Abstract
Nicotinamide mononucleotide (NMN), a crucial intermediate in NAD + synthesis, can rapidly transform into NAD + within the body after ingestion. NMN plays a pivotal role in several important biological processes, including energy metabolism, cellular aging, circadian rhythm regulation, DNA repair, chromatin remodeling, immunity, and inflammation. NMN has emerged as a key focus of research in the fields of biomedicine, health care, and food science. Recent years have witnessed extensive preclinical studies on NMN, offering valuable insights into the pathogenesis of age- and aging-related diseases. Given the sustained global research interest in NMN and the substantial market expectations for the future, here, we comprehensively review the milestones in research on NMN biotherapy over the past 10 years. Additionally, we highlight the current research on NMN in the field of digestive system diseases, identifying existing problems and challenges in the field of NMN research. The overarching aim of this review is to provide references and insights for the further exploration of NMN within the spectrum of digestive system diseases.
Keywords: Nicotinamide adenine dinucleotide (NAD), Nicotinamide mononucleotide (NMN), Biological therapy, Animal experiments, Clinical trials, Digestive system diseases
Introduction
Nicotinamide mononucleotide (NMN) is a crucial intermediate in the biosynthesis of nicotinamide adenine dinucleotide (NAD +), a coenzyme fundamental to cellular metabolism and energy production. NMN is rapidly converted into NAD + upon ingestion, highlighting its pivotal role in maintaining cellular functions and redox balance [1].
NAD + serves as a pivotal coenzyme involved in various metabolic pathways, including glycolysis, the tricarboxylic acid (TCA) cycle, and oxidative phosphorylation, essential for energy production and cellular viability [1]. Additionally, NAD + acts as a signaling molecule regulating critical cellular processes, such as DNA repair, epigenetic modifications, and immune responses, thereby influencing aging processes and metabolic homeostasis [2].
Accumulating evidence suggests that NAD + levels decline with age in a variety of organisms, including humans and rodents, resulting in age-related pathophysiological changes [3–7] and disorders, such as cognitive decline, cancer, metabolic diseases, sarcopenia, and frailty [2]. Notably, these pathological conditions can be slowed down or even reversed by restoring NAD + levels. Therefore, NAD + metabolism has become a key target in research on aging-related diseases and the prolongation of human health. Moreover, researchers are also increasingly concentrating on interventions with NAD + intermediates to attenuate or even prevent age-related functional decline.
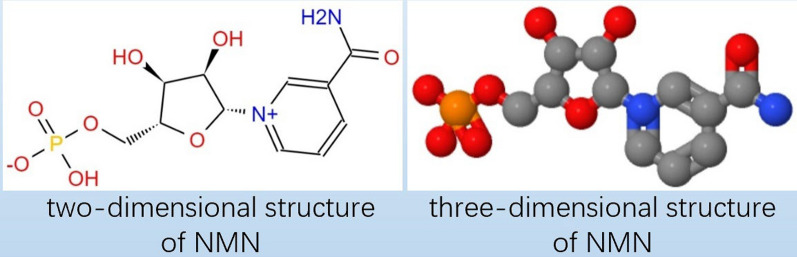
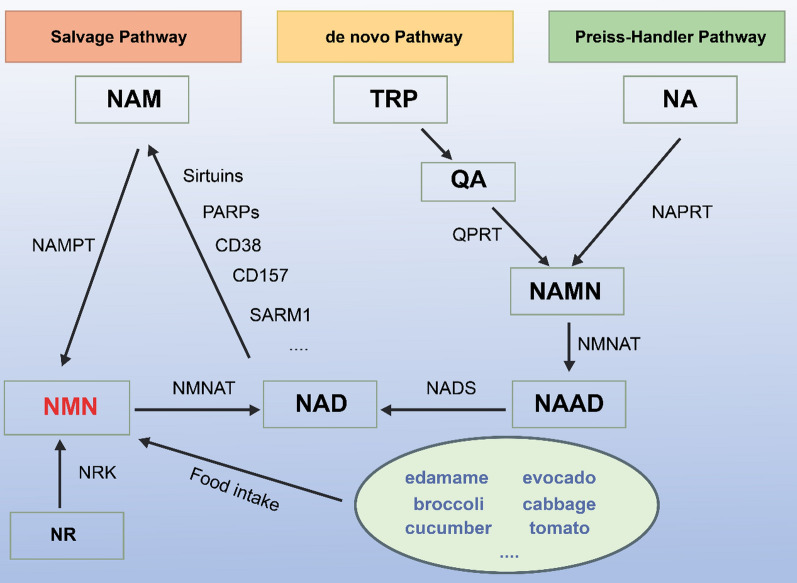
NMN is an acidic water-soluble compound. Chemically, it is a bioactive nucleotide formed by the reaction between phosphate groups and nucleosides composed of ribose and nicotinamide [4]. The molecular formula of NMN is C11H15N2O8P, and its molecular weight is 334.22 g/mol [5]. Figure 1 shows the molecular structure of NMN. Notably, NMN exists in two forms, i.e., α and β isomers, and β-NMN represents its active form [8]. As a natural intermediate of NAD + , NMN is widely present in foods, such as vegetables, fruits, and meat. Moreover, the NMN content is especially high in plant-based foods such as edamame, avocado, broccoli, cabbage, and cucumber [9, 10]. In terms of subcellular distribution, NMN is mainly present in the cytoplasm, nucleus, and mitochondria in mammalian cells. However, in the human body, NMN has also been detected in tissues and body fluids such as the placenta, blood, and urine [11]. In mammals, NMN is synthesized from nicotinamide (NAM) via the rate-limiting enzyme nicotinamide phosphoribosyl transferase (NAMPT). Moreover, it can also be synthesized from nicotinamide riboside (NR) via NR kinase (NRK)-mediated phosphorylation [8, 9]. As previously mentioned, small amounts of NMN can also be ingested via the consumption of NMN-rich foods. Typically, NMN is converted into NAD + via NMN adenylate transferase (NMNAT), which plays a key role in biological regulation and is an important therapeutic target (Fig. 2) [12].
Fig. 1.
The molecular structure of NMN
Fig. 2.
Schematic representation illustrating the conversion of NMN into NAD + via NMNAT. Schematic diagram of NMN synthesis and metabolism. In mammals, NMN can be synthesized from NAM via NAMPT, by NRK-mediated phosphorylation of NR, or through the direct intake of NMN-rich foods. There are three pathways for NAD synthesis: the Salvage Pathway, the de novo Pathway, and the Preiss-Handler Pathway. In the Salvage Pathway, NAM is converted to NMN by NAMPT, which is then converted to NAD by NMNAT. Subsequently, NAD is converted back into NAM by the action of Sirtuins, PARPs, CD38, CD157, SARM1, and other NAD-consuming enzymes, forming a metabolic cycle. In the de novo Pathway, tryptophan from food is converted to quinolinic acid, which is then catalyzed to NAMN by QPRT. In the Preiss-Handler Pathway, NA is converted to NAMN by NAPRT. These two pathways converge when NAMN is transformed into nicotinic acid adenine dinucleotide (NAAD) by NMNAT, and finally into NAD by NADS. NAM: nicotinamide; NAMPT: nicotinamide phosphoribosyl transferase; NR: nicotinamide riboside; NRK: nicotinamide riboside kinase; NMNAT: nicotinamide mononucleotide adenylyl transferase; TRP: tryptophan; QA: quinolinic acid; QPRT: quinolinate phosphoribosyltransferase; NA: nicotinic acid; NAPRT: nicotinic acid phosphoribosyl transferase; NAMN: nicotinate mononucleotide; NMNAT: nicotinamide mononucleotide adenylyl transferases; NAAD: nicotinate adenine dinucleotide; NAD: nicotinamide adenine dinucleotide; NADS, NAD synthase
In recent years, NMN has not only emerged as a key focus of anti-aging research, but has also garnered significant attention due to in vivo studies that have highlighted its wide-ranging pharmacological activities. Thus, NMM has been studied extensively in the field of age-related conditions, such as type 2 diabetes, obesity-related metabolic abnormalities, cardio-cerebral ischemic diseases, and neurodegenerative diseases [1, 8, 10, 11]. Notably, rodent studies have suggested that NMN can effectively enhance NAD + synthesis in peripheral tissues and organs, including the brain [13], blood vessels [14], skeletal muscle [9, 15], adipose tissue [16, 17], pancreas [18], heart [19, 20], liver [9, 21], kidneys [22, 23], and eyes [24, 25], providing several health benefits. In fact, accumulating evidence points to the therapeutic effects of NMN against a variety of key pathophysiological processes in various disease models [1, 12].
This review aimed to comprehensively explore recent advances in NMN biotherapy research, concentrating specifically on its applications in digestive system diseases. By addressing current challenges and emerging trends, novel insights could be provided into NMN's therapeutic efficacy and potential for future research in this dynamic field.
Literature search and selection criteria
Database search
A comprehensive literature search was conducted using multiple databases, including PubMed, Scopus, and Web of Science. The search terms included “NMN,” “nicotinamide mononucleotide,” “biotherapy,” “preclinical studies,” “animal models,” and various specific health conditions (e.g., diabetes, aging, obesity).
Inclusion and exclusion criteria
Inclusion criteria:
Studies published in peer-reviewed journals.
Research involving preclinical models (e.g., mice, rats) where NMN was administered.
Studies demonstrating clear outcomes related to NMN administration (e.g., biochemical, physiological, and molecular effects).
Research that addressed specific mechanisms of action, such as NAD + restoration, SIRT1 activation, and other pathways.
Exclusion criteria:
Non-peer-reviewed articles, reviews, and meta-analyses.
Studies with insufficient methodological details or those that did not include appropriate controls.
Research where NMN was used in combination with other compounds, making it difficult to isolate the effects of NMN alone.
Quality assessment
Each study was assessed for methodological quality, including the design, sample size, duration of treatment, and statistical analysis. Priority was given to studies with robust experimental designs, such as randomized controlled trials (RCTs) in animal models, studies with large sample sizes, and those that included both male and female subjects to ensure generalizability. Studies from renowned research teams and institutions, particularly those with a track record of high-impact publications in the field of aging and metabolic diseases, were given additional consideration.
Quantitative methodology
Data extraction
Detailed information was extracted from each study, including the dosage of NMN, route of administration, duration of the study, animal models used, and the specific health outcomes measured. Quantitative results related to NAD + levels, gene expression, biochemical markers, and physiological effects were documented.
Comparative analysis
Studies were compared based on the consistency of their findings, the dose–response relationship of NMN, and the reproducibility of results across different models and conditions. Meta-analyses of key quantitative outcomes were considered where possible, providing a statistical summary of NMN’s effects on various health parameters.
Representative study selection
From the extensive pool of studies, those that provided clear, reproducible, and significant outcomes were selected as representative examples. Studies were chosen to cover a wide range of conditions affected by NMN, including diabetes, aging, obesity, vascular health, cardiac health, brain health, eye function, kidney function, sepsis, and ovary function. The selection aimed to highlight both the breadth and depth of NMN’s therapeutic potential across different physiological systems and disease models.
Representative pre-clinical studies on NMN biotherapy
The body of literature comprising pre-clinical studies on NMN biotherapy is quite extensive. As it is not feasible to include all preclinical studies on this topic in this review, the contents of the subsequent sections are focused on representative, high-quality studies.
