Abstract
Purpose
To assess quality of life (QOL) data from a double-blind Phase III study evaluating bicalutamide (Casodex™) 80 mg as part of maximum androgen blockade (MAB) in patients with previously untreated advanced prostate cancer.
Methods
Patients with untreated stage C/D prostate cancer were randomized to MAB with bicalutamide plus a luteinizing hormone-releasing hormone agonist (LHRHa) or LHRHa monotherapy. QOL was evaluated at baseline and at weeks 1, 5, and 24 using the Japanese version of the Functional Assessment of Cancer Therapy-Prostate (FACT-P) questionnaire.
Results
A total of 203 patients were assessed for QOL. The MAB group had more rapid and greater improvements in “emotional well-being” and “prostate cancer-specific issues” domain scores than the monotherapy group. Further analysis of “prostate cancer-specific issues” revealed that, compared with monotherapy, MAB provided a greater improvement in “micturition disorder”-related QOL. Complete improvement rates for items related to “pain and micturition disorder” were also higher with MAB. Item scores of “pain and micturition disorder” did not correlate strongly with prostate-specific antigen levels or tumor size. Fewer patients who had deterioration in their “pain and micturition disorder” item scores at week 1 in the MAB group than the monotherapy group.
Conclusions
Maximum androgen blockade with bicalutamide plus LHRHa did not reduce the overall QOL of patients with previously untreated advanced prostate cancer. MAB was superior to monotherapy in achieving early improvement of QOL related to micturition disorder and pain.
Keywords: Advanced prostate cancer, Bicalutamide, Luteinizing hormone releasing hormone agonist, Maximum androgen blockade, Quality of life
Introduction
Maximum androgen blockade (MAB) therapy is the combination of an anti-androgen with orchiectomy or a luteinizing hormone-releasing hormone agonist (LHRHa) to eliminate the exposure of prostate-cancer cells to androgens of both testicular and adrenal origin. In recent years, it has been widely used in Japan to maximize the effect of endocrine therapy on prostate cancer. Although the usefulness of MAB therapy has been disputed for safety and economic reasons (Eisenberger et al. 1998; McLeod et al. 1992), the results of a meta-analysis by the Prostate Cancer Trialists’ Collaborative Group demonstrated that MAB therapy using non-steroidal anti-androgens significantly improved 5-year survival compared with castration alone (Prostate Cancer Trialists’ Collaborative Group 2000). Subsequently, bicalutamide (Casodex™), a non-steroidal anti-androgen with more potent effect and better tolerability than flutamide (Akaza 1999), has been developed and used in MAB therapy. It has been shown that bicalutamide reduces the risk of death by 20% compared with castration alone (Klotz et al. 2004) and has a better safety profile than flutamide, causing diarrhea significantly less frequently (Schellhammer et al. 1997).
We have previously presented the results of a Phase III study in which we directly compared MAB therapy using bicalutamide and an LHRHa with LHRHa monotherapy in patients with previously untreated prostate cancer (Akaza et al. 2004, 2005; Usami et al. 2007). We found that MAB therapy was significantly superior to LHRHa monotherapy in terms of tumor response, time-to-treatment failure, and time-to-progression, yet its safety was comparable to that of LHRHa monotherapy (Akaza et al. 2004, 2005; Usami et al. 2007). The effects of these therapies on quality of life (QOL) was a secondary endpoint in this study; here we present a comprehensive analysis of the effects of MAB therapy using bicalutamide for the prevention of deteriorating QOL caused by micturition disorders (painful micturition, dysuria, pollakiuria, etc.) due to advanced prostate cancer, and by bone pain and fatigue due to bone metastases. We also investigated whether the addition of bicalutamide to LHRHa had the potential to improve the QOL of patients with advanced prostate cancer.
Patients and methods
Patients and study design
This was a multicenter, double-blind, comparative study involving patients with previously untreated advanced prostate cancer (stage C/D) who visited any of the 49 study centers in Japan between February 2000 and December 2001. The patients were randomized to either a combination therapy group receiving LHRHa (subcutaneous administration of goserelin acetate 3.6 mg (Zoladex™) or leuprorelin acetate 3.75 mg once every 4 weeks) and bicalutamide [80 mg/(day p.o.)] or a monotherapy group receiving an LHRHa (as above) and bicalutamide matching placebo (once daily, p.o.). The randomized treatment was continued until December 2003, or until there was evidence of disease progression or any other event leading to withdrawal. Full details of the study design are provided in our previous report (Akaza et al. 2004). Written, informed consent was obtained from all participating patients prior to the start of the study, which was conducted in accordance with the ethical principles of the Declaration of Helsinki and Good Clinical Practice guidelines.
The primary endpoints of this study were percentage of withdrawals due to adverse drug reactions (ADRs) and objective tumor response [overall tumor response and prostate-specific antigen (PSA) normalization rate] at week 12 of the treatment. The secondary endpoints included time-to-treatment failure, time to death, time to progression, adverse events, maximum objective tumor response, subjective response, relationship between changes in PSA and prognosis, objective tumor response rate of bicalutamide as the second-line treatment, anti-androgen withdrawal syndrome, and QOL.
Evaluation of QOL
Quality of life was evaluated at baseline and at weeks 1, 5, and 24 of treatment in both groups using the Japanese version (Hinotsu et al. 1999) of the Functional Assessment of Cancer Therapy-Prostate (FACT-P) questionnaire (Esper et al. 1997). FACT-P is a self-reporting patient QOL questionnaire consisting of five domains; namely, “Physical well-being” (7 items), “Social/family well-being” (9 items), “Emotional well-being” (6 items), “Functional well-being” (7 items), and “Prostate cancer-specific issues” (12 items; see “Appendix”). Patients responded to each item using a 5-point scale ranging from 0 to 4 (0 not at all, 1 a little bit, 2 somewhat, 3 quite a bit, 4 very much). The item scores were calculated from the response according to scoring guidelines. The sum of all the item scores of each domain was the domain score, and the sum of all five domain scores was the total score, with a higher score indicating better QOL.
