Abstract
Purpose
Previous research has documented cognitive deficits in survivors of papillary thyroid carcinoma (PTC).Our longitudinal study with large sample size, aims to assess the impact of menopausal status on cognitive function, elucidate related factors of cognitive impairment, and chart the trajectory of cognitive changes over time in female PTC patients.
Methods
In this longitudinal study, we administered the Montreal Cognitive Assessment (MoCA) to 322 female PTC patients over 40 years old, before surgery and at 3 and 6 months after surgery. Propensity score matching (PSM) was used to adjust for baseline disparities, leading to a final analysis of 228 patients (114 premenopausal and 114 postmenopausal). Cognitive scores were compared between groups using the Mann–Whitney U test, and univariate and multivariate logistic regression analyses were performed to identify independent predictors of cognitive impairment.
Results
Postmenopausal women demonstrated a significantly higher susceptibility to impairment in delayed recall (p = 0.004) and global cognition (p = 0.006) when compared with premenopausal women. Multivariate analysis identified menopause (p < 0.001) and rural residence (p = 0.001) as independent risk factors for cognitive impairment. Furthermore, a gradual improvement in cognitive function over time was observed across both groups over the course of the study.
Conclusions
In female PTC patients, postmenopausal status and rural residence are significant risk factors for cognitive impairment. Postmenopausal women are more susceptible to cognitive deficits than premenopausal women in delayed recall and global cognition.Although cognitive function improves over time, it is crucial for physicians to closely monitor and support these patients to optimize their prognosis.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12905-024-03503-3.
Keywords: Papillary thyroid cancer, Menopausal women, Cognitive function, Longitudinal study, Propensity score matching
Introduction
Papillary thyroid carcinoma (PTC) is recognized as the predominant histological subtype of thyroid cancer (TC), accounting for over 90% of cases and exhibiting a rising incidence trend [1, 2]. In China, TC has become a prevalent malignancy, posing a significant threat to women’s health [3]. Despite the generally favorable prognosis after standard treatment, with a 10-year survival rate exceeding 90%, survivors of TC frequently contend with a diminished quality of life (QOL) [4], a factor partly attributed to cognitive impairments that may arise during treatment.
Cancer-related cognitive impairment (CRCI) is a complex neurocognitive condition arising from the interplay between a cancer diagnosis and its treatment modalities. This condition can lead to deficits in multiple cognitive domains, including learning, memory, attention, executive function, and processing speed [5, 6]. A meta-analysis by Saeed et al. [7] revealed that TC survivors experience significant impairments in attention, concentration, and language abilities, as well as slower processing speeds, compared to individuals without cancer. Furthermore, issues with attention and working memory have been shown to negatively impact the daily functioning of these patients [8].
Postmenopausal women experience significant physiological and hormonal changes that can disrupt daily life, characterized by common symptoms such as hot flashes and insomnia, as well as emotional responses like anxiety and depression [9]. Cognitive decline, including memory loss, is also frequently observed in this demographic. Despite these challenges, there is a notable gap in research focused on cognitive function in menopausal women with PTC. This longitudinal study aims to bridge this gap by investigating the impact of menopause on cognitive function in female PTC patients, identifying risk factors for cognitive decline in this group, and monitoring cognitive function progression over time.
Materials and methods
Study design and participants
This longitudinal study enrolled patients who underwent thyroid surgery between August 1 and November 1, 2023, at the First Affiliated Hospital of China Medical University. Participants completed a self-reporting questionnaire to provide baseline data on health and cognitive parameters prior to surgery. Cognitive function was further assessed through neuropsychological tests administered by two psychologists in a quiet, controlled setting. Postoperatively, patients were followed up, and the questionnaires were readministered at 3 and 6 months after the operation.
According to the 2023 Chinese guidelines on menopause symptom management and menopausal hormone therapy, menopause is defined as the cessation of menstruation for 12 consecutive months in women over 40 years old, excluding cases of pregnancy or other health conditions that may cause amenorrhea [10–12]. Pre-menopausal is defined as the period when a woman has not experienced 12 consecutive months of amenorrhea, excluding cases of pathological amenorrhea.
Given these criteria,this study specifically targets women over 40 years old who are not pregnant and do not have other health conditions that may cause amenorrhea. Enrollment Criteria: (1) Postoperative pathological diagnosis of PTC, (2) Consent to participate in this study. Exclusion Criteria: (1) Prior thyroid surgery, (2) History of strokes, seizures, anxiety, or depression, (3) Pregnancy or other malignancies, or patients receiving hormone replacement therapy or other health conditions that may cause amenorrhea, (4) Incomplete patient information, (5) Diagnosis of follicular thyroid carcinoma (FTC) or medullary thyroid carcinoma (MTC), (6) Age ≤ 40 years old. (7) Male gender.
We systematically collected demographic and clinical data from electronic medical records, including age, comorbidities, education level, marital status, employment status, income status, residence, family cancer history, fine needle aspiration biopsy(FNA), menopausal status, and thyroid function levels of free triiodothyronine, free thyroxine, and thyroid-stimulating hormone. we assessed the history of strokes, seizures, anxiety, or depression by consulting the diagnostic records within the patients’ electronic medical histories. To ensure the accuracy and reliability of our assessment, we had medical professionals review and verify these records.
