Abstract
Background
Osteosarcoma is the most common malignant bone tumor in children and adolescents. Conventional chemotherapy remains unsatisfactory due to drug toxicity and resistance issues. Therefore, there is an urgent need to develop more effective treatments for advanced osteosarcoma. In the current study, we focused on evaluating the anticancer efficacy of avermectin B1, a novel avermectin analog, against osteosarcoma cells.
Methods
The half-inhibitory concentration of avermectin B1 was calculated in three osteosarcoma cell lines. Then, functional experiments were conducted to evaluate the effects of avermectin B1 on cell proliferation, the cell cycle, apoptosis and autophagy. Moreover, the AMPK/ULK1 signaling pathway was detected by Western blot assay. Finally, the in vivo effect of avermectin B1 on tumor growth and metastasis was investigated using the xenograft mouse model. To examine the role of the AMPK/ULK1 pathway, an AMPK-specific inhibitor (dorsomorphin) was used in combination with avermectin B1.
Results
Avermectin B1 inhibited the proliferation of osteosarcoma cells in a dose-dependent manner based on CCK8 and colony formation assays. Then, it was found to inhibit migration and invasion by wound healing assay and cell migration and invasion assay. In addition, avermectin B1 induced osteosarcoma cell apoptosis and autophagy. In vivo, avermectin B1 effectively inhibited osteosarcoma cell growth and pulmonary metastasis. Mechanistically, avermectin B1 activated the AMPK/ULK1 pathway to exert antitumor activity in vitro and in vivo. Dorsomorphin significantly attenuated the Avermectin B1-induced antitumor activities.
Conclusion
Our study suggests that avermectin B1 is a potential agent to treat osteosarcoma cells through the AMPK/ULK1 signaling pathway.
Keywords: Osteosarcoma, Avermectin B1, Autophagy, Apoptosis, AMPK/ULK1 pathway
Introduction
Osteosarcoma is the most common primary malignant tumor of bone, with an incidence of approximately 3 in 1,000,000 [1]. Osteosarcoma occurs in the metaphysis of long bones and is one of the leading causes of cancer-related death in adolescents [1, 2]. With the development of neoadjuvant chemotherapy, the surgical treatment of osteosarcoma has changed from amputation to limb salvage surgery [3]. The 5-year survival rate of osteosarcoma patients has also increased from 20–60-70%, but this survival rate has not improved significantly in the past half century [1, 4]. Early metastasis is one of the characteristics of osteosarcoma. Metastasis can be detected in 20% of patients when clinically diagnosed, and approximately 50% of patients have distant metastases during treatment, of which nearly 70% of metastases occur in the lungs [5, 6]. Moreover, some patients are not sensitive to chemotherapy drugs or have difficulty tolerating the adverse reactions caused by chemotherapy and are prone to distant metastasis and local recurrence, resulting in poor prognosis and a greatly increased amputation rate [7, 8]. Therefore, it is urgent to find new and safe therapeutic drugs for osteosarcoma treatment.
Avermectins (AVMs), FDA-approved antiparasitic drugs, are macrolide antibiotics produced by Streptomyces avermitilis and include avermectin B1 (abamectin) and its synthetic derivative ivermectin [9]. In addition to being widely used as antiparasitic drugs in humans and animals, AVMs can also inhibit the multidrug resistance of tumor cells [10]. In a study on the effects of avermectin C and avermectin B1 on the growth of ascites and solid tumors in mice, Victor A. Drinyaeva [10] found for the first time that AVMs had relatively obvious antitumor effects, and nontoxic doses of AVMs can inhibit tumor growth. Moreover, AVMs and their nontoxic derivatives can also be used as multidrug resistance inhibitors in tumor therapy [11]. In addition, it has been reported that one macrolide compound, abamectin, can inhibit the expression of cytoskeletal proteins in mouse neuroblastoma N2a cells [12]. Avermectin macrocyclic lactone derivatives selamectin and ivermectin could inhibit the migration of breast cancer cells in vitro, while selamectin could inhibit lung metastasis of mouse breast cancer cells in vivo [13]. This research found that ivermectin could inhibit tumor metastasis by the Wnt signaling pathway. Despite the large number of related studies, there are still some key issues of the ability of AVMs to inhibit tumor cell growth that have not been resolved, which is important for the development of new anticancer drugs with increased efficacy and reduced toxic side effects. The role of abamectin B1 in bone tumors, especially osteosarcoma, has not been reported.
Autophagy is an intracellular self-degradative process that plays a key role in regulating cell growth and metabolism [14, 15]. Studies have demonstrated that autophagy is used by tumor cells to repress the initial steps in carcinogenesis and plays an important role in cancer [16, 17]. Wang [18] et al. reported that ivermectin induced PAK1-mediated cytostatic autophagy in breast cancer. They indicated that ivermectin inhibited the Akt/mTOR signaling pathway by increasing ubiquitination-mediated PAK1 degradation, resulting in increased autophagic flux. In addition, other studies have indicated that ivermectin could inhibit the growth of breast cancer and NF2 tumors by inducing cytostatic autophagy [19, 20]. Based on these previous studies on the antitumor effect of AVMs, we reasonably speculated that avermectin B1 had better antitumor activity and that avermectin B1 inhibited the progression of osteosarcoma by inducing autophagy.
Here, our study demonstrates that avermectin B1 exhibits antitumor activity against osteosarcoma and identifies the internal connection between avermectin B1 and autophagy in osteosarcoma.
Materials and methods
Reagents and antibodies
Avermectin B1 (S4999) and dorsomorphin (S7840) were purchased from Selleck (Shanghai, China). Anti-LC3B (3868T), Caspase-9 (9502T), Atg7 (8558T), mTOR (2983 S), p-mTOR (5536 S), ULK1 (8054T), AMPK (5831T), and p-AMPK (2535T) antibodies were purchased from Cell Signaling Technology (CST, Shanghai, China). Anti-β-actin (HRP-60,008), Bax (50599-2-Ig), BAD (10435-1-AP), CDK4 (66950-1-Ig), CDK6 (14052-1-AP), CyclinD1 (60186-1-Ig), CyclinD2 (67048-1-Ig), Atg13 (66708-1-Ig), Atg14 (19491-1-AP) and Beclin1 (66665-1-Ig) antibodies were purchased from Proteintech (Wuhan, Hubei, China).
Cell lines and treatment
The three osteosarcoma cell lines (MNNG, MG63, U2OS) were obtained from the American Type Culture Collection (ATCC) (Manassas, VA, USA). The culture medium for the MNNG and MG63 cell lines was Dulbecco’s modified Eagle’s medium (DMEM), and the U2OS cell line was RPMI-1640 medium. Each medium contained 10% fetal bovine serum (FBS), penicillin (100 U/ml) and streptavidin (100 mg/ml). The cell culture environment was 37 °C in an incubator with 5% CO2.
Determination of IC50 of avermectin B1
Osteosarcoma cells (5000 cells/well) were plated into 96-well plates and incubated with various concentrations of Avermectin B1 (ranging from 0 to 40 mg/mL with a gradient of 5) for 48 h. Then, a Cell Counting Kit (CCK-8) was used to analyze the half-maximal inhibitory concentration (IC50) of avermectin B1.
Proliferation assay
OS cells were plated into 96-well plates (5000 cells/well) and incubated overnight for adherence. Next, the cells were treated with the IC50 of avermectin B1, and the cell proliferation assay was evaluated by CCK-8 assay for 5 days.
Clone formation assay
Cells in the logarithmic growth phase were treated with avermectin B1 for 48 h. Cells were digested with trypsin and plated into 6-well plates (1 × 103 cells/well). After a 10-day incubation, the cells were fixed in methanol at room temperature and stained with 0.1% crystal violet for 30 min. Then, the number of colonies was counted.
