Abstract
Zelkova carpinifolia is a Tertiary relict tree distributed in Hyrcanian and Colchic forests. Most of its habitat has been destroyed in the last century. This study aimed to model potentially suitable habitat areas for Zelkova carpinifolia from the past to the future. The Last Glacial Maximum (LGM) and Future (2061–2080) models include 19 bioclimatic variables from the CCSM4 global circulation model Pearson correlation coefficient was used to assess collinearity between variables and ten variables were selected for distribution modelling. Habitat suitability was estimated using the Biodiversity Modelling (BIOMOD) ensemble modelling method by combining the results of ten algorithm models using the R package “biomod2”. The area under the curve (AUC) of the receiver operating characteristic (ROC) curve and true skills statistics (TSS) were calculated to evaluate the performance of the models. The contributions of the environmental variables were calculated separately for each algorithm model. According to the results obtained, the most effective bioclimatic variable in the distribution of the species is temperature seasonality (Bio4). The modelling results revealed that Zelkova carpinifolia survived in suitable refuge areas in western Asia during the LGM. These distribution areas have remained largely unchanged and even expanded. The future model results predict that the suitable habitats of the species will narrow in the Hyrcanian forests south of Caspian Sea and that more suitable conditions will be found around the Caucasus. Given the increasing destruction of these valuable plant species due to human activities and the expected negative impacts of climate change in the future, it is important to develop policies and strategies for the protection of Zelkova carpinifolia's habitat, the creation of nature reserves, and sustainability.
Keywords: BIOMOD, Ensemble approach, Habitat suitability, Climate change impact, Zelkova carpinifolia
Subject terms: Biodiversity, Biogeography, Ecological modelling
Introduction
Climate change is a significant factor affecting biodiversity. Changes in climate physiologically affect organisms and significantly affect their distribution and migration by causing changes in their habitats1–3. While the climate changes that occurred in the Quaternary had a significant impact on the shaping of today’s vegetation, traces of climate events in the Tertiary period are also evident. The high temperature and humid conditions during the Tertiary period effectively expanded, narrowed or eliminated plant areas4. Subsequently, in the Quaternary, especially with the decrease in temperature during the LGM, the distribution, evolution and diversity of plants, as in previous periods, changed5–7. Climate change, which continues due to the increase in greenhouse gas concentrations today, poses a threat to plants and affects the stability and function of ecosystems8–10. In this century, average global temperature is to increase, precipitation patterns to change, and extreme weather events to increase10–14. The effects and risks of climate change will become more complex as temperatures rise, making it more difficult to manage risks. Considering the studies conducted, changes in climate have irreversible consequences for biodiversity. As a result of climate change, species are expected to differin their habitat areas able to adapt to climatic conditions in a specific region and going out of their climatic niche15,16. Species distribution models (SDMs) are important techniques for understanding, conserving, and managing the effects of climate change on biodiversity and for making spatiotemporal predictions17,18. The number of studies conducted with SDMs has increased significantly in recent years. However, the presence of many modelling algorithms, which are increasing daily, makes it difficult to choose the most appropriate methodology19–23. The BIOMOD, which analyses species distributions using ensemble modelling, offers significant advantages in overcoming these problems by enabling the elimination of methodological uncertainties in models and the investigation of species-environment relationships24.
According to studies that have conducted habitat suitability analysis using ensemble modelling, this model produced meaningful results for Olea europaea, Berberis lycium, and Carijoa riisei1,9,25. However, there have been no studies examining the habitat suitability of Zelkova carpinifolia in the context of climate change. Nevertheless, despite absence of studies examining the habitat suitability of Zelkova carpinifolia in the context of climate change, several studies on species with similar ecological requirements in Hyrcanian forests and their surrounding areas. Models predicting the future distributions of plant species indicate that Hyrcanian Forest tree species will experience negative impacts from climate change in certain regions. However, these species are expected to migrate to higher latitudes, and their range will likely extend to higher elevations2,26–28. This study aimed to model the potentially suitable habitat areas of Zelkova carpinifolia from the LGM to the future using an ensemble modelling approach. Zelkova carpinifolia is a relict genus belonging to the Ulmaceae family. Zelkova carpinifolia, a Tertiary relict, is found on the northern slopes of the Alborz mountain ranges and along the southern coasts of the Caspian Sea, where Hyrcanian forests are located. Zelkova carpinifolia is an important element of the Hyrcanian forests. In addition to the Hyrcanian and Colchic forests, it has a limited distribution. A few isolated populations are also known in Türkiye (Kars, Hakkâri, Siirt, Muş). In Türkiye, it is still very likely places where Zelkova carpinifolia has not yet been discovered29–33. Hyrcanian forests are significant remnants of Arcto-Tertiary forests in western Eurasia34and are home to various thermophilous species26,35–38. Zelkova appears to have constituted a component of humid mesic forests during the Miocene and Pliocene. However, the decline of warm and moist climates in the Cenozoic during the Plio-Pleistocene led to the extinction of this genus in numerous regions39,40. The warm and humid environmental conditions that have prevailed in the distribution area of the species since the late Tertiary have provided a refuge for Zelkova during the ice ages27,31. Zelkova carpinifolia is a tree that prefers moist, humus-rich soils and requires little light. The species is mainly found at altitudes of 100–600 m above sea level, but it is also distributed at altitudes of 1200–1500 m in the Talysh Mountains and the highlands of Anatolia. Zelkova carpinifolia forms pure stands under optimal conditions in Hyrcanian forestsbut is generally found in oak and beech species. In this area, the average annual rainfall is over 1500 mm, and the July temperatures are 22–25 °C. In this area, the species is accompanied by broadleaf Tertiary relics such as Acer velutinum, Albizia julibrissin, Alnus subcordata, Diospyros lotus, Gleditsia caspica, Parrotia persica and Quercus castaneifolia30.
Zelkova carpinifolia is especially threatened by human activities in Hyrcanian forests, where it is widely distributed. The wood of Zelkova carpinifolia is valuable and resistant to moisture and is used in the construction sector and for door frames and windows of mosques in Iran41,42. The population of this species is estimated to have declined by approximately 30% in the past 210 years. The species is categorized as “vulnerable” (VU) according to the IUCN Red List criteria because of its irreversible destruction, and its population is declining43. Considering the limited distribution of Zelkova carpinifolia, the hypothesis of this study that climate change significantly reduces the suitability of the species habitat and causes shifts in distribution patterns, which may threaten the survival of the species. In this context, the aim of this study was to perform a habitat suitability analysis for Zelkova carpinifolia, an endangered relict species, under past, present and future climate conditions using the ensemble approach. The development of conservation strategies for Zelkova carpinfolia involves the use of predictive models based on distribution data. The use of these models can facilitate more evidence-based decision-making with respect to conservation priorities and habitat management, through the analysis of the current and future distributions of the species in question. Consequently, this enables the formulation of efficacious strategies to increase the sustainability of ecosystems and ensure the preservation of biodiversity.
Species occurrence data
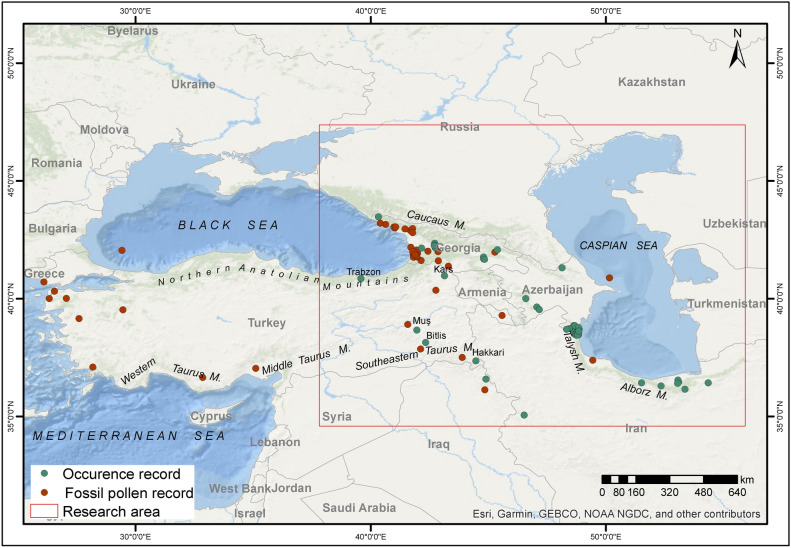
The natural occurrence data of Zelkova carpinifolia were obtained from the Global Biodiversity Information Facility (GBIF)44, and the relevant sections of the "Flora of Turkey and the East Aegean Islands"32. A total of 116 geographically referenced occurrence data points were obtained for Zelkova carpinifolia from the above mentioned sources. Geographically shifting occurrence data to user-friendly and easily accessible locations can result in bias in occurrence data44,45. In terms of the accuracy and reliability, it is important that the occurrence data of the species obtained are minimized in terms of sample bias and autocorrelation and are distributed homogeneously1. In this respect, the occurrence data were filtered to reduce the effects of sample bias and autocorrelation and to ensure their homogeneity. For this purpose, the "Spatially Rarefy Occurrence Data" tool in ArcGIS Pro SDMtoolbox Pro 0.9.1 was used to apply a 5 km2 spatial filter to the species occurrence data46. As a result of the analyses, 51 geographically referenced occurrence datasets were used as the final dataset to model the distribution of Zelkova carpinifolia (Fig. 1).
Fig. 1.
Pollen and occurrence data distribution map for Zelkova carpinifolia. The figure was generated using ArcGIS Pro URL: https://www.esri.com/en-us/arcgis/products/arcgis-pro/overview.
