Abstract
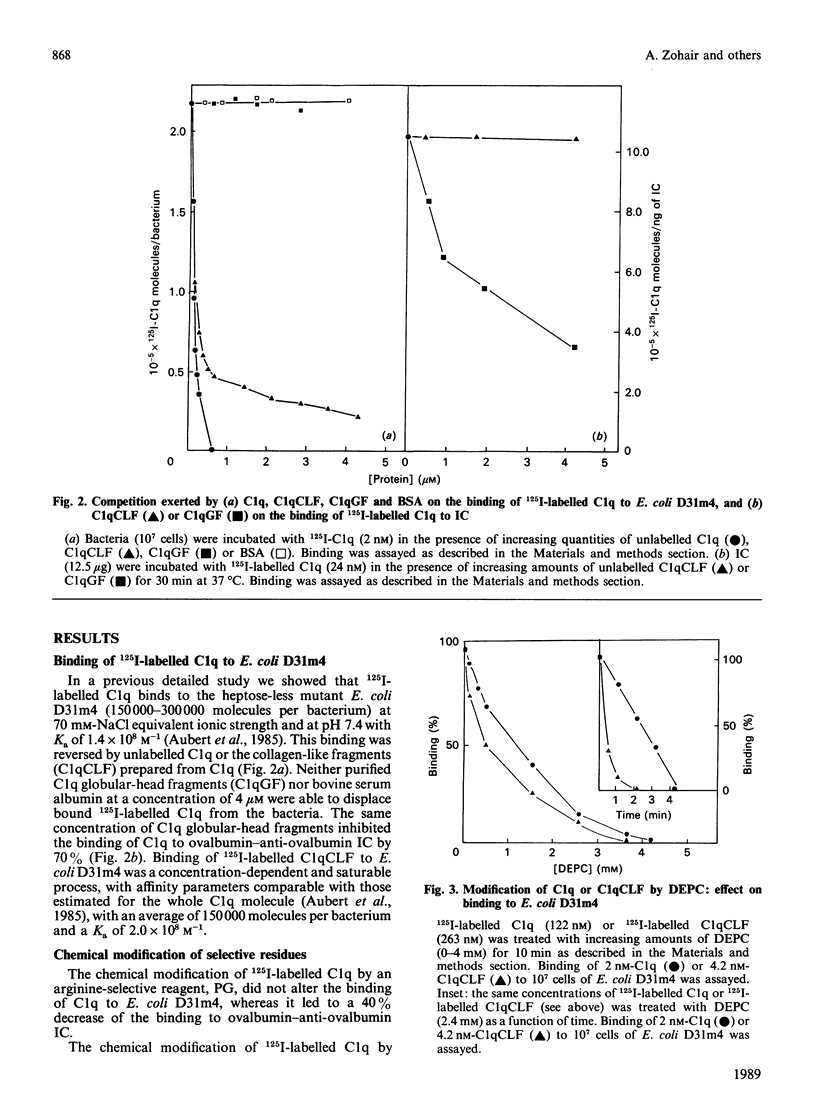
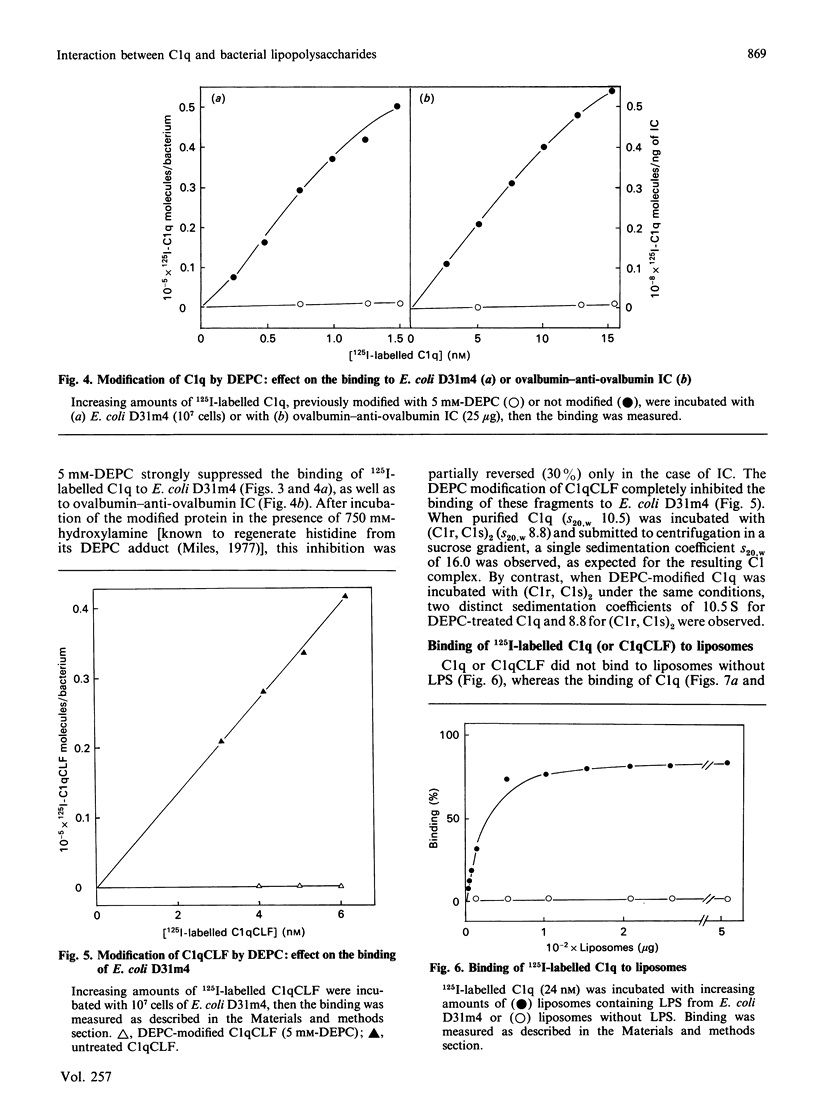
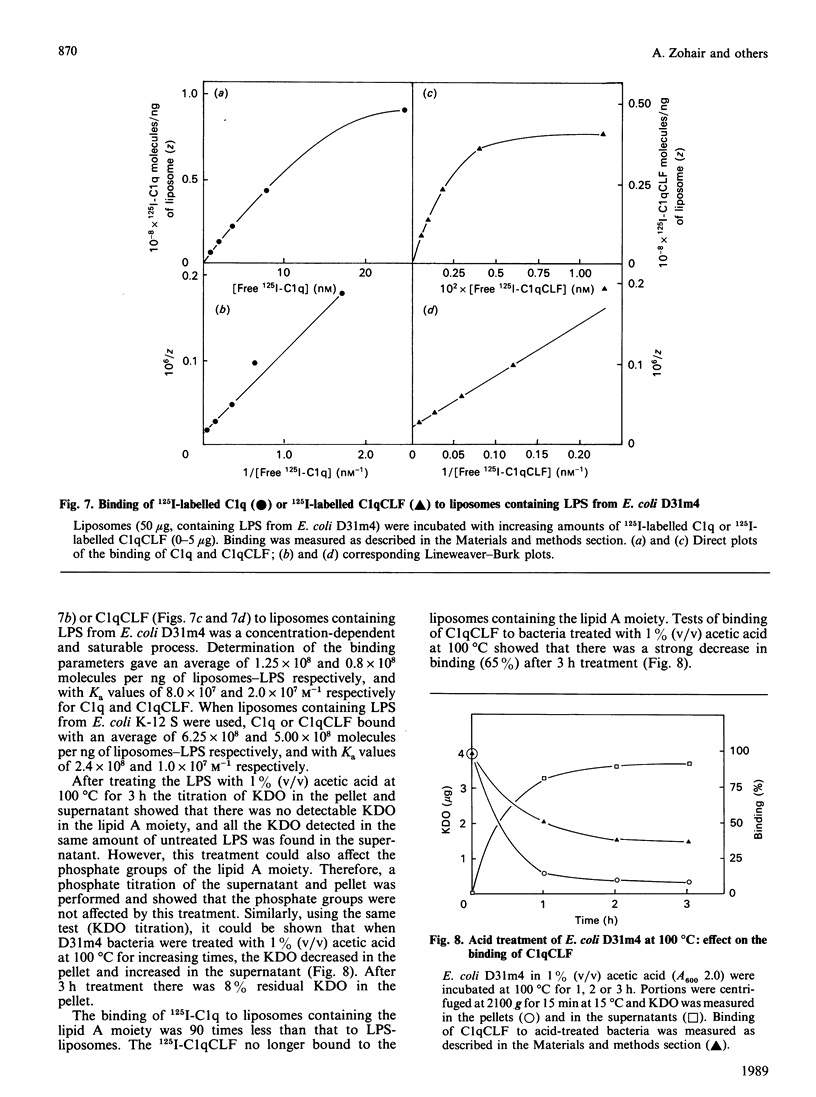
The heptose-less mutant of Escherichia coli, D31m4, bound complement subcomponent C1q and its collagen-like fragments (C1qCLF) with Ka values of 1.4 x 10(8) and 2.0 x 10(8) M-1 respectively. This binding was suppressed by chemical modification of C1q and C1qCLF using diethyl pyrocarbonate (DEPC). To investigate the role of lipopolysaccharides (LPS) in this binding, biosynthetically labelled [14C]LPS were purified from E. coli D31m4 and incorporated into liposomes prepared from phosphatidylcholine (PC) and phosphatidylethanolamine (PE) [PC/PE/LPS, 2:2:1, by wt.]. Binding of C1q or its collagen-like fragments to the liposomes was estimated via a flotation test. These liposomes bound C1q and C1qCLF with Ka values of 8.0 x 10(7) and 2.0 x 10(7) M-1; this binding was totally inhibited after chemical modification of C1q and C1qCLF by DEPC. Liposomes containing LPS purified from the wild-strain E. coli K-12 S also bound C1q and C1qCLF, whereas direct binding of C1q or C1qCLF to the bacteria was negligible. Diamines at concentrations which dissociate C1 into C1q and (C1r, C1s)2, strongly inhibited the interaction of C1q or C1qCLF with LPS. Removal of 3-deoxy-D-manno-octulosonic acid (2-keto-3-deoxyoctonic acid; KDO) from E. coli D31m4 LPS decreases the binding of C1qCLF to the bacteria by 65%. When this purified and modified LPS was incorporated into liposomes, the C1qCLF binding was completely abolished. These results show: (i) the essential role of the collagen-like moiety and probably its histidine residues in the interaction between C1q and the mutant D31m4; (ii) the contribution of LPS, particularly the anionic charges of KDO, to this interaction.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allan R., Rodrick M., Knobel H. R., Isliker H. Inhibition of the interaction between the complement component Clq and immune complexes. Int Arch Allergy Appl Immunol. 1979;58(2):140–148. doi: 10.1159/000232186. [DOI] [PubMed] [Google Scholar]
- Arlaud G. J., Chesne S., Villiers C. L., Colomb M. G. A study on the structure and interactions of the C1 sub-components C1r and C1s in the fluid phase. Biochim Biophys Acta. 1980 Nov 6;616(1):105–115. doi: 10.1016/0005-2744(80)90268-5. [DOI] [PubMed] [Google Scholar]
