Abstract
No meta-analysis has holistically analysed and summarized the efficacy and safety of osilodrostat, a novel dual 11β-hydroxylase (cytochrome P450 family 11 subfamily B member 1 [CYP11B1]) and 18-hydroxylase (aldosterone synthase, CYP11B2) inhibitor in managing Cushing’s syndrome (CS). We undertook this meta-analysis to address this knowledge gap. Electronic databases were searched for randomized controlled trials (RCTs) involving patients with CS receiving osilodrostat in the intervention arm. The primary outcome was to evaluate changes in urine free cortisol (UFC) levels. Secondary outcomes were to evaluate alterations in cortisol levels, androgen levels, mineralocorticoid levels, and adverse events. From initially screened 109 articles, data from 2 RCTs involving 144 patients was analysed. After 8–12 weeks of therapy, the odds of achieving a normal 24-hour UFC was higher in patients receiving oslidrostat as compared to placebo. [odds ratio (OR) 21.94 (95% CI: 8.53–56.43); P < 0.00001; I2 = 0%]. The occurrence of adverse events [OR 1.35 (95% CI: 0.52–3.53); P = 0.54; I2 = 0%; low heterogeneity (LH); High certainty of evidence (HCE)], serious adverse events (SAEs) [OR 1.32 (95% CI: 0.30–5.79); P = 0.72; I2 = 0%; LH; HCE], adrenal insufficiency [OR 5.38 (95% CI: 0.91–31.78); P = 0.06; I2 = 0%; LH; HCE], headache [OR 0.98 (95% CI: 0.35–2.76); P = 0.97; I2 = 0%; LH; HCE], hyperandrogenism [OR 3.68 (95% CI: 0.59–22.80); P = 0.16; I2 = 0%; LH; HCE] and deaths [OR 0.32 (95% CI: 0.01–8.00); P = 0.48; I2 = 0%; LH; HCE] was comparable among the groups. The occurrence of nausea [OR 4.25 (95% CI: 1.26–14.30); P = 0.02; I2 = 0%; LH] and arthralgia [OR 6.54 (95% CI: 1.64–26.13); P = 0.008; I2 = 0%; LH; HCE] was significantly higher in the osilodrostat group as compared to placebo. Osilodrostat has good efficacy and safety in CS and was well tolerated over 48 weeks of use.
Keywords: Adrenal, cushing’s disease, cushing’s syndrome, osilodrostat, pituitary
INTRODUCTION
Osilodrostat (LCI699) was approved for medical management of Cushing’s syndrome (CS) by the European Medicine Agency (EMA) and the United States Food and Drug Agency (USFDA) in early 2020. Osilodrostat (oral medication; peak blood levels at 1 h; half-life of 4 h), is a potent reversible inhibitor of 2 major enzymes involved in adrenal steroidogenesis [11β-hydroxylase cytochrome P450 family 11 subfamily B member 1 (CYP11B1): catalyses the hydroxylation of 11-deoxycortisol to cortisol and of 11-deoxycorticosterone to corticosterone; and 18-hydroxylase (aldosterone synthase, CYP11B2): catalyses the conversion of corticosterone to aldosterone].[1] Thus, osilodrostat happens to be a dual inhibitor of glucocorticoid and mineralocorticoid biosynthetic pathways. In-vitro studies have documented a threefold higher affinity for CYP11B1 with osilodrostat as compared to metyrapone, another drug having a similar mechanism of action.[2] In adrenocortical cancer cell lines, osilodrostat was found to be more potent than ketoconazole and metyrapone with regard to inhibition of cortisol production.[3] Because of its direct action in inhibiting adrenal glucocorticoid synthesis, osilodrostat has been found to be effective in all forms of CS (pituitary, adrenal, and ectopic).
Several randomized controlled trials (RCTs) and real-world studies have been published evaluating the efficacy of osilodrostat in managing CS.[4,5,6] However, to date, no systematic review or meta-analysis is available that has holistically analysed and summarized the clinical efficacy and safety of this novel molecule for the medical management of CS. Hence, the aim of this meta-analysis was to evaluate the efficacy and safety of osilodrostat in managing CS.
