Abstract
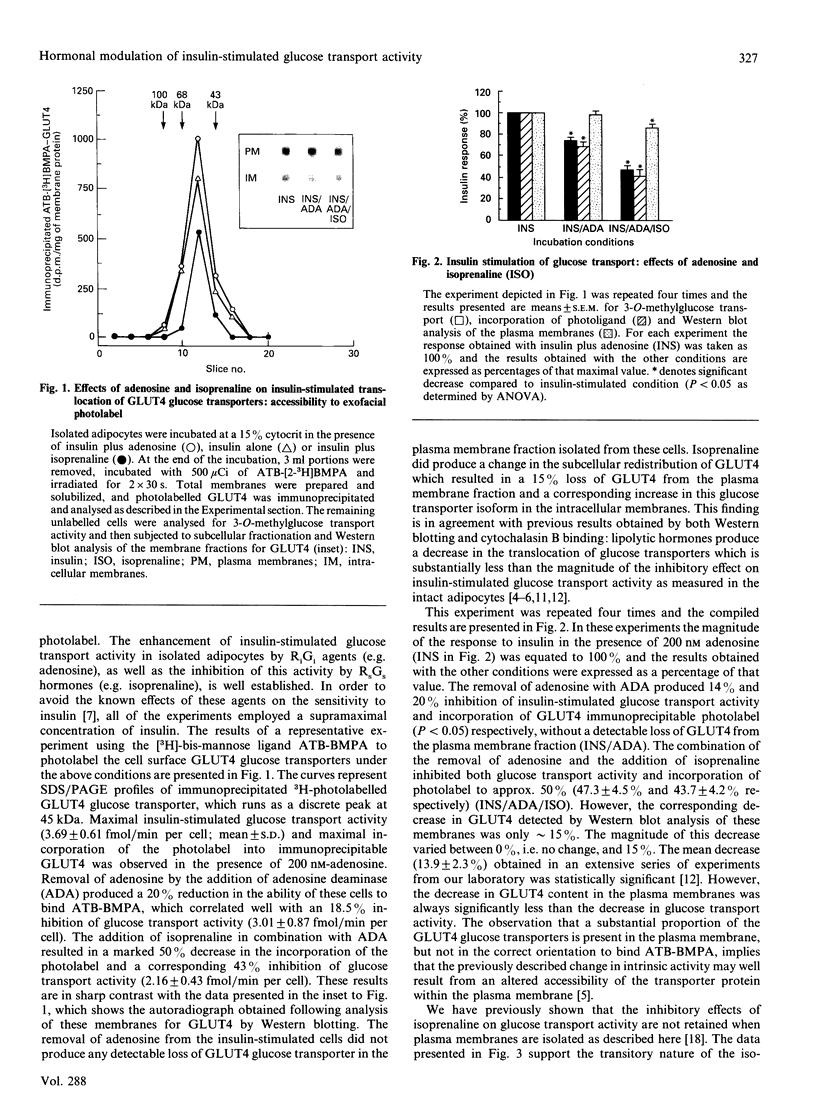
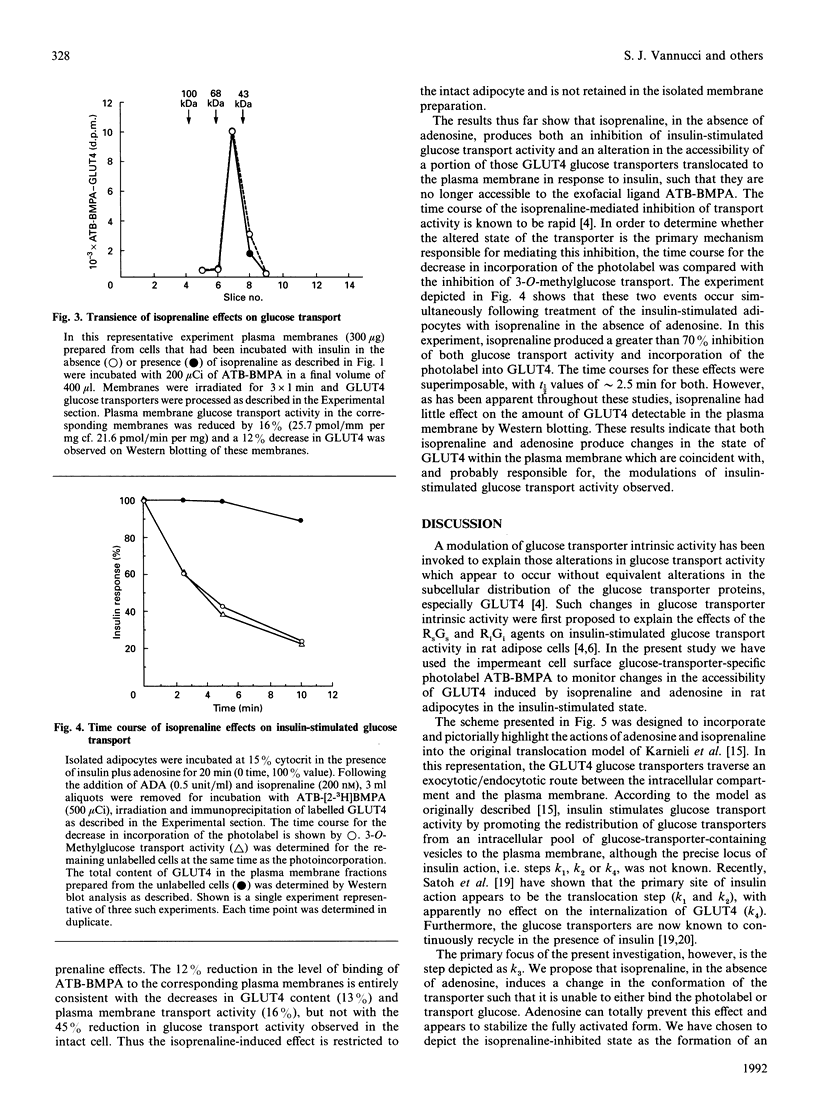
Insulin-stimulated glucose transport activity in rat adipocytes is inhibited by isoprenaline and enhanced by adenosine. Both of these effects occur without corresponding changes in the subcellular distribution of the GLUT4 glucose transporter isoform. In this paper, we have utilized the impermeant, exofacial bis-mannose glucose transporter-specific photolabel, 2-N-4-(1-azi-2,2,2-trifluoroethyl)benzoyl-1,3-bis-(D-mannos- 4-yloxy)-2-propylamine (ATB-BMPA) [Clark & Holman (1990) Biochem. J. 269, 615-622], to examine the cell surface accessibility of GLUT4 glucose transporters under these conditions. Compared with cells treated with insulin alone, adenosine in the presence of insulin increased the accessibility of GLUT4 to the extracellular photolabel by approximately 25%, consistent with its enhancement of insulin-stimulated glucose transport activity; the plasma membrane concentration of GLUT4 as assessed by Western blotting was unchanged. Conversely, isoprenaline, in the absence of adenosine, promoted a time-dependent (t1/2 approximately 2 min) decrease in the accessibility of insulin-stimulated cell surface GLUT4 of > 50%, which directly correlated with the observed inhibition of transport activity; the plasma membrane concentration of GLUT4 decreased by 0-15%. Photolabelling the corresponding plasma membranes revealed that these alterations in the ability of the photolabel to bind to GLUT4 are transient, as the levels of both photolabel incorporation and plasma membrane glucose transport activity were consistent with the observed GLUT4 concentration. These data suggest that insulin-stimulated GLUT4 glucose transporters can exist in two distinct states within the adipocyte plasma membrane, one which is functional and accessible to extracellular substrate, and one which is non-functional and unable to bind extracellular substrate. These effects are only observed in the intact adipocyte and are not retained in plasma membranes isolated from these cells when analysed for their ability to transport glucose or bind photolabel.