Diabetes
As early as 2011, Yoshino et al., from Professor Imai S’s team at Washington University School of Medicine, established a high-fat diet (HFD)- and aging-induced diabetes mouse model. In this study, NMN was intraperitoneally (i.p.) administered to the model mice at a dose of 500 mg/kg/d and could restore NAD + levels and improve glucose tolerance by enhancing insulin sensitivity and insulin secretion. The researchers speculated that the NAD + synthesis impairment mediated by NAMPT is a crucial pathogenic factor in HFD- and aging-induced diabetes. NMN was observed to potentially enhance hepatic insulin sensitivity and restore the expression levels of genes involved in oxidative stress, inflammatory responses, and circadian rhythm, possibly through SIRT1 activation [18].
Using a different modeling approach, Caton et al. [26] established a diabetes model by feeding C57BL/6 mice a fructose-rich diet (FRD) for 16 weeks. They administered a single dose of NMN (500 mg/kg i.p.) and found that NMN could prevent islet dysfunction in the model mice. In vitro experiments also demonstrated that NMN can prevent TNF-α- and IL-1β-mediated islet dysfunction. Interestingly, the FRD- and proinflammatory cytokine-induced dysregulation of genes encoding islet markers was reversed after NMN treatment. This suggests that a potential anti-inflammatory mechanism is responsible for NMN-mediated islet protection.
Aging
A 2014 report by the Aging Research team led by Professor Sinclair of Harvard Medical School showed that NMN can extend the lifespan of mice by targeting the SIRT2 pathway through NAD + and enhancing the expression of the checkpoint kinase BubR1, which declines with age [27]. In another study, also by Professor Sinclair’s group, a 1-week (500 mg/kg/d i.p.) intervention with NMN was found to enhance the oxidative metabolic capacity of mitochondria in the skeletal muscles of elderly mice. Further, NMN restored mitochondrial homeostasis and key biochemical markers of muscle health in 22-month-old mice to levels similar to those in 6-month-old mice. This suggests that NMN may be effective in mitigating the body’s aging-related decline [15].
To elucidate the anti-aging potential of NMN, Mills et al. from Washington University School of Medicine provided normally aging C57BL/6 mice with NMN intervention therapy for up to 12 months. The results showed that NMN did not have any obvious toxic effects in the mice. However, NMN effectively alleviated age-related physiological decline, inhibited age-related weight gain, increased energy metabolism, and promoted physical activity. Furthermore, NMN was found to prevent age-related changes in gene expression in important metabolic organs and also improve mitochondrial oxidative metabolism and nuclear protein imbalance in skeletal muscle cells [9].
Obesity
Mills et al. also examined the effect of long-term NMN intervention on age-related weight gain in normally aging mice. Their findings showed that NMN administration can significantly inhibit age-related weight gain in conventionally fed mice in a dose-dependent manner [9]. Further, studies have shown that NMN can reduce fat mass and liver and plasma triglyceride levels in obese mice, and can also attenuate the abnormalities in glucose tolerance caused by maternal obesity or long-term HFD intake, thereby preventing metabolic impairments in male offspring born to obese mothers [28]. Moreover, NMN demonstrated potential in reducing body weight in offspring born to obese dams by improving hepatic fat metabolism [29].
Vascular health
In a very interesting study, Das et al. from Professor Sinclair's group observed that a 400 mg/kg/d dose of NMN for 2 months could promote angiogenesis by inhibiting SIRT1-mediated Notch signaling, increasing capillary density and thus improving blood flow and endurance in older C57BL/6J mice [30]. Notably, 8 weeks of NMN treatment was also found to restore markers of arterial SIRT1 activity and reduce age-related endothelial dysfunction and arterial stiffness in mice. Additionally, these improvements were associated with the restoration of NO bioavailability, reduction of oxidative stress, and complete or partial normalization of structural proteins in the arterial walls [14]. In line with these findings, one study showed that NMN therapy can reverse age-related changes in neurovascular mRNA expression in mice, and SIRT1 activation contributes to the neurovascular protective effect of NMN [31]. Interestingly, another study demonstrated that NMN supplementation for 2 weeks can preserve cerebral microvascular endothelial function and the neurovascular coupling response, improve cognitive function, and provide cerebrovascular protection in old mice [32].
Cardiac health
A 3-day NMN (500 mg/kg/d i.p.) intervention in heart-specific Ndufs4 knockout mice could reduce the acetylation of cardiac mitochondrial proteins and increase the sensitivity of the mitochondrial permeability transition, thus exerting cardioprotective effects [33]. Moreover, studies in Friedreich ataxic cardiomyopathy mice have shown that NMN intervention can restore the cardiac function of mice with cardiomyopathy to near-normal levels in a SIRT3-dependent manner [19]. Meanwhile, Nadtochiy et al. found that the protective effect of NMN against cardiac ischemia–reperfusion injury is partly mediated via glycolytic stimulation, and the downstream protective mechanism involves increased ATP synthesis during ischemia and/or enhanced acidosis during reperfusion [34].
In line with these findings, research from the Cleveland Clinic has also demonstrated that NMN can maintain cardiac mitochondrial homeostasis, protecting heart-specific KLF4-deficient mice from heart failure due to stress overload [20]. Similarly, Wu et al. revealed that NMN can alleviate isoproterenol-induced cardiac fibrosis by regulating oxidative stress and Smad3 acetylation [35].
Brain health
According to a study by Stein et al., continuous supplementation with NMN during aging can maintain the proliferation and self-renewal of neural stem and progenitor cells (NSPCs) in mice [13]. Moreover, another study from the group led by Imai S showed that NMN can partially attenuate physical activity deficits in adipocyte-specific Nampt knockout mice [36].
In one study focused on the prevention of ischemic brain injury, mice received NMN intervention at the beginning of reperfusion or 30 min after ischemic injury. The results suggested that NMN protects hippocampal CA1 neurons from ischemic cell death and preserves neuronal function after global cerebral ischemia [37]. Consistent with these findings, Wei et al. demonstrated that NMN has shown promise in potentially treating brain injury caused by intracranial hemorrhage through mechanisms involving inhibition of neuroinflammation and oxidative stress [38]. Furthermore, activation of the Nrf2/HO-1 signaling pathway may contribute to the neuroprotective effects of NMN following intracranial hemorrhage [38].
In a mouse model of global cerebral ischemia, a single round of intervention with 62.5 mg/kg NMN was found to normalize mitochondrial NAD + levels, mitochondrial protein acetylation, and reactive oxygen species (ROS) levels in the hippocampus and inhibit mitochondrial fragmentation in this brain region. Subsequent experiments in a transgenic mouse line demonstrated that these effects were dependent on SIRT3. These results suggest that NMN exerts a protective effect against ischemic brain injury via a SIRT3-dependent mechanism [39].
Further, in a mouse model of Alzheimer's disease, NMN was found to reduce the levels of amyloid precursor protein (APP), reverse the oxygen consumption deficit in brain mitochondria, and reduce mitochondrial fragmentation in neurons, thereby improving the mitochondrial respiratory defect [40]. These findings indicated the neuroprotective role of NMN. Further, evidence demonstrated that the age-related decrease in NAD + biosynthesis mediated by NAMPT in the hippocampus (especially the CA1 region) mice can induce cognitive and behavioral changes. However, short-term NMN supplementation (300 mg/kg/d for 3 weeks) can enhance the expression of Cask in the hippocampus of aged mice to attenuate age-related cognitive impairment [41].
Eye function
A study by Mills et al. used fundus biomicroscopy, retinal electroretinography analysis, and tear volume detection to demonstrate the improvement in eye function after the long-term use of NMN [9]. In Nampt knockout mice, NMN supplementation was found to allow cells to bypass the NAMPT pathway, restoring NAD + levels in photoreceptor cells and reducing photoreceptor cell death. Moreover, NMN supplementation could restore normal glycolysis, mitochondrial function, and metabolic stress responses, thereby preventing metabolic dysfunction and cell death [24]. Interestingly, Chen et al. showed that NMN administration can produce a neuroprotective effect on photoreceptors after retinal detachment and oxidative damage, which is associated with increased NAD + and SIRT1 levels in the damaged retina [25].
UVB irradiation induces corneal edema and apoptosis in C57BL/6 mice and decreases the levels of NAD + and NAMPT in corneal endothelial cells. However, subconjunctival NMN administration (500 mM, 5 μL/eye for 2 consecutive days) can effectively prevent UVB-induced corneal tissue injury and endothelial cell apoptosis in mice. In addition, NMN pretreatment can improve the survival rate and inhibit the apoptosis of human corneal endothelial cells subjected to UVB irradiation [42]. Notably, the study by Lee et al. demonstrates that NMN can prevent retinal dysfunction in mouse models of retinal ischemia/reperfusion injury caused by acute intraocular pressure elevation [43].
Kidney function
NMN supplementation was found to restore renal SIRT1 activity and the NAD + pool in 20-month-old mice, and this effect was even more pronounced in 3-month-old mice. In addition, NMN could significantly protect both young (3 months) and old mice (20 months) from cisplatin-induced acute kidney injury, although SIRT1 gene inhibition weakened the renoprotective effects of NMN [23].
Yasuda et al. showed that short-term NMN treatment in early diabetic nephropathy can exert a renoprotective role by upregulating Sirt1 and activating the NAD + rescue pathway [22]. Furthermore, NMN can also ameliorate adriamycin-induced renal injury via the Twist2-mediated epigenetic inhibition of NMN/NAD consumption [44].
Sepsis
Multiple organ failure caused by sepsis is the main cause of death for patients in intensive care units. In this context, key animal model studies by Cao et al. have shown that NMN can prevent mitochondrial dysfunction, inhibit bacterial transmission in mice with sepsis, and control inflammation via SIRT3 signaling. Thus, NMN can reduce multiple organ damage and improve survival rates among mice with sepsis [45]. Further, in mice with sepsis-associated encephalopathy, NMN can activate the NAD + /SIRT1 signaling pathway and reduce hippocampal inflammatory responses and oxidative stress. Accordingly, NMN can improve memory and reduce neuronal damage in these mice [46].
Ovary function
The quality of oocytes is known to decrease with the aging-related decrease in NAD + levels. Evidence shows that NMN treatment can restore NAD + levels and improve oocyte quality in elderly mice, thereby restoring fertility. Interestingly, this effect can also be recapitulated in transgenic mice overexpressing SIRT2 [47]. In line with these findings, Miao et al.'s study—which was published in the same year—reported that NMN (200 mg/kg/d for 10 consecutive days) promotes cytoplasmic and nuclear maturation; inhibits DNA damage and ROS accumulation; and reduces apoptosis by increasing ovulation, fertilization, and meiotic capacity. As a result, NMN can improve the quality of aging oocytes [48].
Other conditions
Interestingly, NMN has been shown to effectively promote the expansion of mesenchymal stromal cells (MSCs) both in vitro and in vivo, stimulate the osteogenic transformation of endogenous MSCs, and protect mouse bones from aging- and radiation-induced damage. From a mechanistic perspective, evidence shows that NMN treatment upregulates SIRT1 expression, regulates the MSCs in aging bone marrow through the SIRT1 pathway, and thus promotes bone formation and reduces adipogenesis [49].
In a rat model of hemorrhagic shock, rats treated with NMN exhibited significant reductions in lactic acidosis and serum IL-6 levels, both of which are strong predictors of patient mortality. Regardless of whether NMN was used as a pretreatment agent or an adjunct treatment during resuscitation, it increased the time for which rats could endure severe shock before needing resuscitation by nearly 25%. Further, it significantly improved survival after resuscitation [50].
NMN restores the function of the NAD + /SIRT1 axis and thereby effectively alleviates HIF-1α-induced adipose fibrosis and inflammation. This demonstrates the role of NMN in regulating HIF-1α and the adipose tissue fibrosis induced by hypoxia [51].
