Abstract
1. The kinetic properties of T-type Ca2+ channels were examined in single ventricular cells from guinea-pig hearts using the cell-attached configuration of the patch-clamp technique. 2. T-type Ca2+ channel activity has been observed in 44 out of 139 patches. The density of these channels was estimated at 0.1-0.3 micron-2. The T-type Ca2+ channel responds to a depolarizing voltage step either with a burst of openings which appears with a distinct delay or with no openings at all. The mean number of bursts per record for the records showing channel activity is 1.1. The probability of observing a blank sweep is high and amounts to 0.65 +/- 0.02 (n = 26). 3. With 110 mM-Ca2+ in the pipette solution, the slope conductance calculated from the current-voltage relationship of the single-channel current in the range between -50 and +10 mV is 6.8 pS. 4. Openings to a subconductance level of about 50% of the main level could be resolved. All possible transitions between the subconductance and the main level were observed, indicating that the cardiac T-type Ca2+ channel possesses a substate. 5. The macroscopic steady-state activation and inactivation, as determined from ensemble-averaged currents, could be described by Boltzmann functions. Half-maximal activation and inactivation occur at -14 and -60.7 mV, the slope parameters of these curves are 10.8 and 5.6 mV respectively. The maximum (peak) open probability is 0.15. 6. The ensemble-averaged current decays monoexponentially. The time constant is strongly voltage dependent and decreases at less negative potentials. 7. The open times are monoexponentially distributed. The mean open time of the channel does not depend on either the holding or the test potential, and has a mean value of 1.4 ms. The distribution of the closed times is biexponential. The fast mean closed time is also voltage independent with a mean value of 0.48 ms. The slow mean closed time increases with voltage from 1.9 ms at -40 mV to 8.8 ms at 0 mV. The mean burst duration also increases with voltage from a value of 4.9 ms at -40 mV to 13.9 ms at -10 mV. 8. The convolution of the first-latency distribution with that of the burst duration closely fits the open probability calculated from the ensemble-averaged current. The mean first latency is also closely correlated with the macroscopic time constant of inactivation.(ABSTRACT TRUNCATED AT 400 WORDS)
Full text
PDF



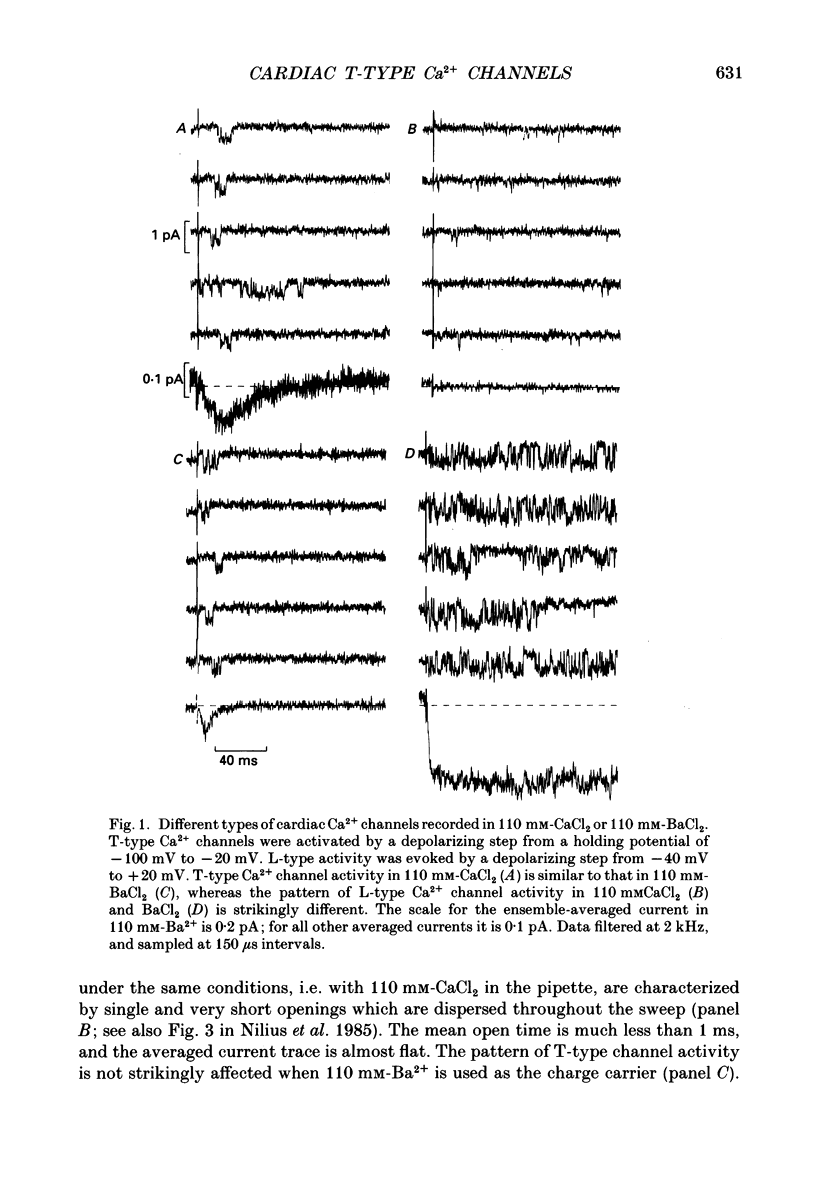

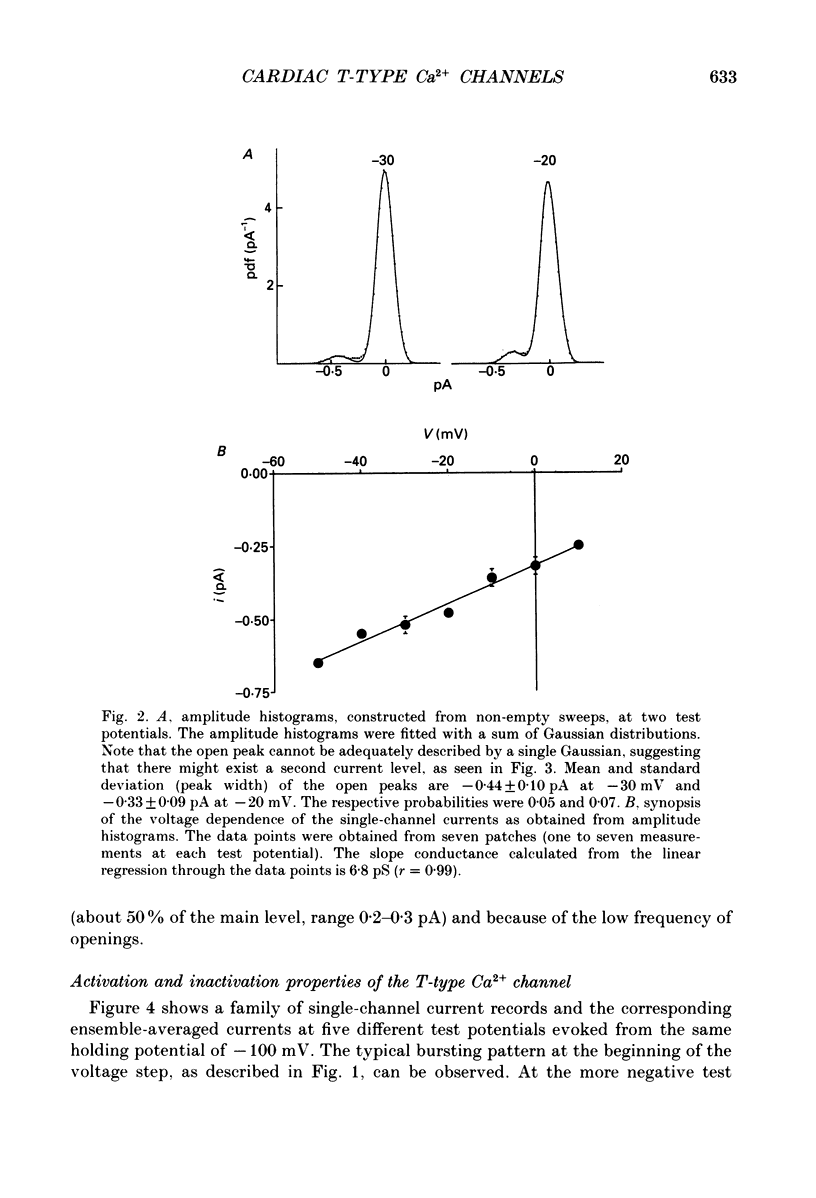
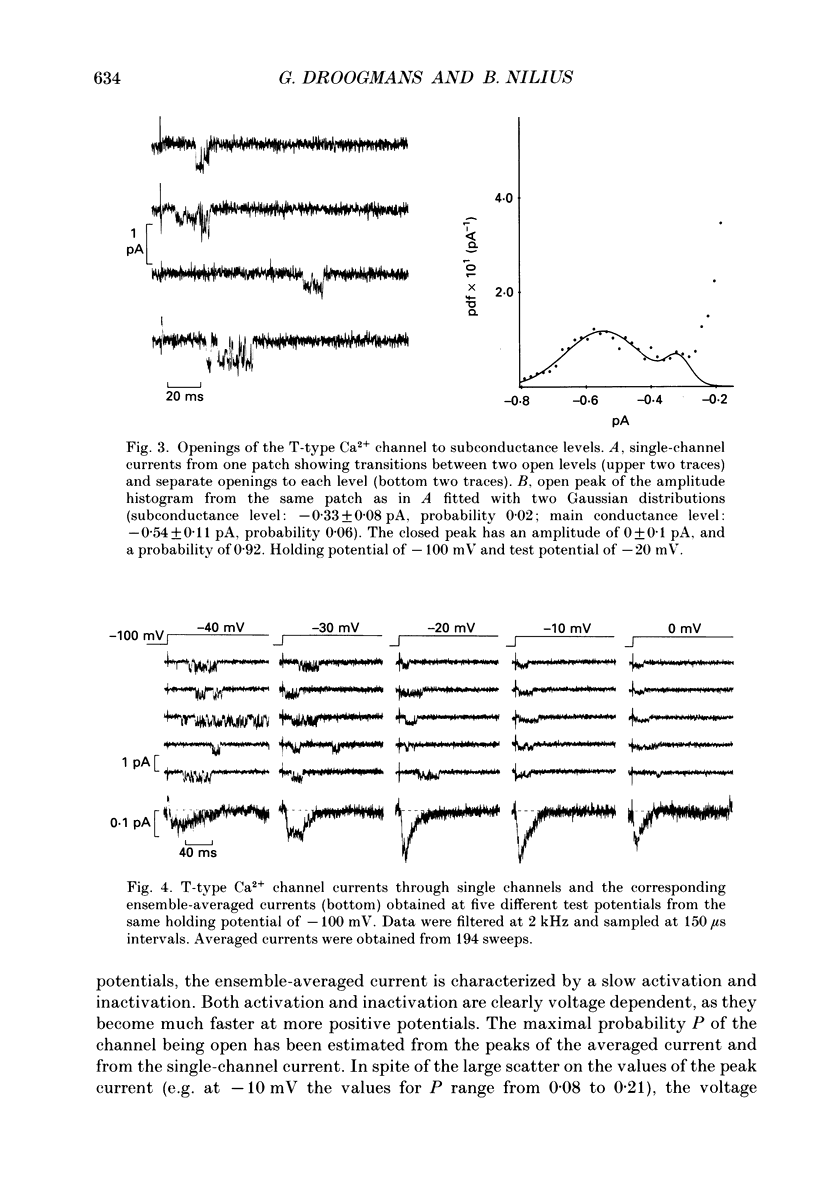

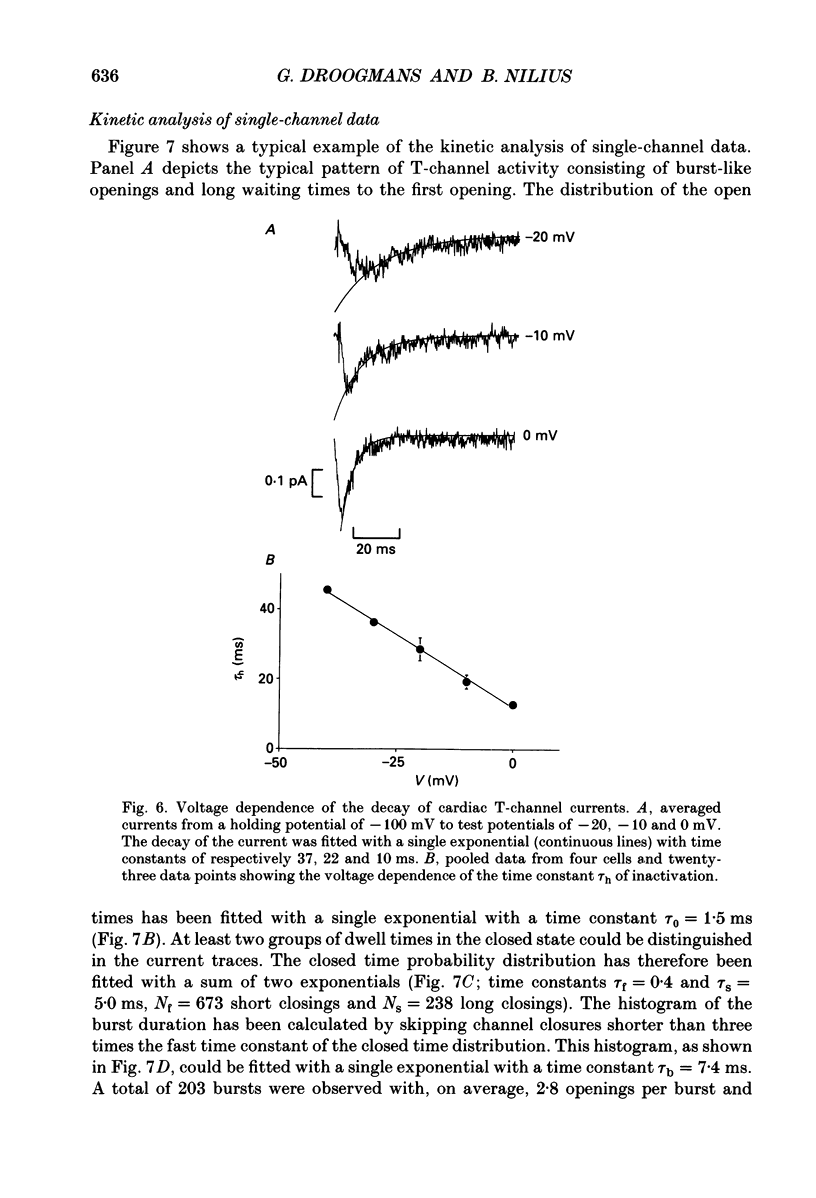











Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aldrich R. W., Corey D. P., Stevens C. F. A reinterpretation of mammalian sodium channel gating based on single channel recording. Nature. 1983 Dec 1;306(5942):436–441. doi: 10.1038/306436a0. [DOI] [PubMed] [Google Scholar]
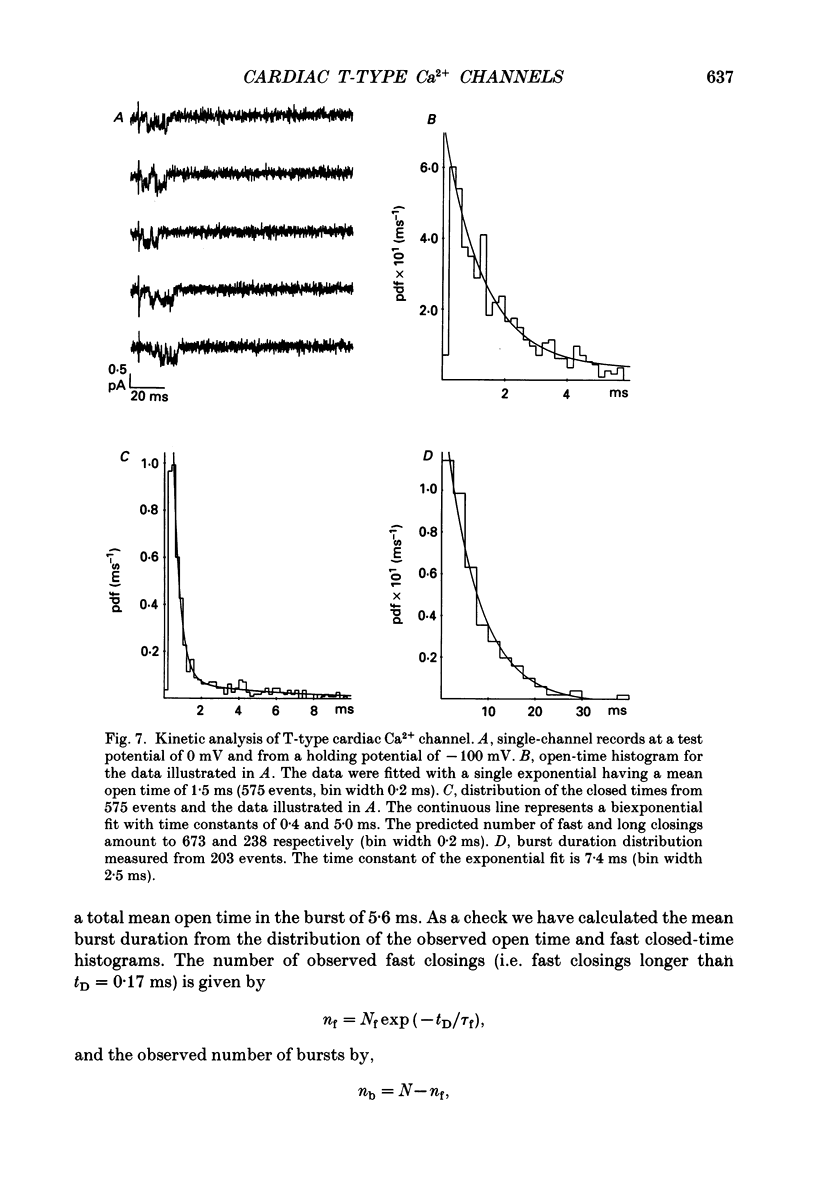
- Bean B. P. Two kinds of calcium channels in canine atrial cells. Differences in kinetics, selectivity, and pharmacology. J Gen Physiol. 1985 Jul;86(1):1–30. doi: 10.1085/jgp.86.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
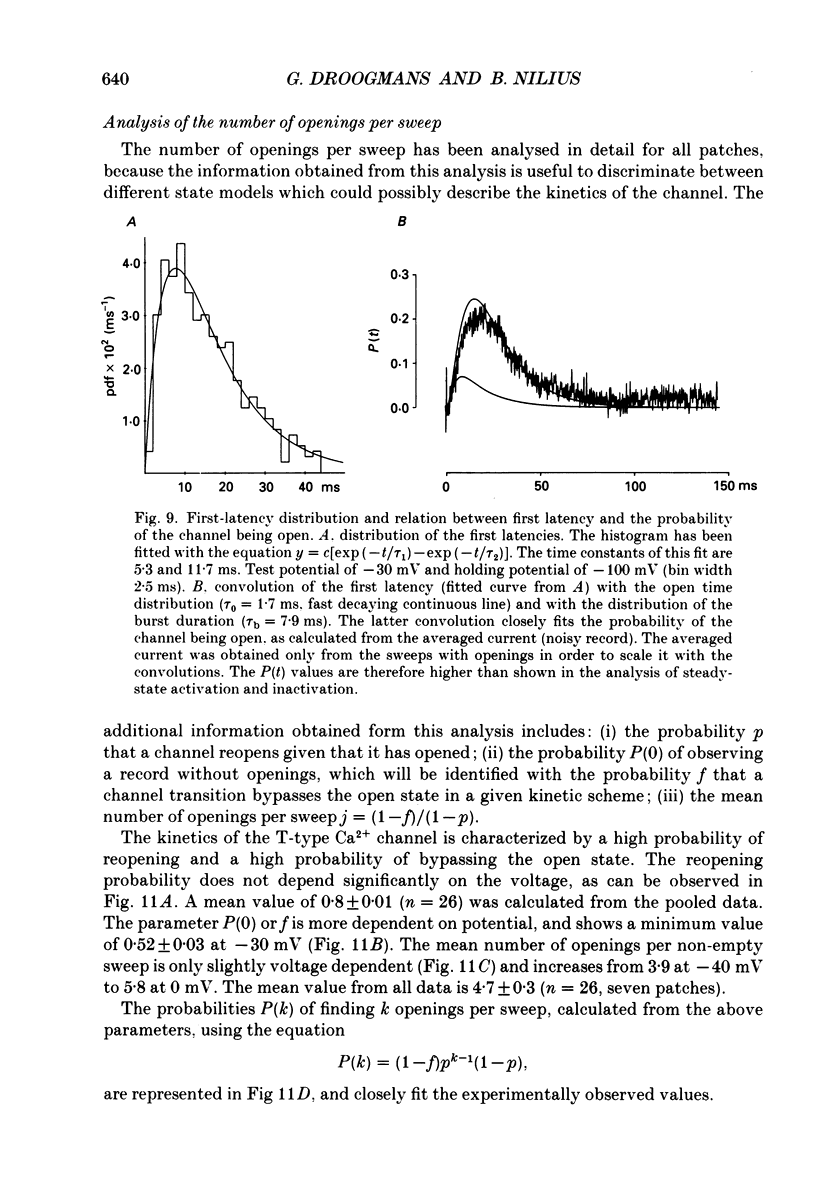
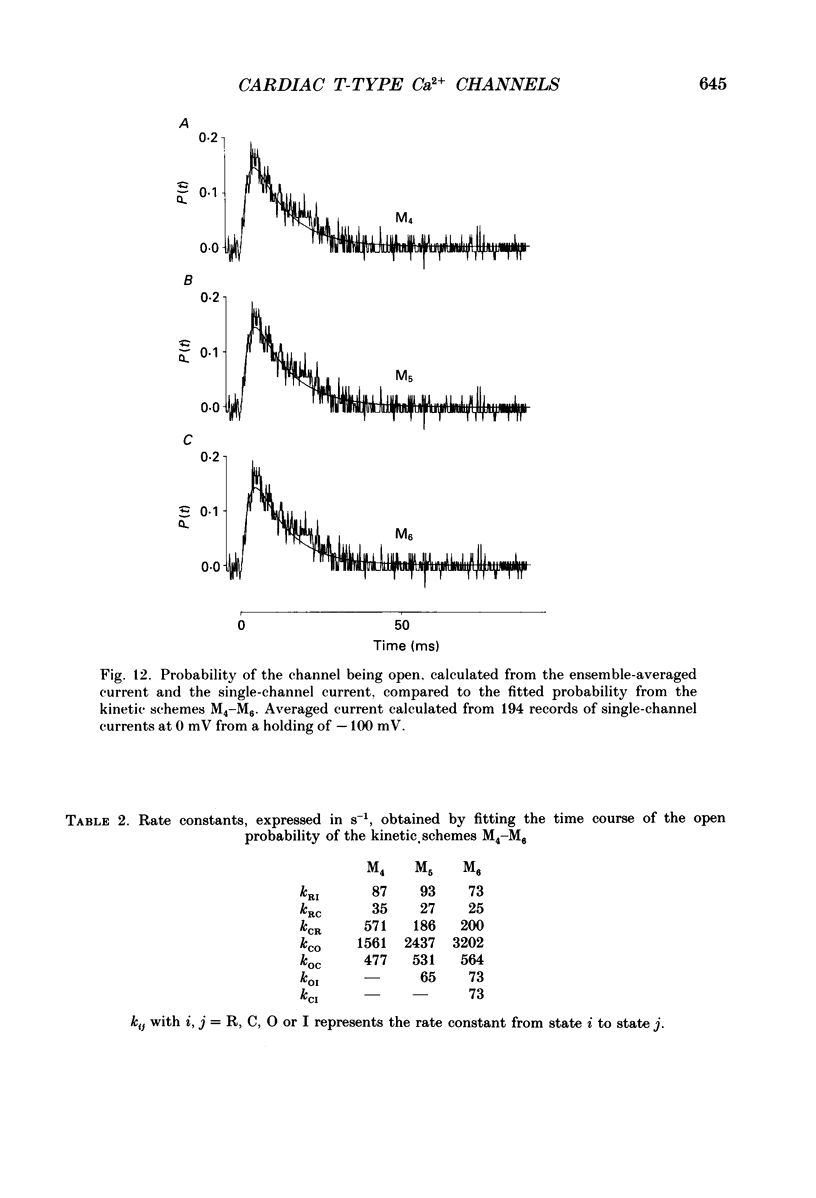
- Bonvallet R. A low threshold calcium current recorded at physiological Ca concentrations in single frog atrial cells. Pflugers Arch. 1987 May;408(5):540–542. doi: 10.1007/BF00585084. [DOI] [PubMed] [Google Scholar]
- Brum G., Osterrieder W., Trautwein W. Beta-adrenergic increase in the calcium conductance of cardiac myocytes studied with the patch clamp. Pflugers Arch. 1984 Jun;401(2):111–118. doi: 10.1007/BF00583870. [DOI] [PubMed] [Google Scholar]
- Carbone E., Lux H. D. A low voltage-activated, fully inactivating Ca channel in vertebrate sensory neurones. Nature. 1984 Aug 9;310(5977):501–502. doi: 10.1038/310501a0. [DOI] [PubMed] [Google Scholar]
