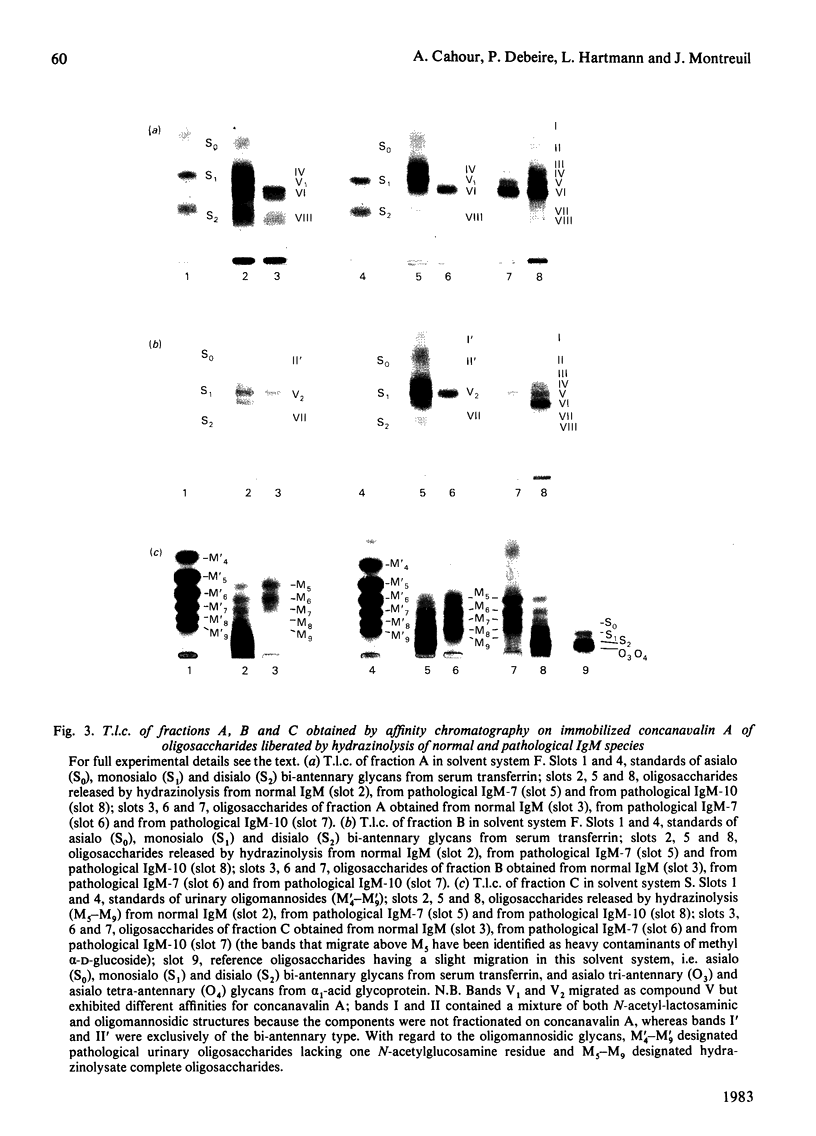
Abstract
The well-known heterogeneity of normal and pathological immunoglobulins M was investigated in a study involving the characterization of their carbohydrate moieties. Oligosaccharide units were released from the native molecule by hydrazinolysis, and they were fractionated by affinity chromatography on a concanavalin A-Sepharose column to yield separate N-acetyl-lactosaminic-type and oligomannosidic-type structures. Further identification of these oligosaccharides was attempted by t.l.c. on silica gel and by determination of their monosaccharide compositions. A comparative study of the oligosaccharide units belonging to each population of immunoglobulin M was possible. Similarities were found in the occurrence of both types of oligosaccharide structures, and, in addition, a common double heterogeneity could be demonstrated for N-acetyl-lactosaminic-type structures: they could be resolved by affinity chromatography into bi-, tri- and tetra-antennary structures, and they also showed differences in N-acetylneuraminic acid content. Though some variations were observed in the exact composition of the oligosaccharide units within each population, it was possible to consider a representative oligosaccharide-unit composition of normal immunoglobulin M as a standard for comparison. On this basis a predominance of multi-antennary structures was observed in the more glycosylated pathological immunoglobulins M (10% carbohydrate content), whereas oligomannosidic structures were increased in pathological immunoglobulins M with a lower content of carbohydrates (7%). These variations are thought to reflect differences in the biosynthetic processing pathway of the carbohydrate units of the pathological immunoglobulins M or the enhanced expression of a molecular clone.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chapman A., Kornfeld R. Structure of the high mannose oligosaccharides of a human IgM myeloma protein. I. The major oligosaccharides of the two high mannose glycopeptides. J Biol Chem. 1979 Feb 10;254(3):816–823. [PubMed] [Google Scholar]
- Chapman A., Kornfeld R. Structure of the high mannose oligosaccharides of a human IgM myeloma protein. II. The minor oligosaccharides of high mannose glycopeptide. J Biol Chem. 1979 Feb 10;254(3):824–828. [PubMed] [Google Scholar]
- Debray H., Montreuil J. Isolation and characterization of surface glycopeptides from adult rat hepatocytes in an established line. Biochimie. 1978;60(8):697–702. doi: 10.1016/s0300-9084(78)80014-5. [DOI] [PubMed] [Google Scholar]
- Fournet B., Montreuil J., Strecker G., Dorland L., Haverkamp J., Vliegenthart F. G., Binette J. P., Schmid K. Determination of the primary structures of 16 asialo-carbohydrate units derived from human plasma alpha 1-acid glycoprotein by 360-MHZ 1H NMR spectroscopy and permethylation analysis. Biochemistry. 1978 Nov 28;17(24):5206–5214. doi: 10.1021/bi00617a021. [DOI] [PubMed] [Google Scholar]
- Hickman S., Kornfeld R., Osterland C. K., Kornfeld S. The structure of the glycopeptides of a human M-immunoglobulin. J Biol Chem. 1972 Apr 10;247(7):2156–2163. [PubMed] [Google Scholar]
- Jouanneau J., Fournet B., Bourrillon R. Localization and overall structure of a mannose-rich glycopeptide from a pathologic immunoglobulin. Biochim Biophys Acta. 1981 Feb 27;667(2):277–284. doi: 10.1016/0005-2795(81)90193-8. [DOI] [PubMed] [Google Scholar]
- Jouanneau J., Razafimahaleo E., Bourrillon R. Glycopeptides des immunoglobulines. Microhétérgénéité des chaînes oligosaccharidiques d'une immunoglobuline M de Waldenström. Eur J Biochem. 1970 Nov;17(1):72–77. doi: 10.1111/j.1432-1033.1970.tb01136.x. [DOI] [PubMed] [Google Scholar]
