Abstract
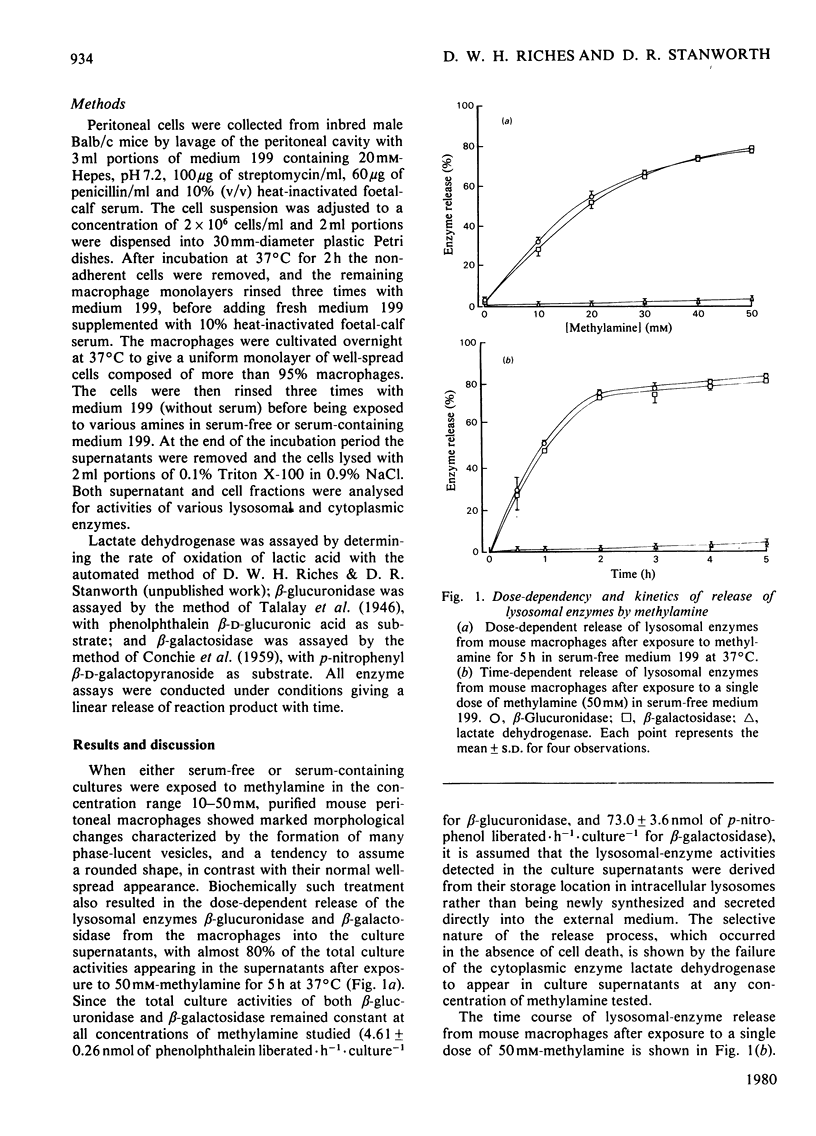
Cultured mouse peritoneal macrophages were found to release substantial amounts of lysosomal beta-glucuronidase and beta-glactosidase activites when exposed to millimolar concentrations of various primary aliphatic monoamines. With methylamine, ethylamine, propylamine and butylamine, lysosomal enzyme release was selective, but further increases in the aliphatic chain length resulted in the compounds becoming lytic. By contrast, structurally related primary aliphatic diamines proved to be inactive at inducing both selective and lytic lysosomal-enzyme discharge.