QOL score and satisfaction rate at baseline
The QOL satisfaction rate (%) (maximum 100%) was based on the total score, the five domain scores, and the 12-item scores of the domain “Prostate cancer-specific issues”.
Effect of treatments on QOL scores
The effect of treatments on QOL was assessed from changes from baseline in total score; the five domain scores; and the 12-item scores of the domain “prostate cancer-specific issues” at weeks 1, 5, and 24. Changes in QOL were compared between the treatment groups at weeks 1, 5, and 24. Missing values at weeks 1, 5, and 24 were imputed using a last value carried forward (LVCF) approach (Yoshida et al. 1997). For comparison, the data were also analyzed without imputation of missing values.
Complete improvement rate for prostate cancer-specific items
The complete improvement rate of items (percentage of patients achieving an increase in an item score from 0 to 3 at baseline to 4 at week 24) was calculated for each of the 12 items of the domain “prostate cancer-specific issues”, and compared between the groups.
Disease-related factors influencing “prostate cancer-specific issues”
To investigate whether item scores of “pain” (items 3–5) and “micturition disorder” (items 9–11: urination difficulty, frequent urination, limited activities due to urination trouble; see “Appendix”) of the domain “prostate cancer-specific issues” correlated with PSA level and tumor size, Spearman’s correlation coefficient was calculated between the item scores and PSA values at baseline and week 24, and between item scores and tumor size at baseline. This was undertaken for all patients, without subdividing by treatment group. We also evaluated Spearman’s correlation coefficient between the changes of item scores from baseline to week 5 and the percentage changes in PSA levels from baseline to week 4, and between the changes of item scores from baseline to week 5 and the percentage changes of tumor size from baseline to week 12.
Disease flare
To verify that the addition of bicalutamide (in the MAB group) suppresses disease flare associated with the initiation of LHRHa therapy, the number of patients whose item scores of “pain” (items 3–5) and “micturition disorder” (items 9–11) of the “prostate cancer-specific issues” domain (see “Appendix”) worsened from baseline to week 1 was compared between the two groups.
Statistical analysis
Changes from baseline in QOL scores at weeks 1, 5, and 24, and intergroup differences in the changes in QOL scores, were compared using t-tests, and intergroup differences in the complete improvement rate of QOL were assessed using the χ 2 test and Fisher’s exact test, all at the 5% significance level. The disease-related factors influencing QOL were assessed using Spearman’s correlation coefficient. Intergroup differences for the purpose of evaluating disease flare were assessed using Fisher’s exact test. Since all analyses were exploratory, the adjustment for multiplicity was not performed.
Results
In total, 205 patients participated in the study (MAB group: n = 102; monotherapy group: n = 103), and 203 of those (MAB: n = 102; monotherapy: n = 101) were available for QOL analysis. There were no significant differences in the distribution of patient characteristics between the two groups (Table 1). The collection rates of QOL questionnaires were 89–100% in both groups throughout the study period.
Table 1.
Patient characteristics
| Characteristic | Number of patients (%) | |
|---|---|---|
| MAB (n = 102) | Monotherapy (n = 101) | |
| Age | ||
| <75 years | 53 (52.0) | 50 (49.5) |
| ≥75 years | 49 (48.0) | 51 (50.5) |
| PSA | ||
| <60 ng/ml | 40 (39.2) | 37 (36.6) |
| ≥60 ng/ml | 62 (60.8) | 64 (63.4) |
| Histopathological class | ||
| Well | 3 (2.9) | 6 (5.9) |
| Moderate | 52 (51.0) | 55 (54.5) |
| Poorly | 47 (46.1) | 40 (39.6) |
| Clinical stage | ||
| C/D1 | 59 (57.8) | 57 (56.4) |
| D2 | 43 (42.2) | 44 (43.6) |
| Disease stage | ||
| T2 | 3 (2.9) | 1 (1.0) |
| T3 | 83 (81.4) | 77 (76.2) |
| T4 | 16 (15.7) | 23 (22.8) |
| Nodal stage | ||
| N0 | 74 (72.5) | 63 (62.4) |
| N1 | 28 (27.5) | 38 (37.6) |
| Metastatic status | ||
| M0 | 59 (57.8) | 58 (57.4) |
| M1 | 43 (42.2) | 43 (42.6) |
| Lesion of metastases | ||
| Bone | 40 (39.2) | 40 (39.6) |
| Lymph node | 28 (27.5) | 38 (37.6) |
| Other | 2 (2.0) | 3 (3.0) |
| LHRHa | ||
| Goserelin acetate | 77 (75.5) | 79 (78.2) |
| Leuprorelin acetate | 25 (24.5) | 22 (21.8) |
| Performance status | ||
| 0, 1 | 99 (97.1) | 99 (98.0) |
| 2 | 3 (2.9) | 2 (2.0) |
LHRHa luteinizing hormone-releasing hormone agonist, MAB maximum androgen blockade, PSA prostate-specific antigen
QOL score and satisfaction rate at baseline
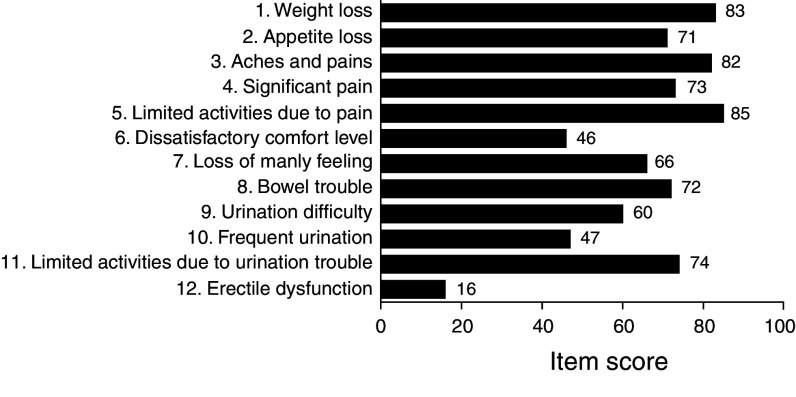
There were no significant differences in baseline QOL score between the two treatment groups. The QOL satisfaction rate overall was 70.3%, and by domain, 86.5% for “physical well-being”, 64.5% for “social/family well-being”, 73.5% for “emotional well-being”, 71.3% for “functional well-being”, and 65.1% for “prostate cancer-specific issues”. Thus, the lowest QOL domain scores were in “social/family well-being” and “prostate cancer-specific issues”. Individual QOL item scores for “prostate cancer-specific issues” at baseline are shown in Fig. 1.