- Carbone E., Lux H. D. Kinetics and selectivity of a low-voltage-activated calcium current in chick and rat sensory neurones. J Physiol. 1987 May;386:547–570. doi: 10.1113/jphysiol.1987.sp016551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carbone E., Lux H. D. Single low-voltage-activated calcium channels in chick and rat sensory neurones. J Physiol. 1987 May;386:571–601. doi: 10.1113/jphysiol.1987.sp016552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavalié A., Pelzer D., Trautwein W. Fast and slow gating behaviour of single calcium channels in cardiac cells. Relation to activation and inactivation of calcium-channel current. Pflugers Arch. 1986 Mar;406(3):241–258. doi: 10.1007/BF00640910. [DOI] [PubMed] [Google Scholar]
- Cota G. Calcium channel currents in pars intermedia cells of the rat pituitary gland. Kinetic properties and washout during intracellular dialysis. J Gen Physiol. 1986 Jul;88(1):83–105. doi: 10.1085/jgp.88.1.83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deitmer J. W. Evidence for two voltage-dependent calcium currents in the membrane of the ciliate Stylonychia. J Physiol. 1984 Oct;355:137–159. doi: 10.1113/jphysiol.1984.sp015411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox A. P., Nowycky M. C., Tsien R. W. Kinetic and pharmacological properties distinguishing three types of calcium currents in chick sensory neurones. J Physiol. 