Abstract
CrossFit® is popular form of fitness training but the cardiorespiratory, hemodynamic and metabolic responses have not been well characterised. We compared responses to a short Fran CrossFit® session (consisting of three rounds of 21, 15 and 9 front squat to press overhead and pull-ups) between sexes to evaluate differences in acute exercise effects. Twenty-five (15 males and 10 females) trained participants performed a prescribed Fran session at maximal exertion. Cardiorespiratory variables were assessed at baseline, during and post-exercise. The rating of perceived exertion, blood pressure, blood lactate and glucose concentrations were measured at baseline and in the recovery period. The males completed Fran session in less time (males 177 ± 15 vs females 206 ± 27 s; mean ± SD), resulting in higher peak oxygen uptake values (50.1 ± 3.3 vs 46.4 ± 2.2 mL kg −1 min −1), heart rate (187 ± 6 vs 180 ± 3 b min−1) and percentage heart rate (96 ± 3 vs 93 ± 1 %) compared to females. In addition, elevated systolic blood pressure (male 154 ± 8 vs females 149 ± 10 mmHg), double product (27,050 ± 1504 vs 25,999 ± 2253 mmHg b∙min −1), blood lactate (15.7 ± 1.7 vs 14.6 ± 2.0 mmol L−1) and glucose (132 ± 15 vs 132 ± 14 mg dL−1) values were observed in both sexes post-exercise. The substantial metabolic demands associated with a Fran session can yield similar elevations in cardiorespiratory and hemodynamic responses in both sexes, meeting the criteria for developing cardiorespiratory fitness. Complementarily, these results suggest that researchers and coaches can use the Fran session at maximal effort to assess and monitor crossfitters' training status throughout a CrossFit® season, guiding efforts towards excellence in this demanding sport.
Keywords: Pulmonary, Cardiovascular, Hemodinamic, Exercise, Recovery, Physical fitness
Highlights
-
•
Male participants demonstrated higher peak oxygen consumption and heart rate values and completed the Fran session in less time than female participants.
-
•
Although blood lactate, glucose, and blood pressure responses after exercise were similar between sexes, ventilatory and performance responses differed.
-
•
These sex-based physiological differences highlight the need for individualized CrossFit® programming and recovery strategies.
-
•
The Fran workout elicited cardiovascular responses consistent with vigorous-intensity exercise and can be used to monitor training status over time.
Abbreviations
| [La−] | Blood lactate concentration |
| DBP | Diastolic blood pressure |
| HR | Heart rate |
| HR | Maximal heart rate |
| RPE | Rating of perceived exertion |
| SBP | Systolic blood pressure |
| O2 | Oxygen uptake |
1. Introduction
Cardiorespiratory fitness measured as maximal exercise capacity is an established predictive marker of cardiovascular disease and all-cause mortality (Clarke et al., 2017). Improvements in maximal exercise capacity are primarily attributed to central adaptations (stroke volume), whereas peripheral adaptations (oxidative capacity) contribute far less (Clarke et al., 2017; Goodman et al., 1999). Conversely, when discussing improvements in submaximal cardiorespiratory fitness, these alterations are closely associated with peripheral adaptations (Clarke et al., 2017; Goodman et al., 1999). Maximal and/or peak oxygen uptake ( O2) is commonly used to assess cardiorespiratory fitness and functional capacity, providing valuable insights into the maximal workload that individuals can sustain during physical exertion (Lopes et al., 2022; Poole and Jones, 2012). In addition, evaluation of respiratory parameters, blood lactate concentration ([La−]) and blood pressure is essential to chacterise the acute physiological impact (Lopes et al., 2022; Rios et al., 2023; Forte et al., 2022) and consequently control chronic exercise adaptations (Gripp et al., 2021).
High-intensity interval exercise sessions of maximal to supramaximal intensities have a similar or even more substantial impact on cardiorespiratory fitness and metabolic adaptations than traditional continuous moderate-intensity training (Gripp et al., 2021; Gillen and Gibala, 2018). These findings underscore the effectiveness of short-duration, high-intensity exercise in inducing favourable exercise adaptations (Gripp et al., 2021; Tabata et al., 1997). Prescribing high-intensity interval exercise is involves manipulating several variables: duration of effort and recovery phases, intensity of effort and recovery, and total number of efforts/recoveries (MacInnis and Gibala, 2017). Furthermore, most high-intensity interval exercise primarily involves cyclical modes of exercise like running or cycling to elicit cardiovascular adaptations (Gillen and Gibala, 2018; Tabata et al., 1997), but strength adaptations are often limited. In contrast, high-intensity functional exercise (e.g. CrossFit®) places emphasis on functional, multi-joint movements and incorporates both aerobic and strength exercises (Claudino et al., 2018; Rios et al., 2021). CrossFit® sessions are adaptable to diverse fitness levels and sex, enhancing muscle recruitment more effectively than cyclical modes of exercise (Claudino et al., 2018). Similar to other high-intensity functional exercise modes, CrossFit® can improve body composition, muscle strength and cardiorespiratory fitness (McDougle et al., 2023).
CrossFit® workouts can also induce blood oxidative stress and raise markers of muscle damage, including interleukin-6 and creatine kinase (McDougle et al., 2023). The extent of these responses varies depending on factors including training intensity, duration, exercises and rest intervals (Rios et al., 2023; McDougle et al., 2023). Performance in CrossFit® is typically assessed by completion time or repetition count, with reference exercise (like Fran) commonly used for evaluating physical fitness (Rios et al., 2023, 2024a; Tibana et al., 2018). Reference workouts such as Fran are standardized in terms of external loads (e.g. males use a fixed weight of 43 kg and females a 30 kg) and repetitions (e.g. 21-15 and 9) to facilitate consistent comparisons of performance among athletes and across various settings worldwide, irrespective of the location or setting in which they are executed (Rios et al., 2023, 2024b). The Fran session has been studied extensively, revealing elevated levels of [La−], glucose levels, heart rate (HR), and peak O2, coupled with a high rating of perceived exertion (RPE) across its three rounds (Rios et al., 2023, 2024a; Tibana et al., 2018). Currently, there is a scarcity of studies that analyze the comprehensive physiological impact of the Fran routine and magnitudes of difference between sexes.