Cell migration and invasion assay
Cells treated with IC25 and IC50 concentrations and control cells for 48 h were washed to remove the avermectin B1, and then seeded in the Transwell chambers (Corning) with the same cell density of 2 × 105 cells/ml using new fresh serum-free medium. The number of viable cells was standardized using the Countess 3/3FL Automated Cell Counter (Invitrogen, USA). 0.2mL (empty penetration to assess cell migration) or 0.1mL (gel penetration) was added slowly and uniformly to the Transwell chambers. The lower chamber was filled with 800 µL of complete medium containing 10% fetal bovine serum. The 24-well plates containing Transwell chambers were incubated for 18–36 h. The time of removal of the chambers was adjusted according to the characteristics of the cells. The cells were fixed in methanol, stained with crystal violet and photographed under a microscope. Five cells were counted randomly in each chamber and the average value was taken. Three replicate wells were set up for each cell type to be tested.
Wound healing assay
OS cells were plated into a 6-well plate to achieve a density of 90%. Then, the cells were scraped/wounded using a yellow tip, washed 3 times with PBS, and photographed under the microscope for 0 h. After treatment with culture medium (1% FBS) containing different concentrations of avermectin B1, the cells were photographed at 18 h and 36 h to compare the migration capacity of each group of cells. Bioassays were replicated three times.
Flow cytometry assay
The cells were inoculated in six-well plates and treated with IC25 or IC50 drug concentrations of avermectin B1. After incubation for 48 h, the wells were digested with trypsin, and the cell suspension was centrifuged for 5 min at 1000 rpm for collection. Then, the cells were stained with Annexin V-FITC and propidium iodide according to the manufacturer’s guidelines. After staining, the cells were read on a flow cytometer (BD Biosciences, Franklin Lakes, NJ, USA). For the cell cycle assay, cells were treated as mentioned above, stained according to the manufacturer’s guidelines (Cell Cycle and Apoptosis Analysis Kit; Beyotime, China) and analyzed by flow cytometry.
Western blot
Cells were lysed in Tissue Protein Extraction Reagent (Thermo Fisher, Waltham, USA) with a cocktail of proteinase inhibitors and phosphatase inhibitors (Roche Applied Science, Indianapolis, USA) to extract the total intracellular proteins. The bicinchoninic acid (BCA) Protein Assay Kit (Thermo Fisher, Waltham, USA) was used to measure the protein concentration. Then, whole cell lysates were separated by 8%, 10% or 12% sodium dodecyl sulfate‒polyacrylamide gel electrophoresis (SDS‒PAGE) and transferred electrophoretically onto polyvinylidene difluoride (PVDF) blotting membranes. The blots were blocked with 5% skimmed milk for 1 h and then incubated with primary antibodies at 4 °C overnight, followed by different secondary antibodies for 1 h at room temperature. Finally. The bands were detected with an enhanced chemiluminescence (ECL) detection system using a multifunctional digital imager to obtain images.
mRFP-GFP-LC3 expressing cell generation and fluorescent LC3 puncta analysis
To perform image-based analysis for autophagy, cells were infected with the tandem GFP-RFP-LC3 adenovirus for 24 h according to the manufacturer’s instructions, then cells were treated and imaged for green fluorescent protein (GFP) and red fluorescent protein (RFP) by using Fluoview FV1000 microscope (Olympus, Tokyo, Japan).
Transmission electron microscopy (TEM)
Cells treated with avermectin B1 were fixed with 2.5% glutaraldehyde, dehydrated, and embedded, and ultrathin sections were prepared and stained with uranyl acetate and lead citrate. Electron micrographs of the ultrastructure of three osteosarcoma cells were taken using transmission electron microscopy. Three micrographs were captured at 6800x and 13000x magnification for each sample in a randomly selected area. The number of autophagic vacuoles (AVs) per unit area of each sample was counted and analyzed.
Xenograft mouse model
Female BALB/c nude mice (20 g, 8 weeks old) were used in vivo, and all animal experiments were performed according to the guidelines approved by the Shanghai Medical Experimental Animal Care Commission. Animals were assigned to experimental groups using simple randomization. MNNG cells (1 × 106 cells/mouse) were injected subcutaneously into the right shoulder of the nude mice to establish a subcutaneous osteosarcoma graft model. The two groups of nude mice were injected intraperitoneally with 200 µL of 0.9% NaCl or 1 mg of avermectin B1 daily. The body weight and tumor volume of the mice were measured every three days. Thirty days after intraperitoneal injection, the mice were euthanized, and the tumor tissue was weighed and stored in 4% tissue fixative for further analysis. In addition, for the lung metastasis study, 2 × 106 MNNG cells were injected into the tail vein of 16 mice according to the above grouping, and treatment was started 1 week later. The body weight of the mice was measured every three days. After 45 days, the mice were sacrificed, and the lung tissue was fixed in 4% tissue fixative. Immunohistochemical staining was performed on the fixed subcutaneous tumor tissue to observe the expression of the relevant proteins in the tumor tissue. The fixed lung tissue was paraffin embedded, sectioned, stained with hematoxylin-eosin (H&E) and immunohistochemically analyzed for histopathological evaluation. All mice were housed and monitored under SPF conditions.
Statistical analysis
Data were analyzed using SPSS (IBM SPSS Statistics 26.0) software. Student’s t test was used to compare the differences between the two groups. One-way ANOVA and post-hoc tests are used to analyze a series of three groups. A value of p < 0.05 was considered statistically significant. Graphs were generated using GraphPad Prism 8 software.
Results
Avermectin B1 inhibits the proliferation of human osteosarcoma cells in vitro
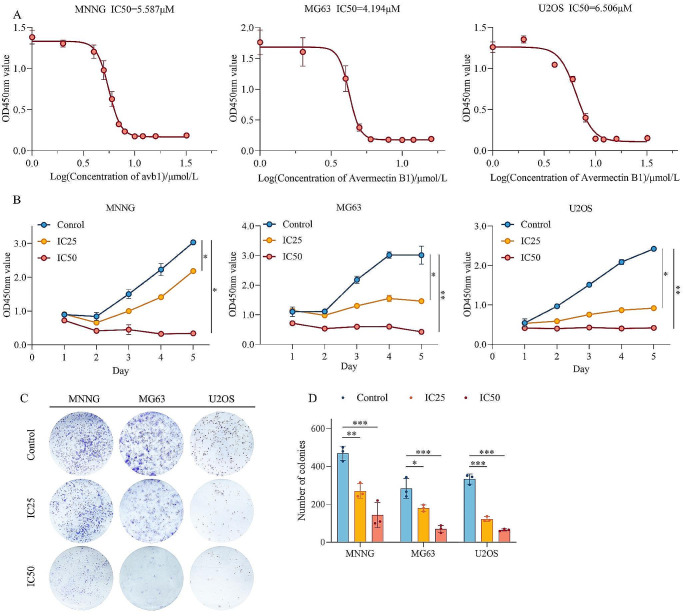
The effect of avermectin B1 on the viability of three osteosarcoma cell lines (MNNG, MG63 and U2OS) was examined using a CCK-8 assay. Cells were treated with different concentrations of avermectin B1 for 48 h, and the results showed that avermectin B1 dose-dependently reduced the viability of MNNG, MG63 and U2OS cells (Fig. 1A). The half-inhibitory concentration IC50 of avermectin B1 was 5.587 µM, 4.194 µM and 6.506 µM for the MNNG, MG63 and U2OS cell lines, respectively. These results showed that the different osteosarcoma cell lines exhibited different sensitivities to avermectin B1. The IC50 for 48 h was selected for subsequent experiments. Next, the proliferative capacity of osteosarcoma cells was assessed by CCK-8 assay, and the results showed that avermectin B1 significantly inhibited the growth of osteosarcoma cells (Fig. 1B). In addition, the results of the colony formation assay further proved that avermectin B1 attenuated the formation of osteosarcoma cell colonies in a dose-dependent manner (Fig. 1C and D). This preliminary investigation of the effects of avermectin B1 on osteosarcoma revealed that it could inhibit the proliferation of osteosarcoma cells.
Fig. 1.
