Abstract
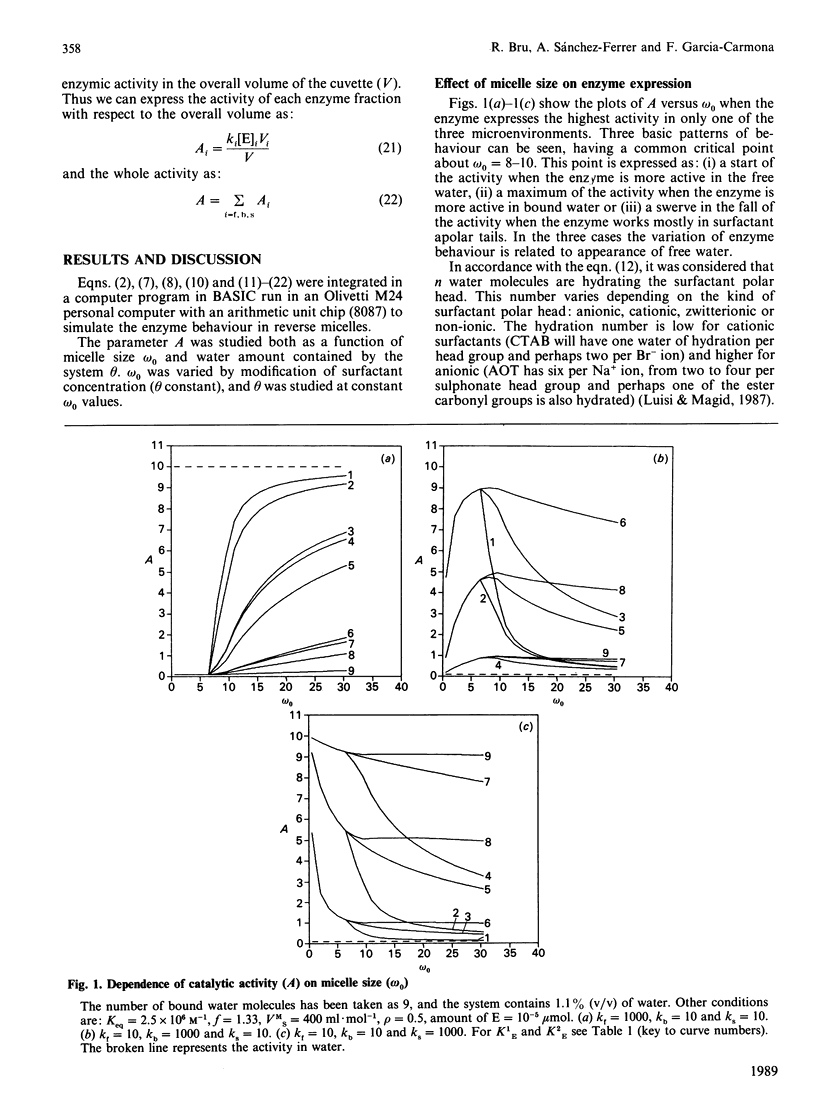
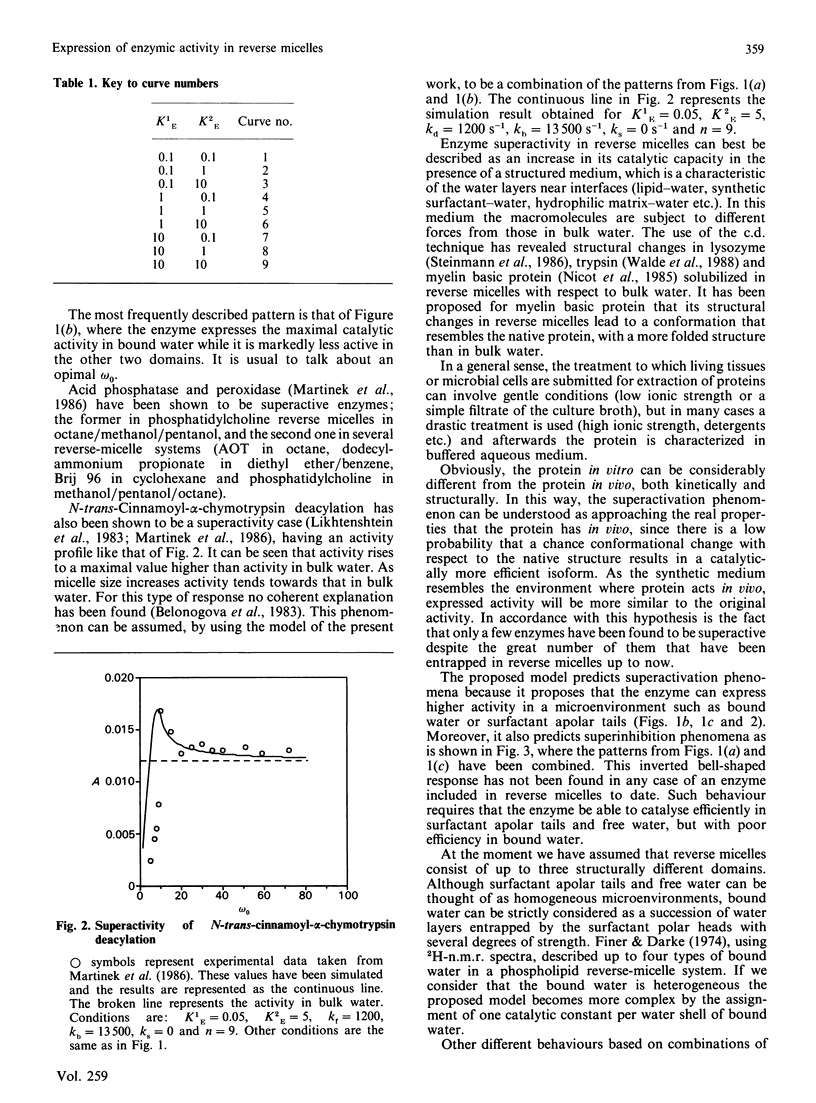
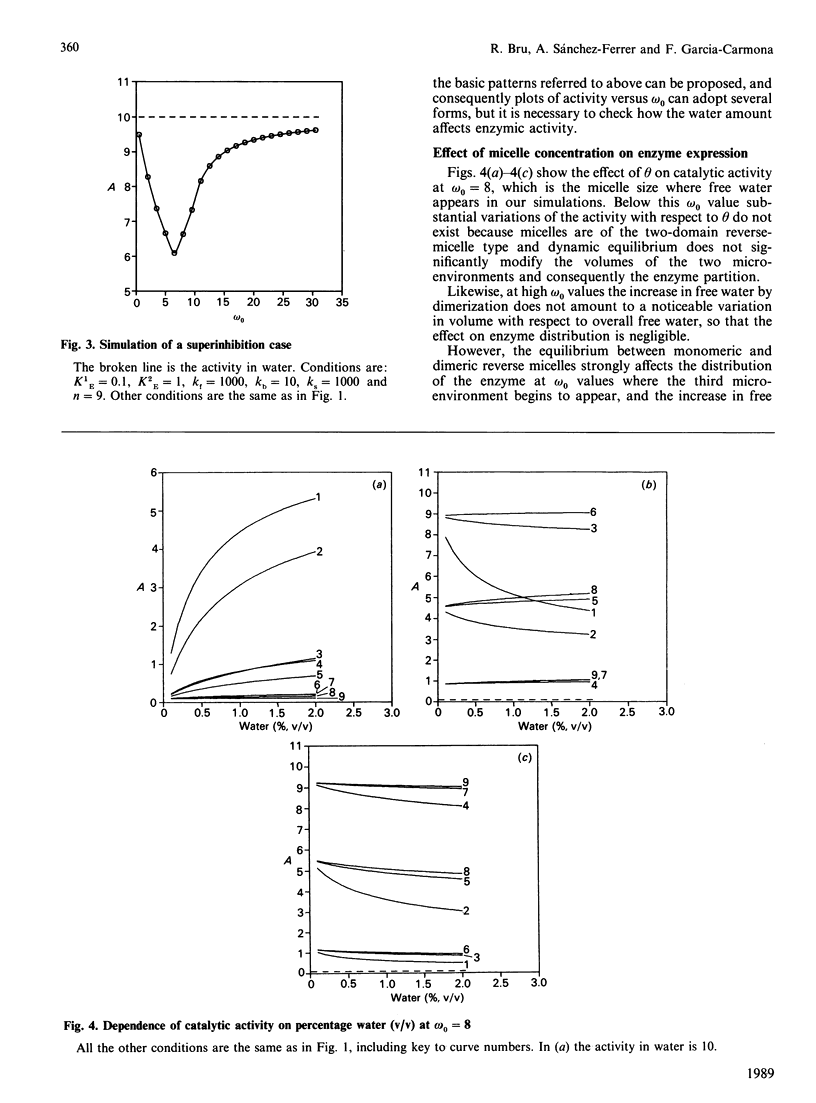
The present work deals with a theoretical model of catalysis by enzymes entrapped in reverse micelles. Three aspects of the enzyme-reverse-micelle system have been considered: structure, dynamics and enzyme distribution and catalysis in reverse micelles. A proposed structural model of reverse micelles [El Seoud (1984) in Reverse Micelles (Luisi, P. L. & Straub, B. E., eds.), p. 81, Plenum Press, New York] consists of three domains: surfactant apolar tails, bound water and free water. Dynamics are based on a dynamic equilibrium of association-dissociation that lead one to consider the dispersed polar phase as a pseudo-continuous phase [Luisi, Giomini, Pileni & Robinson (1988) Biochim. Biophys. Acta 947, 207-246]. Enzyme is distributed among the reverse-micelle domains and it expresses a catalytic constant for each one of them. The overall activity is calculated taking into account the volume in which enzyme is solubilized, and expressed as a function of the whole volume (V). The characteristic parameters of reverse micelles, omega 0 (= [H2O]/[surfactant]) and theta (= % water, v/v), were investigated as modulators of enzymic activity. Three basic patterns of modulation by omega 0 were found depending on which domain the enzyme expressed the highest catalytic constant. Combinations of those basic