Abstract
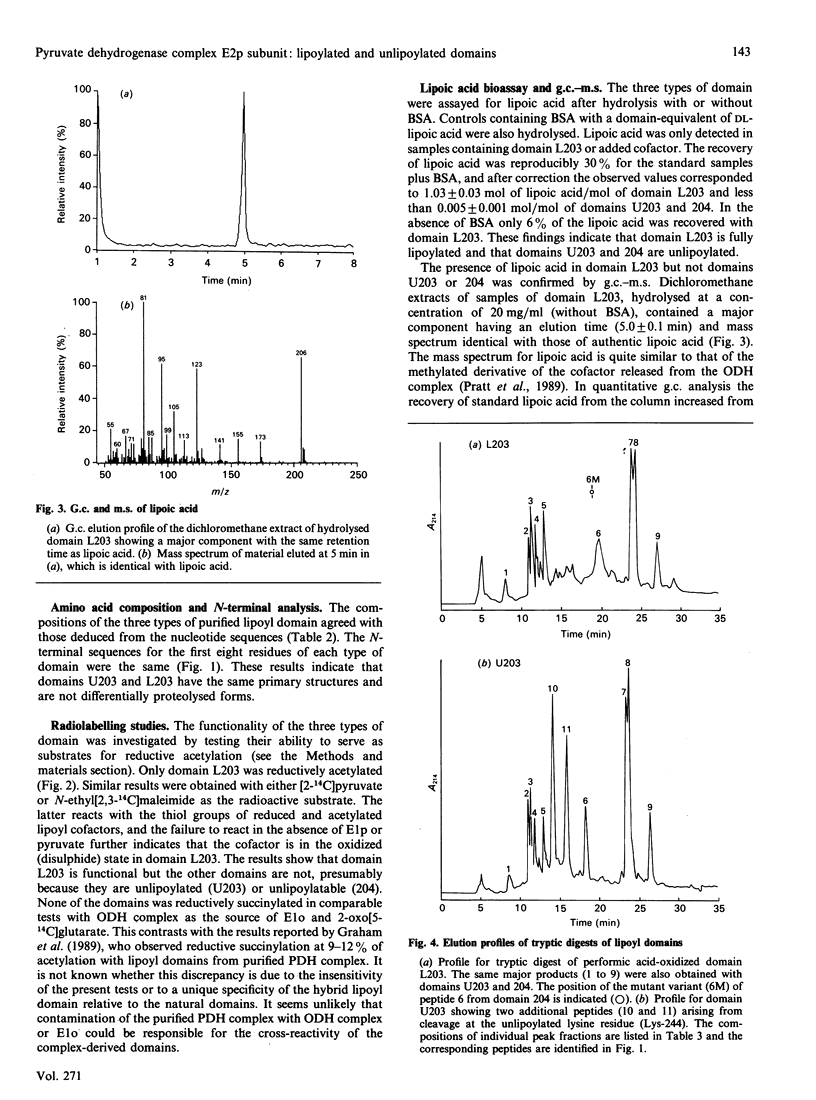
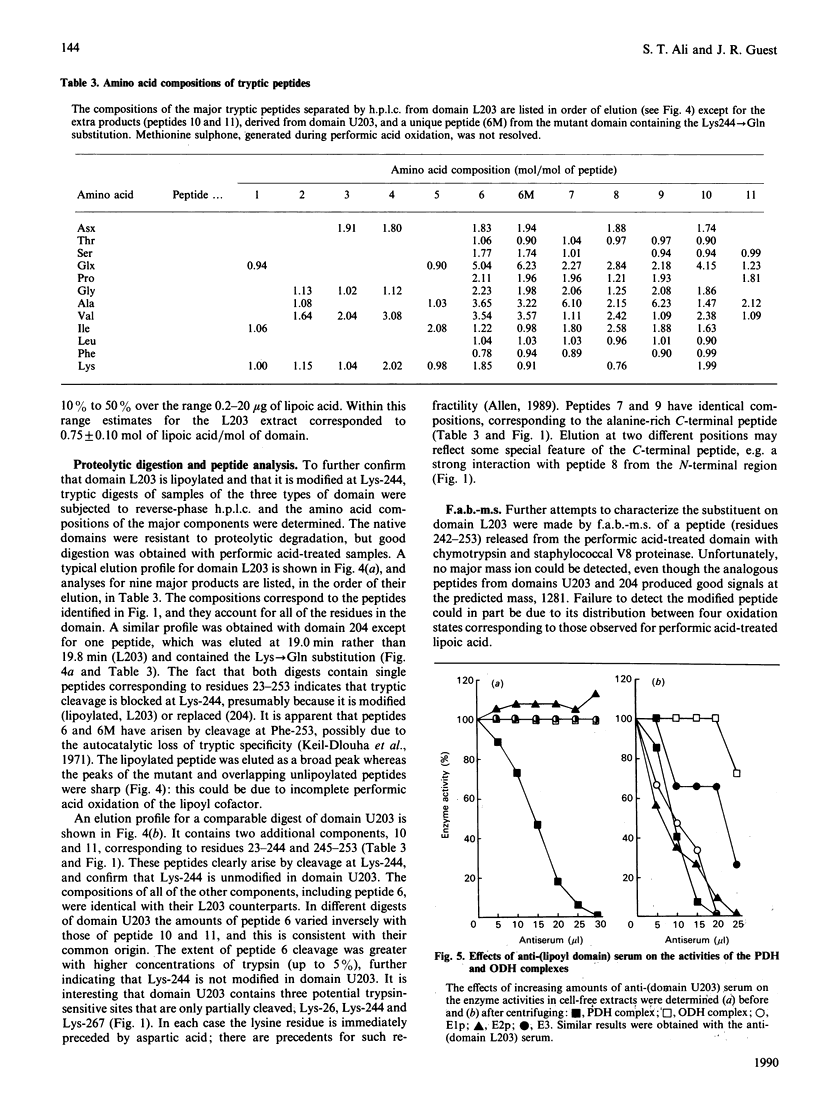
The dihydrolipoamide acetyltransferase subunit (E2p) of the pyruvate dehydrogenase complex of Escherichia coli has three highly conserved and tandemly repeated lipoyl domains, each containing approx. 80 amino acid residues. These domains are covalently modified with lipoyl groups bound in amide linkage to the N6-amino groups of specific lysine residues, and the cofactors perform essential roles in the formation and transfer of acetyl groups by the dehydrogenase (E1p) and acetyltransferase (E2p) subunits. A subgene encoding a hybrid lipoyl domain was previously shown to generate two products when overexpressed, whereas a mutant subgene, in which the lipoyl-lysine codon is replaced by a glutamine codon, expresses only one product. A method has been devised for purifying the three types of independently folded domain from crude extracts of E. coli, based on their pH-(and heat-)stabilities. The domains were characterized by: amino acid and N-terminal sequence analysis, lipoic acid content, acetylation by E1p, tryptic peptide analysis and immunochemical activity. This has shown that the two forms of domain expressed from the parental subgene are lipoylated (L203) and unlipoylated (U203) derivatives of the hybrid lipoyl domain, whereas the mutant subgene produces a single unlipoylatable domain (204) containing the Lys-244----Gln substitution.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ali S. T., Moir A. J., Ashton P. R., Engel P. C., Guest J. R. Octanoylation of the lipoyl domains of the pyruvate dehydrogenase complex in a lipoyl-deficient strain of Escherichia coli. Mol Microbiol. 1990 Jun;4(6):943–950. doi: 10.1111/j.1365-2958.1990.tb00667.x. [DOI] [PubMed] [Google Scholar]
- Allen A. G., Perham R. N., Allison N., Miles J. S., Guest J. R. Reductive acetylation of tandemly repeated lipoyl domains in the pyruvate dehydrogenase multienzyme complex of Escherichia coli is random order. J Mol Biol. 1989 Aug 20;208(4):623–633. doi: 10.1016/0022-2836(89)90153-8. [DOI] [PubMed] [Google Scholar]
- Allison N., Williams C. H., Jr, Guest J. R. Overexpression and mutagenesis of the lipoamide dehydrogenase of Escherichia coli. Biochem J. 1988 Dec 15;256(3):741–749. doi: 10.1042/bj2560741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ambrose M. C., Perham R. N. Spin-label study of the mobility of enzyme-bound lipoic acid in the pyruvate dehydrogenase multienzyme complex of Escherichia coli. Biochem J. 1976 May 1;155(2):429–432. doi: 10.1042/bj1550429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bleile D. M., Hackert M. L., Pettit F. H., Reed L. J. Subunit structure of dihydrolipoyl transacetylase component of pyruvate dehydrogenase complex from bovine heart. J Biol Chem. 1981 Jan 10;256(1):514–519. [PubMed] [Google Scholar]
