ABSTRACT
Male sexual dysfunction, characterised by reduced libido, ejaculatory issues and erectile dysfunction, often results from oxidative stress and enzymatic imbalance, notably involving phosphodiesterase type 5 (PDE5) and nitric oxide synthase (NOS). This study explores the therapeutic potential of β‐caryophyllene (β‐CBP), a sesquiterpene with antioxidant and anti‐inflammatory properties, in mitigating paroxetine‐induced sexual dysfunction in rats. Male Wistar rats were divided into nine treatment groups: control, paroxetine (20 mg/kg/day), sildenafil (20 mg/kg/day), β‐CBP (10 mg/kg/day), β‐CBP (20 mg/kg/day), paroxetine with β‐CBP (10 mg/kg), paroxetine with β‐CBP (20 mg/kg), paroxetine with sildenafil and β‐CBP with sildenafil. Sexual behavioural assays were evaluated, along with oxidative stress markers, including superoxide dismutase (SOD) and catalase (CAT) activity in penile tissue, assessed using spectrophotometric analysis. CB2 receptor expression was significantly increased in β‐CBP‐treated groups, suggesting enhanced cannabinoid receptor‐mediated signalling, which may be linked to improved erectile function. The effects were dose‐dependent, with the 20 mg/kg β‐CBP group displaying the most significant improvements. Additionally, β‐CBP restored antioxidant enzyme activities, including SOD, CAT and reduced glutathione (GSH) levels in penile tissue, effectively reducing oxidative stress. β‐CBP shows promise as a therapeutic agent for male sexual dysfunction by enhancing antioxidative capacity and modulating enzymatic balance.
Keywords: CB2, erectile dysfunction, oxidative stress, paroxetine, β‐caryophyllene
Plain English Summary.
Our study explored how a natural compound called β‐caryophyllene (β‐CBP) might help with sexual dysfunction, a problem that can include low sex drive and difficulty with erections. We used rats in our experiments to see if β‐CBP could counteract the adverse effects of a drug called paroxetine, which can cause these issues. The rats treated with β‐CBP showed significant improvements in sexual performance, likely because β‐CBP reduced oxidative stress and helped restore healthy enzyme function in the body. These results suggest that β‐CBP could be a promising treatment for sexual dysfunction in the future.
1. Introduction
Male sexual dysfunction encompasses a spectrum of disorders such as reduced libido, ejaculatory issues, and erectile dysfunction [1]. These dysfunctions stem from complex pathophysiological factors, including oxidative stress, hormonal imbalances and dysfunction of key enzymes like phosphodiesterase type 5 (PDE5) and nitric oxide synthase (NOS), both critical for sexual health [2]. Oxidative stress, in particular, has been recognised as a significant contributor to the impairment of endothelial function, disrupting nitric oxide (NO) pathways necessary for penile erection and sexual performance [3].
Recent research has highlighted the therapeutic potential of β‐caryophyllene (β‐CBP), a naturally occurring bicyclic sesquiterpene found in essential oils like black pepper, cinnamon and cloves. β‐CBP is renowned for its antioxidant, anti‐inflammatory and neuroprotective properties [4, 5]. It exerts its protective effects by scavenging free radicals, reducing oxidative stress and modulating inflammatory mediators. These properties make β‐CBP a promising candidate for ameliorating sexual dysfunction. Studies have also demonstrated that β‐CBP can improve sexual performance by modulating critical enzymes involved in erectile function, such as PDE5 and NOS, in animal models [2]. Moreover, β‐CBP's antioxidant properties help mitigate oxidative stress, which is linked to the development of sexual dysfunction [6]. Interestingly, olfactory exposure to β‐CBP has been shown to increase testosterone levels, a crucial hormone in sexual performance [7]. In addition to its antioxidative and hormonal effects, β‐CBP acts as an agonist of cannabinoid receptor type 2 (CB2), which is highly expressed in germ cells within the testes. This receptor activation has been shown to influence male reproductive health. Al‐Alami et al. demonstrated that β‐CBP enhances sperm motility while reducing sperm count in rats without affecting spermatogenesis [8]. This suggests that β‐CBP could impact reproductive function beyond its known antioxidant roles, further supporting its potential to address sexual dysfunction.
Given the multifaceted effects of β‐CBP, this study investigates its role in alleviating male sexual dysfunction using a paroxetine‐induced rat model. This model mimics sexual dysfunction symptoms, including decreased libido and impaired sexual behaviour, offering a comprehensive assessment of β‐CBP's efficacy. By examining oxidative stress markers and key enzyme activities, this study seeks to explore the potential of β‐CBP as a novel therapeutic approach for male sexual dysfunction. These findings could provide new insights into the broader implications of β‐CBP on sexual health, highlighting its potential not only for treating erectile dysfunction but also for improving overall male reproductive function.
2. Materials and Methods
The study was conducted in accordance with the Basic & Clinical Pharmacology & Toxicology policy for experimental and clinical studies [9].
2.1. Chemicals and Reagents
The chemical and reagents used included acetylcholine iodide, gallic acid and Folin‐Ciocalteu reagent obtained from Sigma‐Aldrich (St. Louis, MO, United States). β‐CBP (purity C98.5%), metronidazole, Tween 80 and Durcupan™ ACM Fulka embedding mixture were from Sigma‐Aldrich, United States. Thiobarbituric acid (TBA), trichloroacetic acid (TCA), quercetin, 1,1‐diphenyl‐2‐picrylhydrazyl (DPPH) and 2‐deoxyribose were procured from Sigma‐Aldrich Chemie (United States). Sodium carbonate, ferric chloride, aluminium chloride, potassium acetate, potassium ferricyanide, tris salt, ferric sulphate and other reagents used were of analytical grade; glass‐distilled water was also used.
