Abstract
The current therapy for hepatitis B and C is based on systemic administration of recombinant human alpha interferon (r-hIFN-α). However, systemic delivery of r-hIFN-α is associated with severe side effects, but more importantly, it is effective in only a small percentage of patients. In an effort to maximize IFN-α antiviral efficacy, we have explored the therapeutic potential of murine IFN-α2 (mIFNα2) selectively expressed in the liver. To this end, we have developed a helper-dependent adenovirus vector (HD) containing the mIFN-α2 gene under the control of the liver-specific transthyretin promoter (HD-IFN). Comparison with a first-generation adenovirus carrying the same mIFN-α2 expression cassette indicates that at certain HD-IFN doses, induction of antiviral genes can be achieved in the absence of detectable circulating mIFN-α2. Challenge of injected mice with mouse hepatitis virus type 3 showed that HD-IFN provides high liver protection. Moreover, liver protection was also observed in acute nonviral liver inflammation hepatitis induced by concanavalin A at 1 month postinfection. These results hold promise for the development of a gene therapy treatment for chronic viral hepatitis based on liver-restricted expression of IFN-α2.
Interferon (IFN) was discovered by Isaac and Lindenmann in 1957 (18), and recently the U.S. Food and Drug Administration has approved recombinant human IFN-α (r-hIFN-α) for the treatment of hepatitis B virus (HBV) and hepatitis C virus (HCV) infections. IFN-α acts on target cells to confer a state of resistance to viral infectivity at one or more stages of virus entry or replication. These biological effects require binding to the type I IFN receptor complex, which is composed of two subunits, α and β (12). Both subunits undergo rapid ligand-dependent tyrosine phosphorylation, and the α subunit itself acts as a species-specific transducer for type I IFN action (7). At least 30 genes are known to be transcriptionally induced by type I IFNs, including 2′,5′-oligoadenylate synthetase (2′5′OAS), the double-stranded RNA-activated protein kinase, and the IFN-I response factor I (8, 10). 2′5′OAS is important for antiviral response, and its activity is required by cells to activate the endonuclease RNase L, which degrades RNA (31).
The currently available treatment for HCV with r-hIFN-α results in clearance of the virus in only 20% of patients. However, recent clinical trials have shown that a combination of r-hIFN-α and the antiviral drug ribavirin can increase the percentage of recovery up to 40% (9, 30). Although these results appear to be very promising, systemic injection of r-hIFN-α is associated with severe side effects, which worsen in combination with ribavirin, causing the withdrawal of 20% of patients from therapy.
It is not clear why r-hIFN-α treatment is effective in only a minority of patients. One possible explanation has been postulated on the basis of association of specific HCV genotype and lack of sustained response. HCV proteins may block IFN-α-induced antiviral polypeptides, thus allowing virus replication to take place (26, 35, 36). However, the induction of a stronger antiviral response may overcome this blockage, suggesting that treatment of these species may require a higher amount of IFN-α.
These clinical limitations in addition to the high cost of the treatment have prompted research for new delivery systems, either by modifying the recombinant protein (13) or through a gene therapy approach. To this end, the antiviral properties of different IFN-α types have been explored in mice using naked DNA injection into muscle. A prophylactic injection of plasmid DNA expressing different murine IFN-α (mIFN-α) types succeeded in reducing cytomegalovirus replication in the injected mice to a limited extent (42). In the case of HBV, transgenic mice have been developed as a model for viral replication and gene expression. Injection of a first-generation adenovirus (Ad) vector expressing the β-galactosidase gene (Ad-βgal) led to a transient inhibition of HBV virus replication in association with the advent of inflammatory cytokines induced by adenovirus injection (5).
IFN-α exhibits a short half-life in the blood after parenteral protein administration (15, 40), suggesting that the limited performance of IFNs in hepatitis treatment may be caused, at least in part, by insufficient or lack of sustained delivery of the protein to the liver. Because liver-specific gene delivery could overcome these limitations, we have explored the effects of mIFN-α2 gene delivery mediated by Ad vectors that have been shown to transduce mainly in the liver when injected intravenously (17). To further limit mIFN-α2 expression to the target tissue, the mIFN-α2 gene was cloned under the control of the liver-specific transthyretin promoter (TTR) (27). The mIFN-α2 expression cassette was rescued in a first-generation Ad vector, Ad-IFN, and in a helper-dependent (HD) vector, HD-IFN. The HD vectors do not express any viral proteins and result in prolonged transgene expression in immunocompetent mice upon systemic delivery (4, 20, 24, 33). In this study, we show that the combination of an HD vector and liver-specific promoter resulted in intrahepatic IFN-α expression, which protected the liver in acute hepatitis models.
MATERIALS AND METHODS
Cell lines.
293 and 293Cre4 cells (23) were grown in minimal essential medium (MEM) supplemented with 10% heat-inactivated fetal calf serum (FCS). 911 (human embryonic retinoblasts), L-929 (mouse fibroblasts), and HuH-7 (human hepatoma) cells were grown in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% FCS. DBT cells (mouse fibroblasts) were grown in DMEM supplemented with 10% tryptose phosphate broth and 5% fetal calf serum.
Mouse strains.
The mice used in this study were immunocompetent, 6- to 8-week-old (at the time of injection), C57/B6 females purchased from Charles River. Groups of four to five mice received injections in the tail vein of Ad vectors diluted in physiologic solution in volumes of 200 μl. Blood was obtained by retroorbital bleeding, and serum was stored at −80°C. At the indicated time, mice were sacrificed, and organs were rapidly frozen in liquid nitrogen and stored at −80°C.
Construction of mIFN-α2 expression cassette.
