Abstract
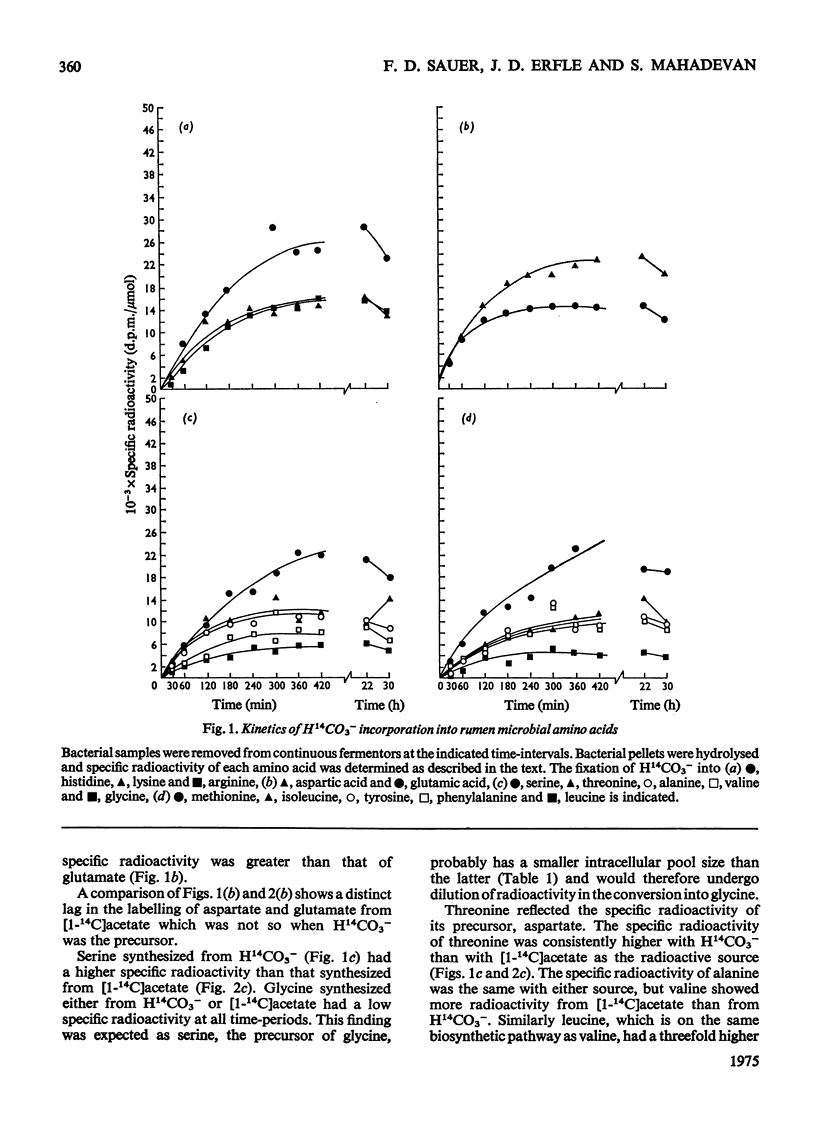
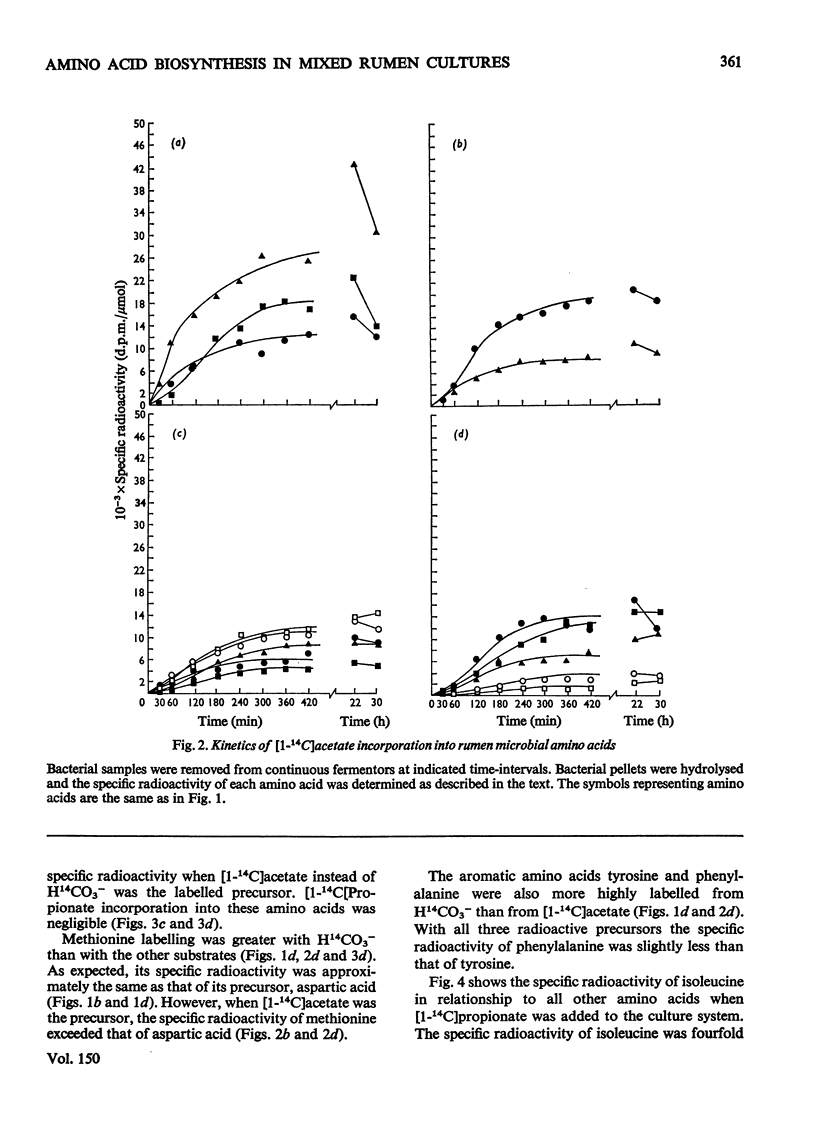
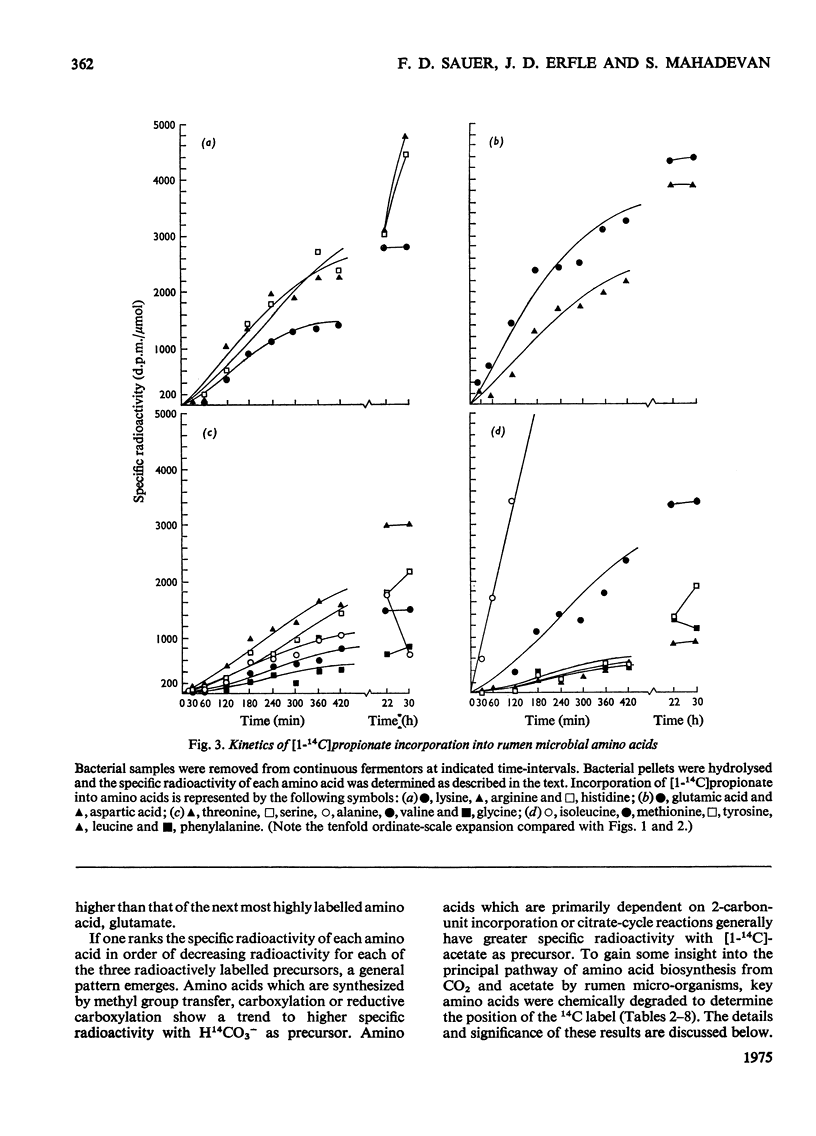
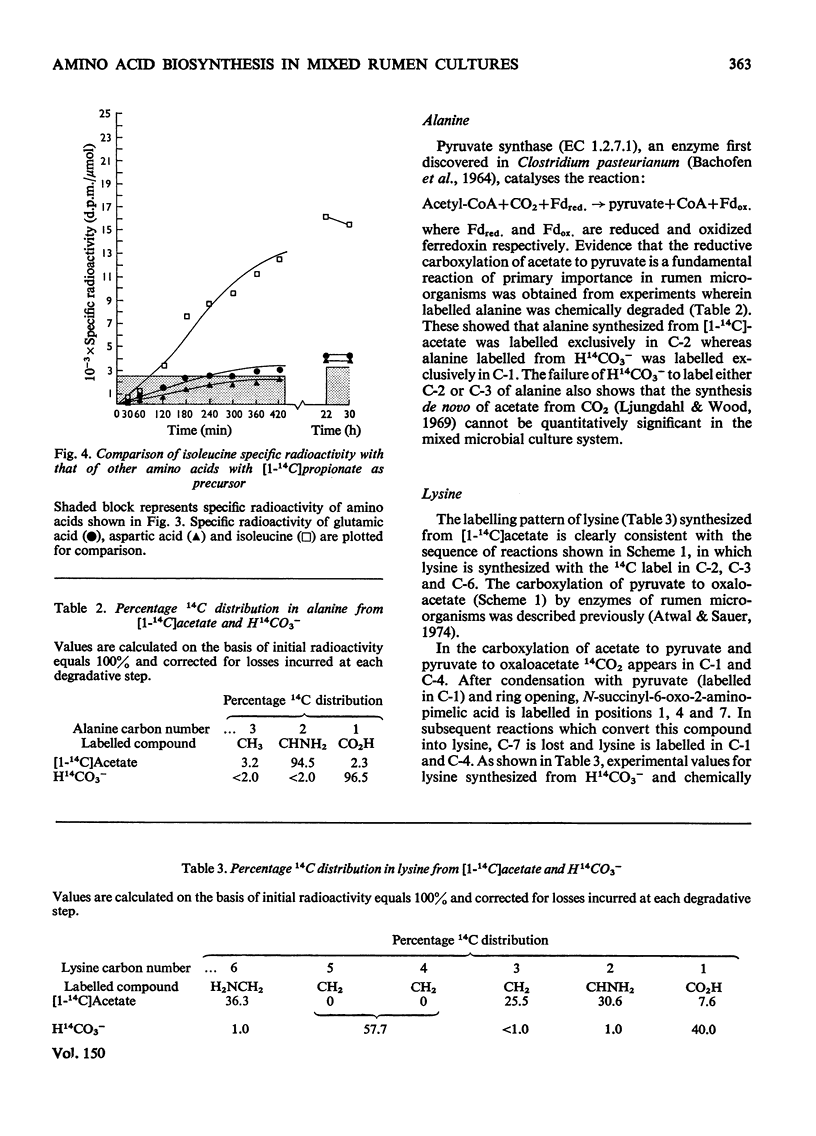
Mixed rumen micro-organisms, maintained in continuous culture readily incorporated labelled HCO3- and acetate into amino acids. Labelled propionate, in contrast, was utilized only for isoleucine biosynthesis, but failed to label other amino acids to any significant extent. Evidence was obtained showing that in these mixed, i.e. symbiotic, cultures foward tricarboxylic acid-cycle reactions only proceed to 2-oxoglutarate. 14C distribution in amino acids clearly shows that 2-oxoglutarate is not oxidized further by tricarboxylic acid-cycle enzymes. Instead, acetate is carboxylated to pyruvate which is then carboxylated to oxaloacetate. Oxaloacetate equilibrates with fumarate and thereby carbon atoms 1 and 4 as well as carbon atoms 2 and 3 are randomized. Evidence was also obtained for the carboxylation of propionate to 2-oxobutyrate, isovalerate to 4-methyl-2-oxopentanoate, phenylacetate and hydroxyphentlacetate to the corresponding phenyl- and hydroxyphenyl-pyruvic acids and succinate to 2-oxoglutarate. Of the amino acid precursors investigated, only 3-hydroxypyruvate, the precursor of serine, appeared to be