Effects of avermectin B1 on the proliferation of osteosarcoma. (A) OS cells were treated with different concentrations of avermectin B1 for 48 h, and cell viability was detected by CCK-8 assay. (B) Assessment of the proliferative capacity of osteosarcoma cells treated with avermectin B1. (C) Colony formation assays in osteosarcoma cells after treatment with avermectin B1 (magnification, ×40). (D) Quantification of the number of colonies. Data are shown as the means ± SDs with scatter plots from at least three independent experiments. Statistical analysis was performed using one-way ANOVA followed by post-hoc tests. *p < 0.05, **p < 0.01, *** p < 0.001
Avermectin B1 inhibits the migration and invasion of human osteosarcoma cells in vitro
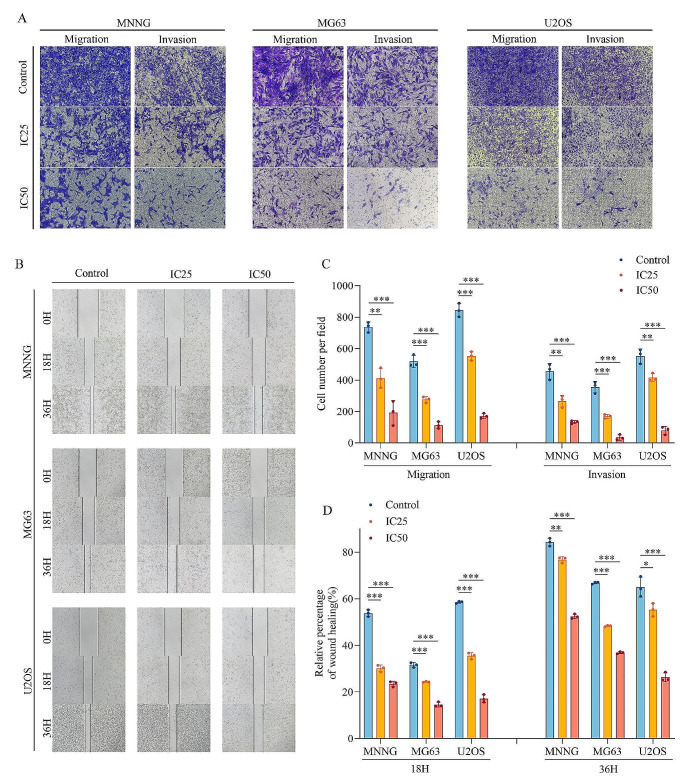
To investigate whether avermectin B1 could inhibit the migration and invasion of osteosarcoma, cells were pretreated with avermectin B1 at different concentrations for 48 h, followed by the Transwell assay. The results showed that avermectin B1 greatly inhibited the migration and invasion of osteosarcoma cells (Fig. 2A and C). To confirm the inhibition of migration and invasion in osteosarcoma cells, a wound healing assay was performed after treatment with avermectin B1 for 48 h (Fig. 2B and D). The results further proved that avermectin B1 inhibited the migration and invasion of osteosarcoma. Altogether, these results suggested that avermectin B1 could dose-dependently inhibit migration and invasion in osteosarcoma cells.
Fig. 2.
Effects of avermectin B1 on the migration and invasion of osteosarcoma. (A) Transwell assays were conducted to determine the migrative and invasive ability of MNNG, MG63 and U2OS cell lines. (B) Wound healing assay of osteosarcoma cell lines treated with avermectin B1. (C) Quantification of migrated and invaded cells. (D) Quantification of the wound healing rate. Data are shown as the means ± SDs with scatter plots from at least three independent experiments. Statistical analysis was performed using one-way ANOVA followed by post-hoc tests. *p < 0.05, **p < 0.01, *** p < 0.001
Avermectin B1 induces cell apoptosis and cell cycle arrest in osteosarcoma in vitro
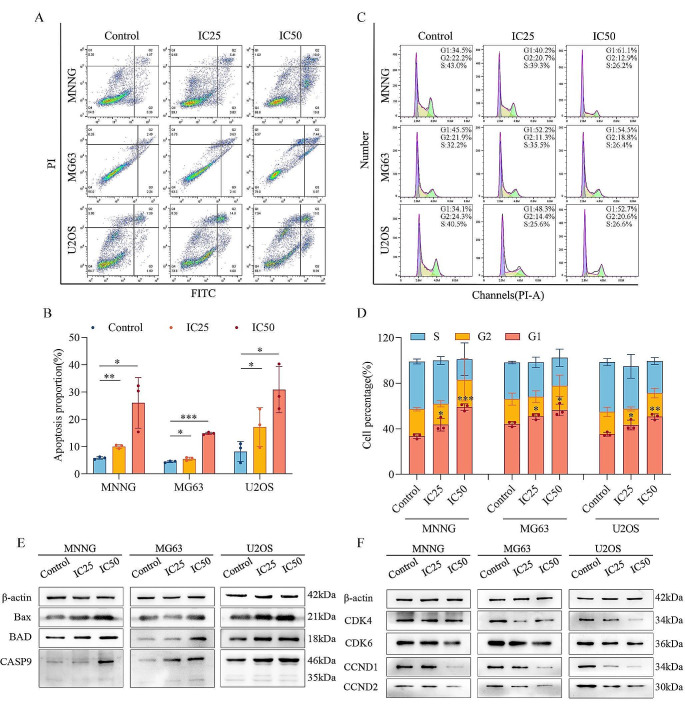
After three osteosarcoma cell lines were treated with avermectin B1 for 48 h, the proportion of apoptotic cells was further evaluated by flow cytometry using Annexin V-FITC/PI staining (Fig. 3A). There was a positive correlation between the proportion of apoptotic cells and the Avermectin B1 concentration (Fig. 3B). The expression of the apoptosis-related proteins Bax, BAD, and caspase-9 was upregulated (Fig. 3E). These experiments showed that avermectin B1 could induce apoptosis in osteosarcoma cells. Checking the cell cycle progression by flow cytometry, the proportion of cells in G1 phase increased after avermectin B1 treatment for 48 h (Fig. 3C and D). Furthermore, the levels of the cyclin-dependent kinases CDK4 and CDK6 were both downregulated after treatment with avermectin B1. Meanwhile, the levels of Cyclin D1 and Cyclin D2 were downregulated (Fig. 3F). Therefore, avermectin B1 treatment could induce cell apoptosis and cell cycle arrest in osteosarcoma cells in vitro.
Fig. 3.
