Abstract
Introduction
Oxylipins are mediators of oxidative stress. To characterize the underlying inflammatory processes and phenotype effect of iron metabolism disorders, we investigated the oxylipin profile in hereditary hemochromatosis (HH) and dysmetabolic iron overload syndrome (DIOS) patients.
Methods
An LC-MS/MS-based method was performed to quantify plasma oxylipins in 20 HH and 20 DIOS patients in fasting conditions and 3 h after an iron-rich meal in HH patients.
Results
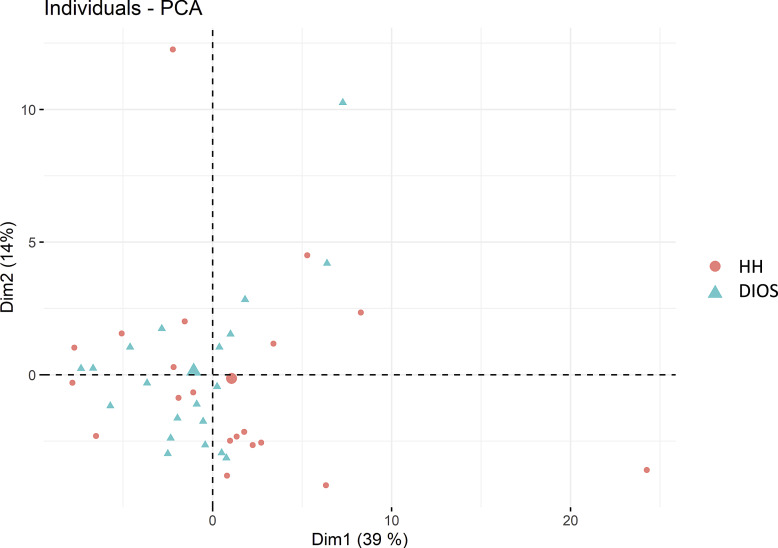
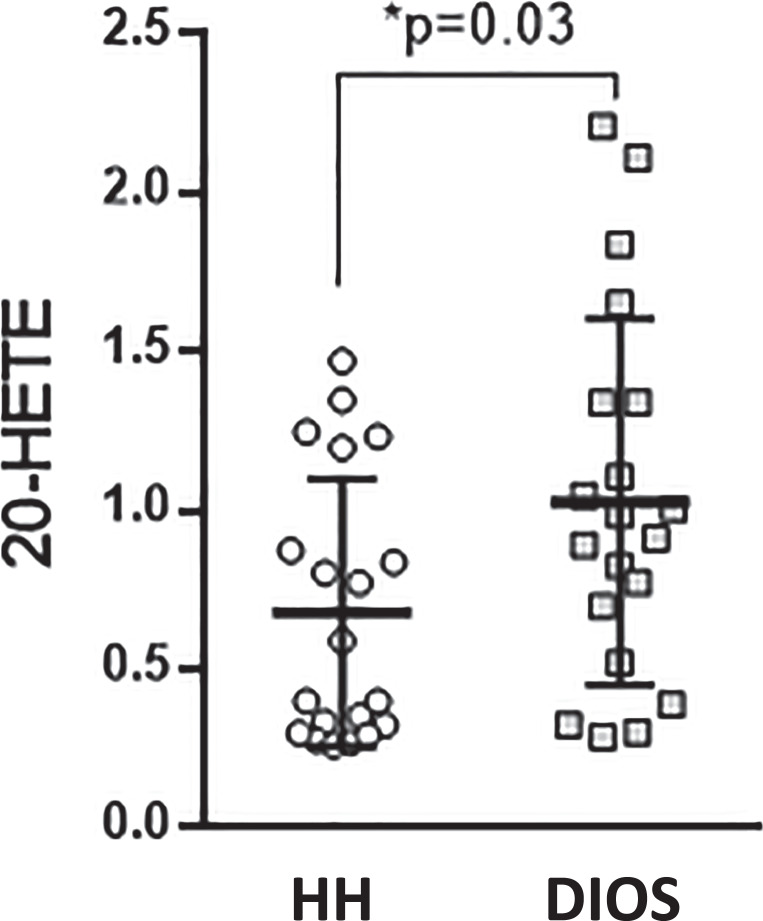
Principal component analysis showed no separation between HH and DIOS, suggesting that the clinical phenotype has no direct impact on oxylipin metabolism. 20-HETE was higher in DIOS and correlated with hypertension (p = 0.03). Different oxylipin signatures were observed in HH before and after the iron-rich meal. Discriminant oxylipins include epoxy fatty acids derived from docosahexaenoic acid and arachidonic acid as well as 13-HODE and 9-HODE. Mediation analysis found no major contribution of dietary iron absorption for 16/22 oxylipins significantly affected by the meal.
Discussion
The oxylipin profiles of HH and DIOS seemed similar except for 20-HETE, possibly reflecting different hypertension prevalence between the two groups. Oxylipins were significantly affected by the iron-rich meal, but the specific contribution of iron was not clear. Although iron may contribute to oxidative stress and inflammation in HH and DIOS, this does not seem to directly affect oxylipin metabolism.
Keywords: Oxylipin, Oxidative stress, Iron meal challenge, Hereditary hemochromatosis, Dysmetabolic iron overload syndrome
Introduction
Normal body iron stores (3–5 g) are strictly regulated in healthy subjects: dietary iron absorption balances uncontrolled losses to prevent iron deficiency and iron overload [1]. Iron is essential to ensure vital functions, but excess iron is toxic. Hereditary hemochromatosis (HH) is due to a homozygous missense substitution (p.Cys.282.Tyr) in the HFE gene promoting massive dietary iron absorption (OMIM 235200): the estimated prevalence is 1 in 379 [2]. Dysmetabolic iron overload syndrome (DIOS) is the most frequent cause of mild iron accumulation (prevalence up to 14.5% in people with metabolic syndrome [3]). There are differences between these two diseases: in DIOS, (i) hepcidin production is preserved, leading to a lower increase in hepatic iron content; (ii) iron accumulation predominates in macrophages, whereas in HH, hypohepcidinemia leads to macrophage iron depletion [4].
