Abstract
A ketogenic diet (KD) can induce weight loss and improve glycemic regulation, potentially reducing the risk of type 2 diabetes development. To elucidate the underlying mechanisms behind these beneficial effects of a KD, we investigated the impact of a KD on organ-specific insulin sensitivity (IS) in skeletal muscle, liver, and adipose tissue. We hypothesized that a KD would increase IS in skeletal muscle. The study included 11 individuals with obesity who underwent a randomized, crossover trial with two 3-week interventions: 1) a KD and 2) a standard diet. Skeletal muscle IS was quantified as the increase in glucose disposal during a hyperinsulinemic-euglycemic clamp (HEC). Hepatic IS and adipose tissue IS were quantified as the relative suppression of endogenous glucose production (EGP) and the relative suppression of palmitate flux during the HEC. The KD led to a 2.2-kg weight loss and increased insulin-stimulated glucose disposal, whereas the relative suppression of EGP during the HEC was similar. In addition, the KD decreased insulin-mediated suppression of lipolysis. In conclusion, a KD increased skeletal muscle IS in individuals with obesity.
Article Highlights
It remains unclear whether the glucose-lowering effect of a ketogenic diet is due to increased glucose disposal in skeletal muscle or due to an increased insulin-mediated suppression of endogenous glucose production.
We investigated how the ketogenic diet affects organ-specific insulin sensitivity.
A 3-week ketogenic diet increased insulin-mediated glucose disposal with no change in insulin-mediated suppression of endogenous glucose production. In addition, a decreased insulin-mediated suppression of lipolysis was observed.
A 3-week ketogenic diet increases skeletal muscle insulin sensitivity in individuals with obesity.
Graphical Abstract
Introduction
A ketogenic diet (KD) is a carbohydrate-restricted diet that has been used to treat drug-resistant epilepsy in children for decades (1). However, evidence for a multitude of beneficial effects of ketone bodies is emerging, including improvement of cardiac (2) and metabolic health (3). The KD induces weight loss (4), improves glycemic control and variability (5–7), and reduces the need for glucose-lowering medications in individuals with type 2 diabetes (8). The mechanism by which the KD improves glucose metabolism is yet to be fully understood. However, the observed reduction in glucose levels suggests a potential enhancement of insulin sensitivity (IS). This is somewhat counterintuitive because a KD inherently involves a high amount of fat intake, which may cause excess lipid accumulation and insulin resistance (IR). This association has been demonstrated numerous times in individuals with overweight, obesity, and type 2 diabetes (9,10). A potential explanation for this apparent paradox is that excess lipid accumulation only occurs with concomitant hyperinsulinemia (11), which can be mitigated by strict limitation of carbohydrate intake.
Because the majority of previous studies assessing the effects of a KD only evaluated the effects on glucose levels, it remains unclear whether the glucose-lowering effect is due to increased glucose disposal in skeletal muscle or due to an increased insulin-mediated suppression of endogenous glucose production (EGP) (12,13). Therefore, we aimed to investigate how the KD affects organ-specific IS (skeletal muscle, liver, and adipose tissue) in individuals with obesity and IR. We hypothesized that a KD would increase insulin-stimulated glucose disposal as a marker of an increase in skeletal muscle IS.
Research Design and Methods
Study Design and Participants
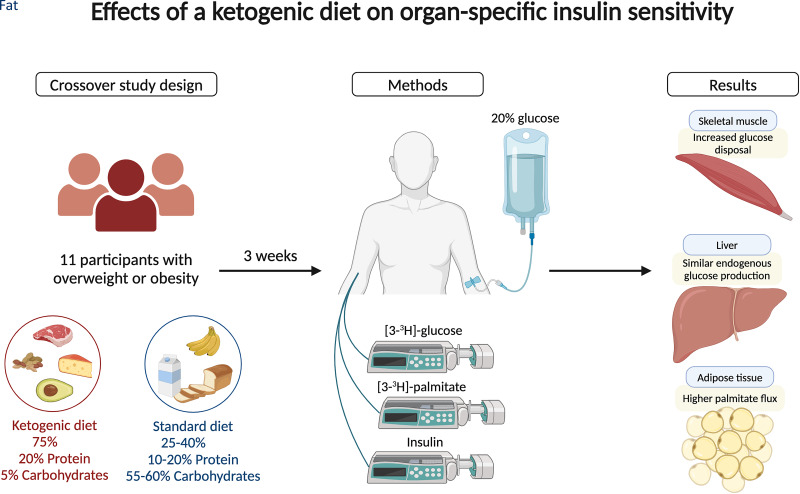
The study followed a randomized controlled crossover design, consisting of two periods in random order, separated by a 1-week washout period: 1) a 3-week KD, and 2) a 3-week standard diet (SDD). Both diet periods were concluded with a metabolic study day. Inclusion criteria were age 50–70 years and BMI 28–40 kg/m2. Exclusion criteria were 1) pharmacological treatment that could affect outcome measures, such as insulin, glucose-lowering medications, statins, or β-blockers; 2) HbA1c >48 mmol/mol; 3) impaired cardiac function; 4) impaired renal function; 5) liver disease; 6) active or prior cancer; 7) anemia; 8) blood donations within the last 3 months; 9) participation in other studies involving radioactive isotopes within the last 6 months; and 10) alcohol abuse. The study protocol was approved by The Central Denmark Region Committees on Health Research Ethics (no. 1–10-72–232-18) and registered at ClinicalTrials.gov (identifier NCT05012748).
All participants were screened for eligibility through assessment of demographic data and medical history, followed by a physical examination and body composition assessment using a whole-body DXA scan (Horizon, Hologic, and Discovery; Hologic). Written informed consent was obtained from the participants who met the inclusion criteria, and they began the first 3-week diet period within 7 days. Participants were instructed to abstain from alcohol during the KD and refrain from strenuous physical activity for 72 h before both study days. Participants were also instructed to fast from 10 p.m. the night before the study day, which was conducted at the Department of Nuclear Medicine and PET Center, Aarhus University Hospital.
