Graphical abstract

Keywords: Aging and aging-associated diseases, Inorganic metal nanoparticles, Biomedical application
Highlights
-
•
Understanding aging and Aging and aging-associated diseases (AAD) are significant for reducing the burden of the elderly.
-
•
The common morphological changes and mechanism of senescent cells are key.
-
•
Inorganic metal nanoparticles (IMNPs) have been widely used due to their superior properties.
-
•
Biomedical applications of IMNPs in aging and AAD is crucial for the further research and development.
Abstract
Aging and aging-associated diseases (AAD), including neurodegenerative disease, cancer, cardiovascular diseases, and diabetes, are inevitable process. With the gradual improvement of life style, life expectancy is gradually extended. However, the extended lifespan has not reduced the incidence of disease, and most elderly people are in ill-health state in their later years. Hence, understanding aging and AAD are significant for reducing the burden of the elderly. Inorganic metal nanoparticles (IMNPs) predominantly include gold, silver, iron, zinc, titanium, thallium, platinum, cerium, copper NPs, which has been widely used to prevent and treat aging and AAD due to their superior properties (essential metal ions for human body, easily synthesis and modification, magnetism). Therefore, a systematic review of common morphological alternations of senescent cells, altered genes and signal pathways in aging and AAD, and biomedical applications of IMNPs in aging and AAD is crucial for the further research and development of IMNPs in aging and AAD. This review focus on the existing research on cellular senescence, aging and AAD, as well as the applications of IMNPs in aging and AAD in the past decade. This review aims to provide cutting-edge knowledge involved with aging and AAD, the application of IMNPs in aging and AAD to promote the biomedical application of IMNPs in aging and AAD.
Introduction
Aging is a natural and unavoidable physiological process, mainly accompanied by physical and cognitive decline, which significantly increases the morbidity and mortality of human diseases and aggravate the economic burden on society [1]. Aging-associated diseases (AAD) are predominantly composed of the following types: Alzheimer's disease, skin aging, wound healing, cancer, cardiovascular diseases, diabetes, nonalcoholic fatty liver disease, obesity, macular degeneration and cataract [2]. The main hallmarks of aging and AAD include DNA damage, telomere deficiency, epigenetic alterations, cell homeostasis disruption, senescence, mitochondrial dysfunction, stem cell capacity disruption, nutritional disorder, and chronic inflammation [3]. The common morphological alterations of senescence cells are composed of enlarged cell size with irregular in shape, depolarization plasma membrane, increased lysosomal proteins and lysosomal content, accumulation of mitochondria, nuclear deformations and transcriptional abnormality [4], [5], [6].
Inorganic metal nanoparticles (IMNPs) were mainly composed of gold, silver, iron, zinc, titanium, thallium, platinum, cerium, and copper NPs. In addition to the metal ions necessary for the human body, IMNPs also have facile synthesis, modifiable, magnetic, antibacterial and other properties, which are gradually widely used in the biomedical field [7]. IMNPs have attracted the attention of researchers because they successfully prevented and treated ageing and AAD, which provide novel strategy to improve the life quality of elderly people (Fig. 1) [8], [9]. IMNPs slowed down the development of ageing and AAD via inhibiting the expression of age-related key pathways, reducing chronic inflammation, decreasing oxidative damage, and other ways [10]. However, a summary of the biomedical application of IMNPs in aging and AAD is still lacking, which is not conducive to broaden their biological applications and accelerate the development of IMNPs-based nanomedicine. Therefore, it is necessary to introduce ageing and AAD, and carefully summarize the application of IMNPs for aging and AAD.
Fig. 1.
The biological application of inorganic metal nanoparticles in aging and AAD.
This review aims to provide a comprehensive overview of recent progress on morphological changes of senescent cells, applications of IMNPs for aging and AAD, focusing on literatures published from 2013 to the present. In the first part of this review, we summarized the common morphological changes of senescent cells, which was positively related to aging. Then, we described different types of aging and AAD via widely used cell lines, animal models, associated proteins, and signaling pathways. Finally, we reviewed the applications of each kind of IMNPs in aging and AAD in detail. This review may provide novel and more comprehensive design ideas for the prevention and treatment of aging and AAD, and broaden the biomedical applications of IMNPs.
Morphological alterations of cellular senescence
Cellular senescence, as an irreversible state, is a primary fundamental ageing mechanism. Senescence cells accelerate the release of pro-inflammatory factors, which is named as the senescence-associated secretory phenotype (SASP) [11]. The accumulation of cellular senescence predominantly contributed to aging and AAD. Senescent cells are alive with many hallmarks, such as cell size and shape, composition of membrane, lysosomal content, number of mitochondria, and nuclear changes [6].
Cell size and shape
The most prominent feature of cellular senescence is enlarged cells with irregular in shape. Cells have their own dedicated mechanisms to control cell size, and cell size is largely dependent on the growth rate. Additionally, ploidy levels are reported to be related to cell size. Study showed that diploid cells grew faster after excluding the influence of original cell size, which indicated that DNA was the key factor in determining cell growth rate [12]. Amazingly, the proteome of the cells did not change regularly, even as the size of the cells increased significantly [13]. mTOR Complex 1 (mTORC1), Rb1, E2F, MAPK p38, YAP/TAZ concentration could reflect cell size, which was considered as size-dependent senescence markers [14]. Multiple types of compounds used to induce different types of senescence were found to inhibit the expression of enzymes Aurora kinase B (AURKB) [15]. Another morphological alteration that reflected cellular senescence was the irregularity of cell. Study showed that a delay in DNA replication caused unbalanced growth, which in turn induced cell swelling [16]. Another mainly cause of alteration in cell shape was cell rearrangement, which was mainly affected by the ATF6a signaling pathway. However, the mechanism of ATF6a signaling pathway in regulating cell morphology remained unclear [17].
Composition of the plasma membrane
Plasma membrane (PM), as a messenger of communication with neighboring cells, has been shown to play a key role in signal transduction. Phosphatidylserine (PtdSer), exposed on the surface of PM, could be recognized by phagocytes as a clearance signal [18]. Oxidoreductases in PM reduced oxidative damage by transferring NAD(P)H, ensuring senescent cell survival [19]. NFAT/ATF3/p53 signaling pathway was positively associated with depolarization of PM, which promoted the senescence of mesenchymal stem cells [20]. Lipid rafts in PM dominantly consisted of cholesterol and sphingolipids, which was closely related to the decrease and dysregulation of aging-dependent signaling pathways, like T-cell receptor signaling [21], [22]. Senescence-associated genes were detected in MRC-5 cell lines, and 841 up-regulated genes (multiple lipid-involved pathways) and 900 down-regulated genes (cell cycle process) were identified. Additionally, phosphatidylcholines accumulation could promote membrane remodeling in cell senescence [23]. The results of Wistar rats with different months old revealed that the alteration of PM level was an important cause of erythrocyte aging. The activities of Na/K ATPase (NKA) and Na/H exchanger (NHE) in old cell population was significantly lower than in the younger group [24]. Sialo glycoproteins and gangliosides in PM could regulate the lifespan of erythrocyte, which was demonstrated by the gradual loss of neuraminidase 1 (NEU1) and NEU3 in the life of red blood cells [25].
Increased lysosomal proteins and lysosomal content
Lysosomes play a vital role in primary degradation and signal transduction. A significantly up-regulation of number and size of lysosomes, and obviously decline of lysosome activity were observed with age increased, which was controlled by transcription factors or transcription repressors, such as transcription Factor EB (TFEB), TFE3 [26]. mTORC1 and PKC were closely related to lysosome biogenesis [27]. ABCA1 could contribute to the accumulation of cholesterol in the lysosome, thus the activity of mTORC1 was further maintained to keep with mTORC1 scaffolding complex enrichment for the enduring secretome of senescent cells [28]. Four proteases as lysosomal proteins like cathepsin B (CTSB), CTSD in MEL-103 cells (senescent cells) were significantly expressed in a RAB27A-dependent manner [29]. In vitro and in vivo studies shown that glycoprotein nonmetastatic melanoma protein B (GPNMB) deletion could shorten the lifespan of human vascular endothelial cells. Overexpression of GPNMB could prolong the lifespan of cells [30]. Lack of BCL2 interacting protein 3 induced lysosomal dysfunction, exacerbating inflammation and muscle atrophy [31]. Moreover, lysosome could maintain its function only under low pH conditions (between 4.5 and 5.5), which was mainly controlled by ATPase. Dysfunction of ATPase or ATPase complex was closely associated with aging and aging-related diseases [32]. Through the process of autophagy, lysosomes could complete the degradation and recovery of the received intracellular components, such as proteins and organelles [33]. Spermidine induced autophagy through post-translational modification of EIF5A, thereby prolonging the lifespan of old human cells [34]. Furthermore, recent studies have found that lysosomes are not only cell degraders, but also center stage of nutrition-dependent signaling pathways, cell growth, and cell differentiation [35], [36]. Lysosomal lipolysis could prolong lifespan of Caenorhabditis elegans via upregulating fat-to-neuron lipid signaling pathway [37].
Accumulation of mitochondria
Mitochondria influenced the development of cell senescence in a myriad ways as a major producer of energy and reactive oxygen species (ROS) [38]. Mitochondrial dysfunction was mainly reflected in the decrease of mitochondrial membrane potential (MMP) and the increase of mitochondrial mass in senescence, which was initially related to the overexpression of ROS and decreased mitophagy, respectively [39]. The results of mice studies demonstrated that the lifespan of mice was significantly extended after 1,4,5-trisphosphate receptor, type 2 (ITPR2) knockout, mainly due to the reduction of contact and material exchange (like lipid and Ca+) between the endoplasmic reticulum (ER) and mitochondria [40]. Additionally, the contacts between mitochondria and ER were proved to play a vital role in aging or senescence processes [41], [42]. The content of mitochondria was negatively associated with the expression of mTORC1 or peroxisome proliferator-activated receptor gamma co-activator 1β (PGC-1β). Inhibiting the expression of mTORC1 or PGC-1β could reverse the senescence process of mouse liver [43]. The activities of Wnt/β-catenin and renin-angiotensin system have been shown to be positively involved with mitochondrial dysfunction and renal fibrosis in both cell line and aging mouse model [44]. Mitokines were a double-edged sword, as evidenced that the increased level maintained mitochondrial function. Conversely, resistance to the action of mitokines might also arise [45]. Inhibition the expression of carnitine palmitoyltransferase 1A could decrease the level of ROS, which contributed to regulate mitochondrial dysfunction and delay the aging process of chondrocyte [46].
Nuclear changes
Nuclear deformations and transcriptional abnormality were also specific features for aging-associated diseases [47]. Reduction of lamin-B1 was considered as a common mark of senescent astrocytes, which could cause nuclear incompleteness [48]. Furthermore, mutations of lamin A and C influenced the integrity of the nuclear, inducing cellular senescence [47]. Deletion of sirtuin 3 (SIRT 3) significantly affected the function of proteins in nuclear envelope, which increased abnormal repeat transcription and accelerated senescence of human mesenchymal stem cells [49]. Other SIRT family members (SIRT1, SIRT6) were also reported to possess anti-vascular aging activity [50]. Cellular experiments showed that LINE-1 RNA deficiency-maintained chromosome homeostasis and reduced the expression of aging-related genes (p16 and p21). Additionally, mouse models indicated that depletion of LINE-1 RNA down-regulated aging-related signaling [51]. MT1-MMP could influence the nuclear lamina structure via overexpressing of p16, p21, and β-galactosidase [52]. TRF2 was tightly related to nuclear deformations and abnormal folding of nuclear matrix, which was considered as a biomarker of cell aging [53]. Lake of arp2/3 complex activated p53 and CdKN1a/p21 in the nuclei, which was tightly associated with the formation of micronuclei [54].
Aging and AAD
With the gradual improvement of life style, life expectancy is gradually extended. However, the extended lifespan has not reduced the incidence of disease, and most elderly people are in ill-health state in their later years. Hence, understanding aging and AAD are significant for reducing the burden of the elderly. Aging is a complex process that consists of the following characteristics: 1) mortality increases exponentially with age; 2) Physiological changes (including progressive decline in renal function and adaptive and innate immunity), which usually lead to functional decline with age; 3) Sensitivity to certain diseases increases with age, such as Alzheimer’s diseases, cancer, cardiovascular diseases, diabetes [55], [56], [57], [58]. Long non-coding RNAs were the molecular marks in aging, which was positively related to the process of aging [59]. There were four classic pathways that altered with the occurrence and development of aging, namely insulin and insulin-like growth factor I (IGF 1), mTORC1, adenosine 5‘-monophosphate (AMP)-activated protein kinase (AMPK) and SIRT1 signaling. Phosphoinositide 3-kinase (PI3K), Akt, and FOXOs in insulin and IGF signaling were primarily concerned with stress defense in aging (Fig. 2). 4EBP1 and S6K1 in mTORC1 signaling were mainly affected protein synthesis. ULK1 and ACC1/2 in AMPK signaling were related to autophagy and lipid metabolism, respectively. PGC1α in SIRT1 signaling contributed to maintain mitochondrial homeostasis [60], [61], [62], [63]. These four signaling pathways were not independent, and they interacted with each other to affect the aging process. Aging as an irreversible deterioration in cell, tissue, and organ levels, is a major cause of many chronic diseases, including Alzheimer's disease, skin aging, cancer, cardiovascular diseases, diabetes, nonalcoholic fatty liver disease, obesity, macular degeneration and cataract [64]. The common characteristics of aging and AAD is genomic instability, telomere attrition, mitochondrial and autophagy disorders, and chronic inflammation [65], [66].
Fig. 2.
Four classic pathways that altered in aging.
Alzheimer diseases
More than 95 % Alzheimer's disease (AD) is caused by aging, specific changes in senescent cells are also observed in AD, including DNA damage, mitochondrial dysfunction, and autophagy depletion [67]. High expression of aging-related proteins was found in AD patients’ plasma, containing IL-6, insulin-like growth factor-binding protein (IGFBP), transforming growth factor (TGF)-β and MMP-10. Additionally, ADAM10, ADAMTS4, and BIN1, as senescence genes, were genetic risk in AD patients (Fig. 3A) [68]. The main characteristics of AD is high content of abnormally folded proteins (amyloid-β and microtubule protein tau) and neuroinflammation [69]. Tumor necrosis factor-α (TNF-α), Interleukin-8 (IL-8), IL-17A, TIMP-4 and matrix metalloproteinase-7 (MMP7) played a pivotal role in neuroinflammation, and significantly upregulated levels of those protein were tightly associated with cognitive decline [70], [71]. Furthermore, the disruption of blood–brain barrier (BBB) activated in inflammation and immune response in the brain, and the formation of BBB was contributed by vascular endothelial growth factor (VEGF), Wnt, netrins and semaphorins [72]. R47H mutation could remarkably increase the expression of pro-inflammatory cytokines and activate AKT signal in microglia, which exacerbated the memory deficit in mouse [73]. Increased wolframin could effectively reduce the expression of tau, and slow down neurodegeneration by reducing ER stress and regulating autophagy-lysosome related proteins [74]. Microgliacyte, astrocytes and neurons activated pyroptosis signaling pathway via regulating different related proteins, slowing down the neurodegenerative process. Microgliacyte mainly acted on all NLRP3 inflammasome-forming proteins, while gasdermin D was detected in three types of cells [75]. miR-204-3p was negatively correlated with memory deficits and oxidative stress in AD mice model, which was targeted to nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 4 [76]. Mild closed head injury in mouse models indicated that overexpression of TDP-43 could induce the production of tau and Aβ, then contributed to cognitive impairment [77]. Autophagy injury promoted the production of IL-1β and IL-18 via acting on NACHT, LRR and PYD domains-containing protein 3 (NLRP3), which activated microglia and regulated neuroinflammation response [78].
Fig. 3.
The relationship between cellular senescence and aging-associated diseases. (A) Alzheimer's disease [69]; (B) Skin aging [81].
