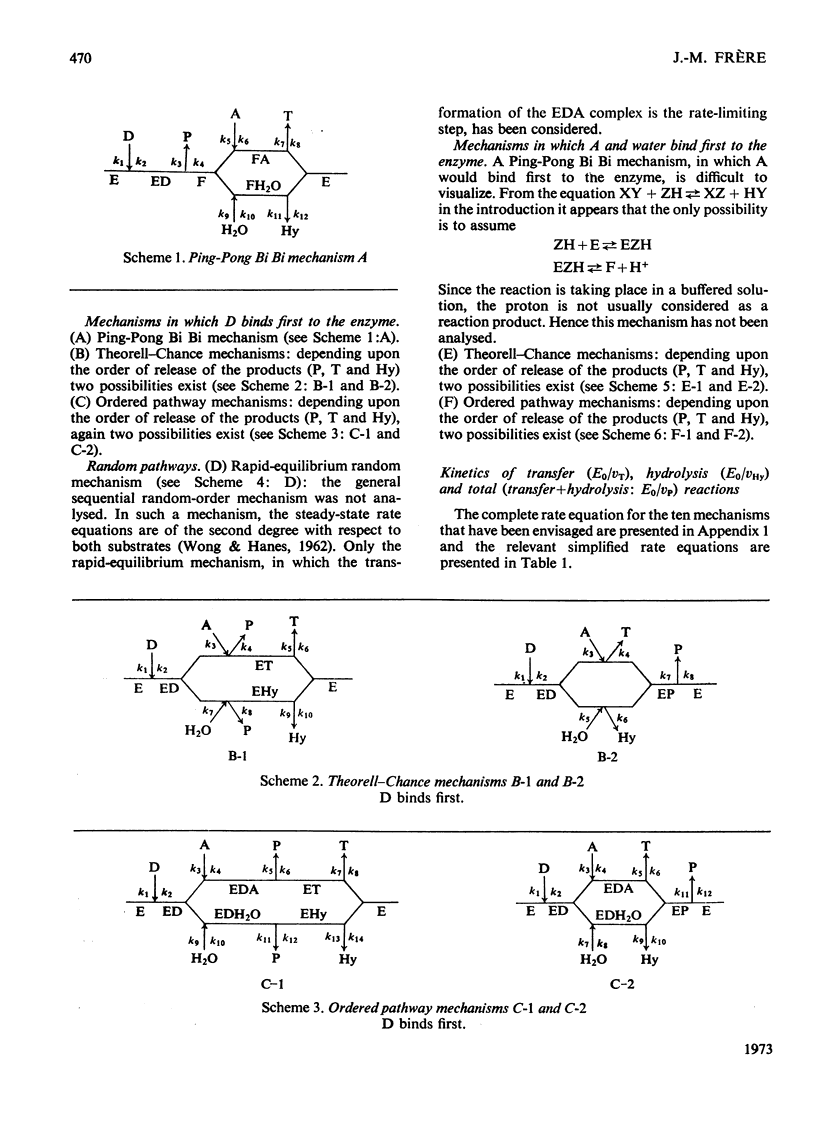
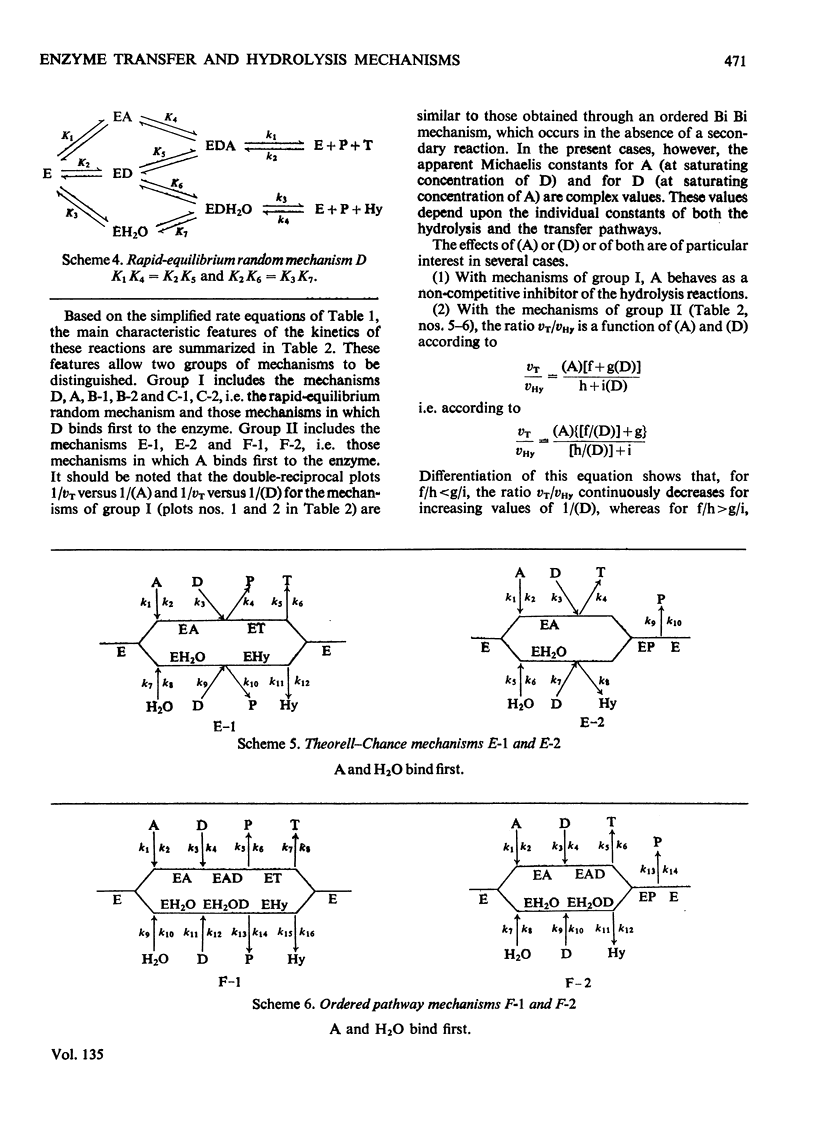
Abstract
The kinetic parameters of ten different enzymic mechanisms in which bimolecular transfer reactions occur concomitantly with the hydrolysis of the donor molecule have been studied. The usefulness of these parameters for making a choice of mechanism is discussed. The analysis has been extended to the use of alternative substrates in bimolecular transfer reactions that proceed without the hydrolysis of the donor molecule.
Full text
PDF










Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- CLELAND W. W. The kinetics of enzyme-catalyzed reactions with two or more substrates or products. I. Nomenclature and rate equations. Biochim Biophys Acta. 1963 Jan 8;67:104–137. doi: 10.1016/0006-3002(63)91800-6. [DOI] [PubMed] [Google Scholar]
- FROMM H. J. THE USE OF ALTERNATIVE SUBSTRATES IN STUDYING ENZYMIC MECHANISMS INVOLVING TWO SUBSTRATES. Biochim Biophys Acta. 1964 Mar 9;81:413–417. doi: 10.1016/0926-6569(64)90126-9. [DOI] [PubMed] [Google Scholar]