synthesized via an oxidative step, i.e. 3-phosphoglyceric acid to 3-phosphohydroxypyruvic acid. Most 2-oxo precursors of amino acids in these organisms appear to be formed via reductive carboxylation of the precursor acid.
Full text
PDF









Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allison M. J. Biosynthesis of amono acids by ruminal microorganisms. J Anim Sci. 1969 Nov;29(5):797–807. doi: 10.2527/jas1969.295797x. [DOI] [PubMed] [Google Scholar]
- Allison M. J., Peel J. L. The biosynthesis of valine from isobutyrate by peptostreptococcus elsdenii and Bacteroides ruminicola. Biochem J. 1971 Feb;121(3):431–437. doi: 10.1042/bj1210431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BACHOFEN R., BUCHANAN B. B., ARNON D. I. FERREDOXIN AS A REDUCTANT IN PYRUVATE SYNTHESIS BY A BACTERIAL EXTRACT. Proc Natl Acad Sci U S A. 1964 Apr;51:690–694. doi: 10.1073/pnas.51.4.690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BUCHANAN B. B., BACHOFEN R., ARNON D. I. ROLE OF FERREDOXIN IN THE REDUCTIVE ASSIMILATION OF CO2 AND ACETATE BY EXTRACTS OF THE PHOTOSYNTHETIC BACTERIUM, CHROMATIUM. Proc Natl Acad Sci U S A. 1964 Sep;52:839–847. doi: 10.1073/pnas.52.3.839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchanan B. B. Orthophosphate requirement for the formation of phosphoenolpyruvate from pyruvate by enzyme preparations from photosynthetic bacteria. J Bacteriol. 