Abstract
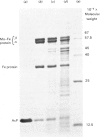
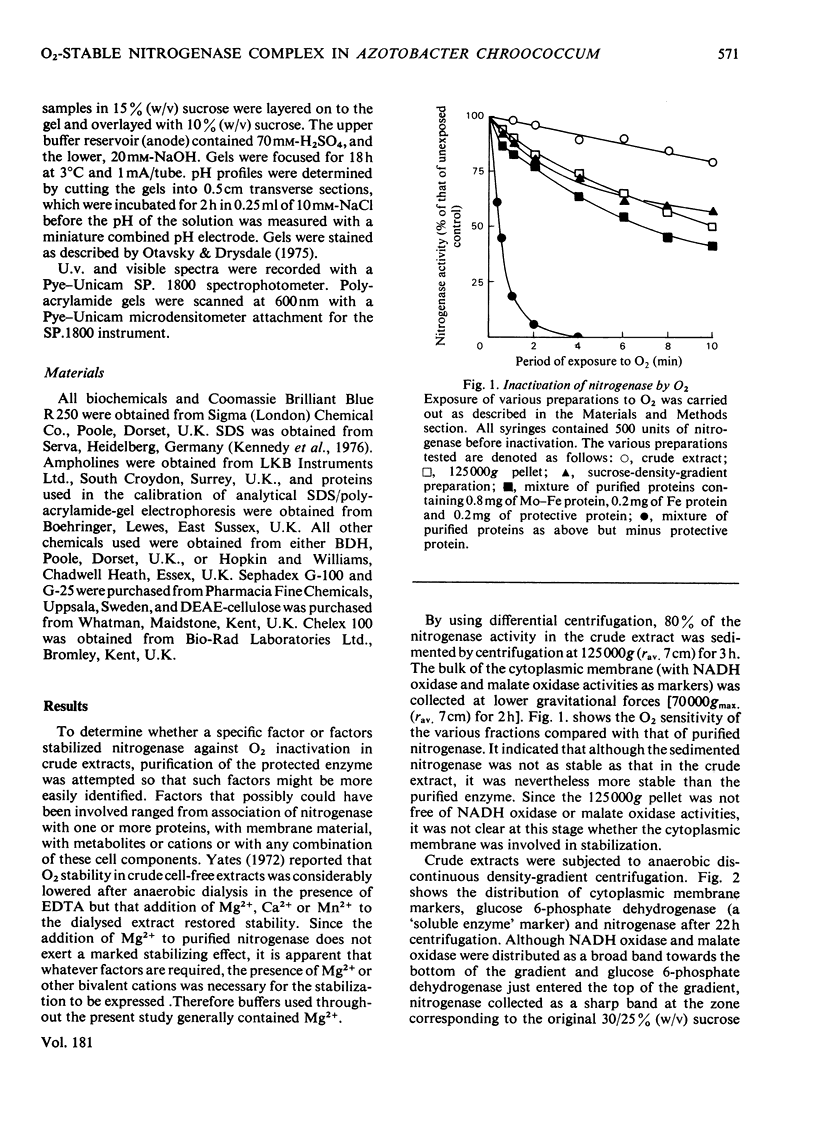
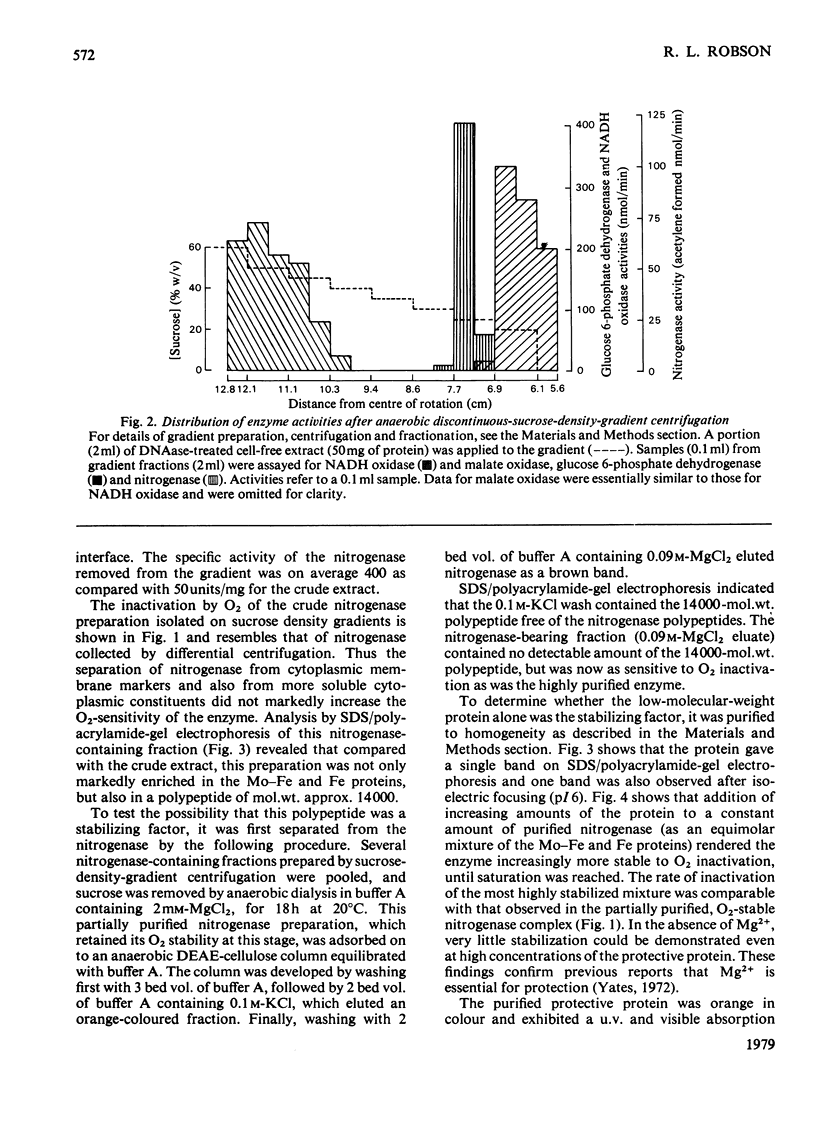
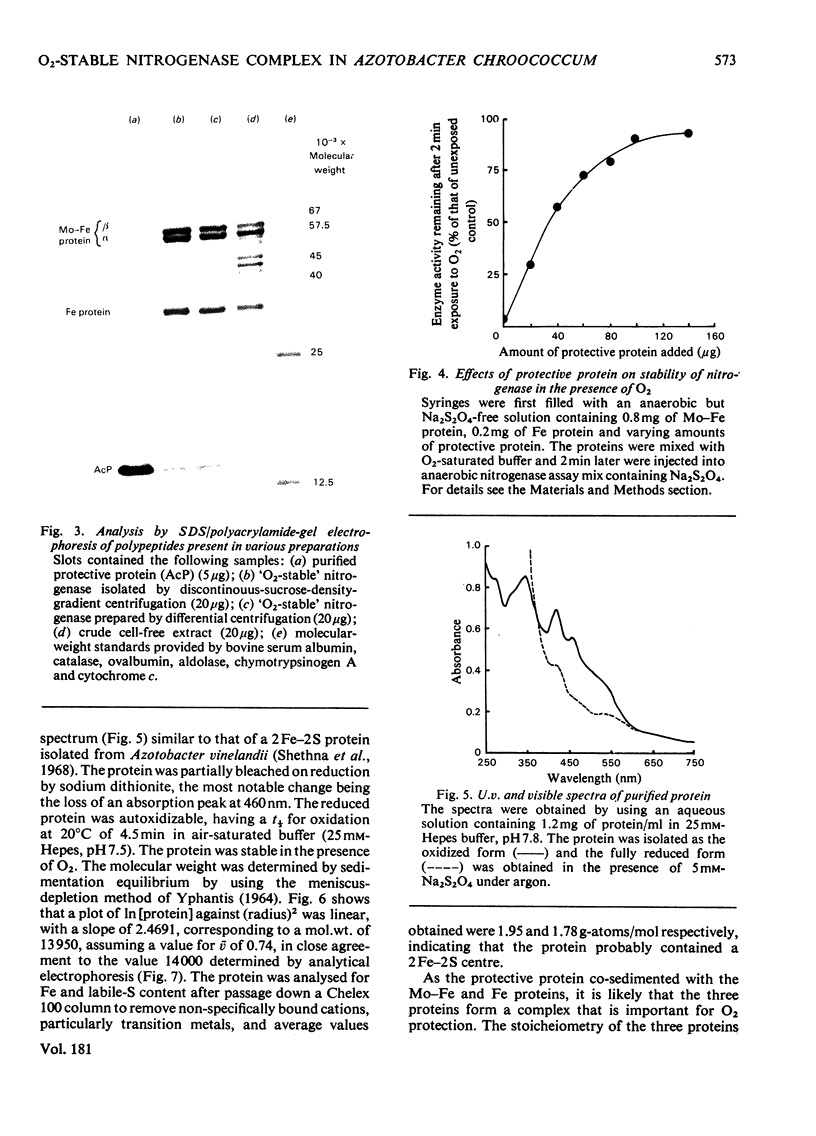
In crude cell-free extracts of Azotobacter chroococcum, nitrogenase was much less sensitive to irreversible inactivation by O2 than was the purified enzyme. When nitrogenase was partially purified by anaerobic discontinuous sucrose-density-gradient centrifugation, O2-tolerance was retained. This preparation was considerably enriched in four polypeptides, three of which were derived from the Mo-Fe(molybdenum-iron) protein and Fe (iron) protein of nitrogenase. The fourth was purified to homogeneity and shown to be an iron-sulphur protein (mol.wt. 14000) probably containing a 2Fe--2S centre. When this protein was added to purified nitrogenase, the enzyme was rendered O2-tolerant, through stabilization was Mg2+-dependent. The isolated O2-tolerant nitrogenase was an equimolar stoicheiometric complex between the MO--Fe, Fe and protective proteins. It is likely that the formation of this complex in vivo is the mechanism of 'conformational protection' in this organism.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BULEN W. A., BURNS R. C., LECOMTE J. R. NITROGEN FIXATION: HYDROSULFITE AS ELECTRON DONOR WITH CELL-FREE PREPARATIONS OF AZOTOBACTER VINELANDII AND RHODOSPIRILLUM RUBRUM. Proc Natl Acad Sci U S A. 1965 Mar;53:532–539. doi: 10.1073/pnas.53.3.532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bulen W. A., LeComte J. R. Nitrogenase complex and its components. Methods Enzymol. 1972;24:456–470. doi: 10.1016/0076-6879(72)24091-5. [DOI] [PubMed] [Google Scholar]
