Abstract
O6-methylguanine DNA methyltransferase (MGMT) promoter methylation is an important clinical biomarker of newly diagnosed glioblastoma. Previous radiological studies using dynamic susceptibility contrast (DSC) magnetic resonance imaging (MRI) perfusion have aimed to predict MGMT methylation status non-invasively in gliomas with radiological characteristics. The possibility of predicting MGMT methylation status using DSC-MRI perfusion with a radiological approach remains controversial. The present study aimed to evaluate the usefulness of MRI perfusion parameters as non-invasive markers to predict MGMT methylation status and prognosis in newly diagnosed glioblastoma patients. Thus, 50 patients with histologically confirmed primary glioblastoma, IDH-wildtype who underwent tumor resection at Osaka University Hospital (Suita, Japan) between January 2017 and January 2023 were included in this study. The mean cerebral blood volume (CBV) ratio (rCBV) and cerebral blood flow (CBF) ratio (rCBF) for tumors with MGMT methylation (mean rCBV:2.09 and mean rCBF:3.08) were significantly higher compared with those for tumors without MGMT methylation (mean rCBV:1.33 and mean rCBF:1.85; P<0.05). While patients with MGMT methylation had longer progression-free survival (PFS) compared with those without MGMT methylation (P<0.05), there was no significant difference in OS with or without MGMT methylation (P=0.06). By contrast, there was no association between MRI perfusion parameters and OS or PFS in patients with glioblastoma. Furthermore, the association between CBV, CBF, MGMT promotor methylation status, OS, and PFS were explored. There was no significant prognostic difference between low vascularity tumors (rCBV <1.3 or rCBF <1.8) with or without MGMT methylation. On the other hand, high vascularity tumors (rCBF ≥1.8) with MGMT promotor methylation were associated to longer OS and PFS compared with those without. However, there was no association between MGMT methylation status and OS or PFS in patients with high rCBV (rCBV ≥1.3). The present study indicated that CBV and CBF could be used to predict the MGMT methylation status in glioblastomas. However, the prognostic value of tumor vascularity and MGMT methylation status may be limited.
Keywords: dynamic susceptibility contrast-enhanced perfusion magnetic resonance imaging, MGMT promoter methylation, glioblastoma
Introduction
Glioblastoma is the most common type of malignant brain tumor. The prognosis of glioblastoma is extremely poor, even with standard treatments, such as chemoradiotherapy. O6-methylguanine DNA methyltransferase (MGMT) promoter methylation is associated with favorable outcomes after temozolomide (TMZ) chemotherapy in patients with newly diagnosed glioblastoma (1). Thus, the evaluation of MGMT methylation status is important for the treatment of these patients.
Several radiological studies have shown the potential of conventional magnetic resonance imaging (MRI) to predict the MGMT methylation status using image texture and deep learning architectures (2–7). Dynamic susceptibility contrast (DSC) MRI offers insight into tumor tissue vascularity by analyzing perfusion. Previous radiological studies using MRI perfusion have aimed to predict MGMT methylation status noninvasively in gliomas using radiological characteristics (5,8–11) and radiomics (12–15). Some reports revealed that DSC-MRI could be used as a noninvasive technique to predict genetic mutations preoperatively without surgical specimen, and to determine molecular characteristics such as IDH mutation status and methylation status of the MGMT promoter in glioblastomas (5,8–10,16). In contrast, other reports have indicated that cerebral blood volume (CBV) did not differ significantly between tumors with methylated or unmethylated MGMT (17,18). The possibility of predicting MGMT methylation status from DSC-MRI perfusion using a radiological approach remains controversial, and there is no expert consensus regarding clinical use.
The potential impact of DSC-MRI perfusion in the prediction of MGMT methylation status in glioblastoma remains disputed. In previous reports, elevated CBV has been associated with decreased survival of glioblastoma patients (19–22). In contrast, MGMT methylation status is highly correlated with survival in glioblastomas with moderate vascularity, but not in those with high vascularity (17,23). Furthermore, patients with glioblastomas showing stable or increasing CBV following chemoradiotherapy experienced significantly improved PFS, particularly in those cases presenting MGMT methylation (24).
This study aimed to evaluate the possibility of using DSC-MRI perfusion as a non-invasive method to predict MGMT methylation status and prognosis in newly diagnosed glioblastoma patients.
Materials and methods
Study design and patient selection
This retrospective study was approved by the Clinical Research Review Committee of Osaka University (Approval No. 22302). The inclusion criteria were as follows: patients who i) have definite pathological results; ii) have MRI images available, including conventional MRI and DSC-MRI, before surgery; iii) did not undergo radiotherapy or chemotherapy before MRI examination; iv) have an available MGMT promoter methylation status. Patients with recurrent tumors, tumors with unsatisfactory images, and young patients aged less than 18 years old were excluded from the study. Fifty patients with histologically confirmed primary glioblastoma, IDH-wildtype (according to the 2021 World Health Organization International Histological Classification of Tumors) who underwent tumor resection at our institution between January 2017 and January 2023 were included in the study (34 men and 16 women; median age, 70.5 years; Table I). All patients were diagnosed according to the 2021 guidelines, regardless of the resection date. DSC-MRI and conventional MRI pulse sequences were acquired preoperatively for all patients. All patients underwent surgical resection with concomitant TMZ treatment and radiotherapy, followed by adjuvant TMZ treatment. Tumor samples were collected after resection.
Table I.
