Abstract
Dermatophytoses are fungal infections of the skin, hair, and nails that affect approximately 25% of the global population. Occlusive clothing, living in a hot humid environment, poor hygiene, proximity to animals, and crowded living conditions are important risk factors. Dermatophyte infections are named for the anatomic area they infect, and include tinea corporis, cruris, capitis, barbae, faciei, pedis, and manuum. Tinea incognito describes steroid-modified tinea. In some patients, especially those who are immunosuppressed or who have a history of corticosteroid use, dermatophyte infections may spread to involve extensive skin areas, and, in rare cases, may extend to the dermis and hair follicle. Over the past decade, dermatophytoses cases not responding to standard of care therapy have been increasingly reported. These cases are especially prevalent in the Indian subcontinent, and Trichophyton indotineae has been identified as the causative species, generating concern regarding resistance to available antifungal therapies. Antifungal-resistant dermatophyte infections have been recently recognized in the United States. Antifungal resistance is now a global health concern. When feasible, mycological confirmation before starting treatment is considered best practice. To curb antifungal-resistant infections, it is necessary for physicians to maintain a high index of suspicion for resistant dermatophyte infections coupled with antifungal stewardship efforts. Furthermore, by forging partnerships with federal agencies, state and local public health agencies, professional societies, and academic institutions, dermatologists can lead efforts to prevent the spread of antifungal-resistant dermatophytes.
1. Introduction
Dermatophytoses are cutaneous infections caused by dermatophytes, fungal pathogens from the Trichophyton, Epidermophyton, and Microsporum genera [1]. Since they require keratin for growth, dermatophytoses are generally limited to skin, hair, and nails, and thrive in warm (25–28 °C), humid environments [2, 3]. Dermatophytoses are often referred to as ‘tinea’ infections (Latin for ‘worm’), referring to the worm-like borders surrounding ring-shaped patches. The word ‘tinea’ is combined with the anatomical site to form the complete nomenclature (Table 1) [4].
Table 1.
Tinea definitions
| Type of tinea infection | Definition |
|---|---|
| Tinea corporis | A superficial fungal infection caused by dermatophytes, with anatomic boundaries defined by the neck, trunk, and extremities |
| Tinea cruris | A superficial fungal infection involving the crural fold. Commonly referred to as ‘jock itch’ |
| Tinea capitis | A dermatophyte infection of the scalp and hair |
| Tinea barbae | A type of dermatophytic folliculitis; a superficial fungal infection of the hair and hair follicles, involving beards and mustaches in adolescents and adults |
| Tinea faciei | A dermatophyte infection of the skin of the face in areas without terminal hairs |
| Tinea incognito | Refers to a tinea corporis, cruris, or faciei infection modified by an anti-inflammatory cream, ointment, or oral therapy that may dampen the local immune response |
| Tinea pedis | Dermatophyte infection involving the foot/feet. Commonly known as ‘athlete’s foot’ |
| Tinea manuum | Dermatophyte infection involving the hand(s) |
| Onychomycosisa | Fungal infection of the nails |
Outside the scope of this review and will not be explored independently
1.1. Prevalence and Main Causal Organisms
An estimated 20–25% of the global population are affected by dermatophytoses [1]. Trichophyton rubrum is the most common causative agent in high-income countries, and Trichophyton mentagrophytes species complex, Microsporum canis, and Epidermophyton floccosum are most prevalent globally. Trichophyton schoenleinii, Trichophyton soudanese, and Trichophyton concentricum are generally limited to parts of Europe, Asia, and Africa [5, 6]. Epidemiology varies over time due to tourism and migration [2, 5, 7]. Taxonomy changes have divided the genera Trichophyton and Microsporum and added the new genera Arthroderma, Nannizzia, Paraphyton and Lophophyton [8].
1.2. Pathogenesis and Virulence Factors
Dermatophytes infect cytokeratin comprising the stratum corneum in the hair, skin, and nails, and adhere to the host via mannan glycoproteins in the fungal cell walls [9–11]. The mold grows as linear hyphae, enabling spread to deeper layers of keratin-rich structures. To utilize keratin for growth, dermatophytes break down disulfide bridges linking epidermal keratins. Keratin is rich in cysteine. Dermatophytes secrete sulfite, a reducing agent that cleaves cystine bonds via cysteine dioxygenase, contributing to keratin degradation [9–12]. Dermatophytes produce biofilms, which have been reported in both Trichophyton and Microsporum species. It is hypothesized that biofilms contribute to dermatophyte antifungal treatment failures [13, 14].
1.3. General Risk Factors
Table 2 lists risk factors contributing to dermatophytosis susceptibility.
Table 2.
Risk factors
| Type of tinea infection | Risk factors |
|---|---|
| All tinea infections | Contact with an individual harboring a dermatophytosis Secondary spread of infection from another affected anatomic region Hyperhidrosis Diabetes mellitus Poor hygiene Obesity Immunosuppression Living in a hot, humid, tropical climate Occlusive clothing/footwear/headwear Contact sports Use of antibiotics or corticosteroids (topical and systemic) Contact with infected animals (pets, stray, farm, laboratory, and wild animals) Being of low socioeconomic status, due to: Crowded living conditions Increased skin-to-skin contact Reduced access to hygienic products, including soaps, shampoos, detergents, etc. Increased proximity to animals, including house pets (i.e. dogs, cats) or livestock (i.e. cattle, horses) [2, 4] |
| Tinea corporis | Tinea gladiatorum is a subtype of tinea corporis caused mainly by T. tonsurans (common among athletes) Skin-to-skin contact during athletics (example, wrestling) Contact with contaminated training equipment |
| Tinea cruris | Moisture in the intertriginous groin area Obesity due to apposition of skin folds |
| Tinea capitis | Decreased sebum production, which leads to decreased fatty acid production and raises the pH of the scalp Significant hormonal changes (pregnancy, menopause, puberty) that decrease sebum production Fomites: sharing combs, hairbrushes, hats, pillows Short hairstyles that allow for ease of colonization of the scalp, including immunosuppression causing impaired hair shaft growth |
| Tinea pedis | Participation in sports, including marathon running [238] Wearing occlusive, closed-toe shoes for long periods of time while working, including miners and soldiers [112, 240, 241] Frequenting public swimming pools, showers, and gyms [5, 111, 242, 243], especially without donning appropriate footwear Cultural practices that involve bare feet, such as entering a place of worship [244] |
| Tinea manuum | Contact with the skin of a foot infected with tinea pedis Contact with infected clothing, towel, or soil [3] Recurrent trauma to the hands, such as in individuals who perform manual labor [134] |
| Tinea barbae | Contact with house pets or other animals via occupational exposure to cattle, horses, etc. [243] Contact with improperly cleaned razor, beard brush, or other facial tool [246] Coarse beard hair |
| Tinea faciei | Skin-to-skin contact, including wrestling [247] |
| Tinea incognito | Misdiagnosis of an existing dermatophyte infection Application of steroid cream, tacrolimus ointment, or other anti-inflammatory topical treatment to a dermatophyte infection Systemic glucocorticoids Immunocompromised |
| Chronic and/or recurrent dermatophytosis | Reduced IFN-γ+ cells, Th1 cells, IL-17+ cells, Th17 cells, elevated IL-4+ cells, increased serum IgE levels and a diminished delayed type hypersensitivity intradermal skin test response [189, 248] History of tinea in a family member [189] History of corticosteroid use [189] Sharing towels in the home [189] Some comorbid conditions, including atopy, diabetes mellitus [137] Poor hygiene Steroid use Low socioeconomic status |
IFN interferon, Th T-helper, IL interleukin, Ig immunoglobulin
1.4. Transmission
Dermatophyte transmission is multifactorial [15]. Dermatophytes likely persist on wet surfaces, including in bathrooms and pool facilities [15–17], and may spread via shared clothing, footwear, towels, and bedding, especially with a comprised skin barrier [15, 18, 19]. Pets and stray, farm, laboratory, and wild animals have been implicated in zoonotic spread [20–22].
1.5. Racial Disparities
There are racial disparities in the prevalence of tinea infections, particularly tinea capitis [23, 24]. In a retrospective population-based study of children < 10 years of age, incident rates per 10,000 enrolled were 252.1 claimants for Black children versus 23.1 claimants for White children (p value not reported) [25]. In a prospective, cross-sectional surveillance study of 10,514 kindergarten fifth-grade children, Black versus White and Hispanic children had higher infection rates (12.9%, 1.1%, and 1.6%, respectively; p < 0.01) [26]. A commercial database study of tinea capitis in children < 18 years of age estimated the incidence rate to be sevenfold higher in Black versus White children [27]. In a cross-sectional study of 351,629 HIV patients in a tertiary healthcare system, dermatophytoses were more common in HIV- versus non-HIV-infected individuals (p < 0.0009) [28].
1.6. Looking Ahead
In a prospective survey-based study of errors made by primary care physicians in managing skin disorders, errors related to diagnosis of dermatophytoses accounted for the largest proportion of errors (32%, 102/319) [29]. In a survey-based study of board-certified dermatologists presented with 13 clinical images of fungal infections, respondents categorized cases with > 75% accuracy in only 31% of cases [30], highlighting that visual inspection alone is unreliable for diagnosis. Dermatologists play a key role not only in accurate diagnosis of superficial fungal infections but also in educating other non-dermatologists about dermatophytoses. The American Academy of Dermatology’s guidelines for the treatment of superficial mycotic infections were last published in 1996, necessitating an updated review on diagnosis and management.
2. Tinea Corporis and Tinea Cruris
2.1. Demographics and Geography
Tinea corporis is most caused by T. rubrum globally. Other causes include T. tonsurans, especially when secondary to tinea capitis infection, and M. canis, particularly with exposure to infected house pets [31, 32] (Table 3).
Table 3.
Predominant organisms responsible for tinea infections worldwide
| Type of tinea infection | Region | Species |
|---|---|---|
| Tinea corporis, tinea cruris, and tinea faciei | Americas | T. rubrum, T. tonsurans, M. canis, E. floccosum |
| Europe | T. rubrum, T. tonsurans, M. canis | |
| Africa | T. rubrum, T. mentagrophytes, M. canis, T. soudanense, T. violaceum, M. audouinii, Nannizzia (Microsporum) gypsea, E. floccosum | |
| Middle East | M. canis, T. violaceum, T. rubrum, | |
| Far East and Southeast Asia | T. mentagrophytes, T. interdigitale, T. rubrum, T. concentricum, E. floccosum | |
| Indian subcontinent | T. rubrum, T. verrucosum, T. mentagrophytes, T. indotineae | |
| Tinea capitis | Americas | T. tonsurans, M. canis |
| Europe | M. canis, T. tonsurans, T. soudanense, T. violaceum, M. audouinii | |
| Africa | T. violaceum, T. soudanense, M. audouinii | |
| Middle East | T. violaceum, M. canis, T. tonsurans | |
| Far East and Southeast Asia | T. violaceum, T. schoenleinii, M. canis, T. tonsurans | |
| Indian subcontinent | T. violaceum, M. audouinii, T. schoenleinii, T. tonsurans | |
| Tinea barbae | Worldwide | T. verrucosum, T. mentagrophytes |
| Tinea pedis | Americas | T. rubrum, T. mentagrophytes, T. interdigitale, E. floccosum |
| Europe | T. rubrum, T. interdigitale | |
| Africa | T. rubrum, T. mentagrophytes, T. interdigitale, T. violaceum | |
| Middle East | T. rubrum, T. mentagrophytes, T. interdigitale | |
| Far East and Southeast Asia | T. rubrum, T. mentagrophytes, T. interdigitale | |
| Indian subcontinent | T. rubrum, T. mentagrophytes, T. interdigitale |
The most common cause of tinea cruris worldwide is T. rubrum. T. mentagrophytes and T. interdigitale are becoming more common infectious etiologies globally, particularly in India [33–35]. Tinea cruris more commonly affects male adolescents and young adult men [36] (Table 3).
2.2. Risk Factors, Clinical Features, and Differential Diagnosis
Risk factors, clinical features, and differential diagnosis of tinea corporis and cruris are listed in Tables 2, 4, and 5, respectively (Figs. 1, 2).
Table 4.
Clinical features
| Type of tinea infection | Clinical features |
|---|---|
| Tinea corporis | Annular, erythematous, and pruritic patches or plaques with scaly and active raised borders, and central clearing as the infection advances [36, 37] Pustules may appear along the border of the patch or plaque Multiple areas can be affected with coalescing plaques |
| Tinea cruris | Annular, erythematous, and pruritic patches or plaques with scaly and active raised borders, and central clearing as the infection advances [36, 37] Always involves the crural fold Infection may spread to the thighs, genital, pubic, perineal, and perianal skin Penis and scrotum in men and labia majora in women are commonly spared [249, 250] Often presents with pruritus Pain may be a prominent feature if maceration or a secondary bacterial infection are present [250, 251] |
| Tinea capitis, endothrix | A type of non-inflammatory tinea capitis Characterized microscopically by arthroconidia within the hair shaft Typically caused by T. tonsurans, T. violaceum, and/or T. soudanese infections. Hair breakage occurs at the level of the scalp, resulting in the appearance of ‘black dots’, which are representative of the broken, distal ends of hairs Does not fluoresce under Wood’s lamp [6, 75] |
| Tinea capitis, ectothrix | A type of non-inflammatory tinea capitis characterized microscopically by arthroconidia outside of the hair shaft, occurring at the mid-follicular level. Hyphae then grow towards the bulb of the hair Typically caused by T. verrucosum, M. canis, M. audouinii, Nannizzia nana, and N. gypsea Often present as scaly patch(es) with accompanying inflammation Hair loss occurs, with breakage of the hair above the level of the scalp by at least 2–3 mm Does fluoresce under Wood’s lamp [6, 74] |
| Tinea capitis, favus | A type of inflammatory tinea capitis characterized microscopically by hyphae and air spaces within hair shafts Almost uniquely caused by T. schoenleinii Begins as erythema surrounding a hair follicle, that progresses to the classic presentation of a scutulum lesion The scutulum (plural scutula) is a concave yellow crust containing fungal hyphae, neutrophils, and epidermal cells that appears on the scalp and cover areas of severe alopecia. Scutula may coalesce to form plaques, which may lead to secondary bacterial infections [94, 252] If left untreated, inflammation may lead to scarring [252] |
| Tinea capitis, kerion | An inflammatory-type presentation of tinea capitis, most often caused by zoophilic dermatophytes [253] Usually presents as a solitary, painful, boggy plaque on the occipital scalp Begins as a folliculitis and a scaly lesion containing short hairs. It then progresses to an erythematous, tender, inflamed boggy plaque, that is covered with pustules producing a copious purulent discharge [254] Patients often experience fever and cervical lymphadenopathy Major and minor features: Major features: tenderness to palpation, alopecia within the lesion, numerous pustules and purulent drainage, and scaling Minor features: a dermatophytid reaction, a type of secondary immunologic reaction caused by dermatophytosis resulting in eczematous scaly patches or plaques at a site distinct from the main infection [255], regional lymphadenopathy (namely cervical), short hairs on dermoscopy, boggy plaques, clear demarcation of borders, overlying erythema, and pruritus [253] |
| Tinea pedis, interdigital | Mainly caused by T. rubrum May present with scaling, erythema, maceration, or fissuring, particularly in the fourth interdigital space [112] Generally spares the dorsal foot, but may spread to adjacent plantar surfaces Typically pruritic and may be foul smelling [112] Further subcategorized into dermatophytosis simplex, a dry phenotype with mild peeling scale, and wet tinea pedis, which presents with wet, macerated interdigital spaces [106] |
| Tinea pedis, hyperkeratotic (‘moccasin-type’) | Typically caused by T. rubrum Named for its distribution Entire plantar surfaces and lateral feet are typically involved bilaterally, while the dorsal feet remain clear [112] Presents on a spectrum, from slight scaling to diffuse hyperkeratosis, with chronic erythema of the plantar surface Occasionally, thick scales and fissures develop along with papules along the line of erythema of the foot [106] |
| Tinea pedis, vesiculobullous (inflammatory) | More commonly caused by T. mentagrophytes Typically involves the medial foot, foot arches, and the sides of the toes Characterized by tense vesicles, pustules, or bullae May be accompanied by burning pain and intense pruritus that may impact ambulation While bullae often contain clear fluid, purulence may ensue with bacterial superinfection, particularly with Staphylococcus aureus or group A Streptococcus infection [106, 112] |
| Tinea pedis, rare ulcerative form | Rare Most often caused by T. interdigitale Presents with ulcers and vesicular lesions that spread rapidly and are prone to bacterial superinfection [112] There may be maceration involving the interdigital spaces and the plantar surface of the foot [112] Immunocompromised patients are at greatest risk [256] |
| Tinea manuum | Typically presents as scaling and erythema of the palm, dorsum, and/or interdigital spaces of one or both hands [257] May resemble tinea corporis with an erythematous plaque and an active border [134] 80% of patients with tinea manuum have co-occurring tinea pedis [2, 239] but tinea manuum may also present independently Clinical features often similar to those of moccasin-type tinea pedis [112] Two feet-one hand syndrome describes involvement of unilateral tinea manuum and bilateral tinea pedis, often with co-occurring onychomycosis, a characteristic distribution that likely results from direct hand-to-foot contact while scratching affected feet [239, 258–260] |
| Tinea barbae, noninflammatory | Affects the hair and hair follicles of beards and mustaches in adult and adolescent males Caused by anthropophilic (humans as primary host) organisms, such as T. tonsurans and T. rubrum Flat, scaly plaques with central clearing, vesicles and pustules may be present at the active border [261] May be difficult to distinguish from tinea faciei [261] |
| Tinea barbae, inflammatory | Affects the hair and hair follicles of beards and mustaches in adult and adolescent males Caused by zoophilic (animals as primary host) organisms, such as T. mentagrophytes and T. verrucosum [262] Kerion lesion develops, with erythema, boggy, tender, weeping nodule or plaque. Pustules and draining sinuses may be present [263, 264] Hair easily plucked and appears brittle Hair loss occurs within the infected areas Pain and pruritus usually mild or non-existent Usually unilateral Constitutional symptoms, including fever, may be present Superinfection of kerion with bacterial organisms may occur May result in alopecia and scar formation if left untreated [262] |
| Tinea faciei | The most frequently misdiagnosed superficial fungal infection [265] Affects the glabrous skin of the face Erythematous, annular plaques with scaly borders [70] Begins with scaly papules that extend outward in a ring, with the central area becoming hypoor hyper-pigmented [70, 265] Pustules may be present [265] |
| Tinea incognito | Classically refers to tinea corporis, cruris, or faciei modified by a topical steroid cream applied due to misdiagnosis of dermatophyte infection as another dermatosis, commonly eczema or psoriasis [104, 105, 266] Also known as ‘steroid-modified tinea’ May also be caused by other anti-inflammatory creams, such as tacrolimus ointment, or systemic glucocorticoids [104] Raised margins and scale typically associated with dermatophytosis may be subtle, making diagnosis challenging May present with inflammatory papules and pustules [267] Affected area may become extensive with unusually shaped borders due to suppression of the local immune response and allowing the fungus to grow [267] Secondary changes caused by long-term steroid use may be present, such as skin atrophy, purpura, or telangiectasia |
Table 5.
Differential diagnosis
| Type of dermatophytosis | Differential diagnosis |
|---|---|
| Tinea corporis | Subacute cutaneous lupus erythematosus Granuloma annulare Erythema annulare centrifugum Nummular eczema Tinea versicolor Cutaneous candidiasis Pityriasis rosea Allergic contact dermatitis Irritant contact dermatitis Atopic dermatitis Seborrheic dermatitis Psoriasis Impetigo Lyme disease |
| Tinea cruris | Candidal intertrigo Seborrheic dermatitis Inverse psoriasis Erythrasma Allergic contact dermatitis Irritant contact dermatitis Lichen simplex chronicus |
| Tinea capitis | Alopecia areata Trichotillomania Seborrheic dermatitis Atopic dermatitis Allergic contact dermatitis Irritant contact dermatitis Psoriasis Traction alopecia Systemic lupus erythematous Psoriasis Pityriasis amiantacea Bacterial folliculitis Sterile folliculitis Pyoderma |
| Tinea pedis | Erythrasma Interdigital Candida infection Atopic dermatitis Allergic contact dermatitis Irritant contact dermatitis Palmoplantar eczema Dyshidrotic eczema Palmoplantar psoriasis Pitted keratolysis Juvenile plantar dermatosis Keratolysis exfoliative Palmoplantar keratoderma Palmoplantar pustulosis Scabies Gram negative toe web infection Plantar psoriasis Friction blisters Autoimmune blistering disorders Pityriasis rubra pilaris |
| Tinea manuum | Psoriasis Allergic contact dermatitis Irritant contact dermatitis Atopic hand dermatitis Occupational dermatitis Dyshidrotic eczema Keratolysis exfoliative |
| Tinea barbae | Bacterial folliculitis Perioral dermatitis Pseudofolliculitis barbae Contact dermatitis Herpes simplex Sporotrichosis Acne vulgaris Acne rosacea |
| Tinea faciei | Atopic dermatitis Seborrheic dermatitis Allergic contact dermatitis Irritant contact dermatitis Psoriasis Rosacea Actinic keratosis Perioral dermatitis Cutaneous lupus erythematosus Polymorphous light eruption Nummular eczema Sarcoidosis Granuloma annulare |
Fig. 1.
Trichophyton tonsurans tinea corporis on the ventral forearm (Dr. Avrom Caplan)
Fig. 2.
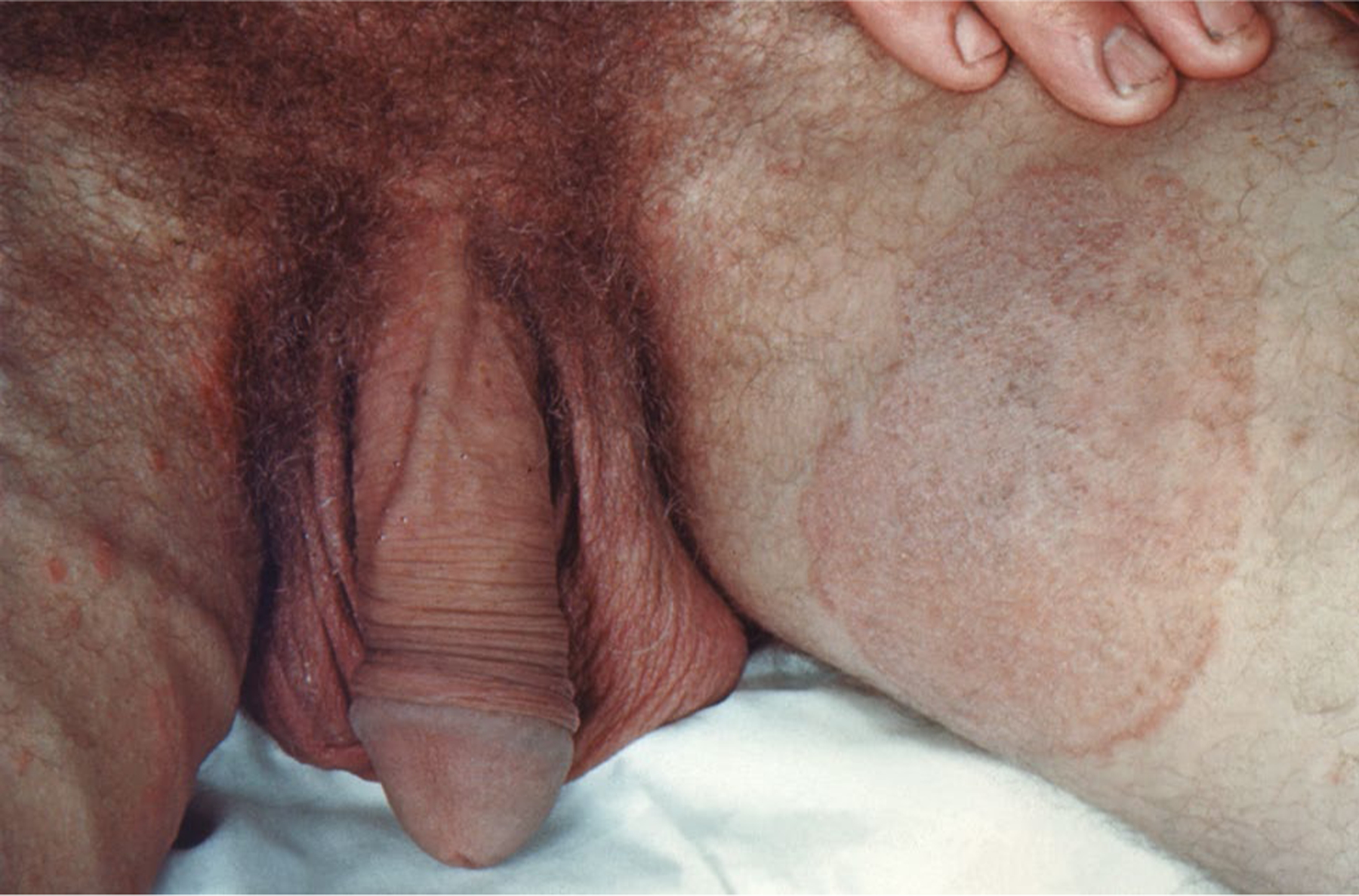
Anterior groin of a male patient with Epidermophyton floccosum tinea cruris (CDC/Dr. Lucille K. Georg)
2.3. Diagnostic Methods
The most rapid and cost-effective diagnostic technique for tinea corporis and cruris is direct microscopic examination with 10–15% potassium hydroxide (KOH) preparation using skin scrapings of the affected area(s) showing segmented hyphae [37]. Taking abundant scrapings from the active edge of a plaque or patch increases sensitivity. A fungal culture of skin scrapings on Mycosel agar media is considered the diagnostic gold standard since it can identify the causative organism; however, turnaround time is typically several weeks, with a significant false negative rate.