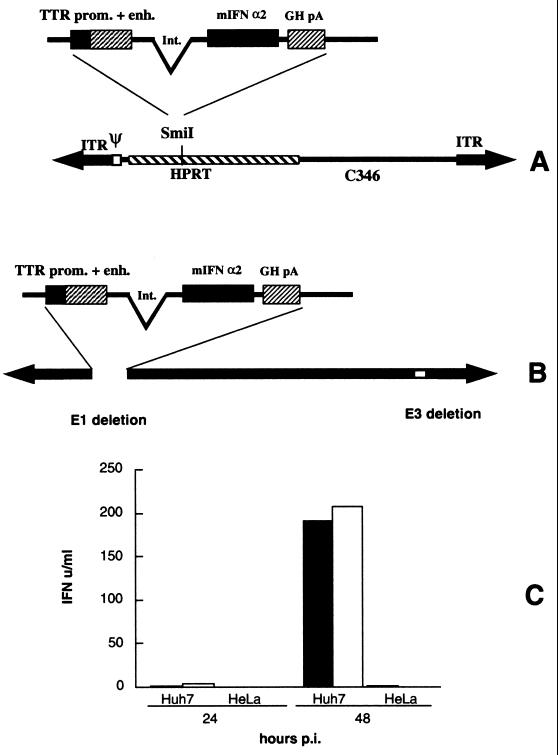
A synthetic mIFN-α2 gene was assembled by PCR with 28 oligonucleotides, 40 nucleotides in length, which collectively encoded both strands of the gene, as previously described (34). The PCR product was then cloned in pBluescript II (pBS-mIFN) and sequenced. The bovine growth hormone poly(A) site was amplified by PCR from plasmid pcDNA (Invitrogen), adding an XbaI site at the 5′ end and a NotI site at the 3′ end. This fragment was cloned downstream of the mIFN-α2 cDNA in pBS-mIFN, generating pBS-mIFN-bGH. The liver-specific TTR gene minimal enhancer and promoter (27) was excised as a HindIII fragment from pTTR-CAT (kindly provided by R. H. Costa) and inserted upstream of the mIFN cDNA in pBS-mIFN-bGH, generating pTTR-mIFN-bGH. To improve the level of expression, an artificial intron was amplified by PCR from pCAT3basic (Invitrogen) and subcloned into the HindIII and EcoRI sites between the TTR promoter and enhancer and the mIFN cDNA, generating pTTR-intr-mIFN-bGH (Fig. 1).
FIG. 1.
Structure of HD-IFN and Ad-IFN and expression of mIFN-α2 in vitro. (A and B) mIFN-α2 expression cassette used in this study. The mIFN-α2 gene was cloned downstream of the TTR promoter (prom.) and enhancer (enh.) followed by the polyadenylation signal of bovine growth hormone (GH pA). Int., intron. (A) To generate the HD-IFN vector, the expression cassette was cloned in the SmiI site of the STK120 backbone vector, which contains the following sequence: the left-terminus Ad5 internal terminal repeats (ITR) and packaging signal (ψ); a fragment of the human hypoxanthine guanine phosphoribosyltransferase (HPRT); a human fragment of the C346 cosmid; and the right-terminus Ad5 ITR sequence. (B) To derive the Ad-IFN vector, the expression cassette was recombined in the E1 region of pHVAd1 vector as described in Materials and Methods. (C) The human cell lines Huh-7 and HeLa were transduced with either HD-IFN (open bars) or Ad-IFN (solid bars) at a multiplicity of infection of 10. Secretion of mIFN-α2 into the cell culture medium was measured by the VSV cythopathic inhibition assay as described in Materials and Methods.
Ad vectors.
To construct pHD-IFN, the mIFN-α2 expression cassette was excised from pTTR-intr-mIFN-bGH using SalI and NotI and subcloned in pABS4 (Microbix). Subsequently, the mIFN-α2 cassette and kanamycin resistance gene were recovered as a PvuII fragment and subcloned in the SmiI site of pSTK120 (24, 29), generating pHD-IFN. Plasmid pAd1-IFN was generated by homologous recombination in Escherichia coli as previously described with slight modifications (6). Briefly, the mIFN-α2 cassette was subcloned in plasmid pΔE1sp1B and recombined in vector pHVAd1 by transformation in E. coli strain BJ 5183 (Fig. 1).
To rescue the helper-dependent HD-IFN vector, plasmid pHD-IFN was cleaved with PmeI and transfected into 293Cre4 cells. Subsequently, the cells were infected with the helper Ad AdLC8cluc (25). The titer of the HD vector during amplification passages on 293Cre4 cells was monitored by infecting HuH-7 cells with lysates from each passage and determining the amount of mIFN-α in cell supernatants at 48 h postinfection by the vesicular stomatitis virus (VSV) inhibition assay. Similarly, pAd-IFN was linearized with PmeI and transfected into 293 cells. Multiple virus passages were performed to reach a high titer. Both Ad vectors were purified on a CsCl density gradient, and physical particles were measured by optical density of DNA. The titer of Ad-IFN, measured as plaque-forming units, was determined on 911 cells by standard plaque assay. Ad-βgal is a first-generation vector that carries the β-galactosidase gene under the transcriptional control of a cytomegalovirus immediate-early promoter (Quantum Biotechnologies).
Cythopathic inhibition assay for IFN.
The viral cytopathic inhibition assay with VSV has been described elsewhere (1). mIFN-α2 activity is expressed in units per milliliter. mIFN-α2 activity was calibrated against the curve obtained with a standard recombinant mIFN-α (Calbiochem).
Intrahepatic mIFN-α2 measurement.
Livers were weighed and homogenized in phosphate-buffered saline with a Polytron homogenizer, and lysates were centrifuged for 30 min at 4°C at 14,000 rpm to eliminate cell debris. Since IFN-α is acid stable, HCl (0.5 N) was added to achieve pH 2.0, and extracts were incubated overnight at 4°C. Neutral pH was reached by adding NaOH, and the extracts were centrifuged for 30 min at 4°C at 14,000 rpm. Clarified extracts were then analyzed by the VSV inhibition assay.
Northern blot analysis and RNase protection assay.
Frozen tissues were mechanically pulverized, and RNA was isolated from tissues with the Ultraspec RNA reagent (Biotecx Laboratories) according to the manufacturer's instructions. Total RNA (20 μg) was used in Northern blot analysis. The intensity of bands was quantified by PhosphorImager analysis. The RNase protection assay for quantification of mRNA was performed with the RiboQuant Multi-Probe RPA assay system (PharMingen) according to the manufacturer's instructions.
Induction of acute hepatitis.
Mouse hepatitis virus type 3 (MHV-3) was amplified on mouse fibroblast DBT cells, and the titer was measured in a standard plaque assay. A total of 100 PFU was injected intraperitoneally (i.p.) in 100 μl of physiologic solution. To measure MHV-3 infectious particles present in the liver, fractions were homogenized with a Polytron homogenizer, and the lysate was centrifuged for 30 min. The titer of MHV-3 in the supernatant was determined by plaque assay. Concanavalin A (ConA; Sigma) was suspended in physiologic solution and injected intravenously (i.v.).
Biochemical and histological analysis.
The extent of hepatocellular injury induced by MHV-3 or ConA injection was monitored by measuring serum alanine aminotransferase (GPT) activity at the indicated time points. GPT activity was measured in a Spotchem model SP-4410 according to the manufacturer's instructions. For histological analysis, liver fractions that had been fixed by immersion in 10% Formalin in 0.1 M phosphate buffer (pH 7.4) were dehydrated in graded alcohol and xylene and embedded in paraffin. Sections were cut and stained with Harris hematoxylin and counterstained with eosin Y. Tissue sections were subsequently washed with distilled water, dehydrated in graded alcohol and xylene, and mounted with Permount. Tissue sections were assessed for the presence of necrosis and inflammatory cell infiltration.