Species fossil data
The identification of palaeontological records belonging to Zelkova carpinifolia, a Tertiary relict, is important for determining the possible refuges of this species in the past. Therefore, 53 fossil pollen/leaf data for the species were obtained from the literature to verify and compare the past distribution models of the species (Fig. 1, Supplemental Table S1). Among the fossil data obtained, 4 are from the early Miocene, 7 from the Miocene, 2 from the middle Miocene, 1 from the late Tortonian, 2 from the late Messinian to the earliest Zanclean, 1 from the late Miocene to the present, 12 from the Pliocene, 22 from the Pleistocene, and 1 from the late Holocene.
Climate data
While different ecological factors affect the distribution of plants, temperature and precipitation are effective factors affecting species distributions when viewed on a large scale. Theoretically, all ecological processes are necessary for a detailed study in ecological niche modelling; however, potential niches and potential distributions can be estimated on climatic variables only47,48. As in many similar studies, the factors considered in species distribution modelling in this study are bioclimatic variables49–52. Within the scope of the study, the “climatic conditions” of the physical geography elements were examined from a geographical perspective and taken as independent variables. In this context, since climatic conditions are the most influential variables on vegetation distribution, bioclimatic variables were primarily considered in this study. In addition, since numerical (ecological, edaphic and topographic) data belonging to the LGM could not be modelled, they were not used in the study.
The prediction performance of species distributions is partially dependent on the selection and resolution of the input data, as well as the magnitude of climate change. In this study, the WorldClim database was used as the source of climate variables44. CCSM4 is widely acknowledged as one of the most effective climate models for simulating the impacts of past and future climate change on plant distributions53. This model has previously been successfully utilized in comparable studies54.
It is considered that there are multiple options available for consideration, including WorldClim 2.1, which represents climate data from 1971 to 2000 and incorporates 23 different global climate models55. It should be noted, however, that this version does not provide data for earlier periods. Furthermore, in light of the climate sensitivity observed in global climate simulations, choose the use of WorldClim 2.1 for current and future climate scenarios. Instead, we elected to employ WorldClim 1.4 for the analysis of past periods in this study. In conclusion, despite the older version of the software, we selected WorldClim 1.4 for use with all timeframes included in our research.
To determine the suitable habitat areas of the species, climate data with a spatial resolution of 2.5 min/ ~ 5 km covering the period 1960–1990 were obtained from the WorldClim 1.4 database; the CCSM4 model with a resolution of 2.5 min/ ~ 5 km was used to determine the suitable habitat areas of the Last Glacial Maximum (approximately 22,000 years ago); and the Community Climate System Model Version 4 (CCSM4) model with a resolution of 2.5 min/ ~ 5 km and Representative Concentration Pathway (RCP) 4.5 and 8.5 scenarios were used for climate projection data for 2061–2080 (https://worldclim.org/). This study includes the CCSM4 model for all periods. In the examination of studies covering similar periods on the subject, the CCSM4 model was found to yield better results; therefore, the CCSM4 model was used in this study3,56,57. The natural distribution data of the bioclimatic species used in the study were masked using ArcGIS Pro software, taking into account the natural distribution data of the species.
Understanding the relationships/interactions between species and the environmental variables surrounding them is important for predicting current and future species distributions. Environmental layers are applied as hypothetical prediction variables using raw data for species distribution model production, and spatial autocorrelation complications can be seen as the degree of dependence associated with the geographical distribution of a particular species58,59. Species distribution models obtained from a large dataset of associated environmental covariates often result in natural multicollinearity; this statistical problem is also defined as a high correlation between covariates in nonexperimental cases where the researcher has no control over the risk associated with hypothetical factors related to independent variables. Therefore, the Pearson correlation test was applied to determine the linear relationships among 19 bioclimatic variables and to eliminate the problem of multicollinearity, and a correlation matrix was created. As a result of the test, data that were highly correlatedwith each other were removed from the model, and bioclimatic data that did not show collinearity with each other with a Pearson correlation coefficient of less than 0.85 were included in the modelling60–66.
As a result of the correlation, 9 variables with a Pearson correlation coefficient of less than 0.85 were Bio2 (mean diurnal range), Bio3 (isothermality), Bio4 (temperature seasonality), Bio8 (mean temperature of wettest quarter), Bio9 (mean temperature of driest quarter), Bio12 (annual precipitation), Bio14 (precipitation of driest month), Bio15 (precipitation seasonality), and Bio19 (precipitation of coldest quarter). When bioclimatic variables, the ecological requirements of Zelkova carpinifolia, which has a humid light requirement, were also taken into account to reduce collinearity the environmental parameters, and it was concluded that the correlation analysis results were appropriate32. This analysis was conducted using the "Principal Component Analysis" tool in SDMtoolbox in ArcGIS Pro.
Ensemble modelling
In species distribution modelling, the ensemble modelling technique reduces the inconsistencies between the predictions of individual models by combining multiple models into a individual ensemble model25,67. The ensemble modelling technique is used in a wide variety of studies and has many important applications in biodiversity studies9,68–70.
The biomod2 package (version 4.2-5.2) was used in R software (version 4.2.2)71 for ensemble distribution modelling of Zelkova carpinifolia[24, 72]. Biomod2 facilitates the application of 10 important algorithms to model species-environment relationships. The ten models used in this study are Artificial Neural Network (ANN)73, classification Tree Analysis (CTA)74, Flexible Discriminant analysis (FDA)75, Generalized AdditiveModel (GAM)76, Generalized Boosted Model (GBM)77, Generalized Linear Model (GLM)78, Multiple Adaptive Regression Splines (MARS)79, Maximum Entropy (MAXENT)80, Random Forest (RF)81, and Surface Response Envelope (SRE)82.
In this study, individual models were first generated using the default biomod settings. Data on species occurrence, including both native and invasive records, were combined with globally generated pseudoabsence data, created randomly. This approach has previously yielded more effective results in predicting potential distributions83,84than real absence data, which can lead to underestimation problems. Each dataset included species occurrence records matched with an equal number of independently generated pseudoabsences. The models were created using 80% of the data (training data), and the performance of the model was evaluated with the remaining 20% (test data). In total, 1000 models were run, including ten different pseudoabsence selections and tenfold cross-validation (10 evaluations) for 10 algorithms85,86. The area under the curve (AUC) of the receiver operating characteristic (ROC) curve and true skills statistics (TSS) were used as evaluation criteria to estimate the accuracy of the models. [21, 87] The AUC defines the success of models with all threshold values. An AUC greater than 0.75 indicates that the model is performing well, and the closer it is to 1, the better and more sensitive the performance is88,89. The TSS takes into account both omission and commission errors and the success obtained as a result of random prediction. The TSS value varies between -1 and + 1, where + 1 indicates the perfect value of the dependent variable3,87. The sensitivity specificity method was used to convert the resulting continuous maps into binary maps that distinguished between unsuitable and suitable areas. This technique is among the most dependable for determining a reclassification threshold. In this modelling framework, sensitivity refers to the ratio of correctly predicted presences, which serves as a measure of omission errors, whereas specificity refers to the ratio of correctly predicted absences, which serves as a measure of commission errors88,90.
Ensemble models were developed for the LGM, current and future (RCP26-RCP4.5-RCP8.5/2070) time periods using the committee average (ca) and weighted mean (wm) approaches independently of the individual modelling outputs91. A permutation approach was used to evaluate the relative impact of climatic variables on the Zelkova carpinifolia models92. Variable importance was calculated using the biomod2 variable importance function. The relevance estimates of the climatic variables used in the models are obtained by dividing the difference between the original prediction and the permuted variable prediction by a negative correlation score (1-correlation score). As a result, a value close to 1 indicates that the variable’s contribution to the model is greater, while a value close to 0 indicates that the variable’s contribution to the model is less (41).
The model results obtained were transferred to ArcGIS Pro software using the reclassify and raster calculator tools to calculate the changes in suitability to visualize and measure the changes in the distribution of Zelkova carpinifolia under past and future climate conditions. When converting continuous predictions into a Boolean classification of " suitable " versus " unsuitable ", the selection of an appropriate threshold is essential. We determined the threshold by maximizing the sum of the training sensitivity and specificity, which optimized the (true skill score) TSS and allowed the generation of binary classifications. The logistic output value p ≥ P is called the appropriate region, whereas the logistic output value p < P is called the inappropriate region93–96. In this study, the suitable area is classified into three categories: low suitability (P ≤ p < 0.5), medium suitability (0.5 ≤ p < 0.7), and high suitability (p ≥ 0.7). Suitability change analysis provides information about the loss and gain of suitable habitats for species in the past and future. In this context, four classes were defined in the suitability change analysis: species loss, species absence, stable and gain, and maps were created9,97–100.
Results and discussion
Model evaluation
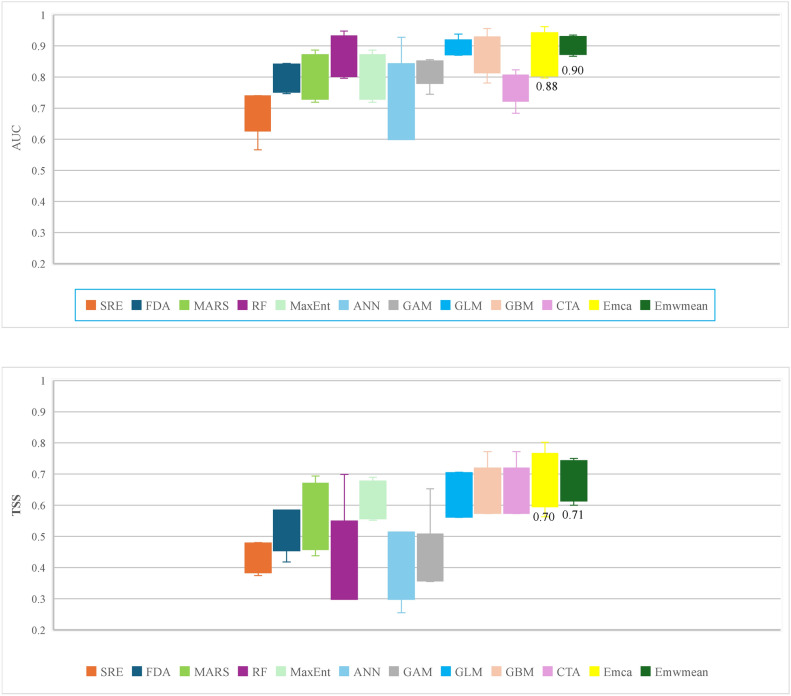
The accuracy of the produced ensemble models was 0.88 for AUC and 0.70 for TSS for committee averaging and 0.90 for AUC and 0.71 for TSS for the weighted mean. The AUC and TSS values indicate that the final models are good for predicting Zelkova carpinifolia. When the individual model accuracies are evaluated, GAM, GBM, MARS, and RF exhibit the best performances, followed by GLM, FDA, MaxEnt, CTA, and ANN. The SRE had the lowest performance (Fig. 2). The high AUC and TSS values for both ensemble models (committee averaging and weighted mean) indicate that they performed well in predicting suitable habitat areas for Zelkova carpinifolia. This suggests that the models effectively captured the relationships between environmental factors and the presence of the species.
