Abstract
Background
The independent effect of waist-to-height ratio (WHtR) and body fat percentage (BF%) on ischemic cardiovascular disease (CVD) remains uncertain.
Objectives
This study aimed to investigate the independent associations of WHtR and BF% with ischemic CVD.
Methods
This prospective cohort study used data from the UK Biobank. BF% was calculated as fat mass divided by body weight, measured by bioimpedance. Cox models estimated hazard ratios (HRs) with 95% confidence intervals (CIs) for overall and sex-specific associations of BF% and WHtR with risks of ischemic CVD and its main subtypes [myocardial infarction (MI) and ischemic stroke (IS)], adjusted for a range of potential confounders, including mutual adjustment for BF% and WHtR.
Results
In total, 468,333 participants without existing CVD were included in the analysis. During 12 y of follow-up, 20,151 ischemic CVD events, 13,604 MIs, and 6681 ISs were recorded. WHtR was linearly associated with ischemic CVD, MI, and IS, with an HR per 5% increase of 1.23 (95% CI: 1.20, 1.25), 1.24 (95% CI: 1.21, 1.27), and 1.22 (95% CI: 1.18, 1.26), respectively, independent of BF%. A stronger association between WHtR and MI was seen in females than in males. The association of BF% with these outcomes was substantially attenuated in both sexes after adjustment for WHtR. For example, in females, the HR (highest compared with lowest fifth) was reduced from 1.94 (95% CI: 1.76, 2.15) to 1.04 (95% CI: 0.90, 1.01) for ischemic CVD, from 2.04 (95% CI: 1.79, 2.32) to 0.97 (95% CI: 0.81, 1.16) for MI, and from 1.81 (95% CI: 1.54, 2.13) to 1.07 (95% CI: 0.85, 1.33) for IS.
Conclusions
WHtR, when used as a proxy measure for central obesity, is linearly associated with ischemic CVD in both sexes, which is independent of BF%. In contrast, the relationship of BF% with these health outcomes is predominantly driven by its correlation with WHtR.
Keywords: body fat percentage, waist-to-height ratio, ischemic cardiovascular diseases, myocardial infarction, ischemic stroke, UK Biobank
Introduction
Obesity is an increasingly urgent public health issue. Obesity-attributable premature deaths and disability-adjusted life years have doubled globally during the past 2 decades, for which ischemic cardiovascular diseases (CVDs), comprising myocardial infarction (MI) and ischemic stroke (IS), are the major components [1,2]. BMI (in kg/m2) is a common measure for obesity, and its association with CVD has been well established [3,4]; nevertheless, it has been criticized when making etiological inferences about anthropometric risk factors for diseases, as it does not consider differences in body composition or body fat distribution (i.e., central obesity) [[5], [6], [7], [8]].
Body fat percentage (BF%, total mass of fat divided by body weight) reflects body composition and therefore is a more accurate measure of general obesity and demonstrates stronger associations with cardiometabolic conditions than BMI [[9], [10], [11]]. Waist-to-height ratio [WHtR, waist circumference (WC) divided by standing height] reflects fat accumulated around the abdominal area. Compared with other central obesity measures, such as WC and waist-to-hip ratio (WHR), WHtR has been shown to be a better predictor of cardiometabolic risk factors and their associated diseases [[12], [13], [14], [15], [16]]. This measure is also less affected by sex or ethnicity [17,18] and has therefore been recommended to measure obesity in the most recent United Kingdom obesity guidelines [19]. Although prior studies have demonstrated that central obesity, compared with general obesity, is a better predictor for cardiometabolic risks, most studies have focused on comparing WC or WHR with BMI [12,20]. Research evidence comparing BF% and WHtR is lacking.
Although some studies have provided evidence for an association of WHtR and BF% with ischemic CVD [[21], [22], [23], [24]], little is known about their independent effects after considering each other. One study in the European Prospective Investigation into Cancer (EPIC)-Norfolk cohort reported no independent effect of BF% after WHR was considered [23]. However, that study had a small sample size (n = 15,000) and did not specifically examine WHtR. The aim of this study was to compare the independent associations of BF% and WHtR with ischemic CVD risk in a large prospective cohort.
Methods
Study design and participants
The UK Biobank is a population-based prospective cohort of 0.5 million participants from England, Scotland, and Wales aged 40 to 69 y at recruitment (2006–2010). At the baseline assessment, a wide range of data related to socioeconomic status, health status, medical history, and lifestyle was collected via a self-administered questionnaire and a nurse-led interview, along with the collection of physical measures and biological samples. Participants’ health is followed up via linkage to electronic health records. The UK Biobank obtained ethical approval from the North West Multicenter Research Ethics Committee, and all participants provided informed consent at baseline assessment.
In this study, we excluded participants if they 1) withdrew their consent during follow-up (n = 1298); 2) were pregnant at baseline (n = 370); 3) had any CVD events at baseline (n = 22,716); or 4) had missing data on anthropometric measures (weight, height, WC, or bioimpedance measures) (n = 9744). In total, 468,333 participants were included in the analysis (Supplemental Figure 1).
Measurement
BF% was measured via bioelectrical impedance analysis (BIA), performed with the Tanita BC418MA Body Composition Analyzer. The analyzer measured body impedance with a high frequency current (50 kHz) and 8 contact electrodes. Participants were asked to place their bare feet on the analyzer platform and to keep their feet still and in good contact with the metallic electrodes while gripping the 2 handles with both hands firmly with palm and thumb contacting the metallic electrodes and arms hanging loosely by their sides. Fat mass, fat-free mass and total body weight were automatically generated by the BIA analyzer.
Height was measured using a Seca 202 telescopic height rod device (Girodmedical). WC was measured using a tape measure around the participants’ middle, just above the hipbones. BF% was computed as fat mass divided by total body weight. WHtR was computed as WC divided by standing height.
The primary outcome was incident ischemic CVD, although its main subtypes (MI and IS) were also analyzed separately. Incident ischemic CVD was defined as hospitalization or death due to MI or IS [25]. Participants were censored at the date of hospitalization, date of death, or date of last follow-up (31 March, 2021 for participants from England and Scotland and 28 February, 2018 for participants from Wales), whichever occurred first. Health outcomes were defined using International Classification of Diseases, Tenth Revision (ICD-10) codes. Briefly, we used ICD-10 codes I21 to I23, I24.1, and I25.2 for MI and I63 to I64 for IS, respectively. The exact ICD-10 codes used are shown in Supplemental Table 1. Outcome events were ascertained via linkage to death registries (primary and/or secondary cause of death) and hospital in-patient databases (primary and/or secondary diagnosis).
Socioeconomic status was assessed using educational attainment (lower than secondary, lower secondary, upper secondary, vocational education, and higher education) and the Townsend Deprivation Index (a postcode-derived measure of socioeconomic status, categorized into quartiles). Regions were determined by the location of the assessment center the participants attended at baseline (London, Southeast, Southwest, Northeast, Northwest, East Midlands, West Midlands, Yorkshire and the Humber, Wales, and Scotland). Individuals were asked to select their self-identified ethnicity from White, Asian, Black, and Mixed or others. Smoking status and alcohol consumption were self-reported and categorized as never, previous, and current users. Physical activity was measured using the International Physical Activity Questionnaire and centrally processed by the UK Biobank; individuals were categorized into 3 physical activity levels (high, moderate, and low) based on the frequency, duration, and intensity of physical activities. BMI was calculated as weight (kg) divided by height (m) squared. Participants also self-reported their diagnoses of hypertension, diabetes, and high cholesterol at baseline. Missing values for categorical variables were treated as a separate “unknown” category.