RESULTS
Construction of Ad vectors.
To obtain liver-specific expression of mIFN-α2, we rescued an HD vector carrying the mIFN-α2 gene under the control of the TTR promoter and enhancer (28) (Fig. 1A). To compare the transduction efficiency of HD-IFN to that of a first-generation Ad vector, Ad-IFN was constructed. This vector is an E1- and E3-deleted Ad in which the same expression cassette present in HD-IFN was inserted in the E1 region (Fig. 1B). The infectious titer of HD-IFN was established on the basis of mIFN-α2 units produced in the supernatant of hepatoma Huh-7 cells infected with different amounts of both virus preparations, and the HD-IFN titer was expressed as plaque-forming unit equivalents (PFU-E).
To assess tissue-specific expression, two different cell lines were infected with both viruses (Fig. 1C). Similar amounts of mIFN-α2 were produced over time by HD-IFN- and Ad-IFN-infected human hepatoma cells (Huh-7). In contrast, mIFN-α2 was not detected upon infection of nonhepatic cells (HeLa). This result indicates that both Ad vectors direct hepatic expression of the mIFN-α2 gene.
Assessment of biological response.
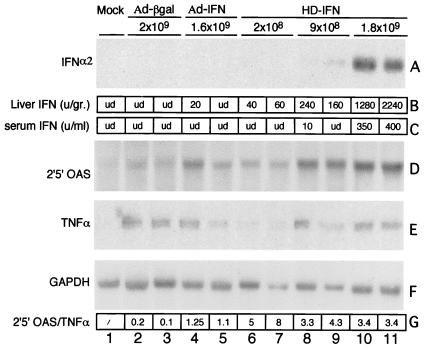
To verify that mIFN-α2 gene delivery could elicit a biological response in vivo, C57/B6 mice were injected with increasing doses of HD-IFN: 2 × 108, 9 × 108, and 1.8 × 109 PFU-E. For comparison, a group of mice were injected with 1.6 × 109 PFU of Ad-IFN. Last, a group of mice were injected with 2 × 109 PFU of the Ad-βgal vector to evaluate induction of antiviral genes mediated by Ad injection in the absence of mIFN-α2 transgene expression. Mice were sacrificed at day 7 postinjection (p.i.), and serum and livers were collected to examine mIFN-α2 content and biological effects. Total liver RNA was prepared from a liver fraction, and mIFN-α2 expression was measured by Northern blot analysis. As shown in Fig. 2A, mIFN-α2 mRNA was detectable only at the highest dose of HD-IFN injected, whereas a faint hybridization signal was detected in livers from mice injected with 9 × 108 PFU-E. No mIFN-α2 transcripts were detected in mice injected with the lowest HD dose, either with Ad-IFN or with Ad-βgal. We next examined the presence of mIFN-α2 in the liver. For this purpose, fractions were homogenized and incubated overnight at low pH to eliminate the acid-labile IFN-γ that could have been released by the inflammatory response against Ad particles (5). The acid-stable mIFN-α2 was measured by the VSV inhibition assay (1). mIFN-α2 was not detected in Ad-βgal-injected mice, and in Ad-IFN-injected mice, an almost undetectable level was observed only in one of two mice examined (Fig. 2B). In contrast, injection of HD-IFN resulted in a dose-response correlation, with the dose of 1.8 × 109 PFU-E yielding almost 1,000-fold-more hepatic mIFN-α2 than the corresponding dose of Ad-IFN. More importantly, injection of the lowest HD-IFN dose resulted in detectable levels of mIFN-α2 in both mice analyzed (in the range of 50 U/g). The presence of mIFN-α2 in the serum was clearly detected only in mice injected with HD-IFN at the dose of 1.8 × 109 PFU-E and almost undetectable at the dose of 9 × 108 PFU-E (Fig. 2C).
FIG. 2.
HD-IFN-induced biological response. C57/B6 mice were injected i.v. with HD-IFN at different doses (2 × 108, 9 × 108, and 1.8 × 109 PFU-E), Ad-IFN (2 × 109 PFU), or Ad-βgal (2 × 109 PFU) or mode injected and euthanized at 1 week p.i. (A) Northern blot analysis was performed with 20 μg of total liver RNA from two representative mice per group. Membrane was hybridized with a radiolabeled mIFN-α2 probe. (B) Intrahepatic mIFN-α2 content, expressed as units of mIFN-α2 per gram of liver. ud, undetectable. (C) mIFN-α2 levels present in the serum, expressed as units per milliliter, were measured at the time of autopsy in the VSV inhibition assay as described in Materials and Methods. (D) Northern blot analysis for expression of 2′5′OAS. (E) Total liver RNA was also analyzed for TNF-α expression in an RNase protection assay. (F) The housekeeping gene glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used for normalization of RNA loading in each lane. (G) The intensities of the 2′5′OAS, TNF-α, and GAPDH bands were quantified with a PhosphorImager and are expressed as the ratio of 2′5′OAS to TNF-α corrected for GAPDH.
To ascertain whether genes normally involved in the antiviral response induced by mIFN-α2 were also activated upon injection of Ad vectors, the same filters were hybridized with the 2′5′OAS probe (Fig. 2D). A weak hybridization signal was observed in mice injected with 2 × 109 PFU of Ad-βgal. The induction of 2′5′OAS may have been triggered by IFN-γ production, as a result of an inflammatory response directed against the first-generation Ad vector (5). At the same dosage, Ad-IFN yielded a slightly higher induction of 2′5′OAS than that mediated by Ad-βgal. This induction may be explained by the low level of expression of the mIFN-α2 gene, as indicated by the presence of mIFN-α2 protein in the liver (Fig. 2B). As observed for the intrahepatic content of mIFN-α2, the induction of 2′5′OAS correlated with the dose of HD-IFN. The injection of 1.8 × 109 PFU-E induced a 25-fold-greater activation of 2′5′OAS than the corresponding dose of Ad-βgal. Also, injection of 2 × 108 PFU-E, which resulted in a small amount of intrahepatic mIFN-α2, induced a significant biological response.
Lastly, the presence of inflammatory cytokines was examined in an RNase protection assay (Fig. 2E). Tumor necrosis factor alpha (TNF-α) was induced by all vectors in a dose-dependent manner, with an almost undetectable signal at the lowest HD-IFN dose. In order to establish a more precise comparison between IFN response consequent to local gene delivery and liver inflammation induced by Ad vectors, the mRNA expression levels of 2′5′OAS and TNF-α were normalized, and their ratio was calculated (Fig. 2G). Interestingly, the HD vectors at all doses tested gave a higher value than the first-generation vectors, and the best conditions (high IFN response and least inflammation) were seen with the lowest viral load. Taken together, these results indicate that the biological response induced by HD-IFN is not a consequence of the inflammation induced by Ad per se but is mainly driven by mIFN-α2 transgene expression.