This study was approved by the Institutional Review Board (IRB) of The First Hospital of China Medical University (IRB No. 2023-338-2), and informed consent was obtained from all participants, and this study adheres to the Declaration of Helsinki.
Instruments
The Montreal Cognitive Assessment (MoCA) Scale Beijing version [13] was employed to assess eight domains of cognitive function, including global cognition, visuospatial/executive, naming, attention, language, abstraction, delayed recall, and orientation [14, 15]. Validated through extensive long-term medical practice, the MoCA is recognized for its high sensitivity and reliability [16, 17]. The specific scoring criteria are detailed in Table 1.
Table 1.
Psychological questionnaire and scoring principles
| Test | Neuropsychological domains | Scoring methodology | Scoring principles | |
|---|---|---|---|---|
| MoCA | Visuospatial/Executive | |||
| Naming | ≥ 26 | Normal | ||
| Attention | Total score are 30 points | 18–25 | Mild cognitive impairment | |
| Language | 1 point is added if education level | 10–17 | Moderate cognitive impairment | |
| Abstraction | is less than 12 years | < 10 | Severe cognitive impairment | |
| Delayed recall | ||||
| Orientation | ||||
| Global cognitiona | ||||
Abbreviation: MoCA Montreal Cognitive Assessment
aGlobal cognition is the sum of scores, the higher the score is, the better the global cognitive function is
Statistical analysis
Continuous variables are presented as mean ± standard deviation (range), and categorical variables are reported as frequencies (percentages). Group differences were assessed by Chi-square test for categorical variables and the Mann–Whitney U test for continuous variables. To control for potential confounders and selection bias, propensity score matching (PSM) was implemented, univariate and multivariate logistic regression analyses were used to explore the factors affecting cognition. All statistical analyses were conducted by SPSS software (version 27.0; IBM SPSS Inc., Chicago, United States) and GraphPad Prism 8. A p-value of less than 0.05 was considered statistically significant.
Results
Baseline demographic and clinicopathologic characteristics
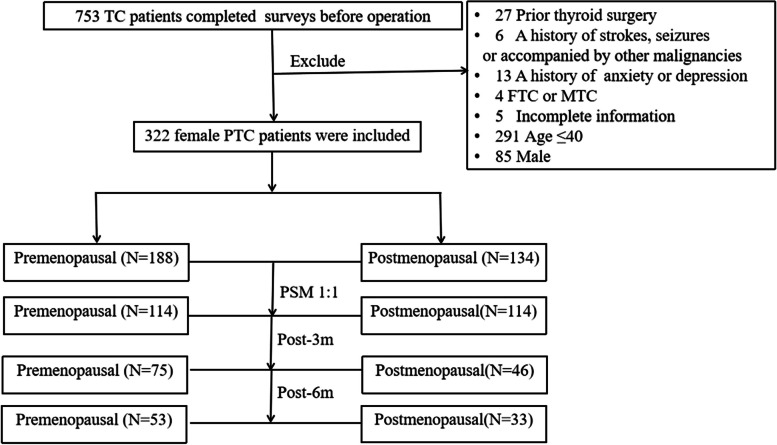
In our study, a total of 753 patients were initially considered. After applying our eligibility criteria and excluding those who did not meet the requirements, we selected 322 female PTC patients over 40 years old for analysis, as shown in Fig. 1. Among these, 134 (41.6%) were postmenopausal with a mean age of 55.1 ± 5.6 years old, ranging from 43 to 77 years old. The premenopausal group comprised 188 (58.4%) participants with a mean age of 46.1 ± 4.0 years old, ranging from 41 to 66 years old. A significant age difference between the two groups was observed (p < 0.001).
Fig. 1.
Flow chart of the studying participants’ selection. Abbreviations:TC, Thyroid cancer; PTC, Papillary thyroid carcinoma; FTC, Follicular thyroid cancers; MTC, Medullary thyroid cancers; m,month; PSM, Propensity Score Matching
Baseline characteristics, including age, comorbidity, education level, employment status, and income status, were compared and showed significant differences (p < 0.05), as detailed in Table 2. To further control for potential confounders and isolate the impact of menopause on cognitive function, we conducted a 1:1 PSM based on comorbidities, education level, employment status, and income status. Post-PSM, the analysis involved an equal number of postmenopausal and premenopausal PTC survivors, with 114 participants in each group, facilitating a more robust comparison. The adjustments made and the resulting group distributions are presented in Table 2 and Fig. 1.
Table 2.