- Fussey S. P., Ali S. T., Guest J. R., James O. F., Bassendine M. F., Yeaman S. J. Reactivity of primary biliary cirrhosis sera with Escherichia coli dihydrolipoamide acetyltransferase (E2p): characterization of the main immunogenic region. Proc Natl Acad Sci U S A. 1990 May;87(10):3987–3991. doi: 10.1073/pnas.87.10.3987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham L. D., Packman L. C., Perham R. N. Kinetics and specificity of reductive acylation of lipoyl domains from 2-oxo acid dehydrogenase multienzyme complexes. Biochemistry. 1989 Feb 21;28(4):1574–1581. doi: 10.1021/bi00430a023. [DOI] [PubMed] [Google Scholar]
- Guest J. R., Angier S. J., Russell G. C. Structure, expression, and protein engineering of the pyruvate dehydrogenase complex of Escherichia coli. Ann N Y Acad Sci. 1989;573:76–99. doi: 10.1111/j.1749-6632.1989.tb14988.x. [DOI] [PubMed] [Google Scholar]
- Guest J. R., Cole S. T., Jeyaseelan K. Organization and expression of the pyruvate dehydrogenase complex genes of Escherichia coli K12. J Gen Microbiol. 1981 Nov;127(1):65–79. doi: 10.1099/00221287-127-1-65. [DOI] [PubMed] [Google Scholar]
- Guest J. R., Lewis H. M., Graham L. D., Packman L. C., Perham R. N. Genetic reconstruction and functional analysis of the repeating lipoyl domains in the pyruvate dehydrogenase multienzyme complex of Escherichia coli. J Mol Biol. 1985 Oct 20;185(4):743–754. doi: 10.1016/0022-2836(85)90059-2. [DOI] [PubMed] [Google Scholar]
- HIRS C. H. The oxidation of ribonuclease with performic acid. J Biol Chem. 1956 Apr;219(2):611–621. [PubMed] [Google Scholar]
- Herbert A. A., Guest J. R. Biochemical and genetic studies with lysine+methionine mutants of Escherichia coli: lipoic acid and alpha-ketoglutarate dehydrogenase-less mutants. J Gen Microbiol. 1968 Oct;53(3):363–381. doi: 10.1099/00221287-53-3-363. [DOI] [PubMed] [Google Scholar]
- Herbert A. A., Guest J. R. Lipoic acid content of Escherichia coli and other microorganisms. Arch Microbiol. 1975 Dec 31;106(3):259–266. doi: 10.1007/BF00446532. [DOI] [PubMed] [Google Scholar]
- Jeyaseelan K., Guest J. R. Isolation and properties of pyruvate dehydrogenase complex mutants of Pseudomonas aeruginosa PAO. J Gen Microbiol. 1980 Oct;120(2):385–392. doi: 10.1099/00221287-120-2-385. [DOI] [PubMed] [Google Scholar]