Physiological factors influence physical fitness differences between males and females given distinct strength and endurance predispositions (Howden et al., 2015; Hottenrott et al., 2021). For example, males typically have a 10 % higher maximal O2 due to their greater heart size, blood volume and hemoglobin than females (Howden et al., 2015; Coe and Astorino, 2023). In addition, sex differences in oxidative capacity and fatigue tolerance are evident and may be explained by several physiological mechanisms. Females generally exhibit a higher proportion of type I (slow-twitch) muscle fibers, which are more oxidative and fatigue-resistant, contributing to greater endurance capabilities. Females tend to rely more on lipid oxidation and less on carbohydrate metabolism during exercise, leading to glycogen sparing and potentially greater fatigue tolerance (Hottenrott et al., 2021; Coe and Astorino, 2023; Jaworowski et al., 2002). However, both groups have demonstrated comparable results in terms of movement economy (Timón et al., 2019). These inherent physiological variations may contribute to sex differences in the adaptive response to high-intensity exercise (Howden et al., 2015; Coe and Astorino, 2023), as the acute exercise response underpins the chronic response.
The acute response to a high-intensity exercise session like a Fran session reveal nuances or variations in this pattern. Current CrossFit® training practices often rely heavily on research conducted primarily on male athletes (Tibana et al., 2018; Rios et al., 2024b; Leitão et al., 2021). For example, one study analyzed [La−], HR and RPE in short and long CrossFit® sessions using only male participants, revealing high cardiovascular and metabolic demands that inform common programming strategies (Tibana et al., 2018). Another study assessed cardiorespiratory and metabolic responses during the Isabel benchmark workout in trained males, demonstrating predominantly anaerobic contributions that guide intensity and recovery prescriptions (Rios et al., 2024b). A third study identified strength, HR recovery and body composition as key predictors of performance in the Fran workout, based on a male-only sample, shaping performance benchmarks still widely applied across sexes (Leitão et al., 2021). Consequently, female athletes may be expected to respond similarly to male, however is this is not the case inaccurately prescribed training may not fully address their unique physiological (e.g cardiovascular response) needs. Prioritizing research on female physiology and exploring sex differences in response to CrossFit® training, we can gain valuable insights into how females differ from males in adapting to this type of exercise. This knowledge can then inform more tailored training methodologies for female athletes to enhance their exercise performance.
Despite the high energy demands of CrossFit® sessions, few studies have investigated the effects of selected CrossFit® exercises on physiological variables including O2, [La−], perceived exertion and HR in real-world contexts (Rios et al., 2023, 2024a; Forte et al., 2022; Tibana et al., 2018). Moreover, data regarding blood pressure responses during these CrossFit® exercises are scarce, necessitating further investigation (Forte et al., 2022). Given CrossFit's increasing popularity among both sexes, with over 15,000 affiliated training centers and a community of 5 million crossfitters (Meier et al., 2023), it is also crucial to explore potential sex differences in these physiological responses, contributing to a better understanding of the sport and for optimal exercise recommendations. Coaches and instructors need this information to provide more informed evidence-based practices and routines for the benefit of both male and female crossfitters. Athletes should be afforded evidence-based information to enhance the effectiveness of their CrossFit® training and reduce the risk of injuries and excessive fatigue. Researchers need this information to formulate research questions and experimental approaches to undertake research of benefit to the CrossFit® community (Rios et al., 2024c). The aim of the study was to assess the cardiorespiratory, hemodynamic and metabolic responses during a high-intensity, short-duration CrossFit® Fran session in trained crossfitters. We also sought to compare these responses between sexes to gain insights into the contrasting acute effects to exercise. Our hypothesis was that a specific CrossFit® session meets the criteria (Ozemek et al., 2025) for developing cardiorespiratory fitness in healthy adults, and that males would exhibit higher O2 (in mL·kg−1·min−1) and HR levels than females, with no differences in hemodynamic and metabolic variables between the sexes.
2. Materials and methods
2.1. Participants
A sample of 25 trained adult participants (15 males and 10 females) who engaged in at least five CrossFit® training sessions per week participated in the study. Table 1 presents their age, anthropometric and physiological characteristics. Recruitment was conducted through announcements at local CrossFit® gyms, with participants meeting the following criteria: (i) more than three years of CrossFit® practice; (ii) no history of cardiopulmonary or intestinal diseases and no musculoskeletal disorders; (iii) age between 18 and 40 years; and (iv) clearance to exercise according to the Physical Activity Readiness Questionnaire. Participants were instructed to abstain from caffeine and alcohol for 24 h before testing, avoid exercise 48 h before experimental sessions, and maintain their regular diet. Informed consent was obtained from all individual participants included in the study. The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Ethics Committee (CEFADE212019) and the guidelines of the World Medical Association for research on humans.
Table 1.
Anthropometric and physiological characteristics of the participants engaged in the current study.
| Variables | Total (n = 25) | Males (n = 15) | Females (n = 10) | p |
|---|---|---|---|---|
| Age (years) | 28.0 ± 5.3 | 29.2 ± 5.8 | 25.9 ± 4.1 | 0.135 |
| Height (cm) | 171.0 ± 8.3 | 176.3 ± 5.6 | 163.1 ± 4.2a | 0.001 |
| Body mass (kg) | 75.6 ± 10.3 | 82.4 ± 7.4 | 65.5 ± 2.8a | 0.001 |
| Body mass index (kg·[m2]−1) | 25.4 ± 1.9 | 26.2 ± 1.9 | 24.1 ± 1.3a | 0.011 |
| Maximal heart rate (b∙min −1) | 189 ± 4 | 188 ± 4 | 190 ± 3 | 0.112 |
| CrossFit® training experience (years) | 5.6 ± 1.7 | 5.5 ± 1.5 | 5.8 ± 2.2 | 0.742 |
Difference to males. Mean ± SD and probabilities (p).
