ABSTRACT
Background
We investigated whether neonates exposed to meconium had a higher risk of respiratory hospitalization in childhood.
Methods
We analyzed a longitudinal cohort of 1,271,563 children with 6,334,857 person‐years of follow‐up between birth and age 5 years in Quebec, Canada from 2006 to 2022. The exposure was meconium aspiration or meconium staining without aspiration at birth. The outcome was hospitalization for bronchiolitis, asthma, and other allergic conditions up to age 5 years. We estimated hazard ratios (HR) and 95% confidence intervals (CI) for the association between meconium exposure and these outcomes using Cox regression models adjusted for patient characteristics. We additionally examined associations according to gestational age.
Results
A total of 6050 children had meconium aspiration (0.5%) and 131,238 were exposed to meconium without aspiration (10.3%). Compared with no exposure, meconium aspiration was associated with 1.24 times the risk of bronchiolitis (95% CI: 1.09–1.42), 1.43 times the risk of asthma (95% CI: 1.20–1.69), and 1.40 times the risk of other allergy hospitalization (95% CI: 1.11–1.76). Meconium aspiration was associated with these outcomes among children born at term (HR: 1.28, 95% CI: 1.14–1.44) and post‐term (HR: 1.56, 95% CI: 1.26–1.94). There was no association for children born preterm. Meconium exposure without aspiration appeared to protect against the outcomes (HR: 0.92, 95% CI: 0.90–0.95), but the association disappeared when we accounted for gestational age.
Conclusion
Meconium aspiration is associated with a higher risk of hospitalization for respiratory or atopic complications, but meconium exposure without aspiration is not associated with these outcomes.
Keywords: allergy and immunology, asthma, bronchiolitis, meconium, meconium aspiration syndrome
1. Introduction
Meconium‐stained amniotic fluid is present in 5% to 20% of deliveries at term [1], but the long‐term outcomes of exposed neonates are not fully understood. Meconium staining occurs when fetal colonic content is released in amniotic fluid during the prenatal period, and is a risk factor for perinatal respiratory complications, including meconium aspiration syndrome [1]. Aspiration of meconium occurs in 0.5 to 2 of every 1000 live births [1, 2], and is associated with local airway inflammation, surfactant inactivation, respiratory distress, and persistent pulmonary hypertension in a newborn [2]. An elevated proportion of children with a history of meconium aspiration syndrome develop chronic respiratory morbidity, including wheezing, alveolar hyperinflation, and exercise‐induced airway hyperreactivity [3, 4, 5, 6].
However, the relationship between meconium exposure and the long‐term risk of developing respiratory or atopic disorders is unclear. Recent studies suggest that exposure to meconium‐stained fluid may have a protective effect on the development of atopy [7, 8, 9, 10, 11]. In data covering 240,000 children followed from birth up to age 18 years, exposure to meconium‐stained fluid was found to be protective against hospitalization for dermatitis, skin rash, asthma, bronchiectasis, and respiratory symptoms such as wheezing [7, 10, 11]. In a separate cohort of more than 60,000 children, exposure to meconium‐stained fluid was associated with a lower risk of wheezing and allergic rhinitis at age 3 years [8, 9]. However, many of these findings could be due to other factors associated with meconium release, including gestational age. Meconium tends to be released later in gestation when fetuses are more mature and less at risk of developing respiratory or atopic disorders [12, 13]. We aimed to determine whether neonatal meconium exposure, with or without aspiration, was associated with the risk of respiratory related hospitalization in childhood independent of other factors.
2. Materials and Methods
2.1. Study Design and Population
We undertook a longitudinal cohort study of 1,271,563 children born between April 2006 and March 2022 in Quebec, Canada. The cohort comprised all children born in hospital, corresponding to more than 98% of all births in Quebec [14]. To construct the cohort, we used the Maintenance and Use of Data for the Study of Hospital Clientele database, which contains clinical records of all hospitalized patients, including newborns and their follow‐up over time. We used anonymized health insurance numbers to follow children from birth up to 5 years of age. Children were linked with their mothers, allowing us to obtain information on maternal pregnancy characteristics. The data did not include stillbirths or children without health insurance numbers.
2.2. Meconium Exposure
The main exposure measure was the presence of meconium in amniotic fluid at the time of birth, classified using diagnostic codes from the International Classification of Diseases, Tenth Revision (ICD‐10) that were in the newborn or maternal delivery chart (E‐Table 1). We considered two types of exposure, including meconium aspiration syndrome identified from the newborn's chart (ICD‐10 P24.0), followed by meconium‐stained amniotic fluid without aspiration identified from the maternal delivery chart (ICD‐10 O68.1, O68.2). We placed infants born at term who were exposed to meconium‐stained fluid and required intubation in the category of meconium aspiration syndrome [15]. The final exposure variable included three separate categories: (1) meconium aspiration; (2) meconium staining without aspiration; and (3) no meconium exposure. These categories were mutually exclusive.