- Keil-Dlouhá V., V, Zylber N., Imhoff J. -M., Tong N. -T., Keil B. Proteolytic activity of pseudotrypsin. FEBS Lett. 1971 Sep 1;16(4):291–295. doi: 10.1016/0014-5793(71)80373-3. [DOI] [PubMed] [Google Scholar]
- Kohara Y., Akiyama K., Isono K. The physical map of the whole E. coli chromosome: application of a new strategy for rapid analysis and sorting of a large genomic library. Cell. 1987 Jul 31;50(3):495–508. doi: 10.1016/0092-8674(87)90503-4. [DOI] [PubMed] [Google Scholar]
- LENNOX E. S. Transduction of linked genetic characters of the host by bacteriophage P1. Virology. 1955 Jul;1(2):190–206. doi: 10.1016/0042-6822(55)90016-7. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Langley D., Guest J. R. Biochemical and genetic characterics of deletion and other mutant strains of Salmonella typhimurium LT2 lacking alpha-keto acid dehydrogenase complex activities,. J Gen Microbiol. 1974 Jun;82(2):319–335. doi: 10.1099/00221287-82-2-319. [DOI] [PubMed] [Google Scholar]
- Marsh P. Ptac-85, an E. coli vector for expression of non-fusion proteins. Nucleic Acids Res. 1986 Apr 25;14(8):3603–3603. doi: 10.1093/nar/14.8.3603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miles J. S., Guest J. R., Radford S. E., Perham R. N. Investigation of the mechanism of active site coupling in the pyruvate dehydrogenase multienzyme complex of Escherichia coli by protein engineering. J Mol Biol. 1988 Jul 5;202(1):97–106. doi: 10.1016/0022-2836(88)90522-0. [DOI] [PubMed] [Google Scholar]
- Miles J. S., Guest J. R. Subgenes expressing single lipoyl domains of the pyruvate dehydrogenase complex of Escherichia coli. Biochem J. 1987 Aug 1;245(3):869–874. doi: 10.1042/bj2450869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Packman L. C., Hale G., Perham R. N. Repeating functional domains in the pyruvate dehydrogenase multienzyme complex of Escherichia coli. EMBO J. 1984 Jun;3(6):1315–1319. doi: 10.1002/j.1460-2075.1984.tb01969.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perham R. N., Packman L. C., Radford S. E. 2-Oxo acid dehydrogenase multi-enzyme complexes: in the beginning and halfway there. Biochem Soc Symp. 1987;54:67–81. [PubMed] [Google Scholar]
- Pratt K. J., Carles C., Carne T. J., Danson M. J., Stevenson K. J. Detection of bacterial lipoic acid. A modified gas-chromatographic-mass-spectrometric procedure. Biochem J. 1989 Mar 15;258(3):749–754. doi: 10.1042/bj2580749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- REED L. J., LEACH F. R., KOIKE M. Studies on a lipoic acid-activating system. J Biol Chem. 1958 May;232(1):123–142. [PubMed] [Google Scholar]