Statistical analysis
For descriptive statistics, mean with SD and frequency with percentage were used for continuous and categorical data, respectively. Cox proportional hazard models were fitted to estimate hazard ratios (HRs) and corresponding 95% confidence intervals (CIs) for the associations of BF% and WHtR with each health outcome. HRs were estimated for sex-specific fifths of each exposure, with the lowest fifth chosen as the reference group, and adjusted for regression dilution bias based on the correlation between the exposure as measured at baseline assessment and repeat assessment [26,27]. All models were stratified by region, age, education, and ethnicity and adjusted for sex, deprivation index (quartiles), smoking status, alcohol consumption, and physical activity. To assess the independent effect of each measure on risk, BF% and WHtR were mutually adjusted. As a sensitivity analysis, we additionally adjusted for BMI to determine if WHtR and BF% had associations with CVD that were, in part, independent of BMI.
Scaled Schoenfeld residuals were used to examine the assumption of proportional hazards (no violation was observed). The correlation between BF% and WHtR was measured with Pearson correlation coefficient. Variance inflation factor (VIF) was calculated to assess collinearity between BF% and WHtR, with VIF >10 indicating presence of collinearity. The floating absolute risk approach was used for visualization [28]. Test for trend was conducted by assigning the median value within each fifth of BF% or WHtR and including this as a continuous variable in the model. We further estimated the association for each 5-unit (%) increase in BF% and WHtR. Subgroup analysis stratified by sex was performed to obtain sex-specific associations. We estimated the P values for sex difference by including an interaction term between sex and exposure (continuous) in the regression model for the overall cohort. Additional subgroup analyses were performed based on age groups (<50, 50–60, ≥60 y), height (less than sex-specific median, greater than or equal to sex-specific median), age × height interaction, smoking status, drinking status, physical activity level, hypertension, diabetes, and high cholesterol.
To assess the potential impact of reverse causation, we excluded participants with outcome events occurring during the first 2 y of follow-up. To assess the impact of comparing sex-combined quintiles with sex-specific quintiles to define BF% and WHtR fifths, we compared the results from each of these models. We also performed a sensitivity analysis by additionally adjusting for hypertension, diabetes, and high cholesterol.
In the UK Biobank, ∼34,000 individuals had their BF% simultaneously measured by BIA and dual-energy X-ray absorptiometry (DXA), which is a more accurate method for body composition measurement than BIA. Although previous research has revealed a strong correlation between body composition data derived from the 2 methods, there remained small individual-level differences [29]. Therefore, we performed a sensitivity analysis investigating the association between ischemic CVD and BIA- and DXA-derived BF% in this subset of participants, adjusted for WHtR and other covariates, to evaluate whether the difference between BIA and DXA would influence the results. Due to the substantially reduced sample size, we categorized BF% into 3 levels instead of 5 levels as done in the primary analysis.
Additionally, we categorized WHtR by 0.5, a commonly used arbitrary cutoff value [19] and investigated the association between this binary WHtR (high compared with low) and the outcomes, adjusted for BF% and other covariates. We calculated risk differences between people with high and low WHtR. We also estimated the fraction of all ischemic CVD events that were attributable to high WHtR in the UK Biobank population (i.e., population attributable fraction), using the same method as used in this article [30]. Because WC is also a commonly used measure for central obesity, we estimated the agreement of WC and WHtR in identifying individuals with obesity. The cutoff values for categorizing WC (high compared with low) were 90 cm and 100 cm for females and males, respectively, as recommended by the International Atherosclerosis Society and International Chair on Cardiometabolic Risk Working Group on Visceral Obesity [31]. Using those with low WHtR and low WC as the reference group, we calculated the HR (95% CI) for ischemic CVDs for the other 3 groups (low WHtR with high WC, high WHtR with low WC, and high WHtR with high WC). All analyses were performed using R software (version 3.6.0; R Development Core Team).
Results
Baseline characteristics of the participants are shown in Table 1 and Supplemental Table 2. The average age of the participants was 56.3 (SD 8.08) y, and 55.4% were female (Table 1). Compared with males, females were less likely to be current smokers, current drinkers, and to have a low physical activity level, hypertension, diabetes, and high cholesterol. Females had a higher mean BF% [36.5 (6.88) compared with 25.2 (5.71)] and lower WHtR [52.1 (7.87) compared with 55.1 (6.41)] than males. Participants with a higher WHtR were more likely to be older, have a lower education level, a lower physical activity level, higher BMI, hypertension, diabetes, and high cholesterol, and were less likely to be current drinkers (Table 1, Supplemental Table 2). In addition, those with a higher BF% were less likely to be current smokers (Supplemental Table 3). The Pearson correlation coefficient between WHtR and BF% was 0.45 in the overall cohort, 0.81 in males, and 0.79 in females. WHtR and BF% in the overall cohort had VIF of 1.37 and 1.37, respectively, indicating an absence of evidence for collinearity, which was similar in both sexes (VIF = 3.25 and 3.26 for WHtR and BF% in males, and 3.26 and 3.27 in females).
TABLE 1.
Baseline characteristics of participants, by sex and fifths of waist-to-height ratio (%)
| Males |
Females |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fifth 1: <49.7 |
Fifth 2: (49.7, 53.0) | Fifth 3: (53.0, 56.0) | Fifth 4: (56.0, 59.9) | Fifth 5: >59.9 |
Overall | Fifth 1: <45.2 |
Fifth 2: (45.2, 49.1) | Fifth 3: (49.1, 53.1) | Fifth 4: (53.1, 58.3) | Fifth 5: >58.3 |
Overall | |
| (n = 41,808) | (n = 41,914) | (n = 41,915) | (n = 41,699) | (n = 41,399) | (n = 208,735) | (n = 51,980) | (n = 52,237) | (n = 51,740) | (n = 51,910) | (n = 51,731) | (n = 259,598) | |