1987 Dec;394:149–172. doi: 10.1113/jphysiol.1987.sp016864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox A. P., Nowycky M. C., Tsien R. W. Single-channel recordings of three types of calcium channels in chick sensory neurones. J Physiol. 1987 Dec;394:173–200. doi: 10.1113/jphysiol.1987.sp016865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagiwara N., Irisawa H., Kameyama M. Contribution of two types of calcium currents to the pacemaker potentials of rabbit sino-atrial node cells. J Physiol. 1988 Jan;395:233–253. doi: 10.1113/jphysiol.1988.sp016916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamill O. P., Marty A., Neher E., Sakmann B., Sigworth F. J. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 1981 Aug;391(2):85–100. doi: 10.1007/BF00656997. [DOI] [PubMed] [Google Scholar]
- Hiriart M., Matteson D. R. Na channels and two types of Ca channels in rat pancreatic B cells identified with the reverse hemolytic plaque assay. J Gen Physiol. 1988 May;91(5):617–639. doi: 10.1085/jgp.91.5.617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horn R., Vandenberg C. A., Lange K. Statistical analysis of single sodium channels. Effects of N-bromoacetamide. Biophys J. 1984 Jan;45(1):323–335. doi: 10.1016/S0006-3495(84)84158-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horn R., Vandenberg C. A. Statistical properties of single sodium channels. J Gen Physiol. 