Fig. 1.
Individual item scores for “prostate cancer-specific issues” and degree of satisfaction at baseline
Effect of treatments on QOL scores
The changes in total and each domain score of FACT-P from baseline are shown in Table 2. The total score tended to increase over time in the MAB group and decreased in the monotherapy group at weeks 1 and 5. A significant difference between the two groups was observed at week 5 (P < 0.001).
Table 2.
Changes in total and individual domain scores of FACT-P
| Domain | Week | Group | Mean ± SD (n) | t-test |
|---|---|---|---|---|
| 1. Physical | 1 | MAB | 0.53 ± 2.90 (93) | P = 0.229* |
| Monotherapy | 0.02 ± 2.86 (94) | |||
| 5 | MAB | 0.56 ± 2.91 (93) | P = 0.209* | |
| Monotherapy | 0.00 ± 3.15 (94) | |||
| 24 | MAB | 0.70 ± 3.94 (93) | P = 0.072* | |
| Monotherapy | −0.27 ± 3.41 (94) | |||
| 2. Social/family | 1 | MAB | −1.15 ± 7.59 (94) | P = 0.711* |
| Monotherapy | −1.55 ± 7.14 (96) | |||
| 5 | MAB | −1.21 ± 8.29 (94) | P = 0.131* | |
| Monotherapy | −2.96 ± 7.64 (96) | |||
| 24 | MAB | −2.31 ± 8.18 (94) | P = 0.724* | |
| Monotherapy | −1.89 ± 8.32 (96) | |||
| 3. Emotional | 1 | MAB | 1.39 ± 3.25 (93) | P = 0.027* |
| Monotherapy | 0.36 ± 3.12 (95) | |||
| 5 | MAB | 1.55 ± 3.89 (93) | P = 0.132* | |
| Monotherapy | 0.70 ± 3.87 (95) | |||
| 24 | MAB | 2.21 ± 4.19 (93) | P = 0.075* | |
| Monotherapy | 1.16 ± 3.82 (95) | |||
| 4. Functional | 1 | MAB | −0.76 ± 3.71 (94) | P = 0.870* |
| Monotherapy | −0.85 ± 3.82 (96) | |||
| 5 | MAB | 0.21 ± 4.05 (94) | P = 0.063† | |
| Monotherapy | −1.11 ± 5.56 (96) | |||
| 24 | MAB | 0.24 ± 5.95 (94) | P = 0.924* | |
| Monotherapy | 0.32 ± 5.44 (96) | |||
| 5. Prostate cancer-specific issues | 1 | MAB | 0.88 ± 5.55 (92) | P = 0.672* |
| Monotherapy | 0.52 ± 5.97 (93) | |||
| 5 | MAB | 3.14 ± 5.92 (92) | P = 0.011* | |
| Monotherapy | 0.90 ± 5.94 (93) | |||
| 24 | MAB | 4.15 ± 7.40 (92) | P = 0.117* | |
| Monotherapy | 2.43 ± 7.37 (93) | |||
| Total score | 1 | MAB | 0.84 ± 12.37 (94) | P = 0.199* |
| Monotherapy | −1.53 ± 12.97 (96) | |||
| 5 | MAB | 4.16 ± 12.72 (94) | P < 0.001* | |
| Monotherapy | −2.52 ± 13.06 (96) | |||
| 24 | MAB | 4.86 ± 18.44 (94) | P = 0.228* | |
| Monotherapy | 1.67 ± 17.97 (96) |
Positive change denotes improvement of QOL
MAB maximum androgen blockade
* Student’s t-test; † Welch’s t-test (when equality of variance was rejected at P = 0.05)
Bold entries indicate P<0.05
Quality of life improvement in the “emotional well-being” domain was seen in both groups at all time points, with a significantly higher score at week 1 in the MAB group compared with the monotherapy group (P = 0.027). The “physical well-being” domain showed a QOL improvement in the MAB group, and a reduced score in the monotherapy group, but there was no significant difference between the two groups. The “functional well-being” domain showed a significant QOL reduction at week 1 in the MAB (P = 0.049) and monotherapy (P = 0.031) groups (no significant intergroup difference) but returned to the baseline level at week 24 in both groups. A reduction in QOL was seen for “social/family well-being” at all time points in both treatment groups, with no significant differences observed between the two groups.
For the “Prostate cancer-specific issues” domain, significant QOL improvement was seen in the MAB group at weeks 5 and 24 (P < 0.001 at both time points) and in the monotherapy group at week 24 (P = 0.002), with a significantly higher score at week 5 with MAB compared with monotherapy (P = 0.011). Intergroup differences in the changes of domain scores (MAB group versus monotherapy group) are shown in Fig. 2.
Fig. 2.
Intergroup differences in domain score at each time point
The changes in 12-item scores of “prostate cancer-specific issues” are shown in Table 3. Improvement in the scores for micturition disorder (items 9, 10, and 11: urination difficulty, frequent urination, limited activities due to urination trouble) were seen at all time points in the MAB group, with significant improvements compared with the monotherapy group for “frequent urination” at week 24 and “limited activities due to urination trouble” at week 1. The “loss of manly feeling” and “erectile dysfunction” items worsened in both groups, although less QOL reduction for “erectile dysfunction” was seen with MAB than with monotherapy and this difference was statistically significant at weeks 5 and 24. For other items, almost all scores tended to increase in both treatment groups with no significant differences between them.