| Age, y | 54.2 (8.42) | 55.6 (8.27) | 56.6 (8.13) | 57.5 (7.90) | 58.0 (7.71) | 56.4 (8.20) | 53.7 (8.03) | 55.6 (8.00) | 56.7 (7.87) | 57.5 (7.73) | 57.5 (7.63) | 56.2 (7.98) |
| Most deprived quartile | 10,141 (24.3%) | 9182 (21.9%) | 9433 (22.5%) | 10,189 (24.4%) | 12,632 (30.5%) | 51,577 (24.7%) | 10,599 (20.4%) | 10,586 (20.3%) | 11,178 (21.6%) | 12,947 (24.9%) | 16,881 (32.6%) | 62,191 (24.0%) |
| White ethnicity | 39,466 (94.4%) | 39,515 (94.3%) | 39,397 (94.0%) | 39,153 (93.9%) | 38,947 (94.1%) | 196,478 (94.1%) | 50,099 (96.4%) | 50,022 (95.8%) | 48,938 (94.6%) | 48,434 (93.3%) | 47,411 (91.6%) | 244,904 (94.3%) |
| Higher education | 19,165 (45.8%) | 16,338 (39.0%) | 14,099 (33.6%) | 12,226 (29.3%) | 9979 (24.1%) | 71,807 (34.4%) | 21,911 (42.2%) | 18,381 (35.2%) | 15,779 (30.5%) | 13,718 (26.4%) | 11,605 (22.4%) | 81,394 (31.4%) |
| Current smoker | 5674 (13.6%) | 5037 (12.0%) | 5060 (12.1%) | 4956 (11.9%) | 4892 (11.8%) | 25,619 (12.3%) | 4392 (8.4%) | 4616 (8.8%) | 4555 (8.8%) | 4599 (8.9%) | 4629 (8.9%) | 22,791 (8.8%) |
| Current drinker | 39,383 (94.2%) | 39,721 (94.8%) | 39,563 (94.4%) | 39,036 (93.6%) | 38,108 (92.1%) | 195,811 (93.8%) | 48,102 (92.5%) | 48,495 (92.8%) | 47,560 (91.9%) | 46,816 (90.2%) | 44,447 (85.9%) | 235,420 (90.7%) |
| Low physical activity | 4624 (11.1%) | 5472 (13.1%) | 6272 (15.0%) | 7265 (17.4%) | 9197 (22.2%) | 32,830 (15.7%) | 5465 (10.5%) | 6298 (12.1%) | 7014 (13.6%) | 7951 (15.3%) | 10,146 (19.6%) | 36,874 (14.2%) |
| Height, cm | 178 (6.68) | 177 (6.72) | 176 (6.58) | 175 (6.66) | 174 (6.75) | 176 (6.81) | 165 (6.11) | 163 (6.01) | 163 (6.10) | 162 (6.16) | 161 (6.24) | 163 (6.29) |
| Weight, kg | 74.2 (8.71) | 80.4 (9.10) | 84.3 (9.56) | 89.3 (10.4) | 101 (15.1) | 85.8 (14.1) | 59.6 (7.04) | 64.6 (7.45) | 69.3 (8.37) | 74.9 (9.69) | 88.4 (14.7) | 71.3 (14.0) |
| Waist circumference, cm | 83.4 (4.90) | 91.0 (3.74) | 95.8 (3.84) | 101 (4.23) | 112 (8.67) | 96.7 (11.1) | 70.0 (3.96) | 77.0 (3.23) | 83.0 (3.52) | 89.7 (4.14) | 103 (8.84) | 84.5 (12.4) |
| BMI, kg/m2 | 23.4 (1.98) | 25.7 (1.79) | 27.3 (1.86) | 29.1 (2.07) | 33.4 (3.90) | 27.8 (4.15) | 21.9 (1.93) | 24.2 (1.94) | 26.2 (2.23) | 28.6 (2.67) | 34.2 (4.85) | 27.0 (5.13) |
| Fat mass, kg | 14.0 (3.97) | 18.3 (3.91) | 21.3 (4.14) | 24.7 (4.64) | 32.5 (8.17) | 22.1 (8.13) | 17.4 (4.62) | 21.7 (4.77) | 25.5 (5.37) | 29.9 (6.22) | 39.7 (9.93) | 26.8 (10.0) |
| Fat-free mass, kg | 60.2 (6.63) | 62.1 (6.80) | 63.1 (6.91) | 64.7 (7.25) | 68.6 (8.41) | 63.7 (7.76) | 42.2 (3.75) | 42.9 (3.85) | 43.8 (4.08) | 45.0 (4.49) | 48.7 (5.83) | 44.5 (5.02) |
| Body fat percentage, % | 18.7 (4.14) | 22.7 (3.46) | 25.1 (3.28) | 27.5 (3.22) | 31.8 (3.94) | 25.2 (5.71) | 28.8 (5.05) | 33.4 (4.30) | 36.5 (4.09) | 39.6 (3.94) | 44.4 (4.33) | 36.5 (6.88) |
| Waist-to-height ratio, % | 47.0 (2.33) | 51.5 (0.93) | 54.5 (0.85) | 57.8 (1.12) | 64.6 (4.48) | 55.1 (6.41) | 42.5 (2.05) | 47.2 (1.11) | 51.0 (1.14) | 55.5 (1.52) | 64.2 (5.14) | 52.1 (7.87) |
| Hypertension | 6520 (15.4%) | 9168 (21.6%) | 11,099 (27.2%) | 13,982 (33.8%) | 18,897 (45.4%) | 59,666 (28.6%) | 5308 (10.2%) | 7877 (15.1%) | 10,785 (20.8%) | 14,626 (28.2%) | 21,446 (41.5%) | 60,042 (23.1%) |
| Diabetes | 1065 (2.5%) | 1424 (3.4%) | 1900 (4.7%) | 2805 (6.8%) | 5362 (12.9%) | 12,556 (6.0%) | 466 (0.9%) | 574 (1.1%) | 913 (1.8%) | 1816 (3.5%) | 5148 (10.0%) | 8917 (3.4%) |
| High cholesterol | 2699 (6.4%) | 4527 (10.7%) | 5482 (13.4%) | 6855 (16.6%) | 8622 (20.7%) | 28,185 (13.5%) | 1812 (3.5%) | 2915 (5.6%) | 4321 (8.4%) | 5967 (11.5%) | 8083 (15.6%) | 23,098 (8.9%) |
Values are mean (SD) or n (%). Fifths are defined with sex-specific cutoff values. Most deprived quartile was defined as in the lowest quartile based on Townsend Deprivation Index, an area-level general index for socioeconomic status. Low physical activity was defined as having low physical activity level based on the International Physical Activity Questionnaire.
Reassessment was performed in a subset of UK Biobank participants in 2013–2014, which demonstrated high correlation with baseline assessment for BF% and WHtR, with Pearson correlation coefficients of 0.82 for BF% (n = 45,814) and 0.79 for WHtR (n = 47,398), which was similar between males and females. The high correlation coefficients indicated high stability of the measures over time.
During a median follow-up of 12.0 (IQR, 11.3–12.7) y, 20,151 incident ischemic CVD events [7118 (35.3%) females], including 13,604 MIs [4269 (31.4%) females] and 6681 ISs [2749 (41.2%) females], were recorded in hospital in-patient and mortality records.
WHtR and ischemic CVDs
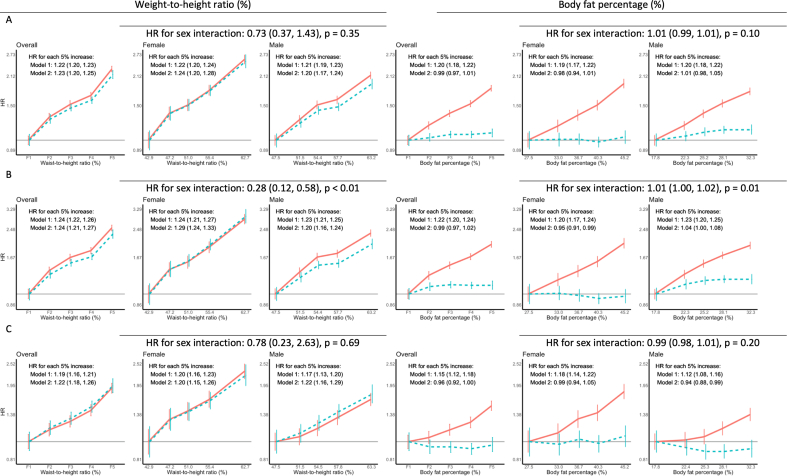
We found consistently positive linear associations of WHtR with incident ischemic CVD, MI, and IS, and the associations remained similar before and after additional adjustment of BF%. In the model adjusted for BF%, each 5% increase in WHtR was associated with higher risks of ischemic CVD (HR: 1.23; 95% CI: 1.20, 1.25), MI (HR: 1.24; 95% CI: 1.21, 1.27), and IS (HR: 1.22; 95% CI: 1.18, 1.26). The associations were similar between females and males for ischemic CVD and IS. However, for MI, the association was stronger in females than in males (HR: 1.29; 95% CI: 1.24, 1.33 compared with HR: 1.20; 95% CI: 1.16, 1.24; P for sex interaction < 0.01) (Figure 1, Supplemental Table 4).
FIGURE 1.
Associations between waist-to-height ratio, body fat percentage, and ischemic cardiovascular diseases (using sex-specific quintiles). Red solid lines show the associations estimated in model 1, i.e., without mutual adjustment of body fat percentage and waist-to-height ratio. Blue dotted lines show the associations estimated in model 2, i.e., with mutual adjustment of body fat percentage and waist-to-height ratio. Models were stratified by region, age group, education, and ethnicity and adjusted for sex, deprivation, smoking status, drinking status, and physical activity. Fifths were defined using sex-specific quintiles. Analysis of the overall cohort was additionally adjusted for sex. HR, hazard ratio.
BF% and ischemic CVDs
Adjustment of WHtR substantially attenuated the association of BF% with all outcomes. The HR for each 5% increase in BF% was reduced from 1.20 (95% CI: 1.18, 1.22) to 0.99 (95% CI: 0.97, 1.01) for ischemic CVD, from 1.22 (95% CI: 1.20, 1.24) to 0.99 (95% CI: 0.97, 1.02) for MI, and from 1.15 (95% CI: 1.12, 1.18) to 0.96 (95% CI: 0.92, 1.00) for IS.