Full text
PDF
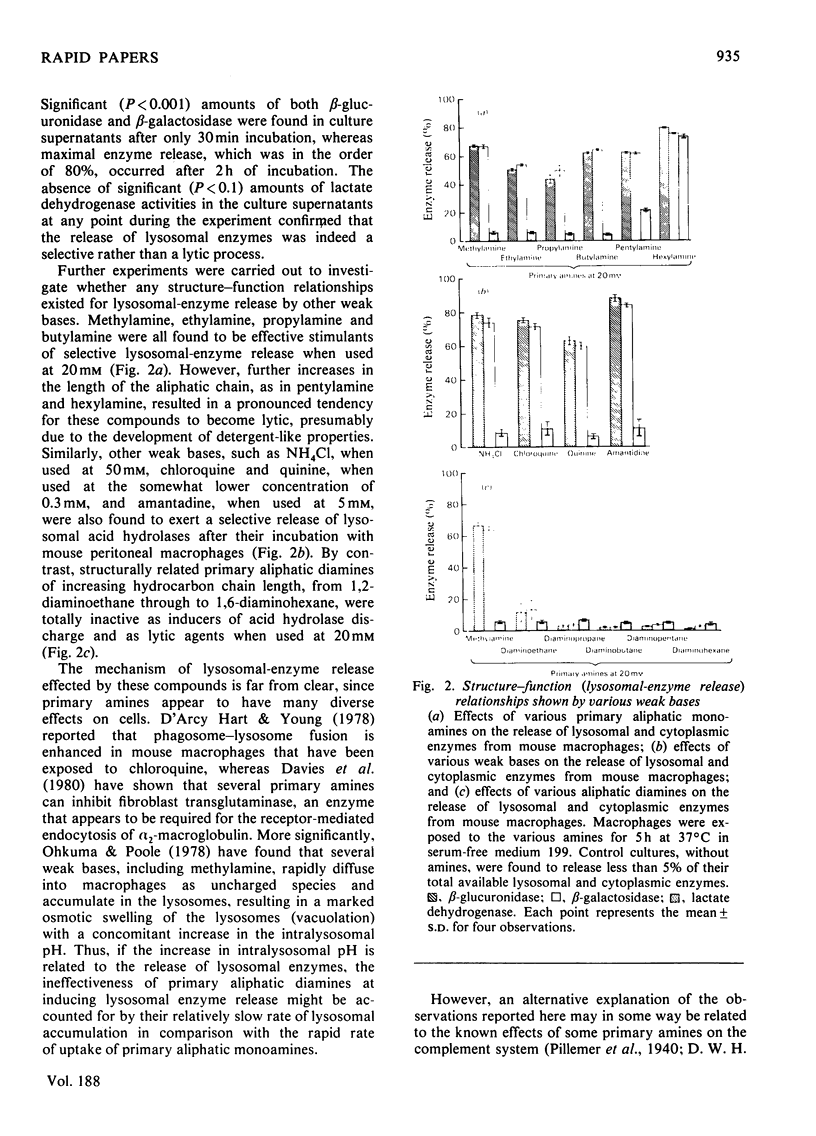

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barber T. A., Burkholder P. M. Enumeration and ultrastructure of C4-producing free alveolar cells from guinea pig lung. J Immunol. 1978 Mar;120(3):716–725. [PubMed] [Google Scholar]
- Bentley C., Fries W., Brade V. Synthesis of factors D, B and P of the alternative pathway of complement activation, as well as of C3, by guinea-pig peritoneal macrophages in vitro. Immunology. 1978 Dec;35(6):971–980. [PMC free article] [PubMed] [Google Scholar]
- Bonney R. J., Wightman P. D., Davies P., Sadowski S. J., Kuehl F. A., Jr, Humes J. L. Regulation of prostaglandin synthesis and of the selective release of lysosomal hydrolases by mouse peritoneal macrophages. Biochem J. 1978 Nov 15;176(2):433–442. doi: 10.1042/bj1760433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CONCHIE J., FINDLAY J., LEVVY G. A. Mammalian glycosidases; distribution in the body. Biochem J. 1959 Feb;71(2):318–325. doi: 10.1042/bj0710318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cardella C. J., Davies P., Allison A. C. Immune complexes induce selective release of lysosomal hydrolases from macrophages. Nature. 1974 Jan 4;247(5435):46–48. doi: 10.1038/247046a0. [DOI] [PubMed] [Google Scholar]
- Davies P. J., Davies D. R., Levitzki A., Maxfield F. R., Milhaud P., Willingham M. C., Pastan I. H. Transglutaminase is essential in receptor-mediated endocytosis of alpha 2-macroglobulin and polypeptide hormones. Nature. 1980 Jan 10;283(5743):162–167. doi: 10.1038/283162a0. [DOI] [PubMed] [Google Scholar]
- Hart P. D., Young M. R. Manipulations of the phagosome-lysosome fusion response in cultured macrophages. Enhancement of fusion by chloroquine and other amines. Exp Cell Res. 1978 Jul;114(2):486–490. doi: 10.1016/0014-4827(78)90516-5. [DOI] [PubMed] [Google Scholar]
- Hsueh W., Gonzalez-Crussi F., Hanneman E. Prostaglandin synthesis in different phases of phagocytosis in lung macrophages. Nature. 1980 Jan 3;283(5742):80–82. doi: 10.1038/283080a0. [DOI] [PubMed] [Google Scholar]
- Humes J. L., Bonney R. J., Pelus L., Dahlgren M. E., Sadowski S. J., Kuehl F. A., Jr, Davies P. Macrophages synthesis and release prostaglandins in response to inflammatory stimuli. Nature. 1977 Sep 8;269(5624):149–151. doi: 10.1038/269149a0. [DOI] [PubMed] [Google Scholar]
