Abstract
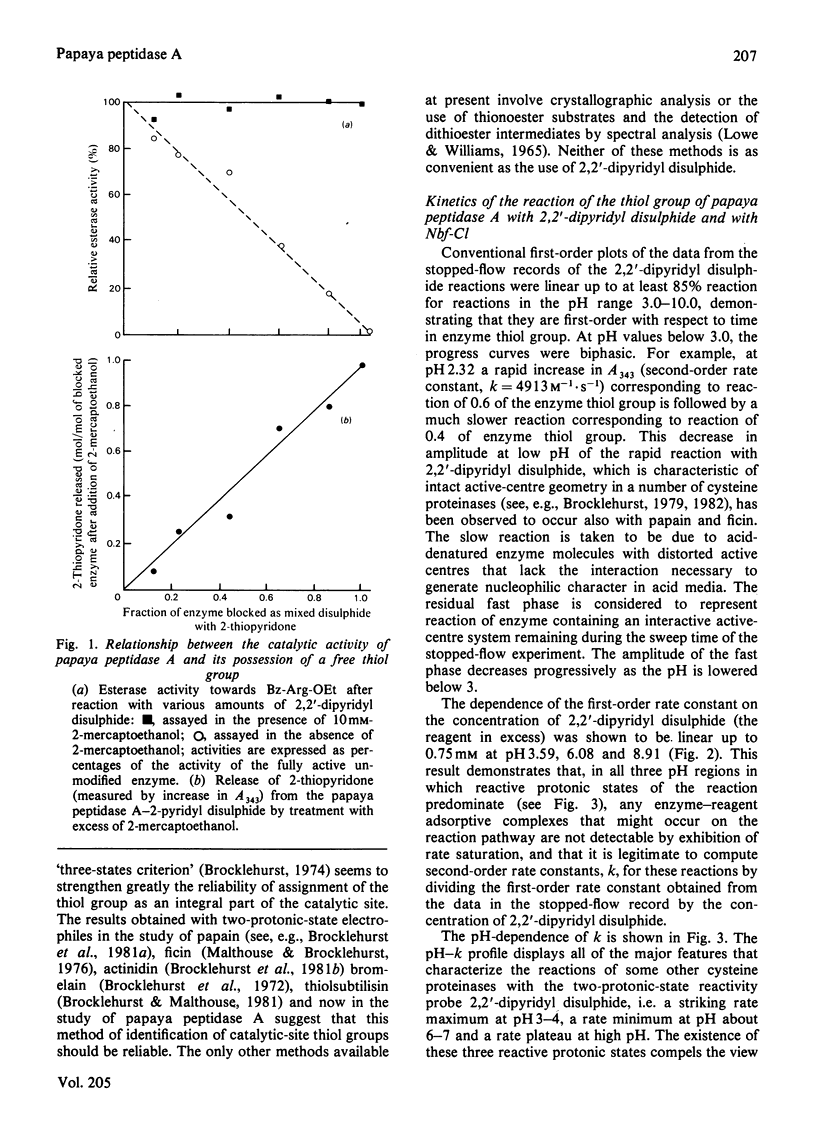
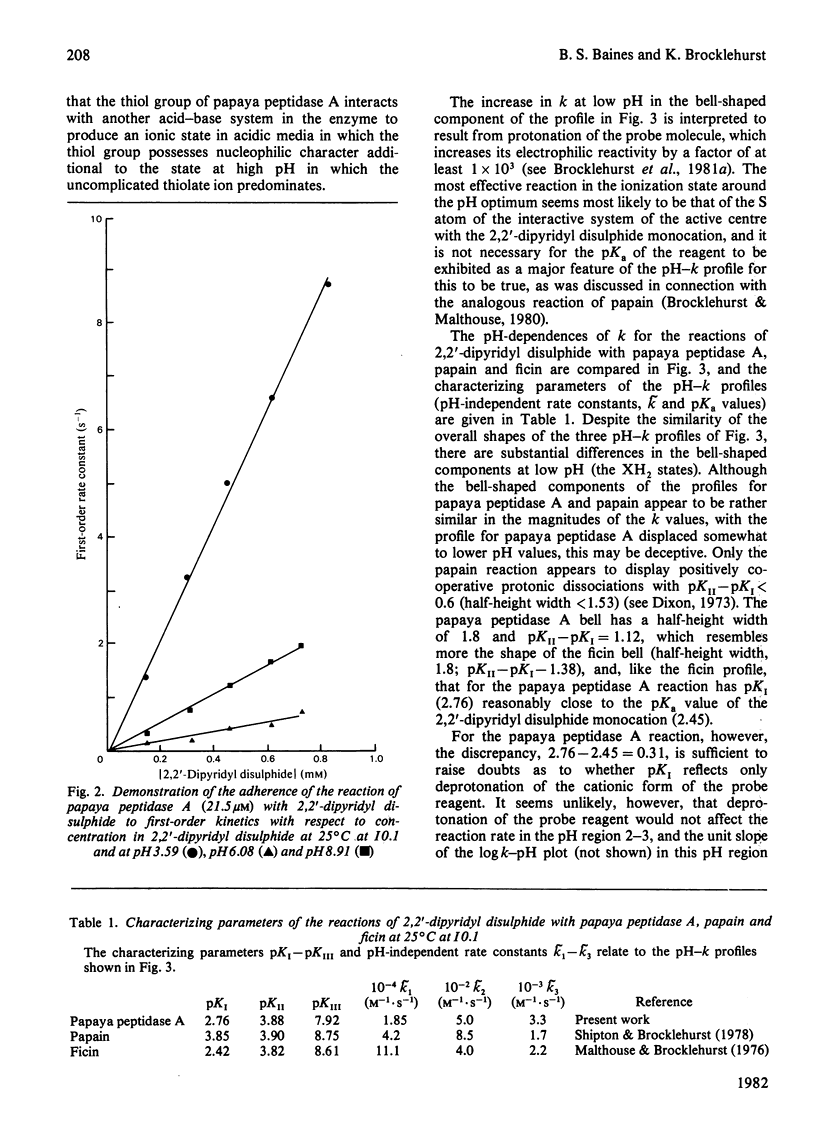
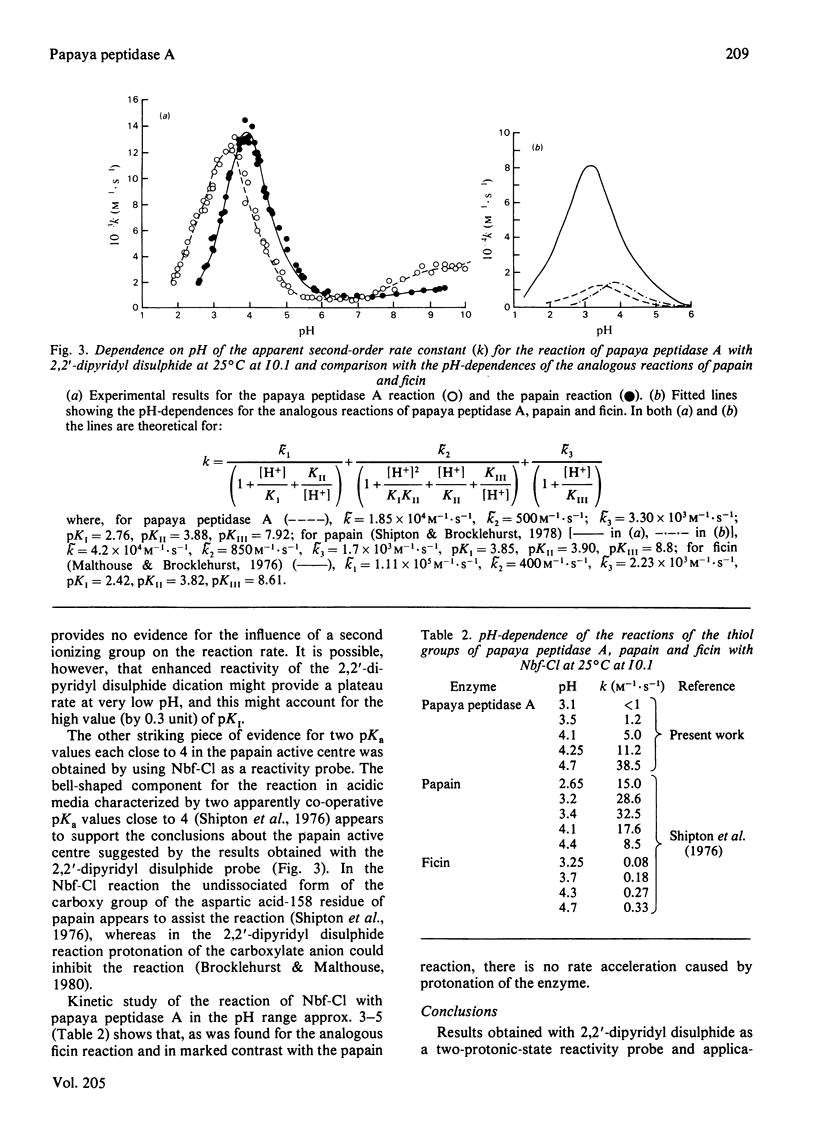
1. The proteinase papaya peptidase A, one of the major components of the latex of Carica papaya L., was shown to contain 1 thiol group per molecule; this thiol group is essential for catalytic activity and is part of the catalytic site. 2. The usefulness of two-protonic-state reactivity probes coupled with modification/activity-loss data in assigning a thiol group as an integral part of the catalytic site as against merely 'essential' for activity is discussed. 3. The active centre of papaya peptidase A was investigated by using 2,2'-dipyridyl disulphide and 4-chloro-7-nitrobenzofurazan as reactivity probes. The presence in the enzyme in weakly acidic media of an interactive system containing a nucleophile S atom (pKI3.9,pKII7.9) was demonstrated. 5. Papaya peptidase A resembles ficin (EC 3.4.22.3) and actinidin (the cysteine proteinase from Actinidin chinenis) in that it does not appear to possess a carboxy group able to influence the reactivity of the thiol group by change of ionization state at pH values of about 4, a situation that contrasts markedly with that which obtains in papain. 6. Implications of the results for possible variations in cysteine proteinase mechanism are discussed.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Angelides K. J., Fink A. L. Cryoenzymology of papain: reaction mechanism with an ester substrate. Biochemistry. 1978 Jun 27;17(13):2659–2668. doi: 10.1021/bi00606a032. [DOI] [PubMed] [Google Scholar]
- Angelides K. J., Fink A. L. Mechanism of Action of papain with a specific anilide substrate. Biochemistry. 1979 May 29;18(11):2355–2363. doi: 10.1021/bi00578a034. [DOI] [PubMed] [Google Scholar]
- Angelides K. J., Fink A. L. Mechanism of thiol protease catalysis: detection and stabilization of a tetrahedral intermediate in papain catalysis. Biochemistry. 1979 May 29;18(11):2363–2369. doi: 10.1021/bi00578a035. [DOI] [PubMed] [Google Scholar]