2.2. Experimental design
All tests were performed in a gym facility under the same environmental conditions (23 °C ambient temperature, 60 % humidity and between 8 and 12 a.m.) and supervised by one experienced researcher. Body mass and height were obtained on arrival before the experimental session using a digital scale with a built-in stadiometer (SECA, model 220). Participants remained seated for 10 min before their resting O2, HR, blood pressure, RPE, [La−] and glucose concentrations were measured. After a 5 min warm-up period for joint mobility and Fran specific exercises with low loads, each participant performed the exercises at maximal exertion, with O2 and HR measured continuously during Fran session. O2, HR, [La−], glucose, blood pressure and RPE responses were also measured immediately after, 5- and 30-min post-exercise.
2.3. Methodology
Fran consists of three rounds with repetitions decreasing from 21 to 15-9, with each round including thrusters (combining front squats and overhead presses) and pull-ups. Males use a fixed weight of 43 kg and females a 30 kg weight (Rios et al., 2023). For thrusters, participants lift a barbell from the floor into a front rack position, perform a deep squat, and smoothly transition into an overhead press without pausing. In pull-ups, participants start hanging from a standard pull-up bar with arms fully extended and feet off the ground. They pull themselves up until their chin clears the bar before returning to the starting position. All evaluations of Fran sessions were supervised to ensure participants met the required intensity (no breaks) and completed the specified number of repetitions. Performance time was quantified using a Seiko stopwatch (Yokohama, Japan).
Respiratory and pulmonary gas exchange data were measured breath-by-breath at rest, during the Fran session, and post-exercise using a K4b2 telemetric portable gas analyzer (Cosmed, Rome, Italy) calibrated using the manufacturer instructions with gases of known concentrations. The highest O2 value from Fran session was defined as their peak O2 and to determine the post-exercise O2, the bi-exponential model was computed using VO2FITTING software for data analysis and treatment, as well as for editing and modelling participants’ responses (Rios et al., 2022):
where O2 (t) represents the relative O2 at the time t, A0 is the O2 at rest, H represents the Heaviside step function, Ap and Asc, τp and τsc, TDp and Tsc are the amplitude, time constant and time delay of the O2 fast and slow components (Rios et al., 2022). HR was measured continuously by a Polar T34 Heart Rate Transmitter (Polar, Kempele, Finland), that telemetrically emitted the data to the K4b2 portable unit. The equation used to calculate the predicted maximal HR (HRmax) for each participant and to which the HR data obtained from the Fran session were compared was 208–0.7 x age (Tanaka et al., 2001).
Systolic (SBP) and diastolic (DBP) blood pressures were measured on the left arm, with the cuff applied approximately 2.5 cm from the forearm. Participants were seated comfortably with their arms and hands resting on the table, avoiding movement and talking during measurements. Each participant's blood pressure was recorded three times, with a 30 s break between readings. The average of these three measurements was used to obtain the criterion value. Measurements were taken at baseline, immediately after Fran session and 5 min post-exercise using an automatic blood pressure monitor (OMRON, model HEM-705CP, Chicago, USA). Mean arterial pressure (Meaney et al., 2000) and double product (Jouven et al., 2005) were calculated using the following formulas: mean arterial pressure = DBP + (SBP - DBP)/3 and double product = HR × SBP. To estimate participants' myocardial O2 (Prado Dantas et al., 2018), the following equation was applied: myocardial O2 = (double product x 0.0014) – 6.37.
Capillary blood samples (5 μL) were collected from the earlobe at baseline, immediately after Fran session and 5 min post-exercise for [La−] and glucose assessment using a Lactate Pro 2 (Arkay, Inc, Kyoto, Japan) and Accu-Chek Aviva (Mannheim, Germany) analyzers. The RPE was determined at baseline, immediately after Fran session and 30 min post-exercise using a Borg scale ranging from 6 to 20 (from very, very light to very and very heavy). To ensure accurate interpretation and consistent reporting, the RPE scale was explained individually to the participants according to established recommendations (Borg, 1970).
2.4. Statistical analysis
All statistical calculations were completed using GraphPad Prism 6 (GraphPad Software, Boston, Massachusetts USA), with mean and standard deviation (SD) values for descriptive analysis. The Shapiro–Wilk test was used to assess data normality and/or two-sided 95 % confidence interval reported for all variables (p ≥ 0.05). Repeated-measures analysis of variance (with a Bonferroni post hoc test) was applied to compare the overall (males and females combined) cardiorespiratory, hemodynamics, metabolic and perceived exertion variables at different time points along with the corresponding partial eta squared values. The independent sample t-test was applied to compare male and female characteristics, cardiorespiratory, hemodynamics, metabolic and perceived exertion variables. The effect size was computed with Cohen's d and interpreted as trivial (<0.2), medium (0.2–0.5), or large (≥0.5). Statistical significance was set at p ≤ 0.05.
3. Results
Table 1 presents the anthropometric and physiological characteristics of the participants, with results showing that males had higher body mass and height than females. The overall mean time to complete Fran session was 189 ± 28 s, ranging 177–199 s. Participants had a mean peak O2 of 48.6 ± 3.4 mL kg−1·min−1 and peak HR of 184 ± 6 b min−1, attaining 95 % of their HRmax during Fran session.
During the Fran session and in the post-exercise period, cardiorespiratory, hemodynamic, and metabolic variables remained elevated compared with baseline (Table 2). Perceived exertion was significantly higher 30 min post-exercise compared to baseline (p = 0.001; 6 ± 0 vs 16 ± 1), with no significant differences immediately after the Fran session (16 ± 1).
Table 2.
