Abstract
Human articular cartilage has a poor ability to self‐repair, meaning small injuries often lead to osteoarthritis, a painful and debilitating condition which is a major contributor to the global burden of disease. Existing clinical strategies generally do not regenerate hyaline type cartilage, motivating research toward tissue engineering solutions. Prospective cartilage tissue engineering therapies can be placed into two broad categories: i) Ex situ strategies, where cartilage tissue constructs are engineered in the lab prior to implantation and ii) in situ strategies, where cells and/or a bioscaffold are delivered to the defect site to stimulate chondral repair directly. While commonalities exist between these two approaches, the core point of distinction—whether chondrogenesis primarily occurs “within” or “without” (outside) the body—can dictate many aspects of the treatment. This difference influences decisions around cell selection, the biomaterials formulation and the surgical implantation procedure, the processes of tissue integration and maturation, as well as, the prospects for regulatory clearance and clinical translation. Here, ex situ and in situ cartilage engineering strategies are compared: Highlighting their respective challenges, opportunities, and prospects on their translational pathways toward long term human cartilage repair.
Keywords: 3D bioprinting, biomaterials, cartilage tissue engineering, in situ tissue engineering, stem cells
This manuscript provides a comprehensive overview of the current state of the art of bioengineering for articular cartilage. Here the intricacies of articular cartilage bioengineering are described, exploring in depth the advances and disadvantages of two strategies of delivering stem cell based approaches, namely, the direct “in situ” delivery and the more traditional bench‐based strategy.

1. Introduction
Articular cartilage is specialized load‐bearing tissue that lines the surface of our long bones and is critical for joint function. Human cartilage has a poor ability to self‐repair, meaning small defects or injuries inevitably worsen over time.[ 1 ] The degeneration of cartilage leads to osteoarthritis, a painful and debilitating condition which is a major contributor to the global burden of disease.[ 2 ] Osteoarthritis afflicts upward of 40 million people in Europe, and more than 50 million in the USA.[ 3 , 4 ] In Australia, at estimated one in eleven people (2.2 million) have the condition.[ 5 ] Despite the prevalence and severity of osteoarthritis, few clinical interventions are effective at preventing its onset, even when an early stage chondral defect has been detected.[ 6 ] Typical interventions include microfracture (where small holes are punched through to underlying bone beneath the defect site to recruit regenerative stem cells from the bone marrow), mosaicplasty (where healthy cartilage is harvested from non‐load bearing regions of the joint and used to tile in the defect area), and matrix‐assisted autologous chondrocyte implantation (MACI, where patient chondrocytes are expanded in the lab and then reimplanted within a collagen matrix).[ 7 ] Some interventions result in initially improved clinical knee‐function scores, but these promising early results generally decline in the long term.[ 8 ] These interventions often fail because they result in a defect filled with fibrocartilage, which is less durable than the hyaline type cartilage, the hallmark of healthy articular cartilage tissue.[ 9 ] There is as yet no clinical strategy which results in long term restoration of functional hyaline cartilage, and this need motivates the development of tissue engineering and biofabrication based approaches to cartilage repair.[ 10 ]
Cartilage tissue engineering is a prospective therapeutic strategy involving some combination of cells, biomaterials, and growth factors to generate new cartilage tissue (chondrogenesis) as a means to achieve cartilage repair.[ 11 ] Cartilage tissue engineering research can be placed into two broad categories: i) Ex situ cartilage engineering, where cartilage tissue constructs are engineered in the lab prior to implantation and ii) in situ cartilage engineering, where cells and/or a bioscaffold are delivered to the defect site to stimulate chondral repair directly. In this review, the characteristic defining a technique as being in situ or ex situ is whether the regenerative cells are situated within the defect site when they begin to produce neo‐cartilage matrix.
Ex situ cartilage can be engineered through a range of methods which differ according to whether the new tissue is organized primarily through the self‐assembly processes of cells or through a biomaterial template, and according to the tools and techniques used to manufacture the construct. Some methods harness the self‐assembly of cell aggregates (for example, pellet culture or cell spheroids), which mimic the tight cell‐cell interactions of development through which articular cartilage originally forms.[ 12 , 13 ] These scaffold‐free are potentially advantageous if they can produce cartilage constructs without the need for implanted biomaterials. Nevertheless, generating engineered cartilage though self‐assembly with high reproducibility and of a therapeutic scale remains a challenge, especially as nutrient diffusion can limit construct size.[ 13 ] The alternative paradigm involves seeding chondrogenic cells upon (or within) a biomaterial scaffold which designates the intended 3D shape. In early cartilage engineering studies, scaffolds were created using techniques (such as solvent casting or electrospinning) which provided only limited control over porosity and shape. In contemporary iterations, advanced fabrication tools are often used to create functional constructs which recapitulate some of the complexity of native tissue. The use of such automated fabrication tools in the context of tissue engineering is termed biofabrication.[ 14 ] Owing to the many advantages afforded by modern fabrication technologies approach, and its prevalence in contemporary literature, we focus on biofabrication based ex situ strategies in this review.
Biofabrication approaches enable the creation of multi‐layered and/or multicomponent structures, tuned to match the biomechanics of native tissue and fashioned with patient‐specific (or defect‐specific) geometries.[ 15 , 16 ] Reproducing both the zonal architecture of articular cartilage, as well as the transition toward osteochondral bone may also become possible.[ 17 ] Following the initial fabrication step(s) the cell‐laden constructs can be cultured in the laboratory for weeks in a controlled environment while exposed to controlled dosages of growth factors and complemented, potentially, with a regimen of dynamic mechanical stimulation.[ 18 ] Encapsulated cells can thus be encouraged to distribute the extracellular matrix (ECM) components of articular cartilage, such as collagen type II and glycosaminoglycans, throughout the construct.[ 19 ] The goal of this process is to create a new piece of cartilage tissue which is as functionally mature as possible, and which can then be implanted to fit the defect site.[ 17 ]
On the contrary, in situ cartilage repair strategies aim to effect chondrogenesis within the defect itself. Here, the bioscaffold milieu (for example, a stem cell‐laden hydrogel) is delivered directly to the defect in an immature state. The biomaterial can be hardened in place, in a way that completely fills the defect geometry.[ 20 ] The native environment helps trigger differentiation of the stem cells toward a chondrogenic linear, and neo‐cartilage formation occurs within the body. Applying this framework to existing clinical treatments, the microfracture technique would be considered an in situ strategy, since bone marrow derived MSCs (BM‐MSCs) deposit new matrix within the defect site. The MACI technique is also a kind of in situ strategy, since ECM production by the chondrocytes is performed within the defect site, rather than during the laboratory culture step. (Chondrocytes are typically seeded on the collagen membrane just one or two days before surgery and the membrane is intended to biodegrade).[ 21 , 22 ]
While several excellent reviews and commentaries on cartilage tissue engineering have been recently published,[ 10 , 11 , 23 ] they typically discuss the field in general terms, and rarely make a strong distinction between strategies of “ex situ” and “in situ” chondrogenesis. In this perspective we highlight stark differences between these strategies across multiple critical processes, including what cell populations are selected for incorporation and how they are processed, how the biomaterial scaffold (if any) is formulated, how the implant is shaped to fit the defect, how maturation of the new tissue is stimulated, and how the new tissue integrates with the host (Figure 1 ). To constrain the scope of our analysis, we focus on a particular clinical scenario where a patient is identified with a partial or full‐thickness chondral defect which does not penetrate the subchondral bone. Our aim is to catalogue a selection of key distinctions between the ex situ and in situ cartilage engineering approaches, both as a means of clarifying differences in process, as well as, in the prospects for long‐term repair and ultimately of clinical translation.
Figure 1.
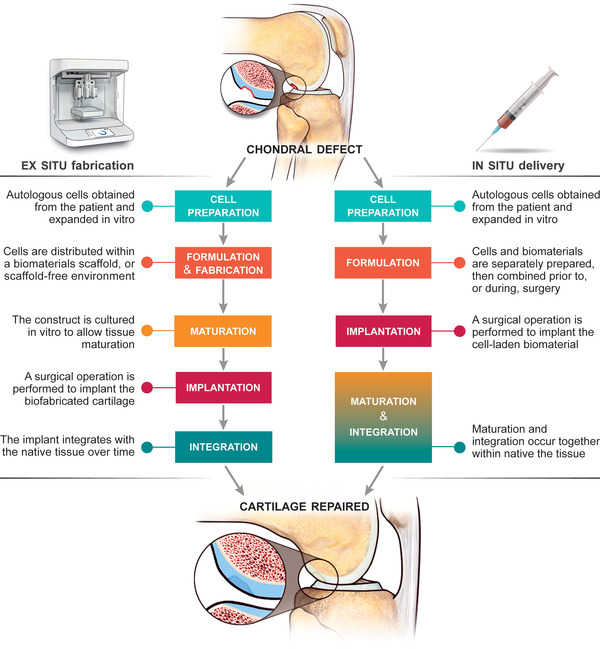
An illustration of the main processes required for each prospective tissue engineering strategy. One key difference is where maturation of the tissue construct takes place: Occurring prior to implantation in the case of the ex situ fabrication strategy, but after implantation in the case of the in situ approach. The repercussions of this central difference are felt up and down the process chain, influencing every other aspect of the intervention.
2. A Brief Introduction to Articular Cartilage and Its Repair
The dense, avascular and aneural cartilage ECM accounts for most of the total volume of articular cartilage, and its main components are water, collagen type II and proteoglycans.[ 24 , 25 ] The resident cells are the chondrocytes, which account for only ≈2% of the total articular cartilage volume. Chondrocytes have a mesenchymal origin and are originally responsible for generating cartilage in fetal development.[ 26 ] In mature tissue, the primary roles of chondrocytes are to maintain the ECM turnover and maintain tissue homeostasis.[ 24 ] Chondrocytes present different morphologies depending on the specific zone in which they are located. At the surface they have a flattened morphology and produce lubricin. In the middle zone, chondrocytes maintain synthesis of proteoglycans and other non‐collagen molecules.[ 27 ] In the deep zone, chondrocytes are arranged in columns, and express collagen type X. Cartilage‐derived stem/progenitor cells (CSPCs) have also been observed in human articular cartilage, but their origin and functions are still a matter of debate.[ 28 ] The poor ability of articular cartilage to naturally self‐repair arises from properties of the local cell populations and their microenvironment.
The avascular nature of cartilage means it is a nutrient poor environment.[ 24 ] Cartilage relies on diffusion of nutrients from the synovial fluid, a process which may be enhanced for large solutes by cyclic loading.[ 29 , 30 ] The lack of available metabolites contributes to the sedentary nature of resident cells. Articular cartilage has an extremely slow rate of ECM production, with the turnover rate of collagen type II estimated at more than one hundred years.[ 31 ]
When a focal injury occurs, there is no acute wound healing response as in most other human tissues, with no migration of monocytes and/or macrophages to the injured cartilage.[ 32 ] This lack of a response is again due to the lack of blood vessels, and the dense matrix which impedes cell migration. Resident articular chondrocytes also do not migrate to the lesion in vivo, and no production of a reparative matrix occurs.[ 33 ]
Cell populations within the tissue also lack regenerative potential. Resident CSPCs are present in very low numbers and while these cells have been shown to produce cartilage ECM in vitro, they require considerable manipulation to do so.[ 24 , 34 ] Other populations of stem cells reside locally in the synovial fluid and the infrapatellar fat pad (IFP), but these typically do not engage in active repair processes to regenerate natural tissue. While partial defects show essentially no ability to self‐repair, defects which are deep enough to release BM‐MSCs do show some limited capacity to heal. Nevertheless, the neo‐tissue generated by BM‐MSCs in vivo is generally fibrocartilage, characterized by high levels of collagen type I which lacks the biomechanical properties necessary for long‐term stability.[ 8 , 35 ]
The primary functions of articular cartilage are biomechanical—the ability to absorb, distribute, and withstand forces applied to the joint and to allow the joint to freely articulate.[ 24 , 36 ] The tissue is made up of approximately 80% fluid and 20% solids (collagen and proteoglycans) and has a distinct zonal structure and stiffness gradient throughout its depth.[ 37 , 38 ] The collagen network in cartilage, especially in the superficial layer, can provide some instantaneous resistance to load at a low strain rate.[ 37 ] However, under prolonged or cyclic compression loads, fluid pressure increases within the cartilage as fluid moves in and out of the tissue.[ 36 ] This motion, along with the low permeability of cartilage resisting the movement of fluid, results in a high frictional drag force which accounts for most of the tissue's load bearing capacity.[ 36 , 38 , 39 ] A striking feature of articular cartilage is its extremely low shear friction coefficient.[ 40 ] It is currently not possible to reproduce all of the complex viscoelastic and depth‐dependent properties of cartilage with synthetic materials, and so the manufacture of an inert prosthetic cartilage layer is infeasible. The most promising strategy to repair articular cartilage is to generate new tissue through biological processes.
The goal of articular cartilage repair is restoration of a functioning joint. The hallmarks of a successful cartilage repair strategy should thus include long term recapitulation of the biomechanical properties of hyaline cartilage (including compressive modulus and the shear friction coefficient); a population of cells which retain a stable chondrocyte lineage in vivo; a recapitulation of the molecular composition of articular cartilage with its abundance of collagen II and proteoglycans, and lack of collagen I; a microstructural and biomechanical integration of the new cartilage tissue both basally and laterally; as well as prevention of further cartilage degradation.
3. A Comparison or Ex Situ and In Situ Tissue Engineering Strategies Applied to Articular Cartilage Repair
3.1. Cell Preparation
This section focuses on how the biofabrication approach (ex situ or in situ) may influence the choice of cells that will be incorporated within the bioscaffold, as well as, the cell culture/differentiation process. Note, this section discusses the process of selecting the cell population, and the cell preparation procedures before they are implanted in the body. The activities of the cells after implantation are discussed in the section on Integration.