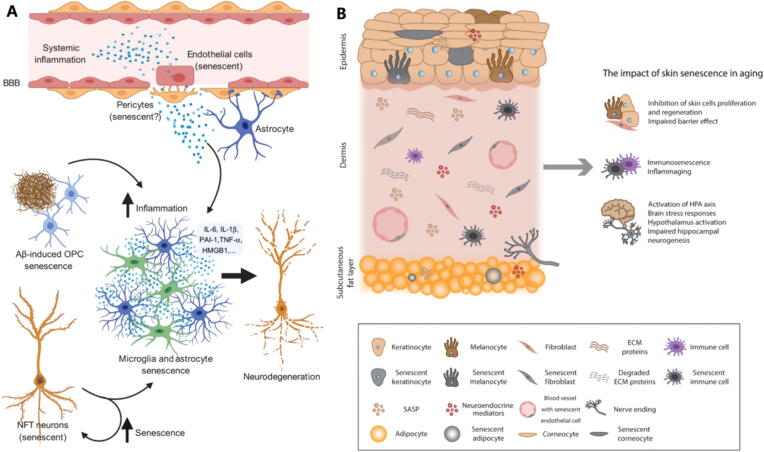
Skin aging and wound healing
Skin aging, like aging, is controlled by both internal and external factors. Cell senescence speeded skin aging via secreting elastin and collagen. Additionally, cell senescence damaged structure and function of skin, including wrinkling dry skin. Interestingly, skin aging might contribute to systemic aging and accelerate age-associated changes [79]. Skin aging exhibited similar changes to aging at the structural, cellular, and molecular levels, which was dominantly included lysosomal mass and mitochondria increase, nuclear deficit (Fig. 3B) [80], [81]. β-galactosidase, p16, p21, p53, lamin B1, as SASP, were also exhibited in skin senescence cells [82]. However, skin aging also possessed its own specific biomarkers, mainly including thioredoxin domain-containing protein 5 (TXNDC5), RhoGDI, ras suppressor protein 1 (RSU1), and Vimentin [83], [84], [85], [86], [87]. Different skin cell types showed different metabolic changes of aging, as evidenced that p16 could only be detected in fibroblasts. Moreover, the level of phosphocholine was much lower in skin melanocytes than that in fibroblasts, and content of phosphocholine obviously enhanced with the process of aging [88]. HLA-E decreased the immune reaction via interacting with NKG2-A/NKG2-B type II integral membrane protein (NKG2A). The expression of HLA-E was gradually increased in dermal fibroblasts but not in melanocytes, which was regulated by p38 MAP kinase signaling pathway [89]. miR-377, as a upstream miRNAs to negatively control DNA methyltransferase 1, was remarkably expressed in aged human skin fibroblasts, which might be a potential target to prevent skin aging [90]. Additionally, various types of miRNA and their directly targets were summarize in human skin senescent cells, including miR-130b, miR-181a, miR-138 [91].
Wound healing played a vital role in restoring the function of skin, cell senescence has been shown to be associated with wound healing. Aging was conducive to the healing of acute wounds, and prolonged the healing time of chronic wounds [92]. In addition, The structure of aged skin changed, mainly manifested in collagen remodeling and hardness increase, which also enhanced the time of wound healing [93]. However, senescent fibroblasts and endothelial cells could promote wound healing via secreting platelet-derived growth factor AA in mouse [94]. Secretion of chemokines (CCL2, CCL21, CXCL12, CXCR4) accelerated inflammatory cells to release growth factors and cytokines, which promote wound healing [95]. Skin senescence mouse model proved that skin aging prolonged the wound healing time. On the contrary, the skin aging sustained senescence even healing [96]. miR-125b was transferred to fibroblasts through exosomes, then inhibited the expression of sirtuin 7, thereby promoting the generation of myofibroblasts and alleviating aging [97]. Nuclear factor erythroid 2-related factor 2, platelet-derived growth factor, Sirt6, and peroxiredoxin-4 (PRDX4) were proved to be the critical regulators of wound closure [98], [99], [100], [101].
Cancer
Senescence response also took place in different types of tumor, with common features such as genomic mutation, epigenetic changes, chronic inflammation, and dysbiosis [102]. However, tissue transcriptomic results showed that the expression trend of tumor and aging related genes was opposite. For example, cell proliferation was significantly inhibited in aging, while cell division was obviously enhanced in cancer (Fig. 4A) [103]. TP53 gene was a key gene in regulating aging and inhibiting tumor, which controlled by multiple types of P53 proteins. The inhibition of p53 expression in hepatic stellate cells could promote the occurrence of cancer [104]. MiR-21, MiR-126, MiR-146a were commonly considered as cancer-associated, inflammation-associated, senescence-associated microRNAs, which were potential anti-cancer targets via NF-κB signaling pathway [105]. The deficiency of integrin-linked kinase contributed to cellular senescence, which also led to senescence of three different gastric cancer cell lines (MKN1, MKN28, AGS cell lines) [106]. Aging could significantly suppress the occurrence and development of tumors, inducing aging become a rational tumor treatment strategy. At present, the dominant approaches to induce tumor senescence were commonly achieved by activating oncogene-related to senescence, chemotherapy or radiotherapy, inhibiting cell cycle and telomerase, modulating epigenome, targeting tumor suppressors or oncogenes [107], [108]. Studies in mice indicated that p38/MAPK and MAPK/ERK signaling pathways were enhanced after chemotherapy, which promoted the transformation of benign papilloma into cancer. Ablation of senescent cells slowed down the progression of papillary carcinoma by regulating p38 MAPK and MAPK/ERK signaling pathways [109]. It was worth noting that senescent cells were always presented in the tumor which was not conducive to anti-cancer treatment. Aging immune cells could lead to the senescence of T cells, macrophages, and thus affect the treatment of tumors [110], [111].
Fig. 4.
The relationship between cellular senescence and AAD. (A) Cancer [110]; (B) Cardiovascular disease [113].
Cardiovascular diseases
Aging was an important factor in promoting cardiovascular disease (involving heart or blood vessels, like heart failure, atherosclerosis), with increasing heart dysfunction and hardening of blood vessels with age (Fig. 4B) [112], [113]. The senescence markers, including cytokines, chemokines, matrix metalloproteases (MMPs) and Toll-like receptors (TLRs) were used to ameliorate atherosclerosis [114]. ASK1-signalosome could activate p38 MAPK signaling pathway via ROS, then promote aging and cardiovascular diseases [115]. Sirtuin family members were pivotal targets for aging. Increasing the activity of sirtuin family members not only slowed down the process of aging but also induced angiogenesis, which contributed to improve the health of cardiovascular patients [116]. The expression of DNA damage response-1 (Redd1) was positively correlated with the aging. Cardiomyocyte senescence model in vivo and in vitro showed that the increase of Redd1 was accompanied by significantly cardiac dysfunction via promoting NF-kB signaling [117]. Serum-and-glucocorticoid inducible kinase-1 obviously inhibited the production of ROS to delay senescence and cardiovascular disease, which could be considered as a potential target in age-related cardiovascular diseases [118]. Deficiency of meteorin-like/Meteorin-β, as a secreted protein in the heart, could directly lead to heart dysfunction, which could be a rational biomarker for heart failure [119]. The dysregulation of phenylalanine in hepatic-cardiac axis played a vital role in cardiac aging via phenylalanine hydroxylase in mice [120]. FKBP5 enhanced inflammation via increasing the interactions with NF-κB regulatory kinases, and triggered aging and acute myocardial infarction [121].
Diabetes
Secretory factors released from senescent cells could lead to pancreatic beta cell dysfunction, insulin resistance, hence accelerated the development of type 2 diabetes. In addition, high blood sugar and metabolic changes in diabetes contributed to cellular aging [122]. Small extracellular vesicles induced formation and migration of angiogenesis via increasing the expression of miR-146a and Src, which might be a potential target for diabetes [123]. In a mouse model of diabetes, it was found that ablation of aging cardiac stem/progenitor cells repaired cardiac dysfunction induced by diabetes [124]. Hyperglycemia could aggravate human umbilical vein endothelial cells senescence, the expression of AQR and its co-expressed genes PLAU were positively related to aging and hyperglycemia process in human umbilical vein endothelial cells [125]. Decoy receptor 2 (DcR2) was highly related to kidney fibrosis and diabetic nephropathy. High expression of DcR2 promoted the expression of aging markers (p16 and p21). Importantly, DcR2 accelerated the progression of diabetes through interaction with peroxiredoxin 1 [126]. Knockdown of sodium-glucose cotransporter-2 (SGLT2) delayed senescence of HK-2 cells via reducing oxidative stress and DNA damage. Additionally, the animal experiment showed that inhibition of SLT2 expression could slow down the development of diabetic nephropathy by activating NRF2 pathway [127]. Increased insulin levels reduced autophagy and promoted aging of bone marrow stromal cells via acting on TGF-β receptor in type 2 diabetes mellitus, which suggested that TGF-β1 pathway was key to the treatment of bone diseases associated with diabetes [128].
Nonalcoholic fatty liver disease
Nonalcoholic fatty liver diseases (NAFLD), as a multifactorial disease, are characterized by a significant increase in lipids in the liver, leading to liver damage and ultimately hepatocellular carcinoma [129]. Researches have proved that senescence are involved in the pathogenesis and development of NAFLD with redundant inflammation and liver fibrosis (Fig. 5A) [130]. NAD+, sirtuin, and mTOR have been shown to play a vital role in senescence and NAFLD [129], [131]. Decreasing the formation of lipid droplets and promoting the lipophagy through AMPK/ULK1 and Akt/mTOR/ULK1 pathways were effective strategies to prevent NAFLD and liver aging [132]. Furthermore, high expression of FGF4 mitigated liver damage in NAFLD model mouse via FGFR4-AMPK-Caspase 6 signal axis [133]. Peroxisome proliferator-activated receptor α deletion disrupted the breakdown of fatty acids, resulting in increased lipid content in the liver of mice [134]. The amount of Gremlin 1 could be used as an indispensable index to predict hepatocyte and NAFLD in clinical patients. In addition, cell experiments proved that BMP4 was a significantly regulatory with anti-aging and anti-inflammatory effects [135]. Sentrin-specific protease 1 (SENP1), as an inhibitor of receptor-interacting serine/threonine-protein kinase 1, was decreased gradually with the aggravation of nonalcoholic steatohepatitis. Knockdown of SENP1 could activate the deSUMOylation of RIPK1, resulting in cell death and inflammation in mouse [136]. As a biomarker of aging, p16 could block the binding site of Foxo1, inhibiting the E2F1/FOXO1/IGF-1 binding at the Foxo1 signaling pathway and alleviating NAFLD [137]. Tubulin beta-4B chain (TUBB4B) was selected as an essential gene for NAFLD-associated hepatocellular carcinoma, which was negatively correlated with apoptosis, cell cycle arrest and cell senescence in HKCI2 and HKCI10 [138].
Fig. 5.
The association between cellular senescence and AAD. (A) Nonalcoholic fatty liver disease [129]; (B) Obesity [143].
Obesity
Obesity and aging are both associated with metabolic disorders, and their common features mainly included chronic inflammation and resident immune cells in adipose tissue (Fig. 5B) [139]. The proinflammatory factors in adipose tissue of obese mice were mainly supplied by senescent cells. Eliminating senescent cells in obese mice reduced circulating inflammatory mediators and alleviated adipose tissue dysfunction [140]. The subcutaneous fat depots of 227 obesity-associated patients showed high expression of senescence-associated β-galactosidase (SA-β-gal), which was related to the activity of insulin-like growth factor-binding protein 3 (IGFBP3), CCL2, plasminogen activator inhibitor 1 (PAI1), and leptin [141]. It was suggested that anti-aging interventions attenuated obesity-related metabolic disorders in severely obese patients. Insulin resistance aggravated the pathological process of obesity. p21 cells were found to be highly expressed in adipose tissue. Reducing the number of p21 cells via silencing NF-κB pathway could prevent and alleviate insulin resistance in obese mice [142]. Obesity and senescence increased the risk of neuropsychiatric disorders, and in vivo experiments showed that senescent cells contributed to anxiety and depression in obese mice [143]. Meg3 knockdown induced liver endothelial cell senescence and enhanced obesity-induced insulin resistance in obese mice [144]. The cellular mechanism of bone fragility in obese patients was described via obese bone marrow stromal stem cells (BM-MSCs) from human. Genes involved in glycolysis and oxidoreductase were predominantly expressed in BM-MSCs with enhancing insulin signaling, thus accelerating cell aging [145].
Macular degeneration and cataract
Aging is the main risk factor for macular degeneration (MD) and cataract. The main hallmark of MD and cataract is the loss of central vision and blindness [146]. Oxidative stress and oxidation products contributed the formation of hyperoxia region, which disrupted oxygen gradient homeostasis in the eyes, which was proved to play a significantly role in age-related MD and cataract [147]. The tear composition of macular patients was basically the same as that of normal people. It was proved that the expression of IL-6 and MCP-1 was directly proportional to the severity of MD through the tears of 19 MD patients [148]. The formation of 7-ketocholesterol was due to the increased oxidative stress, which induced the via oxiapoptophagy PIP2/PIP3/PDK1/PKB (Akt) signaling pathway [149]. Small peptides synthesized in mitochondria were metabolic receptors, which maintained cellular and mitochondrial homeostasis in MD and cataract [150]. The changes of sumoylation enzymes (E1, E 2 and E3) was tightly related to the formation of cataract, which could cause excessive oxidative stress in mouse [151]. Heat shock protein 90 homolog (HSP90) controlled the migration of retinal capillary endothelial cells and the expression of SA-β-Gal through activating NF-κB、HIF1α signal pathways in ARPE-19 cells [152].
Application of inorganic metal nanoparticles for aging and AAD
The human body contains a variety of metallic elements (zinc, iron, copper, etc.) that are essential for sustaining life and optimal functioning, although most of them are present in trace amounts. Zinc is an important element that makes up more than 200 enzymes in the human body, as well as an enzyme activator. Zinc is associated with memory, intelligence and immune function [153]. Iron is an important component of hemoglobin. If the iron supply is insufficient, the synthesis of hemoglobin will be affected and anemia will occur [154]. Copper is also essential to the human body as a unique and highly effective catalyst in biological systems. Copper is an active component of more than 30 enzymes and plays an important regulatory role in human metabolism. Copper involved in hematopoietic process and iron metabolism, involved in some enzyme synthesis and melanin synthesis [155]. IMNPs have been successfully applied in aging and AAD due to their necessity to human body and surface modifiable properties (Table 1) [156].
Table 1.
Summary of representative applications of inorganic NPs in aging and AAD.