MATERIALS AND METHODS
The meta-analysis was carried out according to the recommendations of the Cochrane Handbook for Systematic Reviews of Interventions.[7] The predefined protocol has been registered in the International Prospective Register of Systematic Reviews (PROSPERO), having a registration number CRD42023429490. All randomized controlled trials (RCTs) published till April 2023 were considered for this meta-analysis. This meta-analysis has been reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).[7] Since ethical approval already exists for the individual studies included in the meta-analysis, no separate approval was required for this study.
The patient/population, intervention, comparison and outcomes (PICOS) criteria were used to screen and select the studies for this meta-analysis, with patients (P) being people living with CS; intervention (I) being the use of osilodrostat for managing CS; control (C) being patients either on placebo or any other approved medication for managing CS; outcomes (O) being evaluated were impact on 24-hour urine free cortisol (UFC), serum cortisol, adrenocorticotropic hormone (ACTH), electrolytes, blood pressure, glycaemia, lipid parameters, liver function, renal function, and side effects. Patients with all the different types of CS were considered for this study (pituitary, adrenal or ectopic). Patients with exogenous CS were excluded. Only those studies were included in this meta-analysis that had at least two treatment arms/groups, with one of the groups having patients with CS on osilodrostat either alone or a part of a standard treatment regimen and the other arm/group receiving either placebo or any other medication in place of osilodrostat.
The primary outcome was to find out the percentage of patients able to achieve 24-hour UFC in a normal range. The secondary outcomes of this study were a >50% reduction in UFC from baseline, change in UFC levels, serum cortisol, ACTH, other steroid metabolites, electrolytes, liver parameters, renal parameters, and side effect profile. Analysis of the outcomes was performed based on whether the control group received an active comparator (any other cortisol-lowering medication) – labelled here as the active control group (ACG) or a placebo – labelled as the passive control Group (PCG).
Detailed electronic database of MEDLINE (Via PubMed), Embase (via Ovid SP), Cochrane Central Register of Controlled Trials (CENTRAL) (for trials only), ctri.nic.in, clinicaltrials.gov, global health, and Google Scholar were searched using a Boolean search strategy: (osilodrostat) OR (LCI699). Data extraction was carried out independently by two authors using standard data extraction forms. Details have been elaborated in previous meta-analyses published by our group.[8] Three authors independently assessed the risk of bias using the risk of bias assessment tool in Review Manager (RevMan) Version 5.4 (The Cochrane Collaboration, Oxford, UK 2014) software. Details have been elaborated elsewhere.[9]
Heterogeneity was initially assessed by studying the forest plot generated for the primary and secondary outcomes of this study. Subsequently, heterogeneity was analysed using a Chi-square test on N-1 degrees of freedom, with an alpha of 0.05 used for statistical significance and with the I2 test.[10] Details have been elaborated elsewhere.[8] An overall grading of the evidence (certainty of the evidence) related to each of the primary and secondary outcomes of the meta-analysis was performed using the Grades of Recommendations Assessment, Development and Evaluation (GRADE) approach.[9] Publication bias was assessed by plotting the funnel plot, which specifically targets small study bias, in which small studies tend to show larger estimates of effects and greater variability than larger studies.[11,12] Details have been elaborated previously in a previous meta-analysis published by our group.[8] Data was pooled as a random effect model for the analysis of primary and secondary outcomes. Forest plots were plotted with the left side of the graph favouring osilodrostat and the right side of the graph favouring control using RevMan 5.4 software.
