Abstract
The transcription factor nuclear factor erythroid 2‐related factor 2 (NRF2) has multifarious action with its target genes having redox‐regulating functions and being involved in inflammation control, proteostasis, autophagy, and metabolic pathways. Therefore, the genes controlled by NRF2 are involved in the pathogenesis of myriad diseases, such as cardiovascular diseases, metabolic syndrome, neurodegenerative diseases, autoimmune disorders, and cancer. Amidst this large array of diseases, diabetic neuropathy (DN) occurs in half of patients diagnosed with diabetes and appears as an injury inflicted upon peripheral and autonomic nervous systems. As a complex effector factor, NRF2 has entered the spotlight during the search of new biomarkers and/or new therapy targets in DN. Due to the growing attention for NRF2 as a modulating factor in several diseases, including DN, this paper aims to update the recently discovered regulatory pathways of NRF2 in oxidative stress, inflammation and immunity. It presents the animal models that further facilitated the human studies in regard to NRF2 modulation and the possibilities of using NRF2 as DN biomarker and/or as target therapy.
Keywords: biomarker, diabetic neuropathy, inflammation, NRF2, therapy target
Highlights
Nuclear factor erythroid 2‐related factor 2 (NRF2) is an oxidant stress response transcription factor, and is involved in metabolism, inflammation, autophagy, proteostasis, mitochondrial physiology, and immune responses.
Chronic hyperglycemia in diabetes and prediabetes conditions induces a decrease in NRF2 that leads to an increase of oxidative stress and inflammation.
NRF2 links neuroinflammation and apoptotic pathways and contributes to the development of diabetic neuropathy.
1. INTRODUCTION
The transcription factor nuclear factor erythroid 2‐related factor 2 (NRF2) is involved in myriad cellular processes, from oxidative stress to protein degradation. Due to its structure, it interacts with various molecules to induce these complex cellular functions.
NRF2, belongs to the subfamily of basic leucine zipper (bZIP) transcription factors along with other members such as nuclear factor erythroid‐derived 2 (NFE2) and NRF1, NRF2, and NRF3. It is encoded by the gene nuclear factor, erythroid 2 like 2 (NFE2L2). 1 NRF2 has several NRF2‐ECH homology (Neh) domains that control the NRF2 transcriptional activity. The bZip within Neh1 domain heterodimerizes with sMAF (small musculoaponeurotic fibrosarcoma proteins K, G, F) and other bZip proteins in order to recognize antioxidant response elements (ARE) inducing a subsequent activation of gene transcription. The Neh2 domain interacts with the Kelch domain from Kelch‐like‐ECH‐associated protein 1 (KEAP1) mediating NRF2 ubiquitination and degradation. 2 The group of Neh3‐5 domains are transcriptional activation structures by binding to transcriptional machinery. 3 The Neh6 domain has two redox‐independent degrons (DSGIS and DSAPGS) that bind to E3 ubiquitin ligase β‐transducin repeat‐containing protein, which, in oxidatively stressed cells, mediates NRF2 degradation. 4 The Neh7 domain interacts with retinoic X receptor alpha, repressing NRF2 activity. 5
All the described domains modulate NRF2 activity on target genes at transcriptional and posttranscriptional levels. Studies published in 2020 have shown that there are new NRF2 target genes beyond its already known redox‐regulating capacities. Therefore, genes that are involved in inflammation regulation, proteostasis, unfolded protein response, autophagy, and metabolism were recently reported. 6
As there are many diseases that have oxidative stress‐related pathological background, NRF2‐target genes are essential in various diseases such as cardiovascular diseases, metabolic syndrome, neuronal degeneration, autoimmune disorders, and cancer. 7 , 8 , 9 In various recent animal models, many additional ARE containing genes were found (eg, Nfe2l2‐knockout mice, 10 Keap1‐knockdown mice, Keap1‐knockout mice, 11 constitutive active NRF2‐E79Q‐knockin mice 12 ) alongside the already mentioned oxidative stress target genes.
The International Diabetes Federation reported in 2019 that over 400 million people worldwide are diagnosed with diabetes, 13 distributed as over 100 million individuals in China, 73 million in India, and 30 million in the United States. 14 The same number of individuals are diagnosed with prediabetes 15 , 16 , 17 and the health care system is also burdened by the comorbidities developed by patients with diabetes. 18 Amid the complications triggered by diabetes, the damage inflicted upon peripheral and autonomic nervous systems triggers various forms of neuropathy, and the incidence of this complication is not low as it will occur in half of patients diagnosed with diabetes. 19 , 20
Diabetic neuropathy (DN) affects both central (CNS) and peripheral nervous systems and inflicts deregulations at both levels. 21 Furthermore, central DN has Alzheimer's disease‐like characteristics increasing the risk of dementia, 22 comorbid depression, and anxiety 23 and results from oxytocinergic system deregulation. 24 One of the mechanisms involved in the development of DN is impaired insulin support. 25 Increased glucose leads to increased glutamate levels, which is an excitatory neurotransmitter that induces cognitive dysfunction and CNS damage. 26 Hyperglycemia damages the structure and function of the blood–brain barrier (BBB), affecting CNS. 27 As the BBB is the essential physiological barrier in drug delivery 28 the only drug that is used in DN is epalrestat. This is an aldose reductase inhibitor with proven safety and efficacy. 29 This lack of efficacious medication strengthens the need to seek out further therapy targets in DN, such as Nrf2.
The hyperglycemic conditions generated in diabetes associate mitochondrial deregulations, an increase in the reactive oxygen species (ROS) production and deregulations in the antioxidant defense system. 30 The exacerbated generation of ROS inflicts damages to the peripheral nerves 31 while the chronic hyperglycemic status deregulates mitochondrial functions, triggering injuries in the dorsal root ganglia and axons of sensory neurons, while reducing motor/sensory conduction speed. 32 , 33
The pathophysiology of DN involves complex inflammatory and apoptotic processes. 34 , 35 , 36 Due to the fact that human papillomavirus (HPV) induces an ubiquitary infection causing profound chronic inflammatory processes, 37 HPV infections were recently studied in DN, as histologically, diabetic foot alterations are similar with the lesions induced by HPV 38
Within these intricate inflammatory mechanisms, NRF2 bridges inflammatory and apoptotic pathways and regulates antioxidant proteins (eg, detoxifying enzymes) through the ARE binding site. Moreover, NRF2 links neuroinflammation and apoptotic pathways affecting the progression of DN. 39 The hyperglycemic status developed in diabetes stimulates oxidative stress and inflammation, downregulating the activity of NRF2. Acute hyperglycemia intensifies the expression of NRF2, whereas chronic hyperglycemia decreases it. The NRF2 downregulation induces microvascular changes, leading to DN. 40
Due to its ubiquitous functions, Nrf2 is involved in various diseases. Under normal physiological settings, Nrf2 is inactive and remains in the cytosol. The particularities that Nrf2 displays in diabetes is that the hyperglycemic status is a strong stimulus for oxidative stress and inflammation, and these pathological conditions downregulate the activity of Nrf2 through various neuroinflammatory pathways. Acute hyperglycemia increases Nrf2 expression, but a persistent hyperglycemia decreases its expression. This downregulation of Nrf2 causes various microvascular changes and induces DN therefore distinguishing Nrf2 involvement in DN in comparison with other diseases. 40
A general scheme of NRF2 pathway involvement is depicted in Figure 1.
FIGURE 1.
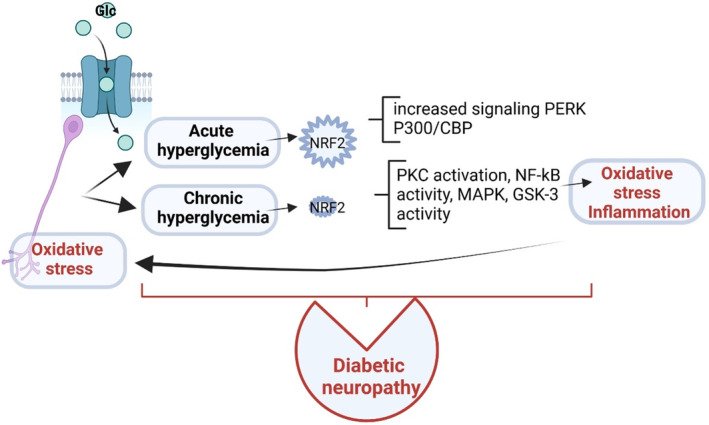
Molecular pathways of NRF2 involvement in DN. Glucose (Glc) intake is increased in diabetes leading to increased oxidative stress at the neuron level. Acute hyperglycemia associates an increase in NRF2 that induces protein kinase RNA‐like endoplasmic reticulum kinase (PERK) and p300/cellular retinol binding (CRB)‐binding protein (P300/CREB) activation. Chronic hyperglycemia induces a decrease in NRF2 that activates protein‐kinase C (PKC), activates NF‐kB, mitogen‐activated protein kinase (MAPK), and glycogen synthase kinase‐3 (GSK‐3). The alteration increases oxidative stress and sustains inflammation contributing to the development of DN. DN, diabetic neuropathy; NF‐κB, nuclear factor‐κB; NRF2, nuclear factor erythroid 2‐related factor 2.
NRF2 research is still an evolving field and new roles of the transcription factor are to be discovered, mainly in the pathology of various diseases. We will review some updated aspects related to NRF2 involvement in DN, the animal models that brought important scientific information on this topic and, finally, the interplay between NRF as marker and/or as therapeutical target.
2. MOLECULAR EVENTS APPENDING TO NRF2 FUNCTION
2.1. NRF2 and noncoding RNA
Noncoding RNAs (ncRNAs) are a group of RNA molecules with heterogenic structures that play a major role in posttranscriptional gene regulation and epigenetic gene silencing. 41 ncRNAs comprise microRNAs (miRNAs), long noncoding RNAs (lncRNAs), small interfering RNAs (siRNAs), circular RNAs (circRNAs), competing endogenous RNAs (ceRNAs) and piwi‐interacting RNAs (piRNAs). 42 miRNAs are structurally shaped by the action of two endonucleases, RNase III enzyme drosha and RNase III endonuclease dicer complex. Over 10 years ago it was demonstrated on an experimental model that a knockdown of the dicer enzyme‐encoding gene performed in pancreatic beta cells induces the development of diabetes. 43 More recently the ncRNA interactome in neuropathic pain was published 44 and it was revealed that microRNA structure assembly involves NRF2/mitogen‐activated protein kinase (MAPK) signal transduction. 45 Thoroughly revised by Pandey et al, 46 the involvement of ncRNA in DN focused on miRNA, lncRNA that is associated with this diabetes complication. Therefore, several years ago upregulated miRNAs were reported in DN 47 with overexpression of miR‐199a‐3p species. This upregulation induces the inhibition of serine protease inhibitor (SerpinE2) contributing to the tissue vascular damage associated with DN. 48 LncRNA MALAT1 was found upregulated in the peripheral‐blood mononuclear cells (PBMC) of DN patients. 49 It is known that MALAT1 can induce neuroinflammation by recruiting EZH2 to the NRF2 promoter and thus impeding Nrf2 expression. 50 Thus, MALAT1 inhibits NRF2, and induces inflammasome activation and ROS production in neuronal‐based in vitro and in vivo settings.
