Abstract
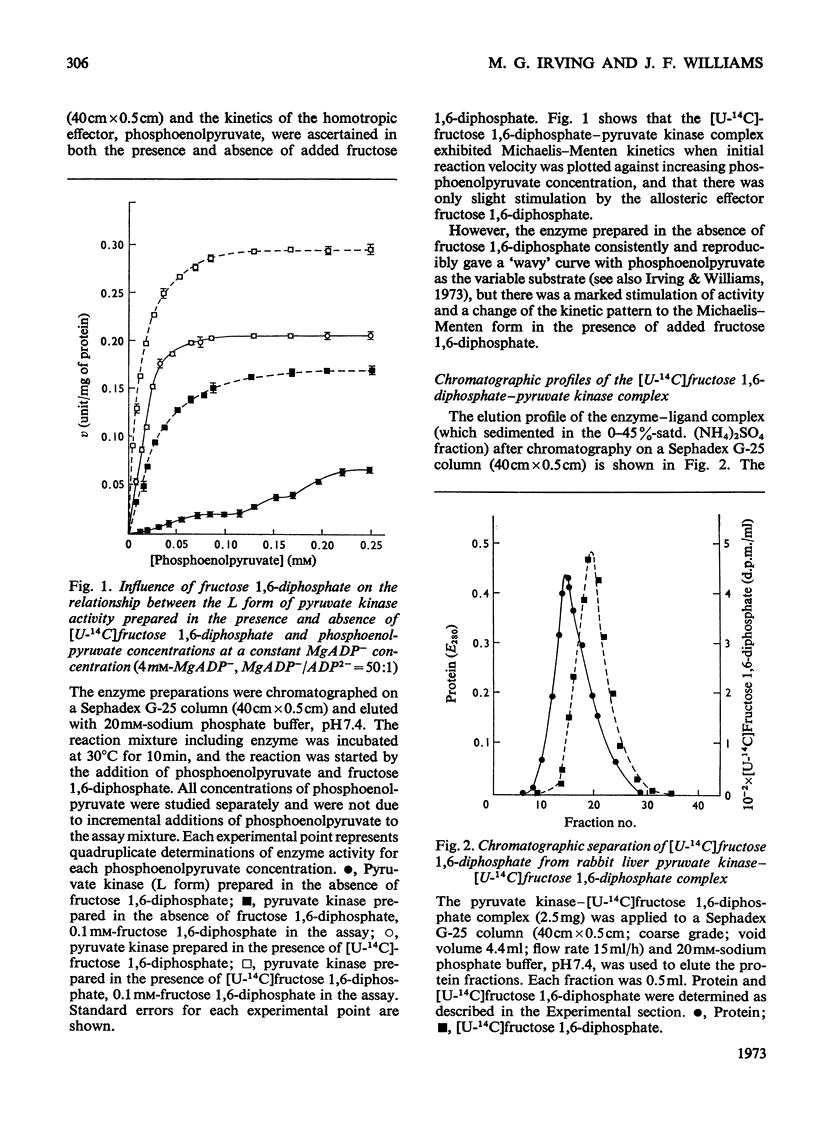
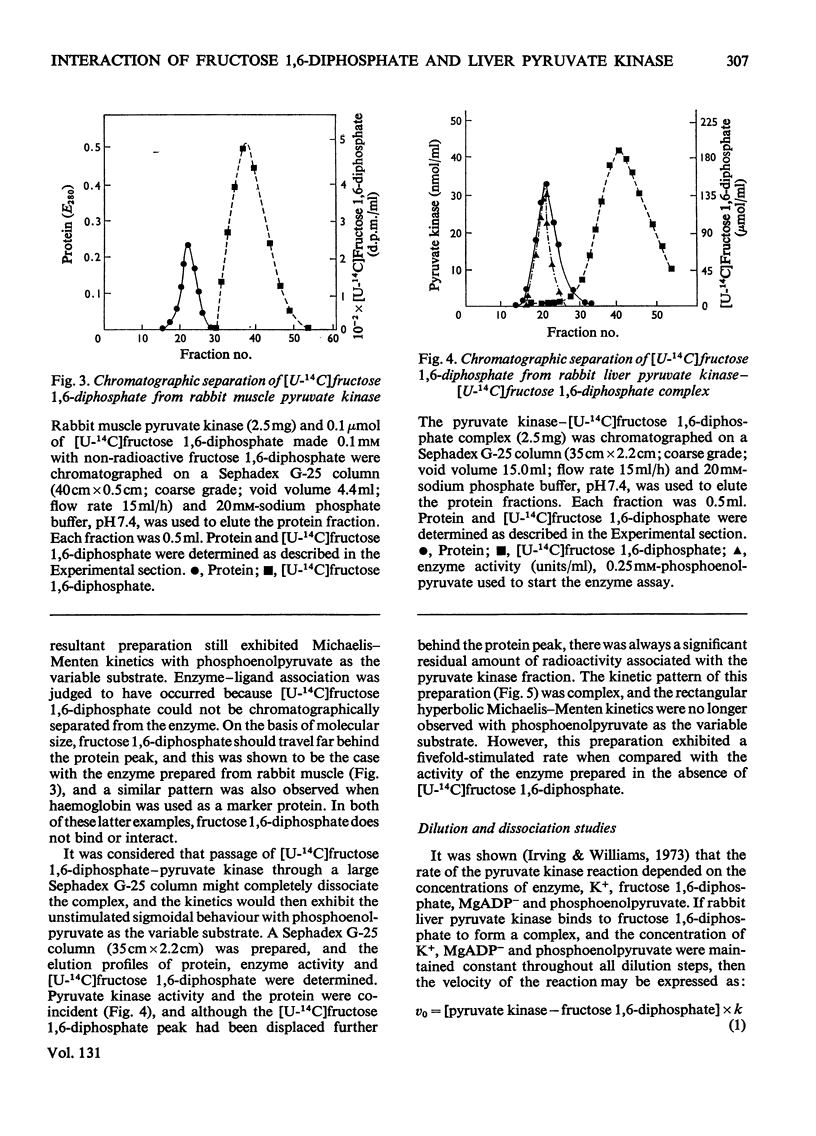
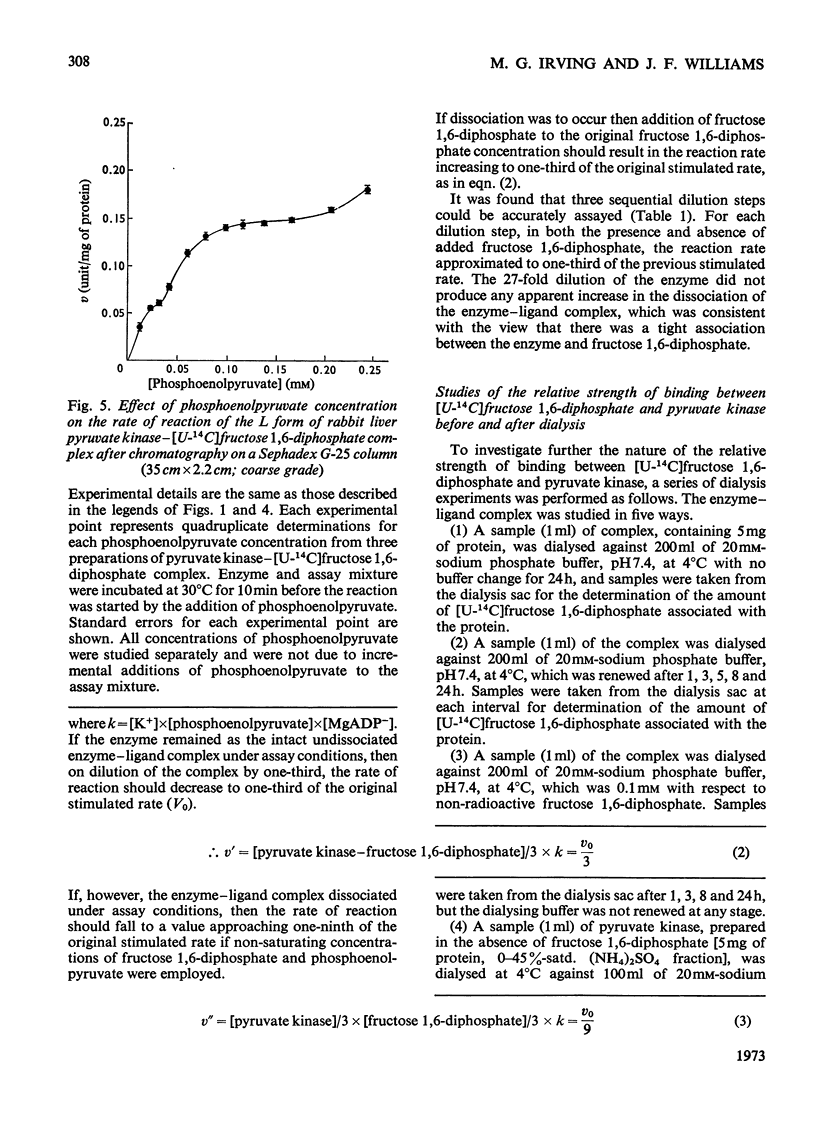
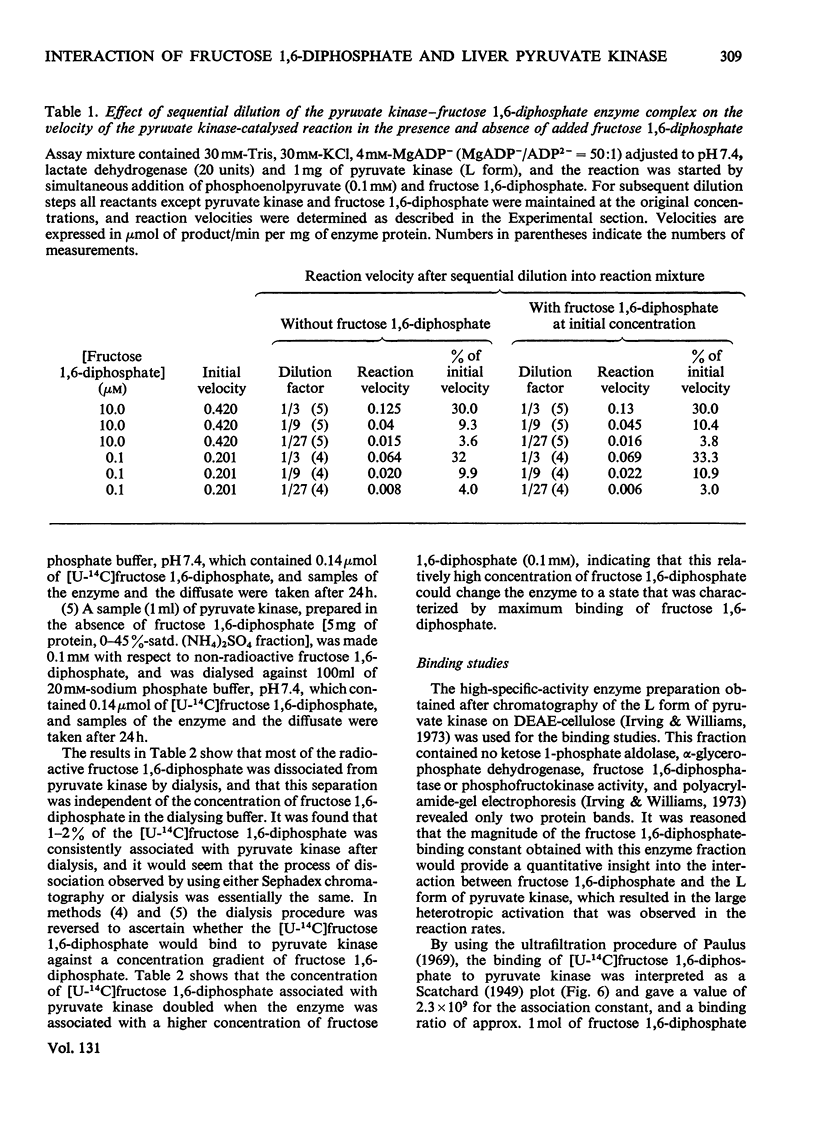
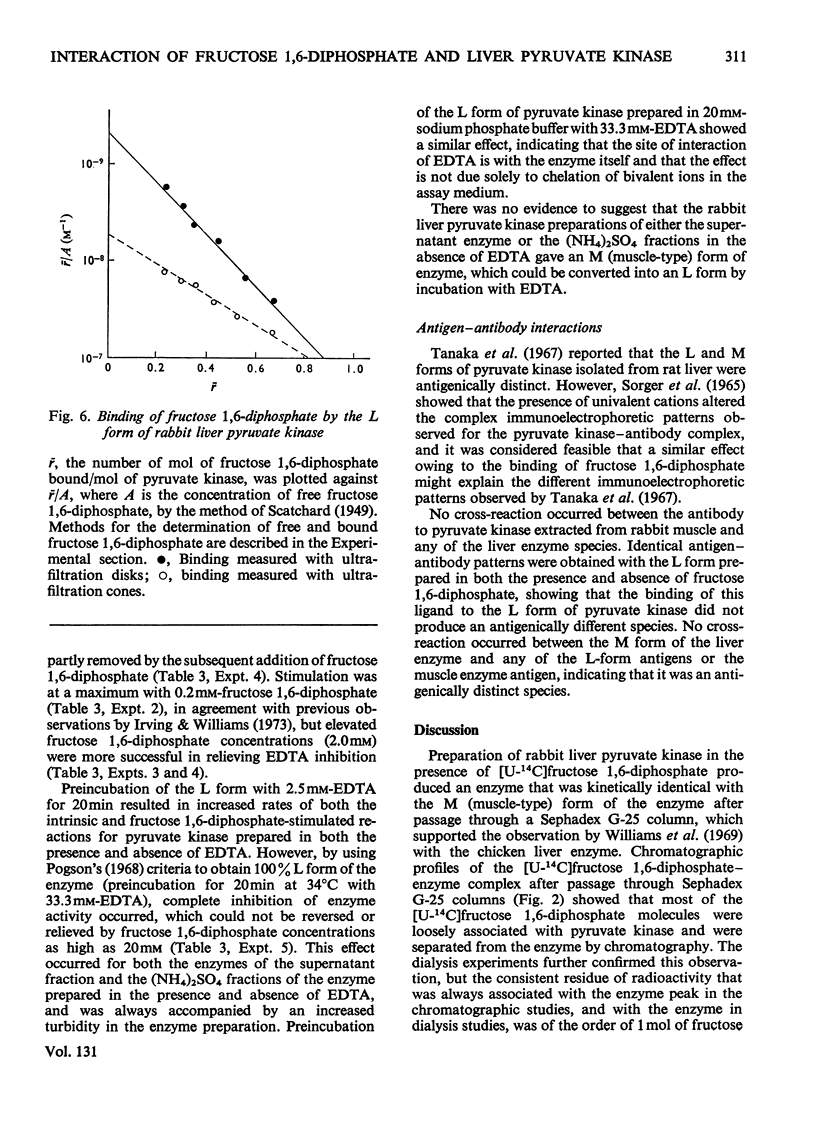
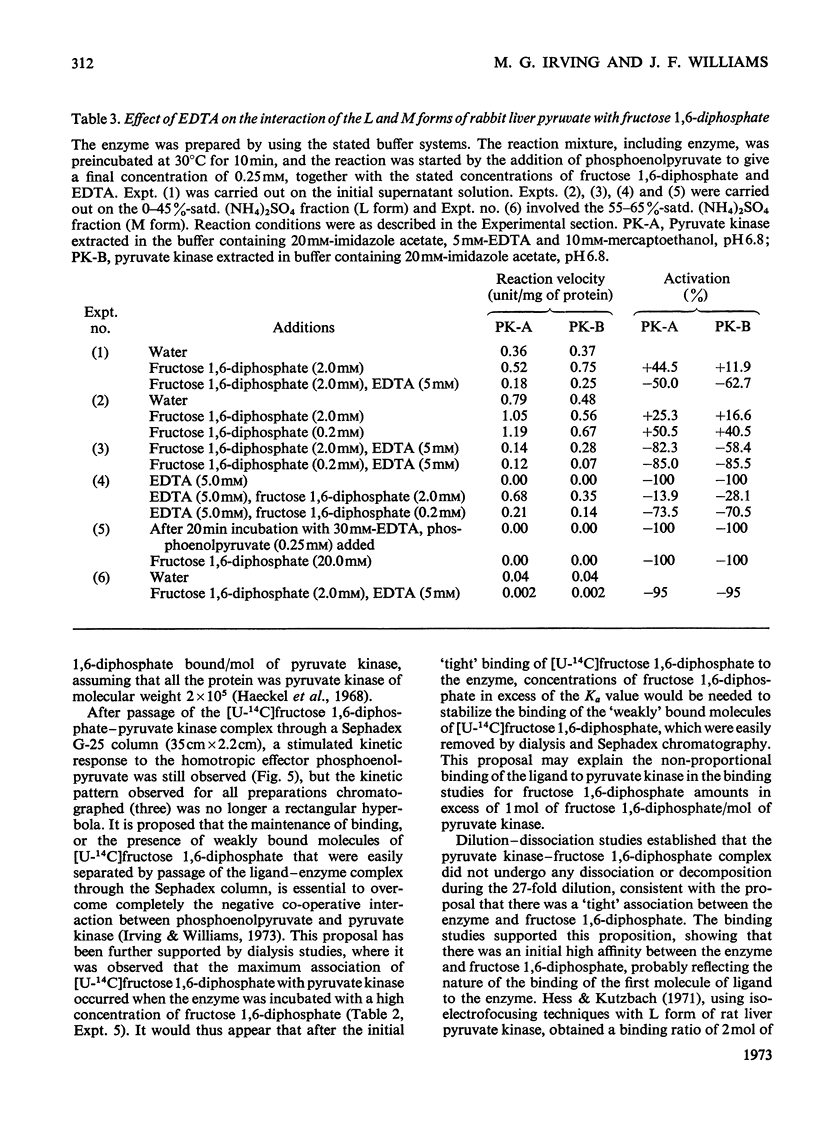
Preparation of the L form of rabbit liver pyruvate kinase (EC 2.7.1.40) in the presence of fructose 1,6-diphosphate yielded an enzyme which was kinetically identical with the M or muscle-type form of pyruvate kinase found in liver. Chromatographic and dialysis studies of this complex showed that most of the fructose 1,6-diphosphate molecules were loosely bound to the enzyme, but dilution–dissociation studies and binding experiments established that there was a high initial affinity between the enzyme and fructose 1,6-diphosphate (Kassoc.