- Arlaud G. J., Sim R. B., Duplaa A. M., Colomb M. G. Differential elution of Clq, Clr and Cls from human Cl bound to immune aggregates. Use in the rapid purification of Cl subcomponents. Mol Immunol. 1979 Jul;16(7):445–450. doi: 10.1016/0161-5890(79)90069-5. [DOI] [PubMed] [Google Scholar]
- Arlaud G. J., Villiers C. L., Chesne S., Colomb M. G. Purified proenzyme C1r. Some characteristics of its activation and subsequent proteolytic cleavage. Biochim Biophys Acta. 1980 Nov 6;616(1):116–129. doi: 10.1016/0005-2744(80)90269-7. [DOI] [PubMed] [Google Scholar]
- Aubert B., Chesne S., Arlaud G. J., Colomb M. G. Antibody-independent interaction between the first component of human complement, C1, and the outer membrane of Escherichia coli D31 m4. Biochem J. 1985 Dec 1;232(2):513–519. doi: 10.1042/bj2320513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boman H. G., Jonsson S., Monner D., Normark S., Bloom G. D. Cell-surface alterations in Escherichia coli K-12 with chromosmal mutations changing ampicillin resistance. Ann N Y Acad Sci. 1971 Jun 11;182:342–357. doi: 10.1111/j.1749-6632.1971.tb30670.x. [DOI] [PubMed] [Google Scholar]
- Boman H. G., Monner D. A. Characterization of lipopolysaccharides from Escherichia coli K-12 mutants. J Bacteriol. 1975 Feb;121(2):455–464. doi: 10.1128/jb.121.2.455-464.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Bragado R., López de Castro J. A., Juárez C., Albar J. P., García Pardo A., Ortí F., Vivanco-Martínez F. Chemical modification of carboxyl groups in human Fcgamma fragment --II. Location of acidic residues involved in complement activation. Mol Immunol. 1982 Apr;19(4):579–588. doi: 10.1016/0161-5890(82)90227-9. [DOI] [PubMed] [Google Scholar]
- Colomb M. G., Arlaud G. J., Villiers C. L. Structure and activation of C1: current concepts. Complement. 1984;1(2):69–80. doi: 10.1159/000467818. [DOI] [PubMed] [Google Scholar]
- Comis A., Easterbrook-Smith S. B. C1q binding to mitochondria: a possible artefact? FEBS Lett. 1985 Jun 3;185(1):105–108. doi: 10.1016/0014-5793(85)80749-3. [DOI] [PubMed] [Google Scholar]
- Comis A., Easterbrook-Smith S. B. Evidence for arginine residues in the immunoglobulin-binding sites of human Clq. Biochim Biophys Acta. 1985 Sep 27;842(1):45–51. doi: 10.1016/0304-4165(85)90291-0. [DOI] [PubMed] [Google Scholar]