2.3. Outcome
The outcome was hospitalization for respiratory or atopic disorders, including bronchiolitis, asthma, and other allergic conditions (anaphylaxis, food allergies, urticaria, other) [16]. We identified the outcomes using ICD‐10 diagnostic codes available in electronic health records (E‐Table 1) [17, 18]. Although bronchiolitis is generally triggered by infection with the respiratory syncytial virus, this pediatric complication is strongly related to asthma in children [19]. We included cases of bronchiolitis between birth and age 2 years, as the majority of hospitalizations for this disorder occur early in childhood [19]. In contrast, we identified hospitalizations for asthma or other allergies between 1 and 5 years of age because these conditions tend to be diagnosed later than bronchiolitis [17].
2.4. Covariates
We accounted for potential confounders, including maternal age (< 25, 25–34, ≥ 35 years), parity (0, 1, ≥ 2), pregnancy‐related morbidity comprising hypertensive disorders, gestational or pre‐existing diabetes, and alcohol, tobacco, or other substance use disorders (yes, no), maternal history of atopy, multiple birth, child sex, socioeconomic disadvantage (yes, no, unknown), gestational age in completed weeks, and time period at birth (2006–2011, 2012–2016, 2017–2022). Socioeconomic disadvantage represented the most disadvantaged quintile of children based on census data for mean neighborhood income, employment, and education [20]. We accounted for the time period at birth to ensure that changes in pediatric care did not drive the results.
2.5. Data Analysis
We calculated hospitalization rates per 1000 person‐years for meconium‐exposed and unexposed children. We plotted the cumulative incidence of hospitalization over time for bronchiolitis, asthma, and other allergies. We used Cox proportional hazards regression to estimate hazard ratios (HR) with 95% confidence intervals (CI) for the association between meconium exposure and the risk of hospitalization between birth and 5 years of age. Models were adjusted for maternal age, parity, pregnancy‐related morbidity, maternal history of atopy, multiple birth, child sex, gestational age, socioeconomic disadvantage, and time period.
In a further set of analyses, we stratified the data by cesarean delivery and presence of chorioamnionitis, as these factors may influence the likelihood of meconium exposure and risk of atopic conditions [1, 21, 22]. We then examined patients at different gestational ages separately (< 37, 37–40, ≥ 41 weeks). The presence of meconium in amniotic fluid increases with gestational age, and term birth reduces the risk of allergies [13, 23]. For the 86,229 preterm infants, we additionally adjusted the analyses for intubation, a marker of preterm neonatal morbidity [24].
We used robust error estimators to account for children with the same mother, and defined the time scale as the number of days between birth and the first hospitalization for any respiratory or atopic condition, death, or the end of the study. Follow‐up ended March 2023. For bronchiolitis, we stopped follow‐up at 2 years of age. For asthma and other allergic conditions, we began follow‐up at 1 year and stopped at 5 years. We censored children who were never hospitalized for any of these conditions before the study end, and accounted for death as a competing event using the Fine and Gray method. We evaluated the proportional hazards assumption graphically in survival curves.
In sensitivity analyses, we examined children who were admitted to a neonatal intensive care unit separately from children who were not. We also restricted the analyses to singletons, as multiple births are more often delivered preterm. We conducted the analysis in SAS v9.4 (SAS Institute Inc., Cary, NC).
3. Results
There were 1,271,563 children and 6,334,857 person‐years of follow‐up in this longitudinal study. A total of 6,050 children had meconium aspiration (0.5%) and 131,238 were exposed to meconium staining without aspiration (10.3%) (E‐Table 2). Children with meconium aspiration were only slightly more likely to be hospitalized during the study than children not exposed to meconium (6.2% vs. 6.0%), while children exposed to meconium without aspiration (5.0%) were less likely to be hospitalized (p‐value < 0.0001). Meconium exposure was more common at later gestational ages.
Children with meconium aspiration had a similar hospitalization rate for respiratory or atopic complications over time compared with children without meconium exposure, while children exposed to meconium‐stained fluid without aspiration had the lowest rate (Figure 1). At age 5 years, there were 62.5 hospitalizations per 1000 children with meconium aspiration (95% CI: 56.5–69.0), 50.6 per 1000 children exposed to meconium without aspiration (95% CI: 49.4–51.8), and 60.0 per 1000 unexposed children (95% CI: 59.6–60.5). For asthma and other allergic conditions, children with meconium aspiration had the highest rates. At age 2 years, the cumulative hospitalization rate for bronchiolitis was 37.4 per 1000 children with meconium aspiration (95% CI: 32.9–42.5), 31.0 per 1000 children exposed to meconium without aspiration (95% CI: 30.1–32.0), and 38.7 per 1000 children not exposed to meconium (95% CI: 38.4–39.1).
Figure 1.
