Abstract
Over 10 million children in the world have epilepsy, with an unknown cause in half of the cases. The gut microbiome has been associated with various neurological disorders, and certain drugs greatly disturb the microbiome. Our aim was to study the association of prenatal and childhood exposure (before the age of two) to antibiotics, proton pump inhibitors (PPIs) and histamine‐2 receptor antagonists, and the risk of childhood epilepsy. Using population‐based registers, we included all live singleton births in Sweden from 2006 to 2017. Exposure was considered prescription(s) to antibiotics, proton pump inhibitors, or H2‐receptor antagonists (separately). Multivariable Cox regression was used to calculate hazard ratios and 95% confidence intervals. 708,903 mother–child dyads were included, and 0.5% of children had an epilepsy diagnosis. Average follow‐up was 3.8 years (IQR 1–6). Prenatal exposure to antibiotics (aHR 1.09, 95% CI 1.01–1.18) and PPIs (aHR 1.38, 95% CI 1.17–1.65) were associated with an increased risk of epilepsy. Exposure to antibiotics (1.11, 95% CI 1.02–1.21), PPIs (3.40, 95% CI 2.47–4.68) and H2RAs (1.65, 95% CI 1.03–2.64) before the age of two was associated with an increased risk of epilepsy after the age of two. Dose response analysis showed that one prescription of antibiotics in pregnancy or early life was not associated with an increased risk of epilepsy, while one prescription of PPIs in pregnancy or early life had an association. To conclude, our results support the hypothesis that microbiome modulating drugs are associated with an increased risk of epilepsy. This needs to be further validated in other studies, ideally including indications for drug use.
Study Highlights.
WHAT IS THE CURRENT KNOWLEDGE ON THE TOPIC?
Antibiotics and proton pump inhibitors modulate the composition of the gut microbiome, and changes in gut microbiome have been associated with epilepsy.
WHAT QUESTION DID THIS STUDY ADDRESS?
Is exposure to microbiome modulating drugs prenatally or in early life associated with the risk of childhood epilepsy?
WHAT DOES THIS STUDY ADD TO OUR KNOWLEDGE?
Our results suggest an association of antibiotics, proton pump inhibitors prenatally and in early life, in addition to H2RAs in early life, with an increased risk of epilepsy in childhood.
HOW MIGHT THIS CHANGE CLINICAL PHARMACOLOGY OR TRANSLATIONAL SCIENCE?
Exposure to microbiome modulating drugs in vulnerable periods, such as in early life, could have potential long‐term effects and must therefore be researched further and only prescribed when necessary. This study may also help shed light on potential novel mechanisms behind childhood epilepsy, which are still poorly understood.
Epilepsy is one of the most common neurologic disorders, affecting over 50 million people worldwide. Many forms of epilepsy have an onset during childhood, and it is estimated that over 10 million children under the age of 15 have active epilepsy [1]. Known risk factors of epilepsy include genetic mutations, chromosomal abnormalities, malformations of the cerebral cortex, and infections of the central nervous system (CNS) [1]. Despite recent advances in both neuroimaging and genetic testing, which have resulted in an increase in the proportion of epilepsies that can be etiologically resolved, up to 40% of epilepsies remain cryptogenic, that is, of unknown cause [2].
Recent studies have highlighted the association of the gut microbiome, the ecosystem of microbes in the gut, with various neurological disorders, including epilepsy [3, 4]. The microbiome can influence inflammation, vagal neuronal activity, hippocampal neurotransmitters, amino acid metabolism, and neuroplasticity [3, 5]. Neuroplasticity and nervous system maturation coincide with maturation of the gut microbiome during the first 2 years of life, where disturbances may have long‐term effects [5, 6, 7].
The gut microbiome can be greatly disturbed by certain drugs, such as antibiotics and proton pump inhibitors (PPIs) [8, 9]. A recent meta‐analysis showed that one in four pregnant women worldwide use antibiotics [10]. Studies have reported an increased risk in symptomatic seizures after antibiotic exposure [3, 11, 12, 13], while others have investigated antibiotics as a potential treatment for epilepsy [3]. Studies on prenatal exposure to antibiotics and the risk of epilepsy, which have a variety of age ranges for the children, sample sizes, and follow‐up time, and mostly originate in Europe and the United States of America, have shown conflicting results [14, 15, 16, 17]. The underlying indication for antibiotics may also affect the risk (including bacterial infections and misdiagnosed viral infections). PPIs and H2‐receptor antagonists (H2RAs) are often used to combat heartburn and other gastric acid‐related diseases [18], indications which are not known to affect neurodevelopment. PPIs were prescribed in approximately 3.8% of pregnancies in Sweden from 2013 to 2017 [19]. Very few studies have looked at the relationship between PPIs or other acid‐suppressive drugs and epilepsy, but prolonged use has been shown to increase the risk of epilepsy in adults [20]. H2RAs are prescribed for similar indications as PPIs but have been shown to be less disruptive to the microbiome [21].
Prenatal and childhood exposure to microbiome modulating drugs and the long‐term effect in children remain understudied. Our aim was to study the association between prenatal and early life exposure to antibiotics, PPIs, and H2RAs and the risk of developing epilepsy in childhood.
MATERIALS AND METHODS
Study design and period
In this population‐based register study with prospectively collected nationwide data, we included all live born singleton infants included in the Swedish Medical Birth Register [22] (established in 1973) from 2006 to 2017. By means of the unique personal identification number, the Medical Birth Register was further linked to other national health registers including the Swedish Patient Register (full nationwide coverage since 1987) [23], the Swedish Cause of Death Register (established 1952) [24] and the Swedish Prescribed Drug Register (established in 2005) [25]. This study was performed in accordance with the Declaration of Helsinki, with ethical permit (2017/2423‐31) from the Swedish Research Council. Informed consent was not required. No data was available on gender, but adult participants are considered as women/mothers in this article.
In‐ and exclusion criteria
Only liveborn singleton births were included in the analysis. Children with diagnoses (cerebral infections, intracerebral hemorrhage, ischemic stroke, other disturbances of cerebral status of newborn, other disorders of brain in diseases classified elsewhere, intrauterine hypoxia, birth asphyxia, Rett syndrome, Struge‐ Weber's syndrome, tuberous sclerosis, cerebral palsy, hydrocephalus, congenital malformations of nervous system, chromosomal malformations and syndromes, malignant neoplasm of brain and convulsions of newborn) inferring a highly elevated risk of epilepsy (see Table S1 ) were excluded at baseline. Children with cerebral infections, intracerebral hemorrhage, or ischemic stroke diagnoses before their epilepsy diagnosis (ICD‐10: G40) were censored if diagnosed before receiving an epilepsy diagnosis. Additionally, children with more than one subtype of epilepsy (to avoid multiple subtype diagnosis and overlapping subgroups) or diagnosed epilepsy in the birth registry (only contains data up to 28 days after birth) were excluded. A full list of exclusion and censoring criteria can be found in Table S1 and Figure S1 .
For women who had multiple children within the time frame, only the first pregnancy was used for analysis.
Exposure
The study exposure, derived from the Swedish Prescribed Drug Register, was defined as at least one dispensed prescription of systemic antibiotics, PPIs, or histamine‐2 receptor antagonist (H2RA) during pregnancy or early life (under the age of 2 years). H2RA were included as they are prescribed for similar indications as PPIs but have different mechanisms of action and are less disruptive of the gut microbiome [21]. In analyses, those without a prescription for the drug of interest were used as references.
Exposure was both estimated prenatally (from last menstrual period to date of birth, grouped into trimesters) and during the first 2 years of life (from birth up to 2nd birthday).