Hepatic protection mediated by HD-IFN.
To investigate the therapeutic potency of mIFN-α2 gene delivery, we assessed the antiviral strength of HD-IFN in acute hepatitis models. For this purpose, we examined the effects of HD-IFN on the infection of susceptible mouse strain C57/B6 with mouse coronavirus MHV-3. Treatment with recombinant mIFN-β type I was shown to prolong survival following MHV-2 exposure, particularly when the treatment was initiated prior to viral infection (19, 22).
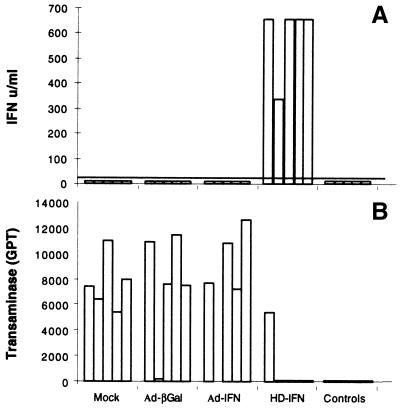
Mice were injected i.v. with different Ad vectors (3 × 109 PFU-E of HD-IFN, 3 × 109 PFU of Ad-IFN, and 3 × 109 PFU of Ad-βgal) and infected i.p. with MHV-3 (100 PFU) at 1 week p.i. At this time point, circulating mIFN-α2 was not detected in either Ad-IFN- or Ad-βgal-injected mice, whereas high levels of serum mIFN-α2 were observed in mice injected with HD-IFN (Fig. 3A). To verify the impact of mIFN-α2 gene delivery on MHV-3-mediated liver damage, transaminase (GPT) levels in the serum were measured 3 days after MHV-3 infection (Fig. 3B). A sharp rise in GPT levels was found in all mock-injected mice and in four of five mice treated with Ad-IFN or Ad-βgal, whereas protection from MHV-3 challenge was observed in 80% of the mice injected with HD-IFN. Ad injection had no effect on the lethality of MHV-3 infection regardless of whether the mice were injected with HD or a first-generation Ad vector (mean survival ± standard deviation: mock, 4.3 ± 0.54 days; Ad-βgal, 4.8 ± 0.83 days; Ad-IFN, 4.8 ± 0.83 days; and HD-IFN, 5.4 ± 1.5 days).
FIG. 3.
Hepatic protection in acute hepatitis induced by MHV-3. Groups of five C57/B6 mice were injected i.v. with Ad-IFN (3 × 109 PFU), Ad-βgal (3 × 109 PFU), or HD-IFN (3 × 109 PFU-E) or mock injected. Untreated mice were used as controls. At day 7 p.i., mice were infected with an i.p. injection of MHV-3 (100 PFU), and transaminase (GPT) levels present in the serum were measured 3 days later. (A) mIFN-α2 present in the serum at 7 days p.i., measured in the VSV inhibition assay. Bars represent individual animals. The horizontal line indicates the limit of detection of the assay. (B) Transaminase (GPT) levels (expressed as units per liter) present in the serum after MHV-3 infection in the same groups of mice as in panel A.
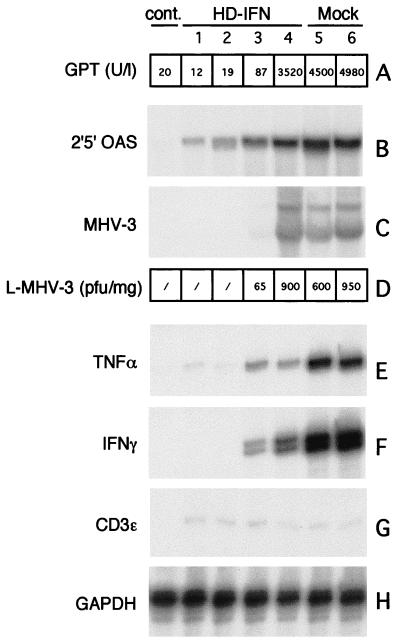
To verify whether hepatic protection could be achieved in the absence of circulating mIFN-α2, a group of mice were injected with a 10-fold-lower dose of HD-IFN (3 × 108 PFU-E). Three days after MHV-3 (100 PFU) infection, the mice were sacrificed, and livers and serum were collected. Although at day 7 p.i. circulating mIFN-α2 was not detected (data not shown), three of four mice preinjected with HD-IFN displayed normal GPT levels in the serum (Fig. 4A). Northern blot analysis of liver RNA with a probe specific for 2′5′OAS gene showed that expression of mIFN-α2-responsive genes had occurred upon HD-IFN injection (Fig. 4B). A stronger 2′5′OAS signal was observed in mice injected with MHV-3 alone. However, in these mice, inflammatory cytokines such as TNF-α and IFN-γ were induced, as measured in an RNase protection assay (Fig. 4E and F), which may explain the stronger 2′5′OAS induction. MHV-3 viral transcripts were detected in mice that displayed an increase in GPT levels, whereas they could not be detected in mice that had been injected with HD-IFN (Fig. 4C). The inhibition of viral replication correlated with the MHV-3 titer in the liver extracts. MHV-3 infectious particles were detected in only one of four HD-IFN-protected mice, whereas significant amounts of virus were found in the livers from all untreated mice (Fig. 4D). T-cell activation, monitored as expression of the CD3ɛ marker, was not different between the groups (Fig. 4G).
FIG. 4.
Hepatic protection in acute hepatitis induced by MHV-3 in the absence of circulating mIFN-α2. C57/B6 mice were mock injected or injected with HD-IFN at the dose of 3 × 108 PFU-E as described in the legend to Fig. 3. Samples were collected at 3 days after MHV-3 infection and analyzed. (A) Transaminase (GPT) levels (expressed as units per liter) present in serum were determined at the time of autopsy (3 days after MHV-3 infection). (B) Northern blot analysis was performed with 20 μg of total RNA from a liver fraction from each mouse. Membrane was hybridized with a radiolabeled 2′5′OAS probe. (C) Northern blot analysis with a 250-bp probe from the 3′ end of the MHV-3 genome, obtained from plasmid DE25 (21). (D) MHV-3 particles present in a liver, expressed per milligram of liver weight. Total liver RNA was also analyzed in an RNase protection assay for expression of inflammatory cytokines such as TNF-α (E) and IFN-γ (F) and for a marker for T-lymphocyte activation, CD3ɛ (G). (H) The housekeeping gene GAPDH was used for normalization. cont., controls.