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Clark A. E., Holman G. D. Exofacial photolabelling of the human erythrocyte glucose transporter with an azitrifluoroethylbenzoyl-substituted bismannose. Biochem J. 1990 Aug 1;269(3):615–622. doi: 10.1042/bj2690615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark A. E., Holman G. D., Kozka I. J. Determination of the rates of appearance and loss of glucose transporters at the cell surface of rat adipose cells. Biochem J. 1991 Aug 15;278(Pt 1):235–241. doi: 10.1042/bj2780235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eckel J., Gerlach-Eskuchen E., Reinauer H. G-protein-mediated regulation of the insulin-responsive glucose transporter in isolated cardiac myocytes. Biochem J. 1990 Dec 15;272(3):691–696. doi: 10.1042/bj2720691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison S. A., Clancy B. M., Pessino A., Czech M. P. Activation of cell surface glucose transporters measured by photoaffinity labeling of insulin-sensitive 3T3-L1 adipocytes. J Biol Chem. 1992 Feb 25;267(6):3783–3788. [PubMed] [Google Scholar]
- Holman G. D., Kozka I. J., Clark A. E., Flower C. J., Saltis J., Habberfield A. D., Simpson I. A., Cushman S. W. Cell surface labeling of glucose transporter isoform GLUT4 by bis-mannose photolabel. Correlation with stimulation of glucose transport in rat adipose cells by insulin and phorbol ester. J Biol Chem. 1990 Oct 25;265(30):18172–18179. [PubMed] [Google Scholar]
- Honnor R. C., Naghshineh S., Cushman S. W., Wolff J., Simpson I. A., Londos C. Cholera and pertussis toxins modify regulation of glucose transport activity in rat adipose cells: evidence for mediation of a cAMP-independent process by G-proteins. Cell Signal. 1992 Jan;4(1):87–98. doi: 10.1016/0898-6568(92)90010-6. [DOI] [PubMed] [Google Scholar]
- James D. E., Brown R., Navarro J., Pilch P. F. Insulin-regulatable tissues express a unique insulin-sensitive glucose transport protein. Nature. 1988 May 12;333(6169):183–185. doi: 10.1038/333183a0. [DOI] [PubMed] [Google Scholar]
- James D. E., Hiken J., Lawrence J. C., Jr Isoproterenol stimulates phosphorylation of the insulin-regulatable glucose transporter in rat adipocytes. Proc Natl Acad Sci U S A. 1989 Nov;86(21):8368–8372. doi: 10.1073/pnas.86.21.8368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joost H. G., Weber T. M., Cushman S. W., Simpson I. A. Activity and phosphorylation state of glucose transporters in plasma membranes from insulin-, isoproterenol-, and phorbol ester-treated rat adipose cells. J Biol Chem. 1987 Aug 15;262(23):11261–11267. [PubMed] [Google Scholar]