The following World Health Organization (WHO) Anatomical Therapeutic Chemical (ATC) classifications were included [26]: Antibacterials for systemic use (J01) and eight classes (J01A: Tetracyclines, J01C: Beta‐lactam antibacterials, Penicillins, J01D: Other beta‐lactam antibacterials, J01E: Sulfonamides and Trimethoprim, J01F: Macrolides, Lincosamides and Streptogramins, J01G: Aminoglycosides antibacterials, J01M: Quinolone antibacterials and J01X: Other antibacterials) and Drugs for peptic ulcer, dyspepsia and GORD and other gastric acid‐related disorders (A02BC proton pump inhibitors and A02BA H2‐receptor antagonists). Furthermore, metronidazole (P01AB01) was merged with J01X [26].
Number of prescriptions and trimester of prescription were recorded for maternal consumption, and number of prescriptions for early life exposure. Information on over‐the‐counter drugs was not available, yet only PPIs and H2RA are available over the counter in Sweden at a higher price.
Outcomes
The main outcome was first diagnosis of epilepsy using the Patient Register and Child Health Services Registry with ICD‐10 code G40 (Epilepsy and recurrent seizures), including subtypes or individual diagnoses as power allows [27]. Epilepsy subtypes were grouped as follows: focal (G400, G401 and G402), generalized (G403, G404, G405 and G407), tonic–clonic seizures (G406) and other (G408 and G409). Children with more than one subtype group were removed from the cohort, except for children with group “other” in combination with one of the other groups where the latter was considered the correct epilepsy subtype.
Outcome was recorded for (1) from birth to the end of the study regarding prenatal exposure, and (2) from the second birthday to the end of the study for early life exposure.
Covariates
The last menstrual period (LMP) date was calculated by extracting the gestational age (as dated at ultrasound, from embryo transfer or last menstrual period) from the date of birth from the Swedish Medical Birth Register. As the date of birth was provided in year/month format due to privacy regulations, it was set as the 15th of each month in all the observations. Maternal exposure was defined as first trimester (LMP 0–97), second trimester (LMP 98–202) and third trimester (LMP 203—delivery). Early childhood exposure was defined as at least one prescription between birth and the second birthday.
Other extracted covariates from the Swedish Medical Birth Register were: (1) maternal factors (age at childbirth (as groups: ≤25, 25–29, 30–34, 35–39, ≥40 years), country of birth (Nordic or non‐Nordic), body mass index (BMI, as groups: <20.0, 20.0–24.9, 25.0–29.9, ≥30.0 kg/m2), parity, use of artificial reproductive technologies (ART) and epilepsy diagnosis (self‐reported), (2) maternal lifestyle factors (any tobacco consumption during pregnancy (smoking and smokeless tobacco)), (3) common maternal chronic comorbidities (hypertension, diabetes, hypo‐ and hyperthyroidism, self‐reported), and (4) child‐related factors (sex of the child), small (<10th percentile)/appropriate/large (over 90th percentile) for gestational age, delivery mode (vaginal, elective‐ or acute c‐section), Apgar score at 5 minutes (lower or higher than 7) and gestational age (preterm (birth before 37 completed gestational weeks) or not).
Statistical analysis
The risk of epilepsy after prenatal or early life (before the second birthday) exposure to antibiotics, PPIs, and H2RA was calculated using multivariable Cox regression models. All children were followed up until first epilepsy diagnosis (using date of admission), the end of the study (December 2018) or death, whichever came first. First, univariable analysis was performed on the above‐mentioned covariates to identify potential confounders. The multivariable models were adjusted for said confounders if P < 0.10 in the univariable analyses, with results presented as adjusted hazard ratios (aHR) with 95% confidence intervals (CI).
Models were reported as unadjusted, reduced (only adjusting for confounders relating to the mother when the exposure was on the mother and not the child, so all variables clearly precede the outcome to limit reverse causation) and fully adjusted (all covariates identified in the univariable analysis).
Likelihood ratio tests were performed to assess the interaction between prenatal exposure to antibiotics and PPIs, and the interaction between early life antibiotics and PPIs. Unadjusted cumulative incidence curves were produced for prenatal and early life exposure to antibiotics, PPIs, and H2RAs.
Sub‐analyses were performed for the following: association of exposure and epilepsy subtype group (with no epilepsy diagnosis as reference), antibiotic subclass and epilepsy risk, maternal diagnosis of epilepsy or not (to minimize potential genetic effects) and epilepsy risk, pregnancy trimester of medication intake (with no exposure in the respective trimester as reference) and epilepsy risk, and age of epilepsy diagnosis (before / after the age of two). When analyzing drug exposure before the age of two, only diagnosis of epilepsy after the age of two was considered.
Absence of reporting of the respective ICD codes was considered absence of the disease/disorder. If a substantial group of individuals had missingness on a certain variable (e.g., BMI was missing in 7% in this cohort), a dummy variable (coded as “Missing”) was created to keep the individuals in the models.
RStudio (version 1.3.1093) [28] was used for all statistical analyses [29, 30, 31, 32, 33, 34]. Figures were made using ggplot2 [35].
RESULTS
A total of 708,903 mother–child dyads were included in the cohort (Figure S1 ). Most mothers were older than 30 (62.7%), normal weight (49.1%) and nulliparous (66.9%) (Table 1 ).
Table 1.
Description of the cohort, stratified by exposure
| Total | Maternal exposure during pregnancy | Child exposure during early life | |||||
|---|---|---|---|---|---|---|---|
| PPI | H2RA | Antibiotics | PPI | H2RA | Antibiotics | ||
