Abstract
Background:
Childhood cancer survivors treated with platinum-based chemotherapy are at risk of treatment-induced hearing loss. Accurate evaluation of hearing thresholds has historically been limited to clinical audiometry, which is logistically challenging and expensive to include in epidemiological studies. We evaluated the feasibility of using a remote, tablet-based hearing assessment in a cohort of pediatric germ cell tumor (GCT) survivors treated with platinum-based chemotherapy.
Methods:
Survivors from the GCT Outcomes and Late effects Data (GOLD) study were recruited to the pilot study (n=100). Study personnel conducted remote hearing assessments of standard and extended high frequency thresholds using validated tablet-based audiometry (SHOEBOX Inc). T-tests and Wilcoxon rank-sum tests evaluated differences in assessment characteristics between children and adults. Agreement between self-reported and measured hearing loss was calculated using Cohen’s kappa.
Results:
We were able to reach 136/168 (81%) eligible participants, of which 100 (74%) agreed to participate. Successful completion of the remote hearing assessment was high (97%; 20 children [ages 7–17], 77 adults [ages 18–31]). Mean assessment length was 37.6 minutes and mean turnaround time was 8.3 days. We observed hearing loss at standard frequencies in 21% of participants. Agreement between self-reported and measured hearing loss was significant (p-value = 1.41 × 10−7), with 83.5% concordance.
Conclusions:
Hearing loss measured using the remote assessment aligns with self-reporting and rates of hearing loss reported in the literature for this population.
Impact:
Remote application of tablet-based audiometry is a feasible and efficacious method for measuring hearing in epidemiologic studies with participants spread across large geographical areas.
Introduction
Recent innovations in portable digital platforms used to collect health data have created opportunities to advance epidemiological studies by providing measurement of biophysical characteristics outside the clinical setting.(1) Direct measurement of health data provides key information that can be used to identify risk factors, uncover windows for intervention, and improve follow-up care.(2) The integration of remote data collection methods with these portable digital technologies can increase study participation while reducing costs to researchers and participants.(3,4) In this context, we describe the remote field application of a digital technology to assess hearing function within childhood cancer survivors, illustrating the potential of remote technologies to broaden data collection in this critical area.
Platinum-based chemotherapy-induced hearing loss, known as ototoxicity, can range from subtle deficits that make it difficult to hear in noisy environments to significant hearing loss that require hearing aids and other hearing assistive technology. The impact of ototoxicity on language acquisition, educational attainment, and overall quality of life has been well documented in survivors of pediatric cancers.(5,6) Most population-based follow-up studies that have assessed hearing loss in childhood cancer survivors have used self-reported hearing loss as the outcome measure and have typically focused on clinically significant hearing loss that would require the use of a hearing aid.(7–12) Measuring hearing directly rather than via self-report offers a more detailed picture of hearing across the acoustic range, especially if the assessment includes the upper frequencies.(13) However, clinical audiometry is logistically challenging and cost prohibitive in large epidemiologic studies, particularly if the participants live in a large geographic area.(14,15) Portable, digital health collection technologies are one potential solution for this problem. Once such example is SHOEBOX® Audiometry, which is a clinically validated and portable automated iPad-based audiometer from SHOEBOX Ltd. (Ottawa, Ontario, Canada). The software has been validated against conventional audiology in children, adolescents, and adults, as well as patients with cognitive impairment and dementia, across a range of settings including clinical and noisy environments, and in various study designs including single visits and longitudinal studies.(16–22)
In our study, we evaluated the efficacy and feasibility of remote audiometry using the SHOEBOX digital application in a cohort of pediatric germ cell tumor (GCT) survivors who were treated with platinum-based chemotherapy, evaluating hearing levels in the standard and extended high frequency (EHF) range. Here, we outline the remote application of this technology and present preliminary findings in a sample of 100 study participants.
Materials and Methods
Study Objectives
The primary objective of this study was to determine the feasibility of implementing remote hearing assessments using the SHOEBOX Audiometry Standard Edition mobile application among survivors of pediatric GCTs. For the purpose of this study, feasibility was defined as the practicality and acceptability of conducting remote hearing assessments using this technology in a population of GCT survivors.
The secondary objective was to evaluate the efficacy of the data from the assessments to gather preliminary estimates of hearing loss. Efficacy was defined as the ability the SHOEBOX Audiometry application to detecting hearing loss across standard and extended high frequencies, as well as across different age groups within the study population, against self-reported hearing loss and prevalence rates reported in the literature for this population.
The GCT Outcomes and Late effects Data (GOLD) study
The GOLD study is a new cohort of GCT survivors recruited from the Children’s Oncology Group registry protocols.(23,24) The GOLD study is aimed at establishing a survivorship cohort for pediatric and adolescent GCTs to assess short term and long term adverse events associated with GCT treatment. Participants were eligible for the study if they previously participated in the germ cell tumor epidemiology study(25) (GaMETES) and provided consent to be contacted for future studies (n=823). We recently conducted a cross-sectional pilot study within this cohort to assess the feasibility of implementing remote hearing assessments using SHOEBOX Audiometry. Participants were eligible for the hearing assessment pilot if they consented to the GOLD study, completed an online questionnaire, and reported chemotherapy treatment for their GCT (Figure 1). Participants were excluded if they were deceased at last known survival status, resided outside the United States, or were not contacted prior to the cutoff date (July 13, 2022) for the pilot study.
Figure 1. CONSORT diagram of study population.