RESULTS
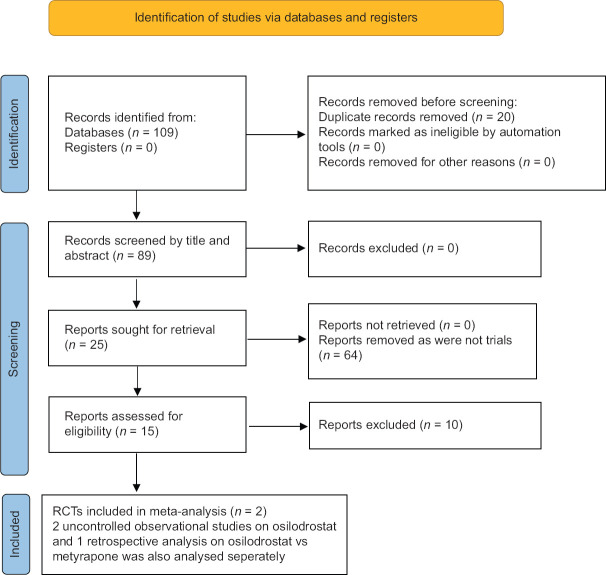
A total of 109 articles were found after the initial search [Figure 1]. Twenty duplicate articles were subsequently removed. Following the screening of the titles and abstracts, followed by full-texts of 89 articles, the search was reduced to 10 studies, which were evaluated in detail for inclusion in this meta-analysis [Figure 1]. LINC stands for LCI699C2302 IN CS. Two LINC RCTs (LINC-3 and LINC-4), which fulfilled all criteria, were analysed in this meta-analysis.[4,5] Studies by Bertagna et al. and Fleseriu et al.[2,6] (LINC 2) did not have a comparator group and were excluded. Only one retrospective study had metyrapone in the ACG and was excluded.[13] The details of the studies included in this meta-analysis have been elaborated in Table 1. The duration of follow-up in the studies LINC-3 and LINC-4 was 48 weeks.[4,5] Gadelha et al.[14] recently published a further 48-week extension follow-up of patients in the LINC-4 study, making the total follow-up duration of 96 weeks. The results from this paper have been merged with LINC-4 data in this systematic review as they represent the same cohort of patients.
Figure 1.
Flowchart elaborating on study retrieval and inclusion in this systematic review. RCT: Randomized controlled trial
Table 1.
Characteristics of patients in the different randomized controlled trials evaluated in this meta-analysis on the use of oslidrostat in Cushing’s disease
| LINC 3[5] | LINC 4[4,14] | |||
|---|---|---|---|---|
|
|
|
|||
| Osilodrostat. (n=36) | Placebo (n=35) | Osilodrostat. (n=36) | Placebo (n=35) | |
| Median age | 41 (37.5–51.5) | 40 (31.5–55) | 41 (21–67) | 37 (19–63) |
| Females | 30 (83%) | 22 (63%) | 43 (89.6%) | 18 (72%) |
| Asian | 7 (9%) | 7 (20%) | 9 (18.8%) | 8 (32%) |
| Time since diagnosis, months Median (IQR) | 53.6 (25.9–94.3) | 76.8 (39.3–133.7) | 69.9 (22.9–92) | 65 (30.4–103.8) |
| Previous pituitary surgery | 32 (89%) | 33 (94%) | 41 (85.4%) | 23 (92%) |
| Previous medical therapy for Cushing’s disease | 26 (72%) | 24 (69%) | 26 (54.2%) | 19 (76%) |
| Previous pituitary irradiation | 6 (17%) | 5 (14%) | 6 (12.5%) | 3 (12%) |
| Mean (SD) 24-h UFC, nmol/L | 890 (1276) | 560 (549) | 421.4 (291.3) | 451.5 (535.1) |
| Study design | ||||
| Screening phase | - 8 to 0 weeks | - 5 to 0 weeks | ||
| Study period 1 | Open label Osilodrostat 2–30 mg BD (0–12 weeks) Osilodrostat therapeutic dose (12–26 weeks) | 2: 1 randomization (osilodrostat 1 mg OD to 20 mg BD: placebo) Osilodrostat therapeutic dose | ||
| Study period 2 | 1:1 randomization* | (12–48 weeks) | ||
| Study period 3 | (osilodrostat: placebo) (26–34 weeks) | 29 patients received osilodrostat for >96 weeks (median: 87.1 weeks); 72.4% of these | ||
| Study period 4 | Osilodrostat (36–48 weeks) | patients achieved normal mUFC | ||
IQR=Interquartile range, SD=Standard deviation; OD: Once daily; mUFC: Mean urinary free cortisol. *Participants were eligible to enter the randomized withdrawal phase at week 26 if they had mean 24-hour UFC concentrations of less than or equal to the ULN at week 24 without a dose increase after week 12
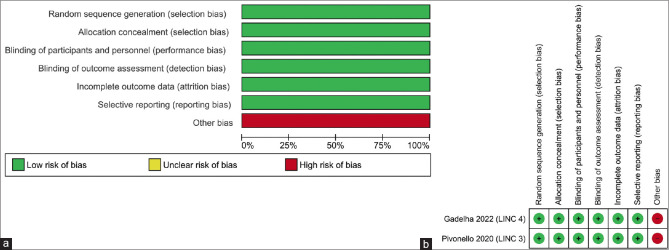
The summaries of the risk of bias of the studies included in the meta-analysis have been elaborated in Figures 2a and b. Random sequence generation, attrition bias, reporting bias, allocation concealment bias (selection bias), performance bias (blinding of participants and investigators), and detection bias (blinding of outcome assessors) were judged to be at low risk of bias in both the studies (100%). Source of funding, especially pharmaceutical, authors from the pharmaceutical organizations, and conflict of interests were looked into in the “other bias” section. Other bias was judged to be at high risk in both studies (100%) [Figure 2a and b].