| (N = 708,903) | (N = 23,386) | (N = 1,059) | (N = 157,409) | (N = 3,423) | (N = 1979) | (N = 214,388) | |
| Epilepsy diagnosis (any age) | 3,404 (0.5%) | 140 (0.6%) | 10 (0.9%) | 860 (0.5%) | |||
| Epilepsy diagnosis after the age of 2 | 2,404 (0.3%) | 45 (1.3%) | 20 (1.0%) | 971 (0.5%) | |||
| Maternal age in years | |||||||
| <25 | 122,730 (17.3%) | 4,050 (17.3%) | 197 (18.6%) | 31,112 (19.8%) | 523 (15.3%) | 343 (17.3%) | 37,291 (17.4%) |
| 25–29 | 141,327 (19.9%) | 5,782 (24.7%) | 286 (27.0%) | 32,676 (20.8%) | 739 (21.6%) | 587 (29.7%) | 43,350 (20.2%) |
| 30–34 | 219,984 (31.0%) | 6,629 (28.3%) | 331 (31.3%) | 45,573 (29.0%) | 987 (28.8%) | 639 (32.3%) | 64,279 (30.0%) |
| >34 | 224,862 (31.7%) | 6,925 (29.6%) | 245 (23.1%) | 48,048 (30.5%) | 1,174 (34.3%) | 410 (20.7%) | 69,468 (32.4%) |
| Maternal BMI | |||||||
| Underweight | 71,141 (10.0%) | 2,222 (9.5%) | 105 (9.9%) | 15,669 (10.0%) | 356 (10.4%) | 208 (10.5%) | 19,986 (9.3%) |
| Normal weight | 348,222 (49.1%) | 9,508 (40.7%) | 426 (40.2%) | 73,445 (46.7%) | 1,678 (49.0%) | 954 (48.2%) | 102,106 (47.6%) |
| Overweight | 161,141 (22.7%) | 6,023 (25.8%) | 256 (24.2%) | 36,531 (23.2%) | 699 (20.4%) | 411 (20.8%) | 50,398 (23.5%) |
| Obese | 77,353 (10.9%) | 3,947 (16.9%) | 173 (16.3%) | 20,057 (12.7%) | 377 (11.0%) | 220 (11.1%) | 25,475 (11.9%) |
| Missing | 51,046 (7.2%) | 1,686 (7.2%) | 99 (9.3%) | 11,707 (7.4%) | 313 (9.1%) | 186 (9.4%) | 16,423 (7.7%) |
| Parity | |||||||
| Multiparous | 234,993 (33.1%) | 8,058 (34.5%) | 428 (40.4%) | 60,650 (38.5%) | 995 (29.1%) | 650 (32.8%) | 92,556 (43.2%) |
| Nulliparous | 473,910 (66.9%) | 15,328 (65.5%) | 631 (59.6%) | 96,759 (61.5%) | 2,428 (70.9%) | 1,329 (67.2%) | 121,832 (56.8%) |
| Nordic country of birth | 539,675 (76.1%) | 14,790 (63.2%) | 657 (62.0%) | 119,616 (76.0%) | 2,685 (78.4%) | 1,582 (79.9%) | 161,031 (75.1%) |
| Assisted conception | 25,595 (3.6%) | 1,066 (4.6%) | 38 (3.6%) | 5,497 (3.5%) | 181 (5.3%) | 100 (5.1%) | 6,731 (3.1%) |
| Delivery mode | |||||||
| Elective c‐section | 46,216 (6.5%) | 2,178 (9.3%) | 96 (9.1%) | 12,103 (7.7%) | 384 (11.2%) | 206 (10.4%) | 15,658 (7.3%) |
| Acute c‐section | 73,095 (10.3%) | 3,058 (13.1%) | 143 (13.5%) | 17,710 (11.3%) | 593 (17.3%) | 336 (17.0%) | 21,694 (10.1%) |
| Vaginal birth | 589,592 (83.2%) | 18,150 (77.6%) | 820 (77.4%) | 127,596 (81.1%) | 2,446 (71.5%) | 1,437 (72.6%) | 177,036 (82.6%) |
| Sex of child | |||||||
| Boy | 364,144 (51.4%) | 11,982 (51.2%) | 517 (48.8%) | 81,117 (51.5%) | 1978 (57.8%) | 1,106 (55.9%) | 117,524 (54.8%) |
| Girl | 344,759 (48.6%) | 11,404 (48.8%) | 542 (51.2%) | 76,292 (48.5%) | 1,445 (42.2%) | 873 (44.1%) | 96,864 (45.2%) |
| Preterm | 34,996 (4.9%) | 1,462 (6.3%) | 61 (5.8%) | 8,517 (5.4%) | 530 (15.5%) | 315 (15.9%) | 10,675 (5.0%) |
| Tobacco use | 51,827 (7.3%) | 2018 (8.6%) | 107 (10.1%) | 15,072 (9.6%) | 231 (6.7%) | 158 (8.0%) | 18,363 (8.6%) |
| Maternal epilepsy | 3,708 (0.5%) | 162 (0.7%) | 6 (0.6%) | 1,011 (0.6%) | 22 (0.6%) | 13 (0.7%) | 1,240 (0.6%) |
| Maternal hypertension | 3,380 (0.5%) | 203 (0.9%) | 11 (1.0%) | 916 (0.6%) | 20 (0.6%) | 21 (1.1%) | 1,138 (0.5%) |
| Maternal diabetes mellitus | 5,814 (0.8%) | 425 (1.8%) | 20 (1.9%) | 1992 (1.3%) | 39 (1.1%) | 21 (1.1%) | 1901 (0.9%) |
| Maternal hypothyroidism | 19,354 (2.7%) | 1,165 (5.0%) | 47 (4.4%) | 5,211 (3.3%) | 159 (4.6%) | 78 (3.9%) | 5,581 (2.6%) |
| Maternal hyperthyroidism | 3,837 (0.5%) | 185 (0.8%) | 13 (1.2%) | 975 (0.6%) | 23 (0.7%) | 14 (0.7%) | 1,232 (0.6%) |
| Apgar <7 at 5 minutes | 4,968 (0.7%) | 219 (0.9%) | 14 (1.3%) | 1,241 (0.8%) | 94 (2.7%) | 39 (2.0%) | 1,370 (0.6%) |
| Large for gestational age | 19,466 (2.7%) | 708 (3.0%) | 42 (4.0%) | 5,200 (3.3%) | 104 (3.0%) | 50 (2.5%) | 6,851 (3.2%) |
| Small for gestational age | 18,974 (2.7%) | 887 (3.8%) | 29 (2.7%) | 4,149 (2.6%) | 264 (7.7%) | 117 (5.9%) | 5,380 (2.5%) |
BMI, Body mass index; c‐section, Caesarean section; PPI, Proton pump inhibitors.
Out of the total cohort, 22.2% of women received antibiotics, 3.3% PPIs, and 0.1% H2RAs during their pregnancy. Regarding early life exposure, for children having reached the age of two, 34.3% of the children received antibiotics before their second birthday, 0.5% PPIs, and 0.3% H2RAs. Average follow‐up time was 3.8 years (IQR 1–6).
Epilepsy diagnosis
In total, 3,404 (0.5%) children were diagnosed with epilepsy, with the majority being diagnosed after the age of two (2,404, 0.3%). Children with epilepsy were more frequently born prematurely (6.8%) and were more likely to have a mother with epilepsy (2%) compared to children without epilepsy (4.9% and 0.5%, respectively) (Table 1 ).
Prenatal exposure and overall risk of epilepsy
The following factors were associated with an increased risk of epilepsy (P < 0.10) in a univariable model for the total cohort and were therefore included in a multivariable model: maternal age, BMI, parity, country of birth, delivery mode, sex of child, preterm birth status, tobacco use during pregnancy, maternal epilepsy, maternal diabetes mellitus, maternal hyperthyroidism, Apgar score at 5 minutes, and being small for gestational age (Table S2 ).
The adjusted multivariable Cox proportional hazard model showed that both prenatal exposure to antibiotics (aHR 1.09, 95% CI 1.01–1.18) and PPIs (aHR 1.38, 95% CI 1.17–1.65) were associated with an increased risk of epilepsy (Table 2 ). Furthermore, in the multivariable model, mother's BMI (overweight; aHR 1.14, 95% CI 1.04–1.24, obese; aHR 1.27, 95% CI 1.14–1.42), Nordic birth country (aHR 1.09, 95% CI 1.01–1.19) preterm birth (aHR 1.26, 95% CI 1.10–1.45), maternal epilepsy (aHR 3.65, 95% CI 2.87–4.64) and being small for gestational age (aHR 1.61, 95% CI 1.36–1.91) were associated with an increased risk of epilepsy, while girls presented with a lower risk than boys (aHR 0.92, 95% CI 0.86–0.99) (Figure 1 ). A reduced model, including only covariates related to the mother and not birth, showed similar results to the fully adjusted model (Table 2 ).
Table 2.