Characteristics of patients with glioblastoma.
| Characteristic | Number of patients | Values | % |
|---|---|---|---|
| Sex | |||
| Male | 34 | 68.0 | |
| Female | 16 | 32.0 | |
| Age, years | |||
| Median | 70.5 | ||
| Range | 24-88 | ||
| MGMT promoter methylation, % | |||
| Median | 0.41 | ||
| Range | 0.00–92.07 | ||
| CBV ratio | |||
| Mean | 1.66 | ||
| Range | 0.51–5.14 | ||
| CBF ratio | |||
| Mean | 2.39 | ||
| Range | 0.49–11.03 | ||
| MTT ratio | |||
| Mean | 0.81 | ||
| Range | 0.44–1.46 | ||
| PFS, months | |||
| Median | 8.6 | ||
| Range | 2.2–60.1 | ||
| Overall survival, months | |||
| Median | 19.5 | ||
| Range | 2.5–60.1 |
Magnetic resonance imaging
All images, including axial T1-, T2-, and T2*-weighted images, fluid-attenuated inversion recovery, and contrast-enhanced T1-weighted sequences (T1Gd) were obtained using a 3-T MR unit (DISCOVERY MR 750; GE Healthcare, Milwaukee, WI, USA) with a 32-channel head coil. Perfusion MRI was performed using a T2*-weighted, single-shot, gradient-recalled, echo-planar imaging (GRE EPI) sequence. The perfusion MRI sequence parameters were as follows: repetition time/echo time, 2000/13.3 ms; matrix, 128×128; flip angle, 60; section thickness, 5 mm; and acquisition time, 90 sec. The contrast, a standard dose of 0.1 mmol/kg body weight of meglumine gadoterate (Guerbet Japan, Tokyo, Japan), was injected at a rate of 2–3 ml/s, followed by saline flush using a power injector.
Imaging analysis
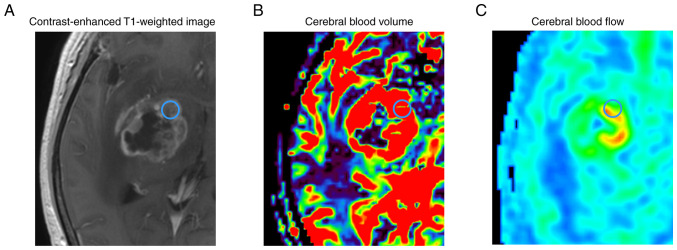
Imaging analysis was performed using Synapse Vincent (Fuji Medical Systems, Tokyo, Japan) in perfusion mode. A single region of interest (ROI) with a diameter of 5 mm (Fig. 1) was set manually on the solid part in each of the enhancing tumor regions from every patient, avoiding areas of cyst formation, hemorrhage, and large vessels, as per previous reports (16,25,26), and the values of CBV, cerebral blood flow (CBF), and mean transit time (MTT) were calculated. The ROI was manually set in the contralateral normal area for each enhanced tumor. The contralateral area was normal and contained no tumor infiltration.
Figure 1.
Tumor in right basal ganglia: (A) Contrast-enhanced T1-weighted image; (B) cerebral blood volume; (C) cerebral blood flow. The blue circle stands for the region of interest delineated.
Disease-to-normal ratios were calculated by dividing the values of CBV, CBF, and MTT for the tumors by the values of the contralateral normal area, and described as rCBV, rCBE, and rMTT, respectively.
Genomic DNA extraction
Tumor samples were immediately frozen and stored at −80°C or immersed in RNAlater Stabilization Solution (Thermo Fisher Scientific, Waltham, MA). Genomic DNA was extracted using a DNeasy Blood & Tissue Kit (Qiagen, Valencia, CA, USA) or NucleoSpin Tissue (Macherey-Nagel, Düren, Germany), as described previously (27).
MGMT promoter methylation analysis
The methylation status of the MGMT promoter (accession number: NM_002412.5) was assessed using quantitative methylation-specific PCR (qMSP). Purified DNA was subjected to bisulfite modification by an EZ DNA Methylation-Gold Kit (Zymo Research, Irvine, CA), according to the manufacturer's instructions. The qMSP was performed on a QuantStudio12K Flex Real-Time PCR System (Thermo Fisher Scientific) with POWER SYBR® Green PCR Master Mix (Thermo Fisher Scientific). The bisulfite-modified DNA was amplified using specific primers for each methylated or unmethylated molecule as listed in Table II. Real-time PCR conditions were as follows: 95°C for 10 min followed by 45 cycles of 95°C for 10 sec, and 60°C for 60 sec. The quantification of methylated and unmethylated sequences was performed by employing the standard curve method as previously described. In the dissociation curve analysis, heterogeneity of the amplified methylated and unmethylated molecules was assessed from melting temperature. The mean ± standard deviation of methylation value was calculated from triplicate PCRs. We used a 1% cut-off value for the determination of MGMT methylation based on an outcome-based study of newly diagnosed GBMs as mentioned in our previous publications (27,28). Sequences of primers used for quantitative methylation-specific PCR are provided in Table II.
Table II.
Sequences of primers used for quantitative methylation specific PCR.
| Gene | Primer | Sequence (5′-3′) |
|---|---|---|
| MGMT promoter | M-forward | TTTCGACGTTCGTAGGTTTTCGC |
| M-reverse | GCACTCTTCCGAAAACGAAACG | |
| U-forward | TTTGTGTTTTGATGTTTGTAGGTTTTTGT | |
| U-reverse | AACTCCACACTCTTCCAAAAACAAAACA |
M Methylated; U, Unmethylated.
Statistical analysis
Statistical analyses were performed using Prism version 9 (GraphPad Software, San Diego, CA, USA). Results were considered statistically significant at a P-value of <0.05. The unpaired t-test was used for comparisons between two groups. Receiver operating characteristic (ROC) curve analysis was performed to compare the performance of each imaging parameter based on each ROI in distinguishing tumors with MGMT methylation from those without MGMT methylation. The Kaplan-Meier method was used to derive OS and PFS curves.
We also attempted to construct a model based on three perfusion parameters to determine MGMT methylation status in glioblastomas by performing multiple logistic regression.
Results
Perfusion MRI parameters and MGMT methylation status
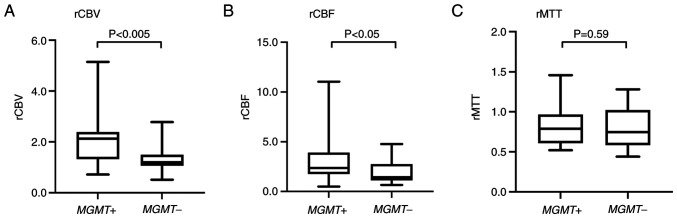
The mean rCBV for tumors with MGMT methylation (2.09; range, 0.72–5.14) was significantly higher than that for tumors without MGMT methylation (1.33; range, 0.51–2.78; P<0.005). The mean rCBF for tumors with MGMT methylation (3.08; range, 0.49–11.03) was significantly higher than that for tumors without MGMT methylation (1.85; range, 0.65–4.77; P<0.05). In contrast, the rMTT for tumors with and without MGMT methylation did not differ (Fig. 2, Table III).