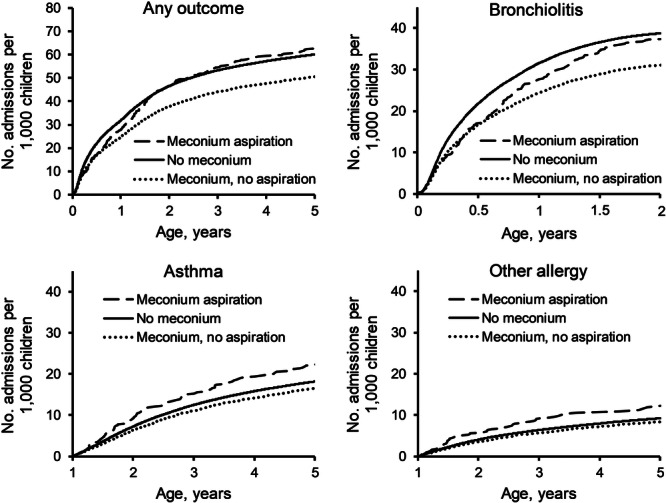
Cumulative rate of childhood hospitalization for respiratory related hospitalization according to meconium exposure. Cumulative rate of hospitalization per 1000 children (y‐axis) according to age in years (x‐axis). The curves represent the hospitalization rate for children with no meconium exposure (solid line), children with meconium aspiration (dashed line), and children with exposure to meconium‐stained fluid (dotted line). Top left panel shows the rate for any respiratory hospitalization, while the three remaining panels show rates for bronchiolitis (top right), asthma (bottom left), and other allergy (bottom right).
Meconium aspiration was associated with a higher risk of hospitalization before 5 years, while exposure to meconium‐stained fluid without aspiration was protective (Table 1). In analyses that did not account for gestational age, meconium aspiration was associated with 1.18 times the risk of any hospitalization (95% CI: 1.06–1.30), whereas exposure to meconium without aspiration seemed to lower the risk (HR 0.92, 95% CI: 0.90–0.95). However, the protective effect of meconium without aspiration disappeared in models that were further adjusted for gestational age, while the association with meconium aspiration strengthened. In models accounting for gestational age, meconium aspiration was associated with 1.24 times the risk of bronchiolitis (95% CI: 1.09–1.42), 1.43 times the risk of asthma (95% CI: 1.20–1.69), and 1.40 times the risk of other allergy (95% CI: 1.11–1.76) compared with no meconium exposure. Meconium‐stained fluid was weakly associated with asthma, with a HR approaching 1 (HR: 1.05, 95% CI: 1.01–1.10), but was not associated with bronchiolitis or other atopic conditions.
Table 1.
Association of meconium exposure with risk of childhood hospitalization.
| Total no. children | No. admissions | Hospitalization rate per 1000 person‐years (95% CI) | Hazard ratio (95% confidence interval) | ||
|---|---|---|---|---|---|
| Not accounting for gestational agea p value | Accounting for gestational ageb p value | ||||
| Any outcome | |||||
| Meconium aspiration | 6050 | 377 | 12.8 (11.6–14.1) | 1.18 (1.06–1.30) 0.0016 | 1.28 (1.16–1.42) < 0.0001 |
| Meconium, no aspiration | 131,238 | 6585 | 10.0 (9.8–10.3) | 0.92 (0.90–0.95) < 0.0001 | 1.00 (0.98–1.03) 0.9227 |
| No meconium | 1,134,275 | 67,607 | 12.0 (11.9–12.1) | Reference | Reference |
| Bronchiolitis | |||||
| Meconium aspiration | 6050 | 229 | 13.7 (12.0–15.5) | 1.14 (1.00–1.29) 0.0535 | 1.24 (1.09–1.42) 0.0010 |
| Meconium, no aspiration | 131,238 | 4171 | 11.3 (11.0–11.7) | 0.92 (0.89–0.95) < 0.0001 | 1.00 (0.97–1.03) 0.9896 |
| No meconium | 1,134,275 | 44,911 | 14.2 (14.1–14.3) | Reference | Reference |
| Asthma | |||||
| Meconium aspiration | 6020 | 132 | 4.3 (3.6–5.1) | 1.28 (1.08–1.52) 0.0052 | 1.43 (1.20–1.69) < 0.0001 |
| Meconium, no aspiration | 131,017 | 2126 | 3.2 (3.0–3.3) | 0.95 (0.91–0.99) 0.0213 | 1.05 (1.01–1.10) 0.0260 |
| No meconium | 1,131,589 | 20,148 | 3.4 (3.4–3.5) | Reference | Reference |
| Other allergy | |||||
| Meconium aspiration | 6005 | 74 | 2.4 (1.9–3.0) | 1.35 (1.07–1.69) 0.0103 | 1.40 (1.11–1.76) 0.0039 |
| Meconium, no aspiration | 130,754 | 1105 | 1.6 (1.5–1.7) | 0.93 (0.87–0.99) 0.0199 | 0.96 (0.91–1.03) 0.2553 |
| No meconium | 1,130,119 | 10,440 | 1.8 (1.7–1.8) | Reference | Reference |
Adjusted for maternal age, parity, pregnancy‐related morbidity, maternal history of atopy, multiple birth, child sex, socioeconomic disadvantage, and time period.
Additionally adjusted for gestational age in completed weeks.
The association between meconium aspiration and hospitalization for respiratory or atopic complications was present for all modes of delivery and regardless of the presence of chorioamnionitis (Table 2). Meconium aspiration was associated with 1.17 times the risk of any hospitalization among children born vaginally (95% CI: 1.03–1.33) and 1.48 times the risk among children born by cesarean (95% CI: 1.25–1.74). Confidence intervals for the association among children affected by chorioamnionitis included the null (HR: 1.23, 95% CI: 0.95–1.59).
Table 2.