Fig. 2.
The true skills statistics (TSS) and the area under the curve (AUC) of the receiver operating characteristic (ROC) curves indicate the model evaluation scores for individual and ensemble models.
Bioclimatic variable contributions
The contributions of the bioclimatic variables included in the model showed differences among the algorithms. (Table 1). When all the algorithms were evaluated together, the variables contributing the most to the 9 selected bioclimatic variables were Bio4 (0.46), Bio12 (0.29), Bio9 (0.29), Bio19 (0.26), and Bio8 (0.13). Considering that warm and humid environmental conditions in the Tertiary provided shelter for the species, it is likely that Bio4 (temperature seasonality), Bio12 (annual precipitation), and Bio9 (mean temperature of Driest Quarter) contributed more to the models.
Table 1.
Algorithmwise values of the bioclimatic variables used for Zelkova carpinifolia modelling (GLM: Generalized Linear Model, GBM: Generalized Boosted Model, GAM: Generalized Additive Model, CTA: Classification Tree Analysis, ANN: Artificial Neural Networks, SRE: Surface Range Envelope, FDA: Flexible Discriminant Analysis, RF: Random Forest, MARS: Multivariate Adaptive Regression Splines, MaxEnt: Maximum Entropy).
| Bioclimatic | Individual species distribution models | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| GLM | GBM | GAM | CTA | ANN | SRE | FDA | RF | MARS | MaxEnt | Mean | |
| Variables | |||||||||||
| Bio2 | 0.00 | 0.04 | 0.19 | 0.10 | 0.09 | 0.12 | 0.13 | 0.12 | 0.00 | 0.13 | 0.09 |
| Bio3 | 0.00 | 0.00 | 0.29 | 0.00 | 0.06 | 0.04 | 0.00 | 0.22 | 0.00 | 0.07 | 0.07 |
| Bio4 | 0.48 | 0.26 | 0.78 | 0.90 | 0.48 | 0.26 | 0.14 | 0.41 | 0.61 | 0.28 | 0.46 |
| Bio8 | 0.00 | 0.13 | 0.20 | 0.00 | 0.18 | 0.34 | 0.00 | 0.16 | 0.12 | 0.13 | 0.13 |
| Bio9 | 0.36 | 0.30 | 0.64 | 0.00 | 0.05 | 0.46 | 0.00 | 0.12 | 0.57 | 0.35 | 0.29 |
| Bio12 | 0.00 | 0.07 | 0.46 | 0.00 | 0.98 | 0.34 | 0.62 | 0.43 | 0.02 | 0.00 | 0.29 |
| Bio14 | 0.05 | 0.03 | 0.31 | 0.00 | 0.18 | 0.09 | 0.18 | 0.21 | 0.00 | 0.02 | 0.11 |
| Bio15 | 0.11 | 0.05 | 0.42 | 0.00 | 0.04 | 0.23 | 0.00 | 0.06 | 0.00 | 0.09 | 0.10 |
| Bio19 | 0.23 | 0.05 | 0.58 | 0.00 | 0.28 | 0.18 | 0.29 | 0.20 | 0.23 | 0.55 | 0.26 |
Zelkova carpinifolia developed in warm and humid environments during the Tertiary period4,101. The most important bioclimatic variables were identified by the models. A lower temperature seasonality (less variation) might be more favourable for Zelkova carpinifolia, potentially aligning with its adaptation to warmer and more moderate climates of the Tertiary. Higher annual and cold-quarter precipitation could be indicative of the humid conditions preferred by the species. A lower mean diurnal range (less extreme daily temperature fluctuations) might be more suitable for Zelkova carpinifolia, again aligning with its potential adaptation to milder climates. Warmer temperatures during the driest period could be favourable for Zelkova carpinifolia, reflecting its possible preference for warm and potentially drought-tolerant conditions39. The model results suggest that temperature seasonality, annual precipitation, precipitation of the coldest quarter, and temperature of the driest quarter are the most important bioclimatic factors influencing the current distribution of Zelkova carpinifolia. These findings likely reflect the species’ adaptation to warm and humid environments.
Fossil occurrences of Zelkova carpinifolia
Zelkova carpinifolia, a member of the Ulmaceae family, is a deciduous tree that has been significant in palaeobotanical studies beacuse of its distinctive leaves and wood structure. Zelkova carpinifolia is native to the Hyrcanian region of Azerbaijan and Iran, as well as the Colchis area of western Georgia. It also grows in the southern Lesser Caucasus (Garabagh, Azerbaijan), eastern Georgia (Babaneuri Strict Nature Reserve), the Zagros Mountains of Iran, and the eastern part of Türkiye36,102–104. In the Caucasus region, the two main population centers for Zelkova carpinifolia are the Hyrcanian forest in southern Azerbaijan and northern Iran. The second is the Colchic forest in western Georgia. Isolated stands of Zelkova carpinifolia can also be found in the Karabakh Mountains, Anatolia, northwestern and eastern Georgia, and Iran105. Fossils of Zelkova species have been discovered across different geological periods. The oldest leaf fossils of Zelkova found in China date back to the Eocene epoch. During the Miocene, Zelkova zelkovifolia fossils were widespread in Europe and Central Asia. Pliocene fossils from Japan resemble extant species such as Zelkova carpinifolia and Zelkova schneideriana. Notably, Zelkova ungeriis a fossil species found in Central Italy during the late Pleistocene106. Fossilized remains of Zelkova carpinifolia have been discovered in various regions of Georgia, particularly in sedimentary rock formations that date back to the Miocene epoch, which lasted from approximately 23–5.3 million years. These fossils are often found in lignite deposits and ancient lake beds, where conditions favour the preservation of plant material. One study evaluated the genetic diversity of Zelkova carpinifolia across the Caucasus and adjacent areas, including Iran28. In this study, long-distance pollen dispersal likely contributed to gene flow among populations of Zelkova carpinifolia. Anatolia serves as a modern refugia for Mediterranean flora from the Neogene and early Pleistocene periods107. Several mesophilic Eurasian plant species, such as Liquidambar, Parrotia, Pterocarya, Zelkova, and the microthermal tree Cedrus, have survived through the European Neogene and still exist in limited areas of the Anatolian landscape and the Hyrcanian zone108. Zelkova carpinifoliais currently found in only two very restricted riparian locations in easternmost Anatolia near Lake Van, although it remains well developed in Abkhazia, the Small Caucasus, and predominantly in the Hyrcanian region109. Additionally, this genus persists in very limited areas of Crete (Zelkova abelicea) and Sicily (Zelkova sicula)110.
The current distribution of ecosystems containing P. fraxinifolia, various Zelkova species (Z. abelicea, Z. carpinifolia, Z. sicula), L. orientalis, and P. persicain the central and eastern Mediterranean regions and the Middle East suggests the potential for these species to re-expanded in Europe. This re-expansion could occur within the context of ongoing climate warming, possibly facilitated by reintroduction efforts to counteract the anticipated increase in aridity over the Mediterranean region111.