The exact mechanisms of iron-induced liver injury remain a subject of debate. In HH, the accumulation of iron in pancreatic cells, hepatocytes, and cardiomyocytes leads to fibrosis and organ failure [5]. In HH and in DIOS, the increase of non-transferrin-bound iron [6] induces oxidative stress [7]. Fatty acids such as palmitic acids increase cellular iron uptake (independently from the transferrin receptor) and increase iron translocation into the mitochondria, inducing the formation of reactive oxygen species and lipid oxidation [8]. In an animal model of dietary iron overload, cellular iron excess was significantly associated with the onset of severe hepatic mitochondrial dysfunction [9]. Another probable mechanism of iron toxicity is the increase of ferroptosis, cell death characterized by lipid peroxide accumulation [10]. In DIOS, macrophage iron storage is assumed to drive a pro-inflammatory phenotype promoting hepatic fibrosis [11], but some studies suggest that polarization to alternative macrophages is not associated with a pro-inflammatory profile [12, 13].
Oxylipins are a large family of lipid mediators that include the well-known eicosanoids (derived from 20 carbons-polyunsaturated fatty acids [PUFAs], e.g., prostaglandin E2 or thromboxane A2) and other PUFA-derived oxygenation metabolites [14]. They are potent signaling molecules produced on demand by various types of cells to regulate a diverse set of homeostatic and most notably inflammatory processes [15]. Free-radical derived oxylipins such as isoprostanes are also gold-standard biomarkers of oxidative stress [16]. Circulating oxylipin profiles can therefore provide a wealth of information regarding inflammation and oxidative stress, two important processes that are perturbed in HH and DIOS [17, 18]. We hypothesized that the circulating oxylipin profile could help to better phenotype and understand the effect of iron in HH and DIOS. To achieve this, we performed MS-based profiling of total plasma oxylipins in HH and DIOS patients using a recently developed and validated method [19]. We also studied the effect of an iron-rich meal intake on the postprandial oxylipin profile in HH patients to further explore the impact of the meal [20, 21] and of iron absorption on oxylipin profiles.
Materials and Methods
Study Design
This is a single-center prospective exploratory study designed as an ancillary analysis of the oxylipin profile during a randomized trial assessing the effect of polyphenols versus placebo on dietary iron absorption in HH and DIOS [22]. Blood samples for oxylipin dosage were collected in EDTA, and plasma was immediately stored at – 80°C after centrifugation. Samples were collected for each participant in fasting conditions (minimum of 6 h overnight fasting). An additional sample was collected for patients with HH 3 h after the consumption of a standardized meal, weighed in grams to ensure a reproducible 43 mg iron dietary intake (online suppl. Table 1; for all online suppl. material, see https://doi.org/10.1159/000536657). During the meal, each patient with HH received a placebo containing 218.9 mg of maltodextrin and 1.1 mg of magnesium stearate.
The study protocol was reviewed and approved by the International Review Board “Comité de Protection des personnes Sud Méditerranée I” (reference 18-10) on February 20, 2018. The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice recommendations and registered on clinicaltrials.gov (NTC03453918) and on the European Clinical Trials Database (EudraCT 2017-A01955-48). All patients gave their written informed consent.
Patients
The inclusion criterion for HH patients was p.Cys.282.Tyr homozygous mutation in the HFE gene. Inclusion criteria for DIOS were based on the study by Lainé et al. [23] including hepatic iron content assessed by magnetic resonance imaging of the liver >50 μmol/g; dry weight with hyperferritinemia >450 μg/L (two concordant determinations); and at least one of the following metabolic features: body mass index (BMI) > 25 kg/m2, abdominal obesity defined by increased waist circumference (>80 cm for women, >94 cm for men), blood pressure >130/85 mm Hg or antihypertensive therapy, high serum triglyceride level (>1.7 mmoL/L), low level of high-density lipoprotein (<1.29 mmoL/L for women, <1.03 mmol/L for men), high level of fasting blood glucose (>5.6 mmoL/L), or type 2 diabetes mellitus. The exclusion criteria were age <18 years; concomitant or use in the last 2 months of drugs or dietary supplements that could affect iron absorption (e.g., ascorbic acid, iron chelators, iron supplements, herbal tea); intestinal malabsorption of any cause; anemia (hemoglobin <9 g/dL); lactating, pregnant or planning pregnancy; alcohol consumption 20 g/day for women, 30 g/day for men; inability to understand and comply with the instructions. Therapeutic phlebotomies before and during the study were not an exclusion criterion.
Oxylipin Dosage
Using computer-generated block randomization, each sample received a number determining the sequence of analysis to preserve the blindness to disease subgroup and fasting/postprandial status. The detailed procedure for plasma total oxylipin extraction and analysis is described in the study by Dalle et al. [24]. Briefly, 100 μL of plasma was first treated with methanol for protein precipitation and hydrolyzed with 0.6 m potassium hydroxide to release the esterified oxylipins. The released oxylipins were then extracted by solid phase extraction and analyzed by liquid chromatography coupled to mass spectrometry using multiple reaction monitoring and electrospray ionization in negative mode.
Statistical Analysis
The number of patients recruited in the main study was determined to show a 30% difference of iron absorption after the meal challenge with procyanidin supplementation compared to a placebo. The results presented here are an ancillary study, and no power calculation was performed specifically for the oxylipin analyses. However, the comparison of the oxylipin profile between HH and DIOS and after the meal challenge in HH patients provided medium to large effect size bounds according to Cohen’s recommendations (0.5–0.8 size effect) [25].
Statistical analysis was performed using Stata 15 (StataCorp, College Station, TX, USA) and R (https://cran.r-project.org/) software. Continuous data were expressed as mean and standard deviation or median and interquartile range. The assumption of normality was studied using the Shapiro-Wilk test. The continuous data were compared between independent groups (between HH and DIOS) using Student’s t test or Mann-Whitney test when the assumptions of the t test were not met. The assumption of homoscedasticity was studied using the Fisher-Snedecor test. Paired comparisons (between HH and DIOS patients, before and after the meal challenge in HH patients) were conducted using paired Student’s t test or Wilcoxon test. Comparisons between independent groups concerning categorical variables were performed using the χ2 test or Fisher’s exact test. The tests were two-sided, with a type I error set at 5%. The individual p values were reported without applying mathematical correction as these analyses were considered as exploratory. Significant results based upon exploratory analyses were clearly labeled as exploratory results. For these analyses, specific attention was given to the magnitude of differences and to clinical relevance. The results were expressed with effect sizes and 95% confidence intervals and interpreted according to the aforementioned rules of thumb reported by Cohen. A principal component analysis (PCA) was used to determine correlated oxylipins and hidden patterns of patients related to them (profiles of correlated oxylipins) and to the meal for HH patients.