Multivariable Cox hazard ratios (aHR) with 95% confidence intervals (CI) for epilepsy at any age, or epilepsy after the age of two. Univariable models, reduced models for prenatal exposure and fully adjusted models
| Overall risk | Risk after the age of 2 years | |||||||
|---|---|---|---|---|---|---|---|---|
| HR univariable | aHR reduceda | aHR fully adjustedb | P | HR univariable | aHR reducedc | aHR fully adjustedd | P | |
| Maternal prenatal exposure | ||||||||
| PPI | 1.46 (1.23–1.73) | 1.41 (1.19–1.67) | 1.38 (1.17–1.65) | 0.0002 | 1.37 (1.11–1.70) | 1.31 (1.06–1.62) | 1.25 (1.01–1.55) | 0.04 |
| Any antibiotics | 1.12 (1.04–1.22) | 1.10 (1.02–1.19) | 1.09 (1.01–1.18) | 0.03 | 1.11 (1.01–1.22) | 1.09 (0.99–1.20) | 1.07 (0.97–1.17) | 0.17 |
| Interaction antibiotics × PPIs | 1.10 (0.78–1.56) | 0.58 | 0.93 (0.60–1.46) | 0.77 | ||||
| PPIs: Only mothers with epilepsy diagnosise | 1.89 (0.68–5.31) | 0.22 | ||||||
| PPIs: Only mothers without epilepsy diagnosise | 1.39 (1.17–1.67) | 0.0002 | ||||||
| Antibiotics: Only mothers with epilepsy diagnosise | 1.02 (0.60–1.72) | 0.95 | ||||||
| Antibiotics: Only mothers without epilepsy diagnosise | 1.10 (1.02–1.19) | 0.02 | ||||||
| Child early life exposure | ||||||||
| PPI | 4.17 (3.10–5.60) | 3.40 (2.47–4.68) | <0.0001 | |||||
| H2RA | 2.76 (1.78–4.28) | 1.65 (1.03–2.64) | 0.04 | |||||
| Any antibiotics | 1.13 (1.04–1.22) | 1.11 (1.02–1.21) | 0.01 | |||||
| Interaction antibiotics × PPIs | 1.41 (0.75–2.68) | 0.29 | ||||||
| PPIs: Only mothers with epilepsy diagnosisf | 4.34 (0.59–32.14) | 0.75 | ||||||
| PPIs: Only mothers without epilepsy diagnosisf | 3.80 (2.81–5.15) | <0.0001 | ||||||
| Antibiotics: Only mothers with epilepsy diagnosisf | 1.13 (0.64–1.97) | 0.68 | ||||||
| Antibiotics: Only mothers without epilepsy diagnosisf | 1.12 (1.03–1.22) | 0.01 | ||||||
All significant levels have now been formatted as bold.
Adjusted for: maternal age, maternal BMI, parity, country of birth, sex of child, tobacco usage, maternal epilepsy, maternal diabetes, maternal hyperthyroidism.
Adjusted for: maternal age, maternal BMI, parity, country of birth, delivery mode, sex of child, preterm status, tobacco usage, maternal epilepsy, maternal diabetes, maternal hyperthyroidism, Apgar score at 5 minutes, and small for gestational age.
Adjusted for: maternal age, maternal BMI, parity, sex of child, tobacco usage, maternal epilepsy, maternal diabetes.
Adjusted for: maternal age, maternal BMI, parity, delivery mode, sex of child, preterm status, tobacco usage, maternal epilepsy, maternal diabetes, Apgar score at 5 minutes, small for gestational age.
Adjusted for: maternal age, maternal BMI, parity, country of birth, delivery mode, sex of child, preterm status, tobacco usage, maternal diabetes, maternal hyperthyroidism, Apgar score at 5 minutes, small for gestational age.
Adjusted for: maternal age, maternal BMI, parity, delivery mode, sex of child, preterm status, tobacco usage, maternal diabetes, Apgar score at 5 minutes, small for gestational age.
Figure 1.
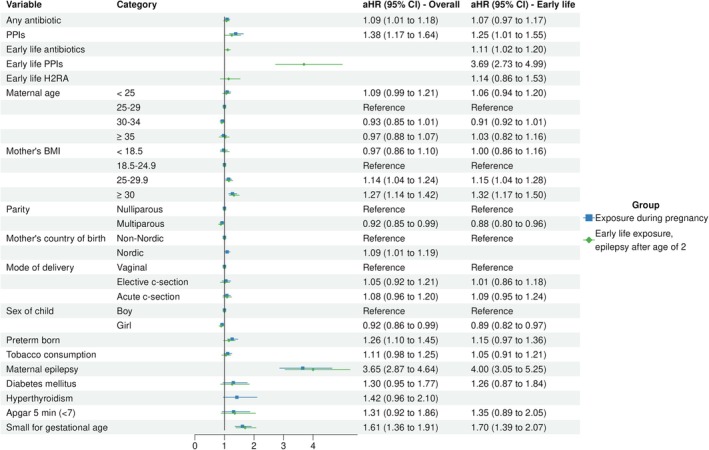
Results from multivariable Cox regression, showing adjusted hazard ratios (aHR) with 95% confidence intervals (CI).
When only looking at mothers that did not have epilepsy, both PPIs (aHR 1.89, 95% CI 0.68–5.31 (maternal epilepsy) vs. aHR 1.39, 95% CI 1.17–1.67 (no maternal epilepsy)) and antibiotics (aHR 1.02, 95% CI 0.60–1.72 (maternal epilepsy) vs. aHR 1.10, 95% CI 1.02–1.19 (no maternal epilepsy)) exposure prenatally were significantly associated with an increased epilepsy risk in the children (Table 2 ). This association was not seen in mothers that had an epilepsy diagnosis (Table 2 ).
Cumulative incidence curves showed an increased incidence of epilepsy for those exposed to antibiotics (Figure 2 a ), PPIs (Figure 2 b ) or H2RAs (Figure 2 c ) compared to unexposed.
Figure 2.
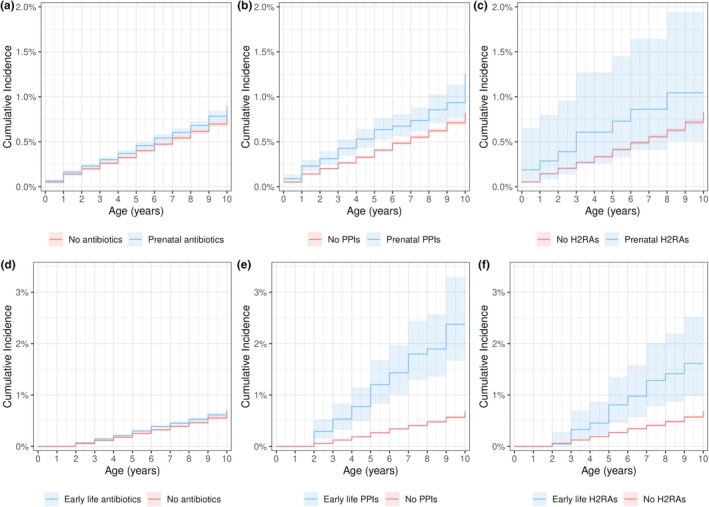
Unadjusted cumulative incidence for prenatal (a) antibiotics, (b) proton pump inhibitors (PPIs) and (c) H2RAs and the risk of epilepsy at any time during the study period. Early life (d) antibiotics, (e) PPIs and (f) H2RAs and the incidence of epilepsy after the age of two.
No interaction was found between prenatal exposure to antibiotics and PPIs (P= 0.58).
Due to the low number of individuals with PPI and H2RA prescriptions, subgroup analysis was not feasible.
Antibiotics exposure by trimester
Out of the three trimesters, exposure to antibiotics in the second trimester was associated with the highest risk of epilepsy (aHR 1.20, 95% CI 1.08–1.33), followed by the first trimester (aHR 1.18, 95% CI 1.05–1.31), with the reference group being women not exposed to antibiotics in the respective trimester. Antibiotics exposure in the third trimester showed no association to an increased risk of epilepsy (Table 3 ).
Table 3.