Baseline characteristics before and after propensity score matching
| Characteristics | Before propensity score matching | After propensity score matching | ||||||
|---|---|---|---|---|---|---|---|---|
| Total (n = 322),% | Postmenopausal (n = 134),% | Premenopausal (n = 188),% | p-Value | Postmenopausal (n = 114),% | Premenopausal (n = 114),% | p-Value | ||
| Age,y, mean ± SD (range, y) | 49.8 ± 6.5(41–77) | 55.1 ± 5.6(43–77) | 46.1 ± 4.0(41–66) | < 0.001 | 54.6 ± 5.6(43–77) | 46.9 ± 4.4(41–66) | < 0.001 | |
| Comorbidities | 0.004 | 1.000 | ||||||
| No | 247(76.7%) | 92(68.7%) | 155(82.4%) | 92(80.7%) | 92(80.7%) | |||
| Yes | 75(23.3%) | 42(31.3%) | 33(17.6%) | 22(19.3%) | 22(19.3%) | |||
| Education level | 0.005 | 0.508 | ||||||
| Elementary/Junior high | 149(46.3%) | 75(56.0%) | 74(39.4%) | 56(49.1%) | 61(53.5%) | |||
| Senior high/College and above | 173(53.7%) | 59(44.0%) | 114(60.6%) | 58(50.9%) | 53(46.5%) | |||
| Marital status | 0.294 | 0.084 | ||||||
| Single,divorced,or Widowed | 34(10.6%) | 17(12.7%) | 17(9.0%) | 16(14.0%) | 8(7.0%) | |||
| Married | 288(89.4%) | 117(87.3%) | 171(91.0%) | 98(86.0%) | 106(93.0%) | |||
| Employment status | < 0.001 | 1.000 | ||||||
| Unemployed | 200(62.1%) | 107(79.9%) | 93(49.5%) | 87(76.3%) | 87(76.3%) | |||
| Employed | 122(37.9%) | 27(20.1%) | 95(50.5%) | 27(23.7%) | 27(23.7%) | |||
| Income status (¥) | 0.006 | 1.000 | ||||||
| < 4000 | 195(60.6%) | 93(69.4%) | 102(54.3%) | 75(65.8%) | 75(65.8%) | |||
| ≥ 4000 | 127(39.4%) | 41(30.6%) | 86(45.7%) | 39(34.2%) | 39(34.2%) | |||
| Residence | 0.541 | 0.201 | ||||||
| Rural | 95(29.5%) | 42(31.3%) | 53(28.2%) | 32(28.1%) | 41(36.0%) | |||
| Urban | 227(70.5%) | 92(68.7%) | 135(71.8%) | 82(71.9%) | 73(64.0%) | |||
| Family history of cancer | 0.191 | 0.164 | ||||||
| No | 272(84.5%) | 109(81.3%) | 163(86.7%) | 96(84.2%) | 103(90.4%) | |||
| Yes | 50(15.5%) | 25(18.7%) | 25(13.3%) | 18(15.8%) | 11(9.6%) | |||
| FNA | 0.447 | 0.888 | ||||||
| No | 103(32.0%) | 46(34.3%) | 57(30.3%) | 37(32.5%) | 38(33.3%) | |||
| Yes | 219(68.0%) | 88(65.7%) | 131(69.7%) | 77(67.5%) | 76(66.7%) | |||
| Thyroidectomy | 0.532 | 1.000 | ||||||
| less than total thyroidectomy | 182(56.5%) | 73(54.5%) | 109(58.0%) | 62(54.4%) | 62(54.4%) | |||
| total thyroidectomy | 140(43.5%) | 61(45.5%) | 79(42.0%) | 52(45.6%) | 52(45.6%) | |||
| Lymph node dissection | 0.157 | 0.120 | ||||||
| Central | 300(93.2%) | 128(95.5%) | 172(91.5%) | 109(95.6%) | 103(90.4%) | |||
| Lateral | 22(6.8%) | 6(4.5%) | 16(8.5%) | 5(4.4%) | 11(9.6%) | |||
| Surgical approach | 0.619 | 0.472 | ||||||
| Remote-access | 32(9.9%) | 12(9.0%) | 20(10.6%) | 11(9.6%) | 8(7.0%) | |||
| Conventional transcervical | 290(90.1%) | 122(91.0%) | 168(89.4%) | 103(90.4%) | 106(93.0%) | |||
| T classification | 0.732 | 0.757 | ||||||
| T1 | 306(95.0%) | 128(95.5%) | 178(94.7%) | 109(95.6%) | 108(94.7%) | |||
| T2 | 16(5.0%) | 6(4.5%) | 10(5.3%) | 5(4.4%) | 6(5.3%) | |||
| N classification | 0.174 | 0.021 | ||||||
| N0 | 210(65.2%) | 95(70.9%) | 115(61.2%) | 83(72.8%) | 63(55.3%) | |||
| N1a | 92(28.6%) | 33(24.6%) | 59(31.4%) | 26(22.8%) | 41(36.0%) | |||
| N1b | 20(6.2%) | 6(4.5%) | 14(7.4%) | 5(4.4%) | 10(8.8%) | |||
| Stage | 0.006 | 0.055 | ||||||
| I | 311(96.6%) | 125(93.3%) | 186(98.9%) | 108(94.7%) | 113(99.1%) | |||
| II | 11(3.4%) | 9(6.7%) | 2(1.1%) | 6(5.3%) | 1(0.9%) | |||
| Risk | 0.900 | 0.531 | ||||||
| low-risk | 251(78.0%) | 106(79.1%) | 145(77.1%) | 92(80.7%) | 85(74.6%) | |||
| Middle-risk | 55(17.1%) | 22(16.4%) | 33(17.6%) | 17(14.9%) | 23(20.2%) | |||
| High-risk | 16(5.0%) | 6(4.5%) | 10(5.3%) | 5(4.4%) | 6(5.3%) | |||
Comorbidities include Hypertension, coronary heart disease, diabetes
Italics indicate statistical significance
Abbreviations: y years, FNA fine needle aspiration biopsy
Univariate and multivariate logistic regression analysis of factors associated with cognitive impairment after PSM
To identify the key determinants of cognitive function, we conducted univariate and multivariate logistic regression analyses, as detailed in Table 3. The univariate analysis revealed several significant predictors, including age (p = 0.003), menopausal status (p = 0.002), education level (p = 0.003), employment status (p = 0.009), and residence (p = 0.003).When these predictors were integrated into a multivariate logistic regression model, it became apparent that menopausal status (p < 0.001) and place of residence (p = 0.001) were the independent risk factors for cognitive impairment.