Table 3.
Changes in individual item scores for the domain “prostate cancer-specific issues”
| Item | Week | Group | Mean ± SD (n) | t-test |
|---|---|---|---|---|
| 1. Weight loss | 1 | MAB | 0.11 ± 0.94 (92) | P = 0.853† |
| Monotherapy | 0.09 ± 0.70 (93) | |||
| 5 | MAB | 0.07 ± 1.10 (92) | P = 0.155* | |
| Monotherapy | 0.28 ± 0.94 (93) | |||
| 24 | MAB | 0.41 ± 1.34 (92) | P = 0.701* | |
| Monotherapy | 0.34 ± 1.09 (93) | |||
| 2. Appetite loss | 1 | MAB | −0.15 ± 1.47 (92) | P = 0.342* |
| Monotherapy | 0.04 ± 1.32 (94) | |||
| 5 | MAB | 0.26 ± 1.52 (92) | P = 0.074* | |
| Monotherapy | −0.13 ± 1.43 (94) | |||
| 24 | MAB | 0.16 ± 1.91 (92) | P = 0.977† | |
| Monotherapy | 0.17 ± 1.46 (94) | |||
| 3. Aches and pains | 1 | MAB | 0.19 ± 1.03 (90) | P = 0.370† |
| Monotherapy | 0.07 ± 0.80 (91) | |||
| 5 | MAB | 0.23 ± 1.36 (90) | P = 0.469† | |
| Monotherapy | 0.11 ± 0.87 (91) | |||
| 24 | MAB | 0.49 ± 1.14 (90) | P = 0.115* | |
| Monotherapy | 0.23 ± 1.04 (91) | |||
| 4. Significant pain | 1 | MAB | 0.20 ± 1.13 (88) | P = 0.951* |
| Monotherapy | 0.19 ± 1.26 (93) | |||
| 5 | MAB | 0.39 ± 1.19 (88) | P = 0.526* | |
| Monotherapy | 0.27 ± 1.29 (93) | |||
| 24 | MAB | 0.55 ± 1.23 (88) | P = 0.176* | |
| Monotherapy | 0.29 ± 1.29 (93) | |||
| 5. Limited activities due to pain | 1 | MAB | 0.06 ± 1.12 (88) | P = 0.509* |
| Monotherapy | −0.04 ± 0.91 (91) | |||
| 5 | MAB | 0.09 ± 1.18 (88) | P = 0.874* | |
| Monotherapy | 0.07 ± 0.90 (91) | |||
| 24 | MAB | 0.27 ± 1.13 (88) | P = 0.298* | |
| Monotherapy | 0.11 ± 0.95 (91) | |||
| 6. Dissatisfactory comfort level | 1 | MAB | −0.16 ± 1.63 (90) | P = 0.162* |
| Monotherapy | 0.16 ± 1.40 (94) | |||
| 5 | MAB | 0.19 ± 1.61 (90) | P = 0.899* | |
| Monotherapy | 0.16 ± 1.50 (94) | |||
| 24 | MAB | 0.14 ± 1.80 (90) | P = 0.195* | |
| Monotherapy | 0.49 ± 1.80 (94) | |||
| 7. Loss of manly feeling | 1 | MAB | −0.44 ± 1.40 (87) | P = 0.322* |
| Monotherapy | −0.22 ± 1.43 (89) | |||
| 5 | MAB | −0.24 ± 1.61 (87) | P = 0.600* | |
| Monotherapy | −0.37 ± 1.65 (89) | |||
| 24 | MAB | −0.44 ± 1.67 (87) | P = 0.313* | |
| Monotherapy | −0.19 ± 1.55 (89) | |||
| 8. Bowel trouble | 1 | MAB | 0.18 ± 1.07 (93) | P = 0.369* |
| Monotherapy | 0.04 ± 1.04 (93) | |||
| 5 | MAB | 0.16 ± 1.14 (93) | P = 0.948* | |
| Monotherapy | 0.17 ± 1.11 (93) | |||
| 24 | MAB | 0.26 ± 1.34 (93) | P = 0.324* | |
| Monotherapy | 0.08 ± 1.17 (93) | |||
| 9. Urination difficulty | 1 | MAB | 0.38 ± 1.06 (89) | P = 0.077* |
| Monotherapy | 0.10 ± 1.01 (87) | |||
| 5 | MAB | 0.87 ± 1.46 (89) | P = 0.221* | |
| Monotherapy | 0.52 ± 1.32 (87) | |||
| 24 | MAB | 1.09 ± 1.50 (89) | P = 0.168* | |
| Monotherapy | 0.78 ± 1.46 (87) | |||
| 10. Frequent urination | 1 | MAB | 0.41 ± 1.16 (91) | P = 0.912* |
| Monotherapy | 0.43 ± 1.30 (89) | |||
| 5 | MAB | 0.69 ± 1.54 (91) | P = 0.063* | |
| Monotherapy | 0.28 ± 1.41 (89) | |||
| 24 | MAB | 0.81 ± 1.71 (91) | P = 0.048* | |
| Monotherapy | 0.34 ± 1.49 (89) | |||
| 11. Limited activities due to urination trouble | 1 | MAB | 0.35 ± 1.20 (88) | P = 0.033* |
| Monotherapy | −0.04 ± 1.26 (91) | |||
| 5 | MAB | 0.41 ± 1.45 (88) | P = 0.175* | |
| Monotherapy | 0.13 ± 1.27 (91) | |||
| 24 | MAB | 0.47 ± 1.57 (88) | P = 0.701† | |
| Monotherapy | 0.38 ± 1.24 (91) | |||
| 12. Erectile dysfunction | 1 | MAB | −0.11 ± 0.91 (84) | P = 0.328† |
| Monotherapy | −0.27 ± 1.19 (82) | |||
| 5 | MAB | −0.01 ± 1.18 (84) | P = 0.016* | |
| Monotherapy | −0.45 ± 1.15 (82) | |||
| 24 | MAB | −0.13 ± 1.13 (84) | P = 0.035* | |
| Monotherapy | −0.51 ± 1.18 (82) |
Positive change denotes improvement of QOL
MAB maximum androgen blockade
* Student’s t-test; † Welch’s t-test (when equality of variance was rejected at p = 0.05)
Bold entries indicate P<0.05
The above data, which were calculated using a LVCF approach, were similar to results obtained without data imputation.
