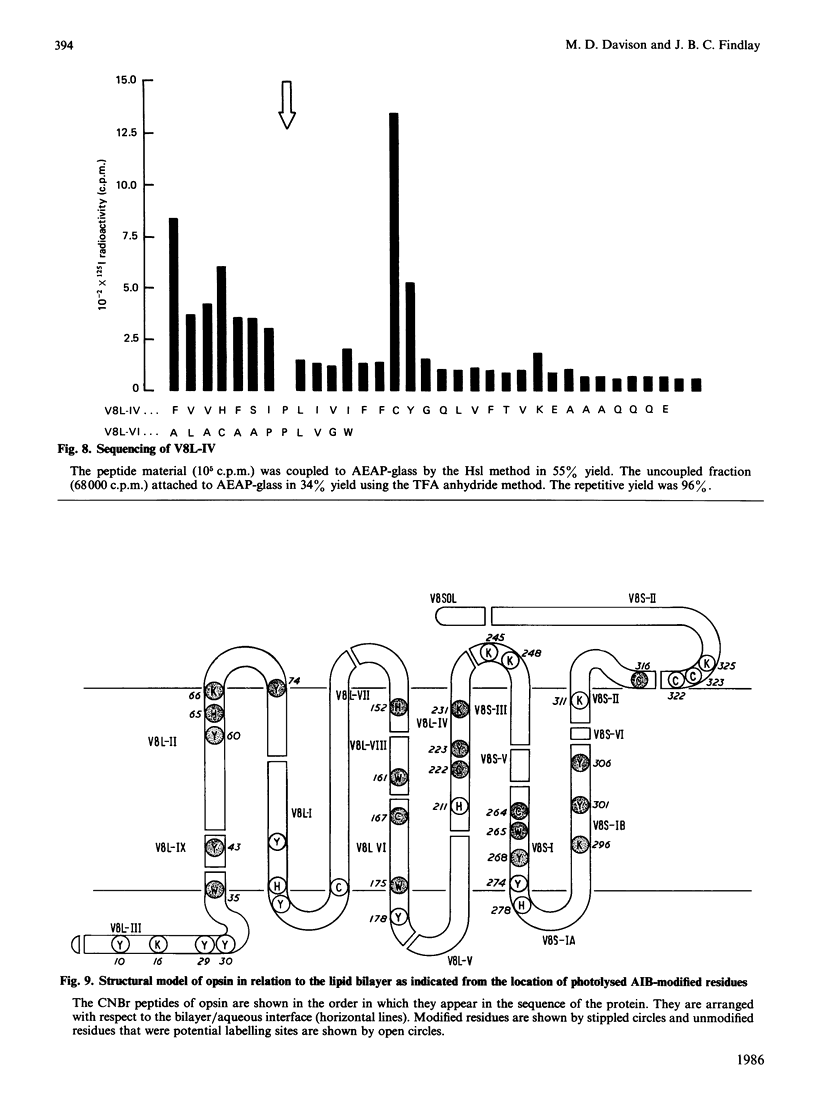
Abstract
Opsin labelled with photoactivated 1-azido-4-[125I]iodobenzene was proteolysed in situ with Staphylococcus aureus V8 proteinase to yield two radioactive membrane-bound fragments. These were separated, cleaved with CNBr and the resultant peptides sequenced in order to locate the radiolabelled residues. In the whole molecule, there was clear evidence for modification of at least 20 sites, identified as derivatives of cysteine, tryptophan, tyrosine, histidine and lysine residues. The probe primary reacted, therefore, with nucleophilic substituents. The positions of the modified sites relative to the confines of the phospholipid bilayer were consistent with all other studies on the disposition of the polypeptide chain. The location of these sites substantiated an earlier suggestion that not all the transmembrane segments should be regarded as continuous regular alpha-helices.