Abstract
Chronic hepatitis B virus (HBV) infection poses significant public health challenges in Canada, particularly among newcomers from regions with high HBV prevalence. In alignment with the World Health Organization’s goal of HBV elimination by 2030, this 2025 guidelines update—developed jointly by the Canadian Association for the Study of the Liver (CASL) and the Association of Medical Microbiology and Infectious Disease (AMMI) Canada—presents recommendations for universal adult HBV screening, vaccination, laboratory assessment, and treatment. These guidelines emphasize patient-centred care, early diagnosis, and expanded antiviral treatment, including for individuals in the indeterminate or grey zone and special populations such as pregnant individuals, children, and those coinfected with HIV, hepatitis C, or hepatitis D. Notably, the guidelines recommend reflex HDV testing and routine use of quantitative HBsAg to support management decisions. These evidence-based recommendations are informed by expert consensus, recent literature, and international standards, with the aim of improving outcomes, reducing stigma, and informing future policy and research priorities.
Keywords: Association of Medical Microbiology and Infectious Disease (AMMI) Canada, Canadian Association for the Study of the Liver (CASL), update, guidelines, hepatitis B
Key Points
Universal screening
Universal vaccination (in those not immune)
Expanded treatment
Urgent need for investment in research on HBV in Canada
Patients at the centre of decision making for their own care, with values and preferences prioritized
Hepatitis B virus (HBV) infection continues to significantly impact the health of Canadians, particularly among newcomers to Canada from regions of increased HBV prevalence. Canada has endorsed the World Health Organization (WHO) Global Health Sector Strategy for HBV elimination by 2030 (1) and has also developed a 2024-2030 action plan (2) to address sexually transmitted and blood-borne infections (STBBI), including HBV. Although there are gaps in HBV surveillance in Canada, limiting a complete understanding of HBV prevalence and trends, the estimated number of people living with chronic HBV is 262,000 (3). According to the Canadian Notifiable Disease Surveillance System, the rate of HBV infection per 100,000 was 9.22 in 2021 (4), or 3,524 cases. This is likely an underestimate, due to gaps in reporting and a failure to diagnose all infected individuals.
Chronic HBV infection in Canada disproportionately affects non-Canadian born individuals (5,6). Approximately 23% of the Canadian population in 2021 was comprosed of immigrants and refugees to Canada (7), with most newcomers having a country of birth associated with an increased prevalence of HBV infection (8). Considering the important economic, social, and cultural contributions of immigrants and newcomers to Canada, it is important to maintain an adequate infrastructure to support immigration-based growth, including within our health care system (9). This requires considering the ways that the health needs of migrants differ from those of individuals born in Canada. Vaccination, early detection, and linkage to care can lead to better outcomes and avoid needless suffering for individuals, elevate the immigrant workforce, and ease the future strain on Canada's health care system by avoiding cases of liver disease and liver cancer.
The HBV guidelines committee have limited the scope of this update to include new recommendations for HBV screening and vaccination, hepatitis D virus (HDV) screening, laboratory assessment of HBV and HDV, treatment of HBV, including management of patients in the indeterminate phase or grey zone, and management of HBV in special populations. The recent WHO recommendations calling for a broader treat-all approach are discussed in the context of the Canadian health care system (10). Finally, the committee highlighted the importance of the patient voice and patient-partnered care (11).
Updated Guidelines Development
The panel responsible for the updated HBV guidelines were chosen in consultation with the Guidelines Committee of the Canadian Association for the Study of the Liver (CASL). Eight authors were chosen based on their expertise in HBV treatment, management of special populations, epidemiology, laboratory assessment, and patient-partnered care. Specific sections of the 2018 HBV Management Guidelines were selected for updating, and recommendations were made based on expert guidance, recent literature, and international guidelines. The recommendation decision process and grading were consistent with the Oxford 2011 Levels of Evidence (12,13) and included iterative discussion and consensus among panel members. These updated guidelines were reviewed, modified, and enhanced through the lens of patient-centred advocacy through the inclusion of the Executive Director for a national coalition of organizations responding to viral hepatitis (Action Hepatitis Canada). Inclusion of such authorities is essential, in that evidence-based recommendations and guidance are complemented by patient and public health considerations. The current document was also reviewed by CASL and Association of Medical Microbiology and Infectious Disease Canada (AMMI) members and revised following further discussion and recommendations.
The recommendations provided in these condensed guidelines serve as an update to relevant sections of the full HBV Management Guidelines (14) and are intended for physicians treating or managing individuals infected with HBV (hepatologists, infectious disease specialists, primary care providers, nurse practitioners), as well as public health practitioners and laboratory specialists. A table summarizing all recommendations developed for the updated guidelines are given in Table 1. The Supplementary Table summarizes recommendations for special populations that are overall unchanged from the 2018 HBV guidelines (14). The guidelines document and recommendations are a living document that will be updated periodically to reflect new research and evidence.
Table 1:
Summary of recommendations for the 2025 HBV guideline update
| Recommendations for HBV screening and testing |
|
| Recommendations for HDV screening and testing |
|
| Recommendations for HBV vaccination |
|
| Recommendations for HBV laboratory assessment |
|
| Recommendations for HBV treatment (Figure 1) |
|
| Recommendations for first-line HBV therapy |
|
| Recommendations for endpoints of HBV therapy |
|
| Recommendations for HCC surveillance |
|
| Recommendations for management of individuals of childbearing age and pregnancy |
|
| Recommendations for management of HBV and HDV coinfection |
|
| Recommendations for management of HBV and HCV coinfection |
|
| Recommendations for management of HBV and HIV co-infection |
|
| Recommendations for management of HBV in pediatric patients |
|
ALT ULN 25 U/L (famales) and 35 U/L (males)
ALT = Alanine transaminase; anti-HBs = Antibody to surface protein; anti-HBe = Antibody to HBeAg; cccDNA = Covalently closed circular DNA; ETV = Entecavir; HBcrAg = xxx; HBeAg = Hepatitis B e antigen; HBsAg = Hepatitis B surface antigen; HBV = Hepatitis B virus; HCC = Hepatocellular carcinoma; HDV = Hepatitis D virus; LHB = Large HBsAg; MHB = Middle HBsAg; NRAg = HBV nucleic acid-related antigen ; MTCT = Mother-to-childtransmission; PEG-IFN = Pegylatedinterferon-alpha; qHBsAg = xxx; TAF = Tenofovir alafenamide; TDF = Tenofovir disoproxil fumarate; ULN = upper limit of normal
Hepatitis B AND D Screening and HBV Vaccination (C Osiowy)
The previous Canadian guidelines (14) included HBV screening recommendations that were risk-based. Examples of factors increasing risk for HBV infection include birth or residency in an HBV-endemic (>2% prevalence) region, injection drug use, multiple sexual partners, incarceration, or close contact with a chronically infected person. Within Western countries having historically low HBV prevalence, such as Canada, immigrants often comprise the majority of the chronic HBV population (15). Recent evidence from the US Centers for Disease Control (16) has shown that risk-based screening alone is insufficient to detect and diagnose ≥90% of individuals affected by HBV, particularly those that are disproportionately affected, largely due to the stigma associated with HBV infection, including the fear of discrimination faced by newcomers and the reluctance by individuals to disclose certain risk factors. To address this barrier and work toward meeting the WHO 2030 HBV elimination goal for HBV diagnosis, the US CDC has recommended universal adult screening (16), such that all individuals over the age of 18 should be offered screening at least once in their lifetime by a triple panel serological screen (hepatitis B surface antigen [HBsAg], antibody to the surface protein [anti-HBs], and total antibody to the core protein [anti-HBc]; see Table 4 of the 2018 guidelines (14), for an explanation of HBV serological markers). We acknowledge that in a low-prevalence population, screening with a triple panel could potentially increase findings of isolated anti-HBc positivity. In addition to a false positive result, this finding is also associated with occult HBV, a state in which replicable virus is present in the liver in HBsAg-negative individuals having detectable or undetectable viral DNA in the serum (17). Isolated anti-HBc positivity may arise due to waning anti-HBs levels following a past infection, passive transfer of anti-HBc to an infant born to an HBsAg-positive gestational parent, passive transfer of anti-HBc after intravenous immunoglobulin administration, acute HBV during the window period when neither HBsAg nor anti-HBs is detectable, or infection with a strain of virus having mutations in the HBsAg protein, resulting in loss of detection by laboratory assays (16,18). Simplifying screening and diagnostic guidelines will be a major step in removing the barriers that prevent HBV infected individuals from receiving a diagnosis. Universal screening is essential to identify asymptomatic individuals before the manifestation of liver-related complications, such as cirrhosis and hepatocellular carcinoma (HCC), resulting in advanced liver disease. An Ontario study showed that 45% of individuals with laboratory-confirmed chronic HBV were diagnosed within 6 months of such complications (19), highlighting the need for earlier diagnosis. Most importantly, universal screening removes the burden of awareness of specific risk criteria and the time needed to assess each patient's individual risk from the provider. In this way, screening and diagnosis of HBV infection becomes truly universal in that all persons are considered equally at risk.
Universal adult screening was determined to be cost effective for the United States, considering their current epidemiology and nucleos(t)ide analogue (NA) treatment costs. Indeed, universal adult screening, in combination with universal adult (aged 19–59 years) vaccination, which was recommended in 2022 (20), was found to be more cost effective than universal vaccination alone. The cost-effectiveness of national universal screening of all Canadian adults is not known; however, there is evidence showing cost-effectiveness of universal screening of immigrants and refugees to Canada (21,22). Cost-effectiveness studies of universal screening of all adults at the provincial and national level are needed to support and encourage screening uptake. Provincial and local laboratory capacity and efficiency, to remove repeat and unnecessary testing during screening, are important considerations for universal adult screening to proceed in Canada.
HDV infection of HBsAg-positive persons is associated with a rapidly progressing viral hepatitis, leading to cirrhosis and liver-related mortality in a higher number of patients compared to chronic HBV alone (13). Current screening recommendations for HDV infection are also based on risk; primarily for HBsAg-positive persons born in regions of high HDV endemicity and those having risk factors similar to HBV infection. Data from several European (23, 24, 25), USA (26) and Asian (27) treatment centres, as well as modelling (28) have shown that risk-based screening leads to very low rates of HDV testing and thus is inadequate for capturing the majority of HDV cases. This evidence and the new availability and promise of HDV treatment options advocates for universal screening for HDV through automatic reflex testing of all HBsAg-positive persons. Modelling and real-world experience, as cited above, have shown that universal reflex testing leads to a higher and more accurate diagnostic yield of HDV cases (29). Universal testing also has the benefit of removing the onus on the health care provider for risk factor awareness and management and reducing feelings of discomfort and stigma that a patient may experience when disclosing risk factors (10). The cost-effectiveness of universal screening for HDV in HBsAg-positive persons in Canada is also not known, and thus cost-effectiveness modelling studies are encouraged to support and confirm the recommendations made. Nevertheless, the overall modest number of additional tests that would be required, together with practices in similar countries and the advantages discussed above, support our recommendation (13,30).
The majority of HDV antibody tests are manual enzyme immunoassays manufactured in Europe, Asia or the US (31), although none are licenced or approved by Health Canada. There is limited diagnostic performance data for these assays, though a systematic review described a pooled sensitivity and specificity of 0.99 (95% CI 0.96 to 1.00) and 0.90 (95% CI 0.79 to 0.96), respectively, for HDV serological assays, including anti-IgM, anti-IgG and total antibody (32). For example, the Wantai HDV IgG test (Wantai BioPharm, Beijing, China) that is used in several Canadian laboratories, was reported to have excellent specificity (99.73%) for HDV anti-IgG among HBsAg-negative and -positive blood donors in China (33). The test was also used to identify the HDV seroprevalence among HBsAg-positive specialist clinic patients in Canada over an 8-year period, resulting in 4.5% (95% CI 4.3 to 5.3) HDV antibody positivity (5). Recently, an automated indirect chemiluminescent immunoassay (CLIA) for HDV total antibody has become available (LIAISON XL Murex anti-HDV assay, DiaSorin Canada Inc., Mississauga, ON). The assay was shown to demonstrate 100% sensitivity and specificity in several studies (34,35). However, the Murex assay demonstrated reduced specificity (98.5%) and sensitivity (90.2%) in comparison to a recently developed prototype assay from Abbott Laboratories (Mississauga, ON) for the ARCHITECT platform (36). The prototype assay, an automated direct CLIA for total HDV antibody, had a specificity of 99.5% among 200 blood donor specimens and a sensitivity of 95.2% with a panel of 62 HDV RNA-positive specimens.
Recommendations for HBV screening and testing
Universal one-time screening of all adults (>18 years) in Canada should be implemented. Screening should be conducted using a triple panel including testing for HBsAg, anti-HBs, and anti-HBc. In consultation with the patient, evidence of a new activity or persistent risk for HBV infection should prompt periodic re-testing if the patient is not immune (i.e., anti-HBs negative). Cost-effectiveness modelling is encouraged to support recommendations at provincial and national levels, and coordinated electronic records are recommended to enhance laboratory capacity and avoid repeat testing. (Strong recommendation; Level 5)
Recommendations for HDV screening and testing
Universal reflex testing of HBsAg-positive individuals for antibody to HDV should be performed for patient samples with no history in the clinical laboratory system. For individuals having a negative result, follow-up testing for antibody to HDV is warranted for evidence of new activity or persistent risk for HDV or a new clinical profile including HBV DNA <2,000 IU/mL and alanine transaminase (ALT) above the ULN. (Strong recommendation; Level 3)
Universal testing of antibody to HDV should be performed using an assay having diagnostic accuracy and performance characteristics of >90% sensitivity and >99% specificity. (Strong recommendation; Level 5)
Universal reflex testing of HDV RNA in newly identified HDV seropositive samples should be performed by specialized or reference laboratories (i.e., the National Microbiology Laboratory). (Strong recommendation; Level 5)
Recommendations for HBV vaccination
Universal infant (ideally at-birth) vaccination is recommended across all Canadian provinces and territories. (Strong recommendation; Level 1)
Universal catch-up (adult) vaccination is recommended for all individuals who have not received a full series of HBV vaccine doses or unsure of their vaccination history. (Strong recommendation; Level 3)
Laboratory Assessment of HBV (C Osiowy)
Previous guidelines described in detail the laboratory assessment of HBV infection. Quantification of HBV DNA continues to be an essential test for management of patient treatment, as the current NA therapies target the viral polymerase, inhibiting HBV replication and reducing HBV DNA as a detectable seromarker. The preferred first-line NA therapies tenofovir disoproxil fumarate (TDF), tenofovir alafenamide (TAF), and entecavir (ETV) display a high resistance barrier in treatment naïve patients, suggesting that sequence analysis for NA resistance may not be necessary; however, HBV DNA genotyping and mutation analysis (Precore/basal core promoter mutations, HBsAg mutations associated with diagnostic or immune escape, and NA resistance in treatment experienced patients) continue to be used selectively by specialists for helping direct treatment and management decisions and interpreting the virological biomarker profile.