Avermectin B1 induces cell apoptosis and cell cycle arrest in osteosarcoma. (A) Flow cytometry was conducted to measure the apoptotic rate of three osteosarcoma cell lines after treatment with IC25 and IC50 of avermectin B1. (B) Quantification of the apoptosis proportion. (C) The cell cycle distribution was detected by flow cytometry after treatment with IC25 and IC50 of avermectin B1. (D) Quantification of the cell cycle percentage. (E) Western blotting was performed to detect the expression levels of the apoptosis-related proteins Bax, BAD, and caspase-9 in avermectin B1-treated cells. (F) The expression levels of the cell cycle-related proteins CDK4, CDK6, Cyclin D1 and Cyclin D2. Data are shown as the means ± SDs with scatter plots from at least three independent experiments. Statistical analysis was performed using one-way ANOVA followed by post-hoc tests. *p < 0.05, **p < 0.01, *** p < 0.001
Avermectin B1 induces autophagy in human osteosarcoma cells
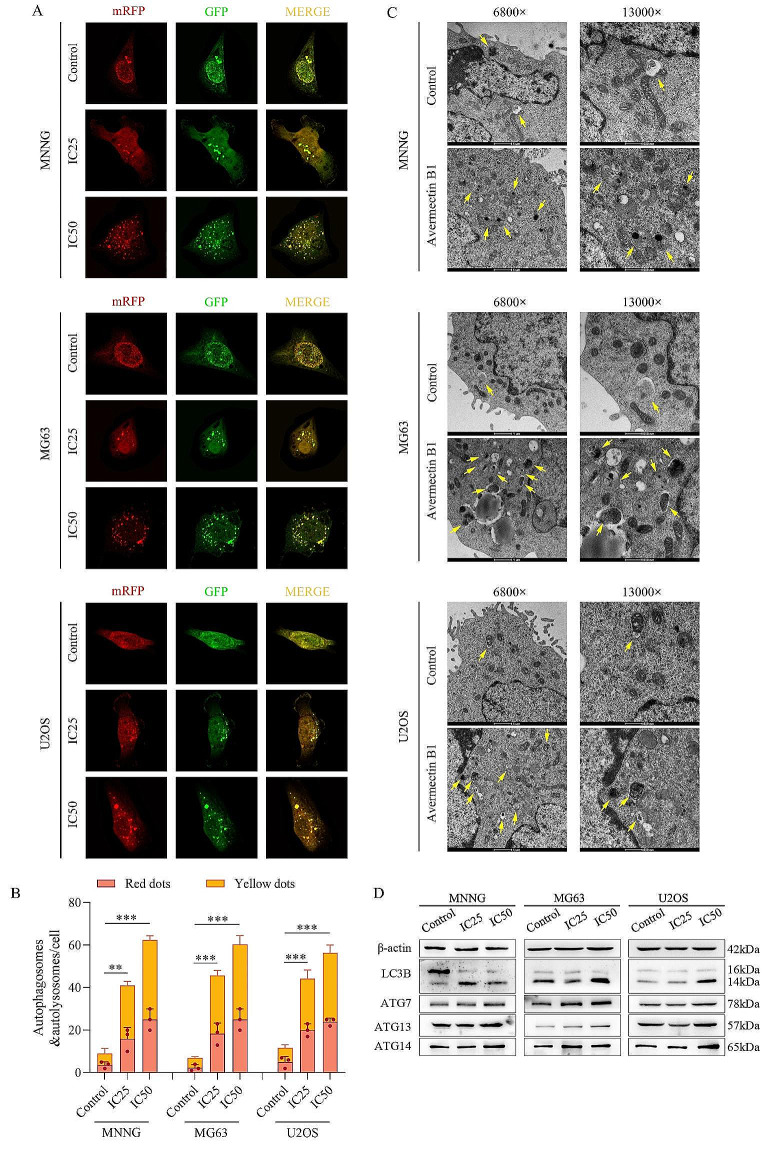
Autophagy plays a crucial role in cellular metastatic homeostasis, but whether avermectin B1 affects autophagy in osteosarcoma lines remains unclear. We utilized the mRFP-GFP-LC3 adenovirus construct to further confirm autophagy induction by the formation punctate signal. The rationale of this assay is based on the pH difference between the acidic autolysosome and the neutral autophagosome. The yellow puncta, formed by merging the green and red signals, are indicators of autophagosomes, while the free red puncta are indicative of autolysosomes. In the present study, after infection with the mRFP-GFP-LC3 adenovirus, it could be observed the successful introduction of this adenovirus could be observed through the presence of both fluorescent proteins. The numbers of yellow and red puncta were both significantly higher than those after treatment with avermectin B1, indicating increased autophagosomes and autolysosomes (Fig. 4A and B). MNNG, MG63 and U2OS cell lines were pretreated with avermectin B1 at the IC50 for 48 h and then observed using TEM. Compared with untreated cells, a greater amount of double-membraned vesicles (autophagosome-like structures) was clearly visible under TEM (Fig. 4C), which contained cytoplasm contents with a high density of electrons and was distinctly recognized as a marker of increased autophagy. Subsequently, Western blot experiments were performed to detect the expression level of the LC3BII/I ratio, which is well accepted as a biomarker for the activated autophagy process. The results showed that avermectin B1 markedly increased the LC3BII/I ratio and the expression of other autophagy-associated proteins, including ATG7, ATG13 and ATG14 (Fig. 4D), which revealed that avermectin B1 induced autophagy.
Fig. 4.
Avermectin B1 induced autophagy in osteosarcoma cells. (A) The LC3 puncta were analyzed by mRFP-GFP-LC3 construct. (B) Quantification of the autophagosomes (yellow dots) and autolysosomes (free red dots). (C) Autophagosome-like structures (indicated by the yellow arrows) were assayed by transmission electron microscopy (magnification, ×6800 and ×13,000). (D) Western blot experiments were performed to measure the expression changes in LC3B and the autophagy-associated proteins ATG7, ATG13 and ATG14. Data are shown as the means ± SDs with scatter plots from at least three independent experiments. Statistical analysis was performed using one-way ANOVA followed by post-hoc tests. **p < 0.01, *** p < 0.001
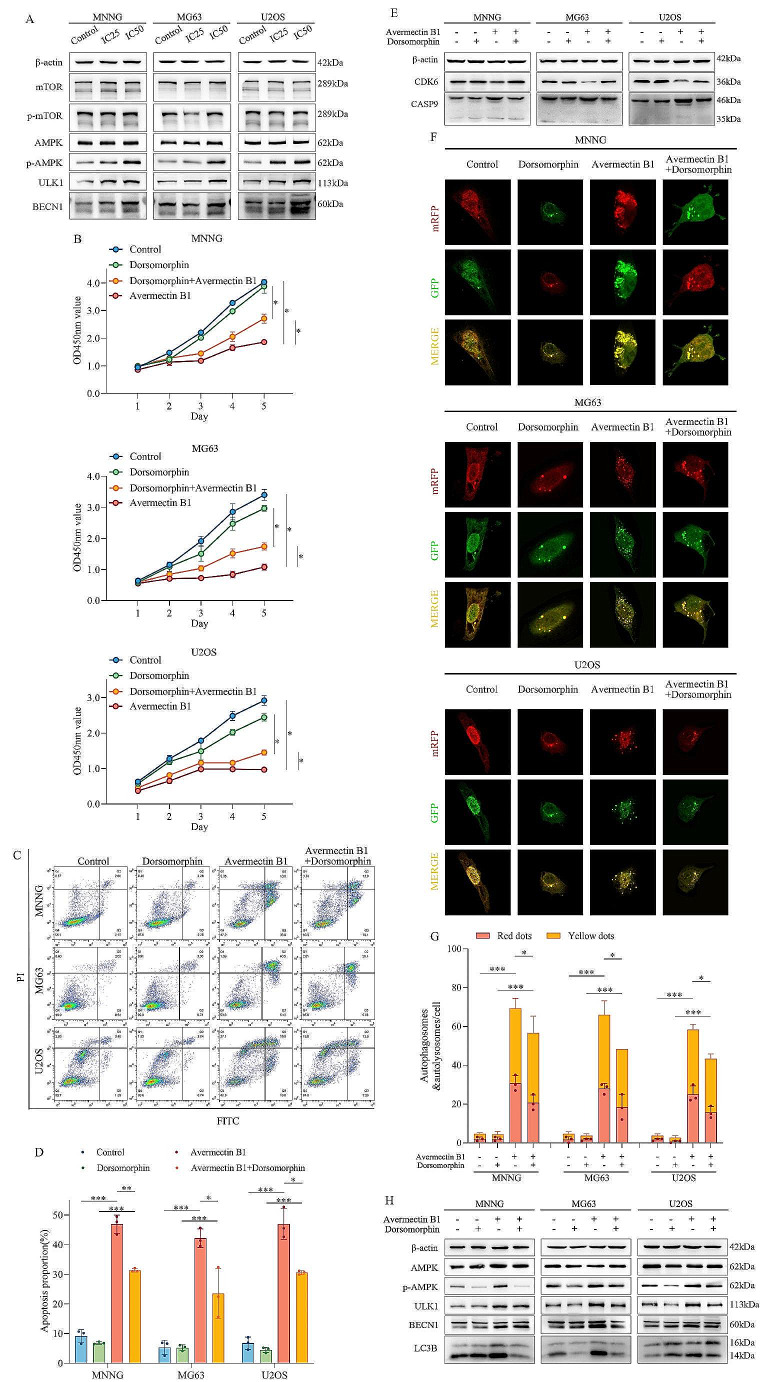
Avermectin B1 promotes autophagy and induces apoptosis through the AMPK/ULK1 pathway
Numerous studies have proven that autophagy involves various signaling pathways, among which the AMPK/ULK1 pathway plays a critical role in the activation of the autophagy process. In this study, we found that avermectin B1 activated p-AMPK, increased ULK1 and Beclin1, and had almost no influence on mTOR and p-mTOR (Fig. 5A). These results suggested that avermectin B1 directly affected ULK1 via p-AMPK without influencing p-mTOR expression. To further clarify the underlying mechanism of avermectin B1 in osteosarcoma, we used the AMPK inhibitor dorsomorphin to restore the antitumor effect promoted by avermectin B1. When Dorsomorphin was combined with Avermectin B1, proliferation experiments and flow cytometry showed that cell viability and apoptosis were restored (Fig. 5B, C and D), and CDK6 and Caspase 9 protein also show a corresponding change (Fig. 5E). The ability of dorsomorphin to also reverse avermectin-induced autophagy can be demonstrated visually by the punctate signaling of autolysosomes and autophagosomes (Fig. 5F and G). Additionally, AMPK/ULK1 pathway proteins and autophagy-related proteins were significantly restored (Fig. 5H). These results implied that avermectin B1 promoted cell autophagy and induced apoptosis through activation of the AMPK/ULK1 pathway.