- Cooper N. R., Morrison D. C. Binding and activation of the first component of human complement by the lipid A region of lipopolysaccharides. J Immunol. 1978 Jun;120(6):1862–1868. [PubMed] [Google Scholar]
- Cooper N. R. The classical complement pathway: activation and regulation of the first complement component. Adv Immunol. 1985;37:151–216. doi: 10.1016/s0065-2776(08)60340-5. [DOI] [PubMed] [Google Scholar]
- Easterbrook-Smith S. B. Evidence for histidine residues in the immunoglobulin-binding site of human Clq. Biosci Rep. 1983 Feb;3(2):135–140. doi: 10.1007/BF01121944. [DOI] [PubMed] [Google Scholar]
- Emanuel E. J., Brampton A. D., Burton D. R., Dwek R. A. Formation of complement subcomponent C1q-immunoglobulin G complex. Thermodynamic and chemical-modification studies. Biochem J. 1982 Aug 1;205(2):361–372. doi: 10.1042/bj2050361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fairbanks G., Steck T. L., Wallach D. F. Electrophoretic analysis of the major polypeptides of the human erythrocyte membrane. Biochemistry. 1971 Jun 22;10(13):2606–2617. doi: 10.1021/bi00789a030. [DOI] [PubMed] [Google Scholar]
- Fraker P. J., Speck J. C., Jr Protein and cell membrane iodinations with a sparingly soluble chloroamide, 1,3,4,6-tetrachloro-3a,6a-diphrenylglycoluril. Biochem Biophys Res Commun. 1978 Feb 28;80(4):849–857. doi: 10.1016/0006-291x(78)91322-0. [DOI] [PubMed] [Google Scholar]
- Galanos C., Lüderitz O., Westphal O. A new method for the extraction of R lipopolysaccharides. Eur J Biochem. 1969 Jun;9(2):245–249. doi: 10.1111/j.1432-1033.1969.tb00601.x. [DOI] [PubMed] [Google Scholar]
- Huang L., Kennel S. J. Binding of immunoglobulin G to phospholipid vesicles by sonication. Biochemistry. 1979 May 1;18(9):1702–1707. doi: 10.1021/bi00576a011. [DOI] [PubMed] [Google Scholar]
- Itaya K., Ui M. A new micromethod for the colorimetric determination of inorganic phosphate. Clin Chim Acta. 1966 Sep;14(3):361–366. doi: 10.1016/0009-8981(66)90114-8. [DOI] [PubMed] [Google Scholar]
- Jansson P. E., Lindberg A. A., Lindberg B., Wollin R. Structural studies on the hexose region of the core in lipopolysaccharides from Enterobacteriaceae. Eur J Biochem. 1981 Apr;115(3):571–577. doi: 10.1111/j.1432-1033.1981.tb06241.x. [DOI] [PubMed] [Google Scholar]
- Karkhanis Y. D., Zeltner J. Y., Jackson J. J., Carlo D. J. A new and improved microassay to determine 2-keto-3-deoxyoctonate in lipopolysaccharide of Gram-negative bacteria. Anal Biochem. 1978 Apr;85(2):595–601. doi: 10.1016/0003-2697(78)90260-9. [DOI] [PubMed] [Google Scholar]