| NPs type | Diseases | Model | Dose | Time | Outcomes | References |
|---|---|---|---|---|---|---|
| RGD-Au NPs | Neurodegenerative diseases | Hutchinson Gilford progeria syndrome mouse model | / | 3 weeks | Enhance hippocampal neurogenesis | [157] |
| Au NPs | Ovarian cancer | Patient-derived xenograft mouse model | 100–300 μg per mice | 14 days | Inhibited the growth of ovarian cancer | [158] |
| Au NPs (5 nm, 20 nm) | Ischemic injury | Oxygen-glucose deprivation/reperfusion cell model | 200–240 µg/L | 48h | Neuroprotective efects | [160] |
| Au NPs (5, 50, 100 nm) | Aging | PC12 cells | 3.12–100 µM | 24h | Au NPs at 100 nm could maintain maximum neuronal activity | [161] |
| Au NPs (5, 40, 100 nm) | Heart disease | Isoproterenol-induced hyperthyroid heart disease in Sprague-Dawley rats | 0.1 mM | 7 days | Size-dependent cardiac protective effect | [163] |
| H9c2 cell | 10 µg/mL | 24h | Decreased autophagy | |||
| Ag NPs | Obesity | High-fat diet mice | 100, 300 mg/kg | 2 weeks | Accelerated formation of steatohepatitis | [164] |
| Ag NPs | Myocardial infarction | Isoproterenol-induced myocardial infarction rat model | 2.5 mg/kg | 14 days | Cardioprotective effects | [165] |
| Ag NPs | Aging | Drosophila | 10–5 µg/mL | 14 days | Shorten lifespan | [166] |
| Ag NPs | Aging | Drosophila | 0.1, 1 µg/mL | 10 days | Shorten lifespan | [167] |
| Ag NPs | Aging | Endothelial cells | 3, 3.6, and 16 μg/mL | 24h | Significant reduced the cells elasticity | [168] |
| Ag NPs | Lung disease | MRC5 cells | 4 µg/mL | 10 days | Accelerated lung cellular senescence | [169] |
| C57BL/6 mice | 0.7 mg/m3 | 4 days | ||||
| Ag NPs | Cancer | MCF-7, A549 and Hep2 cell lines | 10–100 µg/mL | 48h | Good cytotoxicity in all cancer lines | [170] |
| Ag NPs | MCF-7 cell | 12.5 100 mg/mL | 48h | Remarkable cytotoxicity | [171] | |
| ZnO NPs (10–30 and 35–45 nm) |
Aging | Mesenchymal stem cells | 0–5 µg/mL | 24, 48, 72h | Size-dependent cytotoxicity | [172] |
| ZnO NPs | Aging | Drosophila | 0.1, 1, and 10 mM | 48 days | Induced aberrant phenotype in the progeny. | [174] |
| ZnO NPs | Neurodegenerative diseases | Adult and old male mice | 5.6 mg/kg | 4 weeks | Induced neurotoxicity | [175] |
| ZnO NPs | Colon cancer | Human HT29 colon cancer cell | 10–25 µg/mL | 48h | Inhibiting HT29 cell proliferation | [176] |
| ZnO NPs | Skin tumor | Primary mouse keratinocytes | 0.5–8 µg/mL | 48h | Induced cell death | [177] |
| SKH-1 mice | 10 % w/w | h | ||||
| ZnO NPs | Lung cancer | A549 human lung cancer cells | 2.5–7.5 µg/mL | 24h | Decreased cell viability | [178] |
| Fe3O4 NPs | Neurodegenerative diseases | L929 cells | 10 and 100 µg/mL | 24h | Largely reduced the level of H2O2 | [179] |
| Drosophila | 20 µg/mL | 8 days | Prolonged lifespan to 57 days | |||
| Glioma stem cells | 10 µg | 24, 48, 72h | Inhibits the tumourigenicity | [180] | ||
| SPIONs | Cancer | SCC-7 or 4T1 cells | 80–320 µM | 24h | Killed cancer cells under the influence of blue light | [181] |
| Tumor-bearing mouse model | 20–80 mM Fe | 20 days | Prevented cancer development | |||
| SPIONs | Obesity | Human primary adipocytes | / | / | Reduced adipocytes | [182] |
| IONPs | Nonalcoholic fatty liver disease | HepG2 | 5–10 µg/mL | 24h | Aggravated hepatic steatosis and liver injury | [183] |
| C57BL/6J mice | 5 mg Fe/kg | 8 weeks | ||||
| Fe2O3 NPs | Cardiovascular disease | Mice | 25, 5 mg/kg | 30 days | Cardiac dysfunction | [184] |
| TiO2 NPs | Cardiovascular disease | Sprague-Dawley rats | 2–50 mg/kg | 30, 90 days | Induced adverse cardiovascular effects | [185] |
| TiO2 NPs | Cardiovascular disease | H9c2 cell | 20, 40 µg/cm2 | 72h | Reduced metabolic activity and cell proliferation | [186] |
| TiO2 NPs | Gestational diabetes mellitus | Gestational diabetes mellitus mice | 10–250 mg/kg | 20 days | Reproductive toxicity | [187] |
| TiO2 NPs | Cancer | HCT-116 and Caco-2 cell lines | 0.01–1000 mg/L | 72h | Low genotoxicity | [188] |
| TiO2 NPs | Cancer | Liver and lung cancer metastasis mice model | 20 mg/kg | 21 days | Suppressed lung and liver cancer metastasis | [189] |
| TiO2 NPs | Cancer | 4T1 cells | 0.5–32 µg/mL | 2, 4, 6, 12h | Concentration-dependent cytotoxicity | [190] |
| BALB/c Model of 4T1 Mammary Carcinoma |
5 mg/kg | 21 days | Decreased tumor size | |||
| TiOx NPs | Cancer | Cancer stem cells | 200, 400 µg/mL | 48h | Inhibited migration and invasion of pancreatic cancer cells | [191] |
| BALB/c nude mouse tumors |
3 mg/mL | 30 days | Significantly reduced tumor size | |||
| TiO2 NPs | Skin | HaCat keratinocytes | 0.007–50 µg/cm2 | 24, 48, 7 days | Low cytotoxicity | [192] |
| Thallium acetate | Cancer | C6 and U373 cell lines | 5, 10, 50, 100, 200 µM | 24h | Antiproliferative effects in glioblastoma cells | [193] |
| Platinum | Lung cancer | A549 cells | 0.5 µM | 72h | Compound 15 exerted the most cytotoxicity | [199] |
| BALB/c nude mouse tumors |
5, 11.5 mg/kg | 28 days | Markedly inhibited tumor growth | |||
| Platinum complexes | Cancer | HepG2, MCF-7, SGV-7901 cell lines | 1–100 µM | 72h | Potent cytotoxicity | [202] |
| Platinum | Blind retinal diseases | Light-induced retinal degeneration mouse model | 0.3, 1 µM | 3 days | Maintained retinal function | [205] |
| 661 W cells | 1–1 nM | 24, 48h | ||||
| CeO2 NPs | Neurodegenerative diseases | Rat hippocampal culture neurons | 1 µM | 24h | Neuroprotective properties | [206] |
| CeO2 NPs | Neurodegenerative diseases | Scopolamine-induced Alzheimer rat model | 1–10 | 14 days | Enhanced cognitive ability | [207] |
| CeO2 NPs | Cancer | YM1, cancer stem cell like cell lines | 200–100 µg/mL | 24, 48h | Inhibited cell viability | [210] |
| CeO2 NPs | Cancer | MKN28 and BGC823 | 0.01–10 µg/mL | 72h | Suppressed gastric cancer | [211] |
| CeO2 NPs | Cancer | HT-29, NCI-H460, MRC-5, 518A2, HaCaT, DLD1 | 50–500 µM | 72h | Sensitive in HT-29 and 518A2 cell lines | [212] |
| CeO2 NPs | Heart failure | Isoproterenol-induced cardiac toxicity rats model | 0.5, 5 µg/kg | 5 weeks | Cardio protective effect | [213] |
| CeO2 NPs | Diabetes | Streptozotocin-induced rat model | 30 mg/kg | weeks | Attenuate detrimental effects of diabetes | [214] |
| CeO2 NPs | Diabetes | Diabetic mice | 60 mg/kg | 16 days | Reduce fetal abnormalities | [215] |
| CuO NPs | Neurodegenerative diseases | SH-SY5Y cell line | 1, 10, 50 µg/mL | 24h | Adverse effects | [217] |
| Cu/CuO NPs | Cancer | A549 cell line | 0.5–10 ng/mL–10 µg/mL | 72h | Cytotoxicity | [219] |
| Cu NPs | Cancer | SW480 cell line | 4–125 µg/mL | 24h | Significantly inhibited cell viability | [220] |
| Cu NPs | Cancer | MCF-7 and LoVo cell line | 1–50 µg/mL | 48h | Induced apoptosis | [221] |
| CuO NPs | Cardiovascular diseases | EA.hy 926 cell line | 5, 7.5, 10 and 15 µg/mL | 24h | Induced cell death | [224] |
| CuO NPs |
Cardiovascular diseases | Human umbilical vein endothelial cells |
10, 20, 40 µg/mL | 24h | Vascular toxicity | [225] |
Gold NPs
Gold NPs (Au NPs) have been found as the most potential due to their nontoxic feature, tunable size, shape, such as in sensing, image enhancement, and as delivery agents. After Au NPs were injected into the hippocampus of aged mice, Kat2a was activated under the action of electromagnetic field, which stimulated the production of hippocampal neurogenesis and alleviated the symptoms of aging [157]. Au NPs were non-toxic in orthotopic patient-derived xenograft mice model, and could significantly inhibited the size of tumor via down-regulating the expression of insulin growth factor binding protein 2 [158]. The photothermal therapy based on Au NPs could reduce the wrinkles and the size of facial pores by inhibiting the activity of sebaceous glands without obvious adverse reactions in Korean patients [159]. Au NPs at 5 nm reduced the activity of neurons, while Au NPs at 20 nm had a neuronal protective effect on rat cerebral cortical neurons, which activated Akt signaling and reduced the oxidative stress [160]. Au NPs at different sizes (5, 50, 100 nm) could enhance the activity of PIMT enzyme activity in p12 cells, among which 100 nm Au NPs showed the best effect via improving the anti-flutter activity [161]. Au NPs dispersed in acidic condition significantly promoted wound healing via inhibiting bacterial growth [162]. The effect of different sizes of Au NPs (5, 40, 100 nm) on heart was evaluated in isoproterenol-induced rats and H9C2 cells. The cardiac protective Au NPs was size-dependent in rats. When the size of Au NPs was less than 40 nm, it was myocardial protection, while the size was greater than 40 nm, it showed obviously myocardial toxicity. Additionally, Au NPs significantly decrease autophagy in H9C2 cells via reducing the expression of APG7 and caspase 12 [163].
Silver NPs
Owing to their peculiar oxidative effect, silver NPs (Ag NPs) are widely used as antibacterial agents, wound healing and coating over medical devices, and antiviral drugs. Ag NPs by oral administration had no obvious toxicity to normal mice, but could promote the process from steatosis to steatohepatitis in the liver of obese mice, which was mainly caused by enhancing the secretion of SASP (like Cd86, TNF-α and IL-6) and inhibiting fatty acid oxidation [164]. Ag NPs (intraperitoneal (i.p.) injection) significantly ameliorated the myocardial injury caused by isoproterenol, which was mainly reflected in the reduction of plasma biomarkers (Creatine Kinase-MB), oxidative stress, and the inhibition of NF-κB expression [165]. Ag NPs had no influence on the survival rate of Drosophila, while the lifespan of Drosophila remarkably decreased when exposed to Ag NPs. Ag NPs had chronic effects via inducing oxidative stress and activating the nuclear factor erythroid 2-related factor 2 (NRF2) signaling pathway [166]. Meanwhile, long-term exposure to 0.1 and 1 μg/mL Ag NPs significantly reduced the lifespan of Drosophila, primarily by upregulating Transcription regulator galc (GAL4) expression [167]. After treatment with 3 μg/mL Ag NPs for 24h, the elasticity of early endothelial cells was significantly decreased, while the elasticity of late endothelial cells was increased under the same treatment conditions. which may be related to the increase of reactive oxygen species [168]. Ag NPs showed no acute toxicity to MRC5 cells, while treatment with Ag NPs (4 µg/mL) for 10 days induced senescence of lung cells with obvious cellular senescence characteristics, including cell size enlargement and SA-β-gal enhancement. In addition, Ag NPs (exposed in the air) accelerated lung cellular senescence in mice via improving the expression of cyclooxygenase-2/prostaglandin E2 pathway [169]. Ag NPs could decrease the cell viability of various type of cancer cells (MCF-7, A549 and Hep2 cells). When the treated concentration was up to 100 μg/mL, the survival rate of MCF-7, A549 and Hep2 cells only remained 20 %. The inhibitory effect of tumor cells was related to the excessive expression of ROS and cell apoptosis [170]. Ag NPs obtained through biosynthesis method could significantly inhibit the survival rate of MCF-7 cells with the half maximal inhibitory concentration (IC50) value below 65 μg/mL [171].
Zinc oxide NPs
Zinc oxide (ZnO) NPs are widely used in dentistry because of their remarkable optical, magnetic, morphological properties. In recent years, ZnO NPs have shown promising anti-microbial, regenerative, and mechanical properties. ZnO NPs indicated size-dependent cytotoxicity on mesenchymal stem cells. The toxicity of ZnO NPs (10–30 nm) was obviously higher than that in ZnO NPs (35–45 nm). Additionally, both size of ZnO NPs could induced the process of aging in vitro, and the expression of SASP (NF-kB and p53) was significantly higher in the smaller size of ZnO NPs [172]. In addition, ZnO NPs revealed anti-diabetic and anti-aging abilities via inhibiting the expression of inflammation-related proteins (COX-1, COX-2, 15-LOX and sPLA2) [173]. ZnO NPs (0.1, 1, and 10 mM) caused chronic toxicity and induced heritable phenotypic variation in Drosophila, like single or deformed wing [174]. ZnO NPs (5.6 mg/kg body weight, i.p. injection) induced systemic inflammation and oxidative stress in aged mice, causing pathological changes in the hippocampus and neurotoxicity. However, ZnO NPs showed no significant neurotoxicity to adult mice [175]. ZnO NPs inhibited the proliferation of HT29 colon cancer cells in a dose-dependent manner by affecting mitochondrial function [176]. Low exposure to UVB (25 mJ/cm2) or ZnO NPs (1 µg/mL) had no significant effect on cell viability. However, ZnO NPs enhanced the uptake capacity of primary mouse keratinocytes (PMKs). Combined ZnO NPs with ultraviolet radiation B (UVB) exposure obviously decreased the survival rate of PMKs with more than 30 % of cell death via inducing DNA damage and ROS production [177]. ZnO NPs inhibited cell survival of A549 human lung cancer cells via affecting the translation of 1448 genes. After inhibition of TLNRD1 or CCNB1IP1 expression, the toxicity of ZnO NPs was decreased [178].
Iron oxide NPs
Iron oxide NPs (IONPs) are extensively adopted in the field of photodynamic therapy, targeted drug delivery, hyperthermia therapy, and chemotherapy due to their excellent magnetic properties, good biocompatibility, and low price. IONPs showed intrinsic peroxidase-like activities in a dosage dependent manner, which could decompose ROS and decrease the oxidative stress in Drosophila AD model. Iron oxide NPs (range of 10 and 100 µg/mL) did not affect the viability of L929 cells. Iron oxide NPs at 200 µg/mL extended the lifespan of Drosophila to 57 days which was 8 days longer than control flies [179]. Superparamagnetic IONPs (SPIONs) inhibited the proliferation and invasion of glioma stem cells via inhibiting the expression of RN1, CCND1, CDK4 and Ki67 [180]. Ferrihydrite NPs were well biocompatible in SCC-7 and 4T1 cells, but the cell viability significantly decreased when the cells were exposed to ferrihydrite NPs and blue light via promoting DNA fragmentation and glutathione peroxidase 4. Additionally, ferrihydrite NPs promoted the differentiation of macrophages toward tumor-killing type [181]. SPIONs remarkably promoted the expression of SEC16B gene, contributing to the phagocytosis of apoptotic cells, which might be the potential mechanism for SPIONs to treat obesity and type 2 diabetes [182]. Amino-coated IONPs accelerated the formation of lipogenesis and inflammatory responses in high-fat diet mice, but showed no toxicity on normal mice [183]. Mice exposed to 25 and 50 mg/kg Fe2O3 NPs (i.p injection) for 30 days caused seriously cardiac dysfunction, and myocardial cells in the 50 mg/kg group developed necrosis, mainly through upregulation of cytochrome c and caspase 3 [184].
Titanium dioxide NPs
Titanium dioxide NPs (TiO2 NPs) show excellent antibacterial property when exposed to ultraviolet light and are widely used to treat bacterial infections. Treatment with titanium dioxide NPs (TiO2 NPs, 2–50 mg/kg) for 30 days or 90 days decreased myocardial enzyme activity (lactate dehydrogenase, alpha-hydroxybutyrate dehydrogenase and creatine kinase) and promoted the expression of SASP (IL-6 and TNF-α), thus inducing adverse cardiovascular effects [185]. Moreover, TiO2 NPs showed nonnegligible toxicity to H9c2 rat cardio myoblasts. 40 µg/cm2 TiO2 NPs inhibited cell proliferation by 40 % and reduced metabolic activity by 60 %. In addition, oxidative stress was significantly increased after treatment [186]. TiO2 NPs aggravated the increase of blood glucose and decreased of body weight in mice with gestational diabetes. In addition, Ti was detected in both the placenta and the fetus, causing the fetus to develop abnormally. The reproductive toxicity of TiO2 NPs was related to the induction of oxidative stress and the activation of ERS pathway to aggravate apoptosis [187]. The cell viability of HCT 116 and Caco-2 was reduced in a dose-dependent manner by TiO2 NPs. When the concentration was up to 100 mg/L, the survival rate of both cell lines was lower than 30 %, which suggested low genotoxicity of TiO2 NPs [188]. After treated with 20 mg/kg TiO2 NPs (intravenously (i.v.) injection) for 21 days, the body weight of mice decreased significantly, and the inflammatory factors increased significantly, which was the mechanism of TiO2 NPs to inhibit cancer metastasis [189]. TiO2 NPs (8 μg/mL) treated for 24h could decrease the cell viability of 4T1 cell by 50 %, and significantly inhibited tumor growth in 4T1 breast BALB/c mouse tumor model via promoting apoptosis [190]. TiOx NPs combined with 2 or 5Gy radiation treatment decreased the cell viability of pancreatic cancer stem cells by more than 50 %. Furthermore, TiOx NPs (i.v. injection) enhanced the radiation effect to inhibit the tumor size on BALB/c nude mouse via facilitating the production of ROS and restrain the AKT signaling pathway [191]. After 24h of skin exposure to TiO2 NPs, Ti was only found in the stratum corneum and epidermis, and level in the dermis was below limit of detection (5 μg/L). Additionally, TiO2 NPs showed low cytotoxic effect on HaCaT cells when the concentration was up to 44 μg/cm2 [192].
Thallium NPs
Thallium NPs show remarkable potential in MRT applications with long half-life (t1/2 = 73h). Additionally, thallium ions can be used to investigate the membrane potential via live-cell imaging technique. Compared with U373 cells, thallium (I) acetate could significantly inhibit the proliferation of C6 cells, which was concentration-dependent. The survival rate of C6 cells was reduced by 40 % when the concentration of thallium(I) acetate was at 50 μM [193]. Channel activity of TRPM5 and TRPM4 was tightly associated with cardiac arrhythmias and diabetes. A thallium-sensitive dye was developed to detect the activity of TRPM5 or TRPM4 [194]. Furthermore, thallium-201 was considered as a powerful examination for recognizing cardiovascular or diabetes-related diseases. Body mass index, hemoglobin and age were positively correlated with thallium-201 results [195], [196]. Exposure to thallium conditions caused severe metabolic dysfunction and liver dysfunction which deteriorated obesity or NAFLD [197].