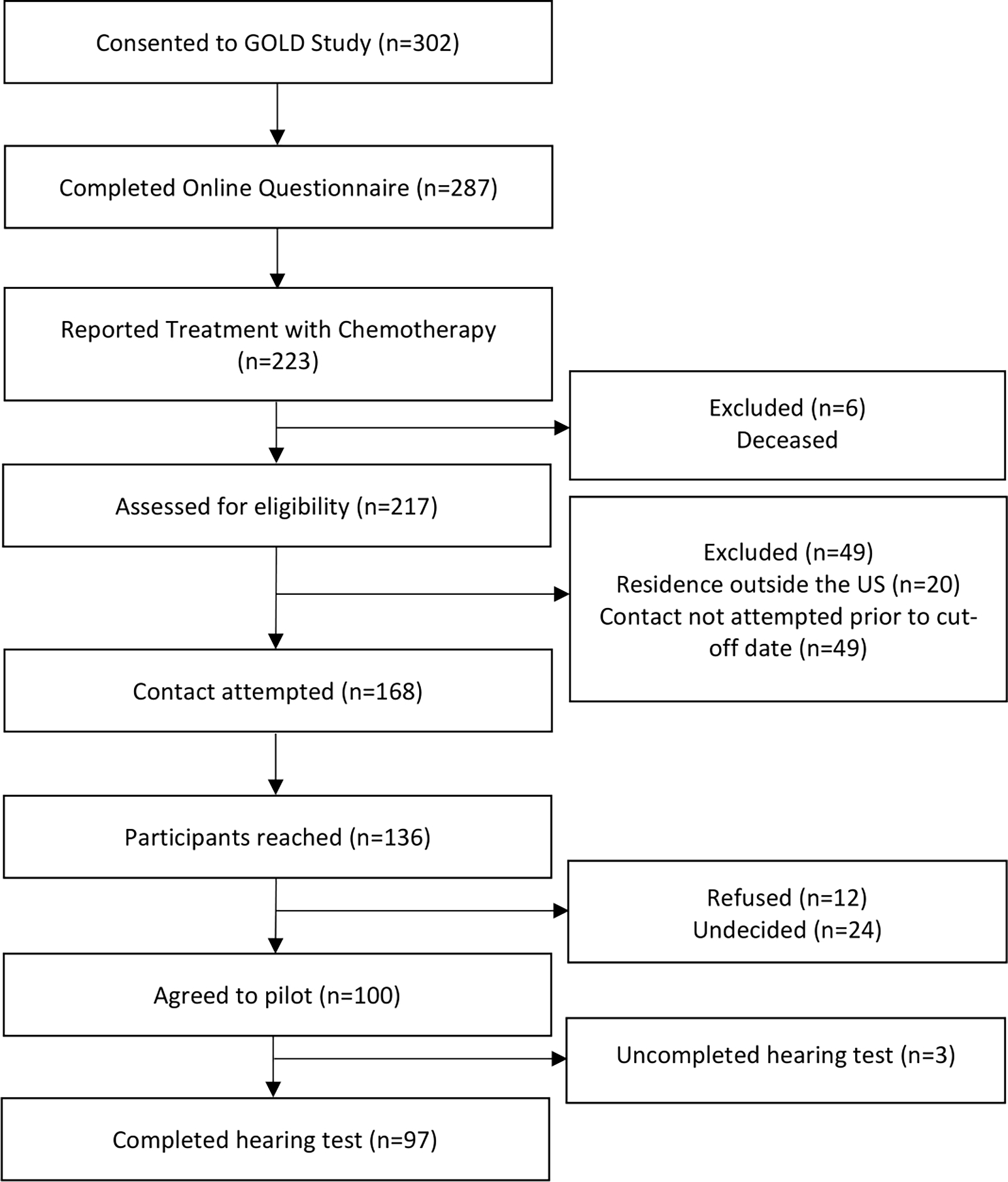
Flow chart depicting study participant inclusion and enrollment at the cut-off date (July 13, 2022).
This study was approved by the Institutional Review Board at the University of Minnesota. Participants aged 18 years and older provided written informed consent for participation in the study. Parents provided informed consent for participants under the age of 18 years. Assent was obtained from children aged 8 – 17 years. This study was performed in accordance with the ethical principles of the Belmont Report and the US Common Rule.
Hearing Assessments
Participants or parents/guardians of participants were contacted via phone to determine whether they were interested in participating in the hearing pilot study. If the participant agreed to take part in the remote hearing assessment, study materials were shipped to participants via FedEx, with a prepaid return label. Equipment required to complete the assessment includes an Apple iPad, DD50 transducers (RadioEar), charger, stand, tape strips, and consent/assent documents. All hearing assessments were conducted at the participant’s home, dorm, or chosen location and guided over the phone by trained study staff. Internet access was not necessary for taking the assessment, all data is stored offline.
Hearing assessments were performed using the SHOEBOX Audiometry Standard Edition mobile application and the Extended High Frequency license, following the manufacturer’s instructions (available from: https://help.shoebox.md/Content/Home.htm).(26) The SHOEBOX Audiometry application allows for the collection of validated subclinical hearing level thresholds at standard frequencies (250–8,000 Hz) and extended high frequencies (10,000–16,000 Hz). We used the “Automatic Pure Tone Test” task to evaluate hearing at ten frequencies: 500, 1,000, 2,000, 3,000, 4,000, 6,000, 8,000, 12,500, 14,000 and 16,000 Hz. In addition to evaluating hearing thresholds for both ears, the SHOEBOX Audiometry application is equipped to detect differences in hearing between the ears, allowing for the identification of unilateral hearing loss. This capability enables comprehensive assessment of hearing function, including the detection of asymmetrical hearing impairments.