Current HBV antiviral treatment is largely limited to NA, which does not inhibit covalently closed circular DNA (cccDNA) transcriptional activity (37). Sterilizing cure of HBV requires the complete elimination of cccDNA, integrated HBV DNA and all HBV viral particles (38). Functional cure, defined as sustained loss of HBsAg expression and undetectable HBV DNA, both based on the sensitivity of current clinical assays, is the interim goal for chronic HBV patients (37). Quantification of cccDNA requires invasive procedures to obtain liver tissue and specialized laboratories to perform analysis, which is known to be challenging and without standardization at present (39). Therefore, other virological or host immune markers have been sought to measure viral transcriptional activity and host immune response to predict the phase of infection and treatment outcome, particularly to estimate the likelihood of stable suppression of viral replication following NA treatment withdrawal. These biomarkers are largely experimental, and the previous guidelines described some of the novel HBV biomarkers being evaluated. The updated guidelines provide recommendations based on more recent data and analysis.
qHBsAg
Functional cure of HBV infection is the goal of therapeutic intervention, as complete cure (i.e., eradication of liver cccDNA) or sterilizing cure (i.e., eradication of integrated viral DNA and all viral particles) are not possible with current NA therapy (40). Partial cure is considered a transitional stage on the way to functional cure and is defined as HBsAg levels <100 IU/mL and undetectable HBV DNA (38), thus quantitative testing of HBsAg is essential for patient management. Ensuring equal access across Canada and laboratory testing capacity for qHBsAg is an issue that must be resolved to meet this goal. Qualitative and quantitative HBsAg (qHBsAg) assays having 10- to 100-fold greater sensitivity (ultrasensitive) than conventional assays, with limits of detection of 50 mIU/mL, have been developed (41, 42, 43), although several are not available for use in Canada. The significance of detecting minute quantities of HBsAg below 50 mIU/mL in determining true HBsAg loss is still to be resolved (44); although further research should elucidate the role of very low HBsAg levels in disease progression and HCC risk (45). HBsAg quantitative cutoff values have been identified to predict clinical relapse, such that a qHBsAg cutoff of <100 IU/mL at end of therapy (EOT) distinguished a lower risk (38% versus 67%, p < 0.01) for hepatitis B e antigen (HBeAg) negative patients to experience off-therapy clinical relapse following long-term control of HBV replication by NA treatment (46). Regarding NA withdrawal strategy, as shown in a large, multicentre cohort, the best candidates for stopping therapy are patients that are virally suppressed, HBeAg-negative, and noncirrhotic with low HBsAg levels, particularly White patients with levels <1,000 IU/mL and Asian patients with levels <100 IU/mL (47). For monitoring the probability of HBsAg loss on therapy, the long-term qHBsAg trajectory was shown to be useful in HBV patients living with HIV co-infection and was also promising in HBV monoinfection (48,49).
The HBsAg detected and measured in serum is comprosed of the three component surface proteins, large (LHB), middle (MHB), and small HBsAg, each of which are present in defined amounts during infection in association with viral or secreted subviral particles (50). An experimental double-antibody sandwich ELISA-specific to LHB and MHB has been developed to quantify levels of both proteins to establish a ratio of each to total HBsAg to more accurately distinguish HBV infection phases (51). The LHB/total HBsAg ratio was thought to reflect the actual state of virus replication compared to HBsAg alone, as LHB and MHB are more abundantly present within HBV Dane particles than subviral HBsAg particles (50) and so LHB levels are relatively associated with HBV DNA levels. LHB is primarily transcribed from cccDNA, not integrated DNA (50), further supporting LHB as a diagnostic biomarker for HBV transcriptional activity. Thus, the quantification of HBsAg isoforms has the potential to direct guidance for optimal treatment initiation and an understanding of disease progression in chronic HBV patients.
Directly associating HBsAg and cccDNA transcriptional activity is challenging due to the additional expression of HBsAg by integrated HBV DNA. This is particularly relevant during the HBeAg-negative phase of infection (52). An experimental multiplex droplet digital PCR assay has been developed to differentiate HBsAg expressed from integrated HBV DNA or from cccDNA, based on the absolute quantity ratio of the two (53). This analysis and others (54) confirm that ongoing transcription and expression of HBsAg from integrated DNA will continue to confound attainment of HBV functional cure.
HBV RNA
Recent reviews analyzing the effectiveness of HBV biomarkers to predict treatment endpoints established the utility of serum HBV RNA quantification to potentially predict virological and clinical relapse based on EOT levels (55,56). Both HBeAg-positive and -negative patients were shown to have a reduced risk of clinical relapse following treatment cessation with low (<2–3 log U/mL) or undetectable HBV RNA at EOT (57, 58, 59, 60). Serum HBV RNA levels also have good prognostic power to predict HBeAg seroconversion and functional cure in in HBeAg-positive and HBeAg-negative chronic HBV patients, respectively (61).
Recent studies have provided a more complete understanding of the composition of serum HBV RNA to support the most accurate means to detect and quantify this biomarker (56,62,63). Most studies agree that HBV serum RNA is comprised primarily of pgRNA, the viral replicative intermediate transcript; however, depending on the method used to quantify HBV RNA, other circulating HBV RNA species, such as mRNA transcripts and spliced or truncated pgRNA, may be detected alongside full-length pgRNA (64,65). Unfortunately, there is no universal standard for pgRNA or total HBV serum RNA and no standardized, commercial assay is available for routine laboratory analysis, making comparisons among studies and laboratories very difficult. At this time, only laboratory-developed assays to quantify serum RNA have been described in Canada (65), although North American commercial molecular assay manufacturers are developing (66) or have investigational (67) quantitative assays.
HBcrAg and NRAg
The only assay offering measurement of HBcrAg, the Lumipulse G HBcrAg assay (Fujirebio Diagnostics, Inc., Malvern, PA, USA), remains unavailable in Canada. The assay detects a group of related HBV proteins all sharing a common 149 amino acids within the core-coding region. As HBeAg is a primary component of HBcrAg, it has been shown that different EOT cutoffs are required for HBeAg-positive or -negative patients to reliably predict virological and clinical relapse (55). Recent studies have shown the utility of HBcrAg measurements in combination with quantitative HBsAg levels to predict HBsAg seroclearance. In a large retrospective analysis of treatment-naïve patients within the ERADICATE-B study, those having high (>1,000 IU/mL) levels of HBsAg had a higher likelihood of reaching HBsAg seroclearance within a mean of 12.8 years, with an adjusted hazard ratio of 1.95, if baseline HBcrAg levels were <10,000 U/mL (68). Undetectable HBcrAg (i.e., <2 log10 U/mL) at NA EOT increased the likelihood of HBsAg loss in patients have HBsAg >10 IU/mL within a large multicentre study, although HBcrAg levels appeared to be dependent on the HBV genotype (69). Low HBcrAg levels (<4 log10 U/mL) at EOT with pegylated interferon (PEG-IFN) in NA-suppressed patients were also shown to be predictive of sustained functional cure (24-week post EOT follow-up), particularly when in combination with high levels of anti-HBs (>2 log10 IU/L), with an AUROC of 0.822 (70). Thus, a combination of biomarker cutoff values, including low levels of HBcrAg, may be used to predict durable HBsAg loss following treatment.
Qualitative detection of HBV nucleic acid-related antigen (NRAg) is also possible using a manual ELISA-based assay (HBV NRAg, Beijing Wantai Biological). The assay detects PreS1 and core antigens, with the biomarkers shown to correlate with HBV DNA levels, regardless of HBeAg status (71). There is a paucity of literature describing the utility of this assay (72); however, the detection of PreS1 protein, or LHB, which shows promise as a predictive biomarker as described earlier, together with core protein detection, may provide a unique measure of transcriptional activity in NA-suppressed patients. Further research is required to fully understand the performance characteristics of the assay and its predictive functions. Assay utility would also be enhanced by quantification of both proteins.
qAHBc
Quantification of antibody specific to the HBV core protein (qAHBc) continues to show promise as a predictor of treatment-induced virological response and HBsAg clearance, as well as spontaneous HBsAg loss. Recently, it was shown that low (<0.1 IU/mL) baseline qAHBc was an independent predictor for HBsAg loss after 48 weeks treatment in NA-suppressed patients having add-on PEG IFN-alpha therapy (73). An investigation of individuals having spontaneously cleared HBsAg demonstrated that qAHBc levels may be an early predictor of eventual HBsAg clearance, as early as 10 years prior to the event (74). As with other predictive biomarkers, a combination, including low qAHBc levels with qHBsAg <100 IU/mL, was superior in identifying >88% of patients who achieved functional cure within 1 year (74). There remain limited options available in Canada for qAHBc testing, with one manual ELISA (Beijing Wantai Biological Pharmacy, Beijing, China) available, having an extremely narrow dynamic range from 0.1 to 2.0 IU/mL, thus requiring extensive pre-dilution to test most samples. A commercial automated assay (Lumipulse G HBcAb-N, Fujirebio, Tokyo, Japan) reporting quantified levels as a cut-off index value is available outside of North America. The lack of standardization in units between both commercial assays further prevents comparisons among laboratories and studies. Further research is needed to translate the ability of qAHBc to reflect the host immune response and predict hepatic inflammation into practical treatment endpoints (75).
Point-of-care testing
At present, there are no Health Canada-approved point-of-care tests available for HBV serological or molecular testing. There are a variety of rapid diagnostic assays for HBsAg that have been pre-qualified by the WHO (76), one of which was successfully used with high uptake and acceptance in an outreach campaign in Toronto, Ontario (77). Hepatitis C (HCV) micro-elimination approaches, such as the use of dried blood spot testing and point-of-care nucleic acid testing (78), serve as an example by which Canada can increase diagnosis of HBV infection in order to meet the 2030 WHO goals for HBV elimination. Such approaches also align with the call by the WHO for broad adoption of simplified testing technologies to improve time to diagnosis and linkage to treatment (79). Although less research has been undertaken for HBV point-of-care testing in Canada, we anticipate a similar utility for HBV point of care testing as demonstrated for HCV.
Recommendations for HBV laboratory assessment
Quantitative HBV DNA testing is recommended to monitor treatment response. For NA-treated patients, the recommendation is to test every 6 months, in order to confirm ongoing adherence. (Strong recommendation; Level 1)
Quantitative measurement of HBsAg is recommended every one to two years to monitor treatment response and in determining functional cure. (Strong recommendation; Level 2)
New biomarkers that are associated with cccDNA transcriptional activity (LHB and MHB expression, serum HBV RNA, HBcrAg, NRAg) or host immune activity (qAHBc) require standardized testing platforms (including analyte standards) or have not been fully studied for their utility in clinical management. Further research on the clinical utility and cost-effectiveness of new HBV biomarkers is required. Future testing algorithms will likely involve a combination of standard and new biomarker assays with results that may provide a scoring system. (Weak recommendation; Level 4)
Point-of-care tests for HBV screening are recommended for community-based testing, and resource-limited or remote regions to remove barriers to diagnosis and treatment uptake. (Strong recommendation; Level 4)
Treatment of HBV Infection (SK Fung)
The guidelines committee continues to recommend TDF and entecavir (ETV), as well as PEG-IFN in selected populations as first-line therapy for chronic HBV. Since publication of the 2018 guidelines, new evidence published include benefits of TAF, lower HCC risk with tenofovir based therapies, the role of quantitative HBsAg, and evidence for expanding treatment in grey zone patients. The guidelines committee calls attention to the necessity for harmonization and availability of these treatments across Canadian publicly funded drug plans among community, hospital, long-term care, and correctional formularies to ensure equitable and uninterrupted coverage for recommended HBV treatment. The NA requires dosage adjustment in individuals with chronic kidney disease and long-term TDF is associated with renal and bone side effects in some patients. Therefore, monitoring of renal function (i.e., estimated glomerular filtration rate [eGFR] in all treated individuals, as well as periodic assessment of bone mineral density (BMD) in TDF treated patients, continue to be recommended, as per 2018 CASL/AMMI HBV management guidelines (14) and Canadian guidelines for osteoporosis management (80).
Tenofovir alafenamide
Long-term follow up from phase 3 registrational trials have demonstrated superior safety of TAF compared to TDF. After 5 years of treatment, patients who received TAF were found to have only minor reductions (<2.5 mL/min) in eGFR and decline in hip or spine BMD was <1% (81). Among those who switched from TDF to TAF, improvement in both bone and renal parameters were also reported at year 5. There was also a modest increase in body weight and LDL-cholesterol in patients who received TAF. However, no increase in cardiovascular events was seen during long-term follow-up in those who received TAF (82). On the other hand, among HBV monoinfected patients, rates of HBV DNA suppression, HBeAg seroconversion, and HBsAg loss were similar between the two groups. In addition, the observed rate of HCC was lower in patients who received tenofovir-based antiviral therapy compared to predicted rates of HCC over the same follow-up period in a group of patients with similar baseline characteristics (83).
Expansion of HBV treatment criteria
There is an increasing awareness of HBV patients in the grey zone. Several studies have reported that 30%–40% of chronic HBV patients cannot be accurately classified into any one phase of chronic infection (84,85). These patients have been termed grey zone patients and their natural history is not entirely benign (86,87). The most common group of grey zone patients include HBeAg-negative patients with normal ALT, mild or no significant hepatic fibrosis and HBV DNA >2,000 IU/mL. Retrospective studies from Taiwan and other countries have demonstrated lower rates of progression to cirrhosis and development of HCC in this group of patients who received long-term antiviral treatment (88,89). Therefore, antiviral treatment should be considered in this group of patients, particularly those who are at high risk for developing cirrhosis and HCC.
Another grey zone group includes older immune-tolerant (i.e., high replicative, non-inflammatory phase) patients. Previous studies of 4-year finite duration antiviral treatment in exclusively immune-tolerant patients suggested no benefit of long-term antiviral treatment with TDF (90,91). More recently, studies from Korea have documented higher than anticipated rates of HCC in this group of patients who are HBeAg-positive, have normal ALT and high-level HBV DNA >6 log IU/mL (92,93). Although treatment cannot be routinely recommended for all HBeAg-positive patients time, the subset who is older (i.e., age >40 years) and those at high risk for complications should be offered antiviral treatment. Although the duration of treatment is likely to be long term, newer agents, in combination with NA therapy in the future, such as antisense oligonucleotides or small interfering RNA may shorten and allow for finite duration antiviral treatment (94). A dynamic summary of new therapies in the HBV pipeline at various stages of clinical development is available from the Hepatitis B Foundation (www.hepb.org/drugwatch).