Fig. 5.
Inhibition of autophagy inhibits the avermectin B1-mediated reduction in cell survival and apoptosis induction in osteosarcoma in vitro. (A) Western blot experiments were conducted to measure the expression changes in AMPK/ULK1 signaling pathway proteins after avermectin B1 treatment. (B) MNNG, MG63 and U2OS cells were treated with IC50 of avermectin B1 and 2 mM dorsomorphin alone or in combination as indicated and subjected to CCK8 assay. (C) Flow cytometry was conducted to measure the apoptotic rate of three osteosarcoma cell lines treated with IC50 of avermectin B1 and 2 mM dorsomorphin alone or in combination. (D) Quantification of the apoptosis proportion. (E) MNNG, MG63 and U2OS cells were treated with the IC50 of avermectin B1 and 2 mM dorsomorphin alone or in combination. Then, Western blotting was performed to detect the expression levels of the apoptosis-related protein caspase-9 and the cell cycle protein CDK6 in avermectin B1-treated cells. β-Actin was used as a loading control. (F) The LC3 puncta were analyzed by mRFP-GFP-LC3 construct when MNNG, MG63 and U2OS cells were treated with avermectin B1 and dorsomorphin alone or in combination. (G) Quantification of the autophagosomes (yellow dots) and autolysosomes (free red dots). (H) The protein expression levels of the AMPK/ULK1 pathway proteins BECN1 and LC3B after treatment with avermectin B1, dorsomorphin or avermectin B1 + dorsomorphin. Data are shown as the means ± SDs with scatter plots from at least three independent experiments. Statistical analysis was performed using Student’s t test. *p < 0.05, **p < 0.01, *** p < 0.001
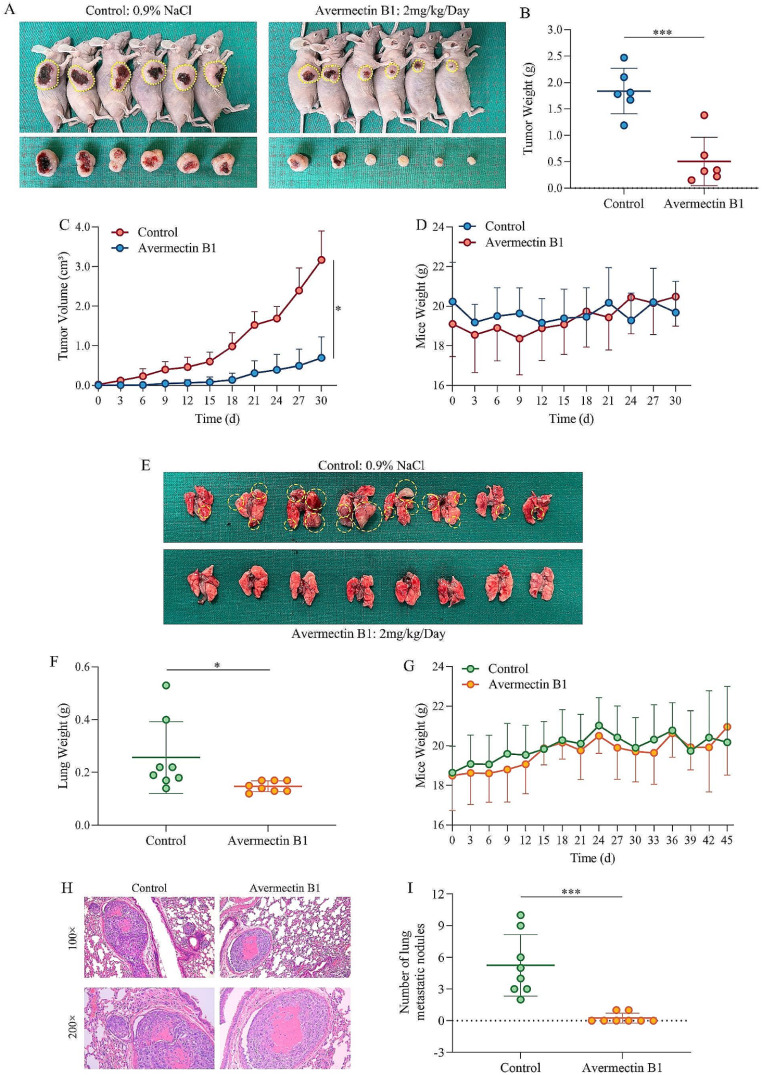
Avermectin B1 inhibits osteosarcoma cell growth and metastasis in vivo
To evaluate the antitumor effect of avermectin B1 in vivo, MNNG cells were subcutaneously transplanted into nude mice or injected into the lateral tail veins of nude mice. Then, these mice were treated with equivalent amounts of saline or avermectin B1 (2 mg/kg/day, n = 6/group). For subcutaneous tumors, the size and weight of the tumors in the 2 mg/kg avermectin B1 group were markedly reduced compared with those of the control group (Fig. 6A, B and C). However, there was no significant decrease in body weight (Fig. 6D). In addition, there were more lung metastases in the control groups in the caudal vein lung metastasis model than in the avermectin B1 group (Fig. 6E). As shown in Fig. 6F and G, the lung weights were lighter than those of the control group, and no significant decrease in body weight was observed. Furthermore, hematoxylin-eosin staining was used to analyze the lung metastasis nodules (Fig. 6H). The histological analysis showed that the avermectin B1-treated group had a lower number of lung metastatic nodules (Fig. 6I). Thus, we concluded that avermectin B1 inhibited osteosarcoma cell growth and metastasis in vivo.
Fig. 6.
