Abstract
1. To study the regulation of calcium influx in non-excitable cells, membrane currents of rat peritoneal mast cells were recorded using the whole-cell patch-clamp technique. At the same time, intracellular calcium concentration ([Ca2+]i) was monitored via the fluorescent calcium-indicator dye Fura-2, which was loaded into cells by diffusion from the patch pipette. 2. Stimulation of mast cells with secretagogues, such as compound 48/80 or substance P, caused release of Ca2+ from internal stores. In addition, external agonists also induced influx of external calcium in 26% of the cells investigated. The agonist-stimulated Ca2+ influx was increased during membrane hyperpolarization and was associated with small whole-cell currents. 3. Likewise, internal application of inositol 1,4,5-trisphosphate (Ins1,4,5P3:0.5-10 microM) elevated [Ca2+]i due both to release of Ca2+ from internal stores and to influx of external calcium. The Ins1,4,5P3-induced influx was greater at more negative membrane potentials, suggesting that Ins1,4,5P3 opened a pathway through which calcium could enter at a rate governed by its electrochemical driving force. 4. Inositol 1,3,4,5-tetrakisphosphate (Ins1,3,4,5P4) did not induce Ca2+ influx by itself nor did it facilitate or enhance Ins1,4,5P3-induced Ca2+ entry. Calcium influx was also induced by inositol 2,4,5-trisphosphate. Since this inositol phosphate is a poor substrate for Ins1,4,5P3 3-kinase it seems unlikely that Ins1,3,4,5P4 plays a role in the regulation of the Ca2(+)-influx pathway in mast cells. 5. The Ins1,4,5P3-induced Ca2+ influx was associated with whole-cell currents of 1-2 pA or less, with no channel activity detectable in whole-cell recordings. The small size of the whole-cell current suggests either that the Ins1,4,5P3-dependent influx occurs via small-conductance channels that are highly calcium specific or that the influx is not via ion channels. 