Table 3.
Univariable and multivariable logistic regression analysis of the factors of cognitive impairment at baseline after propensity score matching
| Characteristics | Univariable analysis | Multivariable analysis | |||||
|---|---|---|---|---|---|---|---|
| β | HR(95%CI) | P-Value | β | HR(95%CI) | P-Value | ||
| Age,y | 0.087 | 1.091(1.030–1.155) | 0.003 | ||||
| Menopausal status | No | Reference | Reference | ||||
| Yes | 1.009 | 2.742(1.435–5.239) | 0.002 | 1.155 | 3.174(1.625–6.201) | < 0.001 | |
| Comorbidities | No | Reference | |||||
| Yes | -0.089 | 0.915(0.427–1.962) | 0.915 | ||||
| Education level | Elementary/Junior high | Reference | |||||
| Senior high/College and above | -0.971 | 0.379(0.200–0.719) | 0.003 | ||||
| Marital status | Single, divorced, or Widowed | Reference | |||||
| Married | 0.080 | 1.083(0.407–2.883) | 0.873 | ||||
| Employment status | Unemployed | Reference | |||||
| Employed | -0.885 | 0.413(0.212–0.805) | 0.009 | ||||
| Income status (¥) | < 4000 | Reference | |||||
| ≥ 4000 | -0.371 | 0.690(0.368–1.295) | 0.248 | ||||
| Residence | Rural | Reference | Reference | ||||
| Urban | -1.232 | 0.292(0.130–0.656) | 0.003 | -1.391 | 0.249(0.108–0.572) | 0.001 | |
| Family history of cancer | No | Reference | |||||
| Yes | -0.029 | 0.972(0.391–2.417) | 0.951 | ||||
| FNA | No | Reference | |||||
| Yes | -0.085 | 0.919(0.477–1.769) | 0.800 | ||||
| Thyroidectomy | less than total thyroidectomy | Reference | |||||
| total thyroidectomy | 0.062 | 1.064(0.576–1.969) | 0.843 | ||||
| Lymph node dissection | Central | Reference | |||||
| Lateral | -0.077 | 0.926(0.286–2.999) | 0.898 | ||||
| Surgical approach | Remote-access | Reference | |||||
| Conventional transcervical | 0.437 | 1.548(0.558–4.291) | 0.401 | ||||
| T classification | T1 | Reference | |||||
| T2 | -0.199 | 0.819(0.210–3.203) | 0.774 | ||||
| N classification | N0 | Reference | |||||
| N1a | -0.723 | 0.485(0.252–0.935) | 0.031 | ||||
| N1b | -0.427 | 0.653(0.193–2.202) | 0.492 | ||||
| Stage | I | Reference | |||||
| II | -0.262 | 0.769(0.145–4.082) | 0.758 | ||||
| Risk | low-risk | Reference | |||||
| Middle-risk | -0.566 | 0.568(0.267–1.205) | 0.141 | ||||
| High-risk | -0.316 | 0.729(0.184–2.882) | 0.652 | ||||
| FT3a(pmol/L) | -0.396 | 0.673(0.367–1.236) | 0.202 | ||||
| FT4a(pmol/L) | -0.054 | 0.948(0.779–1.154) | 0.593 | ||||
| TSHa(mIU/L) | 0.002 | 1.002(0.763–1.317) | 0.987 | ||||
Comorbidities includ Hypertension, coronary heart disease, diabetes
Italics indicate statistical significance
Abbreviations: y years, FNA fine needle aspiration biopsy, FT3 free triiodothyronine, FT4 free thyroxine, TSH Thyroid Stimulating Hormone
aThe analysis was carried out in 197 samples
Cognitive score comparisons before surgery: postmenopausal vs. premenopausal women
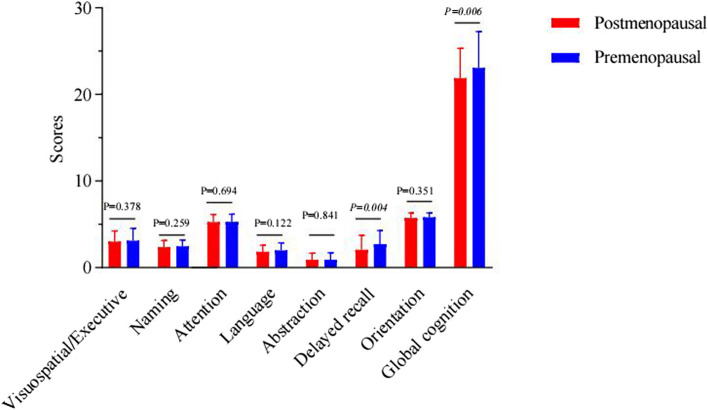
Higher MoCA scores indicate better cognitive performance. Our study identified significant differences in delayed recall (p = 0.004) and global cognition (p = 0.006), with postmenopausal women scoring lower, as detailed in Supplementary Table 1 and illustrated in Fig. 2.