Avermectin B1 inhibits osteosarcoma cell growth and metastasis in vivo. (A) The top panel shows a photograph of a tumor-bearing mouse, and the bottom panel shows a photograph of a subcutaneous tumor. (B) Weight of subcutaneous tumors in the control and avermectin B1 treatment groups in the subcutaneous tumor model. (C) Growth curve of the measured tumor volume in the subcutaneous tumor model. (D, G) Mouse weights separately in the subcutaneous tumor model and lung metastasis model. (E) Lung metastases formed in the avermectin B1-treated and control groups. (F) Lung weight of the lung metastasis model in the control and avermectin B1 treatment groups. (H) Lung metastasis nodules were stained with hematoxylin-eosin and observed by microscopy (magnification, ×100 and ×200). (I) Statistical analysis of lung metastases in the avermectin B1 and control groups in the lung metastasis model. Statistical analysis was performed using Student’s t test. Error bars represent the SD. *p < 0.05, *** p < 0.001
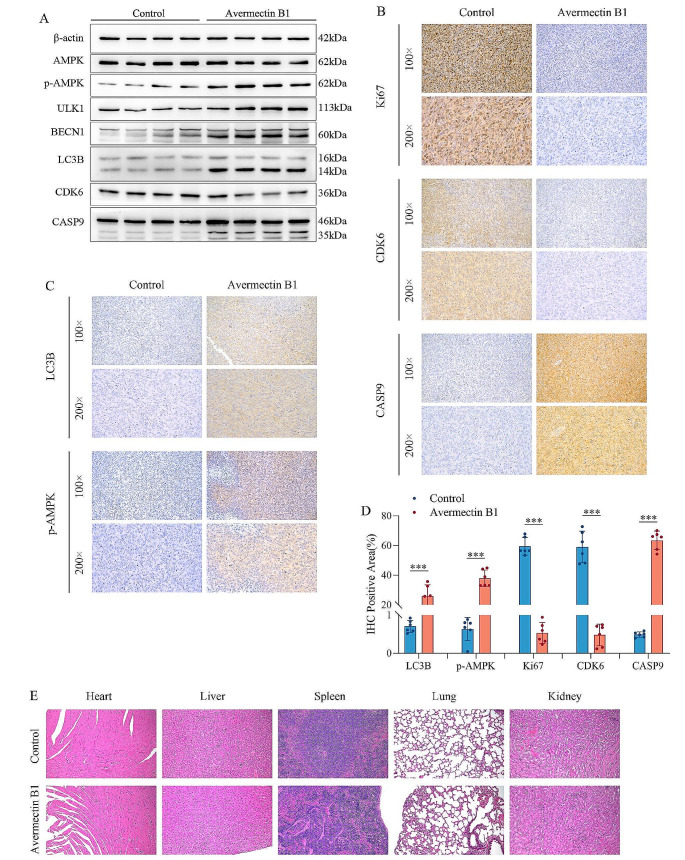
Avermectin B1 activates AMPK/ULK1 signaling pathway in human osteosarcoma cells in vivo
We further explored whether avermectin B1 mediated antitumor activity by regulating the AMPK/ULK1 signaling pathway. The Western blot results (Fig. 7A) showed that the expression of p-AMPK and ULK1 in the avermectin B1-treated group was significantly inhibited compared with that in the control group. In addition, the levels of the autophagy-associated protein Beclin1 and the ratio of LC3 II/I were significantly increased in the Avermectin B1 group compared with the control group. At the same time, the expression levels of CDK6 and Caspase 9 were also consistent with the results of the in vivo experiments. The immunohistochemistry analysis indicated that the staining of p-AMPK, LC3B and Caspase 9 was higher, and the staining of CDK6 and Ki-67 was lower in the avermectin B1 group than in the control group (Fig. 7B, C and D). Moreover, the results of H&E pathological sections of mouse major organs (heart, liver, spleen, lung and kidney) indicated that avermectin B1 treatment had no evident toxicity (Fig. 7E). Taken together, these results suggest that avermectin B1 activates the AMPK/ULK1 pathway to exert antitumor activity and induce autophagy in human osteosarcoma cells in vivo.
Fig. 7.
Avermectin B1 activates AMPK/ULK1 signaling pathway in human osteosarcoma cells in vivo. (A) Western blot analysis of AMPK, p-AMPK, ULK1, BECN1, LC3B, CDK6 and Caspase 9 in subcutaneous tumor tissues. (B, C) Ki67, CDK6, Caspase 9, p-AMPK and LC3B protein expression in tumor tissues was analyzed by immunohistochemistry (×100 and ×200). (D) Percentage of immunohistochemically positive area for Ki67, CDK6, Caspase 9, p-AMPK and LC3B in subcutaneous tumor tissues. (E) H&E staining images of the heart, liver, spleen, lung and kidney in mice. Data are shown as the means ± SDs with scatter plots from at least six independent experiments. Statistical analysis was performed using Student’s t test. *** p < 0.001
Discussion
Osteosarcoma is the most common malignancy arising from bone, accounting for 2.4% percent of pediatric and adolescent cancers worldwide [4]. Current treatment options available for osteosarcoma include surgery and chemotherapy. Chemotherapy is one of the most important methods for the treatment of osteosarcoma. In recent years, many new chemotherapeutic drugs have entered the clinic, but chemotherapy can easily induce drug resistance and cause tumor recurrence and metastasis [1, 5, 6]. Therefore, the development of new drugs that can improve anticancer activity and reduce side effects is an urgent problem to be solved in chemotherapy.
As mentioned above, the broad-spectrum antiparasitic drug avermectin, which is widely used in the field of parasitic control, has many advantages. Some researchers found that avermectin included not only anti-parasitic activity [9] but also antitumor [10, 21] and anti-inflammatory [22] effects and even in the prevention or treatment of alcohol use disorders [23]. It was recently reported that the avermectin family contributed to a promising inhibitory effect on the SARS-CoV-2 virus, which caused a global outbreak in 2020 [24, 25]. Therefore, it is worth developing as a potential new anticancer drug.
Since the first report that avermectin family members could reverse tumor multidrug resistance in 1996 [26], an increasing number of studies have noted the potential of avermectins in the treatment of tumors. Victor A. Drinyaeva [10] studied the natural avermectin mixture avermectin C in ascites and solid tumor growth in mice and found for the first time that avermectin analogs had relatively pronounced antitumor effects. It was reported that a novel avermectin analog named Eprinomectin targeted PC3 metastatic prostate cancer cells by inducing ROS and apoptosis activation and inhibited the activity of alkaline phosphatase, a marker of pluripotent stem cells [27]. In addition, the avermectin macrocyclic lactone derivatives selamectin and ivermectin impaired clonogenic self-renewal in vitro and inhibited tumor growth and metastasis in vivo by transcriptional modulation of genes associated with epithelial-mesenchymal transition and maintenance of a cancer stem cell phenotype in triple-negative breast cancer cells [13]. However, the antitumor effects of avermectin receptors in osteosarcoma have not been reported.
In the current study, we first detected the antitumor properties of avermectin on osteosarcoma in vivo and in vitro. In vitro, the results illustrated that avermectin B1 significantly inhibited cell proliferation in osteosarcoma, and the IC50 values of avermectin B1 in the three cell lines were 5.587, 4.194, and 6.506 µM for 48 h. This result was similar to the dose‒response curve of avermectin on HMLE breast cancer cells [28]. In addition, transwell and wound healing assays showed that avermectin B1 treatment inhibited metastasis in osteosarcoma cells. Furthermore, flow cytometry assays demonstrated that avermectin B1 induces apoptosis and cell cycle arrest in the G1 phase in osteosarcoma cells. This finding was consistent with Zhang’s research [29,] which discovered that several avermectins, represented by avermectin B1 (10 µM), significantly inhibited endogenous transmembrane member 16 A (TMEM16A)-mediated currents and thus proliferation and migration, inducing apoptosis of LA795 mouse lung adenocarcinoma cells. A slight difference was that they tested the impact on proliferation by a BrdU cell proliferation assay compared with our proliferation assay and clone formation assay.
In vivo, Zhang [29] administered avermectin B1 intragastrically to LA795 xenograft nude mice every day for four weeks and injected avermectin B1 (5 mg/kg body weight) intraperitoneally once every 3 days. Otherwise, Drinyaev [10] indicated that although avermectin B1 at doses of 1 mg/kg exhibited no cytotoxicity in vivo [11], it suppressed the growth of ascites Ehrlich carcinoma and P388 lymphoblastic leukemia as well as solid Ehrlich carcinoma. Similarly, we found that avermectin B1 injected intraperitoneally (2 mg/kg body weight) effectively inhibited osteosarcoma cell growth and metastasis in vivo without apparent toxicity. Thus, our results demonstrated that avermectin B1 inhibited proliferation and metastasis and promoted apoptosis in osteosarcoma in vitro and in vivo.