Dieting Periods
All participants received an individualized, isocaloric KD plan based on sex, age, weight, and approximate physical activity level. Each diet plan included options for every meal, ensuring a macronutrient distribution of 5% energy intake (E%) from carbohydrates, 20 E% protein, and 75 E% fat. The diet plans were created by a registered dietitian. A low glycemic index source of carbohydrate was recommended during the KD. Fat intake ranged from 200 to 300 g with 30–40% allocated to saturated fats, 40–50% to monounsaturated fats, and 10–20% to polyunsaturated fats, encompassing both ω-3 and ω-6 fatty acids. Participants were responsible for buying ingredients, preparing meals, and adhering to the diet in their usual environment. During the SDD, participants were instructed to follow the Nordic Nutritional Recommendations (45–60 E% carbohydrates, 10–20 E% protein, 25–40 E% fat).
The participants were randomly assigned in a 1:1 ratio to receive either the KD or SDD in the first study period. To ensure compliance, participants were required to provide daily reports and instructed to measure their plasma levels of β-hydroxybutyrate (OHB) using a flash glucose monitoring system (Freestyle Libre; Abbott Diabetes Care Ltd., Witney, UK) every morning (7 a.m.) and evening (7 p.m.) throughout both 3-week diet periods. Participants who did not increase their OHB levels to 0.3 mmol/L during the first week of the KD would be excluded. They were also instructed to maintain their typical activity levels, ensuring similar levels of physical exertion across the two study periods.
Metabolic Study Day
This study was conducted as part of a larger research setup comprising metabolic examinations and positron emission tomography (PET) scans (14). However, the focus of this specific study was solely on the metabolic examinations. The study days were divided into a basal period (0–360 min) without insulin infusion followed by a clamp period during which a hyperinsulinemic-euglycemic clamp (HEC) was conducted (360–540 min) (Fig. 1). The metabolic examinations included collection of blood samples, indirect calorimetry, HEC, and infusions of radioactive isotope–labeled tracers for measuring glucose and palmitate flux. Additionally, a whole-body DXA scan was performed 1–2 days after each study day to compare the body composition between the two diets.
Figure 1.
Study day design.
Blood and Urine Samples
Upon the arrival of participants, an intravenous catheter was inserted in each antecubital vein, with the right vein designated for infusions and the left vein for the sampling of heated arterialized venous blood. Blood samples were collected according to the study setup shown in Fig. 1. Plasma glucose, OHB, free fatty acid (FFA), lactate, and insulin levels were measured at t = 360 min and 540 min. Total cholesterol, HDL, LDL, triglyceride, hemoglobin, and erythropoietin levels were measured at baseline (t = 0 min). Glucose samples were analyzed immediately using the YSI 2300 STAT Plus glucose analyzer (YSI Life Sciences, Yellow Springs, OH). OHB samples were stored at −20°C, and other samples were stored at −80°C until batch analysis. Plasma OHB concentrations were quantified using hydrophilic interaction liquid chromatography–tandem mass spectrometry (15). FFA concentrations were analyzed using an enzymatic colorimetric method assay for nonesterified fatty acids-HR (Wako Chemicals, Neuss, Germany), the lactate concentrations were analyzed with immobilized enzyme biosensor technology (YSI 2300; YSI Life Sciences), and insulin concentrations were analyzed with Insulin Elisa (Mercodia AB, Uppsala, Sweden). Urine was collected from participants upon arrival until the end of the basal period and again during the entire clamp period for quantification of urine urea nitrogen using absorption photometry (Cobas 6000, Roche) to estimate protein oxidation.
Indirect Calorimetry
Indirect calorimetry was used to measure resting energy expenditure (EE) and respiratory exchange ratio (RER) during the basal period (t = 50 min) and clamp period (t = 450 min) (Jaeger Oxycon Pro, Intramedic). Mean values of a 15-min period collection of respiratory gases were used for calculations.
Hyperinsulinemic-Euglycemic Clamp
During the clamp period, insulin (Humulin Regular) was infused at a continuous rate of 0.6 mU/kg/min. The plasma glucose concentration was measured every 10 min during insulin infusion and a 20% glucose solution was infused at variable rates to maintain euglycemia at 5 mmol/L. The M value was calculated as the mean glucose infusion rate per kilogram of whole-body mass during the last three measurements (t = 520–540 min).
Tracers
At t = 90 min, a 12 µCi bolus of [3-3H]-glucose (GE Healthcare) was administered, followed by continuous infusion at 0.12 µCi/min until t = 540 min. To prevent rapid dilution, [3-3H]glucose was also added to the glucose infusion during the HEC, resulting in a total of 100 µCi [3-3H]glucose in a 500 mL solution of 20% glucose. Additionally, a continuous infusion of [3-3H]-palmitate (GE Healthcare) was infused at 0.3 µCi/min in the basal period (210–270 min) and in the clamp period (480–540 min). Blood samples were drawn at baseline (t = 0 min) and in quadruplicates at the end of the infusions (t = 240–270 and 510–540 min) to measure specific activities of both tracers. The glucose rate of appearance (Ra) and rate of disappearance (Rd) were calculated using Steele’s nonsteady-state equation (16). EGP equals Ra during the basal period and was calculated by subtracting the glucose infusion rate from the Ra during the clamp period. Systemic palmitate flux was determined as described by Christensen et al. (17).
Statistics
Statistical analyses and graphs were generated using Stata 14 (StataCorp, College Station, TX) and GraphPad Prism 9 (GraphPad Software, San Diego, CA). Data are presented as mean ± SD or median (interquartile range), unless stated otherwise.
