Abstract
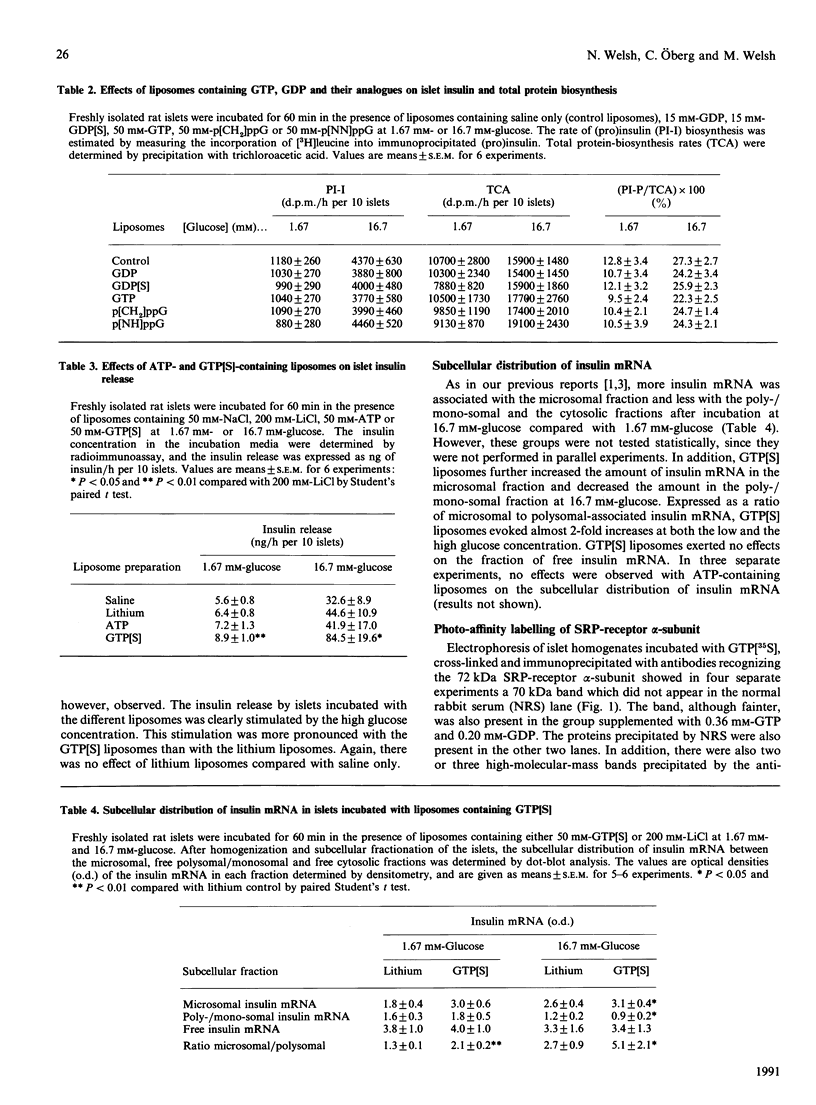
We aimed to elucidate the putative role of GTP-binding proteins in the regulation of insulin biosynthesis. For this purpose, freshly isolated rat islets were incubated in the presence of liposomes containing GDP, guanosine 5'-[beta-thio]diphosphate (GDP[S]), GTP, guanosine 5'-[gamma-thio]triphosphate (GTP[S]), guanosine 5'-[beta gamma-methylene]triphosphate (p[CH2]ppG), guanosine 5'[beta gamma-imido]triphosphate (p[NH]ppG) and ATP, and the effects of the liposomal delivery of these substances on rates of biosynthesis of insulin and total protein were determined. Insulin biosynthesis during a 1 h incubation at 1.67 mM-glucose was stimulated by ATP- and GTP[S]-containing liposomes as compared with control liposomes. At 16.7 mM-glucose, only the GTP[S]-containing liposomes stimulated insulin biosynthesis. No inhibition of islet protein and insulin synthesis was observed with GDP-, GDP[S]-, p[CH2]ppG- and p[NH]ppG-containing liposomes. By determining the subcellular distribution of insulin mRNA, it was found that the mRNA content associated with microsomes was increased and that associated with the cytosolic mono-/poly-somes decreased when the islets were incubated with GTP[S]-containing liposomes, resulting in an approximate doubling of the ratio of microsomal to polysomal-associated insulin mRNA. ATP-containing liposomes produced no effects on the association of insulin mRNA with microsomes. By using photoaffinity labelling and immunoprecipitation techniques, specific binding of GTP[35S] to the alpha-subunit of the signal-recognition particle (SRP) receptor in islet homogenates containing physiological concentrations of GTP and GDP was demonstrated. These findings suggest that the GTP-binding subunit(s) of the SRP receptor, and possibly also of other GTP-binding proteins involved in this process, may regulate insulin biosynthesis by stimulating the translocation of insulin mRNA to the endoplasmic reticulum and by increasing preproinsulin-peptide translocation into the lumen of the reticulum.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ashcroft F. M., Harrison D. E., Ashcroft S. J. Glucose induces closure of single potassium channels in isolated rat pancreatic beta-cells. 1984 Nov 29-Dec 5Nature. 312(5993):446–448. doi: 10.1038/312446a0. [DOI] [PubMed] [Google Scholar]
- Ashcroft S. J. Glucoreceptor mechanisms and the control of insulin release and biosynthesis. Diabetologia. 1980 Jan;18(1):5–15. doi: 10.1007/BF01228295. [DOI] [PubMed] [Google Scholar]
- Bag J., Sarkar S. Cytoplasmic nonpolysomal messenger ribonucleoprotein containing actin messenger RNA in chicken embryonic muscles. Biochemistry. 1975 Aug 26;14(17):3800–3807. doi: 10.1021/bi00688a012. [DOI] [PubMed] [Google Scholar]
- Bernstein H. D., Poritz M. A., Strub K., Hoben P. J., Brenner S., Walter P. Model for signal sequence recognition from amino-acid sequence of 54K subunit of signal recognition particle. Nature. 1989 Aug 10;340(6233):482–486. doi: 10.1038/340482a0. [DOI] [PubMed] [Google Scholar]
