlight the role of certain immune cells such as CD8+ tissue-resident memory T cells and neutrophils, and cytokines like IFN-γ and IL-6 in causing immunotherapy-induced colitis [528–530]. Other mechanisms like loss of self-tolerance, molecular mimicry, and inflammation also contribute to irAEs. For example, in myocarditis, autoreactive T cells targeting specific peptides are activated, a process worsened by the release of self-antigens from dying tumor cells [531]. This is known as epitope spreading. Glucocorticoids are the main treatment for non-endocrine irAEs, and hormonal therapy is used for endocrine disorders. Intravenous immunoglobulins, plasma exchange, and monoclonal antibodies such as infliximab are employed for neurological, hematological, and persistent irAEs [22]. For ICB-induced colitis, fecal microbiota transplantation is utilized [532]. All in all, irAEs are life-threatening reactions that deserve special attention. Thus, it is crucial to formulate personalized strategies for patient categorization and potential biomarkers to investigate the dynamics and resolution timing of irAEs to identify.
Scarcity of robust predictive biomarkers of response and toxicity
Individual biomarkers have been utilized to forecast responses to cancer immunotherapy. Both Microsatellite Instability-High (MSI-H) and Tumor Mutational Burden (TMB) have been associated with enhanced responses to ICB [533, 534]. Nonetheless, the efficacy of TMB as the only biomarker is restricted, as a low TMB can still elicit effective responses, and a high TMB does not assure a response to ICB [22]. Although immune-related biomarkers such as PD-L1, interferon signature, and TIL density have been found to have restrictions when used as the only biomarkers, it is important to note that even though there is an association between PD-L1 expression and improved outcomes in certain types of tumors, substantial responses can still be observed in tumors that do not express PD-L1 [535]. It has been proposed that CAFs, microbiomes, and exosomes derived from tumors could serve as potential biomarkers for tracking the response to immunotherapy in pancreatic cancer [536]. Furthermore, a study on pancreatic cancer patients who received PD-1 inhibitor-based therapies showed that a lower neutrophil-to-lymphocyte ratio predicted better tumor response [537]. Collectively, the progression of predictive biomarkers has been obstructed by the intricate interplay within the pancreatic tumor microenvironment. The field must comprehend these interactions and establish suitable assays for successful biomarker development and codifying combinatorial biomarker strategies. These approaches should be confirmed in upcoming clinical trials.
Lack of integrated regulatory endpoints for cancer immunotherapy
Conventional methodologies for evaluating the efficacy of cancer immunotherapies, such as pembrolizumab and nivolumab, have demonstrated considerable utility [538]. These methodologies encompass metrics such as ORR, PFS, and OS, which have been instrumental in assessing therapeutic outcomes. Nevertheless, these conventional metrics exhibit limitations when applied to the evaluation of cancer immunotherapies. The primary objective of cancer immunotherapy is to induce a durable response and prolong survival, optimally quantified by examining the ‘tail’ of survival curves. However, the extant methodologies for this measurement are deficient [539]. In circumstances where an immunotherapy’s impact takes time to manifest and the rate of successful outcomes is not high, conventional benchmarks such as mPFS and median OS can provide deceptive early indications [473]. This becomes evident in the case of patients with MSI-H PDAC who underwent treatment with pembrolizumab. Despite a relatively low ORR of 18% and a mPFS of just 2.1 months, the responses proved to be quite durable, with a median response duration of 13.4 months [540]. All in all, continued collaboration and optimization of pancreatic cancer immunotherapy endpoints is needed to address the aforementioned issues.
Lack of proper preclinical animal models
Preclinical models are crucial in cancer drug discovery for prioritizing targets and studying various aspects of treatment. These models have contributed to significant discoveries in cancer treatment and immunotherapy, including the effects of CTLA-4 and PD-L1/PD-1 blockade [539]. Nevertheless, the models commonly utilized do not always accurately represent the immune biology of human cancers [541]. This can be attributed to inter-tumoral and intra-tumoral heterogeneity, recapitulation of TME, and serial passaging of tumor cells [542]. For instance, the intricate interplay between tumor and stroma, along with the diverse traits of stromal elements, present substantial obstacles in accurately reproducing the pancreatic cancer microenvironment. Furthermore, the weak immunogenicity and the immunosuppressive characteristics of PDAC complicate preclinical modeling [542]. A problem with frequently utilized preclinical models is their dependence on the inoculation of cancer cell lines. The tumors that develop following this insertion often fail to accurately reproduce the immune context of the tumor, which plays a crucial role in shaping the immune response in human cancers [543]. Moreover, cancer in humans is thought to have evolved over the years, shaping its interaction with the immune response. Genetically engineered mouse models, developed by altering genes and inducing mutations, best represent this disease [544]. However, these models do not mimic the gradual mutation accumulation seen in human cancers, resulting in stable cancers that do not respond well to cancer immunotherapy [539]. Also, a significant obstacle in current attempts to comprehend the occurrence of irAEs is the absence of suitable preclinical animal models. There is a pressing need for the generation of animal models that accurately mimic irAEs, which would facilitate the detailed study of irAEs associated with pancreatic cancer immunotherapy [545]. Thus, there is an unmet need to further develop pancreatic cancer animal models with high-throughput techniques for better mimicking the human pancreas cancer features. It can pave the way for a rapid translation of preclinical findings into clinical settings.
Conclusion and future directions
In summary, the paradigm shift brought about by immunotherapy is fundamentally altering our understanding of cancer treatment. This groundbreaking approach is now being implemented in clinical settings for a variety of solid cancers. Standard therapies have proven ineffective for patients with PDAC, but immunotherapy has demonstrated encouraging results in preclinical stages. Despite these promising results, immunotherapies still face fundamental challenges, which may limit their efficacy in clinical contexts. It is important to consider that each treatment modality has its advantages and disadvantages (Table 10). This article explored a wide range of immunotherapies, such as OVT, and adoptive cell transfer therapies including TCR-engineered T cells, CAR T-cell therapy, CAR NK cell therapy, and CIK cell therapy. Additionally, ICB, immunomodulators, cancer vaccines, and strategies targeting myeloid cells were discussed as potential avenues. Furthermore, this article provided the application of CRISPR/Cas9 technology and gut microbiome in pancreatic cancer immunotherapy. Lastly, strategies for enhancing the effectiveness of immunotherapy and the primary obstacles confronting pancreatic cancer immunotherapy were highlighted.
Table 10.
Advantages and disadvantages/challenges of cancer immunotherapies
| Treatment modality | Strategy | Advantages | Disadvantages/challenges | References |
|---|---|---|---|---|
| Oncolytic Virus Therapy |
Inducing innate and tumor-specific adaptive immune responses Synergistic effects with whole host of other immunological agents (combination therapy) A versatile gene expression platform for the delivery of therapeutic genes |
Poor efficiency in cold tumors Body’s anti-viral immune responses and neutralizing antibodies against oncolytic viruses Potential off-target infections Inadequate tumor cell tropism and transduction Lack of robust predictive biomarkers for patient selection to receive the treatment |
[630–633] | |
| Adoptive Cell Transfer Therapy | TIL therapy |
Different T cell clones (polyclonal) to overcome tumor heterogeneity Better clinical efficacy in tumors with high mutation load A predominantly phenotype of effector memory T cell with cytotoxic functions expressing chemokine receptors for better homing at tumor tissues Low off-target toxicity |
Tissue collection is cumbersome, expensive, and time-consuming Adverse events like thrombocytopenia, anemia, and febrile neutropenia High dose IL-2-induced toxicities. IL-2 is used to improve the in vivo survival and function of TILs Need to have sufficient quality and quantity of TILs for suitable therapeutic response |
[634, 635] |
| TCR therapy |
Large repertoire of targetable tumor antigens Lower epitope density is required to induce T cell activation High avidity of TCR-T cells allows for eradicating several antigen-presenting tumor cells Allogeneic approaches and deleting both endogenous TCRα/β chains help the production of universal T cells to avoid the risk of GVHD |
Choice of target antigen in terms of expression and immunogenicity Sensitization of TME for TCR therapy Unresponsiveness of T cells toward tumor antigen, reduced T cell fitness, short persistence of adoptively transferred T cells, and rapid exhaustion of engineered T cells On-target/Off-target toxicities and toxicity prediction Primary and secondary resistance to therapy Restriction to HLA alleles presenting the tumor epitope |
[269, 581, 636–638] | |
| CAR T cell therapy |
Rapid manufacturing, short treatment time, and usually a single infusion administration T cell activation in a HLA-unrestricted manner Promising outcomes in aggressive form of tumors like GBM and hematological malignancies Binding to the target with high affinity, T cell expansion, and generating memory T cells Available in several platforms like dual specific with many modifications and combinatorial therapies for better efficacy Existence of an acceptable range of antigenic targets |
Antigen escape On-target off-tumor and increased risk of autoimmunity Limited CAR T cell trafficking and tumor infiltration Limited efficacy due to immunosuppressive TME CAR-T cell-associated toxicities like CRS and neurotoxicity Short-term persistence of CAR T cells in vivo GVHD incidence |
[273, 582, 587, 639–641] | |
| CAR NK cell therapy |
Robust response due to various activating receptors Low off-target response due to various inhibitory receptors Low risk of GVHD and CRS Allogeneic (haploidentical) NK cells can be used Provoking an ADCC-oriented cytotoxicity Off-the-shelf therapeutics and administration without HLA matching Recognizing the target in a HLA-unrestricted manner |
Low rate of circulating NK cells NK cell expansion ex vivo is limited and tight Tight conditions in freezing and storage iPSC-derived NK cells often express low levels of endogenous CD16, leading to lower ADCC |
[106, 642, 643] | |
| CIK cell therapy |
HLA-unrestricted tumor-killing activity A heterogeneous population of CD3+ T lymphocytes that can kill heterogeneous tumor cells Applicable with many combinatorial treatment regimens Limited ability to induce GVHD There is no requirement for cellular selection during or after expansion in product manufacturing It has a remarkably safe profile |
Limited understanding of pharmacological mechanisms Limited availability of CIK cell therapy in certain regions Heterogeneity of CIK cells makes it challenging to standardize CIK cell therapy |
[327, 644] | |
| ICT |
Ability to conjugate with a variety of molecules like drugs and nanoparticles Available in several platforms like BsAbs, ScFvs, triabodies, and so on Combinatorial therapy with two or three different immune checkpoint inhibitors or other therapeutic agents Reinvigorating exhausted T cells/NK cell |
Resistance to ICT Development of irAEs Failure of all patients to respond to the treatment The need for predictive biomarkers for optimal patient selection for gaining better clinical outcomes |
[22, 517, 522–524, 527, 645–647] | |
| Cancer Vaccine | Whole tumor cell vaccine |
Containing a vast range of immunogenic epitopes Presenting all potential tumor antigens to immune system Providing a polyvalent anti-tumor immune responses No need for antigen selection during vaccine design |
Poor immunogenicity No standardized dosing regimens |
[648, 649] |
| DC vaccine |
The possibility of loading with a wide range of antigens like tumor lysate, peptides, RNA, DNA, neo-antigens, and viral antigens Promising safety, tolerability, and immunogenicity profile Producing a wide range of cytokines and chemokines by DCs Deploying a wide range of mechanisms by DCs, like cross-presentation, cross-dressing, antigen transfer, and MHCII-restricted presentation |
Inefficient injected DCs migration and homing to lymph nodes Poor efficiency in immunosuppressive TME Optimization of in vitro culture and cytokine cocktails to achieve high-quality DCs Choosing the delivery route of the DC vaccine Selecting antigens and loading strategy for peptide-pulsed DCs in order to overcome tumor heterogeneity Need for effective combination therapies in advanced stages |
[650–652] | |
| Peptide vaccine |
Triggering a more focused immune response against immunodominant epitopes Containing both CD8+ T epitopes and CD4+ T cell epitopes Low risk of autoimmunity Direct presentation on MHC (short peptides) |
Poor immunogenicity Peptide length-dependent vaccine efficacy Immunogenicity differences among different recombinant protein subunits Issues related to HLA restriction Need for appropriate adjuvants |
[649, 652] | |
| Viral and bacterial vector vaccine |
High immunogenicity and long-lasting immune responses Self adjuvanticity Long-lasting transgene expression |
Potential for vector immunogenicity Specialized storage conditions Safety concerns related to oncogene activation in host’s cells and insertional mutagenesis |
[652, 653] | |
| RNA vaccine |
Inducing both humoral and cellular immune responses Providing systemic immunity against metastatic tumors Inducing long-term immunological memory Offering a personalized immunotherapy Lack of accidental infection and insertional mutagenesis Rapid translation of mRNA encoding TSA, TAAs, or personalized neo-antigens into protein in cytoplasm |
Storage conditions and stability Issues related to design, production, and cost Lack of extensive preclinical and clinical trials Limited efficacy due to tumor heterogeneity and immunosuppressive TME Choosing the best vaccine administration route Need for biomarkers for monitoring the treatment response |
[654, 655] | |
| DNA vaccine |
Inducing both humoral and cellular immune responses Delivering multiple antigens simultaneously in the same construct at the same time Safety, stability, easy storage, and fast manufacturing/modifying on a large scale Providing native structure of protein |
Limited migration into the cell nucleus due to extracellular and intracellular barriers Limited immunogenicity in clinical trials Integration of DNA into host genome Possibility for autoimmune reactions |
[652, 656] | |
| Stem cell-based vaccine |
Potential candidate to limit the chances of immune evasion due to shared epitopes between iPSCs and cancer cells Easy production at a short time Potential for triggering a pro-inflammatory profile in TME |
Ethical constraints Limited studies and a need to further prove the results |
[405] |
ADCC: Antibody-dependent cellular cytotoxicity, BsAbs: Bispecific antibodies, CAR: Chimeric antigen receptor, CIK: Cytokine-induced killer, CRS: Cytokine release syndrome, DC: Dendritic cell, GBM: Glioblastoma, GVHD: Graft-versus-host disease, HLA: Human leukocyte antigen, ICT: Immune checkpoint therapy, iPSCs: Induced pluripotent stem cells, irAEs: Immune-related adverse events, MHC: Major histocompatibility complex, NK: Natural killer, ScFvs: Single-chain variable fragments, TAAs: T cell receptor, TCR: T cell receptor, TILs: Tumor-infiltrating lymphocytes, TME: Tumor microenvironment, TSAs: Tumor-specific antigens
The complex nature and heterogeneous composition of cellular elements in the pancreatic tumor microenvironment are of significant importance. There is a complex transition of cell populations as PDAC advances. Employing advanced methods such as single-cell sequencing and multi-omics analysis allows us to delve deeper into the immune cell profile in PDAC, pinpoint cells with higher precision, and chart the single-cell trajectories [546]. This advancement lays a stronger groundwork for developing immunotherapies that target the various elements of the TME.
As we progress in developing immunotherapeutic strategies for the treatment and management of PDAC, it is crucial to prioritize efforts that enhance patients' quality of life. Numerous trials employing immunotherapy in PDAC have had disappointing outcomes, primarily due to the immunosuppressive TME. Therefore, it is imperative to refine and improve existing immunotherapies to effectively address this significant challenge. Furthermore, it is essential to conduct further research on the efficacy of novel immunotherapy targets identified in preclinical studies, thereby validating their potential through human clinical trials. Overall, the open-ended research question remains unanswered as to why many patients with pancreatic cancer do not respond to immunotherapies.
The identification of novel and appropriate molecular targets for targeted immunotherapies is crucial for the success of this immunotherapy in treating pancreatic cancer. While CAR-based therapies have achieved impressive clinical responses in targeting cancer antigens, the efficacy of these therapies in solid cancers has been disappointing, in part due to antigen escape. Targeting heterogeneous pancreatic tumors with immunotherapies will require the identification of novel tumor-specific targets. Therefore, identifying novel and appropriate molecular targets for CAR T cell therapy is essential for the development of effective cancer treatments.
As our understanding of the complex interplay between the immune system and pancreatic cancer continues to evolve, the field of pancreatic cancer immunotherapy is positioned at the forefront of cutting-edge research. These groundbreaking domains of research, such as machine learning and artificial intelligence [547], mutant KRAS peptide-driven vaccines and personalized RNA neo-antigen vaccines [401, 548], single-cell multi-omics-oriented approaches [546, 549], and CRISPR/Cas-based RNA editing [550], are of utmost importance as they define the active research areas of the future, paving the way for gaining better clinical outcomes. Given the role of artificial intelligence in cancer research, researchers used machine learning to analyze complex tumor molecular data from pancreatic cancer patients and found that anti-CD40 therapy reduced T-cell exhaustion in the TME. They identified specific T-cell populations that correlated with improved DFS following anti-CD40 therapy, demonstrating the potential of machine learning in pancreatic cancer immunology research [547]. The creation of multiplexed effector guide arrays (MEGA) has made it possible to effectively control and regulate the T cell transcriptome through the use of CRISPR-Cas13d. With MEGA, genes can be suppressed in primary human T cells without any changes to the DNA, leading to improved T cell function and stronger anti-tumor capabilities. MEGA also enables the regulation of CAR activation and disruption of immunoregulatory metabolic pathways [550], providing a flexible and powerful tool for use in pancreatic cancer immunotherapy.
Acknowledgements
We apologize to the many authors and colleagues whose works are not cited.
Abbreviations
- AEs
Adverse events
- APC
Antigen-presenting cell
- ATRA
All-trans retinoic acid
- Breg
Regulatory B cell
- BsAbs
Bispecific antibodies
- CAR
Chimeric antigen receptor
- CAF
Cancer-associated fibroblast
- cDCs
Conventional dendritic cells
- CRS
Cytokine release syndrome
- CTL
Cytotoxic T lymphocyte
- CTLA-4
Cytotoxic T-lymphocyte-associated protein 4
- CSF1
Colony-stimulating factor 1
- CSF-1R
Colony-stimulating factor 1 receptor
- DC
Dendritic cell
- DFS
Disease-free survival
- ECM
Extracellular matrix
- EGFR
Epidermal growth factor receptor
- EMT
Epithelial-mesenchymal transition
- FAPα
Fibroblast activation protein alpha
- GM-CSF
Granulocyte–macrophage colony-stimulating factor
- ICB
Immune checkpoint blockade
- ICI
Immune checkpoint inhibitor
- IDO
Indoleamine 2, 3-dioxygenase
- IFN-I
Type I interferon
- IFN-γ
Interferon-gamma
- IL
Interleukin
- iPSC
Induced pluripotent stem cell
- LAG-3
Lymphocyte-activation gene 3
- mAb
Monoclonal antibody
- MDSC
Myeloid-derived suppressor cell
- MHC
Major histocompatibility complex
- MMP
Matrix metalloproteinase
- mOS
Median overall survival
- MSC
Mesenchymal stem cell
- MSI-H
Microsatellite Instability-High
- MSLN
Mesothelin
- NK
Natural killer
- ORR
Overall response rate
- OS
Overall survival
- OVT
Oncolytic virus therapy
- PD-1
Programmed cell death protein 1
- PDAC
Pancreatic ductal adenocarcinoma
- PD-L1
Programmed death-ligand 1
- PFS
Progression-free survival
- PSC
Pancreatic stellate cell
- STING
Stimulator of interferon genes
- TA-MSCs
Tumor-associated mesenchymal stem cells
- TAM
Tumor-associated macrophage
- TCR
T-cell receptor
- TGF-β
Transforming growth factor-beta
- TH1
T helper 1
- TH17
T helper 17
- TH2
T helper 2
- TIGIT
T cell immunoreceptor with Ig and ITIM domains
- TIM-3
T-cell immunoglobulin and mucin domain 3
- TIL
Tumor-infiltrating lymphocyte
- TLR
Toll-like receptor
- TMB
Tumor Mutational Burden
- TME
Tumor microenvironment
- Treg
Regulatory T cell
- T-VEC
Talimogene laherparepvec
- VEGF
Vascular endothelial growth factor
- VISTA
V-domain Ig-containing suppressor of T-cell activation
Author contributions
PF: Conceptualization, writing—original draft, writing—review and editing; HK: writing—review and editing, visualization; HN: Writing—review and editing; AAD: Conceptualization, writing—review and editing, and supervision. All authors have read and agreed to the published version of this manuscript.