Platinum NPs
Platinum are widely applied in cancer therapy and photo-thermal therapy because of their ultra-fine structures, large surface area, tuned porosity. Platinum is a component of chemotherapy drugs, and many types of tumors have developed resistance to platinum-based chemotherapy. The latest research shows that it could also act as a catalyst to improve the tumor microenvironment, helping to improve the effectiveness of tumor treatment [198]. A series of platinum derivatives were synthesized and showed potential inhibitory effect on 549 cells. Compound 15 exerted the best effect with IC50 below 0.43 μM, which was much lower than that of cisplatin. In vivo studies indicated that compound 15 (i.v. injection) significantly reduced tumor size without reducing body weight. Anti-tumor effect of compound 15 was closely related to induce mitochondrial apoptosis pathway [199]. Serine biosynthesis and MKK4 kinase activity mediated resistance to platinum-based chemotherapy [200], [201]. The three platinum complexes all showed obvious inhibitory effects on the proliferation of HepG2, MCF-7, SGV-7901 cell lines, among which complex 1 exerted the best inhibitory effect with IC50 below 2.5 μM. Complex 1 significantly promoted the uptake of all three cancer lines and activated caspase-3 to induce cell apoptosis [202]. The THSD7B mutation inhibited pathways associated with cell death and reduced the immune response, leading to platinum resistance in lung cancer patients [203]. The number of toxic and nontoxic seeds in miRNA activity was highly related to platinum resistance in ovarian cancer [204]. Additionally, platinum could obviously reverse the retinal degeneration caused by blue light exposure via decreasing oxidative damage-related cell apoptosis [205].
Cerium oxide NPs
Cerium oxide NPs (CeO2 NPs) exhibited non-negligible anti-inflammatory effect and wound healing ability due to their antibacterial properties, stability, and a high surface area. Because of their remarkable antioxidant properties, CeO2 NPs have been widely used in the treatment of Alzheimer's disease. CeO2 NPs (6 mg/kg, intranasal delivery) significantly improved cognitive ability of Alzheimer rat model, which was in a dose-dependent manner. In addition, prevention or treatment of CeO2 NPs could effectively reduce the proportion of necrotic neuron cells by more than 13 %, which indicated that CeO2 NPs had a strong neuroprotective effect [206], [207], [208]. In addition, CeO2 NPs play an anti-cancer role by increasing antioxidant levels and enhancing tumor cell sensitivity to chemotherapy and radiation therapy [209]. CeO2 NPs induced highly expression of antioxidant genes, like superoxide dismutase 2 (SOD) and catalase-A (CAT) in YM1 and cancer stem cell-like cell lines [210]. CeO2 NPs enhanced the sensitivity of pancreatic cancer cells to radiotherapy by activating the expression of apoptosis signaling kinase 1. High concentration CeO2 NPs (10 µg/mL) significantly inhibited the migration and proliferation of MKN28 and BGC823 cells by promoting DHX15 expression and related pathways [211]. CeO2 NPs showed different toxicities in various human cell lines, of which 518A2 and HT-29 cells were most sensitive with IC50 between 100 and 200 mΜ. However, CeO2 NPs exerted no toxicity in MRC-5 and HaCaT cell lines [212]. CeO2 NPs significantly reversed the cardiac toxicity-caused by isoproterenol via decreasing the oxidative stress. After treatment with 0.5 and 5mg/kg CeO2 NPs, the cardiac markers (CK-MB, LDH) were remarkably decreased with highly expression of antioxidant enzymes [213]. Streptozotocin-induced diabetic rats showed apparently testicular dysfunction, and CeO2 NPs could promote the expression of NFE2-related factor 2, improving sperm parameters, and enhancing the fertility of diabetic rats [214]. Additionally, CeO2 NPs reduced fetal abnormalities induced by gestational diabetes mellitus via decreasing ROS formation [215].
Copper NPs
Copper NPs (Cu NPs) highlighting antimicrobial, anticancer, antiparasitic, antidiabetic, and antioxidant studies owing to their non-toxic and highly stable features. Cu NPs could bind to amyloid deposits and promote the production of ROS, thus exacerbating cognitive dysfunction [216]. Cu oxide (CuO) NPs (50 nm) promoted the oligomerization of Aβ1-42 in SH-SY5Y cells in a dose-dependent manner, then reduced cell survival rate and accelerated the progression of Alzheimer's disease [217]. Cu NPs have become an effective means of tumor treatment by influencing the oxidation level, regulating protein homeostasis, participating in inflammation and inducing cuproptosis in various tumor cells [218]. Cu/CuO NPs showed obviously cytotoxicity to WI-38 cells (human lung normal cells) and A549 cells (lung cancer cells) with IC50 in 0.20 μg/mL and 0.21 μg/mL, respectively [219]. Cu NPs could promote the expression of Bax and p53, and reduce the content of Bcl-2, then inhibited the viability of SW480 cells, which proved the anti-tumor effect of Cu NPs [220]. Meanwhile, Cu NPs significantly increased the expression of apoptosis-related genes (Bax, P53 and Caspases 3) to accelerate the apoptosis of MCF-7 and LoVo cell lines [221]. Cu, as a type of micronutrient, could regulate the homeostasis of enzymes, like superoxide dismutase. In addition, excessive intake of Cu might contribute to vascular dysfunction [222]. Cu sulfide (CuS) NPs activated autophagy in vascular smooth muscle cells via targeting TRPV1, decelerated the atherosclerosis [223]. Exposure to CuO NPs caused vascular endothelial injury, which could be alleviated by promoting the expression of PINK1 and TAX1BP1 and increasing mitophagy [224]. CuO NPs caused DNA damage and cell death of human umbilical vein endothelial cells via activating the p38 mitogen-activated protein kinase signaling pathway [225].
Study on biocompatibility and delivery mechanism of metal nanoparticles
Most metal nanoparticles are widely used in imaging and active or passive tumor cell targeting as carrier and contrast agents owing to their high surface-to-volume ratio, surface plasmon resonance, surface chemistry and multi-functionalization, easy synthesis, and stability [226], [227]. Despite all these advantages, biocompatibility of metal nanoparticles is the most critical factor that must be considered before their clinical applications. In general, cells, tissues or organisms treated with metal nanoparticles show little adverse consequences such as oxidative stress, DNA damage, mutagenesis or apoptosis. It shows that metal nanoparticles have good biocompatibility [228]. For example, Au NPs, Ag NPs, and IONPs are considered to be promising gene carrier for cancer treatment because of their excellent biocompatibility [229]. The cytotoxicity of metal nanoparticles is closely related to their shape, size, surface properties, and chemical composition, which limit their application in biomedical field [230]. Importantly, the biocompatibility of metal nanoparticles is also dependent on the biological environment [231].
When the metal nanoparticles enter the biological environment, they will adsorb various biomolecules such as nucleic acids, proteins, etc., and form a protein-based layer on the surface, which is also known as the “corona” [232]. The protein corona can affect the physicochemical properties, toxicity and biodistribution of metal nanoparticles throughout the body [233]. To reduce the effect of the protein corona, the surface of metal nanoparticles is covered with chitosan, collagen, poly (lactic acid), poly (lactic-co-glycolic acid), polycaprolactone, poly (ethylene glycol), polyvinyl alcohol, polyethyleneimine, quantum dots, polypeptide, lipid NPs, and exosomes. This reduces immune recognition, extends the blood circulation, and enhances efficient biodistribution to the targeted site of metal nanoparticles [234], [235], [236]. Although the surface coating will reduce the recognition of metal nanoparticles by the reticuloendothelial system, complete avoidance is rarely competed. After exposure of metal nanoparticles, cells, tissues or organisms will produce a series of mechanisms, such as ROS, cell cycle arrest, DNA damage (Fig. 6) [231], [237], [238], [239].
Fig. 6.
The systemic delivery of metal nanoparticles in biomedical applications. (A) Au NPs [231]; (B) Ag NPs [237]; (C) ZnO NPs [238]; (D) IONPs [239].
Conclusion and future perspectives
This review aims to give a comprehensive summary of common features of cellular senescence, proteins and signaling pathways in aging and AAD, as well as the biological applications of IMNPs in these diseases. Firstly, we summarized the morphological changes of senescent cells in detail, which evidenced by the enlarged cell size, irregular shape, PM composition change via overexpression of PtdSer, lysosomal proteins increase, mitochondria accumulation, nuclear changes. The morphological changes mainly affected by mTORC1 and SIRT pathways and related proteins. Secondly, aging and AAD were summarized in detail. Common pathophysiological features of aging and AAD included mitochondrial homeostasis imbalance, oxidative stress overexpression, cellular senescence, chronic inflammation, and protein homeostasis degradation. In addition, insulin and IGF 1, mTORC1, AMPK and SIRT1 signaling pathways were considered to be key pathways involved in aging and AAD. Aging was not only the cause of related diseases, but also the occurrence and development of related diseases which eventually accelerated aging. More importantly, aging was a double-edged sword, which aggravated the pathological manifestations of related diseases, making the disease worse. However, aging accelerated the progress of wound healing, and was a new strategy to prevent and treat cancer. Finally, we reviewed the applications of different kinds of IMNPs in aging and AAD. Basically, all IMNPs could improve neurodegenerative diseases (except CuO NPs) and inhibit tumor cell growth. We concluded that there were significant differences in the effects of various types of IMNPs on multiple disease models. For example, IONPs showed obvious cardio toxicity, but they could inhibit the proliferation and growth of tumor cells and slow down the development of neurodegenerative diseases. However, Au and Ag NPs not only reduced myocardial damage, but also be an alternative option to prevent and treat cancer. In future, we should pay attention to the characteristics of IMNPs, and select the IMNPs with the least toxicity and the best efficacy in different diseases. Meaningfully, the molecular mechanism or targets of IMMPs on aging and AAD should be studied. More in-depth research on IMNPs for the treatment of aging and AAD in vivo and in vitro should implemented. Additionally, more clinical or preclinical trials should be carries out in the future to broaden the biological applications of IMNPs.
Author contributions
Yuqing Meng and Chong Qiu collected and analyzed articles, and wrote the manuscript. Jigang Wang reviewed the manuscript. Junzhe Zhang, Yanqing Liu, Yongping Zhu, Haining Lv, Fei Xia, Qiuyan Guo, Qianli Shi assisted in manuscript and provided some helpful suggestions. The manuscript has been read and approved by all co-authors.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
This work was supported by the Fundamental Research Funds for the National Natural Science Foundation (No. 82373821, 82204322, 82204672, and 82104480), Scientific and technological innovation project of China Academy of Chinese Medical Sciences (CI2023E002), the Central Public Welfare Research Institutes of China Academy of Chinese Medical Sciences (No. ZZ15-YQ-065, ZZ15-YQ-062, ZZ16-ND-10-05, ZZ16-ND-10-13, ZZ16-ND-10-17, ZZ17-ND-10-05, ZZ16-ND-10-19, ZZ14-YQ-050, ZZ14-YQ-055, ZZ14-YQ-059, ZZ14-YQ-060, ZZ13-YQ-105 and ZZ16-YQ-046), the Young Elite Scientists Sponsorship Program by CACM (2021QNRC2B29).
Biographies

Yuqing Meng received her Ph.D. degree from State Key Laboratory for Quality Ensurance and Sustainable Use of Dao-di Herbs, Artemisinin Research Center, and Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences. She is an Assistant Researcher in Artemisinin Research Center. Her major research interests include interaction between cells and inorganic nanoparticles and drug targets and mechanisms of natural products.

Junzhe Zhang received his Ph.D. degree in 2019 from Institute of High Energy Physics and University of the Chinese Academy of Sciences. From 2019, he has served as an assistant researcher in State Key Laboratory for Quality Ensurance and Sustainable Use of Dao-di Herbs, Artemisinin Research Center, and Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences. His research focuses on the: the safety and metabolism of inorganic nanomaterials, nano-proteomics, and analytical method development.

Yanqing Liu received her Ph.D. degree in 2018 from Institute of Biophysics, Chinese Academy of Sciences. From 2019, she has served as an assistant researcher in State Key Laboratory for Quality Ensurance and Sustainable Use of Dao-di Herbs, Artemisinin Research Center, and Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences. She is mainly engaged in the pharmacological research of antimalarial drugs and anti-tumor drugs, as well as the study of drug targets and mechanisms.

Yongping Zhu received her Ph.D degree in 2018 from Institute of Biophysics, Chinese Academy of Sciences. From 2018, she has served as an assistant researcher in State Key laboratory for Quality Ensurance and Sustainable Use of Dao-di Herbs, Artemisinin Research Center, and Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences. Her research focuses on the anticancer mechanism of celastrol and the drug targets and mechanisms of other natural products.

Haining Lv obtained Master's (2014) and PhD degree (2017) in Natural Medicine at Peking University, under the supervision of Professors Peng-Fei Tu and Yong Jiang. Currently, he works as a staff scientist in State Key Laboratory for Quality Ensurance and Sustainable Use of Dao-di Herbs, Artemisinin Research Center, and Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences, with Professors Youyou Tu, Jigang Wang, and Chengchao Xu, where his research interests include discovering new biosynthetic enzymes in medicinal plants, exploring their catalytic mechanism, and reconstituting the metabolic pathway in tractable host plants, such as Nicotiana benthamiana, to improve production yields.

Fei Xia received her Ph.D. degree in 2019 from University of Chinese Academy of Sciences. From 2022, she has served as an associate professor in Artemisinin Research Center, and Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences. Her research focuses on synthetic chemistry、nanodrug delivery、target identification and mechanism of active molecules in traditional chinese medicine.

Qiuyan Guo received the Ph.D. degree of pharmacy in 2021 from Tokyo University of Pharmacy and Life Sciences, and the Ph.D. degree of traditional Chinese medicine in 2020 from China Academy of Chinese Medical Sciences. She has served as an assist professor in State Key laboratory for Quality Ensurance and Sustainable Use of Dao-di Herbs, Artemisinin Research Center, and Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences. Her research focuses on the: Pharmacological study on anti-inflammatory and immune traditional Chinese medicine.

Qianli Shi received her Ph.D. degree in 2019 from the Academy of Military Sciences of the Chinese People's Liberation Army. She worked in Artemisinin Research Center, and Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences form 2019. She majors in Pharmacology of Chinese Medicine, focuses on exploring the effective ingredients and mechanism of Chinese medicine.

Chong Qiu received his bachelor degree and doctor degree from School of Pharmaceutical Science, Peking University in 2014 and 2019, respectively. He is an associate professor of State Key Laboratory for Quality Ensurance and Sustainable Use of Dao-di Herbs, Artemisinin Research Center, and Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences. His major research interests include nanocarriers for drugs delivery and anti-cancer mechanisms of natural products.

Jigang Wang received his Ph.D. from National University of Singapore. His research focuses on the mechanism of artemisinin antimalarial and anti-cancer and the drug targets and mechanisms of action of other drugs and natural products. The research group combines chemical biology, proteomics, metabolomics, molecular biology, single-cell sequencing and other related cross-cutting research methods, and conducts systematic research on the targets and mechanisms of action of drugs.
Contributor Information
Chong Qiu, Email: cqiu@icmm.ac.cn.
Jigang Wang, Email: jgwang@icmm.ac.cn.