Participants were instructed to wear the DD50 transducers (RadioEar) over their ears completely, ensuring that the correct side was positioned over each ear, before starting the assessment. Study staff provided verbal guidance over the phone for the entirety of the assessment process. The assessment is a gamified two-choice drag and drop task where participants are asked to drag an icon to a specified location based on their ability to hear a tone. Participants under 12 years of age were administered the “child” test and participants aged 12 and up were administered the “adult” test. The difference in test type reflects the icons of the interface the participant interacts with (a bone and two dogs for the child test, different colored circles for the adult test). Upon completion of the assessment, participants were given the option of a drop off at a FedEx retail location or pickup from their home by a courier scheduled by study staff. Reminders for drop off were sent through the participants preferred contact method. Once the materials were received by study staff, the iPad was connected to the internet and the results were automatically backed up to the cloud. Audiogram results, along with a built-in interpretation by SHOEBOX, were returned to participants via mail.
Audiogram Review
To categorize hearing loss in the standard frequency (SF) range, we calculated a high frequency pure tone average (PTA-HF) for 3000, 4000, 6000, and 8000 Hz with a >20 dB hearing loss (HL) threshold for all ages. In the EHF range, we calculated an EHF pure tone average (PTA-EHF) for 10,000, 12,500, and 16,000 Hz with >20 dB HL threshold for those 25 years of age and under, and >25 dB HL for those over 25 years of age, in line with previous literature.(27,28) Audiograms were considered unevaluable if PTAs were unable to be calculated due to missing data.
Bilateral hearing loss was determined if the PTA exceeded the dB HL threshold for both ears separately. Unilateral hearing loss was determined if the PTA exceeded the dB HL for only one ear. These categories–unilateral and bilateral hearing loss–were mutually exclusive within each PTA frequency grouping. Any hearing loss included individuals with either unilateral or bilateral hearing loss.
Questionnaire
As part of the GOLD study, participants or their parents (depending on age at enrollment and guardianship status if over 18 years of age) completed a self-administered electronic questionnaire via a secure link to a REDCap survey. For participants under 18 years old, parents or guardians completed the survey on their behalf. For adults under guardianship, their guardians also completed the survey. The questionnaire, adapted from materials developed for the Childhood Cancer Survivorship Study(29–31), covered various aspects of health history and behaviors, including medical care, medication use, quality of life, and hearing/speech.
Questions addressing hearing included “Have you ever been told by a doctor or health care professional that you/your child have...” hearing loss not requiring a hearing aid, hearing loss requiring a hearing aid, and deafness in one or both ears not completely corrected by a hearing aid. For analysis purposes, self-reported hearing loss was considered if the response to any of these questions was yes.
Data Analysis
Categorical data were summarized using counts and percentages, while continuous data was summarized using means, standard deviations, medians, and ranges. T-tests for the mean and Wilcoxon rank-sum tests for the median evaluated differences in duration, turnaround time, and time from self-reported to assessed hearing between children and adults. Agreement between self-reported and measured hearing loss was calculated using Cohen’s kappa. The threshold for statistical significance was set at α = 0.05. The analysis was conducted using RStudio version 2022.07.1.
Data Availability
The data that support the findings of in this study are not publicly available due to participant privacy but are available on request from the corresponding author, JNP.
Results
Contact was attempted for 168 participants meeting inclusion criteria prior to the cutoff date for the pilot study, of which 136 participants were reached over the phone (81% contact rate). Of the contacted participants, 100 agreed to participate in the pilot study (74%) and were sent study materials for hearing assessments. The characteristics of the study population are summarized in Table 1. Most participants were adults (77%) at the time of hearing assessments. A majority of participants were male (62%) and had an intracranial tumor location (45%). Hearing assessments were carried out in 35 states across the continental US (Figure 2).
Table 1.
Demographic characteristics of participants in the hearing pilot study.
| Age group (years) |
|||||
|---|---|---|---|---|---|
| Characteristics | Overall | 7–12 | 13–17 | 18–25 | 26–35 |
|
| |||||
| Participants, n | 100 | 8 | 15 | 49 | 28 |
| Age at diagnosis, median (range) | 14 (0–19) | 1 (0–4) | 6 (2–11) | 14 (7–19) | 17 (14–19) |
| Female, n (%) | 38 (38) | 5 (63) | 9 (60) | 20 (41) | 4 (14) |
| Region, n (%) | |||||
| Northa | 38 (38) | 3 (38) | 5 (33) | 16 (33) | 14 (50) |
| Southb | 29 (29) | 3 (38) | 5 (33) | 17 (35) | 4 (14) |
| Eastc | 15 (15) | 1 (13) | 1 (7) | 5 (10) | 8 (29) |
| Westd | 18 (18) | 1 (13) | 4 (27) | 11 (22) | 2 7) |
| Tumor location, n (%) | |||||
| Intracranial | 45 (45) | 1 (12) | 7 47) | 26 (53) | 11 (39) |
| Ovary | 19 (19) | 0 (0) | 2 (13) | 13 (27) | 4 (14) |
| Testis | 19 (19) | 0 (0) | 1 (7) | 5 (10) | 13 (46) |
| Extragonadal | 17 (17) | 7 (88) | 5 (33) | 5 (10) | 0 (0) |
ND, NE, MN, IA, MO, WI, IL, IN, MI, OH
TX, LA, MS, KY, VA, NC, GA, FL, WV
VT, MA, RI, NY, NJ, PA, MD
WA, OR, CA, ID, UT, CO, AZ
Figure 2. Map of states where hearing assessments were completed.
States where at least one assessment was completed are highlighted maroon.