Figure 2.
Boxplots of (A) rCBV), (B) rCBF) and (C) rMTT in tumors with or without MGMT methylation. Box plots show statistical differences in rCBV and rCBF in tumors with or without MGMT methylation. By contrast, rMTT for tumors with and without MGMT methylation did not differ. rCBV, cerebral blood volume ratio; rCBF, cerebral blood flow ratio; rMTT, MTT ratio; MGMT, O6-methylguanine DNA methyltransferase.
Table III.
Correlation between MRI perfusion parameters and MGMT promoter methylation status in patients with glioblastoma.
| MGMT promotor methylation status | Univariate | ||
|---|---|---|---|
|
|
|
||
| Perfusion parameters | Methylated (n=22) | Unmethylated (n=28) | P-value |
| Mean CBV ratio | 2.09 | 1.33 | 0.002 |
| Mean CBF ratio | 3.08 | 1.85 | 0.020 |
| Mean MTT ratio | 0.83 | 0.79 | 0.590 |
CBV, cerebral blood volume; CBF, cerebral blood flow; MTT, mean transit time.
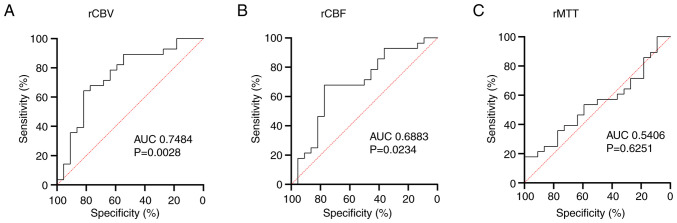
Receiver operating characteristic (ROC) analysis showed that the rCBV [area under the curve (AUC)=0.7484] and rCBF (AUC=0.6883) were more effective in distinguishing between tumors with and without MGMT methylation than the rMTT (AUC=0.5406; Fig. 3).
Figure 3.
Receiver operating characteristic curve showed more reliable predictions that distinguished between tumors with and without MGMT methylation in glioblastomas in terms of (A) rCBV, (B) rCBF and (C) rMTT. rCBV, cerebral blood volume ratio; rCBF, cerebral blood flow ratio; rMTT, MTT ratio; MGMT, O6-methylguanine DNA methyltransferase; AUC, area under the curve.
We attempted to construct a model based on three perfusion parameters to determine MGMT methylation status in glioblastomas by performing multiple logistic regression. The following predictive formula, created using parameters derived from the multiple logistic regression, was obtained to estimate the probability of MGMT methylation (probability range: 0 to 1) for each ROI:
log_odds=0.01832 + 4.743 * rCBV + 1.034 * rCBF + 4.214 * rMTT odds=exp(log_odds)
Probability=odds/(ones(size(odds)) + odds)
Prognosis according to MGMT methylation status
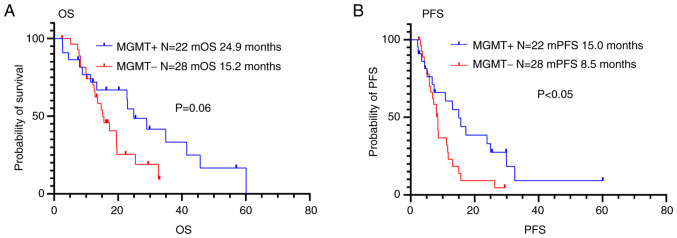
The PFS and OS were 15.0 months and 24.9 months, respectively, in the patients with MGMT methylation, and 8.5 months and 15.2 months, respectively, in the patients without MGMT methylation (Fig. 4 and Table IV). Patients with MGMT methylation had longer PFS than those without MGMT methylation (P<0.05), but there was no significant difference in OS between patients with and without MGMT methylation (P=0.06).
Figure 4.
Kaplan-Meier (A) OS and (B) PFS curves of the patients with glioblastoma grouped according to MGMT methylation status. The patients with MGMT methylation (n=22) had longer PFS compared with those without MGMT methylation (n=28) (P<0.05), but there was no significant difference between patients with and without MGMT methylation in OS. OS, Overall survival; PFS, progression-free survival; MGMT, O6-methylguanine DNA methyltransferase.
Table IV.
Univariate analyses of median survival time and PFS of patients with glioblastoma.
| Variable | No. of cases | MST | P-value (log-rank) | PFS | P-value (log-rank) |
|---|---|---|---|---|---|
| MGMT promoter methylation status | |||||
| Methylated | 22 | 24.9 | 0.06 | 15.0 | 0.03 |
| Unmethylated | 28 | 15.2 | 8.5 | ||
| CBV ratio | |||||
| <1.3 | 23 | 19.5 | 0.53 | 8.3 | 0.72 |
| ≥1.3 | 27 | 14.7 | 11.3 | ||
| CBF ratio | |||||
| <1.8 | 24 | 22.8 | 0.10 | 8.6 | 0.86 |
| ≥1.8 | 26 | 13.6 | 10.9 | ||
| CBV ratio <1.3 | |||||
| MGMT methylated | 5 | 45.7 | 0.15 | 15.7 | 0.23 |
| MGMT unmethylated | 18 | 17.3 | 8.0 | ||
| CBV ratio ≥1.3 | |||||
| MGMT methylated | 17 | 24.9 | 0.07 | 15.0 | 0.06 |
| MGMT unmethylated | 10 | 13.1 | 8.6 | ||
| CBF ratio <1.8 | |||||
| MGMT methylated | 5 | NA | 0.22 | 32.6 | 0.09 |
| MGMT unmethylated | 19 | 19.5 | 8.6 | ||
| CBF ratio ≥1.8 | |||||
| MGMT methylated | 17 | 24.9 | 0.01 | 15.0 | 0.04 |
| MGMT unmethylated | 9 | 10.1 | 8.1 |
NA, not applicable.
