Abstract
The complete primary structure of beta s-crystallin from human lens is reported. The sequence was elucidated by automatic Edman degradation of tryptic and CNBr peptides. The blocked N-terminal dipeptide was identified by fast-atom-bombardment mass spectroscopy. The sequence comparison with other members of crystallin family reveals a closer relationship to human gamma-crystallin (53% identity) than with beta A3/A1 crystallin (37% identity). The structure, evolutionary characteristics and role of beta s-crystallin in lens are discussed.
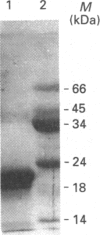
Full text
PDF




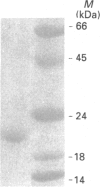
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bax B., Lapatto R., Nalini V., Driessen H., Lindley P. F., Mahadevan D., Blundell T. L., Slingsby C. X-ray analysis of beta B2-crystallin and evolution of oligomeric lens proteins. Nature. 1990 Oct 25;347(6295):776–780. doi: 10.1038/347776a0. [DOI] [PubMed] [Google Scholar]
- Berbers G. A., Hoekman W. A., Bloemendal H., de Jong W. W., Kleinschmidt T., Braunitzer G. Homology between the primary structures of the major bovine beta-crystallin chains. Eur J Biochem. 1984 Mar 15;139(3):467–479. doi: 10.1111/j.1432-1033.1984.tb08029.x. [DOI] [PubMed] [Google Scholar]
- Bindels J. G., Koppers A., Hoenders H. J. Structural aspects of bovine beta-crystallins: physical characterization including dissociation-association behavior. Exp Eye Res. 1981 Sep;33(3):333–343. doi: 10.1016/s0014-4835(81)80056-5. [DOI] [PubMed] [Google Scholar]
- Bloemendal H., Piatigorsky J., Spector A. Recommendations for crystallin nomenclature. Exp Eye Res. 1989 Apr;48(4):465–466. doi: 10.1016/0014-4835(89)90029-8. [DOI] [PubMed] [Google Scholar]
- Blundell T., Lindley P., Miller L., Moss D., Slingsby C., Tickle I., Turnell B., Wistow G. The molecular structure and stability of the eye lens: x-ray analysis of gamma-crystallin II. Nature. 1981 Feb 26;289(5800):771–777. doi: 10.1038/289771a0. [DOI] [PubMed] [Google Scholar]
- Chang T., Chang W. C. Cloning and sequencing of a carp beta s-crystallin cDNA. Biochim Biophys Acta. 1987 Oct 9;910(1):89–92. doi: 10.1016/0167-4781(87)90098-4. [DOI] [PubMed] [Google Scholar]
- Croft L. R. The amino acid sequence of -crystallin (fraction II) from calf lens. Biochem J. 1972 Jul;128(4):961–970. doi: 10.1042/bj1280961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czarnecka E., Gurley W. B., Nagao R. T., Mosquera L. A., Key J. L. DNA sequence and transcript mapping of a soybean gene encoding a small heat shock protein. Proc Natl Acad Sci U S A. 1985 Jun;82(11):3726–3730. doi: 10.1073/pnas.82.11.3726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Driessen H. P., Herbrink P., Bloemendal H., de Jong W. W. Primary structure of the bovine beta-crystallin Bp chain. Internal duplication and homology with gamma-crystallin. Eur J Biochem. 1981 Dec;121(1):83–91. doi: 10.1111/j.1432-1033.1981.tb06433.x. [DOI] [PubMed] [Google Scholar]
- Edman P., Begg G. A protein sequenator. Eur J Biochem. 1967 Mar;1(1):80–91. doi: 10.1007/978-3-662-25813-2_14. [DOI] [PubMed] [Google Scholar]
- Harding J. J., Dilley K. J. Structural proteins of the mammalian lens: a review with emphasis on changes in development, aging and cataract. Exp Eye Res. 1976 Jan;22(1):1–73. doi: 10.1016/0014-4835(76)90033-6. [DOI] [PubMed] [Google Scholar]
- Hewick R. M., Hunkapiller M. W., Hood L. E., Dreyer W. J. A gas-liquid solid phase peptide and protein sequenator. J Biol Chem. 1981 Aug 10;256(15):7990–7997. [PubMed] [Google Scholar]
- Hogg D., Tsui L. C., Gorin M., Breitman M. L. Characterization of the human beta-crystallin gene Hu beta A3/A1 reveals ancestral relationships among the beta gamma-crystallin superfamily. J Biol Chem. 1986 Sep 15;261(26):12420–12427. [PubMed] [Google Scholar]
- Inana G., Piatigorsky J., Norman B., Slingsby C., Blundell T. Gene and protein structure of a beta-crystallin polypeptide in murine lens: relationship of exons and structural motifs. Nature. 1983 Mar 24;302(5906):310–315. doi: 10.1038/302310a0. [DOI] [PubMed] [Google Scholar]