In DN, downregulated miRNAs were also reported, namely miRNA‐146a was found underexpressed in the PBMC of DN patients. miRNA‐146a downregulation activates the nuclear factor‐κB (NF‐κB) gene that induces the overexpression of tumor necrosis factor‐alpha (TNF‐α) and interleukin‐6 (IL‐6), 51 potent inflammatory cytokines that are associated once more with DN. 52
2.2. Epigenetic regulation of NRF2/KEAP1
The NRF2‐KEAP1 pathway is epigenetically regulated by genes that can be reversible or heritable during cell division. The epigenetic regulation of KEAP1/NFE2L2 expression is performed through several mechanisms such as DNA methylation, histone modification, and miRNAs, which are epigenetic modulations that can represent new therapeutic targets. 53 Multiple species of miRNAs have been demonstrated to regulate the NRF2‐KEAP1 pathway. For example, NRF2 overexpression activates the expression of miR‐144‐3p through binding to the AREs in the miR‐144‐3p promoter region. In lung cancer, it was demonstrated that if miR‐144‐3p is targeted, downregulation of NRF2 is induced, subsequently decreasing the expression of genes related to drug resistance. 54 Concomitantly, in lung tumors downregulation of the miR‐27 family and upregulation of miR‐200 family has a modulatory action on the NRF2/KEAP1 axis. 55
The role of lncRNA MALAT1 in immune regulation (eg, inflammatory cytokines induction) and angiogenesis is reported in diabetic complications. 56 , 57 Recent data has shown that lncRNA MALAT1 has an important role in the antioxidant defense system. 58 The hyperglycemia status enhances Sp1 binding at the MALAT1 promoter and elevated lncRNA MALAT1, enhances the binding of Sp1 at the Keap1 promoter, and activates its transcription. High Keap1 expression hinders Nrf2 nuclear movement, obstructing the transcription of the antioxidant response enzymes. Inhibiting lncRNA MALAT1 by its siRNA would lead to inhibition of Keap1 upregulation, thus liberating Nrf2 to translocate into the nucleus and to induce the transcription of antioxidant genes, namely the ones that encode HO1 and Sod2. LncRNA MALAT1 is a highly conserved lncRNA and it is linked to various pathologies including diabetes‐related complications. 59 , 60 If inhibition of lncRNA MALAT1 is performed, diabetic neurodegeneration is alleviated. 61 Moreover, lncRNA antisense noncoding RNA inhibits NF‐kB–mediated signaling reducing the overall proinflammatory processes. 62
3. NRF2 REGULATOR OF IMPORTANT CELLULAR PATHWAY
NRF2 as a transcription factor regulates the expression of hundreds of genes involved in the stress response. Discovered almost 30 years ago, 63 the couple NRF2‐repressor KEAP1 was studied in view of their molecular mechanisms, and their link with various cellular processes within their future clinical application. Thoroughly revised by Kopacz et al there are seminal issues in the NRF2/KEAP1 research domain; solving these issues could bring new therapeutical avenues. 64
3.1. NRF2 and oxidative stress
As already stated, NRF2 regulates antioxidant cellular response; this was its first discovered function, but lately other pathways accompanying cell survival, detoxification, metabolism, autophagy, proteostasis, inflammation, immunity, and differentiation were uncovered. Hence, NRF2 regulates hundreds of genes and is considered to be the hub of a vast regulatory complex system.
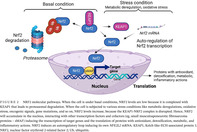
NRF2 regulates the oxidative stress, and the generated ROS are involved in complex physiological/pathological processes starting with normal physiology such as aging, and ending with pathological status such as obesity, cancer, diabetes, and neurodegenerative diseases. 65 NRF2 regulates more than 50 redox homeostasis‐related genes. 66 In cellular conditions that are characterized by the absence of ROS, Keap1 links to NRF2 and promotes its proteasomal degradation. This constitutive degradation of NRF2 guarantees that no augmentation of the NRF2 action will occur on target genes. When ROS accumulate, the Keap1‐NRF2 link is disrupted. During oxidative stress, the generated molecules react with sensor cysteines within KEAP1 (eg, cysteine 151, C151, C273, C288) and NRF2 escapes the degradation. 67 Consequently, the newly synthesized NRF2 builds up in the nucleus activating the cytoprotective genes expression. 3 Hence NRF2, using its sMAF proteins will bind to ARE in the regulatory regions of genes. Table 1 summarizes the genes that are regulated by NRF2 in the oxidative stress context 68 and Figure 2 depicts the molecular events 6 that govern NRF cellular pathways.
TABLE 1.
NRF2 targets in the redox system.
| Molecule (abbreviation) | Action |
|---|---|
| Thioredoxin | Elimination of ROS |
| Thioredoxin reductase | |
| Sulfiredoxin | |
| Peroxiredoxin | |
| Glutathione peroxidase | |
| Superoxide dismutase 1 (SOD1) | |
| Catalase (CAT) | |
| Glutathione S‐transferases (GR) | Reduction of glutathione (GSH) for the GSH regeneration NADPH‐mediated |
| Glutamate cysteine ligase (GCL) | Production of the cellular antioxidant glutathione (GSH) |
| NAD(P)H quinone oxidoreductase 1 (NQO1) | Detoxification/activation of quinones |
| Heme oxygenase 1 (HO‐1) | Degradation of heme to produce biliverdin, ferrous ion, carbon monoxide. |
Abbreviation: ROS, reactive oxygen species.
FIGURE 2.
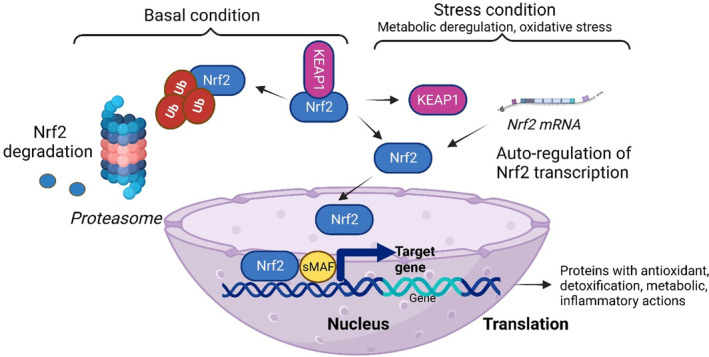
NRF2 molecular pathways. When the cell is under basal conditions, NRF2 levels are low because it is complexed with KEAP1 that leads to proteasomal degradation. When the cell is subjected to various stress conditions like metabolic deregulations, oxidative stress, oncogenic signals, gene mutations, and so on, NRF2 levels increase, because the KEAP1‐NRF2 complex is disrupted. Hence, NRF2 will accumulate in the nucleus, interacting with other transcription factors and cofactors (eg, small musculoaponeurotic fibrosarcoma proteins – sMAF) inducing the transcription of target genes and the translation of proteins with antioxidant, detoxification, metabolic, and inflammatory actions. NRF2 induces an autoregulatory loop inducing its own NFE2L2 mRNA. KEAP1, Kelch‐like‐ECH‐associated protein 1; NRF2, nuclear factor erythroid 2‐related factor 2; Ub, ubiquitin.
NRF2 regulates antioxidant pathways inducing enzymes to produce reduced glutathione (GSH) consumption and regeneration. For the GSH synthesis NRF2 will regulate glutamate‐cysteine ligase catalytic and modulator subunits and glutathione synthetase. Besides the redox enzymes presented in Table 1, glutathione reductase uses NADPH, a cofactor from anabolic reactions. 69
In the landscape of redox complex systems, NRF2 has several targets 6 (Table 1).
In the context of oxidative stress, NRF2‐target genes encode for cytoprotective proteins against toxic and oxidative injuries. Therefore, oxidative stress in pathological conditions can be identified in various diseases such as cardiovascular, metabolic, neuronal degeneration, autoimmune disorders, and cancer. 6 , 65 , 70 , 71 , 72
3.2. NRF2 and metabolic pathways
NRF2 regulates almost 200 genes influencing metabolism and cellular repair. 73 In the context of DN, NRF2 action becomes extremely important in the metabolic area. NRF2 is involved in various layers of metabolic functions. It regulates mitochondrial functions, nutrients uptake, anabolism, macromolecular biosynthesis, and energy metabolism. All these metabolic layers sustain the complex cell physiology. Using chromatin immunoprecipitation sequencing (ChIP‐Seq) analysis in cell lines 74 or in animal models, 75 numerous metabolic‐related genes were revealed as NRF2 targets. Genes involved in glycolysis regulation, pentose phosphate pathway, fatty acid pathways, glutamine, and glutathione metabolism were identified as NRF2 targets. In the brain, NRF2 expression is increased in astrocytes and microglia compared to neurons. 76 It is highly probable that the information circulation between astrocytes and neurons requires an increased metabolism so that astrocytes provide neurons with glycine, glutamate/glutamine and cysteine, namely GSH precursors supporting neuron function. 77
In animal models of diabetic db/db mice that have Keap1 gene knockdown (Keap1flox/−) it was shown that NRF2 activation inhibits gluconeogenesis by suppressing several molecular targets (eg, glucose‐6‐phosphatase, fructose‐1,6‐bisphosphatase 1, phosphoenolpyruvate carboxykinase [PCK1], peroxisome proliferator‐activated receptor g coactivator 1‐a, nuclear receptor subfamily 4, group A, member 2), preventing the initiation of diabetes. 78 Liver‐specific constitutive NRF2 activation induces the reduction of gluconeogenesis through PCK1 and pyruvate dehydrogenase kinase genes. 6
NRF2 is involved in various stages of the glycolysis cycle. Glucose‐6‐phosphate is converted by phosphoglucomutase (PGM) to glucose‐1‐phosphate. Within the glycogen metabolism cycle NRF2 activates PGM5, 1,4‐alpha‐glucan branching enzyme 1, phosphorylase kinase regulatory subunit alpha 1, and glucosidase alpha acid. The liver and the skeletal muscle are the main sites for glycogen synthesis/storage, and NRF2 regulates these processes. 79 In mouse models it was shown that constitutive NRF2 activation liver induces glycogen accumulation maintaining blood glucose during fasting. 79 Meanwhile, in animal models of skeletal muscle‐specific Keap1‐knockout mice, a decreased glycogen content is identified in the skeletal muscle, along with an improved glucose tolerance. 79
The metabolic pathways of iron and oxygen are functionally linked as iron is an essential cofactor element of enzymes involved in the major cellular chains: oxygen transport, oxidative phosphorylation, and metabolite oxidation. NRF2 regulates several genes that further regulate the iron metabolism. Recent studies have shown that genes involved in heme synthesis, hemoglobin catabolism, iron storage, and iron export are controlled by NRF2. Besides cancer, NRF2 activation and iron metabolism deregulation 80 were also noted in diabetes. Ten years ago, it was mentioned that the pathophysiology of diabetes revealed that iron accumulation sustains an increased risk of type 1 and type 2 diabetes. 81 As metabolic stress involves complex processes, one of these is the imbalance of the redox equilibrium that induces tissue damage and pancreatic β‐cell death. These cells are vulnerable to oxidative damage due to the iron accumulation and β‐cell ferroptotic‐like deregulation. Recent studies have shown a link between iron metabolism and redox status in diabetes based on ferroptotic‐like deregulation of pancreatic β‐cells. 82 Ferroptosis (a type of iron‐dependent and nonapoptotic cell death) was identified in the pancreatic β‐cell pathophysiology. The accumulation of toxic ROS and the deregulated ion metabolism 83 are both linked and controlled in diabetes by NRF2.