Multivariable Cox hazard ratios (aHR) with 95% confidence intervals (CI) for antibiotics exposure during pregnancy or early life, by antibiotic subtype or trimester of exposure
| Antibiotics during pregnancy – diagnosis at any agea | Antibiotics <2 years – diagnosis >2 yearsb | |||||
|---|---|---|---|---|---|---|
| Epilepsy diagnosis | aHR, 95% CI | P | Epilepsy diagnosis | aHR, 95% CI | P | |
| (N = 3,462) | (N = 2,455) | |||||
| J01A tetracyclines | 25 (0.7%) | 1.01 (0.68–1.52) | 0.948 | 0 (0%) | NA | NA |
| J01C beta‐lactam antibacterials, Penicillins | 603 (17.7%) | 1.06 (0.97–1.16) | 0.173 | 890 (37.0%) | 0.99 (0.79–1.25) | 0.94 |
| J01D other beta‐lactam antibacterials | 130 (3.8%) | 1.23 (1.03–1.47) | 0.021 | 111 (4.6%) | 1.21 (0.99–1.48) | 0.06 |
| J01E sulfonamides | 16 (0.5%) | 1.05 (0.64–1.71) | 0.850 | 89 (3.7%) | 1.29 (1.04–1.61) | 0.02 |
| J01F macrolides | 54 (1.6%) | 1.56 (1.18–2.05) | 0.002 | 75 (3.1%) | 1.02 (0.81–1.29) | 0.87 |
| J01G aminoglycoside | 1 (0.0%) | NA | NA | 0 (0%) | NA | NA |
| J01M quinolone antibacterials | 13 (0.4%) | NA | NA | 2 (0.1%) | NA | NA |
| J01X other antibacterials and P01AB metronidazole | 256 (7.5%) | 1.20 (1.06–1.37) | 0.005 | 0 (0%) | NA | NA |
| Any antibiotics 1st trimester | 368 (10.8%) | 1.18 (1.05–1.31) | 0.004 | |||
| Any antibiotics 2nd trimester | 408 (12.0%) | 1.20 (1.08–1.33) | 0.0006 | |||
| Any antibiotics 3rd trimester | 315 (9.3%) | 1.06 (0.94–1.19) | 0.370 | |||
All significant levels have now been formatted as bold.
Adjusted for: maternal age, maternal BMI, parity, country of birth, delivery mode, sex of child, preterm status, tobacco usage, maternal epilepsy, maternal diabetes, maternal hyperthyroidism, Apgar score at 5 minutes, and small for gestational age.
Adjusted for: maternal age, maternal BMI, parity, delivery mode, sex of child, preterm status, tobacco usage, maternal epilepsy, maternal diabetes, Apgar score at 5 minutes, and small for gestational age.
Exposure rates by antibiotic class was similar across trimesters, except for Tetracyclines which was highest in the first trimester (5.9%) and lower in the other two (second trimester 0.6%, third trimester 0.4%), and “Other beta‐lactam antibacterials” which were lower in the first (6.2%) and second (8.9%) compared to the third (14.8%) (Figure S2 ).
Antibiotics subgroups and risk of epilepsy
Out of the eight subgroups of antibiotics, three showed an association with an increased risk of epilepsy: “Other beta‐lactam antibacterials” (J01D) (aHR 1.23, 95% CI 1.03–1.47), “Macrolides” (J01F) (aHR 1.56, 95% CI 1.18–2.05), and “Other antibacterials and Metronidazole” (J01X, P01AB) (aHR 1.20, 95% CI 1.06–1.37) (Table 3 ).
Dose response analysis
An increased number of antibiotic prescriptions during pregnancy was associated with an increased risk of epilepsy, rising from one prescription (aHR 1.02, 95% CI 0.93–1.12) to more than three prescriptions (aHR 1.57, 95% CI 1.21–2.04) (Figure 3 , top left). No association was found with a single prescription of antibiotics during pregnancy and the risk of epilepsy. Regarding PPIs, a single prescription increased the risk for epilepsy significantly (aHR 1.44, 95% CI 1.18–1.77) (Figure 3 , top right). The increased risk was not significant for multiple prescriptions, possibly due to lower power as few women had multiple prescriptions.
Figure 3.
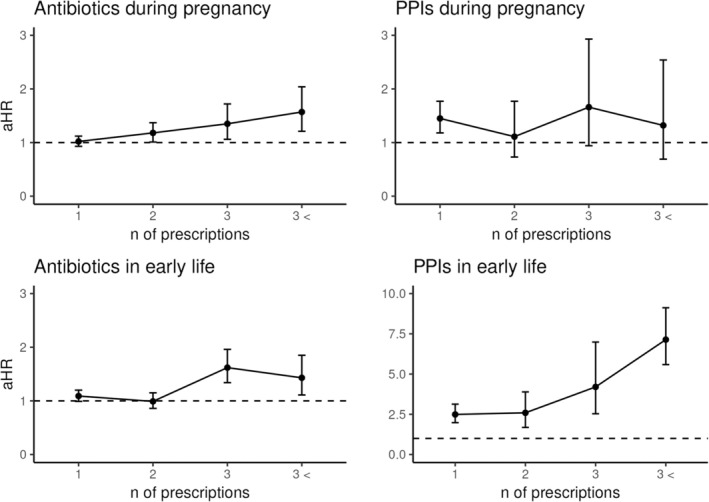
Dose response analysis for antibiotics and proton pump inhibitors (PPIs), both during pregnancy and in early life (before the age of two). Hazard ratios (aHR) and 95% confidence intervals (CI) calculated using multivariable Cox regression adjusted for in pregnancy exposure (maternal age, maternal BMI, parity, country of birth, delivery mode, sex of child, preterm status, tobacco usage, maternal epilepsy, maternal diabetes, maternal hyperthyroidism, Apgar score at 5 minutes, small for gestational age) and early life exposure (maternal age, maternal BMI, parity, delivery mode, sex of child, preterm status, tobacco usage, maternal epilepsy, maternal diabetes, Apgar score at 5 minutes, small for gestational age).
Antibiotics and epilepsy subgroups
Out of the four subgroups of epilepsy, prenatal exposure to antibiotics only showed an association with “Other epilepsy” (G408 (Other epilepsy and recurrent seizures) and G409 (Epilepsy, unspecified), n = 1,555 (46% of all diagnoses)) (aHR 1.22, 95% CI 1.09–1.36) (Table 4 ). Regarding specific individual ICD‐10 codes, only G409 (Epilepsy, unspecified) was associated with an increased risk of epilepsy (aHR 1.15, 95% CI 1.04–1.27) (Table 4 ).
Table 4.
Adjusted multivariable Cox hazard ratios (aHR) with 95% confidence intervals (CI) for antibiotics exposure in pregnancy or early life, by epilepsy diagnosis and diagnosis group
| Group | ICD‐10 code | Everyone (exposure: antibiotics during pregnancy)a | Over 2 (exposure: antibiotics before the age of 2 years)b | ||||||
|---|---|---|---|---|---|---|---|---|---|
| n | % | HR (95% CI) | P | n | % | HR (95% CI) | P | ||
| Focal | G400, G401, G402 | 897 | 26.4 | 1.02 (0.87–1.19) | 0.80 | 571 | 23.8 | 1.11 (0.95–1.29) | 0.19 |
| G400 | 392 | 11.5 | 1.02 (0.81–1.29) | 0.86 | 357 | 14.9 | 0.86 (0.69–1.07) | 0.17 | |
| G401 | 98 | 2.9 | 1.00 (0.62–1.60) | 0.99 | 71 | 3.0 | 1.09 (0.67–1.76) | 0.74 | |
| G402 | 450 | 13.2 | 1.10 (0.89–1.37) | 0.37 | 319 | 13.3 | 1.44 (1.15–1.81) | 0.001 | |
| Generalized | G403, G404, G405, G407 | 930 | 27.3 | 1.00 (0.86–1.17) | 0.18 | 713 | 29.7 | 1.04 (0.87–1.23) | 0.67 |
| G403 | 571 | 16.8 | 0.97 (0.79–1.18) | 0.75 | 420 | 17.5 | 1.00 (0.82–1.22) | 0.98 | |
| G404 | 238 | 7.0 | 0.82 (0.60–1.14) | 0.24 | 75 | 3.1 | 1.03 (0.64–1.66) | 0.89 | |
| G407 | 135 | 4.0 | 1.27 (0.87–1.86) | 0.22 | 92 | 3.8 | 1.01 (0.65–1.55) | 0.98 | |
| Grand mal | G406 | 22 | 0.6 | 0.89 (0.33–2.46) | 0.83 | 14 | 0.6 | 1.07 (0.35–3.32) | 0.90 |
| Other | G408, G409 (and no other) | 1,555 | 45.7 | 1.22 (1.09–1.36) | 0.00 | 1,106 | 46.0 | 1.18 (1.05–1.34) | 0.006 |
| G409 (can be in combination with other diagnoses) | 1946 | 57.2 | 1.15 (1.04–1.27) | 0.01 | 1,390 | 57.8 | 1.15 (1.03–1.28) | 0.01 | |
All significant levels have now been formatted as bold.