Funding
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Hidalgo M. Pancreatic cancer. N Engl J Med. 2010;362:1605–1617. doi: 10.1056/NEJMra0901557. [DOI] [PubMed] [Google Scholar]
- 2.Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73:17–48. doi: 10.3322/caac.21763. [DOI] [PubMed] [Google Scholar]
- 3.Cabasag CJ, Arnold M, Rutherford M, Bardot A, Ferlay J, Morgan E, et al. Pancreatic cancer survival by stage and age in seven high-income countries (ICBP SURVMARK-2): a population-based study. Br J Cancer. 2022;126:1774–1782. doi: 10.1038/s41416-022-01752-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kleeff J, Korc M, Apte M, La Vecchia C, Johnson CD, Biankin AV, et al. Pancreatic cancer. Nat Rev Dis Prim. 2016;2:16022. doi: 10.1038/nrdp.2016.22. [DOI] [PubMed] [Google Scholar]
- 5.Feng Y, Cai L, Pook M, Liu F, Chang C-H, Mouti MA, Nibhani R, Militi S, Dunford J, Philpott M, Fan Y, Fan G-C, Liu Q, Qi J, Wang C, Hong W, Morgan H, Wang M, Sadayappan S, Jegga AG, Oppermann U, Wang Y, Huang W, Jiang LPS. BRD9-SMAD2/3 orchestrates stemness and tumorigenesis in pancreatic ductal adenocarcinoma. Gastroenterology. 2023;166:139–154. doi: 10.1053/j.gastro.2023.09.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Luo J. KRAS mutation in pancreatic cancer. Semin Oncol. 2021;48:10–18. doi: 10.1053/j.seminoncol.2021.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Akkapeddi P, Hattori T, Khan I, Glasser E, Koide A, Ketavarapu G, et al. Exploring switch II pocket conformation of KRAS(G12D) with mutant-selective monobody inhibitors. Proc Natl Acad Sci. 2023;120:e2302485120. doi: 10.1073/pnas.2302485120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mahadevan KK, LeBleu VS, Ramirez EV, Chen Y, Li B, Sockwell AM, et al. Elimination of oncogenic KRAS in genetic mouse models eradicates pancreatic cancer by inducing FAS-dependent apoptosis by CD8+ T cells. Dev Cell. 2023;58:1562–1577.e8. doi: 10.1016/j.devcel.2023.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mahadevan KK, McAndrews KM, LeBleu VS, Yang S, Lyu H, Li B, et al. KRASG12D inhibition reprograms the microenvironment of early and advanced pancreatic cancer to promote FAS-mediated killing by CD8+ T cells. Cancer Cell. 2023;41:1606–1620.e8. doi: 10.1016/j.ccell.2023.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pan M, Jiang C, Zhang Z, Achacoso N, Alexeeff S, Solorzano AV, et al. TP53 gain-of-function and non–gain-of-function mutations are associated with differential prognosis in advanced pancreatic ductal adenocarcinoma. JCO Precis Oncol. 2023;7:e2200570. doi: 10.1200/PO.22.00570. [DOI] [PubMed] [Google Scholar]
- 11.Morton JP, Timpson P, Karim SA, Ridgway RA, Athineos D, Doyle B, et al. Mutant p53 drives metastasis and overcomes growth arrest/senescence in pancreatic cancer. Proc Natl Acad Sci. 2010;107:246–251. doi: 10.1073/pnas.0908428107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ho WJ, Jaffee EM, Zheng L. The tumour microenvironment in pancreatic cancer—clinical challenges and opportunities. Nat Rev Clin Oncol. 2020;17:527–540. doi: 10.1038/s41571-020-0363-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lennon AM, Wolfgang CL, Canto MI, Klein AP, Herman JM, Goggins M, et al. The early detection of pancreatic cancer: what will it take to diagnose and treat curable pancreatic neoplasia? Cancer Res. 2014;74:3381–3389. doi: 10.1158/0008-5472.CAN-14-0734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Foster DS, Jones RE, Ransom RC, Longaker MT, Norton JA. The evolving relationship of wound healing and tumor stroma. JCI Insight. 2018;3:e99911. doi: 10.1172/jci.insight.99911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dvorak HF. Tumors: wounds that do not heal-redux. Cancer Immunol Res. 2015;3:1–11. doi: 10.1158/2326-6066.CIR-14-0209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhao Z, Zheng L, Chen W, Weng W, Song J, Ji J. Delivery strategies of cancer immunotherapy: recent advances and future perspectives. J Hematol Oncol. 2019;12:126. doi: 10.1186/s13045-019-0817-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Baker DJ, Arany Z, Baur JA, Epstein JA, June CH. CAR T therapy beyond cancer: the evolution of a living drug. Nature. 2023;619:707–715. doi: 10.1038/s41586-023-06243-w. [DOI] [PubMed] [Google Scholar]
- 18.Ramezani F, Panahi Meymandi AR, Akbari B, Tamtaji OR, Mirzaei H, Brown CE, et al. Outsmarting trogocytosis to boost CAR NK/T cell therapy. Mol Cancer. 2023;22:183. doi: 10.1186/s12943-023-01894-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhang Y, Zhang Z. The history and advances in cancer immunotherapy: understanding the characteristics of tumor-infiltrating immune cells and their therapeutic implications. Cell Mol Immunol. 2020;17:807–821. doi: 10.1038/s41423-020-0488-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Uslu U, Da T, Assenmacher C-A, Scholler J, Young RM, Tchou J, et al. Chimeric antigen receptor T cells as adjuvant therapy for unresectable adenocarcinoma. Sci Adv. 2023;9:02526. doi: 10.1126/sciadv.ade2526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Garfall AL, Cohen AD, Susanibar-Adaniya SP, Hwang W-T, Vogl DT, Waxman AJ, et al. Anti-BCMA/CD19 CAR T cells with early immunomodulatory maintenance for multiple myeloma responding to initial or later-line therapy. Blood Cancer Discov. 2023;4:118–133. doi: 10.1158/2643-3230.BCD-22-0074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sharma P, Goswami S, Raychaudhuri D, Siddiqui BA, Singh P, Nagarajan A, et al. Immune checkpoint therapy—current perspectives and future directions. Cell. 2023;186:1652–1669. doi: 10.1016/j.cell.2023.03.006. [DOI] [PubMed] [Google Scholar]
- 23.Khalil DN, Smith EL, Brentjens RJ, Wolchok JD. The future of cancer treatment: immunomodulation, CARs and combination immunotherapy. Nat Rev Clin Oncol. 2016;13:273–290. doi: 10.1038/nrclinonc.2016.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Farhangnia P, Mollazadeh Ghomi S, Mollazadehghomi S, Delbandi A-A. Current Clinical Landscape of Immunotherapeutic Approaches in Pancreatic Cancer Treatment. Cham: Springer; 2023. pp. 1–54. [Google Scholar]
- 25.Zheng L, Ding D, Edil BH, Judkins C, Durham JN, Thomas DL, II, et al. Vaccine-induced intratumoral lymphoid aggregates correlate with survival following treatment with a neoadjuvant and adjuvant vaccine in patients with resectable pancreatic adenocarcinoma. Clin Cancer Res. 2021;27:1278–1286. doi: 10.1158/1078-0432.CCR-20-2974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pihlak R, Weaver JMJ, Valle JW, McNamara MG. Advances in molecular profiling and categorisation of pancreatic adenocarcinoma and the implications for therapy. Cancers (Basel) 2018;10:17. doi: 10.3390/cancers10010017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Riquelme E, Maitra A, McAllister F. Immunotherapy for pancreatic cancer: more than just a gut feeling. Cancer Discov. 2018;8:386–388. doi: 10.1158/2159-8290.CD-18-0123. [DOI] [PubMed] [Google Scholar]
- 28.Huber M, Brehm CU, Gress TM, Buchholz M, Alashkar Alhamwe B, von Strandmann EP, et al. The immune microenvironment in pancreatic cancer. Int J Mol Sci. 2020;21:7307. doi: 10.3390/ijms21197307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Timmer FEF, Geboers B, Nieuwenhuizen S, Dijkstra M, Schouten EAC, Puijk RS, et al. Pancreatic cancer and immunotherapy: a clinical overview. Cancers (Basel) 2021;13:4138. doi: 10.3390/cancers13164138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tao J, Yang G, Zhou W, Qiu J, Chen G, Luo W, et al. Targeting hypoxic tumor microenvironment in pancreatic cancer. J Hematol Oncol. 2021;14:14. doi: 10.1186/s13045-020-01030-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chen J, Xiao-Zhong G, Qi X-S. Clinical outcomes of specific immunotherapy in advanced pancreatic cancer: a systematic review and meta-analysis. J Immunol Res. 2017;2017:8282391. doi: 10.1155/2017/8282391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Geboers B, Ruarus AH, Nieuwenhuizen S, Puijk RS, Scheffer HJ, de Gruijl TD, et al. Needle-guided ablation of locally advanced pancreatic cancer: cytoreduction or immunomodulation by in vivo vaccination? Chin Clin Oncol. 2019;8:61. doi: 10.21037/cco.2019.10.05. [DOI] [PubMed] [Google Scholar]
- 33.Galluzzi L, Humeau J, Buqué A, Zitvogel L, Kroemer G. Immunostimulation with chemotherapy in the era of immune checkpoint inhibitors. Nat Rev Clin Oncol. 2020;17:725–741. doi: 10.1038/s41571-020-0413-z. [DOI] [PubMed] [Google Scholar]
- 34.Kleeff J, Beckhove P, Esposito I, Herzig S, Huber PE, Löhr JM, et al. Pancreatic cancer microenvironment. Int J Cancer. 2007;121:699–705. doi: 10.1002/ijc.22871. [DOI] [PubMed] [Google Scholar]
- 35.Feig C, Jones JO, Kraman M, Wells RJB, Deonarine A, Chan DS, et al. Targeting CXCL12 from FAP-expressing carcinoma-associated fibroblasts synergizes with anti-PD-L1 immunotherapy in pancreatic cancer. Proc Natl Acad Sci U S A. 2013;110:20212–20217. doi: 10.1073/pnas.1320318110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Soares KC, Rucki AA, Wu AA, Olino K, Xiao Q, Chai Y, et al. PD-1/PD-L1 blockade together with vaccine therapy facilitates effector T-cell infiltration into pancreatic tumors. J Immunother. 2015;38:1–11. doi: 10.1097/CJI.0000000000000062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Balsano R, Zanuso V, Pirozzi A, Rimassa L, Bozzarelli S. Pancreatic ductal adenocarcinoma and immune checkpoint inhibitors: the gray curtain of immunotherapy and spikes of lights. Curr Oncol. 2023;30:3871–3885. doi: 10.3390/curroncol30040293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Whatcott CJ, Diep CH, Jiang P, Watanabe A, LoBello J, Sima C, et al. Desmoplasia in primary tumors and metastatic lesions of pancreatic cancer. Clin Cancer Res. 2015;21:3561–3568. doi: 10.1158/1078-0432.CCR-14-1051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Protti MP, De Monte L. Cross-talk within the tumor microenvironment mediates Th2-type inflammation in pancreatic cancer. Oncoimmunology. 2012;1:89–91. doi: 10.4161/onci.1.1.17939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Feig C, Gopinathan A, Neesse A, Chan DS, Cook N, Tuveson DA. The pancreas cancer microenvironment. Clin Cancer Res. 2012;18:4266–4276. doi: 10.1158/1078-0432.CCR-11-3114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Le DT, Wang-Gillam A, Picozzi V, Greten TF, Crocenzi T, Springett G, et al. Safety and survival with GVAX pancreas prime and Listeria Monocytogenes-expressing mesothelin (CRS-207) boost vaccines for metastatic pancreatic cancer. J Clin Oncol. 2015;33:1325–1333. doi: 10.1200/JCO.2014.57.4244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chiorazzi M, Martinek J, Krasnick B, Zheng Y, Robbins KJ, Qu R, et al. Autologous humanized PDX modeling for immuno-oncology recapitulates features of the human tumor microenvironment. J Immunother Cancer. 2023;11:e006921. doi: 10.1136/jitc-2023-006921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Truong L-H, Pauklin S. Pancreatic cancer microenvironment and cellular composition: current understandings and therapeutic approaches. Cancers (Basel) 2021;13:5028. doi: 10.3390/cancers13195028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Skorupan N, Palestino Dominguez M, Ricci SL, Alewine C. Clinical strategies targeting the tumor microenvironment of pancreatic ductal adenocarcinoma. Cancers (Basel) 2022;14:4209. doi: 10.3390/cancers14174209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Murakami T, Hiroshima Y, Matsuyama R, Homma Y, Hoffman RM, Endo I. Role of the tumor microenvironment in pancreatic cancer. Ann Gastroenterol Surg. 2019;3:130–137. doi: 10.1002/ags3.12225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sherman MH, Beatty GL. Tumor microenvironment in pancreatic cancer pathogenesis and therapeutic resistance. Annu Rev Pathol. 2023;18:123–148. doi: 10.1146/annurev-pathmechdis-031621-024600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Binnewies M, Roberts EW, Kersten K, Chan V, Fearon DF, Merad M, et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat Med. 2018;24:541–550. doi: 10.1038/s41591-018-0014-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Li J, Byrne KT, Yan F, Yamazoe T, Chen Z, Baslan T, et al. Tumor cell-intrinsic factors underlie heterogeneity of immune cell infiltration and response to immunotherapy. Immunity. 2018;49:178–193.e7. doi: 10.1016/j.immuni.2018.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Stromnes IM, Hulbert A, Pierce RH, Greenberg PD, Hingorani SR. T-cell localization, activation, and clonal expansion in human pancreatic ductal adenocarcinoma. Cancer Immunol Res. 2017;5:978–991. doi: 10.1158/2326-6066.CIR-16-0322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Balli D, Rech AJ, Stanger BZ, Vonderheide RH. Immune cytolytic activity stratifies molecular subsets of human pancreatic cancer. Clin Cancer Res. 2017;23:3129–3138. doi: 10.1158/1078-0432.CCR-16-2128. [DOI] [PubMed] [Google Scholar]
- 51.Carstens JL, Correa de Sampaio P, Yang D, Barua S, Wang H, Rao A, et al. Spatial computation of intratumoral T cells correlates with survival of patients with pancreatic cancer. Nat Commun. 2017;8:15095. doi: 10.1038/ncomms15095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ene-Obong A, Clear AJ, Watt J, Wang J, Fatah R, Riches JC, et al. Activated pancreatic stellate cells sequester CD8+ T cells to reduce their infiltration of the juxtatumoral compartment of pancreatic ductal adenocarcinoma. Gastroenterology. 2013;145:1121–1132. doi: 10.1053/j.gastro.2013.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Balachandran VP, Łuksza M, Zhao JN, Makarov V, Moral JA, Remark R, et al. Identification of unique neoantigen qualities in long-term survivors of pancreatic cancer. Nature. 2017;551:512–516. doi: 10.1038/nature24462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Xiao Z, Todd L, Huang L, Noguera-Ortega E, Lu Z, Huang L, et al. Desmoplastic stroma restricts T cell extravasation and mediates immune exclusion and immunosuppression in solid tumors. Nat Commun. 2023;14:5110. doi: 10.1038/s41467-023-40850-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ermis M, Falcone N, Roberto de Barros N, Mecwan M, Haghniaz R, Choroomi A, et al. Tunable hybrid hydrogels with multicellular spheroids for modeling desmoplastic pancreatic cancer. Bioact Mater. 2023;25:360–373. doi: 10.1016/j.bioactmat.2023.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Raskov H, Orhan A, Christensen JP, Gögenur I. Cytotoxic CD8+ T cells in cancer and cancer immunotherapy. Br J Cancer. 2021;124:359–367. doi: 10.1038/s41416-020-01048-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Orhan A, Vogelsang RP, Andersen MB, Madsen MT, Hölmich ER, Raskov H, et al. The prognostic value of tumour-infiltrating lymphocytes in pancreatic cancer: a systematic review and meta-analysis. Eur J Cancer. 2020;132:71–84. doi: 10.1016/j.ejca.2020.03.013. [DOI] [PubMed] [Google Scholar]
- 58.D’Angelo A, Sobhani N, Roviello G, Bagby S, Bonazza D, Bottin C, et al. Tumour infiltrating lymphocytes and immune-related genes as predictors of outcome in pancreatic adenocarcinoma. PLoS ONE. 2019;14:e0219566. doi: 10.1371/journal.pone.0219566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wartenberg M, Zlobec I, Perren A, Koelzer VH, Gloor B, Lugli A, et al. Accumulation of FOXP3+T-cells in the tumor microenvironment is associated with an epithelial-mesenchymal-transition-type tumor budding phenotype and is an independent prognostic factor in surgically resected pancreatic ductal adenocarcinoma. Oncotarget. 2015;6:4190–4201. doi: 10.18632/oncotarget.2775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ino Y, Yamazaki-Itoh R, Shimada K, Iwasaki M, Kosuge T, Kanai Y, et al. Immune cell infiltration as an indicator of the immune microenvironment of pancreatic cancer. Br J Cancer. 2013;108:914–923. doi: 10.1038/bjc.2013.32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Teramatsu K, Oono T, Oyama K, Fujimori N, Murakami M, Yasumori S, et al. Circulating CD8+CD122+ T cells as a prognostic indicator of pancreatic cancer. BMC Cancer. 2022;22:1134. doi: 10.1186/s12885-022-10207-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lutz V, Hellmund VM, Picard FSR, Raifer H, Ruckenbrod T, Klein M, et al. IL18 receptor signaling regulates tumor-reactive CD8+ T-cell exhaustion via activation of the IL2/STAT5/mTOR pathway in a pancreatic cancer model. Cancer Immunol Res. 2023;11:421–434. doi: 10.1158/2326-6066.CIR-22-0398. [DOI] [PubMed] [Google Scholar]
- 63.Heiduk M, Plesca I, Glück J, Müller L, Digomann D, Reiche C, et al. Neoadjuvant chemotherapy drives intratumoral T cells toward a proinflammatory profile in pancreatic cancer. JCI Insight. 2022;7:e152761. doi: 10.1172/jci.insight.152761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Saravia J, Chapman NM, Chi H. Helper T cell differentiation. Cell Mol Immunol. 2019;16:634–643. doi: 10.1038/s41423-019-0220-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Wei R, Zhang H, Cao J, Qin D, Deng W, Li S. Type 1 T helper cell-based molecular subtypes and signature are associated with clinical outcome in pancreatic ductal adenocarcinoma. Front cell Dev Biol. 2022;10:839893. doi: 10.3389/fcell.2022.839893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Abiko K, Matsumura N, Hamanishi J, Horikawa N, Murakami R, Yamaguchi K, et al. IFN-γ from lymphocytes induces PD-L1 expression and promotes progression of ovarian cancer. Br J Cancer. 2015;112:1501–1509. doi: 10.1038/bjc.2015.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Fukunaga A, Miyamoto M, Cho Y, Murakami S, Kawarada Y, Oshikiri T, et al. CD8+ tumor-infiltrating lymphocytes together with CD4+ tumor-infiltrating lymphocytes and dendritic cells improve the prognosis of patients with pancreatic adenocarcinoma. Pancreas. 2004;28:e26–31. doi: 10.1097/00006676-200401000-00023. [DOI] [PubMed] [Google Scholar]
- 68.Plottel CS, Blaser MJ. Microbiome and malignancy. Cell Host Microbe. 2011;10:324–335. doi: 10.1016/j.chom.2011.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Owyang C, Wu GD. The gut microbiome in health and disease. Gastroenterology. 2014;146:1433–1436. doi: 10.1053/j.gastro.2014.03.032. [DOI] [PubMed] [Google Scholar]
- 70.Pushalkar S, Hundeyin M, Daley D, Zambirinis CP, Kurz E, Mishra A, et al. The pancreatic cancer microbiome promotes oncogenesis by induction of innate and adaptive immune suppression. Cancer Discov. 2018;8:403–416. doi: 10.1158/2159-8290.CD-17-1134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Geller LT, Barzily-Rokni M, Danino T, Jonas OH, Shental N, Nejman D, et al. Potential role of intratumor bacteria in mediating tumor resistance to the chemotherapeutic drug gemcitabine. Science. 2017;357:1156–1160. doi: 10.1126/science.aah5043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Tassi E, Gavazzi F, Albarello L, Senyukov V, Longhi R, Dellabona P, et al. Carcinoembryonic antigen-specific but not antiviral CD4+ T cell immunity is impaired in pancreatic carcinoma patients. J Immunol. 2008;181:6595–6603. doi: 10.4049/jimmunol.181.9.6595. [DOI] [PubMed] [Google Scholar]
- 73.De Monte L, Reni M, Tassi E, Clavenna D, Papa I, Recalde H, et al. Intratumor T helper type 2 cell infiltrate correlates with cancer-associated fibroblast thymic stromal lymphopoietin production and reduced survival in pancreatic cancer. J Exp Med. 2011;208:469–478. doi: 10.1084/jem.20101876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Piro G, Simionato F, Carbone C, Frizziero M, Malleo G, Zanini S, et al. A circulating T(H)2 cytokines profile predicts survival in patients with resectable pancreatic adenocarcinoma. Oncoimmunology. 2017;6:e1322242. doi: 10.1080/2162402X.2017.1322242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.De Monte L, Wörmann S, Brunetto E, Heltai S, Magliacane G, Reni M, et al. Basophil recruitment into tumor-draining lymph nodes correlates with Th2 inflammation and reduced survival in pancreatic cancer patients. Cancer Res. 2016;76:1792–1803. doi: 10.1158/0008-5472.CAN-15-1801-T. [DOI] [PubMed] [Google Scholar]
- 76.Jacenik D, Karagiannidis I, Beswick EJ. Th2 cells inhibit growth of colon and pancreas cancers by promoting anti-tumorigenic responses from macrophages and eosinophils. Br J Cancer. 2023;128:387–397. doi: 10.1038/s41416-022-02056-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ochi A, Nguyen AH, Bedrosian AS, Mushlin HM, Zarbakhsh S, Barilla R, et al. MyD88 inhibition amplifies dendritic cell capacity to promote pancreatic carcinogenesis via Th2 cells. J Exp Med. 2012;209:1671–1687. doi: 10.1084/jem.20111706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Kryczek I, Wei S, Zou L, Altuwaijri S, Szeliga W, Kolls J, et al. Cutting edge: Th17 and regulatory T cell dynamics and the regulation by IL-2 in the tumor microenvironment. J Immunol. 2007;178:6730–6733. doi: 10.4049/jimmunol.178.11.6730. [DOI] [PubMed] [Google Scholar]
- 79.Miyahara Y, Odunsi K, Chen W, Peng G, Matsuzaki J, Wang R-F. Generation and regulation of human CD4+ IL-17-producing T cells in ovarian cancer. Proc Natl Acad Sci. 2008;105:15505–15510. doi: 10.1073/pnas.0710686105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.McAllister F, Bailey JM, Alsina J, Nirschl C, Lankapalli R, Roeser J, et al. Abstract 2867: TH17 cells promote early pancreatic tumorigenesis. Cancer Res. 2013;73:2867. doi: 10.1158/1538-7445.AM2013-2867. [DOI] [Google Scholar]
- 81.Zhang B, Rong G, Wei H, Zhang M, Bi J, Ma L, et al. The prevalence of Th17 cells in patients with gastric cancer. Biochem Biophys Res Commun. 2008;374:533–537. doi: 10.1016/j.bbrc.2008.07.060. [DOI] [PubMed] [Google Scholar]
- 82.He S, Fei M, Wu Y, Zheng D, Wan D, Wang L, et al. Distribution and clinical significance of Th17 cells in the tumor microenvironment and peripheral blood of pancreatic cancer patients. Int J Mol Sci. 2011;12:7424–7437. doi: 10.3390/ijms12117424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Alam MS, Gaida MM, Bergmann F, Lasitschka F, Giese T, Giese NA, et al. Selective inhibition of the p38 alternative activation pathway in infiltrating T cells inhibits pancreatic cancer progression. Nat Med. 2015;21:1337–1343. doi: 10.1038/nm.3957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Sfanos KS, Bruno TC, Maris CH, Xu L, Thoburn CJ, DeMarzo AM, et al. Phenotypic analysis of prostate-infiltrating lymphocytes reveals TH17 and Treg skewing. Clin Cancer Res. 2008;14:3254–3261. doi: 10.1158/1078-0432.CCR-07-5164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Koyama K, Kagamu H, Miura S, Hiura T, Miyabayashi T, Itoh R, et al. Reciprocal CD4+ T-cell balance of effector CD62Llow CD4+ and CD62LhighCD25+ CD4+ regulatory T cells in small cell lung cancer reflects disease stage. Clin Cancer Res. 2008;14:6770–6779. doi: 10.1158/1078-0432.CCR-08-1156. [DOI] [PubMed] [Google Scholar]
- 86.Wilke CM, Kryczek I, Wei S, Zhao E, Wu K, Wang G, et al. Th17 cells in cancer: help or hindrance? Carcinogenesis. 2011;32:643–649. doi: 10.1093/carcin/bgr019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.McAllister F, Bailey JM, Alsina J, Nirschl CJ, Sharma R, Fan H, et al. Oncogenic kras activates a hematopoietic-to-epithelial IL-17 signaling axis in preinvasive pancreatic neoplasia. Cancer Cell. 2014;25:621–637. doi: 10.1016/j.ccr.2014.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Loncle C, Bonjoch L, Folch-Puy E, Lopez-Millan MB, Lac S, Molejon MI, et al. IL17 functions through the novel REG3β–JAK2–STAT3 inflammatory pathway to promote the transition from chronic pancreatitis to pancreatic cancer. Cancer Res. 2015;75:4852–4862. doi: 10.1158/0008-5472.CAN-15-0896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Zhang Y, Zoltan M, Riquelme E, Xu H, Sahin I, Castro-Pando S, et al. Immune cell production of interleukin 17 induces stem cell features of pancreatic intraepithelial neoplasia cells. Gastroenterology. 2018;155:210–223.e3. doi: 10.1053/j.gastro.2018.03.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Wang X, Chen H, Jiang R, Hong X, Peng J, Chen W, et al. Interleukin-17 activates and synergizes with the notch signaling pathway in the progression of pancreatic ductal adenocarcinoma. Cancer Lett. 2021;508:1–12. doi: 10.1016/j.canlet.2021.03.003. [DOI] [PubMed] [Google Scholar]
- 91.Zhang Y, Chandra V, Riquelme Sanchez E, Dutta P, Quesada PR, Rakoski A, et al. Interleukin-17-induced neutrophil extracellular traps mediate resistance to checkpoint blockade in pancreatic cancer. J Exp Med. 2020;217. [DOI] [PMC free article] [PubMed]
- 92.Mucciolo G, Curcio C, Roux C, Li WY, Capello M, Curto R, et al. IL17A critically shapes the transcriptional program of fibroblasts in pancreatic cancer and switches on their protumorigenic functions. Proc Natl Acad Sci. 2021;118:e2020395118. doi: 10.1073/pnas.2020395118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Picard FSR, Lutz V, Brichkina A, Neuhaus F, Ruckenbrod T, Hupfer A, et al. IL-17A-producing CD8+ T cells promote PDAC via induction of inflammatory cancer-associated fibroblasts. Gut. 2023;72:1510–1522. doi: 10.1136/gutjnl-2022-327855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Pando SC, Li L, Howel RM, Mascaro M, Le Roux O, Romanin D, et al. Abstract 1188: IL-17/IL-17RA signaling in the pancreatic epithelium upregulates B7–H4 to promote tumorigenesis. Cancer Res. 2023;83:1188. doi: 10.1158/1538-7445.AM2023-1188. [DOI] [Google Scholar]
- 95.Gnerlich JL, Mitchem JB, Weir JS, Sankpal NV, Kashiwagi H, Belt BA, et al. Induction of Th17 cells in the tumor microenvironment improves survival in a murine model of pancreatic cancer. J Immunol. 2010;185:4063–4071. doi: 10.4049/jimmunol.0902609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Vignali DAA, Collison LW, Workman CJ. How regulatory T cells work. Nat Rev Immunol. 2008;8:523–532. doi: 10.1038/nri2343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Chaudhary B, Elkord E. Regulatory T cells in the tumor microenvironment and cancer progression: role and therapeutic targeting. Vaccines. 2016;4:28. doi: 10.3390/vaccines4030028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Shevach EM. Mechanisms of foxp3+ T regulatory cell-mediated suppression. Immunity. 2009;30:636–645. doi: 10.1016/j.immuni.2009.04.010. [DOI] [PubMed] [Google Scholar]
- 99.Plitas G, Rudensky AY. Regulatory T cells in cancer. Annu Rev Cancer Biol. 2020;4:459–477. doi: 10.1146/annurev-cancerbio-030419-033428. [DOI] [Google Scholar]
- 100.Liyanage UK, Moore TT, Joo H-G, Tanaka Y, Herrmann V, Doherty G, et al. Prevalence of regulatory T cells is increased in peripheral blood and tumor microenvironment of patients with pancreas or breast adenocarcinoma. J Immunol. 2002;169:2756–2761. doi: 10.4049/jimmunol.169.5.2756. [DOI] [PubMed] [Google Scholar]
- 101.Lytle NK, Ferguson LP, Rajbhandari N, Gilroy K, Fox RG, Deshpande A, et al. A multiscale map of the stem cell state in pancreatic adenocarcinoma. Cell. 2019;177:572–586.e22. doi: 10.1016/j.cell.2019.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Hiraoka N, Onozato K, Kosuge T, Hirohashi S. Prevalence of FOXP3+ regulatory T cells increases during the progression of pancreatic ductal adenocarcinoma and its premalignant lesions. Clin Cancer Res. 2006;12:5423–5434. doi: 10.1158/1078-0432.CCR-06-0369. [DOI] [PubMed] [Google Scholar]
- 103.Jang J-E, Hajdu CH, Liot C, Miller G, Dustin ML, Bar-Sagi D. Crosstalk between regulatory T cells and tumor-associated dendritic cells negates anti-tumor immunity in pancreatic cancer. Cell Rep. 2017;20:558–571. doi: 10.1016/j.celrep.2017.06.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Zhang Y, Lazarus J, Steele NG, Yan W, Lee H-J, Nwosu ZC, et al. Regulatory T-cell depletion alters the tumor microenvironment and accelerates pancreatic carcinogenesis. Cancer Discov. 2020;10:422–439. doi: 10.1158/2159-8290.CD-19-0958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Chellappa S, Hugenschmidt H, Hagness M, Line PD, Labori KJ, Wiedswang G, et al. Regulatory T cells that co-express RORγt and FOXP3 are pro-inflammatory and immunosuppressive and expand in human pancreatic cancer. Oncoimmunology. 2016;5:e1102828. doi: 10.1080/2162402X.2015.1102828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Laskowski TJ, Biederstädt A, Rezvani K. Natural killer cells in antitumour adoptive cell immunotherapy. Nat Rev Cancer. 2022;22:557–575. doi: 10.1038/s41568-022-00491-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Myers JA, Miller JS. Exploring the NK cell platform for cancer immunotherapy. Nat Rev Clin Oncol. 2021;18:85–100. doi: 10.1038/s41571-020-0426-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Imai K, Matsuyama S, Miyake S, Suga K, Nakachi K. Natural cytotoxic activity of peripheral-blood lymphocytes and cancer incidence: an 11-year follow-up study of a general population. Lancet (London, England) 2000;356:1795–1799. doi: 10.1016/S0140-6736(00)03231-1. [DOI] [PubMed] [Google Scholar]