It is well-known that abnormal immune activation due to HIV-1 infection leads to the depletion of CD4 + T cells. This results in a series of clinical manifestations associated with acquired immune deficiency syndrome. Fortunately, Mo et al. showed that NMN combined with cART treatment can significantly increase the percentage of CD4 + T cells in HIV-infected huPBL mice. Thus, their findings suggest that NMN treatment can improve the therapeutic effect of cART in restoring CD4 + T cell populations in mice [52].
Overall, following the second wave of NAD + research in the early twenty-first century [53], a large number of experimental studies on NMN have been conducted over the past decade. In particular, two large research teams led by Professor Sinclair at Harvard Medical School and Professor Imai at the University of Washington School of Medicine have made outstanding contributions to the application of NMN in the treatment of diseases related to age and aging. These studies have demonstrated the multiple health benefits of NMN (Table 1).
Table 1.
Overview of experimental studies on NMN conducted following the second wave of NAD + research
| Pre-clinical experiment results | |||
|---|---|---|---|
| Model/Animal | Treatment regimen | Outcomes | Reference |
| C57BL/6 mice | 100 or 300 mg/kg/d (PO) for 12 months | Inhibited weight gain; reduced physiological decline; improved insulin sensitivity and lipid distribution; and improved eye function, physical activity, energy metabolism, and mitochondrial function in skeletal muscle | [9] |
| Namptflox/flox mice | 100 or 300 mg/kg/d (PO) for 12 months | Enhanced hippocampal NAD + levels and maintained the nerve stem/progenitor cell pool | [13] |
| C57BL/6 mice | 300 mg/kg (PO) for 8 weeks | Reduced vascular oxidative stress and attenuated age-related endothelial dysfunction and arterial stiffness | [14] |
| SIRT1-iKO, EglN1 KO, SIRT1-Tg mice | 500 mg/kg/d (IP) for 1 week | Enhanced oxidative metabolism in the mitochondria of skeletal muscle in aged mice | [15] |
| High-fat diet-induced mouse model of diabetes | 500 mg/kg/d (IP) for 7–10 days | Restored NAD + levels and enhanced insulin sensitivity/insulin secretion to improve glucose tolerance | [18] |
| Age-induced mouse model of diabetes | 500 mg/kg/d (IP) for 11 days | ||
| Friedreich ataxic cardiomyopathy mice | 500 mg/kg (IP), twice a week, for 4–5 weeks | Improved diastolic function and normalized systolic function, improved cardiac energy production and utilization, reduced overall energy expenditure, and improved cardiac glucose metabolism | [19] |
| Ischemia–reperfusion or cisplatin-induced mouse models of acute kidney injury and SIRT1± mice | 500 mg/kg/d (IP) for 4 days | Restored SIRT1 activity and NAD + levels in mouse kidneys and protected mice from ischemia–reperfusion or cisplatin-induced acute kidney injury | [23] |
| Rod cell-specific Nampt knockout mice | 150 mg/kg (IP) for 4 weeks; 300 mg/kg (IP) for 10 days | Restored NAD + levels, glycolysis, and mitochondrial function in photoreceptor cells; prevented photoreceptor degeneration; restored vision; and protected the retina from light damage | [24] |
| C57BL/6 mice with retinal detachment | 250 mg or 500 mg/kg/d (IP) for 7 days | Increased NAD + levels and SIRT1 expression; reduced photoreceptor cell death; reduced retinal inflammation; and inhibited oxidative stress | [25] |
| C57BL/6 mice fed a fructose-rich diet | 500 mg/kg (IP) single dose | Inhibited inflammation and improved insulin secretion | [26] |
| SIRT2tg mice and BubR1H/+ mice | 500 mg/kg/d (IP) for 7 days | Increased NAD + and BubR1levels and prolonged the lifespan of SIRT2tg/BubR1H/H mice | [27] |
| C56BL6/J female mice | 500 mg/kg/d (IP) for 21 days | Reduced retroperitoneal fat mass, increased quadriceps muscle weight, improved glucose tolerance, and lowered hepatic and plasma triglyceride levels | [28] |
| C56BL6/J mice fed a high-fat diet | 500 mg/kg/d (IP) for 18 days | Increased liver NAD + levels, decreased liver triglyceride levels, improved glucose tolerance, and decreased body weight | [29] |
| SIRT1-iKO mice | 400 mg/kg/d (PO) for 2 months | Restored SIRT1 activity, increased capillary formation and blood flow in skeletal muscle, and improved endurance and positive muscle response to exercise | [30] |
| C57BL/6 mice | 500 mg/kg/d (IP) for 14 days | Restored the neurovascular mRNA expression in aged mice to the levels seen in young mice and improved microvascular endothelial function and the neurovascular coupling response in the brain | [31] |
| C57BL/6 mice | 500 mg/kg/d (IP) for 14 days | Attenuated the cerebrovascular endothelial dysfunction caused by aging, improved the neurovascular coupling response in elderly mice, and restored NAD + levels and mitochondrial energy in microvascular endothelial cells in the aging brain | [32] |
| Heart-specific Ndufs4 knockout mice | 500 mg/kg/d (IP) for 3days | Reduced the acetylation of cardiac mitochondrial proteins and enhanced the sensitivity of mitochondrial permeability transition | [33] |
| Mouse model of cardiac ischemia–reperfusion injury model and Sirt3−/− Mice | 1 mM via perfusion, single dose | Protected the heart by stimulating glycolytic ATP synthesis during ischemia and by enhancing acidosis during reperfusion | [34] |
| Heart-specific KLF4-deficient mice | 500 mg/kg/d (IP) for 3 days | Restored normal mitochondrial acetylation and protected the heart, improved mitochondrial fatty acid oxidation, preserved mitochondrial ultrastructure; and reduced cell death in stress-overloaded hearts | [35] |
| Mouse model of cardiac fibrosis | 500 mg/kg (IP) every 3 days from 5 days before surgery to 4 weeks after surgery | Reduced cardiac dysfunction, fibrosis, and hypertrophy | [36] |
| Adipocyte-specific Nampt-knockout (ANKO) mice | 500 mg/kg (PO) single dose | Partially attenuated impairments in physical activity | [37] |
| Mouse model of cerebral ischemia | 31.25–50 mg/kg (IP) single dose | Protected hippocampal CA1 neurons from ischemic death and preserved neuronal function | [38] |
| Collagenase-induced mouse model of cerebral hemorrhage | 300 mg single dose via tail vein injection; 300 mg/kg/d for 7 days | Prevented acute brain injury, reduced brain cell death and oxidative stress, inhibited neuroinflammation and pro-inflammatory factor levels, and activated the Nrf2/HO-1 signaling pathway | [39] |
| SIRT3-KO mice | 62.5 mg/kg (IP) single dose | Improved hippocampal mitochondrial NAD + levels, inhibited mitochondrial protein acetylation and ROS levels, and attenuated cerebral ischemic injury | [40] |
| Alzheimer's disease mouse model (AD-Tg mice) | 100 mg/kg every other day (SC) for 28 days | Reduced the level of amyloid precursor protein and improved mitochondrial function | [41] |
| Ca1-specific Nampt-knockout mice | 300 mg/kg/d (PO) for 3 weeks | Enhanced the expression of Cask in the hippocampus of older mice to attenuate age-related cognitive impairment | [42] |
| C57BL/6 mice | 250 mg or 500 mg/kg/d (IP) for 24 h, 3 d, and 7 d | Reduced photoreceptor cell death, inhibited retinal inflammation, and reduced oxidative stress | [43] |
| UVB-irradiated C57BL/6 mice | 500 mM and 5 μL/eye (subconjunctival injection) for 2 days | Prevented corneal tissue injury and endothelial cell apoptosis in mice | [44] |
| Mouse model of retinal ischemia–reperfusion injury | 500 mg/kg/d (IP) for 4 days | Prevented retinal dysfunction and reduced retinal inflammation | [45] |
| Diabetic db/db mice | 500 mg/kg/d (IP) for 14 days | Improved the histopathology associated with diabetic nephropathy, increased Nampt expression, provided long-lasting renal protection, and improved the survival rate | [46] |
| Mouse model of doxorubicin-induced focal glomerulosclerosis | 500 mg/kg/d (IP) for 14 days | Decreased urinary albumin levels; alleviated glomerular sclerosis; increased the concentration of NAD + ; enhanced Sirt1 expression and decreased Claudin-1 expression; and upregulated histone methylation and Dnmt1 expression] | [47] |
| C57BL/6 mouse model of sepsis | 500 mg/kg (IP) single dose | Increased NAD + levels in multiple organs and decreased serum lactate levels, oxidative stress, inflammation, and caspase3 activity | [48] |
| Mouse model of sepsis-associated encephalopathy | 500 mg/kg (IP) single dose | Inhibited the hippocampal inflammatory response; alleviated memory impairment; reduced the apoptosis of hippocampal cells; inhibited microglial activation; and prevented hippocampal neuroinflammation and oxidative stress | [49] |
| SIRT2, NMNAT1, and NMNAT3 transgenic mice | 2 g/L (PO) for 4 weeks; 400 mg/kg (PO) single dose | Improved fertility by restoring NAD + levels and restored oocyte quality, embryo development, and live birth rates | [50] |
| ICR female mice | 200 mg/kg/d (IP) for 14 days | Increased the number of ovulations in old mice, enhanced the meiotic and fertilization ability of aging oocytes, and restored mitochondrial function to inhibit ROS-induced apoptosis in aging oocytes | [51] |
| Prx1-Cre mice ColA1flox−stop−flox−SIRT1 mice | 300 mg/kg/d (IP) for 3 months | Promoted MSC expansion in adult mice, stimulated osteogenesis in older mice, inhibited fat generation in older mice, and protected mouse bones from aging and radiation-induced damage | [52] |
| Hemorrhagic shock model in Long-Evans rats | 400 mg/kg/d (PO) for 5 days | Significantly reduced inflammation, improved cell metabolism, and promoted survival after hemorrhagic shock | [53] |
| Hypoxia-induced mouse model of adipose tissue fibrosis | 500 mg/kg/3d (IP) for 4 weeks | Restored the impaired NAD + /SIRT1 axis; inhibited hypoxia-induced HIF-1α activation; and alleviated hypoxia-induced adipose fibrosis and inflammation | [54] |
| HIV-1-infected humanized mice | 300 mg/kg/d (PO) for 24 days | In combination with cART treatment, significantly increased the percentage of CD4 + T cells in HIV-infected huPBL mice | [55] |
IP: intraperitoneal; SC: Subcutaneous; PO: per oral
Current research efforts are actively investigating the underlying mechanisms of resistance to NMN biotherapy, concentrating on metabolic pathways, cellular signaling, and genetic predispositions that may influence treatment outcomes [29, 70]. Strategic approaches to mitigate resistance include exploring combination therapies to synergistically enhance NMN’s efficacy, optimizing dosage regimens based on individual responses, and developing biomarkers for therapeutic monitoring. These efforts aim to elucidate the complex dynamics of NMN resistance and pave the way for personalized medicine approaches that optimize NMN biotherapy across diverse health conditions.
Recently completed clinical trials
Although numerous animal studies have clearly demonstrated that NMN can alleviate diseases associated with age and aging, its effectiveness and safety in humans are still being explored.
In one clinical trial, researchers evaluated the pharmacokinetics and safety of NMN in a cohort of 10 healthy men aged 40–60 years recruited at the Keio University School of Medicine in Japan. They found that NMN does not cause any obvious clinical symptoms. Specifically, no significant changes in body temperature, blood pressure, heart rate, blood oxygen saturation, and laboratory parameters were observed after NMN administration, indicating that a single oral dose of NMN is safe and can be effectively metabolized. Although this was the first human clinical trial for NMN, the evidence from the trial was weak due to the small number of participants and the lack of a placebo control, long-term treatment regimen, and evaluation of tissue NAD + and plasma NMN levels. Given these limitations, the conclusions of the trial must be validated in the future [54].