Association of meconium exposure with risk of hospitalization according to mode of delivery and presence of chorioamnionitis.
| Hospitalization rate per 1000 person‐years (95% CI) | Hazard ratio (95% confidence interval)a p value | |||||
|---|---|---|---|---|---|---|
| Vaginal delivery | Caesarean delivery | Chorioamnionitis | Vaginal delivery | Caesarean delivery | Chorioamnionitis | |
| Any outcome | ||||||
| Meconium aspiration | 11.7 | 15.0 | 11.8 | 1.17 (1.03–1.33) 0.0155 | 1.48 (1.25–1.74) < 0.0001 | 1.23 (0.95–1.59) 0.1115 |
| Meconium, no aspiration | 10.1 | 10.0 | 9.0 | 1.00 (0.97–1.03) 0.9195 | 1.02 (0.96–1.08) 0.5104 | 0.94 (0.86–1.03) 0.1668 |
| No meconium | 11.5 | 13.3 | 11.7 | Reference | Reference | Reference |
| Bronchiolitis | ||||||
| Meconium aspiration | 12.4 | 16.3 | 11.9 | 1.11 (0.94–1.31) 0.2060 | 1.49 (1.21–1.84) 0.0002 | 1.20 (0.86–1.69) 0.2856 |
| Meconium, no aspiration | 11.6 | 10.6 | 8.6 | 1.00 (0.97–1.04) 0.9342 | 1.00 (0.93–1.08) 0.9388 | 0.87 (0.78–0.98) 0.0262 |
| No meconium | 13.7 | 15.6 | 12.5 | Reference | Reference | Reference |
| Asthma | ||||||
| Meconium aspiration | 4.0 | 4.9 | 4.4 | 1.36 (1.10–1.69) 0.0048 | 1.50 (1.13–1.99) 0.0046 | 1.39 (0.92–2.10) 0.1216 |
| Meconium, no aspiration | 3.0 | 3.6 | 3.5 | 1.03 (0.98–1.09) 0.2188 | 1.12 (1.02–1.23) 0.0126 | 1.07 (0.93–1.23) 0.3620 |
| No meconium | 3.3 | 4.0 | 4.0 | Reference | Reference | Reference |
| Other allergy | ||||||
| Meconium aspiration | 2.4 | 2.4 | 2.5 | 1.43 (1.09–1.89) 0.0112 | 1.31 (0.88–1.96) 0.1898 | 1.34 (0.77–2.32) 0.3014 |
| Meconium, no aspiration | 1.6 | 1.7 | 1.6 | 0.97 (0.90–1.04) 0.3574 | 0.96 (0.84–1.10) 0.5641 | 0.89 (0.72–1.09) 0.2464 |
| No meconium | 1.7 | 1.9 | 2.0 | Reference | Reference | Reference |
Adjusted for maternal age, parity, pregnancy‐related morbidity, maternal history of atopy, multiple birth, child sex, gestational age, socioeconomic disadvantage, and time period.
Compared with no meconium exposure, meconium aspiration was more strongly associated with hospitalization among children born at term (HR: 1.28, 95% CI: 1.14–1.44) or post‐term (HR: 1.56, 95% CI: 1.26–1.94) (Table 3). At term, meconium aspiration was associated with 1.24 times the risk of bronchiolitis (95% CI: 1.07–1.44), 1.48 times the risk of asthma (95% CI: 1.22–1.79), and 1.33 times the risk of other allergy (95% CI: 1.01–1.74) relative to no meconium exposure. Post‐term, meconium aspiration was associated with 1.68 times the risk of bronchiolitis (95% CI: 1.27–2.23) and 1.87 times the risk of other allergy (95% CI: 1.21–2.89). Exposure to meconium‐stained fluid without aspiration was slightly associated with asthma hospitalization among children born at term (HR: 1.06, 95% CI: 1.01–1.12), but not with other outcomes.
Table 3.
Association of meconium exposure with risk of hospitalization among children born at term or post‐term.
| Hazard ratio (95% confidence interval) p value | ||||
|---|---|---|---|---|
| Term (37–40 weeks) | Post‐term (≥ 41 weeks) | |||
| Not accounting for gestational agea | Accounting for gestational ageb | Not accounting for gestational agea | Accounting for gestational ageb | |
| Any outcome | ||||
| Meconium aspiration | 1.21 (1.08–1.36) 0.0013 | 1.28 (1.14–1.44) < 0.0001 | 1.56 (1.25–1.94) < 0.0001 | 1.56 (1.26–1.94) < 0.0001 |
| Meconium, no aspiration | 0.96 (0.93–0.98) 0.0020 | 1.02 (0.99–1.05) 0.2250 | 1.02 (0.96–1.09) 0.4996 | 1.02 (0.96–1.09) 0.4770 |
| No meconium | Reference | Reference | Reference | Reference |
| Bronchiolitis | ||||
| Meconium aspiration | 1.17 (1.00–1.35) 0.0431 | 1.24 (1.07–1.44) 0.0041 | 1.68 (1.26–2.22) 0.0003 | 1.68 (1.27–2.23) 0.0003 |
| Meconium, no aspiration | 0.95 (0.92–0.99) 0.0083 | 1.03 (0.99–1.06) 0.1622 | 1.05 (0.97–1.14) 0.2306 | 1.05 (0.97–1.14) 0.2178 |
| No meconium | Reference | Reference | Reference | Reference |
| Asthma | ||||
| Meconium aspiration | 1.40 (1.15–1.70) 0.0007 | 1.48 (1.22–1.79) < 0.0001 | 1.38 (0.93–2.03) 0.1095 | 1.38 (0.93–2.04) 0.1065 |
| Meconium, no aspiration | 1.00 (0.95–1.06) 0.9225 | 1.06 (1.01–1.12) 0.0179 | 0.99 (0.88–1.10) 0.8285 | 0.99 (0.88–1.10) 0.8423 |
| No meconium | Reference | Reference | Reference | Reference |
| Other allergy | ||||
| Meconium aspiration | 1.29 (0.99–1.69) 0.0614 | 1.33 (1.01–1.74) 0.0404 | 1.86 (1.21–2.88) 0.0051 | 1.87 (1.21–2.89) 0.0050 |
| Meconium, no aspiration | 0.94 (0.87–1.01) 0.0782 | 0.96 (0.90–1.04) 0.3095 | 1.03 (0.89–1.18) 0.6942 | 1.03 (0.89–1.18) 0.6887 |
| No meconium | Reference | Reference | Reference | Reference |
Adjusted for maternal age, parity, pregnancy‐related morbidity, maternal history of atopy, multiple birth, child sex, socioeconomic disadvantage, and time period.