2.2. Determination of DPPH Radical Scavenging Ability
The radical scavenging ability of the sample against DPPH free radical was evaluated as described by Gyamfil et al. [10]. An appropriate sample dilution was mixed with 1 mL of 0.4 mM methanolic solution containing DPPH radical. The mixture was left in the dark for 30 min, and the absorbance was measured at 516 nm. The percentage of DPPH radical scavenging ability was subsequently calculated as a percentage of the control.
2.3. Determination of Fe(II) Chelating Ability
The method of Puntel et al. was used to determine the iron chelating ability of the samples [11]. A mixture containing 150 μL of freshly prepared 500 μM FeSO4, 168 μL of 0.1 M Tris–HCL (pH 7.4) buffer, 218 μL of saline solution and an appropriate concentration of the samples was prepared. After incubation for 5 min, 13 μL of 0.25% 1,10‐phenanthroline (w/v) was added. Absorbance was measured at 510 nm, and the iron chelating ability was subsequently calculated and expressed as a percentage.
2.4. Determination of Ferric‐Reducing Antioxidant Property
The reducing property of the samples was determined by assessing the ability of the samples to minimise FeCl3 solution as described by Oyiazu [12]. A 2.5‐mL aliquot of each sample was mixed with 2.5 mL of 200 mM sodium phosphate buffer (pH 6.6) and 2.5 mL of 1% potassium ferricyanide. Following incubation at 50 °C for 20 min, 2.5 mL of 10% trichloroacetic acid (TCA) was added. After centrifugation, 5 mL of the supernatant was mixed with an equal volume of water and 1 mL of 0.1% ferric chloride. Absorbance was measured at 700 nm.
2.5. Total Antioxidant Capacity
The total antioxidant capacity of the sample (ABTS) was determined against 2,2′‐azino‐bis (ethylbenzothiazoline‐6‐sulphonic acid) (ABTS) radical according to the method described by Re et al. [13]. The ABTS radical was generated by reacting 7 mM ABTS aqueous solution with K2S2O8 (2.45 mM) for 16 h. The absorbance was adjusted at 734 nm, and Trolox equivalent antioxidant capacity (TEAC) was calculated using Trolox as the standard.
2.6. Inhibition of Fenton Reaction (Degradation of Deoxyribose)
The method of Halliwell and Gutteridge was used to determine the ability of the samples to prevent Fe2+/H2O2 induced decomposition of deoxyribose [14]. Samples were added to a reaction mixture containing 120 μL of 20 mM deoxyribose, 400 μL of 0.1 M phosphate buffer and 40 μL of 500 mM FeSO4. After incubation at 37 °C for 30 min, the reaction was stopped, and absorbance was measured at 532 nm using a spectrophotometer.
2.7. Experimental Animal Handling and Feeding
In this study, male and female Wistar rats, each weighing between 200 and 220 g, were utilised. These animals, aged between 9 and 10 weeks, had reached sexual maturity and were sourced from the Federal University of Technology, Akure, Nigeria. All procedures involving the animals adhered to the guidelines set by the National Council for Animal Experiments Control (CONCEA) and the International Guiding Principles for Biomedical Research Involving Animals. Throughout the study, both male and female rats were provided with standard feed.
Following a 2‐week acclimatisation period, the rats were randomly assigned into nine groups, each consisting of six animals (one female in each group), and were fed for 14 days with unrestricted access to food and water. Animal handling strictly followed international standards. The dosage of β‐CBP was based on prior research, with an initial dose of 10 mg/kg/day [15, 16]. However, this study also included a higher dose of 20 mg/kg/day to evaluate a broader dose–response relationship, as the 20 mg/kg dose demonstrated significant effects on erectile function and oxidative stress markers. The inclusion of both dosing regimens aimed to explore the dose‐dependent efficacy of β‐CBP further. Paroxetine (20 mg/kg) and sildenafil citrate (20 mg/kg) were also administered daily, based on previous studies with slight modifications [17, 18].
2.8. Experimental Design
Group I: normal control rats which were administered the vehicle (1% ethanol)
Group II: β‐CBP (10 mg/kg)
Group III: β‐CBP (20 mg/kg)
Group IV: sildenafil citrate (20 mg/kg/day)
Group V: β‐CBP (20 mg/kg/day) + sildenafil citrate (20 mg/kg/day)
Group VI: paroxetine‐induced erectile dysfunction (PIED) rat
Group VII: paroxetine‐induced erectile dysfunction + β‐CBP (10 mg/kg)
Group VIII: paroxetine‐induced erectile dysfunction + (20 mg/kg) of β‐CBP
Group IX: paroxetine‐induced erectile dysfunction + sildenafil citrate (20 mg/kg/day)
All groups received treatments with 1% ethanol as the vehicle. On the 14th day, male rats were paired with females for sexual behavioural assays following the protocol described by Thawatchai et al. and modifications informed by Adefegha et al.
2.9. Sexual Behaviour
The sexual behaviour assay was conducted based on a modified protocol from Thawatchai et al., with additional methodological support from Adefegha et al. This modification involved the use of higher doses of hormones compared to the original study. Specifically, oestradiol benzoate was administered at a dose of 10 μg/mg body weight (five times higher than in Thawatchai et al.), and progesterone was given at 1 mg/kg body weight (double the dose) through subcutaneous and intraperitoneal routes, respectively. While there is a previous study that utilised the modified dosing in the evaluation context of this study [19], the design was informed by existing functional data from Adefegha et al.