3.3. NRF2 in inflammation and immunity
Inflammation is a complex process having various roles, including the elimination of numerous aggressors, activating innate and adaptive immune cells. Many regulatory mechanisms control this inflammatory network, so that the processes resume after the pathogenic insult is eliminated and cell/tissue regeneration occurs. Bypassing these control mechanisms will lead to the chronic form of inflammation. Chronic inflammation is the trigger for many diseases from autoimmune diseases and neurodegenerative diseases to cancer. 84 Regulation of inflammation is performed through toll‐like receptor (TLR) pathways and is NRF2 mediated. TLR pathways controlled by NRF2 establish a connection between immune regulation and the oxidative stress responses via inflammation regulation. This close TLR‐NRF2 link in the inflammatory context is to be found as the basis of many diseases. Targeting this molecular “couple” can lead to the development of future therapeutic strategies. 85
Autoimmune disorders are known to have an excess of ROS generation, proven in the large array of autoimmune diseases from psoriasis to arthritis and from diabetes to Crohn's disease. Other conditions like obesity and aging add immune dysfunction and oxidative stress to these autoimmune deregulations. The accumulation of ROS triggers the activation of inflammatory cells that further sustain the inflammatory status through the secretion of inflammatory cytokines, chemokines, and factors that mediate various signal transduction and transcription pathways (eg, NF‐κB, signal transducer and activator of transcription 3, hypoxia‐inducible factor‐1 alpha, NRF2). The antioxidant enzymes (SOD and catalase) are imbalanced; therefore, therapies that supplement this deregulation of the defensive natural component can alleviate various autoimmune pathologies. 86
The expression of NRF2 in immune cells and in circulating blood cells was reported almost 20 years ago. 63 The highest levels of NRF2 were found in circulating monocytes, neutrophils, and T and B cells, which suggests a clear control of this transcription factor in the physiology of immune cells. 87 Any injury, infection, or chemical/physical trigger will initiate an inflammatory reaction where innate immune cells will be the first line of cells that will arrive at the injured site. Inflammatory cells will eliminate the infectious agent and will initiate the tissue repair processes. 84 ROS are an extended family of signaling molecules and mediators of inflammation. 6 As already stated, chronic inflammation develops as an uncontrolled process that leads to cell damage and cellular proliferation. Alongside many diseases, chronic inflammation is a stepping stone to diabetes. 77 NRF2 modulates redox metabolism alleviating ROS by directly inhibiting proinflammatory cytokine genes or inflammatory NF‐κB signaling. 6 Using ChIP‐seq that analyzes protein–DNA interactions, it was demonstrated that NRF2 binds directly to the promoter regions of IL‐6 and IL‐1β genes. This binding hinders RNA polymerase II enlistment blocking gene induction in innate immune cells, namely in macrophages. 88 NRF2 hinders IL‐6 expression by ARE‐dependent induction suppressing IL‐6 transcription. 89 NRF2 has a modulatory action by suppressing or activating immunity depending on the cellular physiology. For example, NRF2 activation within T cells lowers T activation and differentiation. 90
3.4. In vitro and in vivo models exploring NRF2 molecular events in DN
Animal models have been widely used to study the molecular mechanisms that are controlled by NRF2. In a Caenorhabditis elegans model, the aging processes and related pathologies, including neurological complications of diabetes were studied. Hence α‐dicarbonyls (α‐DCs) accumulate with age and, in mutated animals (glod‐4), hyperesthesia and neuronal damage was shown. TRPA‐1 acts as a sensor for α‐DCs, that has a conserved structure from worms to mammals. Furthermore, TRPA‐1 stimulates SKN‐1/NRF through calcium‐regulated kinase signaling, so that the detoxifying of α‐DCs is activated. Chaudhuri et al have shown in an animal model that TRPA‐1 can be a drug target for diabetes and neurodegenerative diseases. 91
Most animal models developed in this area are rodent models and several mechanisms were studied using these models. Chronic inflammatory status governs major diseases, therefore drugs that alleviate the inflammation are constantly being developed. These drugs are characterized by side effects, hence discovering other treatment approaches in the fight against chronic inflammation is a constant goal of the last decades. Natural‐derived molecules are good candidates in these studies. Recent reports have shown Withaferin A (WA), a bioactive molecule isolated from Withania somnifera, as having an antichronic inflammatory action. WA triggers NF‐kB and NRF2 signaling pathways and has proven its efficacy in both in vitro and in vivo animal models of various diseases including diabetes and neurodegenerative disorders. 92
Another natural polyphenolic derivative of benzoic acid, syringic acid (Syr) was tested in animal models to alleviate the oxidative stress status in diabetes and to improve the complications of DN. Syr was proved to eliminate ROS and modulate various transcriptional factors, including NRF2 in diabetic animal models. 93 Furthermore, Zhao et al found that streptozotocin (STZ)‐induced DN is correlated with antioxidant deficiency in the spinal dorsal root ganglia and the sciatic nerve. 94 More recent data 95 have shown that malondialdehyde (MDA) level in the brain, sciatic nerve, and spinal cord significantly increase in diabetic animals, and that Syr administration reduces MDA level in tissues proving Syr's antioxidant effect. Oxidative stress and impairment of antioxidants contribute to the damage of cognitive functions. 96 , 97 Ogut et al have shown in animal models that Syr can improve learning dysfunction and memory suppression through oxidative stress reduction. 98 Other natural compounds like rosmarinic acid, 99 , 100 , 101 epigallocatechin‐3‐gallate, gallic acid, 102 and ellagic acid 103 were documented as improving memory impairment. This memory impairment due to hyperglycemia is a consequence of cholinergic dysfunction, and therapies that influence cholinesterase activity are to be considered as important in diabetes‐induced neurodegeneration. 104 Some phenolic acids (eg, chlorogenic acid) in animal models reduced acetylcholinesterase in the hippocampi of cognitive deficits animals. 105 Around 20 years ago it was still unclear whether mitochondrial biogenesis has a neuroprotective mechanism in DN. 106 More recent studies have shown that mitochondrial biogenesis promotes cellular viability. 107 Additionally, in neurons, oxidative stress induces mtDNA injury, corroborated with reduction of mitochondrial biogenesis leading to the development of DN. 108 Contrarywise, processes leading to mitochondrial regeneration/biogenesis could decrease the risk of DN. 109 Hence, Syr administration induced an increase in the copy number of mitochondria in the brain and spinal cord. 93 At the genetic level, the process increases gene expression for peroxisome proliferator‐activated receptor‐γ coactivator 1 alpha (PGC‐1α), mitochondrial transcription factor A (TFAM), NRF1 and NRF2, and mitochondrial transcription factor B1. 110 , 111 Indeed, Syr administration increases the expression of PGC‐1α and NRF‐2 mRNA in the brain, inducing mitochondrial biogenesis. Hyperglycemia alters PGC‐1α activator, which consequently leads to TFAM and NRF2 expression impairment and mitochondrial dysfunction. 112 Various neurodegenerative diseases (eg, Alzheimer's, Parkinson's, amyotrophic lateral sclerosis, and Huntington's disease) demonstrate these molecular deregulations, 112 whereas in diabetes, a decrease of PGC‐1α and NRF‐1 gene expression was found. 113 In PGC‐1α‐knockout mice, experimentally triggered diabetes led to more severe DN. 114 It was reported that, in physiological conditions, the blood‐nerve barrier hinders the entrance of circulating T lymphocytes, whereas hyperglycemia damages this nervous‐vascular barrier and favors their entrance. Glycosylated myelin can be an antigen for the immune cells and will be subjected to phagocytosis by the innate immune cells. 115 Inflammatory cytokines, TNF‐α and IL‐1β, were proven to have deleterious effects on neurons and glial cells, and the induced neuroinflammation will lead to demyelination. 115 Modulating these inflammatory cytokines, namely reducing TNF‐α, IL‐1β, and IL‐6 secretion, would lead to a decrease in the sciatic nerve demyelination. 116
In another rat model of diabetes, vitamin D and rosuvastatin were studied for their neurotherapeutic potential. Various parameters were investigated in this animal model, from small/large nerve electrophysiological examination to neuronal inflammation, apoptosis, and various intracellular pathways. This animal model showed that vitamin D and/or rosuvastatin improved diabetes‐induced neuropathy through suppressing Notch1 and Wnt‐10α/β‐catenin pathways that hindered neuronal degeneration. 117 Rutin (a ubiquitous plant flavonoid) was also studied in animal models of DN. Pain assessment, motor nerve conduction velocity, and complex oxidative parameters were assessed in this animal model subjected to rutin therapy. Treatment with rutin and nimesulide (nonsteroidal anti‐inflammatory drug) attenuated all neurological parameters, with the combination having the highest effect on experimental DN. 118
In vitro studies were performed on cultured dorsal root ganglion (DRG) neurons in order to assess various therapeutical compounds. High glucose levels noticeably increased DRG apoptosis by increasing intracellular ROS and activating the NF‐κB signaling pathway. If cells were subjected to quercetin (Q), cinnamaldehyde (C), and hirudin (H), then caspase‐3 activation and apoptosis were significantly reduced. The expression of various molecules decreased after in vitro therapy (NF‐κB, IL‐6, TNF‐α), and NRF2 expression increased. The combination of quercetin, cinnamaldehyde, and hirudin induced the scavenging of ROS, activated NRF‐2 and alleviated inflammatory components. 119 , 120 Likewise, in a high‐glucose DRG neurons model it was shown that cells are sensitive to oxidative stress. In an in vitro model, the antioxidant/antiapoptotic effects of calcitonin gene‐related peptide (CGRP) were studied. The report shows CGRP increased the expression of NRF2 and heme oxygenase‐1 (HO‐1), via PI3K/AKT pathway, reducing apoptosis and oxidative stress. 121
4. NRF2 AS DN DISEASE MARKER AND THERAPY TARGET
As shown previously, NRF2/Keap1 pathway incorporates both metabolic and cytoprotective functions that are deregulated in diabetic conditions. 122 In chronic hyperglycemia, high superoxide anions escape from the electron transport chain within the mitochondria during aerobic respiration 123 and scavenging potency is overridden so that the oxidation processes at cellular levels are enhanced. 124 Soares et al have shown several years ago that diabetic skin has low expression of NRF2 and low antioxidant potency that leads to decreased wound healing. 125 Bone marrow–derived multipotent stromal cells (BMSCs) have a low expression of cytoprotective NRF2/Keap1 in diabetes. If NRF2‐directed transcription is activated, the redox homeostasis is restored in BMSCs and therefore their regenerative potency is recovered. 126 , 127
As previously mentioned, NRF2 is studied as a therapy target. Recent studies have shown that astragaloside II (AS II), a saponin purified from Astragalus spp, can modulate the immune response and tissue regeneration and stop the inflammatory response. AS II was studied in a STZ‐induced DN animal model evaluating its effect on podocyte injury and mitochondrial dysfunction. AS II ameliorated podocyte physiology and apoptosis in DN rats. Furthermore, AS II increased NRF2 expression and decreased Keap1, enabling antioxidative stress. These results have shown that AS II regulates NRF2 and therefore ameliorates podocyte injury in experimental DN. 128
The therapeutic effect of bergenin was reported in 2020 in another animal model of STZ‐induced DN. Using the analysis of cytokines, antioxidant genes, oxidative stress markers, nitric oxide production and MDA/nitrite tests, the bergenin effect on DN symptomatology was evaluated. Bergenin proved to have antioxidant properties modulating gene expression, namely downregulating inducible nitric oxide synthase and upregulating glutathione peroxidase and NRF2 in the nervous system. Moreover, bergenin modulated the pro‐ and anti‐inflammatory cytokines in DN mice. After treatment, the cytokines were predominantly anti‐inflammatory and an upregulation of antioxidant pathways was recorded. 129