NMN has demonstrated potential metabolic benefits in female mice with diet-induced diabetes, although comparisons with male mice are limited [18]. Based on these findings, to examine the effect of NMN on metabolism in humans, Yoshino M et al. conducted a 10-week clinical trial among 25 postmenopausal, prediabetic women who were overweight or obese. They found that NMN can upregulate the expression of platelet-derived growth factor receptor β and other genes related to muscle remodeling and thereby enhance insulin signaling and insulin sensitivity in muscle tissue. This study is undoubtedly the first high-quality clinical study focused on the effects of NMN. Nevertheless, the exact mechanisms responsible for the observed metabolic effects and the potential metabolic benefits of NMN supplementation in other populations remain to be explored [55].
Interestingly, a recent 6-week randomized, double-blind, placebo-controlled trial evaluated the effects of NMN on cardiovascular health in amateur runners. In this study, 48 participants took different doses of NMN orally, trained for 40–60 min each time (5–6 times a week), and then underwent cardiopulmonary exercise tests after 6 weeks. Notably, NMN improved the aerobic capacity of the participants during training. Moreover, the findings suggested that this improvement may be due to the enhanced utilization of oxygen by skeletal muscles [56].
Deteriorating sleep quality and physical/mental fatigue in older adults can reduce the quality of life and increase the risk of death. Hence, a research team from the University of Tsukuba in Japan observed the effects of NMN in 108 participants. They found that compared with the intake of NMN in the morning, the intake of NMN during the afternoon (250 mg orally for 2 weeks) may enhance lower limb function and reduce drowsiness in older adults, thereby improving physical and mental health. Unfortunately, this study did not control for the dietary intake of NNM nor monitor the physiological changes that may be caused by NMN intake. Furthermore, the placebo group also showed improvements in the 5-times sit-to-stand (5-STS) test and sleepiness and sleep disorder scores. Nevertheless, this study was the first to demonstrate that the timely intake of NMN can have a positive effect on older human adults [57].
In the same year, a clinical study by Igarashi M and colleagues at the University of Tokyo in Japan showed that the oral administration of 250 mg/d NMN for 12 weeks was safe and well-tolerated in healthy older men and significantly increased the whole-blood levels of NAD + and NAD + -related metabolites. In addition, NMN partially improved muscle function in this population, as indicated by gait speed and grip strength. Although this study demonstrated the safety and partial benefits of NMN in older men, it had several limitations. Firstly, some unexpected events during the study affected the sample size and corresponding data. Secondly, there were some limitations in statistical analysis during the study. Finally, the study only included older men, and therefore, further research in other populations remained necessary [58].
The research team at Toyama University School of Medicine in Japan also conducted a similar clinical trial. In this study, 30 healthy subjects received oral NMN doses (250 mg/d) for 12 weeks, and physiological and laboratory tests were conducted during this period. Notably, oral NMN administration could significantly increase the blood levels of NAD + , and physiological and laboratory tests showed no abnormalities or any obvious adverse reactions [59].
Meanwhile, a randomized, double-blind, parallel-group, placebo-controlled trial evaluated the safety of large oral doses of NMN in healthy adult men and women. Importantly, no serious adverse events were observed among the participants during the study period, suggesting that 1250 mg/d oral NMN is safe and well-tolerated in healthy adults for at least 4 weeks [60].
In a recent multicenter study from India, 80 healthy middle-aged adults (40–65 years) were treated with different oral doses of NMN daily. The primary objective was to assess blood NAD concentrations, while the secondary objective was to evaluate the safety and tolerability of NMN. The results showed that blood NAD concentrations increased in a dose-dependent manner during treatment. Moreover, NMN had a positive effect on the physical endurance and general health of the subjects, and even doses up to 900 mg/d for 60 days were safe and well-tolerated [61].
Akasaka et al. conducted a 24-week NMN intervention study in 14 male patients with diabetes (aged ≥ 65 years) and reduced grip strength (< 26 kg) or walking speed (< 1.0 m/s). The results showed that supplementation with NMN for 24 weeks was safe but could not improve grip strength or walking speed [62].
A 12-week clinical trial was designed to evaluate the effects of long-term NMN administration on biochemical and metabolic health parameters in adults. The results showed that a 250 mg/d dose of NMN was well-tolerated and did not cause adverse effects. However, NMN strengthened NAD + metabolism in healthy middle-aged adults and showed the potential to alleviate arterial stiffness [63].
Based on these clinical studies (Table 2), it appears that NMN is relatively safe in humans. Additionally, the results of these initial human clinical trials appear promising, although there is still a long way to go. Notably, given the influx of NMN-based anti-aging products in the market, more high-quality clinical studies are urgently required to determine the efficacy and safety of NMN.
Table 2.
Summary of clinical studies evaluating the safety and efficacy of NMN in humans
| Clinical trial results | ||||
|---|---|---|---|---|
| Subjects | Design | Treatment regimen | Outcomes | References |
| 10 healthy men (40–60 years) | Single-arm, non-random intervention | A single oral dose of 100, 250, or 500 mg | NMN did not cause any significant clinical symptoms; body temperature, blood pressure, heart rate, blood oxygen saturation, and related laboratory parameters were not significantly changed; the plasma concentrations of N-methyl-2-pyridine-5-formamide and N-methyl-4-pyridine-5-formamide showed dose-dependent increases; and eye examination findings and sleep quality scores remained unchanged | [57] |
| 25 overweight or obese postmenopausal, prediabetic women | Randomized, double-blind, placebo-controlled | 250 mg/d orally for 10 weeks | NMN increased skeletal muscle insulin signaling, insulin sensitivity, and muscle remodeling in the participants | [58] |
| 48 amateur runners (40 men and 8 women) | Randomized, double-blind, placebo-controlled | 300, 600, or 1200 mg/d orally for 6 weeks | NMN improved the aerobic capacity of the subjects during exercise training | [59] |
| 108 older adults (> 65 years) | Randomized, double-blind, placebo-controlled | 250 mg/d orally for 12 weeks | NMN treatment in the afternoon was effective in improving lower limb function and reducing drowsiness in older adults | [60] |
| 65 healthy older men (> 65 years) | Randomized, double-blind, placebo, parallel control | 250 mg orally for 6 or 12 weeks | NMN significantly increased the levels of whole blood NAD + and NAD + -related metabolites; partially improved muscle function; and was safe and well-tolerated at an oral dose of 250 mg/d for 12 weeks in the test population | [61] |
| 30 healthy adults | Randomized, double-blind, placebo-controlled, parallel-group study | 250 mg/d orally for 12 weeks | NMN significantly increased whole blood NAD + levels, but it did not cause any abnormalities in physiological and laboratory parameters or cause any significant adverse reactions | [62] |
| 32 healthy adult men and women | Randomized, double-blind, placebo-controlled, parallel-group study | 1250 mg/d orally for 4 weeks | Oral NMN (1250 mg/d) was safe and well-tolerated in healthy adults for up to 4 weeks | [63] |
| 80 healthy middle-aged adults (40–65 years) | Randomized, multicenter, double-blind, placebo-controlled, parallel-group study | 300, 600, or 900 mg/d for 60 consecutive days | NAD concentrations in the blood increased in a dose-dependent manner; the clinical efficacy was the highest at an oral dose of 600 mg/day; NMN was safe and well-tolerated at doses of up to 900 mg/day for 60 days; and NMN supplementation had a positive effect on physical endurance and general health in the test population | [64] |
| 14 male diabetes patients with reduced grip strength or walking speed (≥ 65 years) | Double-blind, placebo-controlled | 250 mg/d orally for 24 weeks | Supplementation with NMN for 24 weeks was safe but did not improve grip strength or walking speed | [65] |
| 36 healthy middle-aged subjects (40–59 years) | Randomized, double-blind, placebo-controlled | 250 mg/d orally for 12 weeks | NMN was well-tolerated at an oral dose of 250 mg/d and did not cause adverse events; strengthened NAD + metabolism in healthy middle-aged adults; and showed the potential to alleviate arterial stiffness | [66] |
Status of research on NMN as a therapeutic agent for digestive system diseases
NMN has been extensively studied as a therapeutic agent for various conditions, including aging-related disorders, diabetes, neurodegenerative diseases, and cerebrovascular ischemic diseases [28, 32]. Furthermore, it has also received immense attention in studies on kidney, eye, and reproductive system disorders. However, there is limited research on the use of NMN as an intervention in cases of digestive system diseases, and most existing studies have focused on the liver and intestine.
Liver disease
An HFD severely impairs NAMPT-mediated NAD + biosynthesis in metabolic organs. However, NMN not only reduces glucose intolerance by attenuating the HFD-induced NAD + decline but can also partly enhance insulin sensitivity in the liver by activating SIRT1 [18]. Further, NMN can reverse the activity of endogenous SIRT3 in the liver of Bmal1 knockout mice, restore mitochondrial lipid oxidation in circadian rhythm mutants, and thus enhance hepatic mitochondrial respiratory function [21].
To elucidate the pathogenesis of early alcohol-related liver disease (ALD), Assiri et al. established an animal model of chronic ALD. After treatment with NMN every alternate day (500 mg/kg i.p.), they examined its therapeutic effect using RNA-seq, metabolomic analysis, and other methods. NMN was found to increase liver NAD + levels, prevent ethanol-induced elevations in alanine transaminase (ALT) and aspartate transaminase (AST), and alter 25% of the genes regulating ethanol metabolism. Further, NMN also restored normal Erk1/2 signaling and prevented Atf3 overexpression. These results suggest that NMN supplementation can alter hepatic gene expression and signaling in early ALD, thereby reducing the hepatotoxicity of ethanol [64].
The treatment of liver fibrosis continues to be a significant clinical challenge. A research team from the School of Life Sciences, Tsinghua University, used proteomic analysis to demonstrate that NMN inhibits the secretion of pro-fibrotic proteins and redox enzymes in activated hepatic stellate cells (HSCs). Further, it downregulates the expression of pro-fibrotic genes and causes HSC inactivation. Next, by using a mouse model of liver fibrosis, the researchers revealed that NMN can reduce extracellular matrix accumulation in the liver and also inhibit oxidation-mediated 15-PGDH degradation, thereby promoting PGE2 degradation and inhibiting HSC activation [65].
To further explore the mechanisms underlying liver aging, the researchers performed quantitative proteomic analysis using tissues from aging mice and found that the most upregulated proteins were in fact associated with redox processes. Further analysis revealed that malondialdehyde and protein carbonyl levels increased during aging, and nuclear Nrf2 and its downstream genes were significantly upregulated, suggesting that oxidative stress induced hepatic Nrf2 activation in aging mice. However, NMN intervention successfully counteracted these changes. Further experiments showed that NMN can increase SIRT3 activity and reduce age-related acetylation in Sod2 in mice. However, the effect of NMN on nuclear Nrf2 levels is weaker in SIRT3-deficient mice. Finally, this study demonstrated that NMN supplementation can restore the hepatic redox balance in aging mice via the SIRT3-Nrf2 axis and protect the aging liver from oxidative stress-induced damage [66]. Moreover, in another study, the research team found that protein acetylation plays a key role in maintaining redox homeostasis and energy metabolism during aging. Further, they showed that NMN intervention can hinder the increase in protein acetylation associated with aging and regulate fatty acid β-oxidation, the TCA cycle, and valine degradation, thus attenuating aging-related dysfunction [67].
