Abstract
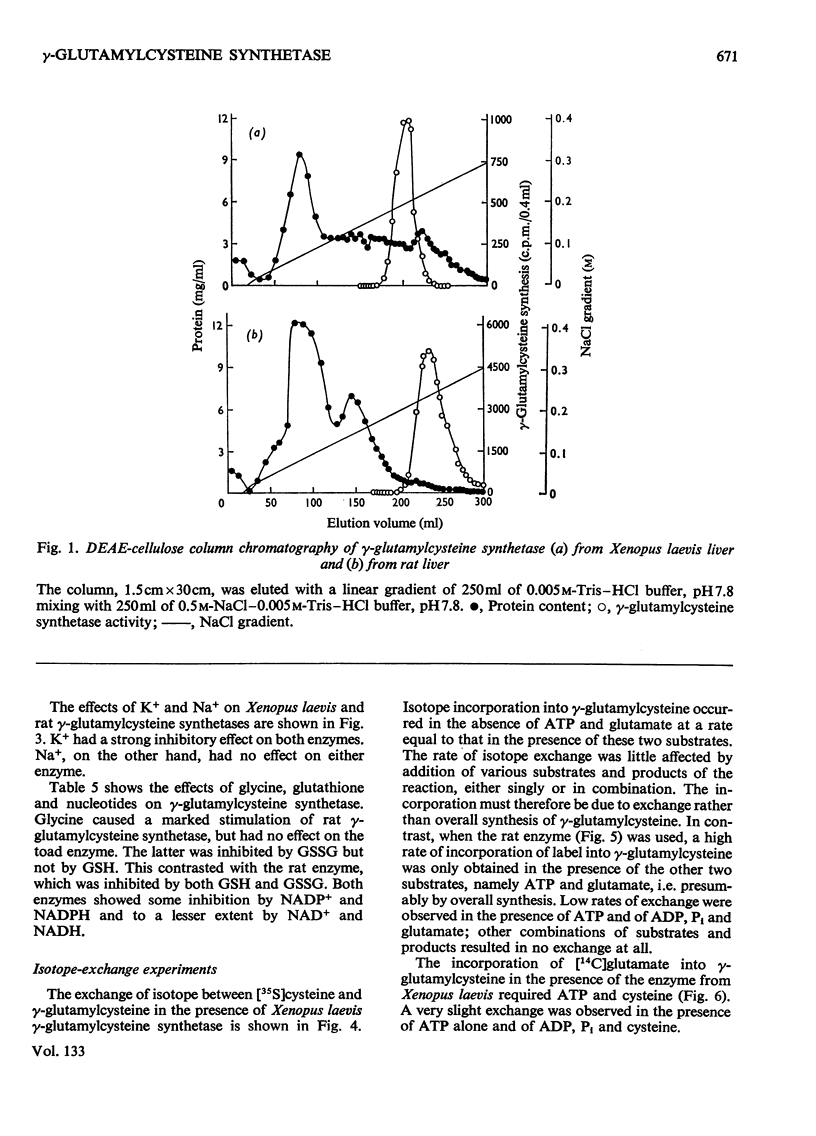
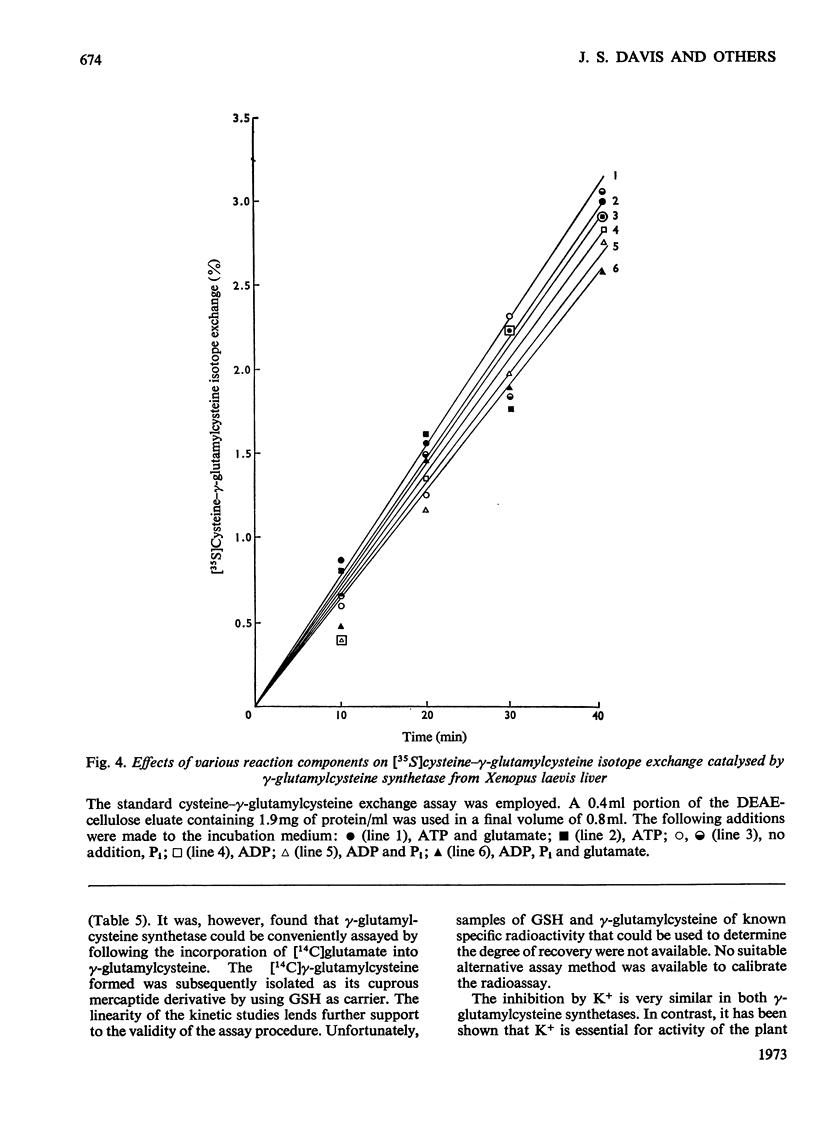
1. An improved radioassay for glutathione synthetase and γ-glutamylcysteine synthetase was developed. 2. Xenopus laevis liver γ-glutamylcysteine synthetase was purified 324-fold by saline–bicarbonate extraction, protamine sulphate precipitation, CM-cellulose and DEAE-cellulose column chromatography, and gel filtration. 3. Rat liver γ-glutamylcysteine synthetase was purified 11400-fold by a procedure similar to that employed for the Xenopus laevis enzyme. 4. Rat liver γ-glutamylcysteine synthetase activity was inhibited by GSH and activated by glycine. These effects, which were not found in the enzyme from Xenopus laevis, may have a regulatory significance. 5. Isotope-exchange experiments revealed fundamental differences in the partial reactions catalysed by the rat and Xenopus laevis synthetases. The enzyme from Xenopus laevis appears to follow a Bi Bi Uni Uni Ping Pong mechanism, with glutamyl–enzyme as intermediate before the addition of cysteine and the release of γ-glutamylcysteine. The results for the rat liver enzyme are consistent with a Tri Tri sequential mechanism.