Participants had a median age at diagnosis of 14 years (range = 0,19), while the median age at completion of the hearing assessment was 24 years (range = 7,31). The response rate was similar between child and adult participants (overall: 74%), as well as the median calls per enrollment (overall median = 2; range = 1,6) (Table 2).
Table 2.
Recruitment and hearing assessment characteristics.
| Age group (years) |
||||
|---|---|---|---|---|
| Characteristics | Overall | Children (age <18) | Adults (age ≥ 18) | p-value |
|
| ||||
| Participants contacted, n | 168 | 38 | 130 | |
| Participants reached, n (%) | 136 (81) | 33 (87) | 103 (79) | |
| Agreed to participate, n (%a) | 100 (74) | 23 (70) | 77 (75) | |
| Calls per enrollmentb, median (range) | 2 (1–6) | 2 (1–4) | 2 (1–6) | |
| Completed assessment, n (%c) | 97 (97) | 20 (87) | 77 (100) | |
| Equipment returned, n (%c) | 100 (100) | 23 (100) | 77 (100) | |
| Assessment duration, meand (s.d.) | 37.6 (10.4) | 38.9 (11.3) | 37.3 (10.1) | 0.56 |
| Assessment duration, mediand (range) | 35 (17–83) | 35 (25–70) | 35 (17–83) | 0.72 |
| Turnaround time, meane (s.d.) | 8.3 (5.7) | 6.2 (1.7) | 8.8 (6.3) | 0.001 |
| Turnaround time, mediane (range) | 6 (3–45) | 6 (4–10) | 7 (3–45) | 0.10 |
| Time from self-reported to assessed hearing (months), meanf (s.d.) | 17.4 (5.6) | 14.4 (5.8) | 18.2 (5.3) | 0.006 |
| Time from self-reported to assessed hearing (months), medianf (range) | 18 (2–29) | 16 (2–22) | 19 (4–29) | 0.003 |
Percentage of those reached
For those with available data
Percentage of those who agreed to participate
Minutes
Days
Months
Hearing assessments
Of the 100 participants who attempted hearing assessments, there were 23 children (age 7–17 years) and 77 adults (age 18–31 years). A total of 87% of children and 100% of adults successfully completed the assessment. All participants returned the study equipment regardless of completion status (100%). The overall median duration of a hearing assessment was 35 minutes, ranging from 17 to 83 minutes. No statistically significant difference in duration was observed between children and adults (mean p-value = 0.56; median p-value = 0.72). The turnaround time, calculated as days between sending materials and receiving them back, ranged from 3 to 45 days. The mean turnaround time was shorter among children as compared with adults (mean <18: 6.2; mean 18+: 8.8; p-value = 0.001). The time from self-reported to assessed hearing, calculated as months between completing the questionnaire and taking the remote hearing assessment, ranged from 2 to 29 months. The mean time from self-reported to assessed hearing was shorter among children than adults (mean<18: 14.4; mean 18+: 18.2; p-value = 0.006).
The prevalence of measured hearing loss varied between SFs and EHFs, as well as among different age groups within our study cohort (Table 3). Overall, 21% of participants exhibited measured hearing loss at SFs, while at EHFs, the prevalence increased to 48%. The proportion of participants with any hearing loss was higher among children as compared to adults at both frequency ranges (Table 3). Any assessed hearing loss at SFs was similar between intracranial and other tumor locations (intracranial: 19%; other: 22%). At EHFs, those with other tumor locations had a higher prevalence of any hearing loss as compared to those with an intracranial tumor (intracranial: 30%; other: 63%). Self-reported hearing loss was present in 25% of participants, and, like measured hearing loss; self-reported hearing loss was higher among children (45%) as compared to adults (19%). Similar to the EHF hearing loss, self-reported hearing loss was observed in almost twice as many participants with other tumor locations as compared to intracranial tumors (intracranial: 16%; other: 31%)
Table 3.
Assessed and self-reported hearing loss among participants.
| Age group | Tumor location | ||||
|---|---|---|---|---|---|
| Characteristics | Overall (n = 97) | Children (n = 20) | Adults (n = 77) | Intracranial (n = 43) | Other (n = 54) |
|
| |||||
| Hearing lossa, n (%) | |||||
| Any hearing loss | 20 (21) | 9 (45) | 11 (14) | 8 (19) | 12 (22) |
| Unilateral hearing loss | 7 (7) | 2 (10) | 5 (6) | 4 (9) | 3 (6) |
| Bilateral hearing loss | 13 (13) | 7 (35) | 6 (8) | 4 (9) | 9 (17) |
| EHF hearing lossb, n (%) | |||||
| Any hearing loss | 47 (48) | 15 (75) | 32 (42) | 13 (30) | 34 (63) |
| Unilateral hearing loss | 14 (14) | 3 (15) | 11 (14) | 6 (14) | 8 (15) |
| Bilateral hearing loss | 33 (34) | 12 (60) | 21 (27) | 7 (16) | 26 (48) |
| Self-reported hearing loss, n (%) | |||||
| Any hearing loss | 24 (25) | 9 (45) | 15 (19) | 7 (16) | 17 (31) |
>20 dB HL pure tone average at 3000, 4000, 6000, & 8000 Hz for all ages
>20 dB HL pure tone average at 10,000, 12,500, & 16,000 Hz for those aged 25 years and under, >25 dB HL for aged 25+ years
The agreement between self-reported and measured hearing loss determinations was statistically significant (p-value = 1.41 × 10−7), with 83.5% of cases in concordance (Table 4). A Cohen’s kappa of 0.531 suggests a moderate level of agreement between the two test types. Some participants exhibited assessed hearing loss without self-reporting it (n=6), while others reported hearing loss despite not showing it on assessment (n=10).