- Joost H. G., Weber T. M., Cushman S. W., Simpson I. A. Insulin-stimulated glucose transport in rat adipose cells. Modulation of transporter intrinsic activity by isoproterenol and adenosine. J Biol Chem. 1986 Aug 5;261(22):10033–10036. [PubMed] [Google Scholar]
- Karnieli E., Zarnowski M. J., Hissin P. J., Simpson I. A., Salans L. B., Cushman S. W. Insulin-stimulated translocation of glucose transport systems in the isolated rat adipose cell. Time course, reversal, insulin concentration dependency, and relationship to glucose transport activity. J Biol Chem. 1981 May 25;256(10):4772–4777. [PubMed] [Google Scholar]
- Kuroda M., Honnor R. C., Cushman S. W., Londos C., Simpson I. A. Regulation of insulin-stimulated glucose transport in the isolated rat adipocyte. cAMP-independent effects of lipolytic and antilipolytic agents. J Biol Chem. 1987 Jan 5;262(1):245–253. [PubMed] [Google Scholar]
- Lawrence J. C., Jr, Hiken J. F., James D. E. Phosphorylation of the glucose transporter in rat adipocytes. Identification of the intracellular domain at the carboxyl terminus as a target for phosphorylation in intact-cells and in vitro. J Biol Chem. 1990 Feb 5;265(4):2324–2332. [PubMed] [Google Scholar]
- Lönnroth P., Appell K. C., Wesslau C., Cushman S. W., Simpson I. A., Smith U. Insulin-induced subcellular redistribution of insulin-like growth factor II receptors in the rat adipose cell. Counterregulatory effects of isoproterenol, adenosine, and cAMP analogues. J Biol Chem. 1988 Oct 25;263(30):15386–15391. [PubMed] [Google Scholar]
- Nishimura H., Saltis J., Habberfield A. D., Garty N. B., Greenberg A. S., Cushman S. W., Londos C., Simpson I. A. Phosphorylation state of the GLUT4 isoform of the glucose transporter in subfractions of the rat adipose cell: effects of insulin, adenosine, and isoproterenol. Proc Natl Acad Sci U S A. 1991 Dec 15;88(24):11500–11504. doi: 10.1073/pnas.88.24.11500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oka Y., Asano T., Shibasaki Y., Lin J. L., Tsukuda K., Katagiri H., Akanuma Y., Takaku F. C-terminal truncated glucose transporter is locked into an inward-facing form without transport activity. Nature. 1990 Jun 7;345(6275):550–553. doi: 10.1038/345550a0. [DOI] [PubMed] [Google Scholar]