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barclay P. L., Findlay J. B. Labelling of the cytoplasmic domains of ovine rhodopsin with hydrophilic chemical probes. Biochem J. 1984 May 15;220(1):75–84. doi: 10.1042/bj2200075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bayley H., Knowles J. R. Photogenerated reagents for membrane labeling. 2. Phenylcarbene and adamantylidene formed within the lipid bilayer. Biochemistry. 1978 Jun 13;17(12):2420–2423. doi: 10.1021/bi00605a026. [DOI] [PubMed] [Google Scholar]
- Brauer A. W., Oman C. L., Margolies M. N. Use of o-phthalaldehyde to reduce background during automated Edman degradation. Anal Biochem. 1984 Feb;137(1):134–142. doi: 10.1016/0003-2697(84)90359-2. [DOI] [PubMed] [Google Scholar]
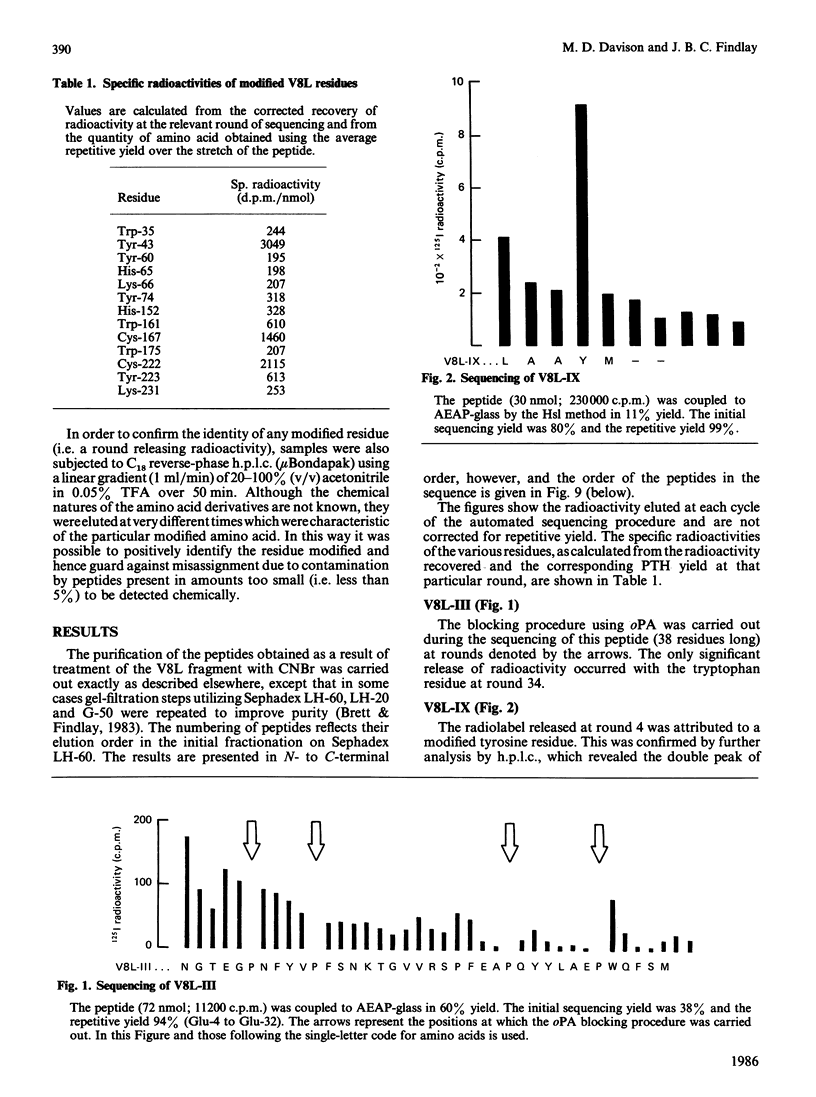
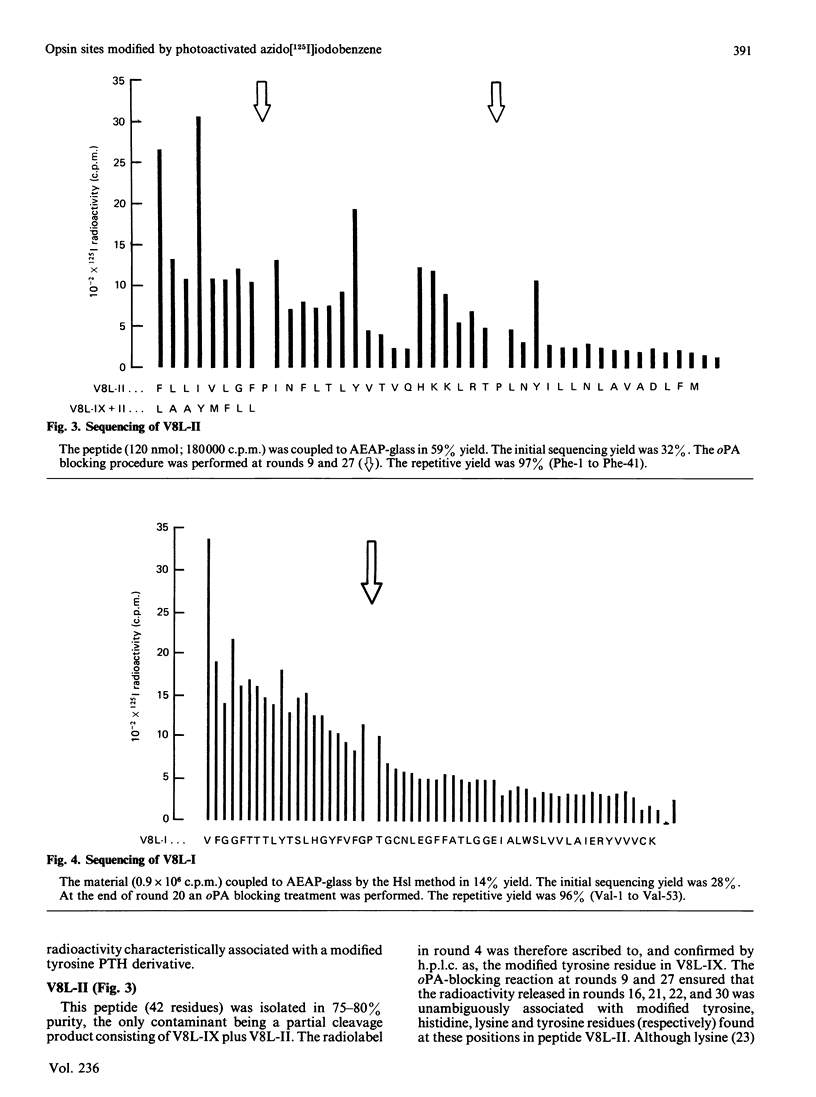
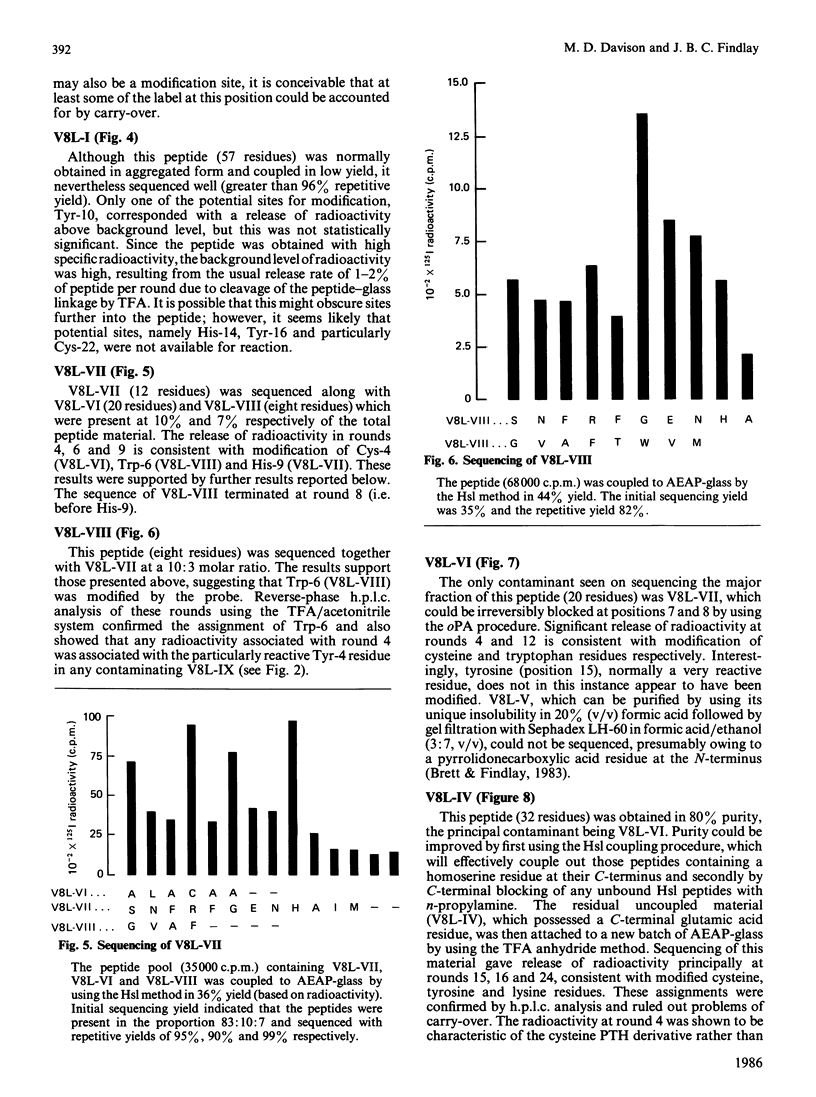
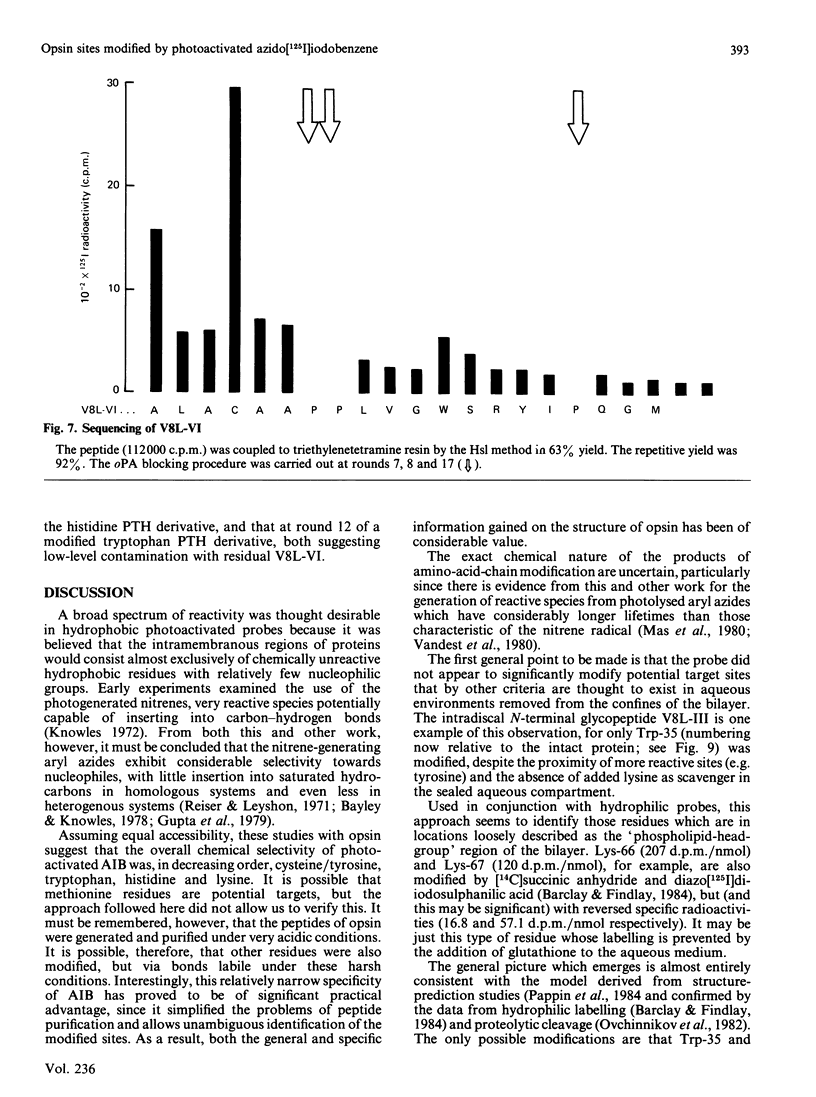
- Brett M., Findlay J. B. Isolation and characterization of the CNBr peptides from the proteolytically derived N-terminal fragment of ovine opsin. Biochem J. 1983 Jun 1;211(3):661–670. doi: 10.1042/bj2110661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunner J., Richards F. M. Analysis of membranes photolabeled with lipid analogues. Reaction of phospholipids containing a disulfide group and a nitrene or carbene precursor with lipids and with gramicidin A. J Biol Chem. 1980 Apr 25;255(8):3319–3329. [PubMed] [Google Scholar]
- Davison M. D., Findlay J. B. Modification of ovine opsin with the photosensitive hydrophobic probe 1-azido-4-[125I]iodobenzene. Labelling of the chromophore-attachment domain. Biochem J. 1986 Mar 1;234(2):413–420. doi: 10.1042/bj2340413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta C. M., Radhakrishnan R., Gerber G. E., Olsen W. L., Quay S. C., Khorana H. G. Intermolecular crosslinking of fatty acyl chains in phospholipids: use of photoactivable carbene precursors. Proc Natl Acad Sci U S A. 1979 Jun;76(6):2595–2599. doi: 10.1073/pnas.76.6.2595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoppe J., Brunner J., Jørgensen B. B. Structure of the membrane-embedded F0 part of F1F0 ATP synthase from Escherichia coli as inferred from labeling with 3-(Trifluoromethyl)-3-(m-[125I]iodophenyl)diazirine. Biochemistry. 1984 Nov 6;23(23):5610–5616. doi: 10.1021/bi00318a035. [DOI] [PubMed] [Google Scholar]