- Kulpa C. F., Jr, Leive L. Mode of insertion of lipopolysaccharide into the outer membrane of escherichia coli. J Bacteriol. 1976 Apr;126(1):467–477. doi: 10.1128/jb.126.1.467-477.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin T. Y., Fletcher D. S. Interaction of human Clq with insoluble immunoglobulin aggregates. Immunochemistry. 1978 Feb;15(2):107–117. doi: 10.1016/0161-5890(78)90050-0. [DOI] [PubMed] [Google Scholar]
- Loos M., Bitter-Suermann D., Dierich M. Interaction of the first (C1), the second (C2) and the fourth (C4) component of complement with different preparations of bacterial lipopolysaccharides and with lipid A. J Immunol. 1974 Mar;112(3):935–940. [PubMed] [Google Scholar]
- MARTIN R. G., AMES B. N. A method for determining the sedimentation behavior of enzymes: application to protein mixtures. J Biol Chem. 1961 May;236:1372–1379. [PubMed] [Google Scholar]
- Miles E. W. Modification of histidyl residues in proteins by diethylpyrocarbonate. Methods Enzymol. 1977;47:431–442. doi: 10.1016/0076-6879(77)47043-5. [DOI] [PubMed] [Google Scholar]
- Morrison D. C., Kline L. F. Activation of the classical and properdin pathways of complement by bacterial lipopolysaccharides (LPS). J Immunol. 1977 Jan;118(1):362–368. [PubMed] [Google Scholar]
- Penefsky H. S. Reversible binding of Pi by beef heart mitochondrial adenosine triphosphatase. J Biol Chem. 1977 May 10;252(9):2891–2899. [PubMed] [Google Scholar]
- Porter R. R., Reid K. B. Activation of the complement system by antibody-antigen complexes: the classical pathway. Adv Protein Chem. 1979;33:1–71. doi: 10.1016/s0065-3233(08)60458-1. [DOI] [PubMed] [Google Scholar]
- Prehm P., Stirm S., Jann B., Jann K. Cell-wall lipopolysaccharide from Escherichia coli B. Eur J Biochem. 1975 Aug 1;56(1):41–55. doi: 10.1111/j.1432-1033.1975.tb02205.x. [DOI] [PubMed] [Google Scholar]
- Reid K. B. Complete amino acid sequences of the three collagen-like regions present in subcomponent C1q of the first component of human complement. Biochem J. 1979 May 1;179(2):367–371. doi: 10.1042/bj1790367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reid K. B., Lowe D. M., Porter R. R. Isolation and characterization of C1q, a subcomponent of the first component of complement, from human and rabbit sera. Biochem J. 1972 Dec;130(3):749–763. doi: 10.1042/bj1300749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosner M. R., Tang J., Barzilay I., Khorana H. G. Structure of the lipopolysaccharide from an Escherichia coli heptose-less mutant. I. Chemical degradations and identification of products. J Biol Chem. 1979 Jul 10;254(13):5906–5917. [PubMed] [Google Scholar]