- Bulen W. A., LeComte J. R. The nitrogenase system from Azotobacter: two-enzyme requirement for N2 reduction, ATP-dependent H2 evolution, and ATP hydrolysis. Proc Natl Acad Sci U S A. 1966 Sep;56(3):979–986. doi: 10.1073/pnas.56.3.979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DOEG K. A., ZIEGLER D. M. Simplified methods for the estimation of iron in mitochondria and submitochondrial fractions. Arch Biochem Biophys. 1962 Apr;97:37–40. doi: 10.1016/0003-9861(62)90041-3. [DOI] [PubMed] [Google Scholar]
- Dalton H., Postgate J. R. Effect of oxygen on growth of Azotobacter chroococcum in batch and continuous cultures. J Gen Microbiol. 1968 Dec;54(3):463–473. doi: 10.1099/00221287-54-3-463. [DOI] [PubMed] [Google Scholar]
- Drozd J., Postgate J. R. Effects of oxygen on acetylene reduction, cytochrome content and respiratory activity of Azotobacter chroococcum. J Gen Microbiol. 1970 Sep;63(1):63–73. doi: 10.1099/00221287-63-1-63. [DOI] [PubMed] [Google Scholar]
- Eady R. R., Smith B. E., Cook K. A., Postgate J. R. Nitrogenase of Klebsiella pneumoniae. Purification and properties of the component proteins. Biochem J. 1972 Jul;128(3):655–675. doi: 10.1042/bj1280655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez-Moreno C. Oxidative inactivation of the Fe-protein from Clostridium pasteurianum nitrogenase. FEBS Lett. 1978 May 15;89(2):304–306. doi: 10.1016/0014-5793(78)80242-7. [DOI] [PubMed] [Google Scholar]