Male rats were trained prior to the experiment to ensure that sexual behaviours could be reliably measured. The training involved exposure to receptive females to allow the males to become familiar with the testing environment and reduce variability in sexual behaviour performance. Female rats were ovariectomised to ensure controlled induction of oestrus using the hormone regimen. Only females confirmed to be in oestrus through vaginal smear cytology were included in the assay, following a previously described methodology [20]. Each oestrus female was paired with a single male from each group. After the male test was completed, the female rats were euthanised in accordance with ethical guidelines through an overdose of pentobarbital administered intraperitoneally. This method ensures a humane and rapid process, minimising any animal distress in line with CONCEA guidelines. The same female was not used for multiple males to avoid potential confounding effects of fatigue or prior mating.
Sexual behaviours were observed using a clear rectangular plastic chamber (60 × 40 × 30 cm) in a designated room. A digital video recorder was positioned to capture all activities without interfering with the behaviour of the animals. The recorded videos were analysed to assess key sexual parameters including mounting number, mounting latency, intromission number and intromission latency. Although we did not assess female sexual behaviours directly, care was taken to ensure that all females were proceptive to the males regardless of the treatment group. This was confirmed by the consistent occurrence of proceptive behaviours, such as solicitation and lordosis, prior to pairing with the males.
2.10. CB2 Receptor Assay
To evaluate the involvement of CB2 receptors in the effect of β‐CBP, a separate group of animals (Group X) was included, which received a selective CB2 receptor antagonist, AM630, administered at a dose of 3 mg/kg intraperitoneally alongside β‐CBP (20 mg/kg). The CB2 receptor antagonist AM630 was administered 30 min prior to each β‐CBP administration to ensure effective CB2 receptor blockade throughout β‐CBP's action. The 3‐mg/kg dose of AM630, primarily used in mouse models, was selected based on modifications from prior studies, such as García‐Gutiérrez et al., which demonstrated its efficacy and receptor‐binding affinity in Swiss ICR mice [21]. We aligned this protocol with previous approaches, such as those outlined by Munawar et al., in which antagonists were pre‐administered to investigate receptor‐specific effects [22].
2.11. Quantification of CB2 Receptor Activity
Penile tissue was collected from all groups, including the CB2 receptor antagonist group, for Western blot analysis. CB2 receptor expression was quantified using a primary antibody specific for CB2 receptors (Santa Cruz Biotechnology, United States; antibody number sc‐293188). The procedure involved the following steps:
Sample preparation: Homogenised penile tissue samples were lysed in RIPA buffer with protease inhibitors.
Protein quantification: Total protein concentration was measured using the BCA protein assay kit (Pierce, United States). A housekeeping protein (β‐actin) was used as a loading control to normalise CB2 receptor expression, ensuring consistent quantification across treatments [23].
SDS‐PAGE and Western Blot: Equal amounts of protein (determined by quantification) were separated by SDS‐PAGE and transferred to PVDF membranes. The membranes were blocked with 5% non‐fat milk and incubated overnight at 4°C with a CB2 primary antibody.
Detection: HRP‐conjugated secondary antibodies were applied, and protein bands were visualised using the ECL detection reagent (GE Healthcare, United States).
Densitometry analysis: Protein bands were quantified using ImageJ software to measure CB2 receptor levels, normalised to the β‐actin control.
2.12. Determination of Penile Superoxide Dismutase (SOD) Activity
Superoxide dismutase (SOD) activity was determined by the method of Mistra and Fridovich [24]. Penile tissue (0.1 mL) was diluted in 0.9 mL of water to make one in 10 dilutions. An aliquot of the diluted homogenate was added to carbonate buffer (0.05 M, pH 10.2) and adrenaline substrate (0.3 M). Absorbance at 480 nm was monitored every 30 s for 150 s.
2.13. Determination of Penile Catalase Activity
This was carried out according to the modified method of Clairborne [25]. Test samples (50 μL) were added to a reacting mixture containing H2O2 and phosphate buffer (pH 7.0). Absorbance at 570 nm was monitored for 3 min at intervals of 150 s.
2.14. Determination of Penile‐Reduced Glutathione (GSH) Level
Reduced GSH level was determined by the method of Jollow et al. [26]. The supernatant (1 mL) was treated with Elman's reagent and phosphate buffer, and absorbance was read at 412 nm.
2.15. Determination of Penile Glutathione‐S‐Transferase (GST) Activity
This was carried out according to Mannervik and Guthenberg's [27] method. Test samples (30 μL) were added to a reacting mixture containing CDNB and phosphate buffer (pH 6.5). Absorbance at 340 nm was monitored for 3 min at 15 s intervals.
2.16. Data Analysis
The results were expressed as the means ± SD. Statistical analysis was determined using JMP Pro (Version 16, JMP, SAS Institute Inc.).
3. Results
3.1. In Vitro Antioxidant Potentials of β‐Caryophyllene
The ferric reducing antioxidant properties (FRAP) analysis of β‐CBP yielded results indicating comparatively lower reducing potentials in contrast to sildenafil citrate, as depicted in Figure 1a. Likewise, the ABTS radical scavenging capacity of β‐CBP is illustrated as Trolox equivalent antioxidant capacity in Figure 1b, revealing the highest ABTS radical scavenging ability. The OH radical scavenging capability of β‐CBP, presented in Figure 1c along with associated IC50 values in Table 1, shows a marked increase in the ‘Sick’ group (18.00 μg/mL), indicating a diminished antioxidant capacity relative to the ‘Control’ group (8.50 μg/mL). This supports the hypothesis that oxidative stress contributes to the altered antioxidant defence in the diseased state.
FIGURE 1.