- Bokoch G. M., Katada T., Northup J. K., Ui M., Gilman A. G. Purification and properties of the inhibitory guanine nucleotide-binding regulatory component of adenylate cyclase. J Biol Chem. 1984 Mar 25;259(6):3560–3567. [PubMed] [Google Scholar]
- Cassel D., Selinger Z. Activation of turkey erythrocyte adenylate cyclase and blocking of the catecholamine-stimulated GTPase by guanosine 5'-(gamma-thio) triphosphate. Biochem Biophys Res Commun. 1977 Aug 8;77(3):868–873. doi: 10.1016/s0006-291x(77)80058-2. [DOI] [PubMed] [Google Scholar]
- Chan S. J., Noyes B. E., Agarwal K. L., Steiner D. F. Construction and selection of recombinant plasmids containing full-length complementary DNAs corresponding to rat insulins I and II. Proc Natl Acad Sci U S A. 1979 Oct;76(10):5036–5040. doi: 10.1073/pnas.76.10.5036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connolly T., Gilmore R. Formation of a functional ribosome-membrane junction during translocation requires the participation of a GTP-binding protein. J Cell Biol. 1986 Dec;103(6 Pt 1):2253–2261. doi: 10.1083/jcb.103.6.2253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connolly T., Gilmore R. The signal recognition particle receptor mediates the GTP-dependent displacement of SRP from the signal sequence of the nascent polypeptide. Cell. 1989 May 19;57(4):599–610. doi: 10.1016/0092-8674(89)90129-3. [DOI] [PubMed] [Google Scholar]
- Connor J., Yatvin M. B., Huang L. pH-sensitive liposomes: acid-induced liposome fusion. Proc Natl Acad Sci U S A. 1984 Mar;81(6):1715–1718. doi: 10.1073/pnas.81.6.1715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freissmuth M., Casey P. J., Gilman A. G. G proteins control diverse pathways of transmembrane signaling. FASEB J. 1989 Aug;3(10):2125–2131. [PubMed] [Google Scholar]
- Gilmore R., Blobel G. Transient involvement of signal recognition particle and its receptor in the microsomal membrane prior to protein translocation. Cell. 1983 Dec;35(3 Pt 2):677–685. doi: 10.1016/0092-8674(83)90100-9. [DOI] [PubMed] [Google Scholar]
- Gomperts B. D. Involvement of guanine nucleotide-binding protein in the gating of Ca2+ by receptors. Nature. 1983 Nov 3;306(5938):64–66. doi: 10.1038/306064a0. [DOI] [PubMed] [Google Scholar]
- Guest P. C., Rhodes C. J., Hutton J. C. Regulation of the biosynthesis of insulin-secretory-granule proteins. Co-ordinate translational control is exerted on some, but not all, granule matrix constituents. Biochem J. 1989 Jan 15;257(2):431–437. doi: 10.1042/bj2570431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halban P. A., Wollheim C. B., Blondel B., Renold A. E. Long-term exposure of isolated pancreatic islets to mannoheptulose: evidence for insulin degradation in the beta cell. Biochem Pharmacol. 1980 Oct 1;29(19):2625–2633. doi: 10.1016/0006-2952(80)90077-5. [DOI] [PubMed] [Google Scholar]
- Hallberg A. Effects of starvation and different culture conditions on the phospholipid content of isolated pancreatic islets. Biochim Biophys Acta. 1984 Dec 6;796(3):328–335. doi: 10.1016/0005-2760(84)90134-6. [DOI] [PubMed] [Google Scholar]