Similar to the overall cohort, the associations of BF% with ischemic CVD, MI, and IS were substantially attenuated among females. For MI, although the HR estimate for 5% increase in BF% (0.95; 95% CI: 0.91, 0.99) implied a possible weak association, HR estimates for the fifths (highest compared with lowest fifth HR: 0.97; 95% CI: 0.81, 1.16) and P value for trend (0.58) suggested null evidence for such an association.
For males, there remained a weak association of BF% with MI after adjustment of WHtR, with HR for each 5% increase in BF% of 1.04 (95% CI: 1.00, 1.08). The HR for 5% increase in BF% was marginally associated with lower risk of IS (HR: 0.94; 95% CI: 0.88, 0.99), suggesting a protective effect; however, the HRs for categorical BF% all including null value and the P value for trend (0.35) both suggested null evidence for such an association. There was no evidence for an association of BF% with overall ischemic CVD after adjusting for WHtR (Figure 1, Supplemental Table 4).
Subgroup analyses
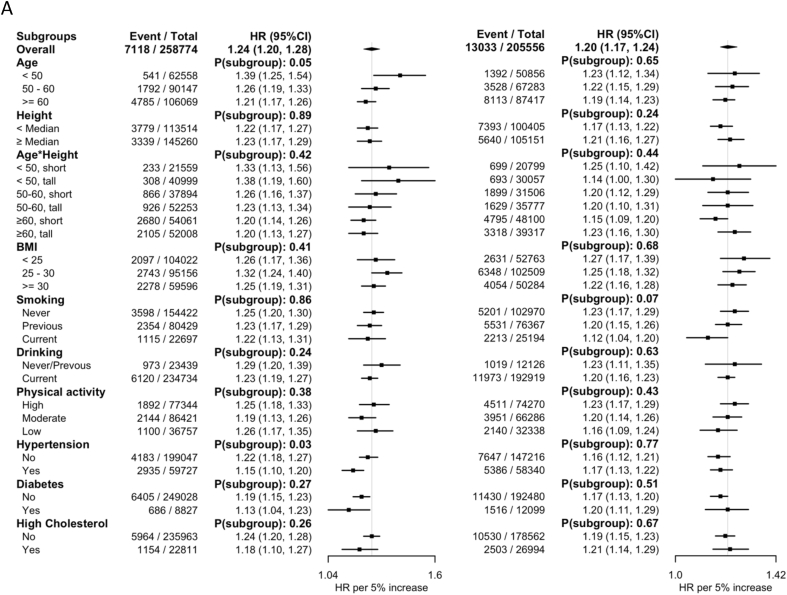
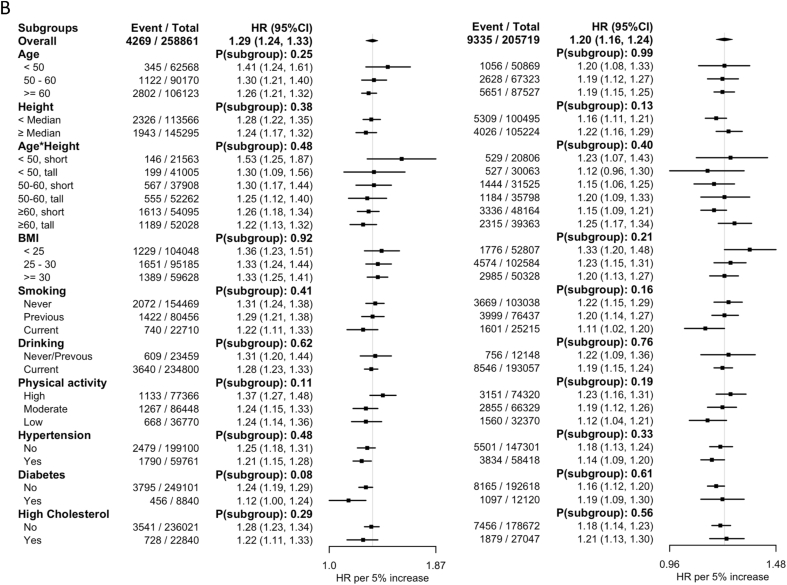
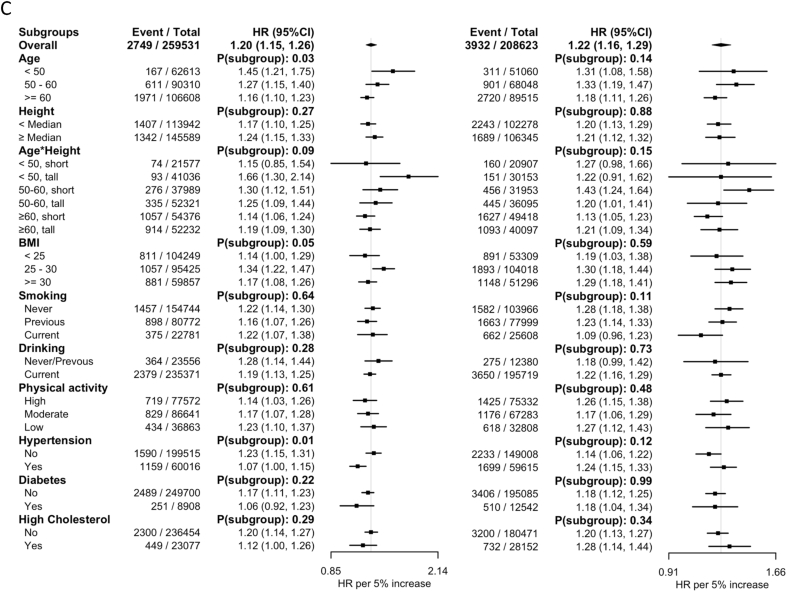
The positive linear associations between WHtR and all health outcomes remained similar in all subgroups of age, height, age × height interaction, smoking, drinking, physical activity level, hypertension, diabetes, and high cholesterol in males (Figure 2). In females, the association between WHtR and IS was stronger in the younger age groups (HR: 1.45; 95% CI: 1.21, 1.17 for those <50 y, HR: 1.27; 95% CI: 1.15, 1.40 for those aged 50–60 y, and HR: 1.16; 95% CI: 1.10, 1.23 for those ≥60 y, P for subgroup difference = 0.03), but weaker in hypertensive individuals than nonhypertensive individuals (HR: 1.07; 95% CI: 1.00, 1.15 compared with HR: 1.23; 95% CI: 1.15, 1.31, P for subgroup difference = 0.01).
FIGURE 2.
Subgroup analysis for sex-specific associations between waist-to-height ratio and ischemic cardiovascular diseases. The associations were estimated in models with mutual adjustment of body fat percentage and waist-to-height ratio. Models are stratified by region, age group, education, and ethnicity and adjusted for deprivation, smoking status, drinking status, physical activity, and body fat percentage. Vertical gray lines indicate the overall association. CI, confidence interval; HR, hazard ratio.
Subgroup analysis for BF% showed null associations with ischemic CVD, MI, and IS in all subgroups for females. For MI, we observed positive associations in males with BMI <25 kg/m2 (HR: 1.11; 95% CI: 1.02, 1.21) and in those without baseline diabetes (HR: 1.06; 95% CI: 1.02, 1.10), as well as a negative association in those with BMI ≥30 kg/m2 (HR: 0.91; 95% CI: 0.84, 0.98) (Supplemental Figure 2).