Jia et al. established an orthotopic liver transplantation model in Sprague–Dawley rats to explore the effects of NMN on ischemia–reperfusion injury caused by a donor liver from a rat who experienced cardiac death. They found that NMN showed potential in reducing liver dysfunction, pathological injury, and apoptosis induced by ischemia–reperfusion injury [68]. Specifically, these benefits may be dependent on enhanced PINK1/Parkin-mediated mitochondrial autophagy and improvements in the antioxidant capacity of the liver via SIRT3 regulation [68].
Xiao et al. studied the effect of NMN on alcohol-induced insulin resistance in hepatocytes. Their findings suggested that NMN exerts some protective effects against alcohol-induced insulin resistance in hepatocytes. Moreover, the mechanisms may be related to the upregulation of the cellular NAD/SIRT1 pathway and the recovery of the PI3K/AKT insulin signaling pathway [69].
Gut and pancreatic diseases
In a mouse model of inflammatory bowel disease (IBD) induced by dextran sodium sulfate, NMN treatment was found to improve intestinal morphology, restore intestinal mucosal barrier function, and reduce the serum expression of proinflammatory factors. In addition, significant beneficial changes in the composition and abundance of intestinal microbiota were observed in mice after NMN treatment [70]. Subsequent research showed that NMN increases the abundance of butyric-producing bacteria and other probiotics and decreases the abundance of several harmful bacteria. In fact, significantly higher levels of bile acid-related metabolites were observed in stool samples from the treatment group than in samples from the control group. In addition, long-term NMN treatment (15 weeks) increased the number of intestinal goblet cells and mucus thickness, as well as the expression of tight junction proteins [71]. These results ultimately indicate that NMN provides intestinal protection by inhibiting intestinal inflammation, reducing the permeability of the intestinal mucosa, and preventing intestinal dysbiosis.
Another study used cellular and animal models of senescence to explore the effects of NMN supplementation on the gut. NMN treatment for 4 months significantly increased the content of NAD + in the jejunum of older mice and improved jejunum structure. In addition, NMN significantly upregulated SIRT3, SIRT6, Nrf2, HO-1, GCLC, SOD2, occludin, and claudin-1 while downregulating TNF-α. In vitro, NMN restored the normal mRNA expression of IL-6, IL-1A, NF-κB1, and claudin-1. The mRNA levels of antioxidant enzymes were also significantly affected, and the vitality and total antioxidant capacity of cells were enhanced after NMN treatment. These results demonstrated the potential of NMN in improving intestinal structure and preventing functional decline [72].
Sleep deprivation impairs intestinal colonization and resistance and causes intestinal dysbiosis in mice. However, NMN supplementation may counteract these negative effects of sleep deprivation by allowing the redistribution of the microbiome-associated bile acid reservoir [73].
Zhao et al. reported that long-term NMN supplementation demonstrated potential in reducing intestinal fibrosis and restoring microecological balance after disruptions induced by ionizing radiation [74].
In an interesting study that used an animal model of acute pancreatitis (AP), fecal microbiome transplantation was found to induce an increase in NMN in AP rats/mice. Further, NMN alleviated AP-related mitochondrial dysfunction, oxidative damage, and inflammation by increasing pancreatic NAD + levels. This suggested that the increase in intestinal microbiota-derived NMN reduces the severity of AP by activating the SIRT3-PRDX5 pathway [75].
Here, we have provided an updated overview of the key preclinical studies reporting the potential of NMN as a therapeutic agent for digestive system disorders. However, the digestive system—as an important physiological organ system—includes a wide range of organs like the esophagus, stomach, small intestine, colon, rectum, liver, biliary tract, and pancreas. Moreover, it performs many important physiological functions including nutrient transport, digestion, absorption, metabolism, immune regulation, internal and external secretion, detoxification, and defecation. Therefore, considerably more effort and time are required to explore the broad applications and therapeutic potential of NMN in digestive system disorders. Surprisingly, a systematic review published in Gastroenterology highlighted that digestive diseases accounted for more than one-third of all diseases globally in 2019, highlighting the enormous challenge associated with their treatment [76]. Unfortunately, studies on NMN in the field of digestive system diseases are currently limited to a small set of diseases or pathological conditions of the liver and intestine. However, on a more positive note, this indicates that several unexplored questions that need to be answered remain and warrant further investigation.
Nevertheless, previous studies have confirmed that NAD + is highly expressed in the liver [9], pancreas [18], and small intestine [77]. Thus, it is essential to explore the potential benefits of NMN in various digestive system diseases and to elucidate the mechanisms. This greatly encourages and promotes our enthusiasm for continued in-depth research in the vast field of digestive system disorders.
Molecular mechanisms of NMN in digestive system diseases
NMN plays a pivotal role in cellular metabolism as a precursor to nicotinamide adenine dinucleotide (NAD +), a coenzyme critical for various metabolic processes. Understanding the molecular mechanisms by which NMN influences digestive system diseases requires examining its impact on cellular pathways relevant to gastrointestinal health [11, 25].
Energy metabolism and gut barrier function
NMN supplementation affects energy metabolism and mitochondrial function, which are crucial for maintaining gut barrier integrity. In conditions, such as inflammatory bowel disease (IBD), disruptions in gut barrier function could lead to increased intestinal permeability and inflammation. NMN enhances mitochondrial function by boosting NAD + levels, thereby promoting cellular energy production [12, 78]. This supports the repair and maintenance of the intestinal epithelial barrier, potentially reducing inflammation and improving gut health.
Anti-inflammatory effects
NAD + is essential for the activity of sirtuins, a class of proteins with anti-inflammatory properties. NMN-mediated elevation of NAD + levels activates sirtuins, which regulate inflammatory responses in the gastrointestinal tract [61]. In diseases, such as ulcerative colitis and Crohn's disease, excessive inflammation contributes to tissue damage. NMN's activation of sirtuins can mitigate inflammation by deacetylating histones and transcription factors, thereby suppressing pro-inflammatory gene expression [28, 31].
Oxidative stress reduction
Oxidative stress plays a significant role in the pathogenesis of digestive system diseases. NMN, through its role in enhancing cellular antioxidant defenses, helps combat oxidative stress [25]. NAD + serves as a cofactor for enzymes such as PARPs (poly ADP-ribose polymerases) and sirtuins, which regulate DNA repair and antioxidant responses. By replenishing NAD + levels, NMN supports these enzymatic activities, thereby reducing oxidative damage to gastrointestinal tissues [29].
Regulation of gut microbiota
Emerging evidence suggests that NAD + metabolism influences the composition and function of the gut microbiota. Dysbiosis, an imbalance in gut microbial communities, is associated with various digestive disorders. NMN's effects on NAD + levels can modulate microbial metabolism and diversity, potentially restoring a healthy gut microbiota composition [32]. This microbial balance is crucial for immune regulation, inflammation modulation, and overall gastrointestinal homeostasis.
Cellular senescence and tissue regeneration
NMN’s ability to enhance NAD + levels supports cellular processes involved in tissue repair and regeneration. In conditions like gastritis or intestinal injury, NMN may promote the proliferation and differentiation of intestinal stem cells [25]. This cellular rejuvenation can accelerate tissue healing and restore normal gastrointestinal function.
Consequently, the molecular mechanisms underlying NMN's actions in digestive system diseases are multifaceted, primarily centered around its role in replenishing cellular NAD + levels. By enhancing energy metabolism, reducing inflammation and oxidative stress, regulating gut microbiota, and promoting tissue regeneration, NMN shows potential therapeutic benefits for conditions affecting the gastrointestinal tract [61]. However, further research, particularly clinical trials in humans with specific digestive disorders, is needed to validate these mechanisms and evaluate NMN's efficacy and safety in clinical settings.
NMN: a balanced assessment
NMN has garnered significant interest in the field of aging and metabolic health due to its role as a precursor to nicotinamide adenine dinucleotide (NAD +), a crucial coenzyme in cellular processes. While several studies suggest NMN supplementation may offer various health benefits [28, 29, 70], it is imperative to critically evaluate these findings and acknowledge the limitations and conflicting results in the existing research.
Potential benefits of NMN
Aging and longevity: NMN has been shown to increase NAD + levels, which tend to decline with age. Research on rodents indicates that NMN supplementation can improve mitochondrial function, enhance physical activity, and potentially extend lifespan [78]. For instance, a study by Mills et al. [9] found that NMN restored NAD + levels and mitigated age-associated physiological decline in mice.
Metabolic health: NMN has demonstrated promise in improving metabolic health markers. Studies suggest NMN can enhance insulin sensitivity, reduce weight gain, and improve lipid profiles in obese and diabetic mice [12]. These findings are promising for conditions, such as type 2 diabetes and metabolic syndrome.
Neuroprotection: There is evidence to suggest NMN may have neuroprotective effects. Research indicates that NMN can cross the blood–brain barrier and may protect against cognitive decline and neurodegenerative diseases by supporting neuronal NAD + levels [77].
Limitations and critique of existing research
Methodological limitations: A significant portion of NMN research has been conducted in animal models, particularly on mice. While these studies provide valuable insights, the results may not fully translate to humans due to physiological differences. Additionally, numerous studies used high doses of NMN, which may not be practical or safe for human consumption.
Small sample sizes and short durations: Human trials on NMN are relatively few, with many having small sample sizes and short study durations. For example, a study by Igarashi et al. [58] involved a small cohort and a brief supplementation period, limiting the ability to draw long-term conclusions.
Potential biases: Some studies may be subjected to publication bias, where positive results are more likely to be published than negative or inconclusive findings. This can skew the perceived efficacy of NMN. Moreover, conflicts of interest, particularly in industry-funded research, can introduce bias.
Conflicting findings: Not all studies uniformly support the benefits of NMN. Some research has shown minimal or no effect on certain health parameters. For instance, a study [70] on healthy older adults found that NMN supplementation did not significantly alter insulin sensitivity or other metabolic markers. These discrepancies highlight the need for further research.
Safety and long-term effects: While NMN is generally considered safe, long-term safety data in humans are limited. Potential side effects and the impact of chronic supplementation are areas that require more extensive investigation.
Investigations into the efficacy of NMN biotherapy in combination with conventional pharmacotherapies have yielded diverse findings. Studies [25, 33] suggested that NMN could exhibit synergistic effects when administered alongside medications targeting metabolic diseases, such as diabetes and cardiovascular diseases. Conversely, the interaction between NMN and certain drugs requires careful consideration due to potential impacts on bioavailability or efficacy. These observations highlight the importance of further research to elucidate optimal therapeutic combinations and potential interactions, aiming to enhance treatment outcomes and ensure patient safety.
Problems and challenges in the field of NMN research
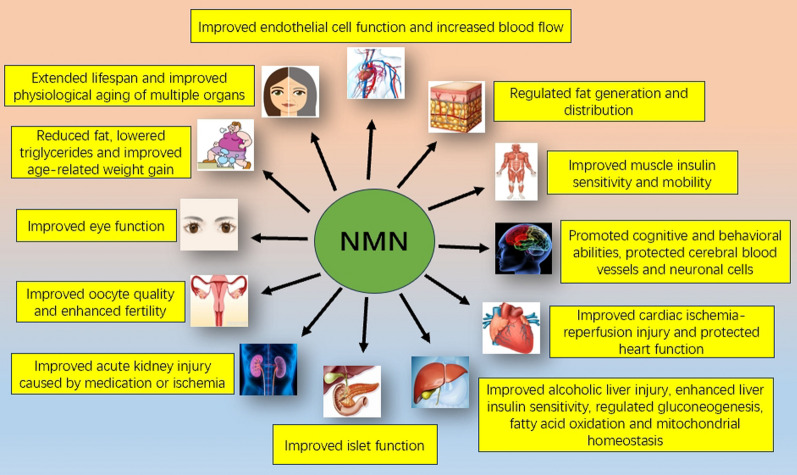
It is undeniable that numerous in vivo experiments conducted in mammals have demonstrated the benefits of NMN supplementation on experimental animals under various disease conditions (Fig. 3). However, it is crucial to acknowledge the existing limitations in current research. Specifically, the signaling molecules downstream of NAD + responsible for mediating the benefits of NMN remain elusive. Additionally, there are complex interactions among various NAD + -dependent biochemical reaction processes, and the metabolism of NMN in the human body may exhibit heterogeneity across different tissues and organs. Moreover, the impact of NMN on physiological function in humans may be the result of interactive effects on multiple organs and aspects [6]. At present, human clinical trial results do not entirely align with the results from previous animal experiments, and the recommended NMN dosage is still under investigation. Therefore, extensive research efforts are needed to address these specific issues and challenges.