A mediation analysis was conducted to assess the respective contributions of iron variation and meal challenge on oxylipin changes at 3 h. A mediation proportion was estimated, indicating how much of the whole increment value provided by an independent variable can be explained by the indirect path in which changes in this independent variable drive a change in the mediator and changes in the mediator that then affect are associated with the outcome. Results were expressed as the mediation proportion of total effect mediated through iron variation and the significance of the mediation analysis associations. Sex and age were considered as adjustment covariates to take into account their possible confounding effect. Furthermore, the mediation proportion of total effect mediated through age was also evaluated. A multilevel mediation analysis was performed with sex and age being integrated.
Results
Between March and July 2018, 91 patients followed up in our department were screened during their routine medical appointment: 20 patients (1 woman) with DIOS and 20 patients (10 women) with HH completed the study. Baseline characteristics are shown in Table 1. Patients with DIOS had a significantly higher BMI, waist circumference, and serum ferritin level. DIOS patients were also more frequently affected by hypertension.
Table 1.
Clinical, demographic, and biological baseline characteristics of the patients
| Parameters | HH, n = 20 | DIOS, n = 20 | p value |
|---|---|---|---|
| Age, years | 55.4±15.3 | 61±7.5 | 0.15 |
| Sex, male, n (%) | 10 (50) | 19 (95) | 0.003 |
| BMI, kg/m2 | 24.9±3.5 | 27.8±3.0 | 0.008 |
| Waist circumference, cm | 90.5±11.8 | 101.5±10.7 | 0.003 |
| Hypertension, n (%) | 3 (15) | 14 (70) | 0.001 |
| Therapeutic phlebotomies, n (%) | 18 (80) | 0 | <0.001 |
| Number of metabolic syndrome criteria, n (%) | |||
| 1 | 1 (5) | ||
| 2 | 2 (10) | ||
| 3 | 4 (20) | ||
| 4 | 8 (40) | ||
| 5 | 5 (25) | ||
| Ferritin, µg/L | 54 [31;85] | 508 [267;597] | <0.001 |
| Hemoglobin, g/dL | 14.8±1.0 | 15.6±1.3 | 0.03 |
| CRP, mg/L | 3.3±1.1 | 3.2±0.9 | 0.7 |
| Fasting venous glycemia, µmol/L | 4.8±0.6 | 5.5±2.2 | 0.2 |
| Haptoglobin, g/L | 1.2±0.4 | 1.2±0.5 | 0.9 |
| LDH, IU/L | 224.7±41.1 | 221.6±36.2 | 0.8 |
| AST, IU/L | 22.4±6.8 | 25.3±6.5 | 0.2 |
| ALT, IU/L | 29.1±12.9 | 42.4±15.4 | 0.005 |
| gGT, IU/L | 33.1±21.3 | 44.2±17.5 | 0.08 |
| ALP, IU/L | 61.2±14 | 68±19.1 | 0.5 |
| Total bilirubin, µmol/L | 9.9±5.3 | 10.5±3.5 | 0.7 |
Data are presented as mean ± standard deviation or median [quartile 1;quartile 3] according to the statistical distribution, unless indicated otherwise.
ALP, alkaline phosphatase; ALT, alanine transaminase; ASP, aspartate transaminase; BMI, body mass index; CRP, C-reactive protein; DIOS, dysmetabolic iron overload syndrome; HH, hereditary hemochromatosis; LDH, lactate dehydrogenase.
Total Oxylipin Profile in Fasted Plasma in DIOS and HH Patients
Among the 133 oxylipins included in the acquisition method, 82 could be quantified (i.e., > lower limit of quantification). Oxylipin profiles of HH and DIOS patients after overnight fasting were analyzed using unsupervised PCA, as shown in Fig. 1). The first two components (that explain, respectively, 39% and 14% of the total variance) show no clear separation of HH and DIOS patients, suggesting very similar oxylipin profiles. The detail of each metabolite contribution to each dimension is available in online supplementary Table 2.
Fig. 1.
PCA of the total plasma oxylipin profile in HH and DIOS patients.
Univariate analysis was used to precisely identify which individual oxylipins may be modulated by health status. No significant difference was observed for most oxylipins except for 20-HETE which was significantly higher in the DIOS group (0.95 [0.61; 1.34] vs. 0.50 [0.31; 1.04], p = 0.03, size effect = −0.68, shown in Fig. 2). The detailed concentration of all the quantified oxylipins is available in online supplementary Table 3.
Fig. 2.
Boxplot showing plasma concentration (nM) of 20-HETE in DIOS and HH patients. DIOS, dysmetabolic iron overload syndrome; HH, hereditary hemochromatosis.
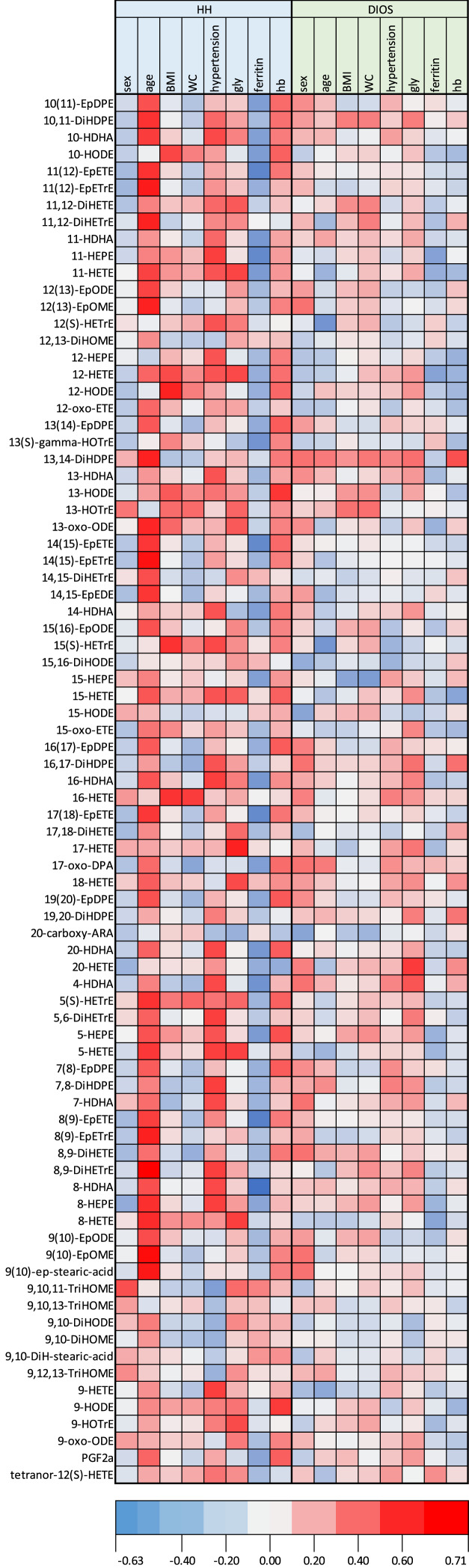
A correlation heat map was built to assess the links between each individual oxylipin and clinical and biological parameters (shown in Fig. 3). Hypertension was positively associated with 20-HETE (R2 = 0.37; p = 0.03) for both HH and DIOS patients. Ferritin was negatively correlated with a high level of many oxylipins in both groups. Age appeared positively correlated with oxylipins in the HH group. BMI and WC showed similar trends of correlations with the oxylipin levels in both diseases.