Polymerase chain reaction (PCR) may rapidly detect the presence of fungal DNA and identify the fungal species [38–43]; however, PCR for dermatophytes is not performed by all laboratories, and widespread adoption has been slow in clinical practice [44, 45].
Reflectance confocal microscopy (RCM) is a non-invasive technique that is being explored for cutaneous fungal infection diagnosis [46–48]. While results are immediate, limitations include difficulty positioning the probe in certain anatomic areas, expensive equipment, and required training.
Dermoscopy is an emerging adjunctive diagnostic technique for dermatophyte infections [49–52]. Useful dermatoscopic features distinguishing tinea corporis from other dermatologic conditions include presence of peripheral dotted vessels, white ‘moth-eaten’ scale with peripheral distribution and outward peeling direction [50].
2.4. Treatment and Management
Treatment for tinea corporis and cruris is always indicated to prevent infection spread to self and others and prevent secondary bacterial infections. In general, mild or localized infection may be treated once to twice daily with topical monotherapy antifungals, including azoles, allylamines, ciclopirox, butenafine, and tolnaftate [53–56], while more disseminated disease is treated with oral antifungals. The American Academy of Pediatrics (AAP) 2021–2024 Report of the Committee on Infectious Diseases recommended application of topical antifungals directly to lesions and 1–2 cm beyond the lesion border. Although clinical resolution may be apparent after 2 weeks of therapy, treatment is recommended for 4–6 weeks. If there is no clinical improvement after 2 weeks of topical therapy, an alternate diagnosis or systemic therapy may be considered [56].
A Cochrane Database Systematic Review of 129 studies involving 18,086 participants with tinea corporis or cruris supported the use of topical terbinafine and naftifine 1%, and clotrimazole 1%, based on clinical and/or mycological cure rates versus placebo. Azoles and benzylamines had similar mycological cures rates (risk ratio [RR] 1.01, 95% confidence interval [CI] 0.94–1.07) [57, 58]. In a mixedtreatment comparison meta-analysis of 65 articles including 14 topical antifungal treatments for tinea corporis, cruris, and pedis, mycological cure at treatment completion was not significantly different among the topical antifungals studied [59]. However, butenafine, naftifine, and terbinafine, a benzylamine and allylamines, respectively, were the most effective among 13 therapies for sustained cure, defined as maintaining cure from infection for ≥ 14 days, compared with azoles. Cost consideration is necessary in most cases since there are limited efficacy data to support one class over another. Therefore, azoles, which are cheaper on average, may be used as first-line therapy, followed by allylamines for first-line treatment failures [58]. Topical antifungals indicated for tinea corporis and cruris are listed in Table 6.
Table 6.
Topical antifungals
| Drugs | Indication | Dose frequency | FDA age approvals |
|---|---|---|---|
| Allylamines | |||
| Naftifine 2% cream | Tinea corporis Tinea cruris Tinea pedis |
Once daily for 2 weeks | Tinea corporis: age ≥2 years Tinea cruris and tinea pedis: age ≥12 years |
| Terbinafine 1% cream | Tinea corporis Tinea cruris Tinea pedis |
Tinea corporis/cruris: once daily for 1 week Tinea pedis (interdigital): twice daily for 1 week Tinea pedis (moccasin): twice daily for 2 weeks |
Tinea corporis, cruris, and pedis: age ≥12 years |
| Azoles and imidazoles | |||
| Clotrimazole 1% cream or solution | Tinea corporis Tinea cruris Tinea pedis |
Tinea corporis and tinea pedis: twice daily for 4 weeks Tinea cruris: twice daily for 2 weeks |
Age ≥ 2 years |
| Econazole 1% cream | Tinea corporis Tinea cruris Tinea pedis |
Tinea corporis/cruris: once daily for 2 weeks Tinea pedis: once daily for 1 month |
Age ≥ 2 years |
| Ketoconazole 2% cream | Tinea corporis Tinea cruris Tinea pedis |
Tinea corporis/tinea cruris: once daily for 2 weeks Tinea pedis: once daily for 6 weeks |
Age ≥ 2 years |
| Luliconazole 1% cream | Tinea corporis Tinea cruris Tinea pedis |
Tinea corporis/cruris: once daily for 1 week Interdigital tinea pedis: once daily for 2 weeks |
Tinea corporis: age ≥ 2 years Tinea cruris and tinea pedis: age ≥ 12 years Age ≥2 years |
| Miconazole 2% cream | Tinea corporis Tinea cruris Tinea pedis |
Tinea corporis: twice daily for 4 weeks Tinea cruris: twice daily for 2 weeks Tinea pedis: twice daily for 4 weeks |
Age ≥ 2 years |
| Oxiconazole 1% cream | Tinea corporis Tinea cruris Tinea pedis |
Tinea corporis/cruris, tinea pedis: once to twice daily for 2 weeks | Age ≥ 2 years |
| Sertaconazole 2% cream | Tinea pedis | Twice daily for 4 weeks | Age ≥ 2 years |
| Sulconazole 1% cream or solution | Tinea corporis Tinea cruris Tinea pedis |
Tinea corporis/cruris: once daily for 3 weeks Tinea pedis: twice daily for 4 weeks |
The safety and effectiveness in pediatric patients have not been established |
|
Benzylamine
|
|||
| Butenafine 1% cream | Tinea corporis Tinea cruris Tinea pedis |
Tinea corporis/cruris: once daily for 2 weeks Interdigital tinea pedis: twice daily for 1 week or once daily 4 weeks |
Age ≥ 12 years |
| Other | |||
| Ciclopirox 0.77% cream | Tinea corporis Tinea cruris Tinea pedis |
Twice daily for 4 weeks | Age ≥ 10 years |
| Ciclopirox 0.77% gel | Tinea corporis Tinea cruris Tinea pedis |
Twice daily for 4 weeks | Age ≥ 16 years |
| Tolnaftate 1% cream or solution | Tinea corporis Tinea cruris Tinea pedis |
Twice daily for 4 weeks | Age ≥ 2 years |
Position statements released in 2019 by the Canadian Pediatric Society recommend that adjunctive topical corticosteroid therapy be avoided due to the risk of skin atrophy [60]. Topical corticosteroid misuse is hypothesized to contribute to antifungal resistance [61, 62]. Combination topical antifungal-corticosteroid products should be avoided. Antifungal monotherapy is effective and may decrease inflammation and pruritus.
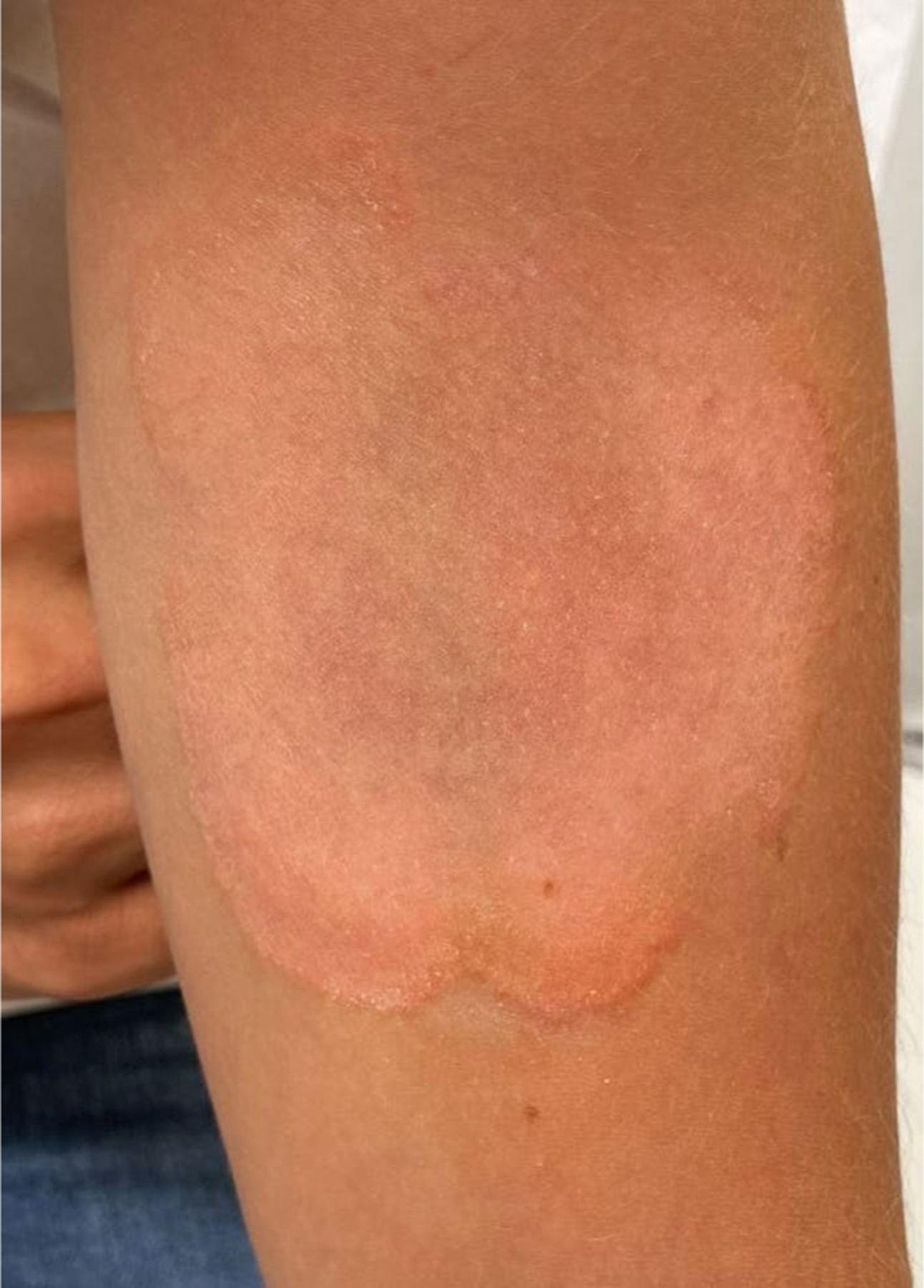
For widespread infection involving multiple areas, disease refractory to topical therapy, or tinea corporis with vellus hair involvement (Fig. 3), oral treatment with either terbinafine or itraconazole is recommended [52]. Griseofulvin and fluconazole are second-line treatments and require longer treatment courses. With partial response, therapy is continued and patient lifestyle factors, including hygiene, which may contribute to incomplete cure are assessed. With no response to treatment, a change to another oral antifungal (i.e. terbinafine to itraconazole) is indicated. Follow-up for at least 4 weeks after clinical cure is recommended to prevent recurrence.
Fig. 3.
Tinea corporis on the lower leg (species unknown) with co-occurring fungal folliculitis (Dr. Avrom Caplan)
Terbinafine is the recommended first-line therapy in treatment-naïve patients with tinea corporis or cruris [53]. In a randomized controlled trial of 50 patients with tinea corporis treated with 500 mg/day of griseofulvin versus 500 mg/day of terbinafine for 6 weeks, successful treatment rates, defined as either clinical and mycological cure or mycological cure with only slight residual clinical signs of infection, were 73% versus 87%, respectively (p value not reported) [63]. In a study of 64 patients diagnosed with tinea and/or tinea cruris receiving terbinafine 250 mg/day versus griseofulvin 500 mg/day for 2 weeks, mycological cure was 87.1% versus 54.8% at 6 weeks follow up (p < 0.05) [64]. In a prospective trial of 60 patients with tinea corporis and/or cruris randomized to terbinafine 250 mg/day versus terbinafine 500 mg/day, complete cures were similar (20% vs. 33.3%; p = 0.82) [65].
In a randomized controlled trial of 78 patients with tinea corporis or cruris treated with itraconazole 100 mg/day or griseofulvin 500 mg/day for 15 days, clinical (72% vs. 51%; p < 0.03) and mycological cure (87% vs. 57%; p < 0.03) were higher for itraconazole versus griseofulvin [66]. A double-blind, randomized controlled trial of 40 patients with clinically and mycologically confirmed tinea corporis or cruris comparing itraconazole 100 mg/day versus griseofulvin 500 mg/day for 15 days showed no difference in complete cure at treatment completion (22.2% vs. 23.8%; p = 1.000) nor at 15-day post-treatment follow-up (55.6% vs. 52.4%; p = 0.672) [67]. Physicians can consider a minimum dose of itraconazole 200 mg to treat tinea corporis and/or cruris, as underdosing may contribute to resistance development.
Recommended oral therapy dosing for tinea corporis and cruris is listed in Table 7 [64, 68, 69].
Table 7.
Oral antifungals
| Drug | Preparation | Indications | Adult dose | Pediatric dose | Duration | Example adverse effects | Laboratory monitoring |
|---|---|---|---|---|---|---|---|
| Griseofulvin microsize | Tablet, suspension | First-line therapy for Trichophyton tinea capitis First-line therapy for Microsporum tinea capitis Second-line therapy for refractory tinea corporis/cruris infection or with vellus hair involvement |
Tinea capitisa Optimal treatment regimen unclear Tinea corporis/cruris 500–1000 mg/day |
Tinea capitis 20–25 mg/kg/day Tinea corporis/cruris 10–20 mg/kg/day |
6–12 weeks 2–4 weeks |
Gastrointestinal distress Headache Hepatotoxicity Granulocytopenia Leukopenia |
Not necessary before initiation of therapy If therapy extends past 8 weeks or multiple courses given, check liver function panel and complete blood count |
| Griseofulvin ultramicrosize | Tablet | First-line therapy for Trichophyton tinea capitis First-line therapy for Microsporum tinea capitis Second-line therapy for refractory tinea corporis/cruris infection or with vellus hair involvement |
Tinea capitisa 10–15 mg/kg/day Tinea corporis/cruris 375–500 mg/day |
Tinea capitis 10–15 mg/kg/day Tinea corporis/cruris 5–15 mg/kg/day |
6–12 weeks 2–4 weeks |
Gastrointestinal distress Headache Hepatotoxicity Granulocytopenia Leukopenia |
Not necessary before initiation of therapy If treatment extends past 8 weeks or multiple courses given, check liver function panel and complete blood count |
| Terbinafine tablets | Tablet | First-line therapy for Trichophyton tinea capitis Second-line therapy for Microsporum tinea capitis First-line therapy for refractory tinea corporis/cruris infection or with vellus hair involvement First-line therapy for refractory tinea pedis |
Tinea capitisa 250 mg/day Tinea corporis/cruris 250 mg/day Tinea pedis 250 mg/day |
Tinea capitis 10–20 kg: 62.5 mg daily 20–40 kg: 125 mg daily ≥40 kg: 250 mg daily Tinea corporis/cruris 10–20 kg: 62.5 mg daily 20–40 kg: 125 mg daily ≥40 kg: 250 mg daily Tinea pedis 10–20 kg: 62.5 mg daily 20–40 kg: 125 mg daily ≥ 40 kg: 250 mg daily |
4–6 weeks (Trichopyton); 8–12 weeks (Microsporum) 1–2 weeks 2 weeks |
Gastrointestinal distress Headache Taste disturbances Hepatotoxicity Pancytopenia |
Liver function panel prior to therapy initiation If treatment extends past 6 weeks, check liver function panel and complete blood count |
| Fluconazole | Tablet, suspension | Second-line therapy for Trichophyton tinea capitis Second-line therapy for Microsporum tinea capitis Second-line therapy for refractory tinea corporis/cruris infection or with vellus hair involvement Second-line therapy for refractory tinea pedis |
Tinea capitisa 6 mg/kg/day− Tinea corporis/cruris 150–200 mg once weekly Tinea pedis 150 mg once weekly |
Tinea capitis 6 mg/kg/day Tinea corporis/cruris 6 mg/kg once weekly Tinea pedis 6 mg/kg once weekly |
3–6 weeks 2–4 weeks 2–6 weeks |
Gastrointestinal distress Headache Hepatotoxicity Prolonged QT interval |
None |
| Itraconazole | Capsule, solution | Second-line therapy for Trichophyton tinea capitis Second-line therapy for Microsporum tinea capitis Alternative first-line therapy for refractory tinea corporis/cruris infection or with vellus hair involvement with contraindication to terbinafine or terbinafine treatment failure Second-line therapy for refractory tinea pedis |
Tinea capitisa 5 mg/kg/day Tinea corporis/cruris 200 mg/day Tinea pedis 200 mg twice daily |
Tinea capitis 3–5 mg/kg/day Tinea corporis/cruris 3–5 mg/kg/day Tinea pedis 3–5 mg/kg/day |
2–6 weeks 1 week (longer courses required in some cases) 1 week |
Gastrointestinal distress Headache Dizziness Hepatotoxicity Cardiotoxicity (triad of hypertension, hypokalemia, peripheraledema) |
Liver function panel prior to therapy initiation If treatment extends past 4 weeks, check liver function panel |
Optimal treatment regimens for tinea capitis in adults are not well known
3. Tinea Capitis
3.1. Demographics and Geography
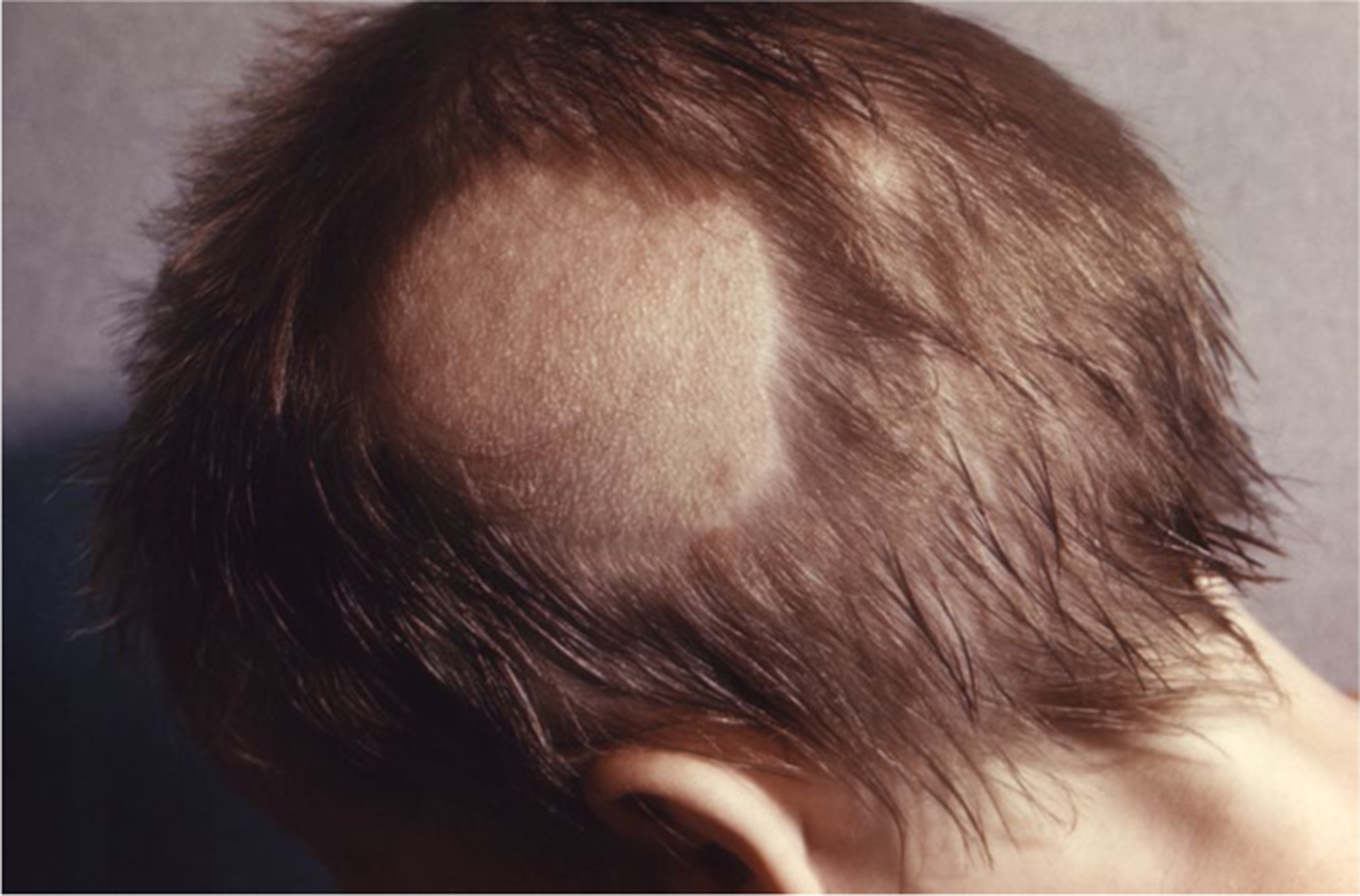
Tinea capitis primarily affects children, particularly in the prepubertal years [70, 71]. Black children are most frequently affected in the US (Fig. 4). In a prospective, cross-sectional surveillance study of 10,514 kindergarten fifth-grade children, Black children had the highest rate of T. tonsurans tinea capitis infection (12.9%) versus Hispanic (1.6%) and White children (1.1%) [p < 0.01] [25]. Although uncommon, tinea capitis may affect adults [72–74].
Fig. 4.
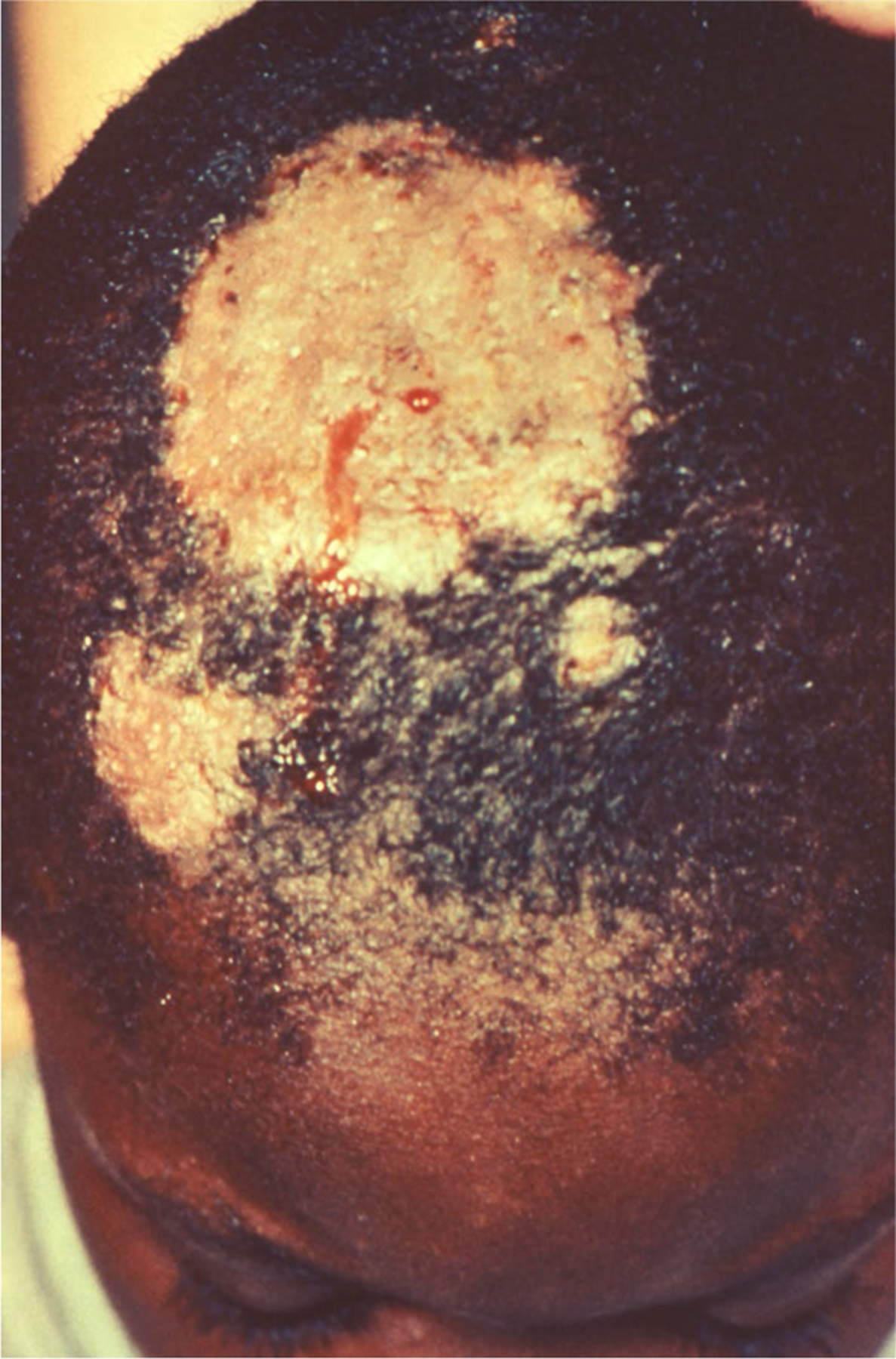
Trichophyton mentagrophytes tinea capitis in the scalp of a Black child (CDC)
Common causative organisms of tinea capitis are listed in Table 3. In general, species within Trichophyton and Microsporum genera cause tinea capitis, including Microsporum audouinii, Trichopython violaceum, or Trichophyton tonsurans. In the US, T. tonsurans is the most common causative species, followed by M. canis (Fig. 5) [23].
Fig. 5.