3.1.1. Overview: Cell Selection for Cartilage Tissue Engineering
The main cell types explored for cartilage engineering are chondrocytes and MSCs. Perinatal cells, as well as fetal or embryonic stem cells and chondroblasts, can also be employed.[ 41 ] While induced pluripotent stem cells demonstrate opportunities for cartilage repair, they still lack long term preclinical studies or clinical trials.[ 42 , 43 ] Each cell source has inherent advantages and disadvantages, and much ongoing research concerns the determination of the most suitable cell type, and the optimal processing conditions, for cartilage engineering.[ 44 , 45 ] While evidence suggests cartilage tissue may be immune privileged,[ 46 ] the majority of existing therapies and ongoing research programs are designed for autologous cells. Chondrocytes, harvested from native tissue and expanded in culture, are employed clinically in the autologous chondrocyte implantation and MACI cell therapies.[ 47 ] Autologous chondrocytes are usually obtained by harvesting cartilage from non‐load bearing regions of the articular surface, but can also be obtained from nasal cartilage[ 48 ] and the ribs.[ 49 ] Expansion of chondrocytes in vitro can be problematic owing to their tendency to de‐differentiate toward a fibroblastic lineage in culture. Recovery of chondrogenic potential may be enabled by culturing chondrocytes on hydrogel substrates, rather than tissue culture plastic. While MACI is a preferred clinical intervention for large chondral defects, this strategy often does not result in effective long‐term repair.[ 50 ] The use of cartilage‐derived cells in clinical trials has decreased from an average of 45% (2006–2012) down to 10–15% (after 2013).[ 47 ]
Currently the most widely studied cell source for articular cartilage repair are MSCs. A recent analysis of clinical trial data found that MSCs were being used in more than half of all trials.[ 47 ] (The rest were cartilage‐derived cells as mentioned above, umbilical cord derived‐cells and studies using multiple cell sources.) Within the synovial joint, sources of MSCs with chondrogenic potential include the cambial layers of the perichondrium and the periosteum, the bone‐marrow stroma, the synovial membrane, the synovial fluid and both the infrapatellar and suprapatellar fat pads.[ 51 , 52 , 53 , 54 ] As such autologous mesenchymal cells can be isolated without the need to compromise the native cartilage.[ 55 ] The increasing use of MSCs has led to production processes in accordance with Good Manufacturing Practice (GMP) to allow the culture processes to be reproducible, robust, efficient, and safe.[ 56 , 57 ] Automated cell culture for scale‐up production under GMP conditions has also been demonstrated.[ 58 ]
Here we follow the convention of denoting terminology of MSCs based on function, using “stem” when discussing progenitor cell functionality of self‐renewal and differentiation, and “stromal” when discussing mesenchymal cells with secretory, immunomodulatory and homing properties.[ 59 , 60 ]
MSC based therapies for cartilage repair have shown promising results in Phase I clinical trials, with a prevalence of studies using cells isolated from bone marrow, adipose tissue and the synovium.[ 61 ] The most commonly used MSCs are derived from the bone marrow (BM‐MSCs).[ 62 ] MSCs derived from the infrapatellar fat pad (IFP‐MSCs) have shown promising chondrogenic capacity,[ 63 , 64 , 65 , 66 ] and can be easily accessed in an arthroscopic procedure. In side‐by‐side comparisons of chondrogenic capacity of different sources of MSCs, synovial‐derived MSCs typically display greater chondrogenic potency than adipose or bone marrow‐derived MSCs.[ 67 , 68 ] Song and Jorgensen summarized the benefit of MSCs in preclinical and clinical studies of osteoarthritis treatment and also compare different MSC sources, with the adipose tissue‐derived MSC being superior since they facilitate tissue regeneration and repair by direct tissue differentiation but also by secreting growth factors, cytokines, angiogenic factors, adipokines, and neurotrophic factors to stimulate restoration of normal tissue function or reduce damage.[ 69 ] A 2018 analysis of the trends in clinical trials for articular cartilage repair found a shift toward using cells derived from adipose tissue, rather than from the cartilage and bone marrow.[ 47 ]
The maintenance of the chondrogenic phenotype and avoidance of hypertrophy of MSCs is a challenge in cell‐based tissue engineered strategies, but is being addressed in several promising recent developments. The development of hypertrophic cartilage ends with the onset of endochondral bone formation which has inferior mechanical properties compared to native cartilage. Several in vitro studies have shown that BM‐MSCs produce type X collagen, a marker for hypertrophic cartilage.[ 70 ] Chondrocytes derived from BM‐MSCs implanted into SCID mice recapitulate endochondral bone formation, resulting in the loss of cartilage that is replaced by bone.[ 71 ] However it may be possible to mitigate hypertrophy through careful choice of growth factors and conditions. Several studies have demonstrated methods to minimize the hypertrophy of MSCs once implanted, through co‐culture protocols, through the appropriate choice of culture substrates and conditions, or through intervention by gene or protein delivery.[ 72 , 73 , 74 ] In one recent example Deng et al. developed improved culture conditions to generate minimally hypertrophic chondrocytes from hBMSCs.[ 74 ] Their method involved modulating TGFβ exposure time as well as inhibiting WNT signaling, a pathway involved in the regulation of chondrocyte hypertrophy. The optimized culture conditions successfully suppressed the expression of hypertrophy genes and deposition of collagen type X in vitro. Furthermore, they found that hBMSCs chondoinduced using these optimized conditions did not undergo ossification when implanted intramuscularly in SCID mice. Ectopic bone formation did occur in the control (non‐optimized) group, representing traditional culture conditions for engineered cartilage. This work strongly suggests that the chondrocytic hypertrophy may not be the inevitable outcome for MSCs, and that a more stable phenotype may be achievable through the choice of appropriate differentiation cues. In general, developing MSC‐based strategies for cartilage repair requires an understanding of the mechanisms by which MSCs mediate tissue regeneration in vivo, and this understanding may hinge upon a better integration of tissue engineering with developmental biology.[ 75 ]
3.1.2. Comparison of Ex Situ and In Situ Strategies
A similar range of cell types have been explored for both the in situ and ex situ cartilage engineering strategies. However, the stage of differentiation of the implanted cells can differ between the two approaches, and this can have a marked influence on the resulting tissue. Fully differentiated MSCs in lab‐made constructs stop producing the soluble factors responsible for collagen type II production.[ 76 ] For the in situ approach, it is still debated if the cells need to be pre‐induced toward the desired lineage or if the tissue microenvironment derived from the host or from the implanted biomaterial, is capable of triggering appropriate repair.[ 77 , 78 ] The use of undifferentiated MSCs has added benefits owing to their various stromal effects on native tissue, including release of anti‐inflammatory factors, cytokines, and trophic factors (Figure 2 ).[ 79 ]
Figure 2.
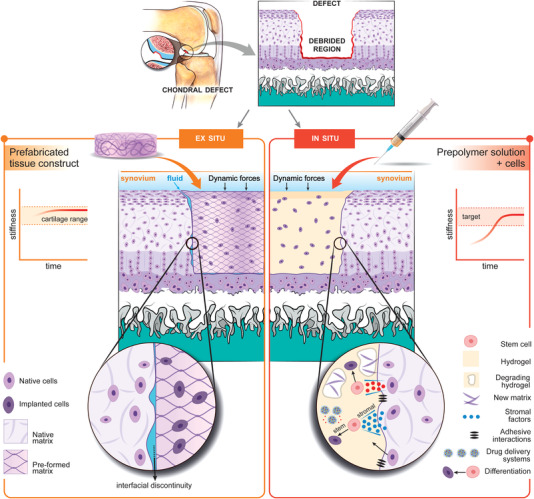
A comparison of the ex situ and in situ cartilage engineering strategies. On the left‐hand‐side we consider an archetypal ex situ cartilage engineering strategy where a cartilage tissue construct is pre‐fabricated and matured in the lab prior to implantation. The pre‐fabricated construct can approximate the stiffness of natural cartilage, however the construct does not exactly match the shape of the debrided chondral defect. Interfacial discontinuities interrupt communication between native and implanted cells, and the infiltration of synovial fluid may negatively impact integration. On the right‐hand‐side we consider an archetypal in situ cartilage engineering strategy where a bioscaffold milieu (hydrogel + mesenchymal stem cells) is delivered arthroscopically to fill the defect site, and the crosslinked in place. The integration of the bioscaffold is an active process as MSCs release stromal factors with anti‐inflammatory and trophic effects upon the host tissue. Growth factor delivery systems as well as exogenous factors contribute to differentiation of MSCs toward the chondrocyte lineage and produce new ECM, which integrates with the ECM of the native tissue. The bioscaffold is initially softer than the surrounding cartilage, but stiffness increases as ECM is released during chondrogenesis.
Whereas an ex situ approach has more opportunities to guide the differentiation along the desired lineage, it is extremely difficult to monitor and control the differentiation of the MSCs after implantation.[ 80 , 81 ] Another challenge is discriminating between the novel tissue created by the host and the cartilage matrix generated directly by the implanted and differentiated cells.[ 23 ]
3.2. Materials Formulation
This section focuses on how the tissue engineering approach (ex situ or in situ) may influence the selection of biomaterials, how they are formulated, and how they are fashioned into an implant.
In general, engineered tissue constructs often employ a biomaterial scaffold to serve as an artificial microenvironment and to provide structural support until native tissue forms. Biomaterials for articular cartilage repair need to satisfy requirements including: Supporting stem cells and/or chondrocytes in a 3D environment; allowing nutrients and newly synthesized matrix molecules to diffuse through the structure; degrading at a suitable rate; and supporting tissue‐specific differentiation. In the case of in situ tissue engineering, the implanted material must also retain suitable mechanical properties while new cartilage is being generated. A wide range of hydrogels have been explored in cartilage tissue engineering, as well as, a suite of other degradable polymer materials which provide mechanical reinforcement.[ 82 , 83 , 84 , 85 ] Naturally‐derived biomaterials may promote induction of chondrogenic differentiation of MSCs.[ 86 ]
Hydrogels are of particular interest in the cartilage engineering field since their highly hydrated environment mimics the native ECM and due to their wide range of biochemical and biomechanical tunability.[ 87 ] Hydrogels are 3D networks of hydrophilic polymers where most of the volume (often more than 90%) is water. This means hydrogels are highly swollen and porous on the molecular scale, allowing diffusive transport of solutes, nutrients and even macromolecules locally produced by cells.[ 84 ] Several chemistries are available which allow cells to be encapsulated with high viability during the polymer cross‐linking process.[ 88 , 89 , 90 , 91 , 92 ] Hydrogels are often used in both in situ and ex situ cartilage engineering strategies as 3D scaffolds for localizing chondrogenic cells, and as a biodegradable template for new hyaline cartilage formation. Several reviews have detailed recent progress in hydrogel design for cartilage repair.[ 84 , 93 , 94 ] Here we focus on how hydrogels are influenced by the choice of ex situ or in situ cartilage repair strategies.
3.2.1. Soft or Stiff? Conflicting Requirements for the Mechanical Properties of Implanted Biomaterials
The biomechanics can be considered from two perspectives: i) How the mechanical properties of the implant affect its participation in load bearing and joint movement and ii) how local stresses and strains affect the differentiation and behavior of encapsulated cells through mechanotransduction mediated signaling pathways.[ 95 ] Mechanotransduction mechanisms that may be relevant during chondrogenesis continue to be elucidated.[ 95 , 96 , 97 ] Numerous studies have consistently found that a softer mechanical environment favors the chondrogenic differentiation of stem cells, compared with stiffer environments.[ 95 ] This requirement for a soft local environment for stem cells stands in conflict with the required load‐bearing properties of the implant as a whole.[ 98 , 99 ] This juxtaposition is not a problem for in vitro engineered cartilage, since constructs can be maintained in culture until robust enough to be implanted and withstand the in vivo loads. Instead, the in situ tissue engineered strategy must adopt a different approach, such as reinforcing a soft, cell‐laden hydrogel with a much stiffer, secondary scaffold to help initially bear the mechanical load.[ 99 , 100 ]
3.2.2. Degradation Rate
The rate of degradation is another important consideration when choosing a biomaterial for cartilage repair and should ideally be tailored to the rate of new matrix formation. In many hydrogel systems degradation rate is closely related to cross‐link density and/or concentration. Several studies have analyzed the relationship between the hydrogel degradation rate and ECM production.[ 101 , 102 , 103 ] Sarem et al., using covalently crosslinked gelatin as a model system, found greater deposition of collagen II in their softer and faster degrading gels (≈0.45 kPa shear modulus and 52% ± 6% mass remaining after 3 weeks incubation) in vitro, compared with their stiffer and slower degrading gels (≈0.75 kPa shear modulus and 68% ± 3% mass remaining after 3 weeks incubation). However, when implanted ectopically in mice their soft gels degraded too quickly (<4 weeks) to retain cartilaginous matrix (i.e., these materials tended to fully degrade in vivo within the four‐week timescale of the experiment).[ 101 ] In another example, MSC chondrogenesis was evaluated in hyaluronic acid hydrogels with tunable crosslink density.[ 103 ] Lower cross‐linked matrix were softer and faster degrading (exhibiting compressive modulus of ≈5 kPa and an enzymatically catalyzed degradation rate of 5.4% ± 0.8% h−1) and showed enhanced chondrogenesis as quantified by the biosynthetic rates of glycosaminoglycan and type II collagen. Gels with a higher degree of cross‐linking were stiffer (≈12 kPa) and slower degrating (3.5% ± 0.1% h−1), resulting in a shift in MSC differentiation toward fibrous phenotypes, with a much lower rate of collagen II production. Our team has studied chondrogenesis of MSCs in gelatin methacrylolyl hydrogels of varying concentrations.[ 102 ] We found that the softest and fastest degrading 6% w/v GelMA materials (≈8 kPa compressive modulus and ≈40% remaining after 28 days in culture) resulted in much higher quality hyaline cartilage in vitro (as quantified by GAG and collagen II production, as well as the lack of collagen X) compared with stiffer and slower grading 10% w/v GelMA (≈35 kPa and ≈60% remaining after 28 days).
While degradable materials are generally proposed for use in the cartilage environment, the rate of degradation in vivo and the fate of degradation products is poorly understood. The degradation rate of biomaterials is typically quantified using subcutaneous implantation models, but such a vascularized environment may not predict degradation rates in avascular cartilage. Likewise, the fate of degradation products within cartilage cannot be presumed to be similar to vascularized tissue, considering the vastly lower rates of molecular transport through cartilage compared to other tissues. The effective pore size in articular cartilage has been estimated at ≈6 nm.[ 104 ] It is unknown which degradation byproducts may be able to diffuse out through the synovial fluid, and which may be trapped within the cartilage itself. Clearance of residual biomaterials is especially important when these molecules may be pro‐inflammatory. Moreover, the ideal degradation rate which retains mechanical load‐bearing capacity throughout the process of neocartilage formation is not well understood. Some efforts have been made to develop more sophisticated models which include simultaneous effects of material degradation and neo‐matrix production on the mechanical properties of engineered tissue.[ 105 , 106 ] While such models can be useful, the ultimate test will likely require long‐term longitudinal studies in animals.
3.2.3. Print Fidelity and Processing Constraints
A perennial challenge in the biofabrication field is to generate 3D structures with high pattern fidelity, while also providing an optimal microenvironment for encapsulated cells. In the case of extrusion based bioprinting, achieving high print fidelity typically involves tailoring the rheological properties of the bio‐ink, by introducing additives or by stabilizing the structure by other means in a layer‐by‐layer fashion. Soft hydrogels can deform or even collapse under their own weight, especially as the size of the structure is increased, requiring the elastic modulus to be increased.[ 107 ]These structural requirements can be in conflict with those which favor cell survival, proliferation and differentiation since MSC chondrogenesis favors a soft microenvironment as discussed above. Thus, satisfying both the requirements for fabrication and biology typically results in a compromise. In the case of in situ delivery however, this requirement is relaxed since the fabrication step involves filling a defect, rather than constructing a freestanding implant. Thus, in situ biofabrication allows for the use of low viscosity, soft biomaterials tailored specifically to the biological requirements, minimally constrained by the fabrication technique.
Some strategies obviate the need for any biomaterials in the bio‐ink. For example, a 3D bioprinting method using scaffold‐free “tissue strands” has been developed where cylindrical filaments were generated by aggregation of primary chondrocytes in an in vitro reservoir.[ 108 ] Tissue strands could be printed without need for a delivering medium or support structure. After culture the sulfated Glycosaminoglycans content was found to be similar to that of native cartilage.