Fig. 2.
Comparisons of cognitive scores in postmenopausal and premenopausal women before operation
Variations in cognitive scores at 3 and 6 months postoperatively
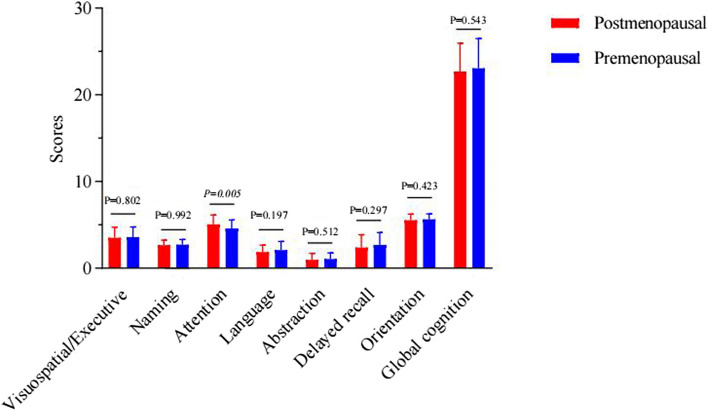
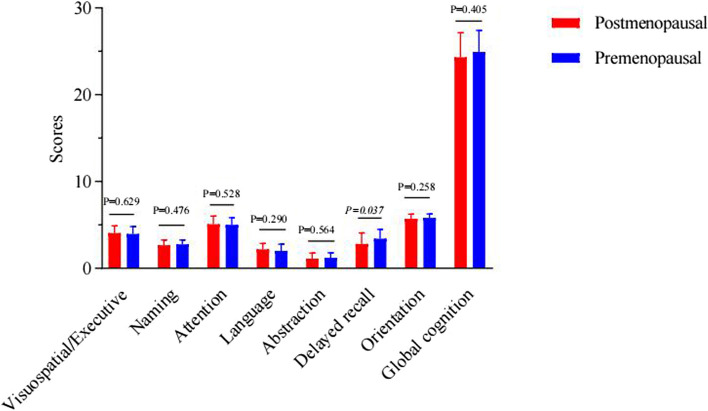
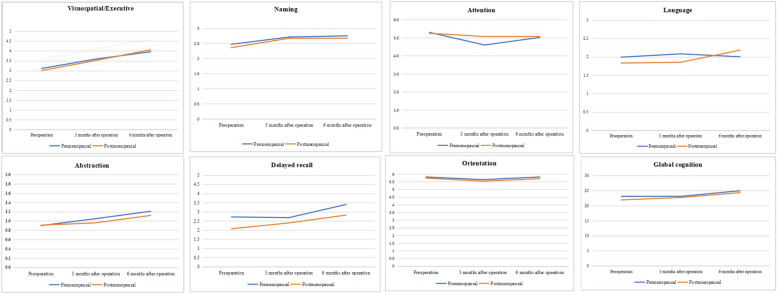
At the 3-month postoperative mark, a significant difference emerged in the attention scores, with postmenopausal women demonstrating superior performance (p = 0.005), as shown in Supplementary Table 2 and Fig. 3. At the 6-month postoperative mark, a significant reversal was noted in delayed recall scores, where premenopausal women outperformed postmenopausal women (p = 0.037), as documented in Supplementary Table 3 and Fig. 4. Furthermore, an analysis of the cognitive function trajectories for both groups over time revealed a steady improvement, as delineated in Supplementary Table 4 and Fig. 5.
Fig. 3.
Comparisons of cognitive scores in postmenopausal and premenopausal women at 3 months after operation
Fig. 4.
Comparisons of cognitive scores in postmenopausal and premenopausal women at 6 months after operation
Fig. 5.
Changes of cognition scores in postmenopausal and premenopausal women over time
Discussion
In this large-scale longitudinal study, we identified that postmenopausal PTC women and who reside in rural areas are risk factors for cognitive impairment. Postmenopausal women performed poorer in global cognition and delayed recall. Nonetheless, a consistent trend of gradual improvement in cognitive function was observed throughout the study period.
Although most TC patients have excellent prognoses, some studies have identified that they are vulnerable to cognitive dysfunction. Jaracz et al. found that DTC (Differentiated thyroid cancer) patients receiving thyroid-stimulating hormone (TSH) inhibition treatment performed worse in executive functions, psychomotor speed and attention, highlighting neuropsychological impairment in DTC patients [18]. Similarly, Samuels et al. discovered that women treated with euthyrox (TSH suppression/replacement treatment) experienced significant impairments in overall health and mood but not in cognition [19]. By comparing 90 healthy individuals with 90 female PTC patients, Jung et al. identified attention and working memory as the most vulnerable cognitive domains [8]. Furthermore, they found that up to 78% of newly diagnosed thyroid cancer patients met both the Global Deficit Score and the International Cancer criteria; older age, depressive symptoms, hypothyroidism status, and low education level were significantly associated with worse cognitive function [20]. Additionally, poor preoperative cognitive function significantly increased the length of hospital stay [21]. As presented in Supplementary Table 5, the majority of previous studies have been limited by their cross-sectional nature and small sample sizes. In contrast, our investigation leverages larger sample size and employs a longitudinal design, which allows us to not only identify risk factors associated with cognitive impairment but also track cognitive function trajectories over time. Additionally, the incorporation of PSM in our study further enhances the comparability of our findings, thereby strengthening the validity of our conclusions.