Fig. 3.
Heatmap correlation analysis (Spearman’s rho) of oxylipin with metabolic parameters and hemoglobin level in DIOS and HH. The correlation between the oxylipin level and metabolic parameters was assessed by Spearman’s rank correlation coefficient. The color intensity represents the degree of correlation. Blue indicates negative correlations, and red indicates positive correlations. Light gray indicates the absence of correlation. BMI, body mass index (kg.m-2); DIOS, dysmetabolic iron overload syndrome; gly, fasting venous glycaemia; hb, hemoglobin at the inclusion visit; HH, hereditary hemochromatosis; WC, waist circumference (cm).
Changes in Plasma Oxylipin Profiles after an Iron-Rich Meal Challenge in HH
The plasma oxylipin profile of HH patients before and after (180 min) an iron-rich meal was analyzed using PCA (shown in Fig. 4). The first two components (explaining, respectively, 41% and 12% of the total variance) showed a relatively good separation of individuals, suggesting distinct oxylipin profiles. The distinction between before and after (180 min) an iron-rich meal by the first PCA component was mainly driven by epoxy fatty acids derived from docosahexaenoic acid (DHA; i.e., EdPDE) and arachidonic acid (i.e., EpETE), while the most important variables on the second component were 13-HODE and 9-HODE (see online suppl. Fig. 1; online suppl. Table 4).
Fig. 4.
PCA of oxylipin profile variations in HH after the iron-rich meal challenge (3 h after the meal).
This multivariate analysis was completed by univariate analysis to assess the effect of the iron-rich meal on each oxylipin taken individually. The 22 identified oxylipins with significant variations after the meal again included 9-HODE and 13-HODE as well as other oxylipins mostly including various hydroxy fatty acids and some of their catabolites (11-HDHA, 11-HEPE, 12(S)-HETrE, 12-HETE, tetranor-12(S)-HETE, 13-HOTrE, 15-HODE, 16-HDHA, 16-HETE, 18-HETE, 9-HOTrE, 9 oxo-ODE), epoxy fatty acids and their dihydroxy derivates (15(16)-EpODE, 15,16-DiHODE, 12,13-DiHOME, 19,20-DiHDPE, 9,10-DiHODE), trihydroxy fatty acids (9,10,11-TriHOME, 9,12,13-TriHOME) as well as one prostaglandin (PGF2α).
The serum iron significantly increased in HH patients after the iron-rich meal (p < 0.001): mean serum iron increased compared to baseline fasting conditions at 120 min (28.64 ± 8.7 vs. 24.73 ± 9.94; 15.8% increase), 180 min (31.02 ± 9.09 vs. 24.96 ± 10.17 mmoL/L; 24.2% increase), and 240 min (31.76 ± 8.06 vs. 25.25 ± 10.20 mmoL/L; 25.7% increase) [22]. To go further in understanding the potential link between the increase of the plasma iron level and alterations of the oxylipin profile, we performed a mediation analysis (Table 2) for the 22 oxylipins with significant variations after the meal. Six oxylipins (11-HEPE, 12(S)-HETrE, 12-HETE, 16-HDHA, 16-HETE, and 18-HETE) with a large size effect variation were mediated by 33–77% through the iron increase after the meal. For the remaining 16 oxylipins, the share of iron variation in the alteration of the oxylipin level was low 0–26%). We found similar results when we added age in the mediation analysis model (data not shown).
Table 2.
Mediation analysis of significant oxylipin variations through iron variations after meal challenge in HH
| Oxylipin | Fasting values | Postprandial values | Size effect | Percentage of oxylipin variations mediated by iron variations |
|---|---|---|---|---|
| 16-HDHA | 0.93 [0.77–1.22] | 0.76 [0.57–1.16] | −0.56 | 77 |
| 12(S)-HETrE | 0.52 [0.44–0.79] | 0.70 [0.57–0.96] | 0.55 | 49 |
| 18-HETE | 0.65 [0.55–0.81] | 0.51 [0.45–0.62] | −0.54 | 49 |
| 16-HETE | 0.37 [0.32–0.43] | 0.31 [0.24–0.36] | −0.68 | 37 |
| 11-HEPE | 0.22 [0.16–0.33] | 0.15 [0.09–0.28] | −0.58 | 37 |
| 12-HETE | 2.78 [2.53–3.48] | 4.94 [2.65–10.27] | 0.75 | 33 |
| 11-HDHA | 1.33 [1.04–1.66] | 0.93 [0.72–1.53] | −0.58 | 26 |
| 13-HODE | 31.76 [27.78–39.51] | 55.82 [44.44–65.82] | 1.64 | 20 |
| 9,10,11-TriHOME | 0.64 [0.53–1.05] | 1.06 [0.84–1.38] | 0.82 | 20 |
| Tetranor-12(S)-HETE | 0.10 [0.09–0.18] | 0.18 [0.13–0.24] | 0.75 | 20 |
| 9,10-DiHODE | 0.24 [0.16–0.43] | 0.55 [0.39–0.70] | 0.98 | 16 |
| 12,13-DiHOME | 3.95 [3.22–5.15] | 6.58 [5.07–8.09] | 0.97 | 16 |
| PGF2a | 4.27 [2.94–6.10] | 3.88 [2.46–5.28] | −0.61 | 16 |
| 9-HOTrE | 1.35 [0.84–1.62] | 6.82 [3.93–9.12] | 2.36 | 11 |
| 9-HODE | 25.96 [19.46–35.50] | 69.967 [47.08–103.69] | 2.08 | 10 |
| 9,12,13-TriHOME | 6.08 [4.21–11.76] | 12.49 [9.77–17.06] | 0.62 | 9 |
| 13-HOTrE | 1.18 [1.00–1.71] | 3.99 [3.11–4.84 | 2.08 | 8 |
| 15(16)-EpODE | 17.61 [9.92–24.58] | 55.45 [29.79–92.41] | 1.38 | 8 |
| 19,20-DiHDPE | 1.11 [0.91–1.31] | 0.95 [0.83–1.06] | −0.54 | 7 |
| 15,16-DiHODE | 6.18 [4.51–8.07] | 23.68 [21.03–30.25] | 2.59 | 4 |
| 9-Oxo-ODE | 4.35 [0.84–1.62] | 13.51 [8.17–15.88] | 1.26 | 4 |
| 15-HODE | 1.41 [1.18–2.02] | 9.19 [6.20–11.18] | 2.50 | 0 |
Data are presented as median [quartile 1 – quartile 3].