Prognosis according to perfusion MRI parameters
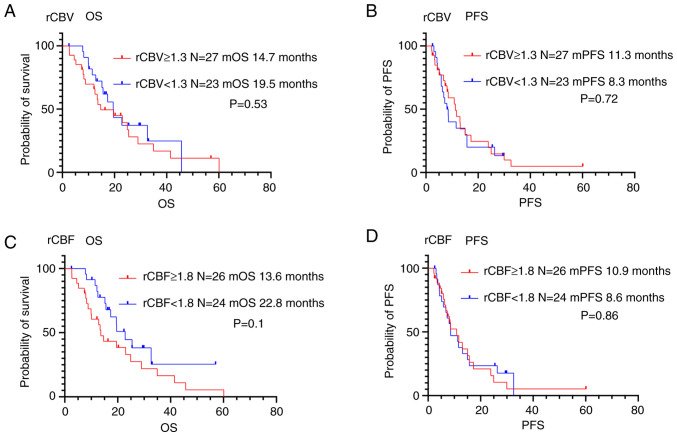
In contrast, there was no association between perfusion MRI parameters and OS or PFS in patients with glioblastoma (Fig. 5 and Table IV).
Figure 5.
Kaplan-Meier survival curves of the patients with glioblastoma grouped according to MR perfusion imaging parameters: (A) rCBV OS and (B) PFS (CBV ≥1.3, n=27; rCBV <1.3, n=27); (C) rCBF OS and (D) PFS (rCBF ≥1.8, n=26; rCBF <1.8, n=24). There was no association between each of the MR perfusion imaging parameters and not only OS but also PFS in patients with glioblastoma. OS, Overall survival; PFS, progression-free survival; MGMT, O6-methylguanine DNA methyltransferase; rCBV, cerebral blood volume ratio; rCBF, cerebral blood flow ratio.
Prognosis according to MGMT methylation status and perfusion MRI parameters
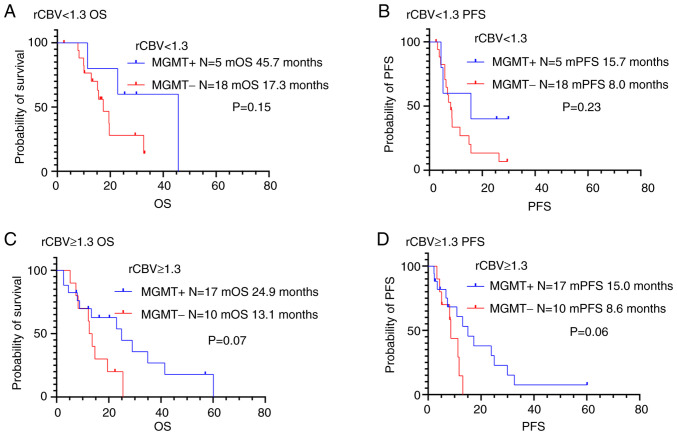
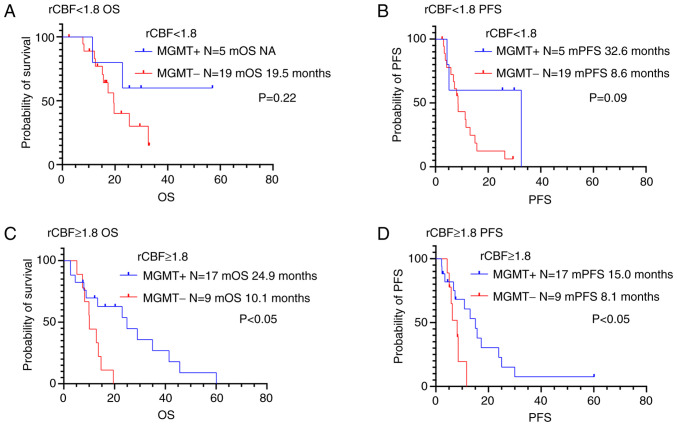
The study investigated the significance in PFS and OS differences between the following two groups: low vascularity tumors with MGMT methylation and low vascularity tumors without MGMT methylation (Figs. 6, 7 and Table IV). Juan-Albarracín et al reported that significant differences were observed in the Kaplan-Meier estimated survival functions for populations divided based on the median rCBV and rCBF (29). They indicated that the median rCBV and rCBF were found to be the relevant prognostic markers in patients with glioblastoma. Previous studies assessed the combined role of tumor vascularity, estimated from perfusion MRI, and MGMT methylation status on OS in patients with glioblastoma (17,23). The classification of tumor vascularity was based on the median rCBV and rCBF values reported by Juan-Albarracín' et al (17,23). We validated thresholds calculated from the current study cohort based on previous reports (17,23,29) and defined the vascular groups using the median rCBV and rCBF. There was no significant association between MGMT methylation status and prognosis in patients with low vascularity tumors (rCBV <1.3 or rCBF <1.8). We also evaluated differences in PFS and OS in high vascularity tumors (rCBV ≥1.3 or rCBF ≥1.8) with methylated and unmethylated MGMT promoters. There was no association between MGMT methylation status and OS or PFS in patients with high rCBV (rCBV ≥1.3). On the other hand, high vascularity tumors (rCBF ≥1.8) with MGMT methylation were associated to longer OS and PFS compared to those without MGMT methylation (P<0.05).
Figure 6.
Kaplan-Meier survival curves of the glioblastoma patients grouped according to MGMT methylation status and tumor vascularity based on rCBV: (A) rCBV <1.3 with MGMT methylation (n=5); (B) rCBV <1.3 without MGMT methylation (n=18); (C) rCBV ≥1.3 with MGMT methylation (n=17); (D) rCBV ≥1.3 without MGMT methylation (n=10). There was no association between MGMT methylation status and prognosis in lower and higher rCBV. OS, Overall survival; PFS, progression-free survival; MGMT, O6-methylguanine DNA methyltransferase; rCBV, cerebral blood volume ratio; rCBF, cerebral blood flow ratio.
Figure 7.