Fig. 3.
Illustrating the beneficial effects of NMN supplementation outlined in numerous in vivo experiments conducted in mammals under various disease conditions
Compared with other well-characterized medications, NMN therapy presents several specific limitations:
Mechanistic uncertainty: In some other medications with well-defined mechanisms of action, the exact signaling pathways downstream of NAD + that mediate NMN’s benefits are not fully understood, making therapeutic outcomes difficult to predict and control.
Complex metabolic interactions: NMN is involved in numerous NAD + -dependent biochemical reactions, leading to complex interactions that have not yet been fully mapped. Other medications often have more targeted mechanisms, resulting in fewer unknowns and more predictable effects [79].
Tissue-specific metabolism: The metabolism of NMN can vary significantly across different tissues and organs, potentially leading to inconsistent therapeutic outcomes [29]. Numerous conventional medications are designed with tissue-specific targeting mechanisms to ensure uniformity in their therapeutic effects.
Clinical efficacy discrepancies: Human clinical trial results for NMN do not always align with those from animal studies. Numerous drugs that have passed multiple phases of clinical trials have well-documented efficacy and safety profiles, whereas NMN still requires extensive clinical validation.
Furthermore, it is imperative to thoroughly evaluate the potential side effects of NMN. While no significant adverse reactions have been reported in animal experiments and human clinical trials to date, Di Stefano et al. [78] found that FK866, an inhibitor of NMNPT (a key rate-limiting enzyme in NMN synthesis), extends the functional survival of cross-cutting axons and their synapses by blocking the elevation of NMN. This delays vincristine-induced neurodegeneration, suggesting that NMN accumulation may promote axonal degeneration after nerve injury. Additionally, some reports suggest that NMN deamidase can delay axonal degeneration in zebrafish larvae and transgenic mice, further indicating that NMN accumulation in axons has a degenerative effect in vivo [79].
When high-dose NMN supplementation surpasses the human body's requirement for niacin equivalents, one of the primary methods via which niacinamide is eliminated is methylation by niacinamide N-methyltransferase. However, whether long-term high-dose NMN supplementation can result in the depletion of methyl donors and potentially cause new abnormalities warrants further attention [12]. Although NAD + supplementation can promote DNA repair [80], certain chemotherapeutic anticancer drugs designed to enhance NAD + consumption may also promote tumor growth [81]. SIRT1, as a downstream target of NAD + , has been found to exhibit either carcinogenic or anticancer effects under different conditions [82].
In recent years, NMN has also garnered attention for its potential application in cancer therapy. Studies [61, 78, 79] have explored NMN’s role in modulating NAD + level to influence cancer cell metabolism and survival pathways. For instance, NMN has exhibited promise in preclinical models of various cancer types, including breast, colon, and pancreatic cancers, by enhancing mitochondrial function, promoting DNA repair mechanisms, and inhibiting tumor growth [12, 82]. Moreover, NMN’s ability to activate sirtuins and regulate cellular senescence pathways suggested a multifaceted approach in cancer treatment [9, 77]. Further clinical trials are warranted to assess the efficacy and safety of NMN in cancer patients. Overall, it is evident that a significant amount of clinical research is still required to elucidate the contraindications and long-term safety of NMN in the treatment of diseases associated with human aging.
While preliminary research and animal studies [25, 70] showed promising results regarding NMN's potential benefits, such as anti-aging effects and metabolic improvements, it is crucial to conduct robust, large-scale clinical trials in human populations. These trials are essential to definitively establish NMN's efficacy, safety profile, and optimal dosage regimens in real-world scenarios. Well-designed clinical trials can provide rigorous evidence that can withstand scientific scrutiny and regulatory requirements. They may address important questions, such as long-term safety, potential side effects, and whether NMN truly delivers on its purported health benefits in diverse populations. Additionally, these trials can explore interactions with other medications and conditions, providing comprehensive insights into NMN's therapeutic potential.
Outlook
Unlike NAM, niacin, and nicotinamidemoside, NMN is the most direct and efficient precursor of NAD + . In fact, it can be used for NAD + synthesis in a wide range of organisms, from yeasts to humans [5–7].
The existence of specific transporters involved in NMN metabolism has been a longstanding scientific question. However, the successful identification of the small intestine-specific NMN transporter Slc12a8 has partly answered this question [77]. Nevertheless, the significance of Slc12a8 in the pathophysiology of aging-related changes remains unclear. Similarly, we are yet to understand whether the uptake of NMN is tissue-specific. Further, questions regarding the dosage and frequency of long-term NMN administration, considering age-related differences or health conditions, need to be answered urgently. The multifunctional role of NMN and its distribution in multiple organelles suggest that NMN may have different biological functions in multiple organelles. Thus, deciphering the mechanistic actions of NMN in each subcellular compartment remains a challenge to be solved. These problems must be addressed through in-depth research, and the journey could be extremely interesting, albeit challenging.
Although numerous preclinical studies offer highly promising possibilities for NMN development, its safety and efficacy in the human body need to be confirmed. Toxicological and clinical evidence supporting the clinical effectiveness of NMN remains scarce. Therefore, in addition to animal studies, a large number of clinical trials should also be conducted to assess the safety and efficacy of NMN, focusing on toxicological parameters and safe metabolic levels in humans [10–12].
These exciting results in the field of NMN biotherapeutics have created new breakthroughs and avenues for research. With more preclinical and clinical research and the exploration of new pharmacological applications of NMN, this compound may someday serve as an "all-in-one" interventional strategy and a gold-standard biotherapeutic agent, ushering in a new era of treatment in the field of aging-related diseases.
Acknowledgements
The authors would like to thank all the reviewers who participated in the review, as well as MJEditor (www.mjeditor.com) for providing English editing services during the preparation of this manuscript.
Author contributions
Li Rong and Guanyi Liao jointly conceived and drafted the review, Yuchen Xie undertook all the works of this major revision, Xinsen Zou and Faguo Yue suggested some important changes to the content of the review, Hong Peng and Tianke Li refined the language, and Jinjun Guo and Li Rong directed and proofread the review throughout. All authors contributed to the article and approved the submitted version.
Funding
Chongqing Natural Science Foundation (CSTB2022NSCQ-MSX1257, CSTB2023NSCQ-MSX0494) and Chongqing Municipal Education Commission Science and Technology Research Plan(KJQN202300475)supported this work.
Availability of data and materials
Date are available by emailing the corresponding author.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publications
The authors agree to publish this manuscript in the Journal of Translational Medicine.
Competing interests
The authors declare that the review was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guanyi Liao and Yuchen Xie contributed equally to this work.
Contributor Information
Jinjun Guo, Email: guojinjun1972@163.com.
Li Rong, Email: lirongcq@126.com.
References
- 1.Rajman L, Chwalek K, Sinclair DA. Therapeutic potential of NAD-boosting molecules: the in vivo evidence. Cell Metab. 2018;27(3):529–47. 10.1016/j.cmet.2018.02.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Covarrubias AJ, Perrone R, Grozio A, Verdin E. NAD+ metabolism and its roles in cellular processes during ageing. Nat Rev Mol Cell Biol. 2021;22(2):119–41. 10.1038/s41580-020-00313-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Imai S, Guarente L. NAD+ and sirtuins in aging and disease. Trends Cell Biol. 2014;24:464–71. 10.1016/j.tcb.2014.04.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Verdin E. NAD(+) in aging, metabolism, and neurodegeneration. Science. 2015;350:1208–13. 10.1126/science.aac4854 [DOI] [PubMed] [Google Scholar]
- 5.Canto C, Menzies KJ, Auwerx J. NAD(+) metabolism and the control of energy homeostasis: a balancing act between mitochondria and the nucleus. Cell Metab. 2015;22:31–53. 10.1016/j.cmet.2015.05.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Xie N, Zhang L, Gao W, Huang C, Huber PE, Zhou X, Li C, Shen G, Zou B. NAD+ metabolism: pathophysiologic mechanisms and therapeutic potential. Signal Transduct Target Ther. 2020;5(1):227. 10.1038/s41392-020-00311-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Amjad S, Nisar S, Bhat AA, Shah AR, Frenneaux MP, Fakhro K, Haris M, Reddy R, Patay Z, Baur J, Bagga P. Role of NAD+ in regulating cellular and metabolic signaling pathways. Mol Metab. 2021;49: 101195. 10.1016/j.molmet.2021.101195 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hong W, Mo F, Zhang Z, Huang M, Wei X. Nicotinamide mononucleotide: a promising molecule for therapy of diverse diseases by targeting NAD+ metabolism. Front Cell Dev Biol. 2020;28(8):246. 10.3389/fcell.2020.00246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mills KF, Yoshida S, Stein LR, Grozio A, Kubota S, Sasaki Y, Redpath P, Migaud ME, Apte RS, Uchida K, Yoshino J, Imai SI. Long-term administration of nicotinamide mononucleotide mitigates age-associated physiological decline in mice. Cell Metab. 2016;24(6):795–806. 10.1016/j.cmet.2016.09.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Poddar SK, Sifat AE, Haque S, Nahid NA, Chowdhury S, Mehedi I. Nicotinamide mononucleotide: exploration of diverse therapeutic applications of a potential molecule. Biomolecules. 2019;9(1):34. 10.3390/biom9010034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nadeeshani H, Li J, Ying T, Zhang B, Lu J. Nicotinamide mononucleotide (NMN) as an anti-aging health product-promises and safety concerns. J Adv Res. 2021;11(37):267–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yoshino J, Baur JA, Imai SI. NAD+ intermediates: the biology and therapeutic potential of NMN and NR. Cell Metab. 