Recommendations for HBV treatment (Figure 1)
Figure 1. Summary of Recommendations for Hepatitis B Treatment Initiation.
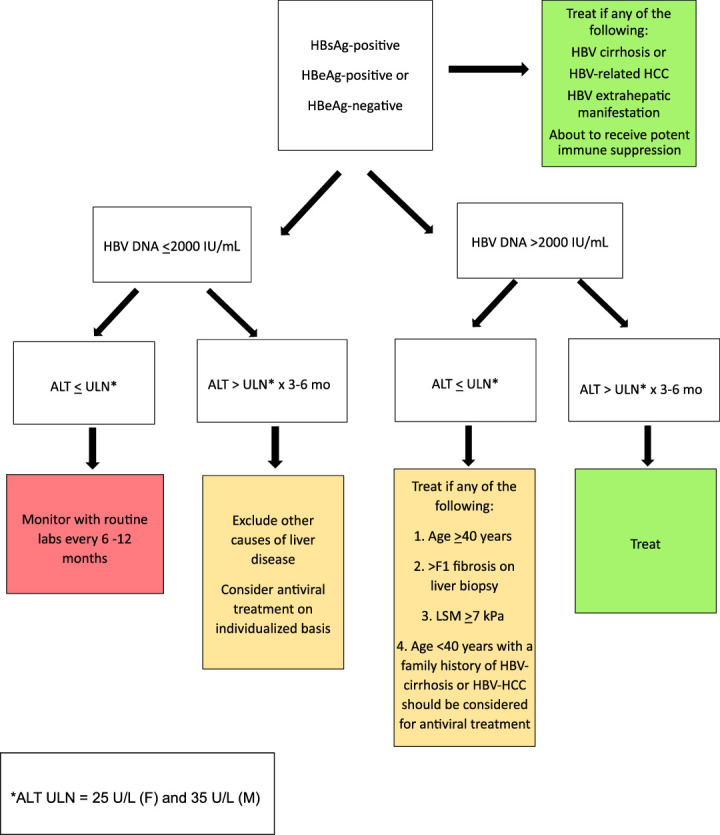
ALT = Alanine transaminase; F = Female; HBeAg = Hepatitis B e antigen; HBsAg = Hepatitis B surface antigen; HBV = Hepatitis B virus; HCC = Hepatocellular carcinoma; M = Male
HBsAg-positive patients with cirrhosis, those with a personal history of HCC, those about to receive highly potent immunosuppressive or immunomodulatory therapies (95), or those with extrahepatic HBV manifestations should receive oral antiviral treatment. (Strong recommendation; Level 1)
HBeAg-positive and HBeAg-negative patients with ALT above the upper limit of normal (ULN)* for 3–6 months or more and HBV DNA >2,000 IU/mL (HBeAg-positive chronic hepatitis and HBeAg-negative chronic hepatitis) should receive antiviral treatment. (Strong recommendation; Level 1)
HBeAg-positive patients over the age of 40 years with normal ALT and HBV DNA >2,000 IU/mL (HBeAg-positive chronic infection, formerly referred to as Immune Tolerant HBV) should receive antiviral treatment. (Strong recommendation; Level 2)
HBeAg-positive patients under the age of 40 years with normal ALT and HBV DNA >2,000 IU/mL (HBeAg-positive chronic infection, formerly referred to as immune tolerant HBV) who have a first-degree relative with a history of HCC or cirrhosis should be considered for antiviral treatment. (Strong recommendation; Level 2)
HBeAg-negative patients over age 40 years with normal ALT and HBV DNA >2,000 IU/mL (HBeAg-negative chronic infection) should receive antiviral treatment to reduce the risk of HCC. (Strong recommendation; Level 2)
HBeAg-negative patients under the age of 40 years with normal ALT and HBV DNA >2,000 IU/mL (HBeAg-negative chronic infection) who have a first-degree relative with a history of HCC or cirrhosis should be considered for antiviral treatment. (Strong recommendation; Level 2)
HBeAg-negative patients with normal ALT and HBV DNA <2,000 IU/mL (formerly referred to as inactive HBV carriers) should be monitored on a regular basis but may be considered for antiviral treatment on an individualized basis. (Strong recommendation; Level 3)
HBeAg-negative patients with ALT above the ULN* and HBV DNA <2,000 IU/mL should be investigated for causes of elevated ALT other than HBV infection but may also be considered for antiviral treatment on an individualized basis. (Strong recommendation; Level 3)
The importance of treatment adherence should be reinforced. If patients experience treatment interruptions (due to loss of medication coverage, etc.) but have not achieved the usual treatment endpoints (i.e., HBeAg seroconversion, low level qHBsAg), they should be counselled about the risk and the importance of regular monitoring for severe rebound flares and relapse of HBV (see Endpoints section).
Recommendations for first-line HBV therapy
TAF, TDF, or ETV are recommended as first-line monotherapy in HBeAg-positive chronic hepatitis and HBeAg-negative chronic HBV patients. (Strong recommendation; Level 1)
TAF or TDF with or without PEG-INF may be used in both HBeAg-positive and -negative chronic hepatitis patients. (Strong recommendation; Level 1)
Recommendations for endpoints of HBV therapy
For patients with HBeAg-positive chronic hepatitis who are noncirrhotic, treatment should continue until confirmed HBeAg loss or seroconversion followed by an additional 12–18 months of consolidation therapy. (Strong recommendation; Level 1)
For patients with HBeAg-negative chronic hepatitis who are noncirrhotic, treatment should continue until confirmed HBsAg loss or quantitative HBsAg <100 IU/mL for Asian patients or quantitative HBsAg <1,000 IU/mL for White patients followed by an additional 12 months of consolidation therapy (47). (Strong recommendation; Level 2)
All HBsAg-positive patients should be monitored closely after stopping NA (i.e., ALT, HBV serology and HBV DNA monthly for 3 months, then once every 3 months for 1 year, then once every 6 months, as per usual standard of care) to exclude severe HBV relapse. (Strong recommendation; Level 2)
For patients with HBV cirrhosis, treatment is of indefinite duration or until HBsAg loss followed by regular monitoring. (Strong recommendation; Level 2)
HBV-Related HCC (CS Coffin)
Chronic HBV is a major risk factor for the development of primary liver cancer (HCC). Current guidelines recommend age, sex and ethnicity specific surveillance for HCC. Various risk calculators have been published to improve risk stratification (96). As defined by these models, a subset of patients without cirrhosis and on long-term antiviral therapy are probably low risk, and potentially may be excluded from HCC surveillance (97). Cirrhosis is the single most important risk factor for HCC. However, the HBV integrates into the host chromosomes potentially within sites impacting tumour suppressor or oncogenes and can lead to pre-cancerous cells and malignant transformation even in young individuals without cirrhosis (98). The seminal Risk Evaluation of Viral Load Elevation and Associated Liver Disease/Cancer-Hepatitis B Virus (REVEAL-HBV) study was based on a community prospective cohort study in Taiwan, and established viral load (i.e., >2,000 IU/mL or 10,000 copies/mL) as independently associated with cirrhosis and HCC (99). The PAGE-B (Platelet Age GEnder–HBVScore) (100) was initially validated in European patients with chronic HBV, and modified PAGE (mPAGE) incorporating albumin was also predictive in Asian patients on antiviral therapy (101). Quantitative hepatitis B surface antigen is a novel biomarker used in monitoring HBV activity (see Laboratory assessment of HBV). Analysis of qHBsAg levels in the REVEAL HBV study cohort showed that in HBeAg-negative individuals, with low viral loads, qHBsAg >1,000 IU/mL was associated with HCC (102). Additional risk factors include viral genotype (A1, C, F), environmental exposures (i.e., aflatoxins derived from fungal contaminants in some food sources in African regions), host genetic factors and viral coinfections (103,104). Individuals of Black ancestry were historically considered to be of higher risk, warranting younger age surveillance. However, in a recent multicentre study of sub-Saharan African and Afro-Surinamese individuals living in Europe, the observed risk was negligible in individuals without advanced fibrosis (105). Obesity and metabolic syndrome affect many patients with HBV leading to metabolic dysfunction-associated steatotic liver disease (MASLD, formerly called non-alcoholic fatty liver disease). MASLD is now the leading cause of end-stage liver disease and HCC in many Western countries. Disease specific HCC risk calculators have been developed (i.e., the Toronto HCC risk index (106)). However, data is lacking to risk-stratify individuals with both MASLD and markers of active HBV infection and/or prior HBV infection (i.e., HBsAg-negative, HBV core antibody positive). The CASL guidelines committee has sponsored updated guidelines for MASLD and HCC with expected timelines for publication in 2026 which will further address HCC surveillance and management.
Recommendations for HCC surveillance
-
HCC surveillance is recommended even in patients who are on antiviral therapy due to residual risk. Abdominal ultrasound and AFP screening every 6 months is recommended in the following patients with chronic infection. (Strong recommendation; Level 3).
All patients with cirrhosis, irrespective of age (even those who clear HBsAg).
Men aged 40 years or older.
Women aged 50 years or older.
Persons of African (Black) ancestry origin aged 30 years or older (due to risk associated with certain African HBV genotypes and environmental factors including aflatoxin exposure).
First-degree family history of HCC (starting at age 40 or 10 years before affected 1st degree family member, whichever is sooner).
All HIV-co-infected patients (starting at age 40).
All HBV/HDV coinfected individuals (starting at age 40, or earlier if advanced >F3 [stage 3] fibrosis).
Risk calculators (i.e., REACH-B, PAGE-B, mPAGE-B) may be useful to support clinical practice and identification of patients who may benefit from HCC screening.
HBV Management in Special Populations (CS Coffin)
The guidelines committee proposed updates for 3 special populations from the 2018 CASL guidelines (i.e., pregnancy, coinfection, and pediatric). The committee did not recommend extensive updates to other special populations as there is insufficient new data published to warrant change in current clinical practice. One committee member (CSC) provided additional support (See Supplementary Table) for prior recommendations and suggested minor re-phrasing and change to timelines in laboratory monitoring for some special populations with additional supporting references (186–207).
Management of HBV in women of child-bearing age and pregnancy and prevention of mother-to-child transmission (CS Coffin)
The vast majority of individuals living with chronic HBV infection globally acquired infection by mother-to-child, or vertical, transmission (107). Infants are at greatest risk of developing chronic HBV infection. All infants, regardless of maternal HBsAg status, should receive the HBV vaccine as soon as possible after birth (within 24 hours) followed by two or three vaccine doses at least four weeks apart. Only a universal strategy will mitigate potential gaps in the care cascade, (i.e., late maternal presentation for prenatal care, system errors in testing and referral) as well as reduce unnecessary stress and anxiety for some families who may have a close household contact living with HBV. Most jurisdictions in Canada only offer birth dose vaccine if the mother is HBsAg-positive. Although the risk of horizontal transmission is much lower, this policy is not patient-centric, potentially impacting infant bonding with other family members who are HBsAg-positive (grandparents, fathers). In addition to the first dose of the HBV vaccine, all infants born to HBsAg-positive mothers should receive hepatitis B immune globulin (HBIG) as soon as possible after birth and testing to confirm they are not infected and HBV immune before 12 months of age.
As in previous guidelines, the committee reiterated the importance of systematic strategies and tracking in all Canadian jurisdictions of pregnant women with chronic HBV, including prenatal and peri-partum HBV management. Although most jurisdictions perform prenatal HBV screening, gaps in testing, follow-up, and linkage to care lead to missed opportunities for timely intervention with antiviral therapy if needed (108,109). Compared to previous guidelines, the updated recommendations promote consideration of: (i) expanding treatment eligibility, (ii) earlier treatment initiation in pregnancy and continued therapy long-term if planning future pregnancies, and (iii) new evidence on the role of TAF in pregnancy.
Screening and assessment
All pregnant women should be offered screening for HBsAg in the first trimester, and this is highly recommended in all Canadian jurisdictions. Additionally, reflex HBV DNA testing should be done to reduce cost and delays in assessment to determine whether antiviral therapy is required during pregnancy. Prenatal screening for HCV antibody (anti-HCV) should be implemented which has been shown to be cost-effective and identify individuals that would have been missed with a risk-based screening strategy (110). This is now recommended in recent guidelines from the Society of Obstetricians and Gynaecologists of Canada (111). All pregnant women should be assessed for active hepatitis and liver disease using non-invasive testing (i.e., liver stiffness measurement (LSM), or ultrasound with shear wave elastography (SWE)) in early pregnancy. Although there is data on LSM and SWE in pregnancy, assessment should be repeated post-partum as a baseline assessment (112).
Antiviral treatment
All pregnant women found to have advanced liver disease (i.e., > stage 1 fibrosis), or meet criteria for treatment, regardless of mother-to-child transmission (MTCT) risk, should be immediately offered therapy with NA that is safe to use in pregnancy (i.e., TDF). Although most women will not have significant HBV, related liver disease, given the well-known safety profile of NA, it is reasonable to discuss benefits of treatment in the context of long-term family planning. Ideally, all women of child-bearing age should be assessed prior to pregnancy to assess for HBV viremia and significant liver disease. Those already on tenofovir-based treatment should continue treatment when they become pregnant, as stopping can lead to detrimental viral and biochemical (ALT) hepatitis flares. There is robust evidence supporting antiviral treatment in all women with HBV DNA >200,000 IU/mL starting in the second trimester (113). Individuals with HBV DNA >200,000 IU/mL and planning a pregnancy in the near future (i.e., 1–2 years) may be offered the opportunity to start treatment before pregnancy or well before the third trimester. Several recent reviews have highlighted clinical and basic science studies showing potential risk of in utero infection (114). Pragmatically, earlier treatment may ensure adherence, and that the patient is able to tolerate therapy in pregnancy and achieve viral load that is well below the threshold associated with MTCT risk at the time of birth. This broader approach will reduce potential unexpected adverse events and risks with a complicated pregnancy, amniocentesis in the second trimester in highly viremic mothers, placental barrier leakage, multiple births, and pre-term birth. The average age for Canadian women to have their first child has increased to 32 years, the risk of pre-term delivery increases over age 35, which may also be compounded in patients with chronic HBV (115,116). The WHO has proposed that in resource limited regions with difficulties accessing infant HBIG and even HBV DNA monitoring, a strategy of treating all HBsAg-positive women of child-bearing age should be adopted (10). In high-resource countries such as Canada, while treatment consideration may be expanded, this may be too liberal since there are few concerns with cold-chain supply and most infants are born in hospital and can receive timely HBV immunoprophylaxis.