- Littman B. H., Ruddy S. Production of the second component of complement by human monocytes: stimulation by antigen-activated lymphocytes or lymphokines. J Exp Med. 1977 May 1;145(5):1344–1352. doi: 10.1084/jem.145.5.1344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller W., Hanauske-Abel H., Loos M. Biosynthesis of the first component of complement by human and guinea pig peritoneal macrophages: evidence for an independent production of the C1 subunits. J Immunol. 1978 Oct;121(4):1578–1584. [PubMed] [Google Scholar]
- Ohkuma S., Poole B. Fluorescence probe measurement of the intralysosomal pH in living cells and the perturbation of pH by various agents. Proc Natl Acad Sci U S A. 1978 Jul;75(7):3327–3331. doi: 10.1073/pnas.75.7.3327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pantalone R., Page R. C. Enzyme production and secretion by lymphokine-activated macrophages. J Reticuloendothel Soc. 1977 May;21(5):343–357. [PubMed] [Google Scholar]
- Schorlemmer H. U., Bitter-Suermann D., Allison A. C. Complement activation by the alternative pathway and macrophage enzyme secretion in the pathogenesis of chronic inflammation. Immunology. 1977 Jun;32(6):929–940. [PMC free article] [PubMed] [Google Scholar]
- Schorlemmer H. U., Burger R., Hylton W., Allison A. C. Induction of lysosomal enzyme release from cultured macrophages by dextran sulfate. Clin Immunol Immunopathol. 1977 Jan;7(1):88–96. doi: 10.1016/0090-1229(77)90033-2. [DOI] [PubMed] [Google Scholar]
- Schorlemmer H. U., Davies P., Allison A. C. Ability of activated complement components to induce lysosomal enzyme release from macrophages. Nature. 1976 May 6;261(5555):48–49. doi: 10.1038/261048a0. [DOI] [PubMed] [Google Scholar]
- Schorlemmer H. U., Edwards J. H., Davies P., Allison A. C. Macrophage responses to mouldy hay dust, Micropolyspora faeni and zymosan, activators of complement by the alternative pathway. Clin Exp Immunol. 1977 Feb;27(2):198–207. [PMC free article] [PubMed] [Google Scholar]
- Unkeless J. C., Gordon S., Reich E. Secretion of plasminogen activator by stimulated macrophages. J Exp Med. 1974 Apr 1;139(4):834–850. doi: 10.1084/jem.139.4.834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Werb Z., Gordon S. Secretion of a specific collagenase by stimulated macrophages. J Exp Med. 1975 Aug 1;142(2):346–360. doi: 10.1084/jem.142.2.346. [DOI] [PMC free article] [PubMed] [Google Scholar]