To evaluate normal distribution, QQ plots were examined. Data were assessed by performing paired t tests or Wilcoxon signed-rank tests. A linear mixed-effects model, with time and treatment as factors, was used to compare changes in hormone and substrate concentrations during the study day at t = 360 min and t = 540 min. The model's assumptions were validated by examining scatter and QQ plots of the predicted versus fitted residuals. When appropriate, data were logarithmically transformed. Interaction between time and treatment are shown as “interaction” and main effects are shown as “treatment,” indicating the two diets, or “time,” indicating effects from the basal period to the clamp period, followed by the corresponding P value. A P value of <0.05 was deemed significant. The participants' sex was not factored into the statistical analysis of the data.
Because this study was part of a larger one aiming to assess changes in myocardial fatty acid metabolism using [11C]palmitate PET scans (14), the data presented here were predefined secondary outcomes without power calculations.
Data and Resource Availability
The data and resources underlying this article will be shared on reasonable request.
Results
Inclusion, Completion, and Compliance
A total of 12 eligible volunteers were screened and recruited. However, one participant was excluded due to a severe migraine attack on the first study day. Therefore, 11 volunteers with obesity were included (n = 6 women; age 57 ± 6 years; BMI 32.5 ± 4.1 kg/m2). One participant did not complete the HEC on the study day after the KD due to discomfort during the PET scans. All participants achieved an OHB level of 0.3 mmol/L within the first week of the KD, resulting in no exclusions. In general, the participants tolerated the KD well, with none reporting severe symptoms.
Biochemistry and Body Composition
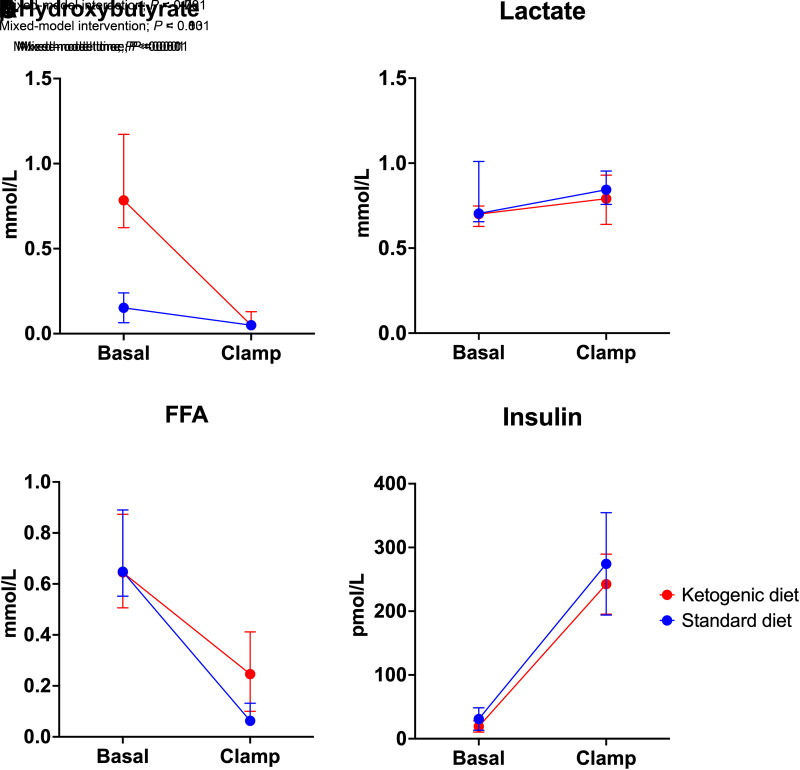
The KD lowered the concentration of triglycerides (Table 1), but no differences were observed in other biomarkers. After 3 weeks of the KD, there was a decrease in body weight (2.2 kg), fat mass (1.1 kg), and lean mass (1.4 kg) (all P < 0.05) compared with the SDD. Plasma OHB concentrations were higher in the basal period during the KD but were fully suppressed to the same concentration during the HEC after both diets (intervention: P < 0.001; time: P < 0.001; interaction: P < 0.001) (Fig. 2A). Plasma lactate concentrations were similar between the two diets (intervention: P = 0.1; time: P = 0.3; interaction: P = 0.7) (Fig. 2B). Plasma FFA concentrations were similar during both periods, but with a reduced suppression during the HEC for the KD (intervention: P = 0.8; time: P < 0.001; interaction: P = 0.2) (Fig. 2C). Plasma insulin concentrations were similar during the basal period with a slightly lower concentration for the KD during the HEC (intervention: P = 0.03, time: P < 0.001; interaction: P = 0.08) (Fig. 2D).
Table 1.
Participant profile
| Screening | KD | SDD | P value* | |
|---|---|---|---|---|
| Age (years) | 56.6 ± 5.8 | |||
| HbA1c (mmol/mol) | 36.7 ± 6.2 | |||
| Weight (kg) | 96.4 ± 12.1 | 93.7 ± 12.2 | 95.9 ± 12.2 | <0.05 |
| BMI (kg/m2) | 32.5 ± 4.1 | 31.6 ± 4.5 | 32.3 ± 4.3 | <0.05 |
| Fat mass (kg) | 36.3 ± 9.2 | 35.1 ± 9.8 | 36.2 ± 9.5 | <0.05 |
| Lean mass (kg) | 57.9 ± 10.6 | 56.5 ± 10.1 | 57.9 ± 10.2 | <0.05 |
| Fat (%) | 37.6 ± 7.9 | 37.2 ± 8.4 | 37.5 ± 7.9 | 0.45 |
| BMD (g/mL) | 1.1 ± 0.1 | 1.2 ± 0.1 | 1.1 ± 0.1 | 0.06 |
| Cholesterol (mmol/L) | 5.8 (5.0–6.4) | 5.3 (3.9–5.8) | 5.3 (4.3–6.3) | 0.60 |
| HDL (mmol/L) | 1.5 ± 0.3 | 1.3 ± 0.4 | 1.4 ± 0.4 | 0.12 |
| LDL (mmol/L) | 3.4 ± 0.8 | 3.7 ± 1.1 | 3.3 ± 0.7 | 0.6 |
| Triglyceride (mmol/L) | 1.6 ± 0.8 | 0.9 ± 0.2 | 1.4 ± 0.7 | <0.05 |
| Hemoglobin (mmol/L) | 8.8 ± 0.7 | 8.4 ± 0.7 | 8.3 ± 0.8 | 0.40 |
| Erythropoietin (IU/L) | 10.3 ± 5.7 | 8.6 ± 2.7 | 10.3 ± 4.7 | 0.23 |
| Hematocrit (%) | 0.4 ± 0.0 | 0.4 ± 0.0 | 0.4 ± 0.0 | 0.3 |
| ALAT (units/L) | 24.0 (19.0–30.0) | 24.0 (18.0–29.0) | 22.0 (18.0–42.0) | 0.75 |
| TSH (103 IU/L) | 2.8 ± 1.4 | 2.3 ± 1.0 | 2.4 ± 1.0 | 0.53 |
| Glucose (t = 360 min) | 4.7 ± 0.4 | 5.2 ± 0.4 | <0.001 |
Comparison is made between the two diet periods. Values are shown as mean ± SD or median (interquartile range). ALAT, alanine aminotransferase; BMD, bone mineral density; TSH, thyroid-stimulating hormone.