- Heding L. G. Determination of total serum insulin (IRI) in insulin-treated diabetic patients. Diabetologia. 1972 Aug;8(4):260–266. doi: 10.1007/BF01225569. [DOI] [PubMed] [Google Scholar]
- Hoenig M., Matschinsky F. M. HPLC analysis of nucleotide profiles in glucose-stimulated perifused rat islets. Metabolism. 1987 Mar;36(3):295–301. doi: 10.1016/0026-0495(87)90192-2. [DOI] [PubMed] [Google Scholar]
- Hortsch M., Avossa D., Meyer D. I. A structural and functional analysis of the docking protein. Characterization of active domains by proteolysis and specific antibodies. J Biol Chem. 1985 Aug 5;260(16):9137–9145. [PubMed] [Google Scholar]
- Howell S. L., Taylor K. W. Potassium ions and the secretion of insulin by islets of Langerhans incubated in vitro. Biochem J. 1968 Jun;108(1):17–24. doi: 10.1042/bj1080017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lernmark A., Nathans A., Steiner D. F. Preparation and characterization of plasma membrane-enriched fractions from rat pancreatic islets. J Cell Biol. 1976 Nov;71(2):606–623. doi: 10.1083/jcb.71.2.606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melançon P., Glick B. S., Malhotra V., Weidman P. J., Serafini T., Gleason M. L., Orci L., Rothman J. E. Involvement of GTP-binding "G" proteins in transport through the Golgi stack. Cell. 1987 Dec 24;51(6):1053–1062. doi: 10.1016/0092-8674(87)90591-5. [DOI] [PubMed] [Google Scholar]
- Nakajima Y., Nakajima S., Inoue M. Pertussis toxin-insensitive G protein mediates substance P-induced inhibition of potassium channels in brain neurons. Proc Natl Acad Sci U S A. 1988 May;85(10):3643–3647. doi: 10.1073/pnas.85.10.3643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oberg C., Welsh N. Nonspecific delivery of substances into pancreatic islet cells by pH-sensitive liposomes in vitro. Diabete Metab. 1990 Jan-Feb;16(1):48–54. [PubMed] [Google Scholar]
- Olson F., Hunt C. A., Szoka F. C., Vail W. J., Papahadjopoulos D. Preparation of liposomes of defined size distribution by extrusion through polycarbonate membranes. Biochim Biophys Acta. 1979 Oct 19;557(1):9–23. doi: 10.1016/0005-2736(79)90085-3. [DOI] [PubMed] [Google Scholar]
- Poste G., Papahadjopoulos D., Vail W. J. Lipid vesicles as carriers for introducing biologically active materials into cells. Methods Cell Biol. 1976;14:33–71. doi: 10.1016/s0091-679x(08)60468-9. [DOI] [PubMed] [Google Scholar]
- Ramsey J. C., Steele W. J. A procedure for the quantitative recovery of homogeneous populations of undegraded free and bound polysomes from rat liver. Biochemistry. 1976 Apr 20;15(8):1704–1712. doi: 10.1021/bi00653a018. [DOI] [PubMed] [Google Scholar]
- Regazzi R., Li G., Ullrich S., Jaggi C., Wollheim C. B. Different requirements for protein kinase C activation and Ca2+-independent insulin secretion in response to guanine nucleotides. Endogenously generated diacylglycerol requires elevated Ca2+ for kinase C insertion into membranes. J Biol Chem. 1989 Jun 15;264(17):9939–9944. [PubMed] [Google Scholar]
- Rothman J. E. Signal-peptide recognition. GTP and methionine bristles. Nature. 1989 Aug 10;340(6233):433–434. doi: 10.1038/340433a0. [DOI] [PubMed] [Google Scholar]