Figure 2.
(a) Risk of bias graph: review authors’ judgements about each risk of bias item presented as percentages across all included studies; (b) Risk of bias summary: review authors’ judgements about each risk of bias item for each included study
Effect of Osilodrostat on Primary and Secondary Outcomes
Proportion of patients with a 24-hour urine free cortisol in normal range
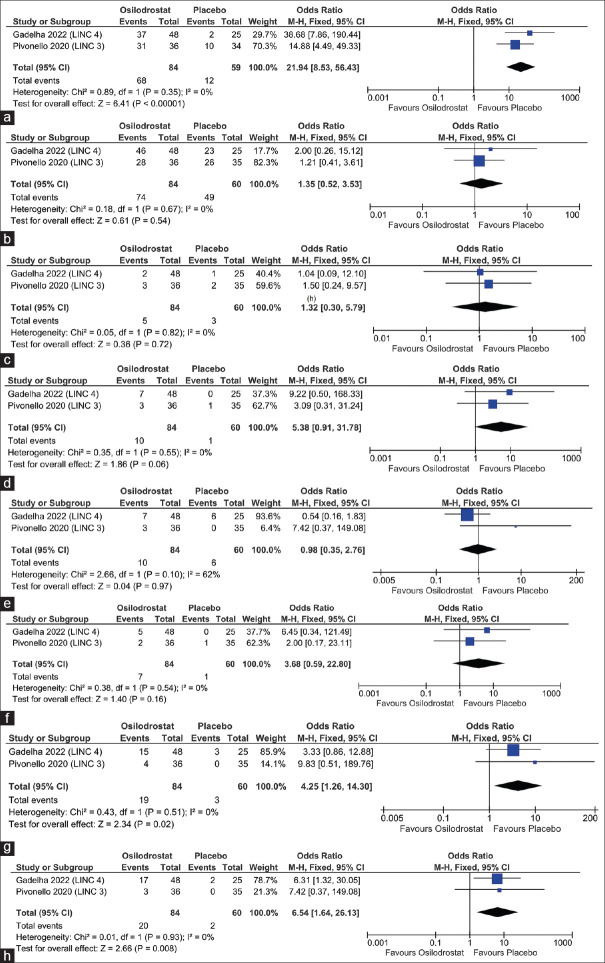
Data from two studies involving 144 people with Cushing’s disease (CD) was analysed to find out the impact of osilodrostat during the randomization phase of treatment. After 8–12 weeks of therapy, the odds of achieving a normal 24-hour UFC were higher in patients receiving osilodrostat as compared to placebo. [Odds ratio (OR) 21.94 (95% CI: 8.53–56.43); P < 0.00001; I2 = 0% (low heterogeneity (LH)); Figure 3a; High certainty of evidence (HCE)]. Other measures of efficacy, like the effect of osilodrostat on mean cortisol values, blood pressure, body mass index (BMI), and quality of life, could not be calculated as the LINC 3 study did not report these outcomes in cases and controls separately.[5]
Figure 3.