Overall physiological demands of Fran at baseline, during and at different post-exercise moments.
| Variable | Baseline | Fran | 0 min | 5 min | η2 |
|---|---|---|---|---|---|
| Oxygen uptake (mL∙kg−1∙min −1) | 6.8 ± 0.8 [6.5–7.1] | 39.4 ± 2.0a [38.5–40.2] | – | 22.0 ± 3.0a, c[21.0–23.0] | 0.99 |
| Carbon dioxide (L∙min −1) | 0.6 ± 0.1 [0.5–0.6] | 3.3 ± 1.0a [3.1–4.0] | – | 2.3 ± 0.5a, c[2.1–2.5] | 0.95 |
| Respiratory frequency (b∙min −1) | 18.0 ± 2.0 [16.8–19.0] | 53.3 ± 8.0a [50.1–56.5] | – | 36.0 ± 6.4a, c[33.2–38.4] | 0.91 |
| Minute ventilation (L∙min −1) | 18.4 ± 3.4 [17.3–20.1] | 109.6 ± 24.0a [99.7–119.4] | – | 80.2 ± 18.0a, c[72.9–87.5] | 0.93 |
| Heart rate (b∙min −1) | 82 ± 10 [78–86] | 178 ± 5a [176–180] | – | 131 ± 14a, c[124–136] | 0.97 |
| Systolic blood pressure (mmHg) | 118 ± 8 [114–121] | – | 152 ± 10a [148–156] | 130 ± 6a,b [128–133] | 0.84 |
| Diastolic blood pressure (mmHg) | 79 ± 2 [78–80] | – | 88 ± 4a [87–90] | 81 ± 2a,b [80–82] | 0.83 |
| Mean arterial pressure (mmHg) | 92 ± 3 [90–93] | – | 110 ± 4a [108–112] | 97 ± 2a,b [96–98] | 0.91 |
| Double product (mmHg·b∙min −1) | 9666 ± 1304 [9128–10205] | – | 26630 ± 1870a [25858–27402] | 16953 ± 1966a,b [16141–17765] | 0.96 |
| Myocardial oxygen uptake (mL·100 g LV·min1) | 7.2 ± 2.0 [6.4–8.0] | – | 31.0 ± 2.6a [30.0–32.0] | 17.3 ± 3.0a,b [16.2–18.5] | 0.96 |
| Blood lactate (mmol∙L−1) | 1.5 ± 0.4 [1.4–1.7] | – | 10.3 ± 2.0a [9.5–11.1] | 15.2 ± 1.8a,b [14.5–16.1] | 0.95 |
| Blood glucose (mg∙dL−1) | 94 ± 8 [91–97] | – | 121 ± 13a [116–126] | 132 ± 14a,b [126–138] | 0.78 |
Differences from baseline (p ≤ 0.05).
Differences immediately post-exercise (p ≤ 0.05). Mean ± SD of males and females combined and [95 % confidence intervals].
Differences from Fran (p ≤ 0.05).
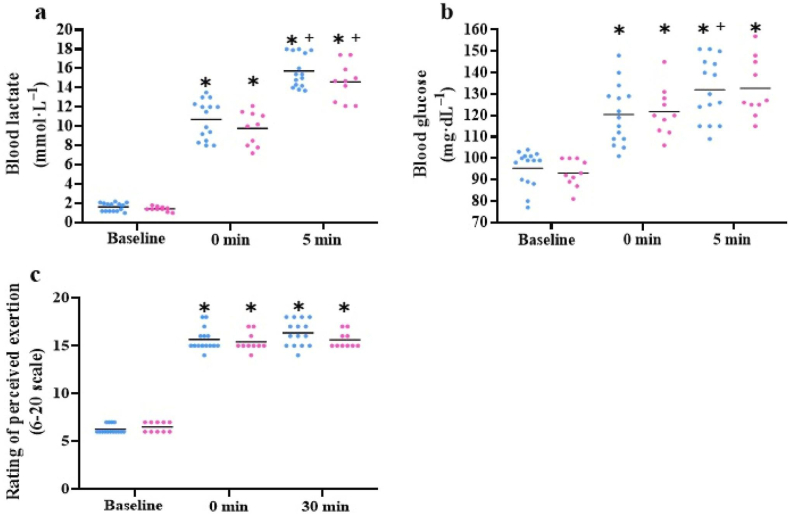
When comparing the sexes, males completed Fran session in less time (177 ± 15 vs 206 ± 27 s; p = 0.006, d = 1.3), resulting in higher peak O2 (50.1 ± 3.3 vs 46.4 ± 2.2 mL kg−1 min−1; p = 0.015, d = 1.3) and HR (187 ± 6 vs 180 ± 3 b min−1; p = 0.039, d = 1.5) than females. The percentage values of HRmax (96 ± 3 vs 93 ± 1 %; p = 0.003, d = 1.3) was also higher in the males. Comparisons of cardiorespiratory, hemodynamic and metabolic responses values at baseline, during Fran and post-exercise in the same sex, and between sexes, at the corresponding time points are shown in Fig. 1, Fig. 2, Fig. 3 (respectively). Cardiorespiratory responses to Fran session were similar in both sexes, showing increases in O2, carbon dioxide, respiratory frequency, minute ventilation and HR during Fran session and 5 min post-exercise (Fig. 1).
Fig. 1.
Cardiorespiratory variables assessed during baseline, Fran and post-exercise for males (blue) and females (pink). ∗ differences from baseline in the same sex (p ≤ 0.05). + differences from Fran in the same sex (p ≤ 0.05). ∗∗ difference between the sexes at the same time point (p ≤ 0.05). Individual and mean values.
Fig. 2.
Hemodynamics variables assessed during baseline and post-exercise for males (blue) and females (pink). ∗ differences from baseline in the same sexes (p ≤ 0.05). + differences from Fran in the same sexes (p ≤ 0.05). Individual and mean values.
Fig. 3.
Metabolic variables and perceived effort assessed during baseline and post-exercise for males (blue) and females (pink). ∗ differences from baseline in the same sexes (p ≤ 0.05). + differences from Fran in the same sexes (p ≤ 0.05). Individual and mean values.
However, males exhibited significantly higher O2 during the Fran session (40.0 ± 1.8 vs 38.4 ± 2.0 mL kg−1 min−1; p = 0.042, d = 1.2), as well as elevated levels of carbon dioxide (3.7 ± 0.4 vs 2.7 ± 0.2 L min−1; p = 0.001, d = 3.2) and minute ventilation (123.9 ± 18.8 vs 88.1 ± 10.9 L min−1; p = 0.001, d = 1.9) during both the Fran session and the recovery period (carbon dioxide: 2.6 ± 0.4 vs 1.8 ± 0.3 L min−1; p = 0.001, d = 3.2; minute ventilation: 89.7 ± 16.1 vs 66.0 ± 7.1 L min−1; p = 0.001, d = 0.1) than females (Fig. 1). In contrast, no significant differences were observed in hemodynamic, metabolic and perceived exertion variables between sexes (Fig. 2, Fig. 3).