Adjusted for: maternal age, maternal BvMI, parity, country of birth, delivery mode, sex of child, preterm status, tobacco usage, maternal epilepsy, maternal diabetes, maternal hyperthyroidism, Apgar score at 5 minutes, small for gestational age. Reference group: no epilepsy diagnosis.
Adjusted for: maternal age, maternal BMI, parity, delivery mode, sex of child, preterm status, tobacco usage, maternal epilepsy, maternal diabetes, Apgar score at 5 minutes, small for gestational age. Reference groups: no epilepsy diagnosis.
Early life exposure and epilepsy after the age of two
In a univariable model, the same factors were associated with an increased risk of epilepsy after the age of two (P < 0.10) as overall epilepsy above, except for country of birth and hyperthyroidism, and the addition of early life H2RA exposure (Table S2 ).
An adjusted multivariable Cox proportional hazard model showed an association with an increased risk of epilepsy after prenatal exposure to PPIs (aHR 1.25, 95% CI 1.01–1.55), as well as early life exposure to antibiotics (aHR 1.11, 95% CI 1.02–1.21), PPIs (aHR 3.40, 95% CI 2.47–4.68) and H2RAs (aHR 1.65, 95% CI 1.03–2.64) (Table 2 ). Other risk factors for epilepsy after the age of two were the same as in the overall analysis above, except for being born prematurely (Figure 1 ).
When comparing mothers with and without epilepsy, PPIs (aHR 4.34, 95% CI 0.59–32.14 (maternal epilepsy) vs aHR 3.80, 95% CI 2.81–5.15 (no maternal epilepsy)) and antibiotics (aHR 1.13, 95% CI 0.64–1.97 (maternal epilepsy) vs aHR 1.12, 95% CI 1.03–1.22 (no maternal epilepsy)), exposure in early life was associated with an increased risk of epilepsy after the age of two (compared to unexposed children) for children whose mothers did not have epilepsy (Table 2 ). However, these results may be underpowered.
Cumulative incidence curves showed a slightly higher incidence for those exposed to antibiotics (Figure 2 d ) or H2RAs (Figure 2 f ) and a much higher incidence for PPIs (Figure 2 e ) compared to unexposed.
No interactions between early life antibiotics and early life PPIs (P = 0.76) were found with a likelihood ratio test.
Dose response analysis
No association was found with one or two prescriptions of antibiotics in early life and epilepsy. The highest association with an increased epilepsy risk was for those with three or more than three antibiotic prescriptions in early life (aHR 1.62, 95% CI 1.34–1.96 (three prescriptions) and aHR 1.43, 95% CI 1.11–1.84 (more than three prescriptions) (Figure 3 bottom left). Regarding early life exposure to PPIs, the risk of epilepsy increased with an increasing number of prescriptions, ranging from one prescription (aHR 2.49, 95% CI 1.98–3.12) to over three prescriptions (aHR 7.15, 95% CI 5.60–9.13) (Figure 3 , bottom right).
Antibiotics and epilepsy subgroups
Sulphonamides were significantly associated with an increased risk of epilepsy (J01E) (aHR 1.29, 95% CI 1.04–1.61) while other classes were not (Table 3 ).
As with prenatal exposure to antibiotics, the only significantly associated group of epilepsy diagnoses with increased risk was with “Other epilepsy” (G408 (Other epilepsy and recurrent seizures) and G409 (Epilepsy, unspecified) combined) (aHR 1.18, 95% CI 1.05–1.34) (Table 4 ).
Regarding individual ICD‐10 codes (not grouped as above), antibiotics exposure was associated with an increased risk of both G402 (Localization‐related (focal) (partial) symptomatic epilepsy and epileptic syndromes with complex partial seizures) (aHR 1.44, 95% CI 1.15–1.81) and G409 (Epilepsy, unspecified) (aHR 1.15, 95% CI 1.03–1.28) (Table 4 ).
DISCUSSION
In our nationwide Swedish cohort, we found that exposure to PPIs and antibiotics, both prenatally and in early life, particularly after multiple prescriptions, was associated with an increased risk of epilepsy. The increased risk remained when excluding mothers with an epilepsy diagnosis, thereby lowering the genetic risk. Furthermore, early life H2RA exposure was associated with an increased risk of epilepsy, but a much smaller effect compared to PPIs, which are prescribed for similar indications. The effect was largest in children exposed to PPIs before the age of 2 years.
Hazard ratios for PPIs were larger than for both antibiotics and H2RAs, which is an indirect support of a potential microbiome‐dependent mechanism, as PPIs are more disruptive to the microbiome than antibiotics and H2RAs [21, 36]. This difference between the effect of PPIs and H2RAs on the microbiome, and its association with the risk of epilepsy is very important, since they are used for similar indications, and H2RAs would therefore be a good substitute for PPIs for those that need gastric acid inhibitors.
Potential mechanisms behind this association may come down to neuroplasticity during the first few years of life. The maternal and infant microbiome has previously been associated with neurodevelopment [37, 38], so we hypothesize that if a non‐optimal microbiome is acquired at birth from the mother, or if an otherwise healthy microbiome is disrupted via drug use in early life, it may have long‐term consequences on neurodevelopment. This could be mediated through a suboptimal microbiome inducing an inflammation response, production of neurotransmitters by bacteria, or other signals that could influence the vagal nerve [37]. Animal studies have suggested a causal relationship between the gut microbiome and seizure susceptibility [3, 39]. Recent Mendelian randomization studies on genomic and clinical data indicate a causative role of the gut microbiome for epilepsy [40, 41, 42, 43]. Clinical studies have shown differences in the composition of the gut microbiome between patients with epilepsy and healthy controls [3, 44, 45, 46, 47, 48]. However, the cohorts were small and heterogeneous, including patients with diverse aetiologies and seizure types usually treated with one or more anti‐seizure medications. Thus, the results currently lack consensus, and further large cohort studies are needed.
We hypothesize that a dysbiosis may be contributing to the clinical picture for certain epilepsy diagnoses, although acknowledging the difficulties in stratifying subgroups of epilepsy in registries. Large clinical studies on the fecal microbiome in patients with a more certain stratification by epilepsy type are necessary to confirm our findings. Even though our results are only exploring potential underlying mechanisms and may have limited individual impact, they are necessary to understand the underlying epidemiological etiology to hopefully contribute to better prevention and early detection in the future.