Table 4.
Agreement between self-reported and assessed hearing loss.
| No (Self-reported) | Yes (Self-reported) | Total | |
|---|---|---|---|
| No (Assessed) | 67 | 10 | 77 |
| Yes (Assessed) | 6 | 14 | 20 |
| Total | 73 | 24 | 97 |
| Agreement | Kappa | z-score | p-value |
|---|---|---|---|
| 83.5% | 0.531 | 5.26 | 1.41 × 10−7 |
Discussion
The purpose of this study was to test the field application of a tablet-based hearing assessment in a study of pediatric GCT survivors treated with platinum-based chemotherapy. Our primary objective was to assess the feasibility of remote audiometry in this population and gauge the willingness of participants to complete the hearing assessment. There was a high level of interest in participating in the assessment among the participants we reached by phone, with a 74% participation rate before the cutoff date among individuals successfully contacted. The remote assessment was also feasible, with a 97% successful completion rate among individuals who attempted the test. Assessments were conducted in 35 states across the U.S., involving both adults and children, and all study and 100% of participants returned the study materials. The results described here provide evidence that remote application of digital technologies for the collection of biophysical data is feasible in epidemiologic studies with geographically diverse participants.
Clinical audiometry, the gold standard for hearing assessment, requires highly calibrated equipment and exceptionally quiet environments, typically achieved with diagnostic audiometers and sound attenuation chambers. (32–34) This makes it non-portable, expensive, and impractical for studies whose participants reside in large geographic areas, particularly in rural and low-income regions with limited access to audiology clinics.(14,15,35) Given the diverse participant distribution in the GOLD study, spanning all 50 U.S. states and Canada, utilizing clinical audiometry was not feasible. This led us to pursue alternative methods for hearing assessment, aiming to generate quantifiable results and including the capability to evaluate hearing in the EHF range.
SHOEBOX® Audiometry has been clinically validated by peer-reviewed research and has been shown to produce accurate sub-clinical hearing thresholds outside of a sound booth, including in relatively noisy environments.(16–22) However, this is the first study to describe a fully remote application where the tools used for the assessment were mailed to study participants and hearing assessments were conducted via phone call. Remote fielding of the application extends the benefits of the technology by not requiring the physical presence of study personnel, reducing travel costs, adding flexibility, and further lowering barriers to study participation. The high completion rate of hearing assessments in the current study may be attributable to the accessibility for participants, as there were no costs and no need to travel. Crucially, all hearing kits were returned by participants, bolstering the applicability of this method epidemiology studies.
The SHOEBOX application contains automated quality-control measures which automatically performed masking in situations of asymmetric hearing loss, flagged frequencies with questionable results, and repeated the process multiple times at different frequencies and intensities to ensure reproducibility of measurements without any manual intervention.(26) Study personnel required minimal training to be able to administer hearing assessments. Its mobile nature and minimized need for advanced training overcomes many of the barriers of clinical audiometry while reserving the accuracy needed for high-quality studies. While comparing SHOEBOX and clinical audiometry was outside the scope of this pilot study, we did find a moderate level of agreement between self-reported hearing loss and assessed hearing loss.
We observed hearing loss in the SF range, affecting one or both ears, in 21% of the study population. This aligns with reported prevalence rates in the literature for this population, though variations are noted based on the type and dosage of chemotherapy administered.(12,36–39) In the EHF range, we observed hearing loss in 48% participants overall. The higher prevalence of hearing loss in the EHF range, compared to the SF range, is consistent with findings from other studies.(40,41) However, studies focused on ototoxicity during pediatric treatment have reported even higher prevalence rates (86%–94%).(40,41) Variables that could account for the difference in ototoxicity prevalence in the EHF range for this cohort compared to other studies include the cumulative platinum exposure and dose intensity, and older age at diagnosis.(36,37) However, it is important to highlight that our investigation focused on long-term measurable hearing outcomes in a large a cohort of GCT survivors, an aspect frequently overlooked in the existing literature, which predominantly focuses on ototoxicity during pediatric treatment. We also observed a higher prevalence of assessed hearing loss in the EHF range for other tumor locations as compared to intracranial tumors, which may be attributable to higher cumulative dosage and a more prolonged exposure to platinum-based chemotherapy, as opposed to the more acute and intense exposure typically associated with intracranial tumors.(42–45)
Based on previous validation studies(16–18,22), SHOEBOX Audiometry has demonstrated high sensitivity and specificity in detecting hearing loss, with values ranging from 87.5% to 100% for sensitivity and from 89% to 100% for specificity. These findings suggest that SHOEBOX Audiometry can be considered reliable in this study as a measure of hearing loss. We found a moderate level of agreement between self-reported hearing loss and assessed hearing loss, with 83.5% of cases showing concordance. Interestingly, six participants exhibited assessed hearing loss without self-reporting it, while 10 participants reported hearing loss despite not showing it on assessment. This may have been due to factors such as the time gap between self-reported and assessed hearing or threshold values close to the cutoff for hearing loss determination. This highlights the importance of objective measures in assessing hearing function, as discrepancies between self-report and objective assessment may have implications for understanding the true prevalence and impact of hearing loss in this population.
While there are many strengths associated with the use of SHOEBOX audiometry for remote hearing assessments, there are also limitations, which must be considered. One limitation of the remote application as implemented in this study is its reliance on shipping through commercial mail and subsequent return of materials by study participants, which is susceptible to delays and potential loss of materials. In our pilot study, we had 5 hearing assessment kits with delayed return when participants did not drop off within the given timeline. However, with follow-up by study staff members, we were able to recover all equipment and no data or materials were lost.