3.2.4. Constraints Due to the Need to Crosslink In Situ
Printing on or within the patient places some limits on the choice of materials, precluding the use of materials which require high temperatures, organic solvents or which produce toxic byproducts during synthesis. Additionally, the in situ approach requires crosslinking reactions to occur relatively rapidly to solidify and fix the material within the surgical timeframe.[ 108 , 109 ] Several chemistries are available which fit this requirement including those based on thermo‐responsive gels,[ 110 ] ionic crosslinking,[ 111 ] and photo‐initiated chemical crosslinking.[ 112 ] As an example, functionalized biopolymers such as gelatin methacryloyl and hyaluronic acid methacrylate can be dissolved with a water soluble photoinitiator and photocrosslinked within seconds of exposure to 400 nm light.[ 113 ] Cells can be damaged in the presence of such reactions, and this cytotoxicity is primarily due to the free radicals generated by the photoinitiator.[ 91 , 114 ] These cytotoxic aspects can be minimized either by sequestering cells away from the photocrosslinkers,[ 114 ] or by tailoring the photocrosslinking conditions to minimize free‐radical flux.[ 113 ]
3.2.5. The Potential of In Vivo Bioprinting to Create More Complex In Situ Constructs
The potential advantages in simplicity offered by the in situ approach can be at the cost of implant complexity. Cartilage consists of multiple zones of varying composition and the ex situ strategy offers more freedom to create a multiphasic construct in terms of materials, cellular content and/or design.[ 24 , 115 ] While the potential benefits of complex chondral implants is a matter of ongoing debate,[ 116 ] and promising results have been achieved using homogenous as well as zonally layered implants.[ 90 , 102 ] the ability to create multiphasic designs is currently limited when the in situ approach is employed. The advent of in situ bioprinting technologies raises the possibility of creating complex, multiphasic and cell‐laden constructs within the defect site.[ 117 , 118 , 119 ] This strategy would circumvent some of the limits of pre‐fabrication by performing the 3D scan after debridement, and using that information to rationally adapt the implant design immediately prior to delivery. The concept of scanning and printing in surgery has already been demonstrated for the treatment of skin wounds using an ink‐jet bioprinter.[ 120 ] An intra‐vital bioprinting concept has also been introduced which allows 3D biomaterial structures to be created within a living animal, without needing a surgical incision to open the tissue.[ 121 ] In situ bioprinting technology is in its infancy however, and will require advances in imaging, artificial intelligence, machine learning and bioprinting hardware to realize its potential. Rapid progress is being made. For example it was recently demonstrated how the movement of the tissue during deposition can be compensated for using closed‐loop feedback and computer vision control systems in an adaptive extrusion printing technology.[ 122 ] Complementary to this is the development of multi‐axis robotic arm based bioprinters which offer the potential to deposit bio‐inks within crevices or under overhangs of native tissue.[ 123 ] Adapting these advances to cartilage repair will require high resolution printing systems, since the heterogeneous layers within articular cartilage have thicknesses on the order of 200 µm, however the lack of a requirement for integrating vascular systems may make cartilage a more accessible target for these technologies compared with other tissues.
3.3. Implantation
This section focuses on the surgical implantation procedure. Simultaneous fabrication and implantation of a scaffold defines the in situ approach, but is challenging step owing to the surgical environment. If the defect is not perfectly filled, this can present barriers for the following steps including complete integration of the implant with the host tissue.
3.3.1. Shape‐Matching
In contrast to the simple cylindrical holes punched out of cartilage for most in vivo studies, chondral lesions often present with extremely heterogeneous topography, showing irregular shape and depth.[ 124 ] While the adoption of additive manufacturing technologies provides new tools to replicate complex geometries in ex situ fabricated implants, this necessitates a highly accurate 3D model of the defect to be filled.[ 125 ] Ascertaining the size and shape of a chondral defect itself is a major challenge. While CT and MRI scans can be used to generate 3D models of cartilage tissue, they can underestimate a lesion size by as much as 60%.[ 128 ] Furthermore, the degenerated cartilage at the fringes of the defect needs to be removed during surgery to allow for graft integration. Precise planning of the debridement step is not possible as it currently relies on the surgeon's in situ assessment of the mechanical integrity of the tissue. As a consequence of these factors, the ex situ tissue‐engineering strategy requires the planned removal of excess healthy cartilage to fit a pre‐fabricated construct, often leaving some inevitable discontinuity at the interface between graft and host.[ 127 ] This practice can lead to cell death at the interface in partial thickness chondral defects[ 128 ] thus hindering integration due to the inability of the cells behind the necrotic area to repopulate the interfacial matrix.[ 129 ]
The in situ tissue engineering strategy is intrinsically shape matching since materials can be crosslinked in situ to fill the defect, regardless of shape.[ 132 ] The surgeon thus has full flexibility to prepare the defect as needed, removing only the damaged tissue to access viable cartilage, without compromising the fit of the bioscaffold.[ 130 ]
3.3.2. Fixation
Robust fixation of implants within the dynamic synovium is a perennial challenge, especially in the case of chondral defects where there is no option to physically anchor to underlying bone. Instead, implants are typically fixed in place with surgical glues, sealants, or suturing,[ 132 , 134 ] and such approaches have had mixed success. For example, fixation to the basal region of the chondral lesion using common fixatives such as fibrin glue did not result in efficient integration in a model of chondral defect in minipigs.[ 136 ] The glue layer may also create a barrier for molecular diffusion and cell migration across the interface,[ 132 , 134 , 135 ] and may also limit integration of neocartilage with native ECM. In situ cartilage engineering strategies face the same fixation challenge, however they allow for the bioscaffold itself to be an adhesive.[ 136 , 137 , 138 , 139 ] The cartilage lesion can be filled with a viscous biomaterial during the process of biofabrication, providing direct fixation of the implant to the cartilage tissue using photoactivation[ 137 ] or enzymatic approaches[ 139 ] thus creating structural and mechanical continuity at the start of the therapy. Realizing the benefits of in situ crosslinking requires great care in practice when delivering cell‐laden biomaterials within a living tissue environment. The defect site is a warm (37 °C), wet environment surrounded by blood and synovial fluid which can dilute the biomaterial, create a movable barrier between the biomaterial and host tissue and/or limit adhesion to the defect.[ 140 , 141 , 142 , 143 , 144 ] Various methods can be used to “dry out” the surgical field which may help with improving contact of the implant with the native tissue.[ 143 , 145 , 146 ]
3.3.3. Open Knee, Arthroscopy, and the Potential for Single‐Stage Procedures
Due to the narrow diameter (≈4 mm) of arthroscopic cannulas, ex situ fabricated cartilage constructs typically require an open or mini‐open knee procedure for implantation.[ 147 ] On the contrary, the in situ strategy has the potential to be paired with arthroscopy techniques.[ 144 , 148 , 149 ] This is highly advantageous as the vast majority of knee cartilage surgeries are performed using arthroscopic methods.[ 150 ] Arthroscopies are shorter and less costly than open surgeries, and lead to faster recovery times with less scarring, and with generally less discomfort and morbidity.[ 151 , 152 ] Developing such a minimally invasive in situ based strategy may ultimately increase the prospects of clinical translation.
A further prospective improvement to the surgical protocol would be to harvest the autologous cells and reimplant them within the same “single‐stage” procedure.[ 153 , 154 ] This possibility is open to in situ tissue engineering strategies, but not to the ex situ approach, since it does not involve a cell culture step. Single‐stage surgery favors cell sources which are easily accessible and able to be rapidly isolated in theatre.[ 153 ] A promising example is the use of adipose derived MSCs that can be isolated arthroscopically from the IFP,[ 155 ] and could successfully drive cartilage repair in preclinical animal models.[ 20 ] Automated devices to isolate stem cells from mixed cell populations are already on the market.[ 156 , 157 ] There is scope to complement this strategy with the incorporation of autologous biological components of medicinal value such as platelet rich plasma (PRP) or bone marrow aspirate concentrate, both of which have shown promise in aiding cartilage repair.[ 158 , 159 , 160 ] From the surgeon and patient's perspective, a minimally invasive, autologous arthroscopic procedure presents a major advantage to save time, reduce cost, and minimize morbidity.
3.3.4. Transport Logistics, Handling, and Sterility
For the ex situ biofabrication strategy, the risk of contamination or damage to the construct is raised due to the long term culture required for in vitro maturation, as well as the extra packaging, transport and handling requirements while preparing the tissue for transplantation. The fabricated construct represents a considerable investment in time and resources, and therefore highly precious and with extremely low error‐tolerance. The components for an in situ tissue engineering are easier to handle and maintain sterility. Since the materials and cells are not combined until implantation, standard protocols can be followed for each. Several methods exist for sterilizing hydrogels while retaining crosslinking and rheological properties, and protocols have been extensively refined for transportation of stem cells for cell therapies.[ 162 , 164 ] Logistics are even simpler for “single‐stage” versions of the in situ strategy where no cell culture is required.
3.3.5. Post‐Operative Care
Following an articular cartilage repair surgery, patients typically follow a rehabilitation program designed to aid the functional recovery and adaptation of the chondral repair tissue. Rehabilitation programs can include protocols for weight bearing, range of motion and motion therapy. Defining an optimum rehabilitation program is challenging owing to the complexity of the repair process, and the consideration of conflicting requirements within the recovering joint. For example, whereas restricting weight‐bearing can protect a new transplant, this restriction can remove mechanical stimuli which are important for the maintenance of healthy articular cartilage, potentially leading to atrophy.[ 163 ] Such restriction might also limit physiological contact forces which are known to enhance chondrogenic differentiation.[ 164 ] Motion therapy is designed to offset some potential complications resulting from being non‐weight bearing by increasing synovial fluid movement and allowing proper joint nutrition. One example of motion therapy is continuous passive motion (CPM), where a device is used to passively move the joint through variable ranges of motion and speeds for periods of hours. Overall, while it is accepted that movement of the joint is beneficial for articular cartilage nutrition, there is limited clinical data to suggest the optimal CPM rehabilitation protocol.[ 167 , 168 ]
In clinical practice rehabilitation programs can be individualized depending on the location of the lesion (e.g., patellofemoral versus tibiofemoral) as well as the patient's age and body mass index.[ 164 , 165 ] Despite some variation in rehabilitation protocols according to the kind of cartilage repair procedure performed, most clinicians conform in avoiding full weight bearing for a minimum of four weeks after surgery, and also in beginning motion therapy within 1 week.[ 164 ] In the longer term, the time to return to active sport can range between 6 and 18 months, with differences based on treatment strategies.[ 168 , 169 ]
Data for post‐operative care following novel tissue engineering implantation is more sparse, and so optimum strategies are yet to be defined.[ 166 ] Though it is well understood that mechanical stimulation is critical for healthy cartilage metabolism, how much joint motion and weight bearing is beneficial (but not detrimental) to the maturation of an in situ engineered bioscaffold is unclear. Some inferences can be made using ex vivo experiments which assess cartilage:scaffold integration under different dynamic loading regimes. In one ex vivo study, in which acellular PVA scaffolds were inserted within a juvenile bovine cartilage explant and cultured for periods of weeks, dynamic loading significantly lower interfacial strength compared with controls in static culture.[ 170 ] The authors highlighted the mismatch between the homogeneous properties of the scaffold and the depth‐dependent properties of the articular cartilage as contributing to the observed abrupt changes in vertical displacement, fluid pressure, and compressive stresses along the interface. These kinds of studies underline the potential importance for non‐weight bearing conditions to allow graft‐host integration for periods of weeks after implantation.
One advantage ascribed to pre‐fabricated scaffolds is their robust mechanical properties which may support earlier weight‐bearing compared to in situ approaches. However, it should be noted that in surveys of current clinical practice, full weight bearing is restricted for a minimum of 4 weeks (and most typically at least 6 weeks) across all surveyed treatment modalities (including microfracture, MACI, and osteochondral grafts).[ 164 ] Thus, placing a premium on immediacy of load‐bearing may not reflect the reality of post‐operative care. On the other hand, motion therapy is widely applied within one week post‐surgery.[ 164 ] With this in mind, the most immediately important mechanical feature of the implant may not be its compressive stiffness (as is typically emphasized in preclinical studies), but rather its ability to withstand the non‐contact, fluidic shear forces generated by motion therapy.
Owing to the lack of clinical evidence, post‐operative care procedures for new treatments are likely to hew close to conventional practice. For example, in a recent clinical trial of a hydrogel scaffold for cartilage repair, CPM was commenced one day after surgery, whilst protected weight bearing did not commence until 6 weeks after surgery.[ 170 ] This protocol broadly matches that recommended by most surgeons following conventional treatments.[ 164 ]
Considering this conservative tendency, the protocols recommended for ex situ and in situ therapies are likely to be similar in the near term. Defining optimum post‐operative care following cartilage engineering therapies will require coordinated efforts of multidisciplinary research teams including surgeons, physiotherapists, and basic scientists. In the preclinical stage this may involve longitudinal studies on large animal models where the joint is periodically imaged by MRI to assess impact of post‐operative procedures.[ 174 ]
3.4. Maturation
In this phase we discuss the development of a cell‐laden bioscaffold toward mature tissue. This necessitates the differentiation of stem cells, the production of proteoglycans and collagen type II (the components predominantly present in native hyaline cartilage)[ 175 ] and the degradation of biomaterial scaffold. In the case of ex situ tissue engineering, this maturation phase occurs in vitro, triggered by exogenous growth factors and/or other stimuli as provided in a bioreactor. Whereas, with in situ tissue engineering strategies, the maturation phase occurs within the living tissue, meaning the bioscaffold must support and orchestrate this process allowing the MSCs to secrete and synthesize proteoglycans and collagens while permitting interaction with the surrounding biophysical cues.
3.4.1. Driving Differentiation: The Need for Growth Factors
Growth factors are considered the gold standard to drive MSCs differentiation in vitro, and although investigations using growth‐factor free chondrogenesis have been proposed,[ 175 , 176 ] the vast majority of studies have used growth factor‐driven MSC differentiation.[ 176 , 177 , 178 ] The TGFβ‐family (including TGFβs and BMPs) is known to bind to receptors type I, II, and III. Activation of these receptors can start cascades which activate SMAD‐dependent and SMAD‐independent signaling, inducing complex‐translocation and subsequent binding to transcription factors (e.g., SOX9), leading to transcription of cartilage ECM components.
In native biological environments, growth factors can be self‐secreted by the cells and the rate of production can be self‐modulated and adapted following cellular responses to achieve cartilage development and homeostasis.[ 179 ] However, this might not entirely satisfy the conditions for differentiation of new MSCs implanted with in situ approaches for cartilage repair, as chondrogenic factors within the mature cartilage tissue are only minimally produced or are bound to the ECM in their inactive form.[ 180 , 181 ] Additionally, the role of those chondrogenic factors is different in an inflamed environment (activation of SMAD linker produced by inflammation can cause post‐translational modifications that disturbs the local TGFβ and BMP signaling pathway), as well as being age‐dependent (e.g., BMP‐7 can be silenced with age).[ 182 ] A further challenge is the possibility of phenotypic instability of stem cells after implantation, where cells may either dedifferentiate, leading to increased production of type I collagen, or undergo hypertrophy, thereby producing mineralized matrix.[ 183 ] In either case, the resulting “mature” tissue possesses inadequate mechanical properties and the therapy is liable to fail.
For these reasons it is advantageous, and potentially necessary, to deliver biologicals (growth factors and or gene regulators, e.g., RNA) to help trigger or enhance chondrogenesis in situ.[ 184 , 185 , 186 ] The maturation rate will then follow the natural cellular process controlled by the local feedback response from the implanted cells and the surrounding environment until a homeostatic state has been reached, whilst taking advantage of the possible nutrient diffusion and mechanical forces within the body.[ 187 , 188 ] In early tissue development, it is expected that the biophysical interaction between the native environment and the bioscaffold will trigger nutrient mass transport by diffusion in the direction of the concentration gradient[ 189 ] whilst in further homeostatic state the mechanical forces in the joint might influence the growth factor maintenance and promote activation.