Microsporum tinea capitis (CDC)
3.2. Risk Factors, Clinical Features, and Differential Diagnosis
The hair shaft infection presents in one of three distinct forms: endothrix, ectothrix, or favus (Fig. 6), which drive the clinical appearance. Endothrix and ectothrix infections are the most common presentations [8, 75]. Kerion is an inflammatory presentation of tinea capitis. Risk factors, clinical features, and differential diagnosis are described in Tables 2, 4, and 5, respectively.
Fig. 6.
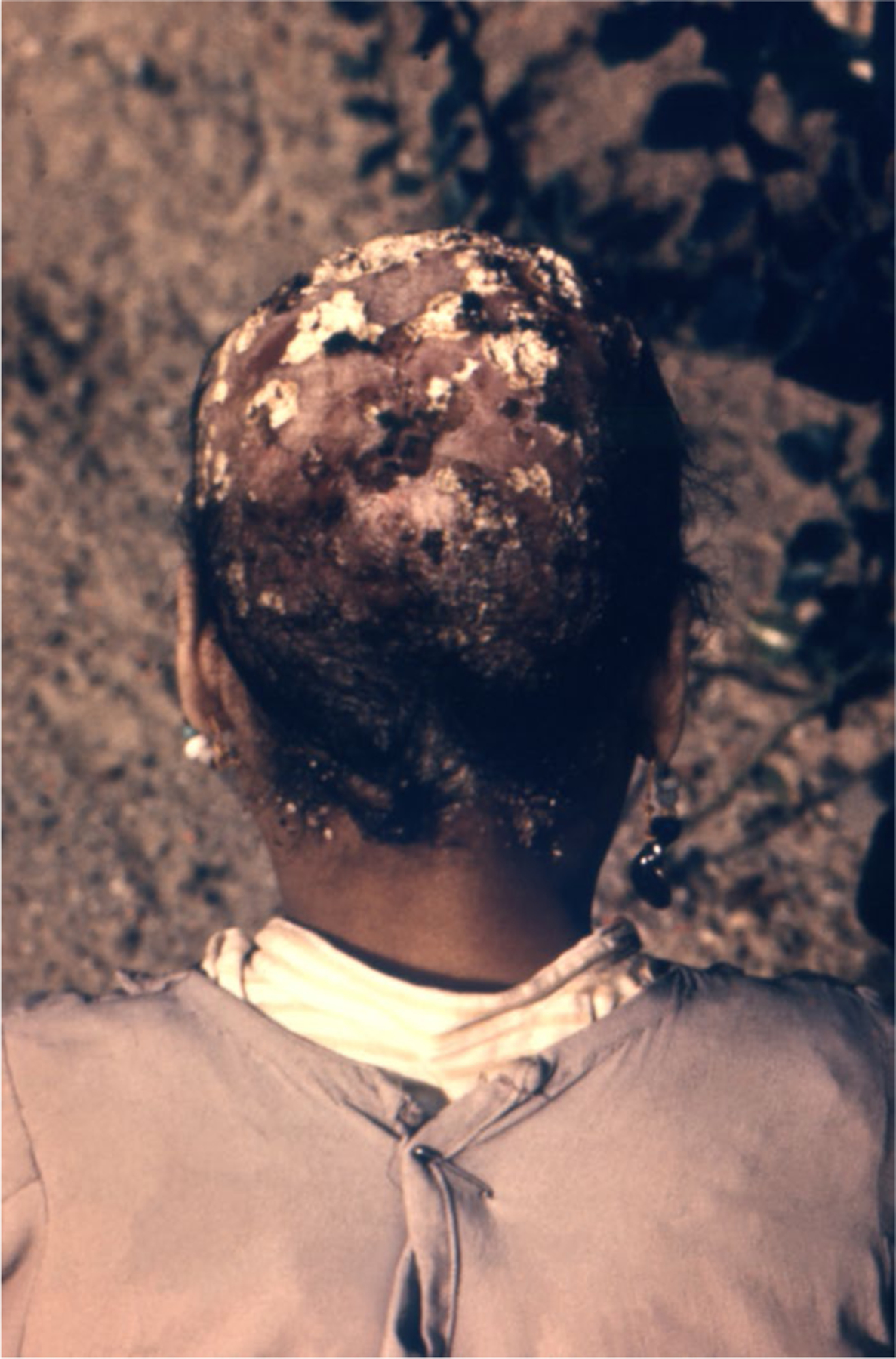
Trichophyton schoenleinii favus accompanied by alopecia (CDC/Dr. Libero Ajello)
3.3. Diagnostic Methods
Physical examination identifies signs of tinea capitis, such as black dots associated with endothrix tinea capitis. Ectothrix infections caused by Microsporum species or T. schoenleinii may fluoresce a green/yellow color or blue/white color, respectively, while endothrix infections do not exhibit fluorescence Wood’s lamp; however, Wood’s lamp examination in not reliable for definitive diagnosis [75].
Fungal culture using Mycosel agar culture remains the gold standard of diagnosis for tinea capitis for species identification. Samples are collected using a sterile toothbrush, moistened cotton swab, or by scraping with a no. 15 blade or glass slide over the affected area. Infected hairs often break off and may be sent along with scrapings [76–78]; however, the results may take 2–4 weeks, which may significantly delay therapy initiation and increase transmission risk [8, 75, 76, 79]. Intense inflammation associated with kerion may cause false negative results and, in general scrapings, should be taken prior to initiating antifungal treatment. A KOH preparation may be used to screen for tinea capitis while fungal cultures are pending. KOH is applied to a glass slide and the proximal ends of a hair shaft are examined micro-scopically. The test is positive if fungal spores are found within or outside of the hair shaft (endothrix and ectothrix, respectively).
PCR may be used for tinea capitis diagnosis, although is rarely employed in clinical practice. In a descriptive cross-sectional study of 115 scalp scrapings and hair fragments from children aged 1–16 years diagnosed with tinea capitis, PCR-ELISA had 84% sensitivity and 85% specificity compared with fungal culture used as the gold standard [80]. In a cross-sectional, descriptive study on 129 hair samples from patients clinically diagnosed with tinea capitis, real-time PCR had 89.3% sensitivity and 75.3% specificity compared with fungal culture (p < 0.05) [81]. A PCR test for M. canis identification achieved 100% sensitivity and specificity in a study of 130 clinical isolates of dermatophytes, 10 yeast/mold isolates, 12 hair and skin samples from animals with/without M. canis infection, and 35 patient specimens, 7 of which were positive for M. canis [82]. M. canis is less susceptible to terbinafine than Trichophyton species, requiring prompt therapy to avoid long-term sequelae, including permanent hair loss.
RCM has been used for tinea capitis diagnosis, whereby hyphae appear linear, with high reflectivity and conidia near the hair shaft present as round and hyperreflective [46, 83, 84]. In one case series including four patients with tinea capitis, diagnosis was achieved in approximately 5 min per patient [46].
Trichoscopy, dermoscopy of the hair and scalp, is a useful diagnostic adjunct. Trichoscopic findings include comma hairs, corkscrew hairs, broken hairs, dystrophic hairs, and/or black dots [85–88]. In a cross-sectional study comparing trichoscopic features of 15 children with tinea capitis with 10 patients with alopecia areata, yellow dots, exclamation mark hairs, and vellus hairs differentiated alopecia areata from tinea captitis [85].
3.4. Treatment and Management
Although mycological confirmation before starting treatment is considered best practice, diagnostic methods may not be readily available or may result in significant delays to therapy initiation. The 2014 British Association of Dermatologists tinea capitis guidelines indicate that it is appropriate to begin treatment when tinea capitis is strongly suspected clinically, including in the presence of scaling or kerion [89].
First-line therapy for tinea capitis is oral antifungal therapy, as topical therapy cannot effectively penetrate the hair shaft. Although many controlled trials described herein are exclusive to pediatric patients, the same medications are used in adult patients. Pediatric treatment regimens are described in Table 7, along with commonly accepted adult dosing recommendations [90].
Griseofulvin and terbinafine are US FDA-approved tinea capitis treatments and are the most commonly used therapies [56]. The AAP 2021–2024 Report of the Committee on Infectious Diseases states that higher doses of griseofulvin than are FDA-approved may be used, and high-dose griseofulvin is the standard of care for pediatric patients with M. canis tinea capitis (griseofulvin microsize 20–25 mg/kg/day, maximum 1 g/day, or ultramicrosize 10–15 mg/kg/day, maximum 750 mg/day, for ≥ 6 weeks and until clinically clear).
For tinea capitis due to Trichophyton, both griseofulvin and terbinafine are acceptable first-line therapies, but choice of agent should be tailored to speciation, if known [91]. A 2016 Cochrane review found that 4 weeks of terbinafine versus 8 weeks of griseofulvin treatment had similar efficacy in achieving complete cure across three studies including 328 participants with Trichophyton tinea capitis (84.2% vs. 79%; RR 1.06, 95% CI 0.98–1.15) [91]. In two randomized, investigator-blinded, active-controlled trials in children aged 4–12 years with clinically diagnosed and KOH-confirmed tinea capitis, terbinafine hydrochloride oral granules (n = 1040, 5–8 mg/kg/day) versus griseofulvin microsize (n = 509, 10–20 mg/kg/day) for 6 weeks had higher complete (45.1% vs. 39.2%; p = 0.024) and mycological cure rates (61.5% vs. 55.5%; p = 0.029) on pooled data at week 10. Terbinafine and griseofulvin had similar efficacy for clinical cure when compared with pooled data at week 10 (63.0% vs. 58.8%; p = 0.10). Terbinafine had greater efficacy for mycological, clinical, and complete cure in children with T. tonsurans tinea capitis specifically (complete cure 52.1% vs. 35.4%; p < 0.0001) [92]. For Microsporum tinea capitis, a meta-analysis of two studies including 334 participants found greater complete cure rates with 6–12 weeks of griseofulvin versus 6 weeks of terbinafine (50.9% vs. 34.7%; RR 0.68, 95% CI 0.53–0.86) [91]. If the exact species is unknown, griseofulvin is recommended as first-line therapy in treating tinea capitis [93].
Due to concerns about resistance of T. tonsurans to griseofulvin, fluconazole and itraconazole may be considered as non-FDA approved therapeutic alternatives [94] and for patients with contraindications to terbinafine and/or griseofulvin, or in patients who cannot tolerate high griseofulvin doses [95]. Fluconazole is FDA-approved for children <2 years of age, for indications other than tinea capitis. A randomized, multicenter, three-arm trial in children aged 3–12 years comparing fluconazole 6 mg/kg/day for 3 weeks followed by 3 weeks of placebo with fluconazole 6 mg/kg/day for 6 weeks and with griseofulvin 11 mg/kg/day for 6 weeks, showed no differences between mycological (44.5%, 49.6%, and 52.2%, respectively; p = 0.40) or clinical cure (87.8%, 89.8%, and 90.9%, respectively; p = 0.61) [95]. For griseofulvin, physicians could consider a dose of 20 mg/kg/day. A randomized controlled trial of 34 children and 1 adult comparing itraconazole 100 mg daily for 6 weeks with ultra-micronized griseofulvin 500 mg daily for 6 weeks for mostly M. canis tinea capitis showed complete cure 8 weeks after therapy completion (88% vs. 88%; p-value not reported) [96]. Another randomized, double-blind controlled trial of weight-based itraconazole and terbinafine each administered for 2 weeks for mostly T. violaceum tinea capitis showed complete cure rates of 85.7% and 77.8%, respectively at week 12 (p > 0.05) [97]. A multicenter, randomized, single-blinded trial of 200 patients treated with griseofulvin microsize 20 mg/kg/day for 6 weeks, terbinafine 62.5–250 mg (weight dependent) for 2–3 weeks, itraconazole 5 mg/kg/day for 2–3 weeks, or fluconazole 6 mg/kg/day for 2–3 weeks showed similar efficacy (measured as a combination of mycological cure and either clinical cure or only a few residual symptoms) among all arms 12 weeks after the start of therapy (griseofulvin: 92%, 95% CI 88.2–95.8%; terbinafine: 94%, 95% CI 90.6–97.4%; itraconazole: 86%, 95% CI 81.1–90.9%; fluconazole: 84%, 95% CI 78.8–89.2%) [98]. Fluconazole and itraconazole are second-line therapies. Dosing of oral antifungals is described in Table 7.
Antifungal shampoos may be used as adjunctive therapy by patients and their household members for tinea capitis in the first 1–2 weeks following diagnosis, to reduce transmission and possible re-infection [36]. In a randomized, double-blind controlled trial of 40 children aged 1–11 years diagnosed with tinea capitis and treated with an 8-week course of ultramicronized griseofulvin, selenium sulfide shampoo 1% and ciclopirox shampoo 1% showed equal efficacy in mycological cure 4 weeks after treatment completion when used as adjunctive treatment twice per week (week 12: 91.7% vs. 95.2%; p = 1) [99]. A study of ketoconazole 2% shampoo used as monotherapy for tinea capitis in 16 Black children aged 3–6 years, over an 8-week treatment period, showed 50% reduced viable arthroconidia numbers after 2 weeks of treatment, clinical cure in 93% of children at week 8, and complete cure in 33% of children 12 months post-treatment [100]. It is recommended that children and their families are instructed on disinfecting hair tools following a diagnosis of tinea capitis, to prevent re-infection or transmission to other household members [101].
It is recommended that patients are reassessed after 1 month of therapy to determine clinical response. If there is no response to therapy despite adherence to prescribed medications, an alternative antifungal is recommended [56].
4. Tinea Barbae/Tinea Faciei
4.1. Demographics and Geography
Table 3 lists the most common causative species of tinea barbae and faciei, by region.
4.2. Risk Factors, Clinical Features, and Differential Diagnosis
Tinea barbae may present as an inflammatory or non-inflammatory morphology, and usually involves an ectothrix infection pattern. Tinea faciei often presents similarly to tinea corporis. Risk factors, clinical features, and differential diagnosis of tinea barbae and are listed in Tables 2, 4, and 5, respectively (Figs. 7, 8).
Fig. 7.
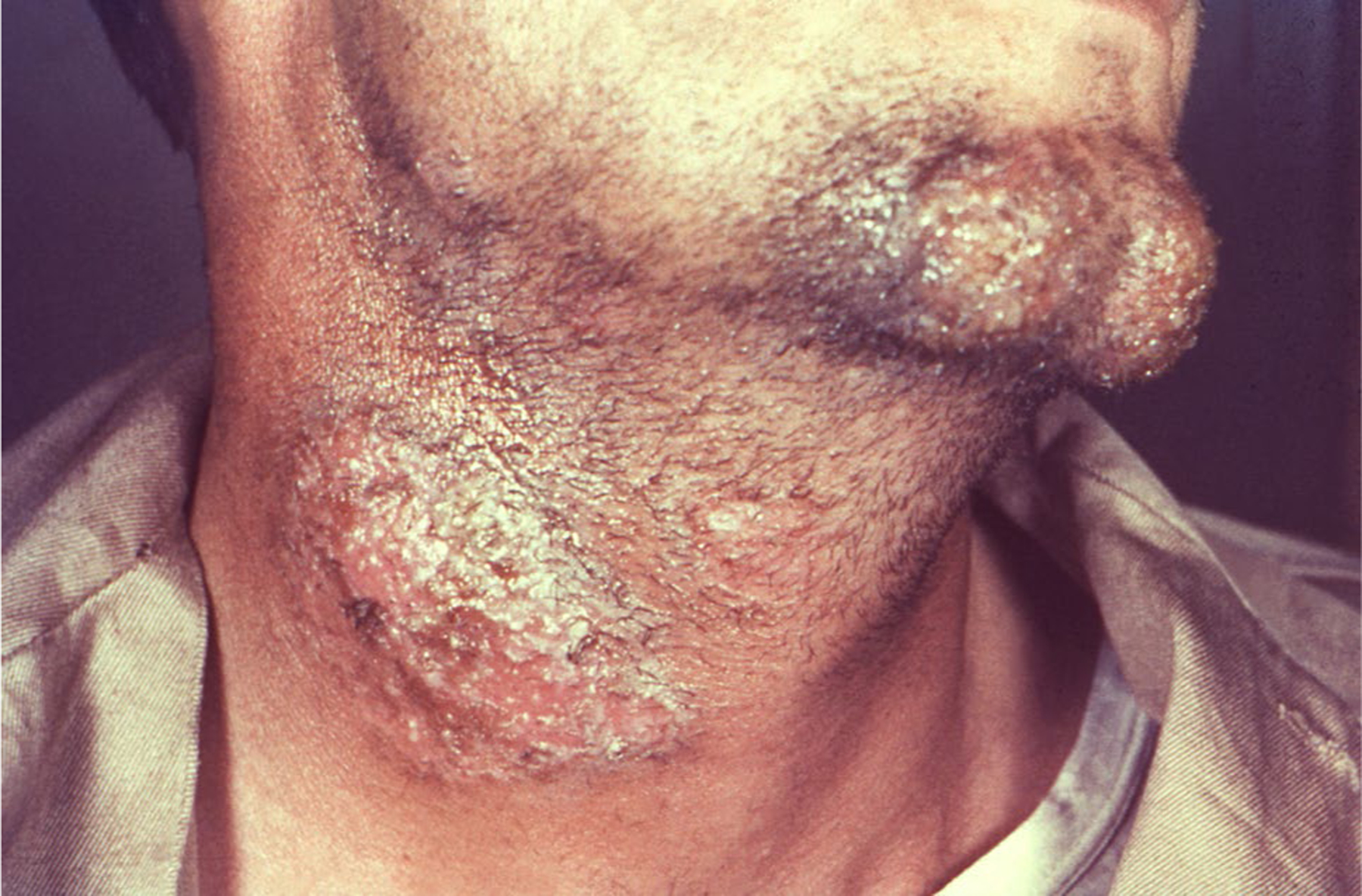
Trichophyton schoenleinii mentagrophytes tinea barbae (CDC/Dr. Lucille K. Georg)
Fig. 8.
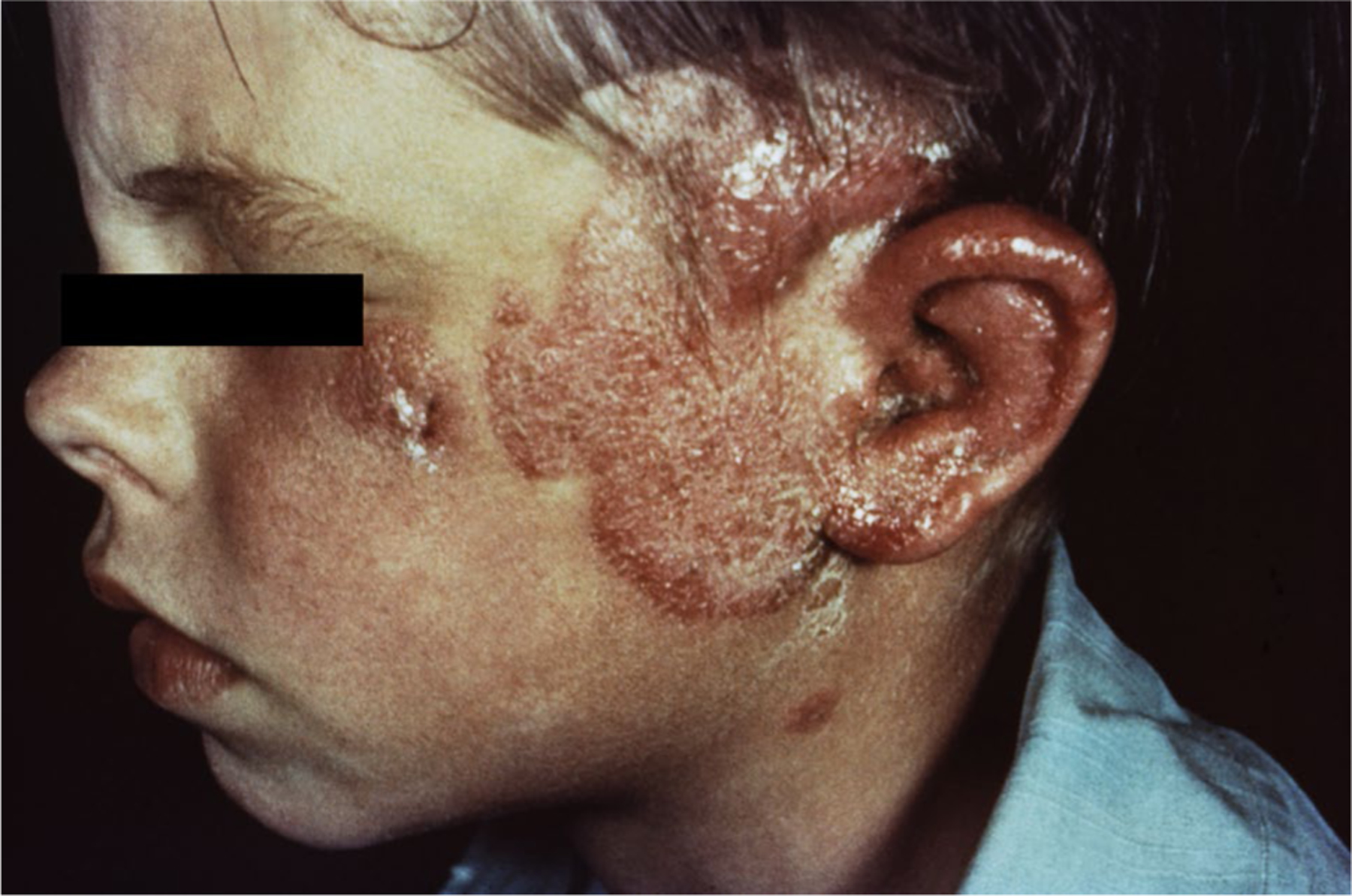
Tinea faciei of unknown species involving the ear (CDC)
4.3. Diagnostic Methods
Diagnostic methods related to tinea capitis, described in Sect. 3.3, apply to tinea barbae, whereas diagnostic methods related to tinea corporis, described in Sect. 2.3, apply to tinea faciei.
4.4. Treatment and Management
Treatment principles for tinea capitis, described in Sect. 3.4, apply to tinea barbae. Oral antifungal therapy is first-line as topical therapy cannot penetrate the hair shaft adequately. Treatment principles for tinea corporis, described in Sect. 2.4 apply to tinea faciei. Topical antifungal therapy is first-line therapy, while oral therapy is reserved for refractory or widespread involvement.
5. Tinea Incognito
5.1. Risk Factors and Clinical Features
Due to dampening of the local immune response, classic clinical features of dermatophytoses may not be present in patients with tinea incognito. Steroid-modified dermatophytosis has been described as ‘tinea pseudoimbricata’ due to its similarity in appearance to tinea imbricata caused by Trichophyton concentricum, which is endemic in parts of Southeast Asia [102], presenting with concentric erythematous rings and associated with moderate to severe pruritus (Fig. 9) [103]. Risk factors and clinical features for tinea incognito are listed in Tables 2 and 4, respectively.
Fig. 9.
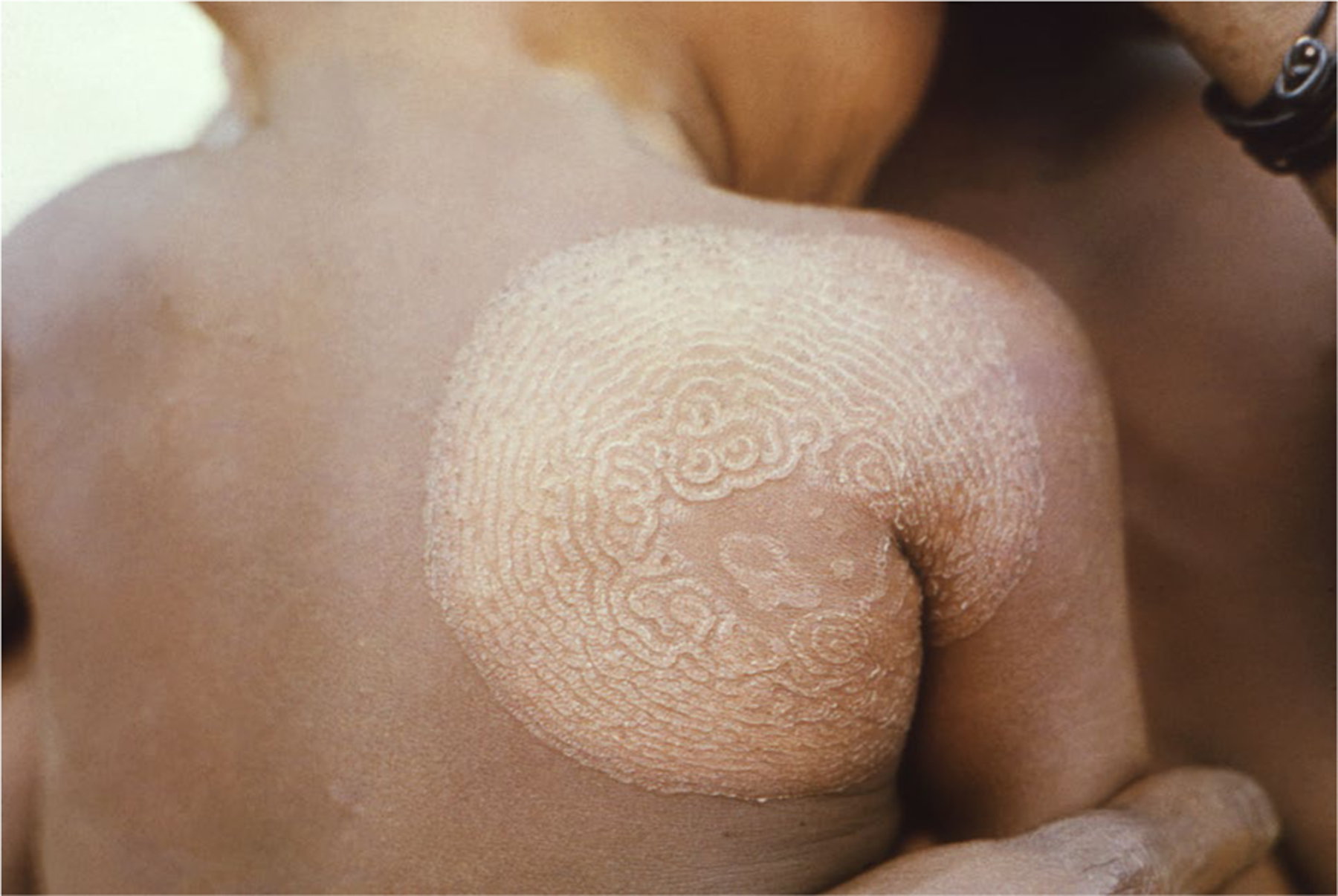
Trichophyton concentricum tinea imbricata on the back of a child in Papua New Guinea (CDC/K. Mae Lennon, Tulane Medical School; Clement Benjamin)
5.2. Diagnostic Methods
Because tinea incognito is a steroid-modified infection and does not present with classic features typically present with dermatophyte infections, diagnosis may be delayed. A high suspicion for tinea incognito is necessary if a patient describes a rash coupled with corticosteroid administration, resulting in a temporary improvement of the lesions, followed by worsening and spread [104].