Complete improvement rate of prostate cancer-specific items
Complete improvement rates at week 24 are shown in Table 4. The complete improvement rates of “aches and pains” and “frequent urination” were significantly higher in the MAB group than in the monotherapy group. The complete improvement rates of all other items, with the exception of “dissatisfactory comfort level”, were higher with MAB than with monotherapy, but none were statistically significant.
Table 4.
Complete improvement rate by item of “prostate cancer-specific issues” at week 24
| Item | MAB % (n/N)a | Monotherapy % (n/N) | Difference | Fisher’s exact test |
|---|---|---|---|---|
| 1. Weight loss | 83.9 (26/31) | 76.7 (23/30) | 7.2 | P = 0.534 |
| 2. Appetite loss | 40.8 (20/49) | 35.1 (13/37) | 5.7 | P = 0.658 |
| 3. Aches and pains | 78.1 (25/32) | 51.5 (17/33) | 26.6 | P = 0.038 |
| 4. Significant pain | 62.8 (27/43) | 40.5 (15/37) | 22.3 | P = 0.072 |
| 5. Limited activities due to pain | 73.1 (19/26) | 47.8 (11/23) | 25.3 | P = 0.086 |
| 6. Dissatisfactory comfort level | 12.7 (8/63) | 25.4 (15/59) | −12.7 | P = 0.104 |
| 7. Loss of manly feeling | 18.4 (9/49) | 14.6 (6/41) | 3.8 | P = 0.779 |
| 8. Bowel trouble | 45.2 (19/42) | 44.7 (17/38) | 0.5 | P = 1.000 |
| 9. Urination difficulty | 70.8 (34/48) | 51.0 (26/51) | 19.8 | P = 0.064 |
| 10. Frequent urination | 37.9 (22/58) | 5.2 (3/58) | 32.7 | P < 0.001 |
| 11. Limited activities due to urination trouble | 66.7 (26/39) | 46.9 (15/32) | 19.8 | P = 0.147 |
| 12. Erectile dysfunction | 2.7 (2/75) | 0.0 (0/64) | 2.7 | P = 0.500 |
MAB maximum androgen blockade
aPercentage (number of completely improved subjects at week 24/number of subjects whose quality of life was impaired at baseline)
Bold entries indicate P<0.05
Disease-related factors influencing “prostate cancer-specific issues”
Correlation coefficients between pain-related items 3, 4, and 5 and PSA values were −0.32, −0.23, and −0.28, respectively. The absolute values of all other correlation coefficients were under 0.2, except for “limited activities due to pain” and tumor size (correlation coefficient −0.20); “aches and pains” and percentage change of PSA value (correlation coefficient −0.28); and “limited activities due to urination trouble” and percentage change of tumor size (correlation coefficient −0.27).
Disease flare
From baseline to week 1, significantly fewer patients in the MAB group compared with the monotherapy group had deterioration in their scores for “urination difficulty” (P = 0.023) and “limited activities due to urination trouble” (P = 0.012) (Table 5). Numerical differences in favor of MAB were also noted over this time period for the three pain-related items (numbers 3–5) and “frequent urination”.
Table 5.
Number of patients whose “pain”- and “micturition-disorder”-related scores on the “prostate cancer-specific issues” domain worsened from baseline to week 1
| Item | MAB % (n/N) | Monotherapy % (n/N) | Fisher’s exact test |
|---|---|---|---|
| 3. Aches and pains | 17.3 (14/81) | 19.0 (15/79) | P = 0.839 |
| 4. Significant pain | 17.1 (14/82) | 21.4 (18/84) | P = 0.557 |
| 5. Limited activities due to pain | 12.8 (10/78) | 18.5 (15/81) | P = 0.386 |
| 9. Urination difficulty | 11.3 (9/80) | 26.3 (20/76) | P = 0.023 |
| 10. Frequent urination | 16.7 (14/84) | 23.1 (18/78) | P = 0.330 |
| 11. Limited activities due to urination trouble | 12.0 (10/83) | 28.0 (23/82) | P = 0.012 |
MAB maximum androgen blockade
Bold entries indicate P<0.05
Discussion
Patients with prostate cancer, especially those with early stage disease, are being identified increasingly early thanks to widely available mass screening programs for prostate cancer and improvements in detection techniques available in Japan as well as in other countries. However, despite these improvements, many patients still present with advanced cancer. Endocrine therapy is prescribed for advanced cancer and for elderly patients, but as this therapy must often be administered for long periods, its effect on patients’ QOL must be fully considered (Yoshida et al. 1997). Many reports have highlighted the deterioration of health-related QOL caused by endocrine therapy for prostate cancer (Wagner et al. 2000; Nagakubo et al. 2002; Lee et al. 2001; Clark et al. 2003; Litwin et al. 2002; Efficace et al. 2003) and ADRs due to androgen depletion (Wagner et al. 2000; Lee et al. 2001); decreases in cognitive function, bone mineral density, muscle strength of upper body, sexual function, and overall QOL (Lee et al. 2001) have been reported.