- 109.Guerra N, Tan YX, Joncker NT, Choy A, Gallardo F, Xiong N, et al. NKG2D-deficient mice are defective in tumor surveillance in models of spontaneous malignancy. Immunity. 2008;28:571–580. doi: 10.1016/j.immuni.2008.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.López-Soto A, Gonzalez S, Smyth MJ, Galluzzi L. Control of metastasis by NK cells. Cancer Cell. 2017;32:135–154. doi: 10.1016/j.ccell.2017.06.009. [DOI] [PubMed] [Google Scholar]
- 111.Davis M, Conlon K, Bohac GC, Barcenas J, Leslie W, Watkins L, et al. Effect of pemetrexed on innate immune killer cells and adaptive immune T cells in subjects with adenocarcinoma of the pancreas. J Immunother. 2012;35:629–640. doi: 10.1097/CJI.0b013e31826c8a4f. [DOI] [PubMed] [Google Scholar]
- 112.Husain Z, Huang Y, Seth P, Sukhatme VP. Tumor-derived lactate modifies antitumor immune response: effect on myeloid-derived suppressor cells and NK cells. J Immunol. 2013;191:1486–1495. doi: 10.4049/jimmunol.1202702. [DOI] [PubMed] [Google Scholar]
- 113.Peng Y-P, Xi C-H, Zhu Y, Yin L-D, Wei J-S, Zhang J-J, et al. Altered expression of CD226 and CD96 on natural killer cells in patients with pancreatic cancer. Oncotarget. 2016;7:66586–66594. doi: 10.18632/oncotarget.11953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Li X, Ni R, Chen J, Liu Z, Xiao M, Jiang F, et al. The presence of IGHG1 in human pancreatic carcinomas is associated with immune evasion mechanisms. Pancreas. 2011;40:753–761. doi: 10.1097/MPA.0b013e318213d51b. [DOI] [PubMed] [Google Scholar]
- 115.Lim SA, Kim J, Jeon S, Shin MH, Kwon J, Kim T-J, et al. Defective localization with impaired tumor cytotoxicity contributes to the immune escape of NK cells in pancreatic cancer patients. Front Immunol. 2019;10:496. doi: 10.3389/fimmu.2019.00496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Huang Q, Huang M, Meng F, Sun R. Activated pancreatic stellate cells inhibit NK cell function in the human pancreatic cancer microenvironment. Cell Mol Immunol. 2019;16:87–89. doi: 10.1038/s41423-018-0014-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Walle T, Kraske JA, Liao B, Lenoir B, Timke C, von Bohlenund Halbach E, et al. Radiotherapy orchestrates natural killer cell dependent antitumor immune responses through CXCL8. Sci Adv. 2023;8:04050. doi: 10.1126/sciadv.abh4050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Ireland L, Luckett T, Schmid MC, Mielgo A. Blockade of stromal Gas6 alters cancer cell plasticity, activates NK cells, and inhibits pancreatic cancer metastasis. Front Immunol. 2020;11:297. doi: 10.3389/fimmu.2020.00297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Dallal RM, Christakos P, Lee K, Egawa S, Son Y-I, Lotze MT. Paucity of dendritic cells in pancreatic cancer. Surgery. 2002;131:135–138. doi: 10.1067/msy.2002.119937. [DOI] [PubMed] [Google Scholar]
- 120.Plesca I, Benešová I, Beer C, Sommer U, Müller L, Wehner R, et al. Clinical significance of tumor-infiltrating conventional and plasmacytoid dendritic cells in pancreatic ductal adenocarcinoma. Cancers (Basel) 2022;14:1216. doi: 10.3390/cancers14051216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Tjomsland V, Sandström P, Spångeus A, Messmer D, Emilsson J, Falkmer U, et al. Pancreatic adenocarcinoma exerts systemic effects on the peripheral blood myeloid and plasmacytoid dendritic cells: an indicator of disease severity? BMC Cancer. 2010;10:87. doi: 10.1186/1471-2407-10-87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Yamamoto T, Yanagimoto H, Satoi S, Toyokawa H, Yamao J, Kim S, et al. Circulating myeloid dendritic cells as prognostic factors in patients with pancreatic cancer who have undergone surgical resection. J Surg Res. 2012;173:299–308. doi: 10.1016/j.jss.2010.09.027. [DOI] [PubMed] [Google Scholar]
- 123.Deicher A, Andersson R, Tingstedt B, Lindell G, Bauden M, Ansari D. Targeting dendritic cells in pancreatic ductal adenocarcinoma. Cancer Cell Int. 2018;18:85. doi: 10.1186/s12935-018-0585-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Takahashi K, Toyokawa H, Takai S, Satoi S, Yanagimoto H, Terakawa N, et al. Surgical influence of pancreatectomy on the function and count of circulating dendritic cells in patients with pancreatic cancer. Cancer Immunol Immunother. 2006;55:775–784. doi: 10.1007/s00262-005-0079-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Bellone G, Carbone A, Smirne C, Scirelli T, Buffolino A, Novarino A, et al. Cooperative induction of a tolerogenic dendritic cell phenotype by cytokines secreted by pancreatic carcinoma cells. J Immunol. 2006;177:3448–3460. doi: 10.4049/jimmunol.177.5.3448. [DOI] [PubMed] [Google Scholar]
- 126.Markowitz J, Wang J, Vangundy Z, You J, Yildiz V, Yu L, et al. Nitric oxide mediated inhibition of antigen presentation from DCs to CD4(+) T cells in cancer and measurement of STAT1 nitration. Sci Rep. 2017;7:15424. doi: 10.1038/s41598-017-14970-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.James CA, Baer JM, Zou C, Panni UY, Knolhoff BL, Hogg GD, et al. Systemic alterations in type-2 conventional dendritic cells lead to impaired tumor immunity in pancreatic cancer. Cancer Immunol Res. 2023;11:1055–1067. doi: 10.1158/2326-6066.CIR-21-0946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Bevan MJ. Cross-priming. Nat Immunol. 2006;7:363–365. doi: 10.1038/ni0406-363. [DOI] [PubMed] [Google Scholar]
- 129.Liang Y, Hannan R, Fu Y-X. Type I IFN activating type I dendritic cells for antitumor immunity. Clin Cancer Res. 2021;27:3818–3824. doi: 10.1158/1078-0432.CCR-20-2564. [DOI] [PubMed] [Google Scholar]
- 130.Lin JH, Huffman AP, Wattenberg MM, Walter DM, Carpenter EL, Feldser DM, et al. Type 1 conventional dendritic cells are systemically dysregulated early in pancreatic carcinogenesis. J Exp Med. 2020;217:e20190673. doi: 10.1084/jem.20190673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Thomas AM, Santarsiero LM, Lutz ER, Armstrong TD, Chen Y-C, Huang L-Q, et al. Mesothelin-specific CD8+ T cell responses provide evidence of in vivo cross-priming by antigen-presenting cells in vaccinated pancreatic cancer patients. J Exp Med. 2004;200:297–306. doi: 10.1084/jem.20031435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Gordon S, Taylor PR. Monocyte and macrophage heterogeneity. Nat Rev Immunol. 2005;5:953–964. doi: 10.1038/nri1733. [DOI] [PubMed] [Google Scholar]
- 133.Mantovani A, Marchesi F, Malesci A, Laghi L, Allavena P. Tumour-associated macrophages as treatment targets in oncology. Nat Rev Clin Oncol. 2017;14:399–416. doi: 10.1038/nrclinonc.2016.217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Najafi A, Keykhaee M, Khorramdelazad H, Karimi MY, Nejatbakhsh Samimi L, Aghamohamadi N, et al. Catalase application in cancer therapy: simultaneous focusing on hypoxia attenuation and macrophage reprogramming. Biomed Pharmacother. 2022;153:113483. doi: 10.1016/j.biopha.2022.113483. [DOI] [PubMed] [Google Scholar]
- 135.Vakilian A, Khorramdelazad H, Heidari P, Sheikh Rezaei Z, Hassanshahi G. CCL2/CCR2 signaling pathway in glioblastoma multiforme. Neurochem Int. 2017;103:1–7. doi: 10.1016/j.neuint.2016.12.013. [DOI] [PubMed] [Google Scholar]
- 136.Hashemi SF, Khorramdelazad H. The cryptic role of CXCL17/CXCR8 axis in the pathogenesis of cancers: a review of the latest evidence. J Cell Commun Signal. 2023;17:409–422. doi: 10.1007/s12079-022-00699-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Mosser DM, Edwards JP. Exploring the full spectrum of macrophage activation. Nat Rev Immunol. 2008;8:958–969. doi: 10.1038/nri2448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Zeng W, Li F, Jin S, Ho P-C, Liu P-S, Xie X. Functional polarization of tumor-associated macrophages dictated by metabolic reprogramming. J Exp Clin Cancer Res. 2023;42:245. doi: 10.1186/s13046-023-02832-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Biswas SK, Mantovani A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nat Immunol. 2010;11:889–896. doi: 10.1038/ni.1937. [DOI] [PubMed] [Google Scholar]
- 140.Yang S, Liu Q, Liao Q. Tumor-associated macrophages in pancreatic ductal adenocarcinoma: origin, polarization, function, and reprogramming. Front cell Dev Biol. 2020;8:607209. doi: 10.3389/fcell.2020.607209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Zhu Y, Herndon JM, Sojka DK, Kim K-W, Knolhoff BL, Zuo C, et al. Tissue-resident macrophages in pancreatic ductal adenocarcinoma originate from embryonic hematopoiesis and promote tumor progression. Immunity. 2017;47:323–338.e6. doi: 10.1016/j.immuni.2017.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Hu H, Hang J-J, Han T, Zhuo M, Jiao F, Wang L-W. The M2 phenotype of tumor-associated macrophages in the stroma confers a poor prognosis in pancreatic cancer. Tumour Biol. 2016;37:8657–8664. doi: 10.1007/s13277-015-4741-z. [DOI] [PubMed] [Google Scholar]
- 143.Gabitass RF, Annels NE, Stocken DD, Pandha HA, Middleton GW. Elevated myeloid-derived suppressor cells in pancreatic, esophageal and gastric cancer are an independent prognostic factor and are associated with significant elevation of the Th2 cytokine interleukin-13. Cancer Immunol Immunother. 2011;60:1419–1430. doi: 10.1007/s00262-011-1028-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Zhang R, Liu Q, Peng J, Wang M, Li T, Liu J, et al. CXCL5 overexpression predicts a poor prognosis in pancreatic ductal adenocarcinoma and is correlated with immune cell infiltration. J Cancer. 2020;11:2371–2381. doi: 10.7150/jca.40517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Liou G-Y, Döppler H, Necela B, Edenfield B, Zhang L, Dawson DW, et al. Mutant KRAS-induced expression of ICAM-1 in pancreatic acinar cells causes attraction of macrophages to expedite the formation of precancerous lesions. Cancer Discov. 2015;5:52–63. doi: 10.1158/2159-8290.CD-14-0474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Griesmann H, Drexel C, Milosevic N, Sipos B, Rosendahl J, Gress TM, et al. Pharmacological macrophage inhibition decreases metastasis formation in a genetic model of pancreatic cancer. Gut. 2017;66:1278–1285. doi: 10.1136/gutjnl-2015-310049. [DOI] [PubMed] [Google Scholar]
- 147.Nywening TM, Belt BA, Cullinan DR, Panni RZ, Han BJ, Sanford DE, et al. Targeting both tumour-associated CXCR2+ neutrophils and CCR2+ macrophages disrupts myeloid recruitment and improves chemotherapeutic responses in pancreatic ductal adenocarcinoma. Gut. 2018;67:1112–1123. doi: 10.1136/gutjnl-2017-313738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Filippini D, Agosto SD, Delfino P, Simbolo M, Piro G, Rusev B, et al. Immunoevolution of mouse pancreatic organoid isografts from preinvasive to metastatic disease. Sci Rep. 2019;9:12286. doi: 10.1038/s41598-019-48663-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Dineen SP, Lynn KD, Holloway SE, Miller AF, Sullivan JP, Shames DS, et al. Vascular endothelial growth factor receptor 2 mediates macrophage infiltration into orthotopic pancreatic tumors in mice. Cancer Res. 2008;68:4340–4346. doi: 10.1158/0008-5472.CAN-07-6705. [DOI] [PubMed] [Google Scholar]
- 150.Boyer S, Lee H-J, Steele N, Zhang L, Sajjakulnukit P, Andren A, et al. Multiomic characterization of pancreatic cancer-associated macrophage polarization reveals deregulated metabolic programs driven by the GM-CSF–PI3K pathway. Elife. 2022;11:e73796. doi: 10.7554/eLife.73796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.LaRue MM, Parker S, Puccini J, Cammer M, Kimmelman AC, Bar-Sagi D. Metabolic reprogramming of tumor-associated macrophages by collagen turnover promotes fibrosis in pancreatic cancer. Proc Natl Acad Sci. 2022;119:e2119168119. doi: 10.1073/pnas.2119168119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Weizman N, Krelin Y, Shabtay-Orbach A, Amit M, Binenbaum Y, Wong RJ, et al. Macrophages mediate gemcitabine resistance of pancreatic adenocarcinoma by upregulating cytidine deaminase. Oncogene. 2014;33:3812–3819. doi: 10.1038/onc.2013.357. [DOI] [PubMed] [Google Scholar]
- 153.Mitchem JB, Brennan DJ, Knolhoff BL, Belt BA, Zhu Y, Sanford DE, et al. Targeting tumor-infiltrating macrophages decreases tumor-initiating cells, relieves immunosuppression, and improves chemotherapeutic responsesmacrophages regulate tumor-initiating cells. Cancer Res. 2013;73:1128–1141. doi: 10.1158/0008-5472.CAN-12-2731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Sanford DE, Belt BA, Panni RZ, Mayer A, Deshpande AD, Carpenter D, et al. Inflammatory monocyte mobilization decreases patient survival in pancreatic cancer: a role for targeting the CCL2/CCR2 axis. Clin Cancer Res. 2013;19:3404–3415. doi: 10.1158/1078-0432.CCR-13-0525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Kalbasi A, Komar C, Tooker GM, Liu M, Lee JW, Gladney WL, et al. Tumor-derived CCL2 mediates resistance to radiotherapy in pancreatic ductal adenocarcinoma. Clin Cancer Res. 2017;23:137–148. doi: 10.1158/1078-0432.CCR-16-0870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Di Mitri D, Mirenda M, Vasilevska J, Calcinotto A, Delaleu N, Revandkar A, et al. Re-education of tumor-associated macrophages by CXCR2 blockade drives senescence and tumor inhibition in advanced prostate cancer. Cell Rep. 2019;28:2156–2168.e5. doi: 10.1016/j.celrep.2019.07.068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Zhang M, Huang L, Ding G, Huang H, Cao G, Sun X, et al. Interferon gamma inhibits CXCL8-CXCR2 axis mediated tumor-associated macrophages tumor trafficking and enhances anti-PD1 efficacy in pancreatic cancer. J Immunother Cancer. 2020;8:e000308. doi: 10.1136/jitc-2019-000308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Piro G, Carbone C, Agostini A, Esposito A, De Pizzol M, Novelli R, et al. CXCR1/2 dual-inhibitor ladarixin reduces tumour burden and promotes immunotherapy response in pancreatic cancer. Br J Cancer. 2023;128:331–341. doi: 10.1038/s41416-022-02028-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Zhu Y, Knolhoff BL, Meyer MA, Nywening TM, West BL, Luo J, et al. CSF1/CSF1R blockade reprograms tumor-infiltrating macrophages and improves response to T-cell checkpoint immunotherapy in pancreatic cancer models. Cancer Res. 2014;74:5057–5069. doi: 10.1158/0008-5472.CAN-13-3723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Veglia F, Sanseviero E, Gabrilovich DI. Myeloid-derived suppressor cells in the era of increasing myeloid cell diversity. Nat Rev Immunol. 2021;21:485–498. doi: 10.1038/s41577-020-00490-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Wang S, Zhao X, Wu S, Cui D, Xu Z. Myeloid-derived suppressor cells: key immunosuppressive regulators and therapeutic targets in hematological malignancies. Biomark Res. 2023;11:34. doi: 10.1186/s40364-023-00475-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Arnoletti JP, Reza J, Rosales A, Monreal A, Fanaian N, Whisner S, et al. Pancreatic ductal adenocarcinoma (PDAC) circulating tumor cells influence myeloid cell differentiation to support their survival and immunoresistance in portal vein circulation. PLoS ONE. 2022;17:e0265725. doi: 10.1371/journal.pone.0265725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Diaz-Montero CM, Salem ML, Nishimura MI, Garrett-Mayer E, Cole DJ, Montero AJ. Increased circulating myeloid-derived suppressor cells correlate with clinical cancer stage, metastatic tumor burden, and doxorubicin-cyclophosphamide chemotherapy. Cancer Immunol Immunother. 2009;58:49–59. doi: 10.1007/s00262-008-0523-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Markowitz J, Brooks TR, Duggan MC, Paul BK, Pan X, Wei L, et al. Patients with pancreatic adenocarcinoma exhibit elevated levels of myeloid-derived suppressor cells upon progression of disease. Cancer Immunol Immunother. 2015;64:149–159. doi: 10.1007/s00262-014-1618-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Bayne LJ, Beatty GL, Jhala N, Clark CE, Rhim AD, Stanger BZ, et al. Tumor-derived granulocyte-macrophage colony-stimulating factor regulates myeloid inflammation and T cell immunity in pancreatic cancer. Cancer Cell. 2012;21:822–835. doi: 10.1016/j.ccr.2012.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Pylayeva-Gupta Y, Lee KE, Hajdu CH, Miller G, Bar-Sagi D. Oncogenic Kras-induced GM-CSF production promotes the development of pancreatic neoplasia. Cancer Cell. 2012;21:836–847. doi: 10.1016/j.ccr.2012.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.King RJ, Shukla SK, He C, Vernucci E, Thakur R, Attri KS, et al. CD73 induces GM-CSF/MDSC-mediated suppression of T cells to accelerate pancreatic cancer pathogenesis. Oncogene. 2022;41:971–982. doi: 10.1038/s41388-021-02132-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Vernon PJ, Loux TJ, Schapiro NE, Kang R, Muthuswamy R, Kalinski P, et al. The receptor for advanced glycation end products promotes pancreatic carcinogenesis and accumulation of myeloid-derived suppressor cells. J Immunol. 2013;190:1372–1379. doi: 10.4049/jimmunol.1201151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Murakami S, Shahbazian D, Surana R, Zhang W, Chen H, Graham GT, et al. Yes-associated protein mediates immune reprogramming in pancreatic ductal adenocarcinoma. Oncogene. 2017;36:1232–1244. doi: 10.1038/onc.2016.288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Zhang W, Nandakumar N, Shi Y, Manzano M, Smith A, Graham G, et al. Downstream of mutant KRAS, the transcription regulator YAP is essential for neoplastic progression to pancreatic ductal adenocarcinoma. Sci Signal. 2014;7:ra42. doi: 10.1126/scisignal.2005049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Hagenbeek TJ, Zbieg JR, Hafner M, Mroue R, Lacap JA, Sodir NM, et al. An allosteric pan-TEAD inhibitor blocks oncogenic YAP/TAZ signaling and overcomes KRAS G12C inhibitor resistance. Nat Cancer. 2023;4:812–828. doi: 10.1038/s43018-023-00577-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Chapeau EA, Sansregret L, Galli GG, Chène P, Wartmann M, Mourikis TP, et al. Direct and selective pharmacological disruption of the YAP–TEAD interface by IAG933 inhibits Hippo-dependent and RAS–MAPK-altered cancers. Nat Cancer. 2024; [DOI] [PMC free article] [PubMed]
- 173.Choueiry F, Torok M, Shakya R, Agrawal K, Deems A, Benner B, et al. CD200 promotes immunosuppression in the pancreatic tumor microenvironment. J Immunother Cancer. 2020;8:e000189. doi: 10.1136/jitc-2019-000189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Pinton L, Solito S, Damuzzo V, Francescato S, Pozzuoli A, Berizzi A, et al. Activated T cells sustain myeloid-derived suppressor cell-mediated immune suppression. Oncotarget. 2016;7:1168–1184. doi: 10.18632/oncotarget.6662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Huang B, Pan P-Y, Li Q, Sato AI, Levy DE, Bromberg J, et al. Gr-1+CD115+ immature myeloid suppressor cells mediate the development of tumor-induced T regulatory cells and T-cell anergy in tumor-bearing host. Cancer Res. 2006;66:1123–1131. doi: 10.1158/0008-5472.CAN-05-1299. [DOI] [PubMed] [Google Scholar]
- 176.Siret C, Collignon A, Silvy F, Robert S, Cheyrol T, André P, et al. Deciphering the crosstalk between myeloid-derived suppressor cells and regulatory T cells in pancreatic ductal adenocarcinoma. Front Immunol. 2020;10:3070. doi: 10.3389/fimmu.2019.03070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Lasser SA, Ozbay Kurt FG, Arkhypov I, Utikal J, Umansky V. Myeloid-derived suppressor cells in cancer and cancer therapy. Nat Rev Clin Oncol. 2024;21:147–164. doi: 10.1038/s41571-023-00846-y. [DOI] [PubMed] [Google Scholar]
- 178.Stromnes IM, Brockenbrough JS, Izeradjene K, Carlson MA, Cuevas C, Simmons RM, et al. Targeted depletion of an MDSC subset unmasks pancreatic ductal adenocarcinoma to adaptive immunity. Gut. 2014;63:1769–1781. doi: 10.1136/gutjnl-2013-306271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Sarhan D, Eisinger S, He F, Bergsland M, Pelicano C, Driescher C, et al. Targeting myeloid suppressive cells revives cytotoxic anti-tumor responses in pancreatic cancer. IScience. 2022;25:105317. doi: 10.1016/j.isci.2022.105317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Giese MA, Hind LE, Huttenlocher A. Neutrophil plasticity in the tumor microenvironment. Blood. 2019;133:2159–2167. doi: 10.1182/blood-2018-11-844548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Xiang Z, Hu T, Wang Y, Wang H, Xu L, Cui N. Neutrophil–lymphocyte ratio (NLR) was associated with prognosis and immunomodulatory in patients with pancreatic ductal adenocarcinoma (PDAC) Biosci Rep. 2020;40:BSR20201190. doi: 10.1042/BSR20201190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Chao T, Furth EE, Vonderheide RH. CXCR2-dependent accumulation of tumor-associated neutrophils regulates T-cell immunity in pancreatic ductal adenocarcinoma. Cancer Immunol Res. 2016;4:968–982. doi: 10.1158/2326-6066.CIR-16-0188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Jin L, Kim HS, Shi J. Neutrophil in the pancreatic tumor microenvironment. Biomolecules. 2021;11:1170. doi: 10.3390/biom11081170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Bellomo G, Rainer C, Quaranta V, Astuti Y, Raymant M, Boyd E, et al. Chemotherapy-induced infiltration of neutrophils promotes pancreatic cancer metastasis via Gas6/AXL signalling axis. Gut. 2022;71:2284–2299. doi: 10.1136/gutjnl-2021-325272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Wang Y, Fang T, Huang L, Wang H, Zhang L, Wang Z, et al. Neutrophils infiltrating pancreatic ductal adenocarcinoma indicate higher malignancy and worse prognosis. Biochem Biophys Res Commun. 2018;501:313–319. doi: 10.1016/j.bbrc.2018.05.024. [DOI] [PubMed] [Google Scholar]
- 186.Masucci MT, Minopoli M, Carriero MV. Tumor associated neutrophils their role in tumorigenesis, metastasis, prognosis and therapy. Front Oncol. 2019;9:1146. doi: 10.3389/fonc.2019.01146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Ng MSF, Kwok I, Tan L, Shi C, Cerezo-Wallis D, Tan Y, et al. Deterministic reprogramming of neutrophils within tumors. Science (80-). 2024;383:eadf6493. doi: 10.1126/science.adf6493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Chen Q, Yin H, Liu S, Shoucair S, Ding N, Ji Y, et al. Prognostic value of tumor-associated N1/N2 neutrophil plasticity in patients following radical resection of pancreas ductal adenocarcinoma. J Immunother cancer. 2022;10:e005798. doi: 10.1136/jitc-2022-005798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Nielsen SR, Strøbech JE, Horton ER, Jackstadt R, Laitala A, Bravo MC, et al. Suppression of tumor-associated neutrophils by lorlatinib attenuates pancreatic cancer growth and improves treatment with immune checkpoint blockade. Nat Commun. 2021;12:3414. doi: 10.1038/s41467-021-23731-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Papayannopoulos V. Neutrophil extracellular traps in immunity and disease. Nat Rev Immunol. 2018;18:134–147. doi: 10.1038/nri.2017.105. [DOI] [PubMed] [Google Scholar]
- 191.Zhang J, Xu X, Shi M, Chen Y, Yu D, Zhao C, et al. CD13(hi) Neutrophil-like myeloid-derived suppressor cells exert immune suppression through Arginase 1 expression in pancreatic ductal adenocarcinoma. Oncoimmunology. 2017;6:e1258504. doi: 10.1080/2162402X.2016.1258504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Jin W, Yin H, Li H, Yu X-J, Xu H-X, Liu L. Neutrophil extracellular DNA traps promote pancreatic cancer cells migration and invasion by activating EGFR/ERK pathway. J Cell Mol Med. 2021;25:5443–5456. doi: 10.1111/jcmm.16555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Castino GF, Cortese N, Capretti G, Serio S, Di Caro G, Mineri R, et al. Spatial distribution of B cells predicts prognosis in human pancreatic adenocarcinoma. Oncoimmunology. 2016;5:e1085147. doi: 10.1080/2162402X.2015.1085147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Takahashi R, Macchini M, Sunagawa M, Jiang Z, Tanaka T, Valenti G, et al. Interleukin-1β-induced pancreatitis promotes pancreatic ductal adenocarcinoma via B lymphocyte-mediated immune suppression. Gut. 2021;70:330–341. doi: 10.1136/gutjnl-2019-319912. [DOI] [PubMed] [Google Scholar]
- 195.Denton AE, Innocentin S, Carr EJ, Bradford BM, Lafouresse F, Mabbott NA, et al. Type I interferon induces CXCL13 to support ectopic germinal center formation. J Exp Med. 2019;216:621–637. doi: 10.1084/jem.20181216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Luo W, Wen T, Qu X. Tumor immune microenvironment-based therapies in pancreatic ductal adenocarcinoma: time to update the concept. J Exp Clin Cancer Res. 2024;43:8. doi: 10.1186/s13046-023-02935-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Mirlekar B, Wang Y, Li S, Zhou M, Entwistle S, De Buysscher T, et al. Balance between immunoregulatory B cells and plasma cells drives pancreatic tumor immunity. Cell Rep Med. 2022;3:100744. doi: 10.1016/j.xcrm.2022.100744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Mirlekar B, Michaud D, Lee SJ, Kren NP, Harris C, Greene K, et al. B cell–derived IL35 drives STAT3-dependent CD8+ T-cell exclusion in pancreatic cancer. Cancer Immunol Res. 2020;8:292–308. doi: 10.1158/2326-6066.CIR-19-0349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Li S, Mirlekar B, Johnson BM, Brickey WJ, Wrobel JA, Yang N, et al. STING-induced regulatory B cells compromise NK function in cancer immunity. Nature. 2022;610:373–380. doi: 10.1038/s41586-022-05254-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Tempero M, Oh D-Y, Tabernero J, Reni M, Van Cutsem E, Hendifar A, et al. Ibrutinib in combination with nab-paclitaxel and gemcitabine for first-line treatment of patients with metastatic pancreatic adenocarcinoma: phase III RESOLVE study. Ann Oncol. 2021;32:600–608. doi: 10.1016/j.annonc.2021.01.070. [DOI] [PubMed] [Google Scholar]
- 201.Ishiwata T, Matsuda Y, Yoshimura H, Sasaki N, Ishiwata S, Ishikawa N, et al. Pancreatic cancer stem cells: features and detection methods. Pathol Oncol Res. 2018;24:797–805. doi: 10.1007/s12253-018-0420-x. [DOI] [PubMed] [Google Scholar]
- 202.Bubin R, Uljanovs R, Strumfa I. Cancer stem cells in pancreatic ductal adenocarcinoma. Int J Mol Sci. 2023;24:7030. doi: 10.3390/ijms24087030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Hermann PC, Huber SL, Herrler T, Aicher A, Ellwart JW, Guba M, et al. Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell. 2007;1:313–323. doi: 10.1016/j.stem.2007.06.002. [DOI] [PubMed] [Google Scholar]
- 204.Chang C-H, Liu F, Militi S, Hester S, Nibhani R, Deng S, et al. The pRb/RBL2-E2F1/4-GCN5 axis regulates cancer stem cell formation and G0 phase entry/exit by paracrine mechanisms. Nat Commun. 2024;15:3580. doi: 10.1038/s41467-024-47680-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Shi Y, Du L, Lin L, Wang Y. Tumour-associated mesenchymal stem/stromal cells: emerging therapeutic targets. Nat Rev Drug Discov. 2017;16:35–52. doi: 10.1038/nrd.2016.193. [DOI] [PubMed] [Google Scholar]
- 206.Ren G, Zhao X, Wang Y, Zhang X, Chen X, Xu C, et al. CCR2-dependent recruitment of macrophages by tumor-educated mesenchymal stromal cells promotes tumor development and is mimicked by TNFα. Cell Stem Cell. 2012;11:812–824. doi: 10.1016/j.stem.2012.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Ren G, Zhang L, Zhao X, Xu G, Zhang Y, Roberts AI, et al. Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell. 2008;2:141–150. doi: 10.1016/j.stem.2007.11.014. [DOI] [PubMed] [Google Scholar]
- 208.Su J, Chen X, Huang Y, Li W, Li J, Cao K, et al. Phylogenetic distinction of iNOS and IDO function in mesenchymal stem cell-mediated immunosuppression in mammalian species. Cell Death Differ. 2014;21:388–396. doi: 10.1038/cdd.2013.149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.De Boeck A, Pauwels P, Hensen K, Rummens J-L, Westbroek W, Hendrix A, et al. Bone marrow-derived mesenchymal stem cells promote colorectal cancer progression through paracrine neuregulin 1/HER3 signalling. Gut. 2013;62:550–560. doi: 10.1136/gutjnl-2011-301393. [DOI] [PubMed] [Google Scholar]
- 210.Beckermann BM, Kallifatidis G, Groth A, Frommhold D, Apel A, Mattern J, et al. VEGF expression by mesenchymal stem cells contributes to angiogenesis in pancreatic carcinoma. Br J Cancer. 2008;99:622–631. doi: 10.1038/sj.bjc.6604508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.McLean K, Gong Y, Choi Y, Deng N, Yang K, Bai S, et al. Human ovarian carcinoma–associated mesenchymal stem cells regulate cancer stem cells and tumorigenesis via altered BMP production. J Clin Invest. 2011;121:3206–3219. doi: 10.1172/JCI45273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Patel SA, Meyer JR, Greco SJ, Corcoran KE, Bryan M, Rameshwar P. Mesenchymal stem cells protect breast cancer cells through regulatory T cells: role of mesenchymal stem cell-derived TGF-beta. J Immunol. 2010;184:5885–5894. doi: 10.4049/jimmunol.0903143. [DOI] [PubMed] [Google Scholar]
- 213.Karnoub AE, Dash AB, Vo AP, Sullivan A, Brooks MW, Bell GW, et al. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature. 2007;449:557–563. doi: 10.1038/nature06188. [DOI] [PubMed] [Google Scholar]