P values were calculated using a paired samples t test and the Wilcoxon rank-sum test (cholesterol, ALAT, and fasting insulin).
Figure 2.
Substrate and hormone concentrations during the study day taken at t = 360 and 540 min. A: Plasma -hydroxybutyrate concentrations were higher in the basal period after the KD compared with the SDD. B: Plasma lactate concentrations were similar. C: Plasma FFA concentrations were similar during both periods. In A–C, data are presented as median with interquartile range. D: Plasma insulin concentrations were similar during the basal period with a slightly lower concentration after the KD during the clamp. Data are presented as mean with SD. All data were analyzed using linear mixed-model analysis.
Insulin Sensitivity
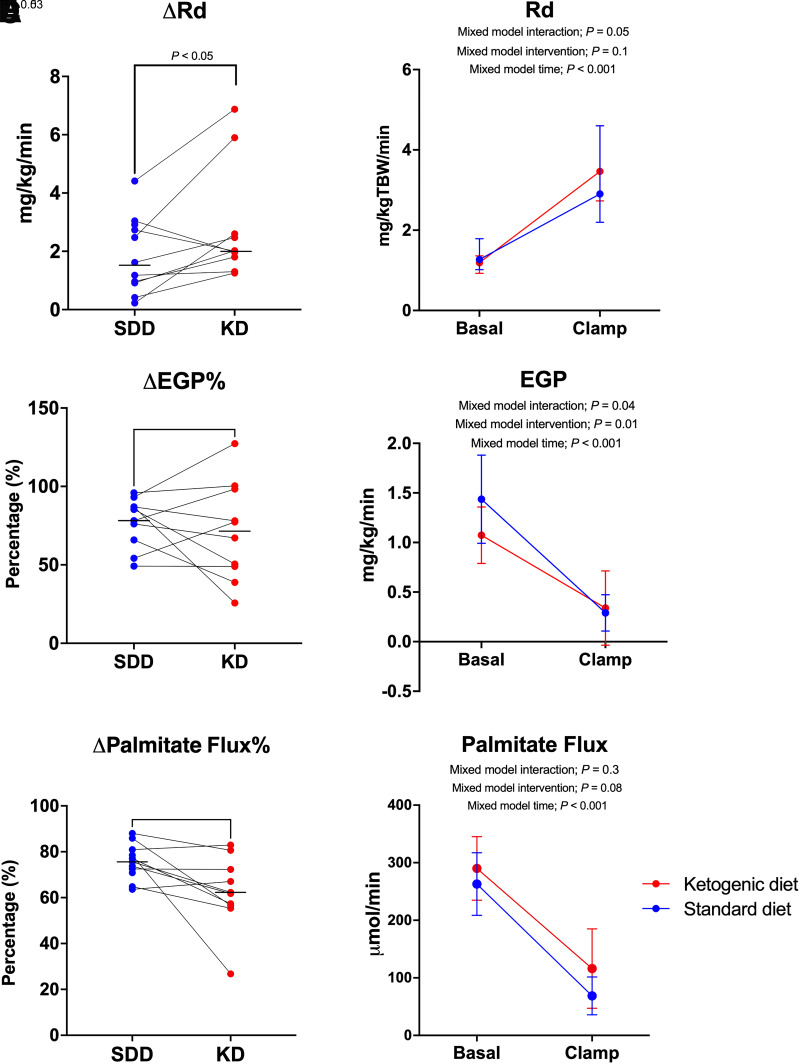
As a measurement of skeletal muscle IS, we evaluated the increase in glucose Rd during the HEC (ΔRd). Glucose ΔRd was higher after the KD compared with the SDD (2.0 [1.7–3.4] vs. 1.6 [0.9–2.9] mg/kg/min; P < 0.05) (Fig. 3A). The glucose Rd was similar after the two diets during the basal period, but there was a more pronounced increase in glucose Rd during insulin infusion after the KD (intervention: P = 0.1; time: P < 0.001; interaction: P = 0.05) (Fig. 3B). Adjusting the ΔRd for the increase in insulin for each participant did not change these results (P < 0.05), and the M value was not significantly different between interventions (KD: 3.0 [2.0–6.3] vs. SDD: 2.6 [1.4–4.7] mg/kg/min; P = 0.06) (Supplementary Material).
Figure 3.
Measurements of organ-specific insulin sensitivity. A: The KD increased glucose disposal during hyperinsulinemia (Rd) compared with the SDD. Data were analyzed with a Wilcoxon test. B: Rd was similar in the basal period with a higher increase in Rd during insulin infusion after the KD. Data are presented as median with interquartile range. C: The KD did not change the relative suppression of EGP (EGP%) during hyperinsulinemia compared with the SDD. Data were analyzed with a paired t test. D: EGP was lower in the basal period and decreased less during the clamp after the KD, compared with the SDD. Data are presented as mean with SD. E: The KD lowered the relative suppression of palmitate flux ( palmitate flux%) compared with the SDD. Data were analyzed with a paired t test. F: The KD induced a slightly lower suppression of palmitate flux from the basal to the clamp period. Data are presented as mean with SD. Abbreviation: TBW, total body weight.