Sensitivity analysis
Additional adjustment of BMI generated broadly consistent results to the primary analysis (Supplemental Table 5), suggesting the associations of WHtR were independent of BMI and BF%. Similar results were also observed in the sensitivity analyses that excluded diagnoses occurring during the first 2 y of follow-up (Supplemental Table 6) and using sex-combined quintiles (Supplemental Table 7), indicating the results were not influenced by reverse causation or the choice of quintiles. Additional adjustment of hypertension, diabetes, and high cholesterol generated qualitatively consistent results that WHtR showed independent and positive associations with the outcomes and the associations of BF were substantially diluted after adjusting for WHtR, but the associations of WHtR were quantitatively weaker (Supplemental Figure 3, Supplemental Table 8). For example, the HR for WHtR–IS association in females dropped from 1.20 (95% CI: 1.15, 1.26) to 1.12 (95% CI: 1.07, 1.18) after additional adjustment for these risk factors. In a subset of 34,394 participants [17,669 (51.4%) females] with both BIA- and DXA-derived BF%, BIA and DXA consistently showed null evidence for associations between BF% and ischemic CVD, MI, and IS (Supplemental Table 9), which was similar to the findings of the primary analysis.
Additional analyses of WHtR
Using 0.5 as the cutoff value for WHtR, we found 54.3% of females and 78.1% of males were categorized as high WHtR (≥0.5). Compared with those with low WHtR (<0.5), females with high WHtR showed 49%, 54%, and 48% higher risks of ischemic CVD (HR: 1.49; 95% CI: 1.38, 1.62), MI (HR: 1.54; 95% CI: 1.39, 1.71), and IS (HR: 1.48; 95% CI: 1.30, 1.68), respectively; the corresponding HR values for males were 1.41 (95% CI: 1.31, 1.51) for ischemic CVD, 1.52 (95% CI: 1.40, 1.66) for MI, and 1.19 (95% CI: 1.06, 1.35) for IS. Compared with males, females had lower WHtR-related risk difference in ischemic CVD (1.41 compared with 2.64 per 1000 person-years) and MI (0.89 compared with 2.05), and comparable risk difference in IS (0.53 compared with 0.58). Among female participants, 23% of ischemic CVD, 25% of MI, and 22% of IS were attributable to high WHtR, whereas in male participants, the attributable fractions were 25%, 30%, and 13%, respectively (Table 2).
TABLE 2.
Associations between categorical waist-to-height ratio (≥0.5 vs. <0.5) and ischemic cardiovascular diseases
| Outcome | Sex | Prevalence of high WHtR (%) | HR (95% CI) | Risk difference (/1000 person) | Risk difference (/1000 person-year) | Population attributable fraction |
|---|---|---|---|---|---|---|
| Ischemic CVD | Females | 54.3 | 1.49 (1.38, 1.62) | 16.65 | 1.41 | 0.23 |
| Males | 78.1 | 1.41 (1.31, 1.51) | 29.97 | 2.64 | 0.25 | |
| Myocardial infarction | Females | 54.3 | 1.54 (1.39, 1.71) | 10.48 | 0.89 | 0.25 |
| Males | 78.1 | 1.52 (1.40, 1.66) | 23.77 | 2.05 | 0.30 | |
| Ischemic stroke | Females | 54.3 | 1.48 (1.30, 1.68) | 6.37 | 0.53 | 0.22 |
| Males | 78.1 | 1.19 (1.06, 1.35) | 6.85 | 0.58 | 0.13 |
Waist-to-height ratio was categorized into binary using a cutoff value of 0.5. Model was stratified by region, age group, education, and ethnicity and adjusted for deprivation, smoking status, drinking status, physical activity, and body fat percentage.
Abbreviations: CI, confidence interval; CVD, cardiovascular disease; HR, hazard ratio; WHtR, waist-to-height ratio.
Using 90 cm and 100 cm for categorizing WC, 30.4% of females and 35.9% of males had high WC. Comparing WHtR and WC in screening obesity, 99.9% of low WHtR individuals also had low WC (99.9% for females and 99.9% for males), whereas only half of individuals with high WHtR had high WC (55.8% for females and 45.9% for males). Therefore, using only WC in screening obesity may miss half of individuals with high risk, compared to WHtR. Compared with those with low WHtR and low WC, people with high WHtR showed significantly higher risks of all outcomes, regardless of WC levels, whereas people with both high WHtR and WC showed higher risks than those with high WHtR but low WC (Table 3).
TABLE 3.
Hazard ratios for ischemic cardiovascular diseases based on waist-to-height ratio and waist circumference
| Females |
Males |
|||
|---|---|---|---|---|
| Cases/total | HR (95% CI) | Cases/total | HR (95% CI) | |
| Ischemic cardiovascular diseases | ||||
| Low WHtR, low WC | 2181/118,119 | Reference | 1804/45,086 | Reference |
| Low WHtR, high WC | 0/44 | — | 0/5 | — |
| High WHtR, low WC | 1826/62,136 | 1.31 (1.20, 1.43) | 5407/86,958 | 1.36 (1.26, 1.46) |
| High WHtR, high WC | 3111/78,475 | 1.85 (1.68, 2.03) | 5822/73,507 | 1.63 (1.51, 1.77) |
| Myocardial infarction | ||||
| Low WHtR, low WC | 1276/118,141 | Reference | 1210/45,108 | Reference |
| Low WHtR, high WC | 0/44 | — | 0/5 | — |
| High WHtR, low WC | 1070/62,166 | 1.32 (1.18, 1.47) | 3904/87,033 | 1.47 (1.35, 1.60) |
| High WHtR, high WC | 1923/78,510 | 2.00 (1.77, 2.25) | 4221/73,573 | 1.74 (1.58, 1.92) |
| Ischemic stroke | ||||
| Low WHtR, low WC | 844/118,349 | Reference | 613/45,451 | Reference |
| Low WHtR, high WC | 0/44 | — | 0/5 | — |
| High WHtR, low WC | 708/62,323 | 1.30 (1.13, 1.50) | 1590/88,259 | 1.13 (1.01, 1.28) |
| High WHtR, high WC | 1197/78,815 | 1.82 (1.57, 2.11) | 1729/74,908 | 1.45 (1.26, 1.68) |
Cutoff values for categorizing WHtR: 0.5 for males and 0.5 for females. Cutoff values for categorizing WC: 90 cm for females and 100 cm for males. —: coefficient cannot be estimated because the case number was zero. Model was stratified by region, age group, education, and ethnicity and adjusted for deprivation, smoking status, drinking status, physical activity, and body fat percentage.
Abbreviations: CI, confidence interval; HR, hazard ratio; WC, waist circumference; WHtR, waist-to-height ratio.
Discussion
In this prospective cohort study of 468,333 participants with a 12-y follow-up, WHtR was an independent risk factor for ischemic CVD and its major subtypes, whereas the association of BF% was substantially attenuated after WHtR was taken into consideration.
Our findings are consistent with previous studies that have reported central obesity to be a risk factor for ischemic CVD, independent of general obesity [7,11,12,32]. Although most previous studies compared BMI with WC or WHR, our study assessed BF% and WHtR, which are more precise measures of general obesity and central obesity, respectively. Before mutual adjustment, BF% and WHtR showed similarly linear positive associations with ischemic CVD in both sexes, with a 5% increase in the exposures associated with ∼20% higher risks of MI and IS. However, adjusting for WHtR almost completely attenuated the associations with BF%, which is consistent with the findings from the EPIC-Norfolk study [23]. This suggests that the observed association of BF% with ischemic CVD is predominantly explained by its correlation with WHtR. By contrast, the associations of WHtR remained qualitatively and quantitatively unchanged, regardless of BF% adjustment. Overall, each 5% increase in WHtR was associated with ∼20% higher risk of ischemic CVD, MI, and IS.
Compared with BMI, BF% is a more accurate measure of body fatness, but it does not distinguish regional fat distribution, i.e., subcutaneous fat and visceral fat, which differ in their metabolic characteristics and cardiometabolic effects [17,33]. WHtR specifically measures abdominal visceral fat. It has been suggested that excess visceral fat deposition reflects ectopic fat deposition, which is a marker for the relative inability of subcutaneous adipose tissue to expand and act as a protective “metabolic sink” [34], thereby leading to a stronger association of CVD with visceral adiposity than with subcutaneous (or “general”) adiposity [33,[35], [36], [37]]. Furthermore, some studies have demonstrated an inverse association between risk of MI with body gluteofemoral fat mass, which is mainly subcutaneous fat, measured with thigh circumference and gynoid to total adipose mass [38,39]. It has also been proposed that excess visceral fat could increase cardiovascular disease risk via altered free fatty acid metabolism, adipokine dysregulation, and/or insulin resistance [40] or by neuroendocrine abnormalities (e.g., overactivated hypothalamic–pituitary–adrenal axis, disturbed gonadal steroids, etc.) that affect energy partition and fat distribution [35]. Although the mechanism is not fully understood and warrants more research, it is recommended that weight management intervention (diet, exercise, etc.) should be targeted at primarily reducing visceral fat [17].