Additionally adjusted for gestational age in completed weeks.
Among preterm births, meconium exposure was less clearly associated with respiratory or atopic conditions (Table 4). Compared with no exposure, meconium exposure overall was only associated with the risk of asthma hospitalization (HR: 1.23, 95% CI: 1.03–1.46), after adjusting for baseline patient characteristics. The association disappeared upon further adjustment for neonatal intubation, which we used as a proxy for respiratory morbidity. However, there was an insufficient number of cases to examine meconium aspiration separately from exposure without aspiration among preterm infants.
Table 4.
Association of meconium exposure with risk of hospitalization among children born preterma.
| Hospitalization rate per 1000 person‐years (95% CI) | Hazard ratio (95% confidence interval) p value | ||
|---|---|---|---|
| Not accounting for intubationb | Accounting for intubationc | ||
| Any outcome | |||
| Meconium | 24.2 (21.7–27.0) | 1.09 (0.98–1.22) 0.1094 | 1.02 (0.92–1.14) 0.6791 |
| No meconium | 21.5 (21.1–22.0) | Reference | Reference |
| Bronchiolitis | |||
| Meconium | 29.3 (25.8–33.4) | 1.09 (0.95–1.24) 0.2154 | 1.02 (0.89–1.16) 0.8162 |
| No meconium | 26.6 (25.9–27.2) | Reference | Reference |
| Asthma | |||
| Meconium | 8.7 (7.3–10.4) | 1.23 (1.03–1.46) 0.0243 | 1.12 (0.94–1.34) 0.2020 |
| No meconium | 7.0 (6.7–7.2) | Reference | Reference |
| Other allergy | |||
| Meconium | 1.7 (1.1–2.5) | 0.73 (0.49–1.08) 0.1121 | 0.69 (0.47–1.03) 0.0708 |
| No meconium | 2.3 (2.2–2.5) | Reference | Reference |
There were 86,229 preterm births, including 108 with meconium aspiration (0.1%) and 2829 without aspiration (3.3%). Meconium aspiration was too rare to examine separately and was combined with other meconium exposures.
Adjusted for maternal age, parity, pregnancy‐related morbidity, maternal history of atopy, multiple birth, child sex, gestational age, socioeconomic disadvantage, and time period.
Additionally adjusted for neonatal intubation.
In sensitivity analyses of children admitted to a neonatal intensive care unit, meconium aspiration was not associated with the risk of any hospitalization in children born at term (HR: 1.11, 95% CI: 0.87–1.42), but was associated with 1.77 times the risk in children born post‐term (95% CI: 1.09–2.89). Among children who were not admitted to a neonatal intensive care unit, meconium aspiration at term was associated 1.25 times the risk of any hospitalization (95% CI: 1.10–1.43), while meconium aspiration post‐term was associated with 1.48 times the risk (95% CI: 1.15–1.89). Meconium exposure was not associated with the risk of hospitalization among preterm births, regardless of neonatal intensive care admission. Restricting the analysis to singletons led to similar results.
4. Discussion
In this cohort of more than 1.2 million children, neonates experiencing meconium aspiration had a higher risk of hospitalization for bronchiolitis, asthma, and other allergic conditions later in childhood. The association was driven primarily by children who were born at term or post‐term, and was present regardless of mode of delivery or the occurrence of chorioamnionitis. While exposure to meconium‐stained fluid without aspiration appeared to protect against subsequent hospitalization for these outcomes, the association disappeared upon adjustment for gestational age. Overall, the findings suggest that meconium aspiration has negative implications for children early in life, whereas exposure to meconium‐stained fluid without aspiration is of limited concern.