Diagnostic methods of tinea incognito mirror those for tinea corporis and cruris, described in Sect. 2.3. It is recommended that diagnostic methods, including KOH preparation and fungal culture, are performed after steroid discontinuation. Dermoscopy may show black dots surrounded by a white-yellow halo, a sign of vellus hair involvement in dermatophytosis [105].
5.3. Treatment and Management
Once tinea incognito has been diagnosed, topical corticosteroids should be stopped and antifungal therapy initiated. If oral corticosteroids are identified as the culprit in cases of tinea incognito, an alternative treatment should be considered. The treatment thereafter follows that of tinea corporis, cruris, and/or faciei, described in Sect. 2.4.
6. Tinea Pedis and Tinea Manuum
6.1. Demographics and Geography
Tinea pedis typically affects post-pubescent adolescents and adults [70] and is most often caused by T. rubrum, T. interdigitale, T. mentagrophytes, and E. floccosum [106]. Common causative species are listed in Table 3. In children, T. tonsurans is the common causative organism [69]. Tinea pedis occurs more often in older adults and may be more common in males than in females [107–111]. Tinea pedis is a common problem among developed and developing countries, with a global prevalence estimate of 3–15% [109–113]. In a study of 6932 participants with tinea pedis in the All of Us database 2017–2021, higher adjusted odds of tinea pedis were observed in Black and Hispanic participants (odds ratio [OR] 1.29, 95% CI 1.20–1.38, and OR 1.38, 95% CI 1.28–1.48, respectively), individuals ≥ 75 years of age (OR 1.45, 95% CI 1.33–1.57), individuals with income <$35,000 (OR 1.09, 95% CI 1.02–1.16), and individuals with a physical disability (OR 1.56, 95% CI 1.08–1.24) [110]. Epidemiology of tinea manuum infections is far less studied than that of tinea pedis.
6.2. Risk Factors, Clinical Features, and Differential Diagnosis
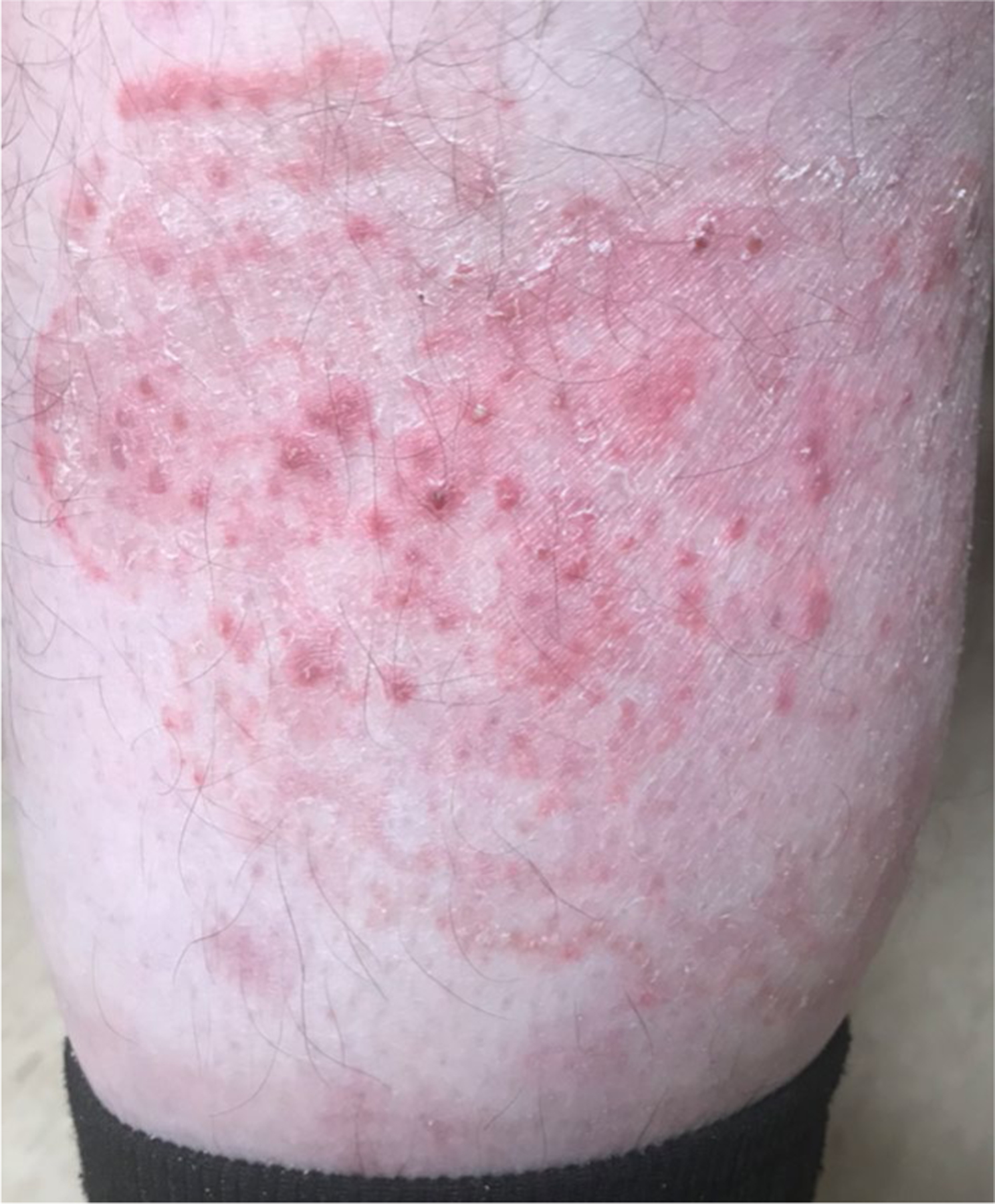
There are three major clinical presentations of tinea pedis, namely interdigital (Fig. 10), hyperkeratotic (‘moccasin-type’), and vesiculobullous (inflammatory) tinea pedis, and a rare ulcerative form. T. rubrum typically causes chronic tinea pedis, whereas T. mentagrophytes is associated with inflammatory lesions [114]. Risk factors, clinical features, and differential diagnosis for tinea pedis (Fig. 11) and manuum (Fig. 12) are listed in Tables 2, 4, and 5, respectively.
Fig. 10.
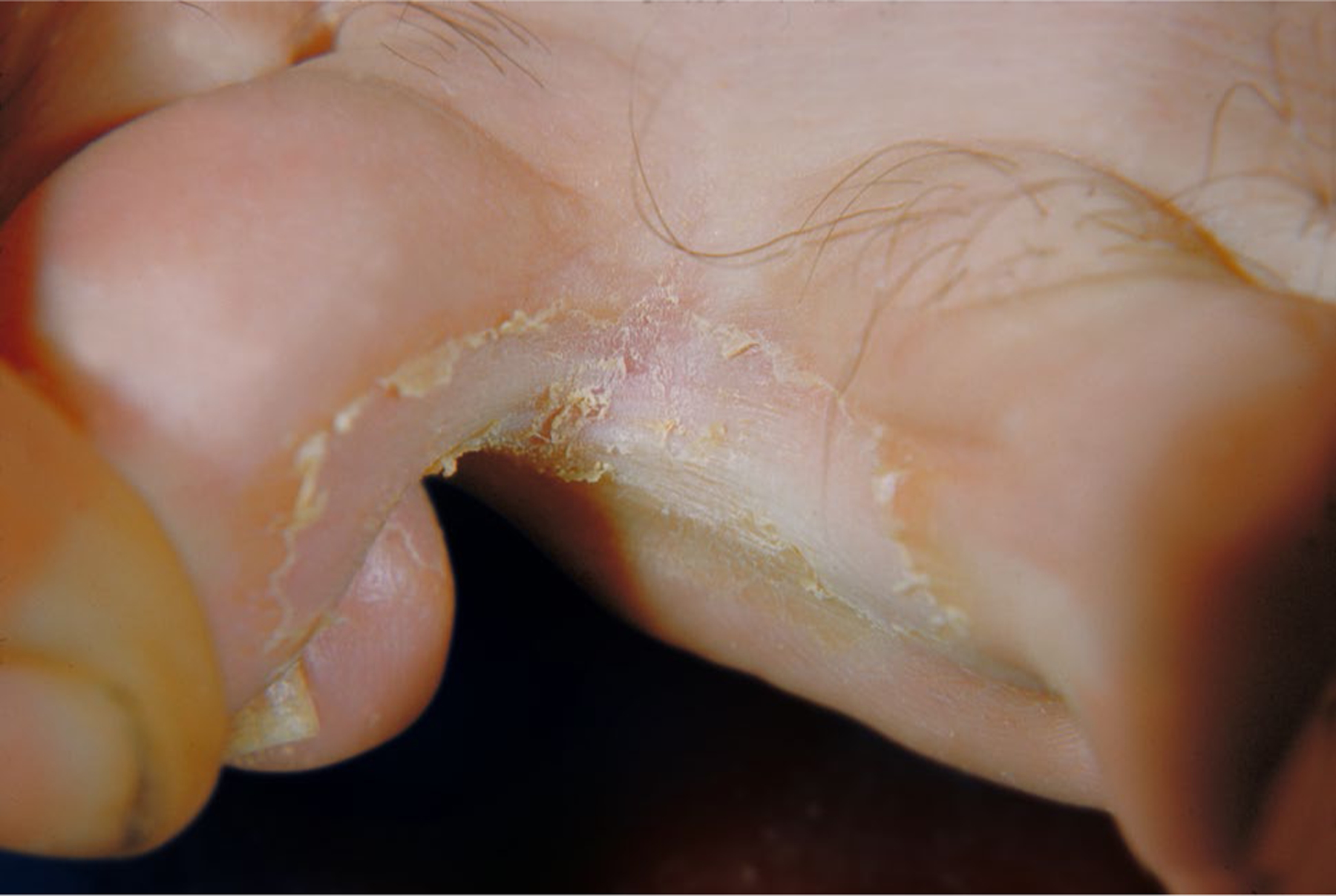
Interdigital tinea pedis of unknown species (CDC/Dr. Lucille K. Georg)
Fig. 11.
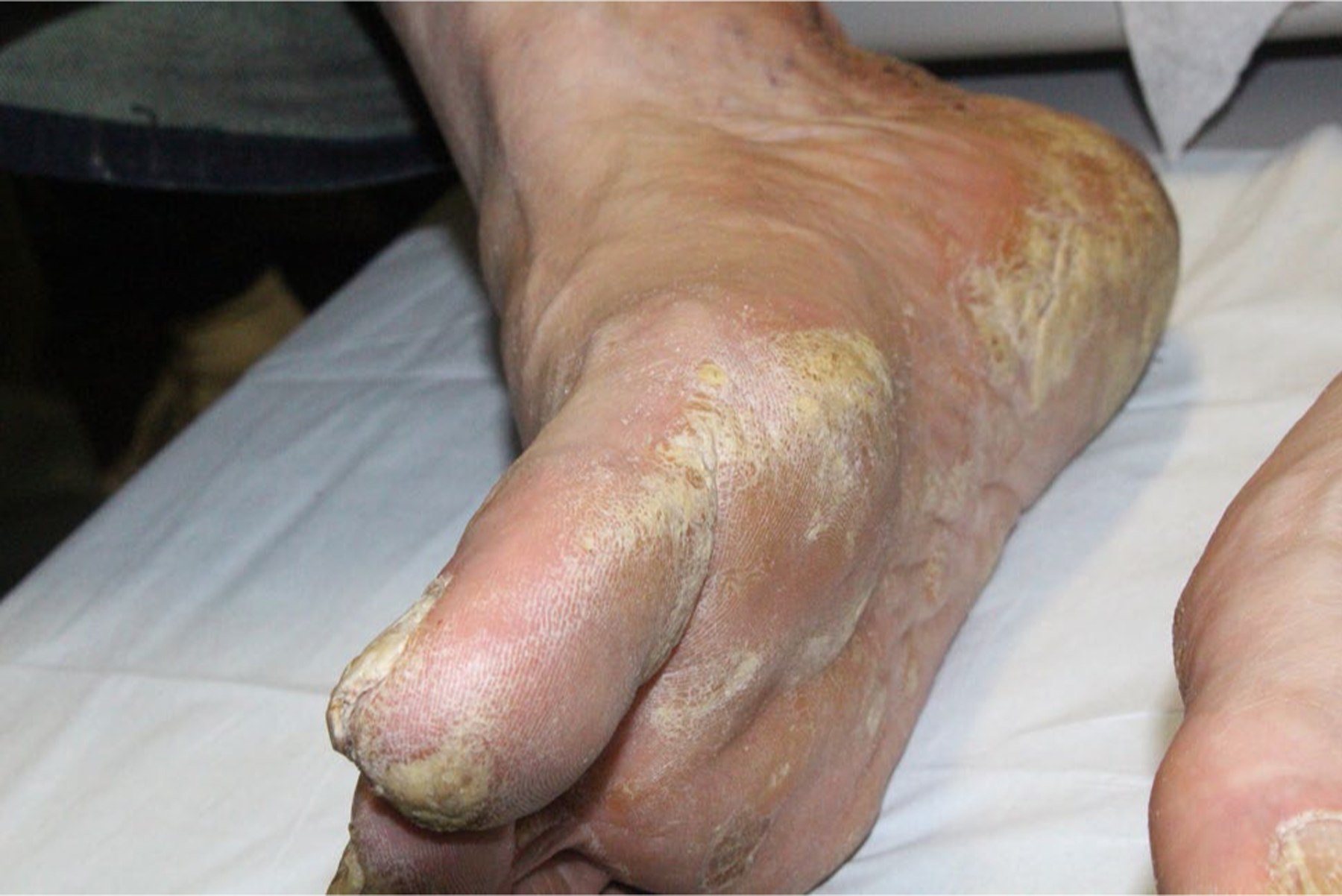
Tinea pedis (Dr. Shari Lipner)
Fig. 12.
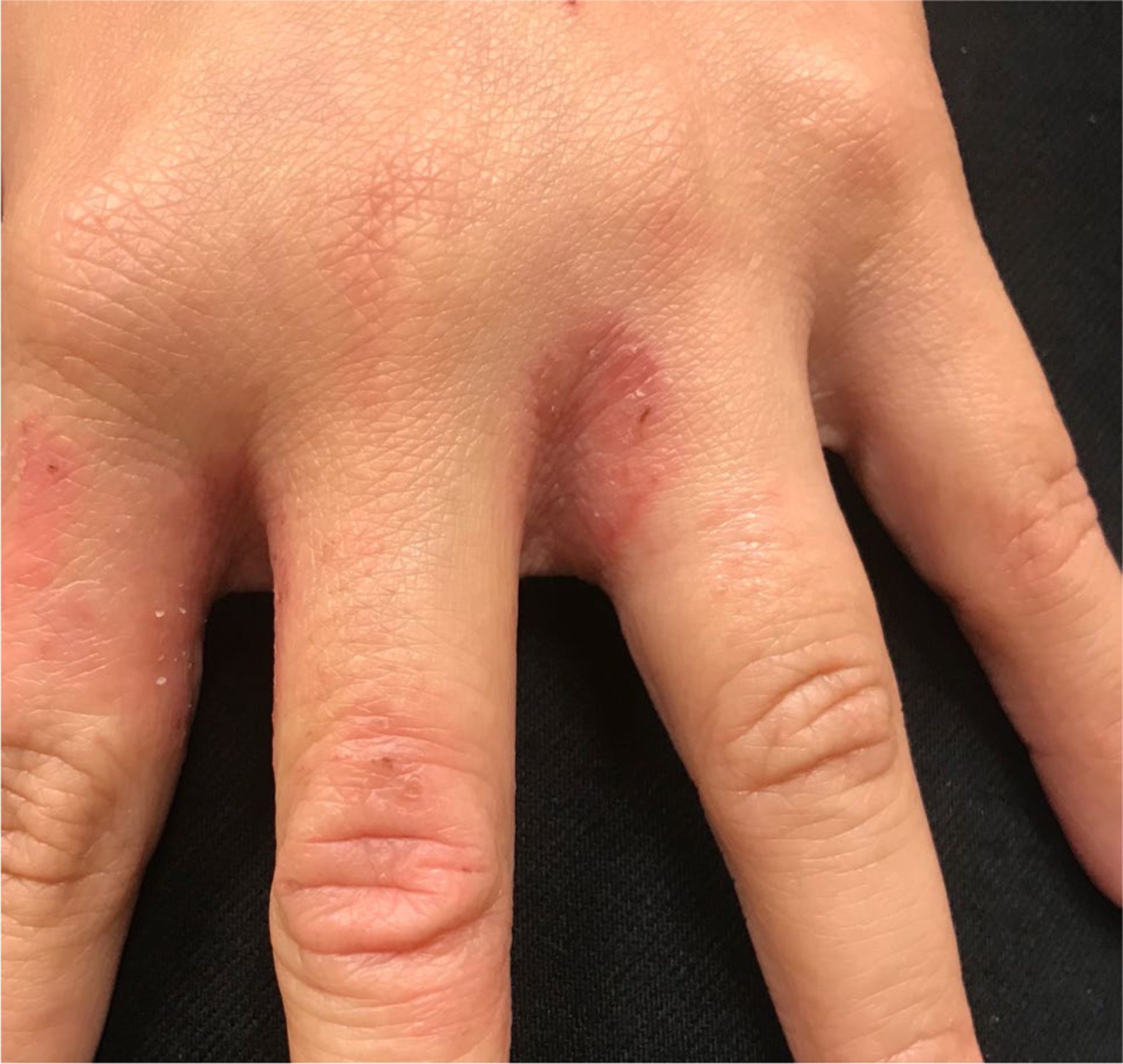
Tinea manuum (Dr. Avrom Caplan)
6.3. Diagnostic Methods
Diagnosis of tinea pedis and tinea manuum is primarily based on clinical features, coupled with a positive fungal culture, KOH preparation demonstrating hyphae, or PCR.
6.4. Treatment
Patient education regarding hygiene and proper foot care is an important aspect of management for tinea pedis, and consequently tinea manuum. Patients are instructed to keep feet clean and dry, including use of sweat-wicking socks to avoid sweat pooling [115, 116]. Patients are advised to wear shoes in communal areas, including in public gyms, showers, and pools. Although onychomycosis is beyond the scope of this review, proper nail care, including keeping nails short and clean, is also important for managing tinea pedis, as coexisting onychomycosis may preclude cure [38, 116, 117].
Topical antifungal therapy is the mainstay of treatment for tinea pedis. Per the AAP 2021–2024 Report of the Committee on Infectious Diseases, 2 weeks of therapy is sufficient for mild tinea pedis in children. Topical treatment in adults is recommended for at least 4 weeks. A Cochrane Library Systematic Review of 67 randomized controlled trials from 1970 to 2005 included participants using topical therapy who had mycologically diagnosed foot and toenail fungal infections. Of these, 11 trials (n = 1144) included in a meta-analysis comparing allylamines and azoles showed an RR of treatment failure of 0.63 (95% CI 0.42–0.94), favoring allylamines [118]. A 2012 systematic review on the efficacy and safety of topical antifungals for the treatment of dermatophytosis included a meta-analysis of 17 randomized controlled trials, including 1781 participants, comparing allylamines, including naftifine and terbinafine (1% or 2%) used for 1–6 weeks, with azoles or imidazoles, including econazole and miconazole (1% or 2%) used for 2–6 weeks, and found no statistically significant difference in mycological cure at the end of treatment [59]. A 2022 systematic review of randomized controlled trials examining 1042 participants with tinea pedis across seven studies found that topical terbinafine and topical butenafine, a benzylamine, were similarly efficacious (RR 1.3, 95% CI 0.4–4.4) [119]. However, limitations included lack of reporting on baseline characteristics in each treatment arm [119]. Taken together, topical treatment with azoles, allylamines, and benzylamines are all effective for tinea pedis treatment. Therefore, choice of therapy may be dictated by cost, availability of a specific class of medication, or physician preference. It has been suggested a cost-effective strategy is to utilize topical azoles as first-line therapy, followed by allylamines in cases of therapy failures [59]; however, more up-to-date studies are needed to validate this recommendation. It is important to ensure that patients complete the treatment regimen to prevent relapses [116].
Newer topical antifungals include sertaconazole 2% cream and luliconazole 1% cream. In a prospective, openlabel, randomized controlled trial of 313 total patients (232 patients with tinea pedis) using sertraconazole solution or cream twice daily for 28 days, efficacy (defined as a negative culture test and reduction of clinical severity) was achieved in 90.6% and 88.9% of patients, respectively [120]. A double-blind, randomized, vehicle-controlled, phase III study of 321 patients with tinea pedis treated with luliconazole cream 1% showed complete clearance at day 42 in 26.4% (28/106) of patients randomized to luliconazole cream 1%, versus 1.9% (2/103) vehicle (p < 0.001) [121]. In two randomized, double-blind, vehicle-controlled studies of 495 subjects with tinea pedis, patients were treated with econazole nitrate 1% foam for 4 weeks, and complete cure (negative KOH, negative fungal culture, complete clinical resolution) was achieved by 24.3% of patients at day 43 for the active drug versus 3.6% for vehicle (p < 0.001) [122].
Tinea pedis refractory to topical therapy may be treated with oral antifungals. First-line therapy for refractory tinea pedis is oral terbinafine. A 2012 Cochrane Library systematic review examining systemic treatments for fungal infections of the skin of the foot included 15 trials and 1438 participants. Of these, two trials of 71 participants compared terbinafine and griseofulvin, with a pooled RR of 2.26 (95% CI 1.49–3.44) favoring terbinafine. There was no significant difference in mycological cure between terbinafine and itraconazole (RR 1.07, 95% CI 0.92–1.25), fluconazole and itraconazole (RR 1.06, 95% CI 0.87–1.30), or fluconazole and ketoconazole (RR 1.04, 95% CI 0.92–1.17) [123]. In addition, clinical subtype of tinea pedis did not influence response to oral therapy [123, 124]. The incidence of liver injury with terbinafine is extremely low, but the drug should be avoided in patients with pre-existing liver disease, and baseline liver function testing is recommended [124–128]. Oral itraconazole may be considered as second-line due to hepatoxicity and cardiotoxicity and frequent drug–drug interactions. Oral ketoconazole should be avoided due to more severe risks of hepatotoxicity and due to more favorable safety profiles of terbinafine and itraconazole [116].
Patient preference is important when prescribing therapy for tinea pedis. In a cross-sectional study evaluating antifungal products for tinea pedis on Amazon.com in 2018, functionality was the most cited positive (14%) and negative (35%) feature for over-the-counter antifungal therapy based on consumer rating and reviews, with specific comments related to itching/burning relief and number of applications required for relief predominating [129]. Non-pharmacologic measures may be used as adjunct therapy, in addition to topical and/or oral antifungal therapy for tinea pedis and manuum. In a cross-sectional survey study of 152 patients with tinea pedis, 55.9% reported use of at least one non-pharmacological therapy for tinea pedis, including cologne, saltwater, vinegar soaks, and henna [130]. Vinegar soaks may be applied using old cotton socks, rather than tub soaks, to increase convenience and compliance [131]. Salicylic acid peels and tea tree oil may also be used to improve clinical symptoms, but are not effective for achieving mycological cure [132, 133].
The treatment algorithm for tinea manuum closely resembles that of tinea pedis, with topical antifungal agents as the preferred choice of therapy. Indications for systemic antifungals for tinea manuum include co-occurring fingernail infection and two feet-one hand syndrome [134, 135].
7. Chronic and Recurrent Dermatophytosis
Chronic and recurrent dermatophytosis is not a strictly defined clinical entity. It has been previously described as a dermatophyte infection persisting for > 6 months, with or without recurrence, even with adequate therapy [136–138]. It may be secondary to host, pathogen, environmental, or therapeutic factors [136].
7.1. Risk Factors for Chronic or Recurrent Infection
Risk factors for chronic or recurrent dermatophytosis are listed in Table 2.