2018;27(3):513–28. 10.1016/j.cmet.2017.11.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Stein LR, Imai S. Specific ablation of Nampt in adult neural stem cells recapitulates their functional defects during aging. EMBO J. 2014;33:1321–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.de Picciotto NE, Gano LB, Johnson LC, Martens CR, Sindler AL, Mills KF, Imai S, Seals DR. Nicotinamide mononucleotide supplementation reverses vascular dysfunction and oxidative stress with aging in mice. Aging Cell. 2016;15:522–30. 10.1111/acel.12461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gomes AP, Price NL, Ling AJ, Moslehi JJ, Montgomery MK, Rajman L, White JP, Teodoro JS, Wrann CD, Hubbard BP, et al. Declining NAD(+) induces a pseudohypoxic state disrupting nuclear-mitochondrial communication during aging. Cell. 2013;155:1624–38. 10.1016/j.cell.2013.11.037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yamaguchi S, Franczyk MP, Chondronikola M, Qi N, Gunawardana SC, Stromsdorfer KL, Porter LC, Wozniak DF, Sasaki Y, Rensing N, Wong M, Piston DW, Klein S, Yoshino J. Adipose tissue NAD+ biosynthesis is required for regulating adaptive thermogenesis and whole-body energy homeostasis in mice. Proc Natl Acad Sci USA. 2019;116(47):23822–8. 10.1073/pnas.1909917116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stromsdorfer KL, Yamaguchi S, Yoon MJ, Moseley AC, Franczyk MP, Kelly SC, Qi N, Imai S, Yoshino J. NAMPT-mediated NAD(+) biosynthesis in adipocytes regulates adipose tissue function and multi-organ insulin sensitivity in mice. Cell Rep. 2016;16:1851–60. 10.1016/j.celrep.2016.07.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yoshino J, Mills KF, Yoon MJ, Imai S. Nicotinamide mononucleotide, a key NAD(+) intermediate, treats the pathophysiology of diet- and age-induced diabetes in mice. Cell Metab. 2011;14:528–36. 10.1016/j.cmet.2011.08.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Martin AS, Abraham DM, Hershberger KA, Bhatt DP, Mao L, Cui H, Liu J, Liu X, Muehlbauer MJ, Grimsrud PA, Locasale JW, Payne RM, Hirschey MD. Nicotinamide mononucleotide requires SIRT3 to improve cardiac function and bioenergetics in a Friedreich’s ataxia cardiomyopathy model. JCI Insight. 2017;2(14): e93885. 10.1172/jci.insight.93885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang R, Shen Y, Zhou L, Sangwung P, Fujioka H, Zhang L, Liao X. Short-term administration of nicotinamide mononucleotide preserves cardiac mitochondrial homeostasis and prevents heart failure. J Mol Cell Cardiol. 2017;112:64–73. 10.1016/j.yjmcc.2017.09.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Peek CB, Affinati AH, Ramsey KM, Kuo HY, Yu W, Sena LA, Ilkayeva O, Marcheva B, Kobayashi Y, Omura C, et al. Circadian clock NAD+ cycle drives mitochondrial oxidative metabolism in mice. Science. 2013;342:1243417. 10.1126/science.1243417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yasuda I, Hasegawa K, Sakamaki Y, Muraoka H, Kawaguchi T, Kusahana E, Ono T, Kanda T, Tokuyama H, Wakino S, Itoh H. Pre-emptive short-term nicotinamide mononucleotide treatment in a mouse model of diabetic nephropathy. J Am Soc Nephrol. 2021;32(6):1355–70. 10.1681/ASN.2020081188 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Guan Y, Wang SR, Huang XZ, Xie QH, Xu YY, Shang D, Hao CM. Nicotinamide mononucleotide, an NAD+ precursor, rescues age-associated susceptibility to AKI in a sirtuin 1-dependent manner. J Am Soc Nephrol. 2017;28(8):2337–52. 10.1681/ASN.2016040385 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lin JB, Kubota S, Ban N, Yoshida M, Santeford A, Sene A, Nakamura R, Zapata N, Kubota M, Tsubota K, et al. NAMPT-mediated NAD(+) biosynthesis is essential for vision in mice. Cell Rep. 2016;17:69–85. 10.1016/j.celrep.2016.08.073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chen X, Amorim JA, Moustafa GA, Lee JJ, Yu Z, Ishihara K, Iesato Y, Barbisan P, Ueta T, Togka KA, Lu L, Sinclair DA, Vavvas DG. Neuroprotective effects and mechanisms of action of nicotinamide mononucleotide (NMN) in a photoreceptor degenerative model of retinal detachment. Aging. 2020;12(24):24504–21. 10.18632/aging.202453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Caton PW, Kieswich J, Yaqoob MM, Holness MJ, Sugden MC. Nicotinamide mononucleotide protects against pro-inflammatory cytokine-mediated impairment of mouse islet function. Diabetologia. 2011;54(12):3083–92. 10.1007/s00125-011-2288-0 [DOI] [PubMed] [Google Scholar]
- 27.North BJ, Rosenberg MA, Jeganathan KB, Hafner AV, Michan S, Dai J, Baker DJ, Cen Y, Wu LE, Sauve AA, van Deursen JM, Rosenzweig A, Sinclair DA. SIRT2 induces the checkpoint kinase BubR1 to increase lifespan. EMBO J. 2014;33(13):1438–53. 10.15252/embj.201386907 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Uddin GM, Youngson NA, Chowdhury SS, Hagan C, Sinclair DA, Morris MJ. Administration of nicotinamide mononucleotide (NMN) reduces metabolic impairment in male mouse offspring from obese mothers. Cells. 2020;9(4):791. 10.3390/cells9040791 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Uddin GM, Youngson NA, Doyle BM, Sinclair DA, Morris MJ. Nicotinamide mononucleotide (NMN) supplementation ameliorates the impact of maternal obesity in mice: comparison with exercise. Sci Rep. 2017;7(1):15063. 10.1038/s41598-017-14866-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Das A, Huang GX, Bonkowski MS, Longchamp A, Li C, Schultz MB, Kim LJ, Osborne B, Joshi S, Lu Y, Treviño-Villarreal JH, Kang MJ, Hung TT, Lee B, Williams EO, Igarashi M, Mitchell JR, Wu LE, Turner N, Arany Z, Guarente L, Sinclair DA. Impairment of an endothelial NAD+-H2S signaling network is a reversible cause of vascular aging. Cell. 2018;173(1):74–89. 10.1016/j.cell.2018.02.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kiss T, Nyúl-Tóth Á, Balasubramanian P, Tarantini S, Ahire C, Yabluchanskiy A, Csipo T, Farkas E, Wren JD, Garman L, Csiszar A, Ungvari Z. Nicotinamide mononucleotide (NMN) supplementation promotes neurovascular rejuvenation in aged mice: transcriptional footprint of SIRT1 activation, mitochondrial protection, anti-inflammatory, and anti-apoptotic effects. Geroscience. 2020;42(2):527–46. 10.1007/s11357-020-00165-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tarantini S, Valcarcel-Ares MN, Toth P, Yabluchanskiy A, Tucsek Z, Kiss T, Hertelendy P, Kinter M, Ballabh P, Süle Z, Farkas E, Baur JA, Sinclair DA, Csiszar A, Ungvari Z. Nicotinamide mononucleotide (NMN) supplementation rescues cerebromicrovascular endothelial function and neurovascular coupling responses and improves cognitive function in aged mice. Redox Biol. 2019;24: 101192. 10.1016/j.redox.2019.101192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Karamanlidis G, Lee CF, Garcia-Menendez L, Kolwicz SC Jr, Suthammarak W, Gong G, Sedensky MM, Morgan PG, Wang W, Tian R. Mitochondrial complex I deficiency increases protein acetylation and accelerates heart failure. Cell Metab. 2013;18(2):239–50. 10.1016/j.cmet.2013.07.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nadtochiy SM, Wang YT, Nehrke K, Munger J, Brookes PS. Cardioprotection by nicotinamide mononucleotide (NMN): involvement of glycolysis and acidic pH. J Mol Cell Cardiol. 2018;121:155–62. 10.1016/j.yjmcc.2018.06.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wu K, Li B, Lin Q, Xu W, Zuo W, Li J, Liu N, Tu T, Zhang B, Xiao Y, Liu Q. Nicotinamide mononucleotide attenuates isoproterenol-induced cardiac fibrosis by regulating oxidative stress and Smad3 acetylation. Life Sci. 2021;1(274): 119299. 10.1016/j.lfs.2021.119299 [DOI] [PubMed] [Google Scholar]
- 36.Yoon MJ, Yoshida M, Johnson S, Takikawa A, Usui I, Tobe K, Nakagawa T, Yoshino J, Imai S. SIRT1-mediated eNAMPT secretion from adipose tissue regulates hypothalamic NAD+ and function in mice. Cell Metab. 2015;21(5):706–17. 10.1016/j.cmet.2015.04.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Park JH, Long A, Owens K, Kristian T. Nicotinamide mononucleotide inhibits post-ischemic NAD(+) degradation and dramatically ameliorates brain damage following global cerebral ischemia. Neurobiol Dis. 2016;95:102–10. 10.1016/j.nbd.2016.07.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wei CC, Kong YY, Li GQ, Guan YF, Wang P, Miao CY. Nicotinamide mononucleotide attenuates brain injury after intracerebral hemorrhage by activating Nrf2/HO-1 signaling pathway. Sci Rep. 2017;7(1):717. 10.1038/s41598-017-00851-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Klimova N, Fearnow A, Long A, Kristian T. NAD+ precursor modulates post-ischemic mitochondrial fragmentation and reactive oxygen species generation via SIRT3 dependent mechanisms. Exp Neurol. 2020;325: 113144. 10.1016/j.expneurol.2019.113144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Long AN, Owens K, Schlappal AE, Kristian T, Fishman PS, Schuh RA. Effect of nicotinamide mononucleotide on brain mitochondrial respiratory deficits in an Alzheimer’s disease-relevant murine model. BMC Neurol. 2015;1(15):19. 10.1186/s12883-015-0272-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Johnson S, Wozniak DF, Imai S. CA1 Nampt knockdown recapitulates hippocampal cognitive phenotypes in old mice which nicotinamide mononucleotide improves. NPJ Aging Mech Dis. 2018;8(4):10. 10.1038/s41514-018-0029-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhao C, Li W, Duan H, Li Z, Jia Y, Zhang S, Wang X, Zhou Q, Shi W. NAD+ precursors protect corneal endothelial cells from UVB-induced apoptosis. Am J Physiol Cell Physiol. 2020;318(4):C796–805. 10.1152/ajpcell.00445.2019 [DOI] [PubMed] [Google Scholar]
- 43.Lee D, Tomita Y, Miwa Y, Shinojima A, Ban N, Yamaguchi S, Nishioka K, Negishi K, Yoshino J, Kurihara T. Nicotinamide mononucleotide prevents retinal dysfunction in a mouse model of retinal ischemia/reperfusion injury. Int J Mol Sci. 2022;23(19):11228. 10.3390/ijms231911228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hasegawa K, Sakamaki Y, Tamaki M, Wakino S. Nicotinamide mononucleotide ameliorates adriamycin-induced renal damage by epigenetically suppressing the NMN/NAD consumers mediated by Twist2. Sci Rep. 2022;12(1):13712. 10.1038/s41598-022-18147-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cao T, Ni R, Ding W, Ji X, Fan GC, Zhang Z, Peng T. Nicotinamide mononucleotide as a therapeutic agent to alleviate multi-organ failure in sepsis. J Transl Med. 2023;21(1):883. 10.1186/s12967-023-04767-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Li HR, Liu Q, Zhu CL, Sun XY, Sun CY, Yu CM, Li P, Deng XM, Wang JF. β-Nicotinamide mononucleotide activates NAD+/SIRT1 pathway and attenuates inflammatory and oxidative responses in the hippocampus regions of septic mice. Redox Biol. 2023;63: 102745. 10.1016/j.redox.2023.102745 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bertoldo MJ, Listijono DR, Ho WJ, et al. NAD+ repletion rescues female fertility during reproductive aging. Cell Rep. 2020;30(6):1670–81. 10.1016/j.celrep.2020.01.058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Miao Y, Cui Z, Gao Q, Rui R, Xiong B. Nicotinamide mononucleotide supplementation reverses the declining quality of maternally aged oocytes. Cell Rep. 2020;32(5): 107987. 10.1016/j.celrep.2020.107987 [DOI] [PubMed] [Google Scholar]