- Simpson I. A., Cushman S. W. Hormonal regulation of mammalian glucose transport. Annu Rev Biochem. 1986;55:1059–1089. doi: 10.1146/annurev.bi.55.070186.005211. [DOI] [PubMed] [Google Scholar]
- Simpson I. A., Yver D. R., Hissin P. J., Wardzala L. J., Karnieli E., Salans L. B., Cushman S. W. Insulin-stimulated translocation of glucose transporters in the isolated rat adipose cells: characterization of subcellular fractions. Biochim Biophys Acta. 1983 Dec 19;763(4):393–407. doi: 10.1016/0167-4889(83)90101-5. [DOI] [PubMed] [Google Scholar]
- Slot J. W., Geuze H. J., Gigengack S., James D. E., Lienhard G. E. Translocation of the glucose transporter GLUT4 in cardiac myocytes of the rat. Proc Natl Acad Sci U S A. 1991 Sep 1;88(17):7815–7819. doi: 10.1073/pnas.88.17.7815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slot J. W., Geuze H. J., Gigengack S., Lienhard G. E., James D. E. Immuno-localization of the insulin regulatable glucose transporter in brown adipose tissue of the rat. J Cell Biol. 1991 Apr;113(1):123–135. doi: 10.1083/jcb.113.1.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith U., Kuroda M., Simpson I. A. Counter-regulation of insulin-stimulated glucose transport by catecholamines in the isolated rat adipose cell. J Biol Chem. 1984 Jul 25;259(14):8758–8763. [PubMed] [Google Scholar]
- Wardzala L. J., Simpson I. A., Rechler M. M., Cushman S. W. Potential mechanism of the stimulatory action of insulin on insulin-like growth factor II binding to the isolated rat adipose cell. Apparent redistribution of receptors cycling between a large intracellular pool and the plasma membrane. J Biol Chem. 1984 Jul 10;259(13):8378–8383. [PubMed] [Google Scholar]
- Weber T. M., Joost H. G., Kuroda M., Cushman S. W., Simpson I. A. Subcellular distribution and phosphorylation state of insulin receptors from insulin- and isoproterenol-treated rat adipose cells. Cell Signal. 1991;3(1):51–58. doi: 10.1016/0898-6568(91)90007-h. [DOI] [PubMed] [Google Scholar]
- Zorzano A., Wilkinson W., Kotliar N., Thoidis G., Wadzinkski B. E., Ruoho A. E., Pilch P. F. Insulin-regulated glucose uptake in rat adipocytes is mediated by two transporter isoforms present in at least two vesicle populations. J Biol Chem. 1989 Jul 25;264(21):12358–12363. [PubMed] [Google Scholar]