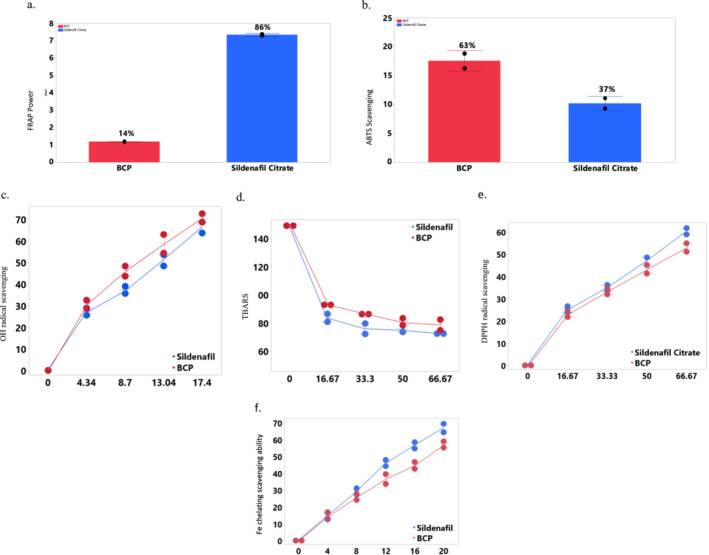
In vitro antioxidant activity of β‐caryophyllene. (a) Ferric reducing antioxidant scavenging ability, (b) ABTS radical scavenging ability of β‐caryophyllene, (c) OH radical scavenging ability, (d) % TBARS, (e) DPPH radical scavenging ability, (f) iron chelation scavenging ability. BCP, β‐caryophyllene. Values represent mean ± standard deviation of triplicate readings. Error bar was constructed using 1 standard deviation from mean. The result in Figure 1 shows the antioxidant activity of β‐caryophyllene and sildenafil citrate. Their antioxidant activity was compared to each other across different assays invitro, as no animals were used.
TABLE 1.
IC50 (μg/mL) of antioxidant activities of β‐CBP and sildenafil citrate in isolated rat's penile tissue.
| Parameters | Control | Sick | β‐CBP | Sildenafil citrate |
|---|---|---|---|---|
| DPPH | 50.00 | 70.00 | 52.92a | 59.14b |
| OH | 8.50 | 18.00 | 10.98a | 12.58b |
| % TBARS | 60.00 | 90.00 | 75.51a | 84.64b |
| Fe2+ chelation | 12.00 | 22.50 | 14.26a | 17.36b |
Note: Values represent means. Within each row, values with different superscript letters (a, b) are significantly different (p < 0.0001).
Furthermore, lipid peroxidation, as displayed in Figure 1d, exhibited a significant increase in the percentage of Thiobarbituric acid reactive substances (TBARS) produced upon the addition of pro‐oxidants. Conversely, a concentration‐dependent reduction in the % TBARS produced was observed upon the inclusion of β‐CBP. Figure 1e showcases the DPPH radical scavenging capacity of β‐CBP, while Table 1 details the corresponding IC50 values. This presentation highlights the concentration‐dependent scavenging of DPPH radicals. In addition, Figure 1f illustrates the iron chelating ability of β‐CBP, where results indicate a concentration‐dependent chelation of Fe2+. The IC50 values in Table 1 provide insight into the chelating potential of β‐CBP.
3.2. Effects of β‐Caryophyllene on Antioxidant Enzymes
Figure 2a illustrates the activity of SOD within liver tissue. The results highlight a significant variance (p < 0.05) between the PIED and control groups. Additionally, all other groups exhibited increased SOD activity, significantly differing (p < 0.05) from both the control and Paroxetine groups. Notably, the β‐CBP groups displayed a further increase compared to the control and PIED groups. Figure 2b presents the impact of β‐CBP on catalase activity in liver tissue. The results reveal a significant rise in catalase activity in the PIED group compared to the control group (p < 0.05). All remaining groups also showed marked differences (p < 0.05) when compared to the control and PIED groups. Moving on to Figure 2c, the influence of β‐CBP on GST activity in liver tissue is depicted. The data illustrates an increase in GST activity in the PIED group, signifying a significant distinction (p < 0.05) from the control group. Furthermore, all groups displayed notable variances (p < 0.05) compared to the normal control and PIED groups.
FIGURE 2.
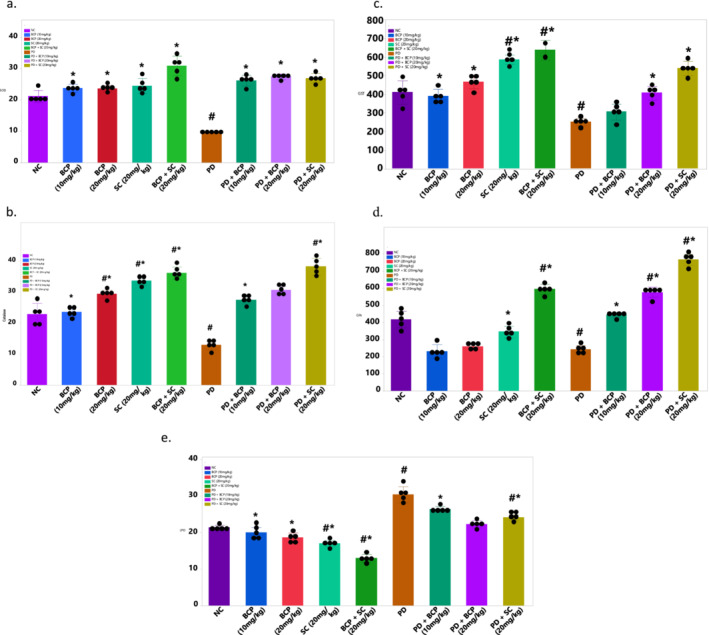
Effect of β‐caryophyllene on antioxidant liver biomarkers of paroxetine‐induced rats. (a) SOD activity, (b) catalase activity, (c) glutathione‐S‐transferase (GST) activity, (d) glutathione peroxidase (GPx) activity, (e) lipid peroxidation activity. Values represent means ± standard deviation of replicate readings (n = 8). Number sign and asterisk indicate significant difference when compared to NC and PD, respectively. p < 0.05 is considered significant.