To assess hepatic IS, we examined the relative suppression of EGP (ΔEGP%) during insulin infusion. When comparing ΔEGP% during insulin infusion, there was no difference between the KD and the SDD (71 ± 31% vs. 78 ± 15%; P = 0.53) (Fig. 3C). However, EGP was lower during the basal period after the KD (1.1 ± 0.3 vs. 1.4 ± 0.4 g/kg/min; P = 0.05), and decreased less during the HEC (intervention: P = 0.01; time: P < 0.001; interaction: P = 0.04) (Fig. 3D). As an estimate of adipose tissue IS, we quantified the insulin-mediated suppression of lipolysis as the relative change in palmitate flux during the HEC. The KD resulted in a reduced suppression of palmitate flux compared with the SDD (62 ± 16 vs. 76 ± 8%; P < 0.05) (Fig. 3E). This was caused by a reduced suppression from basal (290 ± 55 vs. 263 ± 54 mol/min; P < 0.05) to the clamp period (116 ± 69 vs. 69 ± 33 mol/min; P = 0.06) (intervention: P = 0.08; time: P < 0.001; interaction: P = 0.3) (Fig. 3F). Because this was a crossover design, we repeated the aforementioned linear mixed-effect analyses with inclusion of trial order in the analysis; the results did not affect our estimates (data not shown).
Energy Expenditure and Substrate Oxidation
EE was similar after the KD and SDD for both the basal period (1672 ± 223 vs. 1685 ± 234 kcal/24 h; P = 0.75) and the clamp period (1699 ± 241 vs. 1727 ± 247 kcal/24 h; P = 0.11) (intervention: P = 0.7; time: P = 1.0; interaction: P = 0.4) (Fig. 4A), but with a consistently lower RER after the KD during the basal period (0.71 ± 0.05 vs. 0.78 ± 0.05; P < 0.001) and the clamp period (0.76 ± 0.04 vs. 0.84 ± 0.07; P < 0.05) compared with the SDD (intervention: P < 0.001; time: P < 0.01; interaction: P = 0.7) (Fig. 4B). Protein oxidation was higher after the KD during the basal period compared with the SDD (374 ± 113 vs. 260 ± 83 kcal/24 h; P < 0.05), However, the levels were comparable during the clamp period between the two diets (260 ± 62 vs. 221 ± 104 kcal/24 h; P = 0.13) (intervention: P = 0.001; time: P = 0.001; interaction: P = 0.1).
Figure 4.
Measurements with indirect calorimetry. A: EE values were similar after both diets and periods. B: RER was consistently lower during the KD compared with the SDD. C: Protein oxidation (ox) was higher in the basal period after the KD but similar during the clamp. All data are presented as mean with SD.
Discussion
A KD has primarily been used for weight loss in individuals with obesity and IR. However, the precise mechanisms responsible for the beneficial impact on IR remain elusive, especially given the prevailing perception that a high amount of fat intake is unfavorable, primarily due to concerns regarding excess lipid accumulation in nonadipose tissue (lipotoxicity). Our main findings contribute to explaining some of the effects of a KD, with a 2.2-kg weight loss, on glucose and lipid metabolism: 1) the KD increased glucose disposal during hyperinsulinemia; 2) the KD reduced basal EGP and fasting glucose levels but did not affect the suppression of EGP during hyperinsulinemia; and 3) the KD impaired the suppression of lipolysis during hyperinsulinemia.
Insulin-Stimulated Glucose Disposal
IR plays a crucial role in the development of type 2 diabetes, caused by reduced amounts of insulin receptors and impaired insulin binding, signal transduction, and postreceptor processes (18). A KD may increase IS in skeletal muscle by reducing insulin levels through the restriction of carbohydrate intake. Previous studies have demonstrated that a KD can enhance IS in individuals with type 2 diabetes, quantified as a higher M value during an HEC (19,20). The HEC is considered the gold standard for measurement of IS (21), and the M value is often considered representative of skeletal muscle IS, given the significant role of skeletal muscle in glucose disposal during hyperinsulinemic conditions (85–90%) (22,23). However, the M value reflects both insulin-stimulated glucose uptake (glucose Rd) and insulin-suppressed EGP; thus, the independent effects of each parameter cannot be assessed.
In our study, we assessed skeletal muscle IS by measuring the increase in glucose Rd during the HEC, allowing us to isolate the effect of a KD on glucose disposal. The KD resulted in an increased glucose Rd and, although we did not observe a significant effect on the M value, there was a noticeable trend toward an increase (P = 0.06). The absence of statistical significance is likely attributable to the diverging effects of the KD in skeletal muscle and liver, because the effect was less pronounced in the liver, as reflected by lower EGP suppression. Our results also differ from those of previous research demonstrating that a high-fat diet reduced IS in skeletal muscle, whereas a high-carbohydrate diet increased IS (24). However, it is important to note that the diets were hypercaloric, exceeding habitual caloric intake by more than 75%, lasted only 3 days, and included young, healthy men with normal BMI. These factors may explain the different outcomes observed with a high-fat diet. The underlying cause of a KD-related increase in insulin-mediated glucose disposal in our study cannot be determined from our findings. However, the reduced plasma glucose levels during a KD will result in decreased circulating insulin levels. Considering that high insulin levels can induce IR per se (25), it is plausible that the low insulin levels during a KD reduce IR. This notion aligns with findings observed during treatment with sodium-glucose cotransporter 2 inhibitors, where insulin-independent glucose loss in the urine reduces insulin levels and increases IS (26).