For BF% and MI in females, although the HR for 5% increase showed a marginally significant negative association, the HR for categorical BF% and the P values for trend test both suggested null evidence for such an association. Nevertheless, in males, we observed plausible evidence for a weak association of BF% with MI, with a linear association estimate decreasing from 23% to 4% after considering WHtR, for each 5% increase in BF%. This was similar to a previous finding [41] that the association between general obesity (BMI) and MI was slightly higher in males than in females, although the difference between sexes was not significant. Another difference to note is that the authors did not adjust for central obesity. Having said that, it is possible this observed weak association in males was due to residual confounding, given the large attenuation after adjustment for WHtR. Mendelian randomization also shows inconsistent evidence regarding a likely causal role for BF% in coronary artery disease, with some finding a positive association [42] and others no association [43]. Both of these studies selected genetic instrumental variables obtained from sex-combined genome-wide association studies (GWASs), whereas a sex-specific GWAS of BF% indicated its genetic architecture may vary between males and females [44]. Therefore, future Mendelian randomization analysis using sex-specific genetic variants for BF% is warranted to further assess the likely causal role for BF% in CVD in males.
We observed a potential effect modification of sex on the association of WHtR with MI, with a stronger association in females than in males. A previous systematic review similarly found stronger associations of WHtR with CVD in females than in males but did not examine MI specifically [45]. For risk of MI, a previous analysis using UK Biobank data found an interaction between sex and WHR (with stronger association in females than in males) but null evidence for interaction between sex and WHtR, which might be due to short follow-up (7 y), small number of outcome events, and different covariate adjustment [41]. This sex difference is consistent with a study that demonstrated a stronger relationship between central obesity measures and CVD in females than in males, where the majority of CVDs were MI cases [46]. Our finding is supported by prior evidence that central obesity measures and abdominal visceral fat had stronger links with cardiometabolic risk factors in females than in males [47,48]. Future studies are required to confirm this potential sex interaction.
We found evidence for an effect modification of age in the association between WHtR and IS in females, with a stronger association in the younger age groups. Although this finding is of interest, it should be interpreted with caution as this was not the primary objective of this study. Differences in the association of WHtR with coronary heart disease by age and sex have been reported previously, albeit with inconsistent results [14,16]. A recent meta-analysis showed a stronger association of WHtR with CVD in people <60 y than in older people in both sexes [45], which is consistent with our findings and may highlight the importance of maintaining a lower WHtR from early adulthood.
We found evidence for an effect modification of hypertension in the association between WHtR and IS in females, with a strong association in individuals without baseline hypertension and a null association in hypertensive individuals. Similarly, null associations were also observed in individuals with baseline diabetes or high cholesterol, although the difference between these subgroups did not reach statistical significance. First, we should be cautious about the smaller sample size in these subgroups. Second, this suggests the mediatory role of these risk factors in the WHtR–IS association, consistent with previous evidence that has also demonstrated cardiometabolic risk factors as important mediators for the obesity–CVD association [49,50]. This is further supported by a sensitivity analysis that demonstrated a weaker association after additional adjustment for hypertension, diabetes, and high cholesterol. Third, it is also possible that individuals with a diagnosis of hypertension, diabetes, or high cholesterol had adopted lifestyle modification and/or therapeutic treatment, thus reducing their CVD risk. However, we did not observe such differences in males. Although this finding is of interest, it should be interpreted with caution as this was not the primary objective of this study. It warrants more investigations about their roles in the obesity–CVD association, as well as its sex difference.
WHtR has advantages compared to other central obesity measures (WC or WHR). It is easier to measure than WHR (measuring standing height is easier than measuring hip circumference), it is independent of body stature (height), and it has been shown to be an independent predictor of cardiometabolic risk factors. Further, previous evidence suggests that a cutoff value of 0.5 is appropriate to use across different population subgroups (i.e., different ethnic groups) [17,18]. Although we did not look for an optimal threshold below which WHtR was not associated with CVD risk, our analysis showed the commonly used cutoff value of 0.5 was significantly associated with higher CVD risk in both sexes. Finally, WHtR is easy to interpret, such that its health implications can be delivered in a simple public health message: “keep your waist to less than half your height” [19], which is supported by our analysis and is why WHtR has been recommended for obesity screening in the general population.
This study has several strengths, including the large sample size, the prospective design with a long follow-up period, and the ability to perform in-depth sensitivity analyses. However, we acknowledge some limitations. First, although we have adjusted for multiple confounders, residual confounding may still exist (particularly of the relationship of BF% with CVD in males). Second, the UK Biobank is a middle-aged, predominantly White and relatively affluent cohort, which may restrict the external validity of the findings to populations of different ethnicities, age distributions, and socioeconomic status. Third, our primary analysis used BF% derived by BIA, a less accurate body composition measurement than DXA. Although the sensitivity analysis in a subset of participants showed similar findings of null associations with the outcomes between the 2 methods, we still need to be cautious because of the much-reduced sample size and statistical power. Therefore, future investigations with larger sample sizes are warranted to compare BIA and DXA.
In summary, WHtR was linearly positively associated with risk of ischemic CVD in both males and females, whereas BF% showed little or no association after adjustment for WHtR.
Acknowledgments
This research has been conducted using the UK Biobank Resource under Application Number 41115. We sincerely thank all UK Biobank participants, researchers, and staff. This research uses data provided by patients and collected by the NHS as part of their care and support (Copyright © (2023), NHS Digital. Re-used with the permission of the NHS Digital (and/or UK Biobank). All rights reserved). This research used data assets made available by National Safe Haven as part of the Data and Connectivity National Core Study, led by Health Data Research UK in partnership with the Office for National Statistics and funded by UK Research and Innovation.
Author contributions
The authors’ responsibilities were as follows – QF, MW, BL: contributed to the study conception and design; QF: material preparation, data collection, and analysis; QF: wrote the first draft of the manuscript; QF, JB, MC, WO, MW, BL, NA: commented on previous versions of the manuscript; and all authors: read and approved the final manuscript.
Conflict of interest
MW does consultancy for Amgen and Freeline outside the submitted work; no support from any organization for the submitted work; no other relationships or activities that could appear to have influenced the submitted work. All other authors report no conflicts of interests.
Funding
QF, JB, MC, WO, BL, and NA were jointly funded by Wellcome, UKRI, BHF, CRUK, and NIHR (223600/Z/21/Z). MW is supported by an Australian National Health and Medical Research Council Investigator Grant (APP1174120) and Program Grant (APP1149987). The funders of the study had no role in study design, data collection, data analysis, data interpretation, writing of the report, or the decision to submit the manuscript for publication.
Data availability
The data that support the findings of this study are available from the UK Biobank, but restrictions apply to the availability of these data, which were used under license for the current study and so are not publicly available. The UK Biobank resources are, however, available and can be accessed through applications on their website (https:// www.ukbiobank.ac.uk/). Analytic codes are available upon reasonable request.
Conflict of interest
MW does consultancy for Amgen and Freeline outside the submitted work; no support from any organization for the submitted work; no other relationships or activities that could appear to have influenced the submitted work. All other authors report no conflicts of interests.
