Abstract
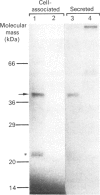
Cathepsin L was partially purified from lysates of freshly isolated macrophages lavaged from lungs of apparently healthy adults and found to be chromatographically and catalytically identical with liver cathepsin L. Western-blotting analysis showed that lung macrophages contain significant levels of a precursor of cathepsin L (43 kDa) in addition to mature enzyme (25 kDa). After culturing for a further 24 h, the precursor disappeared and a new band, corresponding to 34 kDa, appeared, suggesting that the precursor had been processed to an intermediate form of cathepsin L. Biosynthetic labelling of macrophages in vitro with [35S]methionine followed by immunoprecipitation with the cathepsin L antibody confirmed that the cells synthesize cathepsin L as a 43 kDa precursor that is then processed to the mature form (25 kDa) via a 34 kDa intermediate. The precursor, but not the processed forms, was released into the culture medium. During culture in vitro the 34 kDa intermediate accumulated, and little enzyme was processed to the 24 kDa form, consistent with the immunoblot data. Human lung macrophages contain a 1.5 kb transcript of cathepsin L mRNA, whereas none is detectable in human monocytes. These results establish that differentiation of human macrophages within the lung is accompanied by synthesis and expression of an elastinolytic enzyme, cathepsin L. The altered processing of cathepsin L observed during cultivation in vitro suggests caution in the assessment of the elastinolytic potential of human macrophages based on assay in vitro.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baron R., Neff L., Louvard D., Courtoy P. J. Cell-mediated extracellular acidification and bone resorption: evidence for a low pH in resorbing lacunae and localization of a 100-kD lysosomal membrane protein at the osteoclast ruffled border. J Cell Biol. 1985 Dec;101(6):2210–2222. doi: 10.1083/jcb.101.6.2210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Chan S. J., San Segundo B., McCormick M. B., Steiner D. F. Nucleotide and predicted amino acid sequences of cloned human and mouse preprocathepsin B cDNAs. Proc Natl Acad Sci U S A. 1986 Oct;83(20):7721–7725. doi: 10.1073/pnas.83.20.7721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapman H. A., Jr, Allen C. L., Stone O. L., Fair D. S. Human alveolar macrophages synthesize factor VII in vitro. Possible role in interstitial lung disease. J Clin Invest. 1985 Jun;75(6):2030–2037. doi: 10.1172/JCI111922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapman H. A., Jr, Reilly J. J., Jr, Kobzik L. Role of plasminogen activator in degradation of extracellular matrix protein by live human alveolar macrophages. Am Rev Respir Dis. 1988 Feb;137(2):412–419. doi: 10.1164/ajrccm/137.2.412. [DOI] [PubMed] [Google Scholar]
- Chapman H. A., Jr, Stone O. L. Co-operation between plasmin and elastase in elastin degradation by intact murine macrophages. Biochem J. 1984 Sep 15;222(3):721–728. doi: 10.1042/bj2220721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapman H. A., Jr, Stone O. L. Comparison of live human neutrophil and alveolar macrophage elastolytic activity in vitro. Relative resistance of macrophage elastolytic activity to serum and alveolar proteinase inhibitors. J Clin Invest. 1984 Nov;74(5):1693–1700. doi: 10.1172/JCI111586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapman H. A., Jr, Stone O. L., Vavrin Z. Degradation of fibrin and elastin by intact human alveolar macrophages in vitro. Characterization of a plasminogen activator and its role in matrix degradation. J Clin Invest. 1984 Mar;73(3):806–815. doi: 10.1172/JCI111275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Delaissé J. M., Eeckhout Y., Vaes G. Inhibition of bone resorption in culture by inhibitors of thiol proteinases. Biochem J. 1980 Oct 15;192(1):365–368. doi: 10.1042/bj1920365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Etherington D. J., Maciewicz R. A., Mason R. W., Taylor M. A., Wardale R. J. A comparative study of cathepsins N and L and their distribution in different tissues. Prog Clin Biol Res. 1985;180:223–224. [PubMed] [Google Scholar]
- Fertsch D., Schoenberg D. R., Germain R. N., Tou J. Y., Vogel S. N. Induction of macrophage Ia antigen expression by rIFN-gamma and down-regulation by IFN-alpha/beta and dexamethasone are mediated by changes in steady-state levels of Ia mRNA. J Immunol. 1987 Jul 1;139(1):244–249. [PubMed] [Google Scholar]