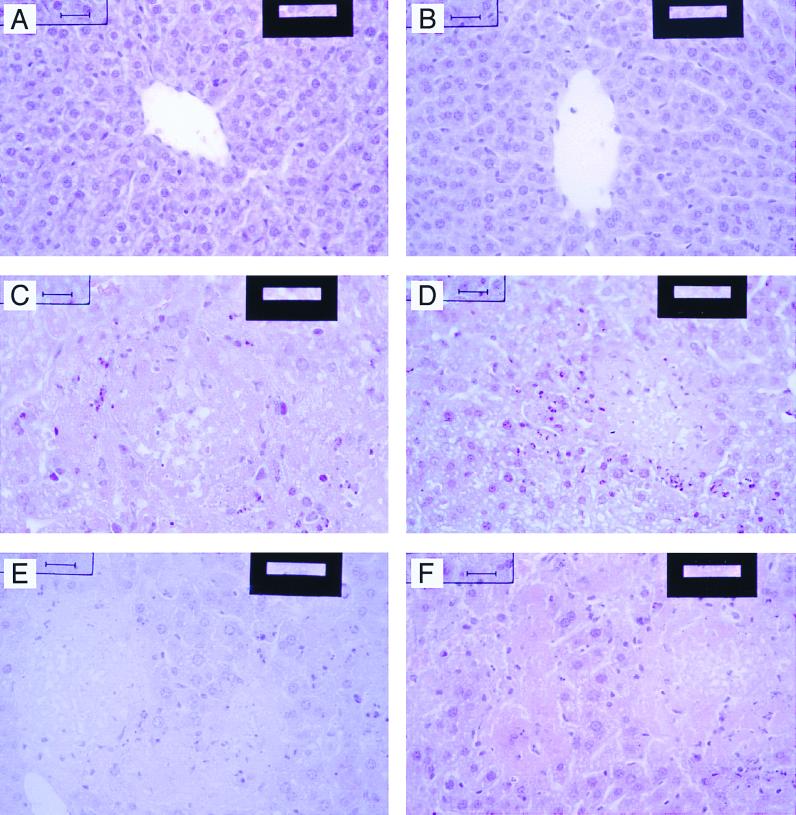
Last, liver histology was performed for HD-IFN- or mock-preinjected mice and the mice preinjected with Ad-βgal (Fig. 5). Liver fractions obtained from HD-IFN-treated mice showed normal architecture, whereas untreated and Ad-βgal-injected mice showed evidence of extensive hepatocellular necrosis. These results show that the hepatic damage consequent to MHV-3 infection is reduced by HD-IFN.
FIG. 5.
Liver histology. (A and B) Liver samples from two animals preinjected with HD-IFN. The hepatic parenchyma is normal; no morphologic patterns of hepatic injury are detectable. (C and D) Liver samples from two animals preinjected with Ad-βgal. (E and F) Liver samples from two animals mock injected. In C, D, E, and F, several areas of zonal necrosis in the hepatic parenchyma are depicted. A main central core of liquefactive necrosis characterizes these areas where nuclei are undetectable, and apoptotic cells with mainly pyknotic nuclei surround them. Hematoxylin and eosin staining. Bars, 35 μm.
To extend these results, the effects of HD-IFN injection were examined in an additional acute hepatitis model. Among the more commonly used models, injection of ConA appears to be the most specific in inducing hepatic injury by T-cell activation which is associated with the production of different inflammatory cytokines, including TNF-α, interleukin-2, and IFN-γ (23). IFN-α/β was shown to protect mice from liver damage induced by ConA challenge, as indicated by a reduced level of serum transaminases (3).
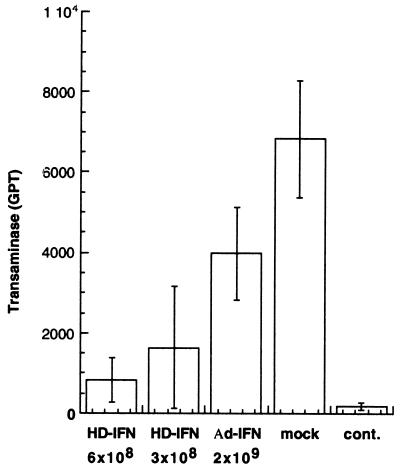
C57/B6 mice were injected with HD-IFN (3 × 108 PFU-E and 6 × 108 PFU-E) and Ad-IFN (2 × 109 PFU). Serum mIFN-α2 was monitored on days 8, 16, and 25, and it was not detected in mice injected with Ad-IFN or with the lower dose of HD-IFN (data not shown). By contrast, a low level of mIFN-α2 was detected in the serum of mice injected with 6 × 108 PFU-E of HD-IFN, which ranged between 12.5 and 5 U of mIFN-α2 per ml. The injected mice were challenged with ConA at day 30 p.i., and GPT levels were measured 24 h after challenge. Mice injected with Ad-IFN at the high dosage (2 × 109 PFU) were only marginally protected compared with mock-treated mice (Fig. 6). On the contrary, an eightfold reduction in GPT levels was observed in mice injected with the more effective HD-IFN virus at the higher dose (6 × 108 PFU-E). Consistently, injection of HD-IFN at the dose of 3 × 108 PFU-E resulted in lower but significant protection. Taken together, these results indicate that HD-IFN exhibits a protective effect against liver injury even at doses that do not yield circulating mIFN-α2 levels.
FIG. 6.
ConA-induced hepatitis. HD-IFN injection protects C57/B6 mice from ConA challenge. C57/B6 mice were injected i.v. with HD-IFN at different dosages (6 × 108 or 3 × 108 PFU-E) or with Ad-IFN at 2 × 109 PFU and challenged with ConA (0.3 g/mouse) 30 days p.i. At 24 h after ConA challenge, transaminases (GPT) (expressed as units per liter) present in the serum were measured. The basal level of serum GPT was measured in untreated mice which were used as controls (cont.). Data are the mean ± standard deviation of serum GPT measured in four animals.
DISCUSSION
We have described the construction of an HD Ad expressing the mIFN-α2 gene. Comparison with a first-generation Ad vector containing the same mIFN-α2 expression cassette indicates that these two vectors function quite differently in vivo, with the HD vector performing more efficiently in inducing an mIFN-α2-mediated biological response and greater therapeutic potency.