References
- 1.He S., Sharpless N.E. Senescence in health and disease. Cell. 2017;169:1000–1011. doi: 10.1016/j.cell.2017.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Saul D., Kosinsky R.L. Epigenetics of aging and aging-associated diseases. Int J Mol Sci. 2021;22:401. doi: 10.3390/ijms22010401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kubben N., Misteli T. Shared molecular and cellular mechanisms of premature ageing and ageing-associated diseases. Nat Rev Mol Cell Bio. 2017;18:595–609. doi: 10.1038/nrm.2017.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Huang W., Hickson L.J., Eirin A., Kirkland J.L., Lerman L.O. Cellular senescence: the good, the bad and the unknown. Nat Rev Nephrol. 2022;18:611–627. doi: 10.1038/s41581-022-00601-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.López-Otín C., Blasco M.A., Partridge L., Serrano M., Kroemer G. Hallmarks of aging: An expanding universe. Cell. 2023;186:243–278. doi: 10.1016/j.cell.2022.11.001. [DOI] [PubMed] [Google Scholar]
- 6.Hernandez-Segura A., Nehme J., Demaria M. Hallmarks of Cellular senescence. Trends Cell Biol. 2018;28:436–453. doi: 10.1016/j.tcb.2018.02.001. [DOI] [PubMed] [Google Scholar]
- 7.Kim S.W., Im G.B., Kim Y.J., Kim Y.H., Lee T.J., Bhang S.H. Bio-application of inorganic nanomaterials in tissue engineering. Adv Exp Med Biol. 2020;1249:115–130. doi: 10.1007/978-981-15-3258-0_8. [DOI] [PubMed] [Google Scholar]
- 8.Bhat B.B., Kamath P.P., Chatterjee S., Bhattacherjee R., Nayak U.Y. Recent updates on nanocosmeceutical skin care and anti-aging products. Curr Pharm Design. 2022;28:1258–1271. doi: 10.2174/1381612828666220321142140. [DOI] [PubMed] [Google Scholar]
- 9.Martano S., Matteis V.D., Cascione M., Rinaldi R. Inorganic nanomaterials versus polymer-based nanoparticles for overcoming neurodegeneration. Nanomaterials. 2022;12:2337. doi: 10.3390/nano12142337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Du C., Zhou L., Qian J., He M., Zhang Z.G., Feng C., et al. Ultrasmall zwitterionic polypeptide-coordinated nanohybrids for highly efficient cancer photothermal ferrotherapy. ACS Appl Mater Inter. 2021;13:44002–44012. doi: 10.1021/acsami.1c11381. [DOI] [PubMed] [Google Scholar]
- 11.Childs B.G., Durik M., Baker D.J., Deursen J.M. Cellular senescence in aging and age-related disease: from mechanisms to therapy. Nat Med. 2015;21:1424–1435. doi: 10.1038/nm.4000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Reiner A.V. DNA content, cell size, and cell senescence. Trends Biochem Sci. 2019;44:645–647. doi: 10.1016/j.tibs.2019.04.013. [DOI] [PubMed] [Google Scholar]
- 13.Lanz M.C., Zatulovskiy E., Swaffer M.P., Zhang L., Ilerten I., Zhang S., et al. Increasing cell size remodels the proteome and promotes senescence. Mol Cell. 2021;82:1–63. doi: 10.1016/j.molcel.2022.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Xie S.C., Swaffer M., Stotheim J.M. Eukaryotic cell size control and its relation to biosynthesis and senescence. Annu Rev Cell Dev Bi. 2022;38:1–29. doi: 10.1146/annurev-cellbio-120219-040142. [DOI] [PubMed] [Google Scholar]
- 15.Sadaie M., Dillon C., Narita M., Young A.R., Cairney C.J., Godwin L.S., et al. Cell-based screen for altered nuclear phenotypes reveals senescence progression in polyploid cells after Aurora kinase B inhibition. Mol Biol Cell. 2015;26:2971–2985. doi: 10.1091/mbc.E15-01-0003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Qiu C., Wu Y., Guo Q., Shi Q., Zhang J., Meng Y., et al. Preparation and application of calcium phosphate nanocarriers in drug delivery. Mater Today Bio. 2022;17 doi: 10.1016/j.mtbio.2022.100501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cormenier J., Martin N., DesléJ S.-C., Pourtier A., Abbadie C., Pluquet O. The ATF6α arm of the unfolded protein response mediates replicative senescence in human fibroblasts through a COX2/prostaglandin E 2 intracrine pathway. Mech Ageing Dev. 2018;170:82–91. doi: 10.1016/j.mad.2017.08.003. [DOI] [PubMed] [Google Scholar]
- 18.Nagata S., Sakuragi T., Segawa K. Flippase and scramblase for phosphatidylserine exposure. Curr Opin Immunol. 2020;62:31–38. doi: 10.1016/j.coi.2019.11.009. [DOI] [PubMed] [Google Scholar]
- 19.Hyun D.H. Plasma membrane redox enzymes: new therapeutic targets for neurodegenerative diseases. Arch Pharm Res. 2019;42:436–445. doi: 10.1007/s12272-019-01147-8. [DOI] [PubMed] [Google Scholar]
- 20.Li M., Gong W., Chen J., Zhang Y., Ma Y., Tu X. PPP3R1 promotes MSCs senescence by inducing plasma membrane depolarization and increasing Ca2+ influx. Int J Mol Sci. 2023;24:4421. doi: 10.3390/ijms24054421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Qiu C., Wu Y.Y., Shi Q.L., Guo Q.Y., Zhang J.Z., Meng Y.Q., et al. Advanced strategies for nucleic acids and small-molecular drugs in combined anticancer therapy. Int J Biol Sci. 2023;19(3):789–810. doi: 10.7150/ijbs.79328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Qiu C., Xia F., Zhang J.Z., Shi Q.L., Meng Y.Q., Wang C., et al. Advanced Strategies for Overcoming Endosomal/Lysosomal Barrier in Nanodrug Delivery. Research (Wash D C) 2023;6:0148. doi: 10.34133/research.0148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Saitou M., Lizardo D.Y., Taskent R.O., Millner A., Gokcumen O., Atilla-Gokcumen G.E. An evolutionary transcriptomics approach links CD36 to membrane remodeling in replicative senescence. Mol Omics. 2018;14:237–246. doi: 10.1039/c8mo00099a. [DOI] [PubMed] [Google Scholar]
- 24.Singh S., Pandey K.B., Rizvi S.I. Erythrocyte senescence and membrane transporters in young and old rats. Arch Physiol Biochem. 2016;122:228–234. doi: 10.1080/13813455.2016.1190761. [DOI] [PubMed] [Google Scholar]
- 25.Avila F., Tringali C., Papini N., Anastasia L., Croci G., Massaccesi L., et al. Identification of lysosomal sialidase NEU1 and plasma membrane sialidase NEU3 in human erythrocytes. J Cell Biochem. 2013;114:204–211. doi: 10.1002/jcb.24355. [DOI] [PubMed] [Google Scholar]
- 26.Yang C., Wang X. Lysosome biogenesis: regulation and functions. J Cell Biol. 2021;220:e202102001. doi: 10.1083/jcb.202102001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li Y., Xu M., Ding X., Yan C., Song Z., Chen L., et al. Protein kinase C controls lysosome biogenesis independently of mTORC1. Nat Cell Biol. 2016;18:1065–1077. doi: 10.1038/ncb3407. [DOI] [PubMed] [Google Scholar]
- 28.Roh K., Noh J., Kim Y., Jang Y., Kim J., Choi H., et al. Lysosomal control of senescence and inflammation through cholesterol partitioning. Nat Metab. 2023;5:398–413. doi: 10.1038/s42255-023-00747-5. [DOI] [PubMed] [Google Scholar]
- 29.Rovira M., Sereda R., Pladevall-Morera D., Ramponi V., Marin I., Maus M., et al. The lysosomal proteome of senescent cells contributes to the senescence secretome. Aging Cell. 2022;21:e13707. doi: 10.1111/acel.13707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Suda M., Shimizu I., Katsuumi G., Hsiao C.L., Yoshida Y., Matsumoto N., et al. Glycoprotein nonmetastatic melanoma protein B regulates lysosomal integrity and lifespan of senescent cells. Sci Rep. 2022;12:6522. doi: 10.1038/s41598-022-10522-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Irazoki A., Martinez-Vicente M., Aparicio P., Aris C., Alibakhshi E., Rubio-Valera M., et al. Coordination of mitochondrial and lysosomal homeostasis mitigates inflammation and muscle atrophy during aging. Aging Cell. 2022;21:e13583. doi: 10.1111/acel.13583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Colacurcio D.J., Nixon R.A. Disorders of lysosomal acidification—the emerging role of v-ATPase in aging and neurodegenerative disease. Ageing Res Rev. 2016;32:75–88. doi: 10.1016/j.arr.2016.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kaushik S., Tasset I., Arias E., Pampliega O., Wong E., Martinez-Vicente M., et al. Autophagy and the hallmarks of aging. Ageing Res Rev. 2021;72 doi: 10.1016/j.arr.2021.101468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhang H., Simon A.K. Polyamines reverse immune senescence via the translational control of autophagy. Autophagy. 2019;16:181–182. doi: 10.1080/15548627.2019.1687967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Perera R.M., Zoncu R. The lysosome as a regulatory hub. Annu Rev Cell Dev Bi. 2016;32:223–253. doi: 10.1146/annurev-cellbio-111315-125125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lawrence R.E., Zoncu R. The lysosome as a cellular centre for signalling, metabolism and quality control. Nat Cell Biol. 2019;21:133–142. doi: 10.1038/s41556-018-0244-7. [DOI] [PubMed] [Google Scholar]
- 37.Savini M., Folick A., Lee Y.T., Jin F., Cuevas A., Tillman M.C., et al. Lysosome lipid signalling from the periphery to neurons regulates longevity. Nat Cell Biol. 2022;24:906–916. doi: 10.1038/s41556-022-00926-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ghosh-Choudhary S.K., Liu J., Finkel T. The role of mitochondria in cellular senescence. FASEB J. 2021;35:e21991. doi: 10.1096/fj.202101462R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Miwa S., Kashyap S., Chini E., Zglinicki T. Mitochondrial dysfunction in cell senescence and aging. J Clin Invest. 2022;132:e158447. doi: 10.1172/JCI158447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ziegler D.V., Vindrieux D., Goehrig D., Jaber S., Collin G., Griveau A., et al. Calcium channel ITPR2 and mitochondria–ER contacts promote cellular senescence and aging. Nat Commun. 2021;12:720. doi: 10.1038/s41467-021-20993-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Janikiewicz J., Szymański J., Malinska D., Patalas-Krawczyk P., Michalska B., Duszyński J., et al. Mitochondria-associated membranes in aging and senescence: structure, function, and dynamics. Cell Death Dis. 2018;9:332. doi: 10.1038/s41419-017-0105-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ahumada-Castro U., Puebla-Huerta A., Cuevas-Espinoza V., Lovy A., Cardenas J.C. Keeping zombies alive: the ER-mitochondria Ca2+ transfer in cellular senescence. Bba-Mol Cell Res. 2021;1868 doi: 10.1016/j.bbamcr.2021.119099. [DOI] [PubMed] [Google Scholar]
- 43.Correia-Melo C., Marques F.D.M., Anderson R., Hewitt G., Hewitt R., Cole J., et al. Mitochondria are required for pro-ageing features of the senescent phenotype. EMBO J. 2016;35:724–742. doi: 10.15252/embj.201592862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Miao J., Liu J., Niu J., Zhang Y., Shen W., Luo C., et al. Wnt/β-catenin/RAS signaling mediates age-related renal fibrosis and is associated with mitochondrial dysfunction. Aging Cell. 2019;18:e13004. doi: 10.1111/acel.13004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Burtscher J., Soltany A., Visavadiya N.P., Burtscher M., Millet G.P., Khoramipour K., et al. Mitochondrial stress and mitokines in aging. Aging Cell. 2023;22:e13770. doi: 10.1111/acel.13770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jiang N., Xing B., Peng R., Shang J., Wu B., Xiao P., et al. Inhibition of Cpt1a alleviates oxidative stress-induced chondrocyte senescence via regulating mitochondrial dysfunction and activating mitophagy. Mech Ageing Dev. 2022;205 doi: 10.1016/j.mad.2022.111688. [DOI] [PubMed] [Google Scholar]
- 47.Sikora E., Bielak-Zmijewska A., Mosieniak G. A common signature of cellular senescence; does it exist? Ageing Res Rev. 2021;71 doi: 10.1016/j.arr.2021.101458. [DOI] [PubMed] [Google Scholar]
- 48.Matias I., Diniz L.P., Damico I.V., Araujo A.P.B., Neves L.D.S., Vargas G., et al. Loss of lamin- B1 and defective nuclear morphology are hallmarks of astrocyte senescence in vitro and in the aging human hippocampus. Aging Cell. 2022;21:e13521. doi: 10.1111/acel.13521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Diao Z., Ji Q., Wu Z., Zhang W., Cai Y., Wang Z., et al. SIRT3 consolidates heterochromatin and counteracts senescence. Nucleic Acids Res. 2021;49:4203–4219. doi: 10.1093/nar/gkab161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kida Y., Goligorsky M.S. Sirtuins, cell senescence, and vascular aging. Can J Cardiol. 2016;32:634–641. doi: 10.1016/j.cjca.2015.11.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Valle F., Reddy P., Yamamoto M., Liu P., Saera-Vila A., Bensaddek D., et al. LINE-1 RNA causes heterochromatin erosion and is a target for amelioration of senescent phenotypes in progeroid syndromes. Sci Transl Med. 2022;14:eabl6057. doi: 10.1126/scitranslmed.abl6057. [DOI] [PubMed] [Google Scholar]
- 52.Gutiérrez-Fernández A., Soria-Valles C., Osorio F.G., Gutiérrez-Abril J., Garabaya C., Aguirre A., et al. Loss of MT1-MMP causes cell senescence and nuclear defects which can be reversed by retinoic acid. EMBO J. 2015;34:1875–1888. doi: 10.15252/embj.201490594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Mitchell T.R., Zhu X.D. Methylated TRF2 associates with the nuclear matrix and serves as a potential biomarker for cellular senescence. Aging. 2014;6:248–263. doi: 10.18632/aging.100650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Copenhaver G.P., Haarer E.L., Theodore C.J., Guo S., Frier R.B., Campellone K.G. Genomic instability caused by Arp2/3 complex inactivation results in micronucleus biogenesis and cellular senescence. Plos Genet. 2023;19:e1010045. doi: 10.1371/journal.pgen.1010045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Partridge L., Deelen J., Slagboom P.E. Facing up to the global challenge of ageing. Nature. 2018;561:1–35. doi: 10.1038/s41586-018-0457-8. [DOI] [PubMed] [Google Scholar]
- 56.Johnson A.A., English B.W., Shokhirev M.N., Sinclair D.A., Cuellar T.L. Human age reversal: fact or fiction? Aging Cell. 2022;21(8):e13664. doi: 10.1111/acel.13664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Khemka S., Reddy A., Garcia R.I., Jacobs M., Reddy R.P., Roghani A.K., et al. Role of diet and exercise in aging, Alzheimer's disease, and other chronic diseases. Ageing Res Rev. 2023;91 doi: 10.1016/j.arr.2023.102091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kuo C.Y., Lin C.H., Lane H.Y. Molecular basis of late-life depression. Int J Mol Sci. 2021;22(14):7421. doi: 10.3390/ijms22147421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sherazi S.A.M., Abbasi A., Jamil A., Uzair M., Ikram A., et al. Molecular hallmarks of long non-coding RNAs in aging and its significant effect on aging-associated diseases. Neural Regen Res. 2023;18:959–968. doi: 10.4103/1673-5374.355751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yang C., Zhang W., Dong X., Fu C., Yuan J., Xu M., et al. A natural product solution to aging and aging-associated diseases. Pharmacol Therapeut. 2020;216 doi: 10.1016/j.pharmthera.2020.107673. [DOI] [PubMed] [Google Scholar]
- 61.Chen C., Zhou M., Ge Y., Wang X. SIRT1 and aging related signaling pathways. Mech Ageing Dev. 2020;187 doi: 10.1016/j.mad.2020.111215. [DOI] [PubMed] [Google Scholar]
- 62.Liu G.Y., Sabatini D.M. mTOR at the nexus of nutrition, growth, ageing and disease. Nat Rev Mol Cell Bio. 2020;21:183–203. doi: 10.1038/s41580-019-0199-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ge Y., Zhou M., Chen C., Wu X., Wang X. Role of AMPK mediated pathways in autophagy and aging. Biochimie. 2022;195:100–113. doi: 10.1016/j.biochi.2021.11.008. [DOI] [PubMed] [Google Scholar]
- 64.Wagner K.H., Cameron-Smith D., Wessner B., Franzke B. Biomarkers of aging: from function to molecular biology. Nutrients. 2016;8:338. doi: 10.3390/nu8060338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Moskalev A., Guvatova Z., Lopes I.D.A., Beckett C.W., Kennedy B.K., De Magalhaes J.P., et al. Targeting aging mechanisms: pharmacological perspectives. Trends Endocrin Met. 2022;33:266–280. doi: 10.1016/j.tem.2022.01.007. [DOI] [PubMed] [Google Scholar]