7.1.1. Immunosuppressed Patients
In immunocompetent hosts, dermatophytosis is typically limited to the epidermis, with epidermal keratinization, body temperature, lipid composition of the stratum corneum, and innate and adaptive immunity preventing more widespread infections and infections extending into the dermis and sub-cutaneous fat [139, 140]. Host immunosuppression also increases the risk of recalcitrant dermatophytosis. In a cross-sectional study of dermatologic conditions in 4679 patients living with HIV in 2013–2018, HIV-infected patients, even if adequately treated, had increased risk of developing tinea corporis, cruris, capitis, and non-specified dermatophytosis (all p < 0.0009), suggesting persistence of immune system susceptibilities despite HIV treatment [28].
7.2. Complications and Impact
7.2.1. Extensive Dermatophytosis
Dermatophytosis may present as either extensive infection or deep and invasive infection. In extensive dermatophytosis, infection is confined to the stratum corneum, but covers a large body surface area or there are numerous discrete lesions [141]. In addition to patients living with HIV/AIDS [142, 143], extensive dermatophytosis has been described in solid organ and bone marrow transplant recipients (Figs. 13, 14), patients with disorders of keratinization, and patients with caspase-recruitment domain-containing protein 9 (CARD9) deficiency [144–148].
Fig. 13.
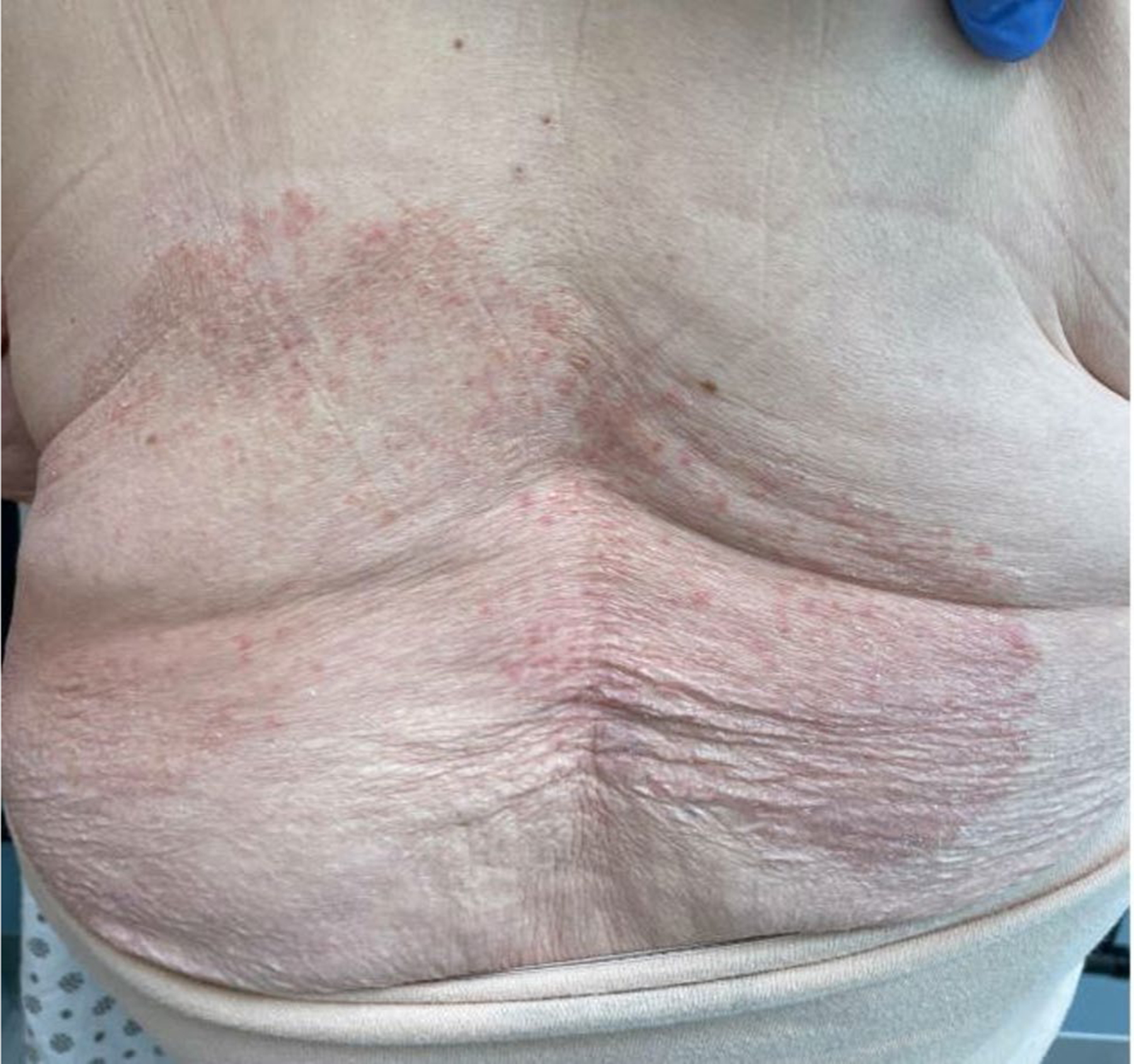
Tinea corporis of the back in a transplant recipient (Dr. Avrom Caplan)
Fig. 14.
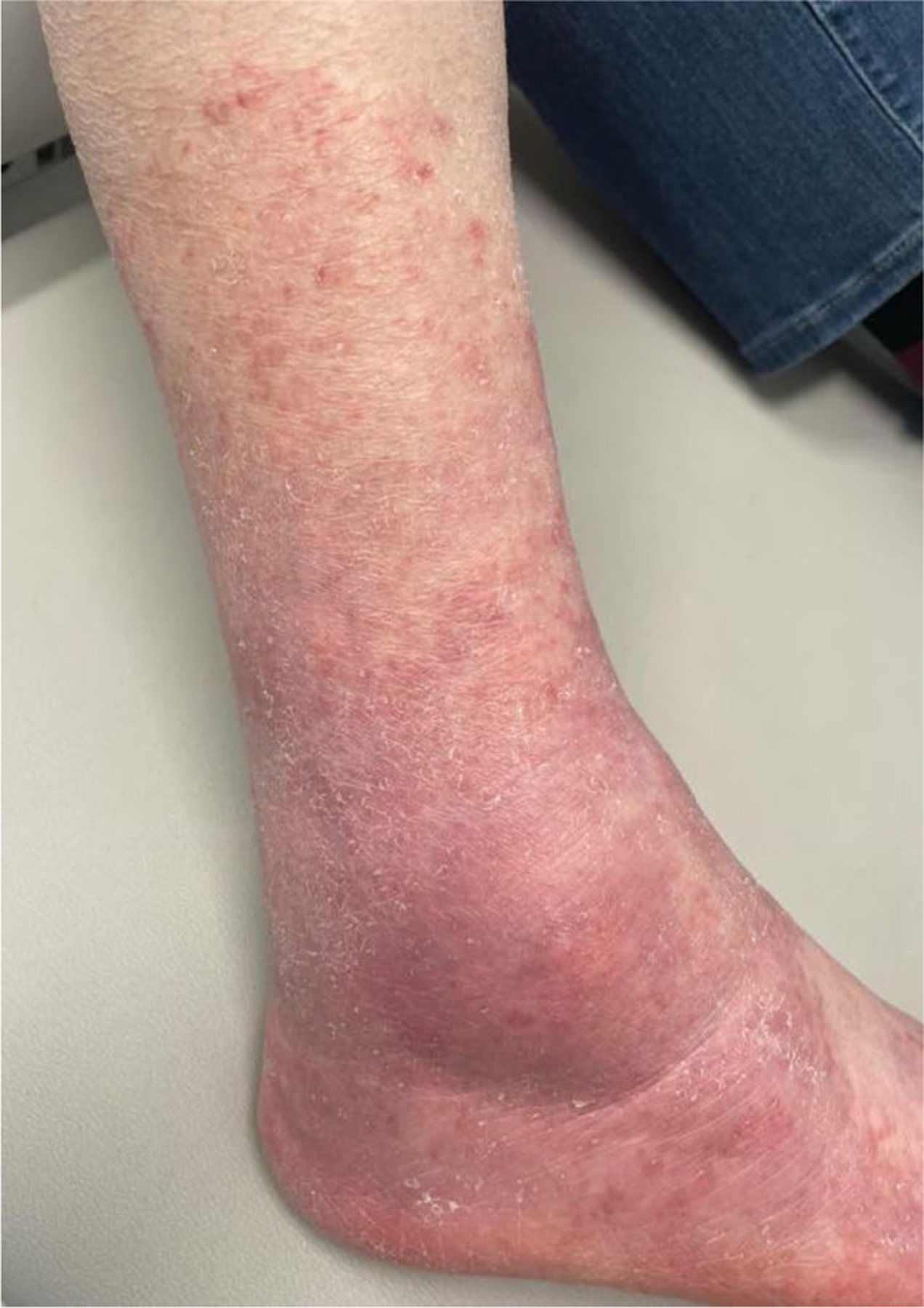
Tinea corporis of the distal leg in a transplant recipient (Dr. Avrom Caplan)
A case series described 13 male patients with severe oral, anal, and genital dermatophyte infections often requiring more than 1 month of antifungal therapy, mainly among men who have sex with men (n = 12) due to Trichophyton mentagrophytes genotype VII. Eight patients had multiple lesions and 7 had co-infection with HIV [149, 150].
7.2.2. Invasive Dermatophytosis
Invasive dermatophytosis presents as either deep dermatophytosis or Majocchi’s granuloma (MG), which are recognized as two separate clinical entities [139, 141]. In deep dermatophytosis, the infection extends from the epidermis to dermis. In a case series of 12 patients with severe dermatophytosis following solid organ transplant, deep dermatophytosis was characterized by discrete nodules (n = 9), an indolent course (median time between transplantation and severe dermatophytosis, on average, of 16 months [range 2–94 months]), and proximity to a superficial dermatophyte infection (n = 12). In a 2016 literature review, severe dermatophytosis was described as “ill-defined infiltrated plaques, nodules, and papules sometimes associated with itching, pain, and discharge” [141].
Deep and invasive dermatophytosis has been reported in solid organ transplant recipients and patients with CARD9 deficiency or HIV/AIDS [151–155], and in patients taking oral or topical corticosteroids and other immunosuppressive agents, or those with atopy, diabetes mellitus, Cushing’s disease, hematologic malignancies and disorders, autoimmune disorders, liver disease, and trauma [156–175]. In extremely rare cases, the infection may spread systemically via lymphatic or vascular systems, or via contiguous spread to bone [151, 176]. Although uncommon, deep dermatophytosis has been reported in immunocompetent hosts without a history of steroid use [177].
MG, also known as nodular granulomatous perifolliculitis, is another type of invasive dermatophytosis whereby dermatophytes invade the dermal and subcutaneous tissue [140]. MG presents as nodules, plaques, and papules in hair-bearing skin, most commonly the forearms, hands, legs, or ankles, including areas where trauma to the skin from shaving may occur [22]. Less frequently, MG may involve the face and/or scalp. It presents as one of two forms—a small, perifollicular papular form presenting with localized dermal infection in mostly healthy people, or a second form presenting with deep, subcutaneous, sometimes fluctuant, plaques and/or nodular lesions in immunocompromised individuals [155, 178–182].
7.2.3. Psychological Burden
Chronic dermatophytosis may profoundly impact quality of life (QoL) [183], likely due to physical symptoms, including pruritus, coupled with psychosocial problems from having rashes involving exposed skin [138], which is amplified with chronic dermatophytoses. Several studies have been conducted in communities across India to evaluate the psychosocial impact of chronic dermatophytoses and to quantitate burden of chronic fungal infection on QoL [138, 184–186]. In a cross-sectional study of the impact of chronic and recurrent dermatophytosis on QoL and psychologic morbidity using the Dermatology Life Quality Index (DLQI) and the Perceived Stress Scale (PSS), including 123 patients age ≥ 12 years, with 65.9% of participants with dermatophytosis for ≥ 6 months, those diagnosed with dermatophytosis for 6–9 months had lower PSS scores versus those with disease for 9–12 months and > 12 months (p < 0.001), with over 55% of patients reporting an ‘extremely large effect’ per the DLQI and involvement of more than one anatomic site a risk factor for higher DLQI score [138]. In a study of 184 children aged 6–12 years with tinea capitis in a semi-urban area of Nigeria, 58.2% had mild-to-severe psychosocial impact, and young females versus males were more often affected (61.5% vs. 41.7%; p = 0.02) [187]. While dermatophytoses have a high prevalence, cultural and socioeconomic differences globally may limit generalizing study findings to US communities. Nevertheless, it is well accepted that chronic and recurrent dermatophytoses have a negative impact on psychosocial wellbeing and QoL. Thus, a multidisciplinary approach to address the psychological impact of disease is needed.
7.3. Recalcitrant Dermatophytosis Treatment Guidelines in India
In response to an evolving landscape of recurrent, chronic, and recalcitrant dermatophytosis affecting the Indian subcontinent in recent years, the Indian Association of Dermatologists, Venereologists and Leprologists created the Task Force Against Recalcitrant Tinea (ITART) in 2017 to develop a consensus on the management of tinea affecting the glabrous skin (tinea corporis, tinea cruris, and tinea faciei), which was updated in 2020 [53]. The task force emphasized the importance of maintaining personal hygiene as a key component of managing tinea infections, including thoroughly drying skin after bathing, thoroughly drying clothes after washing, and avoiding sharing soaps, clothes, and other linens.
The 2020 ITART guidelines recommend that patients follow-up 3 weeks after beginning oral therapy to assess clinical response, and state that with inadequate response to terbinafine 250 mg daily after 3 weeks, the dose may be increased to twice daily. However, the FDA-approved dosage is 250 mg daily. The guidelines also recommend topical and oral antifungal therapy combinations for most patients at a longer duration than would otherwise be recommended to dermatophytosis-naïve patients. The Task Force recommended prescribing dermatophytosis-naïve patients itraconazole 100 mg for 3 weeks, and patients with chronic, steroid-modified, or recalcitrant infections, 100 mg twice daily for 4 weeks [53]. Expert consensus is lacking on the most appropriate dosing regimen of itraconazole for dermatophytoses; however, because of concerns that underdosing could lead to resistance, physicians could consider dosing itraconazole 100 mg twice daily at a minimum. ITART also recommends that topical therapy be applied twice daily and for 2 weeks after clinical resolution.
In 2018, an Expert Consensus on the Management of Dermatophytosis in India, including tinea pedis, was developed [188]. Experts recommended tinea pedis of any duration and severity with combination topical and systemic antifungal therapy. Azoles were recommended as first-line topical therapy. Terbinafine 250 mg once daily was recommended for naïve tinea pedis, and itraconazole 200–400 mg/day was preferred for severe or refractory infection. Recommended treatment for naïve tinea pedis is 2–4 weeks, and > 4 weeks for chronic, severe, or recalcitrant infection [188].
8. Emerging Resistance
Chronic and recurrent dermatophytoses may develop due to resistance to antifungal therapies. In the early 1980s–1990s, antifungal resistance among dermatophytes was hypothesized to be due to non-compliance with prescribed treatment, inadequate drug absorption or blood concentration, and drug–drug interactions; resistance was only demonstrated for griseofulvin and typically related to T. rubrum tinea corporis [189, 190]. Over the past decade, concerning outbreaks have emerged worldwide, with reports of both clinical and laboratory-confirmed resistance to oral terbinafine and other antifungals, most prominently in the Indian subcontinent [191, 192].
8.1. Pathogenesis
Antifungal resistance may develop for a variety of reasons, including host, pathogen, environmental, and pharmacologic factors [136]. Certain dermatophyte strains, including some Trichophyton species, may acquire resistance to antifungal drugs such as terbinafine; these antifungal-resistant strains can cause infection and person-to-person transmission [193]. It is hypothesized that the increased prevalence of antifungal-resistant dermatophyte infections in India stems from overzealous use of antifungal therapies, whether through empiric treatment of suspected but unconfirmed dermatophytosis or through prescription sharing [194–197]. Steroid cream sales in India are estimated at US$329 million, with a large proportion containing an antifungal combination [192]. Corticosteroid-antifungal-antibacterial combination therapies are available over-the-counter in India and other South and Southeast Asian countries at relatively low cost. Additionally, they are overprescribed for the treatment of a variety of dermatoses, even non-fungal dermatoses. These combination topical therapies often contain high-potency steroids, such as clobetasol, and use of these therapies is likely a major driver of resistance [191, 192]. While these medications may temporarily improve clinical symptoms of dermatophyte infections, including pruritus, they are ultimately problematic in altering the classic morphology of cutaneous fungal infections, leading to delayed diagnosis [102, 191, 192, 198, 199]. These combination topical therapies reduce local cellular immunity at sites of dermatophyte infections, allowing for severe dermatophytosis to develop [191, 192].
8.2. Trichophyton indotineae
The pathogen now widely known as Trichophyton indotineae was first identified in 2017 and was referred to as Trichophyton mentagrophytes genotype VIII. Although a member of the T. mentagrophytes species complex, T. indotineae represents a frequently terbinafine-resistant species caused by mutations in the gene encoding squalene epoxidase (SQLE), a key enzyme in the ergosterol biosynthetic pathway whose inhibition results in accumulation of squalene, depletion of ergosterol, and arrest of fungal growth [200]. Mutations in the SQLE gene lead to ineffective enzyme function and cause elevated minimum inhibitory concentrations (MICs) of terbinafine. T. indotineae is distinguished from T. mentagrophytes in part by sequence differences across the genome including the ITS region [200–203]. Therefore, T. indotineae was recognized as a separate species in 2020. This newly identified species is primarily responsible for the epidemic-level prevalence of dermatophyte infections in India [204].
While T. indotineae is believed to have been present in India, Australia, Iran, and Oman since 2004 [196], the first clinical cases of what is now known as T. indotineae dermatophytosis were described in India after 2014, where chronic and recalcitrant dermatophytosis concerned dermatologists for the past decade [53]. T. indotineae dermatophytosis was thereafter recognized in other Asian countries and across Europe [205–215]. The first cases of T. indotineae dermatophytoses in the US were reported in February 2023 in New York City, when two patients with severe tinea infections failed to clear despite treatment with oral terbinafine [197]. As such, T. indotineae dermatophytosis is now recognized as a worldwide problem [216].
Clinically, T. indotineae dermatophytoses evolve differently than classic tinea infections. T. indotineae dermatophytoses present as extensive, severe, and/or refractory tinea corporis, cruris, and/or faciei infections [33], and are characterized by intense pruritus, widespread anatomic involvement, and are highly contagious. These infections are highly inflammatory and lesions may be large and numerous [102, 199].
An added complication of identifying T. indotineae dermatophytoses is that traditional culture-based techniques utilized for dermatophytosis diagnosis fail to correctly identify T. indotineae, instead resulting in diagnoses of T. mentagrophytes or T. interdigitale dermatophytoses [53, 217, 218]. Therefore, T. indotineae dermatophytosis must be confirmed using molecular-based methods, such as PCR, to avoid misdiagnosis and delay of appropriate treatment. In the US, dermatologists are encouraged to contact their state public health department, who can assist in identifying appropriate testing protocol and laboratories that can perform genomic sequencing [197].
Optimal treatment guidelines for T. indotineae dermatophytosis are lacking and current treatment recommendations are based on individual cases or case series. Due to wide-spread resistance to otherwise first-line oral agent terbinafine, oral itraconazole is the drug of choice for treating T. indotineae dermatophytosis. In a 2020 study of 200 patients with microscopy-confirmed tinea infection in India, treatment with 8 weeks of itraconazole was superior to treatment with fluconazole, griseofulvin, and terbinafine (p < 0.001) for both clinical and mycological cure [219]. Although the patients included in this study were not confirmed T. indotineae cases, patients meeting the inclusion criteria had chronic or chronic-relapsing tinea corporis, cruris, or faciei (duration of tinea ≥ 3 months, or tinea present for ≥ 3 months that improved but relapsed within 1 month), suggesting possible T. indotineae infections. A double-blind, randomized trial of 149 adult patients with tinea corporis/cruris involving > 5% body surface area receiving different itraconazole dose regimens (100, 200, 400 mg/day) showed similar complete cure rates for the 100 mg and 200 mg doses (HR 1.44, 95% CI 0.91–2.30; p = 0.12) and treatment duration (mean ± standard deviation [SD] 7.7 ± 4.73 vs. 7.2 ± 3.81 weeks; p = 0.902), with 400 mg superior for both outcomes (100 vs. 400 mg groups; HR 2.87, 95% CI 1.78–4.62, p < 0.01; 200 vs. 400 mg groups (HR 1.99, 95% CI 1.28–3.09; p = 0.02). Although the 200 and 400 mg groups had 63% and 120% higher associated costs versus the 100 mg group in achieving cure [68], use of higher itraconazole doses to achieve complete cure may be more cost-effective overall. A dose of itraconazole 200 mg daily may be used to treat infections unresponsive to first-line oral therapies, with the caveat that treatment may be needed for > 3 months [188, 197]. Topical luliconazole cream may also be used although oral therapy is the mainstay of treatment [220].
Combination therapy with terbinafine and itraconazole has been studied for recalcitrant dermatophytoses, although larger studies are needed to validate its use. In a study of 45 patients with recalcitrant dermatophytosis treated for 4 weeks with terbinafine 250 mg and itraconazole 200 mg once daily versus monotherapy with terbinafine 250 mg twice daily or itraconazole 200 mg twice daily, at 12 weeks, 100% of the combination therapy group achieved complete cure versus 80% and 86.7% in the terbinafine-only and itraconazole-only groups, respectively (p = 0.207) [221]. In a study of 60 patients with clinical and KOH-positive tinea corporis, cruris, or faciei, at 3 weeks, 90% of patients treated with combination therapy with terbinafine 250 mg once daily and itraconazole 200 mg once daily achieved complete cure versus 35% and 50% in the terbinafine and itraconazole monotherapy groups, respectively (p = 0.002) [222].
Resistance to oral itraconazole among dermatophyte infections has been observed [205, 223–225]. In such instances, voriconazole and posaconazole, two oral azole antifungals ordinarily reserved for invasive mold infections, may be utilized. When antifungal therapies fail, physicians should investigate the cause of treatment failure, including adherence to therapy, confirmation of the diagnosis, and whether resistance is suspected. Antifungal susceptibility testing (AFST) could be pursued but is not yet well established in relation to dermatophyte infections.
It is recommended that itraconazole capsules are administered with high-fat foods and an acidic beverage, such as juice, to increase absorption and bioavailability. In contrast, the oral solution should be administered on an empty stomach, either 1 h before, or 2–3 h after, a meal [226]. Triazole antifungals are also associated with substantial drug–drug interactions and adverse effects [227]. For example, coadministering itraconazole with a proton pump inhibitor may reduce bioavailability via gastric acid suppression. Therapeutic drug monitoring may be required for triazoles to ensure adequate dosing, although target serum drug levels have not yet been established for the treatment of resistant dermatophytoses, and the correlation between serum levels and accumulation in tissue has not been established. Given the relatively infrequent use of these antifungals, dermatologists are encouraged to contact infectious disease specialists and pharmacists before prescribing. Issues with insurance coverage may arise when prescribing these less commonly used antifungal therapies, creating a barrier for patients.
8.3. Terbinafine‑Resistant T. rubrum
T. rubrum terbinafine-resistant tinea corporis cases have been reported in the US [223]. In a retrospective study of 271 dermatophyte isolates from North American institutions from 2021 to 2022, 18.6% were resistant to terbinafine, including 21 isolates of T. rubrum [216].
8.4. Implications for Clinical Practice
Empiric treatment of suspected dermatophyte infections is discouraged. At a minimum, dermatologists can confirm mycological diagnosis using KOH with microscopy (Fig. 15), culture, or PCR before prescribing antifungal therapy [228]. If an antifungal-resistant dermatophytosis is suspected and resources are available, advanced molecular techniques (e.g., genomic sequencing of the internal transcribed spacer region) can help identify the causative organism and guide treatment [197, 229]. US dermatologists can contact their state and local health department and Centers for Disease Control and Prevention (CDC) to receive assistance with testing and report unusual cases. If advanced diagnostics are not available, empiric treatment is reasonable.
Fig. 15.
Trichophyton indotineae KOH preparation (Dr. Avrom Caplan). KOH potassium hydroxide
The emergence of antifungal-resistant tinea infections has several implications for clinical practice. First, prescribing combination topical antifungal-steroid medications for dermatophyte infections is strongly discouraged [53, 188, 218]. Dermatologists may educate non-dermatologists and patients on avoiding such therapies. Furthermore, dermatologists can promote the importance of not sharing prescriptions among household members, of proper adherence to recommended therapies, and of seeking examination by a dermatologist if their condition is not improving with recommended therapy [194].
9. Future Directions
As infections with T. indotineae and terbinafine-resistant T. rubrum continue to rise in the US, surveillance efforts will be critical to characterize disease epidemiology, monitor spread, describe treatment practices and outcomes, inform clinical practice, and guide public health interventions [197, 230]. Detection of antifungal-resistant species depends on dermatologists maintaining a high index of suspicion and having adequate access to laboratory testing. In partnership with federal agencies, state and local public health agencies, professional societies, and academic institutions, dermatologists can lead efforts to prevent the spread of antifungalresistant dermatophytes and reduce barriers to testing and appropriate treatment. Staying abreast of updated information regarding new resistant species and changes in treatment guidelines can ensure that dermatologists are providing relevant and informed care to patients.