- Frère J. M., Ghuysen J. M., Perkins H. R., Nieto M. Kinetics of concomitant transfer and hydrolysis reactions catalysed by the exocellular DD-carboxypeptidase-transpeptidase of streptomyces R61. Biochem J. 1973 Nov;135(3):483–492. doi: 10.1042/bj1350483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghuysen J. M., Leyh-Bouille M., Campbell J. N., Moreno R., Frére J. M., Duez C., Nieto M., Perkins H. R. Structure of the wall peptidoglycan of Streptomyces R39 and the specificity profile of its exocellular DD-carboxypeptidase--transpeptidase for peptide acceptors. Biochemistry. 1973 Mar 27;12(7):1243–1251. doi: 10.1021/bi00731a001. [DOI] [PubMed] [Google Scholar]
- Pettersson G. RElationships between rapid equilibrium conditions and linearization of the reciprocal rate equation for the sequential random two-substrate enzyme mechanism. Acta Chem Scand. 1969;23(9):3243–3247. doi: 10.3891/acta.chem.scand.23-3243. [DOI] [PubMed] [Google Scholar]
- WONG J. T., HANES C. S. Kinetic formulations for enzymic reactions involving two substrates. Can J Biochem Physiol. 1962 Jun;40:763–804. [PubMed] [Google Scholar]