The better performance of the HD vector over a first-generation Ad vector carrying the same expression cassette was recently observed with the mouse leptin gene (24). Also in those experiments at 1 week p.i., the transgene mRNA was observed only in the HD-injected mice and not in those injected with the Ad vector. Several factors have been shown to contribute to the weaker Ad-mediated gene expression, including “leaky” viral protein expression, which may trigger an immune response against transduced cells (37, 41). The host immune response may be even more relevant to IFN gene delivery because mIFN-α2 immune modulatory activity may contribute to clearance of the transduced cells. Although we have not examined the extent of clearance of transduced liver cells, the different efficiency of gene expression between the Ad and HD vectors supports the notion that the latter viruses, which do not express Ad proteins, are safer vectors that should guarantee efficient and prolonged expression of the transgene of interest. In this regard, we have observed in preliminary experiments that liver expression of mIFN-α2 mediated by HD-IFN injection is detectable for 3 months (data not shown).
Injection of HD-IFN resulted in high levels of liver mIFN-α2 (Fig. 2). It has been reported previously that after injection of mice with 105 U, homologous IFN-α levels peaked in the liver at 1 h p.i. (104 U/g) and subsequently decreased to baseline levels after 8 h (15). In contrast, injection of HD-IFN (1.8 × 109 PFU-E) resulted in a fivefold-higher mIFN-α2 concentration in the liver than in the serum at 1 week p.i. Moreover, the biological response, measured as 2′5′OAS induction, was dose dependent and observed even at the lowest HD-IFN dose, which did not result in circulating mIFN-α2. The lack of circulating mIFN-α2 at the lowest HD-IFN dose may have different explanations. One obvious possibility is the low sensitivity of the mIFN-α2 detection assay. An alternative explanation may be that mIFN-α2 expressed at low levels is trapped by its own receptors in the liver tissue and released into the bloodstream only when produced above a certain threshold, as observed with the highest HD-IFN dose.
Injection of Ad-βgal, which resulted in a detectable induction of 2′5′OAS (Fig. 2), was not associated with protection against MHV-3 challenge (Fig. 3 and 5). It is interesting that a different result was observed in HBV transgenic mice and in lymphocytic choriomeningitis virus-infected C57/B6 mice; injection of a similar dose of the Ad-βgal vector led to transient inhibition of viral replication in both systems (5, 14). In our experiments, protection against the more aggressive MHV-3 was observed only in the presence of IFN-α expression, indicating that HD-IFN not only induced a stronger 2′5′OAS activation, but it likely elicited a broad spectrum of antiviral pathways which may contribute to the antiviral state observed in the protected mice. Hepatic expression of mIFN-α2 led to liver protection in 80% of the C57/B6 mice infected with MHV-3 (Fig. 3). The reason why not all of the HD-IFN-injected mice were protected from MHV-3 challenge remains elusive. The simplest explanation is that MHV-3 replication may have occurred in tissues, including the peritoneum, where the virus was injected, which were not protected by mIFN-α2. This may have led to greater virus loads in the liver, and gradually the mIFN-α2-mediated block to virus multiplication may have been overcome in an increasing number of cells. A similar mechanism may also explain the minimal increase in survival time observed in the HD-IFN-treated mice, which may have been limited by MHV-3 replication in extrahepatic tissues (11).
In the presence of circulating IFN-α (Fig. 3), peripheral effects such as peritoneal macrophage activation may contribute to liver protection. However, injection of a low HD-IFN dose which resulted in IFN-α being present only in the liver was equally effective, indicating that viral resistance mechanisms activated at the local level are sufficient for liver protection (Fig. 4). In addition, mice protected with a lower dose of HD-IFN showed induction of the 2′5′OAS antiviral gene associated with an absence of MHV-3 transcripts and viral particles, in line with the notion that viral clearance may occur without destruction of potentially infected cells (14). Furthermore, no liver histology alterations were noted in mice pretreated with HD-IFN, indicating that injection of HD-IFN had no negative effects on the liver tissue which could be ascribed to MHV-3 replication in association with the inflammatory response (Fig. 5). It is known that IFN-α promotes a variety of antiviral effects, which include induction of antiviral genes and immunomodulatory effects. The absence of MHV-3 mRNAs in the infected liver indicates that antiviral activities such as those induced by 2′5′OAS may be responsible for MHV-3 clearance. However, we cannot exclude the possibility that virus entry and/or uncoating was also affected by IFN-α expression, as observed for simian virus 40 and retroviruses (38), or that IFN-α may have induced a local immune response that was ultimately responsible for MHV-3 clearance.
HD-IFN also proved efficient in protecting the liver from damage associated with ConA challenge at 30 days p.i., suggesting that transgene expression continued during this period of time (Fig. 6). The anti-inflammatory activity of type I IFNs is thought to be mediated, at least in part, by inhibiting production of inflammatory mediators (16). This protection may have important implications in cirrhosis of the liver, where parenchymal cells are progressively destroyed by activated T cells (39).
The results obtained in this study indicate that the HD-IFN vector is quite effective in protecting the liver from the damage associated with acute hepatitis. We therefore believe that local delivery of IFN-α to the liver tissue with HD Ad vectors might be a promising approach for the treatment of chronic hepatitis. In this context, a more in-depth assessment of the minimal therapeutic dose and potential toxic effects needs to be carried out before this vector can be considered a candidate for the treatment of chronic hepatitis. The development of HD vectors for IFN-α gene delivery to the liver will have to include transcriptional regulatory elements in order to guarantee a minimal effective therapeutic level of IFN-α expression. This additional modification of the HD vector will further increase the safety features of IFN-α hepatic gene delivery. To this end, regulated liver gene expression has recently been demonstrated with an HD vector carrying an RU486-inducible promoter driving transcription of growth hormone (4). Finally, a regulated HD-IFN vector will have to show therapeutic potency in a validated animal model of chronic hepatitis, such as woodchuck hepatitis (32). Utilizing a woodchuck IFN-α homolog to minimize immune responses against IFN-α-transduced cells could provide information on the efficacy of IFN-α gene delivery in a chronically damaged and probably inflamed liver.
An additional feature that renders the delivery of IFN-α to the liver an attractive therapeutic strategy is the observation that antiviral effects are observed with no detectable release of IFN-α into the bloodstream. The restricted expression of IFN-α is particularly relevant in light of the side effects associated with the current treatment of HCV patients, who normally receive 3 × 106 U of r-hIFN-α2 three times per week. Patients receiving higher or more frequent doses suffer from even worse side effects, possibly induced by circulating IFN-α (2). It is likely that selective expression of IFN-α in the liver may render it more tolerable. A further advantage is that liver-specific expression of IFN-α could have autocrine and paracrine effects on the target organ, perhaps resulting in very significant bystander effects and thereby maximizing the therapeutic potency of IFN-α.
ACKNOWLEDGMENTS
We thank M. M. C. Lai, S. Makino, S. Stohlman, and F. Taguchi for providing MHV-3 samples and advice, S. Germoni and M. Aquilina for animal care, and E. Fattori and P. Monaci for critically reading the manuscript. We also thank J. Clench for editorial assistance and M. Emili for graphics.