6. Agonist stimulation also activated large-conductance (ca 50 pS) cation channels, through which divalent cations could permeate; thus, these channels represent a second pathway for Ca2+ influx. The slow speed of activation of the channels by agonists, their activation by internal guanosine 5'-O-(3-thiotriphosphate) (GTP-gamma-S), and the inhibition of agonist activation by internal guanosine 5'-O-(2-thiodiphosphate) (GDP-beta-S) all suggest that the 50 pS channels are regulated by a second messenger and/or a GTP-binding protein. The activity of the 50 pS channel in mast cells is not sensitive to either Ins1,4,5P3 or Ins1,3,4,5P4. Activity of the channel was inhibited by elevated [Ca2+]i.(ABSTRACT TRUNCATED AT 400 WORDS)
Full text
PDF






















Selected References
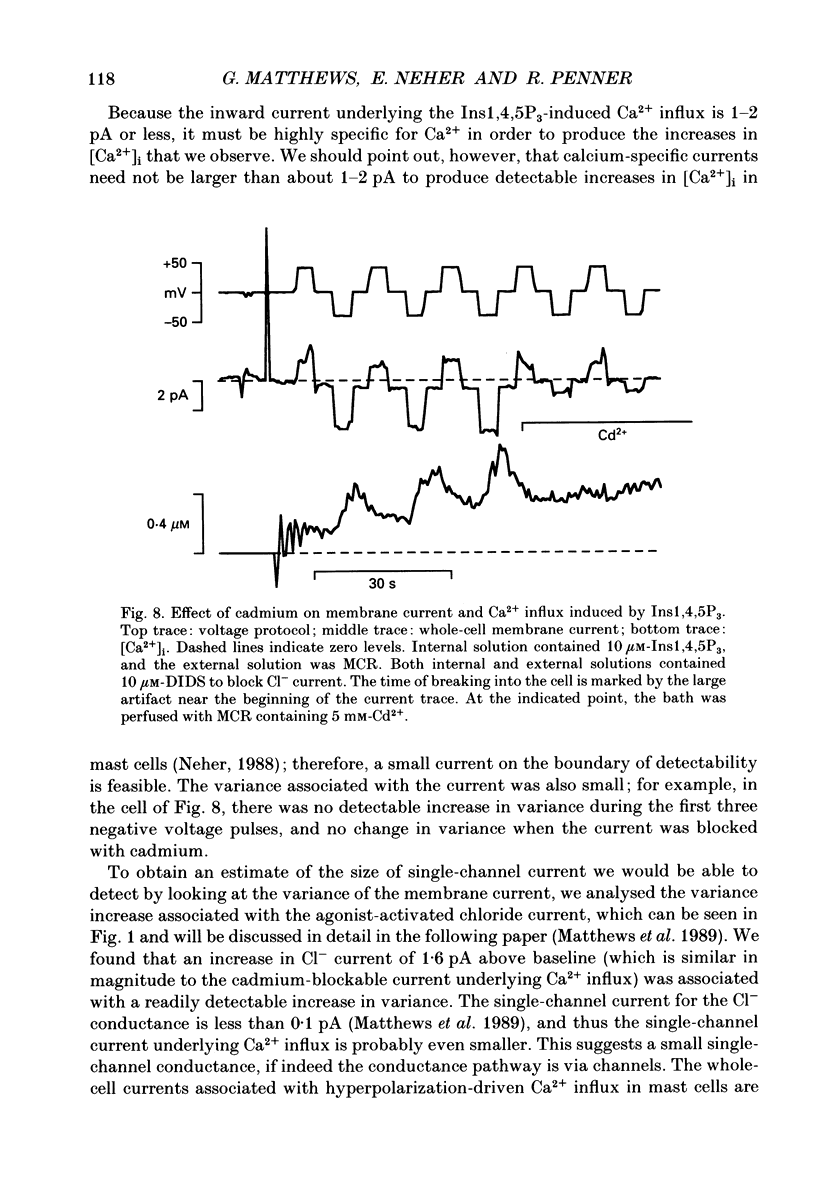
These references are in PubMed. This may not be the complete list of references from this article.
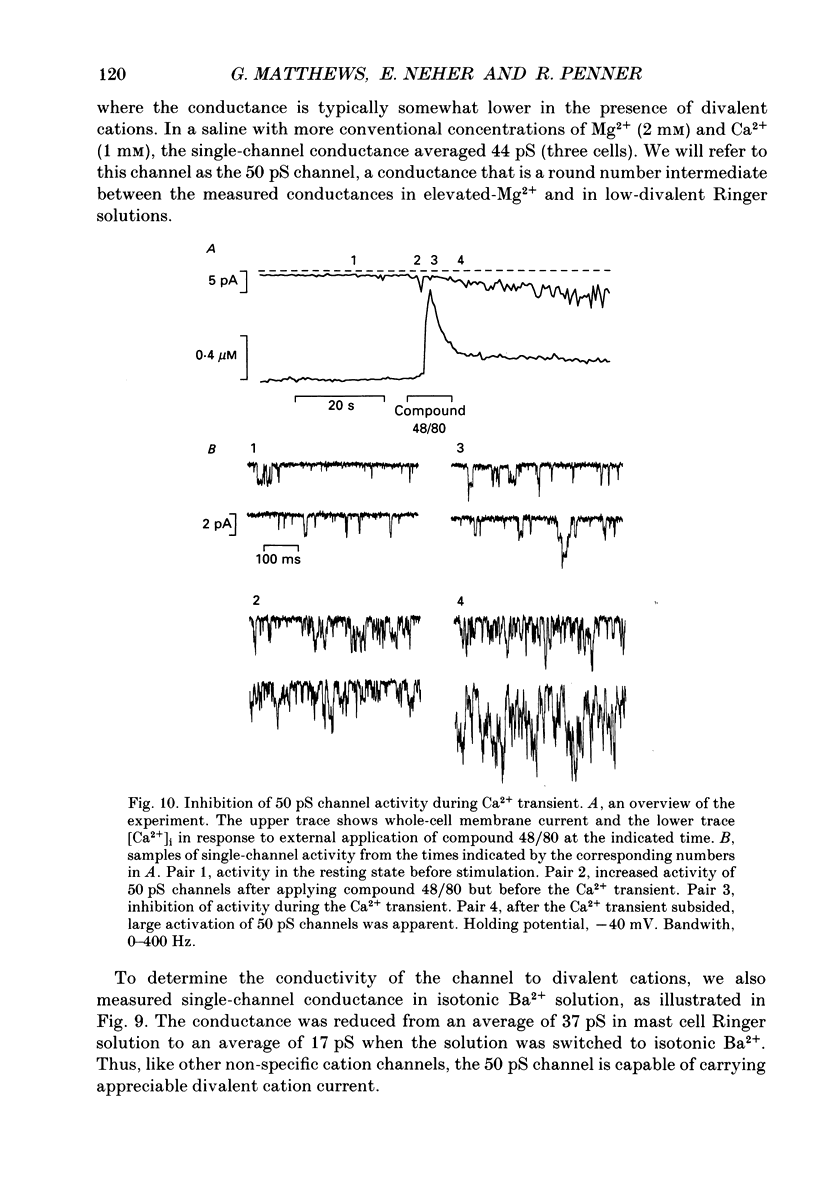
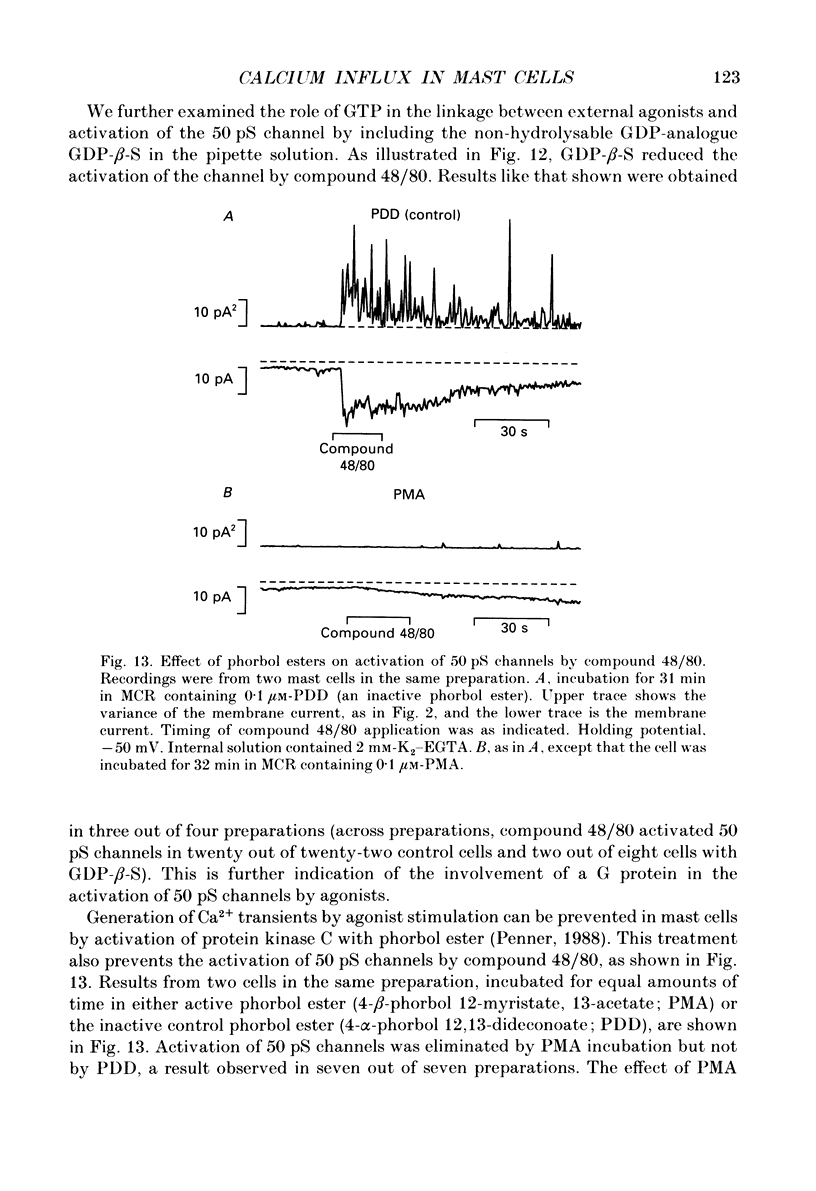
- Almers W., Neher E. The Ca signal from fura-2 loaded mast cells depends strongly on the method of dye-loading. FEBS Lett. 1985 Nov 11;192(1):13–18. doi: 10.1016/0014-5793(85)80033-8. [DOI] [PubMed] [Google Scholar]
- Beaven M. A., Moore J. P., Smith G. A., Hesketh T. R., Metcalfe J. C. The calcium signal and phosphatidylinositol breakdown in 2H3 cells. J Biol Chem. 1984 Jun 10;259(11):7137–7142. [PubMed] [Google Scholar]
- Bennett J. P., Cockcroft S., Gomperts B. D. Rat mast cells permeabilized with ATP secrete histamine in response to calcium ions buffered in the micromolar range. J Physiol. 