- Gal S., Gottesman M. M. The major excreted protein of transformed fibroblasts is an activable acid-protease. J Biol Chem. 1986 Feb 5;261(4):1760–1765. [PubMed] [Google Scholar]
- Hasilik A., Neufeld E. F. Biosynthesis of lysosomal enzymes in fibroblasts. Synthesis as precursors of higher molecular weight. J Biol Chem. 1980 May 25;255(10):4937–4945. [PubMed] [Google Scholar]
- Hinman L. M., Stevens C. A., Matthay R. A., Gee J. B. Elastase and lysozyme activities in human alveolar macrophages. Effects of cigarette smoking. Am Rev Respir Dis. 1980 Feb;121(2):263–271. doi: 10.1164/arrd.1980.121.2.263. [DOI] [PubMed] [Google Scholar]
- Ii K., Hizawa K., Kominami E., Bando Y., Katunuma N. Different immunolocalizations of cathepsins B, H, and L in the liver. J Histochem Cytochem. 1985 Nov;33(11):1173–1175. doi: 10.1177/33.11.4056381. [DOI] [PubMed] [Google Scholar]
- Imort M., Zühlsdorf M., Feige U., Hasilik A., von Figura K. Biosynthesis and transport of lysosomal enzymes in human monocytes and macrophages. Effects of ammonium chloride, zymosan and tunicamycin. Biochem J. 1983 Sep 15;214(3):671–678. doi: 10.1042/bj2140671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joseph L. J., Chang L. C., Stamenkovich D., Sukhatme V. P. Complete nucleotide and deduced amino acid sequences of human and murine preprocathepsin L. An abundant transcript induced by transformation of fibroblasts. J Clin Invest. 1988 May;81(5):1621–1629. doi: 10.1172/JCI113497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirschke H., Kembhavi A. A., Bohley P., Barrett A. J. Action of rat liver cathepsin L on collagen and other substrates. Biochem J. 1982 Feb 1;201(2):367–372. doi: 10.1042/bj2010367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason R. W., Gal S., Gottesman M. M. The identification of the major excreted protein (MEP) from a transformed mouse fibroblast cell line as a catalytically active precursor form of cathepsin L. Biochem J. 1987 Dec 1;248(2):449–454. doi: 10.1042/bj2480449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason R. W., Green G. D., Barrett A. J. Human liver cathepsin L. Biochem J. 1985 Feb 15;226(1):233–241. doi: 10.1042/bj2260233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason R. W., Johnson D. A., Barrett A. J., Chapman H. A. Elastinolytic activity of human cathepsin L. Biochem J. 1986 Feb 1;233(3):925–927. doi: 10.1042/bj2330925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason R. W., Walker J. E., Northrop F. D. The N-terminal amino acid sequences of the heavy and light chains of human cathepsin L. Relationship to a cDNA clone for a major cysteine proteinase from a mouse macrophage cell line. Biochem J. 1986 Dec 1;240(2):373–377. doi: 10.1042/bj2400373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mørland B. Cathepsin B activity in human blood monocytes during differentiation in vitro. Scand J Immunol. 1985 Jul;22(1):9–16. doi: 10.1111/j.1365-3083.1985.tb01854.x. [DOI] [PubMed] [Google Scholar]
- Portnoy D. A., Erickson A. H., Kochan J., Ravetch J. V., Unkeless J. C. Cloning and characterization of a mouse cysteine proteinase. J Biol Chem. 1986 Nov 5;261(31):14697–14703. [PubMed] [Google Scholar]
- Silver I. A., Murrills R. J., Etherington D. J. Microelectrode studies on the acid microenvironment beneath adherent macrophages and osteoclasts. Exp Cell Res. 1988 Apr;175(2):266–276. doi: 10.1016/0014-4827(88)90191-7. [DOI] [PubMed] [Google Scholar]
- Villiger B., Kelley D. G., Engleman W., Kuhn C., 3rd, McDonald J. A. Human alveolar macrophage fibronectin: synthesis, secretion, and ultrastructural localization during gelatin-coated latex particle binding. J Cell Biol. 1981 Sep;90(3):711–720. doi: 10.1083/jcb.90.3.711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welgus H. G., Campbell E. J., Bar-Shavit Z., Senior R. M., Teitelbaum S. L. Human alveolar macrophages produce a fibroblast-like collagenase and collagenase inhibitor. J Clin Invest. 1985 Jul;76(1):219–224. doi: 10.1172/JCI111949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wessel D., Flügge U. I. A method for the quantitative recovery of protein in dilute solution in the presence of detergents and lipids. Anal Biochem. 1984 Apr;138(1):141–143. doi: 10.1016/0003-2697(84)90782-6. [DOI] [PubMed] [Google Scholar]
- White R., Lin H. S., Kuhn C., 3rd Elastase secretion by peritoneal exudative and alveolar macrophages. J Exp Med. 1977 Sep 1;146(3):802–808. doi: 10.1084/jem.146.3.802. [DOI] [PMC free article] [PubMed] [Google Scholar]