The recommended first-line treatment is TDF (117,118), and recent data also show TAF is safe during pregnancy (119, 120, 121). Lamivudine can be used if there is no access to TDF or TAF but is not recommended for long-term use due to risk of antiviral resistance. ETV is not recommended due to the lack of robust safety data. Individuals planning future pregnancies or who become pregnant on ETV should be switched to tenofovir. Interferon (i.e., PEG-IFN) is contraindicated in pregnancy. Breastfeeding is safe and should be encouraged; there is minimal excretion of NA in breast milk and no reports of HBV transmission via human breast milk (122,123).
Monitoring and follow-up
All patients, regardless of treatment, should be monitored during pregnancy and post-partum with HBV DNA and ALT every 3–6 months due to risk of post-partum flares (124). Individuals on treatment should undergo annual assessment of renal function (i.e., eGFR). In women who receive treatment solely to reduce MTCT, treatment can be continued until 3 months (12 weeks) post-partum to reduce the risk of post-partum off-treatment flares. In patients who are considering future pregnancies (i.e., within 3–5 years), continued treatment until they are finished family planning may be considered. Otherwise, all women who stop treatment should be closely monitored with HBV DNA and ALT.
Recommendations for HBV management in pregnancy
All HBsAg-positive persons of child-bearing age planning pregnancy in near future should be assessed (HBV DNA, HBeAg, antibody to HBeAg [anti-HBe], ALT, Fibrosis) and counselled about HBV management. (Strong recommendation; Level 1)
All pregnant persons should be screened for HBsAg in the first trimester of pregnancy. (Strong recommendation; Level 1)
HBsAg-positive pregnant persons should undergo additional assessment for HBeAg, anti-HBe, HBV DNA, and ALT and be referred to a provider with expertise in HBV management. (Strong recommendation; Level 1)
TDF or TAF should be given as the drug of choice for pregnant persons and those of child-bearing potential who require immediate treatment for HBV. (Strong recommendation; Level 1)
HBsAg-positive persons planning to become pregnant in the near future (1–2 years), especially if over age 30 and HBV DNA >200,000 IU/mL, may be considered for immediate treatment, rather than waiting for future pregnancy. (Strong recommendation; Level 2)
HBsAg-positive pregnant persons with HBV DNA of more than 200,000 IU/mL should initiate antiviral therapy as soon as possible and no later than the second trimester to reduce the risk of vertical transmission and to ensure treatment is tolerated. Earlier treatment may be considered in high-risk pregnancies and potential pre-term delivery. (Strong recommendation; Level 2)
TDF or TAF may be continued up to 12 weeks post-partum if given strictly to prevent MTCT. (Strong recommendation; Level 1)
If antiviral treatment is started to reduce MTCT risk, on a case-by-case basis, treatment may be continued until all family planning is completed. (Strong recommendation; Level 5)
After stopping TDF or TAF post-partum, persons should be monitored for ALT flares, with ALT testing every 4 weeks for the first 3 months and then at 6 months, followed by routine monitoring thereafter. (Strong recommendation; Level 2)
All infants born to HBsAg-positive pregnant persons should receive immunoprophylaxis with HBIG and HBV vaccine as soon as possible after birth, and completion of second and third HBV vaccine doses before 6 months. (Strong recommendation; Level 1)
Infants should be tested for HBsAg and anti-HBs between 1 and 4 months after the last dose of vaccine (by age 1 year) to confirm that they are uninfected and immune to HBV. (Strong recommendation; Level 1)
Breastfeeding while receiving TDF or TAF is not contraindicated. (Strong recommendation; Level 1)
Management of HBV Co-Infections
HBV and HDV coinfection (CS Coffin)
HDV causes the most aggressive form of viral hepatitis in humans. Due to shared blood-borne routes of transmission, simultaneous coinfection with HBV and HDV can occur, as well as superinfection of HDV in HBsAg-positive persons (125). The HDV requires the HBV to complete the viral lifecycle, and in clinical practice, assessment of HDV coinfection is only recommended in HBsAg-positive persons. However, case reports of HDV in HBsAg-negative/anti-HBc-positive persons have been described, suggesting low levels of HBsAg (below limits of detection using standard commercial HBsAg ELISAs) may be sufficient for HDV replication (126,127). In these rare circumstances, discussion with a medical microbiologist may be required regarding screening.
Historically there were limited prevalence data on HDV in Canada. Since the last CASL HBV guidelines, updated Canadian epidemiological studies have been published. A retrospective serosurvey of referred HDV testing to the National Microbiology Laboratory estimated HDV antibody positivity at ~5.2% (5). A more recent study based on case series, modelling data, and smaller serosurveys provided estimates of 3%–5% (128). Overall, although HDV is rare in Canada (modelling estimates 10,000–15,000 people), the global prevalence is estimated to be 12–72 million infected persons, which may continue to have significant impact from new cases due to immigration from endemic areas (29). Compared to HBV monoinfected patients, individuals enrolled in the Canadian HBV Network registry that were HBV/HDVcoinfected were more likely to have advanced liver disease, cirrhosis and liver cancer. There are no Health Canada-approved therapies for HDV. PEG-IFN is used off-label in HDV RNA-positive persons but is limited by toxicity, several contraindications, and suboptimal efficacy. There is variation in access to PEG-IFN across Canada, but it is generally available through a special authorization program. PEG-IFN treatment is given for 48 weeks, and those who achieve persistent HDV RNA suppression and ALT normalization show improved clinical outcomes over long-term (10 years) follow-up off-treatment (129,130). Although longer duration (even up to 4 years) has been associated with potential improved response, it is generally not recommended due to toxicity and limited success rates (131). HBV NA are not active against HDV, but treatment is recommended in all HBV/HDV coinfected patients. In theory, the aggressive suppression of HBV replication and prevention of HBV flares in HDV coinfection may help reduce the risk of liver disease progression (132). Early identification of HDV infection will facilitate closer monitoring and enrolment in HCC surveillance programs. Due to viral interactions, many persons with active HDV replication have suppressed HBV viremia and may be misdiagnosed with inactive chronic HBV (i.e., immune control phase). Thus, reflex testing of all HBsAg-positive individuals for antibody to HDV would prevent missed diagnoses of HDV infection, especially by health care providers not familiar with the disease. Thus, updated guidelines advocate for reflex universal one-time screening of all HBsAg-positive individuals and reflex HDV RNA testing in all anti-HDV-positive patients (see Hepatitis B and D screening and HBV vaccination section).
The proposed diagnostic assays and laboratory algorithm are provided in the Hepatitis B and D screening and HBV vaccination section. Repeat HDV antibody testing is not recommended, unless clinical concern and an ongoing risk for superinfection (i.e., HBsAg-positive individual in close contact HBV/HDV coinfected person), as even late transmission can occur leading to fulminant hepatitis (133). The HBV vaccine also protects against HDV, but an HDV-specific vaccine would be of global benefit for the 300 million people with chronic HBV infection.
New therapies for HDV are advancing and revolutionizing the management of this historically difficult to treat disease. Bulevirtide (BLV) is a peptide mimic of the HBV pre-S1 protein and competitively binds to the HBV and host bile acid binding receptor (NTCP), blocking both HDV and HBV entry. BLV was approved by the European Medicines Agency in 2020 for compensated chronic HDV infection and designated orphan drug status by the US Food and Drug Administration. Published randomized controlled trials have shown that BLV at a 2 mg or 10 mg subcutaneous daily dose, given alone or in combination with PEG-IFN, leads to improvement, in liver enzymes, suppression of HDV RNA, histological improvement, and lowering of intrahepatic HDV replication (134, 135, 136). Virological and biochemical response was similar with the 2 mg and 10 mg dose and improved with addition of PEG-IFN (136). Real world studies of BLV monotherapy (given off-label) in persons with decompensated cirrhosis leads to clinical improvement, including resolution of ascites and portal hypertension (137,138). The European Association for the Study of the Liver has published the first dedicated guidelines for HDV management and recommend BLV as a first-line therapy for all persons with HDV infection (13). The duration of BLV treatment is unclear (139) and stated, as long as clinical benefit, can be demonstrated. Since HDV is an RNA virus, without a reservoir, in theory with long-term suppression, hepatocyte turnover would lead to viral clearance. A case report has described cure of HDV infection in an individual with compensated cirrhosis and esophageal varices with clearance of intrahepatic hepatitis delta antigen and HDV RNA levels after 3 years of continuous BLV monotherapy (140). BLV is well tolerated with only mild injection site reactions. Due to competitive inhibition of NTCP, treatment is associated with an asymptomatic increase in bile acids.
A complete list of novel HDV therapies can be found at www.hepb.org/drugwatch. Small phase 2 studies using nucleic acid polymers (NAPS) to block HBsAg release are administered by intravenous or subcutaneous injection. These studies showed long-term HBsAg loss (i.e., functional cure of HDV and HBV) in 7 out of 11 participants (141,142). Recent real world/off-label data with NAPS showed clinical improvement with regression of fibrosis and resolution of portal hypertension (143). Other emerging anti-HDV drugs that have progressed through phase 3 clinical trials (D-LIVR) include a host prenylation inhibitor (Lonafarnib, LNF) boosted with ritonavir (RTV) and offers the advantage of oral dosing (144,145).
Summary of recommendations for HBV and HDV management
All HBsAg-positive/HDV antibody-positive individuals should be considered for anti-HBV NA therapy, regardless of HBV DNA levels or fibrosis stage, but especially if there is detectable viremia. NA are not directly active against HDV but, in theory, will block residual HBV replication which may reduce liver disease risk. (Strong recommendation; Level 2)
PEG-IFN therapy (180 µg once weekly) for 48 weeks should be considered in individuals without contraindications to IFN treatment. (Strong recommendation; Level 1)
As novel HDV therapies (i.e., BLV) are expected to become available in Canada, they should be prioritized for HBV/HDV coinfected individuals, particularly those with advanced liver disease with or without IFN combination therapy. (Strong recommendation; Level 1)
Due to the high risk of rapid disease progression, all HBV/HDV coinfected individuals should be followed by a specialist and monitored closely with HBV DNA, HDV RNA, ALT every 6–12 months, HCC surveillance starting at age 40 (see above), and fibrosis assessment yearly. Late HDV RNA relapse has been described even in those with suppressed HDV RNA on PEG-IFN. (Strong recommendation; Level 2)
HBV and HCV coinfection (CL Cooper)
Due to shared risk factors, HBV and HCV can be found concurrently (146,147). Fibrosis progression is accelerated, and HCC risk is increased (148). Screening for one is recommended in the presence of the other. There is a risk of HBV flare in HBsAg-positive individuals initiating direct-acting antiviral therapy (DAA) for HCV coinfection (149, 150, 151). This risk is low and generally limited to enzymatic and HBV virological laboratory changes but can, in its most severe form, result in clinical disease including fulminant hepatitis (152,153). Co-initiation of an HBV antiviral concurrent with DAA dosing should be considered with HBV nucleoside therapy maintained for 12 weeks following anti-HCV DAA completion (154). Irrespective of whether an HBV antiviral is initiated, liver enzymes should be monitored at least every 4 weeks and HBV DNA levels every 3 months while on DAA therapy, up to 24 weeks following completion. Similarly, ALT monitoring is advised for HBsAg-negative/anti-HBc-positive individuals, especially if cirrhotic. The incidence of HBsAg and/or HBV DNA re-emergent with DAA treatment is uncommon (reported <10% low level HBV viremia) in the anti-HBc-positive population (154, 155, 156).
Recommendations for management of HBV and HCV coinfection
All HCV patients should be tested for HBsAg and anti-HBc prior to initiating HCV DAA therapy. (Strong recommendation; Level 2)
All HBsAg-positive patients, especially those with advanced fibrosis or cirrhosis, should initiate oral anti-HBV NA treatment while undergoing HCV DAA therapy. Empiric NA treatment is recommended given possible delays in HBV DNA testing in some regions. (Strong recommendation; Level 3)
HBsAg-negative and anti-HBc positive (with or without anti-HBs) patients should be monitored for ALT and HBsAg while on HCV DAA until 24 weeks post-treatment. HBsAg and HBV DNA should be measured if ALT fails to normalize or increases during or after completion of DAA treatment. HBV antiviral therapy should be initiated promptly in those with emergence of HBsAg or detectable HBV DNA with elevated ALT. (Strong recommendation; Level 3)
HBV and HIV coinfection (CL Cooper)
Approximately 6%–8% of Canadians living with HIV are HBV co-infected (157,158). Coinfection is associated with more rapid liver fibrosis progression and a heavy burden of HCC, cirrhosis, and end-stage liver disease (159). Antivirals including TDF, TAF, and lamuvidine possess activity against both HIV and HBV. As such, they should be included in HIV antiretroviral regimens. Treatment under the care of physicians with HIV and HBV expertise should be initiated once the diagnosis of HIV and HBV is established and irrespective of CD4+ T cell count. Monitoring for HBV-specific immune reconstitution during the initial 3 months following ARV initiation is required (160,161). However, inclusion of antiretrovirals with HBV activity protect against this risk. There is a small risk for proximal tubular nephropathy (i.e., Fanconi Syndrome) and osteopenia with the use of TDF. This risk is much lower with TAF (162,163). Data demonstrates HIV and HBV efficacy as well as safety in HIV-HBV = coinfected TAF recipients (164,165). Therefore, TAF represents a first-line antiviral in the context of HIV-HBV coinfection.
Dolutegravir/lamivudine represents a first line option for treatment-naïve HIV patients as well as a switch option for patients with fully suppressed HIV viremia (166,167). This is not a first-line option in HIV-HBV-coinfected individuals. If used as part of a switch strategy from a TDF/TAF-containing regimen to dolutegravir/lamivudine, then increased HBV DNA monitoring is recommended to ensure that HBV viremia remains fully suppressed (168).
Cabotegravir/rilpivirine represents an intramuscular injectable alternative to orally administered HIV antiretroviral therapy (169,170). It possesses no HBV antiviral activity. Therefore, if used as part of a switch strategy from a TDF/TAF/lamivudine-containing HIV regimen to Cabotegravir/Rilpivirine then an HBV antiviral should be concurrently initiated to ensure continued HBV antiviral suppression. If Cabotegravir/Rilpivirine is started in an HIV-HBV-coinfected patient not on HIV or HBV active antivirals, then an HBV antiviral should be concurrently initiated to ensure appropriate HBV management is implemented.
Some populations of HIV infected individuals are at greater risk for exposure to HBV (e.g., men who have sex with men or persons who inject drugs (171)). All people living with HIV should be screened for HBV infection and immunity. If anti-HBs negative, then HBV immunization should be pursued as it is effective in preventing infection (see Hepatitis B and D screening and HBV vaccination section) (172). HIV/HBV coinfection is not a contraindication to liver transplantation, assuming all other eligibility criteria are met. Post-transplant outcomes, including survival, are comparable to HBV monoinfection (173,174). Additionally, in HBsAg-positive individuals who would potentially benefit from continuous HIV pre-exposure prophylaxis (i.e., PrEP with TDF and emtricitabine), PrEP may be initiated, regardless of HBV status, but HBV should be closely monitored for virological and biochemical flares, if and when PrEP is discontinued.There is no risk of anti-HBV resistance with TDF treatment interruptions.