Clinical and research implications
Risk increase was only seen for “Other beta‐lactam antibacterials”, Macrolides and “Other antibacterials” prenatally, and Sulfonamides for early life exposure. Macrolides, which showed the largest hazard ratio after prenatal exposure for epilepsy, have been shown to have a strong inhibitory effect on commensal gut bacteria [49], so the class of antibiotics might be considered when prescribing. In addition, confirmation of bacterial infection before treatment is desirable. For antibiotics exposure both prenatally and in early life, the dose response curve suggests that the risk is highest after repeated treatments. PPIs can often be replaced by other drugs or treatments, such as dietary interventions. For PPIs one dose was already significantly associated with an increased risk of epilepsy. This is in line with the knowledge that PPIs are more disruptive to the microbiome than antibiotics [36]. PPIs have been shown to decrease Faecalibacterium, which have anti‐inflammatory properties, and increase beta diversity in the gut [50]. For both PPIs and antibiotics an individualized approach would be most appropriate, where both short‐ and long‐term pros and cons are weighed for the treatments available.
Results regarding the trimester of exposure might allude to the mechanism of action. As the increased risk was during early pregnancy (and not in the third trimester), it could be related to the development of the central nervous system in the fetus. Mice studies have shown that the maternal microbiome and its metabolites can impact the expression of genes related to neural system development and function, especially in male fetuses [51]. Furthermore, the brain keeps developing and maturing drastically in early life, so disturbances may have long‐term effects [5]. The difference in results by trimester is probably not due to the different prevalence of antibiotic types, since no specific antibiotic class was increased in the third trimester.
Strengths and limitations
A major strength of this study is the size of this nation‐wide cohort and the prospectively collected data, limiting the risk of selection bias. Since antibiotics are only available by prescription in Sweden, the risk of misclassification of antibiotics exposure is low. However, PPIs are available over the counter, at a higher price, which causes some nondifferential misclassification, indicating that the true effect of PPIs may be larger than observed. Additionally, we can assume that individuals who use prescribed PPIs are more likely to have more severe symptoms and be long‐term users. Unfortunately, the registries do not contain information on in‐hospital drug use, which causes a risk of misclassification.
The main limitation of this study is the lack of information on the indication of drug use, causing potential confounding by indication, particularly for antibiotics, as several prenatal and early life infections (or misdiagnosed viral infections) [52, 53] may affect the brain and neurodevelopment. PPI use in children is usually off‐label for chronic cough or acid‐related problems in newborns, such as GERD, which can be difficult to diagnose, but often with little benefit and high risk of side effects [54, 55, 56, 57]. Additionally, the low number of individuals with PPIs and H2RAs prescribed could influence our results (broader confidence intervals), and our findings should be validated in other settings, in sufficiently large cohorts. A recent drug utilization study in Sweden shows that H2RAs are seldom prescribed in Sweden, while PPIs are increasingly used [58]. Furthermore, the validity of epilepsy subtype diagnosis may be questionable due to power. Thus, conclusions based on them should be made with caution. Additional covariates, such as information on breastfeeding, would also strengthen future studies on the topic.
CONCLUSION
We found that exposure to PPIs or antibiotics prenatally or in early life was associated with an increased risk of epilepsy. A single prescription of antibiotics during either exposure window was not associated with an increased risk, while an association was found in both windows with a single PPI prescription. H2RAs in early life were associated with an increased risk of epilepsy, but lower compared to PPIs, which are prescribed for similar indications.
Our findings need to be validated in other settings as prescription rates vary globally and infections remain life threatening in certain parts of the world. Furthermore, studies that include indication for drug use and over‐the‐counter use should be performed to assess the safety of these drugs in the pregnant population and children.
FUNDING
This work was supported by funding from the Swedish Research Council (2020–01058) and Open access fee is sponsored by Karolinska Institutet.
CONFLICT OF INTEREST
The authors declared no competing interests for this work.
AUTHOR CONTRIBUTIONS
U.G., R.W., A.G., S.P.N., and N.B. designed the research; U.G. performed the research; U.G., S.P.N., and N.B. analyzed the data; U.G., S.P.N., N.B., R.W., and A.G. wrote the manuscript.
ETHICS STATEMENT
This study was approved by the Swedish Research Council (ethical permit 2017/2423–31) and conducted as per the Declaration of Helsinki.
Supporting information
Data S1.
ACKNOWLEDGMENTS
We would like to thank the Swedish Research Council for funding this study.
DATA AVAILABILITY STATEMENT
Data is located on an access‐controlled server at Karolinska Institutet and is not openly available due to the sensitive personal nature of it. Code is available upon reasonable request to the corresponding author.
References
- 1. Guerrini, R. Epilepsy in children. Lancet 367, 499–524 (2006). [DOI] [PubMed] [Google Scholar]
- 2. Shorvon, S.D. The etiologic classification of epilepsy. Epilepsia 52, 1052–1057 (2011). [DOI] [PubMed] [Google Scholar]
- 3. Lum, G.R. , Olson, C.A. & Hsiao, E.Y. Emerging roles for the intestinal microbiome in epilepsy. Neurobiol. Dis. 135, 104576 (2020). [DOI] [PubMed] [Google Scholar]
- 4. Dahlin, M. & Prast‐Nielsen, S. The gut microbiome and epilepsy. EBioMedicine 44, 741–746 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Damiani, F. , Cornuti, S. & Tognini, P. The gut‐brain connection: exploring the influence of the gut microbiota on neuroplasticity and neurodevelopmental disorders. Neuropharmacology 231, 109491 (2023). [DOI] [PubMed] [Google Scholar]
- 6. Biagioli, V. et al. From birth to weaning: a window of opportunity for microbiota. Nutrients 16, 272 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Thriene, K. & Michels, K.B. Human gut microbiota plasticity throughout the life course. Int. J. Environ. Res. Public Health 20, 1463 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Imhann, F. et al. The influence of proton pump inhibitors and other commonly used medication on the gut microbiota. Gut Microbes 8, 351–358 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Weersma, R.K. , Zhernakova, A. & Fu, J. Interaction between drugs and the gut microbiome. Gut 69, 1510–1519 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Orwa, S.G. , Boven, A. , Pauwels, I. , Versporten, A. , Vlieghe, E. & Brusselaers, N. Global prevalence of antibiotic consumption during pregnancy: a systematic review and meta‐analysis. J. Infect. 89, 106189 (2024). [DOI] [PubMed] [Google Scholar]
- 11. Cannon, J.P. et al. The risk of seizures among the carbapenems: a meta‐analysis. J. Antimicrob. Chemother. 69, 2043–2055 (2014). [DOI] [PubMed] [Google Scholar]
- 12. Sutter, R. , Ruegg, S. & Tschudin‐Sutter, S. Seizures as adverse events of antibiotic drugs: a systematic review. Neurology 85, 1332–1341 (2015). [DOI] [PubMed] [Google Scholar]
- 13. Wanleenuwat, P. , Suntharampillai, N. & Iwanowski, P. Antibiotic‐induced epileptic seizures: mechanisms of action and clinical considerations. Seizure 81, 167–174 (2020). [DOI] [PubMed] [Google Scholar]
- 14. Tao, Q. et al. Prenatal exposure to antibiotics and risk of neurodevelopmental disorders in offspring: a systematic review and meta‐analysis. Front. Neurol. 13, 1045865 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Norgaard, M. et al. Maternal use of antibiotics, hospitalisation for infection during pregnancy, and risk of childhood epilepsy: a population‐based cohort study. PLoS One 7, e30850 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Sassonker‐Joseph, N. et al. Prenatal exposure to antibiotics and development of epilepsy in children. J. Clin. Pharmacol. 61, 18–24 (2021). [DOI] [PubMed] [Google Scholar]
- 17. Choi, A. et al. Association between exposure to antibiotics during pregnancy or early infancy and risk of autism spectrum disorder, intellectual disorder, language disorder, and epilepsy in children: population based cohort study. BMJ 385, e076885 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Attard, C.L. et al. The burden of illness of severe nausea and vomiting of pregnancy in the United States. Am. J. Obstet. Gynecol. 186(5 Suppl Understanding), S220–S227 (2002). [DOI] [PubMed] [Google Scholar]
- 19. Hastie, R. et al. Proton pump inhibitors and preeclampsia risk among 157 720 women. Hypertension 73, 1097–1103 (2019). [DOI] [PubMed] [Google Scholar]
- 20. Liang, C.S. et al. The risk of epilepsy after long‐term proton pump inhibitor therapy. Seizure 87, 88–93 (2021). [DOI] [PubMed] [Google Scholar]
- 21. Zhu, J. et al. Compared to histamine‐2 receptor antagonist, proton pump inhibitor induces stronger oral‐to‐gut microbial transmission and gut microbiome alterations: a randomised controlled trial. Gut 73, 1087‐1097 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Axelsson, O. The Swedish medical birth register. Acta Obstet. Gynecol. Scand. 82, 491–492 (2003). [DOI] [PubMed] [Google Scholar]
- 23. Socialstyrelsen . National Patient Register <https://www.socialstyrelsen.se/en/statistics‐and‐data/registers/national‐patient‐register/> (2022).