patterns lead to other modulation types that can be found experimentally, such as superactivation. Other combinations predict behaviour patterns not described to date, such as superinhibition. Dependence of catalytic activity on theta was only stated at omega 0 values around a critical value, which coincides with the appearance of free water.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Douzou P. Cryoenzymology in aqueous media. Adv Enzymol Relat Areas Mol Biol. 1980;51:1–74. doi: 10.1002/9780470122969.ch1. [DOI] [PubMed] [Google Scholar]
- Finer E. G., Darke A. Phospholipid hydration studied by deuteron magnetic resonace spectroscopy. Chem Phys Lipids. 1974 Feb;12(1):1–16. doi: 10.1016/0009-3084(74)90064-4. [DOI] [PubMed] [Google Scholar]
- HANAHAN D. J. The enzymatic degradation of phosphatidyl choline in diethyl ether. J Biol Chem. 1952 Mar;195(1):199–206. [PubMed] [Google Scholar]
- HIGUCHI W. I., MISRA J. Physical degradation of emulsions via the molecular diffusion route and the possible prevention thereof. J Pharm Sci. 1962 May;51:459–466. doi: 10.1002/jps.2600510514. [DOI] [PubMed] [Google Scholar]
- Hilhorst R., Spruijt R., Laane C., Veeger C. Rules for the regulation of enzyme activity in reserved micelles as illustrated by the conversion of apolar steroids by 20 beta-hydroxysteroid dehydrogenase. Eur J Biochem. 1984 Nov 2;144(3):459–466. doi: 10.1111/j.1432-1033.1984.tb08488.x. [DOI] [PubMed] [Google Scholar]
