Abstract
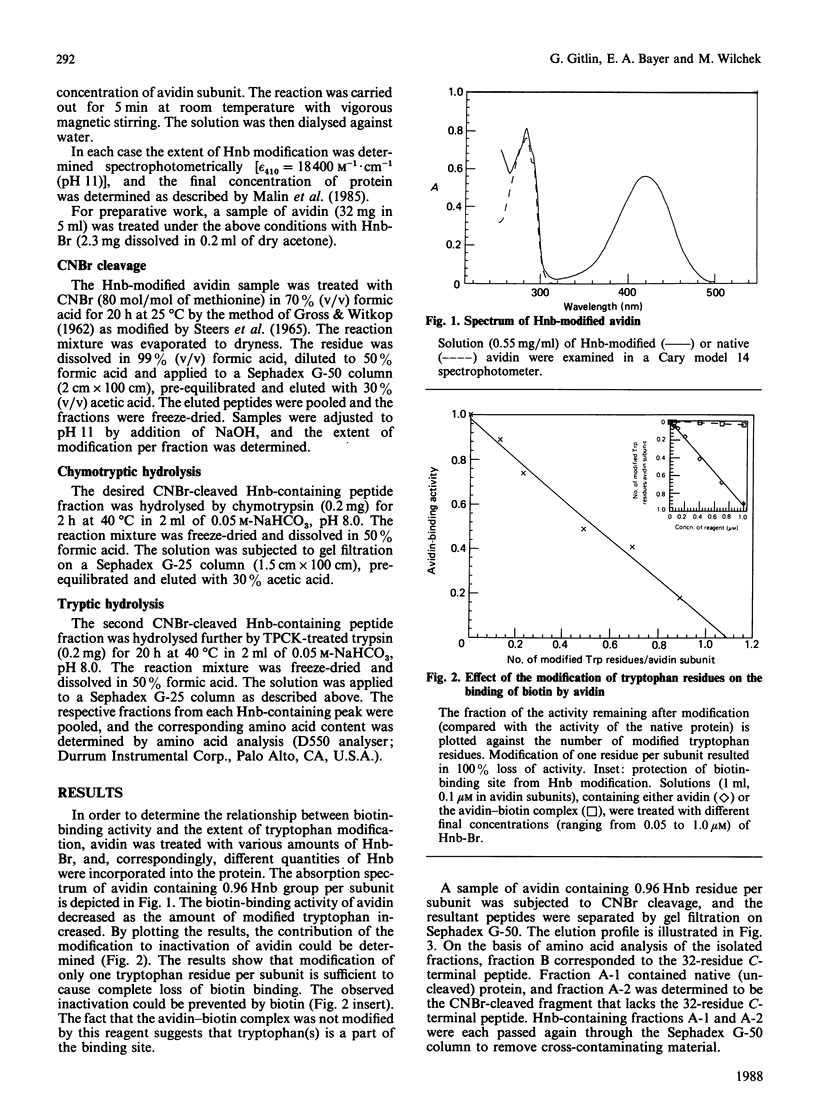
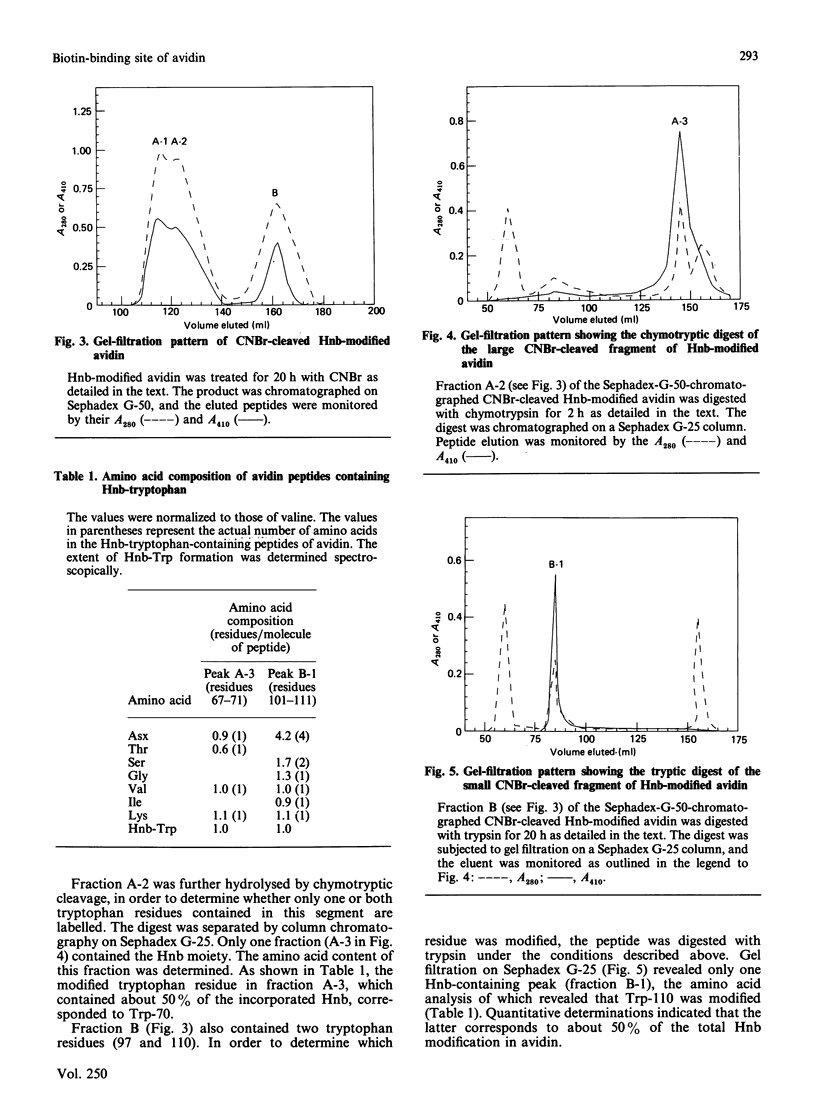
Egg-white avidin was modified with the tryptophan-specific reagent 2-hydroxy-5-nitrobenzyl bromide. The complete loss of biotin-binding activity was achieved upon modification of an average of one tryptophan residue per avidin subunit. The identity of the modified residues was determined by isolating the relevant tryptic and chymotryptic peptides from CNBr-cleaved avidin fragments. The results demonstrate that Trp-70 and Trp-110 are modified in approximately equivalent proportions. It is believed that these residues are located in the active site of avidin and take part in the binding of biotin.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Argaraña C. E., Kuntz I. D., Birken S., Axel R., Cantor C. R. Molecular cloning and nucleotide sequence of the streptavidin gene. Nucleic Acids Res. 1986 Feb 25;14(4):1871–1882. doi: 10.1093/nar/14.4.1871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bayer E. A., Ben-Hur H., Wilchek M. A sensitive enzyme assay for biotin, avidin, and streptavidin. Anal Biochem. 1986 Apr;154(1):367–370. doi: 10.1016/0003-2697(86)90538-5. [DOI] [PubMed] [Google Scholar]
- Bayer E. A., Wilchek M. The use of the avidin-biotin complex as a tool in molecular biology. Methods Biochem Anal. 1980;26:1–45. doi: 10.1002/9780470110461.ch1. [DOI] [PubMed] [Google Scholar]
- DeLange R. J., Huang T. S. Egg white avidin. 3. Sequence of the 78-residue middle cyanogen bromide peptide. Complete amino acid sequence of the protein subunit. J Biol Chem. 1971 Feb 10;246(3):698–709. [PubMed] [Google Scholar]
- FRAENKEL-CONRAT H., SNELL N. S., DUCAY E. D. Avidin. II. Composition and mode of action of avidin A. Arch Biochem Biophys. 1952 Jul;39(1):97–107. doi: 10.1016/0003-9861(52)90264-6. [DOI] [PubMed] [Google Scholar]