- 66.Mohamad Kamal N.S., Safuan S., Shamsuddin S., Foroozandeh P. Aging of the cells: Insight into cellular senescence and detection Methods. Eur J Cell Biol. 2020;99 doi: 10.1016/j.ejcb.2020.151108. [DOI] [PubMed] [Google Scholar]
- 67.Saez-Atienzar S., Masliah E. Cellular senescence and Alzheimer disease: the egg and the chicken scenario. Nat Rev Neurosci. 2020;21:433–444. doi: 10.1038/s41583-020-0325-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Behfar Q., Ramirez Zuniga A., Martino-Adami P.V. Aging, senescence, and dementia. J Prev Alzheimers Dis. 2022;9:523–531. doi: 10.14283/jpad.2022.42. [DOI] [PubMed] [Google Scholar]
- 69.Guerrero A., De Strooper B., Arancibia-Cárcamo I.L. Cellular senescence at the crossroads of inflammation and Alzheimer's disease. Trends Neurosci. 2021;44:714–727. doi: 10.1016/j.tins.2021.06.007. [DOI] [PubMed] [Google Scholar]
- 70.Kivisäkk P., Magdamo C., Trombetta B.A., Noori A., Kuo Y.E., Chibnik L.B., et al. Plasma biomarkers for prognosis of cognitive decline in patients with mild cognitive impairment. Brain Commun. 2022;4:fcac155. doi: 10.1093/braincomms/fcac155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lecca D., Jung Y.J., Scerba M.T., Hwang I., Kim Y.K., Kim S., et al. Role of chronic neuroinflammation in neuroplasticity and cognitive function: a hypothesis. Alzheimers Dement. 2022;18:2327–2340. doi: 10.1002/alz.12610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Sweeney M.D., Zhao Z., Montagne A., Nelson A.R., Zlokovic B.V. Blood-Brain barrier: from physiology to disease and back. Physiol Rev. 2019;99:21–78. doi: 10.1152/physrev.00050.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Sayed F.A., Kodama L., Fan L., Carling G.K., Udeochu J.C., Le D., et al. AD-linked R47H-TREM2 mutation induces disease-enhancing microglial states via AKT hyperactivation. Sci Transl Med. 2021;13:eabe3947. doi: 10.1126/scitranslmed.abe3947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Chen S., Acosta D., Li L., Liang J., Chang Y., et al. Wolframin is a novel regulator of tau pathology and neurodegeneration. Acta Neuropathol. 2022;143:547–569. doi: 10.1007/s00401-022-02417-4. [DOI] [PubMed] [Google Scholar]
- 75.Moonen S., Koper M.J., Schoor E., Schaeverbeke J.M., Vandenberghe R., Arnim C.A.F., et al. Pyroptosis in Alzheimer’s disease: cell type-specific activation in microglia, astrocytes and neurons. Acta Neuropathol. 2022;145:175–195. doi: 10.1007/s00401-022-02528-y. [DOI] [PubMed] [Google Scholar]
- 76.Tao W., Yu L., Shu S., Liu Y., Zhuang Z., Xu S., et al. miR-204-3p/Nox4 mediates memory deficits in a mouse model of Alzheimer’s disease. Mol Ther. 2021;29:396–408. doi: 10.1016/j.ymthe.2020.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Gao F., Hu M., Zhang J., Hashem J., Chen C. TDP-43 drives synaptic and cognitive deterioration following traumatic brain injury. Acta Neuropathol. 2022;144:187–210. doi: 10.1007/s00401-022-02449-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Houtman J., Freitag K., Gimber N., Schmoranzer J., Heppner F.L., Jendrach M. Beclin1-driven autophagy modulates the inflammatory response of microglia via NLRP3. EMBO J. 2019;38:e99430. doi: 10.15252/embj.201899430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ho C.Y., Dreesen O. Faces of cellular senescence in skin aging. Mech Ageing Dev. 2021;198 doi: 10.1016/j.mad.2021.111525. [DOI] [PubMed] [Google Scholar]
- 80.Csekes E, RačkováL. Skin Aging, Cellular Senescence and Natural Polyphenols, Int J Mol Sci 2021; 22: 12641. [DOI] [PMC free article] [PubMed]
- 81.Franco A.C., Aveleira C., Cavadas C. Skin senescence: mechanisms and impact on whole-body aging. Trends Mol Med. 2022;28:97–109. doi: 10.1016/j.molmed.2021.12.003. [DOI] [PubMed] [Google Scholar]
- 82.McCart E.A., Thangapazham R.L., Lombardini E.D., Mog S.R., Panganiban R.A.M., Dickson K.M., et al. Accelerated senescence in skin in a murine model of radiation-induced multi-organ injury. J Radiat Res. 2017;58:636–646. doi: 10.1093/jrr/rrx008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kim S.H., Kim J.H., Suk J.M., Lee Y.I., Kim J., Lee J.H., et al. Identification of skin aging biomarkers correlated with the biomechanical properties. Skin Res Technol. 2021;27:940–947. doi: 10.1111/srt.13046. [DOI] [PubMed] [Google Scholar]
- 84.Cardoso A.L., Fernandes A., Aguilar-Pimentel J.A., de Angelis M.H., Guedes J.R., et al. Towards frailty biomarkers: Candidates from genes and pathways regulated in aging and age-related diseases. Ageing Res Rev. 2018;47:214–277. doi: 10.1016/j.arr.2018.07.004. [DOI] [PubMed] [Google Scholar]
- 85.Negre-Salvayre A., Salvayre R. Post-translational modifications evoked by reactive carbonyl species in ultraviolet-A-exposed skin: implication in fibroblast senescence and skin photoaging. Antioxidants (Basel) 2022;11(11):2281. doi: 10.3390/antiox11112281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Tasakova O.S., Golubtzova N.N., Gunin A.G. Thioredoxin-reductase in fibroblasts of human dermis in the process of aging. Adv Gerontol. 2023;36(4):584–591. [PubMed] [Google Scholar]
- 87.Ahn G.R., Jang Y.N., Lee S.Y., Kim W.J., Han H.S., Yoo K.H., et al. Full-thickness skin rejuvenation by a novel dual-length microneedle radiofrequency device: a proof-of-concept study using human skin. Lasers Surg Med. 2023;55(8):758–768. doi: 10.1002/lsm.23707. [DOI] [PubMed] [Google Scholar]
- 88.Windler C., Gey C., Seeger K. Skin melanocytes and fibroblasts show different changes in choline metabolism during cellular senescence. Mech Ageing Dev. 2017;164:82–90. doi: 10.1016/j.mad.2017.05.001. [DOI] [PubMed] [Google Scholar]
- 89.Pereira B.I., Devine O.P., Vukmanovic-Stejic M., Chambers E.S., Subramanian P., Pate N., et al. Senescent cells evade immune clearance via HLA-E-mediated NK and CD8+ T cell inhibition. Nat Commun. 2019;10:2387. doi: 10.1038/s41467-019-10335-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Xie H.F., Liu Y.Z., Du R., Wang B., Chen M.T., Zhang Y.Y., et al. miR-377 induces senescence in human skin fibroblasts by targeting DNA methyltransferase 1. Cell Death Dis. 2017;8:e2663–e. doi: 10.1038/cddis.2017.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Mancini M., Lena A.M., Saintigny G., MahéC D.N., Melino G., Candi E. MicroRNAs in human skin ageing. Ageing Res Rev. 2014;17:9–15. doi: 10.1016/j.arr.2014.04.003. [DOI] [PubMed] [Google Scholar]
- 92.Wyles S.P., Dashti P., Pirtskhalava T., Tekin B., Inman C., Gomez L.S., et al. Chronic wound model to investigate skin cellular senescence. Aging. 2023;15:2852–2862. doi: 10.18632/aging.204667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Blair M.J., Jones J.D., Woessner A.E., Quinn K.P. Skin structure-function relationships and the wound healing response to intrinsic aging. Adv Wound Care. 2020;9:127–143. doi: 10.1089/wound.2019.1021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Demaria M., Ohtani N., Youssef S.A., Rodier F., Toussaint W., Mitchell J.R., et al. An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev Cell. 2014;31:722–733. doi: 10.1016/j.devcel.2014.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ridiandries A., Tan J., Bursill C. The role of chemokines in wound healing. Int J Mol Sci. 2018;19:3217. doi: 10.3390/ijms19103217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Thanapaul R.J.R., Shvedova M., Shin G.H., Crouch J., Roh D.S. Elevated skin senescence in young mice causes delayed wound healing. GeroScience. 2022;44:1871–1878. doi: 10.1007/s11357-022-00551-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Xia W., Li M., Jiang X., Huang X., Gu S., Ye J., et al. Young fibroblast-derived exosomal microRNA-125b transfers beneficial effects on aged cutaneous wound healing. J Nanobiotechnol. 2022;20:144. doi: 10.1186/s12951-022-01348-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Süntar I., Çetinkaya S., Panieri E., Saha S., Buttari B., Profumo E., et al. Regulatory role of Nrf2 signaling pathway in wound healing process. Molecules. 2021;26:2424. doi: 10.3390/molecules26092424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Kaltalioglu K., Coskun-Cevher S. A bioactive molecule in a complex wound healing process: platelet-derived growth factor. Int J Dermatol. 2015;54:972–977. doi: 10.1111/ijd.12731. [DOI] [PubMed] [Google Scholar]
- 100.Hu X.B., Zhu S., Liu R., Miller J.D., Merkley K., Tilton R.G., et al. Sirt6 deficiency impairs corneal epithelial wound healing. Aging. 2018;10:1932–1946. doi: 10.18632/aging.101513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Yamaguchi R., Guo X., Zheng J., Zhang J., Han J., Shioya A., et al. PRDX4 improved aging-related delayed wound healing in mice. J Invest Dermatol. 2021;141:2720–2729. doi: 10.1016/j.jid.2021.04.015. [DOI] [PubMed] [Google Scholar]
- 102.López-Otín C., Pietrocola F., Roiz-Valle D., Galluzzi L., Kroemer G. Meta-hallmarks of aging and cancer. Cell Metab. 2023;35:12–35. doi: 10.1016/j.cmet.2022.11.001. [DOI] [PubMed] [Google Scholar]
- 103.Chatsirisupachai K., Palmer D., Ferreira S., Magalhães J.P. A human tissue-specific transcriptomic analysis reveals a complex relationship between aging, cancer, and cellular senescence. Aging Cell. 2019;18:e13041. doi: 10.1111/acel.13041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Beck J., Turnquist C., Horikawa I., Harris C. Targeting cellular senescence in cancer and aging: roles of p53 and its isoforms. Carcinogenesis. 2020;41:1017–1029. doi: 10.1093/carcin/bgaa071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Olivieri F., Rippo M.R., Monsurrò V., Salvioli S., Capri M., Procopio A.D., et al. MicroRNAs linking inflamm-aging, cellular senescence and cancer. Ageing Res Rev. 2013;12:1056–1068. doi: 10.1016/j.arr.2013.05.001. [DOI] [PubMed] [Google Scholar]
- 106.Ji C., Zhang M., Hu J., Cao C., Gu Q., Liu Y., et al. The kinase activity of integrin-linked kinase regulates cellular senescence in gastric cancer. Cell Death Dis. 2022;13:577. doi: 10.1038/s41419-022-05020-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Calcinotto A., Kohli J., Zagato E., Pellegrini L., Demaria M., Alimonti A. Cellular senescence: aging, cancer, and injury. Physiol Rev. 2019;99:1047–1078. doi: 10.1152/physrev.00020.2018. [DOI] [PubMed] [Google Scholar]
- 108.Wang L., Lankhorst L., Bernards R. Exploiting senescence for the treatment of cancer. Nat Rev Can. 2022;22:340–355. doi: 10.1038/s41568-022-00450-9. [DOI] [PubMed] [Google Scholar]
- 109.Alimirah F., Pulido T., Valdovinos A., Alptekin S., Chang E., Jones E., et al. Cellular senescence promotes skin carcinogenesis through p38MAPK and p44/42MAPK signaling. Can Res. 2020;80:3606–3619. doi: 10.1158/0008-5472.CAN-20-0108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Ambrosio M., Gil J. Reshaping of the tumor microenvironment by cellular senescence: an opportunity for senotherapies. Dev Cell. 2023;58:1007–1021. doi: 10.1016/j.devcel.2023.05.010. [DOI] [PubMed] [Google Scholar]
- 111.Lian J., Yue Y., Yu W., Zhang Y. Immunosenescence: a key player in cancer development. J Hematol Oncol. 2020;13:151. doi: 10.1186/s13045-020-00986-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Evangelou K., Vasileiou P.V.S., Papaspyropoulos A., Hazapis O., Petty R., Demaria M., et al. Cellular senescence and cardiovascular diseases: moving to the “heart” of the problem. Physiol Rev. 2023;103:609–647. doi: 10.1152/physrev.00007.2022. [DOI] [PubMed] [Google Scholar]
- 113.Mehdizadeh M., Aguilar M., Thorin E., Ferbeyre G., Nattel S. The role of cellular senescence in cardiac disease: basic biology and clinical relevance. Nat Rev Cardiol. 2021;19:250–264. doi: 10.1038/s41569-021-00624-2. [DOI] [PubMed] [Google Scholar]
- 114.Vellasamy D.M., Lee S.J., Goh K.W., Goh B.H., Tang Y.Q., Ming L.C., et al. Targeting immune senescence in atherosclerosis. Int J Mol Sci. 2022;23:13059. doi: 10.3390/ijms232113059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Papaconstantinou J. The role of signaling pathways of inflammation and oxidative stress in development of senescence and aging phenotypes in cardiovascular disease. Cells. 2019;8:1383. doi: 10.3390/cells8111383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Kane A.E., Sinclair D.A. Sirtuins and NAD+ in the Development and Treatment of Metabolic and Cardiovascular Diseases. Circ Res. 2018;123:868–885. doi: 10.1161/CIRCRESAHA.118.312498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Huang P.P., Bai L.J., Liu L.H., Fu J., Wu K.F., Liu H.X., et al. Redd1 knockdown prevents doxorubicin-induced cardiac senescence. Aging. 2021;13:13788–13806. doi: 10.18632/aging.202972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Lauro D., Pastore D., Capuani B., Pacifici F., Palmirotta R., Abete P., et al. Role of serum and glucocorticoid inducible kinase (SGK) 1 in senescence a novel molecular target against age related diseases. Curr Med Chem. 2015;22:3765–3788. doi: 10.2174/0929867322666150812145035. [DOI] [PubMed] [Google Scholar]
- 119.Rupérez C., Ferrer-Curriu G., Cervera-Barea A., Florit L., Guitart-Mampel M., Garrabou G., et al. Meteorin-like/meteorin-β protects heart against cardiac dysfunction. J Exp Med. 2021;218:e20201206. doi: 10.1084/jem.20201206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Czibik G., Mezdari Z., Altintas D.M., Bréhat J., Pini M., et al. Dysregulated phenylalanine catabolism plays a key role in the trajectory of cardiac aging. Circulation. 2021;144:559–574. doi: 10.1161/CIRCULATIONAHA.121.054204. [DOI] [PubMed] [Google Scholar]
- 121.Zannas A.S., Jia M., Hafner K., Baumert J., Wiechmann T., Pape J.C., et al. Epigenetic upregulation of FKBP5 by aging and stress contributes to NF-κB–driven inflammation and cardiovascular risk. P Natl Acad Sci Usa. 2019;116:11370–11379. doi: 10.1073/pnas.1816847116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Narasimhan A., Flores R.R., Robbins P.D., Niedernhofer L.J. Role of Cellular senescence in type II diabetes. Endocrinology. 2021;162:bqab136. doi: 10.1210/endocr/bqab136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Xiao X., Xu M., Yu H., Wang L., Li X., Rak J., et al. Mesenchymal stem cell-derived small extracellular vesicles mitigate oxidative stress-induced senescence in endothelial cells via regulation of miR-146a/Src. Signal Transduct Tar. 2021;6:354. doi: 10.1038/s41392-021-00765-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Marino F., Scalise M., Salerno N., Salerno L., Molinaro C., Cappetta D., et al. Diabetes-induced cellular senescence and senescence-associated secretory phenotype impair cardiac regeneration and function independently of age. Diabetes. 2022;71:1081–1098. doi: 10.2337/db21-0536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Wan Y., Liu Z., Wu A., Khan A.H., Zhu Y., Ding S., et al. Hyperglycemia promotes endothelial cell senescence through AQR/PLAU signaling axis. Int J Mol Sci. 2022;23:2879. doi: 10.3390/ijms23052879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Jia C., Chen K.H., Fei X., Dai H.Z., Yang J., Wang L.M., et al. Decoy receptor 2 mediation of the senescent phenotype of tubular cells by interacting with peroxiredoxin 1 presents a novel mechanism of renal fibrosis in diabetic nephropathy. Kidney Int. 2020;98:645–662. doi: 10.1016/j.kint.2020.03.026. [DOI] [PubMed] [Google Scholar]
- 127.Kim M.N., Moon J.H., Cho Y.M. Sodium-glucose cotransporter-2 inhibition reduces cellular senescence in the diabetic kidney by promoting ketone body-induced NRF2 activation. Diabetes Obes Metab. 2021;23:2561–2571. doi: 10.1111/dom.14503. [DOI] [PubMed] [Google Scholar]