Acknowledgments
This research has been conducted using the UK Biobank Resource under Application Number 41115. We sincerely thank all UK Biobank participants, researchers, and staff. This research uses data provided by patients and collected by the NHS as part of their care and support (Copyright © (2023), NHS Digital. Re-used with the permission of the NHS Digital (and/or UK Biobank). All rights reserved). This research used data assets made available by National Safe Haven as part of the Data and Connectivity National Core Study, led by Health Data Research UK in partnership with the Office for National Statistics and funded by UK Research and Innovation.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.ajcnut.2024.03.018.
Contributor Information
Qi Feng, Email: qifeng@cuhk.edu.hk.
Ben Lacey, Email: ben.lacey@ndph.ox.ac.uk.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.Dai H., Alsalhe T.A., Chalghaf N., Riccò M., Bragazzi N.L., Wu J. The global burden of disease attributable to high body mass index in 195 countries and territories, 1990–2017: an analysis of the Global Burden of Disease Study. PLOS Med. 2020;17(7) doi: 10.1371/journal.pmed.1003198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Roth G.A., Mensah G.A., Johnson C.O., Addolorato G., Ammirati E., Baddour L.M., et al. Global burden of cardiovascular diseases and risk factors, 1990–2019: update from the GBD 2019 study. J. Am. Coll. Cardiol. 2020;76(25):2982–3021. doi: 10.1016/j.jacc.2020.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Aune D., Sen A., Prasad M., Norat T., Janszky I., Tonstad S., et al. BMI and all cause mortality: systematic review and non-linear dose-response meta-analysis of 230 cohort studies with 3.74 million deaths among 30.3 million participants. BMJ. 2016;353:i2156. doi: 10.1136/bmj.i2156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kim M.S., Kim W.J., Khera A.V., Kim J.Y., Yon D.K., Lee S.W., et al. Association between adiposity and cardiovascular outcomes: an umbrella review and meta-analysis of observational and Mendelian randomization studies. Eur. Heart J. 2021;42(34):3388–3403. doi: 10.1093/eurheartj/ehab454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Borga M., West J., Bell J.D., Harvey N.C., Romu T., Heymsfield S.B., et al. Advanced body composition assessment: from body mass index to body composition profiling. J. Investig. Med. 2018;66(5):1–9. doi: 10.1136/jim-2018-000722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nuttall F.Q. Body mass index: obesity, BMI, and health: a critical review. Nutr. Today. 2015;50(3):117–128. doi: 10.1097/NT.0000000000000092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Choi D., Choi S., Son J.S., Oh S.W., Park S.M. Impact of discrepancies in general and abdominal obesity on major adverse cardiac events. J. Am. Heart Assoc. 2019;8(18) doi: 10.1161/JAHA.119.013471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Piché M.E., Tchernof A., Després J.P. Obesity phenotypes, diabetes, and cardiovascular diseases. Circ. Res. 2020;126(11):1477–1500. doi: 10.1161/CIRCRESAHA.120.316101. [DOI] [PubMed] [Google Scholar]
- 9.Zeng Q., Dong S.Y., Sun X.N., Xie J., Cui Y. Percent body fat is a better predictor of cardiovascular risk factors than body mass index. Braz. J. Med. Biol. Res. 2012;45(7):591–600. doi: 10.1590/S0100-879X2012007500059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Si S., Tewara M.A., Ji X., Wang Y., Liu Y., Dai X., et al. Body surface area, height, and body fat percentage as more sensitive risk factors of cancer and cardiovascular disease. Cancer Med. 2020;9(12):4433–4446. doi: 10.1002/cam4.3076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhang X., Shu X.O., Gao Y.T., Yang G., Li H., Zheng W. General and abdominal adiposity and risk of stroke in Chinese women. Stroke. 2009;40(4):1098–1104. doi: 10.1161/STROKEAHA.108.539692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee C.M.Y., Huxley R.R., Wildman R.P., Woodward M. Indices of abdominal obesity are better discriminators of cardiovascular risk factors than BMI: a meta-analysis. J. Clin. Epidemiol. 2008;61(7):646–653. doi: 10.1016/j.jclinepi.2007.08.012. [DOI] [PubMed] [Google Scholar]
- 13.Ashwell M., Gibson S. Waist to height ratio is a simple and effective obesity screening tool for cardiovascular risk factors: analysis of data from the British National Diet and Nutrition Survey of adults aged 19-64 years. Obes. Facts. 2009;2(2):97–103. doi: 10.1159/000203363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rådholm K., Chalmers J., Ohkuma T., Peters S., Poulter N., Hamet P., et al. Use of the waist-to-height ratio to predict cardiovascular risk in patients with diabetes: results from the ADVANCE-ON study. Diabetes Obes. Metab. 2018;20(8):1903–1910. doi: 10.1111/dom.13311. [DOI] [PubMed] [Google Scholar]
- 15.Zhang S., Fu X., Du Z., Guo X., Li Z., Sun G., et al. Is waist-to-height ratio the best predictive indicator of cardiovascular disease incidence in hypertensive adults? A cohort study. BMC Cardiovasc. Disord. 2022;22(1):214. doi: 10.1186/s12872-022-02646-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gelber R.P., Gaziano J.M., Orav E.J., Manson J.E., Buring J.E., Kurth T. Measures of obesity and cardiovascular risk among men and women. J. Am. Coll. Cardiol. 2008;52(8):605–615. doi: 10.1016/j.jacc.2008.03.066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bosomworth N.J. Normal-weight central obesity: unique hazard of the toxic waist. Can. Fam. Physician. 2019;65(6):399–408. [PMC free article] [PubMed] [Google Scholar]
- 18.Browning L.M., Hsieh S.D., Ashwell M. A systematic review of waist-to-height ratio as a screening tool for the prediction of cardiovascular disease and diabetes: 0·5 could be a suitable global boundary value. Nutr. Res. Rev. 2010;23(2):247–269. doi: 10.1017/S0954422410000144. [DOI] [PubMed] [Google Scholar]
- 19.National Institute for Health and Care Excellence (NICE) Obesity: identification, assessment and management. https://www.nice.org.uk/guidance/cg189 [Internet] [cited 12 February 2024]. Clinical Guideline No 189. Available from: [PubMed]
- 20.Carmienke S., Freitag M.H., Pischon T., Schlattmann P., Fankhaenel T., Goebel H., et al. General and abdominal obesity parameters and their combination in relation to mortality: a systematic review and meta-regression analysis. Eur. J. Clin. Nutr. 2013;67(6):573–585. doi: 10.1038/ejcn.2013.61. [DOI] [PubMed] [Google Scholar]
- 21.Jayedi A., Khan T.A., Aune D., Emadi A., Shab-Bidar S. Body fat and risk of all-cause mortality: a systematic review and dose-response meta-analysis of prospective cohort studies. Int. J. Obes. (Lond.) 2022;46(9):1573–1581. doi: 10.1038/s41366-022-01165-5. [DOI] [PubMed] [Google Scholar]
- 22.Jayedi A., Soltani S., Zargar M.S., Khan T.A., Shab-Bidar S. Central fatness and risk of all cause mortality: systematic review and dose-response meta-analysis of 72 prospective cohort studies. BMJ. 2020;370 doi: 10.1136/bmj.m3324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Myint P.K., Kwok C.S., Luben R.N., Wareham N.J., Khaw K.T. Body fat percentage, body mass index and waist-to-hip ratio as predictors of mortality and cardiovascular disease. Heart. 2014;100(20):1613–1619. doi: 10.1136/heartjnl-2014-305816. [DOI] [PubMed] [Google Scholar]
- 24.Oikonomou E.K., Antoniades C. The role of adipose tissue in cardiovascular health and disease. Nat. Rev. Cardiol. 2019;16(2):83–99. doi: 10.1038/s41569-018-0097-6. [DOI] [PubMed] [Google Scholar]