1981 Aug;317:335–345. doi: 10.1113/jphysiol.1981.sp013828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cockcroft S., Howell T. W., Gomperts B. D. Two G-proteins act in series to control stimulus-secretion coupling in mast cells: use of neomycin to distinguish between G-proteins controlling polyphosphoinositide phosphodiesterase and exocytosis. J Cell Biol. 1987 Dec;105(6 Pt 1):2745–2750. doi: 10.1083/jcb.105.6.2745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grynkiewicz G., Poenie M., Tsien R. Y. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem. 1985 Mar 25;260(6):3440–3450. [PubMed] [Google Scholar]
- Hallam T. J., Rink T. J. Receptor-mediated Ca2+ entry: diversity of function and mechanism. Trends Pharmacol Sci. 1989 Jan;10(1):8–10. doi: 10.1016/0165-6147(89)90092-8. [DOI] [PubMed] [Google Scholar]
- Haynes L. W., Kay A. R., Yau K. W. Single cyclic GMP-activated channel activity in excised patches of rod outer segment membrane. Nature. 1986 May 1;321(6065):66–70. doi: 10.1038/321066a0. [DOI] [PubMed] [Google Scholar]
- Hockberger P. E., Swandulla D. Direct ion channel gating: a new function for intracellular messengers. Cell Mol Neurobiol. 1987 Sep;7(3):229–236. doi: 10.1007/BF00711301. [DOI] [PubMed] [Google Scholar]
- Irvine R. F., Brown K. D., Berridge M. J. Specificity of inositol trisphosphate-induced calcium release from permeabilized Swiss-mouse 3T3 cells. Biochem J. 1984 Aug 15;222(1):269–272. doi: 10.1042/bj2220269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irvine R. F., Moor R. M. Micro-injection of inositol 1,3,4,5-tetrakisphosphate activates sea urchin eggs by a mechanism dependent on external Ca2+. Biochem J. 1986 Dec 15;240(3):917–920. doi: 10.1042/bj2400917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kashket E. R., Wong P. T. The intracellular pH of Escherichia coli. Biochim Biophys Acta. 1969 Oct 14;193(1):212–214. doi: 10.1016/0005-2736(69)90074-1. [DOI] [PubMed] [Google Scholar]
- Kennerly D. A., Sullivan T. J., Parker C. W. Activation of phospholipid metabolism during mediator release from stimulated rat mast cells. J Immunol. 1979 Jan;122(1):152–159. [PubMed] [Google Scholar]