=2.3×109), and that binding of the loosely bound fructose 1,6-diphosphate was concentration-dependent and a necessary condition to overcome the co-operative interaction observed with the homotropic effector phosphoenolpyruvate. Preparation of the liver enzyme in the absence of EDTA did not yield a predominantly M form of the enzyme, and incubation of the M form in the presence of EDTA did not convert it into the L form, but resulted in inhibition of enzyme activity. Immunological studies confirmed that the L and M forms in liver were distinct, and that preparation of the L form in the presence of fructose 1,6-diphosphate did not produce an enzyme antigenically different from the L form prepared in the absence of this heterotropic effector.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bailey E., Stirpe F., Taylor C. B. Regulation of rat liver pyruvate kinase. The effect of preincubation, pH, copper ions, fructose 1,6-diphosphate and dietary changes on enzyme activity. Biochem J. 1968 Jul;108(3):427–436. doi: 10.1042/bj1080427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ballard F. J., Hanson R. W. Purification of phosphoenolpyruvate carboxykinase from the cytosol fraction of rat liver and the immunochemical demonstration of differences between this enzyme and the mitochondrial phosphoenolpyruvate carboxykinase. J Biol Chem. 1969 Oct 25;244(20):5625–5630. [PubMed] [Google Scholar]
- Blatt W. F., Robinson S. M., Bixler H. J. Membrane ultrafiltration: the diafiltration technique and its application to microsolute exchange and binding phenomena. Anal Biochem. 1968 Oct 10;26(1):151–173. doi: 10.1016/0003-2697(68)90039-0. [DOI] [PubMed] [Google Scholar]
- Haeckel R., Hess B., Lauterborn W., Wüster K. H. Purification and allosteric properties of yeast pyruvate kinase. Hoppe Seylers Z Physiol Chem. 1968 May;349(5):699–714. doi: 10.1515/bchm2.1968.349.1.699. [DOI] [PubMed] [Google Scholar]