SHOEBOX Audiometry Standard is not designed to capture information on individuals with profound hearing loss (HL >100 dB), which may limit its application in settings where more severe hearing loss is highly prevalent.(26) While our study included participants as young as 7 years old, it is noteworthy that the recommended youngest age cutoff for SHOEBOX audiometry is 6 years of age. Although the application has been tested in patients younger than 6 years old, the effectiveness and reliability in this age group warrant further investigation. Beyond age, it is also important to consider developmental factors when using SHOEBOX audiometry. Children with developmental delays or cognitive impairments may struggle with following instructions or comprehending the testing procedures, potentially affecting the ability to complete the test. Those with accessibility needs may have difficulty, although if they are able to communicate whether they hear a tone or not, assisted mode can be used whereby a parent or guardian can select tone responses. Medical co-morbidities causing fluctuations in hearing levels or temporary hearing loss may influence the hearing results or the ability to complete the test, although the software has not yet been tested in these populations. Children with sensory processing disorders or auditory neuropathy may require more tailored assessment approaches, as standard audiometry may not fully capture their hearing profiles.(46–48) Researchers should consider these practical limitations when considering the applicability of SHOEBOX audiometry in their research.
Overall, the results of this study support the feasibility of using the remote application of digital technology to conduct hearing assessments in epidemiologic studies. The method is efficacious in producing preliminary estimates of hearing loss were in line with self-report and rates reported in the literature for this population. In future analyses in the GOLD study, we will extend this work by increasing the number of study participants and evaluating progression of hearing loss over time. These methods could be applied to other study populations, specifically within childhood cancer or other pediatric conditions. The framework outlined in this pilot study could be applied to the remote collection of other app-based technologies and be applied in other populations. In conclusion, the high participant engagement, logistical advantages, and accurate measurements achieved with SHOEBOX® Audiometry observed in our study contribute to the growing body of evidence supporting the practicality and reliability of remote methods in epidemiological research. The outlined methodology and its strengths, limitations, and future directions offer valuable insights for researchers aiming to employ similar approaches in diverse study populations.
Acknowledgements
Funding
This work was supported Alex’s Lemonade Stand Foundation (POST to PSM), the Children’s Cancer Research Fund, Minneapolis, MN and a Department of Defense Behavioral Health Science Award (W81XWH2210184 to JNP), and National Institutes of Health Grants to the Children’s Oncology Group (U10CA180899 and U10CA180886).
Roles of funders:
The funder did not play a role in the design of the study; the collection, analysis, and interpretation of the data; the writing of the manuscript; and the decision to submit the manuscript for publication.
Footnotes
Conflict of interest: The authors declare no potential conflicts of interest.
References
- 1.Thyagarajan B, Nelson HH, Poynter JN, Prizment AE, Roesler MA, Cassidy E, et al. Field Application of Digital Technologies for Health Assessment in the 10,000 Families Study. Cancer Epidemiol Biomark Prev Publ Am Assoc Cancer Res Cosponsored Am Soc Prev Oncol. 2020. Apr;29(4):744–51. [DOI] [PubMed] [Google Scholar]
- 2.Soucie JM. Public health surveillance and data collection: general principles and impact on hemophilia care. Hematol Amst Neth. 2012. Apr;17 Suppl 1(0 1):S144–146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Senbekov M, Saliev T, Bukeyeva Z, Almabayeva A, Zhanaliyeva M, Aitenova N, et al. The Recent Progress and Applications of Digital Technologies in Healthcare: A Review. Int J Telemed Appl. 2020;2020:8830200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gentili A, Failla G, Melnyk A, Puleo V, Tanna GLD, Ricciardi W, et al. The cost-effectiveness of digital health interventions: A systematic review of the literature. Front Public Health. 2022;10:787135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Knight KRG, Kraemer DF, Neuwelt EA. Ototoxicity in children receiving platinum chemotherapy: underestimating a commonly occurring toxicity that may influence academic and social development. J Clin Oncol Off J Am Soc Clin Oncol. 2005. Dec 1;23(34):8588–96. [DOI] [PubMed] [Google Scholar]
- 6.Grewal S, Merchant T, Reymond R, McInerney M, Hodge C, Shearer P. Auditory late effects of childhood cancer therapy: a report from the Children’s Oncology Group. Pediatrics. 2010. Apr;125(4):e938–950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Trendowski MR, Baedke JL, Sapkota Y, Travis LB, Zhang X, El Charif O, et al. Clinical and genetic risk factors for radiation-associated ototoxicity: A report from the Childhood Cancer Survivor Study and the St. Jude Lifetime Cohort. Cancer. 2021. Nov 1;127(21):4091–102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Weiss A, Sommer G, Kasteler R, Scheinemann K, Grotzer M, Kompis M, et al. Long-term auditory complications after childhood cancer: A report from the Swiss Childhood Cancer Survivor Study. Pediatr Blood Cancer. 2017. Feb;64(2):364–73. [DOI] [PubMed] [Google Scholar]