1974 Sep;119(3):1066–1068. doi: 10.1128/jb.119.3.1066-1068.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchanan B. B. Role of ferredoxin in the synthesis of alpha-ketobutyrate from propionyl coenzyme A and carbon dioxide by enzymes from photosynthetic and nonphotosynthetic bacteria. J Biol Chem. 1969 Aug 10;244(15):4218–4223. [PubMed] [Google Scholar]
- CERIOTTI G. A microchemical determination of desoxyribonucleic acid. J Biol Chem. 1952 Sep;198(1):297–303. [PubMed] [Google Scholar]
- Cooper R. A., Kornberg H. L. Net formation of phosphoenolpyruvate from pyruvate by Escherichia coli. Biochim Biophys Acta. 1965 Jul 8;104(2):618–620. doi: 10.1016/0304-4165(65)90374-0. [DOI] [PubMed] [Google Scholar]
- Emmanuel B., Milligan L. P. Enzymes of the conversion of succinate to glutamate in extracts of rumen microorganisms. Can J Biochem. 1972 Jan;50(1):1–8. doi: 10.1139/o72-001. [DOI] [PubMed] [Google Scholar]
- Evans H. J., Wood H. G. Purification and properties of pyruvate phosphate dikinase from propionic acid bacteria. Biochemistry. 1971 Mar 2;10(5):721–729. doi: 10.1021/bi00781a001. [DOI] [PubMed] [Google Scholar]