1984 Oct;84(4):505–534. doi: 10.1085/jgp.84.4.505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohlhardt M., Fröbe U., Herzig J. W. Properties of normal and non-inactivating single cardiac Na+ channels. Proc R Soc Lond B Biol Sci. 1987 Oct 22;232(1266):71–93. doi: 10.1098/rspb.1987.0062. [DOI] [PubMed] [Google Scholar]
- Kunze D. L., Lacerda A. E., Wilson D. L., Brown A. M. Cardiac Na currents and the inactivating, reopening, and waiting properties of single cardiac Na channels. J Gen Physiol. 1985 Nov;86(5):691–719. doi: 10.1085/jgp.86.5.691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Llinás R., Yarom Y. Properties and distribution of ionic conductances generating electroresponsiveness of mammalian inferior olivary neurones in vitro. J Physiol. 1981 Jun;315:569–584. doi: 10.1113/jphysiol.1981.sp013764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mackie G. O., Meech R. W. Separate sodium and calcium spikes in the same axon. 1985 Feb 28-Mar 6Nature. 313(6005):791–793. doi: 10.1038/313791a0. [DOI] [PubMed] [Google Scholar]
- McCleskey E. W., Fox A. P., Feldman D., Tsien R. W. Different types of calcium channels. J Exp Biol. 1986 Sep;124:177–190. doi: 10.1242/jeb.124.1.177. [DOI] [PubMed] [Google Scholar]
- Mitra R., Morad M. A uniform enzymatic method for dissociation of myocytes from hearts and stomachs of vertebrates. Am J Physiol. 1985 Nov;249(5 Pt 2):H1056–H1060. doi: 10.1152/ajpheart.1985.249.5.H1056. [DOI] [PubMed] [Google Scholar]