- 9.Weiss A, Sommer G, Schindera C, Wengenroth L, Karow A, Diezi M, et al. Hearing loss and quality of life in survivors of paediatric CNS tumours and other cancers. Qual Life Res Int J Qual Life Asp Treat Care Rehabil. 2019. Feb;28(2):515–21. [DOI] [PubMed] [Google Scholar]
- 10.Whelan K, Stratton K, Kawashima T, Leisenring W, Hayashi S, Waterbor J, et al. Auditory complications in childhood cancer survivors: a report from the childhood cancer survivor study. Pediatr Blood Cancer. 2011. Jul 15;57(1):126–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Brinkman TM, Bass JK, Li Z, Ness KK, Gajjar A, Pappo AS, et al. Treatment-induced hearing loss and adult social outcomes in survivors of childhood CNS and non-CNS solid tumors: Results from the St. Jude Lifetime Cohort Study. Cancer. 2015. Nov 15;121(22):4053–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bass JK, Liu W, Banerjee P, Brinkman TM, Mulrooney DA, Gajjar A, et al. Association of Hearing Impairment With Neurocognition in Survivors of Childhood Cancer. JAMA Oncol. 2020. Sep 1;6(9):1363–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tsimpida D, Kontopantelis E, Ashcroft D, Panagioti M. Comparison of Self-reported Measures of Hearing With an Objective Audiometric Measure in Adults in the English Longitudinal Study of Ageing. JAMA Netw Open. 2020. Aug 3;3(8):e2015009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Williams CP, Geiger AM, Norton WE, de Moor JS, Everson NS. Influence of Cost-Related Considerations on Clinical Trial Participation: Results from the 2020 Health Information National Trends Survey (HINTS). J Gen Intern Med. 2023. Apr;38(5):1200–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Legge F, Eaton D, Molife R, Ferrandina G, Judson I, de Bono J, et al. Participation of patients with gynecological cancer in phase I clinical trials: two years experience in a major cancer center. Gynecol Oncol. 2007. Mar;104(3):551–6. [DOI] [PubMed] [Google Scholar]
- 16.Thompson GP, Sladen DP, Borst BJH, Still OL. Accuracy of a Tablet Audiometer for Measuring Behavioral Hearing Thresholds in a Clinical Population. Otolaryngol--Head Neck Surg Off J Am Acad Otolaryngol-Head Neck Surg. 2015. Nov;153(5):838–42. [DOI] [PubMed] [Google Scholar]
- 17.Saliba J, Al-Reefi M, Carriere JS, Verma N, Provencal C, Rappaport JM. Accuracy of Mobile-Based Audiometry in the Evaluation of Hearing Loss in Quiet and Noisy Environments. Otolaryngol--Head Neck Surg Off J Am Acad Otolaryngol-Head Neck Surg. 2017. Apr;156(4):706–11. [DOI] [PubMed] [Google Scholar]
- 18.Bastianelli M, Mark AE, McAfee A, Schramm D, Lefrançois R, Bromwich M. Adult validation of a self-administered tablet audiometer. J Otolaryngol - Head Neck Surg J Oto-Rhino-Laryngol Chir Cervico-Faciale. 2019. Nov 7;48(1):59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yeung J, Javidnia H, Heley S, Beauregard Y, Champagne S, Bromwich M. The new age of play audiometry: prospective validation testing of an iPad-based play audiometer. J Otolaryngol - Head Neck Surg J Oto-Rhino-Laryngol Chir Cervico-Faciale. 2013. Mar 11;42(1):21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Thai-Van H, Joly CA, Idriss S, Melki JB, Desmettre M, Bonneuil M, et al. Online digital audiometry vs. conventional audiometry: a multi-centre comparative clinical study. Int J Audiol. 2023. Apr;62(4):362–7. [DOI] [PubMed] [Google Scholar]
- 21.Kohlert S, Bromwich M. Mobile tablet audiometry in fluctuating autoimmune ear disease. J Otolaryngol - Head Neck Surg J Oto-Rhino-Laryngol Chir Cervico-Faciale. 2017. Mar 7;46(1):18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Frank A, Goldlist S, Mark Fraser AE, Bromwich M. Validation of SHOEBOX QuickTest Hearing Loss Screening Tool in Individuals With Cognitive Impairment. Front Digit Health. 2021;3:724997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Musselman JRB, Spector LG, Krailo MD, Reaman GH, Linabery AM, Poynter JN, et al. The Children’s Oncology Group Childhood Cancer Research Network (CCRN): case catchment in the United States. Cancer. 2014. Oct 1;120(19):3007–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Brown AL, Sok P, Scheurer ME, Rabin KR, Marcotte EL, Hawkins DS, et al. An updated assessment of 43,110 patients enrolled in the Childhood Cancer Research Network: A Children’s Oncology Group report. Cancer. 2022. Jul 15;128(14):2760–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Poynter JN, Richardson M, Roesler M, Krailo M, Amatruda JF, Frazier AL. Family history of cancer in children and adolescents with germ cell tumours: a report from the Children’s Oncology Group. Br J Cancer. 2018. Jan;118(1):121–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.SHOEBOX Help Center [Internet]. [cited 2024 Apr 10]. Available from: https://help.shoebox.md/Content/Home.htm [Google Scholar]