4. Discussion
The main outcomes of this study were: (i) a single CrossFit® Fran session induced significant elevations in cardiorespiratory responses ( O2 and HR) both during and post-exercise; (ii) the Fran session increased both blood pressure and double product, which remained elevated 5 min post-exercise; (iii) these increased cardiorespiratory and hemodynamic responses during exercise were accompanied by increased concentrations of [La−] and glucose, coupled with heightened self-reported perceived exertion post-exercise; and (iv) males exhibited higher values in mean and peak O2, and peak HR during Fran session than females, indicating sex-related differences in performance and physiological stress. Our data indicates that a short-duration (∼3 min) CrossFit® Fran session elicited cardiorespiratory demand matching the vigorous intensity threshold defined by the ACSM guidelines (Ozemek et al., 2025), which can contribute to cardiovascular fitness improvements when performed regularly as part of a weekly training program. The high hemodynamic and metabolic demands of the Fran session confirm its utility and effectiveness in prescribing exercises to elicit positive acute effects, with a greater peripheral response in males than females. However, when applied to less experienced or recreational populations, it is essential that external loads and exercise volume are appropriately scaled to ensure safety and promote progressive adaptation.
The mean and peak O2 and HR values during Fran session, reaching approximately 95 % of HRmax, are consistent with the vigorous intensity thresholds defined by the ACSM guidelines for improving cardiorespiratory fitness (Ozemek et al., 2025). While the session duration is brief, such intensity—when applied regularly—can contribute meaningfully to weekly physical activity targets (Garber et al., 2011). Our results correspond with previous studies assessing O2 (Rios et al., 2023) and HR (Tibana et al., 2018; Leitão et al., 2021) during a Fran session, indicating higher values than maximal effort cycling sprint interval training (Freese et al., 2013; Williams and Kraemer, 2015) and cycling high-intensity interval exercise (Järvinen et al., 2022). The elevated O2 and HR values observed during Fran session may be attributed to the overall exercise volume and the absence of breaks, facilitating an elevated demand on the cardiac, vascular and respiratory systems. The movement of the arms and trunk demands a significant O2 requirement compared with cycling, where they have a lower contribution to the total exercise O2 (Hill et al., 2003). In fact, efforts to complete an exercise session in the shortest time yield an increase in minute ventilation, respiratory frequency and carbon dioxide (Tipton et al., 2017). The increase in minute ventilation is a result of both heightened tidal volume and an elevated respiratory frequency during exercise (Tipton et al., 2017; Guenette and Sheel, 2007). Carbon dioxide is highly related to minute ventilation since this latter variable allows the CO2 removal, preventing or constraining the perturbation in arterial blood gas and acid-base status (Poole and Jones, 2012).
Cardiorespiratory function (e.g. O2, minute ventilation, carbon dioxide, respiratory frequency and HR) remained elevated during the recovery period compared with baseline values, consistent with resistance exercise (Rios et al., 2024a; Thornton and Potteiger, 2002). The high intensity and great volume of muscle contractions (90 repetitions of thrusters plus pull ups) during Fran session contributed to the marked increase in O2 post-exercise compared with baseline (Rios et al., 2023). This increase can possibly be attributed to several factors, including the restoration of muscle adenosine triphosphate and creatine phosphate reserves, the replenishment of O2 reserves in the blood and muscles (Thornton and Potteiger, 2002). The high values of minute ventilation (∼80 L min−1) during the recovery period are additional evidence of the disruption of homeostasis after a single Fran session of maximal effort (Thornton and Potteiger, 2002). This increased minute ventilation may also be interpreted as a compensatory mechanism aimed at restoring the normal pH that may have been disturbed by the accumulation of high [La−] levels post-exercise (∼15 mmol L−1). In this study, the cardiovascular intensity achieved during Fran session was ∼95 % of HRmax, indicating greater hemodynamic stress when compared to other studies evaluating blood pressure after Fran session and high-intensity functional exercises performed at maximal effort (Forte et al., 2022). These results can be explained by the differences in the duration and intensity of CrossFit® exercises, coupled with the lack of inter-round rest (which intensifies mechanical occlusion of blood flow during Fran session). These factors collectively contribute to an increase in cardiovascular function, affecting plasma volume, cardiac output and stroke volume (MacDonald, 2002). While not the main focus of this study, the observed increases in post-exercise O2 could potentially facilitate greater calorie expenditure at rest, offering potential benefits for weight control among crossfitters.
The Fran session elicited an immediate post-exercise increase in [La−] level, indicating a substantial contribution from the anaerobic pathway. This outcome aligns with previous studies that have assessed the impact of a Fran session on [La−] levels (Rios et al., 2023; Tibana et al., 2018; Leitão et al., 2021) and compared to high-intensity functional exercises (Forte et al., 2022; Rios et al., 2021). However, these values were higher than those observed in high-intensity interval exercises (Maté-Muñoz et al., 2018). The Fran session demands total effort and a higher energy/time ratio, intensifying the contribution of the anaerobic pathway, thereby contributing to an increase in metabolic acidosis. In support of this assertion, the anaerobic contribution during Fran session increased carbohydrate metabolism evidenced by increased glucose at the muscular level (see Table 2). This increase in glucose is consistent with previous examination of the Fran session (Rios et al., 2023), and higher than those reported to high-intensity functional exercises (Rios et al., 2024b). In summary, it appears that [La−] and glucose responses depend on both the duration of effort and structure of the CrossFit® exercise session. Efforts performed at maximal intensity yielded higher levels of [La−] and glucose, indicating increased reliance on anaerobic metabolism. Perceived exertion of the participants in this study was "hard" or "very hard", consistent with other studies employing the Borg scale to assess the intensity of Fran session (Rios et al., 2023; Tibana et al., 2018; Leitão et al., 2021) and high-intensity functional exercises (Rios et al., 2024b; Maté-Muñoz et al., 2018). A higher RPE value, combined with elevated metabolic responses after an intense Fran session, indicates the presence of sufficient stimuli to promote neuromuscular and metabolic adaptations associated with improved muscular endurance and fatigue resistance (Rios et al., 2023).
