Abstract
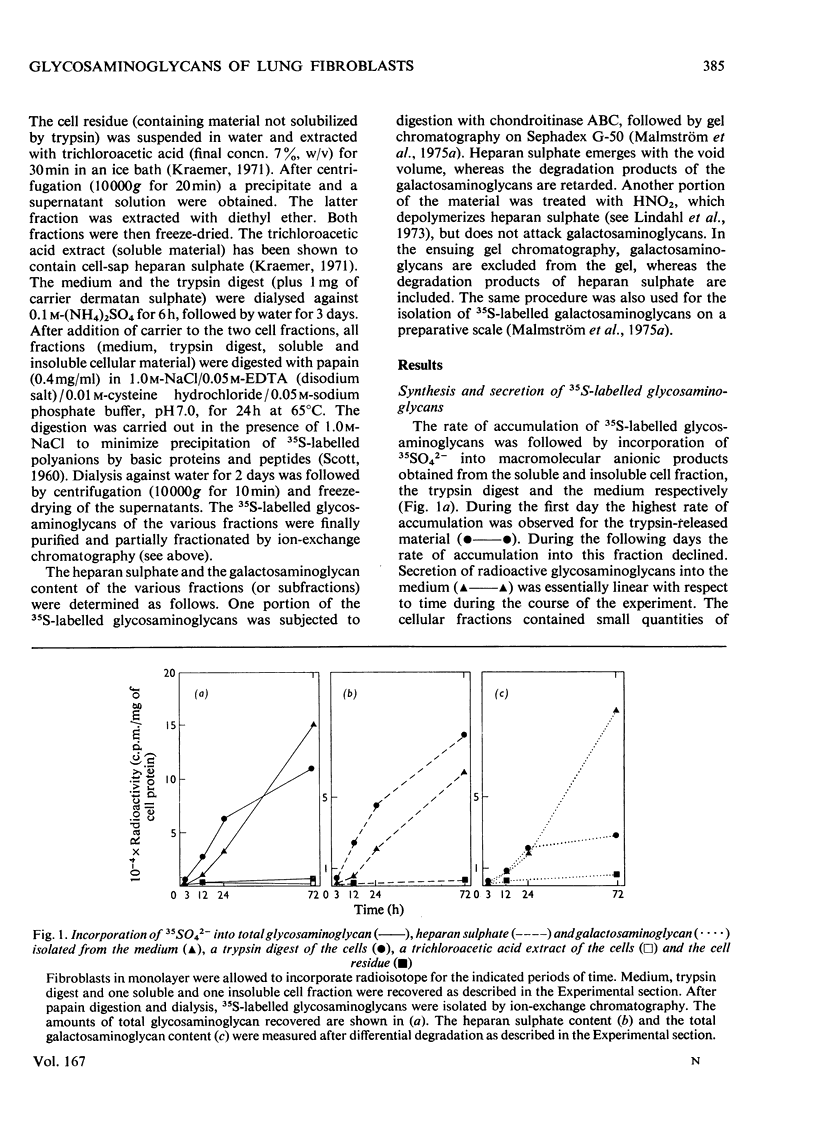
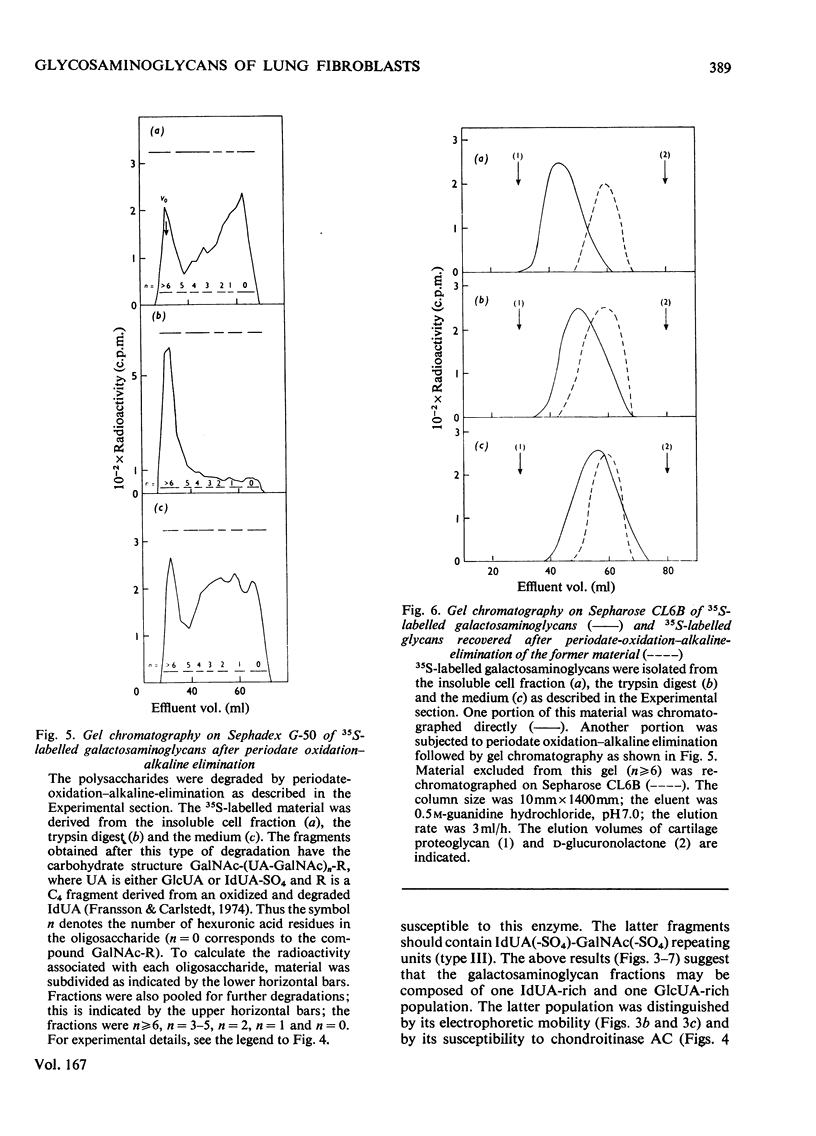
Foetal human lung fibroblasts, grown in monolayer, were allowed to incorporate 35SO42− for various periods of time. 35S-labelled macromolecular anionic products were isolated from the medium, a trypsin digest of the cells in monolayer and the cell residue. The various radioactive polysaccharides were identified as heparan sulphate and a galactosaminoglycan population (chondroitin sulphate and dermatan sulphate) by ion-exchange chromatography and by differential degradations with HNO2 and chondroitinase ABC. Most of the heparan sulphate was found in the trypsin digest, whereas the galactosaminoglycan components were largely confined to the medium. Electrophoretic studies on the various 35S-labelled galactosaminoglycans suggested the presence of a separate chondroitin sulphate component (i.e. a glucuronic acid-rich galactosaminoglycan). The 35S-labelled galactosaminoglycans were subjected to periodate oxidation of l-iduronic acid residues followed by scission in alkali. A periodate-resistant polymer fraction was obtained, which could be degraded to disaccharides by chondroitinase AC. However, most of the 35S-labelled galactosaminoglycans were extensively degraded by periodate oxidation–alkaline elimination. The oligosaccharides obtained were essentially resistant to chondroitinase AC, indicating that the iduronic acid-rich galactosaminoglycans (i.e. dermatan sulphate) were composed largely of repeating units containing sulphated or non-sulphated l-iduronic acid residues. The l-iduronic acid residues present in dermatan sulphate derived from the medium and the trypsin digest contained twice as much ester sulphate as did material associated with the cells. The content of d-glucuronic acid was low and similar in all three fractions. The relative distribution of glycosaminoglycans among the various fractions obtained from cultured lung fibroblasts was distinctly different from that of skin fibroblasts [Malmström, Carlstedt, Åberg & Fransson (1975) Biochem. J. 151, 477–489]. Moreover, subtle differences in co-polymeric structure of dermatan sulphate isolated from the two cell types could be detected.