We conducted this Phase III study to directly compare MAB therapy using bicalutamide and an LHRHa versus LHRHa monotherapy in patients with previously untreated advanced prostate cancer (Akaza et al. 2004, 2005). MAB therapy showed significantly superior efficacy to LHRHa monotherapy and comparable safety. QOL was evaluated up to week 24 as a secondary endpoint of this study to comprehensively analyze the effects of MAB and monotherapy on the QOL of patients with prostate cancer. QOL was assessed using a Japanese version of FACT-P, which is a self-reporting patient QOL measurement tool for prostate cancer. It is used widely internationally, and the prostate cancer-specific domain is known to be especially useful for detecting slight changes in QOL. The reliability and validity of FACT-P have been proven in Japanese patients (Hinotsu et al. 1999) and it can measure QOL relatively easily and quickly. The collection rates of FACT-P questionnaires were at least 89% throughout the study period in both treatment groups, which was considered sufficiently high to obtain reliable data.
Regarding the effect of medical treatment on QOL, the total FACT-P score decreased at weeks 1 and 5 in the monotherapy group, whereas it remained higher than baseline at all time points in the MAB group. Overall, the improvement or maintenance of QOL tended to be greater in the MAB group. This trend became further evident as QOL scores were assessed by domain, clearly showing that MAB provided more rapid and greater improvements in QOL for “emotional well-being” and “prostate cancer-specific issues” compared with monotherapy. The score for the item “I am bothered by side effects of treatment” in the “physical well-being” domain did not differ significantly between the two groups (data not shown). Also, there was no difference between the two groups in “percentage of withdrawals due to ADRs”, the primary safety endpoint of this study [MAB group: 9 withdrawals (8.8%); monotherapy group: 11 withdrawals (10.9%); 95% confidence interval: −10.7, 6.4%] (Akaza et al. 2004, 2005). The frequency of ADRs was also comparable in both groups and there was no marked difference in ADR profile between them. The most common adverse events observed in both treatment groups were nasopharyngitis and hot flushes (Akaza et al. 2004; Usami et al. 2007). There was a low incidence of gynecomastia and breast pain, occurring in only three men receiving the combination therapy, which was as expected from previous studies of hormonal combination therapy (Schellhammer et al. 1997; Kaisary 1996). These results suggested that adding bicalutamide to LHRHa therapy would not adversely affect patients’ QOL.
Regarding “prostate cancer-specific issues”, the MAB group showed a significant improvement in “micturition disorder”-related QOL at all time points, achieving a greater improvement than the monotherapy group. Also, the study clearly showed that the complete improvement rates for items related to “pain” and “micturition disorder” were higher in the MAB group than in the monotherapy group. In contrast, no item was significantly superior for monotherapy compared with MAB therapy. According to the report of Da Silva et al., who evaluated QOL in patients with advanced prostate cancer, 70% of the patients experienced pain and psychological stress, 54% had diminished ability to carry out daily work, 36% had urinary-tract abnormality, and 17% had a disturbance of family and social life, with only 25% engaged in active life (Da Silva et al. 1993). Pain and micturition disorder are considered particularly important. As many of the patients included in the present study had clinical stage C/D1 disease without distant metastases, the deterioration of QOL at baseline was largely due to micturition disorder rather than pain, and the score of frequent urination was markedly decreased. The results of this study demonstrated that MAB was particularly effective in improving micturition disorder-related QOL compared with monotherapy.
The QOL satisfaction rating of the domain “social/family well-being” at baseline was low, and a further deterioration of QOL was observed during the treatment period in both treatment groups. Notably, the satisfaction ratings of the items “I am satisfied with my sex life”, “I get support from my friend”, and “I feel close to my friend” were low at 22.4, 30.0, and 45.1%, respectively (data not shown). The satisfaction rating of “I am satisfied with my sex life” further decreased with medical treatment. Regarding “prostate cancer-specific issues”, the scores for the items “loss of manly feeling” and “erectile dysfunction” worsened after treatment in both groups, with a greater deterioration in the monotherapy group. The deterioration of “social/family well-being” may not be attributable to the effect of medical treatment but due to notification and awareness of their cancer. Besides, considering the pharmacological action of LHRHa, erectile dysfunction is unavoidable and difficult to solve medically. Although it is difficult for physicians to include this aspect in routine clinical practice, marked deterioration of QOL cannot be ignored and needs to be resolved by including appropriate professionals in patient care teams.
To determine prostate cancer-related factors that might influence pain and micturition disorder, the correlation between item scores and PSA or tumor size was assessed. Although there was a weak negative correlation for some combinations (correlation coefficients ranging from −0.20 to −0.32), no significant correlation was observed for most combinations (the absolute correlation coefficient was under 0.2). However, this may be because FACT-P is not a specific tool for quantitatively measuring micturition disorders. Therefore, it is necessary to quantify micturition disorder by measuring urine flow or using the International Prostate Symptom Score to quantitatively assess its correlation with disease-related factors.
During the first few weeks of LHRHa treatment, some patients may experience a temporary worsening of symptoms, termed “disease flare”, due to an initial increase in testosterone levels upon commencing therapy. Disease flare, which often manifests as increased bone pain or difficulty passing urine, may have a negative impact on the patient’s QOL. Anti-androgens such as bicalutamide are sometimes given by physicians at the start of LHRHa therapy to modify this effect (Akaza 1999; Sugiono et al. 2005). As there is a paucity of information, available in the literature regarding how disease-flare suppression affects QOL, this was investigated in our study, as the MAB group received anti-flare protection with bicalutamide, compared with no anti-flare protection in the LHRHa monotherapy group. Fewer patients in the MAB group compared with the monotherapy group reported deterioration in their scores for items related to “pain” and “micturition disorder” at week 1. Moreover, regarding the items “urination difficulty” and “limited activities due to urination trouble”, significant differences were observed in favor of MAB. These results suggest that the addition of bicalutamide to LHRHa therapy suppresses disease flare and, consequently, improves QOL.