- Kornfeld S., Li E., Tabas I. The synthesis of complex-type oligosaccharides. II. Characterization of the processing intermediates in the synthesis of the complex oligosaccharide units of the vesicular stomatitis virus G protein. J Biol Chem. 1978 Nov 10;253(21):7771–7778. [PubMed] [Google Scholar]
- Li E., Tabas I., Kornfeld S. The synthesis of complex-type oligosaccharides. I. Structure of the lipid-linked oligosaccharide precursor of the complex-type oligosaccharides of the vesicular stomatitis virus G protein. J Biol Chem. 1978 Nov 10;253(21):7762–7770. [PubMed] [Google Scholar]
- Narasimhan S., Wilson J. R., Martin E., Schachter H. A structural basis for four distinct elution profiles on concanavalin A--Sepharose affinity chromatography of glycopeptides. Can J Biochem. 1979 Jan;57(1):83–96. doi: 10.1139/o79-011. [DOI] [PubMed] [Google Scholar]
- Palo J., Savolainen H. Thin-layer chromatographic demonstration of aspartylglycosylamine and a novel acidic carbohydrate in human tissues. J Chromatogr. 1972 Feb 23;65(2):447–450. doi: 10.1016/s0021-9673(00)92570-6. [DOI] [PubMed] [Google Scholar]
- Parodi A. J., Leloir L. F. The role of lipid intermediates in the glycosylation of proteins in the eucaryotic cell. Biochim Biophys Acta. 1979 Apr 23;559(1):1–37. doi: 10.1016/0304-4157(79)90006-6. [DOI] [PubMed] [Google Scholar]
- Reading C. L., Penhoet E. E., Ballou C. E. Carbohydrate structure of vesicular stomatitis virus glycoprotein. J Biol Chem. 1978 Aug 25;253(16):5600–5612. [PubMed] [Google Scholar]
- Shimizu A., Putnam F. W., Paul C., Clamp J. R., Johnson I. Structure and role of the five glycopeptides of human IgM immunoglobulins. Nat New Biol. 1971 May 19;231(20):73–76. doi: 10.1038/newbio231073a0. [DOI] [PubMed] [Google Scholar]
- Spik G., Bayard B., Fournet B., Strecker G., Bouquelet S., Montreuil J. Studies on glycoconjugates. LXIV. Complete structure of two carbohydrate units of human serotransferrin. FEBS Lett. 1975 Feb 15;50(3):296–299. doi: 10.1016/0014-5793(75)80513-8. [DOI] [PubMed] [Google Scholar]
- Strecker G., Fournet B., Bouquelet S., Montreuil J., Dhondt J. L., Farriaux J. P. Etude chimique des 'mannosides urinaires excrétés au cours de la mannosidose. Biochimie. 1976;58(5):579–586. doi: 10.1016/s0300-9084(76)80227-1. [DOI] [PubMed] [Google Scholar]
- Tabas I., Kornfeld S. The synthesis of complex-type oligosaccharides. III. Identification of an alpha-D-mannosidase activity involved in a late stage of processing of complex-type oligosaccharides. J Biol Chem. 1978 Nov 10;253(21):7779–7786. [PubMed] [Google Scholar]
- Tabas I., Schlesinger S., Kornfeld S. Processing of high mannose oligosaccharides to form complex type oligosaccharides on the newly synthesized polypeptides of the vesicular stomatitis virus G protein and the IgG heavy chain. J Biol Chem. 1978 Feb 10;253(3):716–722. [PubMed] [Google Scholar]
- Takasaki S., Kobata A. Microdetermination of sugar composition by radioisotope labeling. Methods Enzymol. 1978;50:50–54. doi: 10.1016/0076-6879(78)50006-2. [DOI] [PubMed] [Google Scholar]
- Zanetta J. P., Breckenridge W. C., Vincendon G. Analysis of monosaccharides by gas-liquid chromatography of the O-methyl glycosides as trifluoroacetate derivatives. Application to glycoproteins and glycolipids. J Chromatogr. 1972 Jul 5;69(2):291–304. doi: 10.1016/s0021-9673(00)92897-8. [DOI] [PubMed] [Google Scholar]
- van Halbeek H., Dorland L., Vliegenthart J. F., Spik G., Cheron A., Mohtreuil J. Structure determination of two oligomannoside-type glycopeptides obtained from bovine lactotransferrin, by 500 MHz 1H-NMR spectroscopy. Biochim Biophys Acta. 1981 Jul;675(2):293–296. doi: 10.1016/0304-4165(81)90240-3. [DOI] [PubMed] [Google Scholar]