LGM, current and potential distributions
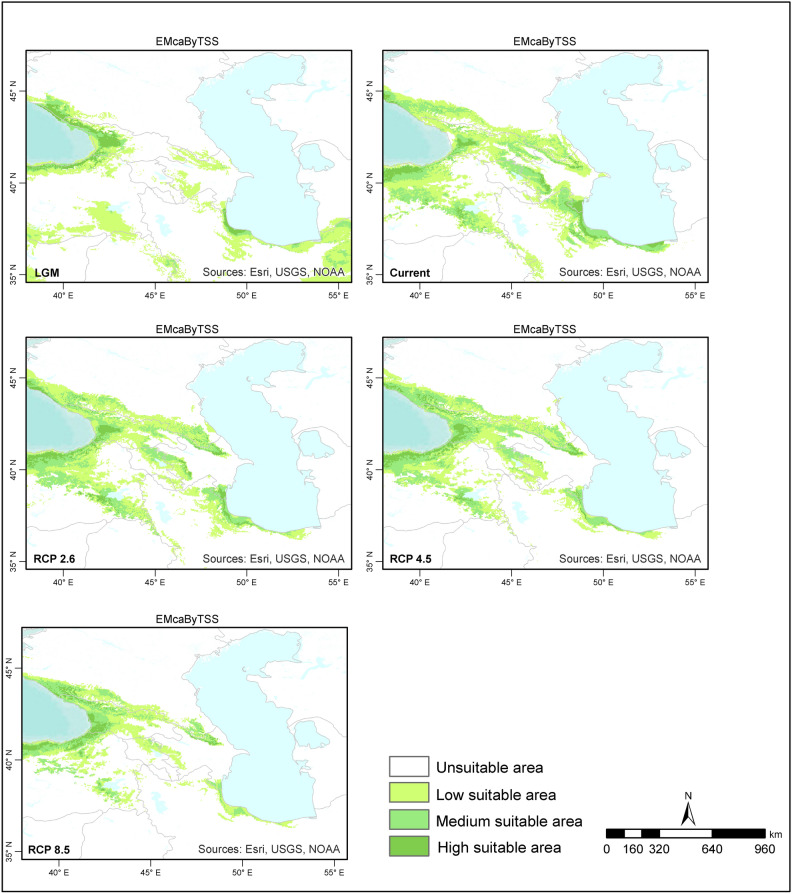
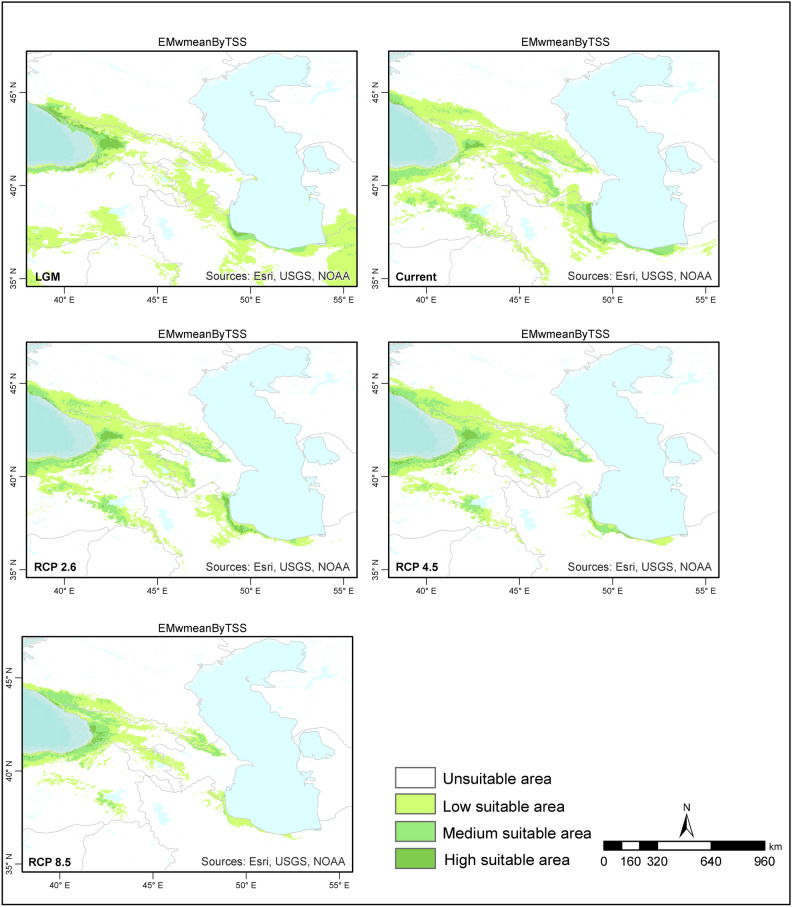
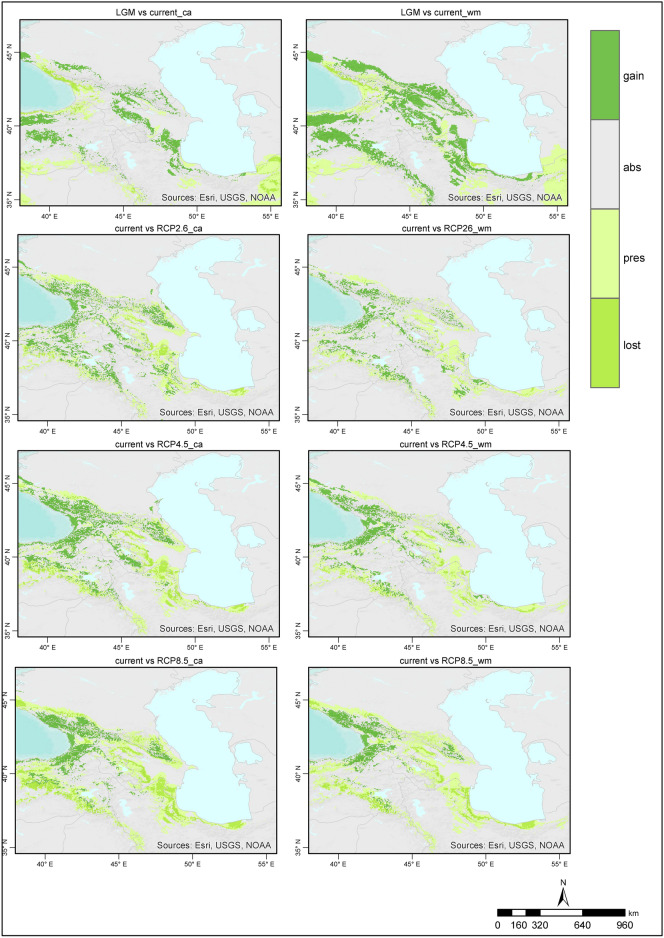
When the ensemble models for the LGM were examined, the species distribution areas covered narrower regions than those in the present day, and the species were distributed at lower altitudes. The majority of suitable habitat areas for Zelkova carpinifolia are located in Colchic forest stands located at lower elevations in Caucasus Mountains and Kaçkar Mountains.Narrower areas of suitable habitat are found Hyracian forests south‒southwest of the Caspian Sea, Elbrus Mountains and Talysh Mountains (Figs. 3–4). When the ensemble models produced according to the committee averaging and weighted mean approaches for the present time period are examined, the habitat areas generally show a similar distributions. However, according to the weighted mean approach, suitable habitat areas for Zelkova carpinifolia are more limited in the Hyrcanian forests in northern Iran and southern Azerbaijan, as well as the Colchic forests of Georgia. When the distribution areas of the species in the western Caspian Sea humid forests were compared with the model results, the model produced according to the weighted mean approach was parallel to the existing distribution of the species. When the two approaches are evaluated together, the species finds suitable habitat areas in Türkiye, Azerbaijan, Iran, Georgia, and Armenia, where it has a natural distribution in Hyracian and Colchic forests. Suitable habitat areas, including the Elbrus Mountains, Talysh Mountains, Caucasus Mountains, Kaçkar Mountains’s lower elevations, and southeastern Türkiye’s humid forest. Zelkova carpinifolia is generally distributed at 100 to 700 m around Hyrcanian forests in the southern Caspian Sea, whereas it is found at relativley altitudes in the Talysh Mountains and Anatolia humid forests. (Figs. 3–4).
Fig. 3.
Predicted habitat suitability for Zelkova carpinifolia under the LGM and future conditions based on the committee average. The figure was generated using R version 4.2.271 and ArcGIS Pro URL: https://www.esri.com/en-us/arcgis/products/arcgis-pro/overview.
Fig. 4.
Predicted habitat suitability for Zelkova carpinifolia under the LGM and future conditions based on the weighted mean. The figure was generated using R version 4.2.271 and ArcGIS Pro URL: https://www.esri.com/en-us/arcgis/products/arcgis-pro/overview.
Compared with those in the present day, the models predict that the distribution of Zelkova carpinifolia during the LGM was more geographically restricted. The species likely occurred at lower elevations. Hyracian forest south of Casipan Sea and Colchic forests in Türkiye and Georgia provided suitable habitat during the colder LGM period.
The current distribution patterns of the models are generally similar to those observed during the LGM, with suitable habitat areas concentrated in the Hyrcanian forests in northern Iran and southern Azerbaijan, as well as the Colchic forests of Georgia and Türkiye. However, the model based on the weighted mean approach predicts a more limited area of suitable habitat in the western Caspian Sea region, specifically in Azerbaijan. This appears to be a more accurate reflection of the actual distribution of Zelkova carpinifolia. The models indicate that suitable habitat for Zelkova carpinifolia is naturally present in Azerbaijan, Iran, and Hyrcanian forests. Additionally, it is found in Türkiye (in the Kaçkar Mountains humid forest on the lower slopes) and in Georgia (in the Kutaisi and Sataplia Nature Reserves, the Akhmeta area, and the Babaneuri Nature Reserve)39. The model results also suggest that the present-day altitude distribution of Zelkova carpinifolia varies depending on its location. For example, around the Caspian Sea, Hyrcanian forests, the species is generally found at lower altitudes (100–700 m). In the Talysh Mountains and Anatolia, the species is found humid forests at higher altitudes than in the Caspian Sea region. These model results provide valuable insights into the response of Zelkova carpinifolia to past and present climatic conditions. The narrower distribution and lower altitude preference during the LGM suggest that the species likely retreated to areas with milder conditions during the colder period. The similar distribution patterns between the LGM and present day, with some adjustments for specific regions, suggest a potential return to suitable pre-LGM habitats as the climate warmed. The variation in altitude distribution across regions might be related to specific microclimates or adaptations within the species. The ensemble models provide a good understanding of the historical and current distribution patterns of Zelkova carpinifolia. This information can be valuable for conservation efforts by identifying areas where the species is likely to occur and prioritizing protection strategies.
According to the future ensemble model results, Zelkova carpinifolia will not lose its current suitable habitat areas in terms of the area it covers. However, shifts in current distribution areas are expected. The species is expected to narrow its possible distribution areas around the Caspian Sea in the south and increase its suitable distribution areas around the Caucasus Mountains in Hyrcanian and Colchic forests. The potential for change in the current suitable habitat areas of the species was lower under the RCP2.6 and RCP4.5 scenarios than under the RCP8.5 scenario. (Figs. 3–4).
The models predict that Zelkova carpinifolia will not experience a complete loss of its current suitable habitat area in terms of total area. However, the models predict shifts in the current distribution areas of Zelkova carpinifolia. This means that the species might have lost suitable habitat in some areas, particularly around the southern Caspian Sea. However, it provides suitable habitat in other areas, particularly around the northern Caucasus Mountains humid forest.