- Haaker H., Veeger C. Involvement of the cytoplasmic membrane in nitrogen fixation by Azotobacter vinelandii. Eur J Biochem. 1977 Jul 1;77(1):1–10. doi: 10.1111/j.1432-1033.1977.tb11634.x. [DOI] [PubMed] [Google Scholar]
- Kelly M. Comparisons and cross reactions of nitrogenase from Klebsiella pneumoniae, Azotobacter chroococcum and Bacillus polymyxa. Biochim Biophys Acta. 1969;191(3):527–540. doi: 10.1016/0005-2744(69)90346-5. [DOI] [PubMed] [Google Scholar]
- Kelly M. Some properties of purified nitrogenase of Azotobacter chroococcum. Biochim Biophys Acta. 1969 Jan 7;171(1):9–22. doi: 10.1016/0005-2744(69)90101-6. [DOI] [PubMed] [Google Scholar]
- Kennedy C., Eady R. R., Kondorosi E., Rekosh D. K. The molybdenum--iron protein of Klebsiella pneumoniae nitrogenase. Evidence for non-identical subunits from peptide 'mapping'. Biochem J. 1976 May 1;155(2):383–389. doi: 10.1042/bj1550383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- NEWTON J. W., WILSON P. W., BURRIS R. H. Direct demonstration of ammonia as an intermediate in nitrogen fixation by Azotobacter. J Biol Chem. 1953 Sep;204(1):445–451. [PubMed] [Google Scholar]
- Otavsky W. I., Drysdale J. W. Recent staining artifacts with LKB "Ampholines" on gel isoelectric-focusing. Anal Biochem. 1975 May 12;65(1-2):533–536. doi: 10.1016/0003-2697(75)90538-2. [DOI] [PubMed] [Google Scholar]
- Petering D., Fee J. A., Palmer G. The oxygen sensitivity of spinach ferredoxin and other iron-sulfur proteins. The formation of protein-bound sulfur-zero. J Biol Chem. 1971 Feb 10;246(3):643–653. [PubMed] [Google Scholar]