- Hess B., Kutzbach C. Identification of two types of liver pyruvate kinase. Hoppe Seylers Z Physiol Chem. 1971 Mar;352(3):453–458. doi: 10.1515/bchm2.1971.352.1.453. [DOI] [PubMed] [Google Scholar]
- Irving M. G., Williams J. F. Kinetic studies on the regulation of rabbit liver pyruvate kinase. Biochem J. 1973 Feb;131(2):287–301. doi: 10.1042/bj1310287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paulus H. A rapid and sensitive method for measuring the binding of radioactive ligands to proteins. Anal Biochem. 1969 Oct 15;32(1):91–100. doi: 10.1016/0003-2697(69)90107-9. [DOI] [PubMed] [Google Scholar]
- Pogson C. I. Adipose-tissue pyruvate kinase. Properties and interconversion of two active forms. Biochem J. 1968 Nov;110(1):67–77. doi: 10.1042/bj1100067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorger G. J., Ford R. E., Evans H. J. Effects of univalent cations on the immunoelectrophoretic behavior of pyruvic kinase. Proc Natl Acad Sci U S A. 1965 Dec;54(6):1614–1621. doi: 10.1073/pnas.54.6.1614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka T., Harano Y., Sue F., Morimura H. Crystallization, characterization and metabolic regulation of two types of pyruvate kinase isolated from rat tissues. J Biochem. 1967 Jul;62(1):71–91. doi: 10.1093/oxfordjournals.jbchem.a128639. [DOI] [PubMed] [Google Scholar]
- Williams J. F., Rienits K. G., Schofield P. J., Clark M. G. The pentose phosphate pathway in rabbit liver. Studies on the metabolic sequence and quantitative role of the pentose phosphate cycle by using a system in situ. Biochem J. 1971 Aug;123(5):923–943. doi: 10.1042/bj1230923. [DOI] [PMC free article] [PubMed] [Google Scholar]