Sex differences can influence the pattern of adaptation to acute and chronic exercise (Howden et al., 2015; Coe and Astorino, 2023). A recent study (Forte et al., 2022) demonstrated no differences in the acute physiological responses (e.g. HR, [La−] and blood pressure) between males and females during and after CrossFit® exercises (like Cindy and Diane). In contrast, a longitudinal study (Poderoso et al., 2019) reported increased testosterone and decreased cortisol levels in males during CrossFit® training, with no hormonal changes evident in females. This study did not explore potential associations between these hormonal changes and other physiological adaptations. Differences in physiological responses between the sexes became apparent when the data were separated and directly compared. We observed a marked difference in the total duration of the Fran session, with males completing it in mean of 177 s compared to females who took 206 s. In addition, the males had higher peak O2 and HR levels than females. Females generally demonstrate relatively lower maximal O2 values than males when cycling or running (Lepers, 2019). This difference has often been attributed to central factors, such as the presence of a smaller heart and lower hemoglobin mass in females, which can limit their capacity to effectively deliver oxygen to skeletal muscles (Howden et al., 2015). Considering the absence of differences in hemodynamic and metabolic function between the sexes, it appears the variation in physiological response between males and females during an acute Fran session is influenced by central and peripheral mechanisms. These peripheral effects refer to local muscular responses, such as greater oxygen extraction, increased metabolite accumulation, and higher reliance on anaerobic glycolysis (Coe and Astorino, 2023; Jaworowski et al., 2002). In our data, males showed higher O2, minute ventilation and carbon dioxide production, suggesting increased muscular metabolic demand and a stronger ventilatory compensatory response to meet the high metabolic cost of the Fran session. Males generally exhibit a higher ATP synthesis capacity to sustain muscle contraction during intense efforts, which is attributed to their superior aerobic and anaerobic power (Jaworowski et al., 2002). This outcome is primarily due to a greater glycolytic capacity, supporting their enhanced ability to generate energy anaerobically compared to females (Jaworowski et al., 2002). Together, these factors likely contributed to the males ability to maintain a higher work rate and complete the session more quickly, highlighting the importance of considering sex-specific physiological traits in performance analysis.
Assessing benchmark sessions like Fran is vital, as they serve as standard exercises used by CrossFit® professionals to gauge fitness levels, featuring specific load parameters and movement patterns (Meier et al., 2023). These results carry practical significance, indicating that researchers and coaches can incorporate the Fran session performed at maximal effort into a comprehensive plan to assess and monitor the training status of crossfitters throughout a CrossFit® training season, providing a strategic guide for achieving excellence in this challenging sport. These findings suggest that the Fran session can be used to individualize training loads based on sex-specific physiological responses. Monitoring performance time and cardiorespiratory variables allows coaches to detect fatigue, guide recovery strategies, and adjust intensity throughout a training cycle. Additionally, repeated Fran assessments may serve as a benchmark to track progress and inform periodization. The time-efficient format makes it a practical tool for routine performance evaluation in both competitive and recreational settings.
Several limitations of the current study warrant acknowledgment. First, we acknowledge that exercise performance in females could be influenced by variations in menstrual cycle phases. Since we did not collect this information, we were unable to address this issue. Secondly, it should be noted that these results are applicable primarily to trained individuals, and thus may not be directly transferable to untrained individuals. Longitudinal studies examining cardiorespiratory, hemodynamic and metabolic responses to multiple days of CrossFit® training, employing consistent methodologies, are essential for gaining a deeper insight into chronic physiological adaptations in individuals of varying sexes and CrossFit® experience levels. Finally, although energy expenditure was not the focus of this study, future research could benefit from quantifying energy cost during and after CrossFit® benchmark sessions, to better understand total session load and its implications for training and recovery.
5. Conclusions
A single session of Fran CrossFit® induces substantial elevations in cardiorespiratory and hemodynamic responses, along with increased post-exercise metabolic activity and perceived exertion, observed in trained crossfitters of both sexes. These responses reached the vigorous intensity threshold defined by the ACSM, indicating that short-duration, high-intensity sessions like Fran can contribute meaningfully to improvements in cardiovascular fitness when performed regularly. Nevertheless, when applying this type of workout to recreational or less experienced populations, careful scaling of intensity and volume is essential to ensure safety and promote long-term adaptation. These outcomes offer valuable insights for coaches and CrossFit® participants, facilitating informed training planning and optimization. However, given the diverse range of workouts and activities within CrossFit®, further research is warranted to comprehensively understand the physiological and performance effects associated with this fitness regimen.
Author contributions
Conceptualization: M.R., D.M-G. and R.J.F.; methodology: M.R., D.M-G., D.B.P. and R.J.F; formal analysis: M.R.; investigation: M.R. and R.C.; resources: M.R. and R.J.F.; writing—original draft preparation, M.R.; writing—review and editing: M.R., R.C., V.M.R., D.M-G., D.B.P and R.J.F.; visualization: M.R.; supervision: D.M-G. and R.J.F.; project administration: M.R. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by national funding through the Portuguese Foundation for Science and Technology, I.P (FCT). and the European Union (EU) to Manoel Rios (FCT202104701BD; https://doi.org/10.54499/2021.04701.BD), Victor Machado Reis (UIDB/04045/2020) and under project/support UIDB/05913/2020—Centre of Research, Education, Innovation and Intervention in Sport (https://doi.org/10.54499/UIDB/05913/2020).
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
The authors express their gratitude to all study participants and collaborators. The authors declare no conflict of interest.
Footnotes
This article is part of a special issue entitled: Sex Differences in Physiology published in Current Research in Physiology.
Current Research in Physiology.
Data availability
Data will be made available on request.