- GREEN N. M. AVIDIN. 3. THE NATURE OF THE BIOTIN-BINDING SITE. Biochem J. 1963 Dec;89:599–609. doi: 10.1042/bj0890599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GREEN N. M. Spectroscopic evidence for the participation of tryptophan residues in the binding of biotin by avidin. Biochim Biophys Acta. 1962 May 7;59:244–246. doi: 10.1016/0006-3002(62)90726-6. [DOI] [PubMed] [Google Scholar]
- GROSS E., WITKOP B. Nonenzymatic cleavage of peptide bonds: the methionine residues in bovine pancreatic ribonuclease. J Biol Chem. 1962 Jun;237:1856–1860. [PubMed] [Google Scholar]
- Gitlin G., Bayer E. A., Wilchek M. Studies on the biotin-binding site of avidin. Lysine residues involved in the active site. Biochem J. 1987 Mar 15;242(3):923–926. doi: 10.1042/bj2420923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green N. M. Avidin. Adv Protein Chem. 1975;29:85–133. doi: 10.1016/s0065-3233(08)60411-8. [DOI] [PubMed] [Google Scholar]
- HORTON H. R., KOSHLAND D. E., Jr A HIGHLY REACTIVE COLORED REAGENT WITH SELECTIVITY FOR THE TRYPTOPHAN RESIDUE IN PROTEINS. 2-HYDROXY-5-NITROBENZYL BROMIDE. J Am Chem Soc. 1965 Mar 5;87:1126–1132. doi: 10.1021/ja01083a033. [DOI] [PubMed] [Google Scholar]
- Malin E. L., Greenberg R., Farrell H. M., Jr Spectrophotometric estimation of protein concentration in the presence of tryptophan modified by 2-hydroxy-5-nitrobenzyl bromide. Anal Biochem. 1985 Feb 1;144(2):356–361. doi: 10.1016/0003-2697(85)90128-9. [DOI] [PubMed] [Google Scholar]
- STEERS E., Jr, CRAVEN G. R., ANFINSEN C. B., BETHUNE J. L. EVIDENCE FOR NONIDENTICAL CHAINS IN THE BETA-GALACTOSIDASE OF ESCHERICHIA COLI K12. J Biol Chem. 1965 Jun;240:2478–2484. [PubMed] [Google Scholar]