- 214.Chaturvedi P, Gilkes DM, Takano N, Semenza GL. Hypoxia-inducible factor-dependent signaling between triple-negative breast cancer cells and mesenchymal stem cells promotes macrophage recruitment. Proc Natl Acad Sci U S A. 2014;111:E2120–E2129. doi: 10.1073/pnas.1406655111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Zhang XH-F, Jin X, Malladi S, Zou Y, Wen YH, Brogi E, et al. Selection of bone metastasis seeds by mesenchymal signals in the primary tumor stroma. Cell. 2013;154:1060–1073. doi: 10.1016/j.cell.2013.07.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Luga V, Zhang L, Viloria-Petit AM, Ogunjimi AA, Inanlou MR, Chiu E, et al. Exosomes mediate stromal mobilization of autocrine Wnt-PCP signaling in breast cancer cell migration. Cell. 2012;151:1542–1556. doi: 10.1016/j.cell.2012.11.024. [DOI] [PubMed] [Google Scholar]
- 217.Ries C, Egea V, Karow M, Kolb H, Jochum M, Neth P. MMP-2, MT1-MMP, and TIMP-2 are essential for the invasive capacity of human mesenchymal stem cells: differential regulation by inflammatory cytokines. Blood. 2007;109:4055–4063. doi: 10.1182/blood-2006-10-051060. [DOI] [PubMed] [Google Scholar]
- 218.Roodhart JML, Daenen LGM, Stigter ECA, Prins H-J, Gerrits J, Houthuijzen JM, et al. Mesenchymal stem cells induce resistance to chemotherapy through the release of platinum-induced fatty acids. Cancer Cell. 2011;20:370–383. doi: 10.1016/j.ccr.2011.08.010. [DOI] [PubMed] [Google Scholar]
- 219.Raghavan S, Snyder CS, Wang A, McLean K, Zamarin D, Buckanovich RJ, et al. Carcinoma-associated mesenchymal stem cells promote chemoresistance in ovarian cancer stem cells via PDGF signaling. Cancers (Basel) 2020;12:2063. doi: 10.3390/cancers12082063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Straussman R, Morikawa T, Shee K, Barzily-Rokni M, Qian ZR, Du J, et al. Tumour micro-environment elicits innate resistance to RAF inhibitors through HGF secretion. Nature. 2012;487:500–504. doi: 10.1038/nature11183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Müerköster S, Wegehenkel K, Arlt A, Witt M, Sipos B, Kruse M-L, et al. Tumor stroma interactions induce chemoresistance in pancreatic ductal carcinoma cells involving increased secretion and paracrine effects of nitric oxide and interleukin-1beta. Cancer Res. 2004;64:1331–1337. doi: 10.1158/0008-5472.CAN-03-1860. [DOI] [PubMed] [Google Scholar]
- 222.Ono M, Kosaka N, Tominaga N, Yoshioka Y, Takeshita F, Takahashi R, et al. Exosomes from bone marrow mesenchymal stem cells contain a microRNA that promotes dormancy in metastatic breast cancer cells. Sci Signal. 2014;7:ra63. doi: 10.1126/scisignal.2005231. [DOI] [PubMed] [Google Scholar]
- 223.Boelens MC, Wu TJ, Nabet BY, Xu B, Qiu Y, Yoon T, et al. Exosome transfer from stromal to breast cancer cells regulates therapy resistance pathways. Cell. 2014;159:499–513. doi: 10.1016/j.cell.2014.09.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Starzyńska T, Dąbkowski K, Błogowski W, Zuba-Surma E, Budkowska M, Sałata D, et al. An intensified systemic trafficking of bone marrow-derived stem/progenitor cells in patients with pancreatic cancer. J Cell Mol Med. 2013;17:792–799. doi: 10.1111/jcmm.12065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Mishra PJ, Mishra PJ, Humeniuk R, Medina DJ, Alexe G, Mesirov JP, et al. Carcinoma-associated fibroblast-like differentiation of human mesenchymal stem cells. Cancer Res. 2008;68:4331–4339. doi: 10.1158/0008-5472.CAN-08-0943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Chen X, Song E. Turning foes to friends: targeting cancer-associated fibroblasts. Nat Rev Drug Discov. 2019;18:99–115. doi: 10.1038/s41573-018-0004-1. [DOI] [PubMed] [Google Scholar]
- 227.Sherman MH, di Magliano MP. Cancer-associated fibroblasts: lessons from pancreatic cancer. Annu Rev Cancer Biol. 2023;7:43–55. doi: 10.1146/annurev-cancerbio-061421-035400. [DOI] [Google Scholar]
- 228.Zhang T, Ren Y, Yang P, Wang J, Zhou H. Cancer-associated fibroblasts in pancreatic ductal adenocarcinoma. Cell Death Dis. 2022;13:897. doi: 10.1038/s41419-022-05351-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Öhlund D, Handly-Santana A, Biffi G, Elyada E, Almeida AS, Ponz-Sarvise M, et al. Distinct populations of inflammatory fibroblasts and myofibroblasts in pancreatic cancer. J Exp Med. 2017;214:579–596. doi: 10.1084/jem.20162024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Gorchs L, Fernández Moro C, Bankhead P, Kern KP, Sadeak I, Meng Q, et al. Human pancreatic carcinoma-associated fibroblasts promote expression of co-inhibitory markers on CD4(+) and CD8(+) T-cells. Front Immunol. 2019;10:847. doi: 10.3389/fimmu.2019.00847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Biffi G, Oni TE, Spielman B, Hao Y, Elyada E, Park Y, et al. IL1-induced JAK/STAT signaling is antagonized by TGFβ to shape CAF heterogeneity in pancreatic ductal adenocarcinoma. Cancer Discov. 2019;9:282–301. doi: 10.1158/2159-8290.CD-18-0710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Raymant M, Astuti Y, Alvaro-Espinosa L, Green D, Quaranta V, Bellomo G, et al. Macrophage-fibroblast JAK/STAT dependent crosstalk promotes liver metastatic outgrowth in pancreatic cancer. Nat Commun. 2024;15:3593. doi: 10.1038/s41467-024-47949-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Vonlaufen A, Phillips PA, Xu Z, Goldstein D, Pirola RC, Wilson JS, et al. Pancreatic stellate cells and pancreatic cancer cells: an unholy alliance. Cancer Res. 2008;68:7707–7710. doi: 10.1158/0008-5472.CAN-08-1132. [DOI] [PubMed] [Google Scholar]
- 234.Schnittert J, Bansal R, Prakash J. Targeting pancreatic stellate cells in cancer. Trends in Cancer. 2019;5:128–142. doi: 10.1016/j.trecan.2019.01.001. [DOI] [PubMed] [Google Scholar]
- 235.Vonlaufen A, Joshi S, Qu C, Phillips PA, Xu Z, Parker NR, et al. Pancreatic stellate cells: partners in crime with pancreatic cancer cells. Cancer Res. 2008;68:2085–2093. doi: 10.1158/0008-5472.CAN-07-2477. [DOI] [PubMed] [Google Scholar]
- 236.Xu Z, Vonlaufen A, Phillips PA, Fiala-Beer E, Zhang X, Yang L, et al. Role of pancreatic stellate cells in pancreatic cancer metastasis. Am J Pathol. 2010;177:2585–2596. doi: 10.2353/ajpath.2010.090899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Provenzano PP, Cuevas C, Chang AE, Goel VK, Von Hoff DD, Hingorani SR. Enzymatic targeting of the stroma ablates physical barriers to treatment of pancreatic ductal adenocarcinoma. Cancer Cell. 2012;21:418–429. doi: 10.1016/j.ccr.2012.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Schneiderhan W, Diaz F, Fundel M, Zhou S, Siech M, Hasel C, et al. Pancreatic stellate cells are an important source of MMP-2 in human pancreatic cancer and accelerate tumor progression in a murine xenograft model and CAM assay. J Cell Sci. 2007;120:512–519. doi: 10.1242/jcs.03347. [DOI] [PubMed] [Google Scholar]
- 239.Cave DD, Di Guida M, Costa V, Sevillano M, Ferrante L, Heeschen C, et al. TGF-β1 secreted by pancreatic stellate cells promotes stemness and tumourigenicity in pancreatic cancer cells through L1CAM downregulation. Oncogene. 2020;39:4271–4285. doi: 10.1038/s41388-020-1289-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Martin JD, Seano G, Jain RK. Normalizing function of tumor vessels: progress, opportunities, and challenges. Annu Rev Physiol. 2019;81:505–534. doi: 10.1146/annurev-physiol-020518-114700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Katsuta E, Qi Q, Peng X, Hochwald SN, Yan L, Takabe K. Pancreatic adenocarcinomas with mature blood vessels have better overall survival. Sci Rep. 2019;9:1310. doi: 10.1038/s41598-018-37909-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Rhim AD, Oberstein PE, Thomas DH, Mirek ET, Palermo CF, Sastra SA, et al. Stromal elements act to restrain, rather than support, pancreatic ductal adenocarcinoma. Cancer Cell. 2014;25:735–747. doi: 10.1016/j.ccr.2014.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Lv X, Li J, Zhang C, Hu T, Li S, He S, et al. The role of hypoxia-inducible factors in tumor angiogenesis and cell metabolism. Genes Dis. 2016;4:19–24. doi: 10.1016/j.gendis.2016.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Keith B, Johnson RS, Simon MC. HIF1α and HIF2α: sibling rivalry in hypoxic tumour growth and progression. Nat Rev Cancer. 2011;12:9–22. doi: 10.1038/nrc3183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Lee KE, Spata M, Bayne LJ, Buza EL, Durham AC, Allman D, et al. Hif1a deletion reveals pro-neoplastic function of b cells in pancreatic neoplasia. Cancer Discov. 2016;6:256–269. doi: 10.1158/2159-8290.CD-15-0822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Fink DM, Steele MM, Hollingsworth MA. The lymphatic system and pancreatic cancer. Cancer Lett. 2016;381:217–236. doi: 10.1016/j.canlet.2015.11.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Shen C-N, Goh K-S, Huang C-R, Chiang T-C, Lee C-Y, Jeng Y-M, et al. Lymphatic vessel remodeling and invasion in pancreatic cancer progression. EBioMedicine. 2019;47:98–113. doi: 10.1016/j.ebiom.2019.08.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Sperveslage J, Frank S, Heneweer C, Egberts J, Schniewind B, Buchholz M, et al. Lack of CCR7 expression is rate limiting for lymphatic spread of pancreatic ductal adenocarcinoma. Int J cancer. 2012;131:E371–E381. doi: 10.1002/ijc.26502. [DOI] [PubMed] [Google Scholar]
- 249.Malik S, Westcott JM, Brekken RA, Burrows FJ. CXCL12 in pancreatic cancer: its function and potential as a therapeutic drug target. Cancers (Basel) 2021;14:86. doi: 10.3390/cancers14010086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Ullman NA, Burchard PR, Dunne RF, Linehan DC. Immunologic strategies in pancreatic cancer: making cold tumors hot. J Clin Oncol. 2022;40:2789–2805. doi: 10.1200/JCO.21.02616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Nisar M, Paracha RZ, Adil S, Qureshi SN, Janjua HA. An extensive review on preclinical and clinical trials of oncolytic viruses therapy for pancreatic cancer. Front Oncol. 2022;12:875188. doi: 10.3389/fonc.2022.875188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Lou E. Oncolytic herpes viruses as a potential mechanism for cancer therapy. Acta Oncol. 2003;42:660–671. doi: 10.1080/0284186031000518. [DOI] [PubMed] [Google Scholar]
- 253.Kaufman HL, Kohlhapp FJ, Zloza A. Oncolytic viruses: a new class of immunotherapy drugs. Nat Rev Drug Discov. 2015;14:642–662. doi: 10.1038/nrd4663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Toda M, Martuza RL, Rabkin SD. Tumor growth inhibition by intratumoral inoculation of defective herpes simplex virus vectors expressing granulocyte-macrophage colony-stimulating factor. Mol Ther. 2000;2:324–329. doi: 10.1006/mthe.2000.0130. [DOI] [PubMed] [Google Scholar]
- 255.McAuliffe PF, Jarnagin WR, Johnson P, Delman KA, Federoff H, Fong Y. Effective treatment of pancreatic tumors with two multimutated herpes simplex oncolytic viruses. J Gastrointest Surg. 2000;4:580–588. doi: 10.1016/S1091-255X(00)80106-7. [DOI] [PubMed] [Google Scholar]
- 256.Hirooka Y, Kasuya H, Ishikawa T, Kawashima H, Ohno E, Villalobos IB, et al. A phase I clinical trial of EUS-guided intratumoral injection of the oncolytic virus, HF10 for unresectable locally advanced pancreatic cancer. BMC Cancer. 2018;18:596. doi: 10.1186/s12885-018-4453-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Cho I-R, Kaowinn S, Moon J, Soh J, Kang HY, Jung C-R, et al. Oncotropic H-1 parvovirus infection degrades HIF-1α protein in human pancreatic cancer cells independently of VHL and RACK1. Int J Oncol Greece. 2015;46:2076–2082. doi: 10.3892/ijo.2015.2922. [DOI] [PubMed] [Google Scholar]
- 258.Bazan-Peregrino M, Garcia-Carbonero R, Laquente B, Álvarez R, Mato-Berciano A, Gimenez-Alejandre M, et al. VCN-01 disrupts pancreatic cancer stroma and exerts antitumor effects. J Immunother cancer. 2021;9:e003254. doi: 10.1136/jitc-2021-003254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Garcia-Carbonero R, Bazan-Peregrino M, Gil-Martín M, Álvarez R, Macarulla T, Riesco-Martinez MC, et al. Phase I, multicenter, open-label study of intravenous VCN-01 oncolytic adenovirus with or without nab-paclitaxel plus gemcitabine in patients with advanced solid tumors. J Immunother Cancer. 2022;10:e003255. doi: 10.1136/jitc-2021-003255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Musher BL, Smaglo BG, Abidi W, Othman M, Patel K, Jawaid S, et al. A phase I/II study of LOAd703, a TMZ-CD40L/4-1BBL-armed oncolytic adenovirus, combined with nab-paclitaxel and gemcitabine in advanced pancreatic cancer. J Clin Oncol. 2022;40:4138. doi: 10.1200/JCO.2022.40.16_suppl.4138. [DOI] [Google Scholar]
- 261.Mahalingam D, Chen S, Xie P, Loghmani H, Heineman T, Kalyan A, et al. Combination of pembrolizumab and pelareorep promotes anti-tumour immunity in advanced pancreatic adenocarcinoma (PDAC) Br J Cancer. 2023;129:782–790. doi: 10.1038/s41416-023-02344-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Chen Y, Chen X, Bao W, Liu G, Wei W, Ping Y. An oncolytic virus–T cell chimera for cancer immunotherapy. Nat Biotechnol. 2024; [DOI] [PubMed]
- 263.Rosenberg SA, Restifo NP, Yang JC, Morgan RA, Dudley ME. Adoptive cell transfer: a clinical path to effective cancer immunotherapy. Nat Rev Cancer. 2008;8:299–308. doi: 10.1038/nrc2355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Brummel K, Eerkens AL, de Bruyn M, Nijman HW. Tumour-infiltrating lymphocytes: from prognosis to treatment selection. Br J Cancer. 2023;128:451–458. doi: 10.1038/s41416-022-02119-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Paijens ST, Vledder A, de Bruyn M, Nijman HW. Tumor-infiltrating lymphocytes in the immunotherapy era. Cell Mol Immunol. 2021;18:842–859. doi: 10.1038/s41423-020-00565-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Kazemi MH, Sadri M, Najafi A, Rahimi A, Baghernejadan Z, Khorramdelazad H, et al. Tumor-infiltrating lymphocytes for treatment of solid tumors: It takes two to tango? Front Immunol. 2022;13:1018962. doi: 10.3389/fimmu.2022.1018962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Hall M, Liu H, Malafa M, Centeno B, Hodul PJ, Pimiento J, et al. Expansion of tumor-infiltrating lymphocytes (TIL) from human pancreatic tumors. J Immunother cancer. 2016;4:61. doi: 10.1186/s40425-016-0164-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Dudley ME, Gross CA, Langhan MM, Garcia MR, Sherry RM, Yang JC, et al. CD8+ enriched “young” tumor infiltrating lymphocytes can mediate regression of metastatic melanoma. Clin Cancer Res. 2010;16:6122–6131. doi: 10.1158/1078-0432.CCR-10-1297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Baulu E, Gardet C, Chuvin N, Depil S. TCR-engineered T cell therapy in solid tumors: state of the art and perspectives. Sci Adv. 2023;9:eadf3700. doi: 10.1126/sciadv.adf3700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Chiorean EG, Chapuis A, Coveler AL, Yeung CCS, Gooley T, Zhen DB, et al. Phase I study of autologous transgenic T cells expressing high affinity mesothelin-specific T-cell receptor (TCR; FH-TCR TMSLN) in patients with metastatic pancreatic ductal adenocarcinoma (mPDA) J Clin Oncol. 2023;41:TPS779. doi: 10.1200/JCO.2023.41.4_suppl.TPS779. [DOI] [Google Scholar]
- 271.Leidner R, Sanjuan Silva N, Huang H, Sprott D, Zheng C, Shih Y-P, et al. Neoantigen T-cell receptor gene therapy in pancreatic cancer. N Engl J Med. 2022;386:2112–2119. doi: 10.1056/NEJMoa2119662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Jackson HJ, Rafiq S, Brentjens RJ. Driving CAR T-cells forward. Nat Rev Clin Oncol. 2016;13:370–383. doi: 10.1038/nrclinonc.2016.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Sterner RC, Sterner RM. CAR-T cell therapy: current limitations and potential strategies. Blood Cancer J. 2021;11:69. doi: 10.1038/s41408-021-00459-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Schäfer D, Tomiuk S, Küster LN, Al Rawashdeh W, Henze J, Tischler-Höhle G, et al. Identification of CD318, TSPAN8 and CD66c as target candidates for CAR T cell based immunotherapy of pancreatic adenocarcinoma. Nat Commun. 2021;12:1453. doi: 10.1038/s41467-021-21774-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Yan T, Zhu L, Chen J. Current advances and challenges in CAR T-Cell therapy for solid tumors: tumor-associated antigens and the tumor microenvironment. Exp Hematol Oncol. 2023;12:14. doi: 10.1186/s40164-023-00373-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Du H, Hirabayashi K, Ahn S, Kren NP, Montgomery SA, Wang X, et al. Antitumor responses in the absence of toxicity in solid tumors by targeting B7–H3 via chimeric antigen receptor T cells. Cancer Cell. 2019;35:221–237.e8. doi: 10.1016/j.ccell.2019.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Hu G, Li G, Wen W, Ding W, Zhou Z, Zheng Y, et al. Case report: B7–H3 CAR-T therapy partially controls tumor growth in a basal cell carcinoma patient. Front Oncol. 2022;12:956593. doi: 10.3389/fonc.2022.956593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Lo A, Wang L-CS, Scholler J, Monslow J, Avery D, Newick K, et al. Tumor-promoting desmoplasia is disrupted by depleting FAP-expressing stromal cells. Cancer Res. 2015;75:2800–2810. doi: 10.1158/0008-5472.CAN-14-3041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Wang L-CS, Lo A, Scholler J, Sun J, Majumdar RS, Kapoor V, et al. Targeting fibroblast activation protein in tumor stroma with chimeric antigen receptor T cells can inhibit tumor growth and augment host immunity without severe toxicity. Cancer Immunol Res. 2014;2:154–166. doi: 10.1158/2326-6066.CIR-13-0027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.te Velde EA, Franke AC, van Hillegersberg R, Elshof SM, de Weger RW, Borel Rinkes IHM, et al. HER-family gene amplification and expression in resected pancreatic cancer. Eur J Surg Oncol. 2009;35:1098–1104. doi: 10.1016/j.ejso.2009.02.013. [DOI] [PubMed] [Google Scholar]
- 281.Komoto M, Nakata B, Amano R, Yamada N, Yashiro M, Ohira M, et al. HER2 overexpression correlates with survival after curative resection of pancreatic cancer. Cancer Sci. 2009;100:1243–1247. doi: 10.1111/j.1349-7006.2009.01176.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Safran H, Steinhoff M, Mangray S, Rathore R, King TC, Chai L, et al. Overexpression of the HER-2/neu oncogene in pancreatic adenocarcinoma. Am J Clin Oncol. 2001;24:496–499. doi: 10.1097/00000421-200110000-00016. [DOI] [PubMed] [Google Scholar]
- 283.Randall J, Cannon TL, Hunt AL, Nutcharoen A, Johnston L, Wang H, et al. Proteomic quantification of HER-2 abundance and activation in pancreatic ductal adenocarcinoma tumor specimens using reverse phase protein array. J Clin Oncol. 2023;41:e16299–e16299. doi: 10.1200/JCO.2023.41.16_suppl.e16299. [DOI] [Google Scholar]
- 284.Randall J, Hunt AL, Nutcharoen A, Johnston L, Chouraichi S, Wang H, et al. Quantitative proteomic analysis of HER2 protein expression in PDAC tumors. Clin. Proteomics. 2024. p. 24. [DOI] [PMC free article] [PubMed]
- 285.Rosewell Shaw A, Porter CE, Yip T, Mah W-C, McKenna MK, Dysthe M, et al. Oncolytic adeno-immunotherapy modulates the immune system enabling CAR T-cells to cure pancreatic tumors. Commun Biol. 2021;4:368. doi: 10.1038/s42003-021-01914-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Feng K, Liu Y, Guo Y, Qiu J, Wu Z, Dai H, et al. Phase I study of chimeric antigen receptor modified T cells in treating HER2-positive advanced biliary tract cancers and pancreatic cancers. Protein Cell. 2018;9:838–847. doi: 10.1007/s13238-017-0440-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Harder J, Ihorst G, Heinemann V, Hofheinz R, Moehler M, Buechler P, et al. Multicentre phase II trial of trastuzumab and capecitabine in patients with HER2 overexpressing metastatic pancreatic cancer. Br J Cancer. 2012;106:1033–1038. doi: 10.1038/bjc.2012.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Han S-H, Ryu KH, Kwon A-Y. The prognostic impact of HER2 genetic and protein expression in pancreatic carcinoma-HER2 protein and gene in pancreatic cancer. Diagnostics (Basel, Switzerland) 2021;11:653. doi: 10.3390/diagnostics11040653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Yeo D, Giardina C, Saxena P, Rasko JEJ. The next wave of cellular immunotherapies in pancreatic cancer. Mol Ther oncolytics. 2022;24:561–576. doi: 10.1016/j.omto.2022.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Liu Y, Guo Y, Wu Z, Feng K, Tong C, Wang Y, et al. Anti-EGFR chimeric antigen receptor-modified T cells in metastatic pancreatic carcinoma: a phase I clinical trial. Cytotherapy. 2020;22:573–580. doi: 10.1016/j.jcyt.2020.04.088. [DOI] [PubMed] [Google Scholar]
- 291.Meril S, Harush O, Reboh Y, Matikhina T, Barliya T, Cohen CJ. Targeting glycosylated antigens on cancer cells using siglec-7/9-based CAR T-cells. Mol Carcinog. 2020;59:713–723. doi: 10.1002/mc.23213. [DOI] [PubMed] [Google Scholar]
- 292.Rodriguez E, Boelaars K, Brown K, Eveline Li RJ, Kruijssen L, Bruijns SCM, et al. Sialic acids in pancreatic cancer cells drive tumour-associated macrophage differentiation via the Siglec receptors Siglec-7 and Siglec-9. Nat Commun. 2021;12:1270. doi: 10.1038/s41467-021-21550-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Wu J, Wang X, Huang Y, Zhang Y, Su S, Shou H, et al. Targeted glycan degradation potentiates cellular immunotherapy for solid tumors. Proc Natl Acad Sci. 2023;120:e2300366120. doi: 10.1073/pnas.2300366120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Chi X, Yang P, Zhang E, Gu J, Xu H, Li M, et al. Significantly increased anti-tumor activity of carcinoembryonic antigen-specific chimeric antigen receptor T cells in combination with recombinant human IL-12. Cancer Med. 2019;8:4753–4765. doi: 10.1002/cam4.2361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Lee EHJ, Murad JP, Christian L, Gibson J, Yamaguchi Y, Cullen C, et al. Antigen-dependent IL-12 signaling in CAR T cells promotes regional to systemic disease targeting. Nat Commun. 2023;14:4737. doi: 10.1038/s41467-023-40115-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Raj D, Nikolaidi M, Garces I, Lorizio D, Castro NM, Caiafa SG, et al. CEACAM7 is an effective target for CAR T-cell therapy of pancreatic ductal adenocarcinoma. Clin Cancer Res. 2021;27:1538–1552. doi: 10.1158/1078-0432.CCR-19-2163. [DOI] [PubMed] [Google Scholar]
- 297.Tomar S, Zhang J, Khanal M, Hong J, Venugopalan A, Jiang Q, et al. Development of highly effective anti-mesothelin hYP218 chimeric antigen receptor T cells with increased tumor infiltration and persistence for treating solid tumors. Mol Cancer Ther. 2022;21:1195–1206. doi: 10.1158/1535-7163.MCT-22-0073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Ko AH, Jordan AC, Tooker E, Lacey SF, Chang RB, Li Y, et al. Dual targeting of mesothelin and CD19 with chimeric antigen receptor-modified t cells in patients with metastatic pancreatic cancer. Mol Ther. 2020;28:2367–2378. doi: 10.1016/j.ymthe.2020.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Lee HH, Kim I, Kim UK, Choi SS, Kim TY, Lee D, et al. Therapeutic effiacy of T cells expressing chimeric antigen receptor derived from a mesothelin-specific scFv in orthotopic human pancreatic cancer animal models. Neoplasia. 2022;24:98–108. doi: 10.1016/j.neo.2021.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Watanabe K, Luo Y, Da T, Guedan S, Ruella M, Scholler J, et al. Pancreatic cancer therapy with combined mesothelin-redirected chimeric antigen receptor T cells and cytokine-armed oncolytic adenoviruses. JCI Insight. 2018;3:e99573. doi: 10.1172/jci.insight.99573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Beatty GL, O’Hara MH, Lacey SF, Torigian DA, Nazimuddin F, Chen F, et al. Activity of mesothelin-specific chimeric antigen receptor T cells against pancreatic carcinoma metastases in a phase 1 trial. Gastroenterology. 2018;155:29–32. doi: 10.1053/j.gastro.2018.03.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Gao Y, Lin H, Guo D, Cheng S, Zhou Y, Zhang L, et al. Suppression of 4.1R enhances the potency of NKG2D-CAR T cells against pancreatic carcinoma via activating ERK signaling pathway. Oncogenesis. 2021;10:62. doi: 10.1038/s41389-021-00353-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Nath S, Daneshvar K, Roy LD, Grover P, Kidiyoor A, Mosley L, et al. MUC1 induces drug resistance in pancreatic cancer cells via upregulation of multidrug resistance genes. Oncogenesis. 2013;2:e51–e51. doi: 10.1038/oncsis.2013.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Posey AD, Jr, Schwab RD, Boesteanu AC, Steentoft C, Mandel U, Engels B, et al. Engineered CAR T cells targeting the cancer-associated Tn-glycoform of the membrane mucin MUC1 control adenocarcinoma. Immunity. 2016;44:1444–1454. doi: 10.1016/j.immuni.2016.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Henry J, Oh D, Eskew J, Baranda J, Rivera IIR, Dumbrava E, et al. 728 Phase 1 study of P-MUC1C-ALLO1 allogeneic CAR-T cells in patients with epithelial-derived cancers. J Immunother Cancer. 2022;10:A761. [Google Scholar]
- 306.Gutierrez R, Shah PD, Hamid O, Garfall AL, Posey A, Bishop MR, et al. Phase I experience with first in class TnMUC1 targeted chimeric antigen receptor T-cells in patients with advanced TnMUC1 positive solid tumors. J Clin Oncol. 2021;39:e14513–e14513. doi: 10.1200/JCO.2021.39.15_suppl.e14513. [DOI] [Google Scholar]
- 307.Wang Y, Chen M, Wu Z, Tong C, Dai H, Guo Y, et al. CD133-directed CAR T cells for advanced metastasis malignancies: a phase I trial. Oncoimmunology. 2018;7:e1440169. doi: 10.1080/2162402X.2018.1440169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Abate-Daga D, Lagisetty KH, Tran E, Zheng Z, Gattinoni L, Yu Z, et al. A novel chimeric antigen receptor against prostate stem cell antigen mediates tumor destruction in a humanized mouse model of pancreatic cancer. Hum Gene Ther. 2014;25:1003–1012. doi: 10.1089/hum.2013.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 309.Dacek MM, Kurtz KG, Wallisch P, Pierre SA, Khayat S, Bourne CM, et al. Potentiating antibody-dependent killing of cancers with CAR T cells secreting CD47-SIRPα checkpoint blocker. Blood. 2023;141:2003–2015. doi: 10.1182/blood.2022016101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 310.Golubovskaya V, Berahovich R, Zhou H, Xu S, Harto H, Li L, et al. CD47-CAR-T cells effectively kill target cancer cells and block pancreatic tumor growth. Cancers (Basel) 2017;9:139. doi: 10.3390/cancers9100139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Qi C, Gong J, Li J, Liu D, Qin Y, Ge S, et al. Claudin18.2-specific CAR T cells in gastrointestinal cancers: phase 1 trial interim results. Nat Med. 2022;28:1189–1198. doi: 10.1038/s41591-022-01800-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Qi C, Xie T, Zhou J, Wang X, Gong J, Zhang X, et al. CT041 CAR T cell therapy for Claudin18.2-positive metastatic pancreatic cancer. J Hematol Oncol. 2023;16:102. doi: 10.1186/s13045-023-01491-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Zhu G, Foletti D, Liu X, Ding S, Melton Witt J, Hasa-Moreno A, et al. Targeting CLDN18.2 by CD3 bispecific and ADC modalities for the treatments of gastric and pancreatic cancer. Sci Rep. 2019;9:8420. doi: 10.1038/s41598-019-44874-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Jiang H, Shi Z, Wang P, Wang C, Yang L, Du G, et al. Claudin18.2-specific chimeric antigen receptor engineered t cells for the treatment of gastric cancer. J Natl Cancer Inst. 2019;111:409–418. doi: 10.1093/jnci/djy134. [DOI] [PubMed] [Google Scholar]
- 315.Zhan X, Wang B, Li Z, Li J, Wang H, Chen L, et al. Phase I trial of Claudin 18.2-specific chimeric antigen receptor T cells for advanced gastric and pancreatic adenocarcinoma. J Clin Oncol. 2019;37:2509. doi: 10.1200/JCO.2019.37.15_suppl.2509. [DOI] [Google Scholar]
- 316.Qi C, Liu C, Gong J, Li J, Liu D, Ge S, et al. Safety, tolerability, and preliminary efficacy results in patients with advanced gastric/gastroesophageal junction adenocarcinoma from a phase Ib/II study of CLDN18.2 CAR T-cell therapy (CT041) J Clin Oncol. 2022;40:4017. doi: 10.1200/JCO.2022.40.16_suppl.4017. [DOI] [Google Scholar]
- 317.Daher M, Rezvani K. Outlook for new CAR-based therapies with a focus on CAR NK cells: what lies beyond CAR-engineered T cells in the race against cancer. Cancer Discov. 2021;11:45–58. doi: 10.1158/2159-8290.CD-20-0556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 318.Pan K, Farrukh H, Chittepu VCSR, Xu H, Pan C, Zhu Z. CAR race to cancer immunotherapy: from CAR T, CAR NK to CAR macrophage therapy. J Exp Clin Cancer Res. 2022;41:119. doi: 10.1186/s13046-022-02327-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 319.Mansour AG, Teng K-Y, Lu T, Barr T, Yu J. Chapter 12 - CAR-NK cell immunotherapy: Development and challenges toward an off-the-shelf product. In: Bonavida B, Jewett ABT-S and C of NKI, editors. Successes Challenges NK Immunother Break Toler to Cancer Resist. Academic Press; 2021. p. 213–30.