- Brocklehurst K., Crook E. M., Kierstan M. The mutability of stem bromelain: evidence for perturbation by structural transitions of the parameters that characterize the reaction of the essential thiol group of bromelain with 2,2'-dipyridyl disulphide. Biochem J. 1972 Jul;128(4):979–982. doi: 10.1042/bj1280979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brocklehurst K., Little G. Reactions of papain and of low-molecular-weight thiols with some aromatic disulphides. 2,2'-Dipyridyl disulphide as a convenient active-site titrant for papain even in the presence of other thiols. Biochem J. 1973 May;133(1):67–80. doi: 10.1042/bj1330067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brocklehurst K., Malthouse J. P. Evidence for a two-state transition in papain that may have no close analogue in ficin. Differences in the disposition of cationic sites and hydrophobic binding areas in the active centres of papain and ficin. Biochem J. 1980 Dec 1;191(3):707–718. doi: 10.1042/bj1910707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brocklehurst K., Malthouse J. P. Evidence that the lack of high catalytic activity of thiolsubtilisin towards specific substrates may be due to an inappropriately located proton-distribution system. Demonstration of highly nucleophilic character of the thiol group of thiolsubtilisin in the catalytically relevant ionization state of the active centre by use of a two-protonic-state reactivity probe. Biochem J. 1981 Mar 1;193(3):819–823. doi: 10.1042/bj1930819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brocklehurst K., Malthouse J. P., Shipton M. Evidence that binding to the s2-subsite of papain may be coupled with catalytically relevant structural change involving the cysteine-25-histidine-159 diad. Kinetics of the reaction of papain with a two-protonic-state reactivity probe containing a hydrophobic side chain. Biochem J. 1979 Nov 1;183(2):223–231. doi: 10.1042/bj1830223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brocklehurst K. Specific covalent modification of thiols: applications in the study of enzymes and other biomolecules. Int J Biochem. 1979;10(4):259–274. doi: 10.1016/0020-711x(79)90088-0. [DOI] [PubMed] [Google Scholar]