Kaplan-Meier survival curves of the glioblastoma patients grouped according to MGMT methylation status and tumor vascularity based on rCBF: (A) rCBF <1.8 with MGMT methylation (n=5); (B) rCBF <1.8 without MGMT methylation (n=19); (C) rCBF ≥1.8 with MGMT methylation (n=17); (D) rCBF ≥1.8 without MGMT methylation (n=9). There was no association between MGMT methylation status, OS and PFS in lower rCBF. However, patients with MGMT methylation had longer OS and PFS compared with those without MGMT methylation in higher vascularized tumors (rCBF ≥1.8; P<0.05). OS, Overall survival; PFS, progression-free survival; MGMT, O6-methylguanine DNA methyltransferase; rCBV, cerebral blood volume ratio; rCBF, cerebral blood flow ratio.
Discussion
Our study indicates that CBV and CBF can be used to predict the MGMT methylation status in glioblastomas. According to our results, the rCBV and rCBF in tumors with MGMT methylation were higher than those in tumors without MGMT methylation. The possibility of predicting the MGMT methylation status from DSC-MRI using a radiological approach remains controversial. In previous reports, the CBV derived from DSC-MRI of glioblastomas with a methylated MGMT promoter were reported to be lower than those corresponding to glioblastomas with unmethylated MGMT (8,10,16). In contrast, other reports have indicated that CBV does not differ significantly between tumors with methylated and unmethylated MGMT (17,18). Using stereotactic image-based histological validation, Song et al reported that CBF showed no statistically significant differences between gliomas with and without MGMT promoter methylation (30). Perfusion parameters are influenced by the location of the tumor in relation to major blood vessels, heterogeneous vascularization of the tumor, tumor necrosis, and intratumoral cystic changes. The DSC-MR perfusion technique is known to be affected by the partial volume effect caused by adjacent tissues. Contouring ROI, excluding necrosis and proximate vascular structures, reduces the partial volume effect caused by adjacent tissues. As mentioned in previous reports (16,25,26), ROIs were drawn to avoid calcification, blood products, dense bone, or large vessels to ensure the accuracy of the measurements. The size of the ROI for the solid part in our study was smaller than that in previous studies (16,25,26). As glioblastomas are heterogeneous tumors, the ROIs in our study were accurately set on the solid part, which contained only enhancing tumor core lesions in each tumor region (Fig. 1) to exclude the effect of tumor heterogeneity. Therefore, rCBV and rCBF may be affected by the definition of the size of ROI. There is a possibility that the prediction of MGMT methylation status could be heavily affected by the method used for ROI design. However, it is still unclear whether rCBV and rCBF were affected by the small ROI or the MGMT methylation. Although it is desirable to perform a regression analysis to clarify whether ROI or MGMT methylation factors were corrected, this makes it very difficult to perform the mentioned analysis, since multiple ROIs or ROIs of different sizes were not set throughout the course of our study.
Meanwhile, Hegi et al have suggested that the methylation status of the MGMT promoter may have prognostic value and, additionally, may be a clinically relevant predictor of the benefit of TMZ chemotherapy (1). HIF-1 was discovered as a molecular target associated with intratumoral hypoxia (31). As previously demonstrated, HIF-1α silencing dramatically increases sensitivity to TMZ in vivo (32). Tang et al showed that the inhibition of HIF-1α through knock-down sensitizes glioma cells to TMZ, with a decrease in MGMT expression (33). Persano er al. showed that HIF-1α suppression promotes the downregulation of MGMT, and this is sufficient to override glioblastoma resistance to TMZ (34). In the present study, glioblastomas with MGMT promoter methylation showed higher rCBV and rCBF than those without. Glioblastomas with maintained perfusion and oxygenation levels may have suppressed HIF-1α expression and downregulated MGMT expression, and may be susceptible to TMZ treatment.
In contrast, whether MRI perfusion parameters correlate with the prognosis of glioblastoma remains controversial. Previous studies have shown that CBV (19–22,35) and CBF (36) have prognostic value. However, no significant association between overall survival time and CBV has been reported in previous studies (37,38). The prognostic correlation between CBV and MGMT methylation status may be influenced by conditions such as tumor vascularity and treatment-induced changes over time. Previous studies have shown a highly significant impact of MGMT status on the prognosis of patients with moderately vascularized tumors, but not in patients with highly vascularized tumors (17,23). Goldman et al reported that treatment-induced changes in CBV affect the prognosis of glioblastoma (24). They reported that glioblastomas that showed stable or increasing CBV following chemoradiotherapy were associated to a significantly improved PFS compared to those with decreased CBV following chemoradiotherapy, particularly in those exhibiting MGMT methylation (24). Batchelor et al found that patients with glioblastoma treated with chemoradiotherapy plus cediranib demonstrated an increase in perfusion and significantly improved survival compared to patients treated with chemoradiotherapy alone. This effect may be due to anti-angiogenic therapy, normalization of blood flow, and enhancement of drug delivery (39). It has been assumed that the methylation of the MGMT promoter induced by the maintained CBV and improved oxygenation enhanced the therapeutic benefits of alkylating agents. In our study, highly vascularized tumors based on rCBF with MGMT methylation were associated to longer OS and PFS than those without MGMT methylation. High CBF tumors may be less hypoxic, leading to MGMT promoter methylation, and improved prognosis with TMZ treatment. The failure to observe a significant difference in OS with and without MGMT methylation can be attributed to the small sample size, which reduced the power (40,41). This can be seen from the P-value of 0.06, which is very close to the significance level. Conversely, even with such a low detection power, a significant difference in OS can be confirmed between patients with and without MGMT methylation who have more highly vascular tumors (rCBF ≥1.8), which may suggest the idea that MGMT methylation status has a stronger effect on OS in cases with higher rCBF. Radiological diagnosis using rCBV and rCBF has the potential to predict MGMT methylation status preoperatively, without reliance on surgical specimens. In our study, there was no association between perfusion MRI parameters and OS or PFS in patients with glioblastoma. Furthermore, there was no significant association between MGMT methylation status and prognosis in patients with lower vascularity tumors based on both the rCBV and the rCBF and those with more highly vascularized tumors based on rCBV. The measurement of cerebral blood perfusion in DSC-MRI is based on the assumption that gadolinium-based contrast agents do not cross the blood-brain barrier. CBV is calculated by the tissue signal change caused by the gadolinium-based contrast agent and the arterial input function. Based on the assumption that the gadolinium-based contrast agents do not cross the blood-brain barrier, the CBV changes caused by the gadolinium-based contrast agents are thought to be due to the gadolinium-based contrast agent stored in the capillaries. However, this assumption does not hold in glioblastoma tumor tissues where the blood-brain barrier has been disrupted. The value of the CBV calculated by the model described above is ambiguous (42). Conversely, CBF is calculated by dividing CBV by MTT (CBF=CBV/MTT), where MTT is the time taken for the tracer to pass through the region of interest. Thus, CBF compensates somewhat for the blood-brain barrier breakdown. It is possible that calculated values with such technical ‘corrections’ more sensitively reflect tumor characteristics. It is not certain that the combination of MRI perfusion parameters with MGMT methylation status can be used to predict the prognosis of glioblastomas. We are skeptical that the combination of perfusion MRI parameters with MGMT methylation status can be used to predict the prognosis of glioblastomas.