- Kuno M., Gardner P. Ion channels activated by inositol 1,4,5-trisphosphate in plasma membrane of human T-lymphocytes. Nature. 1987 Mar 19;326(6110):301–304. doi: 10.1038/326301a0. [DOI] [PubMed] [Google Scholar]
- Kuno M., Okada T., Shibata T. A patch-clamp study: secretagogue-induced currents in rat peritoneal mast cells. Am J Physiol. 1989 Mar;256(3 Pt 1):C560–C568. doi: 10.1152/ajpcell.1989.256.3.C560. [DOI] [PubMed] [Google Scholar]
- Lindau M., Fernandez J. M. IgE-mediated degranulation of mast cells does not require opening of ion channels. Nature. 1986 Jan 9;319(6049):150–153. doi: 10.1038/319150a0. [DOI] [PubMed] [Google Scholar]
- Llano I., Marty A., Tanguy J. Dependence of intracellular effects of GTP gamma S and inositoltrisphosphate on cell membrane potential and on external Ca ions. Pflugers Arch. 1987 Aug;409(4-5):499–506. doi: 10.1007/BF00583807. [DOI] [PubMed] [Google Scholar]
- Matthews G., Neher E., Penner R. Chloride conductance activated by external agonists and internal messengers in rat peritoneal mast cells. J Physiol. 1989 Nov;418:131–144. doi: 10.1113/jphysiol.1989.sp017831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayer M. L., Westbrook G. L. Permeation and block of N-methyl-D-aspartic acid receptor channels by divalent cations in mouse cultured central neurones. J Physiol. 1987 Dec;394:501–527. doi: 10.1113/jphysiol.1987.sp016883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meldolesi J., Pozzan T. Pathways of Ca2+ influx at the plasma membrane: voltage-, receptor-, and second messenger-operated channels. Exp Cell Res. 1987 Aug;171(2):271–283. doi: 10.1016/0014-4827(87)90161-3. [DOI] [PubMed] [Google Scholar]