Recommendations for management of HBV and HIV coinfection
All people living with HIV should be screened for HBV infection and immunity. If anti-HBs and anti-HBc negative, then HBV immunization should be pursued. (Strong recommendation; Level 1)
HIV treatment with HBV-active antivirals should be initiated once the diagnosis of HIV and HBV is established and irrespective of CD4+ T cell count. (Strong recommendation; Level 1)
On-treatment monitoring of HBV is the same as in HBV monoinfection. (Strong recommendation; Level 5)
Patients who have interruptions in HIV therapy that included any HBV active antivirals should be monitored for HBV reactivation if unable to continue with anti-HBV antiviral. (Strong recommendation; Level 4)
All HIV-HBV coinfected patients starting or switching to Cabotegravir/Rilpivirine intramuscular injection treatment for HIV should ensure that HBV antiviral therapy is concurrently initiated or continued. (Strong recommendation; Level 5)
Management of Hepatitis B in Pediatric Patients (F Alvarez)
Prevalence of HBV infection in Canada is relatively low, and incidence is decreasing in pediatric patients thanks to different preventive measures against vertical transmission and vaccination. Vaccination programs in many countries have also decreased the incidence in immigrant children; however, updated data for the pediatric population is needed (175).
Natural history of HBV infection in pediatrics
Most children are asymptomatic; acute hepatitis and its fulminant form are rare events. In patients infected by horizontal transmission, HBeAg seroconversion is observed before age 10 years, in contrast with those infected by vertical transmission, in whom delayed HBeAg seroconversion occurs around puberty (176,177).
Three markers predict HBeAg to anti-HBe seroconversion: increase in serum transaminases, decrease of circulating HBV-DNA, and evidence of inflammatory activity by liver histology. The presence of fibrosis at liver biopsy is infrequent, and the incidence of cirrhosis is around 2%–3%. Half of these children develop HCC many years after HBe seroconversion, an event that very rarely occurs in those without cirrhosis (178,179).
Currently, liver biopsies are rarely indicated in children with chronic HBV infection. Ultrasound-based transient elastography (FibroScan), used to establish the elasticity of the liver and indirectly the degree of fibrosis, can be carried out in children after 3–4 years of age. Regression of fibrosis is observed in patients with HBeAg and HBsAg seroconversion.
HBeAg seroconversion is observed in around 85% of children before the age of 18 years, spontaneously or after treatment. Only 20% of children, whether treated or untreated, seroconvert from HBsAg positive to negative (176).
HDV coinfection is rare and observed mainly in children born in regions with high or intermediate HDV prevalence. These patients show poor prognosis, developing rapid progression to cirrhosis (180).
Almost one in seven children or youth are obese in Canada (181). Childhood obesity is a risk factor for many chronic diseases including steatotic liver disease and/or MASLD in pediatric patients. Further study is required to understand the natural history of MASLD and HBV in children to inform management guidelines. The Canadian Laboratory Initiative on Pediatric Reference Intervals (CALIPER) establishes age- and sex-specific reference intervals, including ALT cut-offs, to improve diagnostic accuracy and disease monitoring in pediatric populations. These intervals are widely adopted by clinical laboratories across Canada.
HBV Treatment in Children
Summary of indications for HBV treatment in children
Two readings of serum ALT levels within 6–12 months over the ULN in a child with elevated HBV DNA levels (ALT >1.5 times normal, during 1 year of observation; HBV DNA >2,000 IU/mL; no liver biopsy required).
Children with cirrhosis, HBV-associated extrahepatic complications (glomerulonephritis, arteritis nodosa), or HIV, HCV, or HDV co-infection and detectable HBV DNA (no liver biopsy required). In those receiving immunosuppressive drugs: Long-term steroids use, solid organ or bone marrow transplantation, treatment of autoimmune diseases, or chemotherapy.
Moderate to severe liver inflammation with >F1 stage fibrosis fibrosis on liver biopsy.
Family history of HCC in children with HBV DNA >2,000 IU/mL.
Liver transplant in HBV-infected recipients, recipient of anti-HBc positive donor organ.
Summary of HBV treatment options in pediatrics
Entecavir and tenofovir are the molecules of choice for the treatment of children with HBV, since they induce a low frequency of viral mutations with few adverse events.
ETV has been used in children older than 2 years of age at a dose of 0.015 mg/kg (maximum of 0.5 mg/day) in children weighing less than 32.5 kg and 0.5 mg/day in those weighing more than 32.6 kg for more than 1 year. HBeAg seroconversion and HBV DNA of less than 50 IU/mL were found in 24.2% versus 3.3% in those receiving placebo at week 48 (p < 0.0008) (182). Emergent resistance was 0.6% and 2.6% at the end of the first and second years of treatment, respectively. Comparable adverse events were reported between those receiving ETV and placebo.
TDF has been used with children older than 2 years of age or having a weight greater than 10 kg. Children older than 12 years of age may be treated with TDF at a dose of 300 mg daily for more than 1 year. After 42 weeks of treatment, HBeAg seroconversion was 21% versus 15% (nonsignificant) in the placebo group. No particular adverse event was recorded in treated patients; however, monitoring of renal function was suggested (183). Long-term TDF can affect bone health; otherwise, studies show that TAF and ETV are safe in pediatrics (184,185).
Monitoring of children with HBV
All children with chronic HBV infection should be monitored annually with complete blood cell count, serum aminotransferases, AFP, HBV profile (HBV serology and HBV DNA), and liver ultrasound and liver stiffness measurement using transient elastography (i.e., TE or FibroScan). TE is endorsed by the WHO based on available evidence suggesting that TE is effective in children although further study is required to determine specific cut-off values (i.e., >7 kpA = F2 fibrosis or >12 kpA = cirrhosis or stage 4). Other non-invasive tests to assess fibrosis include APRI (i.e., >0.5) , regardless of HBV DNA levels. In general, whether HBeAg negative or positive, children with elevated ALT and HBV DNA should be monitored more frequently (i.e., monthly to every 3 months depending on severity of ALT flare). Children who are on NA treatment should be monitored every 3 months. Due to limited data, the current guidelines do not differentiate between children aged 2–11 years and >12 years, hence this is an area requiring further study to understand variations in treatment response and natural history.
Recommendations for management of HBV in pediatric patients
There is no strong evidence at present for treatment of children without complications in the high replicative/non-inflammatory phase (formerly referred to as immune-tolerant) and HBeAg-negative chronic HBV infection with normal ALT (<25 in females, <35 in males) and low level HBV DNA (<2,000 IU/mL) (formerly referred to as inactive chronic carrier phase). (Moderate recommendation; Level 4)
Due to the long-term safety profile of NA, the potential development of curative treatments in the next decade, as well as novel predictive biomarkers of HBV in the liver, on an individualized basis regardless of HBV or ALT levels, taking into account concomitant MASLD and diabetes, a strong family history of HCC or cirrhosis, and infection with genotype C, treatment may be considered in adolescent patients with very high HBV viral load (high replicative/non-inflammatory phase, formerly referred to as immune tolerant phase), under the care of a specialist. (Moderate recommendation; Level 3)
Children with chronic HBV infection, either HBeAg positive or negative, should be followed once a year by ultrasound examination of the liver (ideally with elastography) and serum AFP, in addition to ALT and HBV DNA. (Moderate recommendation; Level 3)
In HBeAg-positive children with serum ALT more than 1.3–2 times the normal values, monitoring every 3 months for at least 1 year, to document decreasing or low levels of serum HBV DNA, is recommended before considering treatment to account for possible spontaneous seroconversion. (Strong recommendation; Level 1)
In HBeAg-negative children ALT above the ULN (>25 in females and >35 in males) on 2 occasions measured 6 months apart and HBV DNA >2,000 IU/mL should be considered for treatment. (Strong recommendation, Level 1)
Treatment should be initiated for children with advanced fibrosis and cirrhosis (based on clinical findings or non-invasive tests) to lower HCC risk. (Strong recommendation; Level 3)
Treatment should be considered for children with extrahepatic disease and coinfection (i.e., HIV, HDV, HCV). (Strong recommendation; Level 2)
All pediatric patients undergoing immunosuppression should be assessed regarding need for HBV treatment (similar to adults). (Strong recommendation; Level 1)
ETV, TDF, and TAF are drugs that are orally administered and exhibit a high genotypic barrier to resistance and a favourable side effect profile. NAs should be continued for 1 year after the disappearance of HBeAg and the appearance of anti-HBe (seroconversion) before attempting cessation. (Strong recommendation; Level 1)
After seroconversion, whether spontaneous or treatment induced, children should be followed every 3 months for at least 1 year. After confirmation of immune inactive status (persistently normal ALT, HBV DNA <2,000 IU/mL), monitoring should be continued every 6 months. (Strong recommendation; Level 2)
Summary and Future Directions
The 2025 CASL/AMMI guidelines provide updated recommendations for the management of chronic HBV in Canada, with a focus on universal adult screening, catch-up vaccination, broader treatment access, and reflex testing for HBD. Reflecting current evidence and aligning with international guidelines, it supports the use of quantitative HBsAg (qHBsAg) testing to guide treatment decisions and promote functional cure. These guidelines also endorse simplified diagnostic pathways and earlier treatment initiation, particularly for individuals who do not clearly meet traditional criteria for treatment (grey zone). Future research priorities include evaluating the cost-effectiveness of screening and vaccination strategies, validating emerging biomarkers, and advancing new antiviral therapies targeting functional or sterilizing cure. To support these efforts, provincial and territorial health systems are encouraged to invest in enhanced HBV surveillance, improved access to recommended HBV/HDV laboratory tests, and the implementation of point-of-care diagnostics, within a patient-centred approach aimed at reducing inequities in access to care and treatment.
Funding Statement
N/A
Ethics Approval:
N/A
Informed Consent:
N/A
Registry and the Registration No. of the Study/Trial:
N/A
Data Accessibility:
N/A
Funding:
N/A
Disclosures:
N/A
Peer Review:
This manuscript was submitted for open feedback by members of the Canadian Association for the Study of the Liver and Association of Medical Microbiologists and Infectious Disease Canada.
Animal Studies:
N/A
Supplemental Material
References
- 1.World Health Organization. Global health sector strategy on viral hepatitis 2016-2021. Towards ending viral hepatitis. https://iris.who.int/handle/10665/246177 [Accessed Jun 05, 2016]. [Google Scholar]
- 2.Public Health Agency of Canada. Canada's sexually transmitted and blood-borne infections (STBBI) action plan 2024–2030. 2024. https://www.canada.ca/en/public-health/services/publications/diseases-conditions/sexually-transmitted-blood-borne-infections-action-plan-2024-2030.html [Accessed Mar 26, 2024].
- 3.Public Health Agency of Canada. Canada's progress towards eliminating viral hepatitis as a public health threat, 2021. 2024. https://www.canada.ca/en/public-health/services/publications/diseases-conditions/progress-towards-eliminating-viral-hepatitis-threat-2021.html [Accessed Sep 17, 2024].
- 4.Government of Canada, Public Health Agency of Canada. Reported cases from 1924 to 2021 in Canada - Notifiable diseases on-line. 2021. https://diseases.canada.ca/notifiable/charts?c=pl [Accessed Mar 27, 2024].
- 5.Osiowy C, Swidinsky K, Haylock-Jacobs S, et al. Molecular epidemiology and clinical characteristics of hepatitis D virus infection in Canada. JHEP Rep. 2022;4:100461. 10.1016/j.jhepr.2022.100461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Coffin CS, Ramji A, Cooper CL, et al. Epidemiologic and clinical features of chronic hepatitis B virus infection in 8 Canadian provinces: A descriptive study by the Canadian HBV network. CMAJ Open. 2019;7:E610–7. 10.9778/cmajo.20190103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Statistics Canada. Immigration and ethnocultural diversity statistics. 2024. https://www.statcan.gc.ca/en/subjects-start/immigration_and_ethnocultural_diversity [Accessed Apr 02, 2021].
- 8.Statistics Canada. Immigrants make up the largest share of the population in over 150 years and continue to shape who we are as Canadians. 2022. https://www150.statcan.gc.ca/n1/daily-quotidien/221026/dq221026a-eng.htm [Accessed Aug 02, 2023].
- 9.Action Hepatitis Canada. Immigration Health is Public Health. 2024. https://www.actionhepatitiscanada.ca/immigrationhealth.html [Accessed Sep 17, 2024].
- 10.World Health Organization. Guidelines for the prevention, diagnosis, care and treatment for people with chronic hepatitis B infection. 2024. https://www.who.int/publications/i/item/9789240090903 [Accessed May 15, 2024]. [PubMed]
- 11.Adda D, Wang S. A declaration from people living with hepatitis B: A call for a whole person approach. J Viral Hepat. 2023;30:630. 10.1111/jvh.13839. [DOI] [PubMed] [Google Scholar]
- 12.Centre for Evidence-Based Medicine. OCEBM Levels of Evidence. 2011. https://www.cebm.ox.ac.uk/resources/levels-of-evidence/ocebm-levels-of-evidence [Accessed May 12, 2024].