- Sasaki T., Yonemasu K. Chemical studies on the isolated collagen-like and globular fragment of complement component C1q. Comparative studies on bovine and human C1q. Biochim Biophys Acta. 1983 Jan 12;742(1):122–128. doi: 10.1016/0167-4838(83)90367-9. [DOI] [PubMed] [Google Scholar]
- Schumaker V. N., Hanson D. C., Kilchherr E., Phillips M. L., Poon P. H. A molecular mechanism for the activation of the first component of complement by immune complexes. Mol Immunol. 1986 May;23(5):557–565. doi: 10.1016/0161-5890(86)90119-7. [DOI] [PubMed] [Google Scholar]
- Siegel R. C., Schumaker V. N. Measurement of the association constants of the complexes formed between intact C1q or pepsin-treated C1q stalks and the unactivated or activated C1r2C1s2 tetramers. Mol Immunol. 1983 Jan;20(1):53–66. doi: 10.1016/0161-5890(83)90105-0. [DOI] [PubMed] [Google Scholar]
- Sim R. B., Porter R. R., Reid K. B., Gigli I. The structure and enzymic activities of the C1r and C1s subcomponents of C1, the first component of human serum complement. Biochem J. 1977 May 1;163(2):219–227. doi: 10.1042/bj1630219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strain S. M., Fesik S. W., Armitage I. M. Structure and metal-binding properties of lipopolysaccharides from heptoseless mutants of Escherichia coli studied by 13C and 31P nuclear magnetic resonance. J Biol Chem. 1983 Nov 25;258(22):13466–13477. [PubMed] [Google Scholar]
- Tschopp J., Müller-Eberhard H. J., Podack E. R. Formation of transmembrane tubules by spontaneous polymerization of the hydrophilic complement protein C9. Nature. 1982 Aug 5;298(5874):534–538. doi: 10.1038/298534a0. [DOI] [PubMed] [Google Scholar]
- Van Schravendijk M. R., Dwek R. A. Interaction of C1q with DNA. Mol Immunol. 1982 Sep;19(9):1179–1187. doi: 10.1016/0161-5890(82)90328-5. [DOI] [PubMed] [Google Scholar]
- Villiers C. L., Arlaud G. J., Colomb M. G. Diamine-induced dissociation of the first component of human complement, C1. Eur J Biochem. 1984 Apr 16;140(2):421–426. doi: 10.1111/j.1432-1033.1984.tb08119.x. [DOI] [PubMed] [Google Scholar]