- Evans M. C., Buchanan B. B., Arnon D. I. A new ferredoxin-dependent carbon reduction cycle in a photosynthetic bacterium. Proc Natl Acad Sci U S A. 1966 Apr;55(4):928–934. doi: 10.1073/pnas.55.4.928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finlayson A. J. Degradation of the carbon skeleton of glutamic acid. Can J Biochem. 1966 Apr;44(4):397–401. doi: 10.1139/o66-049. [DOI] [PubMed] [Google Scholar]
- Gottschalk G., Barker H. A. Presence and stereospecificity of citrate synthase in anaerobic bacteria. Biochemistry. 1967 Apr;6(4):1027–1034. doi: 10.1021/bi00856a011. [DOI] [PubMed] [Google Scholar]
- HOARE D. S. The photo-assimilation of acetate by Rhodospirillum rubrum. Biochem J. 1963 May;87:284–301. doi: 10.1042/bj0870284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoare D. S., Gibson J. Photoassimilation of acetate and the biosynthesis of amino acids by Chlorobium thiosulphatophilum. Biochem J. 1964 Jun;91(3):546–559. doi: 10.1042/bj0910546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jungermann K., Thauer R. K., Decker K. The synthesis of one-carbon units from CO2 in Clostridium kluyveri. Eur J Biochem. 1968 Jan;3(3):351–359. doi: 10.1111/j.1432-1033.1968.tb19536.x. [DOI] [PubMed] [Google Scholar]