Meconium aspiration is known for its acute effects on the neonatal respiratory tract [2], but the long term impact is less understood. Preliminary studies have correlated a history of meconium aspiration syndrome with the prevalence of wheezing, asthma, and bronchial hyperreactivity to exercise [3, 4, 5, 6]. However, these reports either comprised fewer than 100 patients with meconium aspiration, did not include a comparison group, or could not rule out bias. As our sample comprised 6000 children with meconium aspiration, the data provided considerable power and allowed us to account for a range of confounders. Meconium aspiration was associated with elevated risks of hospitalization for bronchiolitis, asthma, and other allergy, suggesting that the long‐term effect on the respiratory tract may be underestimated in previous studies. The findings support the possibility that acute damage caused by meconium to endothelial and epithelial alveolar cells may lead to sustained airway obstruction, pulmonary inflammation, and long‐term lung disease [2]. Invasive mechanical ventilation used to treat meconium aspiration may also worsen inflammation and exacerbate lung injury [25]. Even in the absence of serious tissue injury, mild epithelial damage may lead to the release of alarmins such as thymic stromal lymphopoietin, interleukin‐33, and interleukin‐25, now believed to be the first step in the immunologic cascade leading to allergic sensitization [26].
While meconium has potential to damage the respiratory tract [2], recent cohort studies have not succeeded in demonstrating a harmful effect, instead suggesting that meconium may protect against the development of asthma, bronchiectasis, and wheezing or other respiratory symptoms in childhood [7, 8, 9, 10, 11]. These paradoxical observations have led researchers to suspect that meconium may protect against atopy by increasing the quantity and diversity of organisms in the neonatal gut microbiome, as the microbiome is considered important for the developing immune system [7, 8, 10]. The problem with the hypothesis is that meconium colonization occurs in large part after delivery, once risk of aspiration abates. Moreover, previous studies focused on the presence of meconium in amniotic fluid without distinguishing aspiration from no aspiration. This issue is important because 95% of exposures to meconium do not result in aspiration [1], and most exposures occur at term when meconium is a sign of physiological maturation of the gastrointestinal tract [27], and by extension the respiratory tract. The alleged protective effect of meconium on respiratory outcomes is not surprising because term infants are expected to have mature lungs and a lower risk of respiratory disorders than preterm infants [23]. In our data, the protective association between isolated meconium exposure and respiratory disorders disappeared when we accounted for gestational age, while meconium aspiration at term remained associated with adverse outcomes.
In preterm infants, release of meconium is generally due to infection, hypoxia, or other stresses to the immune system [1], exposures that may influence the risk of respiratory or atopic complications [28]. Preterm infants also have immature respiratory tracts that are prone to reactivity [23]. In our data, the association between meconium and risk of asthma hospitalization disappeared among preterm births when we adjusted for neonatal intubation, a marker of respiratory immaturity or other neonatal morbidity potentially associated with the development of allergies. Overall, our findings suggest that immaturity or other adverse exposures in utero, rather than meconium exposure, are associated with the development of atopic disorders in children born preterm.
Pregnancies complicated by meconium more commonly require cesarean delivery, a risk factor for childhood asthma and other atopic conditions [1, 22]. However, it is unlikely that mode of delivery accounts for the associations as findings were similar when we analyzed cesarean deliveries separately from spontaneous vaginal births. Meconium aspiration was associated with respiratory and atopic conditions in children born by cesarean as well as children born vaginally, while meconium staining without aspiration was not associated with a protective effect in any of these groups. While chorioamnionitis is a risk factor for asthma and is more prevalent in pregnancies complicated by meconium‐stained fluid [1, 21], meconium aspiration did not appear to be associated with subsequent respiratory morbidity in children with past exposure to chorioamnionitis. It is therefore unlikely that chorioamnionitis is a meaningful contributor to our findings.
There are limitations related to the analysis of gestational age in this study. Gestational age may be in the pathway between meconium exposure and respiratory or atopic outcomes. Higher gestational age increases the likelihood of meconium release, but also correlates with maturation of the respiratory system and a lower risk of respiratory morbidity [13, 23]. While addressing the role of gestational age is a necessary component of an analysis of meconium exposure, adjusting or stratifying by gestational age can in some situations lead to collider bias due to unmeasured confounders [29]. It is unclear to what extent gestational age could be a collider in our analysis, however the possibility of bias is minimal among children born at term as unmeasured confounders are less common in this group.
There are other study limitations. We used administrative hospital data where coding errors may have led to nondifferential misclassification of exposures or outcomes. The outcome included respiratory reactivity and atopic conditions severe enough to require hospitalization, but we could not include mild disorders managed in outpatient settings. We lacked power to analyze meconium aspiration separately from meconium exposure without aspiration among children born preterm. Future studies with larger sample sizes will be needed to investigate preterm infants. We could not rule out residual confounding due to lack of information on ethnicity, paternal atopy, and daycare attendance, or the possibility that respiratory morbidity at term may be due to other causes in children with meconium‐stained fluid who were intubated. The data were gathered from a population covered by publicly funded healthcare, but findings may not generalize to other settings.
This study supports the need for continued efforts to optimize perinatal care and minimize the risk of meconium aspiration. Greater awareness of the potential long‐term complications of meconium aspiration is needed, as enhanced follow‐up of exposed children may help identify early signs of respiratory or atopic morbidity. Further research is needed to understand the mechanisms by which meconium aspiration may lead to respiratory or atopic disorders during childhood, with the aim of identifying targets for early prevention.