- 27.Wang M, Ai Y, Han Y, Fan Z, Shi P, Wang H. Extended high-frequency audiometry in healthy adults with different age groups. J Otolaryngol - Head Neck Surg J Oto-Rhino-Laryngol Chir Cervico-Faciale. 2021. Aug 26;50(1):52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hulsker CCC, el Mansori I, Fiocco M, Zsiros J, Wijnen MHW, Looijenga LHJ, et al. Treatment and Survival of Malignant Extracranial Germ Cell Tumours in the Paediatric Population: A Systematic Review and Meta-Analysis. Cancers. 2021. Jul 16;13(14):3561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lone DW, Sadak KT, Miller BS, Sample JM, Hubbard AK, Wolter C, et al. Growth Hormone Deficiency in Childhood Intracranial Germ Cell Tumor Survivors. J Endocrinol Metab. 2022. Jun;12(3):79–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Robison LL, Armstrong GT, Boice JD, Chow EJ, Davies SM, Donaldson SS, et al. The Childhood Cancer Survivor Study: a National Cancer Institute-supported resource for outcome and intervention research. J Clin Oncol Off J Am Soc Clin Oncol. 2009. May 10;27(14):2308–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Leisenring WM, Mertens AC, Armstrong GT, Stovall MA, Neglia JP, Lanctot JQ, et al. Pediatric cancer survivorship research: experience of the Childhood Cancer Survivor Study. J Clin Oncol Off J Am Soc Clin Oncol. 2009. May 10;27(14):2319–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.American National Standards Institute. Maximum Permissible Ambient Noise Levels for Audiometric Test Rooms. New York, NY; 2003. Report No.: ANSI S3.1–1999. [Google Scholar]
- 33.American National Standards Institute. Methods for Manual Pure-Tone Threshold Audiometry. New York, NY; 2004. Report No.: ANSI S3.21–2004. [Google Scholar]
- 34.American National Standards Institute. Specifications for Audiometers. New York, NY; 2004. Report No.: ANSI S3.6–2004. [Google Scholar]
- 35.Bass JK, Bhagat SP. Challenges in ototoxicity monitoring in the pediatric oncology population. J Am Acad Audiol. 2014. Sep;25(8):760–74; quiz 782–3. [DOI] [PubMed] [Google Scholar]
- 36.Sherief LM, Rifky E, Attia M, Ahmed R, Kamal NM, Oshi MAM, et al. Platinum-induced ototoxicity in pediatric cancer survivors: GSTP1 c.313A>G variant association. Medicine (Baltimore). 2022. Nov 11;101(45):e31627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Strebel S, Mader L, Sláma T, Waespe N, Weiss A, Parfitt R, et al. Severity of hearing loss after platinum chemotherapy in childhood cancer survivors. Pediatr Blood Cancer. 2022. Sep;69(9):e29755. [DOI] [PubMed] [Google Scholar]
- 38.Qaddoumi I, Bass JK, Wu J, Billups CA, Wozniak AW, Merchant TE, et al. Carboplatin-associated ototoxicity in children with retinoblastoma. J Clin Oncol Off J Am Soc Clin Oncol. 2012. Apr 1;30(10):1034–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Brooks B, Knight K. Ototoxicity monitoring in children treated with platinum chemotherapy. Int J Audiol. 2018. Sep;57(sup4):S34–40. [DOI] [PubMed] [Google Scholar]
- 40.Knight KR, Kraemer DF, Winter C, Neuwelt EA. Early changes in auditory function as a result of platinum chemotherapy: use of extended high-frequency audiometry and evoked distortion product otoacoustic emissions. J Clin Oncol Off J Am Soc Clin Oncol. 2007. Apr 1;25(10):1190–5. [DOI] [PubMed] [Google Scholar]
- 41.Abujamra AL, Escosteguy JR, Dall’Igna C, Manica D, Cigana LF, Coradini P, et al. The use of high-frequency audiometry increases the diagnosis of asymptomatic hearing loss in pediatric patients treated with cisplatin-based chemotherapy. Pediatr Blood Cancer. 2013. Mar;60(3):474–8. [DOI] [PubMed] [Google Scholar]
- 42.Goldman S, Bouffet E, Fisher PG, Allen JC, Robertson PL, Chuba PJ, et al. Phase II Trial Assessing the Ability of Neoadjuvant Chemotherapy With or Without Second-Look Surgery to Eliminate Measurable Disease for Nongerminomatous Germ Cell Tumors: A Children’s Oncology Group Study. J Clin Oncol Off J Am Soc Clin Oncol. 2015. Aug 1;33(22):2464–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Fangusaro J, Wu S, MacDonald S, Murphy E, Shaw D, Bartels U, et al. Phase II Trial of Response-Based Radiation Therapy for Patients With Localized CNS Nongerminomatous Germ Cell Tumors: A Children’s Oncology Group Study. J Clin Oncol Off J Am Soc Clin Oncol. 2019. Dec 1;37(34):3283–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Billmire D, Vinocur C, Rescorla F, Colombani P, Cushing B, Hawkins E, et al. Malignant mediastinal germ cell tumors: an intergroup study. J Pediatr Surg. 2001. Jan;36(1):18–24. [DOI] [PubMed] [Google Scholar]
- 45.Rogers PC, Olson TA, Cullen JW, Billmire DF, Marina N, Rescorla F, et al. Treatment of Children and Adolescents With Stage II Testicular and Stages I and II Ovarian Malignant Germ Cell Tumors: A Pediatric Intergroup Study—Pediatric Oncology Group 9048 and Children’s Cancer Group 8891. J Clin Oncol. 2004. Sep;22(17):3563–9. [DOI] [PubMed] [Google Scholar]
- 46.Starr A, Picton TW, Sininger Y, Hood LJ, Berlin CI. Auditory neuropathy. Brain J Neurol. 1996. Jun;119 ( Pt 3):741–53. [DOI] [PubMed] [Google Scholar]
- 47.Rance G, Starr A. Pathophysiological mechanisms and functional hearing consequences of auditory neuropathy. Brain J Neurol. 2015. Nov;138(Pt 11):3141–58. [DOI] [PubMed] [Google Scholar]
- 48.Passarello N, Tarantino V, Chirico A, Menghini D, Costanzo F, Sorrentino P, et al. Sensory Processing Disorders in Children and Adolescents: Taking Stock of Assessment and Novel Therapeutic Tools. Brain Sci. 2022. Oct 31;12(11):1478. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of in this study are not publicly available due to participant privacy but are available on request from the corresponding author, JNP.