- LEWIS D., ELSDEN S. R. The fermentation of L-threonine, L-serine, L-cysteine and acrylic acid by a gram-negative coccus. Biochem J. 1955 Aug;60(4):683–692. doi: 10.1042/bj0600683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ljungdahl L. G. Total synthesis of acetate from CO2 by heterotrophic bacteria. Annu Rev Microbiol. 1969;23:515–538. doi: 10.1146/annurev.mi.23.100169.002503. [DOI] [PubMed] [Google Scholar]
- Milligan L. P. Carbon dioxide fixing pathways of glutamic acid synthesis in the rumen. Can J Biochem. 1970 Apr;48(4):463–468. doi: 10.1139/o70-075. [DOI] [PubMed] [Google Scholar]
- Purser D. B., Buechler S. M. Amino acid composition of rumen organisms. J Dairy Sci. 1966 Jan;49(1):81–84. doi: 10.3168/jds.S0022-0302(66)87791-3. [DOI] [PubMed] [Google Scholar]
- SLYTER L. L., NELSON W. O., WOLIN M. J. MODIFICATIONS OF A DEVICE FOR MAINTENANCE OF THE RUMEN MICROBIAL POPULATION IN CONTINUOUS CULTURE. Appl Microbiol. 1964 Jul;12:374–377. doi: 10.1128/am.12.4.374-377.1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sauer F., Erfle J. D., Binns M. R. Turnover rates and intracellular pool size distribution of citrate cycle intermediates in normal, diabetic and fat-fed rats estimated by computer analysis from specific activity decay data of 14C-labeled citrate cycle acids. Eur J Biochem. 1970 Dec;17(2):350–363. doi: 10.1111/j.1432-1033.1970.tb01173.x. [DOI] [PubMed] [Google Scholar]