- 13.European Association for the Study of the Liver. EASL clinical practice guidelines on hepatitis delta virus. J Hepatol. 2023;79:433–460. 10.1016/j.jhep.2023.05.001. [DOI] [PubMed] [Google Scholar]
- 14.Coffin CS, Fung SK, Alvarez F, et al. Management of hepatitis B virus infection: 2018 guidelines from the Canadian Association for the Study of Liver Disease and Association of Medical Microbiology and Infectious Disease Canada. Can Liver J. 2018;1:156–217. 10.3138/canlivj.2018-0008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Razavi-Shearer D, Gamkrelidze I, Pan CQ, et al. The impact of immigration on hepatitis B burden in the United States: A modelling study. Lancet Reg Health Am. 2023;22:100516. 10.1016/j.lana.2023.100516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Conners EE, Panagiotakopoulos L, Hofmeister MG, et al. Screening and testing for hepatitis B virus infection: CDC recommendations - United States, 2023. MMWR Recomm Rep. 2023;72:1–5. 10.15585/mmwr.rr7201a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Saitta C, Pollicino T, Raimondo G. Occult hepatitis B virus infection: An update. Viruses. 2022;14:1504. 10.3390/v14071504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gish RG, Basit SA, Ryan J, Dawood A, Protzer U. Hepatitis B core antibody: Role in clinical practice in 2020. Curr Hepatology Rep. 2020;19:254–265. 10.1007/s11901-020-00522-0. [DOI] [Google Scholar]
- 19.Lapointe-Shaw L, Chung H, Holder L, et al. Diagnosis of chronic hepatitis B pericomplication: Risk factors and trends over time. Hepatology. 2021;73:2141–2154. 10.1002/hep.31557. [DOI] [PubMed] [Google Scholar]
- 20.Weng MK, Doshani M, Khan MA, et al. Universal hepatitis B vaccination in adults aged 19–59 years: Updated recommendations of the advisory committee on immunization practices — United States, 2022. MMWR Morb Mortal Wkly Rep. 2022;71:477–483. 10.15585/mmwr.mm7113a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wong WWL, Woo G, Heathcote EJ, Krahn M. Cost effectiveness of screening immigrants for hepatitis B. Liver Int. 2011;31:1179–1190. 10.1111/j.1478-3231.2011.02559.x. [DOI] [PubMed] [Google Scholar]
- 22.Rossi C, Schwartzman K, Oxlade O, Klein MB, Greenaway C. Hepatitis B screening and vaccination strategies for newly arrived adult Canadian immigrants and refugees: A cost-effectiveness analysis. PLoS One. 2013;8:e78548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Palom A, Rando-Segura A, Vico J, et al. Implementation of anti-HDV reflex testing among HBsAg-positive individuals increases testing for hepatitis D. JHEP Rep. 2022;4:100547. 10.1016/j.jhepr.2022.100547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Parfut A, Tripon S, Gantner P, et al. Impact of anti-HDV reflex testing at HBs antigen positive discovery in a single center france: Support for primary HDV screening in france. J Clin Virol. 2024;171:105650. 10.1016/j.jcv.2024.105650. [DOI] [PubMed] [Google Scholar]
- 25.Cossiga V, Brusa S, Montalti R, et al. Anti-HDV reflex testing in HBsAg-positive subjects: An efficacious strategy to identify HDV infection. Liver Int. 2024;44:148–154. 10.1111/liv.15746. [DOI] [PubMed] [Google Scholar]
- 26.Wong RJ, Kaufman HW, Niles JK, et al. Low performance of hepatitis delta virus testing among 2 national cohorts of chronic hepatitis B patients in the United States. Am J Gastroenterol. 2022;117:2067–2070. 10.14309/ajg.0000000000001947. [DOI] [PubMed] [Google Scholar]
- 27.Lai JC, Wong GL, Wong VW, Yip TC. Unmet need in screening for hepatitis D virus: Time to take action. J Hepatol. 2024;80:e277–8. 10.1016/j.jhep.2023.12.006. [DOI] [PubMed] [Google Scholar]
- 28.Razavi HA, Buti M, Terrault NA, et al. Hepatitis D double reflex testing of all hepatitis B carriers in low-HBV- and high-HBV/HDV-prevalence countries. J Hepatol. 2023;79:576–580. 10.1016/j.jhep.2023.02.041. [DOI] [PubMed] [Google Scholar]
- 29.Polaris Observatory Collaborators. Adjusted estimate of the prevalence of hepatitis delta virus in 25 countries and territories. J Hepatol. 2024;80:232–242. 10.1016/j.jhep.2023.10.043. [DOI] [PubMed] [Google Scholar]
- 30.Abbas Z, Abbas M. Is there a need for universal double reflex testing of HBsAg-positive individuals for hepatitis D infection? World J Hepatol. 2024;16:300–303. 10.4254/wjh.v16.i3.300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Stockdale AJ, Kreuels B, Henrion MY, et al. The global prevalence of hepatitis D virus infection: Systematic review and meta-analysis. J Hepatol. 2020;73:523–532. 10.1016/j.jhep.2020.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pan Z, Chen S, Xu L, et al. Diagnostic efficacy of serological antibody detection tests for hepatitis delta virus: A systematic review and meta-analysis. Viruses. 2023;15:2345. 10.3390/v15122345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chang L, Yan Y, Ji H, et al. Low seroprevalence of hepatitis delta virus co-infection in hepatitis B virus-infected blood donors in China: A multicenter study. Front Microbiol. 2022;13:992817. 10.3389/fmicb.2022.992817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chow S, Atienza EE, Cook L, et al. Comparison of enzyme immunoassays for detection of antibodies to hepatitis D virus in serum. Clin Vaccine Immunol. 2016;23:732–734. 10.1128/CVI.00028-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rocco C, Bonavolta R, Vallefuoco L, et al. Comparison of anti-hepatitis D virus (HDV) ETI-AB-DELTAK-2 assay and the novel LIAISON® XL MUREX anti-HDV assay in the diagnosis of HDV infection. Diagn Microbiol Infect Dis. 2019;95:114873. 10.1016/j.diagmicrobio.2019.114873. [DOI] [PubMed] [Google Scholar]
- 36.Qiu X, Hadji A, Olivo A, et al. Evaluation of a fully automated high-throughput serology assay for detection of hepatitis D virus antibodies. J Clin Virol. 2024;173:105689. 10.1016/j.jcv.2024.105689. [DOI] [PubMed] [Google Scholar]
- 37.Ghany MG, Lok AS. Functional cure of hepatitis B requires silencing covalently closed circular and integrated hepatitis B virus DNA. J Clin Invest. 2022;132:e163175. 10.1172/JCI163175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ghany MG, Buti M, Lampertico P, Lee HM. 2022 AASLD-EASL HBV-HCV Treatment Endpoints Conference Faculty. Guidance on treatment endpoints and study design for clinical trials aiming to achieve cure in chronic hepatitis B and D: Report from the 2022 AASLD-EASL HBV-HDV treatment endpoints conference. J Hepatol. 2023;79:1254–1269. 10.1016/j.jhep.2023.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Allweiss L, Testoni B, Yu M, et al. Quantification of the hepatitis B virus cccDNA: Evidence-based guidelines for monitoring the key obstacle of HBV cure. Gut. 2023;72:972–983. 10.1136/gutjnl-2022-328380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Feld JJ, Lok AS, Zoulim F. New perspectives on development of curative strategies for chronic hepatitis B. Clin Gastroenterol Hepatol. 2023;21:2040–2050. 10.1016/j.cgh.2023.02.032. [DOI] [PubMed] [Google Scholar]
- 41.Yang R, Song G, Guan W, Wang Q, Liu Y, Wei L. The Lumipulse G HBsAg-quant assay for screening and quantification of the hepatitis B surface antigen. J Virol Methods. 2016;228:39–47. 10.1016/j.jviromet.2015.11.016. [DOI] [PubMed] [Google Scholar]
- 42.Prakash A, Ponnuvel S, Devadasan JDC, et al. ARCHITECT HBsAg next assay is positioned better to resolve and refine challenging weak reactive clinical samples. J Clin Virol. 2023;166:105524. 10.1016/j.jcv.2023.105524. [DOI] [PubMed] [Google Scholar]
- 43.Wong DK, Inoue T, Mak L, et al. A longitudinal study to detect hepatitis B surface and core-related antigens in chronic hepatitis B patients with hepatitis B surface antigen seroclearance using highly sensitive assays. J Clin Virol. 2023;160:105375. 10.1016/j.jcv.2022.105375. [DOI] [PubMed] [Google Scholar]
- 44.Ghany MG, Buti M, Lampertico P, Lee HM. Ultrasensitive HBsAg assays-not yet ready for primetime. J Hepatol. 2024. online ahead of print: 10.1016/j.jhep.2024.03.001. [DOI] [PubMed] [Google Scholar]
- 45.Tu T, Ajoyan H, George J. Novel assays to solve the clinical and scientific challenges of chronic hepatitis B. Clin Liver Dis. 2023;27:837–855. 10.1016/j.cld.2023.05.002. [DOI] [PubMed] [Google Scholar]
- 46.Liu Y, Jeng W, Peng C, Chien R, Liaw Y. Higher end-of-treatment HBsAg levels is associated with later onset but not severe relapse in HBeAg-negative chronic hepatitis B patients stopping antivirals. Aliment Pharmacol Ther. 2024;59:762. 10.1111/apt.17880. [DOI] [PubMed] [Google Scholar]
- 47.Hirode G, Choi HSJ, Chen C, et al. Off-therapy response after nucleos(t)ide analogue withdrawal in patients with chronic hepatitis B: An international, multicenter, multiethnic cohort (RETRACT-B study). Gastroenterology. 2022;162:757–71.e4. 10.1053/j.gastro.2021.11.002. [DOI] [PubMed] [Google Scholar]
- 48.Begré L, Boyd A, Salazar-Vizcaya L, et al. Long-term quantitative hepatitis B surface antigen (HBsAg) trajectories in persons with and without HBsAg loss on tenofovir-containing antiretroviral therapy. HIV Med. 2024;25:291–298. 10.1111/hiv.13561. [DOI] [PubMed] [Google Scholar]
- 49.Lee SK, Kwon JH, Nam SW. Baseline and reduction at 1 year of hepatitis B surface antigenlevel predicts functional cure and low HBsAg titer: A long-termkinetics of hepatitis B surface antigen. J Hepatol. 2024;80:S762 (FRI–364). 10.1016/S0168-8278(24)02131-7. [DOI] [Google Scholar]
- 50.Lazarevic I, Banko A, Miljanovic D, Cupic M. Hepatitis B surface antigen isoforms: Their clinical implications, utilisation in diagnosis, prevention and new antiviral strategies. Pathogens. 2024;13:46. 10.3390/pathogens13010046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zeng Y, Zhu C, Huang E, et al. Detection of serum large and middle hepatitis B virus surface proteins: A novel potential diagnostic and prognostic biomarker for chronic hepatitis B. Clin Chim Acta. 2024;553:117739. 10.1016/j.cca.2023.117739. [DOI] [PubMed] [Google Scholar]
- 52.Meier M, Calabrese D, Suslov A, Terracciano LM, Heim MH, Wieland S. Ubiquitous expression of HBsAg from integrated HBV DNA in patients with low viral load. J Hepatol. 2021;75:840–847. 10.1016/j.jhep.2021.04.051. [DOI] [PubMed] [Google Scholar]
- 53.Grudda T, Hwang HS, Taddese M, et al. Integrated hepatitis B virus DNA maintains surface antigen production during antiviral treatment. J Clin Invest. 2022;132:e161818. 10.1172/JCI161818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Gao N, Guan G, Xu G, et al. Integrated HBV DNA and cccDNA maintain transcriptional activity in intrahepatic HBsAg-positive patients with functional cure following PEG-IFN-based therapy. Aliment Pharmacol Ther. 2023;58:1086–1098. 10.1111/apt.17670. [DOI] [PubMed] [Google Scholar]
- 55.Zeng G, Koffas A, Mak L, Gill US, Kennedy PTF. Utility of novel viral and immune markers in predicting HBV treatment endpoints: A systematic review of treatment discontinuation studies. JHEP Rep. 2023;5:100720. 10.1016/j.jhepr.2023.100720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wu L, Yang Z, Zheng M. Biogenesis of serum HBV RNA and clinical phenomena of serum HBV RNA in chronic hepatitis B patients before and after receiving nucleos(t)ide analogues therapy. J Viral Hepat. 2024. online ahead of print: 10.1111/jvh.13926. [DOI] [PubMed] [Google Scholar]
- 57.Fan R, Peng J, Xie Q, et al. Combining hepatitis B virus RNA and hepatitis B core-related antigen: Guidance for safely stopping nucleos(t)ide analogues in hepatitis B e antigen-positive patients with chronic hepatitis B. J Infect Dis. 2020;222:611–618. 10.1093/infdis/jiaa136. [DOI] [PubMed] [Google Scholar]
- 58.Xie Y, Li M, Ou X, et al. HBeAg-positive patients with HBsAg < 100 IU/mL and negative HBV RNA have lower risk of virological relapse after nucleos(t)ide analogues cessation. J Gastroenterol. 2021;56:856-867. 10.1007/s00535-021-01812-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Xia M, Chi H, Wu Y, et al. Serum hepatitis B virus RNA level is associated with biochemical relapse in patients with chronic hepatitis B infection who discontinue nucleos(t)ide analogue treatment. Aliment Pharmacol Ther. 2021;54:709–714. 10.1111/apt.16538. [DOI] [PubMed] [Google Scholar]
- 60.Kaewdech A, Assawasuwannakit S, Sripongpun P, Chamroonkul N, Tangkijvanich P, Piratvisuth T. Clinical utility of SCALE-B to predict hepatitis B virus relapse, hepatitis B surface antigen loss after antiviral cessation in Asian patients after 2-year follow-up. Front Med (Lausanne). 2022;9:859430. 10.3389/fmed.2022.859430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Yu X, Pfefferkorn M, van Bömmel F, Zhang X, Berg T. Clinical applications of circulating HBV RNA as a potential surrogate biomarker for intrahepatic cccDNA transcriptional activity. Gut. 2024;73:563–566. 10.1136/gutjnl-2023-331217. [DOI] [PubMed] [Google Scholar]
- 62.Deng R, Liu S, Shen S, Guo H, Sun J. Circulating HBV RNA: From biology to clinical applications. Hepatology. 2022;76:1520–1530. 10.1002/hep.32479. [DOI] [PubMed] [Google Scholar]
- 63.Vachon A, Seo GE, Patel NH, et al. Hepatitis B virus serum RNA transcript isoform composition and proportion in chronic hepatitis B patients by nanopore long-read sequencing. Front Microbiol. 2023;14:1233178. 10.3389/fmicb.2023.1233178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wang J, Shen T, Huang X, et al. Serum hepatitis B virus RNA is encapsidated pregenome RNA that may be associated with persistence of viral infection and rebound. J Hepatol. 2016;65:700–710. 10.1016/j.jhep.2016.05.029. [DOI] [PubMed] [Google Scholar]
- 65.Vachon A, Giles E, Patel N, et al. Analytical and clinical validation of 3′ RACE RT-qPCR assay for detection and quantification of hepatitis B virus (HBV) serum RNA. J Clin Virol Plus. 2022;2:100126. 10.1016/j.jcvp.2022.100126. [DOI] [Google Scholar]
- 66.Butler EK, Gersch J, McNamara A, et al. Hepatitis B virus serum DNA and RNA levels in nucleos(t)ide analog-treated or untreated patients during chronic and acute infection. Hepatology. 2018;68:2106–2117. 10.1002/hep.30082. [DOI] [PubMed] [Google Scholar]
- 67.Testoni B, Scholtès C, Plissonnier M, et al. Quantification of circulating HBV RNA expressed from intrahepatic cccDNA in untreated and NUC treated patients with chronic hepatitis B. Gut. 2024;73:659–667. 10.1136/gutjnl-2023-330644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Tseng T, Chiang C, Liu C, et al. Low hepatitis B core-related antigen levels correlate higher spontaneous seroclearance of hepatitis B surface antigen in chronic hepatitis B patients with high hepatitis B surface antigen levels. Gastroenterology. 2023;164:669–79.e6. 10.1053/j.gastro.2023.01.005. [DOI] [PubMed] [Google Scholar]
- 69.Sonneveld MJ, Chiu S, Park JY, et al. Probability of HBsAg loss after nucleo(s)tide analogue withdrawal depends on HBV genotype and viral antigen levels. J Hepatol. 2022;76:1042–1050. 10.1016/j.jhep.2022.01.007. [DOI] [PubMed] [Google Scholar]
- 70.Huang D, Wu D, Wang P, et al. End-of-treatment HBcrAg and HBsAb levels identify durable functional cure after peg-IFN-based therapy in patients with CHB. J Hepatol. 2022;77:42–54. 10.1016/j.jhep.2022.01.021. [DOI] [PubMed] [Google Scholar]
- 71.Beijing Wantai Biological Pharmcy Enterprise. WANTAI HBV NRAg ELISA. 2007. https://www.ystwt.cn/hepatitis-b/ [Accessed Apr 02, 2025].