AFST for dermatophytes was rarely performed before the emergence of T. indotineae [231] but is becoming an increasingly important component of managing recalcitrant tinea infections [232]. The European Committee on Antimicrobial Susceptibility Testing (EUCAST) provides guidelines for conducting in vitro AFST. A multicenter validation of six EUCAST methods was performed, whereby 10 laboratories with minimal experience with dermatophyte testing performed susceptibility testing of terbinafine, itraconazole, voriconazole and amorolfine against a blinded panel of 38 terbinafine wild-type and target gene mutant isolates, showing a low error rate and favoring a chloramphenicol and cycloheximide supplemented growth media and spectrophotometric endpoint reading using 50% inhibition [233], a potential standardized method for AFST. Although adoption of this method worldwide may generate reproducible susceptibility data, it requires prolonged incubation periods and specialized laboratory equipment with staff who are trained in at least basic laboratory-based work, and many laboratories throughout the world follow the guidelines outlined by the Clinical and Laboratory Standards Institute rather than EUCAST [233, 234]. The terbinafine containing agar method (TCAM) may be used to identify susceptibility to terbinafine. In one study of 20 molecularly identified isolates, including isolates of T. indotineae, eight clinical microbiology laboratories received blinded samples. All laboratories correctly identified the terbinafine-resistant isolates (n = 5) and the terbinafine-susceptible isolates (n = 15) [231], highlighting the reliability and ease of performing the TCAM. However, TCAM is limited to testing terbinafine susceptibility, precluding use in suspected itraconazole-resistant and other suspected antifungal-resistant dermatophytoses [231]. Molecular methods may be used to detect resistant dermatophytes and may identify molecular mechanisms and target gene mutations that confer antifungal resistance, but these methods are not widely available [235]. In one study of a commercially available PCR assay used to identify Trichophyton isolates with common SQLE mutations, including Phe397Leu and Leu393Phe, the assay identified all 40 isolates of T. indotineae harboring a mutant SQLE gene out of 90 total T. indotineae isolates [234]. While this method may potentially provide rapid information about antifungal resistance, it is currently considered an adjunct to culture-based susceptibility testing techniques, due to the potential for a range of mutations in resistant dermatophytes that are not yet available in molecular assays [235]. Additional large, prospective studies are needed to confirm the reliability of molecular methods to detect antifungal-resistant dermatophytes.
10. Conclusion
Antifungal stewardship is a cornerstone of preventing the spread of antifungal-resistant dermatophytoses [229, 236, 237]. Confirmation of dermatophyte infections with appropriate mycological testing is necessary, followed by adherence to recommended treatment guidelines and consensus statements issued by trusted medical organizations and societies. High clinical suspicion for resistant isolates of dermatophytes is necessary if patients do not improve with the recommended course of therapy. Furthermore, dermatologist-led engagements with regulatory bodies and public health agencies could help address barriers to providing testing and appropriate treatment and to address the public health threat posed by the spread of these infections [197]. Antifungal stewardship extends beyond specialist care by a dermatologist, to both patients and non-dermatologists, who must be educated on the risks of inappropriate prescribing practices related to antifungal therapy.
Key Points.
Dermatophytoses are fungal infections of the skin, hair, and nails.
Over the past decade, some dermatophytoses have become resistant to antifungal therapies, resulting in chronic or recurrent infections.
Antifungal stewardship is a key aspect of managing and preventing treatment-resistant dermatophytoses.
Funding
No sources of funding were used to assist in the preparation of this review.
Financial disclosures
Shari R. Lipner has served as a consultant for Ortho-Dermatologics, Eli Lilly, Moberg Pharmaceuticals, and Belle-Torus Corporation.
Footnotes
Declarations
Prior publication This work has not been previously published.
Conflicts of interest Rachel C. Hill, Avrom S. Caplan, Boni Elewski, Jeremy A.W. Gold, Shawn R. Lockhart, Dallas J. Smith, and Shari R. Lipner have no conflicts of interest to declare.
Ethics approval No ethical approval was required as this was a review study.
Consent to participate/publish Patient consent was not required as this was a review study.
Institutional Review Board approval status Not applicable.
Reprint requests Shari R. Lipner, MD, PhD.
Publisher's Disclaimer: Disclaimer The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Data/code availability
Data/code sharing are not applicable to this article as no datasets or code were generated and/or analyzed during the current study.
References
- 1.Chanyachailert P, Leeyaphan C, Bunyaratavej S. Cutaneous fungal infections caused by dermatophytes and non-dermatophytes: an updated comprehensive review of epidemiology, clinical presentations, and diagnostic testing. J Fungi (Basel). 2023;9(6):669. 10.3390/jof9060669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Havlickova B, Czaika VA, Friedrich M. Epidemiological trends in skin mycoses worldwide [published correction appears in Mycoses. 2009 Jan;52(1):95]. Mycoses. 2008;51(Suppl 4):2–15. 10.1111/j.1439-0507.2008.01606.x. [DOI] [PubMed] [Google Scholar]
- 3.Hainer BL. Dermatophyte infections. Am Fam Physician. 2003;67(1):101–8. [PubMed] [Google Scholar]
- 4.Osman M, Kasir D, Rafei R, et al. Trends in the epidemiology of dermatophytosis in the Middle East and North Africa region. Int J Dermatol 2022;61(8):935–68. 10.1111/ijd.15967. [DOI] [PubMed] [Google Scholar]
- 5.Ameen M Epidemiology of superficial fungal infections. Clin Dermatol 2010;28(2):197–201. 10.1016/j.clindermatol.2009.12.005. [DOI] [PubMed] [Google Scholar]
- 6.Grigoryan KV, Tollefson MM, Olson MA, Newman CC. Pediatric tinea capitis caused by Trichophyton violaceum and Trichophyton soudanense in Rochester, Minnesota, United States. Int J Dermatol 2019;58(8):912–5. 10.1111/ijd.14352257. [DOI] [PubMed] [Google Scholar]
- 7.Hay RJ. Tinea Capitis: current status. Mycopathologia 2017;182(1–2):87–93. 10.1007/s11046-016-0058-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.de Hoog GS, Dukik K, Monod M, et al. Toward a novel multilocus phylogenetic taxonomy for the dermatophytes. Mycopathologia 2017;182(1–2):5–31. 10.1007/s11046-016-0073-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kasperova A, Kunert J, Raska M. The possible role of dermatophyte cysteine dioxygenase in keratin degradation. Med Mycol 2013;51(5):449–54. 10.3109/13693786.2013.794310. [DOI] [PubMed] [Google Scholar]
- 10.Kasperova A, Cahlikova R, Kunert J, Sebela M, Novak Z, Raska M. Exposition of dermatophyte Trichophyton mentagrophytes to l-cystine induces expression and activation of cysteine dioxygenase. Mycoses. 2014;57(11):672–8. 10.1111/myc.12220. [DOI] [PubMed] [Google Scholar]
- 11.Nenoff P, Krüger C, Ginter-Hanselmayer G, Tietz HJ. Mycology—an update. Part 1: dermatomycoses: causative agents, epidemiology and pathogenesis. J Dtsch Dermatol Ges 2014;12(3):188–212. 10.1111/ddg.12245. [DOI] [PubMed] [Google Scholar]
- 12.Grumbt M, Monod M, Yamada T, Hertweck C, Kunert J, Staib P. Keratin degradation by dermatophytes relies on cysteine dioxygenase and a sulfite efflux pump. J Investig Dermatol 2013;133(6):1550–5. 10.1038/jid.2013.41. [DOI] [PubMed] [Google Scholar]
- 13.Danielli LJ, Lopes W, Vainstein MH, Fuentefria AM, Apel MA. Biofilm formation by Microsporum canis. Clin Microbiol Infect 2017;23(12):941–2. 10.1016/j.cmi.2017.06.006. [DOI] [PubMed] [Google Scholar]
- 14.Costa-Orlandi CB, Sardi JC, Santos CT, Fusco-Almeida AM, Mendes-Giannini MJ. In vitro characterization of Trichophyton rubrum and T. mentagrophytes biofilms. Biofouling. 2014;30(6):719–27. 10.1080/08927014.2014.919282. [DOI] [PubMed] [Google Scholar]
- 15.Jazdarehee A, Malekafzali L, Lee J, Lewis R, Mukovozov I. Transmission of onychomycosis and dermatophytosis between household members: a scoping review. J Fungi (Basel). 2022;8(1):60. 10.3390/jof8010060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Raboobee N, Aboobaker J, Peer AK. Tinea pedis et unguium in the Muslim community of Durban, South Africa. Int J Dermatol 1998;37(10):759–65. 10.1046/j.1365-4362.1998.00550.x. [DOI] [PubMed] [Google Scholar]
- 17.Watanabe S, Anzawa K, Mochizuki T. High prevalence of superficial white onychomycosis by Trichophyton interdigitale in a Japanese nursing home with a geriatric hospital. Mycoses. 2017;60(10):634–7. 10.1111/myc.12625. [DOI] [PubMed] [Google Scholar]
- 18.Tuknayat A, Bhalla M, Kaur A, Garg S. Familial dermatophytosis in India: a study of the possible contributing risk factors. J Clin Aesthet Dermatol 2020;13(2):58–60. [PMC free article] [PubMed] [Google Scholar]
- 19.Baumgardner DJ. Fungal infections from human and animal contact. J Patient Cent Res Rev 2017;4(2):78–89. 10.17294/2330-0698.1418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tan JS. Human zoonotic infections transmitted by dogs and cats. Arch Intern Med 1997;157(17):1933–43. [PubMed] [Google Scholar]
- 21.Filipello Marchisio V, Preve L, Tullio V. Fungi responsible for skin mycoses in Turin (Italy). Mycoses. 1996;39(3–4):141–50. 10.1111/j.1439-0507.1996.tb00117.x. [DOI] [PubMed] [Google Scholar]
- 22.Boral H, Durdu M, Ilkit M. Majocchi’s granuloma: current perspectives. Infect Drug Resist 2018;11:751–60. 10.2147/IDR.S145027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mirmirani P, Tucker LY. Epidemiologic trends in pediatric tinea capitis: a population-based study from Kaiser Permanente Northern California. J Am Acad Dermatol 2013;69(6):916–21. 10.1016/j.jaad.2013.08.031. [DOI] [PubMed] [Google Scholar]
- 24.Silverberg NB, Weinberg JM, DeLeo VA. Tinea capitis: focus on African American women. J Am Acad Dermatol 2002;46(2 Suppl Understanding):S120–4. 10.1067/mjd.2002.120793. [DOI] [PubMed] [Google Scholar]
- 25.Lobato MN, Vugia DJ, Frieden IJ. Tinea capitis in California children: a population-based study of a growing epidemic. Pediatrics. 1997;99(4):551–4. 10.1542/peds.99.4.551. [DOI] [PubMed] [Google Scholar]
- 26.Abdel-Rahman SM, Farrand N, Schuenemann E, et al. The prevalence of infections with Trichophyton tonsurans in schoolchildren: the CAPITIS study. Pediatrics. 2010;125(5):966–73. 10.1542/peds.2009-2522. [DOI] [PubMed] [Google Scholar]
- 27.Hennessee IP, Benedict K, Dulski TM, Lipner SR, Gold JAW. Racial disparities, risk factors, and clinical management practices for tinea capitis: an observational cohort study among US children with Medicaid. J Am Acad Dermatol 2023;89(6):1261–4. 10.1016/j.jaad.2023.07.1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bender AM, Tang O, Khanna R, Ständer S, Kang S, Kwatra SG. Racial differences in dermatologic conditions associated with HIV: a cross-sectional study of 4679 patients in an urban tertiary care center. J Am Acad Dermatol 2020;82(5):1117–23. 10.1016/j.jaad.2019.08.072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pariser RJ, Pariser DM. Primary care physicians’ errors in handling cutaneous disorders. A prospective survey. J Am Acad Dermatol 1987;17(2 Pt 1):239–45. 10.1016/s0190-9622(87)70198-4. [DOI] [PubMed] [Google Scholar]
- 30.Yadgar RJ, Bhatia N, Friedman A. Cutaneous fungal infections are commonly misdiagnosed: a survey-based study. J Am Acad Dermatol 2017;76(3):562–3. 10.1016/j.jaad.2016.09.041. [DOI] [PubMed] [Google Scholar]
- 31.Yee G, Al Aboud AM. Tinea corporis. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024. [Updated 2023 Aug 8]. https://www.ncbi.nlm.nih.gov/books/NBK544360/. [PubMed] [Google Scholar]
- 32.Leung AK, Lam JM, Leong KF, Hon KL. Tinea corporis: an updated review. Drugs Context. 2020. 10.7573/dic.2020-5-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Khurana A, Sardana K, Chowdhary A. Antifungal resistance in dermatophytes: recent trends and therapeutic implications. Fungal Genet Biol 2019;132: 103255. 10.1016/j.fgb.2019.103255. [DOI] [PubMed] [Google Scholar]
- 34.Pathania S, Rudramurthy SM, Narang T, Saikia UN, Dogra S. A prospective study of the epidemiological and clinical patterns of recurrent dermatophytosis at a tertiary care hospital in India. Indian J Dermatol Venereol Leprol 2018;84(6):678–84. 10.4103/ijdvl.IJDVL_645_17. [DOI] [PubMed] [Google Scholar]
- 35.Rudramurthy SM, Shankarnarayan SA, Dogra S, et al. Mutation in the squalene epoxidase gene of trichophyton interdigitale and trichophyton rubrum associated with allylamine resistance. Antimicrob Agents Chemother. 2018;62(5):e02522–e2617. 10.1128/AAC.02522-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ely JW, Rosenfeld S, Seabury SM. Diagnosis and management of tinea infections. Am Fam Physician. 2014;90(10):702–10. [PMC free article] [PubMed] [Google Scholar]
- 37.Moriarty B, Hay R, Morris-Jones R. The diagnosis and management of tinea. BMJ 2012;345: e4380. 10.1136/bmj.e4380. [DOI] [PubMed] [Google Scholar]
- 38.Lipner SR, Scher RK. Onychomycosis: clinical overview and diagnosis. J Am Acad Dermatol 2019;80(4):835–51. 10.1016/j.jaad.2018.03.062. [DOI] [PubMed] [Google Scholar]
- 39.Gupta AK, Cooper EA, Summerbell RC, Nakrieko KA. Utility of polymerase chain reaction for assessment of onychomycosis during topical therapy. Int J Dermatol 2022;61(11):1385–9. 10.1111/ijd.16241. [DOI] [PubMed] [Google Scholar]
- 40.Gupta AK, Hall DC, Cooper EA, Ghannoum MA. Diagnosing onychomycosis: what’s new? J Fungi (Basel). 2022;8(5):464. 10.3390/jof8050464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Gupta AK, Versteeg SG, Shear NH. Onychomycosis in the 21st century: an update on diagnosis, epidemiology, and treatment. J Cutan Med Surg 2017;21(6):525–39. 10.1177/1203475417716362. [DOI] [PubMed] [Google Scholar]
- 42.Gupta AK, Nakrieko KA. Onychomycosis infections do polymerase chain reaction and culture reports agree? J Am Podiatr Med Assoc 2017;107(4):280–6. 10.7547/15-136. [DOI] [PubMed] [Google Scholar]
- 43.Litz CE, Cavagnolo RZ. Polymerase chain reaction in the diagnosis of onychomycosis: a large, single-institute study. Br J Dermatol 2010;163(3):511–4. 10.1111/j.1365-2133.2010.09852.x. [DOI] [PubMed] [Google Scholar]
- 44.Garg J, Tilak R, Garg A, Prakash P, Gulati AK, Nath G. Rapid detection of dermatophytes from skin and hair. BMC Res Notes. 2009;2:60. 10.1186/1756-0500-2-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Verrier J, Monod M. Diagnosis of dermatophytosis using molecular biology. Mycopathologia. 2017;182(1–2):193–202. 10.1007/s11046-016-0038-z. [DOI] [PubMed] [Google Scholar]
- 46.Cinotti E, Perrot JL, Labeille B, Cambazard F. Reflectance confocal microscopy for cutaneous infections and infestations. J Eur Acad Dermatol Venereol 2016;30(5):754–63. 10.1111/jdv.13254. [DOI] [PubMed] [Google Scholar]
- 47.Markus R, Huzaira M, Anderson RR, González S. A better potassium hydroxide preparation? In vivo diagnosis of tinea with confocal microscopy. Arch Dermatol 2001;137(8):1076–8. [PubMed] [Google Scholar]
- 48.Liansheng Z, Xin J, Cheng Q, Zhiping W, Yanqun L. Diagnostic applicability of confocal laser scanning microscopy in tinea corporis. Int J Dermatol 2013;52(10):1281–2. 10.1111/j.1365-4632.2011.05144.x. [DOI] [PubMed] [Google Scholar]
- 49.Lim SS, Shin K, Mun JH. Dermoscopy for cutaneous fungal infections: a brief review. Health Sci Rep 2022;5(1): e464. 10.1002/hsr2.464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Lekkas D, Ioannides D, Lazaridou E, et al. Dermatoscopy of tinea corporis. J Eur Acad Dermatol Venereol 2020;34(6):e278–80. 10.1111/jdv.16277. [DOI] [PubMed] [Google Scholar]
- 51.Gómez-Moyano E, Crespo Erchiga V, Martínez Pilar L, et al. Using dermoscopy to detect tinea of vellus hair. Br J Dermatol 2016;174(3):636–8. 10.1111/bjd.14085. [DOI] [PubMed] [Google Scholar]
- 52.Knöpfel N, del Pozo LJ, Escudero Mdel M, Martín-Santiago A. Dermoscopic visualization of vellus hair involvement in tinea corporis: a criterion for systemic antifungal therapy? Pediatr Dermatol 2015;32(5):e226–7. 10.1111/pde.12648. [DOI] [PubMed] [Google Scholar]
- 53.Rengasamy M, Shenoy MM, Dogra S, et al. Indian Association of Dermatologists, Venereologists and Leprologists (IADVL) task force against recalcitrant tinea (ITART) consensus on the management of glabrous tinea (INTACT). Indian Dermatol Online J 2020;11(4):502–19. 10.4103/idoj.IDOJ_233_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Drake LA, Dinehart SM, Farmer ER, et al. Guidelines of care for superficial mycotic infections of the skin: tinea corporis, tinea cruris, tinea faciei, tinea manuum, and tinea pedis. Guidelines/Outcomes Committee. American Academy of Dermatology. J Am Acad Dermatol 1996;34(2):282–6. 10.1016/s0190-9622(96)80135-6. [DOI] [PubMed] [Google Scholar]
- 55.Antifungal agents for common outpatient paediatric infections. Canadian Paediatric Society. https://cps.ca/en/documents/posit ion/antifungal-agents-common-infections. Accessed 6 Nov 2023. [Google Scholar]
- 56.Kimberlin DW, et al. editors. Red book: 2021–2024 Report of the Committee on Infectious Diseases. 32nd ed. Itasca: American Academy of Pediatrics; 2021. [Google Scholar]
- 57.El-Gohary M, van Zuuren EJ, Fedorowicz Z, et al. Topical antifungal treatments for tinea cruris and tinea corporis. Cochrane Database Syst Rev 2014;8:CD009992. 10.1002/14651858.CD009992.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.van Zuuren EJ, Fedorowicz Z, El-Gohary M. Evidence-based topical treatments for tinea cruris and tinea corporis: a summary of a cochrane systematic review. Br J Dermatol 2015;172(3):616–41. 10.1111/bjd.13441. [DOI] [PubMed] [Google Scholar]
- 59.Rotta I, Ziegelmann PK, Otuki MF, Riveros BS, Bernardo NL, Correr CJ. Efficacy of topical antifungals in the treatment of dermatophytosis: a mixed-treatment comparison meta-analysis involving 14 treatments. JAMA Dermatol 2013;149(3):341–9. 10.1001/jamadermatol.2013.1721. [DOI] [PubMed] [Google Scholar]
- 60.Greenberg HL, Shwayder TA, Bieszk N, Fivenson DP. Clotrimazole/betamethasone diproprionate: a review of costs and complications in the treatment of common cutaneous fungal infections. Pediatr Dermatol 2002;19(1):78–81. 10.1046/j.1525-1470.2002.00027.x. [DOI] [PubMed] [Google Scholar]
- 61.Rosen T, Elewski BE. Failure of clotrimazole-betamethasone dipropionate cream in treatment of Microsporum canis infections. J Am Acad Dermatol 1995;32(6):1050–1. 10.1016/0190-9622(95)91360-2. [DOI] [PubMed] [Google Scholar]
- 62.Alston SJ, Cohen BA, Braun M. Persistent and recurrent tinea corporis in children treated with combination antifungal/corticosteroid agents. Pediatrics. 2003;111(1):201–3. 10.1542/peds.111.1.201. [DOI] [PubMed] [Google Scholar]
- 63.Cole GW, Stricklin G. A comparison of a new oral antifungal, terbinafine, with griseofulvin as therapy for tinea corporis. Arch Dermatol 1989;125(11):1537–9. [PubMed] [Google Scholar]
- 64.Voravutinon V Oral treatment of tinea corporis and tinea cruris with terbinafine and griseofulvin: a randomized double blind comparative study. J Med Assoc Thai 1993;76(7):388–93. [PubMed] [Google Scholar]
- 65.Lal NR, Basu D, Saha A, Ghosh R, Verma R, Bandyopadhyay D. Efficacy and safety of 250 mg versus 500 mg oral terbinafine in the treatment of tinea corporis and cruris: a randomised, assessor-blinded comparative study. Indian J Dermatol Venereol Leprol 2023;89(5):665–71. 10.25259/IJDVL_74_2022. [DOI] [PubMed] [Google Scholar]
- 66.Bourlond A, Lachapelle JM, Aussems J, et al. Double-blind comparison of itraconazole with griseofulvin in the treatment of tinea corporis and tinea cruris. Int J Dermatol 1989;28(6):410–2. 10.1111/j.1365-4362.1989.tb02491.x. [DOI] [PubMed] [Google Scholar]
- 67.Panagiotidou D, Kousidou T, Chaidemenos G, et al. A comparison of itraconazole and griseofulvin in the treatment of tinea corporis and tinea cruris: a double-blind study. J Int Med Res 1992;20(5):392–400. 10.1177/030006059202000504. [DOI] [PubMed] [Google Scholar]
- 68.Khurana A, Agarwal A, Agrawal D, et al. Effect of different itraconazole dosing regimens on cure rates, treatment duration, safety, and relapse rates in adult patients with Tinea corporis/cruris: a randomized clinical trial. JAMA Dermatol 2022;158(11):1269–78. 10.1001/jamadermatol.2022.3745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Farag A, Taha M, Halim S. One-week therapy with oral terbinafine in cases of tinea cruris/corporis. Br J Dermatol 1994;131(5):684–6. 10.1111/j.1365-2133.1994.tb04983.x. [DOI] [PubMed] [Google Scholar]
- 70.Hawkins DM, Smidt AC. Superficial fungal infections in children. Pediatr Clin N Am 2014;61(2):443–55. 10.1016/j.pcl.2013.12.003. [DOI] [PubMed] [Google Scholar]
- 71.Gupta AK, Friedlander SF, Simkovich AJ. Tinea capitis: an update. Pediatr Dermatol 2022;39(2):167–72. 10.1111/pde.14925. [DOI] [PubMed] [Google Scholar]
- 72.Hillary T, Suys E. An outbreak of tinea capitis in elderly patients. Int J Dermatol 2014;53(2):e101–3. 10.1111/j.1365-4632.2012.05817.x. [DOI] [PubMed] [Google Scholar]
- 73.Takwale A, Agarwal S, Holmes SC, Berth-Jones J. Tinea capitis in two elderly women: transmission at the hairdresser. Br J Dermatol 2001;144(4):898–900. 10.1046/j.13652133.2001.04154.x. [DOI] [PubMed] [Google Scholar]
- 74.Cremer G, Bournerias I, Vandemeleubroucke E, Houin R, Revuz J. Tinea capitis in adults: misdiagnosis or reappearance? Dermatology. 1997;194(1):8–11. 10.1159/000246048. [DOI] [PubMed] [Google Scholar]
- 75.Elsaie ML. Update on Tinea capitis diagnosis and treatment. Cutis. 2022;110(5):238–40. 10.12788/cutis.0648. [DOI] [PubMed] [Google Scholar]
- 76.Gupta AK, Mays RR, Versteeg SG, et al. Tinea capitis in children: a systematic review of management. J Eur Acad Dermatol Venereol 2018;32(12):2264–74. 10.1111/jdv.15088. [DOI] [PubMed] [Google Scholar]