A few limitations and caveats in the current study should be noted and addressed. As previously mentioned, this study was limited by its small sample size, leading to potential bias in our results. First, as the ROIs in our study were accurately set on the solid part in each enhancing tumor region to exclude the effect of tumor heterogeneity while avoiding areas of cyst formation, hemorrhage and large vessels, the size of the ROIs was smaller than that in previous studies (16,25,26). Therefore, there is a possibility that the prediction for MGMT methylation status may have been heavily affected by the method used for ROI design. Second, while the methylation status of the MGMT promoter may have prognostic value, there was no significant difference in the OS of patients with and without MGMT methylation in our study. Future large-scale studies are required to validate the proposed prognostic value of CBF and MGMT methylation status.
In conclusion, we aimed to evaluate whether DSC-MRI could be employed as a non-invasive method to predict MGMT methylation status and prognosis in newly diagnosed glioblastoma patients. Our study indicates that rCBV and rCBF can be used to predict the MGMT methylation status preoperatively, offering the possibility to change clinical management in patients affected by glioblastoma. However, we are not certain that the combination of MRI perfusion parameters with MGMT methylation status can be used to predict prognosis in these patients.
Acknowledgements
Not applicable.
Funding Statement
This work was supported by Japan Science and Technology Agency (grant no. JPMJPF2009).
Availability of data and materials
The data generated in the present study are not publicly available due to them containing information that could compromise research participant privacy/consent but may be requested from the corresponding author.
Authors' contributions
YO conceived and designed the study. DC, YO, RU, HiK, RH, NoK, NaK, YK and HaK acquired the data. DC, YO and SY analyzed and interpreted the data and drafted the manuscript. AA and NT contributed to the methodology for radiological analysis. DC and YO confirmed the authenticity of all the raw data. All authors provided critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of Osaka University Hospital (approval no. 22302). Written informed consent was obtained from all patients.
Patient consent for publication
Patients provided written informed consent for publication of their data.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Hegi ME, Diserens AC, Gorlia T, Hamou MF, de Tribolet N, Weller M, Kros JM, Hainfellner JA, Mason W, Mariani L, et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med. 2005;352:997–1003. doi: 10.1056/NEJMoa043331. [DOI] [PubMed] [Google Scholar]
- 2.Ahn SS, Shin NY, Chang JH, Kim SH, Kim EH, Kim DW, Lee SK. Prediction of methylguanine methyltransferase promoter methylation in glioblastoma using dynamic contrast-enhanced magnetic resonance and diffusion tensor imaging. J Neurosurg. 2014;121:367–373. doi: 10.3171/2014.5.JNS132279. [DOI] [PubMed] [Google Scholar]
- 3.Doniselli FM, Pascuzzo R, Agro M, Aquino D, Anghileri E, Farinotti M, Pollo B, Paterra R, Cuccarini V, Moscatelli M, et al. Development of A Radiomic Model for MGMT Promoter Methylation Detection in Glioblastoma Using Conventional MRI. Int J Mol Sci. 2023;25:138. doi: 10.3390/ijms25010138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Drabycz S, Roldan G, de Robles P, Adler D, McIntyre JB, Magliocco AM, Cairncross JG, Mitchell JR. An analysis of image texture, tumor location, and MGMT promoter methylation in glioblastoma using magnetic resonance imaging. Neuroimage. 2010;49:1398–1405. doi: 10.1016/j.neuroimage.2009.09.049. [DOI] [PubMed] [Google Scholar]
- 5.Han Y, Yan LF, Wang XB, Sun YZ, Zhang X, Liu ZC, Nan HY, Hu YC, Yang Y, Zhang J, et al. Structural and advanced imaging in predicting MGMT promoter methylation of primary glioblastoma: A region of interest based analysis. BMC Cancer. 2018;18:215. doi: 10.1186/s12885-018-4114-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kanas VG, Zacharaki EI, Thomas GA, Zinn PO, Megalooikonomou V, Colen RR. Learning MRI-based classification models for MGMT methylation status prediction in glioblastoma. Comput Methods Programs Biomed. 2017;140:249–257. doi: 10.1016/j.cmpb.2016.12.018. [DOI] [PubMed] [Google Scholar]
- 7.Sanada T, Kinoshita M, Sasaki T, Yamamoto S, Fujikawa S, Fukuyama S, Hayashi N, Fukai J, Okita Y, Nonaka M, et al. Prediction of MGMT promotor methylation status in glioblastoma by Contrast-enhanced T1-weighted intensity image. Neurooncol Adv. 2024;6:vdae016. doi: 10.1093/noajnl/vdae016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ozturk K, Soylu E, Cayci Z. Correlation between dynamic susceptibility contrast perfusion MRI and genomic alterations in glioblastoma. Neuroradiology. 2021;63:1801–1810. doi: 10.1007/s00234-021-02674-2. [DOI] [PubMed] [Google Scholar]