- Mitra R., Morad M. Two types of calcium channels in guinea pig ventricular myocytes. Proc Natl Acad Sci U S A. 1986 Jul;83(14):5340–5344. doi: 10.1073/pnas.83.14.5340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nilius B., Hess P., Lansman J. B., Tsien R. W. A novel type of cardiac calcium channel in ventricular cells. Nature. 1985 Aug 1;316(6027):443–446. doi: 10.1038/316443a0. [DOI] [PubMed] [Google Scholar]
- Nilius B., Vereecke J., Carmeliet E. Different conductance states of the bursting Na channel in guinea-pig ventricular myocytes. Pflugers Arch. 1989 Jan;413(3):242–248. doi: 10.1007/BF00583536. [DOI] [PubMed] [Google Scholar]
- Nowycky M. C., Fox A. P., Tsien R. W. Three types of neuronal calcium channel with different calcium agonist sensitivity. Nature. 1985 Aug 1;316(6027):440–443. doi: 10.1038/316440a0. [DOI] [PubMed] [Google Scholar]
- Patlak J., Horn R. Effect of N-bromoacetamide on single sodium channel currents in excised membrane patches. J Gen Physiol. 1982 Mar;79(3):333–351. doi: 10.1085/jgp.79.3.333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pietrobon D., Prod'hom B., Hess P. Conformational changes associated with ion permeation in L-type calcium channels. Nature. 1988 May 26;333(6171):373–376. doi: 10.1038/333373a0. [DOI] [PubMed] [Google Scholar]