- 49.Song J, Li J, Yang F, Ning G, Zhen L, Wu L, Zheng Y, Zhang Q, Lin D, Xie C, Peng L. Nicotinamide mononucleotide promotes osteogenesis and reduces adipogenesis by regulating mesenchymal stromal cells via the SIRT1 pathway in aged bone marrow. Cell Death Dis. 2019;10(5):336. 10.1038/s41419-019-1569-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sims CA, Guan Y, Mukherjee S, Singh K, Botolin P, Davila A Jr, Baur JA. Nicotinamide mononucleotide preserves mitochondrial function and increases survival in hemorrhagic shock. JCI Insight. 2018;3(17): e120182. 10.1172/jci.insight.120182 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wu K, Li B, Ma Y, Tu T, Lin Q, Zhu J, Zhou Y, Liu N, Liu Q. Nicotinamide mononucleotide attenuates HIF-1α activation and fibrosis in hypoxic adipose tissue via NAD+/SIRT1 axis. Front Endocrinol. 2023;26(14):1099134. 10.3389/fendo.2023.1099134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Mo Y, Yue M, Yim LY, Zhou R, Yu C, Peng Q, Zhou Y, Luk TY, Lui GC, Huang H, Lim CYH, Wang H, Liu L, Sun H, Wang J, Song Y, Chen Z. Nicotinamide mononucleotide impacts HIV-1 infection by modulating immune activation in T lymphocytes and humanized mice. EBioMedicine. 2023;98: 104877. 10.1016/j.ebiom.2023.104877 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Imai S, Armstrong CM, Kaeberlein M, Guarente L. Transcriptional silencing and longevity protein Sir2 is an NAD-dependent histone deacetylase. Nature. 2000;403:795–800. 10.1038/35001622 [DOI] [PubMed] [Google Scholar]
- 54.Irie J, Inagaki E, Fujita M, Nakaya H, Mitsuishi M, Yamaguchi S, Yamashita K, Shigaki S, Ono T, Yukioka H, Okano H, Nabeshima YI, Imai SI, Yasui M, Tsubota K, Itoh H. Effect of oral administration of nicotinamide mononucleotide on clinical parameters and nicotinamide metabolite levels in healthy Japanese men. Endocr J. 2020;67(2):153–60. 10.1507/endocrj.EJ19-0313 [DOI] [PubMed] [Google Scholar]
- 55.Yoshino M, Yoshino J, Kayser BD, Patti GJ, Franczyk MP, Mills KF, Sindelar M, Pietka T, Patterson BW, Imai SI, Klein S. Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science. 2021;372(6547):1224–9. 10.1126/science.abe9985 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Liao B, Zhao Y, Wang D, Zhang X, Hao X, Hu M. Nicotinamide mononucleotide supplementation enhances aerobic capacity in amateur runners: a randomized, double-blind study. J Int Soc Sports Nutr. 2021;18(1):54. 10.1186/s12970-021-00442-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kim M, Seol J, Sato T, Fukamizu Y, Sakurai T, Okura T. Effect of 12-week intake of nicotinamide mononucleotide on sleep quality, fatigue, and physical performance in older japanese adults: a randomized, double-blind placebo-controlled study. Nutrients. 2022;14(4):755. 10.3390/nu14040755 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Igarashi M, Nakagawa-Nagahama Y, Miura M, Kashiwabara K, Yaku K, Sawada M, Sekine R, Fukamizu Y, Sato T, Sakurai T, Sato J, Ino K, Kubota N, Nakagawa T, Kadowaki T, Yamauchi T. Chronic nicotinamide mononucleotide supplementation elevates blood nicotinamide adenine dinucleotide levels and alters muscle function in healthy older men. NPJ Aging. 2022;8(1):5. 10.1038/s41514-022-00084-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Okabe K, Yaku K, Uchida Y, Fukamizu Y, Sato T, Sakurai T, Tobe K, Nakagawa T. Oral administration of nicotinamide mononucleotide is safe and efficiently increases blood nicotinamide adenine dinucleotide levels in healthy subjects. Front Nutr. 2022;11(9): 868640. 10.3389/fnut.2022.868640 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Fukamizu Y, Uchida Y, Shigekawa A, Sato T, Kosaka H, Sakurai T. Safety evaluation of β-nicotinamide mononucleotide oral administration in healthy adult men and women. Sci Rep. 2022;12(1):14442. 10.1038/s41598-022-18272-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Yi L, Maier AB, Tao R, Lin Z, Vaidya A, Pendse S, Thasma S, Andhalkar N, Avhad G, Kumbhar V. The efficacy and safety of β-nicotinamide mononucleotide (NMN) supplementation in healthy middle-aged adults: a randomized, multicenter, double-blind, placebo-controlled, parallel-group, dose-dependent clinical trial. Geroscience. 2023;45(1):29–43. 10.1007/s11357-022-00705-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Akasaka H, Nakagami H, Sugimoto K, Yasunobe Y, Minami T, Fujimoto T, Yamamoto K, Hara C, Shiraki A, Nishida K, Asano K, Kanou M, Yamana K, Imai SI, Rakugi H. Effects of nicotinamide mononucleotide on older patients with diabetes and impaired physical performance: a prospective, placebo-controlled, double-blind study. Geriatr Gerontol Int. 2023;23(1):38–43. 10.1111/ggi.14513 [DOI] [PubMed] [Google Scholar]
- 63.Katayoshi T, Uehata S, Nakashima N, Nakajo T, Kitajima N, Kageyama M, Tsuji-Naito K. Nicotinamide adenine dinucleotide metabolism and arterial stiffness after long-term nicotinamide mononucleotide supplementation: a randomized, double-blind, placebo-controlled trial. Sci Rep. 2023;13(1):2786. 10.1038/s41598-023-29787-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Assiri MA, Ali HR, Marentette JO, Yun Y, Liu J, Hirschey MD, Saba LM, Harris PS, Fritz KS. Investigating RNA expression profiles altered by nicotinamide mononucleotide therapy in a chronic model of alcoholic liver disease. Hum Genomics. 2019;13(1):65. 10.1186/s40246-019-0251-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Zong Z, Liu J, Wang N, Yang C, Wang Q, Zhang W, Chen Y, Liu X, Deng H. Nicotinamide mononucleotide inhibits hepatic stellate cell activation to prevent liver fibrosis via promoting PGE2 degradation. Free Radic Biol Med. 2021;162:571–81. 10.1016/j.freeradbiomed.2020.11.014 [DOI] [PubMed] [Google Scholar]
- 66.Luo C, Ding W, Yang C, et al. Nicotinamide mononucleotide administration restores redox homeostasis via the Sirt3-Nrf2 axis and protects aged mice from oxidative stress-induced liver injury. J Proteome Res. 2022;21(7):1759–70. 10.1021/acs.jproteome.2c00167 [DOI] [PubMed] [Google Scholar]
- 67.Luo C, Ding W, Zhu S, Chen Y, Liu X, Deng H. Nicotinamide mononucleotide administration amends protein acetylome of aged mouse liver. Cells. 2022;11(10):1654. 10.3390/cells11101654 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Zhixing J, Ying C, Huiyuan Li, Degong J. Nicotinamide mononucleotide attenuates ischemia-reperfusion injury induced by donor liver from cardiac death through Sirt3. Organ Transplantat. 2022;13(5):618–25. [Google Scholar]
- 69.Lin X, Liping H, Gang L. Nicotinamide mononucleotide alleviates ethanol-induced insulin resistance. Chinese Pharmacolog Bulletin. 2022;38(11):1633–8. [Google Scholar]
- 70.Huang P, Wang X, Wang S, et al. Treatment of inflammatory bowel disease: potential effect of NMN on intestinal barrier and gut microbiota. Curr Res Food Sci. 2022;5(5):1403–11. 10.1016/j.crfs.2022.08.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Huang P, Jiang A, Wang X, et al. NMN maintains intestinal homeostasis by regulating the gut microbiota. Front Nutr. 2021;29(8): 714604. 10.3389/fnut.2021.714604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ru M, Wang W, Zhai Z, et al. Nicotinamide mononucleotide supplementation protects the intestinal function in aging mice and D-galactose induced senescent cells. Food Funct. 2022;13(14):7507–19. 10.1039/D2FO00525E [DOI] [PubMed] [Google Scholar]
- 73.Fang D, Xu T, Sun J, Shi J, Li F, Yin Y, Wang Z, Liu Y. Nicotinamide mononucleotide ameliorates sleep deprivation-induced gut microbiota dysbiosis and restores colonization resistance against intestinal infections. Adv Sci. 2023;10(9): e2207170. 10.1002/advs.202207170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zhao X, Ji K, Zhang M, Huang H, Wang F, Liu Y, Liu Q. NMN alleviates radiation-induced intestinal fibrosis by modulating gut microbiota. Int J Radiat Biol. 2023;99(5):823–34. 10.1080/09553002.2023.2145029 [DOI] [PubMed] [Google Scholar]
- 75.Liu LW, Xie Y, Li GQ, Zhang T, Sui YH, Zhao ZJ, Zhang YY, Yang WB, Geng XL, Xue DB, Chen H, Wang YW, Lu TQ, Shang LR, Li ZB, Li L, Sun B. Gut microbiota-derived nicotinamide mononucleotide alleviates acute pancreatitis by activating pancreatic SIRT3 signalling. Br J Pharmacol. 2023;180(5):647–66. 10.1111/bph.15980 [DOI] [PubMed] [Google Scholar]
- 76.Wang Y, Huang Y, Chase RC, Li T, Ramai D, Li S, Huang X, Antwi SO, Keaveny AP, Pang M. Global burden of digestive diseases: a systematic analysis of the global burden of diseases study, 1990 to 2019. Gastroenterology. 2023;165(3):773–83. 10.1053/j.gastro.2023.05.050 [DOI] [PubMed] [Google Scholar]
- 77.Grozio A, Mills KF, Yoshino J, Bruzzone S, Sociali G, Tokizane K, Lei HC, Cunningham R, Sasaki Y, Migaud ME, Imai SI. Slc12a8 is a nicotinamide mononucleotide transporter. Nat Metab. 2019;1(1):47–57. 10.1038/s42255-018-0009-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Di Stefano M, Nascimento-Ferreira I, Orsomando G, Mori V, Gilley J, Brown R, Janeckova L, Vargas ME, Worrell LA, Loreto A, Tickle J, Patrick J, Webster JR, Marangoni M, Carpi FM, Pucciarelli S, Rossi F, Meng W, Sagasti A, Ribchester RR, Magni G, Coleman MP, Conforti L. A rise in NAD precursor nicotinamide mononucleotide (NMN) after injury promotes axon degeneration. Cell Death Differ. 2015;22(5):731–42. 10.1038/cdd.2014.164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Di Stefano M, Loreto A, Orsomando G, Mori V, Zamporlini F, Hulse RP, Webster J, Donaldson LF, Gering M, Raffaelli N, Coleman MP, Gilley J, Conforti L. NMN deamidase delays wallerian degeneration and rescues axonal defects caused by NMNAT2 deficiency in vivo. Curr Biol. 2017;27(6):784–94. 10.1016/j.cub.2017.01.070 [DOI] [PubMed] [Google Scholar]
- 80.Li J, Bonkowski MS, Moniot S, Zhang D, Hubbard BP, Ling AJ, Rajman LA, Qin B, Lou Z, Gorbunova V, Aravind L, Steegborn C, Sinclair DA. A conserved NAD+ binding pocket that regulates protein-protein interactions during aging. Science. 2017;355(6331):1312–7. 10.1126/science.aad8242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Gujar AD, Le S, Mao DD, Dadey DY, Turski A, Sasaki Y, Aum D, Luo J, Dahiya S, Yuan L, Rich KM, Milbrandt J, Hallahan DE, Yano H, Tran DD, Kim AH. An NAD+-dependent transcriptional program governs self-renewal and radiation resistance in glioblastoma. Proc Natl Acad Sci USA. 2016;113(51):E8247–56. 10.1073/pnas.1610921114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Chalkiadaki A, Guarente L. The multifaceted functions of sirtuins in cancer. Nat Rev Cancer. 2015;15(10):608–24. 10.1038/nrc3985 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Date are available by emailing the corresponding author.