It is generally accepted that members of the avermectin family, including avermectin B1, act as antiparasitic drugs by inhibiting the nervous system through the opening of ligand-gated chloride channels, including glutamate (GluCl) and γ-aminobutyric acid (GABA) gated channels [30, 31]. Other effects and mechanisms of avermectin B1 drugs have been explored. For example, avermectin B1 attenuated lipopolysaccharide-induced inflammatory damage in vitro by downregulating the NF-κB and MAPK activation pathways [32]. Although a series of derivatives of avermectin B1, such as ivermectin, moxidectin and eprinomectin, have successively shown mechanisms of anticancer action [21, 27, 33], the exact antitumor mechanism of avermectin B1 is not clear. Zhang [29] proposed that the avermectin family, including avermectin B1, inhibits TMEM16A-encoded Ca2+-activated Cl− channels and that endogenous TMEM16A-mediated currents underlie their anticancer effects. On the basis of previous studies [34–37], it has been suggested that avermectin B1 can induce apoptosis and autophagy in different cells by ROS accumulation to mediate mTOR signaling pathway suppression. Our results verified that avermectin B1 inhibits the growth of osteosarcoma by inducing cytostatic autophagy both in vivo and in vitro. In contrast to the cytotoxicity of avermectin B1 via the PI3K/AKT pathway reported by Zhu [35, 37] et al., we found that avermectin B1 activated the AMPK/ULK1 signaling pathway to exert antitumor activity and induce autophagy in osteosarcoma. Some studies have shown that AMPK negatively regulates aerobic glycolysis (the Warburg effect) in cancer cells and suppresses tumor growth in vivo [38]. In addition, AMPK activation is one of the key pathways involved in autophagy [39]. AMPK inhibited the mTOR pathway and activated ULK1-induced autophagy [40]. Additionally, AMPK directly phosphorylates and activates ULK1 at Ser317, Ser555 and Ser777 to initiate autophagy [41]. However, some studies have reported that inactivation of mTOR is not always required for autophagy [42, 43]. Consistent with this finding, our results showed that AMPK activation after avermectin B1 treatment induced autophagy through phosphorylation of ULK1, independent of mTOR inhibition. Furthermore, the role of AMPK/ULK1 activation in avermectin B1-induced autophagy was confirmed by the pharmacological inhibitor dorsomorphin. The AMPK/ULK1 pathway is one of the important signaling pathways for autophagy in most cancer cells, and many anticancer drugs exert antitumor activity by activating the AMPK/ULK1 pathway [39]. Our study suggested that avermectin B1 might promote cell autophagy and induce apoptosis through the AMPK/ULK1 signaling pathway. These results provide further evidence that the AMPK/ULK1 pathway is a potential target for developing novel anticancer agents.
Conclusion
In summary, it was observed that abamectin has antitumor activity against osteosarcoma cells. These results suggested that the underlying mechanism of abamectin-induced autophagy was induced by activating the AMPK/ULK1 pathway, and it is evident that abamectin has the potential to be an effective anticancer therapy for osteosarcoma.
Acknowledgements
Not applicable.
Author contributions
X.F. and Z.L.: Methodology, Data curation, Writing-Original draft preparation. Z.P.: Visualization, Investigation. Y.L.: Data curation. C.T.: Formal analysis. D.C.: Conceptualization. D.C. and Q.Y.: Writing Reviewing and Editing, Funding acquisition.
Funding
This work was supported by the grants from the National Natural Science (81872182, 81802685).
Data availability
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
All animal experiments were performed according to the guidelines approved by the Shanghai Medical Experimental Animal Care Commission.
Patient consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xiang Fei and Zhaohui Li contributed equally to this work.
Contributor Information
Dongdong Cheng, Email: 1988noodles@163.com.
Qingcheng Yang, Email: tjyqc@163.com.
References
- 1.Strauss SJ, Frezza AM, Abecassis N, Bajpai J, Bauer S, Biagini R et al (2021) Bone sarcomas: ESMO-EURACAN-GENTURIS-ERN PaedCan Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol 32(12):1520–1536. 10.1016/j.annonc.2021.08.1995 [DOI] [PubMed] [Google Scholar]
- 2.Sadykova LR, Ntekim AI, Muyangwa-Semenova M, Rutland CS, Jeyapalan JN, Blatt N et al (2020) Epidemiology and risk factors of Osteosarcoma. Cancer Invest 38(5):259–269. 10.1080/07357907.2020.1768401 [DOI] [PubMed] [Google Scholar]
- 3.Harrison DJ, Geller DS, Gill JD, Lewis VO, Gorlick R (2018) Current and future therapeutic approaches for osteosarcoma. Expert Rev Anticancer Ther 18(1):39–50. 10.1080/14737140.2018.1413939 [DOI] [PubMed] [Google Scholar]
- 4.Meltzer PS, Helman LJ (2021) New Horizons in the Treatment of Osteosarcoma. N Engl J Med 385(22):2066–2076. 10.1056/NEJMra2103423 [DOI] [PubMed] [Google Scholar]
- 5.Whelan JS, Davis LE (2018) Osteosarcoma, Chondrosarcoma, and Chordoma. J Clin Oncol 36(2):188–193. 10.1200/jco.2017.75.1743 [DOI] [PubMed] [Google Scholar]
- 6.Lilienthal I, Herold N (2020) Targeting Molecular mechanisms Underlying Treatment Efficacy and Resistance in Osteosarcoma: a review of current and future strategies. Int J Mol Sci 21(18). 10.3390/ijms21186885 [DOI] [PMC free article] [PubMed]
- 7.Grünewald TG, Alonso M, Avnet S, Banito A, Burdach S, Cidre-Aranaz F et al (2020) Sarcoma treatment in the era of molecular medicine. EMBO Mol Med 12(11):e11131. https://doi.org/10.15252/emmm201911131 [DOI] [PMC free article] [PubMed]
- 8.Sayles LC, Breese MR, Koehne AL, Leung SG, Lee AG, Liu HY et al (2019) Genome-informed targeted therapy for Osteosarcoma. Cancer Discov 9(1):46–63. https://doi.org/10.1158/2159–8290.CD-17-1152 [DOI] [PMC free article] [PubMed]
- 9.Burg RW, Miller BM, Baker EE, Birnbaum J, Currie SA, Hartman R et al (1979) Avermectins, new family of potent anthelmintic agents: producing organism and fermentation. Antimicrob Agents Chemother 15(3):361–367. 10.1128/aac.15.3.361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Drinyaev VA, Mosin VA, Kruglyak EB, Novik TS, Sterlina TS, Ermakova NV et al (2004) Antitumor effect of avermectins. Eur J Pharmacol 501(1–3):19–23. 10.1016/j.ejphar.2004.08.009 [DOI] [PubMed] [Google Scholar]
- 11.Korystov YN, Ermakova NV, Kublik LN, Levitman M, Shaposhnikova VV, Mosin VA et al (2004) Avermectins inhibit multidrug resistance of tumor cells. Eur J Pharmacol 493(1–3):57–64. 10.1016/j.ejphar.2004.03.067 [DOI] [PubMed] [Google Scholar]
- 12.Sun YJ, Long DX, Li W, Hou WY, Wu YJ, Shen JZ (2010) Effects of avermectins on neurite outgrowth in differentiating mouse neuroblastoma N2a cells. Toxicol Lett 192(2):206–211. 10.1016/j.toxlet.2009.10.021 [DOI] [PubMed] [Google Scholar]
- 13.Kwon YJ, Petrie K, Leibovitch BA, Zeng L, Mezei M, Howell L et al (2015) Selective inhibition of SIN3 Corepressor with avermectins as a Novel Therapeutic Strategy in Triple-negative breast Cancer. Mol Cancer Ther 14(8):1824–1836. 10.1158/1535-7163.MCT-14-0980-T [DOI] [PMC free article] [PubMed]