This study indicates that postmenopausal status is significantly associated with poorer cognitive performance in female PTC patients over 40 years old. This finding aligns with previous research in other cancer populations, where significant alterations in executive function, working memory, and attention have been observed during treatment in postmenopausal breast cancer patients [22]. Moreover, a decline in global cognitive function, memory, language and communication, and sensorimotor function has been noted following chemotherapy [23]. This aligns with the findings of Yamamoto et al., who conducted a questionnaire survey involving 876 breast cancer patients experiencing cognitive impairment. They discovered that fewer family members, a history of breast cancer surgery, severe menopausal symptoms, and psychological distress were significantly associated with cognitive difficulties [24]. High cognitive fatigue has also been linked to severe menopausal and depressive symptoms, negatively impacting the quality of life [25]. However, there is a scarcity of research focusing on the impact of menopausal status on cognition in TC patients. Given that both thyroid and breast cancer are prevalent in women, and considering the work and life changes, along with physiological and psychological challenges faced by postmenopausal women, they may be more susceptible to the side effects of TC treatment [26]. Therefore, prioritizing the mental health of postmenopausal women is imperative.
Estrogen plays a pivotal role in neuroprotection, enhancing neuronal connectivity and synaptic plasticity, which are crucial for preserving cognitive function. Study has demonstrated that menopause is a dynamic neurological transition that significantly affects brain structure, connectivity, and metabolic profiles [27]. Observations from longitudinal studies have noted a progressive decline in cognitive domains such as learning, memory, and attention/working memory as women transition from premenopause to postmenopause [28]. The decline is partly attributable to the marked reduction in ovarian function and estrogen levels [29]. Furthermore, the prevalence of age-related cardiovascular and cerebrovascular diseases, including diabetes and hypertension, increases with advancing age. The hippocampus, a region of the brain critical for learning, memory, and cognitive function, is disproportionately susceptible to hypoxic-ischemic injury owing to alterations in cerebral blood flow, resulting in a pronounced increase in cognitive impairment among the elderly [30]. Estrogen acts as a protective factor for cardiovascular health and cognitive function [31–33]. Consequently, the reduction in estrogen production that occurs in postmenopausal women can significantly exacerbate their risk of cognitive decline.
Our study revealed that residing in rural areas is a significant determinant of cognitive function, partly due to the fact that cognitive function is intricately linked to cultural, educational, and social knowledge, as well as experiential factors. Individuals in rural settings frequently encounter lower levels of education and have more limited access to disease-related information compared to their urban counterparts. Therefore, in the clinical context, it is crucial to augment informational support and extend empathetic care to comprehensively enhance the quality of life for these patients.
We observed a gradual improvement in cognitive function scores over time, supporting previous findings that DTC survivors undergoing TSH suppression therapy initially experience short-term memory impairment, attention deficits, word selection anomia, and depressive symptoms, which tend to ameliorate with time except for word selection anomia [34]. Enhanced comprehension and acceptance of their condition, coupled with reduced anxiety and psychological distress, may be associated with the observed changes, similar variations were noted in a comprehensive five-year follow-up study [35]. Furthermore, postoperative complications, including hypoparathyroidism [36], recurrent laryngeal nerve injury [37], scarring issues [38] contribute to immediate fluctuations in cognitive performance. Additionally, resuming regular work and daily activities can further promote the improvement of cognitive function.
Limitations
There are several limitations. As a single-center study, our findings may not be generalizable to other populations or healthcare settings. Additionally, the determination of menopausal status relied on self-report, without corroboration from hormonal or imaging studies, potentially introducing subjective bias. We also did not account for changes in menopausal status during the follow-up period, and baseline variables such as body mass index (BMI), smoking, and alcohol consumption were not systematically collected, which could impact the analysis’s comprehensiveness. The sample size was determined by practical availability rather than a calculated estimation. Furthermore, some patients were loss to follow-up during the postoperative period. In future studies, we plan to address these limitations by incorporating multi-center data, employing objective measures to define menopausal status, collecting a broader range of baseline characteristics. We will also implement scientific follow-up methods to minimize patient loss to follow-up. Moreover, we plan to utilize advanced molecular techniques such as gene expression analysis, proteomics, and metabolomics to uncover the molecular mechanisms underlying cognitive health management in this patient population.
Conclusions
Female PTC patients who are postmenopausal and reside in rural areas are at an elevated risk of cognitive deficits. Compared to premenopausal women, postmenopausal individuals exhibit greater susceptibility to impairments in delayed recall and overall cognitive function. Despite these challenges, there is evidence that cognitive function tends to improve over time. Therefore, it is crucial for clinicians to prioritize these patients by providing robust informational support and tailored nursing interventions to optimize their overall prognosis.
Supplementary Information
Acknowledgements
Not applicable.
Authors’ contributions
All authors contributed to the study conception and design. Material preparation were performed by Zhiqiang Gui, Yang Shi, data collection were performed by Chun Xu,Qingshu Wu and data analysis were performed by Yuenan Zheng, Jie Zhao.The first draft of the manuscript was written by Yuenan Zheng, Jie Zhao. Manuscript polishing and revision were performed by Hao Zhang, Zhihong Wang and Liang He. All authors commented on previous versions of the manuscript. Lili zhu provided tremendous help during the revision of the article. All authors read and approved the final manuscript.
