Abstract
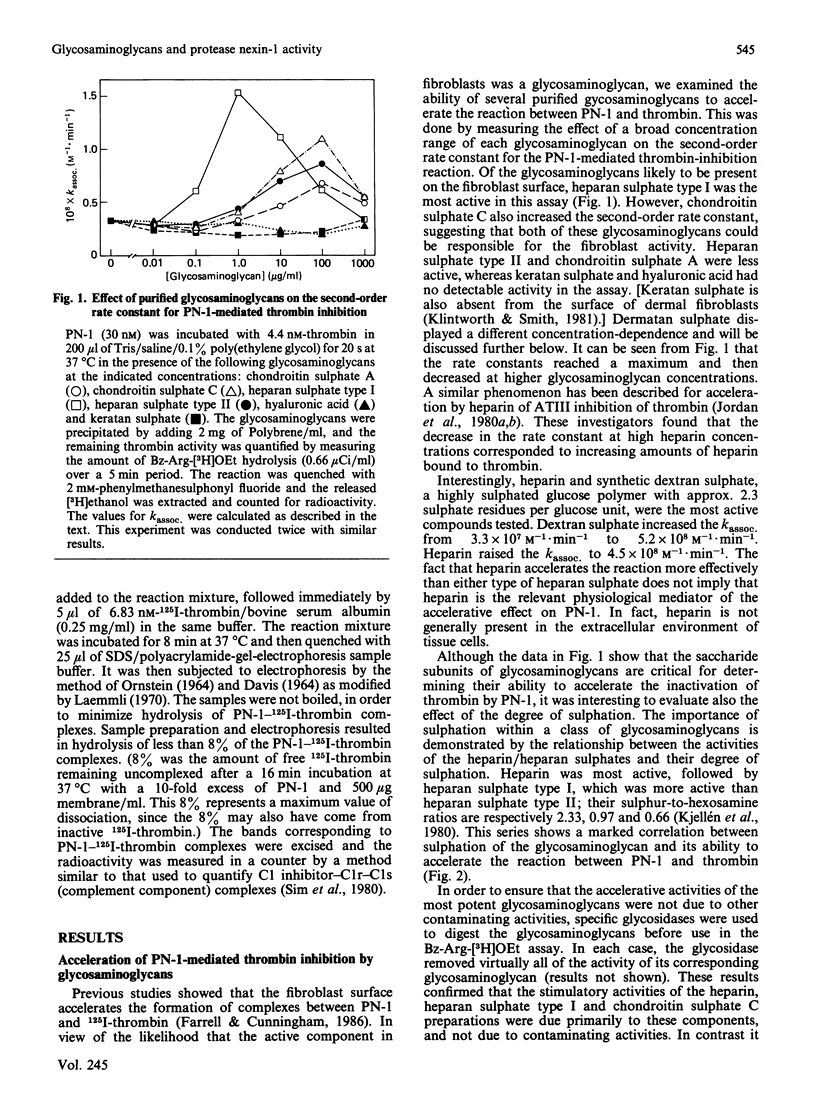
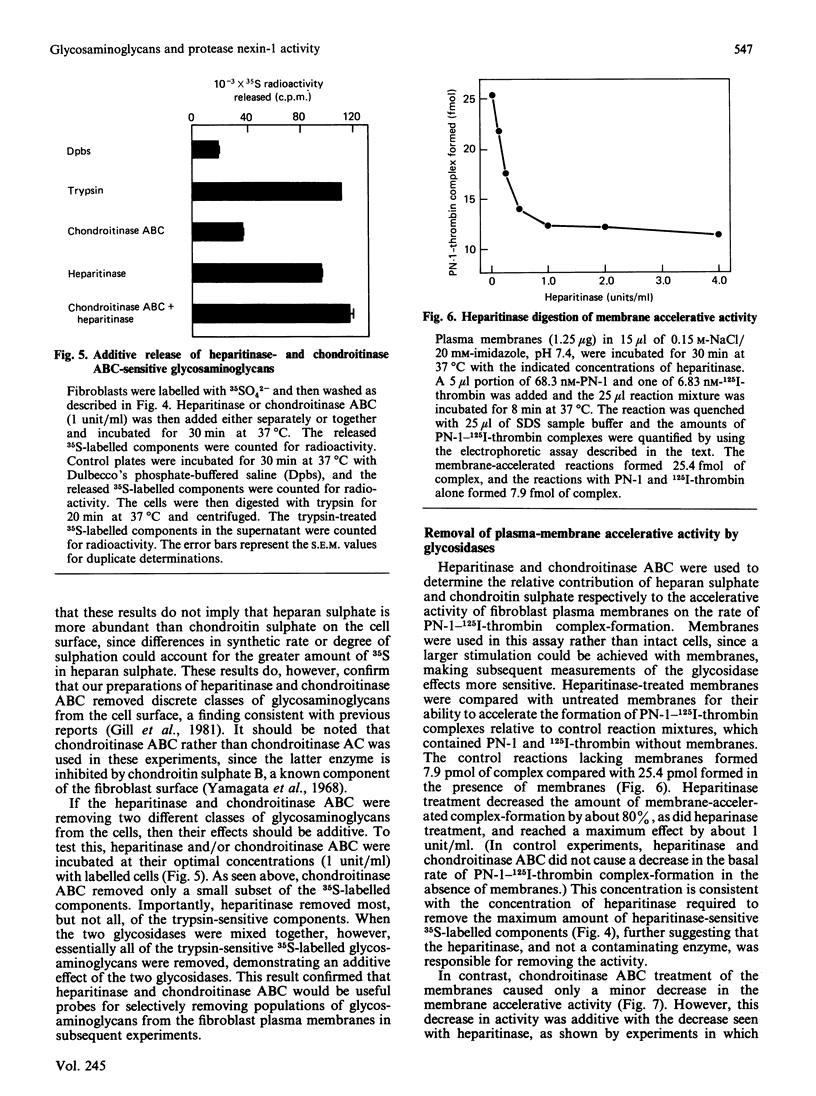
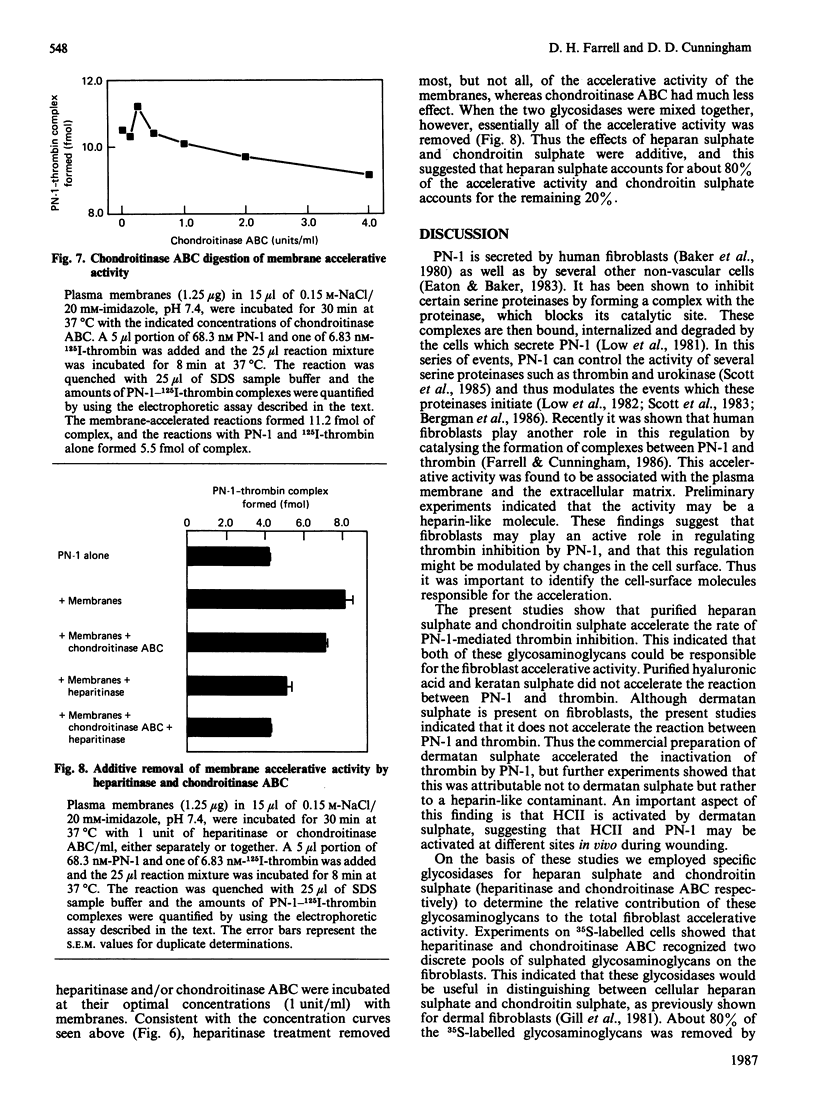
Protease nexin-1 (PN-1) is a proteinase inhibitor that is secreted by human fibroblasts in culture. PN-1 inhibits certain regulatory serine proteinases by forming a covalent complex with the catalytic-site serine residue; the complex then binds to the cell surface and is internalized and degraded. The fibroblast surface was recently shown to accelerate the rate of complex-formation between PN-1 and thrombin. The present paper demonstrates that the accelerative activity is primarily due to cell-surface heparan sulphate, with a much smaller contribution from chondroitin sulphate. This conclusion is supported by the effects of purified glycosaminoglycans on the second-order rate constant for the inhibition of thrombin by PN-1. Also, treatment of 35SO4(2-)-labelled cells with heparitin sulphate lyase or chondroitin sulphate ABC lyase demonstrated two discrete pools of 35S-labelled glycosaminoglycans; subsequent treatment of plasma membranes with these glycosidases showed that heparitin sulphate lyase treatment abolished about 80% of the accelerative activity and chondroitin sulphate ABC lyase removed the remaining 20%. These results show that two components are responsible for the acceleration of PN-1-thrombin complex-formation by human fibroblasts. Although dermatan sulphate is also present on fibroblasts, it did not accelerate the inhibition of thrombin by PN-1.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baker J. B., Low D. A., Simmer R. L., Cunningham D. D. Protease-nexin: a cellular component that links thrombin and plasminogen activator and mediates their binding to cells. Cell. 1980 Aug;21(1):37–45. doi: 10.1016/0092-8674(80)90112-9. [DOI] [PubMed] [Google Scholar]
- Bergman B. L., Scott R. W., Bajpai A., Watts S., Baker J. B. Inhibition of tumor-cell-mediated extracellular matrix destruction by a fibroblast proteinase inhibitor, protease nexin I. Proc Natl Acad Sci U S A. 1986 Feb;83(4):996–1000. doi: 10.1073/pnas.83.4.996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chase T., Jr, Shaw E. Comparison of the esterase activities of trypsin, plasmin, and thrombin on guanidinobenzoate esters. Titration of the enzymes. Biochemistry. 1969 May;8(5):2212–2224. doi: 10.1021/bi00833a063. [DOI] [PubMed] [Google Scholar]