- 25.Feng Q., Kim J.H., Omiyale W., Bešević J., Conroy M., May M., et al. Raw and cooked vegetable consumption and risk of cardiovascular disease: a study of 400,000 adults in UK Biobank. Front. Nutr. 2022;9 doi: 10.3389/fnut.2022.831470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Clarke R., Shipley M., Lewington S., Youngman L., Collins R., Marmot M., et al. Underestimation of risk associations due to regression dilution in long-term follow-up of prospective studies. Am. J. Epidemiol. 1999;150(4):341–353. doi: 10.1093/oxfordjournals.aje.a010013. [DOI] [PubMed] [Google Scholar]
- 27.MacMahon S., Peto R., Cutler J., Collins R., Sorlie P., Neaton J., et al. Blood pressure, stroke, and coronary heart disease. Part 1, prolonged differences in blood pressure: prospective observational studies corrected for the regression dilution bias. Lancet. 1990;335(8692):765–774. doi: 10.1016/0140-6736(90)90878-9. [DOI] [PubMed] [Google Scholar]
- 28.Plummer M. Improved estimates of floating absolute risk. Stat. Med. 2004;23(1):93–104. doi: 10.1002/sim.1485. [DOI] [PubMed] [Google Scholar]
- 29.Achamrah N., Colange G., Delay J., Rimbert A., Folope V., Petit A., et al. Comparison of body composition assessment by DXA and BIA according to the body mass index: a retrospective study on 3655 measures. PLOS ONE. 2018;13(7) doi: 10.1371/journal.pone.0200465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chan K.H., Wright N., Xiao D., Guo Y., Chen Y., Du H., et al. Tobacco smoking and risks of more than 470 diseases in China: a prospective cohort study. Lancet Public Health. 2022;7(12):e1014–e1026. doi: 10.1016/S2468-2667(22)00227-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ross R., Neeland I.J., Yamashita S., Shai I., Seidell J., Magni P., et al. Waist circumference as a vital sign in clinical practice: a Consensus Statement from the IAS and ICCR Working Group on Visceral Obesity. Nat. Rev. Endocrinol. 2020;16(3):177–189. doi: 10.1038/s41574-019-0310-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.O’Donnell M.J., Xavier D., Liu L., Zhang H., Chin S.L., Rao-Melacini P., et al. Risk factors for ischaemic and intracerebral haemorrhagic stroke in 22 countries (the INTERSTROKE study): a case-control study. Lancet. 2010;376(9735):112–123. doi: 10.1016/S0140-6736(10)60834-3. [DOI] [PubMed] [Google Scholar]
- 33.Piché M.E., Poirier P., Lemieux I., Després J.P. Overview of epidemiology and contribution of obesity and body fat distribution to cardiovascular disease: an update. Prog. Cardiovasc. Dis. 2018;61(2):103–113. doi: 10.1016/j.pcad.2018.06.004. [DOI] [PubMed] [Google Scholar]
- 34.Després J.P., Lemieux I., Bergeron J., Pibarot P., Mathieu P., Larose E., et al. Abdominal obesity and the metabolic syndrome: contribution to global cardiometabolic risk. Arterioscler. Thomb. Vasc. Biol. 2008;28(6):1039–1049. doi: 10.1161/ATVBAHA.107.159228. [DOI] [PubMed] [Google Scholar]
- 35.Després J.P. Body fat distribution and risk of cardiovascular disease: an update. Circulation. 2012;126(10):1301–1313. doi: 10.1161/CIRCULATIONAHA.111.067264. [DOI] [PubMed] [Google Scholar]
- 36.Powell-Wiley T.M., Poirier P., Burke L.E., Després J.P., Gordon-Larsen P., Lavie C.J., et al. Obesity and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2021;143(21):e984–e1010. doi: 10.1161/CIR.0000000000000973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.McCarty M.F. A paradox resolved: the postprandial model of insulin resistance explains why gynoid adiposity appears to be protective. Med. Hypotheses. 2003;61(2):173–176. doi: 10.1016/s0306-9877(02)00238-4. [DOI] [PubMed] [Google Scholar]
- 38.Heitmann B.L., Frederiksen P. Thigh circumference and risk of heart disease and premature death: prospective cohort study. BMJ. 2009;339 doi: 10.1136/bmj.b3292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wiklund P., Toss F., Jansson J.H., Eliasson M., Hallmans G., Nordström A., et al. Abdominal and gynoid adipose distribution and incident myocardial infarction in women and men. Int. J. Obes. (Lond). 2010;34(12):1752–1758. doi: 10.1038/ijo.2010.102. [DOI] [PubMed] [Google Scholar]
- 40.Ortega-Loubon C., Fernández-Molina M., Singh G., Correa R. Obesity and its cardiovascular effects. Diabetes Metab. Res. Rev. 2019;35(4) doi: 10.1002/dmrr.3135. [DOI] [PubMed] [Google Scholar]
- 41.Peters S.A.E., Bots S.H., Woodward M. Sex differences in the association between measures of general and central adiposity and the risk of myocardial infarction: results from the UK Biobank. J. Am. Heart Assoc. 2018;7(5) doi: 10.1161/JAHA.117.008507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Martin S., Tyrrell J., Thomas E.L., Bown M.J., Wood A.R., Beaumont R.N., et al. Disease consequences of higher adiposity uncoupled from its adverse metabolic effects using Mendelian randomisation. eLife. 2022;11 doi: 10.7554/eLife.72452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhuang Z., Yao M., Wong J.Y.Y., Liu Z., Huang T. Shared genetic etiology and causality between body fat percentage and cardiovascular diseases: a large-scale genome-wide cross-trait analysis. BMC Med. 2021;19(1):100. doi: 10.1186/s12916-021-01972-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hübel C., Gaspar H.A., Coleman J.R.I., Finucane H., Purves K.L., Hanscombe K.B., et al. Genomics of body fat percentage may contribute to sex bias in anorexia nervosa. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2019;180(6):428–438. doi: 10.1002/ajmg.b.32709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Xue R., Li Q., Geng Y., Wang H., Wang F., Zhang S. Abdominal obesity and risk of CVD: a dose-response meta-analysis of thirty-one prospective studies. Br. J. Nutr. 2021;126(9):1420–1430. doi: 10.1017/S0007114521000064. [DOI] [PubMed] [Google Scholar]
- 46.Carlsson A.C., Riserus U., Ärnlöv J., Borné Y., Leander K., Gigante B., et al. Prediction of cardiovascular disease by abdominal obesity measures is dependent on body weight and sex – results from two community based cohort studies. Nutr. Metab. Cardiovasc. Dis. 2014;24(8):891–899. doi: 10.1016/j.numecd.2014.02.001. [DOI] [PubMed] [Google Scholar]
- 47.Sorimachi H., Omote K., Omar M., Popovic D., Verbrugge F.H., Reddy Y.N.V., et al. Sex and central obesity in heart failure with preserved ejection fraction. Eur. J. Heart Fail. 2022;24(8):1359–1370. doi: 10.1002/ejhf.2563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Tanaka S., Togashi K., Rankinen T., Pérusse L., Leon A.S., Rao D.C., et al. Sex differences in the relationships of abdominal fat to cardiovascular disease risk among normal-weight white subjects. Int. J. Obes. Relat. Metab. Disord. 2004;28(2):320–323. doi: 10.1038/sj.ijo.0802545. [DOI] [PubMed] [Google Scholar]
- 49.Koliaki C., Liatis S., Kokkinos A. Obesity and cardiovascular disease: revisiting an old relationship. Metabolism. 2019;92:98–107. doi: 10.1016/j.metabol.2018.10.011. [DOI] [PubMed] [Google Scholar]
- 50.Mathieu P., Poirier P., Pibarot P., Lemieux I., Després J.P. Visceral obesity: the link among inflammation, hypertension, and cardiovascular disease. Hypertension. 2009;53(4):577–584. doi: 10.1161/HYPERTENSIONAHA.108.110320. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data that support the findings of this study are available from the UK Biobank, but restrictions apply to the availability of these data, which were used under license for the current study and so are not publicly available. The UK Biobank resources are, however, available and can be accessed through applications on their website (https:// www.ukbiobank.ac.uk/). Analytic codes are available upon reasonable request.