As mentioned earlier, QOL is affected by many factors other than medical treatment and it may be necessary to investigate the validity of the methodology by thoroughly reviewing the questionnaire and selecting the appropriate domains to detect differences in QOL. We therefore hope that the results of our study can be validated by a future prospective clinical study, taking the above-mentioned points into consideration.
Conclusions
Bicalutamide plus LHRHa combination therapy (MAB) and LHRHa monotherapy did not reduce the overall QOL of patients with previously untreated advanced prostate cancer. MAB was superior to monotherapy in achieving early improvement of QOL related to micturition disorder and pain.
Casodex and Zoladex are trademarks of the AstraZeneca group of companies.
Acknowledgments
We thank all the patients for their participation in this study. We also thank the doctors and support staff in each institute, and Chris Rapier PhD for providing editorial assistance. Financial sponsorship for this trial was provided by AstraZeneca.
Appendix
Question items in the FACT-P questionnaire (version 4A).
| Not at all | A little bit | Some-what | Quite a bit | Very much | |
|---|---|---|---|---|---|
| Physical well-being | |||||
| (1) I have a lack of energy | 0 | 1 | 2 | 3 | 4 |
| (2) I have nausea | 0 | 1 | 2 | 3 | 4 |
| (3) Because of my physical condition, I have trouble meeting the needs of my family | 0 | 1 | 2 | 3 | 4 |
| (4) I have pain | 0 | 1 | 2 | 3 | 4 |
| (5) I am bothered by side effects of treatment | 0 | 1 | 2 | 3 | 4 |
| (6) I feel ill | 0 | 1 | 2 | 3 | 4 |
| (7) I am forced to spend time in bed | 0 | 1 | 2 | 3 | 4 |
| Social/family well-being | |||||
| (1) I feel close to my friends | 0 | 1 | 2 | 3 | 4 |
| (2) I feel close to my familya | 0 | 1 | 2 | 3 | 4 |
| (3) I get emotional support from my family | 0 | 1 | 2 | 3 | 4 |
| (4) I get support from my friends | 0 | 1 | 2 | 3 | 4 |
| (5) My family has accepted my illness | 0 | 1 | 2 | 3 | 4 |
| (6) I am satisfied with family communication about my illness | 0 | 1 | 2 | 3 | 4 |
| (7) Although I am ill, my family has a satisfactory lifea | 0 | 1 | 2 | 3 | 4 |
| (8) I feel close to my partner (or the person who is my main support) | 0 | 1 | 2 | 3 | 4 |
| Regardless of your current level of sexual activity, please answer the following question. If you prefer not to answer it, please check this box □ and go to the next section | |||||
| (9) I am satisfied with my sex life | 0 | 1 | 2 | 3 | 4 |
| Emotional well-being | |||||
| (1) I feel sad | 0 | 1 | 2 | 3 | 4 |
| (2) I am satisfied with how I am coping with my illness | 0 | 1 | 2 | 3 | 4 |
| (3) I am losing hope in the fight against my illness | 0 | 1 | 2 | 3 | 4 |
| (4) I feel nervous | 0 | 1 | 2 | 3 | 4 |
| (5) I worry about dying | 0 | 1 | 2 | 3 | 4 |
| (6) I worry that my condition will get worse | 0 | 1 | 2 | 3 | 4 |
| Functional well-being | |||||
| (1) I am able to work (include work at home) | 0 | 1 | 2 | 3 | 4 |
| (2) My work (include work at home) is fulfilling | 0 | 1 | 2 | 3 | 4 |
| (3) I am able to enjoy life | 0 | 1 | 2 | 3 | 4 |
| (4) I have accepted my illness | 0 | 1 | 2 | 3 | 4 |
| (5) I am sleeping well | 0 | 1 | 2 | 3 | 4 |
| (6) I am enjoying the things I usually do for fun | 0 | 1 | 2 | 3 | 4 |
| (7) I am content with the quality of my life right now | 0 | 1 | 2 | 3 | 4 |
| Additional concernsb | |||||
| (1) I am losing weight | 0 | 1 | 2 | 3 | 4 |
| (2) I have a good appetite | 0 | 1 | 2 | 3 | 4 |
| (3) I have aches and pains that bother me | 0 | 1 | 2 | 3 | 4 |
| (4) I have certain parts of my body where I experience significant pain | 0 | 1 | 2 | 3 | 4 |
| (5) My pain keeps me from doing things I want to do | 0 | 1 | 2 | 3 | 4 |
| (6) I am satisfied with my present comfort level | 0 | 1 | 2 | 3 | 4 |
| (7) I am able to feel like a man | 0 | 1 | 2 | 3 | 4 |
| (8) I have trouble moving my bowels | 0 | 1 | 2 | 3 | 4 |
| (9) I have difficulty urinating | 0 | 1 | 2 | 3 | 4 |
| (10) I urinate more frequently than usual | 0 | 1 | 2 | 3 | 4 |
| (11) My problems with urinating limit my activities | 0 | 1 | 2 | 3 | 4 |
| (12) I am able to have and maintain an erection | 0 | 1 | 2 | 3 | 4 |
Above is a list of statements that other people with your illness have said are important. By circling one (1) number per line, please indicate how true each statement has been for you during the past 7 days. A higher FACT-P score indicated better quality of life. As the FACT-P questionnaire contains positive and negative items, the scores of the negative items (physical well-being items 1–7; emotional well-being items 1 and 3–6; additional concerns items 1, 3–5, 8–11) were reversed as part of the scoring calculation
FACT-P are owned and copyrighted by, and the intellectual property of, David Cella, PhD
aItems 2 and 7 of the social/family well-being domain were specific to the Japanese version of FACT-P
bProstate cancer-specific issues
References
- Akaza H (1999) Prostate cancer therapy and anti-androgens: recent trends. Jpn J Urol Surg 12:623–624 [Google Scholar]