- 320.Liu E, Tong Y, Dotti G, Shaim H, Savoldo B, Mukherjee M, et al. Cord blood NK cells engineered to express IL-15 and a CD19-targeted CAR show long-term persistence and potent antitumor activity. Leukemia. 2018;32:520–531. doi: 10.1038/leu.2017.226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 321.Wang K, Wang L, Wang Y, Xiao L, Wei J, Hu Y, et al. Reprogramming natural killer cells for cancer therapy. Mol Ther. 2024; [DOI] [PubMed]
- 322.Xia N, Haopeng P, Gong JU, Lu J, Chen Z, Zheng Y, et al. Robo1-specific CAR-NK immunotherapy enhances efficacy of 125I seed brachytherapy in an orthotopic mouse model of human pancreatic carcinoma. Anticancer Res Greece. 2019;39:5919–5925. doi: 10.21873/anticanres.13796. [DOI] [PubMed] [Google Scholar]
- 323.Teng K-Y, Mansour AG, Zhu Z, Li Z, Tian L, Ma S, et al. Off-the-shelf prostate stem cell antigen-directed chimeric antigen receptor natural killer cell therapy to treat pancreatic cancer. Gastroenterology. 2022;162:1319–1333. doi: 10.1053/j.gastro.2021.12.281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 324.Da Y, Liu Y, Hu Y, Liu W, Ma J, Lu N, et al. STING agonist cGAMP enhances anti-tumor activity of CAR-NK cells against pancreatic cancer. Oncoimmunology. 2022;11:2054105. doi: 10.1080/2162402X.2022.2054105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.Nianias A, Themeli M. Induced pluripotent stem cell (iPSC)–derived lymphocytes for adoptive cell immunotherapy: recent advances and challenges. Curr Hematol Malig Rep. 2019;14:261–268. doi: 10.1007/s11899-019-00528-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.Zhu T, Wu X, Liao Y, Yan Y, Yu M, Wang L, et al. The role of innate immune cells as modulators of the tumor microenvironment in the metastasis and treatment of pancreatic cancer. Clin Cancer Bull. 2023;2:2. doi: 10.1007/s44272-023-00005-5. [DOI] [Google Scholar]
- 327.Gao X, Mi Y, Guo N, Xu H, Xu L, Gou X, et al. Cytokine-induced killer cells as pharmacological tools for cancer immunotherapy. Front Immunol. 2017;8:774. doi: 10.3389/fimmu.2017.00774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 328.Chung MJ, Park JY, Bang S, Park SW, Song SY. Phase II clinical trial of ex vivo-expanded cytokine-induced killer cells therapy in advanced pancreatic cancer. Cancer Immunol Immunother. 2014;63:939–946. doi: 10.1007/s00262-014-1566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 329.Wang M, Shi S, Qi J, Tang X, Tian J. S-1 plus CIK as second-line treatment for advanced pancreatic cancer. Med Oncol. 2013;30:747. doi: 10.1007/s12032-013-0747-9. [DOI] [PubMed] [Google Scholar]
- 330.Wang Z, Liu Y, Li R, Shang Y, Zhang Y, Zhao L, et al. Autologous cytokine-induced killer cell transfusion increases overall survival in advanced pancreatic cancer. J Hematol Oncol. 2016;9:6. doi: 10.1186/s13045-016-0237-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 331.Korman AJ, Garrett-Thomson SC, Lonberg N. The foundations of immune checkpoint blockade and the ipilimumab approval decennial. Nat Rev Drug Discov. 2022;21:509–528. doi: 10.1038/s41573-021-00345-8. [DOI] [PubMed] [Google Scholar]
- 332.Li X, Gulati M, Larson AC, Solheim JC, Jain M, Kumar S, et al. Immune checkpoint blockade in pancreatic cancer: trudging through the immune desert. Semin Cancer Biol. 2022;86:14–27. doi: 10.1016/j.semcancer.2022.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 333.Keir ME, Butte MJ, Freeman GJ, Sharpe AH. PD-1 and its ligands in tolerance and immunity. Annu Rev Immunol. 2008;26:677–704. doi: 10.1146/annurev.immunol.26.021607.090331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 334.Cercek A, Lumish M, Sinopoli J, Weiss J, Shia J, Lamendola-Essel M, et al. PD-1 blockade in mismatch repair-deficient, locally advanced rectal cancer. N Engl J Med. 2022;386:2363–2376. doi: 10.1056/NEJMoa2201445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 335.Migden MR, Rischin D, Schmults CD, Guminski A, Hauschild A, Lewis KD, et al. PD-1 blockade with cemiplimab in advanced cutaneous squamous-cell carcinoma. N Engl J Med. 2018;379:341–351. doi: 10.1056/NEJMoa1805131. [DOI] [PubMed] [Google Scholar]
- 336.Zhang Y, Chen X, Mo S, Ma H, Lu Z, Yu S, et al. PD-L1 and PD-L2 expression in pancreatic ductal adenocarcinoma and their correlation with immune infiltrates and DNA damage response molecules. J Pathol Clin Res. 2022;8:257–267. doi: 10.1002/cjp2.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Chouari T, La Costa FS, Merali N, Jessel M-D, Sivakumar S, Annels N, et al. Advances in immunotherapeutics in pancreatic ductal adenocarcinoma. Cancers (Basel) 2023;15:4265. doi: 10.3390/cancers15174265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 338.Chaib S, López-Domínguez JA, Lalinde-Gutiérrez M, Prats N, Marin I, Boix O, et al. The efficacy of chemotherapy is limited by intratumoral senescent cells expressing PD-L2. Nat Cancer. 2024;5:448–462. doi: 10.1038/s43018-023-00712-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 339.Wang X, Li X, Wei X, Jiang H, Lan C, Yang S, et al. PD-L1 is a direct target of cancer-FOXP3 in pancreatic ductal adenocarcinoma (PDAC), and combined immunotherapy with antibodies against PD-L1 and CCL5 is effective in the treatment of PDAC. Signal Transduct Target Ther. 2020;5:38. doi: 10.1038/s41392-020-0144-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 340.Zhang X, Lao M, Xu J, Duan Y, Yang H, Li M, et al. Combination cancer immunotherapy targeting TNFR2 and PD-1/PD-L1 signaling reduces immunosuppressive effects in the microenvironment of pancreatic tumors. J Immunother Cancer. 2022;10:e003982. doi: 10.1136/jitc-2021-003982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 341.Panni RZ, Herndon JM, Zuo C, Hegde S, Hogg GD, Knolhoff BL, et al. Agonism of CD11b reprograms innate immunity to sensitize pancreatic cancer to immunotherapies. Sci Transl Med. 2019;11:eaau9240. doi: 10.1126/scitranslmed.aau9240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 342.Piper M, Hoen M, Darragh LB, Knitz MW, Nguyen D, Gadwa J, et al. Simultaneous targeting of PD-1 and IL-2Rβγ with radiation therapy inhibits pancreatic cancer growth and metastasis. Cancer Cell. 2023;41:950–969.e6. doi: 10.1016/j.ccell.2023.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 343.Bengsch F, Knoblock DM, Liu A, McAllister F, Beatty GL. CTLA-4/CD80 pathway regulates T cell infiltration into pancreatic cancer. Cancer Immunol Immunother. 2017;66:1609–1617. doi: 10.1007/s00262-017-2053-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 344.Ware MB, Phillips M, McQuinn C, Zaidi MY, Knochelmann HM, Greene E, et al. Dual IL-6 and CTLA-4 blockade regresses pancreatic tumors in a T cell and CXCR3-dependent manner. JCI Insight. 2023;8:e155006. doi: 10.1172/jci.insight.155006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Seifert L, Plesca I, Müller L, Sommer U, Heiduk M, von Renesse J, et al. LAG-3-expressing tumor-infiltrating T cells are associated with reduced disease-free survival in pancreatic cancer. Cancers (Basel) 2021;13:1297. doi: 10.3390/cancers13061297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 346.Gulhati P, Schalck A, Jiang S, Shang X, Wu C-J, Hou P, et al. Targeting T cell checkpoints 41BB and LAG3 and myeloid cell CXCR1/CXCR2 results in antitumor immunity and durable response in pancreatic cancer. Nat Cancer. 2023;4:62–80. doi: 10.1038/s43018-022-00500-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 347.Farhangnia P, Akbarpour M, Yazdanifar M, Aref AR, Delbandi A-A, Rezaei N. Advances in therapeutic targeting of immune checkpoints receptors within the CD96-TIGIT axis: clinical implications and future perspectives. Expert Rev Clin Immunol. 2022; [DOI] [PubMed]
- 348.Heiduk M, Klimova A, Reiche C, Digomann D, Beer C, Aust DE, et al. TIGIT expression delineates T-cell populations with distinct functional and prognostic impact in pancreatic cancer. Clin Cancer Res. 2023;29:2638–2650. doi: 10.1158/1078-0432.CCR-23-0258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 349.Banta KL, Xu X, Chitre AS, Au-Yeung A, Takahashi C, O’Gorman WE, et al. Mechanistic convergence of the TIGIT and PD-1 inhibitory pathways necessitates co-blockade to optimize anti-tumor CD8(+) T cell responses. Immunity. 2022;55:512–526.e9. doi: 10.1016/j.immuni.2022.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 350.Pearce H, Croft W, Nicol SM, Margielewska-Davies S, Powell R, Cornall R, et al. Tissue-resident memory T cells in pancreatic ductal adenocarcinoma coexpress PD-1 and TIGIT and functional inhibition is reversible by dual antibody blockade. Cancer Immunol Res. 2023;11:435–449. doi: 10.1158/2326-6066.CIR-22-0121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 351.Freed-Pastor WA, Lambert LJ, Ely ZA, Pattada NB, Bhutkar A, Eng G, et al. The CD155/TIGIT axis promotes and maintains immune evasion in neoantigen-expressing pancreatic cancer. Cancer Cell. 2021;39:1342–1360.e14. doi: 10.1016/j.ccell.2021.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 352.Peng H, Li L, Zuo C, Chen MY, Zhang X, Myers NB, et al. Combination TIGIT/PD-1 blockade enhances the efficacy of neoantigen vaccines in a model of pancreatic cancer. Front Immunol. 2022;13:1039226. doi: 10.3389/fimmu.2022.1039226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 353.Werba G, Weissinger D, Kawaler EA, Zhao E, Kalfakakou D, Dhara S, et al. Single-cell RNA sequencing reveals the effects of chemotherapy on human pancreatic adenocarcinoma and its tumor microenvironment. Nat Commun. 2023;14:797. doi: 10.1038/s41467-023-36296-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 354.Martin AS, Molloy M, Ugolkov A, von Roemeling RW, Noelle RJ, Lewis LD, et al. VISTA expression and patient selection for immune-based anticancer therapy. Front Immunol. 2023;14:1086102. doi: 10.3389/fimmu.2023.1086102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 355.ElTanbouly MA, Zhao Y, Nowak E, Li J, Schaafsma E, Le Mercier I, et al. VISTA is a checkpoint regulator for naïve T cell quiescence and peripheral tolerance. Science. 2020;367. [DOI] [PMC free article] [PubMed]
- 356.Rezagholizadeh F, Tajik F, Talebi M, Taha SR, Shariat Zadeh M, Farhangnia P, et al. Unraveling the potential of CD8, CD68, and VISTA as diagnostic and prognostic markers in patients with pancreatic ductal adenocarcinoma. Front Immunol. 2024;15:1283364. doi: 10.3389/fimmu.2024.1283364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 357.Hou Z, Pan Y, Fei Q, Lin Y, Zhou Y, Liu Y, et al. Prognostic significance and therapeutic potential of the immune checkpoint VISTA in pancreatic cancer. J Cancer Res Clin Oncol. 2021;147:517–531. doi: 10.1007/s00432-020-03463-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 358.Blando J, Sharma A, Higa MG, Zhao H, Vence L, Yadav SS, et al. Comparison of immune infiltrates in melanoma and pancreatic cancer highlights VISTA as a potential target in pancreatic cancer. Proc Natl Acad Sci. 2019;116:1692–1697. doi: 10.1073/pnas.1811067116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 359.Allard B, Allard D, Buisseret L, Stagg J. The adenosine pathway in immuno-oncology. Nat Rev Clin Oncol. 2020;17:611–629. doi: 10.1038/s41571-020-0382-2. [DOI] [PubMed] [Google Scholar]
- 360.Xia C, Yin S, To KKW, Fu L. CD39/CD73/A2AR pathway and cancer immunotherapy. Mol Cancer. 2023;22:44. doi: 10.1186/s12943-023-01733-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 361.Chen Q, Pu N, Yin H, Zhang J, Zhao G, Lou W, et al. CD73 acts as a prognostic biomarker and promotes progression and immune escape in pancreatic cancer. J Cell Mol Med. 2020;24:8674–8686. doi: 10.1111/jcmm.15500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 362.Jacoberger-Foissac C, Cousineau I, Bareche Y, Allard D, Chrobak P, Allard B, et al. CD73 inhibits cGAS–STING and cooperates with CD39 to promote pancreatic cancer. Cancer Immunol Res. 2023;11:56–71. doi: 10.1158/2326-6066.CIR-22-0260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 363.Miller RA, Luke JJ, Hu S, Mahabhashyam S, Jones WB, Marron T, et al. Anti-CD73 antibody activates human B cells, enhances humoral responses and induces redistribution of B cells in patients with cancer. J Immunother cancer. 2022;10:e005802. doi: 10.1136/jitc-2022-005802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 364.Bendell J, LoRusso P, Overman M, Noonan AM, Kim D-W, Strickler JH, et al. First-in-human study of oleclumab, a potent, selective anti-CD73 monoclonal antibody, alone or in combination with durvalumab in patients with advanced solid tumors. Cancer Immunol Immunother. 2023;72:2443–2458. doi: 10.1007/s00262-023-03430-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 365.Farhangnia P, Ghomi SM, Akbarpour M, Delbandi A-A. Bispecific antibodies targeting CTLA-4: game-changer troopers in cancer immunotherapy. Front Immunol. 2023;14:1155778. doi: 10.3389/fimmu.2023.1155778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 366.Long AW, Xu H, Santich BH, Guo H, Hoseini SS, de Stanchina E, et al. Heterodimerization of T cell engaging bispecific antibodies to enhance specificity against pancreatic ductal adenocarcinoma. J Hematol Oncol. 2024;17:20. doi: 10.1186/s13045-024-01538-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 367.Segal NH, Melero I, Moreno V, Steeghs N, Marabelle A, Rohrberg K, et al. CEA-CD3 bispecific antibody cibisatamab with or without atezolizumab in patients with CEA-positive solid tumours: results of two multi-institutional Phase 1 trials. Nat Commun. 2024;15:4091. doi: 10.1038/s41467-024-48479-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 368.Schram AM, Odintsov I, Espinosa-Cotton M, Khodos I, Sisso WJ, Mattar MS, et al. Zenocutuzumab, a HER2xHER3 bispecific antibody, is effective therapy for tumors driven by NRG1 gene rearrangements. Cancer Discov. 2022;12:1233–1247. doi: 10.1158/2159-8290.CD-21-1119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 369.Thakur A, Ung J, Tomaszewski EN, Schienschang A, LaBrie TM, Schalk DL, et al. Priming of pancreatic cancer cells with bispecific antibody armed activated T cells sensitizes tumors for enhanced chemoresponsiveness. Oncoimmunology. 2021;10:1930883. doi: 10.1080/2162402X.2021.1930883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 370.Oberg H-H, Peipp M, Kellner C, Sebens S, Krause S, Petrick D, et al. Novel bispecific antibodies increase γδ T-cell cytotoxicity against pancreatic cancer cells. Cancer Res. 2014;74:1349–1360. doi: 10.1158/0008-5472.CAN-13-0675. [DOI] [PubMed] [Google Scholar]
- 371.Akce M, Hu-Lieskovan S, Reilley M, Strauss JF, Specht JM, Stein MN, et al. A phase 1 multiple-ascending dose study to evaluate the safety and tolerability of XmAb23104 (PD-1 x ICOS) in subjects with selected advanced solid tumors (DUET-3) J Clin Oncol. 2022;40:2604. doi: 10.1200/JCO.2022.40.16_suppl.2604. [DOI] [Google Scholar]
- 372.Kvarnhammar AM, Veitonmäki N, Hägerbrand K, Dahlman A, Smith KE, Fritzell S, et al. The CTLA-4 x OX40 bispecific antibody ATOR-1015 induces anti-tumor effects through tumor-directed immune activation. J Immunother Cancer. 2019;7:103. doi: 10.1186/s40425-019-0570-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 373.Gang J, Guo S, Zhang Y, Ma Y, Guo X, Zhou X, et al. A phase II study of KN046 monotherapy as 2nd line and above treatment for unresectable locally advanced or metastatic pancreatic ductal adenocarcinoma (PDAC) J Clin Oncol. 2022;40:e16305–e16305. doi: 10.1200/JCO.2022.40.16_suppl.e16305. [DOI] [Google Scholar]
- 374.Lin MJ, Svensson-Arvelund J, Lubitz GS, Marabelle A, Melero I, Brown BD, et al. Cancer vaccines: the next immunotherapy frontier. Nat Cancer. 2022;3:911–926. doi: 10.1038/s43018-022-00418-6. [DOI] [PubMed] [Google Scholar]
- 375.Hardacre JM, Mulcahy M, Small W, Talamonti M, Obel J, Krishnamurthi S, et al. Addition of algenpantucel-L immunotherapy to standard adjuvant therapy for pancreatic cancer: a phase 2 study. J Gastrointest Surg. 2013;17:91–94. doi: 10.1007/s11605-012-2064-6. [DOI] [PubMed] [Google Scholar]
- 376.Hewitt DB, Nissen N, Hatoum H, Musher B, Seng J, Coveler AL, et al. A phase 3 randomized clinical trial of chemotherapy with or without algenpantucel-L (hyperacute-pancreas) immunotherapy in subjects with borderline resectable or locally advanced unresectable pancreatic cancer. Ann Surg. 2022;275:45–53. doi: 10.1097/SLA.0000000000004669. [DOI] [PubMed] [Google Scholar]
- 377.Jaffee EM, Hruban RH, Biedrzycki B, Laheru D, Schepers K, Sauter PR, et al. Novel allogeneic granulocyte-macrophage colony-stimulating factor-secreting tumor vaccine for pancreatic cancer: a phase I trial of safety and immune activation. J Clin Oncol. 2001;19:145–156. doi: 10.1200/JCO.2001.19.1.145. [DOI] [PubMed] [Google Scholar]
- 378.Laheru D, Lutz E, Burke J, Biedrzycki B, Solt S, Onners B, et al. Allogeneic granulocyte macrophage colony-stimulating factor-secreting tumor immunotherapy alone or in sequence with cyclophosphamide for metastatic pancreatic cancer: a pilot study of safety, feasibility, and immune activation. Clin Cancer Res. 2008;14:1455–1463. doi: 10.1158/1078-0432.CCR-07-0371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 379.Le DT, Lutz E, Uram JN, Sugar EA, Onners B, Solt S, et al. Evaluation of ipilimumab in combination with allogeneic pancreatic tumor cells transfected with a GM-CSF gene in previously treated pancreatic cancer. J Immunother. 2013;36:382–389. doi: 10.1097/CJI.0b013e31829fb7a2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 380.Lutz ER, Wu AA, Bigelow E, Sharma R, Mo G, Soares K, et al. Immunotherapy converts nonimmunogenic pancreatic tumors into immunogenic foci of immune regulation. Cancer Immunol Res. 2014;2:616–631. doi: 10.1158/2326-6066.CIR-14-0027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 381.Heumann T, Judkins C, Li K, Lim SJ, Hoare J, Parkinson R, et al. A platform trial of neoadjuvant and adjuvant antitumor vaccination alone or in combination with PD-1 antagonist and CD137 agonist antibodies in patients with resectable pancreatic adenocarcinoma. Nat Commun. 2023;14:3650. doi: 10.1038/s41467-023-39196-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 382.McCormick KA, Coveler AL, Rossi GR, Vahanian NN, Link C, Chiorean EG. Pancreatic cancer: update on immunotherapies and algenpantucel-L. Hum Vaccin Immunother. 2016;12:563–575. doi: 10.1080/21645515.2015.1093264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 383.Rong Y, Qin X, Jin D, Lou W, Wu L, Wang D, et al. A phase I pilot trial of MUC1-peptide-pulsed dendritic cells in the treatment of advanced pancreatic cancer. Clin Exp Med. 2012;12:173–180. doi: 10.1007/s10238-011-0159-0. [DOI] [PubMed] [Google Scholar]
- 384.Koido S, Homma S, Okamoto M, Takakura K, Mori M, Yoshizaki S, et al. Treatment with chemotherapy and dendritic cells pulsed with multiple Wilms’ tumor 1 (WT1)-specific MHC class I/II-restricted epitopes for pancreatic cancer. Clin Cancer Res. 2014;20:4228–4239. doi: 10.1158/1078-0432.CCR-14-0314. [DOI] [PubMed] [Google Scholar]
- 385.Mayanagi S, Kitago M, Sakurai T, Matsuda T, Fujita T, Higuchi H, et al. Phase I pilot study of Wilms tumor gene 1 peptide-pulsed dendritic cell vaccination combined with gemcitabine in pancreatic cancer. Cancer Sci. 2015;106:397–406. doi: 10.1111/cas.12621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 386.Takakura K, Koido S, Kan S, Yoshida K, Mori M, Hirano Y, et al. Prognostic markers for patient outcome following vaccination with multiple MHC Class I/II-restricted WT1 peptide-pulsed dendritic cells plus chemotherapy for pancreatic cancer. Anticancer Res Greece. 2015;35:555–562. [PubMed] [Google Scholar]
- 387.Tsukinaga S, Kajihara M, Takakura K, Ito Z, Kanai T, Saito K, et al. Prognostic significance of plasma interleukin-6/-8 in pancreatic cancer patients receiving chemoimmunotherapy. World J Gastroenterol. 2015;21:11168–11178. doi: 10.3748/wjg.v21.i39.11168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 388.Yanagisawa R, Koizumi T, Koya T, Sano K, Koido S, Nagai K, et al. WT1-pulsed dendritic cell vaccine combined with chemotherapy for resected pancreatic cancer in a phase I study. Anticancer Res Greece. 2018;38:2217–2225. doi: 10.21873/anticanres.12464. [DOI] [PubMed] [Google Scholar]
- 389.Kobayashi M, Shimodaira S, Nagai K, Ogasawara M, Takahashi H, Abe H, et al. Prognostic factors related to add-on dendritic cell vaccines on patients with inoperable pancreatic cancer receiving chemotherapy: a multicenter analysis. Cancer Immunol Immunother. 2014;63:797–806. doi: 10.1007/s00262-014-1554-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 390.Lau SP, van Montfoort N, Kinderman P, Lukkes M, Klaase L, van Nimwegen M, et al. Dendritic cell vaccination and CD40-agonist combination therapy licenses T cell-dependent antitumor immunity in a pancreatic carcinoma murine model. J Immunother Cancer. 2020;8:e000772. doi: 10.1136/jitc-2020-000772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 391.Kimura Y, Tsukada J, Tomoda T, Takahashi H, Imai K, Shimamura K, et al. Clinical and immunologic evaluation of dendritic cell-based immunotherapy in combination with gemcitabine and/or S-1 in patients with advanced pancreatic carcinoma. Pancreas. 2012;41:195–205. doi: 10.1097/MPA.0b013e31822398c6. [DOI] [PubMed] [Google Scholar]
- 392.Nagaraj S, Ziske C, Strehl J, Messmer D, Sauerbruch T, Schmidt-Wolf IGH. Dendritic cells pulsed with alpha-galactosylceramide induce anti-tumor immunity against pancreatic cancer in vivo. Int Immunol. 2006;18:1279–1283. doi: 10.1093/intimm/dxl059. [DOI] [PubMed] [Google Scholar]
- 393.Bernhardt SL, Gjertsen MK, Trachsel S, Møller M, Eriksen JA, Meo M, et al. Telomerase peptide vaccination of patients with non-resectable pancreatic cancer: a dose escalating phase I/II study. Br J Cancer. 2006;95:1474–1482. doi: 10.1038/sj.bjc.6603437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 394.Middleton G, Silcocks P, Cox T, Valle J, Wadsley J, Propper D, et al. Gemcitabine and capecitabine with or without telomerase peptide vaccine GV1001 in patients with locally advanced or metastatic pancreatic cancer (TeloVac): an open-label, randomised, phase 3 trial. Lancet Oncol. 2014;15:829–840. doi: 10.1016/S1470-2045(14)70236-0. [DOI] [PubMed] [Google Scholar]
- 395.Asahara S, Takeda K, Yamao K, Maguchi H, Yamaue H. Phase I/II clinical trial using HLA-A24-restricted peptide vaccine derived from KIF20A for patients with advanced pancreatic cancer. J Transl Med. 2013;11:291. doi: 10.1186/1479-5876-11-291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 396.Pant S, Wainberg ZA, Weekes CD, Furqan M, Kasi PM, Devoe CE, et al. Lymph-node-targeted, mKRAS-specific amphiphile vaccine in pancreatic and colorectal cancer: the phase 1 AMPLIFY-201 trial. Nat Med. 2024 [DOI] [PMC free article] [PubMed]
- 397.Cappello P, Rolla S, Chiarle R, Principe M, Cavallo F, Perconti G, et al. Vaccination with ENO1 DNA prolongs survival of genetically engineered mice with pancreatic cancer. Gastroenterology. 2013;144:1098–1106. doi: 10.1053/j.gastro.2013.01.020. [DOI] [PubMed] [Google Scholar]