- Radford S. E., Perham R. N., Ullrich S. J., Appella E. Antibodies against an inter-domain segment of polypeptide chain inhibit active-site coupling in the pyruvate dehydrogenase multienzyme complex. FEBS Lett. 1989 Jul 3;250(2):336–340. doi: 10.1016/0014-5793(89)80750-1. [DOI] [PubMed] [Google Scholar]
- Russell G. C., Williamson R. A., Guest J. R. Partial complementation of pyruvate dehydrogenase deficiency by independently expressed lipoyl and catalytic domains of the dihydrolipoamide acetyltransferase component. FEMS Microbiol Lett. 1989 Aug;51(3):267–271. doi: 10.1016/0378-1097(89)90408-4. [DOI] [PubMed] [Google Scholar]
- Stephens P. E., Darlison M. G., Lewis H. M., Guest J. R. The pyruvate dehydrogenase complex of Escherichia coli K12. Nucleotide sequence encoding the dihydrolipoamide acetyltransferase component. Eur J Biochem. 1983 Jul 1;133(3):481–489. doi: 10.1111/j.1432-1033.1983.tb07490.x. [DOI] [PubMed] [Google Scholar]
- Stephens P. E., Darlison M. G., Lewis H. M., Guest J. R. The pyruvate dehydrogenase complex of Escherichia coli K12. Nucleotide sequence encoding the pyruvate dehydrogenase component. Eur J Biochem. 1983 Jun 1;133(1):155–162. doi: 10.1111/j.1432-1033.1983.tb07441.x. [DOI] [PubMed] [Google Scholar]
- Stephens P. E., Lewis H. M., Darlison M. G., Guest J. R. Nucleotide sequence of the lipoamide dehydrogenase gene of Escherichia coli K12. Eur J Biochem. 1983 Oct 3;135(3):519–527. doi: 10.1111/j.1432-1033.1983.tb07683.x. [DOI] [PubMed] [Google Scholar]
- Stepp L. R., Bleile D. M., McRorie D. K., Pettit F. H., Reed L. J. Use of trypsin and lipoamidase to study the role of lipoic acid moieties in the pyruvate and alpha-ketoglutarate dehydrogenase complexes of Escherichia coli. Biochemistry. 1981 Aug 4;20(16):4555–4560. doi: 10.1021/bi00519a007. [DOI] [PubMed] [Google Scholar]
- Texter F. L., Radford S. E., Laue E. D., Perham R. N., Miles J. S., Guest J. R. Site-directed mutagenesis and 1H NMR spectroscopy of an interdomain segment in the pyruvate dehydrogenase multienzyme complex of Escherichia coli. Biochemistry. 1988 Jan 12;27(1):289–296. doi: 10.1021/bi00401a044. [DOI] [PubMed] [Google Scholar]
- Yeaman S. J. The 2-oxo acid dehydrogenase complexes: recent advances. Biochem J. 1989 Feb 1;257(3):625–632. doi: 10.1042/bj2570625. [DOI] [PMC free article] [PubMed] [Google Scholar]