- Kuntz I. D., Jr, Kauzmann W. Hydration of proteins and polypeptides. Adv Protein Chem. 1974;28:239–345. doi: 10.1016/s0065-3233(08)60232-6. [DOI] [PubMed] [Google Scholar]
- Kurganov B. I., Tsetlin L. G., Malakhova E. A., Chebotareva N. A., Lankin V. Z., Glebova G. D., Berezovsky V. M., Levashov A. V., Martinek K. A novel approach to study of action of water-insoluble inhibitors of enzymic reactions. J Biochem Biophys Methods. 1985 Aug;11(2-3):177–184. doi: 10.1016/0165-022x(85)90053-3. [DOI] [PubMed] [Google Scholar]
- Luisi P. L., Giomini M., Pileni M. P., Robinson B. H. Reverse micelles as hosts for proteins and small molecules. Biochim Biophys Acta. 1988 Feb 24;947(1):209–246. doi: 10.1016/0304-4157(88)90025-1. [DOI] [PubMed] [Google Scholar]
- Luisi P. L., Magid L. J. Solubilization of enzymes and nucleic acids in hydrocarbon micellar solutions. CRC Crit Rev Biochem. 1986;20(4):409–474. doi: 10.3109/10409238609081999. [DOI] [PubMed] [Google Scholar]
- Malakhova E. A., Kurganov B. I., Levashov A. V., Berezin I. V., Martinek K. Novyi podkhod k izucheniiu fermentativnykh reaktsii s uchastiem nerastvorimykh v vode substratov. Pankreaticheskaia lipaza, vkliuchennaia v obrashchennye mitselly poverkhnostno-aktivnogo veshchestva v organicheskom rastvoritele. Dokl Akad Nauk SSSR. 1983;270(2):474–477. [PubMed] [Google Scholar]