REFERENCES
- 1.Armstrong J A. Cytopathic effect inhibition assay for interferon: microculture plate assay. Methods Enzymol. 1981;78:381–387. doi: 10.1016/0076-6879(81)78145-x. [DOI] [PubMed] [Google Scholar]
- 2.Ascione A, De Luca M, Canestrini C, Di Costanzo G G, Raimondo G, Longo G, Manns M P, Tillmann H L, Forte G B, Rocco P, Biceglia O, Faleo D, Vinelli F, Cela E M, Amitrano L, Addario L, Gigliotti T. Efficacy of high dose of recombinant alpha 2b interferon on long term response in chronic hepatitis C and cirrhosis: prospective randomized multicentre study. Ital J Gastroenterol Hepatol. 1998;30:517–523. [PubMed] [Google Scholar]
- 3.Bahr G M, Pouillart P R, Chedid L A. Enhancement in vivo of the antiinflammatory and antitumor activities of type I interferon by association with the synthetic immunomodulator murabutide. J Interferon Cytokine Res. 1996;16:297–306. doi: 10.1089/jir.1996.16.297. [DOI] [PubMed] [Google Scholar]
- 4.Burcin M M, Schiedner G, Kochanek S, Tsai S Y, O'Malley B W. Adenovirus-mediated regulable target gene expression in vivo. Proc Natl Acad Sci USA. 1999;96:355–360. doi: 10.1073/pnas.96.2.355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cavanaugh V J, Guidotti L G, Chisari F V. Inhibition of hepatitis B virus replication during adenovirus and cytomegalovirus infections in transgenic mice. J Virol. 1998;72:2630–2637. doi: 10.1128/jvi.72.4.2630-2637.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chartier C, Degryse E, Gantzer M, Dieterle A, Pavirani A, Mehtali M. Efficient generation of recombinant adenovirus vectors by homologous recombination in Escherichia coli. J Virol. 1996;70:4805–4810. doi: 10.1128/jvi.70.7.4805-4810.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Colamonici O R, Porterfield B, Domanski P, Constantinescu S, Pfeffer L M. Complementation of the interferon alpha response in resistant cells by expression of the cloned subunit of the interferon alpha receptor. A central role of this subunit in interferon alpha signaling. J Biol Chem. 1994;269:9598–9602. [PubMed] [Google Scholar]
- 8.David M. Transcription factors in interferon signaling. Pharmacol Ther. 1995;65:149–161. doi: 10.1016/0163-7258(94)00050-d. [DOI] [PubMed] [Google Scholar]
- 9.Davis G L, Esteban-Mur R, Rustgi V, Hoefs J, Gordon S C, Trepo C, Shiffman M L, Zeuzem S, Craxi A, Ling M H, Albrecht J. Interferon alfa-2b alone or in combination with ribavirin for the treatment of relapse of chronic hepatitis C. International Hepatitis Interventional Therapy Group. N Engl J Med. 1998;339:1493–1499. doi: 10.1056/NEJM199811193392102. [DOI] [PubMed] [Google Scholar]
- 10.Der S D, Zhou A, Williams B R, Silverman R H. Identification of genes differentially regulated by interferon alpha, beta, or gamma using oligonucleotide arrays. Proc Natl Acad Sci USA. 1998;95:15623–15628. doi: 10.1073/pnas.95.26.15623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ding J W, Ning Q, Liu M F, Lai A, Leibowitz J, Peltekian K M, Cole E H, Fung L S, Holloway C, Marsden P A, Yeger H, Phillips M J, Levy G A. Fulminant hepatic failure in murine hepatitis virus strain 3 infection: tissue-specific expression of a novel fgl2 prothrombinase. J Virol. 1997;71:9223–9230. doi: 10.1128/jvi.71.12.9223-9230.1997. . (Erratum, 72:3504, 1998.) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Domanski P, Colamonici O R. The type-I interferon receptor. The long and short of it. Cytokine Growth Factor Rev. 1996;7:143–151. doi: 10.1016/1359-6101(96)00017-2. [DOI] [PubMed] [Google Scholar]
- 13.Eto T, Takahashi H. Enhanced inhibition of hepatitis B virus production by asialoglycoprotein receptor-directed interferon. Nat Med. 1999;5:577–581. doi: 10.1038/8462. [DOI] [PubMed] [Google Scholar]
- 14.Guidotti L G, Rochford R, Chung J, Shapiro M, Purcell R, Chisari F V. Viral clearance without destruction of infected cells during acute HBV infection. Science. 1999;284:825–829. doi: 10.1126/science.284.5415.825. [DOI] [PubMed] [Google Scholar]
- 15.Heremans H, Billiau A, De Somer P. Interferon in experimental viral infections in mice: tissue interferon levels resulting from the virus infection and from exogenous interferon therapy. Infect Immun. 1980;30:513–522. doi: 10.1128/iai.30.2.513-522.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Heremans H, Dijkmans R, Sobis H, Vandekerckhove F, Billiau A. Regulation by interferons of the local inflammatory response to bacterial lipopolysaccharide. J Immunol. 1987;138:4175–4179. [PubMed] [Google Scholar]
- 17.Huard J, Lochmuller H, Acsadi G, Jani A, Massie B, Karpati G. The route of administration is a major determinant of the transduction efficiency of rat tissues by adenoviral recombinants. Gene Ther. 1995;2:107–115. [PubMed] [Google Scholar]
- 18.Isaacs A, Lindenmann J. I. Virus interference. Proc R Soc Lond. 1957;147:258–267. [PubMed] [Google Scholar]
- 19.Kato Y, Noda Y, Unoura M, Tanaka N, Kobayashi K, Hattori N, Hatano K, Kobayashi S. Effect of exogenous mouse interferon on murine fulminant hepatitis induced by mouse hepatitis virus type 2. Dig Dis Sci. 1986;31:177–180. doi: 10.1007/BF01300705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Maione, D., M. Wiznerowicz, P. Delmastro, R. Cortese, G. Ciliberto, N. La Monica, and R. Savino. Prolonged expression and effective readministration of erythropoietin delivered with a fully deleted adenoviral vector. Hum. Gene Ther. in press. [DOI] [PubMed]
- 21.Makino S, Shieh C K, Soe L H, Baker S C, Lai M M. Primary structure and translation of a defective interfering RNA of murine coronavirus. Virology. 1988;166:550–560. doi: 10.1016/0042-6822(88)90526-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Minagawa H, Takenaka A, Mohri S, Mori R. Protective effect of recombinant murine interferon beta against mouse hepatitis virus infection. Antiviral Res. 1987;8:85–95. doi: 10.1016/0166-3542(87)90079-9. [DOI] [PubMed] [Google Scholar]