Funding
The Natural Science Foundation of Liaoning Province (Grant Number JYTMS20230080).
Natural Science Foundation of Liaoning Province (Grant Number 2023010724-JH3/107).
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
This study was approved by the Institutional Review Board (IRB) of The First Hospital of China Medical University (IRB No. 2023-338-2).The study was conducted in strict adherence to the relevant guidelines and regulations, and in accordance with the principles of the Helsinki Declaration. Written or oral informed consent was obtained from all participants.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Zhihong Wang, Hao Zhang and Liang He as the corresponding authors contributed to work equally.
Contributor Information
Zhihong Wang, Email: wangzhihongcmu@163.com.
Hao Zhang, Email: haozhang@cmu.edu.cn.
Liang He, Email: hl_31@163.com.
References
- 1.Megwalu UC, Moon PK. Thyroid cancer incidence and mortality trends in the United States: 2000–2018. Thyroid. 2022;32:560–70. 10.1089/thy.2021.0662. [DOI] [PubMed] [Google Scholar]
- 2.Pizzato M, et al. The epidemiological landscape of thyroid cancer worldwide: GLOBOCAN estimates for incidence and mortality rates in 2020. Lancet Diabetes Endocrinol. 2022;10:264–72. 10.1016/s2213-8587(22)00035-3. [DOI] [PubMed] [Google Scholar]
- 3.Zheng RS, et al. Cancer incidence and mortality in China, 2022. Zhonghua Zhong Liu Za Zhi. 2024;46:221–31. 10.3760/cma.j.cn112152-20240119-00035. [DOI] [PubMed] [Google Scholar]
- 4.Oerlemans S, et al. Self-perceived cognitive functioning and quality of life among cancer survivors: results from the PROFILES registry. J Cancer Surviv. 2022;16:303–13. 10.1007/s11764-021-01023-9. [DOI] [PubMed] [Google Scholar]
- 5.Lomeli N, Lepe J, Gupta K, Bota DA. Cognitive complications of cancer and cancer-related treatments - novel paradigms. Neurosci Lett. 2021;749:135720. 10.1016/j.neulet.2021.135720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wefel JS, Kesler SR, Noll KR, Schagen SB. Clinical characteristics, pathophysiology, and management of noncentral nervous system cancer-related cognitive impairment in adults. CA Cancer J Clin. 2015;65:123–38. 10.3322/caac.21258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Saeed O, et al. Cognitive functioning in thyroid cancer survivors: a systematic review and meta-analysis. J Cancer Surviv. 2019;13:231–43. 10.1007/s11764-019-00745-1. [DOI] [PubMed] [Google Scholar]
- 8.Jung MS, Visovatti M. Post-treatment cognitive dysfunction in women treated with thyroidectomy for papillary thyroid carcinoma. Support Care Cancer. 2017;25:915–23. 10.1007/s00520-016-3481-y. [DOI] [PubMed] [Google Scholar]
- 9.Metcalf CA, Duffy KA, Page CE, Novick AM. Cognitive problems in perimenopause: a review of recent evidence. Curr Psychiatry Rep. 2023;25:501–11. 10.1007/s11920-023-01447-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Menopause Subgroup, Chinese Society of Obstetrics and Gynecology, Chinese Medical Association. The 2023 Chinese menopause symptom management and menopausal hormone therapy guidelines. Zhonghua Fu Chan Ke Za Zhi. 2023;58:4–21. 10.3760/cma.j.cn112141-20221118-00706. [DOI] [PubMed]
- 11.Nguyen PN, Nguyen VT. Evaluating clinical features in intracavitary uterine pathologies among Vietnamese women presenting with Peri-and postmenopausal bleeding: a bicentric observational descriptive analysis. J Midlife Health. 2022;13:225–32. 10.4103/jmh.jmh_81_22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nguyen PN, Nguyen VT. Additional value of Doppler ultrasound to B-mode ultrasound in assessing for uterine intracavitary pathologies among perimenopausal and postmenopausal bleeding women: a multicentre prospective observational study in Vietnam. J Ultrasound. 2023;26:459–69. 10.1007/s40477-022-00732-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yu J, Li J, Huang X. The Beijing version of the Montreal cognitive assessment as a brief screening tool for mild cognitive impairment: a community-based study. BMC Psychiatry. 2012;12:156. 10.1186/1471-244x-12-156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tian R, Guo Y, Ye P, Zhang C, Luo Y. The validation of the Beijing version of the Montreal cognitive assessment in Chinese patients undergoing hemodialysis. PLoS ONE. 2020;15:e0227073. 10.1371/journal.pone.0227073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sun R, Ge B, Wu S, Li H, Lin L. Optimal cut-off MoCA score for screening for mild cognitive impairment in elderly individuals in China: a systematic review and meta-analysis. Asian J Psychiatr. 2023;87:103691. 10.1016/j.ajp.2023.103691. [DOI] [PubMed] [Google Scholar]
- 16.Jia X, et al. A comparison of the Mini-Mental State Examination (MMSE) with the Montreal Cognitive Assessment (MoCA) for mild cognitive impairment screening in Chinese middle-aged and older population: a cross-sectional study. BMC Psychiatry. 2021;21:485. 10.1186/s12888-021-03495-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yang YM, et al. Trends in cognitive function assessed by a battery of neuropsychological tests after mild acute ischemic stroke. J Stroke Cerebrovasc Dis. 2020;29:104887. 10.1016/j.jstrokecerebrovasdis.2020.104887. [DOI] [PubMed] [Google Scholar]