- 128.Zhang P., Zhang H.G., Lin J.L., Xiao T., Xu R.Y., et al. Insulin impedes osteogenesis of BMSCs by inhibiting autophagy and promoting premature senescence via the TGF-β1 pathway. Aging. 2020;12:2084–2100. doi: 10.18632/aging.102723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.He Y., Su Y., Duan C., Wang S., He W., Zhang Y., et al. Emerging role of aging in the progression of NAFLD to HCC. Ageing Res Rev. 2023;84 doi: 10.1016/j.arr.2022.101833. [DOI] [PubMed] [Google Scholar]
- 130.Papatheodoridi A.M., Chrysavgis L., Koutsilieris M., Chatzigeorgiou A. The role of senescence in the development of nonalcoholic fatty liver disease and progression to nonalcoholic steatohepatitis. Hepatology. 2019;71:363–374. doi: 10.1002/hep.30834. [DOI] [PubMed] [Google Scholar]
- 131.Zhang C.Y., Tan X.H., Yang H.H., Jin L., Hong J.R., Zhou Y., et al. COX-2/sEH dual inhibitor alleviates hepatocyte senescence in NAFLD mice by restoring autophagy through Sirt1/PI3K/AKT/mTOR. Int J Mol Sci. 2022;23:8267. doi: 10.3390/ijms23158267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Gao Y., Zhang W., Zeng L.Q., Bai H., Li J., Zhou J., et al. Exercise and dietary intervention ameliorate high-fat diet-induced NAFLD and liver aging by inducing lipophagy. Redox Biol. 2020;36 doi: 10.1016/j.redox.2020.101635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Song L., Wang L., Hou Y., Zhou J., Chen C., Ye X., et al. FGF4 protects the liver from nonalcoholic fatty liver disease by activating the AMP-activated protein kinase–Caspase 6 signal axis. Hepatology. 2022;76:1105–1120. doi: 10.1002/hep.32404. [DOI] [PubMed] [Google Scholar]
- 134.Montagner A., Polizzi A., FouchéE D.S., Lippi Y., Lasserre F., et al. Liver PPARα is crucial for whole-body fatty acid homeostasis and is protective against NAFLD. Gut. 2016;65:1202–1214. doi: 10.1136/gutjnl-2015-310798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Baboota R.K., Rawshani A., Bonnet L., Li X., Yang H., Mardinoglu A., et al. BMP4 and Gremlin 1 regulate hepatic cell senescence during clinical progression of NAFLD/NASH. Nat Metab. 2022;4:1007–1021. doi: 10.1038/s42255-022-00620-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Yan L., Zhang T., Wang K., Chen Z., Yang Y., Shan B., et al. SENP1 prevents steatohepatitis by suppressing RIPK1-driven apoptosis and inflammation. Nat Commun. 2022;13:7153. doi: 10.1038/s41467-022-34993-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Kundu D., Kennedy L., Zhou T., Ekser B., Meadows V., Sybenga A., et al. p16 INK4A drives nonalcoholic fatty liver disease phenotypes in high fat diet fed mice through biliary E2F1/FOXO1/IGF-1 signaling. Hepatology. 2023;78:243–257. doi: 10.1097/HEP.0000000000000307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Yang Z., Gao S., Wong C.C., Liu W., Chen H., et al. TUBB4B is a novel therapeutic target in non-alcoholic fatty liver disease-associated hepatocellular carcinoma. J Pathol. 2023;260:71–83. doi: 10.1002/path.6065. [DOI] [PubMed] [Google Scholar]
- 139.Reyes-Farias M., Fos-Domenech J., Serra D., Herrero L., Sánchez-Infantes D. White adipose tissue dysfunction in obesity and aging. Biochem Pharmacol. 2021;192 doi: 10.1016/j.bcp.2021.114723. [DOI] [PubMed] [Google Scholar]
- 140.Palmer A.K., Xu M., Zhu Y., Pirtskhalava T., Weivoda M.M., Hachfeld C.M., et al. Targeting senescent cells alleviates obesity-induced metabolic dysfunction. Aging Cell. 2019;18:e12950. doi: 10.1111/acel.12950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Rouault C., Marcelin G., Adriouch S., Rose C., Genser L., Ambrosini M., et al. Senescence-associated β-galactosidase in subcutaneous adipose tissue associates with altered glycaemic status and truncal fat in severe obesity. Diabetologia. 2020;64:240–254. doi: 10.1007/s00125-020-05307-0. [DOI] [PubMed] [Google Scholar]
- 142.Wang L., Wang B., Gasek N.S., Zhou Y., Cohn R.L., Martin D.E., et al. Targeting p21Cip1 highly expressing cells in adipose tissue alleviates insulin resistance in obesity. Cell Metab. 2022;34:75–89. doi: 10.1016/j.cmet.2021.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Ogrodnik M., Zhu Y., Langhi L.G.P., Tchkonia T., Krüger P., Fielder E., et al. Obesity-induced cellular senescence drives anxiety and impairs neurogenesis. Cell Metab. 2019;29:1061–1077. doi: 10.1016/j.cmet.2018.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Cheng X., Shihabudeen M.S., Moran M., Viana M.P., Schlichte S.L., et al. Long non-coding RNA Meg3 deficiency impairs glucose homeostasis and insulin signaling by inducing cellular senescence of hepatic endothelium in obesity. Redox Biol. 2021;40 doi: 10.1016/j.redox.2021.101863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Tencerova M., Frost M., Figeac F., Nielsen T.K., Ali D., Lauterlein J.J.L., et al. Obesity-associated hypermetabolism and accelerated senescence of bone marrow stromal stem cells suggest a potential mechanism for bone fragility. Cell Rep. 2019;27:2050–2062. doi: 10.1016/j.celrep.2019.04.066. [DOI] [PubMed] [Google Scholar]
- 146.Weinberg J., Gaur M., Swaroop A., Taylor A. Proteostasis in aging-associated ocular disease. Mol Aspects Med. 2022;88 doi: 10.1016/j.mam.2022.101157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Terao R., Ahmed T., Suzumura A., Terasaki H. Oxidative stress-induced cellular senescence in aging retina and age-related macular degeneration. Antioxidants. 2022;11:2189. doi: 10.3390/antiox11112189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Engelbrecht C., Sardinha L.R., Rizzo L.V. Cytokine and chemokine concentration in the tear of patients with age-related cataract. Curr Eye Res. 2020;45:1101–1106. doi: 10.1080/02713683.2020.1715445. [DOI] [PubMed] [Google Scholar]
- 149.Nury T., Yammine A., Ghzaiel I., Sassi K., Brahmi F., et al. Attenuation of 7-ketocholesterol- and 7β-hydroxycholesterol-induced oxiapoptophagy by nutrients, synthetic molecules and oils: Potential for the prevention of age-related diseases. Ageing Res Rev. 2021;68 doi: 10.1016/j.arr.2021.101324. [DOI] [PubMed] [Google Scholar]
- 150.Dabravolski S.A. Mitochondria-derived peptides in healthy ageing and therapy of age-related diseases. Adv Protein Chem Str. 2023;136:197–215. doi: 10.1016/bs.apcsb.2023.02.015. [DOI] [PubMed] [Google Scholar]
- 151.Nie Q., Wang L., Gong X., Xiang J.W., Xiao Y., Xie J., et al. Altered expression patterns of the sumoylation enzymes E1, E2 and E3 are associated with glucose oxidase- and UVA-induced cataractogenesis. Curr Mol Med. 2019;18:542–549. doi: 10.2174/1566524019666190111152324. [DOI] [PubMed] [Google Scholar]
- 152.Chen D.D., Peng X.Y., Wang Y.X., Jiang M.J., Xue M.J., et al. HSP90 acts as a senomorphic target in senescent retinal pigmental epithelial cells. Aging. 2021;13:21547–21570. doi: 10.18632/aging.203496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Sun Y., Zhao J., Song X., Sun Z., Zhang R., Zhong J., et al. Effects of marginal zinc deficiency on learning and memory ability after birth. Food Funct. 2022;13(13):7204–7214. doi: 10.1039/d2fo01074g. [DOI] [PubMed] [Google Scholar]
- 154.Pires I.S., Berthiaume F., Palmer A.F. Engineering Therapeutics to detoxify hemoglobin, heme, and iron. Annu Rev Biomed Eng. 2023;25:1–21. doi: 10.1146/annurev-bioeng-081622-031203. [DOI] [PubMed] [Google Scholar]
- 155.Hong Q., Yang H., Fang Y., Li W., Zhu C., Wang Z., et al. Adaptable graphitic C6N6-based copper single-atom catalyst for intelligent biosensing. Nat Commun. 2023;14(1):2780. doi: 10.1038/s41467-023-38459-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Yadav S, Maurya PK. Biomedical applications of metal oxide nanoparticles in aging and age-associated diseases. 3 Biotech 2021; 11: 338. [DOI] [PMC free article] [PubMed]
- 157.Chang Y., Cho B., Lee E., Kim J., Yoo J., Sung J.S., et al. Electromagnetized gold nanoparticles improve neurogenesis and cognition in the aged brain. Biomaterials. 2021;278 doi: 10.1016/j.biomaterials.2021.121157. [DOI] [PubMed] [Google Scholar]
- 158.Hossen M.N., Wang L., Dwivedi S.K.D., Zhang Y., Rao G., Elechalwar C.K., et al. Gold nanoparticles disrupt the IGFBP2/mTOR/PTEN axis to inhibit ovarian cancer growth. Adv Sci. 2022;9:e2200491. doi: 10.1002/advs.202200491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Choi Y.J., Kim J.S., Kim W.S. Photothermal therapy using gold nanoparticles and a long-pulsed 755-nm alexandrite laser to treat facial photoaging in Asian skin: a prospective clinical trial. Laser Surg Med. 2022;54:1060–1070. doi: 10.1002/lsm.23581. [DOI] [PubMed] [Google Scholar]
- 160.Zheng Y., Wu Y., Liu Y., Guo Z., Bai T., Zhou P., et al. Intrinsic effects of gold nanoparticles on oxygen-glucose deprivation/reperfusion injury in rat cortical neurons. Neurochem Res. 2019;44:1549–1566. doi: 10.1007/s11064-019-02776-7. [DOI] [PubMed] [Google Scholar]
- 161.Huang T.H., Liu C., Wang J.J., Pan S.N., Han Y., Pickard C.J., et al. Metallic aluminum suboxides with ultrahigh electrical conductivity at high pressure. Research. 2022;2022:9798758. doi: 10.34133/2022/9798758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Selmani A., Jeitler R., Auinger M., Tetyczka C., Banzer P., Kantor B., et al. Investigation of the influence of wound-treatment-relevant buffer systems on the colloidal and optical properties of gold nanoparticles. Nanomaterials. 2023;13:1878. doi: 10.3390/nano13121878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Tian S.J., Peng H., Liu H.Z., Zhou J.C., Zhang J.Y. Scalable fabrication of metallic conductive fibers from rheological tunable semi-liquid metals. Research. 2022;2022:9890686. doi: 10.34133/2022/9890686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Jia J., Li F., Zhou H., Bai Y., Liu S., Jiang Y., et al. Oral exposure to silver nanoparticles or silver ions may aggravate fatty liver disease in overweight mice. Environ Sci Technol. 2017;51:9334–9343. doi: 10.1021/acs.est.7b02752. [DOI] [PubMed] [Google Scholar]
- 165.Arozal W., Monayo E.R., Barinda A.J., Perkasa D.P., Soetikno V., Nafrialdi N., et al. Protective effects of silver nanoparticles in isoproterenol-induced myocardial infarction in rats. Front Med. 2022;9 doi: 10.3389/fmed.2022.867497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Mao B.H., Chen Z.Y., Wang Y.J., Yan S.J. Silver nanoparticles have lethal and sublethal adverse effects on development and longevity by inducing ROS-mediated stress responses. Sci Rep. 2018;8 doi: 10.1038/s41598-018-20728-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Tian H., Eom H.J., Moon S., Lee J., Choi J., Chung Y.D. Development of biomarker for detecting silver nanoparticles exposure using a GAL4 enhancer trap screening in Drosophila. Environ Toxicol Phar. 2013;36:548–556. doi: 10.1016/j.etap.2013.05.013. [DOI] [PubMed] [Google Scholar]
- 168.Kolodziejczyk A.M., Kucinska M., Jakubowska A., Sokolowska P., Rosowski M., Tkacz-Szczesna B., et al. Endothelial cell aging detection by means of atomic force spectroscopy. J Mol Recognit. 2020;33:e2853. doi: 10.1002/jmr.2853. [DOI] [PubMed] [Google Scholar]
- 169.Chen B., Chai Q., Xu S., Li Q., Wu T., Chen S., et al. Silver nanoparticle-activated COX2/PGE2 axis involves alteration of lung cellular senescence in vitro and in vivo. Ecotox Environ Safe. 2020;204 doi: 10.1016/j.ecoenv.2020.111070. [DOI] [PubMed] [Google Scholar]
- 170.Venugopal K., Ahmad H., Manikandan E., Thanigai Arul K., Kavitha K., Moodley M.K., et al. The impact of anticancer activity upon Beta vulgaris extract mediated biosynthesized silver nanoparticles (ag-NPs) against human breast (MCF-7), lung (A549) and pharynx (Hep-2) cancer cell lines. J Photoch Photobio B. 2017;173:99–107. doi: 10.1016/j.jphotobiol.2017.05.031. [DOI] [PubMed] [Google Scholar]
- 171.Alharbi N.S., Khaled J.M., Alanazi K., Kadaikunnan S., Alobaidi A.S. Biosynthesis of silver nanoparticles (Ag-NPs) using Senna alexandrina grown in Saudi Arabia and their bioactivity against multidrug-resistant pathogens and cancer cells. Saudi Pharm J. 2023;31:911–920. doi: 10.1016/j.jsps.2023.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Deylam M., Alizadeh E., Sarikhani M., Hejazy M., Firouzamandi M. Zinc oxide nanoparticles promote the aging process in a size-dependent manner. J Mater Sci-Mater M. 2021;32:128. doi: 10.1007/s10856-021-06602-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Rehman H., Ali W., Zaman Khan N., Aasim M., Khan T., Ali K.A. Delphinium uncinatum mediated biosynthesis of zinc oxide nanoparticles and in-vitro evaluation of their antioxidant, cytotoxic, antimicrobial, anti-diabetic, anti-inflammatory, and anti-aging activities. Saudi J Biol Sci. 2023;30 doi: 10.1016/j.sjbs.2022.103485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Anand A.S., Prasad D.N., Singh S.B., Kohli E. Chronic exposure of zinc oxide nanoparticles causes deviant phenotype in Drosophila melanogaster. J Hazard Mater. 2017;327:180–186. doi: 10.1016/j.jhazmat.2016.12.040. [DOI] [PubMed] [Google Scholar]
- 175.Tian L., Lin B., Wu L., Li K., Liu H., Yan J., et al. Neurotoxicity induced by zinc oxide nanoparticles: age-related differences and interaction. Sci Rep. 2015;5:16117. doi: 10.1038/srep16117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Subramaniam V.D., Ramachandran M., Marotta F., Banerjee A., Sun X.F., Pathak S. Comparative study on anti-proliferative potentials of zinc oxide and aluminium oxide nanoparticles in colon cancer cells. Acta Biomedica. 2019;90:241–247. doi: 10.23750/abm.v90i2.6939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Pal A., Alam S., Mittal S., Arjaria N., Shankar J., Kumar M., et al. UVB irradiation-enhanced zinc oxide nanoparticles-induced DNA damage and cell death in mouse skin. Mutat Res Genet Toxicol Environ Mutagen. 2016;807:15–24. doi: 10.1016/j.mrgentox.2016.06.005. [DOI] [PubMed] [Google Scholar]
- 178.Wei S.S., Guo W.H., Qian Y., Xiang J., Liu K.L., Gao X.J., et al. Ribosome profiling reveals translatome remodeling in cancer cells in response to zinc oxide nanoparticles. Aging. 2021;13:23119–23132. doi: 10.18632/aging.203606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Zhang Y., Wang Z., Li X., Wang L., Yin M., Wang L., et al. Dietary iron oxide nanoparticles delay aging and ameliorate neurodegeneration in drosophila. Adv Mater. 2016;28:1387–1393. doi: 10.1002/adma.201503893. [DOI] [PubMed] [Google Scholar]
- 180.Pan Z., Shi Z., Wei H., Sun F., Song J., Huang Y., et al. Magnetofection Based on superparamagnetic iron oxide nanoparticles weakens glioma stem cell proliferation and invasion by mediating high expression of MicroRNA-374a. J Cancer. 2016;7:1487–1496. doi: 10.7150/jca.15515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Yang Y., Tian Q., Wu S., Li Y., Yang K., Yan Y., et al. Blue light-triggered Fe2+-release from monodispersed ferrihydrite nanoparticles for cancer iron therapy. Biomaterials. 2021;271 doi: 10.1016/j.biomaterials.2021.120739. [DOI] [PubMed] [Google Scholar]
- 182.Sharifi S., Daghighi S., Motazacker M.M., Badlou B., Sanjabi B., Akbarkhanzadeh A., et al. Superparamagnetic iron oxide nanoparticles alter expression of obesity and T2D-associated risk genes in human adipocytes. Sci Rep. 2013;3:2173. doi: 10.1038/srep02173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Zhu M., Chen H., Zhou S., Zheng L., Li X., Chu R., et al. Iron oxide nanoparticles aggravate hepatic steatosis and liver injury in nonalcoholic fatty liver disease through BMP-SMAD-mediated hepatic iron overload. Nanotoxicology. 2021;15:761–778. doi: 10.1080/17435390.2021.1919329. [DOI] [PubMed] [Google Scholar]