In 2022, a set of new cinnamaldehyde compounds were evaluated in vitro, ex vivo, and in vivo for the capacity to restore peripheral nerve degeneration. Between them, the highest potency was demonstrated by a compound having a naphthaldehyde group. Its hydrophobic interactions with transient receptor potential cation channel subfamily A member 1 had the highest binding affinity. This compound hindered the oxidative stress in Schwann cells alleviating neurodegenerative processes. 130 Trichostatin A (TSA) is a reversible inhibitor of class I and II histone deacetylases, regulating peripheral neurodegeneration. TSA was recently shown to inhibit overall peripheral neurodegeneration (eg, myelin fragmentation, axonal degradation, trans‐dedifferentiation/proliferation of Schwann cells). 131 Moreover, additional studies have shown that the effect of TSA is associated with the Keap1‐NRF2 pathway. 132
In a high‐glucose rat model, the effect of hirudin on oxidative stress/apoptosis of spinal dorsal root ganglion cells was studied. This recent report outlines that the activity of spinal dorsal root ganglion cells is supported by hirudin due to its ROS scavenging potency, NRF‐2/HO‐1 pathway up‐regulation, NF‐κ B pathway inhibition, and caspase‐3/apoptosis downregulation. 133 In another DN rat model published in 2023, the translocator protein agonist Ro5‐4864 was proven to alleviate neuropathic pain. Besides increased myelin sheath thickness and protein expression, the compound improved the expression of NRF2, cytoplasmic HO‐1, and NQO1. 134 The bioflavonoid morin has a scavenging effect on ROS and it also proved its efficacy in DN animal models. Administration of morin reduced hyperalgesia and allodynia ameliorating mitochondrial SOD, membrane depolarization, and total ROS. The mechanism that morin triggered is based on the increased NRF2‐antioxidant defenses. 135
Gangliosides are highly involved in neurological diseases and their administration showed beneficial effects since the 80's. 136 More recently, the mechanisms of mono‐sialo‐tetrahexosyl‐ganglioside ganglioside in the treatment of DN were studied. Actually, this ganglioside attenuates neuroinflammation. 137 At least in Alzheimer's disease, one of the mechanisms of ganglioside action is through the NRF‐2/ARE signaling pathway. 138 Lipoic acid is a dithiol compound that reduces ROS, chelates metal ions, and regenerates vitamin C, vitamin E, and glutathione. Through its action, it enhances the antioxidant system, a process that was reported as relying on NRF‐2‐gene expression. Therefore, lipoic acid is another NRF2‐mediated compound that is beneficial in DN, and these results were reported in several clinical trials of DN patients. 139 Several phytochemicals were tested for their antioxidant potency and their neuroregenerative action. Polyphenols are known to interact with ROS and apigenin has proven its neuroprotective effect against oxidative stress through extracellular signal‐regulated kinase‐independent processes. 140 Sinomenine is a known alkaloid with anti‐inflammatory, immunosuppressive, and neuroregeneration capacities. 141 Sinomenine induces mechanisms that rely on mediating ferroptosis of hippocampal neurons. The actions of sinomenine were studied on cellular models of HT‐22 cells and in DN animal models. Sinomenine reduced hippocampus neuronal ferroptosis through the enhancement of EGF expression that activated the NRF2/HO‐1 signaling pathway. The in vitro results were confirmed in the DN animal model. 142
NRF2 is a marker that indicates whether the cell functions normally or has a profoundly deregulated cellular physiology. NRF2 is similarly a therapy target, as its upregulation can balance the pathological state induced by various factors, hyperglycemia included.
In Table 2 we present the expression of Nrf2 in different diseases including DN and the drugs that target NRF2.
TABLE 2.
Expression of NRF2 in various diseases and updated drugs that aim to regulate NRF2.
| Pathology | Nrf2 status | Action | Drug targeting Nrf2 | Drug action | Ref |
|---|---|---|---|---|---|
| Prostate cancer and castration‐resistant prostate cancer | Suppressed | Increased autophagy, apoptosis, and necrotic ptosis, favoring tumor metastasis and drug resistance | Neoadjuvant endocrine/chemo therapy | Drugs activate Nrf‐2, which induces the p120‐Nrf1 nuclear accumulation, significantly inhibiting NF‐κB DNA‐binding activity by decreasing the transactivation of androgen receptor | 143 |
| Acute myeloid leukemia | Overexpressed inducing autophagy, cytokines, and kinases' expression. | Nrf2 target genes (eg, NADPH quinone oxidoreductase I, glutamate‐cysteine ligase catalytic subunit, glutamate‐cysteine ligase modifier subunit) have NF‐κB binding site and hence these two signaling pathways cooperate in promoting chemoresistance to bortezomib treatment | 144 | ||
| Skin pathology induced by UVB irradiation | Nrf2 degraded by UVB | Nrf2 has a photo‐protective effect through the suppression of MMPs expression | 145 | ||
| Neuronal inflammation | Suppressed | Nrf2 suppression favors oxidative stress and inflammation through the activation of NOX4/ROS/NF‐kB pathway | 146 | ||
| Animal model of subdued to spinal cord injury Nrf2 deficient | Suppressed | Nrf2 suppression induces significant elevation in NF‐κB activation, TNF‐α production, and expression of MMP9 | 147 | ||
| Different in vitro, in vivo models and humans with Huntington disease | Suppressed | Nrf2 suppression induces neuroinflammation processes in microglia and macrophages | Triazole derivatives | Nrf2 induction inhibits proinflammatory cytokines | 148, 149 |
| Multiple sclerosis | Suppressed | Nrf2 suppression induces low control of antioxidative pathways | Dimethyl fumarate, approved by US Food and Drug Administration | Induces anti‐inflammatory action via Nrf2 antioxidant pathway | 150, 151 |
| Diabetes‐associated gastric pathology | Suppressed | Nrf2 suppression reduces the expression of HMOX1 gene with anti‐inflammatory activity | 152 | ||
| Obesity‐induced diabetes | Suppressed | Nrf2 suppression lowers the control on insulin secretion and insulin resistance, inducing hyperglycemia | 78 | ||
| Diabetic skin ulcers | Suppressed | Nrf2 suppression and/or knockdown delays wound healing, increases oxidative DNA damage, reduced TGF‐β1 levels, high MMP9 expression and increased apoptosis | 153 | ||
| Diabetic nephropathy | Suppressed | Increased oxidative damage and inflammation | Epigallocatechin gallate | Induces activation of Nrf2, ERK and p38 MAPK signaling pathways for anti‐inflammation action | 154 |
| Neuropathic inflammation | Suppressed | Increased oxidative damage and inflammation | Rosmarinic acid | Induces Nrf2 activation and attenuates chronic constriction injury –induced neuropathic inflammation | 155 |
Abbreviations: MMP, matrix metalloproteinase; NF‐κB, nuclear factor‐κB; NRF2, nuclear factor erythroid 2‐related factor 2; ROS, reactive oxygen species; TGF‐β1, transforming growth factor‐beta1; TNF‐α, tumor necrosis factor‐alpha; UVB, ultraviolet B.
5. NRF2 DETRIMENTAL IN HUMAN PATHOLOGY
As already stated, Nrf2 is located at the intersection of crucial signaling pathways, regulating various cellular functions, beyond the redox balance, and is involved in cellular metabolism, proteostasis, mitochondrial function, and inflammation. However, due to its complex involvement, the specific time frame for NRF2 activation or inhibition is of seminal importance. NRF2 signaling has an intrinsic dualism mediating beneficial effects but also some detrimental effects in biological processes associated with aging, neurodegeneration, metabolic diseases, long‐COVID‐19, and carcinogenesis. 156 For example, Nrf2 has a context‐dependent manner of action in the cardiovascular system. Recent emerging data show the unexpected role of Nrf2 in mediating cardiovascular maladaptive remodeling and dysfunction in vascular diseases. All these findings gain clinical relevance having Nrf2 pathways as targets in cardiovascular diseases. 157 In bacterial infections, a recent report has shown another detrimental effect attributed to Nrf2. Besides various roles, immunity maintains the host's iron homeostasis sequestrating iron from pathogens and hence controlling the infection. Nucleotide‐oligomerization domain‐like receptors (NLRs) are highly involved in this process. Moreover, it was shown that NLRP6 alters the host resistance to bacterial infection. NLRP6 deficiency upsurges the iron exporter ferroportin‐mediated iron efflux through a NRF2‐dependent process. Therefore, NLRP6 becomes a therapeutic target for limiting bacterial iron acquisition. 158
As mentioned before, increased NRF2 activity is beneficial for proper health, nevertheless, the fact that NRF2 becomes activated in neoplastic transformed cells is detrimental. Tumor cells display persistent NRF2 activation that govern their therapeutic resistance and aggressiveness. This persistent activation of NRF2 confers molecular advantages for these transformed cells in terms of genetic, metabolic, and immunological characteristics underlaying the detrimental action of NRF2 in tumorigenesis. 159
Recent cumulative evidence points out that Nrf2 activators and their involvement in human pathology should be kept in mind before introducing them in clinical practice.
6. CONCLUSION
NRF2 is one of the recent leading subjects of extensive research. Studies regarding NRF2 concentrate upon inflammation, metabolism, and oxidative stress as foremost factors in human pathologies. First identified as an oxidant stress response transcription factor, recent studies have shown its ubiquitous involvement in regulation of seminal cellular processes such as metabolism, inflammation, autophagy, proteostasis, mitochondrial physiology, and last but not least immune responses. These key regulator functions of NRF2 are sustained by the fact that this transcription factor is centered in a complex regulatory network that controls important functions. Additionally, due to its central role, NRF2's interconnections contribute to the initiation and development of metabolism‐ or inflammation‐associated diseases, diabetes included.
As diabetes and prediabetes are alarmingly increasing worldwide, and knowing that half of the patients will sustain injuries of the peripheral and autonomic nervous systems, developing various forms of DN, the studies of the intimate molecular mechanisms are seminal. The pathophysiology of DN involves inflammatory and apoptotic complex pathways. which are bridged by NRF2. Through its ARE binding site, NRF2 regulates antioxidant proteins (eg, detoxifying enzymes). Moreover, NRF2 links neuroinflammation and apoptotic pathways affecting the progression of DN.
There are several challenges regarding the current research in Nrf2. Therefore, when evaluating the pharmacodynamic action of NRF2 inducers, genetic and biochemical biomarkers should be evaluated. For example, the Nrf2 induction has to evaluate NQO1 transcript, epigenetic regulators like histone deacetylase, oxidative stress markers, pro‐ and anti‐inflammatory cytokines, NF‐κB signaling molecules and many other indicators of a proper functioning of induced Nrf2. Moreover, the action of inducers should be closely followed in a dose–response manner and standardized methodologies should be used to validate the obtained results. In the testing workflow, another general issue is that animal models do not exactly reproduce human pathology. Therefore, animal models that best recapitulate human diseases in terms of Nrf2‐related pathogenic mechanisms are to be obtained in the future. In these preclinical studies, new Nrf2 modulators can be tested. It is well known that noncommunicable diseases are characterized by multiple pathways and high complexity. As Nrf2 is involved in various pathologies, its mechanism can be similar or different. Therefore, the Nrf2‐intimate molecular mechanisms that lead to a specific disease should be thoroughly analyzed to identify new Nrf2 modulators or even to repurpose existing drugs. Deciphering the molecular mechanisms of Nrf2 is also important for the evaluation of additional networks. These networks can be disturbed when triggering Nrf2, so any potential unwanted side effects of a drug can occur and hence should be foreseen. Another issue that requires attention is Nrf2's partner, Keap1, which should also be investigated in order to detect structural subtleties as well as high‐ and low‐affinity active sites that could hinder/modulate the Keap1 interaction with Nrf2. When using tentative drugs that aim at active residues, these drugs can also affect other proteins, hence the importance to thoroughly study the structural subtleties should be highlighted. Another issue that challenges the Nrf2 domain is that Nrf2 activators cannot cross the blood–brain barrier in neurological disorders. 160 Furthermore, the positive results of Nrf2 targeted drugs obtained in preclinical settings still need to be validated in clinical trials.