In this study comprising a large population of children with more than 6 million person‐years of follow‐up, meconium aspiration was associated with an elevated risk of hospitalization for bronchiolitis, asthma, and other allergic conditions up to 5 years of age. In contrast, meconium exposure without aspiration was not associated with these outcomes once we accounted for gestational age. The results suggest that, apart from risk of aspiration, the presence of meconium at time of birth has no major implications for morbidity in children. It is unlikely that isolated meconium exposures have a direct impact on respiratory or atopic disorders.
Author Contributions
Gabriel Côté‐Corriveau: conceptualization, methodology, writing – review and editing, visualization. Ariane Lasry: conceptualization, writing – original draft, methodology, visualization. Philippe Bégin: writing – review and editing, conceptualization. Marianne Bilodeau‐Bertrand: conceptualization, writing – original draft, methodology, formal analysis, visualization, investigation, data curation. Jessica Healy‐Profitós: writing – review and editing. Nathalie Auger: conceptualization, funding acquisition, writing – original draft, methodology, software, supervision, project administration, resources, validation, visualization.
Ethics Statement
The institutional review board of the University of Montreal Hospital Centre provided a waiver from informed consent and ethical review as the data were anonymized.
Conflicts of Interest
The authors declare no conflicts of interest.
Supporting information
E‐Table 1 Diagnostic and intervention codes for meconium exposure and study outcomes. E‐Table 2 Characteristics of children according to meconium exposure.
Acknowledgments
This study was supported by the Canadian Institutes of Health Research [grant number PJT‐162300 to Nathalie Auger] and Fonds de recherche du Québec‐Santé [grant number 296785 to Nathalie Auger].
Data Availability Statement
The data that support the findings of this study are available from the Institut de la statistique du Québec. Restrictions apply to the availability of these data, which were used under license for this study. Access to the data may be requested from the Institut de la statistique du Québec (https://statistique.quebec.ca/research/#/accueil).
References
- 1. Gallo D. M., Romero R., Bosco M., et al., “Meconium‐Stained Amniotic Fluid,” American Journal of Obstetrics and Gynecology 228, no. 5S (2023): S1158–S1178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Osman A., Halling C., Crume M., Al Tabosh H., Odackal N., and Ball M. K., “Meconium Aspiration Syndrome: A Comprehensive Review,” Journal of Perinatology 43, no. 10 (2023): 1211–1221. [DOI] [PubMed] [Google Scholar]
- 3. Vázquez Nava F., Salas Ramírez E., Sánchez Núncio H. R., et al., “Meconium Aspiration Syndrome, Parental Atopy and Asthma Symptoms in Children Under Two Years Old,” Revista alergia Mexico (Tecamachalco, Puebla, Mexico: 1993) 53, no. 4 (2006): 130–135. [PubMed] [Google Scholar]
- 4. Gupta A. K. and Anand N. K., “Wheezy Baby Syndrome—A Possible Sequelae of Neonatal Meconium Aspiration Syndrome,” The Indian Journal of Pediatrics 58, no. 4 (1991): 525–527. [PubMed] [Google Scholar]
- 5. Macfarlane P. I. and Heaf D. P., “Pulmonary Function in Children After Neonatal Meconium Aspiration Syndrome,” Archives of Disease in Childhood 63, no. 4 (1988): 368–372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Djemal N., Ben Ammar H., Masmoudi K., et al., “[Pulmonary Function in Children After Neonatal Meconium Aspiration Syndrome],” Archives de pediatrie: organe officiel de la Societe francaise de pediatrie 15, no. 2 (2008): 105–110. [DOI] [PubMed] [Google Scholar]
- 7. Krieger Y., Horev A., Wainstock T., Sheiner E., and Walfisch A., “Meconium‐Stained Amniotic Fluid as a Protective Factor Against Childhood Dermatitis and Skin Rash‐Related Hospitalization in the Offspring—A Population‐Based Cohort Analysis,” Journal of the European Academy of Dermatology and Venereology 34, no. 2 (2020): 319–324. [DOI] [PubMed] [Google Scholar]
- 8. Murata T., Kyozuka H., Fukuda T., et al., “Meconium‐Stained Amniotic Fluid During Labor May be a Protective Factor for the Offspring's Childhood Wheezing up to 3 Years of Age: The Japan Environment and Children's Study,” European Journal of Pediatrics 181, no. 8 (2022): 3153–3162. [DOI] [PubMed] [Google Scholar]
- 9. Murata T., Kyozuka H., Fukuda T., et al., “Meconium‐Stained Amniotic Fluid and Offspring Allergies: The Japan Environment and Children's Study,” Pediatric Allergy and Immunology 34, no. 5 (2023): e13956. [DOI] [PubMed] [Google Scholar]
- 10. Paz Levy D., Walfisch A., Wainstock T., et al., “Meconium‐Stained Amniotic Fluid Exposure Is Associated With a Lower Incidence of Offspring Long‐Term Infectious Morbidity,” American Journal of Reproductive Immunology 81, no. 6 (2019): e13108. [DOI] [PubMed] [Google Scholar]