- Hoppe J., Friedl P., Jørgensen B. B. [125I]Iodonaphtylazide labeling selectively a cysteine residue in the F0 of the ATP-synthase from E. coli is unsuitable for topographic studies of membrane proteins. FEBS Lett. 1983 Aug 22;160(1-2):239–242. doi: 10.1016/0014-5793(83)80974-0. [DOI] [PubMed] [Google Scholar]
- Hoppe J., Montecucco C., Friedl P. Labeling of subunit b of the ATP synthase from Escherichia coli with a photoreactive phospholipid analogue. J Biol Chem. 1983 Mar 10;258(5):2882–2885. [PubMed] [Google Scholar]
- Horn M. J., Laursen R. A. Solid-phase edman degradation: attachment of carboxyl-terminal homoserine peptides to an insoluble resin. FEBS Lett. 1973 Nov 1;36(3):285–288. doi: 10.1016/0014-5793(73)80392-8. [DOI] [PubMed] [Google Scholar]
- Lin L. F., Lees M. B. Interactions of dicyclohexylcarbodiimide with myelin proteolipid. Proc Natl Acad Sci U S A. 1982 Feb;79(3):941–945. doi: 10.1073/pnas.79.3.941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mas M. T., Wang J. K., Hargrave P. A. Topography of rhodopsin in rod outer segment disk membranes. Photochemical labeling with N-(4-azido-2-nitrophenyl)-2-aminoethanesulfonate. Biochemistry. 1980 Feb 19;19(4):684–691. doi: 10.1021/bi00545a012. [DOI] [PubMed] [Google Scholar]
- Ross A. H., Radhakrishnan R., Robson R. J., Khorana H. G. The transmembrane domain of glycophorin A as studied by cross-linking using photoactivatable phospholipids. J Biol Chem. 1982 Apr 25;257(8):4152–4161. [PubMed] [Google Scholar]
- Vandest P., Labbe J. P., Kassab R. Photoaffinity labelling of arginine kinase and creatine kinase with a gamma-P-substituted arylazido analogue of ATP. Eur J Biochem. 1980 Mar;104(2):433–442. doi: 10.1111/j.1432-1033.1980.tb04445.x. [DOI] [PubMed] [Google Scholar]
- Zimmerman C. L., Appella E., Pisano J. J. Rapid analysis of amino acid phenylthiohydantoins by high-performance liquid chromatography. Anal Biochem. 1977 Feb;77(2):569–573. doi: 10.1016/0003-2697(77)90276-7. [DOI] [PubMed] [Google Scholar]