Interestingly, a direct effect of ketone bodies on IS has been reported. Thus, ingestion of ketone salts enhanced glucose tolerance after an oral glucose tolerance test in one study (27), and an acute infusion of OHB increased insulin-stimulated glucose uptake during an HEC in another study (28). However, in the latter study, IR was induced through a stimulation of lipolysis by a growth hormone infusion, and the increase in glucose uptake was likely mediated through the efficient suppression of lipolysis achieved with the OHB infusion. Others have demonstrated an inability of an OHB infusion to influence IS measured during an HEC, but this outcome was observed during relatively low circulating ketone body levels (0.4 mmol/L) (29). Overall, a KD may enhance skeletal muscle IS by lowering insulin levels and promoting weight loss (30).
Another noteworthy finding is the lower RER after the KD during the HEC. Given that IR in skeletal muscle is a key factor in the onset of type 2 diabetes (31), one might anticipate that an enhancement in skeletal muscle IS during insulin stimulation would result in a more pronounced increase in glucose oxidation after a KD. This aligns with the concept of metabolic flexibility, wherein increased IS typically improves the ability to switch to glucose oxidation during insulin infusion (32). In our study, however, the increase in glucose oxidation during the HEC was similar regardless of the preceding diet, indicating that a KD does not improve overall metabolic flexibility. It is important to note that RER can drop below 0.7 during a KD, due to the conversion of fatty acids into ketone bodies (33). This process uses oxygen but does not necessarily result in an equivalent amount of CO2 production. Instead, some ketone bodies accumulate or are eliminated through urine or breath. When the production of ketone bodies exceeds their use, the overall RER can reach levels below 0.7 (33). However, this should not significantly affect the RER measurement during the HEC, when ketogenesis is fully suppressed. A potential explanation for the similar change in RER could be that skeletal muscle only accounts for approximately 30% of the resting EE (34,35). Consequently, an increase in skeletal muscle IS after a KD may not be sufficient to significantly increase glucose oxidation and, thus, RER measurements during an HEC.
Insulin-Mediated Suppression of EGP
EGP encompasses gluconeogenesis and hepatic glycogenolysis. Both processes are stimulated by glucagon and inhibited by insulin; therefore, EGP can be viewed as a marker of overall hepatic IS (36). However, it is important to note that gluconeogenesis tends to maintain a consistent level under typical physiological circumstances, suggesting that alterations in EGP primarily stem from changes in glycogenolysis (37). In individuals with type 2 diabetes, the inability of insulin to suppress these processes results in elevated plasma glucose levels (38). In our study, a KD resulted in decreased basal EGP, whereas no effect of the KD was observed during the HEC. Moreover, there was no difference in the relative inhibition of EGP, suggesting a lack of change in hepatic IS during a KD. The reduction in EGP and subsequent plasma glucose levels during the basal period may be attributed to the adaptive process by which the body becomes accustomed to using ketone bodies as its primary oxidative fuel source, thereby reducing the demand for glucose. The underlying mechanism is likely similar to what occurs during 48 h of fasting, when a 20% reduction in plasma glucose and a 25% decrease in glucose production and use have been reported (39).
The effects of a high-fat, a high-carbohydrate, and an intermediate-fat, intermediate-carbohydrate diet on IS during an HEC have been previously reported (40). In that study, six participants adhered to each of the three diets for 11 days. Although all three diets reduced EGP during the HEC, the KD resulted in the least suppression of EGP, demonstrating a difference between the diets. Conversely, we found no difference in our study. The disparity between our findings could potentially be attributed to variations in insulin infusion rates, because they used a lower rate of insulin infusion (20 mU/body surface area m2/min) compared with our slightly higher infusion rate (23.6 mU/body surface area m2/min). This distinction is crucial because higher levels of insulin may override smaller differences in hepatic IR if the insulin dose exceeds what is needed for maximal suppression of EGP (41). It is also worth considering the variation in participant characteristics, given that the Bisschop et al. study (40) included normal-weight individuals in contrast to our participants with obesity. However, it is unlikely that a KD would induce a greater degree of hepatic IR in lean individuals compared with those who already exhibit some level of IR.
Insulin-Mediated Suppression of Lipolysis
Insulin efficiently suppresses lipolysis in adipose tissue, with lean and insulin-responsive individuals demonstrating greater efficacy compared with those with obesity and type 2 diabetes (42). Consequently, impaired adipose tissue IS contributes to elevated circulating FFA levels.
In our study, we assessed adipose tissue IS by quantifying lipolysis inhibition during the HEC using the FFA tracer [3-3H]-palmitate instead of a glycerol tracer. We preferred the FFA tracer because it reflects the amount of FFA delivered to other organs, which we find more physiological relevant for whole-body substrate metabolism. We observed a reduced insulin-mediated suppression of lipolysis after a KD. This aligns with a previous investigation in which, during an HEC, the FFA concentration was less reduced in individuals on a high-fat diet compared with a high-carbohydrate diet (72% vs. 86%) (40). The adverse impact of a KD on adipose tissue IS raises concerns because increased levels of circulating FFAs can induce IR in other tissues through lipotoxicity (10). However, this concern may be mitigated during a KD, where low insulin levels may allow excess FFA to be oxidized and used for energy instead of accumulating in nonadipose tissue. Therefore, elevated FFA levels may be harmful only in conjunction with increased insulin levels, rather than inherently, challenging the notion that IR in adipose tissue per se significantly mediates IR in other organs. This is also supported by data from studies using sodium-glucose cotransporter 2 inhibitors, where increased circulating FFA levels were seen together with an increase in skeletal muscle IS (26,43).
Limitations
There are certain limitations to consider in our study. First, the sample size was relatively small; however, the crossover design increases the statistical strength. Second, the KD was standardized to match the expected EE of each participant, but the participants still had a weight loss of 2.2 kg, which could have affected our findings because weight loss can increase IS (30). However, a DXA scan might overstate changes in lean body mass after a KD, due to alterations in the hydration status (44). During a KD, glycogen depots are depleted and, because glycogen binds water, the subsequent water loss will lead to a lower estimate of lean mass measured with DXA (45). Therefore, the recorded weight loss may not accurately reflect true weight loss.