Forest plot highlighting the impact of osilodrostat as compared to the placebo group on (a) Percentage of patients achieving normal 24-h UFC; (b) Total adverse events; (c) Serious adverse events; (d) adrenal insufficiency (e) headache (f) hyperandrogenism (g) deaths (h) nausea
Safety
Data from two studies (144 patients) was analysed to evaluate the impact of osilodrostat on the occurrence of adverse events over 48 weeks of treatment. The occurrence of adverse events was not statistically different in patients receiving osilodrostat as compared to the control group [OR 1.35 (95% CI: 0.52–3.53); P = 0.54; I2 = 0%; LH; Figure 3b; HCE]. The occurrence of serious adverse events (SAEs) [OR 1.32 (95% CI: 0.30–5.79); P = 0.72; I2 = 0%; LH; Figure 3c; HCE], adrenal insufficiency [OR 5.38 (95% CI: 0.91–31.78); P = 0.06; I2 = 0%; LH; Figure 3d; HCE], headache [OR 0.98 (95% CI: 0.35–2.76); P = 0.97; I2 = 0%; LH; Figure 3e; HCE], hyperandrogenism [OR 3.68 (95% CI: 0.59–22.80); P = 0.16; I2 = 0%; LH; Figure 3f; HCE] and deaths [OR 0.32 (95% CI: 0.01–8.00); P = 0.48; I2 = 0%; LH; HCE] was comparable among the groups. Nausea, vomiting, and asthenia were the most commonly noted adverse events. The occurrence of nausea [OR 4.25 (95% CI: 1.26–14.30); P = 0.02; I2 = 0%; LH; Figure 3g] and arthralgia [OR 6.54 (95% CI: 1.64–26.13); P = 0.008; I2 = 0%; LH; Figure 3h; HCE] was significantly higher in the osilodrostat group as compared to placebo. As with any treatment inhibiting adrenal glucocorticoid steroidogenesis, the risk of Nelson’s syndrome [high ACTH, hyperpigmentation, and corticotroph tumour progression (CTP)] remains with the use of osilodrostat. There is a single report of the need for pituitary surgery for CTP after 4 years of therapy with osilodrostat for persistent CD in a patient from the LINC3 study.[15] In the LINC3 study, pituitary enlargement has been noted in as many as 37% of patients following 48 months of osilodrostat therapy; pituitary enlargement was significant enough to warrant osilodrostat stoppage in 2.9% of patients.[5] Patients with known prolongation of the QTc interval need close monitoring, as osilodrostat can lead to additional QTc prolongation, which is dose-dependent and has been noted to be 5.3 msec at the full dose of 30 mg twice daily.[16]
The key summary of findings of the study focussing on the percentage of patients achieving normal 24-hour UFC and the side effect profile has been elaborated in Table 2. Funnel plots were plotted to evaluate the presence of publication bias, and have been elaborated in Supplementary Figure 1 (90.8KB, tif) . Key outcomes of observational studies have been elaborated in Table 3.
Table 2.
Summary of findings of the key outcomes of this systematic review
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Certainty of the evidence (GRADE) | |
|---|---|---|---|---|---|
|
| |||||
| Risk with placebo | Risk with Osilodrostat | ||||
| Complete responders (mean 24-h UFC < ULN) in the randomization phase | 203 per 1,000 | 849 per 1,000 (685 to 935) | OR 21.94 (8.53 to 56.43) | 143 (2 RCTs) | ⨁⨁⨁⨁ High |
| Adverse events | 817 per 1,000 | 857 per 1,000 (698 to 940) | OR 1.35 (0.52 to 3.53) | 144 (2 RCTs) | ⨁⨁⨁⨁ High |
| Serious Adverse events | 50 per 1,000 | 65 per 1,000 (16 to 234) | OR 1.32 (0.30 to 5.79) | 144 (2 RCTs) | ⨁⨁⨁⨁ High |
| Adrenal insufficiency | 17 per 1,000 | 84 per 1,000 (15 to 350) | OR 5.38 (0.91 to 31.78) | 144 (2 RCTs) | ⨁⨁⨁⨁ High |
| Nausea | 50 per 1,000 | 183 per 1,000 (62 to 429) | OR 4.25 (1.26 to 14.30) | 144 (2 RCTs) | ⨁⨁⨁⨁ High |
| Arthralgia | 33 per 1,000 | 184 per 1,000 (54 to 474) | OR 6.54 (1.64 to 26.13) | 144 (2 RCTs) | ⨁⨁⨁⨁ High |
| Headache | 100 per 1,000 | 98 per 1,000 (37 to 235) | OR 0.98 (0.35 to 2.76) | 144 (2 RCTs) | ⨁⨁⨁⨁ High |
| Hyperandrogenism | 17 per 1,000 | 59 per 1,000 (10 to 279) | OR 3.68 (0.59 to 22.80) | 144 (2 RCTs) | ⨁⨁⨁⨁ High |
| Death | 17 per 1,000 | 5 per 1,000 (0 to 119) | OR 0.32 (0.01 to 8.00) | 144 (2 RCTs) | ⨁⨁⨁⨁ High |
CI=Confidence interval, OR=Odds ratio, ACG=Active control group, PCG=Placebo control group. *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI)
Table 3.