- 77.Le M, Gabrielli S, Ghazawi FM, Alkhodair R, Sheppard DC, Jafarian F. Efficacies and merits of the cotton swab technique for diagnosing tinea capitis in the pediatric population. J Am Acad Dermatol 2020;83(3):920–2. 10.1016/j.jaad.2020.01.009. [DOI] [PubMed] [Google Scholar]
- 78.Nasir S, Ralph N, O’Neill C, Cunney R, Lenane P, O’Donnell B. Trends in tinea capitis in an Irish pediatric population and a comparison of scalp brushings versus scalp scrapings as methods of investigation. Pediatr Dermatol 2014;31(5):622–3. 10.1111/pde.12093. [DOI] [PubMed] [Google Scholar]
- 79.Wahbah HR, Atallah RB, Eldahshan RM, Elsaie ML. A prospective clinical and trichoscopic study of tinea capitis in children during treatment. Dermatol Ther 2022;35(7): e15582. 10.1111/dth.15582. [DOI] [PubMed] [Google Scholar]
- 80.Wiegand C, Mugisha P, Mulyowa GK, et al. Identification of the causative dermatophyte of tinea capitis in children attending Mbarara Regional Referral Hospital in Uganda by PCR-ELISA and comparison with conventional mycological diagnostic methods. Med Mycol 2017;55(6):660–8. 10.1093/mmy/myw112. [DOI] [PubMed] [Google Scholar]
- 81.Ndiaye M, Sacheli R, Diongue K, et al. Evaluation of the Multiplex Real-Time PCR DermaGenius® Assay for the detection of dermatophytes in hair samples from Senegal. J Fungi (Basel). 2021;8(1):11. 10.3390/jof8010011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Brillowska-Dabrowska A, Michałek E, Saunte DM, Nielsen SS, Arendrup MC. PCR test for Microsporum canis identification. Med Mycol 2013;51(6):576–9. 10.3109/13693786.2012.755741. [DOI] [PubMed] [Google Scholar]
- 83.Wei LW, Qiao JJ. Mini-review: the diagnostic methods of Tinea capitis. Mycopathologia. 2023;188(5):563–9. 10.1007/s11046-023-00731-3. [DOI] [PubMed] [Google Scholar]
- 84.Veasey JV, Meneses OMS, da Silva FO. Reflectance confocal microscopy of tinea capitis: comparing images with results of dermoscopy and mycological exams. Int J Dermatol 2019;58(7):849–51. 10.1111/ijd.14241. [DOI] [PubMed] [Google Scholar]
- 85.Hughes R, Chiaverini C, Bahadoran P, Lacour JP. Corkscrew hair: a new dermoscopic sign for diagnosis of tinea capitis in black children. Arch Dermatol 2011;147(3):355–6. 10.1001/archdermatol.2011.31. [DOI] [PubMed] [Google Scholar]
- 86.Mapelli ET, Gualandri L, Cerri A, Menni S. Comma hairs in tinea capitis: a useful dermatoscopic sign for diagnosis of tinea capitis. Pediatr Dermatol 2012;29(2):223–4. 10.1111/j.1525-1470.2011.01598.x. [DOI] [PubMed] [Google Scholar]
- 87.Slowinska M, Rudnicka L, Schwartz RA, et al. Comma hairs: a dermatoscopic marker for tinea capitis: a rapid diagnostic method. J Am Acad Dermatol 2008;59(5 Suppl):S77–9. 10.1016/j.jaad.2008.07.009. [DOI] [PubMed] [Google Scholar]
- 88.Ekiz O, Sen BB, Rifaioğlu EN, Balta I. Trichoscopy in paediatric patients with tinea capitis: a useful method to differentiate from alopecia areata. J Eur Acad Dermatol Venereol 2014;28(9):1255–8. 10.1111/jdv.12246. [DOI] [PubMed] [Google Scholar]
- 89.Fuller LC, Barton RC, Mohd Mustapa MF, Proudfoot LE, Punjabi SP, Higgins EM. British Association of Dermatologists’ guidelines for the management of tinea capitis 2014. Br J Dermatol 2014;171(3):454–63. 10.1111/bjd.13196. [DOI] [PubMed] [Google Scholar]
- 90.Elewski BE, Hughey LC, Sobera JO. Fungal diseases. In: Bolognia JL, Jorizzo JL, Schaffer JV, editors. Dermatology, 3rd ed., Vol 2. Elsevier Limited; 2012. p. 1251. [Google Scholar]
- 91.Chen X, Jiang X, Yang M, et al. Systemic antifungal therapy for tinea capitis in children. Cochrane Database Syst Rev 2016;2016(5):CD004685. 10.1002/14651858.CD004685.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Elewski BE, Cáceres HW, DeLeon L, et al. Terbinafine hydrochloride oral granules versus oral griseofulvin suspension in children with tinea capitis: results of two randomized, investigatorblinded, multicenter, international, controlled trials. J Am Acad Dermatol 2008;59(1):41–54. 10.1016/j.jaad.2008.02.019. [DOI] [PubMed] [Google Scholar]
- 93.Tey HL, Tan AS, Chan YC. Meta-analysis of randomized, controlled trials comparing griseofulvin and terbinafine in the treatment of tinea capitis. J Am Acad Dermatol 2011;64(4):663–70. 10.1016/j.jaad.2010.02.048. [DOI] [PubMed] [Google Scholar]
- 94.Elewski BE. Tinea capitis: a current perspective. J Am Acad Dermatol 2000;42(1 Pt 1):1–24. 10.1016/s01909622(00)90001-x. [DOI] [PubMed] [Google Scholar]
- 95.Foster KW, Friedlander SF, Panzer H, Ghannoum MA, Elewski BE. A randomized controlled trial assessing the efficacy of fluconazole in the treatment of pediatric tinea capitis. J Am Acad Dermatol 2005;53(5):798–809. 10.1016/j.jaad.2005.07.028. [DOI] [PubMed] [Google Scholar]
- 96.López-Gómez S, Del Palacio A, Van Cutsem J, Soledad Cuétara M, Iglesias L, Rodriguez-Noriega A. Itraconazole versus griseofulvin in the treatment of tinea capitis: a double-blind randomized study in children. Int J Dermatol 1994;33(10):743–7. 10.1111/j.1365-4362.1994.tb01525.x. [DOI] [PubMed] [Google Scholar]
- 97.Jahangir M, Hussain I, Ul Hasan M, Haroon TS. A double-blind, randomized, comparative trial of itraconazole versus terbinafine for 2 weeks in tinea capitis. Br J Dermatol 1998;139(4):672–4. 10.1046/j.1365-2133.1998.02465.x. [DOI] [PubMed] [Google Scholar]
- 98.Gupta AK, Adam P, Dlova N, et al. Therapeutic options for the treatment of tinea capitis caused by Trichophyton species: griseofulvin versus the new oral antifungal agents, terbinafine, itraconazole, and fluconazole. Pediatr Dermatol 2001;18(5):433–8. 10.1046/j.1525-1470.2001.01978.x. [DOI] [PubMed] [Google Scholar]
- 99.Chen C, Koch LH, Dice JE, et al. A randomized, double-blind study comparing the efficacy of selenium sulfide shampoo 1% and ciclopirox shampoo 1% as adjunctive treatments for tinea capitis in children. Pediatr Dermatol 2010;27(5):459–62. 10.1111/j.1525-1470.2010.01093.x. [DOI] [PubMed] [Google Scholar]
- 100.Greer DL. Successful treatment of tinea capitis with 2% ketoconazole shampoo. Int J Dermatol 2000;39(4):302–4. 10.1046/j.1365-4362.2000.00885.x. [DOI] [PubMed] [Google Scholar]
- 101.Winge MC, Chryssanthou E, Wahlgren CF. Combs and hair-trimming tools as reservoirs for dermatophytes in juvenile tinea capitis. Acta Derm Venereol 2009;89(5):536–7. 10.2340/00015555-0694. [DOI] [PubMed] [Google Scholar]
- 102.Uhrlaß S, Verma SB, Gräser Y, et al. Trichophyton indotineae-an emerging pathogen causing recalcitrant dermatophytoses in india and worldwide—a multidimensional perspective. J Fungi (Basel). 2022;8(7):757. 10.3390/jof8070757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Singal A, Jakhar D, Kaur I, Pandhi D, Das S. Tinea Pseudoimbricata as a unique manifestation of steroid abuse: a clinico-mycological and dermoscopic study from a Tertiary Care Hospital. Indian Dermatol Online J 2019;10(4):422–5. 10.4103/idoj.IDOJ_385_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Nowowiejska J, Baran A, Flisiak I. Tinea incognito-a great physician pitfall. J Fungi (Basel). 2022;8(3):312. 10.3390/jof8030312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Piccolo V, Corneli P, Russo T, Zalaudek I, Alfano R, Argenziano G. Dermoscopy as a useful tool in diagnosis of tinea incognito. Int J Dermatol 2019;58(2):e32–4. 10.1111/ijd.14278. [DOI] [PubMed] [Google Scholar]
- 106.Al Hasan M, Fitzgerald SM, Saoudian M, Krishnaswamy G. Dermatology for the practicing allergist: Tinea pedis and its complications. Clin Mol Allergy. 2004;2(1):5. 10.1186/1476-7961-2-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Perea S, Ramos MJ, Garau M, Gonzalez A, Noriega AR, del Palacio A. Prevalence and risk factors of tinea unguium and tinea pedis in the general population in Spain. J Clin Microbiol 2000;38(9):3226–30. 10.1128/JCM.38.9.3226-3230.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Rogers D, Kilkenny M, Marks R. The descriptive epidemiology of tinea pedis in the community. Australas J Dermatol 1996;37(4):178–84. 10.1111/j.1440-0960.1996.tb01049.x. [DOI] [PubMed] [Google Scholar]
- 109.Panackal AA, Halpern EF, Watson AJ. Cutaneous fungal infections in the United States: Analysis of the National Ambulatory Medical Care Survey (NAMCS) and National Hospital Ambulatory Medical Care Survey (NHAMCS), 1995–2004. Int J Dermatol 2009;48(7):704–12. 10.1111/j.1365-4632.2009.04025.x. [DOI] [PubMed] [Google Scholar]
- 110.Moseley I, Ragi SD, Ouellette S, Rao B. Tinea pedis in under-represented groups: all of Us database analysis. Mycoses. 2023;66(1):29–34. 10.1111/myc.13522. [DOI] [PubMed] [Google Scholar]
- 111.Nowicka D, Nawrot U. Tinea pedis—an embarrassing problem for health and beauty—a narrative review. Mycoses. 2021;64(10):1140–50. 10.1111/myc.13340. [DOI] [PubMed] [Google Scholar]
- 112.Ilkit M, Durdu M. Tinea pedis: the etiology and global epidemiology of a common fungal infection. Crit Rev Microbiol 2015;41(3):374–88. 10.3109/1040841X.2013.856853. [DOI] [PubMed] [Google Scholar]
- 113.Leung AK, Barankin B, Lam JM, Leong KF, Hon KL. Tinea pedis: an updated review. Drugs Context. 2023. 10.7573/dic.2023-5-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Rinaldi MG. Dermatophytosis: epidemiological and microbiological update. J Am Acad Dermatol 2000;43(5 Suppl):S120–4. 10.1067/mjd.2000.110378. [DOI] [PubMed] [Google Scholar]
- 115.Diseases Hygiene-related. Centers for Disease Control and Prevention. https://www.cdc.gov/hygiene/disease/index.html. Accessed 6 Nov 2023. [Google Scholar]
- 116.Gupta AK, Cooper EA, Ryder JE, Nicol KA, Chow M, Chaudhry MM. Optimal management of fungal infections of the skin, hair, and nails. Am J Clin Dermatol 2004;5(4):225–37. 10.2165/00128071-200405040-00003. [DOI] [PubMed] [Google Scholar]
- 117.Lipner SR, Scher RK. Onychomycosis: treatment and prevention of recurrence. J Am Acad Dermatol 2019;80(4):853–67. 10.1016/j.jaad.2018.05.1260. [DOI] [PubMed] [Google Scholar]
- 118.Crawford F, Hollis S. Topical treatments for fungal infections of the skin and nails of the foot. Cochrane Database Syst Rev 2007;2007(3): CD001434. 10.1002/14651858.CD001434.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Ward H, Parkes N, Smith C, Kluzek S, Pearson R. Consensus for the treatment of Tinea pedis: a systematic review of randomised controlled trials. J Fungi (Basel). 2022;8(4):351. 10.3390/jof8040351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Borelli C, Klövekorn G, Ernst TM, Bödeker RH, Korting HC, Neumeister C. Comparative study of 2% sertaconazole solution and cream formulations in patients with tinea corporis, tinea pedis interdigitalis, or a corresponding candidosis. Am J Clin Dermatol 2007;8(6):371–8. 10.2165/00128071-200708060-00007. [DOI] [PubMed] [Google Scholar]
- 121.Jarratt M, Jones T, Adelglass J, et al. Efficacy and safety of oncedaily luliconazole 1% cream in patients ≥12 years of age with interdigital tinea pedis: a phase 3, randomized, double-blind, vehicle-controlled study. J Drugs Dermatol 2014;13(7):838–46. [PubMed] [Google Scholar]
- 122.Elewski BE, Vlahovic TC. Econazole nitrate foam 1% for the treatment of tinea pedis: results from two double-blind, vehicle-controlled, phase 3 clinical trials. J Drugs Dermatol 2014;13(7):803–8. [PubMed] [Google Scholar]
- 123.Bell-Syer SE, Hart R, Crawford F, Torgerson DJ, Tyrrell W, Russell I. Oral treatments for fungal infections of the skin of the foot. Cochrane Database Syst Rev 2002;2:CD003584. 10.1002/14651858.CD003584. [DOI] [PubMed] [Google Scholar]
- 124.Echuri H, Sivesind TE, Dellavalle RP. From the cochrane library: oral treatments for fungal infections of the skin of the foot. J Am Acad Dermatol 2022;87(5):e183–4. 10.1016/j.jaad.2022.07.006. [DOI] [PubMed] [Google Scholar]
- 125.Ishack S, Miller RC, Lipner SR. Insights into the “fear factor” regarding terbinafine-associated hepatoxicity in an assessment of online information. J Am Acad Dermatol 2022;86(6):e269–71. 10.1016/j.jaad.2022.02.006. [DOI] [PubMed] [Google Scholar]
- 126.Wang Y, Geizhals S, Lipner SR. Retrospective analysis of laboratory abnormalities in patients prescribed terbinafine for onychomycosis. J Am Acad Dermatol 2021;84(2):497–9. 10.1016/j.jaad.2020.04.172. [DOI] [PubMed] [Google Scholar]
- 127.Wang Y, Lipner SR. Retrospective analysis of abnormal laboratory test results in pediatric patients prescribed terbinafine for superficial fungal infections. J Am Acad Dermatol 2021;85(4):1042–4. 10.1016/j.jaad.2021.01.073. [DOI] [PubMed] [Google Scholar]
- 128.Wang Y, Lipner SR. Retrospective analysis of laboratory abnormalities in patients with preexisting liver and hematologic diseases prescribed terbinafine for onychomycosis. J Am Acad Dermatol 2021;84(1):220–1. 10.1016/j.jaad.2020.09.004. [DOI] [PubMed] [Google Scholar]
- 129.Kang R, Lipner S. Consumer preferences of antifungal products for treatment and prevention of tinea pedis. J Dermatol Treat. 2019;30(8):745–9. 10.1080/09546634.2019.1572862. [DOI] [PubMed] [Google Scholar]
- 130.Kara Polat A, Akın Belli A, Göre Karaali M, Koku Aksu AE. The attitudes, behaviors, and opinions about non-pharmacological agents in patients with tinea pedis. Dermatol Ther 2020;33(6): e14041. 10.1111/dth.14041. [DOI] [PubMed] [Google Scholar]
- 131.Kelly S, Liu D, Wang T, Rajpara A, Franano C, Aires D. Vinegar sock soak for tinea pedis or onychomycosis. J Am Acad Dermatol 2023;89(1): e23. 10.1016/j.jaad.2017.09.043. [DOI] [PubMed] [Google Scholar]
- 132.Tong MM, Altman PM, Barnetson RS. Tea tree oil in the treatment of tinea pedis. Australas J Dermatol 1992;33(3):145–9. 10.1111/j.1440-0960.1992.tb00103.x. [DOI] [PubMed] [Google Scholar]
- 133.Saoji V, Madke B. Efficacy of salicylic acid peel in dermatophytosis. Indian J Dermatol Venereol Leprol 2021;87(5):671–5. 10.4103/ijdvl.IJDVL_853_18. [DOI] [PubMed] [Google Scholar]
- 134.Chamorro MJ, House SA. Tinea manuum. In: StatPearls. Treasure Island: StatPearls Publishing; 2023. [PubMed] [Google Scholar]
- 135.Sahoo AK, Mahajan R. Management of tinea corporis, tinea cruris, and tinea pedis: a comprehensive review. Indian Dermatol Online J 2016;7(2):77–86. 10.4103/2229-5178.178099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Dogra S, Ramam M. Difficult dermatophytosis. JAMA Dermatol 2022;158(11):1243–4. 10.1001/jamadermatol.2022.3736. [DOI] [PubMed] [Google Scholar]
- 137.Sentamilselvi G, Kamalam A, Ajithadas K, Janaki C, Thambiah AS. Scenario of chronic dermatophytosis: an Indian study. Mycopathologia. 1997;140(3):129–35. 10.1023/a:1006843418759. [DOI] [PubMed] [Google Scholar]
- 138.Das A, Fatima F, Sil A, Podder I, Jafferany M. Therapeutic management of psychological morbidity and impaired quality of life in patients with persistent dermatophytoses. Dermatol Ther 2020;33(6): e14124. 10.1111/dth.14124. [DOI] [PubMed] [Google Scholar]
- 139.Kershenovich R, Sherman S, Reiter O, et al. A unique clinicopathological manifestation of fungal infection: a case series of deep dermatophytosis in immunosuppressed patients. Am J Clin Dermatol 2017;18(5):697–704. 10.1007/s40257-017-0276-y. [DOI] [PubMed] [Google Scholar]
- 140.Ilkit M, Durdu M, Karakaş M. Majocchi’s granuloma: a symptom complex caused by fungal pathogens. Med Mycol 2012;50(5):449–57. 10.3109/13693786.2012.669503. [DOI] [PubMed] [Google Scholar]
- 141.Rouzaud C, Chosidow O, Brocard A, et al. Severe dermatophytosis in solid organ transplant recipients: a French retrospective series and literature review. Transpl Infect Dis 2018. 10.1111/tid.12799. [DOI] [PubMed] [Google Scholar]
- 142.Lowinger-Seoane M, Torres-Rodríguez JM, Madrenys-Brunet N, Aregall-Fusté S, Saballs P. Extensive dermatophytoses caused by Trichophyton mentagrophytes and Microsporum canis in a patient with AIDS. Mycopathologia. 1992;120(3):143–6. 10.1007/BF00436391. [DOI] [PubMed] [Google Scholar]
- 143.del Palacio A, Pereiro-Miguens M, Gimeno C, et al. Widespread dermatophytosis due to Microsporum (Trichophyton) gallinae in a patient with AIDS—a case report from Spain. Clin Exp Dermatol 1992;17(6):449–53. 10.1111/j.1365-2230.1992.tb00258.x. [DOI] [PubMed] [Google Scholar]
- 144.Grossman ME, Pappert AS, Garzon MC, Silvers DN. Invasive Trichophyton rubrum infection in the immunocompromised host: report of three cases. J Am Acad Dermatol 1995;33(2 Pt 1):315–8. 10.1016/0190-9622(95)90270-8. [DOI] [PubMed] [Google Scholar]
- 145.Engelhard D, Or R, Naparstek E, Leibovici V. Treatment with itraconazole of widespread tinea corporis due to Trichophyton rubrum in a bone marrow transplant recipient. Bone Marrow Transplant. 1988;3(5):517–9. [PubMed] [Google Scholar]
- 146.Machan M, Kestenbaum T, Fraga GR. Diffuse hyperkeratosis in a deaf and blind 48-year-old woman-quiz case Diagnosis: keratitis-ichthyosis-deafness (KID) syndrome with secondary dermatophytosis. Arch Dermatol 2012;148(10):1199–200. 10.1001/archderm.148.10.1199-c. [DOI] [PubMed] [Google Scholar]
- 147.Jachiet M, Lanternier F, Rybojad M, et al. Posaconazole treatment of extensive skin and nail dermatophytosis due to autosomal recessive deficiency of CARD9. JAMA Dermatol 2015;151(2):192–4. 10.1001/jamadermatol.2014.2154. [DOI] [PubMed] [Google Scholar]
- 148.Budihardja D, Freund V, Mayser P. Widespread erosive tinea corporis by Arthroderma benhamiae in a renal transplant recipient: case report. Mycoses. 2010;53(6):530–2. 10.1111/j.1439-0507.2009.01736.x. [DOI] [PubMed] [Google Scholar]
- 149.Jabet A, Dellière S, Seang S, et al. Sexually transmitted Trichophyton mentagrophytes genotype VII infection among men who have sex with men. Emerg Infect Dis 2023;29(7):1411–4. 10.3201/eid2907.230025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Kupsch C, Czaika VA, Deutsch C, Gräser Y. Trichophyton mentagrophytes—a new genotype of zoophilic dermatophyte causes sexually transmitted infections. J Dtsch Dermatol Ges 2019;17(5):493–501. 10.1111/ddg.13776. [DOI] [PubMed] [Google Scholar]
- 151.Lanternier F, Pathan S, Vincent QB, et al. Deep dermatophytosis and inherited CARD9 deficiency. N Engl J Med 2013;369(18):1704–14. 10.1056/NEJMoa1208487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Rajpara V, Frankel S, Rogers C, Nouri K. Trichophyton tonsurans associated tinea corporis infection with the development of Majocchi’s granuloma in a renal transplant patient. J Drugs Dermatol 2005;4(6):767–9. [PubMed] [Google Scholar]
- 153.Steiner UC, Trüeb RM, Schad K, et al. Trichophyton rubruminduced Majocchi’s granuloma in a heart transplant recipient. A therapeutic challenge. J Dermatol Case Rep 2012;6(3):70–2. 10.3315/jdcr.2012.1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Voisard JJ, Weill FX, Beylot-Barry M, Vergier B, Dromer C, Beylot C. Dermatophytic granuloma caused by Microsporum canis in a heart-lung recipient. Dermatology. 1999;198(3):317–9. 10.1159/000018141. [DOI] [PubMed] [Google Scholar]
- 155.Romero FA, Deziel PJ, Razonable RR. Majocchi’s granuloma in solid organ transplant recipients. Transpl Infect Dis 2011;13(4):424–32. 10.1111/j.1399-3062.2010.00596.x. [DOI] [PubMed] [Google Scholar]
- 156.Smith KJ, Welsh M, Skelton H. Trichophyton rubrum showing deep dermal invasion directly from the epidermis in immunosuppressed patients. Br J Dermatol 2001;145(2):344–8. 10.1046/j.1365-2133.2001.04331.x. [DOI] [PubMed] [Google Scholar]
- 157.Demidovich CW, Kornfeld BW, Gentry RH, Fitzpatrick JE. Deep dermatophyte infection with chronic draining nodules in an immunocompromised patient. Cutis. 1995;55(4):237–40. [PubMed] [Google Scholar]
- 158.Swart E, Smit FJ. Trichophyton violaceum abscesses. Br J Dermatol 1979;101(2):177–84. 10.1111/j.1365-2133.1979.tb05605.x. [DOI] [PubMed] [Google Scholar]
- 159.Nir-Paz R, Elinav H, Pierard GE, et al. Deep infection by Trichophyton rubrum in an immunocompromised patient. J Clin Microbiol 2003;41(11):5298–301. 10.1128/JCM.41.11.5298-5301.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Seddon ME, Thomas MG. Invasive disease due to Epidermophyton floccosum in an immunocompromised patient with Behçet’s syndrome. Clin Infect Dis 1997;25(1):153–4. 10.1086/516887. [DOI] [PubMed] [Google Scholar]
- 161.Lowther AL, Somani AK, Camouse M, Florentino FT, Somach SC. Invasive Trichophyton rubrum infection occurring with infliximab and long-term prednisone treatment. J Cutan Med Surg 2007;11(2):84–8. 10.2310/7750.2007.00009. [DOI] [PubMed] [Google Scholar]
- 162.Kwon KS, Jang HS, Son HS, et al. Widespread and invasive Trichophyton rubrum infection mimicking Kaposi’s sarcoma in a patient with AIDS. J Dermatol 2004;31(10):839–43. 10.1111/j.1346-8138.2004.tb00612.x. [DOI] [PubMed] [Google Scholar]
- 163.Muñoz-Pèrez MA, Rodriguez-Pichardo A, Camacho F, Rios JJ. Extensive and deep dermatophytosis caused by Trichophyton mentagrophytes var. interdigitalis in an HIV-1 positive patient. J Eur Acad Dermatol Venereol 2000;14(1):61–3. 10.1046/j.1468-3083.2000.00015.x. [DOI] [PubMed] [Google Scholar]
- 164.Tsang P, Hopkins T, Jimenez-Lucho V. Deep dermatophytosis caused by Trichophyton rubrum in a patient with AIDS. J Am Acad Dermatol 1996;34(6):1090–1. 10.1016/s0190-9622(96)90298-4. [DOI] [PubMed] [Google Scholar]
- 165.Tateishi Y, Sato H, Akiyama M, et al. Severe generalized deep dermatophytosis due to Trichophyton rubrum (trichophytic granuloma) in a patient with atopic dermatitis. Arch Dermatol 2004;140(5):624–5. 10.1001/archderm.140.5.624. [DOI] [PubMed] [Google Scholar]
- 166.Erbağci Z Deep dermatophytoses in association with atopy and diabetes mellitus: Majocchi’s granuloma tricophyticum or dermatophytic pseudomycetoma? Mycopathologia. 2002;154(4):163–9. 10.1023/a:1016328001146. [DOI] [PubMed] [Google Scholar]
- 167.Peixoto I, Maquine G, Francesconi VA, Francesconi F. Dermatophytosis caused by Tricophyton rubrum as an opportunistic infection in patients with Cushing disease. An Bras Dermatol 2010;85(6):888–90. 10.1590/s0365-05962010000600017. [DOI] [PubMed] [Google Scholar]
- 168.Baker RL, Para MF. Successful use of ketoconazole for invasive cutaneous Trichophyton rubrum infection. Arch Intern Med 1984;144(3):615–7. [PubMed] [Google Scholar]
- 169.Elewski BE, Sullivan J. Dermatophytes as opportunistic pathogens. J Am Acad Dermatol 1994;30(6):1021–2. 10.1016/s0190-9622(09)80146-1. [DOI] [PubMed] [Google Scholar]