3.4.2. The Challenge of Controlled Release of Growth Factors In Situ
The production of neo‐cartilage tissue within a native environment, comes with various challenges. For instance, in ex situ fabrication approaches the replenishment of growth factors is done periodically by replacing the cell culture media. However, in situ engineering strategies may require pre‐loading with sufficient factors to maintain a chondrogenic phenotype for the duration of the chondrogenic process. To this end, the fabrication of growth factor delivery systems, or cellular genetic modifications to deliver chondrogenic factors, has been the focus of several investigations without a consistent agreement in method, loaded concentration, and optimal release profile.[ 190 , 191 , 192 , 193 ] From the growth factor delivery perspective, control over the spatio‐temporal release of factors to the cells must be aligned with cellular requirements, which can be complex or even impossible to achieve. Pivotal components of the design of a delivery system include the growth factors’ intrinsic characteristics (e.g., molecular weight, structure), material selection (e.g., water‐solubility), and the system itself (e.g., microparticles); only if these components are in synchrony with the cellular processes can tissue maturation be achieved.[ 197 ]
The successful strategy for growth factor delivery must be carefully designed to present the factor in their active conformational state to be recognizable for the cell receptors.[ 194 , 195 , 196 ] The preservation of growth factors is often a difficult task as several growth factors have short half‐lives or are highly sensitive to degradation and denaturation.[ 197 ] In the case of RNA integrating strategies, the challenge relies on its high sensitivity to degradative enzymes and hydrolysis leading to an inherent instability.[ 198 , 199 ] To cope with these scenarios, some approaches have tended to increase the amount of growth factors within bioscaffolds, at the expense of potential detrimental effects.[ 200 ] In particular, for 3D bioprinting the inclusion of growth factors must avoid high temperatures or the use of organic solvents to prevent denaturation, using water‐soluble hydrogels to retain or encapsulate the factors can avoid this problem.[ 201 , 202 ]
3.4.3. Biomechanical Cues
In in vitro tissue engineering, recapitulating mechanical stimuli has been shown to enhance cartilage formation.[ 18 , 203 ] As an example, Behrendt et al., have reported that simulated joint loading conditions could induce tissue maturation by activation of TGFβ‐1 in cell‐laden hydrogels.[ 204 ] In vitro bioreactors are designed to recapitulate the biomechanical and biochemical milieu of the joint. The most sophisticated bioreactors can combine multiple kinds of biomechanical stress (e.g., shear and compression),[ 208 ] but these are still highly simplistic representations of the in vivo system. Bioscaffolds undergoing in situ chondrogenesis within the defect site are exposed to a plethora of biomechanical and biochemical stimuli, including tissue specific oxygen levels, nutrient diffusion gradients, and exogenous growth factors.[ 206 ] As an example of the such dynamic complexity within the synovial joint, it has been shown that under loading of healthy cartilage, latent TGFβ (in the synovial fluid and bound to the ECM), becomes active by dissociation from the latency‐associated peptide complex, and this contributes to maintenance of a homeostatic state.[ 182 , 207 ]
3.4.4. Other Challenges and Unknowns
Spatiotemporal control is more challenging when multiple growth factors are employed to enhance chondrogenic tissue formation.[ 208 , 209 ] For example, TGFβ‐1 can enhance BMP‐2 induced chondrogenesis.[ 210 , 211 ] Furthermore, the precise time of uptake and production of each of the chondrogenic factors during the chondrogenic process and their role in late stages of maturation remains largely unknown. Some chondrogenic factors play dual roles within the cartilage environment (e.g., TGFβs have a chondrocyte phenotype maintenance role in healthy cartilage but are also speculated to contribute to degenerative conditions such as osteoarthritis) therefore their contribution to the MSC differentiation upon in situ implantation remains unclear.[ 182 ] The synergy between uptake and release for each factor must be refined and new cell interrogation approaches, such as real‐time tracking of growth factor production/consumption versus chondrogenic expression and ECM component secretion (e.g., glycosaminoglycans, collagens) must be developed.[ 212 ] Additionally, the investigation of how inflammation at the site of a cartilage injury may affect de‐novo tissue formation has yet to be elucidated, some discussion around this has been included in the section on Integration.
3.5. Integration
As noted by Khan et al., “Cartilage repair without vertical and lateral integration is destined for failure.”[ 213 ] This section describes how the in situ biofabrication approach impacts the integration of an implant in a chondral defect where integration is the processes leading to the basolateral, morphomechanical continuity between exogenous implants, and endogenous cartilage tissue in absence of a foreign body response.
3.5.1. Failures of Integration for Pre‐Fabricated Cartilage Constructs
In the current state of the art, ex situ biofabrication strategies have shown promise for their ability to create complex tissue analogues ex vivo.[ 90 ] However, the integration of these pre‐formed constructs with the native tissue remains a formidable challenge, despite the exploration of a range of sophisticated strategies including chemotactic biomolecules, interfacial adhesives, ECM digestion, and extracellular vesicles.[ 214 , 215 ] As an example. the lack of integration in osteochondral implants could be responsible for unbalanced mechanical stress, synovial fluid infiltration, and accumulation of fibrotic tissue, which in turn can cause deterioration of the implanted bioscaffold.[ 215 , 216 , 217 ] One potential explanation for the failure of ex situ engineered cartilage constructs is due to the relatively inactive microenvironment at the interface—where a pre‐formed construct is next to a cartilage tissue, with both sides populated by mature but sedentary chondrocytes. On the contrary, the in situ tissue engineering strategy has the potential to affect an active processes of integration at the interface by taking advantage of both the stem and stromal effects of implanted mesenchymal cells.
One critically important, but often overlooked, requirement is the need for the implant to retain its shape throughout the degradation and remodeling process. Especially when in soft environments, cells undergoing differentiation and producing ECM can induce remodeling of their surroundings which may result in a change, including shrinkage, of the implant from its original size and shape both in vitro and in vivo.[ 215 , 218 , 219 ] Any gaps resulting from this remodeling may limit integration between the implant and host. Mechanical reinforcement scaffolds can potentially play a secondary role here by maintaining shape integrity during these dynamic processes.
Besides these challenges, one limitation in the field is the lack of standardization of analytical techniques which specifically investigate the interaction between the exogenous implant and the endogenous tissue at the interface. Currently, the measurement of the “push‐out” displacement forces of circular implants in cartilage explants is emerging as a common approach to quantify biomechanical integration,[ 221 , 223 ] though no standard protocol has yet been defined.
3.5.2. Active Matrix Deposition Aids Integration
The ability of MSCs to actively deposit matrix during chondrogenic differentiation represents an integrative advantage with respect to a pre‐fabricated tissue. Seminal work has shown that the active deposition of collagen fibers contributes to an increased functional integration at cartilage interfaces as measured by adhesion strength.[ 222 ] In another classic study, where engineered cartilage constructs were inserted in rings of cartilage explants, tissue remodeling, and adhesion strength was higher for immature constructs (cultured for 5 days prior to insertion) than for mature constructs (cultured for 5 days prior to implantation) or even for cartilage explants.[ 223 ] The authors concluded that the “most important factor for integrative tissue repair is the presence of biosynthetically active cells, capable of proliferating, filling the gaps at tissue interface, and progressively forming cartilaginous tissue.” In support of this, the proliferative status and high cell density at the interface has been reported to improve integrative cartilage repair in a cartilage ring bovine repair model,[ 224 ] where the interface was pretreated with collagenase. Such collagenase treatment can also enhance cell proliferation.[ 228 ] In fact, previous studies showed a key role of metalloproteinases in the chondrogenic process where impairment of ECM production occurred after inhibition of MMP‐2 and upregulation of collagenases MMP‐2, MMP9, and MMP‐13 was observed in chondrogenic cells.[ 226 ]
The active integration process can be modulated by the physical properties of the scaffold material (e.g., porosity, permeability, and stiffness), however. This was shown in a cartilage integration model using collagen depositing chondrocytes embedded in concentrations of agarose ranging from 0.5 kPa (0.5% w/v) to 23 kPa (10% w/v).[ 227 ] At week 12, the quality of integration at the hydrogel‐cartilage boundary was generally higher for the lower stiffness samples, as quantified by integration strength. In this case the higher concentration hydrogels (with their initial high stiffness and lower permeability) may have impeded diffusion of ECM to the interface and/or slowed nutrient transport which would further limit tissue development and regeneration.
3.5.3. Mutual Influence of Implanted MSCs and the Host Environment
The in situ cartilage engineering approach allows the application of mesenchymal cells capable of both “stem” and “stromal” functions. The stem function is evident in the in situ differentiation of mesenchymal cells and the production of cartilage matrix. As recently demonstrated in an animal study, the native cartilage niche is the determining factor capable of regulating BM‐MSCs differentiation down a chondrogenic lineage, and the implanted stem cells are the cell types responsible for producing neocartilage in the chondral defect.[ 228 ] Implanted mesenchymal cells can also release a range of stromal factors which can modulate inflammation, and perform trophic and chemotactic activities.[ 229 , 230 ] At the interface between the defect and the native cartilage, these factors stimulate cartilage ECM remodeling and CSPCs for differentiation, a peculiar feature that the mature chondrocytes do not display.
The anti‐inflammatory capability of MSCs has been demonstrated in clinical results using MSCs as advanced medicinal products (ATMP), where MSCs were found to play a critical role in the two phases of inflammation essential for an effective healing process and the prevention of further degeneration of the cartilage tissue.[ 231 ] This capability has been demonstrated to be superior to derived products like PRP.[ 232 ] MSCs can also play a key role in the response to tissue injuries by inducing repair/regeneration processes at the injury site,[ 233 , 234 ] and are known to release trophic factors involved in cell survival, proliferation, and differentiation such as FGF, IGF, and TGFβs.[ 235 , 236 ] MSCs have also shown the ability to induce cartilage regeneration by performing trophic functions on resident cells.[ 237 ] In cartilage generation studies in vitro MSCs have been proven to improve cartilage formation of chondrocyte in co‐culture experiments when compared to the role of individual populations.[ 238 , 239 , 240 , 241 ] Cartilage defects treated with arthroscopic delivery of LI‐labelled synovium derived MSCs in pigs showed that the MSCs triggered cartilage repair even though their presence was trackable only up to 7 days.[ 242 ] Regeneration was assessed over 3 months with newly synthesized cartilage matrix observed in the treated condition but absent in the untreated group, a possible indication of their role in the stimulation of resident cell regenerative capacity. The trophic effects of MSCs have been exploited in co‐culture with chondrocytes in vivo on biodegradable scaffolds[ 246 ] and also in a recent clinical trial where allogeneic MSCs were implanted in a mixture with autologous chondrons.[ 244 ]
MSCs also have a direct chemotactic action on the host's resident cells and can trigger their recruitment to the wound site, thus contributing further to the integration process. Chemotactic molecules such as CCL2/MCP‐1, CXCL8/IL‐8, CXCL12/SDF‐1 are expressed in MSCs[ 248 ] and they have been shown to stimulate the migration of bone marrow MSCs, synovium derived MSCs, adipose derived MSCs, and chondroprogenitor cells.[ 246 ]
To conclude, the complete regeneration process may be coordinated by multiple mechanisms, and stem cells may play different roles in different stages of the actual repair process. Modulating the two main roles of MSCs (stem and stromal) might lead to the desired regeneration effect.[ 250 ]
3.6. Validation and Clinical Translation
3.6.1. Overview
This section is focused on how the preclinical validation and clinical translation stages of product development may differ between the ex situ and in situ approaches. It includes discussion of biological and biomechanical characterization, quality control aspects, regulatory considerations, clinical trial design, cost, and uptake.
3.6.2. Animal Models
One question pertinent to the translation of cartilage engineering in general is, what animal models can be used which best mimic the integration and remodeling characteristics of human cartilage? Small animal models, such as rabbits, are often used in early stage research however these models are problematic owing to their greater self‐repair capacity compared with humans.[ 248 ] Large animal models such as sheep, pig, and horse are potential candidates suitable for studies on defect diameters comparable to those found in humans.[ 249 , 250 ] However, these models still come with limitations, most notably cost, ethical concerns, non‐representative biomechanical strains, difficulties to accurately control postoperative joint loading during rehabilitation and potentially different rates of healing.[ 251 ]
3.6.3. Selection of Patients
The selection of patients that will benefit from this type of in situ or ex situ tissue engineering strategy must be determined. The available studies allow us to draw some indications. As reported in their systematic review, Filardo and colleagues reported no major adverse events related to the treatment or cell harvest in studies using MSCs as cellular therapies to treat cartilage lesions.[ 252 , 253 ] Clinical benefit is also reported regardless of cell‐source or administration method, with better outcomes in patients with young age, lower BMI, and smaller focal lesion size. These results must be considered also from a cost‐effectiveness and efficacy standpoint.
Autologous cell‐based therapies may be subject to variability due to the varying chondrogenic potential of the individual patient's cells, that is, due to sex, age, and genetic differences.[ 79 ] For in situ cartilage regeneration strategies, there is an additional factor of variability associated with the maturation process in vivo. Identification of specific markers of integrative potential could enable preclinical screening to assess suitability of patients for an in situ tissue engineering therapy.
Only the combined assessment of the balance between the outcomes from different studies and the costs of the products will consolidate the promising results of experimental trials in clinical practice[ 257 ] and will help to determine the specific cohort of eligible patients.
Achieving this will require robust, prospective randomized control clinical trials which are adequately powered, with patients undergoing precise post‐operative rehabilitation regimens and long term follow‐up, but such studies are still lacking in the literature on stem cell therapies for cartilage repair.[ 258 ]
3.6.4. Quality Control Measures
The regulation of personalized medical devices is complicated by the challenge of implementing quality control mechanisms which are valid for “batches of one” products made on demand.[ 256 ] This challenge may be exacerbated for treatments incorporating autologous cells considering the additional sources of patient variability: Unknowns around the capacity of that patient's stem cells to undergo chondrogenic differentiation in situ as well as unknowns around the inflammatory environment of that patient's knee joint.
An in situ approach also influences the implementation of quality control measures. Whereas a bench based fabricated tissue can undergo non‐destructive testing to ensure it meets certain mechanical and biological criteria prior to implantation, an in situ approach relies on more significant remodeling after implantation, limiting the quality control testing that can be performed prior to implantation.
These additional challenges may be alleviated by developments in novel non‐destructive techniques for interrogating 3D tissues.[ 257 ] Magnetic resonance spectroscopy, imaging and elastography have been utilized to assess ECM composition, tissue microstructure, porosity and tissue stiffness.[ 258 ] Magnetic resonance imaging has also been used in a pilot human clinical study to assess defect filling, tissue maturation, tissue integration, and new tissue development over 6 months.[ 259 ] Dalecki et al. obtained a 3D quantitative visualization of collagen microstructure based on ultrasonic integrated backscatter coefficient parametric images and demonstrated a correlation between cell localization and collagen fiber density or thickness.[ 260 ] ECM deposition and remodeling can be tracked using reporter gene‐based imaging in vivo.[ 257 ]
3.6.5. Regulatory Challenges
While the regulatory frameworks around new treatments can differ greatly depending on the classification of the medical device, general requirements include regulatory approval, certification of the manufacturing facility, and certification of the quality control processes. The requirements for a particular strategy depend strongly upon which classification is applied. The in situ cartilage engineering approach bears some similarities with existing, clinically approved treatments (such as MACI) which may aid its pathway through regulation. The avoidance of a pre‐fabrication step also simplifies the process in that it reduces the requirements around certifying a clinical grade manufacturing facility. It also simplifies the logistics of preparing the surgery: A pre‐fabricated “tissue” would need to be transported from the manufacturing facility under a highly controlled environment in a time‐sensitive manner.
The burden of proof may be higher for in situ tissue engineered treatments where stem cells implanted into the body are still in a multipotent state, raising questions about potential hyperplasia, differentiation along undesirable pathways, and/or tumorigenic transformation.[ 261 , 262 ] In general, a regenerative event always seeks to maintain or to re‐establish the form and the function of tissue cells, a process called morphostasis.[ 263 ] However, regeneration could involve the emergence of abnormal growth, which may lead to the onset of cells with an aggressive phenotype.[ 264 ] Cells incorporated in ex situ fabricated tissue constructs would likely be at a more terminal stage of differentiation and so pose less of a risk of undesired transformation.
On the other hand, treatments which employ a “single‐stage” process, where the patient's cells are harvested, processed and reimplanted without leaving the operating theatre, may be subject to a less stringent regulatory pathway considering that a GMP cell processing facility would not be required. Clinical grade cell culture is generally expensive, time consuming and associated with intrinsic risks due to the substantial manipulation and ex vivo cell expansion.