- 18.Jaracz J, Kucharska A, Rajewska-Rager A, Lacka K. Cognitive functions and mood during chronic thyrotropin-suppressive therapy with L-thyroxine in patients with differentiated thyroid carcinoma. J Endocrinol Invest. 2012;35:760–5. 10.3275/8013. [DOI] [PubMed] [Google Scholar]
- 19.Samuels MH, et al. The effects of levothyroxine replacement or suppressive therapy on health status, mood, and cognition. J Clin Endocrinol Metab. 2014;99:843–51. 10.1210/jc.2013-3686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jung MS, et al. Cognitive impairment in women newly diagnosed with thyroid cancer before treatment. Support Care Cancer. 2022;30:8959–67. 10.1007/s00520-022-07299-6. [DOI] [PubMed] [Google Scholar]
- 21.Larrabee K, et al. Cognitive function and postoperative outcomes in patients with head and neck cancer. Laryngoscope. 2023;133:2999–3005. 10.1002/lary.30677. [DOI] [PubMed] [Google Scholar]
- 22.Bender CM, et al. Patterns of change in cognitive function with anastrozole therapy. Cancer. 2015;121:2627–36. 10.1002/cncr.29393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Merriman JD, et al. Trajectories of self-reported cognitive function in postmenopausal women during adjuvant systemic therapy for breast cancer. Psychooncology. 2017;26:44–52. 10.1002/pon.4009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yamamoto S, Masutani E, Arao H. Self-reported cognitive decline in Japanese patients with breast cancer treated with endocrine therapy. Breast Cancer. 2020;27:670–82. 10.1007/s12282-020-01062-7. [DOI] [PubMed] [Google Scholar]
- 25.Miura K, Ando S, Imai T. The association of cognitive fatigue with menopause, depressive symptoms, and quality of life in ambulatory breast cancer patients. Breast Cancer. 2016;23:407–14. 10.1007/s12282-014-0578-3. [DOI] [PubMed] [Google Scholar]
- 26.Uygur MM, Yoldemir T, Yavuz DG. Thyroid disease in the perimenopause and postmenopause period. Climacteric. 2018;21:542–8. 10.1080/13697137.2018.1514004. [DOI] [PubMed] [Google Scholar]
- 27.Mosconi L, et al. Menopause impacts human brain structure, connectivity, energy metabolism, and amyloid-beta deposition. Sci Rep. 2021;11:10867. 10.1038/s41598-021-90084-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Maki PM, et al. Cognitive changes during the menopausal transition: a longitudinal study in women with and without HIV. Menopause. 2021;28:360–8. 10.1097/gme.0000000000001725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Maki PM, Jaff NG. Brain fog in menopause: a health-care professional’s guide for decision-making and counseling on cognition. Climacteric. 2022;25:570–8. 10.1080/13697137.2022.2122792. [DOI] [PubMed] [Google Scholar]
- 30.Johnson AC. Hippocampal vascular supply and its role in vascular cognitive impairment. Stroke. 2023;54:673–85. 10.1161/strokeaha.122.038263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Meng Q, et al. Attenuation of estrogen and its receptors in the post-menopausal stage exacerbates dyslipidemia and leads to cognitive impairment. Mol Brain. 2023;16:80. 10.1186/s13041-023-01068-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ryczkowska K, Adach W, Janikowski K, Banach M, Bielecka-Dabrowa A. Menopause and women’s cardiovascular health: is it really an obvious relationship? Arch Med Sci. 2023;19:458–66. 10.5114/aoms/157308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Engler-Chiurazzi EB, Brown CM, Povroznik JM, Simpkins JW. Estrogens as neuroprotectants: estrogenic actions in the context of cognitive aging and brain injury. Prog Neurobiol. 2017;157:188–211. 10.1016/j.pneurobio.2015.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jin S, et al. Naming difficulties after thyroid stimulating hormone suppression therapy in patients with differentiated thyroid carcinoma: a prospective cohort study. Endocrine. 2019;65:327–37. 10.1007/s12020-019-01943-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kim BH, et al. Longitudinal changes in quality of life before and after thyroidectomy in patients with differentiated thyroid cancer. J Clin Endocrinol Metab. 2023. 10.1210/clinem/dgad748. [DOI] [PubMed] [Google Scholar]
- 36.Büttner M, et al. Quality of life in patients with hypoparathyroidism after treatment for thyroid cancer. J Clin Endocrinol Metab. 2020;105:dgaa597. 10.1210/clinem/dgaa597. [DOI] [PubMed] [Google Scholar]
- 37.Noto B, Asmus I, Schäfers M, Görlich D, Riemann B. Predictors of anxiety and depression in differentiated thyroid cancer survivors: results of a cross-sectional study. Thyroid. 2022;32:1077–85. 10.1089/thy.2022.0067. [DOI] [PubMed] [Google Scholar]
- 38.Kurumety SK, et al. Post-thyroidectomy neck appearance and impact on quality of life in thyroid cancer survivors. Surgery. 2019;165:1217–21. 10.1016/j.surg.2019.03.006. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.