Figure 2d demonstrates the effect of β‐CBP on glutathione peroxidase (GPx) in liver tissue. The results exhibit a significant elevation in GPx activity in the PIED group (p < 0.05), and all other groups displayed marked differences (p < 0.05) in comparison to the normal control and PIED groups. Figure 2e portrays the effects of β‐CBP on lipid peroxidation within liver homogenate. β‐CBP effectively reduced lipid peroxidation activity in comparison to the PD group. These findings indicate significant distinctions (p < 0.05) among all groups, with the PIED groups treated with β‐CBP displaying a noteworthy difference (p < 0.05) compared to the control groups.
3.3. CB2 Receptor Expression
Figure 3 presents the relative expression levels of CB2 receptors across different experimental groups. The CB2 receptor expression was significantly increased in the groups treated with β‐CBP compared to the normal control group. Notably, the β‐CBP (20 mg/kg) group exhibited the highest expression levels among all the groups, including the combination treatment with sildenafil citrate. The PIED rat group showed reduced CB2 receptor expression, which was partially restored by β‐CBP treatment in a dose‐dependent manner. Interestingly, the co‐administration of β‐CBP with the CB2 antagonist AM630 resulted in a notable decrease in CB2 receptor expression, underscoring the role of CB2 receptor activation in mediating the observed effects.
FIGURE 3.
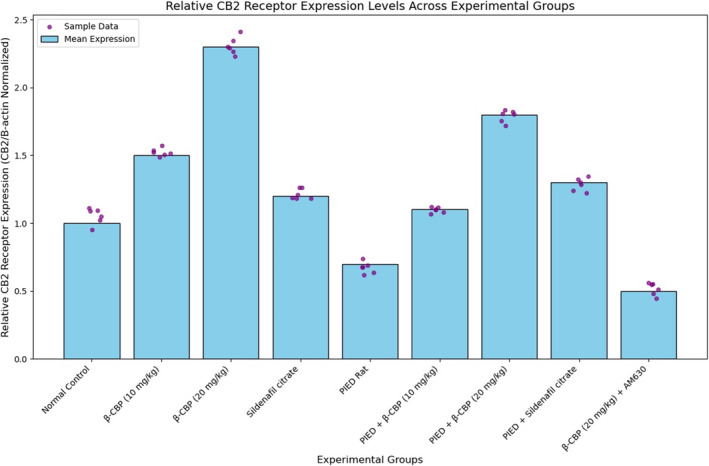
CB2 receptor expression levels in different experimental groups. Values are expressed relative to the normal control group. Error bars represent the standard deviation of the mean (n = 6). Statistical significance was determined at p < 0.05.
4. Discussion
Male sexual dysfunction, including conditions such as erectile dysfunction and reduced libido, arises from various underlying pathophysiological mechanisms. Among these, oxidative stress has been identified as a significant factor contributing to sexual dysfunction, particularly in its impairment of endothelial function and the disruption of NO‐mediated pathways essential for erection [3]. Given the centrality of oxidative stress in sexual dysfunction, addressing oxidative damage through antioxidant therapies has garnered attention as a potential intervention.
In this context, β‐CBP has emerged as a promising therapeutic candidate due to its well‐documented antioxidant properties. As a natural bicyclic sesquiterpene, β‐CBP exerts multifaceted protective effects through its ability to scavenge free radicals, inhibit lipid peroxidation, and modulate inflammatory responses [28]. This study aimed to investigate the potential of β‐CBP in ameliorating male sexual dysfunction, specifically using a paroxetine‐induced model that mimics sexual dysfunction symptoms such as diminished libido and compromised erectile function. Previous studies have shown that paroxetine‐induced sexual dysfunction is associated with oxidative stress, as it promotes the generation of reactive oxygen species (ROS) that disrupt key enzymatic pathways, such as NOS and PDE5, leading to impaired penile tissue integrity [29, 30].
Oxidative stress is known to disrupt penile function by promoting the formation of ROS [31], which in turn impairs the activity of key enzymes such as PDE5 and NOS. These enzymes are critical to regulating penile blood flow and smooth muscle relaxation, which are necessary for normal erectile function. The in vitro assays revealed that β‐CBP exhibits significant antioxidant activity, though it showed lower reducing potentials than Sildenafil Citrate in the FRAP assay (Figure 1a). However, β‐CBP demonstrated a strong ABTS radical scavenging ability (Figure 1b), highlighting its capacity to neutralise these radicals. Its concentration‐dependent scavenging of OH radicals (Figure 1c and Table 1) and the ability to reduce TBARS production (Figure 1d) underscore its potential to counteract oxidative damage effectively. The DPPH scavenging assay (Figure 1e) further confirms β‐CBP's ability to donate hydrogen atoms, a key mechanism for neutralising radicals. This aligns with recent research suggesting that phenolic compounds, like β‐CBP, exert antioxidant effects by donating hydrogen atoms to reactive oxygen species [32, 33].