- 72.Vachon A, Osiowy C. Novel biomarkers of hepatitis B virus and their use in chronic hepatitis B patient management. Viruses. 2021;13:951. 10.3390/v13060951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Wang W, Jia R, Gao Y, et al. Quantitative anti-HBc combined with quantitative HBsAg can predict HBsAg clearance in sequential combination therapy with PEG-IFN-α in NA-suppressed chronic hepatitis B patients. Front Immunol. 2022;13:894410. 10.3389/fimmu.2022.894410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Kan K, Wong DK, Hui RW, Seto WK, Yuen M, Mak L. Anti-HBc: A significant host predictor of spontaneous HBsAg seroclearance in chronic hepatitis B patients - a retrospective longitudinal study. BMC Gastroenterol. 2023;23:348. 10.1186/s12876-023-02983-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Brakenhoff SM, de Knegt RJ, Oliveira J, et al. Levels of antibodies to hepatitis B core antigen are associated with liver inflammation and response to peginterferon in patients with chronic hepatitis B. J Infect Dis. 2023;227:113–122. 10.1093/infdis/jiac210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Jackson K, Gish RG. Point of care diagnostic testing for hepatitis B virus. Curr Hepatology Rep. 2020;19:245–253. 10.1007/s11901-020-00531-z. [DOI] [Google Scholar]
- 77.Mendlowitz AB, Mandel E, Capraru CI, et al. Factors associated with knowledge and awareness of hepatitis B in individuals of Chinese descent: Results from a mass point of care testing and outreach campaign in Toronto, Canada. Can Liver J. 2024;7:28–39. 10.3138/canlivj-2023-0015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Lettner B, Mason K, Greenwald ZR, et al. Rapid hepatitis C virus point-of-care RNA testing and treatment at an integrated supervised consumption service in Toronto, Canada: A prospective, observational cohort study. Lancet Reg Health Am. 2023;22:100490. 10.1016/j.lana.2023.100490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.World Health Organization. Updated recommendations on simplified service delivery and diagnostics for hepatitis C infection. 2022. https://iris.who.int/bitstream/handle/10665/357086/9789240052697-eng.pdf?sequence=1https://iris.who.int/bitstream/handle/10665/357086/9789240052697-eng.pdf?sequence=1 [Accessed Sep 17, 2024]. [PubMed]
- 80.Morin SN, Feldman S, Funnell L, et al. Clinical practice guideline for management of osteoporosis and fracture prevention in Canada: 2023 update. CMAJ. 2023;195:E1333–48. 10.1503/cmaj.221647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Chan HLY, Buti M, Lim Y, et al. Long-term treatment with tenofovir alafenamide for chronic hepatitis B results in high rates of viral suppression and favorable renal and bone safety. Am J Gastroenterol. 2024;119:486–496. 10.14309/ajg.0000000000002468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Fung SK, Pan CQ, Wong GL, et al. Atherosclerotic cardiovascular disease risk profile of patients with chronic hepatitis B treated with tenofovir alafenamide or tenofovir disoproxil fumarate for 96 weeks. Aliment Pharmacol Ther. 2024;59:217–229. 10.1111/apt.17764. [DOI] [PubMed] [Google Scholar]
- 83.Lim Y, Chan HLY, Ahn SH, et al. Tenofovir alafenamide and tenofovir disoproxil fumarate reduce incidence of hepatocellular carcinoma in patients with chronic hepatitis B. JHEP Rep. 2023;5:100847. 10.1016/j.jhepr.2023.100847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Yao K, Liu J, Wang J, et al. Distribution and clinical characteristics of patients with chronic hepatitis B virus infection in the grey zone. J Viral Hepat. 2021;28:1025–1033. 10.1111/jvh.13511. [DOI] [PubMed] [Google Scholar]
- 85.Gan QY, Wang JX, Qian F, et al. Clinical and histological features of patients with chronic hepatitis B virus infection in the grey zone. J Viral Hepat. 2023;30:803–809. 10.1111/jvh.13873. [DOI] [PubMed] [Google Scholar]
- 86.Hsu YC, Chen CY, Chang I-W, et al. Once-daily tenofovir disoproxil fumarate in treatment-naive Taiwanese patients with chronic hepatitis B and minimally raised alanine aminotransferase (TORCH-B): A multicentre, double-blind, placebo-controlled, parallel-group, randomised trial. Lancet Infect Dis. 2021;21:823–833. 10.1016/S1473-3099(20)30692-7. [DOI] [PubMed] [Google Scholar]
- 87.Wang J, Yan X, Zhu L, et al. Significant histological disease of patients with chronic hepatitis B virus infection in the grey zone. Aliment Pharmacol Ther. 2023;57:464–474. 10.1111/apt.17272. [DOI] [PubMed] [Google Scholar]
- 88.Huang DQ, Tran A, Yeh M, et al. Antiviral therapy substantially reduces HCC risk in patients with chronic hepatitis B infection in the indeterminate phase. Hepatology. 2023;78:1558–1568. 10.1097/HEP.0000000000000459. [DOI] [PubMed] [Google Scholar]
- 89.Jeng W, Lok AS. Should treatment indications for chronic hepatitis B be expanded? Clin Gastroenterol Hepatol. 2021;19:2006–2014. 10.1016/j.cgh.2020.04.091. [DOI] [PubMed] [Google Scholar]
- 90.Chan HLY, Chan CK, Hui AJ, et al. Effects of tenofovir disoproxil fumarate in hepatitis B e antigen-positive patients with normal levels of alanine aminotransferase and high levels of hepatitis B virus DNA. Gastroenterology. 2014;146:1240–1248. 10.1053/j.gastro.2014.01.044. [DOI] [PubMed] [Google Scholar]
- 91.Wong VW, Hui AJ, Wong GL, et al. Four-year outcomes after cessation of tenofovir in immune-tolerant chronic hepatitis B patients. J Clin Gastroenterol. 2018;52:347–352. 10.1097/MCG.0000000000000852. [DOI] [PubMed] [Google Scholar]
- 92.Kim G, Lim Y, Han S, et al. High risk of hepatocellular carcinoma and death in patients with immune-tolerant-phase chronic hepatitis B. Gut. 2018;67:945–952. 10.1136/gutjnl-2017-314904. [DOI] [PubMed] [Google Scholar]
- 93.Kim H, Kim G, Park J, Kang H, Lee E, Lim Y. Cost-effectiveness of antiviral treatment in adult patients with immune-tolerant phase chronic hepatitis B. Gut. 2021;70:2172–2182. 10.1136/gutjnl-2020-321309. [DOI] [PubMed] [Google Scholar]
- 94.Yuen M, Lim S, Plesniak R, et al. Efficacy and safety of bepirovirsen in chronic hepatitis B infection. N Engl J Med. 2022;387:1957–1968. 10.1056/NEJMoa2210027. [DOI] [PubMed] [Google Scholar]
- 95.Papatheodoridis GV, Lekakis V, Voulgaris T, et al. Hepatitis B virus reactivation associated with new classes of immunosuppressants and immunomodulators: A systematic review, meta-analysis, and expert opinion. J Hepatol. 2022;77:1670–1689. 10.1016/j.jhep.2022.07.003. [DOI] [PubMed] [Google Scholar]
- 96.Kim H, Yu X, Kramer J, et al. Comparative performance of risk prediction models for hepatitis B-related hepatocellular carcinoma in the United States. J Hepatol. 2022;76:294–301. 10.1016/j.jhep.2021.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Wu X, Xu X, Zhou J, et al. Hepatocellular carcinoma prediction model performance decreases with long-term antiviral therapy in chronic hepatitis B patients. Clin Mol Hepatol. 2023;29:747–762. 10.3350/cmh.2023.0121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Mason WS, Gill US, Litwin S, et al. HBV DNA integration and clonal hepatocyte expansion in chronic hepatitis B patients considered immune tolerant. Gastroenterology. 2016;151:986–98.e4. 10.1053/j.gastro.2016.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Chen C, Yang H, Su J, et al. Risk of hepatocellular carcinoma across a biological gradient of serum hepatitis B virus DNA level. JAMA. 2006;295:65–73. 10.1001/jama.295.1.65. [DOI] [PubMed] [Google Scholar]
- 100.Papatheodoridis G, Dalekos G, Sypsa V, et al. PAGE-B predicts the risk of developing hepatocellular carcinoma in Caucasians with chronic hepatitis B on 5-year antiviral therapy. J Hepatol. 2016;64:800–806. 10.1016/j.jhep.2015.11.035. [DOI] [PubMed] [Google Scholar]
- 101.Kim JH, Kim YD, Lee M, et al. Modified PAGE-B score predicts the risk of hepatocellular carcinoma in Asians with chronic hepatitis B on antiviral therapy. J Hepatol. 2018;69:1066–1073. 10.1016/j.jhep.2018.07.018. [DOI] [PubMed] [Google Scholar]
- 102.Tseng T, Liu C, Yang H, et al. High levels of hepatitis B surface antigen increase risk of hepatocellular carcinoma in patients with low HBV load. Gastroenterology. 2012;142:1140–1149. [DOI] [PubMed] [Google Scholar]
- 103.Lee M, Yang H, Liu J, et al. Prediction models of long-term cirrhosis and hepatocellular carcinoma risk in chronic hepatitis B patients: Risk scores integrating host and virus profiles. Hepatology. 2013;58:546–554. 10.1002/hep.26385. [DOI] [PubMed] [Google Scholar]
- 104.Kedar Mukthinuthalapati VVP, Sewram V, Ndlovu N, et al. Hepatocellular carcinoma in Sub-Saharan Africa. JCO Glob Oncol. 2021;7:756–766. 10.1200/GO.20.00425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Patmore LA, van Eekhout KMA, Buti M, et al. Hepatocellular carcinoma risk in Sub-Saharan African and Afro-Surinamese individuals with chronic hepatitis B living in Europe. J Hepatol. 2024;80:243–250. 10.1016/j.jhep.2023.10.019. [DOI] [PubMed] [Google Scholar]
- 106.Sharma SA, Kowgier M, Hansen BE, et al. Toronto HCC risk index: A validated scoring system to predict 10-year risk of HCC in patients with cirrhosis. J Hepatol. 2017;68:P92–9. 10.1016/j.jhep.2017.07.033. [DOI] [PubMed] [Google Scholar]
- 107.World Health Organization. WHO | Hepatitis B Fact Sheet. 2024. https://www.who.int/news-room/fact-sheets/detail/hepatitis-b [Accessed Jul 05, 2024].