- 398.Mandili G, Curcio C, Bulfamante S, Follia L, Ferrero G, Mazza E, et al. In pancreatic cancer, chemotherapy increases antitumor responses to tumor-associated antigens and potentiates DNA vaccination. J Immunother Cancer. 2020;8:e001071. doi: 10.1136/jitc-2020-001071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 399.Gong Y, Zhou Q, Liao Y, Mai C, Chen T, Tang Y, et al. Optimized construction of MUC1-VNTRn DNA vaccine and its anti-pancreatic cancer efficacy. Oncol Lett. 2017;13:2198–2206. doi: 10.3892/ol.2017.5717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 400.Geng F, Dong L, Bao X, Guo Q, Guo J, Zhou Y, et al. CAFs/tumor cells co-targeting DNA vaccine in combination with low-dose gemcitabine for the treatment of Panc02 murine pancreatic cancer. Mol Ther Oncolytics. 2022;26:304–313. doi: 10.1016/j.omto.2022.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 401.Rojas LA, Sethna Z, Soares KC, Olcese C, Pang N, Patterson E, et al. Personalized RNA neoantigen vaccines stimulate T cells in pancreatic cancer. Nature. 2023;618:144–150. doi: 10.1038/s41586-023-06063-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 402.Travieso T, Li J, Mahesh S, Mello JDFRE, Blasi M. The use of viral vectors in vaccine development. NPJ Vaccines. 2022;7:75. doi: 10.1038/s41541-022-00503-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 403.Raman V, Howell LM, Bloom SMK, Hall CL, Wetherby VE, Minter LM, et al. Intracellular Salmonella delivery of an exogenous immunization antigen refocuses CD8 T cells against cancer cells, eliminates pancreatic tumors and forms antitumor immunity. Front Immunol. 2023;14:1228532. doi: 10.3389/fimmu.2023.1228532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 404.Schmitz-Winnenthal FH, Hohmann N, Niethammer AG, Friedrich T, Lubenau H, Springer M, et al. Anti-angiogenic activity of VXM01, an oral T-cell vaccine against VEGF receptor 2, in patients with advanced pancreatic cancer: a randomized, placebo-controlled, phase 1 trial. Oncoimmunology. 2015;4:e1001217. doi: 10.1080/2162402X.2014.1001217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 405.Kooreman NG, Kim Y, de Almeida PE, Termglinchan V, Diecke S, Shao N-Y, et al. Autologous iPSC-based vaccines elicit anti-tumor responses in vivo. Cell Stem Cell. 2018;22:501–513.e7. doi: 10.1016/j.stem.2018.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 406.Ouyang X, Liu Y, Zhou Y, Guo J, Wei T-T, Liu C, et al. Antitumor effects of iPSC-based cancer vaccine in pancreatic cancer. Stem Cell Rep. 2021;16:1468–1477. doi: 10.1016/j.stemcr.2021.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 407.Beatty GL, Chiorean EG, Fishman MP, Saboury B, Teitelbaum UR, Sun W, et al. CD40 agonists alter tumor stroma and show efficacy against pancreatic carcinoma in mice and humans. Science (80-) 2011;331:1612–1616. doi: 10.1126/science.1198443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 408.Morrison AH, Diamond MS, Hay CA, Byrne KT, Vonderheide RH. Sufficiency of CD40 activation and immune checkpoint blockade for T cell priming and tumor immunity. Proc Natl Acad Sci. 2020;117:8022–8031. doi: 10.1073/pnas.1918971117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 409.Byrne KT, Vonderheide RH. CD40 stimulation obviates innate sensors and drives T cell immunity in cancer. Cell Rep. 2016;15:2719–2732. doi: 10.1016/j.celrep.2016.05.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 410.O’Hara MH, O’Reilly EM, Varadhachary G, Wolff RA, Wainberg ZA, Ko AH, et al. CD40 agonistic monoclonal antibody APX005M (sotigalimab) and chemotherapy, with or without nivolumab, for the treatment of metastatic pancreatic adenocarcinoma: an open-label, multicentre, phase 1b study. Lancet Oncol. 2021;22:118–131. doi: 10.1016/S1470-2045(20)30532-5. [DOI] [PubMed] [Google Scholar]
- 411.Sockolosky JT, Dougan M, Ingram JR, Ho CCM, Kauke MJ, Almo SC, et al. Durable antitumor responses to CD47 blockade require adaptive immune stimulation. Proc Natl Acad Sci. 2016;113:E2646–E2654. doi: 10.1073/pnas.1604268113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 412.Liu M, O’Connor RS, Trefely S, Graham K, Snyder NW, Beatty GL. Metabolic rewiring of macrophages by CpG potentiates clearance of cancer cells and overcomes tumor-expressed CD47−mediated ‘don’t-eat-me’ signal. Nat Immunol. 2019;20:265–275. doi: 10.1038/s41590-018-0292-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 413.Okada H, Takahashi K, Yaku H, Kobiyama K, Iwaisako K, Zhao X, et al. In situ vaccination using unique TLR9 ligand K3-SPG induces long-lasting systemic immune response and synergizes with systemic and local immunotherapy. Sci Rep. 2022;12:2132. doi: 10.1038/s41598-022-05702-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 414.Khalil DN, Suek N, Campesato LF, Budhu S, Redmond D, Samstein RM, et al. In situ vaccination with defined factors overcomes T cell exhaustion in distant tumors. J Clin Invest. 2019;129:3435–3447. doi: 10.1172/JCI128562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 415.Urman A, Ding Y, Wang H, Burkhart RA, He J, Jaffee EM, et al. Abstract CT134: Safety and immunologic impact of neoadjuvant/adjuvant GM-CSF-secreting allogenic pancreatic tumor cell vaccine (GVAX) combined with cyclophosphamide, pembrolizumab, and macrophage-targeting CSF1R inhibitor IMC-CS4 in pancreatic adenocarcin. Cancer Res. 2024;84:CT134. doi: 10.1158/1538-7445.AM2024-CT134. [DOI] [Google Scholar]
- 416.Astuti Y, Raymant M, Quaranta V, Clarke K, Abudula M, Smith O, et al. Efferocytosis reprograms the tumor microenvironment to promote pancreatic cancer liver metastasis. Nat Cancer. 2024; [DOI] [PMC free article] [PubMed]
- 417.Xue M, Zhu Y, Jiang Y, Han L, Shi M, Su R, et al. Schwann cells regulate tumor cells and cancer-associated fibroblasts in the pancreatic ductal adenocarcinoma microenvironment. Nat Commun. 2023;14:4600. doi: 10.1038/s41467-023-40314-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 418.Huang H, Zhang Y, Gallegos V, Sorrelle N, Zaid MM, Toombs J, et al. Targeting TGFβR2-mutant tumors exposes vulnerabilities to stromal TGFβ blockade in pancreatic cancer. EMBO Mol Med. 2019;11:e10515. doi: 10.15252/emmm.201910515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 419.Grauel AL, Nguyen B, Ruddy D, Laszewski T, Schwartz S, Chang J, et al. TGFβ-blockade uncovers stromal plasticity in tumors by revealing the existence of a subset of interferon-licensed fibroblasts. Nat Commun. 2020;11:6315. doi: 10.1038/s41467-020-19920-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 420.Sherman MH, Yu RT, Engle DD, Ding N, Atkins AR, Tiriac H, et al. Vitamin D receptor-mediated stromal reprogramming suppresses pancreatitis and enhances pancreatic cancer therapy. Cell. 2014;159:80–93. doi: 10.1016/j.cell.2014.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 421.Steele NG, Biffi G, Kemp SB, Zhang Y, Drouillard D, Syu L, et al. Inhibition of hedgehog signaling alters fibroblast composition in pancreatic cancer. Clin Cancer Res. 2021;27:2023–2037. doi: 10.1158/1078-0432.CCR-20-3715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 422.Das S, Shapiro B, Vucic EA, Vogt S, Bar-Sagi D. Tumor cell-derived IL1β promotes desmoplasia and immune suppression in pancreatic cancer. Cancer Res. 2020;80:1088–1101. doi: 10.1158/0008-5472.CAN-19-2080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 423.Oberstein PE, Rahma O, Beri N, Stoll-Dastice A, Kawaler EA, Dolgalev I, et al. Abstract PR005: primary results of PanCAN-SR1, a phase 1b study evaluating gemcitabine, nab-paclitaxel, canakinumab, and spartalizumab to target IL-1β and PD-1 in metastatic pancreatic cancer with correlative tissue and blood biomarker analysis. Cancer Res. 2022;82:PR005. doi: 10.1158/1538-7445.PANCA22-PR005. [DOI] [Google Scholar]
- 424.Liu J, Wang Y, Mu C, Li M, Li K, Li S, et al. Pancreatic tumor eradication via selective Pin1 inhibition in cancer-associated fibroblasts and T lymphocytes engagement. Nat Commun. 2022;13:4308. doi: 10.1038/s41467-022-31928-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 425.Koikawa K, Kibe S, Suizu F, Sekino N, Kim N, Manz TD, et al. Targeting Pin1 renders pancreatic cancer eradicable by synergizing with immunochemotherapy. Cell. 2021;184:4753–4771.e27. doi: 10.1016/j.cell.2021.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 426.Kocher HM, Basu B, Froeling FEM, Sarker D, Slater S, Carlin D, et al. Phase I clinical trial repurposing all-trans retinoic acid as a stromal targeting agent for pancreatic cancer. Nat Commun. 2020;11:4841. doi: 10.1038/s41467-020-18636-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 427.Dubiella C, Pinch BJ, Koikawa K, Zaidman D, Poon E, Manz TD, et al. Sulfopin is a covalent inhibitor of Pin1 that blocks Myc-driven tumors in vivo. Nat Chem Biol. 2021;17:954–963. doi: 10.1038/s41589-021-00786-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 428.Aoki S, Inoue K, Klein S, Halvorsen S, Chen J, Matsui A, et al. Placental growth factor promotes tumour desmoplasia and treatment resistance in intrahepatic cholangiocarcinoma. Gut. 2022;71:185–193. doi: 10.1136/gutjnl-2020-322493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 429.Kim DK, Jeong J, Lee DS, Hyeon DY, Park GW, Jeon S, et al. PD-L1-directed PlGF/VEGF blockade synergizes with chemotherapy by targeting CD141+ cancer-associated fibroblasts in pancreatic cancer. Nat Commun. 2022;13:6292. doi: 10.1038/s41467-022-33991-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 430.Zimmermannova O, Ferreira AG, Ascic E, Velasco Santiago M, Kurochkin I, Hansen M, et al. Restoring tumor immunogenicity with dendritic cell reprogramming. Sci Immunol. 2023;8:eadd4817. doi: 10.1126/sciimmunol.add4817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 431.Hegde S, Krisnawan VE, Herzog BH, Zuo C, Breden MA, Knolhoff BL, et al. Dendritic cell paucity leads to dysfunctional immune surveillance in pancreatic cancer. Cancer Cell. 2020;37:289–307.e9. doi: 10.1016/j.ccell.2020.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 432.Hosein AN, Dougan SK, Aguirre AJ, Maitra A. Translational advances in pancreatic ductal adenocarcinoma therapy. Nat Cancer. 2022;3:272–286. doi: 10.1038/s43018-022-00349-2. [DOI] [PubMed] [Google Scholar]
- 433.Chamma H, Vila IK, Taffoni C, Turtoi A, Laguette N. Activation of STING in the pancreatic tumor microenvironment: a novel therapeutic opportunity. Cancer Lett. 2022;538:215694. doi: 10.1016/j.canlet.2022.215694. [DOI] [PubMed] [Google Scholar]
- 434.Jing W, McAllister D, Vonderhaar EP, Palen K, Riese MJ, Gershan J, et al. STING agonist inflames the pancreatic cancer immune microenvironment and reduces tumor burden in mouse models. J Immunother Cancer. 2019;7:115. doi: 10.1186/s40425-019-0573-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 435.Zhang S, Song D, Yu W, Li J, Wang X, Li Y, et al. Combining cisplatin and a STING agonist into one molecule for metalloimmunotherapy of cancer. Natl Sci Rev. 2024;11:nwae020. doi: 10.1093/nsr/nwae020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 436.Twyman-Saint Victor C, Rech AJ, Maity A, Rengan R, Pauken KE, Stelekati E, et al. Radiation and dual checkpoint blockade activate non-redundant immune mechanisms in cancer. Nature. 2015;520:373–377. doi: 10.1038/nature14292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 437.Nywening TM, Wang-Gillam A, Sanford DE, Belt BA, Panni RZ, Cusworth BM, et al. Targeting tumour-associated macrophages with CCR2 inhibition in combination with FOLFIRINOX in patients with borderline resectable and locally advanced pancreatic cancer: a single-centre, open-label, dose-finding, non-randomised, phase 1b trial. Lancet Oncol. 2016;17:651–662. doi: 10.1016/S1470-2045(16)00078-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 438.Noel M, O’Reilly EM, Wolpin BM, Ryan DP, Bullock AJ, Britten CD, et al. Phase 1b study of a small molecule antagonist of human chemokine (C-C motif) receptor 2 (PF-04136309) in combination with nab-paclitaxel/gemcitabine in first-line treatment of metastatic pancreatic ductal adenocarcinoma. Invest New Drugs. 2020;38:800–811. doi: 10.1007/s10637-019-00830-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 439.Steele CW, Karim SA, Leach JDG, Bailey P, Upstill-Goddard R, Rishi L, et al. CXCR2 inhibition profoundly suppresses metastases and augments immunotherapy in pancreatic ductal adenocarcinoma. Cancer Cell. 2016;29:832–845. doi: 10.1016/j.ccell.2016.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 440.Vétizou M, Pitt JM, Daillère R, Lepage P, Waldschmitt N, Flament C, et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science (80-) 2015;350:1079–1084. doi: 10.1126/science.aad1329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 441.Sivan A, Corrales L, Hubert N, Williams JB, Aquino-Michaels K, Earley ZM, et al. Commensal bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science. 2015;350:1084–1089. doi: 10.1126/science.aac4255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 442.Aghamajidi A, Maleki VS. The effect of the gut microbiota on systemic and anti-tumor immunity and response to systemic therapy against cancer. Cancers (Basel) 2022;14:3563. doi: 10.3390/cancers14153563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 443.Routy B, Lenehan JG, Miller WH, Jamal R, Messaoudene M, Daisley BA, et al. Fecal microbiota transplantation plus anti-PD-1 immunotherapy in advanced melanoma: a phase I trial. Nat Med. 2023;29:2121–2132. doi: 10.1038/s41591-023-02453-x. [DOI] [PubMed] [Google Scholar]
- 444.Choi Y, Lichterman JN, Coughlin LA, Poulides N, Li W, Del Valle P, et al. Immune checkpoint blockade induces gut microbiota translocation that augments extraintestinal antitumor immunity. Sci Immunol. 2024;8:eabo2003. doi: 10.1126/sciimmunol.abo2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 445.Helmink BA, Khan MAW, Hermann A, Gopalakrishnan V, Wargo JA. The microbiome, cancer, and cancer therapy. Nat Med. 2019;25:377–388. doi: 10.1038/s41591-019-0377-7. [DOI] [PubMed] [Google Scholar]
- 446.Routy B, Le Chatelier E, Derosa L, Duong CPM, Alou MT, Daillère R, et al. Gut microbiome influences efficacy of PD-1–based immunotherapy against epithelial tumors. Science (80-) 2018;359:91–97. doi: 10.1126/science.aan3706. [DOI] [PubMed] [Google Scholar]
- 447.Hanahan D. Hallmarks of cancer: new dimensions. Cancer Discov. 2022;12:31–46. doi: 10.1158/2159-8290.CD-21-1059. [DOI] [PubMed] [Google Scholar]
- 448.Kharofa J, Haslam D, Wilkinson R, Weiss A, Patel S, Wang K, et al. Analysis of the fecal metagenome in long-term survivors of pancreas cancer. Cancer. 2023;129:1986–1994. doi: 10.1002/cncr.34748. [DOI] [PubMed] [Google Scholar]
- 449.Li Y, Chang RB, Stone ML, Delman D, Markowitz K, Xue Y, et al. Multimodal immune phenotyping reveals microbial-T cell interactions that shape pancreatic cancer. Cell Rep Med. 2024;5:101397. doi: 10.1016/j.xcrm.2024.101397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 450.Merali N, Chouari T, Kayani K, Rayner CJ, Jiménez JI, Krell J, et al. A comprehensive review of the current and future role of the microbiome in pancreatic ductal adenocarcinoma. Cancers (Basel) 2022;14:1020. doi: 10.3390/cancers14041020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 451.Ghaddar B, Biswas A, Harris C, Omary MB, Carpizo DR, Blaser MJ, et al. Tumor microbiome links cellular programs and immunity in pancreatic cancer. Cancer Cell. 2022;40:1240–1253.e5. doi: 10.1016/j.ccell.2022.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 452.Tintelnot J, Xu Y, Lesker TR, Schönlein M, Konczalla L, Giannou AD, et al. Microbiota-derived 3-IAA influences chemotherapy efficacy in pancreatic cancer. Nature. 2023;615:168–174. doi: 10.1038/s41586-023-05728-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 453.Alam A, Levanduski E, Denz P, Villavicencio HS, Bhatta M, Alhorebi L, et al. Fungal mycobiome drives IL-33 secretion and type 2 immunity in pancreatic cancer. Cancer Cell. 2022;40:153–167.e11. doi: 10.1016/j.ccell.2022.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 454.Sethi V, Kurtom S, Tarique M, Lavania S, Malchiodi Z, Hellmund L, et al. Gut microbiota promotes tumor growth in mice by modulating immune response. Gastroenterology. 2018;155:33–37.e6. doi: 10.1053/j.gastro.2018.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 455.Thomas RM, Gharaibeh RZ, Gauthier J, Beveridge M, Pope JL, Guijarro MV, et al. Intestinal microbiota enhances pancreatic carcinogenesis in preclinical models. Carcinogenesis. 2018;39:1068–1078. doi: 10.1093/carcin/bgy073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 456.Derosa L, Routy B, Desilets A, Daillère R, Terrisse S, Kroemer G, et al. Microbiota-centered interventions: the next breakthrough in immuno-oncology? Cancer Discov. 2021;11:2396–2412. doi: 10.1158/2159-8290.CD-21-0236. [DOI] [PubMed] [Google Scholar]
- 457.Baruch EN, Youngster I, Ben-Betzalel G, Ortenberg R, Lahat A, Katz L, et al. Fecal microbiota transplant promotes response in immunotherapy-refractory melanoma patients. Science (80-) 2021;371:602–609. doi: 10.1126/science.abb5920. [DOI] [PubMed] [Google Scholar]
- 458.Davar D, Dzutsev AK, McCulloch JA, Rodrigues RR, Chauvin J-M, Morrison RM, et al. Fecal microbiota transplant overcomes resistance to anti–PD-1 therapy in melanoma patients. Science (80-) 2021;371:595–602. doi: 10.1126/science.abf3363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 459.Riquelme E, Zhang Y, Zhang L, Montiel M, Zoltan M, Dong W, et al. Tumor microbiome diversity and composition influence pancreatic cancer outcomes. Cell. 2019;178:795–806.e12. doi: 10.1016/j.cell.2019.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 460.Spencer CN, McQuade JL, Gopalakrishnan V, McCulloch JA, Vetizou M, Cogdill AP, et al. Dietary fiber and probiotics influence the gut microbiome and melanoma immunotherapy response. Science (80-) 2021;374:1632–1640. doi: 10.1126/science.aaz7015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 461.Huang Y, Zhu N, Zheng X, Liu Y, Lu H, Yin X, et al. Intratumor microbiome analysis identifies positive association between megasphaera and survival of Chinese patients with pancreatic ductal adenocarcinomas. Front Immunol. 2022;13:785422. doi: 10.3389/fimmu.2022.785422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 462.Ghaffari S, Khalili N, Rezaei N. CRISPR/Cas9 revitalizes adoptive T-cell therapy for cancer immunotherapy. J Exp Clin Cancer Res. 2021;40:269. doi: 10.1186/s13046-021-02076-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 463.Katti A, Diaz BJ, Caragine CM, Sanjana NE, Dow LE. CRISPR in cancer biology and therapy. Nat Rev Cancer. 2022;22:259–279. doi: 10.1038/s41568-022-00441-w. [DOI] [PubMed] [Google Scholar]
- 464.Chugh S, Barkeer S, Rachagani S, Nimmakayala RK, Perumal N, Pothuraju R, et al. Disruption of C1galt1 gene promotes development and metastasis of pancreatic adenocarcinomas in mice. Gastroenterology. 2018;155:1608–1624. doi: 10.1053/j.gastro.2018.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 465.Wang SC, Nassour I, Xiao S, Zhang S, Luo X, Lee J, et al. SWI/SNF component ARID1A restrains pancreatic neoplasia formation. Gut. 2019;68:1259–1270. doi: 10.1136/gutjnl-2017-315490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 466.Li M, Xie H, Liu Y, Xia C, Cun X, Long Y, et al. Knockdown of hypoxia-inducible factor-1 alpha by tumor targeted delivery of CRISPR/Cas9 system suppressed the metastasis of pancreatic cancer. J Control Release. 2019;304:204–215. doi: 10.1016/j.jconrel.2019.05.019. [DOI] [PubMed] [Google Scholar]
- 467.Rao CV, Janakiram NB, Madka V, Kumar G, Scott EJ, Pathuri G, et al. Small-molecule inhibition of GCNT3 disrupts mucin biosynthesis and malignant cellular behaviors in pancreatic cancer. Cancer Res. 2016;76:1965–1974. doi: 10.1158/0008-5472.CAN-15-2820. [DOI] [PubMed] [Google Scholar]
- 468.Zhang D, Li L, Jiang H, Knolhoff BL, Lockhart AC, Wang-Gillam A, et al. Constitutive IRAK4 activation underlies poor prognosis and chemoresistance in pancreatic ductal adenocarcinoma. Clin Cancer Res. 2017;23:1748–1759. doi: 10.1158/1078-0432.CCR-16-1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 469.Zhang J, Zhang S, Dörflein I, Ren X, Pfeffer S, Britzen-Laurent N, et al. Impact of CRISPR/Cas9-mediated CD73 knockout in pancreatic cancer. Cancers (Basel) 2023;15:4842. doi: 10.3390/cancers15194842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 470.Gu Y, Zhang Z, Camps MGM, Ossendorp F, Wijdeven RH, ten Dijke P. Genome-wide CRISPR screens define determinants of epithelial-mesenchymal transition mediated immune evasion by pancreatic cancer cells. Sci Adv. 2023;9:eadf9915. doi: 10.1126/sciadv.adf9915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 471.Witz A, Dardare J, Francois A, Husson M, Rouyer M, Demange J, et al. CRISPR/Cas9-mediated knock-in of BRCA1/2 mutations restores response to olaparib in pancreatic cancer cell lines. Sci Rep. 2023;13:18741. doi: 10.1038/s41598-023-45964-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 472.Heumann T, Azad N. Next-generation immunotherapy for pancreatic ductal adenocarcinoma: navigating pathways of immune resistance. Cancer Metastasis Rev. 2021;40:837–862. doi: 10.1007/s10555-021-09981-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 473.Bockorny B, Grossman JE, Hidalgo M. Facts and hopes in immunotherapy of pancreatic cancer. Clin Cancer Res. 2022;28:4606–4617. doi: 10.1158/1078-0432.CCR-21-3452. [DOI] [PubMed] [Google Scholar]
- 474.Wang J, Saung MT, Li K, Fu J, Fujiwara K, Niu N, et al. CCR2/CCR5 inhibitor permits the radiation-induced effector T cell infiltration in pancreatic adenocarcinoma. J Exp Med. 2022;219:e20211631. doi: 10.1084/jem.20211631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 475.Byrne KT, Betts CB, Mick R, Sivagnanam S, Bajor DL, Laheru DA, et al. Neoadjuvant selicrelumab, an agonist CD40 antibody, induces changes in the tumor microenvironment in patients with resectable pancreatic cancer. Clin Cancer Res. 2021;27:4574–4586. doi: 10.1158/1078-0432.CCR-21-1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 476.Vonderheide RH. CD40 agonist antibodies in cancer immunotherapy. Annu Rev Med. 2020;71:47–58. doi: 10.1146/annurev-med-062518-045435. [DOI] [PubMed] [Google Scholar]
- 477.Jian C-Z, Lin L, Hsu C-L, Chen Y-H, Hsu C, Tan C-T, et al. A potential novel cancer immunotherapy: agonistic anti-CD40 antibodies. Drug Discov Today. 2024;29:103893. doi: 10.1016/j.drudis.2024.103893. [DOI] [PubMed] [Google Scholar]