- 23.Mizuhara H, Kuno M, Seki N, Yu W G, Yamaoka M, Yamashita M, Ogawa T, Kaneda K, Fujii T, Senoh H, Fujiwara H. Strain difference in the induction of T-cell activation-associated, interferon gamma-dependent hepatic injury in mice. Hepatology. 1998;27:513–519. doi: 10.1002/hep.510270227. [DOI] [PubMed] [Google Scholar]
- 24.Morsy M A, Gu M, Motzel S, Zhao J, Lin J, Su Q, Allen H, Franlin L, Parks R J, Graham F L, Kochanek S, Bett A J, Caskey C T. An adenoviral vector deleted for all viral coding sequences results in enhanced safety and extended expression of a leptin transgene. Proc Natl Acad Sci USA. 1998;95:7866–7871. doi: 10.1073/pnas.95.14.7866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Parks R J, Chen L, Anton M, Sankar U, Rudnicki M A, Graham F L. A helper-dependent adenovirus vector system: removal of helper virus by Cre-mediated excision of the viral packaging signal. Proc Natl Acad Sci USA. 1996;93:13565–13570. doi: 10.1073/pnas.93.24.13565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Polyak S J, Paschal D M, McArdle S, Gale M J, Jr, Moradpour D, Gretch D R. Characterization of the effects of hepatitis C virus nonstructural 5A protein expression in human cell lines and on interferon-sensitive virus replication. Hepatology. 1999;29:1262–1271. doi: 10.1002/hep.510290438. [DOI] [PubMed] [Google Scholar]
- 27.Qian X, Samadani U, Porcella A, Costa R H. Decreased expression of hepatocyte nuclear factor 3 alpha during the acute-phase response influences transthyretin gene transcription. Mol Cell Biol. 1995;15:1364–1376. doi: 10.1128/mcb.15.3.1364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Qin X Q, Tao N, Dergay A, Moy P, Fawell S, Davis A, Wilson J M, Barsoum J. Interferon-beta gene therapy inhibits tumor formation and causes regression of established tumors in immune-deficient mice. Proc Natl Acad Sci USA. 1998;95:14411–14416. doi: 10.1073/pnas.95.24.14411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Recchia A, Parks R J, Lamartina S, Toniatti C, Pieroni L, Palombo F, Ciliberto G, Graham F L, Cortese R, La Monica N, Colloca S. Site-specific integration mediated by a hybrid adenovirus/adeno-associated virus vector. Proc Natl Acad Sci USA. 1999;96:2615–2620. doi: 10.1073/pnas.96.6.2615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Reichard O, Norkrans G, Fryden A, Braconier J H, Sonnerborg A, Weiland O. Randomised, double-blind, placebo-controlled trial of interferon alpha-2b with and without ribavirin for chronic hepatitis C. The Swedish Study Group. Lancet. 1998;351:83–87. doi: 10.1016/s0140-6736(97)06088-1. [DOI] [PubMed] [Google Scholar]
- 31.Rice A P, Duncan R, Hershey J W, Kerr I M. Double-stranded RNA-dependent protein kinase and 2-5A system are both activated in interferon-treated, encephalomyocarditis virus-infected HeLa cells. J Virol. 1985;54:894–898. doi: 10.1128/jvi.54.3.894-898.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Roggendorf M, Tolle T K. The woodchuck: an animal model for hepatitis B virus infection in man. Intervirology. 1995;38:100–112. doi: 10.1159/000150418. [DOI] [PubMed] [Google Scholar]
- 33.Schiedner G, Morral N, Parks R J, Wu Y, Koopmans S C, Langston C, Graham F L, Beaudet A L, Kochanek S. Genomic DNA transfer with a high-capacity adenovirus vector results in improved in vivo gene expression and decreased toxicity. Nat Genet. 1998;18:180–183. doi: 10.1038/ng0298-180. . (Erratum, 18:298.) [DOI] [PubMed] [Google Scholar]
- 34.Stemmer W P, Crameri A, Ha K D, Brennan T M, Heyneker H L. Single-step assembly of a gene and entire plasmid from large numbers of oligodeoxyribonucleotides. Gene. 1995;164:49–53. doi: 10.1016/0378-1119(95)00511-4. [DOI] [PubMed] [Google Scholar]
- 35.Tan S L, Nakao H, He Y, Vijaysri S, Neddermann P, Jacobs B L, Mayer B J, Katze M G. NS5A, a nonstructural protein of hepatitis C virus, binds growth factor receptor-bound protein 2 adaptor protein in a Src homology 3 domain/ligand-dependent manner and perturbs mitogenic signaling. Proc Natl Acad Sci USA. 1999;96:5533–5538. doi: 10.1073/pnas.96.10.5533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Taylor D R, Shi S T, Romano P R, Barber G N, Lai M M. Inhibition of the interferon-inducible protein kinase PKR by HCV E2 protein. Science. 1999;285:107–110. doi: 10.1126/science.285.5424.107. [DOI] [PubMed] [Google Scholar]
- 37.Tripathy S K, Black H B, Goldwasser E, Leiden J M. Immune responses to transgene-encoded proteins limit the stability of gene expression after injection of replication-defective adenovirus vectors. Nat Med. 1996;2:545–550. doi: 10.1038/nm0596-545. [DOI] [PubMed] [Google Scholar]
- 38.Vilcek J, Ganes C S. Interferon and other cytokines. In: Fields B N, Knipe D M, Howley P M, editors. Fields virology. 3rd ed. Philadelphia, Pa: Lippincott-Raven; 1996. pp. 375–399. [Google Scholar]
- 39.Wermers G W, Band H, Yunis E J. Role of the HLA system in antigen recognition and disease. New York, N.Y: Raven Press; 1998. [Google Scholar]
- 40.Wills R J. Clinical pharmacokinetics of interferons. Clin Pharmacokinet. 1990;19:390–399. doi: 10.2165/00003088-199019050-00003. [DOI] [PubMed] [Google Scholar]
- 41.Yang Y, Nunes F A, Berencsi K, Furth E E, Gonczol E, Wilson J M. Cellular immunity to viral antigens limits E1-deleted adenoviruses for gene therapy. Proc Natl Acad Sci USA. 1994;91:4407–4411. doi: 10.1073/pnas.91.10.4407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yeow W S, Lawson C M, Beilharz M W. Antiviral activities of individual murine IFN-alpha subtypes in vivo: intramuscular injection of IFN expression constructs reduces cytomegalovirus replication. J Immunol. 1998;160:2932–2939. [PubMed] [Google Scholar]