Conclusions
A 3-week KD led to an increase in skeletal muscle IS, no change in hepatic IS, and a decrease in adipose tissue IS. These results highlight the seemingly opposite adaptive effects of the KD on organ-specific IS. Although the positive effects of the KD on skeletal muscle IS indicate potential metabolic benefits of this dietary approach, further studies with longer durations and hard end points, such as the onset of type 2 diabetes, are needed to establish the efficacy of the KD.
This article contains supplementary material online at https://doi.org/10.2337/figshare.26317192.
Article Information
Funding. This research was funded by the Novo Nordisk Foundation grants NNF19OC0058872 (to N.M.) and NNF18OC0052953 (to L.C.G.).
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. T.V.L., K.M.L., C.B.A., L.C.G., E.S., and N.M. conceptualized the study with relevant methodology. T.V.L., K.M.L., M.G.B.P., and M.L.G.K. performed the study. T.V.L. and M.G.B.P. performed the formal analyses. L.C.G. conceptualized the study with relevant methodology. T.V.L. wrote the original draft of the manuscript. All authors contributed to reviewing and editing the final manuscript. T.V.L. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Funding Statement
This research was funded by the Novo Nordisk Foundation grants NNF19OC0058872 (to N.M.) and NNF18OC0052953 (to L.C.G.).
References
- 1. Ye F, Li X-J, Jiang W-L, Sun H-B, Liu J. Efficacy of and patient compliance with a ketogenic diet in adults with intractable epilepsy: a meta-analysis. J Clin Neurol 2015;11:26–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Ferrannini E, Mark M, Mayoux E. CV Protection in the EMPA-REG OUTCOME trial: a “thrifty substrate” hypothesis. Diabetes Care 2016;39:1108–1114 [DOI] [PubMed] [Google Scholar]
- 3. Dowis K, Banga S. The potential health benefits of the ketogenic diet: a narrative review. Nutrients 2021;13:1654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Kosinski C, Jornayvaz FR. Effects of ketogenic diets on cardiovascular risk factors: evidence from animal and human studies. Nutrients 2017;9:517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Dashti HM, Al-Zaid NS, Mathew TC, et al. Long term effects of ketogenic diet in obese subjects with high cholesterol level. Mol Cell Biochem 2006;286:1–9 [DOI] [PubMed] [Google Scholar]
- 6. Hussain TA, Mathew TC, Dashti AA, Asfar S, Al-Zaid N, Dashti HM. Effect of low-calorie versus low-carbohydrate ketogenic diet in type 2 diabetes. Nutrition 2012;28:1016–1021 [DOI] [PubMed] [Google Scholar]
- 7. Tay J, Luscombe-Marsh ND, Thompson CH, et al. A very low-carbohydrate, low-saturated fat diet for type 2 diabetes management: a randomized trial. Diabetes Care 2014;37:2909–2918 [DOI] [PubMed] [Google Scholar]
- 8. Mayer SB, Jeffreys AS, Olsen MK, McDuffie JR, Feinglos MN, Yancy WS, Jr. Two diets with different haemoglobin A1c and antiglycaemic medication effects despite similar weight loss in type 2 diabetes. Diabetes Obes Metab 2014;16:90–93 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Shulman GI. Ectopic fat in insulin resistance, dyslipidemia, and cardiometabolic disease. N Engl J Med 2014;371:1131–1141 [DOI] [PubMed] [Google Scholar]
- 10. Ferrannini E, Barrett EJ, Bevilacqua S, DeFronzo RA. Effect of fatty acids on glucose production and utilization in man. J Clin Invest 1983;72:1737–1747 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Czech MP. Insulin action and resistance in obesity and type 2 diabetes. Nat Med 2017;23:804–814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Yuan X, Wang J, Yang S, et al. Effect of the ketogenic diet on glycemic control, insulin resistance, and lipid metabolism in patients with T2DM: a systematic review and meta-analysis. Nutr Diabetes 2020;10:38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Møller N. Ketone body, 3-hydroxybutyrate: minor metabolite—major medical manifestations. J Clin Endocrinol Metab 2020;105:dgaa370. [DOI] [PubMed] [Google Scholar]
- 14. Luong TV, Pedersen MGB, Abild CB, et al. A ketogenic diet lowers myocardial fatty acid oxidation but does not affect oxygen consumption: a study in overweight humans. Obesity (Silver Spring) 2024;32:506–516 [DOI] [PubMed] [Google Scholar]
- 15. Sørensen LK, Rittig NF, Holmquist EF, et al. Simultaneous determination of β-hydroxybutyrate and β-hydroxy-β-methylbutyrate in human whole blood using hydrophilic interaction liquid chromatography electrospray tandem mass spectrometry. Clin Biochem 2013;46:1877–1883 [DOI] [PubMed] [Google Scholar]
- 16. Steele R. Influences of glucose loading and of injected insulin on hepatic glucose output. Ann N Y Acad Sci 1959;82:420–430 [DOI] [PubMed] [Google Scholar]
- 17. Christensen B, Nellemann B, Larsen MS, et al. Whole body metabolic effects of prolonged endurance training in combination with erythropoietin treatment in humans: a randomized placebo controlled trial. Am J Physiol Endocrinol Metab 2013;305:E879–E889 [DOI] [PubMed] [Google Scholar]
- 18. Kahn CR. The molecular mechanism of insulin action. Annu Rev Med 1985;36:429–451 [DOI] [PubMed] [Google Scholar]
- 19. Boden G, Sargrad K, Homko C, Mozzoli M, Stein TP. Effect of a low-carbohydrate diet on appetite, blood glucose levels, and insulin resistance in obese patients with type 2 diabetes. Ann Intern Med 2005;142:403–411 [DOI] [PubMed] [Google Scholar]