Summary of key outcomes on uncontrolled studies with osilodrostat in Cushing’s syndrome
| Study design | Patients | Study duration (weeks) | Efficacy | Side effects | |
|---|---|---|---|---|---|
| LINC 1[2] | Proof of concept | 12 patients with CD | 10 | 91.7% achieved normal UFC levels at day 70 100% achieved normalized UFC levels or achieved a >50% reduction in UFC at day 70 | Fatigue (58%), nausea (42%), headache (25%), hypokalemia (25%), diarrhea (25%), dizziness (17%), arthralgia (17%) |
| LINC 2[6] | Phase II open-label prospective study | 19 patients with CD | 22 | 78.9% achieved normal UFC at 22 weeks | Adrenal insufficiency (32%), nausea (32%), diarrhea (32%), fatigue (32%) |
| Detomas et al.[13] | Retrospective analysis | 16 patients with endogenous CS (pituitary n=7, adrenal n=4, ectopic CS n=5). Osilodrostat=8 Metyrapone=8 | 12 | At 12 weeks, a more pronounced decrease of cortisol was found in the metyrapone than in the osilodrostat group (-51.1% vs -25.8%, compared to baseline cortisol) | At 8 weeks and 12 weeks, 2 patients under metyrapone and 3 patients under osilodrostat required potassium replacement therapy. A progressive increase in the QTc interval was identified in the osilodrostat group, but not in the metyrapone one (455±23 ms vs 432±3 ms) |
CD=Cushing’s disease, UFC=Urinary free cortisol, CS=Cushing’s syndrome, ms=Milli-second
DISCUSSION
Medical management of CS is warranted to control hypercortisolism as a part of pre-operative preparation for a definitive surgical cure, for patients not fit or willing for surgical treatment, and for managing residual hypercortisolism in post-pituitary/adrenal/paraneoplastic tumour surgery. Currently, available medical therapy targeting the pituitary gland in the setting of CD is pasireotide and cabergoline. Medical therapy targeting adrenal glucocorticoid steroidogenesis to decrease hypercortisolism includes ketoconazole, metyrapone, mitotane, and etomidate. However, none of these agents have been perfect till date and have their share of major and minor side effects. Mifepristone is the third class of agent, being a direct glucocorticoid receptor antagonist. Osilodrostat is a welcome addition to the class of medications inhibiting adrenal glucocorticoid steroidogenesis, which is the most popular class for its ability to reduce hypercortisolism in all the different types of CS.
This is the first meta-analysis to highlight the efficacy and safety of osilodrostat in managing CS with efficacy data available till up to 96 weeks of clinical use. This meta-analysis provides reassuring data on the safety of osilodrostat in clinical practice. Because of its mechanism of action, androgenic side effects like hirsutism and acne are expected in women on osilodrostat. However, increased occurrence of hyperandrogenism was not noted with the use of osilodrostat in the controlled phase of different RCTs published till date. Mechanistically, the short half-life and immediate onset of action of osilodrostat make it an attractive agent for the rapid management of hypercortisolism in patients with CS. This can be an effective agent for pre-operative rapid control of hypercortisolism before surgical intervention in patients of CS with severe hypercortisolism.
Because of its mechanism of action, increased circulating levels of 11-deoxycortisol have been noted with osilodrostat use, which cross-reacts with cortisol assays during immunoassays.[17] Hence, serum cortisol levels in patients on osilodrostat should ideally be measured using tandem mass spectrometry and not immunoassays to avoid labomas.[18] However, tandem mass spectrometry is not easily available in clinical practice, and hence, this becomes a limitation with osilodrostat use similar to that of metyrapone. 11-deoxycorticosterone and ACTH levels are also elevated use of osilodrostat. All these hormone changes are reversible with osilodrostat stoppage.