β‐CBP also significantly influenced antioxidant enzyme activity. The study found increased activity of SOD, catalase, GST and GPx in liver tissue (Figure 2a–e), indicating an enhancement of antioxidant defences. This suggests that β‐CBP reduces oxidative stress by boosting these crucial enzyme activities. Notably, β‐CBP administration restored oxidative balance in the penile tissues of paroxetine‐induced dysfunction rats, as evidenced by increased non‐protein thiol levels. Non‐protein thiols, including GPx, play a crucial role in cellular antioxidant defence by detoxifying peroxides and preventing lipid peroxidation [34], thus protecting penile tissues from oxidative damage. Additionally, β‐CBP modulated CB2 receptor expression, which is involved in reproductive health. The upregulation of CB2 receptors in β‐CBP‐treated groups suggests that β‐CBP may exert protective effects beyond its antioxidant activity by influencing cannabinoid‐mediated pathways. This finding aligns with previous research indicating that β‐CBP selectively binds to CB2 receptors, reducing inflammation and oxidative damage [35, 36]. The significant increase in CB2 receptor expression in the β‐CBP‐treated groups implies that β‐CBP enhances cannabinoid receptor‐mediated signalling, potentially contributing to improved erectile function.
Iron‐induced lipid peroxidation accelerates the formation of free radicals, contributing to cellular and tissue damage [37]. β‐CBP's iron chelation capacity, demonstrated by its concentration‐dependent inhibition of Fe2+ (Figure 1f and Table 1), further supports its role as an antioxidant. Chelating agents are valuable secondary antioxidants because they lower the redox potential of metal ions, stabilising the oxidised forms and reducing oxidative stress [38]. The chelation of Fe2+ by β‐CBP, demonstrated through concentration‐dependent inhibition of lipid peroxidation, underscores its potential as a secondary antioxidant that stabilises oxidised metal ions and further mitigates oxidative stress.
Regarding receptor activity, β‐CBP significantly upregulated CB2 receptor expression compared to normal control and PIED rat groups (Figure 3). This finding aligns with previous studies demonstrating β‐CBP's modulation of CB2 receptor activity, contributing to its therapeutic effects [35, 36]. The upregulation of CB2 receptors suggests that β‐CBP enhances cannabinoid receptor‐mediated signalling pathways, which may help mitigate oxidative stress and improve erectile function. Interestingly, co‐administration of β‐CBP with the CB2 antagonist AM630 resulted in reduced CB2 receptor expression, underscoring the role of CB2 receptor activation in β‐CBP's effects. AM630's inhibition of CB2 receptor‐mediated responses correlates with the lower CB2 receptor expression observed in the PIED + AM630 group. This contrast highlights the specific involvement of CB2 receptors in mediating the protective effects of β‐CBP.
However, despite these promising findings, the study has some limitations that must be acknowledged. One key limitation is the lack of power calculations conducted prior to the experiment. This could impact the robustness of the findings and highlights the need for future studies to incorporate power analyses to ensure adequate sample sizes for detecting treatment effects. The potential colour interference at higher concentrations is a noted limitation, which affects the accuracy of the DPPH assay. Moreover, while β‐CBP demonstrated significant antioxidative and protective effects, further research is needed to elucidate the precise mechanisms by which it modulates CB2 receptor activity and its long‐term impact on male reproductive health.
5. Conclusion
This study demonstrates that β‐CBP exhibits potent antioxidant properties, including free radical scavenging, lipid peroxidation inhibition and iron chelation. Enhanced antioxidant enzyme activities, including SOD, catalase, GST and GPx, in paroxetine‐induced erectile dysfunction models accompanied these effects. Additionally, β‐CBP upregulated CB2 receptor expression, suggesting its involvement in cannabinoid‐mediated pathways, which may contribute to its protective effects on male sexual function. Despite lower reducing potential in comparison to sildenafil citrate, β‐CBP displayed significant antioxidant efficacy, indicating its potential as a therapeutic agent for ameliorating oxidative stress‐related male sexual dysfunction. Future research should focus on elucidating the precise mechanisms underlying β‐CBP's interaction with CB2 receptors and further validate its long‐term benefits in reproductive health.
Author Contributions
Adefegha S. A and Ganiyu Oboh. contributed to the study's conception, supervision and design. Material preparation and data collection were performed by Olopade E. O. and Fakayode A. E. The first draft of the manuscript was written by Alao J. O. Data analysis was performed by Olopade E. O. and Adepoju A. E. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
Acknowledgements
Open access publishing facilitated by Auckland University of Technology, as part of the Wiley ‐ Auckland University of Technology agreement via the Council of Australian University Librarians.
Funding: The authors received no specific funding for this work.