- Römisch K., Webb J., Herz J., Prehn S., Frank R., Vingron M., Dobberstein B. Homology of 54K protein of signal-recognition particle, docking protein and two E. coli proteins with putative GTP-binding domains. Nature. 1989 Aug 10;340(6233):478–482. doi: 10.1038/340478a0. [DOI] [PubMed] [Google Scholar]
- Thomas P. S. Hybridization of denatured RNA and small DNA fragments transferred to nitrocellulose. Proc Natl Acad Sci U S A. 1980 Sep;77(9):5201–5205. doi: 10.1073/pnas.77.9.5201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson R. C., Karim A. M. The accuracy of protein biosynthesis is limited by its speed: high fidelity selection by ribosomes of aminoacyl-tRNA ternary complexes containing GTP[gamma S]. Proc Natl Acad Sci U S A. 1982 Aug;79(16):4922–4926. doi: 10.1073/pnas.79.16.4922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vallar L., Biden T. J., Wollheim C. B. Guanine nucleotides induce Ca2+-independent insulin secretion from permeabilized RINm5F cells. J Biol Chem. 1987 Apr 15;262(11):5049–5056. [PubMed] [Google Scholar]
- Waldo G. L., Evans T., Fraser E. D., Northup J. K., Martin M. W., Harden T. K. Identification and purification from bovine brain of a guanine-nucleotide-binding protein distinct from Gs, Gi and Go. Biochem J. 1987 Sep 1;246(2):431–439. doi: 10.1042/bj2460431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welsh M., Hammer R. E., Brinster R. L., Steiner D. F. Stimulation of growth hormone synthesis by glucose in islets of Langerhans isolated from transgenic mice. J Biol Chem. 1986 Oct 5;261(28):12915–12917. [PubMed] [Google Scholar]
- Welsh M., Scherberg N., Gilmore R., Steiner D. F. Translational control of insulin biosynthesis. Evidence for regulation of elongation, initiation and signal-recognition-particle-mediated translational arrest by glucose. Biochem J. 1986 Apr 15;235(2):459–467. doi: 10.1042/bj2350459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welsh N., Hallberg A., Sandler S., Hellerström C. Use of liposomes to introduce substances into pancreatic islet cells. Diabetes. 1988 Aug;37(8):1123–1128. doi: 10.2337/diab.37.8.1123. [DOI] [PubMed] [Google Scholar]
- Welsh N., Sjöholm A. Polyamines and insulin production in isolated mouse pancreatic islets. Biochem J. 1988 Jun 15;252(3):701–707. doi: 10.1042/bj2520701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welsh N., Welsh M., Steiner D. F., Hellerström C. Mechanisms of leucine- and theophylline-stimulated insulin biosynthesis in isolated rat pancreatic islets. Biochem J. 1987 Aug 15;246(1):245–248. doi: 10.1042/bj2460245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White B. A., Bancroft F. C. Cytoplasmic dot hybridization. Simple analysis of relative mRNA levels in multiple small cell or tissue samples. J Biol Chem. 1982 Aug 10;257(15):8569–8572. [PubMed] [Google Scholar]
- von Heijne G. Signal sequences. The limits of variation. J Mol Biol. 1985 Jul 5;184(1):99–105. doi: 10.1016/0022-2836(85)90046-4. [DOI] [PubMed] [Google Scholar]