- Chen L. B., Buchanan J. M. Mitogenic activity of blood components. I. Thrombin and prothrombin. Proc Natl Acad Sci U S A. 1975 Jan;72(1):131–135. doi: 10.1073/pnas.72.1.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- Danø K., Andreasen P. A., Grøndahl-Hansen J., Kristensen P., Nielsen L. S., Skriver L. Plasminogen activators, tissue degradation, and cancer. Adv Cancer Res. 1985;44:139–266. doi: 10.1016/s0065-230x(08)60028-7. [DOI] [PubMed] [Google Scholar]
- Eaton D. L., Baker J. B. Evidence that a variety of cultured cells secrete protease nexin and produce a distinct cytoplasmic serine protease-binding factor. J Cell Physiol. 1983 Nov;117(2):175–182. doi: 10.1002/jcp.1041170207. [DOI] [PubMed] [Google Scholar]
- Farrell D. H., Cunningham D. D. Human fibroblasts accelerate the inhibition of thrombin by protease nexin. Proc Natl Acad Sci U S A. 1986 Sep;83(18):6858–6862. doi: 10.1073/pnas.83.18.6858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farrell D. H., Van Nostrand W. E., Cunningham D. D. A simple two-step purification of protease nexin. Biochem J. 1986 Aug 1;237(3):907–912. doi: 10.1042/bj2370907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fenton J. W., 2nd, Fasco M. J., Stackrow A. B. Human thrombins. Production, evaluation, and properties of alpha-thrombin. J Biol Chem. 1977 Jun 10;252(11):3587–3598. [PubMed] [Google Scholar]
- Fraker P. J., Speck J. C., Jr Protein and cell membrane iodinations with a sparingly soluble chloroamide, 1,3,4,6-tetrachloro-3a,6a-diphrenylglycoluril. Biochem Biophys Res Commun. 1978 Feb 28;80(4):849–857. doi: 10.1016/0006-291x(78)91322-0. [DOI] [PubMed] [Google Scholar]