- 9.Paech D, Windschuh J, Oberhollenzer J, Dreher C, Sahm F, Meissner JE, Goerke S, Schuenke P, Zaiss M, Regnery S, et al. Assessing the predictability of IDH mutation and MGMT methylation status in glioma patients using relaxation-compensated multipool CEST MRI at 7.0 T. Neuro Oncol. 2018;20:1661–1671. doi: 10.1093/neuonc/noy073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ryoo I, Choi SH, Kim JH, Sohn CH, Kim SC, Shin HS, Yeom JA, Jung SC, Lee AL, Yun TJ, et al. Cerebral blood volume calculated by dynamic susceptibility contrast-enhanced perfusion MR imaging: Preliminary correlation study with glioblastoma genetic profiles. PLoS One. 2013;8:e71704. doi: 10.1371/journal.pone.0071704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shen N, Zhang S, Cho J, Li S, Zhang J, Xie Y, Wang Y, Zhu W. Application of cluster analysis of time evolution for magnetic resonance Imaging-derived oxygen extraction fraction mapping: A Promising strategy for the genetic profile prediction and grading of glioma. Front Neurosci. 2021;15:736891. doi: 10.3389/fnins.2021.736891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Do DT, Yang MR, Lam LHT, Le NQK, Wu YW. Improving MGMT methylation status prediction of glioblastoma through optimizing radiomics features using genetic algorithm-based machine learning approach. Sci Rep. 2022;12:13412. doi: 10.1038/s41598-022-17707-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Li ZC, Bai H, Sun Q, Li Q, Liu L, Zou Y, Chen Y, Liang C, Zheng H. Multiregional radiomics features from multiparametric MRI for prediction of MGMT methylation status in glioblastoma multiforme: A multicentre study. Eur Radiol. 2018;28:3640–3650. doi: 10.1007/s00330-017-5302-1. [DOI] [PubMed] [Google Scholar]
- 14.Sasaki T, Kinoshita M, Fujita K, Fukai J, Hayashi N, Uematsu Y, Okita Y, Nonaka M, Moriuchi S, Uda T, et al. Radiomics and MGMT promoter methylation for prognostication of newly diagnosed glioblastoma. Sci Rep. 2019;9:14435. doi: 10.1038/s41598-019-50849-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Xi YB, Guo F, Xu ZL, Li C, Wei W, Tian P, Liu TT, Liu L, Chen G, Ye J, et al. Radiomics signature: A potential biomarker for the prediction of MGMT promoter methylation in glioblastoma. J Magn Reson Imaging. 2018;47:1380–1387. doi: 10.1002/jmri.25860. [DOI] [PubMed] [Google Scholar]
- 16.Lu J, Li X, Li H. Perfusion parameters derived from MRI for preoperative prediction of IDH mutation and MGMT promoter methylation status in glioblastomas. Magn Reson Imaging. 2021;83:189–195. doi: 10.1016/j.mri.2021.09.005. [DOI] [PubMed] [Google Scholar]
- 17.Fuster-Garcia E, Lorente Estelles D, Alvarez-Torres MDM, Juan-Albarracin J, Chelebian E, Rovira A, Acosta CA, Pineda J, Oleaga L, Molla-Olmos E, et al. MGMT methylation may benefit overall survival in patients with moderately vascularized glioblastomas. Eur Radiol. 2021;31:1738–1747. doi: 10.1007/s00330-020-07297-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Moon WJ, Choi JW, Roh HG, Lim SD, Koh YC. Imaging parameters of high grade gliomas in relation to the MGMT promoter methylation status: The CT, diffusion tensor imaging, and perfusion MR imaging. Neuroradiology. 2012;54:555–563. doi: 10.1007/s00234-011-0947-y. [DOI] [PubMed] [Google Scholar]
- 19.Bonekamp D, Deike K, Wiestler B, Wick W, Bendszus M, Radbruch A, Heiland S. Association of overall survival in patients with newly diagnosed glioblastoma with contrast-enhanced perfusion MRI: Comparison of intraindividually matched T1-and T2 (*)-based bolus techniques. J Magn Reson Imaging. 2015;42:87–96. doi: 10.1002/jmri.24756. [DOI] [PubMed] [Google Scholar]
- 20.Hirai T, Murakami R, Nakamura H, Kitajima M, Fukuoka H, Sasao A, Akter M, Hayashida Y, Toya R, Oya N, et al. Prognostic value of perfusion MR imaging of high-grade astrocytomas: Long-term Follow-up study. AJNR Am J Neuroradiol. 2008;29:1505–1510. doi: 10.3174/ajnr.A1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jain R, Poisson L, Narang J, Gutman D, Scarpace L, Hwang SN, Holder C, Wintermark M, Colen RR, Kirby J, et al. Genomic mapping and survival prediction in glioblastoma: Molecular subclassification strengthened by hemodynamic imaging biomarkers. Radiology. 2013;267:212–220. doi: 10.1148/radiol.12120846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Law M, Young RJ, Babb JS, Peccerelli N, Chheang S, Gruber ML, Miller DC, Golfinos JG, Zagzag D, Johnson G. Gliomas: Predicting time to progression or survival with cerebral blood volume measurements at dynamic Susceptibility-weighted Contrast-enhanced perfusion MR imaging. Radiology. 2008;247:490–498. doi: 10.1148/radiol.2472070898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Alvarez-Torres MDM, Fuster-Garcia E, Balana C, Puig J, Garcia-Gomez JM. Lack of benefit of extending temozolomide treatment in patients with high vascular glioblastoma with methylated MGMT. Cancers (Basel) 2021;13 doi: 10.3390/cancers13215420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Goldman J, Hagiwara A, Yao J, Raymond C, Ong C, Bakhti R, Kwon E, Farhat M, Torres C, Erickson LG, et al. Paradoxical association between relative cerebral blood volume dynamics following chemoradiation and increased Progression-free survival in newly diagnosed IDH Wild-Type MGMT promoter methylated glioblastoma with measurable disease. Front Oncol. 2022;12:849993. doi: 10.3389/fonc.2022.849993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shen N, Zhao L, Jiang J, Jiang R, Su C, Zhang S, Tang X, Zhu W. Intravoxel incoherent motion Diffusion-weighted imaging analysis of diffusion and microperfusion in grading gliomas and comparison with arterial spin labeling for evaluation of tumor perfusion. J Magn Reson Imaging. 2016;44:620–632. doi: 10.1002/jmri.25191. [DOI] [PubMed] [Google Scholar]
