Abstract
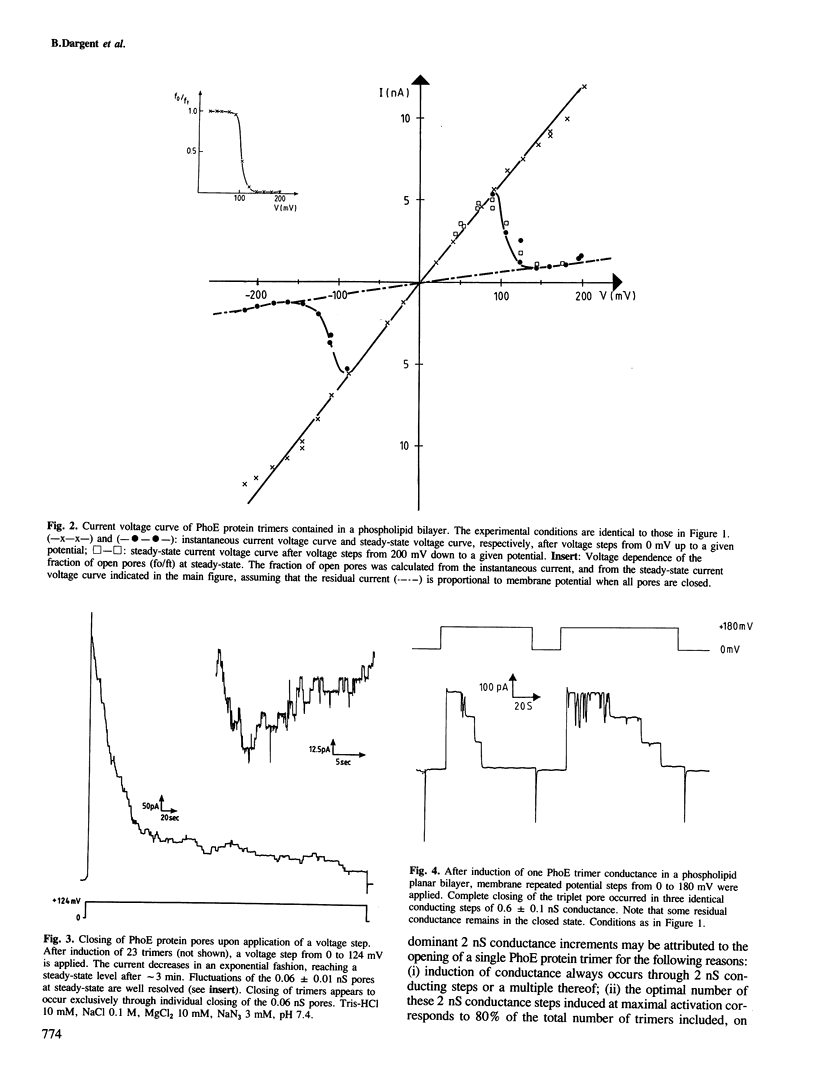
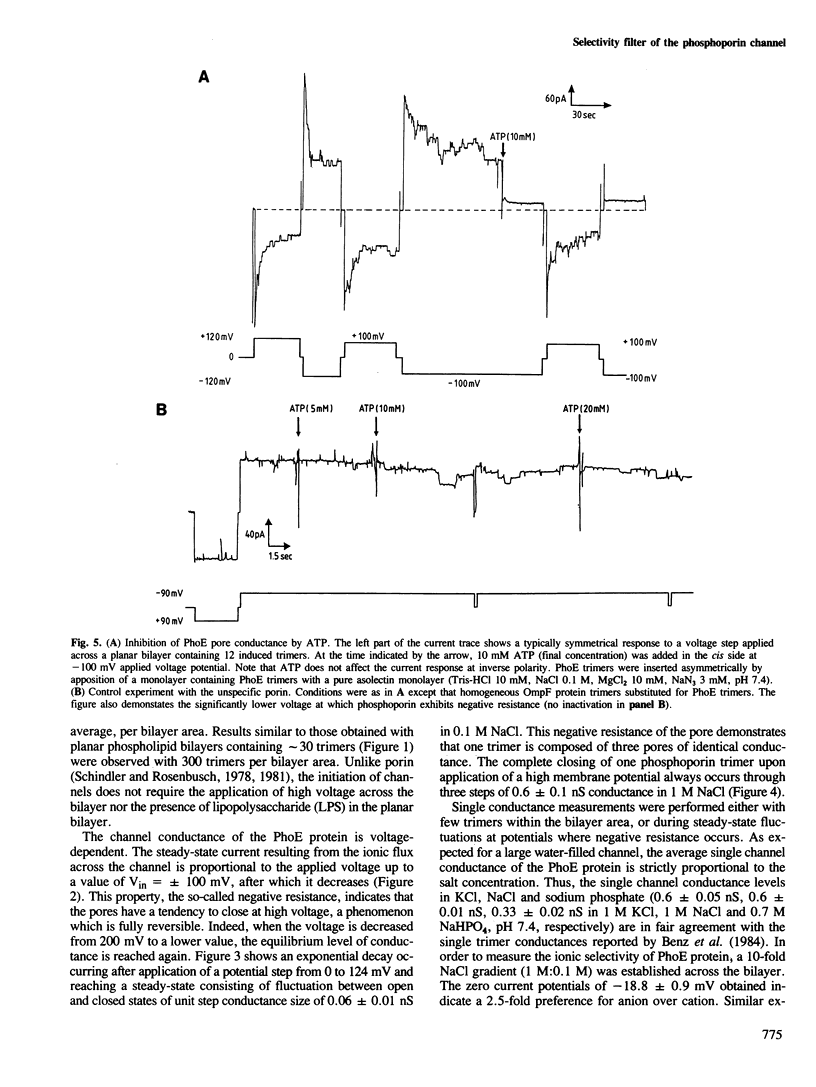
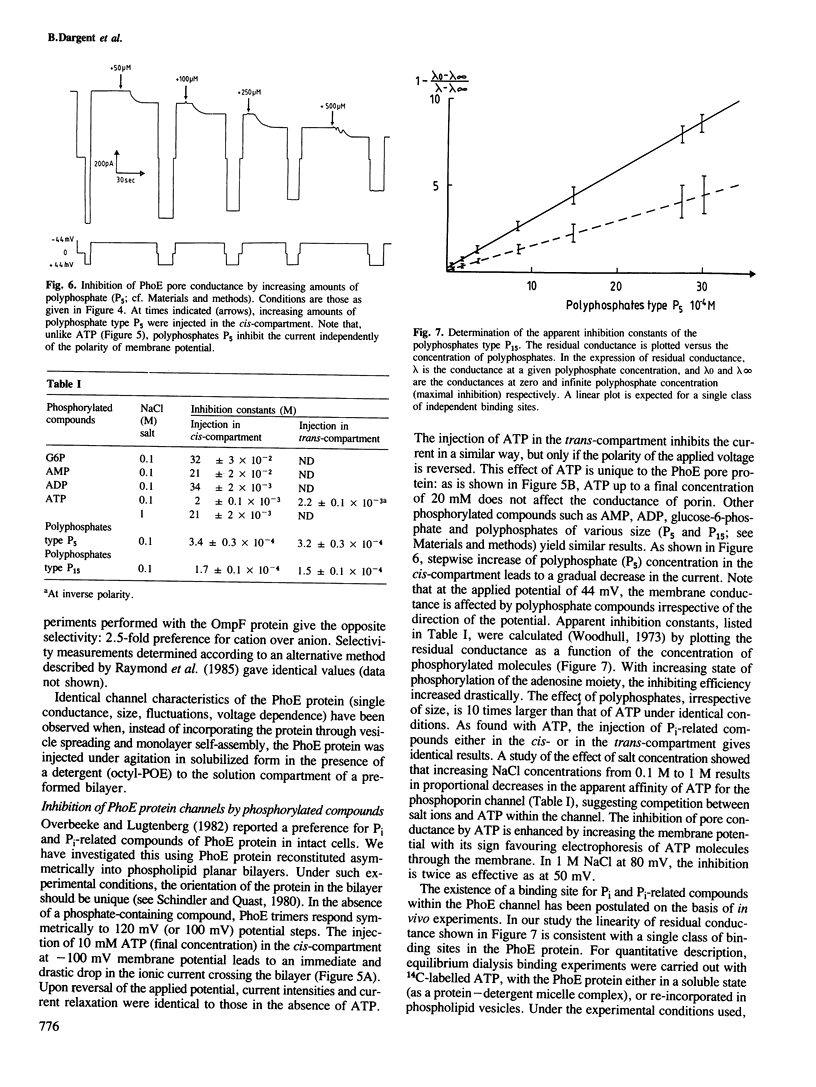
Phosphoporin, an Escherichia coli outer membrane-spanning protein re-incorporated in phospholipid planar bilayers generates aqueous channels similar to those of matrix porin. One phosphoporin trimer contains three pores which are induced simultaneously but fluctuate separately between open and closed states. Membrane potential shifts this two-state equilibrium in favour of closed channels. This negative resistance occurs at lower potentials than with matrix porin channels. The phosphoporin channel is poorly anion selective for small solutes. Polyphosphates and other phosphorylated molecules specifically inhibit phosphoporin pore conductance to small ions, a property which is specific to phosphoporin. There is an excellent correlation between the effect of such solutes measured in planar bilayers and their inhibitory effect on beta-lactam antibiotic uptake in vivo by phosphoporin. It is concluded that the phosphoporin channel contains a selectivity filter which is only efficient for larger molecules, most probably through basic residues.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Benz R., Darveau R. P., Hancock R. E. Outer-membrane protein PhoE from Escherichia coli forms anion-selective pores in lipid-bilayer membranes. Eur J Biochem. 1984 Apr 16;140(2):319–324. doi: 10.1111/j.1432-1033.1984.tb08104.x. [DOI] [PubMed] [Google Scholar]
- Benz R., Schmid A., Hancock R. E. Ion selectivity of gram-negative bacterial porins. J Bacteriol. 1985 May;162(2):722–727. doi: 10.1128/jb.162.2.722-727.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bezanilla F., Armstrong C. M. Negative conductance caused by entry of sodium and cesium ions into the potassium channels of squid axons. J Gen Physiol. 