- 20. Goss AM, Gower B, Soleymani T, et al. Effects of weight loss during a very low carbohydrate diet on specific adipose tissue depots and insulin sensitivity in older adults with obesity: a randomized clinical trial. Nutr Metab (Lond) 2020;17:64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. DeFronzo RA, Tobin JD, Andres R. Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am J Physiol 1979;237:E214–E223 [DOI] [PubMed] [Google Scholar]
- 22. Klén R, Honka M-J, Hannukainen JC, et al. Predicting skeletal muscle and whole-body insulin sensitivity using NMR-metabolomic profiling. J Endocr Soc 2020;4:bvaa026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Defronzo RA. Banting Lecture. From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes 2009;58:773–795 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Lundsgaard A-M, Sjøberg KA, Høeg LD, et al. Opposite regulation of insulin sensitivity by dietary lipid versus carbohydrate excess. Diabetes 2017;66:2583–2595 [DOI] [PubMed] [Google Scholar]
- 25. Rizza RA, Mandarino LJ, Genest J, Baker BA, Gerich JE. Production of insulin resistance by hyperinsulinaemia in man. Diabetologia 1985;28:70–75 [DOI] [PubMed] [Google Scholar]
- 26. Merovci A, Solis-Herrera C, Daniele G, et al. Dapagliflozin improves muscle insulin sensitivity but enhances endogenous glucose production. J Clin Invest 2014;124:509–514 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Myette-Côté É, Neudorf H, Rafiei H, Clarke K, Little JP. Prior ingestion of exogenous ketone monoester attenuates the glycaemic response to an oral glucose tolerance test in healthy young individuals. J Physiol 2018;596:1385–1395 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Høgild ML, Hjelholt AJ, Hansen J, et al. Ketone body infusion abrogates growth hormone-induced lipolysis and insulin resistance. J Clin Endocrinol Metab 2023;108:653–664 [DOI] [PubMed] [Google Scholar]
- 29. Bratusch-Marrain PR, DeFronzo RA. Failure of hyperketonemia to alter basal and insulin-mediated glucose metabolism in man. Horm Metab Res 1986;18:185–189 [DOI] [PubMed] [Google Scholar]
- 30. Clamp LD, Hume DJ, Lambert EV, Kroff J. Enhanced insulin sensitivity in successful, long-term weight loss maintainers compared with matched controls with no weight loss history. Nutr Diabetes 2017;7:e282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. DeFronzo RA, Tripathy D. Skeletal muscle insulin resistance is the primary defect in type 2 diabetes. Diabetes Care 2009;32(Suppl 2):S157–S163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Goodpaster BH, Sparks LM. Metabolic flexibility in health and disease. Cell Metab 2017;25:1027–1036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Schutz Y, Ravussin E. Respiratory quotients lower than 0.70 in ketogenic diets. Am J Clin Nutr 1980;33:1317–1319 [DOI] [PubMed] [Google Scholar]
- 34. Frontera WR, Ochala J. Skeletal muscle: a brief review of structure and function. Calcif Tissue Int 2015;96:183–195 [DOI] [PubMed] [Google Scholar]
- 35. Zurlo F, Larson K, Bogardus C, Ravussin E. Skeletal muscle metabolism is a major determinant of resting energy expenditure. J Clin Invest 1990;86:1423–1427 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Sharabi K, Tavares CDJ, Rines AK, Puigserver P. Molecular pathophysiology of hepatic glucose production. Mol Aspects Med 2015;46:21–33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Nuttall FQ, Ngo A, Gannon MC. Regulation of hepatic glucose production and the role of gluconeogenesis in humans: is the rate of gluconeogenesis constant? Diabetes Metab Res Rev 2008;24:438–458 [DOI] [PubMed] [Google Scholar]
- 38. Golay A, Swislocki AL, Chen YD, Reaven GM. Relationships between plasma-free fatty acid concentration, endogenous glucose production, and fasting hyperglycemia in normal and non-insulin-dependent diabetic individuals. Metabolism 1987;36:692–696 [DOI] [PubMed] [Google Scholar]
- 39. Browning JD, Baxter J, Satapati S, Burgess SC. The effect of short-term fasting on liver and skeletal muscle lipid, glucose, and energy metabolism in healthy women and men. J Lipid Res 2012;53:577–586 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Bisschop PH, de Metz J, Ackermans MT, et al. Dietary fat content alters insulin-mediated glucose metabolism in healthy men. Am J Clin Nutr 2001;73:554–559 [DOI] [PubMed] [Google Scholar]
- 41. James DE, Stöckli J, Birnbaum MJ. The aetiology and molecular landscape of insulin resistance. Nat Rev Mol Cell Biol 2021;22:751–771 [DOI] [PubMed] [Google Scholar]
- 42. Groop LC, Bonadonna RC, DelPrato S, et al. Glucose and free fatty acid metabolism in non-insulin-dependent diabetes mellitus. Evidence for multiple sites of insulin resistance. J Clin Invest 1989;84:205–213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Ferrannini E, Baldi S, Frascerra S, et al. Shift to fatty substrate utilization in response to sodium-glucose cotransporter 2 inhibition in subjects without diabetes and patients with type 2 diabetes. Diabetes 2016;65:1190–1195 [DOI] [PubMed] [Google Scholar]
- 44. St-Onge M-P, Wang Z, Horlick M, Wang J, Heymsfield SB. Dual-energy X-ray absorptiometry lean soft tissue hydration: independent contributions of intra- and extracellular water. Am J Physiol Endocrinol Metab 2004;287:E842–E847 [DOI] [PubMed] [Google Scholar]
- 45. Kreitzman SN, Coxon AY, Szaz KF. Glycogen storage: illusions of easy weight loss, excessive weight regain, and distortions in estimates of body composition. Am J Clin Nutr 1992;56:292S–293S [DOI] [PubMed] [Google Scholar]