- 14.Vargas JNS, Hamasaki M, Kawabata T, Youle RJ, Yoshimori T (2023) The mechanisms and roles of selective autophagy in mammals. Nat Rev Mol Cell Biol 24(3):167–185. 10.1038/s41580-022-00542-2 [DOI] [PubMed]
- 15.Mizushima N (2018) A brief history of autophagy from cell biology to physiology and disease. Nat Cell Biol 20(5):521–527. 10.1038/s41556-018-0092-5 [DOI] [PubMed] [Google Scholar]
- 16.Debnath J, Gammoh N, Ryan KM Autophagy and autophagy-related pathways in cancer. Nat Rev Mol Cell Biol 2023:1–16 10.1038/s41580-023-00585-z [DOI] [PMC free article] [PubMed]
- 17.Kimmelman AC, White E (2017) Autophagy and Tumor Metabolism. Cell Metab 25(5):1037–1043. 10.1016/j.cmet.2017.04.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang K, Gao W, Dou Q, Chen H, Li Q, Nice EC et al (2016) Ivermectin induces PAK1-mediated cytostatic autophagy in breast cancer. Autophagy 12(12):2498–2499. 10.1080/15548627.2016.1231494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dou Q, Chen HN, Wang K, Yuan K, Lei Y, Li K et al (2016) Ivermectin induces Cytostatic Autophagy by blocking the PAK1/Akt Axis in breast Cancer. Cancer Res 76(15):4457–4469. 10.1158/0008-5472.Can-15-2887 [DOI] [PubMed] [Google Scholar]
- 20.Hashimoto H, Messerli SM, Sudo T, Maruta H (2009) Ivermectin inactivates the kinase PAK1 and blocks the PAK1-dependent growth of human ovarian cancer and NF2 tumor cell lines. Drug Discov Ther 3(6):243–246 [PubMed] [Google Scholar]
- 21.Tang M, Hu X, Wang Y, Yao X, Zhang W, Yu C et al (2021) Ivermectin, a potential anticancer drug derived from an antiparasitic drug. Pharmacol Res 163:105207. 10.1016/j.phrs.2020.105207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ci X, Li H, Yu Q, Zhang X, Yu L, Chen N et al (2009) Avermectin exerts anti-inflammatory effect by downregulating the nuclear transcription factor kappa-B and mitogen-activated protein kinase activation pathway. Fundam Clin Pharmacol 23(4):449–455. 10.1111/j.1472-8206.2009.00684.x [DOI] [PubMed] [Google Scholar]
- 23.Asatryan L, Yardley MM, Khoja S, Trudell JR, Hyunh N, Louie SG et al (2014) Avermectins differentially affect ethanol intake and receptor function: implications for developing new therapeutics for alcohol use disorders. Int J Neuropsychopharmacol 17(06):907–916. 10.1017/s1461145713001703 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Caly L, Druce JD, Catton MG, Jans DA, Wagstaff KM (2020) The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro. Antiviral Res 178:104787. 10.1016/j.antiviral.2020.104787 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.González-Paz L, Hurtado-León ML, Lossada C, Fernández-Materán FV, Vera-Villalobos J, Loroño M et al (2021) Comparative study of the interaction of ivermectin with proteins of interest associated with SARS-CoV-2: a computational and biophysical approach. Biophys Chem 278:106677. 10.1016/j.bpc.2021.106677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Didier A, Loor F (1996) The abamectin derivative ivermectin is a potent P-glycoprotein inhibitor. Anticancer Drugs 7(7):745–751. 10.1097/00001813-199609000-00005 [DOI] [PubMed] [Google Scholar]
- 27.Samy A, Bakthavachalam V, Vudutha M, Vinjamuri S, Chinnapaka S, Munirathinam G (2020) Eprinomectin, a novel semi-synthetic macrocylic lactone is cytotoxic to PC3 metastatic prostate cancer cells via inducing apoptosis. Toxicol Appl Pharmacol 401:115071. 10.1016/j.taap.2020.115071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gupta PB, Onder TT, Jiang G, Tao K, Kuperwasser C, Weinberg RA et al (2009) Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell 138(4):645–659. 10.1016/j.cell.2009.06.034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang X, Zhang G, Zhai W, Zhao Z, Wang S, Yi J (2020) Inhibition of TMEM16A ca(2+)-activated Cl(-) channels by avermectins is essential for their anticancer effects. Pharmacol Res 156:104763. 10.1016/j.phrs.2020.104763 [DOI] [PubMed] [Google Scholar]
- 30.Campbell WC, Fisher MH, Stapley EO, Albers-Schönberg G, Jacob TA (1983) Ivermectin: a potent new antiparasitic agent. Science 221(4613):823–828. 10.1126/science.6308762 [DOI] [PubMed] [Google Scholar]
- 31.Cully DF, Vassilatis DK, Liu KK, Paress PS, Van der Ploeg LH, Schaeffer JM et al (1994) Cloning of an avermectin-sensitive glutamate-gated chloride channel from Caenorhabditis elegans. Nature 371(6499):707–711. 10.1038/371707a0 [DOI] [PubMed] [Google Scholar]
- 32.Zhang X, Li J, Chen C, Ci X, Yu Q, Zhang X et al (2011) Protective effect of abamectin on acute lung injury induced by lipopolysaccharide in mice. Fundam Clin Pharmacol 25(6):700–707. 10.1111/j.1472-8206.2010.00896.x [DOI] [PubMed] [Google Scholar]
- 33.Song D, Liang H, Qu B, Li Y, Liu J, Chen C et al (2018) Moxidectin inhibits glioma cell viability by inducing G0/G1 cell cycle arrest and apoptosis. Oncol Rep 40(3):1348–1358. 10.3892/or.2018.6561 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liang Y, Dong B, Pang N, Hu J (2019) ROS generation and DNA damage contribute to abamectin-induced cytotoxicity in mouse macrophage cells. Chemosphere 234:328–337. 10.1016/j.chemosphere.2019.06.031 [DOI] [PubMed] [Google Scholar]
- 35.Zhu S, Zhou J, Zhou Z, Zhu Q (2019) Abamectin induces apoptosis and autophagy by inhibiting reactive oxygen species-mediated PI3K/AKT signaling in MGC803 cells. J Biochem Mol Toxicol 33(7):e22336. 10.1002/jbt.22336 [DOI] [PubMed] [Google Scholar]
- 36.Liang Y, Dong B, Pang N, Hu J (2020) Abamectin induces cytotoxicity via the ROS, JNK, and ATM/ATR pathways. Environ Sci Pollut Res Int 27(12):13726–13734. 10.1007/s11356-019-06869-2 [DOI] [PubMed] [Google Scholar]
- 37.Zhu S, Zhou J, Sun X, Zhou Z, Zhu Q (2020) ROS accumulation contributes to abamectin-induced apoptosis and autophagy via the inactivation of PI3K/AKT/mTOR pathway in TM3 leydig cells. J Biochem Mol Toxicol 34(8):e22505. 10.1002/jbt.22505 [DOI] [PubMed] [Google Scholar]
- 38.Faubert B, Boily G, Izreig S, Griss T, Samborska B, Dong Z et al (2013) AMPK is a negative regulator of the Warburg effect and suppresses tumor growth in vivo. Cell Metab 17(1):113–124. 10.1016/j.cmet.2012.12.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kim J, Kundu M, Viollet B, Guan KL (2011) AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat Cell Biol 13(2):132–141. 10.1038/ncb2152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Roach PJ (2011) AMPK -> ULK1 -> autophagy. Mol Cell Biol 31(15):3082–3084. 10.1128/mcb.05565-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jang JE, Eom JI, Jeung HK, Cheong JW, Lee JY, Kim JS et al (2017) AMPK-ULK1-Mediated Autophagy confers resistance to BET inhibitor JQ1 in Acute myeloid leukemia stem cells. Clin Cancer Res 23(11):2781–2794. 10.1158/1078 [DOI] [PubMed]
- 42.Xie CM, Liu XY, Sham KW, Lai JM, Cheng CH (2014) Silencing of EEF2K (eukaryotic elongation factor-2 kinase) reveals AMPK-ULK1-dependent autophagy in colon cancer cells. Autophagy 10(9):1495–1508. 10.4161/auto.29164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mao K, Klionsky DJ (2011) AMPK activates autophagy by phosphorylating ULK1. Circ Res 108(7):787–788. 10.1161/RES.0b013e3182194c29 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No datasets were generated or analysed during the current study.