The RCP 2.6 and RCP 4.5 scenarios represent lower levels of greenhouse gas emissions and are predicted to have a lower impact on Zelkova carpinifolia's distribution than RCP8.5. This scenario represents a higher level of greenhouse gas emissions and is predicted to have a more significant impact on the species distribution. These model results suggest that Zelkova carpinifolia might be able to adapt to some degree of climate change, but its distribution is likely affected. The predicted northwards shift in suitable habitat suggests that Zelkova carpinifolia might experience more favourable climatic conditions as it moves northwards due to a warming climate. Identifying areas where the species might lose suitable habitat allows proactive conservation measures to be taken in those regions. Considering the impacts of different climate change scenarios allows flexible and adaptable conservation planning.
To estimate the possible habitat areas of Zelkova carpinifolia in the Quaternary addressing the Tertiary conditions that were effective in determining the current distribution area. Zelkova carpinifolia responded to climate changes in the Tertiary. It is thought that the Hyrcanian and Colchic forests, which include Zelkova carpinifolia, emerged in the Upper Pliocene during the upheavals (formation) of the Caucasus Mountain ranges in the Miocene-Pliocene period28,112,113. Zelkova carpinifolia, which was distributed in the Hyrcanian and Colchic regions during the interglacial period of the Tertiary, was able to continue to exist in these refugia during the LGM. However, during the Holocene, climate changes affected the distribution of Zelkova carpinifolia in Georgia and Iran26,114,115. The Hyrcanian and Colchic forests are now completely separated from each other and consist of different plant communities with a few common relict plant species116.
The ensemble models produced according to the committee averaging and weighted mean approaches for the LGM time period showed similar suitable habitat areas for the species. The model results for the LGM are consistent with the fossil pollen data for the LGM. This indicates that there is a equilibrium relationship between climate and species among all the ecological factors that affect species distribution117.
The relatively slow change since the Last Glacial Maximum can be associated with a significant narrowing of the distribution area of species affected by climate change during the Tertiary. During the Tertiary, the distribution of the species decreased, and the species survived in suitable refugia in western Asia during the LGM. During the Holocene, climate changes affected the distribution of Zelkova carpinifolia in Georgia and Iran26,114,115. The Hyrcanian and Colchic forests are now completely separated from each other and consist of different plant communities with a few common relict plant species116. As a result, the suitable refugia of the species in the LGM have remained unchanged and even expanded their habitat areas. Today, Zelkova carpinifolia is generally found in lowland areas and on the slopes of mountains that are not too high. This situation is effective in species with a fragmented distribution even in suitable habitat areas, which are more exposed to anthropogenic activities such as logging for timber, grazing, agriculture, use as fuel, and opening up settlement areas27,115,34,35.
According to the results of future models, it is predicted that the suitable habitat areas of Zelkova carpinifolia will narrow around the Caspian Sea and its surroundings and that more suitable conditions will be found around the Caucasus. These results are consistent with studies conducted for species with similar climate requirements in our study area. For example, climate change is predicted to have a positive effecton the suitable habitat areas of Acer mazandaranicum, Populus caspica, and Taxus baccata, which are distributed in Hyrcanian forests119,34,35,119. Again, shifts are expected in the future distribution areas of the relict Gleditsia caspica, which is distributed in Hyrcanian forests120.
When studies of species with similar ecological requirements to those of Zelkova carpinifolia have been conducted, it is predicted that Zelkova schneideriana will lose some of its suitable habitat areas in the future, whereas shifts in the distribution area of Zelkova serrata are expected121–123.
Species suitability changes
According to the species suitability change analysis, the gains and losses of the species in stable areas in the past and future were analysed both spatially and proportionally (Table 2, Fig. 5).
Table 2.
Statistical analysis of the changes in the suitability of Zelkova carpinifolia.
| Scenario | Ensemble Type |
Loss (km2) |
Loss % |
Absent (km2) |
Absent (%) |
Stable (km2) |
Stable (%) |
Gain (km2) |
Gain (%) |
|---|---|---|---|---|---|---|---|---|---|
|
LGM Current |
Committee averaging |
157,322.4 | 8.0 | 1,494,887.0 | 76.3 | 25,648.5 | 1.3 | 281,916.4 | 14.4 |
|
Current-RCP2.6 2070 |
Committee averaging |
117,678.5 | 6.0 | 1,703,189.8 | 87.5 | 22,912.1 | 1.2 | 103,181.6 | 5.3 |
|
Current-RCP4.5 2070 |
Committee averaging |
139,989.4 | 7.2 | 1,665,532.3 | 85.5 | 39,547.2 | 2.0 | 101,910.6 | 5.2 |
|
Current-RCP8.5 2070 |
Committee averaging |
185,703.2 | 9.5 | 1,611,732.4 | 82.8 | 76,474.5 | 3.9 | 73,058.9 | 3.8 |
|
LGM Current |
Weighted mean |
222,430.9 | 11.3 | 1,519,185.8 | 77.5 | 17,136.2 | 0.9 | 201,603.1 | 10.3 |
|
Current-RCP2.6 2070 |
Weighted mean |
122,563.7 | 6.3 | 1,750,199.5 | 90.1 | 6293.1 | 0.3 | 63,075.3 | 3.2 |
|
Current-RCP4.5 2070 |
Weighted mean |
143,156.0 | 7.4 | 1,707,859.1 | 87.9 | 17,601.5 | 0.9 | 73,477.5 | 3.8 |
|
Current-RCP8.5 2070 |
Weighted mean |
186,805.4 | 9.6 | 1,653,622.6 | 85.1 | 48,981.4 | 2.5 | 52,697.9 | 2.7 |
Fig. 5.
Changes in the predicted suitability of LGM-current and current-future climatic conditions for Zelkova carpinifolia based on the committee average and weighted mean. The figure was generated using R version 4.2.271 and ArcGIS Pro URL: https://www.esri.com/en-us/arcgis/products/arcgis-pro/overview.
The results indicated that it was determined the suitable distribution areas of the species changed depending on the climatic conditions. According to the ca method, the loss rates in the LGM to current period are 8.0%, absent rates 76.3%, stable areas are 1.3%, gain areas are 14.4%. Under the current to future period (RCP 2.6) loss areas are 6.0%, absent areas are 87.5%, stable areas are 1.2%, gain areas are 5.3%. Under the current to future period (RCP 8.5), loss rates are 9.5%, absent areas are 82.7%, stable areas are 3.9%, gain areas are 3.8%. According to the wm method, loss areas in the LGM to current period are 11.3%, absent areas are 77.5%, stable areas are 0.9%, gain areas are 10.3%. According to the wm method under the current to future period (RCP 2.6) loss areas are 6.3%, absent areas are 90.1%, stable areas are 03%, gain areas are 2.0%. Under the current to future period (RCP 8.5), loss areas are 9.6%, absent areas are 85.1%, stable areas are 2.5%, and gain areas are 2.7% (Table 2). According to both ensemble types, suitable habitat areas are expected to increase from the LGM to the present day and from the present day to the future. However, the gain rates of suitable habitat areas are decreasing and tend to decrease from the past to the future. According to precipitation trend analyses over the last few decades, the amount of annual total precipitation has increased, and there has been a decreasing trend in consecutive dry days (CDD) and an increasing trend in consecutive wet days (CWD). Changes in precipitation and temperature trends may stress Zelkova carpinifolia and restrict its spread in stable areas in the future under current climatic conditions.
Conclusions
Climate change poses a greater threat to endangered species with limited distributions, especially in mountainous areas. Zelkova carpinifolia is a relict species distributed in mountainous areas in Western Asia. Owing to climate change and anthropogenic impacts, Zelkova carpinifolia is listed as vulnerable on the IUCN Red List of Threatened Species. In the context of climate change, planning or implementing conservation measures for Zelkova carpinifolia first requires predicting the species’s past, present, and future geographical distributions. Habitat suitability models are important tools for guiding conservation actions by analysing the relationships between environmental factors and the presence or abundance of species. These predictions provide valuable information about the distribution of species and increase the effectiveness of conservation strategies.
In this study, the potentially suitable habitat areas of Zelkova carpinifolia from the late Quaternary to the present day and the potential future distributions of the species in response to climate change were modelled using an ensemble modelling approach under different climate scenarios. The species distribution predictions generated by combining the advantages of different models and methods increased the accuracy of the models and made the results more reliable. The wm and ca methods were used in the ensemble modelling approach, and the results were generally found to be similar. This indicates that the projection results are consistent. The species finds suitable distribution areas under temperate conditions in terms of climate requirements. However, it is predicted that some of the currently suitable habitats will not be suitable in the future due to climate change. The model results revealed that the distribution of the species was strongly affected by temperature seasonality (Bio4). Climate change can affect the distribution of Zelkova carpinifolia, a vulnerable species. Therefore, there is a need for a sustainable ecosystem that protects the current areas where Zelkova carpinifolia is distributed and provides ecological conditions suitable for this species. This sustainable ecosystem should ensure the continuation of biodiversity, protect the current distribution areas of Zelkova carpinifolia and provide suitable ecological conditions.
Additional information
Correspondence and requests for materials should be addressed to D.E.K.
Supplementary Information
Below is the link to the electronic supplementary material.
Author contributions
Derya Evrim Koç, Beyza Ustaoğlu, Demet Biltekin contributed with the collection, organization and analysis of the data and wrote the manuscript draft.
Data availability
All the data generated/ analyzed during the study (i.e., the environmental variables and occurrence data), are available with the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-024-78733-4.
References
- 1.Khan, S. & Verma, S. (2022). Ensemble modelling to predict the impact of future climate change on the global distribution of Olea europaea subsp. cuspidata. Frontiers in Forests and Global Change. 5, 977691; 10.3389/ffgc.2022.977691.