- Prod'hom B., Pietrobon D., Hess P. Direct measurement of proton transfer rates to a group controlling the dihydropyridine-sensitive Ca2+ channel. Nature. 1987 Sep 17;329(6136):243–246. doi: 10.1038/329243a0. [DOI] [PubMed] [Google Scholar]
- Reuter H., Kokubun S., Prod'hom B. Properties and modulation of cardiac calcium channels. J Exp Biol. 1986 Sep;124:191–201. doi: 10.1242/jeb.124.1.191. [DOI] [PubMed] [Google Scholar]
- Satin L. S., Cook D. L. Evidence for two calcium currents in insulin-secreting cells. Pflugers Arch. 1988 Apr;411(4):401–409. doi: 10.1007/BF00587719. [DOI] [PubMed] [Google Scholar]
- Scanley B. E., Fozzard H. A. Low conductance sodium channels in canine cardiac Purkinje cells. Biophys J. 1987 Sep;52(3):489–495. doi: 10.1016/S0006-3495(87)83237-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tytgat J., Nilius B., Vereecke J., Carmeliet E. The T-type Ca channel in guinea-pig ventricular myocytes is insensitive to isoproterenol. Pflugers Arch. 1988 Jun;411(6):704–706. doi: 10.1007/BF00580872. [DOI] [PubMed] [Google Scholar]
- Vandenberg C. A., Horn R. Inactivation viewed through single sodium channels. J Gen Physiol. 1984 Oct;84(4):535–564. doi: 10.1085/jgp.84.4.535. [DOI] [PMC free article] [PubMed] [Google Scholar]