- 170.Faergemann J, Gisslén H, Dahlberg E, Westin J, Roupe G. Trichophyton rubrum abscesses in immunocompromised patients. A case report. Acta Derm Venereol 1989;69(3):244–7. [PubMed] [Google Scholar]
- 171.Lestringant GG, Lindley SK, Hillsdon-Smith J, Bouix G. Deep dermatophytosis to Trichophyton rubrum and T. verrucosum in an immunosuppressed patient. Int J Dermatol 1988;27(10):707–9. 10.1111/j.1365-4362.1988.tb01269.x. [DOI] [PubMed] [Google Scholar]
- 172.Marconi VC, Kradin R, Marty FM, Hospenthal DR, Kotton CN. Disseminated dermatophytosis in a patient with hereditary hemochromatosis and hepatic cirrhosis: case report and review of the literature. Med Mycol 2010;48(3):518–27. 10.3109/13693780903213512. [DOI] [PubMed] [Google Scholar]
- 173.Gupta S, Kumar B, Radotra BD, Rai R. Majocchi’s granuloma trichophyticum in an immunocompromised patient. Int J Dermatol 2000;39(2):140–1. 10.1046/j.1365-4362.2000.00894.x. [DOI] [PubMed] [Google Scholar]
- 174.Wu LC, Sun PL, Chang YT. Extensive deep dermatophytosis cause by Trichophyton rubrum in a patient with liver cirrhosis and chronic renal failure. Mycopathologia. 2013;176(5–6):457–62. 10.1007/s11046-013-9696-2. [DOI] [PubMed] [Google Scholar]
- 175.Wang R, Huang C, Zhang Y, Li R. Invasive dermatophyte infection: a systematic review. Mycoses. 2021;64(4):340–8. 10.1111/myc.13212. [DOI] [PubMed] [Google Scholar]
- 176.Boudghene Stambouli O, Amrani N, Boudghéne Stambouli K, Bouali F. Dermatophytic disease with deficit in CARD9: a new case with a brain impairment. J Mycol Med 2017;27(2):250–3. 10.1016/j.mycmed.2017.01.001. [DOI] [PubMed] [Google Scholar]
- 177.Simonsen JK, Mose KF, Kristensen L, Herlin LK. Deep dermatophytosis in an immunocompetent adult with no prior history of skin disease. Med Mycol Case Rep 2023;39:31–3. 10.1016/j.mmcr.2023.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Smith KJ, Neafie RC, Skelton HG 3rd, Barrett TL, Graham JH, Lupton GP. Majocchi’s granuloma. J Cutan Pathol 1991;18(1):28–35. 10.1111/j.1600-0560.1991.tb00598.x. [DOI] [PubMed] [Google Scholar]
- 179.Allen GE. Majocchi’s granuloma. Br J Dermatol 1966;78(10):544. [PubMed] [Google Scholar]
- 180.Radentz WH, Yanase DJ. Papular lesions in an immunocompromised patient. Trichophyton rubrum granulomas (Majocchi’s granuloma). Arch Dermatol 1993;129(9):1189–93. 10.1001/archderm.1993.01680300119022. [DOI] [PubMed] [Google Scholar]
- 181.Lepage JC. Source of Majocchi’s granuloma. J Am Acad Dermatol 1983;8(2):260. 10.1016/s0190-9622(83)80194-7. [DOI] [PubMed] [Google Scholar]
- 182.Sequeira M, Burdick AE, Elgart GW, Berman B. New-onset Majocchi’s granuloma in two kidney transplant recipients under tacrolimus treatment. J Am Acad Dermatol 1998;38(3):486–8. 10.1016/s0190-9622(98)70511-0. [DOI] [PubMed] [Google Scholar]
- 183.Gupta MA, Gupta AK. Psychiatric and psychological co-morbidity in patients with dermatologic disorders: epidemiology and management. Am J Clin Dermatol 2003;4(12):833–42. 10.2165/00128071-200304120-00003. [DOI] [PubMed] [Google Scholar]
- 184.Narang T, Bhattacharjee R, Singh S, et al. Quality of life and psychological morbidity in patients with superficial cutaneous dermatophytosis. Mycoses. 2019;62(8):680–5. 10.1111/myc.12930. [DOI] [PubMed] [Google Scholar]
- 185.Patro N, Panda M, Jena AK. The menace of superficial dermatophytosis on the quality of life of patients attending referral hospital in eastern India: a cross-sectional observational study. Indian Dermatol Online J 2019;10(3):262–6. 10.4103/idoj.IDOJ_342_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Mushtaq S, Faizi N, Amin SS, Adil M, Mohtashim M. Impact on quality of life in patients with dermatophytosis. Australas J Dermatol 2020;61(2):e184–8. 10.1111/ajd.13191. [DOI] [PubMed] [Google Scholar]
- 187.Fienemika AE, Okeafor CU. The identification and grading of the psychosocial impact of Tinea capitis in primary school children in a semi-urban area of Rivers State, Nigeria. Niger Postgrad Med J 2017;24(1):20–4. 10.4103/npmj.npmj_172_16. [DOI] [PubMed] [Google Scholar]
- 188.Rajagopalan M, Inamadar A, Mittal A, et al. Expert consensus on the management of dermatophytosis in India (ECTODERM India). BMC Dermatol 2018;18(1):6. 10.1186/s12895-018-0073-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Jones HE. Problems of resistant dermatophytes. J Am Acad Dermatol 1990;23(4 Pt 2):779–81. 10.1016/01909622(90)70287-r. [DOI] [PubMed] [Google Scholar]
- 190.Artis WM, Odle BM, Jones HE. Griseofulvin-resistant dermatophytosis correlates with in vitro resistance. Arch Dermatol 1981;117(1):16–9. [PubMed] [Google Scholar]
- 191.Bishnoi A, Vinay K, Dogra S. Emergence of recalcitrant dermatophytosis in India. Lancet Infect Dis 2018;18(3):250–1. 10.1016/S1473-3099(18)30079-3. [DOI] [PubMed] [Google Scholar]
- 192.Verma SB. Emergence of recalcitrant dermatophytosis in India. Lancet Infect Dis 2018;18(7):718–9. 10.1016/S1473-3099(18)30338-4. [DOI] [PubMed] [Google Scholar]
- 193.Mukherjee PK, Leidich SD, Isham N, Leitner I, Ryder NS, Ghannoum MA. Clinical Trichophyton rubrum strain exhibiting primary resistance to terbinafine. Antimicrob Agents Chemother. 2003;47(1):82–6. 10.1128/AAC.47.1.82-86.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Gupta AK, Venkataraman M, Hall DC, Cooper EA, Summerbell RC. The emergence of Trichophyton indotineae: implications for clinical practice. Int J Dermatol 2023;62(7):857–61. 10.1111/ijd.16362. [DOI] [PubMed] [Google Scholar]
- 195.Jabet A, Normand AC, Brun S, et al. Trichophyton indotineae, from epidemiology to therapeutic. J Mycol Med 2023;33(3): 101383. 10.1016/j.mycmed.2023.101383. [DOI] [PubMed] [Google Scholar]
- 196.Jabet A, Brun S, Normand AC, et al. Extensive dermatophytosis caused by terbinafine-resistant Trichophyton indotineae, France. Emerg Infect Dis 2022;28(1):229–33. 10.3201/eid2801.210883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field: first reported US cases of tinea caused by Trichophyton indotineae— New York City, December 2021–March 2023. MMWR Morb Mortal Wkly Rep 2023;72(19):536–7. 10.15585/mmwr.mm7219a4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Gupta YK, Ramachandran SS. Fixed dose drug combinations: issues and challenges in India. Indian J Pharmacol 2016;48(4):347–9. 10.4103/0253-7613.186200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Verma S, Madhu R. The Great Indian epidemic of superficial dermatophytosis: an appraisal. Indian J Dermatol 2017;62(3):227–36. 10.4103/ijd.IJD_206_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Singh A, Masih A, Khurana A, et al. High terbinafine resistance in Trichophyton interdigitale isolates in Delhi, India harbouring mutations in the squalene epoxidase gene. Mycoses. 2018;61(7):477–84. 10.1111/myc.12772. [DOI] [PubMed] [Google Scholar]
- 201.Kano R, Kimura U, Kakurai M, et al. Trichophyton indotineae sp. nov.: a new highly terbinafine-resistant anthropophilic dermatophyte species. Mycopathologia. 2020;185(6):947–58. 10.1007/s11046-020-00455-8. [DOI] [PubMed] [Google Scholar]
- 202.Tang C, Kong X, Ahmed SA, et al. Taxonomy of the Trichophyton mentagrophytes/T. interdigitale species complex harboring the highly virulent Multiresistant Genotype T. indotineae. Mycopathologia. 2021;186(3):315–26. 10.1007/s11046-021-00544-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Gupta AK, Venkataraman M. Antifungal resistance in superficial mycoses. J Dermatol Treat. 2022;33(4):1888–95. 10.1080/09546634.2021.1942421. [DOI] [PubMed] [Google Scholar]
- 204.Caplan AS, Zakhem GA, Pomeranz MK. Trichophyton mentagrophytes internal transcribed spacer genotype VIII. JAMA Dermatol 2023;159(10):1130. 10.1001/jamadermatol.2023.2645. [DOI] [PubMed] [Google Scholar]
- 205.Fattahi A, Shirvani F, Ayatollahi A, et al. Multidrug-resistant Trichophyton mentagrophytes genotype VIII in an Iranian family with generalized dermatophytosis: report of four cases and review of literature. Int J Dermatol 2021;60(6):686–92. 10.1111/ijd.15226. [DOI] [PubMed] [Google Scholar]
- 206.Nenoff P, Verma SB, Ebert A, et al. Spread of terbinafine-resistant Trichophyton mentagrophytesType VIII (India) in Germany- “The Tip of the Iceberg?”. J Fungi (Basel). 2020;6(4):207. 10.3390/jof6040207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Dellière S, Joannard B, Benderdouche M, et al. Emergence of difficult-to-treat tinea corporis caused by Trichophyton mentagrophytes complex isolates, Paris, France. Emerg Infect Dis 2022;28(1):224–8. 10.3201/eid2801.210810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Sacheli R, Hayette MP. Antifungal resistance in dermatophytes: genetic considerations, clinical presentations and alternative therapies. J Fungi (Basel). 2021;7(11):983. 10.3390/jof7110983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Klinger M, Theiler M, Bosshard PP. Epidemiological and clinical aspects of Trichophyton mentagrophytes/Trichophyton interdigitale infections in the Zurich area: a retrospective study using genotyping. J Eur Acad Dermatol Venereol 2021;35(4):1017–25. 10.1111/jdv.17106. [DOI] [PubMed] [Google Scholar]
- 210.Siopi M, Efstathiou I, Theodoropoulos K, Pournaras S, Meletiadis J. Molecular epidemiology and antifungal susceptibility of Trichophyton isolates in Greece: emergence of terbinafineresistant Trichophytonmentagrophytes type VIII locally and globally. J Fungi (Basel). 2021;7(6):419. 10.3390/jof7060419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Astvad KMT, Hare RK, Jørgensen KM, Saunte DML, Thomsen PK, Arendrup MC. Increasing terbinafine resistance in Danish Trichophyton isolates 2019–2020. J Fungi (Basel). 2022;8(2):150. 10.3390/jof8020150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Kong X, Tang C, Singh A, et al. Antifungal susceptibility and mutations in the squalene epoxidase gene in dermatophytes of the Trichophyton mentagrophytes species complex. Antimicrob Agents Chemother. 2021;65(8): e0005621. 10.1128/AAC.00056-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Posso-De Los Rios CJ, Tadros E, Summerbell RC, Scott JA. Terbinafine resistant Trichophyton indotineae isolated in patients with superficial dermatophyte infection in Canadian patients. J Cutan Med Surg 2022;26(4):371–6. 10.1177/12034754221077891. [DOI] [PubMed] [Google Scholar]
- 214.Ngo TMC, Ton Nu PA, Le CC, Ha TNT, Do TBT, Tran TG. First detection of Trichophyton indotineae causing tinea corporis in Central Vietnam. Med Mycol Case Rep 2022;36:37–41. 10.1016/j.mmcr.2022.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Saunte DML, Pereiro-Ferreirós M, Rodríguez-Cerdeira C, et al. Emerging antifungal treatment failure of dermatophytosis in Europe: take care or it may become endemic. J Eur Acad Dermatol Venereol 2021;35(7):1582–6. 10.1111/jdv.17241. [DOI] [PubMed] [Google Scholar]
- 216.Cañete-Gibas CF, Mele J, Patterson HP, et al. Terbinafineresistant dermatophytes and the presence of Trichophyton indotineae in North America. J Clin Microbiol 2023;61(8): e0056223. 10.1128/jcm.00562-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.“Tinea gone wild: the emergence of Trichophyton indotineae as a global phenomenon. American Academy of Dermatology. https://www.aad.org/dw/dw-insights-and-inquiries/archi ve/2023/tinea-gone-wild-trichophyton-indotineae. Accessed 6 Nov 2023. [Google Scholar]
- 218.Verma SB, Panda S, Nenoff P, et al. The unprecedented epidemic-like scenario of dermatophytosis in India: II diagnostic methods and taxonomical aspects. Indian J Dermatol Venereol Leprol 2021;87(3):326–32. 10.25259/IJDVL_302_20. [DOI] [PubMed] [Google Scholar]
- 219.Singh S, Chandra U, Anchan VN, Verma P, Tilak R. Limited effectiveness of four oral antifungal drugs (fluconazole, griseofulvin, itraconazole and terbinafine) in the current epidemic of altered dermatophytosis in India: results of a randomized pragmatic trial. Br J Dermatol 2020;183(5):840–6. 10.1111/bjd.19146. [DOI] [PubMed] [Google Scholar]
- 220.Gueneau R, Joannard B, Haddad N, et al. Extensive dermatophytosis caused by terbinafine-resistant Trichophyton indotineae, successfully treated with topical voriconazole. Int J Antimicrob Agents. 2022;60(5–6): 106677. 10.1016/j.ijantimicag.2022.106677. [DOI] [PubMed] [Google Scholar]
- 221.Hassaan ZRAA Mohamed HAK, Eldahshan RM Elsaie ML. Comparison between the efficacy of terbinafine and itraconazole orally vs. the combination of the two drugs in treating recalcitrant dermatophytosis. Sci Rep 2023;13(1):19037. 10.1038/s41598-023-46361-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Sharma P, Bhalla M, Thami GP, Chander J. Evaluation of efficacy and safety of oral terbinafine and itraconazole combination therapy in the management of dermatophytosis. J Dermatol Treat. 2020;31(7):749–53. 10.1080/09546634.2019.1612835. [DOI] [PubMed] [Google Scholar]
- 223.Gu D, Hatch M, Ghannoum M, Elewski BE. Treatment-resistant dermatophytosis: a representative case highlighting an emerging public health threat. JAAD Case Rep 2020;6(11):1153–5. 10.1016/j.jdcr.2020.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Chandrashekar BS, Poojitha DS. Evaluation of efficacy and safety of oral voriconazole in the management of recalcitrant and recurrent dermatophytosis. Clin Exp Dermatol 2022;47(1):30–6. 10.1111/ced.14799. [DOI] [PubMed] [Google Scholar]
- 225.What Do You Need To Know About Emerging, Antifungal Medication-Resistant Dermatophyte Infections? American Academy of Dermatology. https://www.aad.org/dw/monthly/2023/septe mber/clinical-applications-antifungal-medication-resistant-infections. Accessed 6 Nov 2023. [Google Scholar]
- 226.Dominguez-Gil Hurlé A, et al. Therapeutic drug monitoring of itraconazole and the relevance of pharmacokinetic interactions. Clin Microbiol Infect 2006;12:97–106.16460557 [Google Scholar]
- 227.Sporanox [package insert]. Titusville: Janssen Pharmaceutical Companies; 2023 [Google Scholar]
- 228.Gold JAW, Benedict K, Dulski TM, Lipner SR. Inadequate diagnostic testing and systemic antifungal prescribing for tinea capitis in an observational cohort study of 3.9 million children, United States. J Am Acad Dermatol 2023;89(1):133–5. 10.1016/j.jaad.2023.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Elewski B A call for antifungal stewardship. Br J Dermatol 2020;183(5):798–9. 10.1111/bjd.19387. [DOI] [PubMed] [Google Scholar]
- 230.Emerging severe and antimicrobial-resistant ringworm infections. Centers for Disease Control https://www.cdc.gov/fungal/disea ses/ringworm/dermatophyte-resistance.html. Accessed 6 Nov 2023. [Google Scholar]
- 231.Bidaud AL, Normand AC, Jabet A, et al. Reliability of a terbinafine agar containing method for the screening of dermatophyte resistance. Med Mycol 2023;61(5):myad043. 10.1093/mmy/myad043. [DOI] [PubMed] [Google Scholar]
- 232.Gupta AK, Renaud HJ, Quinlan EM, Shear NH, Piguet V. The Growing problem of antifungal resistance in onychomycosis and other superficial mycoses. Am J Clin Dermatol 2021;22(2):149–57. 10.1007/s40257-020-00580-6. [DOI] [PubMed] [Google Scholar]
- 233.Arendrup MC, Jørgensen KM, Guinea J, et al. Multicentre validation of a EUCAST method for the antifungal susceptibility testing of microconidia-forming dermatophytes. J Antimicrob Chemother. 2020;75(7):1807–19. 10.1093/jac/dkaa111. [DOI] [PubMed] [Google Scholar]
- 234.Singh A, Singh P, Dingemans G, Meis JF, Chowdhary A. Evaluation of DermaGenius® resistance real-time polymerase chain reaction for rapid detection of terbinafine-resistant Trichophyton species. Mycoses. 2021;64(7):721–6. 10.1111/myc.13271. [DOI] [PubMed] [Google Scholar]
- 235.Rogers TR, Verweij PE, Castanheira M, et al. Molecular mechanisms of acquired antifungal drug resistance in principal fungal pathogens and EUCAST guidance for their laboratory detection and clinical implications. J Antimicrob Chemother. 2022;77(8):2053–73. 10.1093/jac/dkac161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Core Elements of Antibiotic Stewardship. Centers for Disease Control https://www.cdc.gov/antibiotic-use/core-elements/index.html. Accessed 6 Nov 2023. [Google Scholar]
- 237.Hamdy RF, Zaoutis TE, Seo SK. Antifungal stewardship considerations for adults and pediatrics. Virulence. 2017;8(6):658–72. 10.1080/21505594.2016.1226721. (Epub 2016 Sep 2. PMID: 27588344; PMCID: PMC5626349). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Lacroix C, Baspeyras M, de La Salmonière P, et al. Tinea pedis in European marathon runners. J Eur Acad Dermatol Venereol 2002;16(2):139–42. 10.1046/j.1468-3083.2002.00400.x. [DOI] [PubMed] [Google Scholar]
- 239.Zhan P, Geng C, Li Z, et al. The epidemiology of tinea manuum in Nanchang area, South China. Mycopathologia. 2013;176(1– 2):83–8. 10.1007/s11046-013-9673-9236. [DOI] [PubMed] [Google Scholar]
- 240.Caputo R, De Boulle K, Del Rosso J, Nowicki R. Prevalence of superficial fungal infections among sports-active individuals: results from the Achilles survey, a review of the literature. J Eur Acad Dermatol Venereol 2001;15(4):312–6. [PubMed] [Google Scholar]
- 241.Cohen AD, Wolak A, Alkan M, Shalev R, Vardy DA. Prevalence and risk factors for tinea pedis in Israeli soldiers. Int J Dermatol 2005;44(12):1002–5. 10.1111/j.1365-4632.2005.02281.x. [DOI] [PubMed] [Google Scholar]
- 242.Gentles JC, Evans EG. Foot infections in swimming baths. Br Med J 1973;3(5874):260–2. 10.1136/bmj.3.5874.260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Seebacher C, Bouchara JP, Mignon B. Updates on the epidemiology of dermatophyte infections. Mycopathologia. 2008;166(5– 6):335–52. 10.1007/s11046-008-9100-9. [DOI] [PubMed] [Google Scholar]
- 244.Veraldi S, Esposito L, Gorani A. Tinea pedis acquired in mosques? Mycoses. 2018;61(11):794–5. 10.1111/myc.12775. [DOI] [PubMed] [Google Scholar]
- 245.Rutecki GW, Wurtz R, Thomson RB. From animal to man: Tinea Barbae. Curr Infect Dis Rep 2000;2(5):433–7. 10.1007/s11908-000-0073-1. [DOI] [PubMed] [Google Scholar]
- 246.Müller VL, Kappa-Markovi K, Hyun J, et al. Tinea capitis et barbae caused by Trichophyton tonsurans: a retrospective cohort study of an infection chain after shavings in barber shops. Mycoses. 2021;64(4):428–36. 10.1111/myc.13231. [DOI] [PubMed] [Google Scholar]
- 247.Adams BB. Tinea corporis gladiatorum. J Am Acad Dermatol 2002;47(2):286–90. 10.1067/mjd.2002.120603. [DOI] [PubMed] [Google Scholar]
- 248.Jha K, Shaw D, Karim A, et al. Immunological response and clinical profile in patients with recurrent dermatophytosis. Mycoses. 2021;64(11):1429–41. 10.1111/myc.13322. [DOI] [PubMed] [Google Scholar]
- 249.Zhao D, Chen B, Wang YT, Jiao CH. Topical clotrimazole cream for the treatment of tinea cruris: a retrospective study. Medicine (Baltimore). 2020;99(47): e23189. 10.1097/MD.0000000000023189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Gupta AK, Chaudhry M, Elewski B. Tinea corporis, tinea cruris, tinea nigra, and piedra. Dermatol Clin 2003;21(3):395–v. 10.1016/s0733-8635(03)00031-7. [DOI] [PubMed] [Google Scholar]
- 251.Martin A, Kobayashi G. Superficial fungal infection: dermatophytosis, tinea nigra, Piedra. Fitzpatrick’s dermatology in general medicine. 5th ed. New York: McGraw-Hill; 1999. p. 2337–57. [Google Scholar]
- 252.Ilkit M Favus of the scalp: an overview and update. Mycopathologia. 2010;170(3):143–54. 10.1007/s11046-010-9312-7. [DOI] [PubMed] [Google Scholar]
- 253.John AM, Schwartz RA, Janniger CK. The kerion: an angry tinea capitis. Int J Dermatol 2018;57(1):3–9. 10.1111/ijd.13423. [DOI] [PubMed] [Google Scholar]
- 254.Zaraa I, Hawilo A, Aounallah A, et al. Inflammatory Tinea capitis: a 12-year study and a review of the literature. Mycoses. 2013;56(2):110–6. 10.1111/j.1439-0507.2012.02219.x. [DOI] [PubMed] [Google Scholar]
- 255.Topaloğlu Demir F, Karadag AS. Are dermatophytid reactions in patients with kerion celsi much more common than previously thought? A Prospective Study. Pediatr Dermatol 2015;32(5):635–40. 10.1111/pde.12515. [DOI] [PubMed] [Google Scholar]
- 256.Legge BS, Grady JF, Lacey AM. The incidence of tinea pedis in diabetic versus nondiabetic patients with interdigital macerations: a prospective study. J Am Podiatr Med Assoc 2008;98(5):353–6. 10.7547/0980353. [DOI] [PubMed] [Google Scholar]
- 257.Veraldi S, Schianchi R, Benzecry V, Gorani A. Tinea manuum: a report of 18 cases observed in the metropolitan area of Milan and review of the literature. Mycoses. 2019;62(7):604–8. 10.1111/myc.12914. [DOI] [PubMed] [Google Scholar]
- 258.Mizumoto J Two Feet-One Hand Syndrome. Cureus. 2021;13(12): e20758. 10.7759/cureus.20758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Seeburger J, Scher RK. Long-term remission of two feet-one hand syndrome. Cutis. 1998;61(3):149–51. [PubMed] [Google Scholar]
- 260.Daniel CR 3rd, Gupta AK, Daniel MP, Daniel CM. Two feet-one hand syndrome: a retrospective multicenter survey. Int J Dermatol 1997;36(9):658–60. 10.1046/j.1365-4362.1997.00237.x. [DOI] [PubMed] [Google Scholar]
- 261.Bonifaz A, Ramírez-Tamayo T, Saúl A. Tinea barbae (tinea sycosis): experience with nine cases. J Dermatol 2003;30(12):898– 903. 10.1111/j.1346-8138.2003.tb00345.x. [DOI] [PubMed] [Google Scholar]
- 262.Paller AS, and Mancini AJ. 17—Skin disorders caused by fungi. Paller, Mancini, and Hurwitz Clinical Pediatric Dermatology. Sixth ed. Elsevier Inc; 2022. p. 467–96 (e5). [Google Scholar]
- 263.Kuruvella T, Pandey S. Tinea Barbae. In: StatPearls. Treasure Island: StatPearls Publishing; 2023. [PubMed] [Google Scholar]
- 264.Singh S, Sondhi P, Yadav S, Ali F. Tinea barbae presenting as kerion. Indian J Dermatol Venereol Leprol 2017;83(6):741. 10.4103/ijdvl.IJDVL_1104_16. [DOI] [PubMed] [Google Scholar]
- 265.Lin RL, Szepietowski JC, Schwartz RA. Tinea faciei, an often deceptive facial eruption. Int J Dermatol 2004;43(6):437–40. 10.1111/j.1365-4632.2004.02339.x. [DOI] [PubMed] [Google Scholar]
- 266.Amina A, Sana M. Tinea faciei incognito. Indian Pediatr 2019;56(5):433. [PubMed] [Google Scholar]
- 267.Feder HM Jr. Tinea incognito misdiagnosed as erythema migrans. N Engl J Med 2000;343(1):69. 10.1056/NEJM200007063430116. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data/code sharing are not applicable to this article as no datasets or code were generated and/or analyzed during the current study.