References
- Borg G. Perceived exertion as an indicator of somatic stress. Scand. J. Rehabil. Med. 1970;2:92–98. [PubMed] [Google Scholar]
- Clarke J., De Lannoy L., Ross R. Comparison of measures of maximal and submaximal fitness in response to exercise. Med. Sci. Sports Exerc. 2017;49:711–716. doi: 10.1249/MSS.0000000000001164. [DOI] [PubMed] [Google Scholar]
- Claudino J.G., Gabbett T.J., Bourgeois F., Souza H.d.S., Miranda R.C., Mezêncio B., Soncin R., Cardoso Filho C.A., Bottaro M., Hernandez A.J. CrossFit overview: systematic review and meta-analysis. Sports. Med. Open. 2018;4:1–14. doi: 10.1186/s40798-018-0124-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coe L.N., Astorino T.A. Sex differences in hemodynamic response to high‐intensity interval exercise. Scand. J. Med. Sci. Sports. 2023 doi: 10.1111/sms.14495. [DOI] [PubMed] [Google Scholar]
- Forte L.D., Freire Y.G., Júnior J.S., Melo D.A., Meireles C.L. Physiological responses after two different CrossFit workouts. Biol. Sport. 2022;39:231–236. doi: 10.5114/biolsport.2021.102928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freese E.C., Gist N.H., Cureton K.J. Physiological responses to an acute bout of sprint interval cycling. J. Strength Condit Res. 2013;27:2768–2773. doi: 10.1519/JSC.0b013e318281575c. [DOI] [PubMed] [Google Scholar]
- Garber C.E., Blissmer B., Deschenes M.R., Franklin B.A., Lamonte M.J., Lee I.-M., Nieman D.C., Swain D.P. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011;43:1334–1359. doi: 10.1249/MSS.0b013e318213fefb. [DOI] [PubMed] [Google Scholar]
- Gillen J.B., Gibala M.J. Interval training: a time-efficient exercise strategy to improve cardiometabolic health. Appl. Physiol. Nutr. Metabol. 2018;43:iii–iv. doi: 10.1139/apnm-2018-0453. [DOI] [PubMed] [Google Scholar]
- Goodman J.M., Pallandi D.V., Reading J.R., Plyley M.J., Liu P.P., Kavanagh T. Central and peripheral adaptations after 12 weeks of exercise training in post-coronary artery bypass surgery patients. J. Cardiopulm. Rehabil. Prev. 1999;19:144–150. doi: 10.1097/00008483-199905000-00001. [DOI] [PubMed] [Google Scholar]
- Gripp F., Nava R.C., Cassilhas R.C., Esteves E.A., Magalhães C.O.D., Dias-Peixoto M.F., de Castro Magalhães F., Amorim F.T. HIIT is superior than MICT on cardiometabolic health during training and detraining. Eur. J. Appl. Physiol. 2021;121:159–172. doi: 10.1007/s00421-020-04502-6. [DOI] [PubMed] [Google Scholar]
- Guenette J.A., Sheel A. Physiological consequences of a high work of breathing during heavy exercise in humans. J. Sci. Med. Sport. 2007;10:341–350. doi: 10.1016/j.jsams.2007.02.003. [DOI] [PubMed] [Google Scholar]
- Hill D.W., Halcomb J.N., Stevens E.C. Oxygen uptake kinetics during severe intensity running and cycling. Eur. J. Appl. Physiol. 2003;89:612–618. doi: 10.1007/s00421-002-0779-x. [DOI] [PubMed] [Google Scholar]
- Hottenrott L., Möhle M., Ide A., Ketelhut S., Stoll O., Hottenrott K. Recovery from different high-intensity interval training protocols: comparing well-trained women and men. Sports. 2021;9:34. doi: 10.3390/sports9030034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howden E.J., Perhonen M., Peshock R.M., Zhang R., Arbab-Zadeh A., Adams-Huet B., Levine B.D. Females have a blunted cardiovascular response to one year of intensive supervised endurance training. J. Appl. Physiol. 2015;119:37–46. doi: 10.1152/japplphysiol.00092.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Järvinen L., Lundin Petersdotter S., Chaillou T. High-intensity resistance exercise is not as effective as traditional high-intensity interval exercise for increasing the cardiorespiratory response and energy expenditure in recreationally active subjects. Eur. J. Appl. Physiol. 2022:1–16. doi: 10.1007/s00421-021-04849-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaworowski A., Porter M.M., Holmbäck A.M., Downham D., Lexell J. Enzyme activities in the tibialis anterior muscle of young moderately active men and women: relationship with body composition, muscle cross-sectional area and fibre type composition. Acta Physiol. Scand. 2002;176:215–225. doi: 10.1046/j.1365-201X.2002.t01-2-01004.x. [DOI] [PubMed] [Google Scholar]
- Jouven X., Empana J.P., Schwartz P.J., Desnos M., Courbon D., Ducimetière P. Heart-rate profile during exercise as a predictor of sudden death. N. Engl. J. Med. 2005;352:1951–1958. doi: 10.1056/NEJMoa043012. [DOI] [PubMed] [Google Scholar]
- Leitão L., Dias M., Campos Y., Vieira J.G., Sant'Ana L., Telles L.G., Tavares C., Mazini M., Novaes J., Vianna J. Physical and physiological predictors of FRAN CrossFit® WOD athlete's performance. Int. J. Environ. Res. Publ. Health. 2021;18:4070. doi: 10.3390/ijerph18084070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lepers R. Sex difference in triathlon performance. Front. Physiol. 2019;10:973. doi: 10.3389/fphys.2019.00973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopes A.M., Rios M., Beleza J., Carvalho D.D., Monteiro S., Montanha T., Martins S., Guimarães J.T., Fernandes R.J., Magalhães J. Adding protein to a carbohydrate pre-exercise beverage does not influence running performance and metabolism. J. Sports Med. Phys. Fit. 2022;12 doi: 10.23736/S0022-4707.22.13714-X. [DOI] [PubMed] [Google Scholar]
- MacDonald J.R. Potential causes, mechanisms, and implications of post exercise hypotension. J. Hum. Hypertens. 2002;16:225–236. doi: 10.1038/sj.jhh.1001377. [DOI] [PubMed] [Google Scholar]