- Brocklehurst K. Two-protonic-state electrophiles as probes of enzyme mechanisms. Methods Enzymol. 1982;87:427–469. doi: 10.1016/s0076-6879(82)87026-2. [DOI] [PubMed] [Google Scholar]
- Dixon H. B. Shapes of curves of pH-dependence of reactions. Biochem J. 1973 Jan;131(1):149–154. doi: 10.1042/bj1310149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWE G., WILLIAMS A. A STUDY OF SOME THIOL ESTER HYDROLYSES AS MODELS FOR THE DEACYLATION STEP OF PAPAIN-CATALYSED HYDROLYSES. Biochem J. 1965 Jul;96:194–198. doi: 10.1042/bj0960194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malthouse J. P., Brocklehurst K. Preparation of fully active ficin from Ficus glabrata by covalent chromatography and characterization of its active centre by using 2,2'-depyridyl disulphide as a reactivity probe. Biochem J. 1976 Nov;159(2):221–234. doi: 10.1042/bj1590221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson G. W. Isolation and characterization of papaya peptidase A from commercial chymopapain. Biochemistry. 1975 Aug 12;14(16):3695–3700. doi: 10.1021/bi00687a028. [DOI] [PubMed] [Google Scholar]
- Shipton M., Brochlehurst K. Characterization of the papain active centre by using two-protonic-state electrophiles as reactivity probes. Evidence for nucleophilic reactivity in the un-interrupted cysteine-25-histidine-159 interactive system. Biochem J. 1978 May 1;171(2):385–401. doi: 10.1042/bj1710385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shipton M., Stuchbury T., Brocklehurst K. 4-Chloro-7-nitrobenzo-2-oxa-1,3-diazole as a reactivity probe for the investigation of the thiol proteinases. evidence that ficin and bromelain may lack carboxyl groups conformationally equivalent to that of aspartic acid-158 of papain. Biochem J. 1976 Nov;159(2):235–244. doi: 10.1042/bj1590235. [DOI] [PMC free article] [PubMed] [Google Scholar]