- Gill P. J., Adler J., Silbert C. K., Silbert J. E. Removal of glycosaminoglycans from cultures of human skin fibroblasts. Biochem J. 1981 Jan 15;194(1):299–307. doi: 10.1042/bj1940299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guenther J., Nick H., Monard D. A glia-derived neurite-promoting factor with protease inhibitory activity. EMBO J. 1985 Aug;4(8):1963–1966. doi: 10.1002/j.1460-2075.1985.tb03878.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kjellén L., Oldberg A., Hök M. Cell-surface heparan sulfate. Mechanisms of proteoglycan-cell association. J Biol Chem. 1980 Nov 10;255(21):10407–10413. [PubMed] [Google Scholar]
- Kleinman H. K., Silbert J. E., Silbert C. K. Heparan sulfate of skin fibroblasts grown in culture. Connect Tissue Res. 1975;4(1):17–23. doi: 10.3109/03008207509152193. [DOI] [PubMed] [Google Scholar]
- Klintworth G. K., Smith C. F. Difference between the glycosaminoglycans synthetized by corneal and cutaneous fibroblasts in culture. Lab Invest. 1981 Jun;44(6):553–559. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Low D. A., Baker J. B., Koonce W. C., Cunningham D. D. Released protease-nexin regulates cellular binding, internalization, and degradation of serine proteases. Proc Natl Acad Sci U S A. 1981 Apr;78(4):2340–2344. doi: 10.1073/pnas.78.4.2340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Low D. A., Scott R. W., Baker J. B., Cunningham D. D. Cells regulate their mitogenic response to thrombin through release of protease nexin. Nature. 1982 Jul 29;298(5873):476–478. doi: 10.1038/298476a0. [DOI] [PubMed] [Google Scholar]
- Marcum J. A., Fritze L., Galli S. J., Karp G., Rosenberg R. D. Microvascular heparin-like species with anticoagulant activity. Am J Physiol. 1983 Nov;245(5 Pt 1):H725–H733. doi: 10.1152/ajpheart.1983.245.5.H725. [DOI] [PubMed] [Google Scholar]
- Marcum J. A., Rosenberg R. D. Anticoagulantly active heparin-like molecules from vascular tissue. Biochemistry. 1984 Apr 10;23(8):1730–1737. doi: 10.1021/bi00303a023. [DOI] [PubMed] [Google Scholar]
- Marcum J. A., Rosenberg R. D. Heparinlike molecules with anticoagulant activity are synthesized by cultured endothelial cells. Biochem Biophys Res Commun. 1985 Jan 16;126(1):365–372. doi: 10.1016/0006-291x(85)90615-1. [DOI] [PubMed] [Google Scholar]
- ORNSTEIN L. DISC ELECTROPHORESIS. I. BACKGROUND AND THEORY. Ann N Y Acad Sci. 1964 Dec 28;121:321–349. doi: 10.1111/j.1749-6632.1964.tb14207.x. [DOI] [PubMed] [Google Scholar]