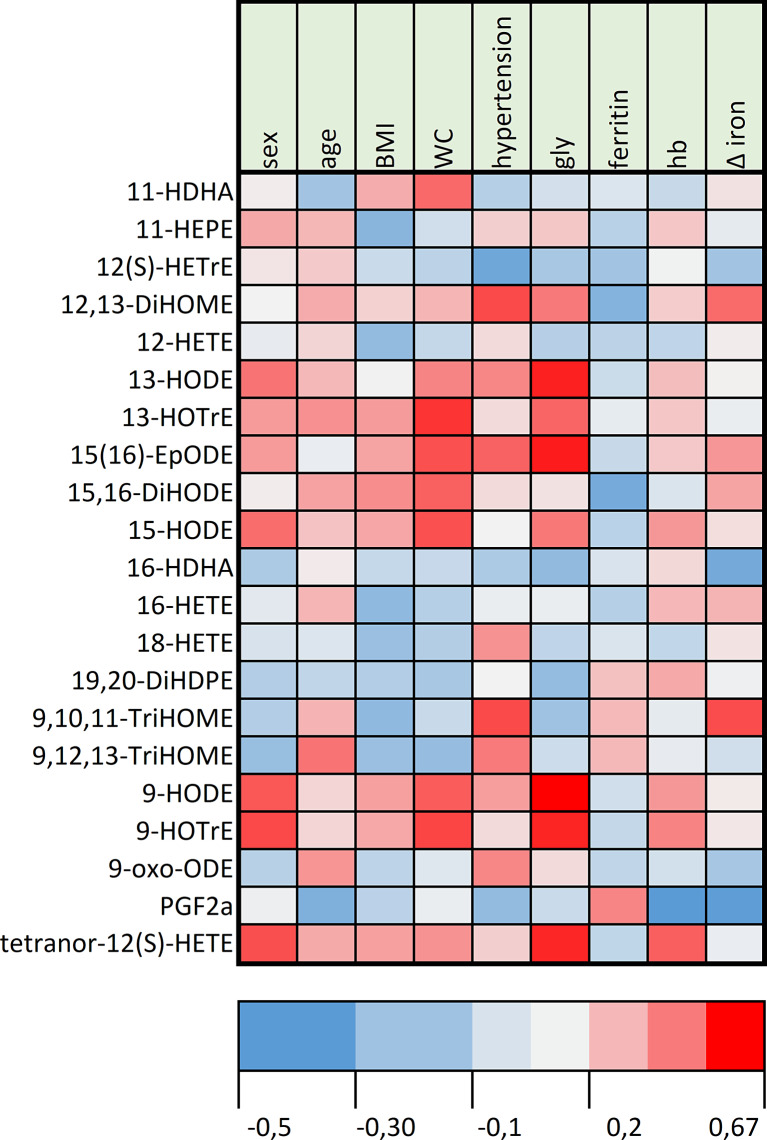
Figure 5 shows a heatmap representation of significant oxylipin variations with iron absorption and the main metabolic parameters of the patients. The serum ferritin level was negatively correlated to the oxylipin variations. BMI and waist circumference showed similar trends of correlations with oxylipin variations.
Fig. 5.
Heatmap correlation analysis (Spearman’s rho) of oxylipin variations with metabolic parameters after the iron meal challenge in HH. The correlation between the oxylipin level, metabolic parameters, and serum iron variations after the meal at 180 min was assessed by Spearman’s rank correlation coefficient. The color intensity represents the degree of correlation. Blue indicates negative correlations, and red indicates positive correlations. Light gray indicates the absence of correlation. BMI, body mass index (kg.m-2); gly, fasting venous glycaemia; hb, hemoglobin at the inclusion visit; WC, waist circumference (cm); Δ iron, serum iron increase at 180 min after meal consumption.
Discussion
HH and DIOS are the two most common iron overload diseases. Excess iron is toxic for the body as free iron induces the production of radical oxygen species and enhances ferroptosis, a specific cell death characterized by the accumulation of lipid peroxide. Inflammation is another key factor in these diseases [26], contributing in particular to the onset of organ damage, especially liver fibrosis and cirrhosis. Eicosanoids and other oxylipins are important lipid mediators involved in inflammation and oxidative stress. We discuss here how oxylipin profiles are impacted by health status in HH and DIOS patients, modulated by a meal and dietary iron absorption in HH patients, and what it may indicate in terms of inflammation and oxidative stress.
In our study, the overall plasma oxylipin profile was not different between DIOS and HH patients. However, iron metabolism was markedly different in DIOS and HH which might have influenced oxylipin metabolism differentially. Briefly, in HH, hepatocytes are the main site of iron accumulation, whereas in DIOS, iron is captured in macrophages. In a previous pilot study comparing the oxylipin profile in patients with DIOS or metabolic syndrome without iron overload and healthy controls, we found slight alterations of the oxylipin profile [27]. When looking at individual oxylipins, we observed that the plasma levels of 20-HETE were significantly higher in patients with DIOS than in HH patients. 20-HETE is an hydroxylated metabolite of arachidonic acid, produced by cytochrome P450 [28]. It is a key regulator of vascular tone and consequently plays a critical role in hypertension onset. The elevation of 20-HETE is known to increase renal and peripheral vascular resistance and in turn to the development of hypertension [29]. The higher prevalence of hypertension in DIOS patients compared to HH patients might thus explain the higher 20-HETE [30]. In conclusion, oxylipin metabolism in HH and DIOS does not seem to be influenced by iron metabolism itself but may be rather linked to high blood pressure [24].