- 2.Koc, D. E., Svenning, J. C. & Avcı, M. Climate change impacts on the potential distribution of Taxus baccata L. in the Eastern Mediterranean and the Bolkar Mountains (Turkey) from Last Glacial Maximum to the future. Eurasian Journal of Forest Science. 6(3), 69–82 (2018).
- 3.Koç, D. E., Biltekin, D. & Ustaoğlu, B. Modelling potential distribution of Carpinus betulus in Anatolia and its surroundings from the Last Glacial Maximum to the future. Arab. J. Geosci.14(12), 1186. 10.1007/s12517-021-07444-1 (2021). [Google Scholar]
- 4.Millar, C. I. Tertiary Vegetation History. Sierra Nevada Ecosystem Project: Final report to Congress, vol. II, Assessments and scientific basis for management options. University of (California, Centers for Water and Wildland Resources, 71–122, 1996).
- 5.Biltekin, D. et al. Vegetation and climate record from Abric Romaní (Capellades, northeast Iberia) during the Upper Pleistocene (MIS 5d–3). Quatern. Sci. Rev.220, 154–164 (2019). [Google Scholar]
- 6.Hewitt, G. M. The genetic legacy of the Quaternary ice ages. Nature. 405(6789), 907–913; 10. 1038/35016000 (2000). [DOI] [PubMed]
- 7.Svenning, J.C. & Skov, F. Ice age legacies in the geographic distribution of tree species richness in Europe. Global Ecology and Biogeography. 16, 234–245; doi. org/10. 1111/j. 1466- 8238. 2006. 00280. x (2007).
- 8.Scholze, M., Knorr, W., Arnell, N. W. & Prentice, I. C. A climate-change risk analysis for world ecosystems. Proc. Natl. Acad. Sci.103(35), 13116–13120 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Anjum, N., Ridwan, Q., Akhter, F. & Hanief, M. Predicting current and future distribution pattern of Berberis lycium Royle concerning climate change using an ensemble modelling approach. Geology, Ecology, and Landscapes. 1–10; 10.1080/24749508.2023.2287293 (2023).
- 10.Lee, H. et al. IPCC, 2023: Climate Change 2023: Synthesis Report, Summary for Policymakers. Contribution of Working Groups I, II and III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change (ed. H. Lee & J. Romero) IPCC, (Geneva, Switzerland, 2023).
- 11.Zhou, T. New physical science behind climate change: What does IPCC AR6 tell us?. The Innovation.2(4), 100173. 10.1016/j.xinn.2021.100173 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hama, A. A. & Khwarahm, N. R. Predictive mapping of two endemic oak tree species under climate change scenarios in a semiarid region: range overlap and implications for conservation. Ecological Informatics. 73, 101930; 10.1016/j.ecoinf.2022.101930 (2023).
- 13.Bahşi, K., Ustaoğlu, B., Aksoy, S. & Sertel, E. Estimation of emissions from crop residue burning in Türkiye using remotely sensed data and the Google Earth Engine platform. Geocarto International. 38(1), 2157052; 10.1080/10106049.2022.2157052 (2023).
- 14.Ustaoğlu, B., Tunçat, K. A. & Koç, D. E. Impacts of Climate Change on Precipitation and Temperature Climatology in Türkiye from Present to Future Perspective in Urban Commons, Future Smart Cities and Sustainability (ed. Chatterjee, U., Bandyopadhyay, N., Setiawati, M. D., Sarkar, S. 403–426 (Springer International Publishing, 2023).
- 15.Lovejoy, T. E. & Hannah, L. Climate Change and Biodiversity (Teri Press, 2006). [Google Scholar]
- 16.Bellard, C., Bertelsmeier, C., Leadley, P., Thuiller, W. & Courchamp, F. Impacts of climate change on the future of biodiversity. Ecol. Lett.15(4), 365–377. 10.1111/j.1461-0248.2011.01736.x (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Srivastava, V., Lafond, V. & Griess, V.C. Species distribution models (SDM): applications, benefits and challenges in invasive species management. CABI Reviews. 1–13; 10.1079/PAVSNNR201914020 (2019).
- 18.Sarkar, D., Jagannivsan, H., Debnath, A. & Talukdar, G. A systematic review on the potential impact of future climate change on India’s biodiversity using species distribution model (SDM) studies: trends, and data gaps. Biodiversity and Conservation. 1–17 (2024).
- 19.Elith, J. et al. Novel methods improve prediction of species’ distributions from occurrence data. Ecography29, 129–151 (2006). [Google Scholar]
- 20.Heikkinen, R. K. et al. Methods and uncertainties in bioclimatic envelope modelling under climate change. Prog. Phys. Geogr.30(6), 751–777 (2006). [Google Scholar]
- 21.Guisan, A. & Zimmermann, N. E. Predictive habitat distribution models in ecology. Ecological Modelling. 135(2–3), 147–186; 10. 1016/S0304 - 3800(00) 00354 – 9 (2000).
- 22.Sequeira, A. M., Bouchet, P. J., Yates, K. L., Mengersen, K. & Caley, M. J. Transferring biodiversity models for conservation: Opportunities and challenges. Methods Ecol. Evol.9(5), 1250–1264 (2018). [Google Scholar]
- 23.Araújo, M. B., et al. Standards for distribution models in biodiversity assessments. Science Advances. 5, eaat4858; 10.1126/sciadv.aat4858 (2019). [DOI] [PMC free article] [PubMed]
- 24.Thuiller, W., Cade, B., Engler, R. & Araújo, M. B. BIOMOD a platform for ensemble forecasting of species distributions. Ecography32, 369–373 (2009). [Google Scholar]
- 25.Mohamed Nisin, K. M. N., Sreenath, K. R. & Sreeram, M. P. Change in habitat suitability of the invasive Snowflake coral (Carijoa riisei) during climate change: an ensemble modelling approach. Ecological Informatics. 76, 102145; 10.1016/j.ecoinf.2023.102145 (2023).
- 26.Alavi, S. J., Ahmadi, K., Hosseini, S. M., Tabari, M. & Nouri, Z. The response of English yew (Taxus baccata L.) to climate change in the Caspian Hyrcanian mixed forest ecoregion. Regional Environmental Change. 19, 1495–1506 (2019).
- 27.Taleshi, H. et al. Climate change impacts on the distribution and diversity of major tree species in the temperate forests of Northern Iran. Reg. Environ. Change19(8), 2711–2728 (2019). [Google Scholar]
- 28.Ahmadi, K., et al. The potential impact of future climate on the distribution of European yew (Taxus baccata L.) in the Hyrcanian Forest region (Iran). International Journal of Biometeorology. 64, 1451–1462 (2020). [DOI] [PubMed]
- 29.Kvavadze, E. V. & Connor, S. E. Zelkova carpinifolia (Pallas) K. Koch in Holocene sediments of Georgia—an indicator of climatic optima. Review of Palaeobotany and Palynology. 133(1–2), 69–89 (2005).
- 30.Kozlowski G. & Gratzfeld J. Zelkova – An Ancient Tree. Global Status and Conservation Action. (Natural History Museum Fribourg, Switzerland 2013).
- 31.Maharramova, E. H., Safarov, H. M., Kozlowski, G., Borsch, T. & Muller, L. A. Analysis of nuclear microsatellites reveals limited differentiation between Colchic and Hyrcanian populations of the wind-pollinated relict tree Zelkova carpinifolia (Ulmaceae). Am. J. Bot.102(1), 119–128 (2015). [DOI] [PubMed] [Google Scholar]
- 32.Geven, F. & Adigüzel, N. Zelkova carpinifolia (Pall.) C. Koch (Ulmaceae) in Turkey (relict tree): floristics, ecology, distribution and threats. In International Forestry Symposium. 147–154 (2016).
- 33.Uzun, A. & Galalaey, A. M. K. Zelkova carpinifolia (Ulmaceae), a relict tree, first recorded in Iraq. Folia Geobot.57(4), 303–317 (2022). [Google Scholar]
- 34.Scharnweber, T., Rietschel, M. & Manthey, M. Degradation stages of the Hyrcanian forests in southern Azerbaijan. Archiv für Naturschutz und Landschaftsforschung.46(2), 133–156 (2007). [Google Scholar]
- 35.Alipour, S. et al. Predicting past, current and future suitable habitat for endemic Hyrcanian species Populus caspica Bornm. New Forest.54(2), 325–342 (2023). [Google Scholar]
- 36.Ahmadi, A., Kavosi, M. R. & Soltanloo, H. Zelkova carpinifolia reservoir from Hyrcanian Forests, Northern Iran, a new sacrifice of Ophiostoma novo-ulmi. Biodiversitas. Journal of Biological Diversity, 15(1), (2014).
- 37.Haidarian, A. M,. Tamartash, R., Jafarian, Z. & Tarkesh, E. M., Tatian, M. R. Predicting the impacts of climate change on Persian oak (Quercus brantii) using species distribution modelling in central Zagros for conservation planning. Iran J Env Stu.43, 497–511 (2017).
- 38.Sagheb Talebi, K., Sajedi, T. & Pourhashemi, M. Forests of Iran: A Treasure from the Past, a Hope for the Future) (Springer, 2014). [Google Scholar]
- 39.Denk, T., Frotzler, N. & Davitashvili, N. Vegetational patterns and distribution of relict taxa in humid temperate forests and wetlands of Georgia (Transcaucasia). Biol. J. Lin. Soc.72(2), 287–332 (2001). [Google Scholar]
- 40.Wang, Y. F., Ferguson, D. K., Zetter, R., Denk, T. & Garfı` G Leaf architecture and epidermal characters in Zelkova, Ulmaceae. Bot J Linn Soc. 136, 255–265 (2001).