Data Availability Statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
References
- 1. Thawatchai P., Jintanaporn W., Sitthichai I., Supaporn M., and Wipawee T., “Moringa oleifera Leaves Extract Attenuates Male Sexual Dysfunction,” American Journal of Neuroscience 3, no. 1 (2012): 17–24, 10.3844/amjnsp.2012.17.24. [DOI] [Google Scholar]
- 2. Adefegha S. A., Oboh G., and Olopade E. O., “β‐Caryophyllene Improves Sexual Performance via Modulation of Crucial Enzymes Relevant to Erectile Dysfunction in Rats,” Toxicology Research 37, no. 2 (2021): 249–260, 10.1007/s43188-020-00061-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Chen M., Zhang Z., Zhou R., Li B., Jiang J., and Shi B., “The Relationship Between Oxidative Balance Score and Erectile Dysfunction in the U.S. Male Adult Population,” Scientific Reports 14, no. 1 (2024): 10746, 10.1038/s41598-024-61287-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Chang H. J., Kim J. M., Lee J. C., Kim W. K., and Chun H. S., “Protective Effect of β‐Caryophyllene, a Natural Bicyclic Sesquiterpene, Against Cerebral Ischemic Injury,” Journal of Medicinal Food 16, no. 6 (2013): 471–480, 10.1089/jmf.2012.2283. [DOI] [PubMed] [Google Scholar]
- 5. Di Sotto A., Maffei F., Hrelia P., Castelli F., Sarpietro M. G., and Mazzanti G., “Genotoxicity Assessment of β‐Caryophyllene Oxide,” Regulatory Toxicology and Pharmacology 66, no. 3 (2013): 264–268, 10.1016/j.yrtph.2013.04.006. [DOI] [PubMed] [Google Scholar]
- 6. Gushiken L. F. S., Beserra F. P., Hussni M. F., et al., “Beta‐Caryophyllene as an Antioxidant, Anti‐inflammatory and Re‐ Epithelialization Activities in a Rat Skin Wound Excision Model,” Oxidative Medicine and Cellular Longevity 2022 (2022): 1–21, 10.1155/2022/9004014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Tarumi W. and Shinohara K., “Olfactory Exposure to β‐Caryophyllene Increases Testosterone Levels in Women's Saliva,” Sexual Medicine 8, no. 3 (2020): 525–531, 10.1016/j.esxm.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Al‐Alami Z. M., Shraideh Z. A., and Taha M. O., “β‐Caryophyllene as Putative Male Contraceptive: Enhances Spermatogenesis but Not Spermiogenesis in Albino Rats,” Medicinal Chemistry Research 24, no. 11 (2015): 3876–3884, 10.1007/s00044-015-1428-3. [DOI] [Google Scholar]
- 9. Tveden‐Nyborg P., Bergmann T. K., Jessen N., Simonsen U., and Lykkesfeldt J., “BCPT 2023 Policy for Experimental and Clinical Studies,” Basic & Clinical Pharmacology & Toxicology 133, no. 4 (2023): 391–396, 10.1111/bcpt.13944. [DOI] [PubMed] [Google Scholar]
- 10. Gyamfi M. A., Yonamine M., and Aniya Y., “Free‐Radical Scavenging Action of Medicinal Herbs From Ghana,” General Pharmacology: The Vascular System 32, no. 6 (1999): 661–667, 10.1016/S0306-3623(98)00238-9. [DOI] [PubMed] [Google Scholar]
- 11. Puntel R. L., Nogueira C. W., and Rocha J. B. T., “Krebs Cycle Intermediates Modulate Thiobarbituric Acid Reactive Species (TBARS) Production in Rat Brain in Vitro,” Neurochemical Research 30, no. 2 (2005): 225–235, 10.1007/s11064-004-2445-7. [DOI] [PubMed] [Google Scholar]
- 12. Oyaizu M., “Studies on Products of Browning Reaction. Antioxidative Activities of Products of Browning Reaction Prepared From Glucosamine,” Japanese Journal of Nutrition and Dietetics 44, no. 6 (1986): 307–315, 10.5264/eiyogakuzashi.44.307. [DOI] [Google Scholar]
- 13. Re R., Pellegrini N., Proteggente A., Pannala A., Yang M., and Rice‐Evans C., “Antioxidant Activity Applying an Improved ABTS Radical Cation Decolorisation Assay,” Free Radical Biology and Medicine 26, no. 9–10 (1999): 1231–1237, 10.1016/S0891-5849(98)00315-3. [DOI] [PubMed] [Google Scholar]
- 14. Halliwell B. and Gutteridge J. M. C., “Formation of a Thiobarbituric‐Acid‐Reactive Substance From Deoxyribose in the Presence of iron Salts: The Role of Superoxide and Hydroxyl Radicals,” FEBS Letters 128, no. 2 (1981): 347–352, 10.1016/0014-5793(81)80114-7. [DOI] [PubMed] [Google Scholar]
- 15. Abbas M. A., Taha M. O., Zihlif M. A., and Disi A. M., “β‐Caryophyllene Causes Regression of Endometrial Implants in a Rat Model of Endometriosis Without Affecting Fertility,” European Journal of Pharmacology 702, no. 1–3 (2013): 12–19, 10.1016/j.ejphar.2013.01.011. [DOI] [PubMed] [Google Scholar]
- 16. Horváth B., Mukhopadhyay P., Kechrid M., et al., “β‐Caryophyllene Ameliorates Cisplatin‐Induced Nephrotoxicity in a Cannabinoid 2 Receptor‐Dependent Manner,” Free Radical Biology & Medicine 52, no. 8 (2012): 1325–1333, 10.1016/j.freeradbiomed.2012.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. He J., Zhou S., Li X., et al., “Pharmacokinetic Evaluation of β‐Caryophyllene Alcohol in Rats and Beagle Dogs,” Xenobiotica 48, no. 8 (2018): 845–850, 10.1080/00498254.2017.1367441. [DOI] [PubMed] [Google Scholar]
- 18. Adefegha S. A., Oyeleye S. I., Dada F. A., Olasehinde T. A., and Oboh G., “Modulatory Effect of Quercetin and Its Glycosylated Form on key Enzymes and Antioxidant Status in Rats Penile Tissue of Paroxetine‐Induced Erectile Dysfunction,” Biomedicine & Pharmacotherapy 107 (2018): 1473–1479, 10.1016/j.biopha.2018.08.128. [DOI] [PubMed] [Google Scholar]