Future studies will have to focus on the mechanisms that can be modulated at the mitochondrial genes, mitochondrial biogenesis, and ATP content of brain and spinal cord tissues to find new therapeutic properties of drugs to be used in DN. All these studies can have NRF2 activators as a starting point to alleviate the progression of DN through NRF2‐mediated mechanisms.
Without being exhaustive, our paper focused on the recent published data regarding the contribution of NRF2 in the installment and progression of DN.
AUTHOR CONTRIBUTIONS
Monica Neagu, Carolina Constantin, Constantin Caruntu: Literature search, data collection, original draft preparation; Mihaela Surcel, Adriana Munteanu, and Monica Neagu: Writing—review and editing. Monica Neagu, Carolina Constantin, Constantin Caruntu, Cristian Scheau, and Ilinca Savulescu‐Fiedler: conceptualization, methodology, supervision, project administration, writing—review and editing. All authors have read and agreed to the published version of the manuscript.
FUNDING INFORMATION
The work and open access fee was supported by a grant of the Romanian Ministry of Research, Innovation and Digitization, CCCDI‐UEFISCDI, project number [PN‐III‐P2‐2.1‐PED‐2021‐2243 and 31PFE/30.12.2021], within PNCDI III.
DISCLOSURE
The authors declare no conflict of interest.
INSTITUTIONAL REVIEW BOARD STATEMENT
Considering that only previously published papers were used for the systematic review, there are no ethical concerns with this study. The institutional review board confirmed that no ethical approval was needed.
INFORMED CONSENT STATEMENT
Not applicable.
Neagu M, Constantin C, Surcel M, et al. Diabetic neuropathy: A NRF2 disease? Journal of Diabetes. 2024;16(9):e13524. doi: 10.1111/1753-0407.13524
DATA AVAILABILITY STATEMENT
The data presented in this study are available on request from the corresponding author.
REFERENCES
- 1. Jaramillo MC, Zhang DD. The emerging role of the NRF2‐Keap1 signaling pathway in cancer. Genes Dev. 2013;27:2179‐2191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Taguchi K, Yamamoto M. The KEAP1‐NRF2 system in cancer. Front Oncol. 2017;7:85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Canning P, Sorrell FJ, Bullock AN. Structural basis of Keap1 interactions with NRF2. Free Radic Biol Med. 2015;88:101‐107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. McMahon M, Thomas N, Itoh K, Yamamoto M, Hayes JD. Redox‐regulated turnover of NRF2 is determined by at least two separate protein domains, the redox‐sensitive Neh2 degron and the redox‐insensitive Neh6 degron. J Biol Chem. 2004;279:31556‐31567. [DOI] [PubMed] [Google Scholar]
- 5. Wang H, Liu K, Geng M, et al. RXRalpha inhibits the NRF2‐ARE signaling pathway through a direct interaction with the Neh7 domain of NRF2. Cancer Res. 2013;73:3097‐3108. [DOI] [PubMed] [Google Scholar]
- 6. He F, Ru X, Wen T. NRF2, a transcription factor for stress response and beyond. Int J Mol Sci. 2020;21:4777. doi: 10.3390/ijms21134777 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Xiaoyu W, Jiajia W, Yang Y, Qihai G, Jianmei G. Activation of Nrf2 signaling: a key molecular mechanism of protection against cardiovascular diseases by natural products. Front Pharmacol. 2022;13:1‐19. doi: 10.3389/fphar.2022.1057918 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. He WJ, Lv CH, Chen Z, et al. The regulatory effect of phytochemicals on chronic diseases by targeting Nrf2‐ARE signaling pathway. Antioxidants (Basel). 2023;12(2):236. doi: 10.3390/antiox12020236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Pouremamali F, Pouremamali A, Dadashpour M, Soozangar N, Jeddi F. An update of Nrf2 activators and inhibitors in cancer prevention/promotion. Cell Commun Signal. 2022;20(1):100. doi: 10.1186/s12964-022-00906-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Wakabayashi N, Itoh K, Wakabayashi J, et al. Keap1‐null mutation leads to postnatal lethality due to constitutive NRF2 activation. Nat Genet. 2003;35:238‐245. [DOI] [PubMed] [Google Scholar]
- 11. Wu KC, Cui JY, Klaassen CD. Beneficial role of NRF2 in regulating NADPH generation and consumption. Toxicol Sci. 2011;123:590‐600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. He F, Antonucci L, Yamachika S, et al. NRF2 activates growth factor genes and downstream AKT signaling to induce mouse and human hepatomegaly. J Hepatol. 2020;72(6):1182‐1195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. International Diabetes Federation . IDF Diabetes Atlas. 8th edition: key messages ed. International Diabetes Federation; 2019. Available from IDF https://diabetesatlas.org/key-messages.html [Google Scholar]
- 14. World Health Organization. Diabetes WHO 2018. Available from https://www.who.int/news-room/fact-sheets/detail/diabetes. [Google Scholar]
- 15. Wang L, Gao P, Zhang M, et al. Prevalence and ethnic pattern of diabetes and prediabetes in China in 2013. JAMA. 2017;317(24):2515‐2523. doi: 10.1001/jama.2017.7596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Anjana RM, Deepa M, Pradeepa R, et al. Prevalence of diabetes and prediabetes in 15 states of India: results from the ICMR‐INDIAB population‐based cross‐sectional study. Lancet Diabetes Endocrinol. 2017;5(8):585‐596. doi: 10.1016/S2213-8587(17)30174-2 [DOI] [PubMed] [Google Scholar]
- 17. Centers for Disease Control and Prevention . Prediabetes: your chance to prevent type 2 diabetes CDC. https://www.cdc.gov/diabetes/basics/prediabetes.html (updated December 30, 2022)
- 18. Schlesinger S, Neuenschwander M, Barbaresko J, et al. Prediabetes and risk of mortality, diabetes‐related complications and comorbidities: umbrella review of meta‐analyses of prospective studies. Diabetologia. 2022;65(2):275‐285. doi: 10.1007/s00125-021-05592-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Callaghan BC, Price RS, Chen KS, Feldman EL. The importance of rare subtypes in diagnosis and treatment of peripheral neuropathy: a review. JAMA Neurol. 2015;72:1510‐1518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Căruntu C, Negrei C, Boda D, Constantin C, Căruntu A, Neagu M. Biotechnological advances for diagnosis of peripheral diabetic neuropathy. Roum Biotech Lett. 2014;19(6):9846‐9858. [Google Scholar]
- 21. Sameni H, Panahi M. The effect of co‐administration of 4‐methylcatechol and progesterone on sciatic nerve function and neurohistological alterations in streptozotocin‐induced diabetic neuropathy in rats. Cell Journal (Yakhteh). 2011;13(1):31. [PMC free article] [PubMed] [Google Scholar]
- 22. Selvarajah D, Wilkinson ID, Davies J, Gandhi R, Tesfaye S. Central nervous system involvement in diabetic neuropathy. Curr Diab Rep. 2011;11(4):310‐322. doi: 10.1007/s11892-011-0205-z [DOI] [PubMed] [Google Scholar]
- 23. Poulsen KM, Pachana NA, McDermott BM. Health professionals' detection of depression and anxiety in their patients with diabetes: the influence of patient, illness and psychological factors. J Health Psychol. 2016;21(8):1566‐1575. [DOI] [PubMed] [Google Scholar]
- 24. Kobylinska L, Ghita MA, Caruntu C, et al. Preliminary insights in oxytocin association with the onset of diabetic neuropathy. Acta Endocrinol (Buchar). 2017;13(2):249‐253. doi: 10.4183/aeb.2017.249 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. King MR, Anderson NJ, Liu C, et al. Activation of the insulin‐signaling pathway in sciatic nerve and hippocampus of type 1 diabetic rats. Neuroscience. 2015;303:220‐228. doi: 10.1016/j.neuroscience.2015.06.060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Kawamura T, Umemura T, Hotta H. Cognitive impairment in diabetic patients: can diabetic control prevent cognitive decline? J Diabetes Investig. 2012;3(5):413‐423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Wątroba M, Grabowska AD, Szukiewicz D. Effects of diabetes mellitus‐related Dysglycemia on the functions of blood‐brain barrier and the risk of dementia. Int J Mol Sci. 2023;24(12):10069. doi: 10.3390/ijms241210069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Henrich‐Noack P, Nikitovic D, Neagu M, et al. The blood‐brain barrier and beyond: Nano‐based neuropharmacology and the role of extracellular matrix. Nanomedicine. 2019;17:359‐379. doi: 10.1016/j.nano.2019.01.016 [DOI] [PubMed] [Google Scholar]
- 29. Zhang T, Wu J, Yao X, et al. The aldose reductase inhibitor Epalrestat maintains blood‐brain barrier integrity by enhancing endothelial cell function during cerebral ischemia. Mol Neurobiol. 2023;60(7):3741‐3757. doi: 10.1007/s12035-023-03304-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Rovira‐Llopis S, Bañuls C, Diaz‐Morales N, Hernandez Mijares A, Rocha M, Victor VM. Mitochondrial dynamics in type 2 diabetes: pathophysiological implications. Redox Biol. 2017;11:637‐645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Yagihashi S, Mizukami H, Sugimoto K. Mechanism of diabetic neuropathy: where are we now and where to go? J Diabetes Investig. 2011;2(1):18‐32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Edwards JL, Quattrini A, Lentz SI, et al. Diabetes regulates mitochondrial biogenesis and fission in mouse neurons. Diabetologia. 2010;53:160‐169. doi: 10.1007/s00125-009-1553-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Yang YH, Wang Z, Zheng J, Wang R. Protective effects of gallic acid against spinal cord injury‐induced oxidative stress. Mol Med Rep. 2015;12(2):3017‐3024. doi: 10.3892/mmr.2015.3738 [DOI] [PubMed] [Google Scholar]
- 34. Sanaye MM, Kavishwar SA. Diabetic neuropathy: review on molecular mechanisms. Curr Mol Med. 2023;23(2):97‐110. doi: 10.2174/1566524021666210816093111 [DOI] [PubMed] [Google Scholar]
- 35. Baum P, Toyka KV, Blüher M, Kosacka J, Nowicki M. Inflammatory mechanisms in the pathophysiology of diabetic peripheral neuropathy (DN)‐new aspects. Int J Mol Sci. 2021;22(19):10835. doi: 10.3390/ijms221910835 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Eid SA, Rumora AE, Beirowski B, et al. New perspectives in diabetic neuropathy. Neuron. 2023;111(17):2623‐2641. doi: 10.1016/j.neuron.2023.05.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Boda D, Neagu M, Constantin C, et al. HPV strain distribution in patients with genital warts in a female population sample. Oncol Lett. 2016;12(3):1779‐1782. doi: 10.3892/ol.2016.4903 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Li L, Iwamoto S, Cha J, Falanga V. Diabetic foot ulcer calluses show histological similarities to HPV infection without evidence of HPV involvement by nested PCR. Int J Low Extrem Wounds. 2010;9(2):84‐89. doi: 10.1177/1534734610371599 [DOI] [PubMed] [Google Scholar]