Osilodrostat is a weak inhibitor of CYP2D6 and CYP3A4/5 and a moderate inhibitor of CYP2C19 and CYP1A2.[18] Hence dose needs to be adjusted in people on other medications with interfere with cytochrome metabolism, like proton pump inhibitors, theophylline, rifampicin, quinolones, and isoniazid, among others.[19] Simultaneous use of osilodrostat and oral contraceptives does not affect their half-life and metabolism.[20] Based on the available clinical data, the recommended dose of osilodrostat initiation to control hypercortisolism in CS is 2 mg twice daily, which is gradually up-titrated over several weeks with the aim to normalize 24-hour UFC on patients, without precipitating clinical and biochemical features of hypocortisolism like asthenia and hyperkalaemia among others. Commonly used maintenance dose is 4–14 mg twice daily with a maximum dose of 30 mg twice daily.[1] Osilodrostat has three active metabolites [M34.5 (51%), M16.5 (9%), and M24.9 (7%) of administered dose], which have longer half-lives making twice daily dosing feasible, which significantly improves patient compliance and satisfaction vis a viz four doses/day and six doses/day for ketoconazole and metyrapone respectively.[21] Hirsutism and acne (androgenic side effects) may limit the long-term use of osilodrostat in women, similar to that of metyrapone. Women may actually prefer ketoconazole because of its anti-androgenic benefits also. However, liver toxicity, which can rarely be severe and life-threatening, is a major red flag against long-term ketoconazole use.
Limitations of this systematic review include the small number of controlled trials available for analysis of outcomes with the use of osilodrostat. However, data from uncontrolled studies shows good efficacy and durability of osilodrostat in controlling hypercortisolism.
Our analysis shows that osilodrostat is a potent, effective, safe, and well-tolerated agent in controlling hypercortisolism in CS. Osilodrostat is a welcome addition to the medical armamentarium for managing CS.
Authors’ contribution
The study was conceptualized by DD. The study protocol was planned out by LN and DD. Literature search was done by NR, VS and CS. Data extraction was done by VS, CS and SB. Data analysis was done by DD and LN. All authors contributed equally to the manuscript preparation.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Funnel plot of all the included studies in the meta-analysis (assessing the publication bias) of the main outcomes assessed (a) Percentage of patients achieving normal 24-h UFC; (b) Total adverse events; (c) Serious adverse events; (d) adrenal insufficiency (e) nausea (f) arthralgias (g) hyperandrogenism
Acknowledgment
None.
REFERENCES
- 1.Martino M, Aboud N, Lucchetti B, Salvio G, Arnaldi G. Osilodrostat oral tablets for adults with Cushing's disease. Expert Rev Endocrinol Metab. 2022;17:99–109. doi: 10.1080/17446651.2022.2044789. [DOI] [PubMed] [Google Scholar]
- 2.Bertagna X, Pivonello R, Fleseriu M, Zhang Y, Robinson P, Taylor A, et al. LCI699, a potent 11β- hydroxylase inhibitor, normalizes urinary cortisol in patients with Cushing's disease: Results from a multicenter, proof-of-concept study. J Clin Endocrinol Metab. 2014;99:1375–83. doi: 10.1210/jc.2013-2117. [DOI] [PubMed] [Google Scholar]
- 3.Creemers SG, Feelders RA, de Jong FH, Franssen GJH, de Rijke YB, van Koetsveld PM, et al. Osilodrostat is a potential novel steroidogenesis inhibitor for the treatment of Cushing syndrome: An in vitro study. J Clin Endocrinol Metab. 2019;104:3437–49. doi: 10.1210/jc.2019-00217. [DOI] [PubMed] [Google Scholar]
- 4.Gadelha M, Bex M, Feelders RA, Heaney AP, Auchus RJ, Gilis-Januszewska A, et al. Randomized trial of osilodrostat for the treatment of cushing disease. J Clin Endocrinol Metab. 2022;107:e2882–95. doi: 10.1210/clinem/dgac178. doi: 10.1210/clinem/dgac178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pivonello R, Fleseriu M, Newell-Price J, Bertagna X, Findling J, Shimatsu A, et al. Efficacy and safety of osilodrostat in patients with Cushing's disease (LINC 3): A multicentre phase III study with a double-blind, randomised withdrawal phase. Lancet Diabetes Endocrinol. 2020;8:748–61. doi: 10.1016/S2213-8587(20)30240-0. [DOI] [PubMed] [Google Scholar]