3.6.6. Ease of Translation and Clinical Acceptance
Translation of the ex situ tissue engineering strategies are complicated due to the requirement for fabrication equipment and in vitro pre‐maturation in a GMP grade facility, along with stringent transportation requirements from the lab to clinic. The in situ tissue engineering strategy greatly simplifies this complexity, reducing cost (less equipment, lab space, fabrication team, time, transport, consumables, and material needed) allowing the end product/process to be more accessible. The development of an arthroscopic surgical approach would further reduce cost, and could improve the adoption of the technique considering it is greatly preferred by surgeons. We agree with Bernhard and Vunjak‐Novakovic that “[t]he simplest and most robust method for achieving durable cartilage repair will certainly have the highest chance of clinical acceptance.”[ 265 ]
3.6.7. Results from Pre‐Clinical and Clinical Trials
Here we briefly highlight the status of both ex situ and in situ cartilage repair strategies in pre‐clinical and human clinical trials.
Ex situ strategies (especially using advanced fabrication techniques) are primarily in the pre‐clinical stage, and typically target osteochondral defects. A large number of such biofabrication studies assess capacity for repair in small animal models, especially rabbits, and these have been reviewed by others.[ 115 , 266 ] Only a few of these advanced strategies have been applied to large animal models, and none yet to humans. For example, He et al generated cartilage ex situ by culturing autologous BM‐MSCs on a PGA/PLA scaffold for various time periods (2, 4, or 8 weeks) before implanting in a pig model of an osteochondral defect. Six months after implantation, the mechanical properties, collagen content and GAG content of the repaired cartilage approached that of native cartilage. Interestingly, the maturity level of the ex situ engineered cartilage had a significant influence on the repaired results, with the 4 and 8 week cultured constructs superior to the 2 week constructs. Satisfactory integration with native cartilage was also reported. This level of integration is in contrast to an early study where scaffold‐free cartilage engineered in vitro was implanted in a minipigs, resulting in gross repair of cartilage defects but poor integration with native cartilage.[ 267 ] Further in vivo studies (especially in large animal models) are required to understand the potential for cartilage repair using ex situ engineered cartilage implants.
Studies applying in situ cartilage engineering strategies tend to dominate in late pre‐clinical and human clinical trials. A recent (2021) systematic review and meta‐analysis included 50 clinical trials of hydrogel implants for cartilage repair, comprising 2846 patients in total.[ 268 ] Of these studies, 24 involved cell‐based hydrogel implants (986 patients) while 1860 patients received hydrogel without cells. All of these included trials would qualify as in situ cartilage engineering according to our distinction. Most trials were for variations of the MACI/MACT treatment (where autologous chondrocytes are seeded on hydrogel implants) or variations of material aided microfracture (where an implanted or injected material is used to help localize and/or stimulate repair by the released BM‐MSCs). In either case the hydrogel implants were often commercially available products (e.g., Cartipatch, Hyalograft C, Tisseel fibrin, Bioseed C, CaReS, Novacart, and ChondroDux). Studies where MSCs are retrieved and reimplanted are more rare. In one notable example, a human trial was performed where autologous chondrocytes and bone marrow cells were retrieved and re‐implanted with a 3D printed degradable scaffold, all in single‐stage surgery.[ 157 ] Improved IKDC scores were noted from 12 months. Two years after surgery, haline‐like cartilage in over half of patients, but most also contained fibrocartilage.
In Table 1 below, summarize the outcomes from a selection of representative pre‐clinical and clinical trials.
Table 1.
Selected results from pre‐clinical and clinical trials of ex situ and in situ cartilage repair strategies
| Reference | Repair category | Method/material | Cell source and preparation | Model, defect type | Study length | Outcome |
|---|---|---|---|---|---|---|
| Ex situ | ||||||
| Mainil‐Varlet et al. 2001 [267] | In vitro cartilage | Implantation of cartilage construct engineered in an in vitro bioreactor. No biomaterials used. | Allogeneic pig chondrocytes cultured for 3–4 weeks in a scaffold‐free bioreactor system. | Minipig. Chondral, full thickness, and superficial osteochondral defects | 6 month | Tissue engineered implants resulted gross repair of cartilage defects but poor integration with native cartilage. |
| He at al., 2017 [269] | In vitro cartilage | In vitro cartilage generated with 2, 4, or 8 week culture of BM‐MSCs on PGA/PLA scaffold | Autologous BM‐MSCs | Pig model. Osteochondral defect. | 6 month | Mechanical properties, collagen content, and GAG content approached that of native cartilage. 4 and 8 week cultured constructs superior to 2 week constructs. |
| In situ | ||||||
| Nehrer et al, 2009 [270] | MACI/MACT | Hyaluronan‐based scaffold seeded with autologous chondrocytes. (Hyalograft C) | Arthroscopic biopsy to collect cartilage and isolate chondrocytes. | Human. Full thickness. | 7 years | Significant increase in Lysholm score from 1 year post‐surgery (young patients only) |
| Crawford et al., 2012 [271] | MACI/MACT | Chondrocytes seeded on a bovine type‐I collagen matrix. (NeoCart) | Arthroscopic biopsy to collect chondrocytes. | Human. Full thickness. | 2 years | Significantly higher “change from baseline” IKDC score for the NeoCart group vs microfracture |
| Sharma et al., 2013 [259] | Material aided Mfx | Hydrogel implantation in conjunction with microfracture. Used a novel chondroitin sulfate adhesive, photo‐polymerized in situ. (CondroDux) | Infiltration of bone marrow MSCs from microfracture procedure | Human. Full thickness. | 6 month | Implant + MFx showed higher tissue fill compared to MFx only control. Pain reduced in treatment group versus controls, but similar IKDC scores. |
| Wong et al., 2013 [272] | Intra‐articular injection | Cell‐recipient group received intra‐articular injection of cultured MSCs with hyaluronic acid, control group only received hyaluronic acid | Autologous BM‐MSCs expanded in culture for 3 weeks | Human. Osteoarthritic knees. | 2 years | Clinical (inc. IKDC) and MOCART scores significantly higher for cell‐recipient group compared with hydrogel only group one year post‐treatment. |
| Jo et al., 2014 [273] | Intra‐articular injection | Arthroscopic intra‐articular injection of autologous adipose tissue derived MSCs in three dose cohorts | Autologous AD‐MSCs from abdominal subcutaneous fats and expanded in culture for 3 weeks | Human; patients with osteoarthritis | 6 month. | High‐dose group showed improved WOMAC score and decreased defect size 6 months after injection. |
| Mumme et al., 2016 [274] | MACI/MACT | Chondrocytes seeded into collagen type I/III membrane. (Chondro‐Gide) | Autologous nasal chondrocytes seeded onto membrane 2 weeks prior to implantation. | Human. Full thickness cartilage defect. | 2 years | Significant increase in IKDC scores and KOOS scores. Significant decrease in MOCART. (No control group) |
| D'Ambrosi et al., 2019 [275] | Material aided Mfx | Arthroscopic treatment using resorbable polylactide‐coglycolide copolymer implant. (TruFit) | Infiltration of MSCs from Mfx | Human. Full thickness focal lesion. | 8 years | Significant increase in HSS and KOOS score compared with baseline. (No control group.) |
| Słynarski et al., 2020 [154] | Single‐stage surgery | 3D printed scaffold made of polyethylene glycol terephthalate and polybutylene terephthalate. (INSTRUCT) | Chondrocytes and bone marrow cells retreived and re‐implanted in single‐stage surgery | Human. Osteochondral defect. | 2 years | Hyaline‐like cartilage in over half of patients, but most also contained fibrocartilage. Improved IKDC score from 12 months. |
| Sennett et al., 2021 [276] | Composite material aided Mfx | Composite scaffold of a woven PCL mesh + hydrogel (RADA self‐assembled peptide or thermogelling hyaluronic acid) in conjunction with microfracture | Infiltration of BM‐MSCs from Mxf, or injection of autologous bone marrow aspirate. | Minipig. Full thickness. | 12 month | Long term results counter to short term – no benefit to PCL scaffolds or hydrogels observed and worse outcomes than microfracture treatment alone |
| Both in situ and ex situ | ||||||
| Di Bella et al., 2018 [20] | In situ surgical printing of MSCs | Compared in situ printed HA‐GelMA bioink and bench‐based printed bioscaffold pre‐seeded with MSCs | Sheep adipose‐derived stem cells. Ex situ group (MSCs seeded 1 week prior to implantation) | Sheep. Full thickness | 8 weeks | Improved neocartilage observed in treatment group, significantly higher O'Driscoll score than microfracture group but not ex situ group or control |
Mfx = microfracture; IKDC = International Knee Documentation Committee subjective knee evaluation score; KOOS = Knee Injury and Osteoarthritis Outcome score, MOCAT = Magnetic Resonance Observation of Cartilage Repair Tissue; WOMAC = Western Ontario and McMaster Universities Arthritis Index; HSS = Hospital for Special Surgery Score.
4. Conclusion
Current tissue engineering strategies are generally limited in their ability to affect long term repair of hyaline type articular cartilage and to achieve contiguous integration with host tissue. In this perspective review we have compared the challenges and opportunities of ex situ and in situ tissue engineering for cartilage repair (Table 2 ).
Table 2.
A summary of key findings from our comparison of ex situ and in situ tissue engineering strategies for cartilage repair
| Ex situ | In situ | |
|---|---|---|
| Cell preparation |
|
|
| Materials formulation |
|
|
| Implantation and post‐operative care |
|
|
| Maturation |
|
|
| Integration |
|
|
| Validation and clinical translation |
|
|
The ex situ strategy benefits from its ability to create functional constructs which recapitulate the complexity of native tissue, including multi‐layered and/or multicomponent structures, tuned to match the biomechanics of native tissue. They also arguably benefit from a more reproducible and controlled production, since the fabrication step can be automated using additive manufacturing tools, and the maturation phase proceeds in a controlled environment. Following this process, ex situ strategies can produce biomechanically mature constructs with high compressive strength. Nevertheless, seamless integration of ex situ constructs remains a challenge.
The in situ tissue engineering strategy can take advantage of the beneficial activity of encapsulated mesenchymal cells, possibly resulting in a more active integrative process than occurs when pre‐fabricated constructs are implanted. The stromal effects of undifferentiated MSCs may reduce inflammation, and stimulate activity of native cells through the release of trophic factors. Differentiation and activity of implanted MSCs can be advantageously modulated by the biomechanical and biochemical environment. Crucially, the generation of new matrix in situ has been shown to improve integration with host tissue. The act of fashioning the implant in situ can also aid integration due to the ease of creating a “perfect fit” implant, as long as shape integrity can be maintained during the remodeling process. To be widely adopted, the approach should be amenable to arthroscopic delivery. While methods, where cells are minimally manipulated, especially “one‐stage” procedures where autologous cells do not leave the surgical field, have advantages in terms of rapid translation.
To realize these opportunities, several challenges with the in situ tissue engineering strategy should be overcome. The relatively soft environment favorable to neocartilage formation is not suitable for load bearing, and so reinforcement structures may be required to maintain mechanical integrity of the implant in situ. Monitoring and validating the production of matrix in situ, and distinguishing which cells are responsible for neocartilage formation is another area that needs further development. Due to the potential variability in regenerative capacity between patients, it may be important to select prospective patients by screening biomarkers of autologous cells.
Studies which directly compare ex situ and in situ cartilage engineering strategies (i.e., including similar cell sources and biomaterial components for each case) are currently lacking. There is also potential for hybrid strategies which marry the best features of both ex situ and in situ chondrogenesis. For example, where a cartilage construct is created using ex situ fabrication and matured in vitro, but where integration is aided using adhesive gel loaded with MSCs. Much further work is required to understand whether true human cartilage repair is most likely to eventuate through creating new cartilage tissue “within” or “without” the patient's body.
Conflict of Interest
The authors declare no conflict of interest.
Acknowledgements
Funding for this work was gratefully received from the Victorian Medical Research Acceleration Fund (2018, Round 2), NHMRC‐MRFF Investigator Grant (Di Bella) (#1193897), and the RMIT Vice‐Chancellor's Fellowship scheme. The authors are grateful to Prof. Enrico Lucarelli for feedback and useful discussions. Graphics for figures 1 and 2 were kindly provided by Maria Pia Cumani (Rizzoli Orthopaedic Institute, Bologna, Italy). After initial online publication, the acknowledgemet to Maria Pia Cumani was added on December 21, 2022.
Open access publishing facilitated by The University of Melbourne, as part of the Wiley ‐ The University of Melbourne agreement via the Council of Australian University Librarians.