- Morris A. P., Gallacher D. V., Irvine R. F., Petersen O. H. Synergism of inositol trisphosphate and tetrakisphosphate in activating Ca2+-dependent K+ channels. Nature. 1987 Dec 17;330(6149):653–655. doi: 10.1038/330653a0. [DOI] [PubMed] [Google Scholar]
- Nakamura T., Ui M. Simultaneous inhibitions of inositol phospholipid breakdown, arachidonic acid release, and histamine secretion in mast cells by islet-activating protein, pertussis toxin. A possible involvement of the toxin-specific substrate in the Ca2+-mobilizing receptor-mediated biosignaling system. J Biol Chem. 1985 Mar 25;260(6):3584–3593. [PubMed] [Google Scholar]
- Penner R., Matthews G., Neher E. Regulation of calcium influx by second messengers in rat mast cells. Nature. 1988 Aug 11;334(6182):499–504. doi: 10.1038/334499a0. [DOI] [PubMed] [Google Scholar]
- Penner R. Multiple signaling pathways control stimulus-secretion coupling in rat peritoneal mast cells. Proc Natl Acad Sci U S A. 1988 Dec;85(24):9856–9860. doi: 10.1073/pnas.85.24.9856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penner R., Neher E. Secretory responses of rat peritoneal mast cells to high intracellular calcium. FEBS Lett. 1988 Jan 4;226(2):307–313. doi: 10.1016/0014-5793(88)81445-5. [DOI] [PubMed] [Google Scholar]
- Penner R., Neher E. The role of calcium in stimulus-secretion coupling in excitable and non-excitable cells. J Exp Biol. 1988 Sep;139:329–345. doi: 10.1242/jeb.139.1.329. [DOI] [PubMed] [Google Scholar]
- Penner R., Pusch M., Neher E. Washout phenomena in dialyzed mast cells allow discrimination of different steps in stimulus-secretion coupling. Biosci Rep. 1987 Apr;7(4):313–321. doi: 10.1007/BF01121453. [DOI] [PubMed] [Google Scholar]
- Snyder P. M., Krause K. H., Welsh M. J. Inositol trisphosphate isomers, but not inositol 1,3,4,5-tetrakisphosphate, induce calcium influx in Xenopus laevis oocytes. J Biol Chem. 1988 Aug 15;263(23):11048–11051. [PubMed] [Google Scholar]