- Inana G., Shinohara T., Maizel J. V., Jr, Piatigorsky J. Evolution and diversity of the crystallins. Nucleotide sequence of a beta-crystallin mRNA from the mouse lens. J Biol Chem. 1982 Aug 10;257(15):9064–9071. [PubMed] [Google Scholar]
- Ingolia T. D., Craig E. A. Four small Drosophila heat shock proteins are related to each other and to mammalian alpha-crystallin. Proc Natl Acad Sci U S A. 1982 Apr;79(7):2360–2364. doi: 10.1073/pnas.79.7.2360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- McDevitt D. S., Croft L. R. On the existence of gamma-crystallin in the bird lens. Exp Eye Res. 1977 Nov;25(5):473–481. doi: 10.1016/0014-4835(77)90176-2. [DOI] [PubMed] [Google Scholar]
- Meakin S. O., Du R. P., Tsui L. C., Breitman M. L. Gamma-crystallins of the human eye lens: expression analysis of five members of the gene family. Mol Cell Biol. 1987 Aug;7(8):2671–2679. doi: 10.1128/mcb.7.8.2671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quax-Jeuken Y., Driessen H., Leunissen J., Quax W., de Jong W., Bloemendal H. beta s-Crystallin: structure and evolution of a distinct member of the beta gamma-superfamily. EMBO J. 1985 Oct;4(10):2597–2602. doi: 10.1002/j.1460-2075.1985.tb03976.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siezen R. J., Fisch M. R., Slingsby C., Benedek G. B. Opacification of gamma-crystallin solutions from calf lens in relation to cold cataract formation. Proc Natl Acad Sci U S A. 1985 Mar;82(6):1701–1705. doi: 10.1073/pnas.82.6.1701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slingsby C., Bateman O. A. Quaternary interactions in eye lens beta-crystallins: basic and acidic subunits of beta-crystallins favor heterologous association. Biochemistry. 1990 Jul 17;29(28):6592–6599. doi: 10.1021/bi00480a007. [DOI] [PubMed] [Google Scholar]
- Slingsby C., Driessen H. P., Mahadevan D., Bax B., Blundell T. L. Evolutionary and functional relationships between the basic and acidic beta-crystallins. Exp Eye Res. 1988 Mar;46(3):375–403. doi: 10.1016/s0014-4835(88)80027-7. [DOI] [PubMed] [Google Scholar]
- Summers L. J., Slingsby C., Blundell T. L., den Dunnen J. T., Moormann R. J., Schoenmakers J. G. Structural variation in mammalian gamma-crystallins based on computer graphics analyses of human, rat and calf sequences. 1. Core packing and surface properties. Exp Eye Res. 1986 Jul;43(1):77–92. doi: 10.1016/s0014-4835(86)80047-1. [DOI] [PubMed] [Google Scholar]
- Thomson J. A., Siezen R. J., Kaplan E. D., Messmer M., Chakrabarti B. Comparative studies of beta s-crystallins from human, bovine, rat and rabbit lenses. Curr Eye Res. 1989 Feb;8(2):139–149. doi: 10.3109/02713688908995185. [DOI] [PubMed] [Google Scholar]
- Wistow G. J., Piatigorsky J. Lens crystallins: the evolution and expression of proteins for a highly specialized tissue. Annu Rev Biochem. 1988;57:479–504. doi: 10.1146/annurev.bi.57.070188.002403. [DOI] [PubMed] [Google Scholar]
- Wistow G., Slingsby C., Blundell T., Driessen H., De Jong W., Bloemendal H. Eye-lens proteins: the three-dimensional structure of beta-crystallin predicted from monomeric gamma-crystallin. FEBS Lett. 1981 Oct 12;133(1):9–16. doi: 10.1016/0014-5793(81)80460-7. [DOI] [PubMed] [Google Scholar]
- Zarina S., Zaidi Z. H., Wania J. H. Free amino acids composition of aqueous humor from Pakistani subjects with senile cataract. J Pak Med Assoc. 1987 Mar;37(3):67–70. [PubMed] [Google Scholar]
- Zigler J. S., Jr, Horwitz J., Kinoshita J. H. Studies on the low molecular weight proteins of human lens. Exp Eye Res. 1981 Jan;32(1):21–30. doi: 10.1016/s0014-4835(81)80035-8. [DOI] [PubMed] [Google Scholar]
- Zigler J. S., Jr, Russell P., Horwitz J., Reddy V. N., Kinoshita J. H. Further studies on low molecular weight crystallins: relationship between the bovine beta s, the human 24kD protein and the gamma-crystallins. Curr Eye Res. 1986 May;5(5):395–401. doi: 10.3109/02713688609025179. [DOI] [PubMed] [Google Scholar]
- van Dam A. F. Purification and composition of beta-s-crystallin. Exp Eye Res. 1966 Oct;5(4):255–266. doi: 10.1016/s0014-4835(66)80035-0. [DOI] [PubMed] [Google Scholar]