O'Connell C. D., Duchi S., Onofrillo C., Caballero‐Aguilar L. M., Trengove A., Doyle S. E., Zywicki W. J., Pirogova E., Di Bella C., Within or Without You? A Perspective Comparing In Situ and Ex Situ Tissue Engineering Strategies for Articular Cartilage Repair. Adv. Healthcare Mater. 2022, 11, 2201305. 10.1002/adhm.202201305
References
- 1. Davies‐Tuck M. L., Wluka A. E., Wang Y., Teichtahl A. J., Jones G., Ding C., Cicuttini F. M., Osteoarthritis Cartilage 2008, 16, 337. [DOI] [PubMed] [Google Scholar]
- 2. Long H., Liu Q., Yin H., Wang K., Diao N., Zhang Y., Lin J., Guo A., Arthritis Rheumatol. 2022, 74, 1172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Kingsbury S. R., Gross H. J., Isherwood G., Conaghan P. G., Rheumatology 2014, 53, 937. [DOI] [PubMed] [Google Scholar]
- 4. Barbour K. E., Helmick C. G., Boring M., Brady T. J., MMWR. Morb. Mortal. Wkly. Rep. 2017, 66, 246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Australian Bureau of Statistics , National Health Survey: First Results 2017–18, Canberra, 2018. [Google Scholar]
- 6. Smith G. D., Knutsen G., Richardson J. B., J. Bone Jt. Surg., Br. Vol. 2005, 87, 445. [DOI] [PubMed] [Google Scholar]
- 7. Montgomery S. R., Foster B. D., Ngo S. S., Terrell R. D., Wang J. C., Petrigliano F. A., Mcallister D. R., Knee Surg. Sports Traumatol. Arthrosc. 2014, 22, 2070. [DOI] [PubMed] [Google Scholar]
- 8. Mithoefer K., Mcadams T., Williams R. J., Kreuz P. C., Mandelbaum B. R., Am. J. Sports Med. 2009, 37, 2053. [DOI] [PubMed] [Google Scholar]
- 9. Liu Y., Shah K. M., Luo J., Front. Bioeng. Biotechnol. 2021, 9, 770655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Stampoultzis T., Karami P., Pioletti D. P., Curr. Res. Transl. Med. 2021, 69, 103299. [DOI] [PubMed] [Google Scholar]
- 11. Vinatier C., Mrugala D., Jorgensen C., Guicheux J., Noël D., Trends Biotechnol. 2009, 27, 307. [DOI] [PubMed] [Google Scholar]
- 12. Zhang L., Hu J., Athanasiou K. A., Crit. Rev. Biomed. Eng. 2009, 37, 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Cooper S. M., Rainbow R. S., Tissue Eng., Part B 2021, 28, 995. [DOI] [PubMed] [Google Scholar]
- 14. Groll J., Boland T., Blunk T., Burdick J. A., Cho D.‐W., Dalton P. D., Derby B., Forgacs G., Li Q., Mironov V. A., Moroni L., Nakamura M., Shu W., Takeuchi S., Vozzi G., Woodfield T. B. F., Xu T., Yoo J. J., Malda J., Biofabrication 2016, 8, 013001. [DOI] [PubMed] [Google Scholar]
- 15. Castilho M., Mouser V., Chen M., Malda J., Ito K., Acta Biomater. 2019, 95, 297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Askari M., Naniz M. A., Kouhi M., Saberi A., Zolfagharian A., Bodaghi M., Biomater. Sci. 2021, 9, 535. [DOI] [PubMed] [Google Scholar]
- 17. Groen W. M., Diloksumpan P., van Weeren P. R., Levato R., Malda J., J. Orthop. Res. 2017, 35, 2089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Meinert C., Schrobback K., Hutmacher D. W., Klein T. J., Sci. Rep. 2017, 7, 16997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Hauptstein J., Böck T., Bartolf‐Kopp M., Forster L., Stahlhut P., Nadernezhad A., Blahetek G., Zernecke‐Madsen A., Detsch R., Jüngst T., Blunk T., Adv. Healthcare Mater. 2020, 9, 2000737. [DOI] [PubMed] [Google Scholar]
- 20. Di Bella C., Duchi S., O'connell C. D., Blanchard R., Augustine C., Yue Z., Thompson F., Richards C., Beirne S., Onofrillo C., Bauquier S. H., Ryan S. D., Pivonka P., Wallace G. G., Choong P. F., J. Tissue Eng. Regener. Med. 2018, 12, 611. [DOI] [PubMed] [Google Scholar]
- 21. Brittberg M., Am. J. Sports Med. 2010, 38, 1259. [DOI] [PubMed] [Google Scholar]
- 22. Nixon A. J., Rickey E., Butler T. J., Scimeca M. S., Moran N., Matthews G. L., Osteoarthritis Cartilage 2015, 23, 648. [DOI] [PubMed] [Google Scholar]
- 23. Malda J., Groll J., Van Weeren P. R., Nat. Rev. Rheumatol. 2019, 15, 571. [DOI] [PubMed] [Google Scholar]
- 24. Fox A. J. S., Bedi A., Rodeo S. A., Sports Health 2009, 1, 461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Chen S., Fu P., Wu H., Pei M., Cell Tissue Res. 2017, 370, 53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Muir H., BioEssays 1995, 17, 1039. [DOI] [PubMed] [Google Scholar]
- 27. Goldring M. B., Ther. Adv. Musculoskelet. Dis. 2012, 4, 269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Jiang Y., Tuan R S., Nat. Rev. Rheumatol. 2015, 11, 206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. O'hara B. P., Urban J. P., Maroudas A., Ann. Rheum. Dis. 1990, 49, 536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Mauck R. L., Hung C. T., Ateshian G. A., J. Biomech. Eng. 2003, 125, 602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Maroudas A., Palla G., Gilav E., Connect. Tissue Res. 1992, 28, 161. [DOI] [PubMed] [Google Scholar]
- 32. Van Der Kraan P. M., Tissue Eng. Regener. Med. 2019, 16, 327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Gelse K., Riedel D., Pachowsky M., Hennig F. F., Trattnig S., Welsch G. H., J Orthop Res 2015, 33, 390. [DOI] [PubMed] [Google Scholar]
- 34. Williams R., Khan I. M., Richardson K., Nelson L., Mccarthy H. E., Analbelsi T., Singhrao S. K., Dowthwaite G. P., Jones R. E., Baird D. M., Lewis H., Roberts S., Shaw H. M., Dudhia J., Fairclough J., Briggs T., Archer C. W., PLoS One 2010, 5, e13246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Falah M., Nierenberg G., Soudry M., Hayden M., Volpin G., Int. Orthop. 2010, 34, 621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Lu X. L., Mow V. C., Med. Sci. Sports Exercise 2008, 40, 193. [DOI] [PubMed] [Google Scholar]
- 37. Quiroga J. M. P., Wilson W., Ito K., Van Donkelaar C. C., Biomech. Model. Mechanobiol. 2017, 16, 151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Wong M., Carter D. R, Bone 2003, 33, 1. [DOI] [PubMed] [Google Scholar]
- 39. Park S., Krishnan R., Nicoll S. B., Ateshian G. A., J. Biomech. 2003, 36, 1785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Jahn S., Klein J., Phys. Today 2018, 71, 48. [Google Scholar]
- 41. Farr J., Yao J. Q., Cartilage 2011, 2, 346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Lee J., Smeriglio P., Chu C. R., Bhutani N., Stem Cell Res. Ther. 2017, 8, 244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Nguyen D., Hgg D. A., Forsman A., Ekholm J., Nimkingratana P., Brantsing C., Kalogeropoulos T., Zaunz S., Concaro S., Brittberg M., Lindahl A., Gatenholm P., Enejder A., Simonsson S., Sci. Rep. 2017, 7, 658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Steinert A. F., Ghivizzani S. C., Rethwilm A., Tuan R. S., Evans C. H., Nöth U., Arthritis Res. Ther. 2007, 9, 213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Evenbratt H., Andreasson L., Bicknell V., Brittberg M., Mobini R., Simonsson S., Cell Regen. 2022, 11, 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Smith B., Sigal I. R., Grande D. A., Immunol. Res. 2015, 63, 181. [DOI] [PubMed] [Google Scholar]
- 47. Negoro T., Takagaki Y., Okura H., Matsuyama A., npj Regener. Med. 2018, 3, 17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Acevedo Rua L., Mumme M., Manferdini C., Darwiche S., Khalil A., Hilpert M., Buchner D. A., Lisignoli G., Occhetta P., Von Rechenberg B., Haug M., Schaefer D. J., Jakob M., Caplan A., Martin I., Barbero A., Pelttari K., Sci. Transl. Med. 2021, 13, eaaz4499. [DOI] [PubMed] [Google Scholar]
- 49. Huwe L. W., Sullan G. K., Hu J. C., Athanasiou K. A., Tissue Eng., Part A 2018, 24, 516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Brittberg M., Recker D., Ilgenfritz J., Saris D. B. F., Am. J. Sports Med. 2018, 46, 1343. [DOI] [PubMed] [Google Scholar]
- 51. Hulme C. H., Perry J., Mccarthy H. S., Wright K. T., Snow M., Mennan C., Roberts S., Emerging Top. Life Sci. 2021, 5, 575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Urlić I., Ivković A., Cells 2021, 10, 2496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Li N., Gao J., Mi L., Zhang G., Zhang L., Zhang N., Huo R., Hu J., Xu K., Stem Cell Res. Ther. 2020, 11, 381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Hu H., Liu W., Sun C., Wang Q., Yang W., Zhang Z., Xia Z., Shao Z., Wang B., Aging Dis. 2021, 12, 886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Chen F. H., Rousche K. T., Tuan R. S., Nat. Clin. Pract. Rheumatol. 2006, 2, 373. [DOI] [PubMed] [Google Scholar]
- 56. Sensebé L., Bourin P., Tarte K., Hum. Gene Ther. 2011, 22, 19. [DOI] [PubMed] [Google Scholar]
- 57. Lam A. T. L., Reuveny S., Oh S. K.‐W., Stem Cell Res. 2020, 44, 101738. [DOI] [PubMed] [Google Scholar]
- 58. Fitzgerald J. C., Duffy N., Cattaruzzi G., Vitrani F., Paulitti A., Mazzarol F., Mauro P., Sfiligoj A., Curcio F., Jones D. M., Mcinerney V., Krawczyk J., Kelly J., Finnerty A., Mcdonagh K., Mccabe U., Duggan M., Connolly L., Shaw G., Murphy M., Barry F., Front. Bioeng. Biotechnol. 2022, 10, 10.3389/fbioe.2022.834267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Viswanathan S., Shi Y., Galipeau J., Krampera M., Leblanc K., Martin I., Nolta J., Phinney D. G., Sensebe L., Cytotherapy 2019, 21, 1019. [DOI] [PubMed] [Google Scholar]
- 60. Caplan A. I., Stem Cells Transl. Med. 2017, 6, 1445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. wai Lee W. Y., Wang B., J. Orthop. Transl. 2017, 9, 76. [Google Scholar]
- 62. Goldberg A., Mitchell K., Soans J., Kim L., Zaidi R., J Orthop. Surg. Res. 2017, 12, 39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Buckley C. T., Vinardell T., Thorpe S. D., Haugh M. G., Jones E., Mcgonagle D., Kelly D. J., J. Biomech. 2010, 43, 920. [DOI] [PubMed] [Google Scholar]
- 64. Ye K., Felimban R., Traianedes K., Moulton S. E., Wallace G. G., Chung J., Quigley A., Choong P. F. M., Myers D. E., PLoS One 2014, 9, e99410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Felimban R., Ye K., Traianedes K., Di Bella C., Crook J., Wallace G. G., Quigley A., Choong P. F. M., Myers D. E., Tissue Eng., Part A 2014, 20, 2213. [DOI] [PubMed] [Google Scholar]
- 66. Francis S. L., Di Bella C., Wallace G. G., Choong P. F. M., Front. Surg. 2018, 5, 70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Sakaguchi Y., Sekiya I., Yagishita K., Muneta T., Arthritis Rheum. 2005, 52, 2521. [DOI] [PubMed] [Google Scholar]
- 68. Vinardell T., Sheehy E. J., Buckley C. T., Kelly D. J., Tissue Eng., Part A 2012, 18, 1161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Song Y., Jorgensen C., Biomedicines 2022, 10, 1278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Dennis J. E., Carbillet J.‐P., Caplan A. I., Charbord P., Cells Tissues Organs 2002, 170, 73. [DOI] [PubMed] [Google Scholar]
- 71. Pelttari K., Winter A., Steck E., Goetzke K., Hennig T., Ochs B. G., Aigner T., Richter W., Arthritis Rheum. 2006, 54, 3254. [DOI] [PubMed] [Google Scholar]
- 72. Chen S., Fu P., Cong R., Wu H., Pei M., Genes Dis. 2015, 2, 76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Weissenberger M., Weissenberger M. H., Gilbert F., Groll J., Evans C. H., Steinert A. F., BMC Musculoskeletal Disord. 2020, 21, 109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Deng Y., Lei G., Lin Z., Yang Y., Lin H., Tuan R. S., Biomaterials 2019, 192, 569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Sheng G., BMC Dev. Biol. 2015, 15, 26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Gadjanski I., Spiller K., Vunjak‐Novakovic G., Stem Cell Rev. Rep. 2012, 8, 863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Discher D. E., Mooney D. J., Zandstra P. W., Science 2009, 324, 1673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Peerani R., Zandstra P. W., J. Clin. Invest. 2010, 120, 60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Chen F. H., Tuan R. S., Arthritis Res. Ther. 2008, 10, 223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Smith G. D., Taylor J., Almqvist K. F., Erggelet C., Knutsen G., Portabella M. G., Smith T., Richardson J. B., Arthroscopy 2005, 21, 1462. [DOI] [PubMed] [Google Scholar]
- 81. Mainil‐Varlet P., Aigner T., Brittberg M., Bullough P., Hollander A., Hunziker E., Kandel R., Nehrer S., Pritzker K., Roberts S., Stauffer E., J. Bone Jt. Surg. 2003, 85, 45. [PubMed] [Google Scholar]
- 82. Fu J., He P., Wang D. A., in Tissue Eng. Artif. Organs, Wiley‐VCH Verlag GmbH & Co. KGaA, Weinheim, Germany, 2017, pp. 243–295. [Google Scholar]
- 83. Bhardwaj N., Devi D., Mandal B. B., Macromol. Biosci. 2015, 15, 153. [DOI] [PubMed] [Google Scholar]
- 84. Vega S., Kwon M., Burdick J., Eur. Cells Mater. 2017, 33, 59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Armiento A. R., Stoddart M. J., Alini M., Eglin D., Acta Biomater. 2017, 65, 1. [DOI] [PubMed] [Google Scholar]
- 86. Burnsed O. A., Schwartz Z., Marchand K. O., Hyzy S. L., Olivares‐Navarrete R., Boyan B. D., Acta Biomater. 2016, 43, 139. [DOI] [PubMed] [Google Scholar]
- 87. Bian L., Guvendiren M., Mauck R. L., Burdick J. A., Proc. Natl. Acad. Sci. U. S. A. 2013, 110, 10117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Yue K., Santiago G. T.‐d., Alvarez M. M., Tamayol A., Annabi N., Khademhosseini A., Biomaterials 2015, 73, 254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Liu S. Q., Tian Q., Hedrick J. L., Hui J. H. P., Rachel Ee P. L., Yang Y. Y., Biomaterials 2010, 31, 7298. [DOI] [PubMed] [Google Scholar]
- 90. Levato R., Webb W. R., Otto I. A., Mensinga A., Zhang Y., van Rijen M., van Weeren R., Khan I. M., Malda J., Acta Biomater. 2017, 61, 41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Lee C., O'connell C. D., Onofrillo C., Choong P. F. M., Di Bella C., Duchi S., Stem Cells Transl. Med. 2020, 9, 302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Müller M., Öztürk E., Arlov Ø., Gatenholm P., Zenobi‐Wong M., Ann. Biomed. Eng. 2017, 45, 210. [DOI] [PubMed] [Google Scholar]
- 93. Pascual‐Garrido C., Guilak F., Rai M. F., Harris M. D., Lopez M. J., Todhunter R. J., Clohisy J. C., J. Orthop. Res. 2018, 36, 1807. [DOI] [PubMed] [Google Scholar]
- 94. Hafezi M., Nouri Khorasani S., Zare M., Esmaeely Neisiany R., Davoodi P., Polymers (Basel) 2021, 13, 4199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Zhang Y., Chen S., Pei M., Eur. Cells Mater. 2016, 31, 59. [DOI] [PubMed] [Google Scholar]