- 24. Brooke, H.L. et al. The Swedish cause of death register. Eur. J. Epidemiol. 32, 765–773 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Wettermark, B. et al. The new Swedish Prescribed Drug Register‐‐opportunities for pharmacoepidemiological research and experience from the first six months. Pharmacoepidemiol. Drug Saf. 16, 726–735 (2007). [DOI] [PubMed] [Google Scholar]
- 26. Methodology, W.C.C.f.D.S . ATC/DDD Index 2022 <https://www.whocc.no/atc_ddd_index/> (2021).
- 27. Socialstyrelsen . Klassifikation av sjukdomar och hälsoproblem 1997 (Socialstyrelsen, 1996). https://www.socialstyrelsen.se/globalassets/sharepoint‐dokument/artikelkatalog/klassifikationer‐och‐koder/1999‐4‐2_199942.pdf [Google Scholar]
- 28. RStudio Team . RStudio: Integrated Development for R (RStudio, PBC, Boston, MA, 2020). [Google Scholar]
- 29. Müller, R.S. DBI: R Database Interface (2024).
- 30. Hazan, G. et al. The impact of maternal hyperemesis gravidarum on early childhood respiratory morbidity. Pediatr. Pulmonol. 59, 707–714 (2024). [DOI] [PubMed] [Google Scholar]
- 31. Wickham, H. et al. Welcome to the {tidyverse}. J. Open Source Softw. 4, 1686 (2019). [Google Scholar]
- 32. Hadley Wickham, E.M. & Smith, D. Haven: Import and Export ‘SPSS’, ‘Stata’ and ‘SAS’ Files (2023).
- 33. Joe Conway, D.E. , Nishiyama, T. , Prayaga, S.K. & Tiffin, N. RPostgreSQL: R Interface to the ‘PostgreSQL’ Database System (2024).
- 34. Rich, B. table1: Tables of Descriptive Statistics in HTML (2023).
- 35. Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer‐Verlag, New York, NY, 2016). [Google Scholar]
- 36. Brusselaers, N. Prescribed drugs and the microbiome. Gastroenterol. Clin. North Am. 48, 331–342 (2019). [DOI] [PubMed] [Google Scholar]
- 37. Frerichs, N.M. , de Meij, T.G.J. & Niemarkt, H.J. Microbiome and its impact on fetal and neonatal brain development: current opinion in pediatrics. Curr. Opin. Clin. Nutr. Metab. Care 27, 297–303 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Sun, Z. et al. Revealing the importance of prenatal gut microbiome in offspring neurodevelopment in humans. EBioMedicine 90, 104491 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Darch, H. & McCafferty, C.P. Gut microbiome effects on neuronal excitability & activity: implications for epilepsy. Neurobiol. Dis. 165, 105629 (2022). [DOI] [PubMed] [Google Scholar]
- 40. Qiu, Y. et al. Causal links between gut microbiomes, cytokines and risk of different subtypes of epilepsy: a Mendelian randomization study. Front. Neurosci. 18, 1397430 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Ouyang, Y. et al. Genetically proxied gut microbiota, gut metabolites with risk of epilepsy and the subtypes: a bi‐directional Mendelian randomization study. Front Mol Neurosci 15, 994270 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Wang, X. et al. Anatomizing causal relationships between gut microbiota, plasma metabolites, and epilepsy: a Mendelian randomization study. Neurochem. Int. 183, 105924 (2024). [DOI] [PubMed] [Google Scholar]
- 43. Zeng, Y. , Cao, S. & Yang, H. Roles of gut microbiome in epilepsy risk: a Mendelian randomization study. Front. Microbiol. 14, 1115014 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Amlerova, J. et al. Evidences for a role of gut microbiota in pathogenesis and Management of Epilepsy. Int. J. Mol. Sci. 22, 5576 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Peng, A. et al. Altered composition of the gut microbiome in patients with drug‐resistant epilepsy. Epilepsy Res. 147, 102–107 (2018). [DOI] [PubMed] [Google Scholar]
- 46. Lindefeldt, M. et al. The ketogenic diet influences taxonomic and functional composition of the gut microbiota in children with severe epilepsy. NPJ Biofilms Microbiomes 5, 5 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Zhang, Y. et al. Altered gut microbiome composition in children with refractory epilepsy after ketogenic diet. Epilepsy Res. 145, 163–168 (2018). [DOI] [PubMed] [Google Scholar]
- 48. Xie, G. et al. Ketogenic diet poses a significant effect on imbalanced gut microbiota in infants with refractory epilepsy. World J. Gastroenterol. 23, 6164–6171 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Maier, L. et al. Unravelling the collateral damage of antibiotics on gut bacteria. Nature 599, 120–124 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Kiecka, A. & Szczepanik, M. Proton pump inhibitor‐induced gut dysbiosis and immunomodulation: current knowledge and potential restoration by probiotics. Pharmacol. Rep. 75, 791–804 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Husso, A. et al. Impacts of maternal microbiota and microbial metabolites on fetal intestine, brain, and placenta. BMC Biol. 21, 207 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Yates, E.F. & Mulkey, S.B. Viral infections in pregnancy and impact on offspring neurodevelopment: mechanisms and lessons learned. Pediatr. Res. 96, 64–72 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. San Martin‐Gonzalez, N. et al. Maternal respiratory viral infections during pregnancy and offspring's neurodevelopmental outcomes: a systematic review. Neurosci. Biobehav. Rev. 149, 105178 (2023). [DOI] [PubMed] [Google Scholar]
- 54. Ward, R.M. & Kearns, G.L. Proton pump inhibitors in pediatrics: mechanism of action, pharmacokinetics, pharmacogenetics, and pharmacodynamics. Paediatr. Drugs 15, 119–131 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Tafuri, G. et al. Off‐label use of medicines in children: can available evidence avoid useless paediatric trials? The case of proton pump inhibitors for the treatment of gastroesophageal reflux disease. Eur. J. Clin. Pharmacol. 65, 209–216 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Dipasquale, V. et al. A narrative review on efficacy and safety of proton pump inhibitors in children. Front. Pharmacol. 13, 839972 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Pasman, E.A. et al. Proton pump inhibitors in children: the good, the bad, and the ugly. Curr. Allergy Asthma Rep. 20, 39 (2020). [DOI] [PubMed] [Google Scholar]
- 58. Brusselaers, N. et al. Trends in proton pump inhibitor use in Sweden by sex and age: a drug utilisation study. Drug Saf. 48, 389–400 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data S1.
Data Availability Statement
Data is located on an access‐controlled server at Karolinska Institutet and is not openly available due to the sensitive personal nature of it. Code is available upon reasonable request to the corresponding author.