- Akaza H, Yamaguchi A, Matsuda T et al (2004) Superior anti-tumor efficacy of bicalutamide 80 mg in combination with a luteinizing hormone-releasing hormone (LHRH) agonist versus LHRH agonist monotherapy as first-line treatment for advanced prostate cancer: interim results of a randomized study in Japanese patients. Jpn J Clin Oncol 34:20–28 [DOI] [PubMed] [Google Scholar]
- Akaza H, Yoshida H, Takimoto Y et al (2005) Bicalutamide 80 mg in combination with an LHRHa versus LHRHa monotherapy in previously untreated advanced prostate cancer: a double-blind, placebo controlled trial. J Clin Oncol 23:16S [Google Scholar]
- Clark JA, Bokhour BG, Inui TS, Silliman RA, Talcott JA (2003) Measuring patients’ perceptions of the outcomes of treatment for early prostate cancer. Med Care 41:923–936 [DOI] [PubMed] [Google Scholar]
- Da Silva FC, Reis E, Costa T, Denis L (1993) Quality of life in patients with prostatic cancer. A feasibility study. The Members of Quality of Life Committee of the EORTC Genitourinary Group. Cancer 71:1138–1142 [DOI] [PubMed] [Google Scholar]
- Efficace F, Bottomley A, van Andel G (2003) Health related quality of life in prostate carcinoma patients: a systematic review of randomized controlled trials. Cancer 97:377–388 [DOI] [PubMed] [Google Scholar]
- Eisenberger MA, Blumenstein BA, Crawford ED et al (1998) Bilateral orchiectomy with or without flutamide for metastatic prostate cancer. N Engl J Med 339:1036–1042 [DOI] [PubMed] [Google Scholar]
- Esper P, Mo F, Chodak G, Sinner M, Cella D, Pienta KJ (1997) Measuring quality of life in men with prostate cancer using the functional assessment of cancer therapy-prostate instrument. Urology 50:920–928 [DOI] [PubMed] [Google Scholar]
- Hinotsu A, Niimi M, Akaza H et al (1999) Development of Japanese version of QOL questionnaire for bladder and prostate cancer patients using FACT-Bl and P: pilot study. Gan To Kagaku Ryoho 26:657–666 [PubMed] [Google Scholar]
- Kaisary AV (1996) Compliance with hormonal treatment for prostate cancer. Br J Hosp Med 55:359–366 [PubMed] [Google Scholar]
- Klotz L, Schellhammer P, Carroll K (2004) A re-assessment of the role of combined androgen blockade for advanced prostate cancer. BJU Int 93:1177–1182 [DOI] [PubMed] [Google Scholar]
- Lee WR, Hall MC, McQuellon RP, Case LD, McCullough DL (2001) A prospective quality-of-life study in men with clinically localized prostate carcinoma treated with radical prostatectomy, external beam radiotherapy, or interstitial brachytherapy. Int J Radiat Oncol Biol Phys 51:614–623 [DOI] [PubMed] [Google Scholar]
- Litwin MS, Lubeck DP, Spitalny GM, Henning JM, Carroll PR (2002) Mental health in men treated for early stage prostate carcinoma: a posttreatment, longitudinal quality of life analysis from the Cancer of the Prostate Strategic Urologic Research Endeavor. Cancer 95:54–60 [DOI] [PubMed] [Google Scholar]
- McLeod DG, Crawford ED, Blumenstein BA, Eisenberger MA, Dorr FA (1992) Controversies in the treatment of metastatic prostate cancer. Cancer 70:324–328 [DOI] [PubMed] [Google Scholar]
- Nagakubo I, Horiba M, Arima S, Morikawa T, Kato S (2002) Assessment of QOL over time in 146 patients treated with radical prostatectomy. Jpn J Urol Surg 15:1031–1039 [Google Scholar]
- Prostate Cancer Trialists’ Collaborative Group (2000) Maximum androgen blockade in advanced prostate cancer: an overview of the randomised trials. Lancet 355:1491–1498 [PubMed] [Google Scholar]
- Schellhammer PF, Sharifi R, Block NL, Soloway MS, Venner PM, Patterson AL, Sarosdy MF, Vogelzang NJ, Schellenger JJ, Kolvenbag GJ (1997) Clinical benefits of bicalutamide compared with flutamide in combined androgen blockade for patients with advanced prostatic carcinoma: final report of a double-blind, randomized, multicenter trial. Casodex Combination Study Group. Urology 50:330–336 [DOI] [PubMed] [Google Scholar]
- Sugiono M, Winkler MH, Okeke AA, Benney M, Gillatt DA (2005) Bicalutamide vs cyproterone acetate in preventing flare with LHRH analogue therapy for prostate cancer-a pilot study. Prostate Cancer Prostatic Dis 8:91–94 [DOI] [PubMed] [Google Scholar]
- Usami M, Akaza H, Arai Y, Hirano Y, Kagawa S, Kanetake H, Naito S, Sumiyoshi Y, Takimoto Y, Terai A, Yoshida H, Ohashi Y (2007) Bicalutamide 80 mg combined with a luteinizing hormone-releasing hormone agonist (LHRH-A) versus LHRH-A monotherapy in advanced prostate cancer: findings from a phase III randomized, double-blind, multicenter trial in Japanese patients. Prostate Cancer Prostatic Dis 10:194–201 [DOI] [PubMed] [Google Scholar]
- Wagner G, Béjin A, Fugl-Meyer AR et al (2000) Symptom score and quality of life. In: Jardin A, Wagner G, Khoury S, Giuliano F, Padma-Nathan H, Rosen R (eds) Erectile dysfunction. Health Publication Ltd, Plymouth, pp 102–113 [Google Scholar]
- Yoshida M, Ueda S, Machida J et al (1997) Antitumour effect of leuprorelin acetate on prostate cancer and its effect on QOL. Jpn J Urol Surg 10:799–807 [Google Scholar]