- 96. Martínez‐Moreno D., Jiménez G., Gálvez‐Martín P., Rus G., Marchal J. A., Biochim. Biophys. Acta Mol. Basis Dis. 2019, 1865, 1067. [DOI] [PubMed] [Google Scholar]
- 97. Athanasiou K. A., Responte D. J., Brown W. E., Hu J. C., J. Biomech. Eng. 2015, 137, 020901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Darling E. M., Athanasiou K. A., Ann. Biomed. Eng. 2003, 31, 1114. [DOI] [PubMed] [Google Scholar]
- 99. Cipollaro L., Ciardulli M. C., Porta G. D., Peretti G. M., Maffulli N., Br. Med. Bull. 2019, 132, 53. [DOI] [PubMed] [Google Scholar]
- 100. Visser J., Melchels F. P. W., Jeon J. E., Van Bussel E. M., Kimpton L. S., Byrne H. M., Dhert W. J. A., Dalton P. D., Hutmacher D. W., Malda J., Nat. Commun. 2015, 6, 6933. [DOI] [PubMed] [Google Scholar]
- 101. Sarem M., Arya N., Heizmann M., Neffe A. T., Barbero A., Gebauer T. P., Martin I., Lendlein A., Shastri V. P., Acta Biomater. 2018, 69, 83 [DOI] [PubMed] [Google Scholar]
- 102. Onofrillo C., Duchi S., Francis S., O'connell C. D., Aguilar L. M. C., Doyle S., Yue Z., Wallace G. G., Choong P. F., Di Bella C., Biomaterials 2021, 264, 120383. [DOI] [PubMed] [Google Scholar]
- 103. Seong W., Chuan T., Kurisawa M., Spector M., Toh W. S., Lim T. C., Kurisawa M., Spector M., Biomaterials 2012, 33, 3835. [DOI] [PubMed] [Google Scholar]
- 104. Didomenico C. D., Lintz M., Bonassar L. J., Nat. Rev. Rheumatol. 2018, 14, 393. [DOI] [PubMed] [Google Scholar]
- 105. Bawolin N. K., Zhang W. J., Chen X. B., J. Biomimetics, Biomater., Tissue Eng. 2010, 6, 19. [Google Scholar]
- 106. Xu X., Jagota A., Peng S., Luo D., Wu M., Hui C.‐Y., Langmuir 2013, 29, 8665. [DOI] [PubMed] [Google Scholar]
- 107. Yu Y., Moncal K. K., Li J., Peng W., Rivero I., Martin J. A., Ozbolat I. T., Sci. Rep. 2016, 6, 28714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Liao Y., He Q., Zhou F., Zhang J., Liang R., Yao X., Bunpetch V., Li J., Zhang S., Ouyang H., Polym. Rev. 2020, 60, 203. [Google Scholar]
- 109. Nezhad‐Mokhtari P., Ghorbani M., Roshangar L., Rad J. S., Int. J. Biol. Macromol. 2019, 139, 760. [DOI] [PubMed] [Google Scholar]
- 110. Cochis A., Grad S., Stoddart M. J., Farã S., Altomare L., Azzimonti B., Alini M., Rimondini L., Sci. Rep. 2017, 7, 45018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Bidarra S. J., Barrias C. C., Granja P. L., Acta Biomater. 2014, 10, 1646. [DOI] [PubMed] [Google Scholar]
- 112. Lim K. S., Galarraga J. H., Cui X., Lindberg G. C. J., Burdick J. A., Woodfield T. B. F., Chem. Rev. 2020, 120, 10662. [DOI] [PubMed] [Google Scholar]
- 113. O'connell C. D., Zhang B., Onofrillo C., Duchi S., Blanchard R., Quigley A., Bourke J., Gambhir S., Kapsa R., Di Bella C., Choong P., Wallace G. G., Soft Matter 2018, 14, 2142. [DOI] [PubMed] [Google Scholar]
- 114. Duchi S., Onofrillo C., O’Connell C. D., Blanchard R., Augustine C., Quigley A. F., Kapsa R. M. I. I., Pivonka P., Wallace G., Di Bella C., et al., Sci. Rep. 2017, 7, 5837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Doyle S. E., Snow F., Duchi S., O'Connell C. D., Onofrillo C., Di Bella C., Pirogova E., Int. J. Mol. Sci. 2021, 22, 12420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Schuurman W., Klein T. J., Dhert W. J. A., Van Weeren P. R., Hutmacher D. W., Malda J., J. Tissue Eng. Regener. Med. 2012, 9, 669. [DOI] [PubMed] [Google Scholar]
- 117. Singh S., Choudhury D., Yu F., Mironov V., Naing M. W., Acta Biomater. 2020, 101, 14. [DOI] [PubMed] [Google Scholar]
- 118. Samandari M., Mostafavi A., Quint J., Memić A., Tamayol A., Trends Biotechnol. 2022, 40, 1229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Wu Y., Ravnic D. J., Ozbolat I. T., Trends Biotechnol. 2020, 38, 594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Albanna M., Binder K. W., Murphy S. V., Kim J., Qasem S A., Zhao W., Tan J., El‐Amin I. B., Dice D. D., Marco J., Green J., Xu T., Skardal A., Holmes J. H., Jackson J. D., Atala A., Yoo J. J., Sci. Rep. 2019, 9, 1856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Urciuolo A., Poli I., Brandolino L., Raffa P., Scattolini V., Laterza C., Giobbe G. G., Zambaiti E., Selmin G., Magnussen M., Brigo L., De Coppi P., Salmaso S., Giomo M., Elvassore N., Nat. Biomed. Eng. 2020, 4, 901. [DOI] [PubMed] [Google Scholar]
- 122. Zhu Z., Guo S.‐Z., Hirdler T., Eide C., Fan X., Tolar J., Mcalpine M. C., Adv. Mater. 2018, 1707495, 1707495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Albouy M., Desanlis A., Brosset S., Auxenfans C., Courtial E. J., Eli K., Cambron S., Palmer J., Vidal L., Thepot A., Santos M. D., Marquette C. A., Plast. Reconstr. Surg. 2022, 10, e4056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Chilelli B. J., Cole B. J., Farr J., Lattermann C., Gomoll A. H., Instr. Course Lect. 2017, 66, 507. [PubMed] [Google Scholar]
- 125. Kang H.‐W., Lee S. J., Ko I. K., Kengla C., Yoo J. J., Atala A., Nat. Biotechnol. 2016, 34, 312. [DOI] [PubMed] [Google Scholar]
- 126. Gomoll A. H., Yoshioka H., Watanabe A., Dunn J. C., Minas T., Cartilage 2011, 2, 389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Di Bella C., Fosang A., Donati D. M., Wallace G. G., Choong P. F. M., Front. Surg. 2015, 2, 39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Hunziker E. B., Quinn T. M., J. Bone Jt. Surg. 2003, 85, 85. [Google Scholar]
- 129. Tew S. R., Kwan A. P. L., Hann A., Thomson B. M., Archer C. W., Arthritis Rheum. 2000, 43, 215. [DOI] [PubMed] [Google Scholar]
- 130. Gomoll A. H., Farr J., Gillogly S. D., Kercher J. S., Minas T., Instr. Course Lect. 2011, 60, 461. [PubMed] [Google Scholar]
- 131. Filardo G., Drobnic M., Perdisa F., Kon E., Hribernik M., Marcacci M., Osteoarthritis Cartilage 2014, 22, 557. [DOI] [PubMed] [Google Scholar]
- 132. Drobnic M., Perdisa F., Kon E., Cefalã F., Marcacci M., Filardo G., Knee Surg., Sports Traumatol., Arthroscopy 2018, 26, 2774. [DOI] [PubMed] [Google Scholar]
- 133. Lotz B., Bothe F., Deubel A. K., Hesse E., Renz Y., Werner C., Schäfer S., Böck T., Groll J., Von Rechenberg B., Richter W., Hagmann S., Int. J. Biomater. 2021, 2021, 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. Brittberg M., Biomaterials 1997, 18, 235. [DOI] [PubMed] [Google Scholar]
- 135. Tarafder S., Park G. Y., Felix J., Lee C. H., Acta Biomater. 2020, 117, 77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Broguiere N., Cavalli E., Salzmann G. M., Applegate L. A., Zenobi‐Wong M., ACS Biomater. Sci. Eng. 2016, 2, 2176. [DOI] [PubMed] [Google Scholar]
- 137. Lim K. S., Abinzano F., Bernal P. N., Albillos Sanchez A., Atienza‐Roca P., Otto I. A., Peiffer Q. C., Matsusaki M., Woodfield T. B. F., Malda J., Levato R., Adv. Healthcare Mater. 2020, 9, 1901792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. ztürk E., Stauber T., Levinson C., Cavalli E., Arlov O., Zenobi‐Wong M., Biomed. Mater. 2020, 15, 045019. [DOI] [PubMed] [Google Scholar]
- 139. Trengove A., Duchi S., Onofrillo C., O'connell C. D., Di Bella C., O'connor A. J., Front. Med. Technol. 2021, 3, 773673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Trengove A., Di Bella C., O'connor A. J., Tissue Eng., Part B 2022, 28, 114. [DOI] [PubMed] [Google Scholar]
- 141. Englert C., Mcgowan K. B., Klein T. J., Giurea A., Schumacher B. L., Sah R. L., Arthritis Rheum. 2005, 52, 1091. [DOI] [PubMed] [Google Scholar]
- 142. Ding S.‐L., Liu X., Zhao X.‐Y., Wang K.‐T., Xiong W., Gao Z.‐L., Sun C.‐Y., Jia M.‐X., Li C., Gu Q., Zhang M.‐Z., Bioact. Mater. 2022, 17, 81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Yuk H., Varela C. E., Nabzdyk C. S., Mao X., Padera R. F., Roche E. T., Zhao X., Nature 2019, 575, 169. [DOI] [PubMed] [Google Scholar]
- 144. Hua Y., Xia H., Jia L., Zhao J., Zhao D., Yan X., Zhang Y., Tang S., Zhou G., Zhu L., Lin Q., Sci. Adv. 2021, 7, eabg0628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Penton J. L., Flick T. R., Savoie F. H., Heard W. M., Sherman W. F., Orthop. J. Sports Med. 2021, 9, 232596712110354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146. Heng C. H. Y., Snow M., Dave L. Y. H., Arthrosc. Tech. 2021, 10, e751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147. Safran M. R., Kim H., Zaffagnini S., J. Am. Acad. Orthop. Surg. 2008, 16, 306. [DOI] [PubMed] [Google Scholar]
- 148. Dijkstra K., Huitsing R. P. J., Custers R. J. H., Kouwenhoven J. W. M., Bleys R. L. A. W., Vonk L. A., Saris D. B. F., Tissue Eng., Part C 2019, 25, 379. [DOI] [PubMed] [Google Scholar]
- 149. Whyte G. P., Gobbi A., Sadlik B., Arthrosc. Tech. 2016, 5, e913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Siemieniuk R. A. C., Harris I. A., Agoritsas T., Poolman R. W., Brignardello‐Petersen R., Van De Velde S., Buchbinder R., Englund M., Lytvyn L., Quinlan C., Helsingen L., Knutsen G., Olsen N. R., Macdonald H., Hailey L., Wilson H. M., Lydiatt A., Kristiansen A., Br. J. Sports Med. 2018, 52, 313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. Millar N. L., Wu X., Tantau R., Silverstone E., Murrell G. A. C., Clin. Orthop. Relat. Res. 2009, 467, 966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152. Goyal D., J. Arthrosc. Surg. Sports Med. 2020, 1, 159. [Google Scholar]
- 153. Francis S. L., Duchi S., Onofrillo C., Di Bella C., Choong P. F. M., Stem Cells Int. 2018, 2018, 10.1155/2018/8947548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154. Ynarski K., De Jong W. C., Snow M., Hendriks J. A. A., Wilson C. E., Verdonk P., Am. J. Sports Med. 2020, 48, 1327. [DOI] [PubMed] [Google Scholar]
- 155. Dragoo J. L., Chang W., Am. J. Sports Med. 2017, 45, 3119. [DOI] [PubMed] [Google Scholar]
- 156. Tremolada C., Colombo V., Ventura C., Curr. Stem Cell Rep. 2016, 2, 304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157. Reinhardt M., Bader A., Giri S., Expert Rev. Med. Devices 2015, 12, 353. [DOI] [PubMed] [Google Scholar]
- 158. Beigi M.‐H., Atefi A., Ghanaei H.‐R., Labbaf S., Ejeian F., Nasr‐Esfahani M.‐H., J. Tissue Eng. Regener. Med. 2018, 12, 1327. [DOI] [PubMed] [Google Scholar]
- 159. Bellisari F. C., De Marino L., Arrigoni F., Mariani S., Bruno F., Palumbo P., De Cataldo C., Sgalambro F., Catallo N., Zugaro L., Di Cesare E., Splendiani A., Masciocchi C., Giovagnoni A., Barile A., Radiol. Med. 2021, 126, 1085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160. Snow M., Williams R., Pagkalos J., Grover L., Cartilage 2021, 12, 226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161. Yu N.‐H., Chun S. Y., Ha Y.‐S., Kim H. T., Kim D. H., Kim J., Chung J.‐W., Lee J. N., Song P. H., Yoo E. S., Kim B. S., Kwon T. G., Tissue Eng. Regener. Med. 2018, 15, 639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162. Aoyama T., in Mesenchymal Stem Cells, (Ed: Pham P. V.), IntechOpen, London: 2017, Ch. 13. [Google Scholar]
- 163. Hinterwimmer S., Krammer M., Krötz M., Glaser C., Baumgart R., Reiser M., Eckstein F., Arthritis Rheum. 2004, 50, 2516. [DOI] [PubMed] [Google Scholar]
- 164. Memmel C., Krutsch W., Koch M., Riedl M., Henssler L., Zeman F., Knorr C., Alt V., Pfeifer C., Appl. Sci. 2021, 11, 8873. [Google Scholar]
- 165. Hurley E. T., Davey M. S., Jamal M. S., Manjunath A. K., Alaia M. J., Strauss E. J., Cartilage 2021, 13, 907S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166. Musumeci G., World J. Orthop. 2014, 5, 80.24829869 [Google Scholar]
- 167. Wolf M. T., Zhang H., Sharma B., Marcus N. A., Pietzner U., Fickert S., Lueth A., Albers G. H. R., Elisseeff J. H., Cartilage 2020, 11, 447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168. Hambly K., Silvers H. J., Steinwachs M., Cartilage 2012, 3, 50S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169. Wagner K. R., Kaiser J. T., DeFroda S. F., Meeker Z. D., Cole B. J., Arthrosc. Sports Med. Rehabil. 2022, 4, e115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170. Chen T., McCarthy M. M., Guo H., Warren R., Maher S. A., J. Biomech. Eng. 2018, 140, 10.1115/1.4040121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171. Kajabi A. W., Casula V., Sarin J K., Ketola J. H., Nykã¤Nen O., Te Moller N. C. R., Mancini I. A. D., Visser J., Brommer H., Van Weeren P. R., Malda J., TöYrã¤S J., Nieminen M. T., Nissi M. J., J. Orthop. Res. 2021, 39, 63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172. DuRaine G. D., Darling E. M., Reddi A. H., Hu J. C., Athanasiou K. A., Articular Cartilage, 2nd ed., CRC Press, Boca Raton, FL: 2017. [Google Scholar]
- 173. Han L., Wang M., Li P., Gan D., Yan L., Xu J., Wang K., Fang L., Chan C. W., Zhang H., Yuan H., Lu X., ACS Appl. Mater. Interfaces 2018, 10, 28015. [DOI] [PubMed] [Google Scholar]
- 174. Shen H., Lin H., Sun A. X., Song S., Zhang Z., Dai J., Tuan R. S., J. Mater. Chem. B 2018, 6, 908. [DOI] [PubMed] [Google Scholar]
- 175. Leijten J., Teixeira L. S. M., Bolander J., Ji W., Vanspauwen B., Lammertyn J., Schrooten J., Luyten F. P., Sci. Rep. 2016, 6, 36011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176. Im G.‐I., Jung N.‐H., Tae S.‐K., Tissue Eng. 2006, 12, 527. [DOI] [PubMed] [Google Scholar]
- 177. Kim H.‐J., Im G.‐I., J. Orthop. Res. 2009, 27, 612. [DOI] [PubMed] [Google Scholar]
- 178. Yu D.‐A., Han J., Kim B.‐S., Int. J. Stem Cells 2012, 5, 16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179. Fortier L. A., Barker J. U., Strauss E. J., Mccarrel T. M., Cole B. J., Clin. Orthop. Relat. Res. 2011, 469, 2706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180. Ruiz M., Maumus M., Fonteneau G., Pers Y.‐M., Ferreira R., Dagneaux L., Delfour C., Houard X., Berenbaum F., Rannou F., Jorgensen C., Osteoarthritis Cartilage 2019, 27, 493. [DOI] [PubMed] [Google Scholar]
- 181. Pfeifer C. G., Karl A., Kerschbaum M., Berner A., Lang S., Schupfner R., Koch M., Angele P., Nerlich M., Mueller M. B., Int. J. Stem Cells 2019, 12, 139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182. Van Der Kraan P. M., J. Bone Metab. 2018, 25, 65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183. Kwon H., Paschos N. K., Hu J. C., Athanasiou K., Cell. Mol. Life Sci. 2016, 73, 1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184. Lam J., Lu S., Kasper F. K, Mikos A. G., Adv. Drug Delivery Rev. 2015, 84, 123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185. Yang Z., Zhao T., Gao C., Cao F., Li H., Liao Z., Fu L., Li P., Chen W., Sun Z., Jiang S., Tian Z., Tian G., Zha K., Pan T., Li X., Sui X., Yuan Z., Liu S., Guo Q., ACS Appl. Mater. Interfaces 2021, 13, 23369. [DOI] [PubMed] [Google Scholar]