- 11. Rodavsky G., Sheiner E., Walfisch A., et al., “Meconium Stained Amniotic Fluid Exposure and Long‐Term Respiratory Morbidity in the Offspring,” Pediatric Pulmonology 56, no. 7 (2021): 2328–2334. [DOI] [PubMed] [Google Scholar]
- 12. Gonçalves C., Wandalsen G., Lanza F., Goulart A. L., Solé D., and Dos Santos A., “Repercussions of Preterm Birth on Symptoms of Asthma, Allergic Diseases and Pulmonary Function, 6–14 Years Later,” Allergologia et Immunopathologia 44, no. 6 (2016): 489–496. [DOI] [PubMed] [Google Scholar]
- 13. Bhat R. Y. and Rao A., “Meconium‐Stained Amniotic Fluid and Meconium Aspiration Syndrome: A Prospective Study,” Annals of Tropical Paediatrics 28, no. 3 (2008): 199–203. [DOI] [PubMed] [Google Scholar]
- 14. Auger N., Maniraho A., Ayoub A., and Arbour L., “Association of Maternal Cancer With Congenital Anomalies in Offspring,” Paediatric and Perinatal Epidemiology 38, no. 2 (2024): 121–129. [DOI] [PubMed] [Google Scholar]
- 15. Kalra V., Leegwater A. J., Vadlaputi P., Garlapati P., Chawla S., and Lakshminrusimha S., “Neonatal Outcomes of Non‐Vigorous Neonates With Meconium‐Stained Amniotic Fluid Before and After Change in Tracheal Suctioning Recommendation,” Journal of Perinatology 42, no. 6 (2022): 769–774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Soullane S., Bégin P., Lewin A., Lee G. E., and Auger N., “Increased Risk of Allergy Hospitalization After Cesarean Delivery,” Annals of Allergy, Asthma & Immunology 127, no. 1 (2021): 142–144. [DOI] [PubMed] [Google Scholar]
- 17. Gabryszewski S. J., Dudley J., Shu D., et al., “Patterns in the Development of Pediatric Allergy,” Pediatrics 152, no. 2 (2023): e2022060531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Pelletier J. H., Au A. K., Fuhrman D. Y., et al., “Healthcare Use in the Year Following Bronchiolitis Hospitalization,” Hospital Pediatrics 12, no. 11 (2022): 937–949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Jartti T. and Gern J. E., “Role of Viral Infections in the Development and Exacerbation of Asthma in Children,” Journal of Allergy and Clinical Immunology 140, no. 4 (2017): 895–906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Pampalon R., Hamel D., Gamache P., Philibert M. D., Raymond G., and Simpson A., “An Area‐Based Material and Social Deprivation Index for Public Health in Quebec and Canada,” Canadian Journal of Public Health 103, no. 8 Suppl 2 (2012): S17–S22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Getahun D., Strickland D., Zeiger R. S., et al., “Effect of Chorioamnionitis on Early Childhood Asthma,” Archives of Pediatrics & Adolescent Medicine 164, no. 2 (2010): 187–192. [DOI] [PubMed] [Google Scholar]
- 22. Sandall J., Tribe R. M., Avery L., et al., “Short‐Term and Long‐Term Effects of Caesarean Section on the Health of Women and Children,” The Lancet 392, no. 10155 (2018): 1349–1357. [DOI] [PubMed] [Google Scholar]
- 23. Caffarelli C., Gracci S., Giannì G., and Bernardini R., “Are Babies Born Preterm High‐Risk Asthma Candidates?,” Journal of Clinical Medicine 12, no. 16 (2023): 5400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Sweet D. G., Carnielli V. P., Greisen G., et al., “European Consensus Guidelines on the Management of Respiratory Distress Syndrome: 2022 Update,” Neonatology 120, no. 1 (2023): 3–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Silva P. L., Ball L., Rocco P. R. M., and Pelosi P., “Physiological and Pathophysiological Consequences of Mechanical Ventilation,” Seminars in Respiratory and Critical Care Medicine 43, no. 3 (2022): 321–334. [DOI] [PubMed] [Google Scholar]
- 26. Whetstone C. E., Ranjbar M., Omer H., Cusack R. P., and Gauvreau G. M., “The Role of Airway Epithelial Cell Alarmins in Asthma,” Cells 11, no. 7 (2022): 1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Mazor M., Hershkovitz R., Bashiri A., et al., “Meconium Stained Amniotic Fluid In Preterm Delivery Is an Independent Risk Factor for Perinatal Complications,” European Journal of Obstetrics & Gynecology and Reproductive Biology 81, no. 1 (1998): 9–13. [DOI] [PubMed] [Google Scholar]
- 28. Lao T. T. and Annie Hui S. Y., “The Obstetric Aspects of Maternal Asthma,” Best Practice & Research Clinical Obstetrics & Gynaecology 85, no. Pt A (2022): 57–69. [DOI] [PubMed] [Google Scholar]
- 29. Wilcox A. J., Weinberg C. R., and Basso O., “On the Pitfalls of Adjusting for Gestational Age at Birth,” American Journal of Epidemiology 174, no. 9 (2011): 1062–1068. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
E‐Table 1 Diagnostic and intervention codes for meconium exposure and study outcomes. E‐Table 2 Characteristics of children according to meconium exposure.
Data Availability Statement
The data that support the findings of this study are available from the Institut de la statistique du Québec. Restrictions apply to the availability of these data, which were used under license for this study. Access to the data may be requested from the Institut de la statistique du Québec (https://statistique.quebec.ca/research/#/accueil).