Full text
PDF









Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andrews P. Estimation of molecular size and molecular weights of biological compounds by gel filtration. Methods Biochem Anal. 1970;18:1–53. [PubMed] [Google Scholar]
- CLELAND W. W. The kinetics of enzyme-catalyzed reactions with two or more substrates or products. I. Nomenclature and rate equations. Biochim Biophys Acta. 1963 Jan 8;67:104–137. doi: 10.1016/0006-3002(63)91800-6. [DOI] [PubMed] [Google Scholar]
- CLIFFE E. E., WALEY S. G. Acidic peptides of the lens. 6. Metabolism of gamma-glutamyl peptides in subcellular fractions of rabbit liver. Biochem J. 1961 Apr;79:118–128. doi: 10.1042/bj0790118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell P. N., Cooper C., Hicks M. Studies on the role of the morphological constituents of the microsome fraction from rat liver in protein synthesis. Biochem J. 1964 Aug;92(2):225–234. doi: 10.1042/bj0920225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies J. W., Cocking E. C. Liquid scintillation counting of 14C and 3H samples using glass-fibre or filter-paper discs. Biochim Biophys Acta. 1966 Feb 28;115(2):511–513. doi: 10.1016/0304-4165(66)90456-9. [DOI] [PubMed] [Google Scholar]
- JOHNSTON R. B., BLOCH K. Enzymatic synthesis of glutathione. J Biol Chem. 1951 Jan;188(1):221–240. [PubMed] [Google Scholar]
- Jackson R. C. Studies in the enzymology of glutathione metabolism in human erythrocytes. Biochem J. 1969 Feb;111(3):309–315. doi: 10.1042/bj1110309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- MANDELES S., BLOCK K. Enzymatic synthesis of gamma-glutamylcysteine. J Biol Chem. 1955 Jun;214(2):639–646. [PubMed] [Google Scholar]
- Orlowski M., Meister A. Isolation of highly purified gamma-glutamylcysteine synthetase from rat kidney. Biochemistry. 1971 Feb 2;10(3):372–380. doi: 10.1021/bi00779a003. [DOI] [PubMed] [Google Scholar]
- Orlowski M., Meister A. Partial reactions catalyzed by -glutamylcysteine synthetase and evidence for an activated glutamate intermediate. J Biol Chem. 1971 Dec 10;246(23):7095–7105. [PubMed] [Google Scholar]
- Rathbun W. B. Gamma-glutamyl-cysteine synthetase from bovine lens. I. Purification and properties. Arch Biochem Biophys. 1967 Oct;122(1):62–72. doi: 10.1016/0003-9861(67)90124-5. [DOI] [PubMed] [Google Scholar]
- Rathbun W. B. Gamma-glutamyl-cysteine synthetase from bovine lens. II. Cysteine analogue studies. Arch Biochem Biophys. 1967 Oct;122(1):73–84. doi: 10.1016/0003-9861(67)90125-7. [DOI] [PubMed] [Google Scholar]
- SNOKE J. E., YANARI S., BLOCH K. Synthesis of glutathione from gamma-glutamylcysteine. J Biol Chem. 1953 Apr;201(2):573–586. [PubMed] [Google Scholar]
- WEBSTER G. C. Peptide-bond synthesis in higher plants. I. The synthesis of glutathione. Arch Biochem Biophys. 1953 Dec;47(2):241–250. doi: 10.1016/0003-9861(53)90464-0. [DOI] [PubMed] [Google Scholar]
- WEBSTER G. C., VARNER J. E. Peptidebond synthesis in higher plants. II. Studies on the mechanism of synthesis of gamma-glutamylcysteine. Arch Biochem Biophys. 1954 Sep;52(1):22–32. doi: 10.1016/0003-9861(54)90085-5. [DOI] [PubMed] [Google Scholar]
- Widnell C. C., Tata J. R. A procedure for the isolation of enzymically active rat-liver nuclei. Biochem J. 1964 Aug;92(2):313–317. doi: 10.1042/bj0920313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- YANARI S., SNOKE J. E., BLOCH K. Energy sources in glutathione synthesis. J Biol Chem. 1953 Apr;201(2):561–571. [PubMed] [Google Scholar]