- 186. Trippel S., Ghivizzani S., Nixon A., Gene Ther. 2004, 11, 351. [DOI] [PubMed] [Google Scholar]
- 187. Lee J. M., Ryu J. H., Kim E. A., Jo S., Kim B.‐S., Lee H., Im G.‐I., Biomaterials 2015, 39, 173. [DOI] [PubMed] [Google Scholar]
- 188. Vanden Berg‐Foels W. S., Tissue Eng. Part B Rev. 2014, 20, 28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189. Rouwkema J., Koopman B. F. J. M., Blitterswijk C. A. V., Dhert W. J. A., Malda J., Biotechnol. Genet. Eng. Rev. 2009, 26, 163. [DOI] [PubMed] [Google Scholar]
- 190. Fisher M. B., Belkin N. S., Milby A. H., Henning E. A., Söegaard N., Kim M., Pfeifer C., Saxena V., Dodge G. R., Burdick J. A., Schaer T. P., Steinberg D. R., Mauck R. L., Cartilage 2016, 7, 174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191. Choi B., Kim S., Fan J., Kowalski T., Petrigliano F., Evseenko D., Lee M., Biomater. Sci. 2015, 3, 742. [DOI] [PubMed] [Google Scholar]
- 192. Fan W., Yuan L., Li J., Wang Z., Chen J., Guo C., Mo X., Yan Z., Mater. Sci. Eng. C 2020, 110, 110705. [DOI] [PubMed] [Google Scholar]
- 193. Shi S., Chan A G., Mercer S., Eckert G. J., Trippel S. B., J. Orthop. Res. 2014, 32, 54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194. Aguilar L. M. C., Silva S. M., Moulton S. E., J. Controlled Release 2019, 306, 40. [DOI] [PubMed] [Google Scholar]
- 195. Li J., Dong S., Stem Cells Int. 2016, 2016, 2470351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196. Augustyniak E., Trzeciak T., Richter M., Kaczmarczyk J., Suchorska W., Int. Orthop. 2015, 39, 995. [DOI] [PubMed] [Google Scholar]
- 197. Runser S., Cerletti N., Biotechnol. Appl. Biochem. 1995, 22, 39. [PubMed] [Google Scholar]
- 198. Kim N. J., Yoo J. J., Atala A., Lee S. J., Small RNA Delivery for In Situ Tissue Regeneration, Elsevier Inc., Amsterdam: 2016. [Google Scholar]
- 199. Wadhwa A., Aljabbari A., Lokras A., Foged C., Thakur A., Pharmaceutics 2020, 12, 102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200. Mobasheri A., Choi H., Martín‐vasallo P., Biology 2020, 9, 330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201. Aguilar L. M. C., Duchi S., Onofrillo C., O'Connell C. D., Di Bella C., Moulton S. E., J. Colloid Interface Sci. 2021, 587, 240. [DOI] [PubMed] [Google Scholar]
- 202. Caballero‐Aguilar L. M., Duchi S., Quigley A., Onofrillo C., Di Bella C., Moulton S. E., MethodsX 2021, 8, 101324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203. Shahin K., Doran P. M., in Cartil. Tissue Eng., 2015, pp. 221–233. [Google Scholar]
- 204. Behrendt P., Ladner Y., Stoddart M. J., Lippross S., Alini M., Eglin D., Armiento A. R., Am. J. Sports Med. 2020, 48, 210. [DOI] [PubMed] [Google Scholar]
- 205. Vainieri M. L., Wahl D., Alini M., Van Osch G. J. V. M., Grad S., Acta Biomater. 2018, 81, 256. [DOI] [PubMed] [Google Scholar]
- 206. Murdock M. H., Badylak S. F., Curr. Opin. Biomed. Eng. 2017, 1, 4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207. Albro M. B., Cigan A. D., Nims R. J., Yeroushalmi K. J., Oungoulian S. R., Hung C. T., Ateshian G. A., Osteoarthritis Cartilage 2012, 20, 1374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208. Thielen N., Van Der Kraan P., Van Caam A., Cells 2019, 8, 969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209. Keller B., Yang T., Chen Y., Munivez E., Bertin T., Zabel B., Lee B., PLoS One 2011, 6, e16421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210. Shintani N., Siebenrock K. A., Hunziker E. B., PLoS One 2013, 8, e53086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211. Bosnakovski D., Mizuno M., Kim G., Takagi S., Okumura M., Fujinaga T., Biotechnol. Bioeng. 2006, 93, 1152. [DOI] [PubMed] [Google Scholar]
- 212. Loebel C., Saleh A. M., Jacobson K. R., Daniels R., Mauck R L., Calve S., Burdick J. A., Nat. Protoc. 2022, 17, 618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213. Khan I., Gilbert S., Singhrao S., Duance V., Archer C., Eur. Cells Mater. 2008, 16, 26. [DOI] [PubMed] [Google Scholar]
- 214. Jelodari S., Sadrabadi A. E., Zarei F., Jahangir S., Azami M., Sheykhhasan M., Hosseini S., Biomed Res. Int. 2022, 2022, 7638245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215. Diloksumpan P., Abinzano F., de Ruijter M., Mensinga A., Plomp S., Khan I., Brommer H., Smit I., Castilho M. D., van Weeren P. R., Malda J., Levato R., J. Trial Error 2021, 2, 7. [Google Scholar]
- 216. Bothe F., Deubel A. K., Hesse E., Lotz B., Groll J., Werner C., Richter W., Hagmann S., Int. J. Mol. Sci. 2019, 20, 653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217. Vansusante J., Buma P., Homminga G., Vandenberg W., Veth R., Biomaterials 1998, 19, 2367. [DOI] [PubMed] [Google Scholar]
- 218. Li F., Levinson C., Truong V. X., Laurent‐Applegate L. A., Maniura‐Weber K., Thissen H., Forsythe J. S., Zenobi‐Wong M., Frith J. E., Biomater. Sci. 2020, 8, 1711. [DOI] [PubMed] [Google Scholar]
- 219. Kim S. A., Sur Y. J., Cho M. L., Go E. J., Kim Y. H., Shetty A. A., Kim S. J., Sci. Rep. 2020, 10, 10678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220. Radhakrishnan J., Manigandan A., Chinnaswamy P., Subramanian A., Sethuraman S., Biomaterials 2018, 162, 82. [DOI] [PubMed] [Google Scholar]
- 221. Sahu N., Miller A., Viljoen H. J., Subramanian A., Tissue Eng., Part A 2019, 25, 1538. [DOI] [PubMed] [Google Scholar]
- 222. Dimicco M. A., Sah R. L., J. Orthop. Res. 2001, 19, 1105. [DOI] [PubMed] [Google Scholar]
- 223. Obradovic B., Martin I., Padera R. F., Treppo S., Freed L. E., Vunjak‐Navakovic G., J. Orthop. Res. 2001, 19, 1089. [DOI] [PubMed] [Google Scholar]
- 224. Bos P. K., Degroot J., Budde M., Verhaar J. A. N., Van Osch G. J. V. M., Arthritis Rheum. 2002, 46, 976. [DOI] [PubMed] [Google Scholar]
- 225. Lee M. C., Sung K. L. P., Kurtis M. S., Akeson W. H., Sah R. L., Clin. Orthop. Relat. Res. 2000, 370, 286. [DOI] [PubMed] [Google Scholar]
- 226. Kondo S., Shukunami C., Morioka Y., Matsumoto N., Takahashi R., Oh J., Atsumi T., Umezawa A., Kudo A., Kitayama H., Hiraki Y., Noda M., J. Cell Sci. 2007, 120, 849. [DOI] [PubMed] [Google Scholar]
- 227. Yang Y.‐H. K., Ogando C. R., Barabino G. A., J. Funct. Biomater. 2020, 11, 5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228. Hou M., Tian B., Bai B., Ci Z., Liu Y., Zhang Y., Zhou G., Cao Y., Bioact. Mater. 2022, 13, 149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229. Caplan A., J. Pathol. 2009, 217, 318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230. Le H., Xu W., Zhuang X., Chang F., Wang Y., Ding J., J. Tissue Eng. 2020, 11, 204173142094383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231. Planat‐Benard V., Varin A., Casteilla L., Front. Immunol. 2021, 12, 626755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232. Rikkers M., Dijkstra K., Terhaard B. F., Admiraal J., Levato R., Malda J., Vonk L. A., Cartilage 2021, 13, 991S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233. Caplan A. I., Cell Stem Cell 2008, 3, 229. [DOI] [PubMed] [Google Scholar]
- 234. Caplan A. I., Tissue Eng., Part B 2009, 15, 195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235. Rehman J., Traktuev D., Li J., Merfeld‐Clauss S., Temm‐Grove C. J., Bovenkerk J. E., Pell C. L., Johnstone B. H., Considine R. V., March K. L., Circulation 2004, 109, 1292. [DOI] [PubMed] [Google Scholar]
- 236. Kinnaird T., Stabile E., Burnett M. S., Lee C. W., Barr S., Fuchs S., Epstein S. E., Circ. Res. 2004, 94, 678. [DOI] [PubMed] [Google Scholar]
- 237. Gnecchi M., Zhang Z., Ni A., Dzau V. J., Circ. Res. 2008, 103, 1204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238. Wu L., Leijten J. C. H., Georgi N., Post J. N., Van Blitterswijk C. A., Karperien M., Tissue Eng., Part A 2011, 17, 1425. [DOI] [PubMed] [Google Scholar]
- 239. Acharya C., Adesida A., Zajac P., Mumme M., Riesle J., Martin I., Barbero A., J. Cell. Physiol. 2012, 227, 88. [DOI] [PubMed] [Google Scholar]
- 240. Meretoja V. V., Dahlin R. L., Kasper F. K., Mikos A. G., Biomaterials 2012, 33, 6362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241. Wang M., Rahnama R., Cheng T., Grotkopp E., Jacobs L., Limburg S., Kim H. T., Kuo A. C., J. Orthop. Res. 2013, 31, 1936. [DOI] [PubMed] [Google Scholar]
- 242. Nakamura T., Sekiya I., Muneta T., Hatsushika D., Horie M., Tsuji K., Kawarasaki T., Watanabe A., Hishikawa S., Fujimoto Y., Tanaka H., Kobayashi E., Cytotherapy 2012, 14, 327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243. Dahlin R. L., Kinard L. A., Lam J., Needham C. J., Lu S., Kasper F. K., Mikos A. G., Biomaterials 2015, 35, 7460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244. De Windt T. S., Vonk L. A., Slaper‐Cortenbach I. C. M., Van Den Broek M. P. H., Nizak R., Van Rijen M. H. P., De Weger R. A., Dhert W. J. A., Saris D. B. F., Stem Cells 2017, 35, 256. [DOI] [PubMed] [Google Scholar]
- 245. Croitoru‐Lamoury J., Lamoury F. M. J., Zaunders J. J., Veas L. A., Brew B. J., J. Interferon Cytokine Res. 2007, 27, 53. [DOI] [PubMed] [Google Scholar]
- 246. Zhang S., Hu B., Liu W., Wang P., Lv X., Chen S., Liu H., Shao Z., Semin. Arthritis Rheum. 2020, 50, 198. [DOI] [PubMed] [Google Scholar]
- 247. Jiang S., Tian G., Li X., Yang Z., Wang F., Tian Z., Huang B., Wei F., Zha K., Sun Z., Sui X., Liu S., Guo W., Guo Q., Stem Cells Int. 2021, 2021, 8882505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248. Cook J. L., Hung C. T., Kuroki K., Stoker A. M., Cook C. R., Pfeiffer F. M., Sherman S. L., Stannard J. P., Bone Jt. Res. 2014, 3, 89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249. Chu C. R., Szczodry M., Bruno S., Tissue Eng., Part B 2010, 16, 105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250. Takroni T., Laouar L., Adesida A., Elliott J. A. W., Jomha N. M., J. Exp. Orthop. 2016, 3, 35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251. Ribitsch I., Baptista P. M., Lange‐Consiglio A., Melotti L., Patruno M., Jenner F., Schnabl‐Feichter E., Dutton L. C., Connolly D. J., Van Steenbeek F. G., Dudhia J., Penning L. C., Front. Bioeng. Biotechnol. 2020, 8, 972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252. Filardo G., Madry H., Jelic M., Roffi A., Cucchiarini M., Kon E., Knee Surg., Sports Traumatol., Arthroscopy 2013, 21, 1717. [DOI] [PubMed] [Google Scholar]
- 253. Filardo G., Perdisa F., Roffi A., Marcacci M., Kon E., J. Orthop. Surg. Res. 2016, 11, 42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254. Mastbergen S. C., Saris D. L. B. F., Lafeber F. P. J. G., Nat. Rev. Rheumatol. 2013, 9, 277. [DOI] [PubMed] [Google Scholar]
- 255. Pastides P., Chimutengwende‐Gordon M., Maffulli N., Khan W., Osteoarthritis Cartilage 2013, 21, 646. [DOI] [PubMed] [Google Scholar]
- 256. Hourd P., Medcalf N., Segal J., Williams D. J., Regener. Med. 2015, 10, 863. [DOI] [PubMed] [Google Scholar]
- 257. Mansour J. M., Lee Z., Welter J. F., Ann. Biomed. Eng. 2016, 44, 733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258. Kotecha M., Klatt D., Magin R. L., Tissue Eng., Part B 2013, 19, 470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259. Sharma B., Fermanian S., Gibson M., Unterman S., Herzka D. A., Cascio B., Coburn J., Hui A. Y., Marcus N., Gold G. E., Elisseeff J. H., Sci. Transl. Med. 2013, 5, 167ra6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260. Mercado K. P., Helguera M.A., Hocking D. C., Dalecki D., Tissue Eng., Part C 2015, 21, 671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261. Baksh S. C., Fuchs E., Cell 2020, 182, 1377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262. Gilbert F., O'Connell C. D., Mladenovska T., Dodds S., Sci. Eng. Ethics 2017, 24, 73. [DOI] [PubMed] [Google Scholar]
- 263. Morgan T. H., Regeneration, MacMillan, London: 1901. [Google Scholar]
- 264. Lambrou G. I., Remboutsika E., Front. Physiol. 2014, 5, 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265. Bernhard J. C., Vunjak‐Novakovic G., Stem Cell Res. Ther. 2016, 7, 56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266. O'shea D. G., Curtin C. M., O'brien F. J., Biomater. Sci. 2022, 2462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267. Mainil‐Varlet P., Rieser F., Grogan S., Mueller W., Saager C., Jakob R. P., Osteoarthritis Cartilage 2001, 9, S6. [DOI] [PubMed] [Google Scholar]
- 268. Hosseini S. A., Mohammadi R., Noruzi S., Ganji R., Oroojalian F., Sahebkar A., Jt., Bone, Spine 2021, 88, 105096. [DOI] [PubMed] [Google Scholar]
- 269. He A., Liu L., Luo X., Liu Y., Liu Y., Liu F., Wang X., Zhang Z., Zhang W., Liu W., et al, Sci. Rep. 2017, 7, 40489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270. Nehrer S., Dorotka R., Domayer S., Stelzeneder D., Kotz R., Am. J. Sports Med. 2009, 37, 81. [DOI] [PubMed] [Google Scholar]
- 271. Crawford D. C., Deberardino T. M., Williams R. J., J. Bone Jt. Surg. 2012, 94, 979. [DOI] [PubMed] [Google Scholar]
- 272. Wong K. L., Lee K. B. L., Tai B. C., Law P., Lee E. H., Hui J. H. P., Arthroscopy 2013, 29, 2020. [DOI] [PubMed] [Google Scholar]
- 273. Jo C. H., Lee Y. G., Shin W. H., Kim H., Chai J. W., Jeong E. C., Kim J. E., Shim H., Shin J. S., Shin I. S., Ra J. C., Oh S., Yoon K. S., Stem Cells 2014, 32, 1254. [DOI] [PubMed] [Google Scholar]
- 274. Mumme M., Barbero A., Miot S., Wixmerten A., Feliciano S., Wolf F., Asnaghi A. M., Baumhoer D., Bieri O., Kretzschmar M., Pagenstert G., Haug M., Schaefer D. J., Martin I., Jakob M., Lancet 2016, 388, 1985. [DOI] [PubMed] [Google Scholar]
- 275. D'Ambrosi R., Giacco F., Ragone V., Ursino N., Int. Orthop. 2019, 43, 2183. [DOI] [PubMed] [Google Scholar]
- 276. Sennett M., Friedman J., Ashley B., Stoeckl B., Patel J., Alini M., Cucchiarini M., Eglin D., Madry H., Mata A., Semino C., Stoddart M., Johnstone B., Moutos F., Estes B., Guilak F., Mauck R., Dodge G., Eur. Cells Mater. 2021, 41, 40. [DOI] [PMC free article] [PubMed] [Google Scholar]