- 26.Wang X, Chen XZ, Shi L, Dai JP. Glioma grading and IDH1 mutational status: Assessment by intravoxel incoherent motion MRI. Clin Radiol. 2019;74:651.e7–651.e14. doi: 10.1016/j.crad.2019.03.020. [DOI] [PubMed] [Google Scholar]
- 27.Okita Y, Nonaka M, Shofuda T, Kanematsu D, Yoshioka E, Kodama Y, Mano M, Nakajima S, Kanemura Y. (11)C-methinine uptake correlates with MGMT promoter methylation in nonenhancing gliomas. Clin Neurol Neurosurg. 2014;125:212–216. doi: 10.1016/j.clineuro.2014.08.004. [DOI] [PubMed] [Google Scholar]
- 28.Sasaki T, Fukai J, Kodama Y, Hirose T, Okita Y, Moriuchi S, Nonaka M, Tsuyuguchi N, Terakawa Y, Uda T, et al. Characteristics and outcomes of elderly patients with diffuse gliomas: A Multi-institutional cohort study by Kansai Molecular Diagnosis Network for CNS tumors. J Neurooncol. 2018;140:329–339. doi: 10.1007/s11060-018-2957-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Juan-Albarracin J, Fuster-Garcia E, Perez-Girbes A, Aparici-Robles F, Alberich-Bayarri A, Revert-Ventura A, Marti-Bonmati L, Garcia-Gomez JM. Glioblastoma: Vascular habitats detected at preoperative dynamic susceptibility-weighted Contrast-enhanced perfusion MR imaging predict survival. Radiology. 2018;287:944–954. doi: 10.1148/radiol.2017170845. [DOI] [PubMed] [Google Scholar]
- 30.Song S, Shan Y, Wang L, Cheng Y, Yang H, Zhao G, Wang Z, Lu J. MGMT promoter methylation status shows no effect on [(18)F]FET uptake and CBF in gliomas: A stereotactic Image-based histological validation study. Eur Radiol. 2022;32:5577–5587. doi: 10.1007/s00330-022-08606-9. [DOI] [PubMed] [Google Scholar]
- 31.Onnis B, Rapisarda A, Melillo G. Development of HIF-1 inhibitors for cancer therapy. J Cell Mol Med. 2009;13:2780–2786. doi: 10.1111/j.1582-4934.2009.00876.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li L, Lin X, Shoemaker AR, Albert DH, Fesik SW, Shen Y. Hypoxia-inducible Factor-1 inhibition in combination with temozolomide treatment exhibits robust antitumor efficacy in vivo. Clin Cancer Res. 2006;12:4747–4754. doi: 10.1158/1078-0432.CCR-05-2842. [DOI] [PubMed] [Google Scholar]
- 33.Tang JH, Ma ZX, Huang GH, Xu QF, Xiang Y, Li N, Sidlauskas K, Zhang EE, Lv SQ. Downregulation of HIF-1a sensitizes U251 glioma cells to the temozolomide (TMZ) treatment. Exp Cell Res. 2016;343:148–158. doi: 10.1016/j.yexcr.2016.04.011. [DOI] [PubMed] [Google Scholar]
- 34.Persano L, Pistollato F, Rampazzo E, Della Puppa A, Abbadi S, Frasson C, Volpin F, Indraccolo S, Scienza R, Basso G. BMP2 sensitizes glioblastoma Stem-like cells to Temozolomide by affecting HIF-1α stability and MGMT expression. Cell Death Dis. 2012;3:e412. doi: 10.1038/cddis.2012.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Burth S, Kickingereder P, Eidel O, Tichy D, Bonekamp D, Weberling L, Wick A, Low S, Hertenstein A, Nowosielski M, et al. Clinical parameters outweigh Diffusion- and Perfusion-derived MRI parameters in predicting survival in newly diagnosed glioblastoma. Neuro Oncol. 2016;18:1673–1679. doi: 10.1093/neuonc/now122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gerstner ER, Zhang Z, Fink JR, Muzi M, Hanna L, Greco E, Prah M, Schmainda KM, Mintz A, Kostakoglu L, et al. ACRIN 6684: Assessment of tumor hypoxia in newly diagnosed glioblastoma using 18F-FMISO PET and MRI. Clin Cancer Res. 2016;22:5079–5086. doi: 10.1158/1078-0432.CCR-15-2529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Paik W, Kim HS, Choi CG, Kim SJ. Pre-operative perfusion skewness and kurtosis are potential predictors of Progression-free survival after partial resection of newly diagnosed glioblastoma. Korean J Radiol. 2016;17:117–126. doi: 10.3348/kjr.2016.17.1.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.White ML, Zhang Y, Kazmi SAJ, Aizenberg M, Shonka N, Yu F, Appiah AK. Evaluating survival in subjects with astrocytic brain tumors by dynamic Susceptibility-weighted perfusion MR imaging. PLoS One. 2021;16:e0244275. doi: 10.1371/journal.pone.0244275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Batchelor TT, Gerstner ER, Emblem KE, Duda DG, Kalpathy-Cramer J, Snuderl M, Ancukiewicz M, Polaskova P, Pinho MC, Jennings D, et al. Improved tumor oxygenation and survival in glioblastoma patients who show increased blood perfusion after cediranib and chemoradiation. Proc Natl Acad Sci USA. 2013;110:19059–19064. doi: 10.1073/pnas.1318022110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Willan AR, Pinto EM. The value of information and optimal clinical trial design. Stat Med. 2005;24:1791–1806. doi: 10.1002/sim.2069. [DOI] [PubMed] [Google Scholar]
- 41.Zhang L, Cui L, Yang B. Optimal flexible sample size design with robust power. Stat Med. 2016;35:3385–3396. doi: 10.1002/sim.6931. [DOI] [PubMed] [Google Scholar]
- 42.Nagahama H, Shonai T, Takashima H, Hirano T, Suzuki J, Sakurai Y. MRI of Perfusion: Principles and Clinical Applications. Igaku Butsuri. 2016;36:103–109. doi: 10.11323/jjmp.36.2_103. (In Japanese) [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data generated in the present study are not publicly available due to them containing information that could compromise research participant privacy/consent but may be requested from the corresponding author.