- 19. Ågmo A., Turi A. L., Ellingsen E., and Kaspersen H., “Preclinical Models of Sexual Desire: Conceptual and Behavioral Analyses,” Pharmacology Biochemistry and Behavior 78, no. 3 (2004): 379–404, 10.1016/j.pbb.2004.04.013. [DOI] [PubMed] [Google Scholar]
- 20. Bertolin K. and Murphy B. D., “Monitoring Mouse Estrous Cycles,” in The Guide to Investigation of Mouse Pregnancy, (Elsevier, 2014), 475–477, 10.1016/B978-0-12-394445-0.00039-4. [DOI] [Google Scholar]
- 21. Ross R. A., Brockie H. C., Stevenson L. A., et al., “Agonist‐Inverse Agonist Characterisation at CB1 and CB2 Cannabinoid Receptors of L759633, L759656 and AM630,” British Journal of Pharmacology 126, no. 3 (1999): 665–672, 10.1038/sj.bjp.0702351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Munawar N., Oriowo M. A., and Masocha W., “Antihyperalgesic Activities of Endocannabinoids in a Mouse Model of Antiretroviral‐Induced Neuropathic Pain,” Frontiers in Pharmacology 8 (2017): 8, 10.3389/fphar.2017.00136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Argenziano M., Pota V., Di Paola A., et al., “CB2 Receptor as Emerging Anti‐inflammatory Target in Duchenne Muscular Dystrophy,” IJMS. 24, no. 4 (2023): 3345, 10.3390/ijms24043345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Misra H. P. and Fridovich I., “The Role of Superoxide Anion in the Autoxidation of Epinephrine and a Simple Assay for Superoxide Dismutase,” Journal of Biological Chemistry 247, no. 10 (1972): 3170–3175. [PubMed] [Google Scholar]
- 25. Claiborne A., “Catalase Activity,” in Handbook Methods for Oxygen Radical Research, 1st ed. (CRC Press, 1985), 2. [Google Scholar]
- 26. Jollow D. J., Mitchell J. R., Zampaglione N., and Gillette J. R., “Bromobenzene‐Induced Liver Necrosis. Protective Role of Glutathione and Evidence for 3,4‐Bromobenzene Oxide as the Hepatotoxic Metabolite,” Pharmacology 11, no. 3 (1974): 151–169, 10.1159/000136485. [DOI] [PubMed] [Google Scholar]
- 27. Mannervik B. and Guthenberg C., “Glutathione Transferase (Human Placenta),” in Methods in Enzymology, vol. 77, (Elsevier, 1981), 231–235, 10.1016/S0076-6879(81)77030-7. [DOI] [PubMed] [Google Scholar]
- 28. Scandiffio R., Geddo F., Cottone E., et al., “Protective Effects of (E)‐β‐Caryophyllene (BCP) in Chronic Inflammation,” Nutrients 12, no. 11 (2020): 3273, 10.3390/nu12113273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Ademosun A. O., Adebayo A. A., and Oboh G., “Anogeissus leiocarpus Attenuates Paroxetine‐Induced Erectile Dysfunction in Male Rats via Enhanced Sexual Behavior, Nitric Oxide Level and Antioxidant Status,” Biomedicine & Pharmacotherapy 111 (2019): 1029–1035, 10.1016/j.biopha.2019.01.022. [DOI] [PubMed] [Google Scholar]
- 30. Saikia Q., Hazarika A., and Kalita J. C., “Isoliquiritigenin Ameliorates Paroxetine‐Induced Sexual Dysfunction in Male Albino Mice,” Reproductive Toxicology 117 (2023): 108341, 10.1016/j.reprotox.2023.108341. [DOI] [PubMed] [Google Scholar]
- 31. Roychoudhury S., Chakraborty S., Choudhury A. P., et al., “Environmental Factors‐Induced Oxidative Stress: Hormonal and Molecular Pathway Disruptions in Hypogonadism and Erectile Dysfunction,” Antioxidants 10, no. 6 (2021): 837, 10.3390/antiox10060837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Kruk J., Aboul‐Enein B. H., Duchnik E., and Marchlewicz M., “Antioxidative Properties of Phenolic Compounds and Their Effect on Oxidative Stress Induced by Severe Physical Exercise,” Journal of Physiological Sciences 72, no. 1 (2022): 19, 10.1186/s12576-022-00845-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Kumar V., Mathela C. S., Kumar M., and Tewari G., “Antioxidant Potential of Essential Oils From Some Himalayan Asteraceae and Lamiaceae Species,” Medicine in Drug Discovery 1 (2019): 100004, 10.1016/j.medidd.2019.100004. [DOI] [Google Scholar]
- 34. Jomova K., Alomar S. Y., Alwasel S. H., Nepovimova E., Kuca K., and Valko M., “Several Lines of Antioxidant Defense Against Oxidative Stress: Antioxidant Enzymes, Nanomaterials With Multiple Enzyme‐Mimicking Activities, and Low‐Molecular‐Weight Antioxidants,” Archives of Toxicology 98, no. 5 (2024): 1323–1367, 10.1007/s00204-024-03696-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Hashiesh H. M., Sharma C., Goyal S. N., et al., “A Focused Review on CB2 Receptor‐Selective Pharmacological Properties and Therapeutic Potential of β‐Caryophyllene, a Dietary Cannabinoid,” Biomedicine & Pharmacotherapy 140 (2021): 111639, 10.1016/j.biopha.2021.111639. [DOI] [PubMed] [Google Scholar]
- 36. Ricardi C., Barachini S., Consoli G., Marazziti D., Polini B., and Chiellini G., “Beta‐Caryophyllene, a Cannabinoid Receptor Type 2 Selective Agonist, in Emotional and Cognitive Disorders,” International Journal of Molecular Sciences 25, no. 6 (2024): 3203, 10.3390/ijms25063203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Chaudhary P., Janmeda P., Docea A. O., et al., “Oxidative Stress, Free Radicals and Antioxidants: Potential Crosstalk in the Pathophysiology of Human Diseases,” Frontiers in Chemistry 11 (2023): 1158198, 10.3389/fchem.2023.1158198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Gulcin İ. and Alwasel S. H., “Metal Ions, Metal Chelators and Metal Chelating Assay as Antioxidant Method,” PRO 10, no. 1 (2022): 132, 10.3390/pr10010132. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.