1972 Nov;60(5):588–608. doi: 10.1085/jgp.60.5.588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorset D. L., Engel A., Häner M., Massalski A., Rosenbusch J. P. Two-dimensional crystal packing of matrix porin. A channel forming protein in Escherichia coli outer membranes. J Mol Biol. 1983 Apr 25;165(4):701–710. doi: 10.1016/s0022-2836(83)80275-7. [DOI] [PubMed] [Google Scholar]
- Eisenman G., Horn R. Ionic selectivity revisited: the role of kinetic and equilibrium processes in ion permeation through channels. J Membr Biol. 1983;76(3):197–225. doi: 10.1007/BF01870364. [DOI] [PubMed] [Google Scholar]
- Engel A., Massalski A., Schindler H., Dorset D. L., Rosenbusch J. P. Porin channel triplets merge into single outlets in Escherichia coli outer membranes. Nature. 1985 Oct 17;317(6038):643–645. doi: 10.1038/317643a0. [DOI] [PubMed] [Google Scholar]
- Garavito R. M., Rosenbusch J. P. Isolation and crystallization of bacterial porin. Methods Enzymol. 1986;125:309–328. doi: 10.1016/s0076-6879(86)25027-2. [DOI] [PubMed] [Google Scholar]
- Hille B. Potassium channels in myelinated nerve. Selective permeability to small cations. J Gen Physiol. 1973 Jun;61(6):669–686. doi: 10.1085/jgp.61.6.669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korteland J., Tommassen J., Lugtenberg B. PhoE protein pore of the outer membrane of Escherichia coli K12 is a particularly efficient channel for organic and inorganic phosphate. Biochim Biophys Acta. 1982 Sep 9;690(2):282–289. doi: 10.1016/0005-2736(82)90332-7. [DOI] [PubMed] [Google Scholar]
- Latorre R., Miller C. Conduction and selectivity in potassium channels. J Membr Biol. 1983;71(1-2):11–30. doi: 10.1007/BF01870671. [DOI] [PubMed] [Google Scholar]