- MacInnis M.J., Gibala M.J. Physiological adaptations to interval training and the role of exercise intensity. J. Physiol. 2017;595:2915–2930. doi: 10.1113/jp273196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maté-Muñoz J.L., Lougedo J.H., Barba M., Cañuelo-Márquez A.M., Guodemar-Pérez J., García-Fernández P., Lozano-Estevan M.d.C., Alonso-Melero R., Sánchez-Calabuig M.A., Ruíz-López M. Cardiometabolic and muscular fatigue responses to different crossfit® workouts. J. Sports Sci. Med. 2018;17:668. [PMC free article] [PubMed] [Google Scholar]
- McDougle J.M., Mangine G.T., Townsend J.R., Jajtner A.R., Feito Y. Acute physiological outcomes of high-intensity functional training: a scoping review. PeerJ. 2023;11 doi: 10.7717/peerj.14493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meaney E., Alva F., Moguel R., Meaney A., Alva J., Webel R. Formula and nomogram for the sphygmomanometric calculation of the mean arterial pressure. Heart. 2000;84:64. doi: 10.1136/heart.84.1.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meier N., Schlie J., Schmidt A. CrossFit(®): 'unknowable' or predictable?-A systematic review on predictors of CrossFit(®) performance. Sports (Basel) 2023;11 doi: 10.3390/sports11060112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozemek C., Bonikowske A., Christle J., Gallo P. Lippincott Williams & Wilkins; 2025. ACSM's Guidelines for Exercise Testing and Prescription. [Google Scholar]
- Poderoso R., Cirilo-Sousa M., Júnior A., Novaes J., Vianna J., Dias M., Leitão L., Reis V., Neto N., Vilaça-Alves J. Gender differences in chronic hormonal and immunological responses to CrossFit®. Int. J. Environ. Res. Publ. Health. 2019;16:2577. doi: 10.3390/ijerph16142577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poole D.C., Jones A.M. Oxygen uptake kinetics. Compr. Physiol. 2012;2:933–996. doi: 10.1002/cphy.c100072. [DOI] [PubMed] [Google Scholar]
- Prado Dantas T.S., Aidar F.J., de Souza R.F., de Matos Gama D., Pires Ferreira A.R., de Almeida Barros N., Matos dos Santos M.D., Oliveira Barros G., Rodrigues Santos C.R., da Silva Júnior W.M. Evaluation of a CrossFit® session on post-exercise blood pressure. J Exerc Physiol onl. 2018;21 [Google Scholar]
- Rios M., Macan T., Stevanović-Silva J., Nhusawi K., Fernandes R.J., Beleza J., Ascensão A., Magalhães J. Acute CrossFit® workout session impacts blood redox marker modulation. Physiologia. 2021;1:13–21. doi: 10.3390/physiologia1010004. [DOI] [Google Scholar]
- Rios M., Reis V.M., Soares S., Moreira-Gonçalves D., Fernandes R.J. Pros and cons of two methods of anaerobic alactic energy assessment in a high-intensity CrossFit® workout. Oxygen. 2022;2:621–627. [Google Scholar]
- Rios M., Zacca R., Azevedo R., Fonseca P., Pyne D.B., Reis V.M., Moreira-Gonçalves D., Fernandes R.J. Bioenergetic analysis and fatigue assessment during the workout fran in experienced crossfitters. Int. J. Sports Physiol. Perform. 2023;1:1–7. doi: 10.1123/ijspp.2022-0411. [DOI] [PubMed] [Google Scholar]
- Rios M., Becker K.M., Monteiro A.S., Fonseca P., Pyne D.B., Reis V.M., Moreira-Gonçalves D., Fernandes R.J. Effect of the fran CrossFit workout on oxygen uptake kinetics, energetics, and postexercise muscle function in trained CrossFitters. Int. J. Sports Physiol. Perform. 2024:1–8. doi: 10.1123/ijspp.2023-0201. [DOI] [PubMed] [Google Scholar]
- Rios M., Becker K.M., Cardoso F., Pyne D.B., Reis V.M., Moreira-Gonçalves D., Fernandes R.J. Assessment of cardiorespiratory and metabolic contributions in an extreme intensity CrossFit(®) benchmark workout. Sensors (Basel) 2024;24 doi: 10.3390/s24020513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rios M., Pyne D.B., Fernandes R.J. The effects of CrossFit(®) practice on physical fitness and overall quality of life. Int. J. Environ. Res. Publ. Health. 2024;22 doi: 10.3390/ijerph22010019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabata I., Irisawa K., Kouzaki M., Nishimura K., Ogita F., Miyachi M. Metabolic profile of high intensity intermittent exercises. Med. Sci. Sports Exerc. 1997;29:390–395. doi: 10.1097/00005768-199703000-00015. [DOI] [PubMed] [Google Scholar]
- Tanaka H., Monahan K.D., Seals D.R. Age-predicted maximal heart rate revisited. J. Am. Coll. Cardiol. 2001;37:153–156. doi: 10.1016/s0735-1097(00)01054-8. [DOI] [PubMed] [Google Scholar]
- Thornton M.K., Potteiger J.A. Effects of resistance exercise bouts of different intensities but equal work on EPOC. Med. Sci. Sports Exerc. 2002;34:715–722. doi: 10.1097/00005768-200204000-00024. [DOI] [PubMed] [Google Scholar]
- Tibana R.A., De Sousa N.M.F., Prestes J., Voltarelli F.A. Lactate. Heart rate and rating of perceived exertion responses to shorter and longer duration CrossFit® training sessions. J Funct Morphol. 2018;3:60. doi: 10.3390/jfmk3040060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Timón R., Olcina G., Camacho-Cardeñosa M., Camacho-Cardenosa A., Martinez-Guardado I., Marcos-Serrano M. 48-hour recovery of biochemical parameters and physical performance after two modalities of CrossFit workouts. Biol. Sport. 2019;36:283–289. doi: 10.5114/biolsport.2019.85458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tipton M.J., Harper A., Paton J.F., Costello J.T. The human ventilatory response to stress: rate or depth? J. Physiol. 2017;595:5729–5752. doi: 10.1113/JP274596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams B.M., Kraemer R.R. Comparison of cardiorespiratory and metabolic responses in kettlebell high-intensity interval training versus sprint interval cycling. J. Strength Condit Res. 2015;29:3317–3325. doi: 10.1519/JSC.0000000000001193. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data will be made available on request.