Full text
PDF







Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Cöster L., Malmström A., Sjöberg I., Fransson L. The co-polymeric structure of pig skin dermatan sulphate. Distribution of L-iduronic acid sulphate residues in co-polymeric chains. Biochem J. 1975 Feb;145(2):379–389. doi: 10.1042/bj1450379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fransson L. A., Carlstedt I. Alkaline and smith degradation of oxidized dermatan sulphate-chondroitin sulphate copolymers. Carbohydr Res. 1974 Sep;36(2):349–358. doi: 10.1016/s0008-6215(00)83056-6. [DOI] [PubMed] [Google Scholar]
- Fransson L. A. Interaction between dermatan sulphate chains. I. Affinity chromatography of copolymeric galactosaminioglycans on dermatan sulphate-substituted agarose. Biochim Biophys Acta. 1976 Jun 23;437(1):106–115. doi: 10.1016/0304-4165(76)90351-2. [DOI] [PubMed] [Google Scholar]
- Fransson L. A. Periodate oxidation of L-iduronic acid residues in dermatan sulphate. Carbohydr Res. 1974 Sep;36(2):339–348. doi: 10.1016/s0008-6215(00)83055-4. [DOI] [PubMed] [Google Scholar]
- Kraemer P. M. Heparan sulfates of cultured cells. II. Acid-soluble and -precipitable species of different cell lines. Biochemistry. 1971 Apr 13;10(8):1445–1451. doi: 10.1021/bi00784a027. [DOI] [PubMed] [Google Scholar]
- Kresse H., von Figura K., Buddecke E., Fromme H. G. Metabolism of sulfated glycosaminoglycans in cultivated bovine arterial cells. I. Characterization of different pools of sulfated glycosaminoglycans. Hoppe Seylers Z Physiol Chem. 1975 Jun;356(6):929–941. doi: 10.1515/bchm2.1975.356.s1.929. [DOI] [PubMed] [Google Scholar]
- LAGUNOFF D., WARREN G. Determination of 2-deoxy-2-sulfoaminohexose content of mucopolysaccharides. Arch Biochem Biophys. 1962 Dec;99:396–400. doi: 10.1016/0003-9861(62)90285-0. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lindahl U., Bäckström G., Jansson L., Hallén A. Biosynthesis of heparin. II. Formation of sulfamino groups. J Biol Chem. 1973 Oct 25;248(20):7234–7241. [PubMed] [Google Scholar]
- Malmström A., Fransson L. A. Biosynthesis of dermatan sulfate. I. Formation of L-iduronic acid residues. J Biol Chem. 1975 May 10;250(9):3419–3425. [PubMed] [Google Scholar]
- Malström A., Carlstedt I., Aberg L., Fransson L. A. The copolymeric structure of dermatan sulphate produced by cultured human fibroblasts. Different distribution of iduronic acid and glucuronic acid-containing units in soluble and cell-associated glycans. Biochem J. 1975 Dec;151(3):477–489. doi: 10.1042/bj1510477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SCOTT J. E. Aliphatic ammonium salts in the assay of acidic polysaccharides from tissues. Methods Biochem Anal. 1960;8:145–197. doi: 10.1002/9780470110249.ch4. [DOI] [PubMed] [Google Scholar]
- Saito H., Uzman B. G. Production and secretion of chondroitin sulfates and dermatan sulfate by established mammalian cell lines. Biochem Biophys Res Commun. 1971 May 21;43(4):723–728. doi: 10.1016/0006-291x(71)90675-9. [DOI] [PubMed] [Google Scholar]
- Wessler E. Analytical and preparative separation of acidic glycosaminoglycans by electrophoresis in barium acetate. Anal Biochem. 1968 Dec;26(3):439–444. doi: 10.1016/0003-2697(68)90205-4. [DOI] [PubMed] [Google Scholar]
- Wight T. N., Ross R. Proteoglycans in primate arteries. II. Synthesis and secretion of glycosaminoglycans by arterial smooth muscle cells in culture. J Cell Biol. 1975 Dec;67(3):675–686. doi: 10.1083/jcb.67.3.675. [DOI] [PMC free article] [PubMed] [Google Scholar]