- 39. Chandrasekaran K, Zilliox LA, James W, Russell JW. Chapter 10 Diabetic Neuropathy – Research, In Microvascular Disease in Diabetes, Book Editor(s): Francesco Tecilazich MD, PhD. 13 March 2020 Online ISBN:9781119309642. doi: 10.1002/9781119309642 © 2020 John Wiley & Sons, Inc. [DOI]
- 40. Kumar A, Mittal R. NRF2: a potential therapeutic target for diabetic neuropathy. Inflammopharmacol. 2017;25:393‐402. doi: 10.1007/s10787-017-0339-y [DOI] [PubMed] [Google Scholar]
- 41. Dinescu S, Ignat S, Lazar A, Constantin C, Neagu M, Costache M. Epitranscriptomic signatures in lncRNAs and their possible roles in cancer. Genes. 2019;10(52):1‐27. doi: 10.3390/genes10010052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Neagu M, Constantin C, Cretoiu SM, Zurac S. miRNAs in the diagnosis and prognosis of skin cancer. Front Cell Dev Biol. 2020;8:71. doi: 10.3389/fcell.2020.00071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Kalis M, Bolmeson C, Esguerra JL, et al. Beta‐cell specific deletion of Dicer1 leads to defective insulin secretion and diabetes mellitus. PloS One. 2011;6(12):e29166. doi: 10.1371/journal.pone.0029166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Gada Y, Pandey A, Jadhav N, Ajgaonkar S, Mehta D, Nair S. New vistas in microRNA regulatory Interactome in neuropathic pain. Front Pharmacol. 2022;12:778014. doi: 10.3389/fphar.2021.778014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Gandhi M, Nair S. New vistas in malignant mesothelioma: MicroRNA architecture and NRF2/MAPK signal transduction. Life Sci. 2020;257:118123. doi: 10.1016/j.lfs.2020.118123 [DOI] [PubMed] [Google Scholar]
- 46. Pandey A, Ajgaonkar S, Jadhav N, et al. Current insights into miRNA and lncRNA dysregulation in diabetes: signal transduction, clinical trials and biomarker discovery. Pharmaceuticals (Basel). 2022;15(10):1269. doi: 10.3390/ph15101269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Li YB, Wu Q, Liu J, Fan YZ, Yu KF, Cai Y. miR‐199a‐3p is involved in the pathogenesis and progression of diabetic neuropathy through downregulation of SerpinE2. Mol Med Rep. 2017;16(3):2417‐2424. doi: 10.3892/mmr.2017.6874 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Simeoli R, Fierabracci A. Insights into the role of MicroRNAs in the onset and development of diabetic neuropathy. Int J Mol Sci. 2019;20(18):4627. doi: 10.3390/ijms20184627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Ashjari D, Karamali N, Rajabinejad M, et al. The axis of long non‐coding RNA MALAT1/miR‐1‐3p/CXCR4 is dysregulated in patients with diabetic neuropathy. Heliyon. 2022;8(3):e09178. doi: 10.1016/j.heliyon.2022.e09178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Cai LJ, Tu L, Huang XM, et al. LncRNA MALAT1 facilitates inflammasome activation via epigenetic suppression of Nrf2 in Parkinson's disease. Mol Brain. 2020;13(1):130. doi: 10.1186/s13041-020-00656-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Wang GF, Xu N. Expression and clinical significance of MicroRNA 146a in peripheral blood mononuclear cells from type 2 diabetic neuropathy patients. Int J Clin Exp Med. 2018;11:7165‐7173. [Google Scholar]
- 52. Chanda D, Ray S, Chakraborti D, Sen S, Mitra A. Interleukin‐6 levels in patients with diabetic polyneuropathy. Cureus. 2022;14(2):e21952. doi: 10.7759/cureus.21952 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Camiña N, Penning TM. Genetic and epigenetic regulation of the NRF2‐KEAP1 pathway in human lung cancer. Br J Cancer. 2022;126(9):1244‐1252. doi: 10.1038/s41416-021-01642-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Yin Y, Liu H, Xu J, et al. miR‐144‐3p regulates the resistance of lung cancer to cisplatin by targeting Nrf2. Oncol Rep. 2018;40:3479‐3488. [DOI] [PubMed] [Google Scholar]
- 55. Fabrizio FP, Sparaneo A, Castellana S, et al. 17P – REDOXI‐miRNA of Keap1/Nrf2 axis in lung tumors. Ann Oncol. 2019;30(2):ii5. doi: 10.1093/annonc/mdz072.015 [DOI] [Google Scholar]
- 56. Biswas S, Thomas AA, Chen S, et al. MALAT1: an epigenetic regulator of inflammation in diabetic retinopathy. Sci Rep. 2018;8:6526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Martens CR, Bansal SS, Accornero F. Cardiovascular inflammation: RNA takes the lead. J Mol Cell Cardiol. 2019;129:247‐256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Radhakrishnan R, Kowluru RA. Long noncoding RNA MALAT1 and regulation of the antioxidant defense system in diabetic retinopathy. Diabetes. 2021;70(1):227‐239. doi: 10.2337/db20-0375 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Gordon AD, Biswas S, Feng B, Chakrabarti S. MALAT1: a regulator of inflammatory cytokines in diabetic complications. Endocrinol Diabetes Metab. 2018;1(2):e00010. doi: 10.1002/edm2.10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Ahmadi A, Kaviani S, Yaghmaie M, et al. Altered expression of MALAT1 lncRNA in chronic lymphocytic leukemia patients, correlation with cytogenetic findings. Blood Res. 2018;53:320‐324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Zhang YL, Hu HY, You ZP, Li BY, Shi K. Targeting long non‐coding RNA MALAT1 alleviates retinal neurodegeneration in diabetic mice. Int J Ophthalmol. 2020;13:213‐219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Abdulle LE, Hao JL, Pant OP, et al. MALAT1 as a diagnostic and therapeutic target in diabetes‐related complications: a promising Long‐noncoding RNA. Int J Med Sci. 2019;16(4):548‐555. doi: 10.7150/ijms.30097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Moi P, Chan K, Asunis I, Cao A, Kan YW. Isolation of NF‐E2‐related factor 2 (Nrf2), a NF‐E2‐like basic leucine zipper transcriptional activator that binds to the tandem NF‐E2/AP1 repeat of the beta‐globin locus control region. Proc Natl Acad Sci U S A. 1994;91(21):9926‐9930. doi: 10.1073/pnas.91.21.9926 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Kopacz A, Rojo AI, Patibandla C, et al. Overlooked and valuable facts to know in the NRF2/KEAP1 field. Free Radic Biol Med. 2022;192:37‐49. doi: 10.1016/j.freeradbiomed.2022.08.044 [DOI] [PubMed] [Google Scholar]
- 65. Ion A, Popa IM, Papagheorghe LML, et al. Proteomic approaches to biomarker discovery in cutaneous T‐cell lymphoma. Disease Markers. 2016;2016:1‐8. | Article ID 9602472. doi: 10.1155/2016/9602472 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Heurtaux T, Bouvier DS, Benani A, et al. Normal and pathological NRF2 Signalling in the central nervous system. Antioxidants (Basel). 2022;11(8):1426. doi: 10.3390/antiox11081426 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Hayes JD, Dinkova‐Kostova AT. The NRF2 regulatory network provides an interface between redox and intermediary metabolism. Trends Biochem Sci. 2014;39:199‐218. [DOI] [PubMed] [Google Scholar]
- 68. Baird L, Lleres D, Swift S, Dinkova‐Kostova AT. Regulatory flexibility in the NRF2‐mediated stress response is conferred by conformational cycling of the Keap1‐NRF2 protein complex. Proc Natl Acad Sci U S A. 2013;110:15259‐15264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Chen CT, Shih YR, Kuo TK, Lee OK, Wei YH. Coordinated changes of mitochondrial biogenesis and antioxidant enzymes during osteogenic differentiation of human mesenchymal stem cells. Stem Cells. 2008;26:960‐968. [DOI] [PubMed] [Google Scholar]
- 70. Panieri E, Pinho SA, Afonso GJM, Oliveira PJ, Cunha‐Oliveira T, Saso L. NRF2 and mitochondrial function in cancer and cancer stem cells. Cell. 2022;11(15):2401. doi: 10.3390/cells11152401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Scheau C, Draghici C, Ilie MA, et al. Neuroendocrine factors in melanoma pathogenesis. Cancers (Basel). 2021;13(9):2277. doi: 10.3390/cancers13092277 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Diaconeasa A, Boda D, Neagu M, et al. The role of confocal microscopy in the dermato‐oncology practice. J Med Life. 2011;4(1):63‐74. [PMC free article] [PubMed] [Google Scholar]
- 73. He F, Antonucci L, Karin M. NRF2 as a regulator of cell metabolism and inflammation in cancer. Carcinogenesis. 2020;41(4):405‐416. doi: 10.1093/carcin/bgaa039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Chorley BN, Campbell MR, Wang X, et al. Identification of novel NRF2‐regulated genes by ChIP‐Seq: influence on retinoid X receptor alpha. Nucl Acids Res. 2012;40(15):7416‐7429. doi: 10.1093/nar/gks409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Fu J, Xiong Z, Huang C, et al. Hyperactivity of the transcription factor Nrf2 causes metabolic reprogramming in mouse esophagus. J Biol Chem. 2019;294(1):327‐340. doi: 10.1074/jbc.RA118.005963 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Zhang Y, Chen K, Sloan SA, et al. An RNA‐sequencing transcriptome and splicing database of glia, neurons, and vascular cells of the cerebral cortex. J Neurosci. 2014;34(36):11929‐11947. doi: 10.1523/JNEUROSCI.1860-14.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Cuadrado A, Rojo AI, Wells G, et al. Therapeutic targeting of the NRF2 and KEAP1 partnership in chronic diseases. Nat Rev Drug Discov. 2019;18(4):295‐317. doi: 10.1038/s41573-018-0008-x [DOI] [PubMed] [Google Scholar]
- 78. Uruno A, Furusawa Y, Yagishita Y, et al. The Keap1‐Nrf2 system prevents onset of diabetes mellitus. Mol Cell Biol. 2013;33(15):2996‐3010. doi: 10.1128/MCB.00225-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Uruno A, Yagishita Y, Katsuoka F, et al. Nrf2‐mediated regulation of skeletal muscle glycogen metabolism. Mol Cell Biol. 2016;36(11):1655‐1672. doi: 10.1128/MCB.01095-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Kerins MJ, Ooi A. The roles of NRF2 in modulating cellular iron homeostasis. Antioxid Redox Signal. 2018;29(17):1756‐1773. doi: 10.1089/ars.2017.7176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Simcox JA, McClain DA. Iron and diabetes risk. Cell Metab. 2013;17:329‐341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Elumalai S, Karunakaran U, Moon JS, Won KC. Ferroptosis signaling in pancreatic β‐cells: novel insights & therapeutic targeting. Int J Mol Sci. 2022;23(22):13679. doi: 10.3390/ijms232213679 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Dixon SJ, Lemberg KM, Lamprecht MR, et al. Ferroptosis: an iron‐dependent form of nonapoptotic cell death. Cell. 2012;149(5):1060‐1072. doi: 10.1016/j.cell.2012.03.042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Caruntu C, Boda D, Dumitrascu G, Constantin C, Neagu M. Proteomics focusing on immune markers in psoriatic arthritis. Biomark Med. 2015;9(6):513‐528. doi: 10.2217/BMM.14.76 [DOI] [PubMed] [Google Scholar]