- Rosenberg R. D., Damus P. S. The purification and mechanism of action of human antithrombin-heparin cofactor. J Biol Chem. 1973 Sep 25;248(18):6490–6505. [PubMed] [Google Scholar]
- Scott R. W., Bergman B. L., Bajpai A., Hersh R. T., Rodriguez H., Jones B. N., Barreda C., Watts S., Baker J. B. Protease nexin. Properties and a modified purification procedure. J Biol Chem. 1985 Jun 10;260(11):7029–7034. [PubMed] [Google Scholar]
- Scott R. W., Eaton D. L., Duran N., Baker J. B. Regulation of extracellular plasminogen activator by human fibroblasts. The role of protease nexin. J Biol Chem. 1983 Apr 10;258(7):4397–4403. [PubMed] [Google Scholar]
- Sim R. B., Arlaud G. J., Colomb M. G. Kinetics of reaction of human C1-inhibitor with the human complement system proteases C1r and C1s. Biochim Biophys Acta. 1980 Apr 11;612(2):433–449. doi: 10.1016/0005-2744(80)90126-6. [DOI] [PubMed] [Google Scholar]
- Thom D., Powell A. J., Lloyd C. W., Rees D. A. Rapid isolation of plasma membranes in high yield from cultured fibroblasts. Biochem J. 1977 Nov 15;168(2):187–194. doi: 10.1042/bj1680187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tollefsen D. M., Blank M. K. Detection of a new heparin-dependent inhibitor of thrombin in human plasma. J Clin Invest. 1981 Sep;68(3):589–596. doi: 10.1172/JCI110292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tollefsen D. M., Pestka C. A., Monafo W. J. Activation of heparin cofactor II by dermatan sulfate. J Biol Chem. 1983 Jun 10;258(11):6713–6716. [PubMed] [Google Scholar]
- Wight T. N. Proteoglycans in pathological conditions: atherosclerosis. Fed Proc. 1985 Feb;44(2):381–385. [PubMed] [Google Scholar]
- Witt I., Kaiser C., Schrenk W. J., Wunderwald P. Amino acid composition and N-terminal sequence of antithrombin BM. Thromb Res. 1983 Dec 1;32(5):513–518. doi: 10.1016/0049-3848(83)90261-x. [DOI] [PubMed] [Google Scholar]
- Yamagata T., Saito H., Habuchi O., Suzuki S. Purification and properties of bacterial chondroitinases and chondrosulfatases. J Biol Chem. 1968 Apr 10;243(7):1523–1535. [PubMed] [Google Scholar]