- Reid M. S., Bieleski R. L. A simple apparatus for vertical flat-sheet polyacrylamide gel electrophoresis. Anal Biochem. 1968 Mar;22(3):374–381. doi: 10.1016/0003-2697(68)90278-9. [DOI] [PubMed] [Google Scholar]
- Senior P. J., Dawes E. A. Poly- -hydroxybutyrate biosynthesis and the regulation of glucose metabolism in Azotobacter beijerinckii. Biochem J. 1971 Nov;125(1):55–66. doi: 10.1042/bj1250055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shethna Y. I., DerVartanian D. V., Beinert H. Non heme (iron-sulfur) proteins of Azotobacter vinelandii. Biochem Biophys Res Commun. 1968 Jun 28;31(6):862–868. doi: 10.1016/0006-291x(68)90531-7. [DOI] [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]
- YPHANTIS D. A. EQUILIBRIUM ULTRACENTRIFUGATION OF DILUTE SOLUTIONS. Biochemistry. 1964 Mar;3:297–317. doi: 10.1021/bi00891a003. [DOI] [PubMed] [Google Scholar]
- Yates M. G. Effect of non-haem iron proteins and cytochrome C from Azotobacter upon the activity and oxygen sensitivity of Azobacter nitrogenase. FEBS Lett. 1970 Jun 27;8(5):281–285. doi: 10.1016/0014-5793(70)80287-3. [DOI] [PubMed] [Google Scholar]
- Yates M. G., Planqué K. Nitrogenase from Azotobacter chroococcum. Purification and properties of the component proteins. Eur J Biochem. 1975 Dec 15;60(2):467–476. doi: 10.1111/j.1432-1033.1975.tb21025.x. [DOI] [PubMed] [Google Scholar]
- Yates M. G. The effect of ATP upon the oxygen sensitivity of nitrogenase from Azotobacter chroococcum. Eur J Biochem. 1972 Sep 18;29(2):386–392. doi: 10.1111/j.1432-1033.1972.tb02000.x. [DOI] [PubMed] [Google Scholar]