- 85. Mohan S, Gupta D. Crosstalk of toll‐like receptors signaling and NRF2 pathway for regulation of inflammation. Biomed Pharmacother. 2018;108:1866‐1878. doi: 10.1016/j.biopha.2018.10.019 [DOI] [PubMed] [Google Scholar]
- 86. Srivastava S, Singh D, Patel S, Singh MR. Role of enzymatic free radical scavengers in management of oxidative stress in autoimmune disorders. Int J Biol Macromol. 2017;101:502‐517. doi: 10.1016/j.ijbiomac.2017.03.100 [DOI] [PubMed] [Google Scholar]
- 87. Bagger FO, Sasivarevic D, Sohi SH, et al. BloodSpot: a database of gene expression profiles and transcriptional programs for healthy and malignant haematopoiesis. Nucleic Acids Res. 2016;44(D1):D917‐D924. doi: 10.1093/nar/gkv1101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Kobayashi EH, Suzuki T, Funayama R, et al. Nrf2 suppresses macrophage inflammatory response by blocking proinflammatory cytokine transcription. Nat Commun. 2016;7:11624. doi: 10.1038/ncomms11624 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Bambouskova M, Gorvel L, Lampropoulou V, et al. Electrophilic properties of itaconate and derivatives regulate the IκBζ‐ATF3 inflammatory axis. Nature. 2018;556(7702):501‐504. doi: 10.1038/s41586-018-0052-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Suzuki T, Murakami S, Biswal SS, et al. Systemic activation of NRF2 alleviates lethal autoimmune inflammation in scurfy mice. Mol Cell Biol. 2017;37(15):e00063‐17. doi: 10.1128/MCB.00063-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Chaudhuri J, Bose N, Gong J, et al. A Caenorhabditis elegans model elucidates a conserved role for TRPA1‐Nrf signaling in reactive α‐Dicarbonyl detoxification. Curr Biol. 2016;26(22):3014‐3025. doi: 10.1016/j.cub.2016.09.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Logie E, Vanden BW. Tackling chronic inflammation with Withanolide phytochemicals‐a Withaferin a perspective. Antioxidants (Basel). 2020;9(11):1107. doi: 10.3390/antiox9111107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Srinivasulu C, Ramgopal M, Ramanjaneyulu G, Anuradha C, Suresh KC. Syringic acid (SA)–a review of its occurrence, biosynthesis, pharmacological and industrial importance. Biomed Pharmacother. 2018;108:547‐557. [DOI] [PubMed] [Google Scholar]
- 94. Zhao WC, Zhang B, Liao MJ, et al. Curcumin ameliorated diabetic neuropathy partially by inhibition of NADPH oxidase mediating oxidative stress in the spinal cord. Neurosci Lett. 2014;560:81‐85. doi: 10.1016/j.neulet.2013.12.019 [DOI] [PubMed] [Google Scholar]
- 95. Rashedinia M, Alimohammadi M, Shalfroushan N, et al. Neuroprotective effect of Syringic acid by modulation of oxidative stress and mitochondrial mass in diabetic rats. Biomed Res Int. 2020;2020:1‐12. doi: 10.1155/2020/8297984 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Duffy SL, Lagopoulos J, Hickie IB, et al. Glutathione relates to neuropsychological functioning in mild cognitive impairment. Alzheimers Dement. 2014;10(1):67‐75. doi: 10.1016/j.jalz.2013.01.005 [DOI] [PubMed] [Google Scholar]
- 97. Adabizadeh M, Mehri S, Rajabpour M, Abnous K, Rashedinia M, Hosseinzadeh H. The effects of crocin on spatial memory impairment induced by hyoscine: role of NMDA, AMPA, ERK, and CaMKII proteins in rat hippocampus. Iran J Basic Med Sci. 2019;22(6):601‐609. doi: 10.22038/ijbms.2019.30138.7266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Ogut E, Sekerci R, Akcay G, et al. Protective effects of syringic acid on neurobehavioral deficits and hippocampal tissue damages induced by sub‐chronic deltamethrin exposure. Neurotoxicol Teratol. 2019;76:106839. doi: 10.1016/j.ntt.2019.106839 [DOI] [PubMed] [Google Scholar]
- 99. Hasanein P, Mahtaj AK. Ameliorative effect of rosmarinic acid on scopolamine‐induced memory impairment in rats. Neurosci Lett. 2015;585:23‐27. doi: 10.1016/j.neulet.2014.11.027 [DOI] [PubMed] [Google Scholar]
- 100. Sharifi‐Rad J, Ezzat SM, El Bishbishy MH, et al. Rosmarinus plants: key farm concepts towards food applications. Phytother Res. 2020;34(7):1474‐1518. doi: 10.1002/ptr.6622 [DOI] [PubMed] [Google Scholar]
- 101. Fonteles AA, de Souza CM, de Sousa Neves JC, et al. Rosmarinic acid prevents against memory deficits in ischemic mice. Behav Brain Res. 2016;297:91‐103. doi: 10.1016/j.bbr.2015.09.029 [DOI] [PubMed] [Google Scholar]
- 102. Mansouri MT, Naghizadeh B, Ghorbanzadeh B, Farbood Y, Sarkaki A, Bavarsad K. Gallic acid prevents memory deficits and oxidative stress induced by intracerebroventricular injection of streptozotocin in rats. Pharmacol Biochem Behav. 2013;111:90‐96. doi: 10.1016/j.pbb.2013.09.002 [DOI] [PubMed] [Google Scholar]
- 103. Khodaei F, Rashedinia M, Heidari R, Rezaei M, Khoshnoud MJ. Ellagic acid improves muscle dysfunction in cuprizone‐induced demyelinated mice via mitochondrial Sirt3 regulation. Life Sci. 2019;237:116954. doi: 10.1016/j.lfs.2019.116954 [DOI] [PubMed] [Google Scholar]
- 104. Ademiluyi AO, Oboh G, Boligon AA, Athayde ML. Dietary supplementation with fermented legumes modulate hyperglycemia and acetylcholinesterase activities in Streptozotocin‐induced diabetes. Pathophysiology. 2015;22(4):195‐201. doi: 10.1016/j.pathophys.2015.08.003 [DOI] [PubMed] [Google Scholar]
- 105. Esmaeil Jamaat E, Kiasalari Z, Sanaeirad A, Roghani M. The effect of chlorogenic acid on learning and memory and acetylchoinesterase activity in rats with cognitive deficit induced by intracerebroventricular streptozotocin. J Basic Clin Pathophysiol. 2018;6(2):17‐22. [Google Scholar]
- 106. Rolo AP, Palmeira CM. Diabetes and mitochondrial function: role of hyperglycemia and oxidative stress. Toxicol Appl Pharmacol. 2006;212(2):167‐178. doi: 10.1016/j.taap.2006.01.003 [DOI] [PubMed] [Google Scholar]
- 107. Niknahad H, Jamshidzadeh A, Heidari R, Zarei M, Ommati MM. Ammonia‐induced mitochondrial dysfunction and energy metabolism disturbances in isolated brain and liver mitochondria, and the effect of taurine administration: relevance to hepatic encephalopathy treatment. Clin Exp Hepatol. 2017;3(3):141‐151. doi: 10.5114/ceh.2017.68833 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Szendroedi J, Phielix E, Roden M. The role of mitochondria in insulin resistance and type 2 diabetes mellitus. Nat Rev Endocrinol. 2011;8(2):92‐103. doi: 10.1038/nrendo.2011.138 [DOI] [PubMed] [Google Scholar]
- 109. Valenti D, De Rasmo D, Signorile A, et al. Epigallocatechin‐3‐gallate prevents oxidative phosphorylation deficit and promotes mitochondrial biogenesis in human cells from subjects with Down's syndrome. Biochim Biophys Acta. 2013;1832(4):542‐552. doi: 10.1016/j.bbadis.2012.12.011 [DOI] [PubMed] [Google Scholar]
- 110. Golpich M, Amini E, Mohamed Z, Azman Ali R, Mohamed Ibrahim N, Ahmadiani A. Mitochondrial dysfunction and biogenesis in neurodegenerative diseases: pathogenesis and treatment. CNS Neurosci Ther. 2017;23(1):5‐22. doi: 10.1111/cns.12655 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Kim JA, Wei Y, Sowers JR. Role of mitochondrial dysfunction in insulin resistance. Circ Res. 2008;102(4):401‐414. doi: 10.1161/CIRCRESAHA.107.165472 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Fernyhough P. Mitochondrial dysfunction in diabetic neuropathy: a series of unfortunate metabolic events. Curr Diab Rep. 2015;15(11):89. doi: 10.1007/s11892-015-0671-9 [DOI] [PubMed] [Google Scholar]
- 113. Patti ME, Butte AJ, Crunkhorn S, et al. Coordinated reduction of genes of oxidative metabolism in humans with insulin resistance and diabetes: potential role of PGC1 and NRF1. Proc Natl Acad Sci U S A. 2003;100(14):8466‐8471. doi: 10.1073/pnas.1032913100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Choi J, Chandrasekaran K, Inoue T, Muragundla A, Russell JW. PGC‐1α regulation of mitochondrial degeneration in experimental diabetic neuropathy. Neurobiol Dis. 2014;64:118‐130. doi: 10.1016/j.nbd.2014.01.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Shi X, Chen Y, Nadeem L, Xu G. Beneficial effect of TNF‐α inhibition on diabetic peripheral neuropathy. J Neuroinflammation. 2013;10:69. doi: 10.1186/1742-2094-10-69 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Cuevas A, Saavedra N, Salazar LA, Abdalla DS. Modulation of immune function by polyphenols: possible contribution of epigenetic factors. Nutrients. 2013;5(7):2314‐2332. doi: 10.3390/nu5072314 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. El‐Sawaf ES, Saleh S, Abdallah DM, Ahmed KA, El‐Abhar HS. Vitamin D and rosuvastatin obliterate peripheral neuropathy in a type‐2 diabetes model through modulating Notch1, Wnt‐10α, TGF‐β and NRF‐1 crosstalk. Life Sci. 2021;279:119697. doi: 10.1016/j.lfs.2021.119697 [DOI] [PubMed] [Google Scholar]
- 118. Mittal R, Kumar A, Singh DP, Bishnoi M, Nag TC. Ameliorative potential of rutin in combination with nimesulide in STZ model of diabetic neuropathy: targeting Nrf2/HO‐1/NF‐kB and COX signalling pathway. Inflammopharmacology. 2018;26(3):755‐768. doi: 10.1007/s10787-017-0413-5 [DOI] [PubMed] [Google Scholar]
- 119. Shi Y, Liang XC, Zhang H, Sun Q, Wu QL, Qu L. Combination of quercetin, cinnamaldehyde and hirudin protects rat dorsal root ganglion neurons against high glucose‐induced injury through Nrf‐2/HO‐1 activation and NF‐κB inhibition. Chin J Integr Med. 2017;23(9):663‐671. doi: 10.1007/s11655-017-2405-0 [DOI] [PubMed] [Google Scholar]