- 41.Bétrisey, S., Yousefzadeh, H. & Kozlowski, G. Zelkova carpinifolia. The IUCN Red List of Threatened Species. e.T31303A131166773; 10.2305/IUCN.UK.2018- 2.RLTS.T31303A131166773.en (2018).
- 42.Browicz, K. & Zielińsk, J. Zelkova Spach., Flora of Turkey and the East Aegean Islands Vol.7 (ed. Davis P., Coode M. & Cullen J.) 648–649 (Edinburgh: Edinburgh University Press.1982).
- 43.Global Biodiversity Information Facility, GBIF Occurrence Download 10.15468/dl.q4n46a (18 January 2024).
- 44.Hijmans, R. J., Cameron, S. E., Parra, J. L., Jones, P. G. & Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Clim. 25, 1965–1978; 10.1002/joc.1276 (2005).
- 45.Ahmad, R., et al. Global distribution modelling, invasion risk assessment and niche dynamics of Leucanthemum vulgare (Ox-eye Daisy) under climate change. Sci. Rep.9, 11395; 10.1038/s41598-019-47859-1 (2019). [DOI] [PMC free article] [PubMed]
- 46.Brown, J. L., Bennett, J. R. & French, C. M. SDMtoolbox 2.0: the next generation Python-based GIS toolkit for landscape genetic, biogeographic and species distribution model analyses. PeerJ. 5, e4095; 10.7717/peerj.4095 (2017). [DOI] [PMC free article] [PubMed]
- 47.Drew, C. A. & Perera, A. H. Expert Knowledge As a Basis for Landscape Ecological Predictive Models in Predictive Species and Habitat Modeling in Landscape Ecology: Concepts and Applications 229-248 (New York, Springer. 2010).
- 48.Bobrowski, M., Gerlitz, L. & Schickhoff, U. Modelling the Potential Distribution of Betula Utilis in the Himalaya. Global Ecology and Conservation.11, 69–83 (2017). [Google Scholar]
- 49.Akyol, A., Örücü, Ö. K. & Arslan, E. S. Habitat suitability mapping of stone pine (Pinus pinea L.) under the effects of climate change. Biologia. 75, 2175–2187 (2020).
- 50.Özdemir, S., Gülsoy, S. & Mert, A. Predicting the effect of climate change on the potential distribution of Crimean Juniper. Kastamonu University Journal of Forestry Faculty.20(2), 133–142 (2020). [Google Scholar]
- 51.Mirhashemi, H., et al. Species distribution models of Brant’s oak (Quercus brantii Lindl.): The impact of spatial database on predicting the impacts of climate change. Ecological Engineering. 194, 107038; 10.1016/j.ecoleng.2023.107038 (2023).
- 52.Piwowarczyk, R. & Kolanowska, M. Effect of global warming on the potential distribution of a holoparasitic plant (Phelypaea tournefortii): both climate and host distribution matter. Scientific Reports. 13(1), 10741; /10.1038/s41598-023-37897-1 (2023). [DOI] [PMC free article] [PubMed]
- 53.Abdelaal, M., Fois, M., Fenu, G. & Bacchetta, G. Using MaxEnt modeling to predict the potential distribution of the endemic plant Rosa arabica Crép. Egypt. Ecological informatics50, 68–75 (2019). [Google Scholar]
- 54.Li, J., Fan, G. & He, Y. Predicting the current and future distribution of three Coptis herbs in China under climate change conditions, using the MaxEnt model and chemical analysis. Sci. Total Environ.698, 134141 (2020). [DOI] [PubMed] [Google Scholar]
- 55.Fick, S. E. & Hijmans, R. J. WorldClim 2: New 1-km spatial resolution climate surfaces for global land areas. International Journal of Climatology, 37(12), 4302–4315 https:// doi. org/ 10. 1002/ joc. 5086 (2017).
- 56.Duan, X., Li, J. & Wu, S. Maxent modelling to estimate the impact of climate factors on distribution of Pinus densiflora. Forests.13(3), 402. 10.3390/f13030402 (2022). [Google Scholar]
- 57.Li, M., et al. Predictive modelling of the distribution of Clematis sect. Fruticella s. str. under climate change reveals a range expansion during the Last Glacial Maximum. PeerJ. 8, e8729; 10.7717/peerj.8729 (2020). [DOI] [PMC free article] [PubMed]
- 58.Remya, K., Ramachandran, A. & Jayakumar, A. S. Predicting the current and future suitable habitat distribution of Myristica dactyloides Gaertn. using MaxEnt model in the Eastern Ghats, India. Ecological Engineering. 82, 184–188 (2015).
- 59.Moya, W., Jacome, G. & Yoo, C. Past, current, and future trends of red spiny lobster based on PCA with MaxEnt model in Galapagos Islands. Ecuador. Ecology and Evolution.7(13), 4881–4890 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Bystriakova, N., Peregrym, M., Erkens, R. H., Bezsmertna, O. & Schneider, H. Sampling bias in geographic and environmental space and its effect on the predictive power of species distribution models. Systematics and biodiversity.10(3), 305–315 (2012). [Google Scholar]
- 61.Ma, B. & Sun, J. Predicting the distribution of Stipa purpurea across the Tibetan Plateau via the MaxEnt model. BMC Ecology.18, 1–12 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Su, H., Bista, M. & Li, M. Mapping habitat suitability for Asiatic black bear and red panda in Makalu Barun National Park of Nepal from Maxent and GARP models. Scientific Reports11(1), 14135. 10.1038/s41598-021-93540-x (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Qin, A., Liu, B., Guo, Q., Bussmann, R.W., Ma, F, Jian, Z., Xu, G. & Pei, S. Maxent modeling for predicting impacts of climate change on the potential distribution of Thuja Sutchuenensis Franch., an extremely endangered conifer from southwestern China. Glob. Ecol. Conserv. 10, 139–146 (2017).
- 64.Dagtekin, D., Şahan, E. A., Denk, T., Köse, N. & Dalfes, H. N. Past, Present and future distributions of Oriental Beech (Fagus orientalis) under climate change projections. PLoS ONE15, e0242280 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Özdel, M. M., Ustaoğlu, B. & Cürebal, İ. Modeling of the Potential Distribution Areas Suitable for Olive (Olea europaea L.) in Türkiye from a Climate Change Perspective. Agriculture, 14(9), 1629 (2024).
- 66.Li, W., Xu, Z., Shi, M. & Chen, J. Prediction of potential geographical distribution patterns of Salix tetrasperma Roxb. Asia under different climate scenarios. Acta Ecol. Sin, 39, 3224–3234. (2019).
- 67.Araújo, M. B. & New, M. Ensemble forecasting of species distributions. Trends in Ecology & Evolution.22(1), 42–47 (2007). [DOI] [PubMed] [Google Scholar]
- 68.Pearson, R. G. et al. Model-based uncertainty in species range prediction. J. Biogeogr.33(10), 1704–1711 (2006). [Google Scholar]
- 69.Li, Y. P., Gao, X., An, Q., Sun, Z. & Wang, H. B. (2022). Ecological niche modelling based on ensemble algorithms to predicting current and future potential distribution of African swine fever virus in China. Scientific Reports. 12(1), 15614; 10.1038/s41598-022-20008-x. [DOI] [PMC free article] [PubMed]
- 70.Guo, L., Gao, Y., He, P., He, Y. & Meng, F. Modelling for predicting the potential geographical distribution of three Ephedra herbs in China. Plants.12(4), 787. 10.3390/plants12040787 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.R Core Team R: A language and environment for statistical computing (4.2.2). R Foundation for Statistical Computing. https://www.r-project.org/ (2022).
- 72.Thuiller, W., Georges, D., Gueguen, M., Engler, R., Breiner, F., Lafourcade, B., Patin, R., Blancheteau, H. biomod2: Ensemble Platform for Species Distribution Modelling. R Package Version 4.2.2. Available online at: https://CRAN.R-project.org/package=biomod2 (2024).
- 73.Ripley, B. Pattern Recognition and Neural Networks (Cambridge University, 1996). [Google Scholar]
- 74.Breiman, L., Friedman, J.H., Olshean, R. A. & Stone, C. J. Classification and Regression Trees. (Belmont, CA:Wadsworth, 1984).
- 75.Hastie, T., Tibshirani, R. & Buja, A. Flexible discriminant analysis by optimal scoring. J. Am. Stat. Assoc. 89, 1255–1270; 10.1080/01621459.1994.10476866 (1994).
- 76.Hastie, T. J. & Tibshirani, R. Generalized Additive Models (Chapman and Hall, 1990). [DOI] [PubMed] [Google Scholar]
- 77.Ridgeway, G. The state of boosting. Comput. Sci. Stat.31, 172–181 (1999). [Google Scholar]
- 78.McCullagh, P. & Nelder, J. A. Generalized Linear Models 2nd edn. (Chapman and Hall, 1989). [Google Scholar]
- 79.Friedman, J. H. Multivariate adaptive regression splines. The Annals of Statistics.19(1), 1–67 (1991). [Google Scholar]
- 80.Phillips, S. J., Anderson, R. P. & Schapire, R. E. Maximum entropy modelling of species geographic distributions. Ecol. Model. 190, 231–259; 10.1016/j.ecolmodel.2005.03.026 (2006).
- 81.Breiman, L. Random forests. Machine Learning.45, 5–32. 10.1023/A:1010933404324 (2001). [Google Scholar]
- 82.Busby, J. R. BIOCLIM—a bioclimate analysis and prediction system, in Nature Conservation: Cost Effective Biological Surveys and Data Analysis (ed. Margules, C. R. & Austin, M. P.) 64–68 (Canberra: CSIRO, 1991).