The iron-rich meal challenge induced a substantial increase in serum iron levels, reflecting dietary iron absorption [22] which was two-fold higher in HH patients compared with DIOS patients. Thus, we chose to assess the changes in plasma oxylipin in HH patients. The meal was associated with a marked alteration of the oxylipin signature. However, it was difficult to determine if the oxylipin shift was due to iron absorption or to the meal intake. A couple of previous studies investigated the immediate oxylipin signature in healthy [21] and obese subjects [20].
In our study, we identified a significant increase of 9-HODE and 13-HODE after the iron-rich meal challenge in HH patients. These oxidized linoleic acid metabolites are pro-inflammatory molecules [31] involved in atherogenesis [32] through the decreased secretion of the fibrinolytic inhibitor plasminogen activator inhibitor-1 [33] and the inhibition of platelet activation [34]. Moreover, 9-HODE and 13-HODE are involved in the regulation of innate immune cells (macrophages and monocytes) through the stimulation of lipid uptake and apoptosis [35, 36]. In macrophages, 9-HODE increases the expression of pro-inflammatory cytokines (TNF-α, MIP2-α) [37]. Additionally, 13-HODE inhibits the neutrophil production of leukotriene B4 which is implicated in the recruitment and activation of leucocytes enhancing tissue inflammation through the increased production of pro-inflammatory cytokines [38].
In a single healthy patient study, Gouveia et al. [21] found a postprandial decrease of 9-HODE, but the result was significant only at 1 h after the meal (340 g banana providing an estimated amount of 0.68 mg of nonheme iron with low fat intake). Strassburg et al. [20] showed that variations of oxylipins after the meal challenge were correlated to the fatty acid meal composition: the consumption of a saturated fatty acid-rich meal induced an increase of 9-HODE, which is in line with the high proportion of linoleic acid in our standardized meal (estimate fat intake in our study provided in online supplementary Table 1: 38 g of lipid including 3.8 g of linoleic acid, which was 2.5 times lower than the fat shake provided by Strassburg et al. in their study with no estimation of the amount of iron provided by the meal). In contrast, we found a significant increase of 12,13-DiHOME after the meal, whereas Strassburg and Gouveia found a significant decrease in the postprandial state. 12,13-DiHOME is a metabolite of linoleic acid formed by the hydroxylation of 12,13 EpOME. The biological functions of 12,13-Di-HOME are still being debated: a lipid infusion was shown to increase the plasma level of 12,13-Di-HOME in healthy volunteers [39], although increased concentration of 12,13-Di-HOME has been associated with improved metabolic health mediated by the brown adipose tissue [40].
We found low to no correlation between the amount of absorbed dietary iron (represented by the serum iron variations) on the one hand and the oxylipin profile variations on the other hand (Table 2), suggesting that the shift of oxylipins observed was rather due to the meal intake than the iron itself. To the best of our knowledge, the link between iron absorption and postprandial alterations of oxylipin signature was never assessed before. The most correlated variation of oxylipin was the decrease of 16-HDHA, which is an autoxidation product of DHA, a long chain ω3 PUFA. 18-HETE was significantly decreased after the meal, a phenomenon shown to be correlated to insulin resistance [41]. 12(S)-HETrE is the product of the nonheme oxygenase oxidation of ω-6 PUFA which has been shown to play a protective role in cardio protection through the inhibition of thrombosis, especially in the case of vascular damage [42]. To date, none of these oxylipins have been investigated as a cofactor of iron overload damage in chronic or acute conditions. As hepatic steatosis appears to be a cofactor of the onset of cirrhosis in HH, a recent study investigating lipid metabolism in a small cohort of HH patients treated by phlebotomies highlighted an increased level of triglycerides and of polyunsaturated-fatty acids compared to healthy subjects [43]. Our results supported no increase of pro-oxidative oxylipin in the postprandial state despite the large amount of absorbed iron. We hypothesize that this might reflect that iron toxicity in HH is due to chronic accumulation inducing an increase of free iron, leading to organ damage such as heart and liver fibrosis. The increase of non-transferrin bound iron in homozygous HFE patients was recently shown in non-iron-depleted patients [44], reflecting chronic iron accumulation. The authors confirmed a strong decrease in free iron after therapeutic phlebotomy; however, most of the patients in our cohort were also treated by maintenance bloodletting. In chronic hepatitis C, a pilot study of iron depletion by therapeutic phlebotomies showed a decrease of 8-isoprostane that may reflect a decrease of oxidative stress [45]. However, the reduction in oxidative markers in the initial phase must be weighed against possible rebound phenomena during the maintenance phase [46]. Thus, a comparison of the oxylipin profile in asymptomatic HH patients and in symptomatic patients (with clinical and biochemical abnormalities) at different phases of iron depletion treatment could be of great interest to further explore the underlying mechanism driving liver injury. The effect of therapeutic phlebotomy itself on immediate oxidative stress may also be investigated.
To conclude, the oxylipin profiles were very similar between HH and DIOS patients except for a higher 20-HETE level in DIOS, which is more likely to reflect the difference in blood pressure. In HH patients, significant changes in oxylipin signature were found after an iron-rich meal challenge, but it was not possible to determine specifically the contribution of iron in this effect. In conclusion, in HH and DIOS, high levels of serum iron do not induce strong changes in the oxylipin metabolism of patients.
Acknowledgments
We want to thank Justine Bertrand-Michel and Pauline Le Fadouer from the MetaToul platform UMR 5504, UMR 792, CNRS, INRAE, INSA 135 Avenue de Rangueil 31077 Toulouse, for performing the oxylipin analyses. We would like to thank the “Derives Resiniques et Terpeniques” company (DRT) who made a generous grant to support this work.
Statement of Ethics
The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice recommendations and registered on clinicaltrials.gov (NTC03453918) and on the European Clinical Trials Database (EudraCT 2017-A01955-48). The study protocol was reviewed and approved by the International Review Board “Comité de Protection des personnes Sud Méditerranée I” (reference 18-10) on February 20, 2018. All patients gave their written informed consent.
Conflict of Interest Statement
The authors have no conflicts of interest to declare. The DRT company had no role in conceiving and conducting the study; in collection, management, analysis, or interpretation of the data; or in preparation, review, and submission of the manuscript for publication.
Funding Sources
The research was supported by Clermont-Ferrand University Hospital (Grant No. AOI-2016-RUIVARD). The company DRT provided a grant to support the research (payment to Clermont-Ferrand University Hospital).