- 478.Liu H-C, Davila Gonzalez D, Viswanath DI, Vander Pol RS, Saunders SZ, Di Trani N, et al. Sustained intratumoral administration of agonist CD40 antibody overcomes immunosuppressive tumor microenvironment in pancreatic cancer. Adv Sci. 2023;10:2206873. doi: 10.1002/advs.202206873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 479.Beatty GL, Torigian DA, Chiorean EG, Saboury B, Brothers A, Alavi A, et al. A phase I study of an agonist CD40 monoclonal antibody (CP-870,893) in combination with gemcitabine in patients with advanced pancreatic ductal adenocarcinoma. Clin Cancer Res. 2013;19:6286–6295. doi: 10.1158/1078-0432.CCR-13-1320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 480.Winograd R, Byrne KT, Evans RA, Odorizzi PM, Meyer ARL, Bajor DL, et al. Induction of T-cell immunity overcomes complete resistance to PD-1 and CTLA-4 blockade and improves survival in pancreatic carcinoma. Cancer Immunol Res. 2015;3:399–411. doi: 10.1158/2326-6066.CIR-14-0215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 481.Knopf P, Stowbur D, Hoffmann SHL, Hermann N, Maurer A, Bucher V, et al. Acidosis-mediated increase in IFN-γ-induced PD-L1 expression on cancer cells as an immune escape mechanism in solid tumors. Mol Cancer. 2023;22:207. doi: 10.1186/s12943-023-01900-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 482.Jardim-Perassi BV, Irrera P, Oluwatola OE, Abrahams D, Estrella VC, Ordway B, et al. L-DOS47 elevates pancreatic cancer tumor pH and enhances response to immunotherapy. Biomedicines. 2024;12:461. doi: 10.3390/biomedicines12020461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 483.Jacobetz MA, Chan DS, Neesse A, Bapiro TE, Cook N, Frese KK, et al. Hyaluronan impairs vascular function and drug delivery in a mouse model of pancreatic cancer. Gut. 2013;62:112–120. doi: 10.1136/gutjnl-2012-302529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 484.Blair AB, Wang J, Davelaar J, Baker A, Li K, Niu N, et al. Dual stromal targeting sensitizes pancreatic adenocarcinoma for anti-programmed cell death protein 1 therapy. Gastroenterology. 2022;163:1267–1280.e7. doi: 10.1053/j.gastro.2022.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 485.Le DT, Brockstedt DG, Nir-Paz R, Hampl J, Mathur S, Nemunaitis J, et al. A live-attenuated Listeria vaccine (ANZ-100) and a live-attenuated Listeria vaccine expressing mesothelin (CRS-207) for advanced cancers: phase I studies of safety and immune induction. Clin Cancer Res. 2012;18:858–868. doi: 10.1158/1078-0432.CCR-11-2121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 486.Decout A, Katz JD, Venkatraman S, Ablasser A. The cGAS–STING pathway as a therapeutic target in inflammatory diseases. Nat Rev Immunol. 2021;21:548–569. doi: 10.1038/s41577-021-00524-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 487.Barber GN. STING: infection, inflammation and cancer. Nat Rev Immunol. 2015;15:760–770. doi: 10.1038/nri3921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 488.Diamond MS, Kinder M, Matsushita H, Mashayekhi M, Dunn GP, Archambault JM, et al. Type I interferon is selectively required by dendritic cells for immune rejection of tumors. J Exp Med. 2011;208:1989–2003. doi: 10.1084/jem.20101158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 489.Fuertes MB, Kacha AK, Kline J, Woo S-R, Kranz DM, Murphy KM, et al. Host type I IFN signals are required for antitumor CD8+ T cell responses through CD8α+ dendritic cells. J Exp Med. 2011;208:2005–2016. doi: 10.1084/jem.20101159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 490.Demaria O, De Gassart A, Coso S, Gestermann N, Di Domizio J, Flatz L, et al. STING activation of tumor endothelial cells initiates spontaneous and therapeutic antitumor immunity. Proc Natl Acad Sci. 2015;112:15408–15413. doi: 10.1073/pnas.1512832112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 491.Ager CR, Reilley MJ, Nicholas C, Bartkowiak T, Jaiswal AR, Curran MA. Intratumoral STING activation with T-cell checkpoint modulation generates systemic antitumor immunity. Cancer Immunol Res. 2017;5:676–684. doi: 10.1158/2326-6066.CIR-17-0049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 492.Corrales L, Glickman LH, McWhirter SM, Kanne DB, Sivick KE, Katibah GE, et al. Direct activation of STING in the tumor microenvironment leads to potent and systemic tumor regression and immunity. Cell Rep. 2015;11:1018–1030. doi: 10.1016/j.celrep.2015.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 493.Weiss JM, Guérin MV, Regnier F, Renault G, Galy-Fauroux I, Vimeux L, et al. The STING agonist DMXAA triggers a cooperation between T lymphocytes and myeloid cells that leads to tumor regression. Oncoimmunology. 2017;6:e1346765. doi: 10.1080/2162402X.2017.1346765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 494.Fu J, Kanne DB, Leong M, Glickman LH, McWhirter SM, Lemmens E, et al. STING agonist formulated cancer vaccines can cure established tumors resistant to PD-1 blockade. Sci Transl Med. 2015;7:283ra52. doi: 10.1126/scitranslmed.aaa4306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 495.Ager CR, Boda A, Rajapakshe K, Lea ST, Di Francesco ME, Jayaprakash P, et al. High potency STING agonists engage unique myeloid pathways to reverse pancreatic cancer immune privilege. J Immunother cancer. 2021;9. [DOI] [PMC free article] [PubMed]
- 496.Uslu U, Sun L, Castelli S, Finck AV, Assenmacher C-A, Young RM, et al. The STING agonist IMSA101 enhances chimeric antigen receptor T cell function by inducing IL-18 secretion. Nat Commun. 2024;15:3933. doi: 10.1038/s41467-024-47692-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 497.Bear AS, Vonderheide RH, O’Hara MH. Challenges and opportunities for pancreatic cancer immunotherapy. Cancer Cell. 2020;38:788–802. doi: 10.1016/j.ccell.2020.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 498.DeNardo DG, Galkin A, Dupont J, Zhou L, Bendell J. GB1275, a first-in-class CD11b modulator: rationale for immunotherapeutic combinations in solid tumors. J Immunother cancer. 2021;9:e003005. doi: 10.1136/jitc-2021-003005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 499.Park H, Bendell JC, Messersmith WA, Rasco DW, De Bono JS, Strickler JH, et al. Preliminary clinical and biologic results of GB1275, a first-in-class oral CD11b modulator, alone and with pembrolizumab, in advanced solid tumors (KEYNOTE A36) J Clin Oncol. 2021;39:2505. doi: 10.1200/JCO.2021.39.15_suppl.2505. [DOI] [Google Scholar]
- 500.Noel MS, Hezel AF, Linehan D, Wang-Gillam A, Eskens F, Sleijfer S, et al. Orally administered CCR2 selective inhibitor CCX872-b clinical trial in pancreatic cancer. J Clin Oncol. 2017;35:276. doi: 10.1200/JCO.2017.35.4_suppl.276. [DOI] [Google Scholar]
- 501.Le D, Gutierrez ME, Saleh M, Chen E, Mallick AB, Pishvaian MJ, et al. Abstract CT124: A phase Ib/II study of BMS-813160, a CC chemokine receptor (CCR) 2/5 dual antagonist, in combination with chemotherapy or nivolumab in patients (pts) with advanced pancreatic or colorectal cancer. Cancer Res. 2018;78:CT124. doi: 10.1158/1538-7445.AM2018-CT124. [DOI] [Google Scholar]
- 502.Seo YD, Jiang X, Sullivan KM, Jalikis FG, Smythe KS, Abbasi A, et al. Mobilization of CD8+ T cells via CXCR4 blockade facilitates PD-1 checkpoint therapy in human pancreatic cancer. Clin Cancer Res. 2019;25:3934–3945. doi: 10.1158/1078-0432.CCR-19-0081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 503.Sanmamed MF, Carranza-Rua O, Alfaro C, Oñate C, Martín-Algarra S, Perez G, et al. Serum interleukin-8 reflects tumor burden and treatment response across malignancies of multiple tissue origins. Clin Cancer Res. 2014;20:5697–5707. doi: 10.1158/1078-0432.CCR-13-3203. [DOI] [PubMed] [Google Scholar]
- 504.Cabel L, Proudhon C, Romano E, Girard N, Lantz O, Stern M-H, et al. Clinical potential of circulating tumour DNA in patients receiving anticancer immunotherapy. Nat Rev Clin Oncol. 2018;15:639–650. doi: 10.1038/s41571-018-0074-3. [DOI] [PubMed] [Google Scholar]
- 505.Biasci D, Smoragiewicz M, Connell CM, Wang Z, Gao Y, Thaventhiran JED, et al. CXCR4 inhibition in human pancreatic and colorectal cancers induces an integrated immune response. Proc Natl Acad Sci. 2020;117:28960–28970. doi: 10.1073/pnas.2013644117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 506.Bockorny B, Semenisty V, Macarulla T, Borazanci E, Wolpin BM, Stemmer SM, et al. BL-8040, a CXCR4 antagonist, in combination with pembrolizumab and chemotherapy for pancreatic cancer: the COMBAT trial. Nat Med. 2020;26:878–885. doi: 10.1038/s41591-020-0880-x. [DOI] [PubMed] [Google Scholar]
- 507.Zboralski D, Hoehlig K, Eulberg D, Frömming A, Vater A. Increasing tumor-infiltrating T cells through inhibition of CXCL12 with NOX-A12 synergizes with PD-1 blockade. Cancer Immunol Res. 2017;5:950–956. doi: 10.1158/2326-6066.CIR-16-0303. [DOI] [PubMed] [Google Scholar]
- 508.Halama N, Williams A, Prüfer U, Frömming A, Beyer D, Eulberg D, et al. Abstract CT117: Phase 1/2 study with CXCL12 inhibitor NOX-A12 and pembrolizumab in patients with microsatellite-stable, metastatic colorectal or pancreatic cancer. Cancer Res. 2020;80:0117. doi: 10.1158/1538-7445.AM2020-CT117. [DOI] [Google Scholar]
- 509.Waickman AT, Alme A, Senaldi L, Zarek PE, Horton M, Powell JD. Enhancement of tumor immunotherapy by deletion of the A2A adenosine receptor. Cancer Immunol Immunother. 2012;61:917–926. doi: 10.1007/s00262-011-1155-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 510.Thompson EA, Powell JD. Inhibition of the adenosine pathway to potentiate cancer immunotherapy: potential for combinatorial approaches. Annu Rev Med. 2021;72:331–348. doi: 10.1146/annurev-med-060619-023155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 511.Hay CM, Sult E, Huang Q, Mulgrew K, Fuhrmann SR, McGlinchey KA, et al. Targeting CD73 in the tumor microenvironment with MEDI9447. Oncoimmunology. 2016;5:e1208875. doi: 10.1080/2162402X.2016.1208875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 512.Bendell JC, LoRusso P, Overman MJ, Noonan AM, Kim D-W, Strickler J, et al. Safety and efficacy of the anti-CD73 monoclonal antibody (mAb) oleclumab ± durvalumab in patients (pts) with advanced colorectal cancer (CRC), pancreatic ductal adenocarcinoma (PDAC), or EGFR-mutant non-small cell lung cancer (EGFRm NSCLC) J Clin Oncol. 2021;39:9047. doi: 10.1200/JCO.2021.39.15_suppl.9047. [DOI] [Google Scholar]
- 513.Manji GA, Wainberg ZA, Krishnan K, Giafis N, Udyavar A, Quah CS, et al. ARC-8: phase I/Ib study to evaluate safety and tolerability of AB680 + chemotherapy + zimberelimab (AB122) in patients with treatment-naive metastatic pancreatic adenocarcinoma (mPDAC) J Clin Oncol. 2021;39:404. doi: 10.1200/JCO.2021.39.3_suppl.404. [DOI] [Google Scholar]
- 514.Evans RA, Diamond MS, Rech AJ, Chao T, Richardson MW, Lin JH, et al. Lack of immunoediting in murine pancreatic cancer reversed with neoantigen. JCI insight. 2016;1:e88328. doi: 10.1172/jci.insight.88328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 515.Łuksza M, Sethna ZM, Rojas LA, Lihm J, Bravi B, Elhanati Y, et al. Neoantigen quality predicts immunoediting in survivors of pancreatic cancer. Nature. 2022;606:389–395. doi: 10.1038/s41586-022-04735-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 516.Lin S, Hong J, Wu S, Zhu C, Liu F, Lin W, et al. BCL2A1 neoepitope–elicited cytotoxic T lymphocytes are a promising individualized immunotherapy of pancreatic cancer. J Leukoc Biol. 2024;0092. [DOI] [PubMed]
- 517.Sharma P, Hu-Lieskovan S, Wargo JA, Ribas A. Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell. 2017;168:707–723. doi: 10.1016/j.cell.2017.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 518.Luke JJ, Bao R, Sweis RF, Spranger S, Gajewski TF. WNT/β-catenin pathway activation correlates with immune exclusion across human cancers. Clin Cancer Res. 2019;25:3074–3083. doi: 10.1158/1078-0432.CCR-18-1942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 519.Sharma P, Siddiqui BA, Anandhan S, Yadav SS, Subudhi SK, Gao J, et al. The next decade of immune checkpoint therapy. Cancer Discov. 2021;11:838–857. doi: 10.1158/2159-8290.CD-20-1680. [DOI] [PubMed] [Google Scholar]
- 520.Peng W, Chen JQ, Liu C, Malu S, Creasy C, Tetzlaff MT, et al. Loss of PTEN promotes resistance to T cell-mediated immunotherapy. Cancer Discov. 2016;6:202–216. doi: 10.1158/2159-8290.CD-15-0283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 521.Kim I-K, Diamond MS, Yuan S, Kemp SB, Kahn BM, Li Q, et al. Plasticity-induced repression of Irf6 underlies acquired resistance to cancer immunotherapy in pancreatic ductal adenocarcinoma. Nat Commun. 2024;15:1532. doi: 10.1038/s41467-024-46048-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 522.Schneider BJ, Lacchetti C, Bollin K. Management of the top 10 most common immune-related adverse events in patients treated with immune checkpoint inhibitor therapy. JCO Oncol Pract. 2022;18:431–444. doi: 10.1200/OP.21.00776. [DOI] [PubMed] [Google Scholar]
- 523.Martins F, Sofiya L, Sykiotis GP, Lamine F, Maillard M, Fraga M, et al. Adverse effects of immune-checkpoint inhibitors: epidemiology, management and surveillance. Nat Rev Clin Oncol. 2019;16:563–580. doi: 10.1038/s41571-019-0218-0. [DOI] [PubMed] [Google Scholar]
- 524.Ramos-Casals M, Brahmer JR, Callahan MK, Flores-Chávez A, Keegan N, Khamashta MA, et al. Immune-related adverse events of checkpoint inhibitors. Nat Rev Dis Prim. 2020;6:38. doi: 10.1038/s41572-020-0160-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 525.Brudno JN, Kochenderfer JN. Toxicities of chimeric antigen receptor T cells: recognition and management. Blood. 2016;127:3321–3330. doi: 10.1182/blood-2016-04-703751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 526.Maher VE, Fernandes LL, Weinstock C, Tang S, Agarwal S, Brave M, et al. Analysis of the association between adverse events and outcome in patients receiving a programmed death protein 1 or programmed death ligand 1 antibody. J Clin Oncol. 2019;37:2730–2737. doi: 10.1200/JCO.19.00318. [DOI] [PubMed] [Google Scholar]
- 527.Morad G, Helmink BA, Sharma P, Wargo JA. Hallmarks of response, resistance, and toxicity to immune checkpoint blockade. Cell. 2021;184:5309–5337. doi: 10.1016/j.cell.2021.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 528.Sasson SC, Slevin SM, Cheung VTF, Nassiri I, Olsson-Brown A, Fryer E, et al. Interferon-gamma-producing CD8(+) tissue resident memory T cells are a targetable hallmark of immune checkpoint inhibitor-colitis. Gastroenterology. 2021;161:1229–1244.e9. doi: 10.1053/j.gastro.2021.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 529.Zhou Y, Medik YB, Patel B, Zamler DB, Chen S, Chapman T, et al. Intestinal toxicity to CTLA-4 blockade driven by IL-6 and myeloid infiltration. J Exp Med. 2023;220. [DOI] [PMC free article] [PubMed]
- 530.Luoma AM, Suo S, Williams HL, Sharova T, Sullivan K, Manos M, et al. Molecular pathways of colon inflammation induced by cancer immunotherapy. Cell. 2020;182:655–671.e22. doi: 10.1016/j.cell.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 531.Axelrod ML, Meijers WC, Screever EM, Qin J, Carroll MG, Sun X, et al. T cells specific for α-myosin drive immunotherapy-related myocarditis. Nature. 2022;611:818–826. doi: 10.1038/s41586-022-05432-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 532.Wang Y, Wiesnoski DH, Helmink BA, Gopalakrishnan V, Choi K, DuPont HL, et al. Fecal microbiota transplantation for refractory immune checkpoint inhibitor-associated colitis. Nat Med. 2018;24:1804–1808. doi: 10.1038/s41591-018-0238-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 533.Le DT, Uram JN, Wang H, Bartlett BR, Kemberling H, Eyring AD, et al. PD-1 blockade in tumors with mismatch-repair deficiency. N Engl J Med. 2015;372:2509–2520. doi: 10.1056/NEJMoa1500596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 534.Marabelle A, Fakih M, Lopez J, Shah M, Shapira-Frommer R, Nakagawa K, et al. Association of tumour mutational burden with outcomes in patients with advanced solid tumours treated with pembrolizumab: prospective biomarker analysis of the multicohort, open-label, phase 2 KEYNOTE-158 study. Lancet Oncol. 2020;21:1353–1365. doi: 10.1016/S1470-2045(20)30445-9. [DOI] [PubMed] [Google Scholar]
- 535.Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443–2454. doi: 10.1056/NEJMoa1200690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 536.Tsunedomi R, Shindo Y, Nakajima M, Yoshimura K, Nagano H. The tumor immune microenvironment in pancreatic cancer and its potential in the identification of immunotherapy biomarkers. Expert Rev Mol Diagn. 2023;23:1121–1134. doi: 10.1080/14737159.2023.2281482. [DOI] [PubMed] [Google Scholar]
- 537.Qiu X, Shi Z, Tong F, Lu C, Zhu Y, Wang Q, et al. Biomarkers for predicting tumor response to PD-1 inhibitors in patients with advanced pancreatic cancer. Hum Vaccin Immunother. 2023;19:2178791. doi: 10.1080/21645515.2023.2178791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 538.Tang J, Shalabi A, Hubbard-Lucey VM. Comprehensive analysis of the clinical immuno-oncology landscape. Ann Oncol Off J Eur Soc Med Oncol. 2018;29:84–91. doi: 10.1093/annonc/mdx755. [DOI] [PubMed] [Google Scholar]
- 539.Hegde PS, Chen DS. Top 10 challenges in cancer immunotherapy. Immunity. 2020;52:17–35. doi: 10.1016/j.immuni.2019.12.011. [DOI] [PubMed] [Google Scholar]
- 540.Marabelle A, Le DT, Ascierto PA, Di Giacomo AM, De Jesus-Acosta A, Delord J-P, et al. Efficacy of pembrolizumab in patients with noncolorectal high microsatellite instability/mismatch repair-deficient cancer: results from the phase II KEYNOTE-158 study. J Clin Oncol. 2019;38:1–10. doi: 10.1200/JCO.19.02105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 541.Ebert PJR, Cheung J, Yang Y, McNamara E, Hong R, Moskalenko M, et al. MAP kinase inhibition promotes T cell and anti-tumor activity in combination with PD-L1 checkpoint blockade. Immunity. 2016;44:609–621. doi: 10.1016/j.immuni.2016.01.024. [DOI] [PubMed] [Google Scholar]
- 542.Yu Y, Yang G, Huang H, Fu Z, Cao Z, Zheng L, et al. Preclinical models of pancreatic ductal adenocarcinoma: challenges and opportunities in the era of precision medicine. J Exp Clin Cancer Res. 2021;40:8. doi: 10.1186/s13046-020-01787-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 543.Fridman WH, Pagès F, Sautès-Fridman C, Galon J. The immune contexture in human tumours: impact on clinical outcome. Nat Rev Cancer. 2012;12:298–306. doi: 10.1038/nrc3245. [DOI] [PubMed] [Google Scholar]
- 544.Kersten K, de Visser KE, van Miltenburg MH, Jonkers J. Genetically engineered mouse models in oncology research and cancer medicine. EMBO Mol Med. 2017;9:137–153. doi: 10.15252/emmm.201606857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 545.Wei SC, Duffy CR, Allison JP. Fundamental mechanisms of immune checkpoint blockade therapy. Cancer Discov. 2018;8:1069–1086. doi: 10.1158/2159-8290.CD-18-0367. [DOI] [PubMed] [Google Scholar]
- 546.Montagne JM, Jaffee EM, Fertig EJ. Multiomics empowers predictive pancreatic cancer immunotherapy. J Immunol. 2023;210:859–868. doi: 10.4049/jimmunol.2200660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 547.Blise KE, Sivagnanam S, Betts CB, Betre K, Kirchberger N, Tate BJ, et al. Machine learning links T-cell function and spatial localization to neoadjuvant immunotherapy and clinical outcome in pancreatic cancer. Cancer Immunol Res. 2024;OF1–15. [DOI] [PMC free article] [PubMed]
- 548.Haldar SD, Judkins C, Ferguson A, Abou Diwan E, Lim SJ, Wang H, et al. A phase I study of a mutant KRAS-targeted long peptide vaccine in patients at high risk of developing pancreatic cancer. J Clin Oncol. 2023;41:TPS758. doi: 10.1200/JCO.2023.41.4_suppl.TPS758. [DOI] [Google Scholar]
- 549.Yousuf S, Qiu M, Voith von Voithenberg L, Hulkkonen J, Macinkovic I, Schulz AR, et al. Spatially resolved multi-omics single-cell analyses inform mechanisms of immune dysfunction in pancreatic cancer. Gastroenterology. 2023;165:891–908. doi: 10.1053/j.gastro.2023.05.036. [DOI] [PubMed] [Google Scholar]
- 550.Tieu V, Sotillo E, Bjelajac JR, Chen C, Malipatlolla M, Guerrero JA, et al. A versatile CRISPR-Cas13d platform for multiplexed transcriptomic regulation and metabolic engineering in primary human T cells. Cell. 2024;187:1278–1295.e20. doi: 10.1016/j.cell.2024.01.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 551.Zhang Y, Yan W, Mathew E, Bednar F, Wan S, Collins MA, et al. CD4+ T lymphocyte ablation prevents pancreatic carcinogenesis in mice. Cancer Immunol Res. 2014;2:423–435. doi: 10.1158/2326-6066.CIR-14-0016-T. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 552.Burrack AL, Rollins MR, Spartz EJ, Mesojednik TD, Schmiechen ZC, Raynor JF, et al. CD40 agonist overcomes T cell exhaustion induced by chronic myeloid cell IL-27 production in a pancreatic cancer preclinical model. J Immunol. 2021;206:1372–1384. doi: 10.4049/jimmunol.2000765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 553.Beatty GL, Winograd R, Evans RA, Long KB, Luque SL, Lee JW, et al. Exclusion of T cells from pancreatic carcinomas in mice is regulated by Ly6C(low) F4/80(+) extratumoral macrophages. Gastroenterology. 2015;149:201–210. doi: 10.1053/j.gastro.2015.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 554.Stromnes IM, Schmitt TM, Hulbert A, Brockenbrough JS, Nguyen H, Cuevas C, et al. T cells engineered against a native antigen can surmount immunologic and physical barriers to treat pancreatic ductal adenocarcinoma. Cancer Cell. 2015;28:638–652. doi: 10.1016/j.ccell.2015.09.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 555.Keenan BP, Saenger Y, Kafrouni MI, Leubner A, Lauer P, Maitra A, et al. A Listeria vaccine and depletion of T-regulatory cells activate immunity against early stage pancreatic intraepithelial neoplasms and prolong survival of mice. Gastroenterology. 2014;146:1784–94.e6. doi: 10.1053/j.gastro.2014.02.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 556.Pylayeva-Gupta Y, Das S, Handler JS, Hajdu CH, Coffre M, Koralov SB, et al. IL35-producing B cells promote the development of pancreatic neoplasia. Cancer Discov. 2016;6:247–255. doi: 10.1158/2159-8290.CD-15-0843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 557.Gunderson AJ, Kaneda MM, Tsujikawa T, Nguyen AV, Affara NI, Ruffell B, et al. Bruton tyrosine kinase-dependent immune cell cross-talk drives pancreas cancer. Cancer Discov. 2016;6:270–285. doi: 10.1158/2159-8290.CD-15-0827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 558.Zhang Y, Velez-Delgado A, Mathew E, Li D, Mendez FM, Flannagan K, et al. Myeloid cells are required for PD-1/PD-L1 checkpoint activation and the establishment of an immunosuppressive environment in pancreatic cancer. Gut. 2017;66:124–136. doi: 10.1136/gutjnl-2016-312078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 559.Long KB, Gladney WL, Tooker GM, Graham K, Fraietta JA, Beatty GL. IFNγ and CCL2 cooperate to redirect tumor-infiltrating monocytes to degrade fibrosis and enhance chemotherapy efficacy in pancreatic carcinoma. Cancer Discov. 2016;6:400–413. doi: 10.1158/2159-8290.CD-15-1032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 560.Sherman MH, Yu RT, Tseng TW, Sousa CM, Liu S, Truitt ML, et al. Stromal cues regulate the pancreatic cancer epigenome and metabolome. Proc Natl Acad Sci. 2017;114:1129–1134. doi: 10.1073/pnas.1620164114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 561.Sousa CM, Biancur DE, Wang X, Halbrook CJ, Sherman MH, Zhang L, et al. Pancreatic stellate cells support tumour metabolism through autophagic alanine secretion. Nature. 2016;536:479–483. doi: 10.1038/nature19084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 562.Zhang Y, Recouvreux MV, Jung M, Galenkamp KMO, Li Y, Zagnitko O, et al. Macropinocytosis in cancer-associated fibroblasts is dependent on CaMKK2/ARHGEF2 signaling and functions to support tumor and stromal cell fitness. Cancer Discov. 2021;11:1808–1825. doi: 10.1158/2159-8290.CD-20-0119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 563.Francescone R, Barbosa Vendramini-Costa D, Franco-Barraza J, Wagner J, Muir A, Lau AN, et al. Netrin G1 promotes pancreatic tumorigenesis through cancer-associated fibroblast-driven nutritional support and immunosuppression. Cancer Discov. 2021;11:446–479. doi: 10.1158/2159-8290.CD-20-0775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 564.Ferrara B, Pignatelli C, Cossutta M, Citro A, Courty J, Piemonti L. The extracellular matrix in pancreatic cancer: description of a complex network and promising therapeutic options. Cancers (Basel) 2021;13:4442. doi: 10.3390/cancers13174442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 565.Lee JJ, Perera RM, Wang H, Wu D-C, Liu XS, Han S, et al. Stromal response to Hedgehog signaling restrains pancreatic cancer progression. Proc Natl Acad Sci U S A. 2014;111:E3091–E3100. doi: 10.1073/pnas.1411679111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 566.Tian C, Clauser KR, Öhlund D, Rickelt S, Huang Y, Gupta M, et al. Proteomic analyses of ECM during pancreatic ductal adenocarcinoma progression reveal different contributions by tumor and stromal cells. Proc Natl Acad Sci U S A. 2019;116:19609–19618. doi: 10.1073/pnas.1908626116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 567.Kai F, Drain AP, Weaver VM. The extracellular matrix modulates the metastatic journey. Dev Cell. 2019;49:332–346. doi: 10.1016/j.devcel.2019.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 568.Tian C, Huang Y, Clauser KR, Rickelt S, Lau AN, Carr SA, et al. Suppression of pancreatic ductal adenocarcinoma growth and metastasis by fibrillar collagens produced selectively by tumor cells. Nat Commun. 2021;12:2328. doi: 10.1038/s41467-021-22490-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 569.Bhattacharjee S, Hamberger F, Ravichandra A, Miller M, Nair A, Affo S, et al. Tumor restriction by type I collagen opposes tumor-promoting effects of cancer-associated fibroblasts. J Clin Invest. 2021;131. [DOI] [PMC free article] [PubMed]
- 570.Katz SC, Moody AE, Guha P, Hardaway JC, Prince E, LaPorte J, et al. HITM-SURE: hepatic immunotherapy for metastases phase Ib anti-CEA CAR-T study utilizing pressure enabled drug delivery. J Immunother Cancer. 2020;8:e001097. doi: 10.1136/jitc-2020-001097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 571.Botta GP, Becerra CR, Jin Z, Kim DW, Zhao D, Lenz H-J, et al. Multicenter phase Ib trial in the U.S. of salvage CT041 CLDN18.2-specific chimeric antigen receptor T-cell therapy for patients with advanced gastric and pancreatic adenocarcinoma. J Clin Oncol. 2022;40:2538. doi: 10.1200/JCO.2022.40.16_suppl.2538. [DOI] [Google Scholar]