- Läuger P. Ion transport through pores: a rate-theory analysis. Biochim Biophys Acta. 1973 Jul 6;311(3):423–441. doi: 10.1016/0005-2736(73)90323-4. [DOI] [PubMed] [Google Scholar]
- Mizuno T., Chou M. Y., Inouye M. A comparative study on the genes for three porins of the Escherichia coli outer membrane. DNA sequence of the osmoregulated ompC gene. J Biol Chem. 1983 Jun 10;258(11):6932–6940. [PubMed] [Google Scholar]
- Neuhaus J. M., Schindler H., Rosenbusch J. P. The periplasmic maltose-binding protein modifies the channel-forming characteristics of maltoporin. EMBO J. 1983;2(11):1987–1991. doi: 10.1002/j.1460-2075.1983.tb01689.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikaido H., Rosenberg E. Y. Porin channels in Escherichia coli: studies with liposomes reconstituted from purified proteins. J Bacteriol. 1983 Jan;153(1):241–252. doi: 10.1128/jb.153.1.241-252.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikaido H., Vaara M. Molecular basis of bacterial outer membrane permeability. Microbiol Rev. 1985 Mar;49(1):1–32. doi: 10.1128/mr.49.1.1-32.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Overbeeke N., Lugtenberg B. Recognition site for phosphorus-containing compounds and other negatively charged solutes on the PhoE protein pore of the outer membrane of Escherichia coli K12. Eur J Biochem. 1982 Aug;126(1):113–118. doi: 10.1111/j.1432-1033.1982.tb06754.x. [DOI] [PubMed] [Google Scholar]
- Raymond L., Slatin S. L., Finkelstein A. Channels formed by colicin E1 in planar lipid bilayers are large and exhibit pH-dependent ion selectivity. J Membr Biol. 1985;84(2):173–181. doi: 10.1007/BF01872215. [DOI] [PubMed] [Google Scholar]