Author Contributions
Hervé Lobbes: conceptualization, methodology, formal analysis, investigation, writing – original draft and review and editing, and funding acquisition. Céline Dalle: investigation, resources, validation, and writing – review and editing. Bruno Pereira: methodology, data curation, formal analysis, software, and writing – review and editing. Marc Ruivard and Andrzej Mazur: conceptualization, project administration, funding acquisition, mentorship, and writing – review and editing. Cécile Gladine: conceptualization, methodology, formal analysis, resources, mentorship, writing – original draft and review and editing, and funding acquisition.
Funding Statement
The research was supported by Clermont-Ferrand University Hospital (Grant No. AOI-2016-RUIVARD). The company DRT provided a grant to support the research (payment to Clermont-Ferrand University Hospital).
Data Availability Statement
The data that support the findings of this study are not publicly available due to local legal constraints but are available from the corresponding author (Dr. Cécile Gladine, cecile.gladine@inrae.fr) upon reasonable request.
Supplementary Material
Supplementary Material
Supplementary Material
Supplementary Material
Supplementary Material
References
- 1. Anderson GJ, Frazer DM. Current understanding of iron homeostasis. Am J Clin Nutr. 2017;106:S1559–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Wallace DF, Subramaniam VN. The global prevalence of HFE and non-HFE hemochromatosis estimated from analysis of next-generation sequencing data. Genet Med. 2016;18(6):618–26. [DOI] [PubMed] [Google Scholar]
- 3. Bozzini C, Girelli D, Olivieri O, Martinelli N, Bassi A, De Matteis G, et al. Prevalence of body iron excess in the metabolic syndrome. Diabetes Care. 2005;28(8):2061–3. [DOI] [PubMed] [Google Scholar]
- 4. Valenti L, Corradini E, Adams LA, Aigner E, Alqahtani S, Arrese M, et al. Consensus Statement on the definition and classification of metabolic hyperferritinaemia. Nat Rev Endocrinol. 2023;19(5):299–310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Brissot P, Pietrangelo A, Adams PC, de Graaff B, McLaren CE, Loréal O. Haemochromatosis. Nat Rev Dis Primers. 2018;4:18016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Loréal O, Gosriwatana I, Guyader D, Porter J, Brissot P, Hider RC. Determination of non-transferrin-bound iron in genetic hemochromatosis using a new HPLC-based method. J Hepatol. 2000;32(5):727–33. [DOI] [PubMed] [Google Scholar]
- 7. Ahmed U, Latham PS, Oates PS. Interactions between hepatic iron and lipid metabolism with possible relevance to steatohepatitis. World J Gastroenterol. 2012;18(34):4651–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Yao D, Shi W, Gou Y, Zhou X, Yee Aw T, Zhou Y, et al. Fatty acid-mediated intracellular iron translocation: a synergistic mechanism of oxidative injury. Free Radic Biol Med. 2005;39(10):1385–98. [DOI] [PubMed] [Google Scholar]
- 9. Bacon BR, Park CH, Brittenham GM, O’Neill R, Tavill AS. Hepatic mitochondrial oxidative metabolism in rats with chronic dietary iron overload. Hepatology. 1985;5(5):789–97. [DOI] [PubMed] [Google Scholar]
- 10. Wang H, An P, Xie E, Wu Q, Fang X, Gao H, et al. Characterization of ferroptosis in murine models of hemochromatosis. Hepatology. 2017;66(2):449–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Vinchi F, Costa da Silva M, Ingoglia G, Petrillo S, Brinkman N, Zuercher A, et al. Hemopexin therapy reverts heme-induced proinflammatory phenotypic switching of macrophages in a mouse model of sickle cell disease. Blood. 2016;127(4):473–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Corna G, Campana L, Pignatti E, Castiglioni A, Tagliafico E, Bosurgi L, et al. Polarization dictates iron handling by inflammatory and alternatively activated macrophages. Haematologica. 2010;95(11):1814–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Lahaye C, Gladine C, Pereira B, Berger J, Chinetti-Gbaguidi G, Lainé F, et al. Does iron overload in metabolic syndrome affect macrophage profile? A case control study. J Trace Elem Med Biol. 2021;67:126786. [DOI] [PubMed] [Google Scholar]
- 14. Gladine C, Ostermann AI, Newman JW, Schebb NH. MS-based targeted metabolomics of eicosanoids and other oxylipins: analytical and inter-individual variabilities. Free Radic Biol Med. 2019;144:72–89. [DOI] [PubMed] [Google Scholar]
- 15. Dennis EA, Norris PC. Eicosanoid storm in infection and inflammation. Nat Rev Immunol. 2015;15(8):511–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Milne GL, Sanchez SC, Musiek ES, Morrow JD. Quantification of F2-isoprostanes as a biomarker of oxidative stress. Nat Protoc. 2007;2(1):221–6. [DOI] [PubMed] [Google Scholar]
- 17. Dongiovanni P, Fracanzani AL, Fargion S, Valenti L. Iron in fatty liver and in the metabolic syndrome: a promising therapeutic target. J Hepatol. 2011;55(4):920–32. [DOI] [PubMed] [Google Scholar]
- 18. Fernandez M, Lokan J, Leung C, Grigg A. A critical evaluation of the role of iron overload in fatty liver disease. J Gastroenterol Hepatol. 2022;37(10):1873–83. [DOI] [PubMed] [Google Scholar]
- 19. Mainka M, Dalle C, Pétéra M, Dalloux-Chioccioli J, Kampschulte N, Ostermann AI, et al. Harmonized procedures lead to comparable quantification of total oxylipins across laboratories. J Lipid Res. 2020;61(11):1424–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Strassburg K, Esser D, Vreeken RJ, Hankemeier T, Müller M, van Duynhoven J, et al. Postprandial fatty acid specific changes in circulating oxylipins in lean and obese men after high-fat challenge tests. Mol Nutr Food Res. 2014;58(3):591–600. [DOI] [PubMed] [Google Scholar]
- 21. Gouveia-Figueira S, Späth J, Zivkovic AM, Nording ML. Profiling the oxylipin and endocannabinoid metabolome by UPLC-ESI-MS/MS in human plasma to monitor postprandial inflammation. PLoS One. 2015;10(7):e0132042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Lobbes H, Gladine C, Mazur A, Pereira B, Dualé C, Cardot J-M, et al. Effect of procyanidin on dietary iron absorption in hereditary hemochromatosis and in dysmetabolic iron overload syndrome: a crossover double-blind randomized controlled trial. Clin Nutr. 2020;39(1):97–103. [DOI] [PubMed] [Google Scholar]