- Martinek K., Levashov A. V., Klyachko N. L., Pantin V. I., Berezin I. V. The principles of enzyme stabilization. VI. Catalysis by water-soluble enzymes entrapped into reversed micelles of surfactants in organic solvents. Biochim Biophys Acta. 1981 Jan 15;657(1):277–294. doi: 10.1016/0005-2744(81)90151-0. [DOI] [PubMed] [Google Scholar]
- Martinek K., Levashov A. V., Klyachko N., Khmelnitski Y. L., Berezin I. V. Micellar enzymology. Eur J Biochem. 1986 Mar 17;155(3):453–468. doi: 10.1111/j.1432-1033.1986.tb09512.x. [DOI] [PubMed] [Google Scholar]
- Nicot C., Vacher M., Vincent M., Gallay J., Waks M. Membrane proteins in reverse micelles: myelin basic protein in a membrane-mimetic environment. Biochemistry. 1985 Nov 19;24(24):7024–7032. doi: 10.1021/bi00345a041. [DOI] [PubMed] [Google Scholar]
- Samama J. P., Lee K. M., Biellmann J. F. Enzymes and microemulsions. Activity and kinetic properties of liver alcohol dehydrogenase in ionic water-in-oil microemulsions. Eur J Biochem. 1987 Mar 16;163(3):609–617. doi: 10.1111/j.1432-1033.1987.tb10910.x. [DOI] [PubMed] [Google Scholar]
- Steinmann B., Jäckle H., Luisi P. L. A comparative study of lysozyme conformation in various reverse micellar systems. Biopolymers. 1986 Jun;25(6):1133–1156. doi: 10.1002/bip.360250612. [DOI] [PubMed] [Google Scholar]
- Walde P., Peng Q., Fadnavis N. W., Battistel E., Luisi P. L. Structure and activity of trypsin in reverse micelles. Eur J Biochem. 1988 Apr 15;173(2):401–409. doi: 10.1111/j.1432-1033.1988.tb14013.x. [DOI] [PubMed] [Google Scholar]