- 120. Shi Y, Liang XC, Zhang H, Wu QL, Qu L, Sun Q. Quercetin protects rat dorsal root ganglion neurons against high glucose‐induced injury in vitro through Nrf‐2/HO‐1 activation and NF‐κB inhibition. Acta Pharmacol Sin. 2013;34(9):1140‐1148. doi: 10.1038/aps.2013.59 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Liu Y, Zhang S, Xue J, et al. CGRP reduces apoptosis of DRG cells induced by high‐glucose oxidative stress injury through PI3K/AKT induction of Heme Oxygenase‐1 and Nrf‐2 expression. Oxid Med Cell Longev. 2019;2019:1‐9. doi: 10.1155/2019/2053149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Kensler TW, Wakabayashi N, Biswal S. Cell survival responses to environmental stresses via the Keap1‐Nrf2‐ARE pathway. Annu Rev Pharmacol Toxicol. 2007;47:89‐116. doi: 10.1146/annurev.pharmtox.46.120604.141046 [DOI] [PubMed] [Google Scholar]
- 123. Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circ Res. 2010;107:1058‐1070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Evans JL, Goldfifine ID, Maddux BA, Grodsky GM. Are oxidative stress activated signaling pathways mediators of insulin resistance and beta‐cell dysfunction? Diabetes. 2003;52:1‐8. [DOI] [PubMed] [Google Scholar]
- 125. Soares MA, Cohen OD, Low YC, et al. Restoration of Nrf2 signaling normalizes the regenerative niche. Diabetes. 2016;65(3):633‐646. doi: 10.2337/db15-0453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Rabbani PS, Soares MA, Hameedi SG, et al. Dysregulation of Nrf2/Keap1 redox pathway in diabetes affects multipotency of stromal cells. Diabetes. 2019;68(1):141‐155. doi: 10.2337/db18-0232 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Ding Y, Ding X, Zhang H, Li S, Yang P, Tan Q. Relevance of NLRP3 Inflammasome‐related pathways in the pathology of diabetic wound healing and possible therapeutic targets. Oxid Med Cell Longev. 2022;2022:1‐15. doi: 10.1155/2022/9687925 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Su J, Gao C, Xie L, et al. Astragaloside II ameliorated Podocyte injury and mitochondrial dysfunction in Streptozotocin‐induced diabetic rats. Front Pharmacol. 2021;12:638422. doi: 10.3389/fphar.2021.638422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Villarreal CF, Santos DS, Lauria PSS, et al. Bergenin reduces experimental painful diabetic neuropathy by restoring redox and immune homeostasis in the nervous system. Int J Mol Sci. 2020;21(14):4850. doi: 10.3390/ijms21144850 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. Chun YL, Park KH, Pallavi B, et al. Novel Cinnamaldehyde derivatives inhibit peripheral nerve degeneration by targeting Schwann cells. Antioxidants (Basel). 2022;11(10):1846. doi: 10.3390/antiox11101846 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Kim M, Park C, Jung J, Yeo SG. The histone deacetylase class I, II inhibitor trichostatin a delays peripheral neurodegeneration. J Mol Histol. 2019;50(2):167‐178. doi: 10.1007/s10735-019-09815-1 [DOI] [PubMed] [Google Scholar]
- 132. Li LH, Peng WN, Deng Y, Li JJ, Tian XR. Action of trichostatin a on Alzheimer's disease‐like pathological changes in SH‐SY5Y neuroblastoma cells. Neural Regen Res. 2020;15(2):293‐301. doi: 10.4103/1673-5374.265564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Liu W, Liang XC, Shi Y. Effects of Hirudin on high glucose‐induced oxidative stress and inflammatory pathway in rat dorsal root ganglion neurons. Chin J Integr Med. 2020;26(3):197‐204. doi: 10.1007/s11655-019-2712-8 [DOI] [PubMed] [Google Scholar]
- 134. Gao N, Ma B, Jia H, Hao C, Jin T, Liu X. Translocator protein alleviates allodynia and improves Schwann cell function against diabetic peripheral neuropathy via activation of the Nrf2‐dependent antioxidant system and promoting autophagy. Diabet Med. 2023;40(6):e15090. doi: 10.1111/dme.15090 [DOI] [PubMed] [Google Scholar]
- 135. Bachewal P, Gundu C, Yerra VG, Kalvala AK, Areti A, Kumar A. Morin exerts neuroprotection via attenuation of ROS induced oxidative damage and neuroinflammation in experimental diabetic neuropathy. Biofactors. 2018;44(2):109‐122. doi: 10.1002/biof.1397 [DOI] [PubMed] [Google Scholar]
- 136. Pozza G, Saibene V, Comi G, Canal N. The effect of ganglioside administration in human diabetic peripheral neuropathy. In: Rapport MM, Gorio A, eds. Gangliosides in Neurological and Neuromuscular Function, Development and Repair. Raven Press; 1981. [Google Scholar]
- 137. Coelho MA, Jeyaraman M, Jeyaraman N, et al. Application of Sygen® in diabetic peripheral neuropathies‐a review of biological interactions. Bioengineering (Basel). 2022;9(5):217. doi: 10.3390/bioengineering9050217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Wang X, Li B, Yu X, Zhou Y, Gao Y. The neuroprotective effect of GM‐1 ganglioside on the amyloid‐Beta‐induced oxidative stress in PC‐12 cells mediated by Nrf‐2/ARE signaling pathway. Neurochem Res. 2022;47(8):2405‐2415. doi: 10.1007/s11064-022-03635-8 [DOI] [PubMed] [Google Scholar]
- 139. Golbidi S, Badran M, Laher I. Diabetes and alpha lipoic acid. Front Pharmacol. 2011;17(2):69. doi: 10.3389/fphar.2011.00069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Kim M, Jung J, Jeong NY, Chung HJ. The natural plant flavonoid apigenin is a strong antioxidant that effectively delays peripheral neurodegenerative processes. Anat Sci Int. 2019;94(4):285‐294. doi: 10.1007/s12565-019-00486-2 [DOI] [PubMed] [Google Scholar]
- 141. Tang J, Raza A, Chen J, Xu H. A systematic review on the Sinomenine derivatives. Mini Rev Med Chem. 2018;18(11):906‐917. doi: 10.2174/1389557517666171123212557 [DOI] [PubMed] [Google Scholar]
- 142. Chen J, Guo P, Han M, Chen K, Qin J, Yang F. Cognitive protection of sinomenine in type 2 diabetes mellitus through regulating the EGF/Nrf2/HO‐1 signaling, the microbiota‐gut‐brain axis, and hippocampal neuron ferroptosis. Phytother Res. 2023;37:3323‐3341. doi: 10.1002/ptr.7807 [DOI] [PubMed] [Google Scholar]
- 143. Zhang Y, Xin Z, Dong B, Xue W. Combination of the NRF2 inhibitor and autophagy inhibitor significantly inhibited Tumorigenicity of castration‐resistant prostate cancer. Comput Math Methods Med. 2022;2022:1‐11. doi: 10.1155/2022/4182401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Rushworth SA, Zaitseva L, Murray MY, Shah NM, Bowles KM, MacEwan DJ. The high Nrf2 expression in human acute myeloid leukemia is driven by NF‐κB and underlies its chemo‐resistance. Blood. 2012;120(26):5188‐5198. doi: 10.1182/blood-2012-04-422121 [DOI] [PubMed] [Google Scholar]
- 145. Saw CL, Yang AY, Huang MT, et al. Nrf2 null enhances UVB‐induced skin inflammation and extracellular matrix damages. Cell Biosci. 2014;4:39. doi: 10.1186/2045-3701-4-39 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146. Dai Y, Zhang H, Zhang J, Yan M. Isoquercetin attenuates oxidative stress and neuronal apoptosis after ischemia/reperfusion injury via Nrf2‐mediated inhibition of the NOX4/ROS/NF‐κB pathway. Chem Biol Interact. 2018;284:32‐40. doi: 10.1016/j.cbi.2018.02.017 [DOI] [PubMed] [Google Scholar]
- 147. Mao L, Wang H, Qiao L, Wang X. Disruption of Nrf2 enhances the upregulation of nuclear factor‐kappaB activity, tumor necrosis factor‐α, and matrix metalloproteinase‐9 after spinal cord injury in mice. Mediators Inflamm. 2010;2010:238321. doi: 10.1155/2010/238321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148. Quinti L, Dayalan Naidu S, Träger U, et al. KEAP1‐modifying small molecule reveals muted NRF2 signaling responses in neural stem cells from Huntington's disease patients. Proc Natl Acad Sci U S A. 2017;114(23):E4676‐E4685. doi: 10.1073/pnas.1614943114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149. Quinti L, Casale M, Moniot S, et al. SIRT2‐ and NRF2‐targeting Thiazole‐containing compound with therapeutic activity in Huntington's disease models. Cell Chem Biol. 2016;23(7):849‐861. doi: 10.1016/j.chembiol.2016.05.015 [DOI] [PubMed] [Google Scholar]
- 150. Linker RA, Lee DH, Ryan S, et al. Fumaric acid esters exert neuroprotective effects in neuroinflammation via activation of the Nrf2 antioxidant pathway. Brain. 2011;134(Pt 3):678‐692. doi: 10.1093/brain/awq386 [DOI] [PubMed] [Google Scholar]
- 151. Gold R, Linker RA, Stangel M. Fumaric acid and its esters: an emerging treatment for multiple sclerosis with antioxidative mechanism of action. Clin Immunol. 2012;142(1):44‐48. doi: 10.1016/j.clim.2011.02.017 [DOI] [PubMed] [Google Scholar]
- 152. Choi KM, Kashyap PC, Dutta N, et al. CD206‐positive M2 macrophages that express heme oxygenase‐1 protect against diabetic gastroparesis in mice. Gastroenterology. 2010;138:2399‐2409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153. Long M, Vega MR, Wen Q, et al. An essential role of NRF2 in diabetic wound healing. Diabetes. 2016;65:780‐793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154. Sun W, Liu X, Zhang H, et al. Epigallocatechin gallate upregulates NRF2 to prevent diabetic nephropathy via disabling KEAP1. Free Radic Biol Med. 2017;108:840‐857. doi: 10.1016/j.freeradbiomed.2017.04.365 [DOI] [PubMed] [Google Scholar]
- 155. Ghasemzadeh Rahbardar M, Amin B, Mehri S, Mirnajafi‐Zadeh SJ, Hosseinzadeh H. Anti‐inflammatory effects of ethanolic extract of Rosmarinus officinalis L. and rosmarinic acid in a rat model of neuropathic pain. Biomed Pharmacother. 2017;86:441‐449. doi: 10.1016/j.biopha.2016.12.049 [DOI] [PubMed] [Google Scholar]
- 156. Gallorini M, Carradori S, Panieri E, Sova M, Saso L. Modulation of NRF2: biological dualism in cancer, targets and possible therapeutic applications. Antioxid Redox Signal. 2023. doi: 10.1089/ars.2022.0213 [DOI] [PubMed] [Google Scholar]
- 157. Wu W, Hendrix A, Nair S, Cui T. Nrf2‐mediated dichotomy in the vascular system: mechanistic and therapeutic perspective. Cell. 2022;11(19):3042. doi: 10.3390/cells11193042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158. Deng Q, Yang S, Sun L, et al. A detrimental role of NLRP6 in host iron metabolism during salmonella infection. Redox Biol. 2022;49:102217. doi: 10.1016/j.redox.2021.102217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159. Kitamura H, Takeda H, Motohashi H. Genetic, metabolic and immunological features of cancers with NRF2 addiction. FEBS Lett. 2022;596(16):1981‐1993. doi: 10.1002/1873-3468.14458 [DOI] [PubMed] [Google Scholar]
- 160. Saha S, Buttari B, Panieri E, Profumo E, Saso L. An overview of Nrf2 signaling pathway and its role in inflammation. Molecules. 2020;25(22):5474. doi: 10.3390/molecules25225474 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.