- 23. Lainé F, Ruivard M, Loustaud‐Ratti V, Bonnet F, Calès P, Bardou‐Jacquet E, et al. Metabolic and hepatic effects of bloodletting in dysmetabolic iron overload syndrome: a randomized controlled study in 274 patients. Hepatology. 2017;65(2):465–74. [DOI] [PubMed] [Google Scholar]
- 24. Dalle C, Tournayre J, Mainka M, Basiak-Rasała A, Pétéra M, Lefèvre-Arbogast S, et al. The plasma oxylipin signature provides a deep phenotyping of metabolic syndrome complementary to the clinical criteria. Int J Mol Sci. 2022;23(19):11688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Cohen J. A power primer. Psychol Bull. 1992;112(1):155–9. [DOI] [PubMed] [Google Scholar]
- 26. Esser N, Legrand-Poels S, Piette J, Scheen AJ, Paquot N. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Res Clin Pract. 2014;105(2):141–50. [DOI] [PubMed] [Google Scholar]
- 27. Dalle C, Lahaye C, Bosviel R, Ruivard M, Gladine C, Mazur A. MS-Based lipidomic profiling of oxylipins supports mild inflammation in dysmetabolic iron overload syndrome affected patients (P08-046-19). Curr Dev Nutr. 2019;3:nzz044.P08–046-19 (Supplement_1):nzz044. [Google Scholar]
- 28. Roman RJ. P-450 metabolites of arachidonic acid in the control of cardiovascular function. Physiol Rev. 2002;82(1):131–85. [DOI] [PubMed] [Google Scholar]
- 29. Roman RJ, Fan F. 20-HETE: hypertension and beyond. Hypertension. 2018;72(1):12–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Waldman M, Peterson SJ, Arad M, Hochhauser E. The role of 20-HETE in cardiovascular diseases and its risk factors. Prostaglandins Other Lipid Mediat. 2016;125:108–17. [DOI] [PubMed] [Google Scholar]
- 31. Pecorelli A, Cervellati C, Cordone V, Amicarelli F, Hayek J, Valacchi G. 13-HODE, 9-HODE and ALOX15 as potential players in Rett syndrome OxInflammation. Free Radic Biol Med. 2019;134:598–603. [DOI] [PubMed] [Google Scholar]
- 32. Vangaveti V, Baune BT, Kennedy RL. Hydroxyoctadecadienoic acids: novel regulators of macrophage differentiation and atherogenesis. Ther Adv Endocrinol Metab. 2010;1(2):51–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Marx N, Bourcier T, Sukhova GK, Libby P, Plutzky J. PPARgamma activation in human endothelial cells increases plasminogen activator inhibitor type-1 expression: PPARgamma as a potential mediator in vascular disease. Arterioscler Thromb Vasc Biol. 1999;19(3):546–51. [DOI] [PubMed] [Google Scholar]
- 34. Coene MC, Bult H, Claeys M, Laekeman GM, Herman AG. Inhibition of rabbit platelet activation by lipoxygenase products of arachidonic and linoleic acid. Thromb Res. 1986;42(2):205–14. [DOI] [PubMed] [Google Scholar]
- 35. Nagy L, Tontonoz P, Alvarez JG, Chen H, Evans RM. Oxidized LDL regulates macrophage gene expression through ligand activation of PPARgamma. Cell. 1998;93(2):229–40. [DOI] [PubMed] [Google Scholar]
- 36. Fu Y, Luo N, Lopes-Virella MF, Garvey WT. The adipocyte lipid binding protein (ALBP/aP2) gene facilitates foam cell formation in human THP-1 macrophages. Atherosclerosis. 2002;165(2):259–69. [DOI] [PubMed] [Google Scholar]
- 37. Warner DR, Liu H, Miller ME, Ramsden CE, Gao B, Feldstein AE, et al. Dietary linoleic acid and its oxidized metabolites exacerbate liver injury caused by ethanol via induction of hepatic proinflammatory response in mice. Am J Pathol. 2017;187(10):2232–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Iversen L, Fogh K, Bojesen G, Kragballe K. Linoleic acid and dihomogammalinolenic acid inhibit leukotriene B4 formation and stimulate the formation of their 15-lipoxygenase products by human neutrophils in vitro. Evidence of formation of antiinflammatory compounds. Agents Actions. 1991;33(3–4):286–91. [DOI] [PubMed] [Google Scholar]
- 39. Edwards LM, Lawler NG, Nikolic SB, Peters JM, Horne J, Wilson R, et al. Metabolomics reveals increased isoleukotoxin diol (12,13-DHOME) in human plasma after acute Intralipid infusion. J Lipid Res. 2012;53(9):1979–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Macêdo APA, Muñoz VR, Cintra DE, Pauli JR. 12,13-diHOME as a new therapeutic target for metabolic diseases. Life Sci. 2022;290:120229. [DOI] [PubMed] [Google Scholar]
- 41. Shoieb SM, El-Sherbeni AA, El-Kadi AOS. Subterminal hydroxyeicosatetraenoic acids: crucial lipid mediators in normal physiology and disease states. Chem Biol Interact. 2019;299:140–50. [DOI] [PubMed] [Google Scholar]
- 42. Yeung J, Holinstat M. Who is the real 12-HETrE? Prostaglandins Other Lipid Mediat. 2017;132:25–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Seeßle J, Gan-Schreier H, Kirchner M, Stremmel W, Chamulitrat W, Merle U. Plasma Lipidome, PNPLA3 polymorphism and hepatic steatosis in hereditary hemochromatosis. BMC Gastroenterol. 2020;20(1):230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Infanti L, Leitner G, Moe MK, Pehlic V, Benkert P, Cattaneo M, et al. Indices of iron homeostasis in asymptomatic subjects with HFE mutations and moderate ferritin elevation during iron removal treatment. Blood Cells Mol Dis. 2022;97:102689. [DOI] [PubMed] [Google Scholar]
- 45. Kaito M, Iwasa M, Kobayashi Y, Fujita N, Tanaka H, Gabazza EC, et al. Iron reduction therapy by phlebotomy reduces lipid peroxidation and oxidative stress in patients with chronic hepatitis C. J Gastroenterol. 2006;41(9):921–2. [DOI] [PubMed] [Google Scholar]
- 46. Shizukuda Y, Bolan CD, Nguyen TT, Botello G, Tripodi DJ, Yau Y-Y, et al. Oxidative stress in asymptomatic subjects with hereditary hemochromatosis. Am J Hematol. 2007;82(3):249–50. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data that support the findings of this study are not publicly available due to local legal constraints but are available from the corresponding author (Dr. Cécile Gladine, cecile.gladine@inrae.fr) upon reasonable request.