- 572.Chan LY, Dass SA, Tye GJ, Imran SAM, Wan Kamarul Zaman WS, Nordin F. CAR-T Cells/-NK cells in cancer immunotherapy and the potential of MSC to enhance its efficacy: a review. Biomedicines. 2022;10:804. doi: 10.3390/biomedicines10040804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 573.Rafiq S, Hackett CS, Brentjens RJ. Engineering strategies to overcome the current roadblocks in CAR T cell therapy. Nat Rev Clin Oncol. 2020;17:147–167. doi: 10.1038/s41571-019-0297-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 574.Young RM, Engel NW, Uslu U, Wellhausen N, June CH. Next-generation CAR T-cell therapies. Cancer Discov. 2022;12:1625–1633. doi: 10.1158/2159-8290.CD-21-1683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 575.Tokarew N, Ogonek J, Endres S, von Bergwelt-Baildon M, Kobold S. Teaching an old dog new tricks: next-generation CAR T cells. Br J Cancer. 2019;120:26–37. doi: 10.1038/s41416-018-0325-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 576.Tomasik J, Jasiński M, Basak GW. Next generations of CAR-T cells—new therapeutic opportunities in hematology? Front Immunol. 2022;13:1034707. doi: 10.3389/fimmu.2022.1034707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 577.Khawar MB, Sun H. CAR-NK cells: from natural basis to design for kill. Front Immunol. 2021;12:707542. doi: 10.3389/fimmu.2021.707542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 578.Raftery MJ, Franzén AS, Pecher G. CAR NK cells: the future is now. Annu Rev Cancer Biol. 2023;7:229–246. doi: 10.1146/annurev-cancerbio-061521-082320. [DOI] [Google Scholar]
- 579.McLellan AD, Ali Hosseini Rad SM. Chimeric antigen receptor T cell persistence and memory cell formation. Immunol Cell Biol. 2019;97:664–674. doi: 10.1111/imcb.12254. [DOI] [PubMed] [Google Scholar]
- 580.Cerwenka A, Lanier LL. Natural killer cell memory in infection, inflammation and cancer. Nat Rev Immunol. 2016;16:112–123. doi: 10.1038/nri.2015.9. [DOI] [PubMed] [Google Scholar]
- 581.Depil S, Duchateau P, Grupp SA, Mufti G, Poirot L. “Off-the-shelf” allogeneic CAR T cells: development and challenges. Nat Rev Drug Discov. 2020;19:185–199. doi: 10.1038/s41573-019-0051-2. [DOI] [PubMed] [Google Scholar]
- 582.Ghassemi S, Durgin JS, Nunez-Cruz S, Patel J, Leferovich J, Pinzone M, et al. Rapid manufacturing of non-activated potent CAR T cells. Nat Biomed Eng. 2022;6:118–128. doi: 10.1038/s41551-021-00842-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 583.Pfefferle A, Huntington ND. You have got a fast CAR: chimeric antigen receptor NK cells in cancer therapy. Cancers (Basel) 2020;12:706. doi: 10.3390/cancers12030706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 584.Basar R, Daher M, Rezvani K. Next-generation cell therapies: the emerging role of CAR-NK cells. Blood Adv. 2020;4:5868–5876. doi: 10.1182/bloodadvances.2020002547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 585.Anderson ND, Birch J, Accogli T, Criado I, Khabirova E, Parks C, et al. Transcriptional signatures associated with persisting CD19 CAR-T cells in children with leukemia. Nat Med. 2023;29:1700–1709. doi: 10.1038/s41591-023-02415-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 586.Liu E, Marin D, Banerjee P, Macapinlac HA, Thompson P, Basar R, et al. Use of CAR-transduced natural killer cells in CD19-positive lymphoid tumors. N Engl J Med. 2020;382:545–553. doi: 10.1056/NEJMoa1910607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 587.Ghilardi G, Fraietta JA, Gerson JN, Van Deerlin VM, Morrissette JJD, Caponetti GC, et al. T cell lymphoma and secondary primary malignancy risk after commercial CAR T cell therapy. Nat Med. 2024;30:984–989. doi: 10.1038/s41591-024-02826-w. [DOI] [PubMed] [Google Scholar]
- 588.Bonifant CL, Jackson HJ, Brentjens RJ, Curran KJ. Toxicity and management in CAR T-cell therapy. Mol Ther Oncolytics. 2016;3:16011. doi: 10.1038/mto.2016.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 589.Wang L, Zhang L, Dunmall LC, Wang YY, Fan Z, Cheng Z, et al. The dilemmas and possible solutions for CAR-T cell therapy application in solid tumors. Cancer Lett. 2024;591:216871. doi: 10.1016/j.canlet.2024.216871. [DOI] [PubMed] [Google Scholar]
- 590.Al-Haideri M, Tondok SB, Safa SH, Maleki AH, Rostami S, Jalil AT, et al. CAR-T cell combination therapy: the next revolution in cancer treatment. Cancer Cell Int. 2022;22:365. doi: 10.1186/s12935-022-02778-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 591.Ko AH, Kim K-P, Siveke JT, Lopez CD, Lacy J, O’Reilly EM, et al. Atezolizumab plus PEGPH20 versus chemotherapy in advanced pancreatic ductal adenocarcinoma and gastric cancer: MORPHEUS phase Ib/II umbrella randomized study platform. Oncologist. 2023;28:553–e472. doi: 10.1093/oncolo/oyad022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 592.Wang J, Gai J, Zhang T, Niu N, Qi H, Thomas DL, et al. Neoadjuvant radioimmunotherapy in pancreatic cancer enhances effector T cell infiltration and shortens their distances to tumor cells. Sci Adv. 2024;10:eadk1827. doi: 10.1126/sciadv.adk1827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 593.Lee V, Ding D, Rodriguez C, Onners B, Narang A, Meyer J, et al. A phase 2 study of cyclophosphamide (CY), GVAX, pembrolizumab (Pembro), and stereotactic body radiation (SBRT) in patients (pts) with locally advanced pancreas cancer (LAPC) J Clin Oncol. 2021;39:4134. doi: 10.1200/JCO.2021.39.15_suppl.4134. [DOI] [Google Scholar]
- 594.Halama N, Prüfer U, Froemming A, Beyer D, Eulberg D, Jungnelius JU, et al. 613P - Phase I/II study with CXCL12 inhibitor NOX-A12 and pembrolizumab in patients with microsatellite-stable, metastatic colorectal or pancreatic cancer. Ann Oncol. 2019;30:v231. doi: 10.1093/annonc/mdz246.090. [DOI] [Google Scholar]
- 595.Overman M, Javle M, Davis RE, Vats P, Kumar-Sinha C, Xiao L, et al. Randomized phase II study of the Bruton tyrosine kinase inhibitor acalabrutinib, alone or with pembrolizumab in patients with advanced pancreatic cancer. J Immunother Cancer. 2020;8:e000587. doi: 10.1136/jitc-2020-000587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 596.Wang-gillam A, Lim K-H, McWilliams RR, Suresh R, Lockhart AC, Brown A, et al. Phase I dose escalation and expansion study of defactinib, pembrolizumab, and gemcitabine in patients with advanced treatment-refractory pancreatic cancer. J Clin Oncol. 2022;40:4146. doi: 10.1200/JCO.2022.40.16_suppl.4146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 597.Razak AR, Cleary JM, Moreno V, Boyer M, Calvo Aller E, Edenfield W, et al. Safety and efficacy of AMG 820, an anti-colony-stimulating factor 1 receptor antibody, in combination with pembrolizumab in adults with advanced solid tumors. J Immunother Cancer. 2020;8:e001006. doi: 10.1136/jitc-2020-001006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 598.Johnson M, Dudek AZ, Sukari A, Call J, Kunk PR, Lewis K, et al. ARRY-382 in combination with pembrolizumab in patients with advanced solid tumors: results from a phase 1b/2 study. Clin Cancer Res. 2022;28:2517–2526. doi: 10.1158/1078-0432.CCR-21-3009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 599.Voissière A, Gomez-Roca C, Chabaud S, Rodriguez C, Nkodia A, Berthet J, et al. The CSF-1R inhibitor pexidartinib affects FLT3-dependent DC differentiation and may antagonize durvalumab effect in patients with advanced cancers. Sci Transl Med. 2024;16:eadd1834. doi: 10.1126/scitranslmed.add1834. [DOI] [PubMed] [Google Scholar]
- 600.Kamath SD, Kalyan A, Kircher S, Nimeiri H, Fought AJ, Benson A, 3rd, et al. Ipilimumab and gemcitabine for advanced pancreatic cancer: A phase Ib study. Oncologist. 2020;25:e808–e815. doi: 10.1634/theoncologist.2019-0473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 601.Wainberg ZA, Hochster HS, Kim EJ, George B, Kaylan A, Chiorean EG, et al. Open-label, phase I study of nivolumab combined with nab-paclitaxel plus gemcitabine in advanced pancreatic cancer. Clin Cancer Res. 2020;26:4814–4822. doi: 10.1158/1078-0432.CCR-20-0099. [DOI] [PubMed] [Google Scholar]
- 602.Agarwal P, Qi H, Munjal K, Gai J, Ferguson A, Parkinson R, et al. Overall survival (OS) and pathologic response rate from a phase II clinical trial of neoadjuvant GVAX pancreas vaccine (with cyclophosphamide) in combination with nivolumab and stereotactic body radiation therapy (SBRT) followed by definitive resection fo. J Clin Oncol. 2023;41:e16309–e16309. doi: 10.1200/JCO.2023.41.16_suppl.e16309. [DOI] [Google Scholar]
- 603.Padrón LJ, Maurer DM, O’Hara MH, O’Reilly EM, Wolff RA, Wainberg ZA, et al. Sotigalimab and/or nivolumab with chemotherapy in first-line metastatic pancreatic cancer: clinical and immunologic analyses from the randomized phase 2 PRINCE trial. Nat Med. 2022;28:1167–1177. doi: 10.1038/s41591-022-01829-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 604.Tsujikawa T, Crocenzi T, Durham JN, Sugar EA, Wu AA, Onners B, et al. Evaluation of cyclophosphamide/GVAX pancreas followed by Listeria-Mesothelin (CRS-207) with or without nivolumab in patients with pancreatic cancer. Clin Cancer Res. 2020;26:3578–3588. doi: 10.1158/1078-0432.CCR-19-3978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 605.Brar G, Xie C, Floudas CS, Morelli MP, Fioravanti S, Walker M, et al. Immune checkpoint inhibition (ICI) in combination with SBRT in patients with advanced pancreatic adenocarcinoma (aPDAC) J Clin Oncol. 2019;37:192. doi: 10.1200/JCO.2019.37.4_suppl.192. [DOI] [Google Scholar]
- 606.Koenig JL, Pappas L, Yeap BY, Clark JW, Weekes CD, Allen JN, et al. A phase II study of nivolumab and ipilimumab with radiation therapy in patients with metastatic, microsatellite stable pancreatic adenocarcinoma. J Clin Oncol. 2023;41:4143. doi: 10.1200/JCO.2023.41.16_suppl.4143. [DOI] [Google Scholar]
- 607.Meng J, Lv Y, Bao W, Meng Z, Wang S, Wu Y, et al. Generation of whole tumor cell vaccine for on-demand manipulation of immune responses against cancer under near-infrared laser irradiation. Nat Commun. 2023;14:4505. doi: 10.1038/s41467-023-40207-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 608.Lutz E, Yeo CJ, Lillemoe KD, Biedrzycki B, Kobrin B, Herman J, et al. A lethally irradiated allogeneic granulocyte-macrophage colony stimulating factor-secreting tumor vaccine for pancreatic adenocarcinoma. A Phase II trial of safety, efficacy, and immune activation. Ann Surg. 2011;253:328–335. doi: 10.1097/SLA.0b013e3181fd271c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 609.Batchu RB, Gruzdyn OV, Qazi AM, Mahmud EM, Mostafa G, Weaver DW, et al. Pancreatic cancer cell lysis by cell-penetrating peptide-MAGE-A3–induced cytotoxic T lymphocytes. JAMA Surg. 2016;151:1086–1088. doi: 10.1001/jamasurg.2016.2346. [DOI] [PubMed] [Google Scholar]
- 610.Morse MA, Nair SK, Boczkowski D, Tyler D, Hurwitz HI, Proia A, et al. The feasibility and safety of immunotherapy with dendritic cells loaded with CEA mRNA following neoadjuvant chemoradiotherapy and resection of pancreatic cancer. Int J Gastrointest Cancer. 2002;32:1–6. doi: 10.1385/IJGC:32:1:1. [DOI] [PubMed] [Google Scholar]
- 611.Muscarella P, Bekaii-Saab T, McIntyre K, Rosemurgy A, Ross SB, Richards DA, et al. A phase 2 randomized placebo-controlled adjuvant trial of GI-4000, a recombinant yeast expressing mutated RAS proteins in patients with resected pancreas cancer. J Pancreat cancer. 2021;7:8–19. doi: 10.1089/pancan.2020.0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 612.Palmer DH, Valle JW, Ma YT, Faluyi O, Neoptolemos JP, Jensen Gjertsen T, et al. TG01/GM-CSF and adjuvant gemcitabine in patients with resected RAS-mutant adenocarcinoma of the pancreas (CT TG01-01): a single-arm, phase 1/2 trial. Br J Cancer. 2020;122:971–977. doi: 10.1038/s41416-020-0752-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 613.Cohn A, Morse MA, O’Neil B, Whiting S, Coeshott C, Ferraro J, et al. Whole recombinant saccharomyces cerevisiae yeast expressing RAS mutations as treatment for patients with solid tumors bearing RAS mutations: results from a phase 1 trial. J Immunother. 2018;41:141–150. doi: 10.1097/CJI.0000000000000219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 614.Abou-Alfa GK, Chapman PB, Feilchenfeldt J, Brennan MF, Capanu M, Gansukh B, et al. Targeting mutated K-ras in pancreatic adenocarcinoma using an adjuvant vaccine. Am J Clin Oncol. 2011;34:321–325. doi: 10.1097/COC.0b013e3181e84b1f. [DOI] [PubMed] [Google Scholar]
- 615.Maki RG, Livingston PO, Lewis JJ, Janetzki S, Klimstra D, Desantis D, et al. A phase I pilot study of autologous heat shock protein vaccine HSPPC-96 in patients with resected pancreatic adenocarcinoma. Dig Dis Sci. 2007;52:1964–1972. doi: 10.1007/s10620-006-9205-2. [DOI] [PubMed] [Google Scholar]
- 616.Kameshima H, Tsuruma T, Kutomi G, Shima H, Iwayama Y, Kimura Y, et al. Immunotherapeutic benefit of α-interferon (IFNα) in survivin2B-derived peptide vaccination for advanced pancreatic cancer patients. Cancer Sci. 2013;104:124–129. doi: 10.1111/cas.12046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 617.Miyazawa M, Ohsawa R, Tsunoda T, Hirono S, Kawai M, Tani M, et al. Phase I clinical trial using peptide vaccine for human vascular endothelial growth factor receptor 2 in combination with gemcitabine for patients with advanced pancreatic cancer. Cancer Sci. 2010;101:433–439. doi: 10.1111/j.1349-7006.2009.01416.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 618.Niethammer AG, Lubenau H, Mikus G, Knebel P, Hohmann N, Leowardi C, et al. Double-blind, placebo-controlled first in human study to investigate an oral vaccine aimed to elicit an immune reaction against the VEGF-Receptor 2 in patients with stage IV and locally advanced pancreatic cancer. BMC Cancer. 2012;12:361. doi: 10.1186/1471-2407-12-361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 619.Brett BT, Smith SC, Bouvier CV, Michaeli D, Hochhauser D, Davidson BR, et al. Phase II study of anti-gastrin-17 antibodies, raised to G17DT, in advanced pancreatic cancer. J Clin Oncol. 2002;20:4225–4231. doi: 10.1200/JCO.2002.11.151. [DOI] [PubMed] [Google Scholar]
- 620.Gilliam AD, Broome P, Topuzov EG, Garin AM, Pulay I, Humphreys J, et al. An international multicenter randomized controlled trial of G17DT in patients with pancreatic cancer. Pancreas. 2012;41:374–379. doi: 10.1097/MPA.0b013e31822ade7e. [DOI] [PubMed] [Google Scholar]
- 621.Dalgleish AG. A multicenter randomized, open-label, proof-of-concept, phase II trial comparing gemcitabine with and without IMM-101 in advanced pancreatic cancer. J Clin Oncol. 2015;33:336. doi: 10.1200/jco.2015.33.3_suppl.336. [DOI] [Google Scholar]
- 622.Dalgleish AG, Stebbing J, Adamson DJ, Arif SS, Bidoli P, Chang D, et al. Randomised, open-label, phase II study of gemcitabine with and without IMM-101 for advanced pancreatic cancer. Br J Cancer. 2016;115:789–796. doi: 10.1038/bjc.2016.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 623.Borazanci EH, Jameson G, Korn RL, Caldwell L, Ansaldo K, Hendrickson K, et al. Abstract CT152: a Phase II pilot trial of nivolumab (N) + albumin bound paclitaxel (AP) + paricalcitol (P) + cisplatin (C) + gemcitabine (G) (NAPPCG) in patients with previously untreated metastatic pancreatic ductal adenocarcinoma (PDAC) Cancer Res. 2019;79:CT152. doi: 10.1158/1538-7445.AM2019-CT152. [DOI] [Google Scholar]
- 624.Oberstein PE, Rahma OE, Beri N, Stoll-D’Astice AC, Duliege A-M, Nazeer S, et al. A phase 1b study evaluating IL-1β and PD-1 targeting with chemotherapy in metastatic pancreatic cancer (PanCAN-SR1) J Clin Oncol. 2022;40:557. doi: 10.1200/JCO.2022.40.4_suppl.557. [DOI] [Google Scholar]
- 625.Oberstein PE, Rahma OE, Beri N, Stoll-D’Astice AC, Duliege A-M, Kawaler E, et al. Initial efficacy and biomarker analysis of a phase 1b study targeting IL-1β and PD-1 with chemotherapy in metastatic pancreatic cancer (PanCAN-SR1) J Clin Oncol. 2022;40:e16287–e16287. doi: 10.1200/JCO.2022.40.16_suppl.e16287. [DOI] [Google Scholar]
- 626.Muth ST, Saung MT, Blair AB, Henderson MG, Thomas DL, Zheng L. CD137 agonist-based combination immunotherapy enhances activated, effector memory T cells and prolongs survival in pancreatic adenocarcinoma. Cancer Lett. 2021;499:99–108. doi: 10.1016/j.canlet.2020.11.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 627.Zhong P, Nakata K, Oyama K, Higashijima N, Sagara A, Date S, et al. Blockade of histamine receptor H1 augments immune checkpoint therapy by enhancing MHC-I expression in pancreatic cancer cells. J Exp Clin Cancer Res. 2024;43:138. doi: 10.1186/s13046-024-03060-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 628.Li P, Rozich N, Wang J, Wang J, Xu Y, Herbst B, et al. Anti-IL-8 antibody activates myeloid cells and potentiates the anti-tumor activity of anti-PD-1 antibody in the humanized pancreatic cancer murine model. Cancer Lett. 2022;539:215722. doi: 10.1016/j.canlet.2022.215722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 629.Wan Z, Huang H, West RE, III, Zhang M, Zhang B, Cai X, et al. Overcoming pancreatic cancer immune resistance by codelivery of CCR2 antagonist using a STING-activating gemcitabine-based nanocarrier. Mater Today. 2023;62:33–50. doi: 10.1016/j.mattod.2022.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 630.Chen L, Zuo M, Zhou Q, Wang Y. Oncolytic virotherapy in cancer treatment: challenges and optimization prospects. Front Immunol. 2023;14:1308890. doi: 10.3389/fimmu.2023.1308890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 631.Reale A, Vitiello A, Conciatori V, Parolin C, Calistri A, Palù G. Perspectives on immunotherapy via oncolytic viruses. Infect Agent Cancer. 2019;14:5. doi: 10.1186/s13027-018-0218-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 632.Goradel NH, Baker AT, Arashkia A, Ebrahimi N, Ghorghanlu S, Negahdari B. Oncolytic virotherapy: challenges and solutions. Curr Probl Cancer. 2021;45:100639. doi: 10.1016/j.currproblcancer.2020.100639. [DOI] [PubMed] [Google Scholar]
- 633.Jin K-T, Du W-L, Liu Y-Y, Lan H-R, Si J-X, Mou X-Z. Oncolytic virotherapy in solid tumors: the challenges and achievements. Cancers (Basel) 2021;13:588. doi: 10.3390/cancers13040588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 634.Wang S, Sun J, Chen K, Ma P, Lei Q, Xing S, et al. Perspectives of tumor-infiltrating lymphocyte treatment in solid tumors. BMC Med. 2021;19:140. doi: 10.1186/s12916-021-02006-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 635.Chesney J, Lewis KD, Kluger H, Hamid O, Whitman E, Thomas S, et al. Efficacy and safety of lifileucel, a one-time autologous tumor-infiltrating lymphocyte (TIL) cell therapy, in patients with advanced melanoma after progression on immune checkpoint inhibitors and targeted therapies: pooled analysis of consecutive cohorts. J Immunother Cancer. 2022;10:e005755. doi: 10.1136/jitc-2022-005755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 636.Kunert A, Straetemans T, Govers C, Lamers C, Mathijssen R, Sleijfer S, et al. TCR-engineered T cells meet new challenges to treat solid tumors: choice of antigen, t cell fitness, and sensitization of tumor milieu. Front Immunol. 2013;4:363. doi: 10.3389/fimmu.2013.00363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 637.Debets R, Donnadieu E, Chouaib S, Coukos G. TCR-engineered T cells to treat tumors: seeing but not touching? Semin Immunol. 2016;28:10–21. doi: 10.1016/j.smim.2016.03.002. [DOI] [PubMed] [Google Scholar]
- 638.Chandran SS, Klebanoff CA. T cell receptor-based cancer immunotherapy: emerging efficacy and pathways of resistance. Immunol Rev. 2019;290:127–147. doi: 10.1111/imr.12772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 639.Choi BD, Gerstner ER, Frigault MJ, Leick MB, Mount CW, et al. Intraventricular CARv3-TEAM-E T cells in recurrent glioblastoma. N Engl J Med. 2024;390:1290–1298. doi: 10.1056/NEJMoa2314390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 640.Spiegel JY, Patel S, Muffly L, Hossain NM, Oak J, Baird JH, et al. CAR T cells with dual targeting of CD19 and CD22 in adult patients with recurrent or refractory B cell malignancies: a phase 1 trial. Nat Med. 2021;27:1419–1431. doi: 10.1038/s41591-021-01436-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 641.Bagley SJ, Logun M, Fraietta JA, Wang X, Desai AS, Bagley LJ, et al. Intrathecal bivalent CAR T cells targeting EGFR and IL13Rα2 in recurrent glioblastoma: phase 1 trial interim results. Nat Med. 2024; [DOI] [PubMed]
- 642.Ebrahimiyan H, Tamimi A, Shokoohian B, Minaei N, Memarnejadian A, Hossein-Khannazer N, et al. Novel insights in CAR-NK cells beyond CAR-T cell technology; promising advantages. Int Immunopharmacol. 2022;106:108587. doi: 10.1016/j.intimp.2022.108587. [DOI] [PubMed] [Google Scholar]
- 643.Marin D, Li Y, Basar R, Rafei H, Daher M, Dou J, et al. Safety, efficacy and determinants of response of allogeneic CD19-specific CAR-NK cells in CD19+ B cell tumors: a phase 1/2 trial. Nat Med. 2024;30:772–784. doi: 10.1038/s41591-023-02785-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 644.Cappuzzello E, Vigolo E, D’Accardio G, Astori G, Rosato A, Sommaggio R. How can Cytokine-induced killer cells overcome CAR-T cell limits. Front Immunol. 2023;14:1229540. doi: 10.3389/fimmu.2023.1229540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 645.Karasarides M, Cogdill AP, Robbins PB, Bowden M, Burton EM, Butterfield LH, et al. Hallmarks of resistance to immune-checkpoint inhibitors. Cancer Immunol Res. 2022;10:372–383. doi: 10.1158/2326-6066.CIR-20-0586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 646.Fares CM, Van Allen EM, Drake CG, Allison JP, Hu-Lieskovan S, Allen EM Van, et al. Mechanisms of resistance to immune checkpoint blockade: why does checkpoint inhibitor immunotherapy not work for all patients? Am Soc Clin Oncol Educ B Am Soc Clin Oncol; 2019;147–64. [DOI] [PubMed]
- 647.Cogdill AP, Andrews MC, Wargo JA. Hallmarks of response to immune checkpoint blockade. Br J Cancer. 2017;117:1–7. doi: 10.1038/bjc.2017.136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 648.Kaczmarek M, Poznańska J, Fechner F, Michalska N, Paszkowska S, Napierała A, et al. Cancer vaccine therapeutics: limitations and effectiveness—a literature review. Cells. 2023;12:2159. doi: 10.3390/cells12172159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 649.Liu J, Fu M, Wang M, Wan D, Wei Y, Wei X. Cancer vaccines as promising immuno-therapeutics: platforms and current progress. J Hematol Oncol. 2022;15:28. doi: 10.1186/s13045-022-01247-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 650.Saxena M, Balan S, Roudko V, Bhardwaj N. Towards superior dendritic-cell vaccines for cancer therapy. Nat Biomed Eng. 2018;2:341–346. doi: 10.1038/s41551-018-0250-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 651.Perez CR, De Palma M. Engineering dendritic cell vaccines to improve cancer immunotherapy. Nat Commun. 2019;10:5408. doi: 10.1038/s41467-019-13368-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 652.Fan T, Zhang M, Yang J, Zhu Z, Cao W, Dong C. Therapeutic cancer vaccines: advancements, challenges, and prospects. Signal Transduct Target Ther. 2023;8:450. doi: 10.1038/s41392-023-01674-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 653.Shanmugaraj B, Priya LB, Mahalakshmi B, Subbiah S, Hu R-M, Velmurugan BK, et al. Bacterial and viral vectors as vaccine delivery vehicles for breast cancer therapy. Life Sci. 2020;250:117550. doi: 10.1016/j.lfs.2020.117550. [DOI] [PubMed] [Google Scholar]
- 654.Wang B, Pei J, Xu S, Liu J, Yu J. Recent advances in mRNA cancer vaccines: meeting challenges and embracing opportunities. Front Immunol. 2023;14:1246682. doi: 10.3389/fimmu.2023.1246682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 655.Sayour EJ, Boczkowski D, Mitchell DA, Nair SK. Cancer mRNA vaccines: clinical advances and future opportunities. Nat Rev Clin Oncol. 2024; [DOI] [PubMed]
- 656.Kutzler MA, Weiner DB. DNA vaccines: ready for prime time? Nat Rev Genet. 2008;9:776–788. doi: 10.1038/nrg2432. [DOI] [PMC free article] [PubMed] [Google Scholar]