- 83.Capinha, C., Leung, B. & Anastácio, P. Predicting worldwide invasiveness forfour major problematic decapods: an evaluation of using different calibrationsets. Ecography34, 448–459 (2011). [Google Scholar]
- 84.Gama, M., Crespo, D., Dolbeth, M. & Anastácio, P. Predicting global habitat suitability for Corbicula fluminea using species distribution models: The importance of different environmental datasets. Ecological Modelling319, 163–169 (2016). [Google Scholar]
- 85.Beaumont, L. J. et al. Different climatic envelopes among invasive populations may lead tounderestimations of current and future biological invasions. Divers. Distrib.15(3), 409–420 (2009). [Google Scholar]
- 86.Barbet-Massin, M., Jiguet, F., Albert, C. H. & Thuiller, W. Selecting pseudoabsences for species distribution models: How, where and how many? Methods in Ecology and Evolution. 3(2), 327–338; 10. 1111/j. 2041- 210X. 2011. 00172. X (2012).
- 87.Allouche, O., Tsoar, A. & Kadmon, R. Assessing the accuracy of species distribution models: prevalence, kappa and the true skill statistic (TSS). J. Appl. Ecol.43, 1223–1232 (2006). [Google Scholar]
- 88.Liu, C., Berry, P. M., Dawson, T. P. & Pearson, R. G. Selecting thresholdsof occurrence in the prediction of species distributions. Ecography28, 385–393 (2005). [Google Scholar]
- 89.Elith, J. Quantitative Methods for Modelling Species Habitat: Comparative Performance and an Application to Australian Plants in Quantitative Methods for Conservation Biology (ed. Ferson, S. & Burgman, M.) 39–58 (Springer, New York, NY. 10.1007/0-387-22648-6_4 2000).
- 90.Phillips, S. J. & Dudík, M. Modeling of species distributions with Maxent: new extensions and a comprehensive evaluation. Ecography.31(2), 161–175 (2008). [Google Scholar]
- 91.Marmion, M., Parviainen, M., Luoto, M., Heikkinen, R. K. & Thuiller, W. Evaluation of consensus methods in predictive species distribution modelling. Diversity and Distributions15(1), 59–69 (2009). [Google Scholar]
- 92.Elith, J., Ferrier, S., Huettmann, F. & Leathwick, J. The evaluation strip: a new and robustmethod for plotting predicted responses fromspecies distribution models. Ecol. Model. 186, 280–289; 10.1016/j.ecolmodel.2004.12.007 (2005).
- 93.Zhong, Y., Xue, Z., Jiang, M., Liu, B. & Wang, G. The application of species distribution modeling in wetland restoration: A case study in the Songnen Plain. Northeast China. Ecological Indicators121, 107137 (2021). [Google Scholar]
- 94.Chen, B. et al. Distribution pattern and change prediction of Saposhnikovia divaricata suitable area in China under climate change. Ecological Indicators143, 109311 (2022). [Google Scholar]
- 95.de Andrade, A. F. A., Velazco, S. J. E. & Júnior, P. D. M. Enmtml: An R package for a straightforward construction of complex ecological niche models. Environ. Modell. Softw.125, 104615 (2020). [Google Scholar]
- 96.Jiménez-Valverde, A. & Lobo, J. M. Threshold criteria for conversion of probability of species presence to either–or presence–absence. Acta Oecologica31, 361–369 (2007).97. Guisan, A., Thuiller, W. & Zimmermann, N. E. Habitat Suitability and Distribution Models with Applications in R. (Cambridge, Cambridge University Press. 2017).
- 97.Mori, E. et al. How the South was won: current and potential range expansion of the crested porcupine in Southern Italy. Mammalian Biology.101, 11–19 (2021). [Google Scholar]
- 98.Kumari, P., et al. (2022). Modelling of Valeriana wallichii habitat suitability and niche dynamics in the Himalayan region under anticipated climate change. Biology. 11(4), 498; 10.3390/biology11040498 (2022). [DOI] [PMC free article] [PubMed]
- 99.Wani, Z. A., et al. Changing climatic scenarios anticipate dwindling of suitable habitats for endemic species of himalaya-predictions of ensemble modelling using Aconitum heterophyllum as a model plant. Sustainability. 14, 8491; 10.3390/su141 48491 (2022).
- 100.Naciri, Y. et al. Species delimitation in the East Asian species of the relict tree genus Zelkova (Ulmaceae): A complex history of diversification and admixture among species. Molecular Phylogenetics and Evolution134, 172–185. 10.1016/j.ympev.2019.02.010 (2019). [DOI] [PubMed] [Google Scholar]
- 101.Gulisashvili, V. Z.. Dendroflora of the Caucasus. (Academy of Sciences of Georgian SSR, Tbilisi, Georgia, 1961).
- 102.Kvavadze, E. V. & S. E. Connor. Zelkova carpinifolia (Pallas) K. Koch in Holocene sediments of Georgia—An indicator of climatic optima. Review of Palaeobotany and Palynology,133, 69–89 (2005).
- 103.Akhani, H., M. Djamali, A. Ghorbanalizadeh & E. Ramezani. Plant biodiversity of Hyrcanian relict forests, N Iran: An overview of the flora, vegetation, palaeoecology and conservation. Pakistan Journal of Botany,42, 231–258 (2010).105. Zhang, M. L., et al. Cenozoic evolutionary history of Zelkova (Ulmaceae), evidenced from ITS, trnL-trnF, psbA-trnH, and rbcL. Tree Genetics & Genomes.13, 101; 10.1007/s11295-017-1182-4 (2017).
- 104.Follieri, M., Magri, D. & Sadori, L. Late Pleistocene Zelkova extinction in Central Italy. New Phytologist.103(1), 269–273. 10.1111/j.1469-8137.1986.tb00613.x (1986). [Google Scholar]
- 105.Biltekin, et al. Anatolia: a long-time plant refuge area documented by pollen records over the last 23 million years. Review of Palaeobotany and Palynology. 215, 1–22. (2015).
- 106.Quézel, P. & Médail, F. Ecologie Et Biogéographie Des Forêts Du Bassinméditerranéen 571 (Elsevier, 2003). [Google Scholar]
- 107.Zohary, M. Geobotanical Foundations of the Middle East. 739 (Fischer, Stuttgart, 1973).
- 108.Di Pasquale, G., Garfi, G. & Quézel, P. Sur la présence d’un Zelkova nouveau en Sicile sud-orientale (Ulmaceae). Biocosme Mésogéen8–9, 401–409 (1992). [Google Scholar]
- 109.Giorgi, F. & Lionello, P. Climate change projections for the Mediterranean region. Global and Planetary Change.63(2–3), 90–104 (2008). [Google Scholar]
- 110.Avdeev, B. & Niemi, N. A. Rapid Pliocene exhumation of the central Greater Caucasus constrained by low‐temperature thermochronometry. Tectonics. 30(2), 10.1029/2010TC002808 (2011).
- 111.Sosson, M. et al. Subductions, obduction and collision in the Lesser Caucasus (Armenia, Azerbaijan, Georgia), new insights. Geological Society, London, Special Publications.340(1), 329–352 (2010). [Google Scholar]
- 112.Djamali, M. et al. A late Pleistocene long pollen record from Lake Urmia. NW Iran. Quaternary Research.69(3), 413–420 (2008). [Google Scholar]
- 113.Ramezani, E., Marvie Mohadjer, M. R., Knapp, H. D., Ahmadi, H. & Joosten, H. The late-Holocene vegetation history of the Central Caspian (Hyrcanian) forests of northern Iran. The Holocene. 18(2), 307–321; (2008).
- 114.Nakhutsrishvili, G., Zazanashvili, N., Batsatsashvili, K. & Montalvo, C. S. Colchic and Hyrcanian forests of the Caucasus: similarities, differences and conservation status. Flora Mediterranea. 25 (Special Issue), 185–192 (2015).
- 115.Nogués-Bravo, D. Predicting the past distribution of species climatic niches. Global Ecology and Biogeography.18(5), 521–531. 10.1111/j.1466-8238.2009.00476.x (2009). [Google Scholar]
- 116.Akhani, H., Djamali, M., Ghorbanalizadeh, A. & Ramezani, E. Plant biodiversity of Hyrcanian relict forests, N Iran: an overview of the flora, vegetation, palaeoecology and conservation. Pakistan Journal of Botany.42(1), 231–258 (2010). [Google Scholar]
- 117.Yousefzadeh, H., et al. Potential effects of climate change on future distribution of an endangered tree species, Acer mazandaranicum, in the Hyrcanian forest. Forest Ecology and Management. 555, 121654; 10.1016/j.foreco.2023.121654 (2024).
- 118.Soofi, M. et al. Livestock grazing in protected areas and its effects on large mammals in the Hyrcanian forest. Iran. Biological Conservation.217, 377–382 (2018). [Google Scholar]
- 119.Zhou, Y., Lu, X. & Zhang, G. Potentially differential impacts on niche overlap between Chinese endangered Zelkova schneideriana and its associated tree species under climate change. Frontiers in Ecology and Evolution. 11, 11:1218149; 10.3389/fevo.2023.1218149 (2023).
- 120.Sun, J., et al. Modelling the potential distribution of Zelkova schneideriana under different human activity intensities and climate change patterns in China. Global Ecology and Conservation, 21, e00840; 10.1016/j.gecco.2019.e00840 (2020).
- 121.Cao, C. & Tao, J. Predicting the areas of suitable distribution for Zelkova serrata in China under climate change. Sustainability.13(3), 1493. 10.3390/su13031493 (2021). [Google Scholar]
- 122.Nastos, P. T. & Zerefos, C. S. Spatial and temporal variability of consecutive dry and wet days in Greece. Atmospheric Research.94(4), 616–628 (2009). [Google Scholar]
- 123.Parey, S. Generating a set of temperature time series representative of recent past and near future climate. Frontiers in Environmental Science.7, 99 (2019). [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All the data generated/ analyzed during the study (i.e., the environmental variables and occurrence data), are available with the corresponding author on reasonable request.