- 184.Manickam V., Periyasamy M., Dhakshinamoorthy V., Panneerselvam L., Perumal E. Recurrent exposure to ferric oxide nanoparticles alters myocardial oxidative stress, apoptosis and necrotic markers in male mice. Chem-Biol Interact. 2017;278:54–64. doi: 10.1016/j.cbi.2017.10.003. [DOI] [PubMed] [Google Scholar]
- 185.Chen Z., Wang Y., Zhuo L., Chen S., Zhao L., Luan X., et al. Effect of titanium dioxide nanoparticles on the cardiovascular system after oral administration. Toxicol Lett. 2015;239:123–130. doi: 10.1016/j.toxlet.2015.09.013. [DOI] [PubMed] [Google Scholar]
- 186.Huerta-García E., Zepeda-Quiroz I., Sánchez-Barrera H., Colín-Val Z., Alfaro-Moreno E., Ramos-Godinez M., et al. Internalization of titanium dioxide nanoparticles is cytotoxic for H9c2 rat cardiomyoblasts. Molecules. 2018;23:1955. doi: 10.3390/molecules23081955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Chen L., Nie P.H., Yao L.Y., Tang Y.Z., Hong W.D., Liu W.T., et al. TiO2 NPs induce the reproductive toxicity in mice with gestational diabetes mellitus through the effects on the endoplasmic reticulum stress signaling pathway. Ecotox Environ Safe. 2021;226 doi: 10.1016/j.ecoenv.2021.112814. [DOI] [PubMed] [Google Scholar]
- 188.Ferrante M., Grasso A., Salemi R., Libra M., Tomasello B., Fiore M., et al. DNA damage and apoptosis as in-vitro effect biomarkers of titanium dioxide nanoparticles (TiO2-NPs) and the food additive E171 toxicity in colon cancer cells: HCT-116 and Caco-2. Int J Env Res Pub He. 2023;20:2002. doi: 10.3390/ijerph20032002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Zhao F., Wang C., Yang Q., Han S., Hu Q., Fu Z. Titanium dioxide nanoparticle stimulating pro-inflammatory responses in vitro and in vivo for inhibited cancer metastasis. Life Sci. 2018;202:44–51. doi: 10.1016/j.lfs.2018.03.058. [DOI] [PubMed] [Google Scholar]
- 190.Iqbal H., Razzaq A., Uzair B., Ul Ain N., Sajjad S., Althobaiti N.A., et al. Breast Cancer inhibition by biosynthesized titanium dioxide nanoparticles is comparable to free doxorubicin but appeared safer in BALB/c mice. Materials. 2021;14:3155. doi: 10.3390/ma14123155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Salah M., Akasaka H., Shimizu Y., Morita K., Nishimura Y., Kubota H., et al. Reactive oxygen species-inducing titanium peroxide nanoparticles as promising radiosensitizers for eliminating pancreatic cancer stem cells. J Exp Clin Canc Res. 2022;41:146. doi: 10.1186/s13046-022-02358-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Crosera M., Prodi A., Mauro M., Pelin M., Florio C., Bellomo F., et al. Titanium dioxide nanoparticle penetration into the skin and effects on HaCaT cells. Int J Env Res Pub He. 2015;12:9282–9297. doi: 10.3390/ijerph120809282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Rangel-López E., Robles-Bañuelos B., Guadiana-Ramírez N., Alvarez-Garduño V., Galván-Arzate S., Zazueta C., et al. Thallium induces antiproliferative and cytotoxic activity in glioblastoma C6 and U373 cell cultures via apoptosis and changes in cell cycle. Neurotox Res. 2022;40:814–824. doi: 10.1007/s12640-022-00514-6. [DOI] [PubMed] [Google Scholar]
- 194.Philippaert K., Kerselaers S., Voets T., Vennekens R. A thallium-based screening procedure to identify molecules that modulate the activity of Ca2+-activated monovalent cation-selective channels. Slas Discov. 2018;23:341–352. doi: 10.1177/2472555217748932. [DOI] [PubMed] [Google Scholar]
- 195.Lin J.D., Pei D., Chen F.Y., Wu C.Z., Lu C.H., Huang L.Y., et al. Comparison between machine learning and multiple linear regression to identify abnormal thallium myocardial perfusion scan in Chinese type 2 diabetes. Diagnostics. 2022;12:1619. doi: 10.3390/diagnostics12071619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Hayashi T., Joki N., Tanaka Y., Iwasaki M., Kubo S., Matsukane A., et al. Thallium-201 washout rate of stress myocardial perfusion imaging as a predictor of mortality in diabetic kidney disease patients initiating hemodialysis: an observational, follow-up study. Clin Exp Nephrol. 2017;22:142–150. doi: 10.1007/s10157-017-1414-9. [DOI] [PubMed] [Google Scholar]
- 197.Xie Z., Aimuzi R., Si M., Qu Y., Jiang Y. Associations of metal mixtures with metabolic-associated fatty liver disease and non-alcoholic fatty liver disease: NHANES 2003–2018. Front Public Health. 2023;11:1133194. doi: 10.3389/fpubh.2023.1133194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Garcia-Peiro J.I., Bonet-Aleta J., Santamaria J., Hueso J.L. Platinum nanoplatforms: classic catalysts claiming a prominent role in cancer therapy. Chem Soc Rev. 2022;51:7662–7681. doi: 10.1039/d2cs00518b. [DOI] [PubMed] [Google Scholar]
- 199.Wang X., Liu Z., Wang Y., Gou S. Platinum(IV) Prodrugs with cancer stem cell inhibitory effects on lung cancer for overcoming drug resistance. J Med Chem. 2022;65:7933–7945. doi: 10.1021/acs.jmedchem.2c00472. [DOI] [PubMed] [Google Scholar]
- 200.Jiang Y., Song L., Lin Y., Nowialis P., Gao Q., Li T., et al. ROS-mediated SRMS activation confers platinum resistance in ovarian cancer. Oncogene. 2023;42:1672–1684. doi: 10.1038/s41388-023-02679-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Nyen T.V., Planque M., Wagensveld L., Duarte J.A.G., Zaal E.A., Talebi A., et al. Serine metabolism remodeling after platinum-based chemotherapy identifies vulnerabilities in a subgroup of resistant ovarian cancers. Nat Commun. 2022;13:4578. doi: 10.1038/s41467-022-32272-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Zhao J., Hua W., Xu G., Gou S. Biotinylated platinum(IV) complexes designed to target cancer cells. J Inorg Biochem. 2017;176:175–180. doi: 10.1016/j.jinorgbio.2017.08.017. [DOI] [PubMed] [Google Scholar]
- 203.Yao Z.F., Lin A.Q., Yi Y.L., Shen W.T., Zhang J., Luo P. THSD7B mutation induces platinum resistance in small cell lung cancer patients. Drug Des Dev Ther. 2022;16:1679–1695. doi: 10.2147/DDDT.S363665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Patel M., Wang Y., Bartom E.T., Dhir R., Nephew K.P., et al. The Ratio of toxic-to-nontoxic miRNAs predicts platinum sensitivity in ovarian cancer. Cancer Res. 2021;81:3985–4000. doi: 10.1158/0008-5472.CAN-21-0953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Su L., Gong X., Fan R., Ni T., Yang F., Zhang X., et al. Mechanism of action of platinum nanoparticles implying from antioxidant to metabolic programming in light-induced retinal degeneration model. Redox Biol. 2023;65 doi: 10.1016/j.redox.2023.102836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Hanzha V.V., Rozumna N.M., Kravenska Y.V., Spivak M.Y., Lukyanetz E.A. The effect of cerium dioxide nanoparticles on the viability of hippocampal neurons in Alzheimer’s disease modeling. Front Cell Neurosci. 2023;17:1131168. doi: 10.3389/fncel.2023.1131168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Danish S.M., Gupta A., Khan U.A., Hasan N., Ahmad F.J., Warsi M.H., et al. Intranasal cerium oxide nanoparticles ameliorate cognitive function in rats with Alzheimer’s via anti-oxidative pathway. Pharmaceutics. 2022;14:756. doi: 10.3390/pharmaceutics14040756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Abdel Gaber S.A., Hamza A.H., Tantawy M.A., Toraih E.A., Ahmed H.H. Germanium dioxide nanoparticles mitigate biochemical and molecular changes characterizing Alzheimer’s disease in rats. Pharmaceutics. 2023;15:1386. doi: 10.3390/pharmaceutics15051386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Gao Y., Gao F., Chen K., Ma J.L. Cerium oxide nanoparticles in cancer. Oncotargets Ther. 2014;7:835–840. doi: 10.2147/OTT.S62057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Javid H., Hashemy S.I., Heidari M.F., Esparham A., Gorgani-Firuzjaee S., Siemianowicz K. The anticancer role of cerium oxide nanoparticles by inducing antioxidant activity in esophageal cancer and cancer stem-like ESCC spheres. Biomed Res Int. 2022;2022:1–11. doi: 10.1155/2022/3268197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Wason M., Lu H., Yu L., Lahiri S., Mukherjee D., Shen C., et al. Cerium oxide nanoparticles sensitize pancreatic cancer to radiation therapy through oxidative activation of the JNK apoptotic pathway. Cancers. 2018;10:303. doi: 10.3390/cancers10090303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Pešić M., Podolski-Renić A., Stojković S., Matović B., Zmejkoski D., Kojić V., et al. Anti-cancer effects of cerium oxide nanoparticles and its intracellular redox activity. Chem-Biol Interact. 2015;232:85–93. doi: 10.1016/j.cbi.2015.03.013. [DOI] [PubMed] [Google Scholar]
- 213.El Shaer S.S., Salaheldin T.A., Saied N.M., Abdelazim S.M. In vivo ameliorative effect of cerium oxide nanoparticles in isoproterenol-induced cardiac toxicity. Exp Toxicol Pathol. 2017;69:435–441. doi: 10.1016/j.etp.2017.03.001. [DOI] [PubMed] [Google Scholar]
- 214.Artimani T., Amiri I., Soleimani Asl S., Saidijam M., Hasanvand D., Afshar S. Amelioration of diabetes-induced testicular and sperm damage in rats by cerium oxide nanoparticle treatment. Andrologia. 2018;50:e13089. doi: 10.1111/and.13089. [DOI] [PubMed] [Google Scholar]
- 215.Vafaei-Pour Z., Shokrzadeh M., Jahani M., Shaki F. Embryo-protective effects of cerium oxide nanoparticles against gestational diabetes in mice. Iran J Pharm Res. 2018;17:964–975. [PMC free article] [PubMed] [Google Scholar]
- 216.Mathys Z.K., White A.R. Copper and Alzheimer’s disease. Adv Neurobiol. 2017;18:199–216. doi: 10.1007/978-3-319-60189-2_10. [DOI] [PubMed] [Google Scholar]
- 217.Jaragh-Alhadad L.A., Falahati M. Copper oxide nanoparticles promote amyloid-β-triggered neurotoxicity through formation of oligomeric species as a prelude to Alzheimer's diseases. Int J Biol Macromol. 2022;207:121–129. doi: 10.1016/j.ijbiomac.2022.03.006. [DOI] [PubMed] [Google Scholar]
- 218.Tsymbal S., Li G., Agadzhanian N., Sun Y., Zhang J., Dukhinova M., et al. Recent advances in copper-based organic complexes and nanoparticles for tumor theranostics. Molecules. 2022;27:7066. doi: 10.3390/molecules27207066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Fahmy H.M., Ebrahim N.M., Gaber M.H. In-vitro evaluation of copper/copper oxide nanoparticles cytotoxicity and genotoxicity in normal and cancer lung cell lines. J Trace Elem Med Bio. 2020;60 doi: 10.1016/j.jtemb.2020.126481. [DOI] [PubMed] [Google Scholar]
- 220.Ghasemi P., Shafiee G., Ziamajidi N., Abbasalipourkabir R. Copper nanoparticles induce apoptosis and oxidative stress in SW-480 human colon cancer cell line. Biol Trace Elem Res. 2022;20:3746–3754. doi: 10.1007/s12011-022-03458-2. [DOI] [PubMed] [Google Scholar]
- 221.Al-zharani M., Qurtam A.A., Daoush W.M., Eisa M.H., Aljarba N.H., Alkahtani S., et al. Antitumor effect of copper nanoparticles on human breast and colon malignancies. Environ Sci Pollut R. 2020;28:1587–1595. doi: 10.1007/s11356-020-09843-5. [DOI] [PubMed] [Google Scholar]
- 222.Kitala K., Tanski D., Godlewski J., Krajewska-Włodarczyk M., Gromadziński L., Majewski M. Copper and zinc particles as regulators of cardiovascular system function—a review. Nutrients. 2023;15:3040. doi: 10.3390/nu15133040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Gao W., Sun Y., Cai M., Zhao Y., Cao W., Liu Z., et al. Copper sulfide nanoparticles as a photothermal switch for TRPV1 signaling to attenuate atherosclerosis. Nat Commun. 2018;9:231. doi: 10.1038/s41467-017-02657-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Fan Y., Cheng Z., Mao L., Xu G., Li N., Zhang M., et al. PINK1/TAX1BP1-directed mitophagy attenuates vascular endothelial injury induced by copper oxide nanoparticles. J Nanobiotechnol. 2022;20:149. doi: 10.1186/s12951-022-01338-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.He H., Zou Z., Wang B., Xu G., Chen C., Qin X., et al. Copper oxide nanoparticles induce oxidative DNA damage and cell death via copper ion-mediated P38 MAPK activation in vascular endothelial cells. Int J Nanomed. 2020;15:3291–3302. doi: 10.2147/IJN.S241157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Alam A.M., Shon Y.S. Water-soluble noble metal nanoparticle catalysts capped with small organic molecules for organic transformations in water. ACS Appl Nano Mater. 2021;4(4):3294–3318. doi: 10.1021/acsanm.1c00335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Itterheimová P., Dosedělová V., Kubáň P. Use of metal nanoparticles for preconcentration and analysis of biological thiols. Electrophoresis. 2023;44(1–2):135–157. doi: 10.1002/elps.202200142. [DOI] [PubMed] [Google Scholar]
- 228.Kus-Liśkiewicz M., Fickers P., Ben T.I. Biocompatibility and cytotoxicity of gold nanoparticles: recent advances in methodologies and regulations. Int J Mol Sci. 2021;22(20):10952. doi: 10.3390/ijms222010952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Sharma A.R., Lee Y.H., Bat-Ulzii A., Bhattacharya M., Chakraborty C., Lee S.S. Recent advances of metal-based nanoparticles in nucleic acid delivery for therapeutic applications. J Nanobiotechnology. 2022;20(1):501. doi: 10.1186/s12951-022-01650-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Xiong P., Huang X., Ye N., Lu Q., Zhang G., Peng S., et al. Cytotoxicity of metal-based nanoparticles: from mechanisms and methods of evaluation to pathological manifestations. Adv Sci (Weinh) 2022;9(16):e2106049. doi: 10.1002/advs.202106049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Singh P., Pandit S., Mokkapati V.R.S.S., Garg A., Ravikumar V., Mijakovic I. Gold nanoparticles in diagnostics and therapeutics for human cancer. Int J Mol Sci. 2018;19(7):1979. doi: 10.3390/ijms19071979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Wang C., Chen B., He M., Hu B. Composition of intracellular protein corona around nanoparticles during internalization. ACS Nano. 2021;15(2):3108–3122. doi: 10.1021/acsnano.0c09649. [DOI] [PubMed] [Google Scholar]
- 233.Hajipour M.J., Safavi-Sohi R., Sharifi S., Mahmoud N., Ashkarran A.A., Voke E., et al. An overview of nanoparticle protein corona literature. Small. 2023;19(36):e2301838. doi: 10.1002/smll.202301838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Esmaeili Y., Khavani M., Bigham A., Sanati A., Bidram E., Shariati L., et al. Mesoporous silica@chitosan@gold nanoparticles as “on/off” optical biosensor and pH-sensitive theranostic platform against cancer. Int J Biol Macromol. 2022;202:241–255. doi: 10.1016/j.ijbiomac.2022.01.063. [DOI] [PubMed] [Google Scholar]
- 235.Li B., Chen X., Qiu W., Zhao R., Duan J., Zhang S., et al. Synchronous disintegration of ferroptosis defense axis via engineered exosome-conjugated magnetic nanoparticles for glioblastoma therapy. Adv Sci (Weinh) 2022;9(17):e2105451. doi: 10.1002/advs.202105451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Obisesan O.S., Ajiboye T.O., Mhlanga S.D., Mufhandu H.T. Biomedical applications of biodegradable polycaprolactone-functionalized magnetic iron oxides nanoparticles and their polymer nanocomposites. Colloids Surf B Biointerfaces. 2023;227 doi: 10.1016/j.colsurfb.2023.113342. [DOI] [PubMed] [Google Scholar]
- 237.Quevedo A.C., Lynch I., Valsami-Jones E. Cellular repair mechanisms triggered by exposure to silver nanoparticles and ionic silver in embryonic zebrafish cells. Environ Sci Nano. 2021;8:2507. [Google Scholar]
- 238.Mortezagholi B., Movahed E., Fathi A., Soleimani M., Forutan Mirhosseini A., Zeini N., et al. Plant-mediated synthesis of silver-doped zinc oxide nanoparticles and evaluation of their antimicrobial activity against bacteria cause tooth decay. Microsc Res Tech. 2022;85(11):3553–3564. doi: 10.1002/jemt.24207. [DOI] [PubMed] [Google Scholar]
- 239.Geppert M., Himly M. Iron Oxide Nanoparticles in Bioimaging - An Immune Perspective. Front Immunol. 2021;12 doi: 10.3389/fimmu.2021.688927. [DOI] [PMC free article] [PubMed] [Google Scholar]