- 6.Fleseriu M, Biller BMK, Bertherat J, Young J, Hatipoglu B, Arnaldi G, et al. Long-term efficacy and safety of osilodrostat in Cushing's disease: Final results from a Phase II study with an optional extension phase (LINC 2) Pituitary. 2022;25:959–70. doi: 10.1007/s11102-022-01280-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Higgins JP, Altman DG, Gotzsche PC, Jüni P, Moher D, Oxman AD, et al. The cochrane collaboration's tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi: 10.1136/bmj.d5928. doi: 10.1136/bmj.d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dutta D, Agarwal A, Maisnam I, Singla R, Khandelwal D, Sharma M. Efficacy and safety of the novel dipeptidyl peptidase-4 inhibitor gemigliptin in the management of type 2 diabetes: A meta-analysis. Endocrinol Metab (Seoul) 2021;36:374–87. doi: 10.3803/EnM.2020.818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dutta D, Surana V, Bhattacharya S, Aggarwal S, Sharma M. Efficacy and safety of novel non-steroidal mineralocorticoid receptor antagonist finerenone in the management of diabetic kidney disease: A meta-analysis. Indian J Endocrinol Metab. 2022;26:198–205. doi: 10.4103/ijem.ijem_376_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and metaanalyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ. 2009;339:b2700. doi: 10.1136/bmj.b2700. doi: 10.1136/bmj.b2700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. BMJ (Clinical Research Ed). 2008;336:924–6. doi: 10.1136/bmj.39489.470347.AD. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Song F, Eastwood AJ, Gilbody S, Duley L, Sutton AJ. Publication and related biases. Health Technol Assess. 2000;4:1–115. [PubMed] [Google Scholar]
- 13.Detomas M, Altieri B, Deutschbein T, Fassnacht M, Dischinger U. Metyrapone versus osilodrostat in the short-term therapy of endogenous Cushing's syndrome: Results from a single center cohort study. Front Endocrinol (Lausanne) 2022;13:903545. doi: 10.3389/fendo.2022.903545. doi: 10.3389/fendo.2022.903545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gadelha M, Snyder PJ, Witek P, Bex M, Belaya Z, Turcu AF, et al. Long-term efficacy and safety of osilodrostat in patients with Cushing's disease: Results from the LINC 4 study extension. Front Endocrinol (Lausanne) 2023;14:1236465. doi: 10.3389/fendo.2023.1236465. doi: 10.3389/fendo.2023.1236465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Assié G, Bahurel H, Coste J, Silvera S, Kujas M, Dugué MA, et al. Corticotroph tumor progression after adrenalectomy in Cushing's disease: A reappraisal of Nelson's syndrome. J Clin Endocrinol Metab. 2007;92:172–9. doi: 10.1210/jc.2006-1328. [DOI] [PubMed] [Google Scholar]
- 16.Dougherty JA, Desai DS, Herrera JB. Osilodrostat: A novel steroidogenesis inhibitor to treat Cushing's disease. Ann Pharmacother. 2021;55:1050–60. doi: 10.1177/1060028020968808. [DOI] [PubMed] [Google Scholar]
- 17.Tritos NA. Adrenally directed medical therapies for Cushing syndrome. J Clin Endocrinol Metab. 2021;106:16–25. doi: 10.1210/clinem/dgaa778. [DOI] [PubMed] [Google Scholar]
- 18.Dutta D, Chowdhury S. Endocrine labomas. Indian J Endocr Metab. 2012;16:S275–8. doi: 10.4103/2230-8210.104059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Armani S, Ting L, Sauter N, Darstein C, Tripathi AP, Wang L, et al. Drug interaction potential of osilodrostat (LCI699) based on its effect on the pharmacokinetics of probe drugs of cytochrome p450 enzymes in healthy adults. Clin Drug Investig. 2017;37:465–72. doi: 10.1007/s40261-017-0497-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Perosevic M, Tritos NA. Clinical utility of osilodrostat in Cushing's disease: Review of currently available literature. Drug Des Devel Ther. 2023;17:1303–12. doi: 10.2147/DDDT.S315359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Duggan S. Osilodrostat: First approval. Drugs. 2020;80:495–500. doi: 10.1007/s40265-020-01277-0. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Funnel plot of all the included studies in the meta-analysis (assessing the publication bias) of the main outcomes assessed (a) Percentage of patients achieving normal 24-h UFC; (b) Total adverse events; (c) Serious adverse events; (d) adrenal insufficiency (e) nausea (f) arthralgias (g) hyperandrogenism