- Schindler H. Formation of planar bilayers from artificial or native membrane vesicles. FEBS Lett. 1980 Dec 15;122(1):77–79. doi: 10.1016/0014-5793(80)80405-4. [DOI] [PubMed] [Google Scholar]
- Schindler H., Quast U. Functional acetylcholine receptor from Torpedo marmorata in planar membranes. Proc Natl Acad Sci U S A. 1980 May;77(5):3052–3056. doi: 10.1073/pnas.77.5.3052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schindler H., Rosenbusch J. P. Matrix protein from Escherichia coli outer membranes forms voltage-controlled channels in lipid bilayers. Proc Natl Acad Sci U S A. 1978 Aug;75(8):3751–3755. doi: 10.1073/pnas.75.8.3751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schindler H., Rosenbusch J. P. Matrix protein in planar membranes: clusters of channels in a native environment and their functional reassembly. Proc Natl Acad Sci U S A. 1981 Apr;78(4):2302–2306. doi: 10.1073/pnas.78.4.2302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tommassen J., Lugtenberg B. PHO-regulon of Escherichia coli K12: a minireview. Ann Microbiol (Paris) 1982 Mar-Apr;133(2):243–249. [PubMed] [Google Scholar]
- Tommassen J., Pugsley A. P., Korteland J., Verbakel J., Lugtenberg B. Gene encoding a hybrid OmpF--PhoE pore protein in the outer membrane of Escherichia coli K12. Mol Gen Genet. 1984;197(3):503–508. doi: 10.1007/BF00329950. [DOI] [PubMed] [Google Scholar]
- Tommassen J., van der Ley P., van Zeijl M., Agterberg M. Localization of functional domains in E. coli K-12 outer membrane porins. EMBO J. 1985 Jun;4(6):1583–1587. doi: 10.1002/j.1460-2075.1985.tb03820.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodhull A. M. Ionic blockage of sodium channels in nerve. J Gen Physiol. 1973 Jun;61(6):687–708. doi: 10.1085/jgp.61.6.687. [DOI] [PMC free article] [PubMed] [Google Scholar]