- 108.Biondi MJ, Marchand-Austin A, Cronin K, et al. Prenatal hepatitis B screening, and hepatitis B burden among children, in Ontario: A descriptive study. CMAJ. 2020;192:E1299–305. 10.1503/cmaj.200290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Matthews PC, Ocama P, Wang S, et al. Enhancing interventions for prevention of mother-to-child- transmission of hepatitis B virus. JHEP Rep. 2023;5:100777. 10.1016/j.jhepr.2023.100777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Thompson LA, Plitt SS, Doucette K, et al. Evaluation and comparison of risk-based and universal prenatal HCV screening programs in Alberta, Canada. J Hepatol. 2023;79:1121–1128. 10.1016/j.jhep.2023.05.044. [DOI] [PubMed] [Google Scholar]
- 111.Atkinson A, Bjurman N, Yudin M, Elwood C. Clinical consensus statement no. 458: Hepatitis C virus in pregnancy. J Obstet Gynaecol Can. 2025;47:102780. 10.1016/j.jogc.2025.102780. [DOI] [PubMed] [Google Scholar]
- 112.Stenberg Ribeiro M, Hagström H, Stål P, Ajne G. Transient liver elastography in normal pregnancy - a longitudinal cohort study. Scand J Gastroenterol. 2019;54:761–765. 10.1080/00365521.2019.1629007. [DOI] [PubMed] [Google Scholar]
- 113.Funk AL, Lu Y, Yoshida K, et al. Efficacy and safety of antiviral prophylaxis during pregnancy to prevent mother-to-child transmission of hepatitis B virus: A systematic review and meta-analysis. Lancet Infect Dis. 2021;21:70–84. 10.1016/S1473-3099(20)30586-7. [DOI] [PubMed] [Google Scholar]
- 114.Joshi SS, Coffin CS. Hepatitis B and pregnancy: Virologic and immunologic characteristics. Hepatol Commun. 2020;4:157–171. 10.1002/hep4.1460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Ma X, Sun D, Li C, Ying J, Yan Y. Chronic hepatitis B virus infection and preterm labor(birth) in pregnant women-an updated systematic review and meta-analysis. J Med Virol. 2018;90:93–100. 10.1002/jmv.24927. [DOI] [PubMed] [Google Scholar]
- 116.Xu C, Bao Y, Zuo J, et al. Maternal chronic hepatitis B virus infection and the risk of preterm birth: A retrospective cohort analysis in Chinese women. J Viral Hepat. 2021;28:1422–1430. 10.1111/jvh.13585. [DOI] [PubMed] [Google Scholar]
- 117.Pan CQ, Duan Z, Dai E, et al. Tenofovir to prevent hepatitis B transmission in mothers with high viral load. N Engl J Med. 2016;374:2324–2334. 10.1056/NEJMoa1508660. [DOI] [PubMed] [Google Scholar]
- 118.Jourdain G, Ngo-Giang-Huong N, Harrison L, et al. Tenofovir versus placebo to prevent perinatal transmission of hepatitis B. N Engl J Med. 2018;378:911–923. 10.1056/NEJMoa1708131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Ding Y, Cao L, Zhu L, et al. Efficacy and safety of tenofovir alafenamide fumarate for preventing mother-to-child transmission of hepatitis B virus: A national cohort study. Aliment Pharmacol Ther. 2020;52:1377–1386. 10.1111/apt.16043. [DOI] [PubMed] [Google Scholar]
- 120.Huang J, Cheng C, Li K, Zhu C, Liu Y. Effectiveness and safety of tenofovir alafenamide fumarate in the prevention of perinatal hepatitis B transmission: A meta-analysis. Dig Dis Sci. 2024;69:978–988. 10.1007/s10620-023-08258-9. [DOI] [PubMed] [Google Scholar]
- 121.Pan CQ, Zhu L, Yu AS, Zhao Y, Zhu B, Dai E. Tenofovir alafenamide versus tenofovir disoproxil fumarate for preventing vertical transmission in chronic hepatitis B mothers: A systematic review and meta-analysis. Clin Infect Dis. 2024;ahead of print: 10.1093/cid/ciae288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Li B, Liu Z, Liu X, et al. Efficacy and safety of tenofovir disoproxil fumarate and tenofovir alafenamide fumarate in preventing HBV vertical transmission of high maternal viral load. Hepatol Int. 2021;15:1103–1108. 10.1007/s12072-021-10235-1. [DOI] [PubMed] [Google Scholar]
- 123.Ehrhardt S, Xie C, Guo N, Nelson K, Thio CL. Breastfeeding while taking lamivudine or tenofovir disoproxil fumarate: A review of the evidence. Clin Infect Dis. 2015;60:275–278. 10.1093/cid/ciu798. [DOI] [PubMed] [Google Scholar]
- 124.Samadi Kochaksaraei G, Castillo E, Sadler MD, et al. Real-world clinical and virological outcomes in a retrospective multiethnic cohort study of 341 untreated and tenofovir disoproxil fumarate-treated chronic hepatitis B pregnant patients in North America. Aliment Pharmacol Ther. 2020;52:1707–1716. 10.1111/apt.16123. [DOI] [PubMed] [Google Scholar]
- 125.Asselah T, Rizzetto M. Hepatitis D virus infection. N Engl J Med. 2023;389:58–70. 10.1056/NEJMra2212151. [DOI] [PubMed] [Google Scholar]
- 126.Delfino CM, Eirin ME, Berini C, et al. HDAg-L variants in covert hepatitis D and HBV occult infection among Amerindians of Argentina: New insights. J Clin Virol. 2012;54:223–228. 10.1016/j.jcv.2012.04.014. [DOI] [PubMed] [Google Scholar]
- 127.Aguilera A, Rodríguez-Calviño J, de Mendoza C, Soriano V. Short article: Hepatitis delta in patients with resolved hepatitis B virus infection. Eur J Gastroenterol Hepatol. 2018;30:1063–1065. 10.1097/MEG.0000000000001187. [DOI] [PubMed] [Google Scholar]
- 128.Wong RJ, Hirode G, Feld J, et al. An updated assessment of hepatitis delta prevalence among adults in Canada: A meta-analysis. J Viral Hepat. 2024;31:324–341. 10.1111/jvh.13939. [DOI] [PubMed] [Google Scholar]
- 129.Wranke A, Hardtke S, Heidrich B, et al. Ten-year follow-up of a randomized controlled clinical trial in chronic hepatitis delta. J Viral Hepat. 2020;27:1359–1368. 10.1111/jvh.13366. [DOI] [PubMed] [Google Scholar]
- 130.Wedemeyer H, Yurdaydìn C, Dalekos GN, et al. Peginterferon plus adefovir versus either drug alone for hepatitis delta. N Engl J Med. 2011;364:322–331. 10.1056/NEJMoa0912696. [DOI] [PubMed] [Google Scholar]
- 131.Heller T, Rotman Y, Koh C, et al. Long-term therapy of chronic delta hepatitis with peginterferon alfa. Aliment Pharmacol Ther. 2014;40:93–104. 10.1111/apt.12788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Wranke A, Lobato C, Ceausu E, et al. Long-term outcome of hepatitis delta in different regions world-wide: Results of the Hepatitis Delta International Network. Liver Int. 2024. ahead of print: 10.1111/liv.16006. [DOI] [PubMed] [Google Scholar]
- 133.Osiowy C, Andonov A, Fonseca K, et al. Transmission of hepatitis D virus between spouses: A longitudinal study of the first reported Canadian case. IDCases. 2017;8:37–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Wedemeyer H, Aleman S, Brunetto MR, et al. A phase 3, randomized trial of bulevirtide in chronic hepatitis D. N Engl J Med. 2023;389:22–32. 10.1056/NEJMoa2213429. [DOI] [PubMed] [Google Scholar]
- 135.Wedemeyer H, Schöneweis K, Bogomolov P, et al. Safety and efficacy of bulevirtide in combination with tenofovir disoproxil fumarate in patients with hepatitis B virus and hepatitis D virus coinfection (MYR202): A multicentre, randomised, parallel-group, open-label, phase 2 trial. Lancet Infect Dis. 2023;23:117–129. 10.1016/S1473-3099(22)00318-8. [DOI] [PubMed] [Google Scholar]
- 136.Asselah T, Chulanov V, Lampertico P, et al. Bulevirtide combined with pegylated interferon for chronic hepatitis D. N Engl J Med. 2024;391:133–143. 10.1056/NEJMoa2314134. [DOI] [PubMed] [Google Scholar]
- 137.Degasperi E, Anolli MP, Uceda Renteria SC, et al. Bulevirtide monotherapy for 48 weeks in patients with HDV-related compensated cirrhosis and clinically significant portal hypertension. J Hepatol. 2022;77:1525–1531. 10.1016/j.jhep.2022.07.016. [DOI] [PubMed] [Google Scholar]
- 138.Lampertico P, Roulot D, Wedemeyer H. Bulevirtide with or without pegIFNα for patients with compensated chronic hepatitis delta: From clinical trials to real-world studies. J Hepatol. 2022;77:1422–1430. 10.1016/j.jhep.2022.06.010. [DOI] [PubMed] [Google Scholar]
- 139.Jachs M, Panzer M, Hartl L, et al. Long-term follow-up of patients discontinuing bulevirtide treatment upon long-term HDV-RNA suppression. JHEP Rep. 2023;5:100751. 10.1016/j.jhepr.2023.100751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Anolli MP, Degasperi E, Allweiss L, et al. A 3-year course of bulevirtide monotherapy may cure HDV infection in cirrhotics. J Hepatol. 2023. ahead of print: 10.1016/j.jhep.2022.12.023. [DOI] [PubMed] [Google Scholar]
- 141.Bazinet M, Pântea V, Cebotarescu V, et al. Safety and efficacy of REP 2139 and pegylated interferon alfa-2a for treatment-naive patients with chronic hepatitis B virus and hepatitis D virus co-infection (REP 301 and REP 301-LTF): A non-randomised, open-label, phase 2 trial. Lancet Gastroenterol Hepatol. 2017;2:877–889. 10.1016/S2468-1253(17)30288-1. [DOI] [PubMed] [Google Scholar]
- 142.Bazinet M, Pântea V, Cebotarescu V, et al. Persistent control of hepatitis B virus and hepatitis delta virus infection following REP 2139-ca and pegylated interferon therapy in chronic hepatitis B virus/hepatitis delta virus coinfection. Hepatol Commun. 2021;5:189–202. 10.1002/hep4.1633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Stern C, Bourliere M, Loustaud-Ratti V, et al. OS-034 safety and efficacy of REP 2139-mg in hepatitis D patients with advanced liver disease: An international compassionate use program. J Hepatol. 2024;80:S28. 10.1016/S0168-8278(24)00475-6. [DOI] [PubMed] [Google Scholar]
- 144.Yurdaydin C, Keskin O, Yurdcu E, et al. A phase 2 dose-finding study of lonafarnib and ritonavir with or without interferon alpha for chronic delta hepatitis. Hepatology. 2022;75:1551–1565. 10.1002/hep.32259. [DOI] [PubMed] [Google Scholar]
- 145.Etzion, O, Hamid SS, Asselah T, et al. Week 48 results of the phase 3 D-LIVR study, a randomized double-blind, placebo-controlled trial evaluating the safety and efficacy of Lonafarnib-boosted with Ritonavir with or without Peginterferon Alfa in patients with chronic hepatitis delta, J Hepatol. 2023;78:S10. 10.1016/S0168-8278(23)00451-8. [DOI] [Google Scholar]
- 146.De Monte A, Courjon J, Anty R, et al. Direct-acting antiviral treatment in adults infected with hepatitis C virus: Reactivation of hepatitis B virus coinfection as a further challenge. J Clin Virol. 2016;78:27–30. 10.1016/j.jcv.2016.02.026. [DOI] [PubMed] [Google Scholar]
- 147.Konstantinou D, Deutsch M. The spectrum of HBV/HCV coinfection: Epidemiology, clinical characteristics, viral interactions and management. Ann Gastroenterol. 2015;28:221–228. [PMC free article] [PubMed] [Google Scholar]
- 148.Caccamo G, Saffioti F, Raimondo G. Hepatitis B virus and hepatitis C virus dual infection. World J Gastroenterol. 2014;20:14559–14567. 10.3748/wjg.v20.i40.14559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Collins JM, Raphael KL, Terry C, et al. Hepatitis B virus reactivation during successful treatment of hepatitis C virus with sofosbuvir and simeprevir. Clin Infect Dis. 2015;61:1304–1306. 10.1093/cid/civ474. [DOI] [PubMed] [Google Scholar]
- 150.Bersoff-Matcha SJ, Cao K, Jason M, et al. Hepatitis B virus reactivation associated with direct-acting antiviral therapy for chronic hepatitis C virus: A review of cases reported to the U.S. Food and Drug Administration adverse event reporting system. Ann Intern Med. 2017;166:792–798. 10.7326/M17-0377. [DOI] [PubMed] [Google Scholar]
- 151.Yeh M, Huang C, Huang C, et al. Hepatitis B-related outcomes following direct-acting antiviral therapy in Taiwanese patients with chronic HBV/HCV co-infection. J Hepatol. 2020;73:62–71. 10.1016/j.jhep.2020.01.027. [DOI] [PubMed] [Google Scholar]
- 152.Ende AR, Kim NH, Yeh MM, Harper J, Landis CS. Fulminant hepatitis B reactivation leading to liver transplantation in a patient with chronic hepatitis C treated with simeprevir and sofosbuvir: A case report. J Med Case Rep. 2015;9:164. 10.1186/s13256-015-0630-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Mücke MM, Backus LI, Mücke VT, et al. Hepatitis B virus reactivation during direct-acting antiviral therapy for hepatitis C: A systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2018;3:172–180. 10.1016/S2468-1253(18)30002-5. [DOI] [PubMed] [Google Scholar]
- 154.Calvaruso V, Ferraro D, Licata A, et al. HBV reactivation in patients with HCV/HBV cirrhosis on treatment with direct-acting antivirals. J Viral Hepat. 2018;25:72–79. 10.1111/jvh.12754. [DOI] [PubMed] [Google Scholar]
- 155.Ogawa E, Furusyo N, Murata M, Toyoda K, Hayashi T, Ura K. Potential risk of HBV reactivation in patients with resolved HBV infection undergoing direct-acting antiviral treatment for HCV. Liver Int. 2018;38:76–83. 10.1111/liv.13496. [DOI] [PubMed] [Google Scholar]
- 156.Wu T, Kwok RM, Tran TT. Isolated anti-HBc: The relevance of hepatitis B core antibody-A review of new issues. Am J Gastroenterol. 2017;112:1780–1788. 10.1038/ajg.2017.397. [DOI] [PubMed] [Google Scholar]
- 157.Public Health Agency of Canada. Chapter 13: HIV/AIDS Epi updates, April 2012 – HIV/AIDS in Canada among people from countries where HIV is endemic. 2014. https://www.canada.ca/en/public-health/services/hiv-aids/publications/epi-updates/chapter-13-hiv-aids-canada-among-people-from-countries-hiv-endemic.html [Accessed May 28, 2024].
- 158.Rana U, Driedger M, Sereda P, et al. Characteristics and outcomes of antiretroviral-treated HIV-HBV co-infected patients in Canada? BMC Infect Dis. 2019;19:982. 10.1186/s12879-019-4617-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Singh KP, Crane M, Audsley J, Avihingsanon A, Sasadeusz J, Lewin SR. HIV-hepatitis B virus coinfection: Epidemiology, pathogenesis, and treatment. AIDS. 2017;31:2035–2052. 10.1097/QAD.0000000000001574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.European AIDS Clinical Society. Guidelines 8.1. 2016. https://www.eacsociety.org/media/guidelines_8.1-english.pdf [Accessed Apr 15, 2024].
- 161.European AIDS Clinical Society. Guidelines V. 12.0. 2023. https://www.eacsociety.org/media/guidelines-12.0.pdf [Accessed May 28, 2024].
- 162.Agarwal K, Brunetto M, Seto WK, et al. 96 weeks treatment of tenofovir alafenamide vs. tenofovir disoproxil fumarate for hepatitis B virus infection. J Hepatol. 2018;68:672–681. 10.1016/j.jhep.2017.11.039. [DOI] [PubMed] [Google Scholar]
- 163.Lampertico P, Buti M, Fung S, et al. Switching from tenofovir disoproxil fumarate to tenofovir alafenamide in virologically suppressed patients with chronic hepatitis B: A randomised, double-blind, phase 3, multicentre non-inferiority study. Lancet Gastroenterol Hepatol. 2020;5:441–453. 10.1016/S2468-1253(19)30421-2. [DOI] [PubMed] [Google Scholar]
- 164.Gallant J, Brunetta J, Crofoot G, et al. Brief report: Efficacy and safety of switching to a single-tablet regimen of elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide in HIV-1/hepatitis B-coinfected adults. J Acquir Immune Defic Syndr. 2016;73:294–298. 10.1097/QAI.0000000000001069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Huang Y, Cheng C, Liou B, et al. Efficacy and safety of elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide as maintenance treatment in HIV/HBV-coinfected patients. J Acquir Immune Defic Syndr. 2021;86:473–481. 10.1097/QAI.0000000000002589. [DOI] [PubMed] [Google Scholar]
- 166.Cahn P, Sierra Madero J, Arribas JR, et al. Three-year durable efficacy of dolutegravir plus lamivudine in antiretroviral therapy - naive adults with HIV-1 infection. AIDS. 2022;36:39–48. 10.1097/QAD.0000000000003070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Llibre JM, Brites C, Cheng C, et al. Efficacy and safety of switching to the 2-drug regimen dolutegravir/lamivudine versus continuing a 3- or 4-drug regimen for maintaining virologic suppression in adults living with human immunodeficiency virus 1 (HIV-1): Week 48 results from the phase 3, noninferiority SALSA randomized trial. Clin Infect Dis. 2023;76:720–729. 10.1093/cid/ciac130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Clinical Info HIV.gov. What to Start: Advantages and Disadvantages of Antiretroviral Components for Initial Therapy | NIH. 2021. https://clinicalinfo.hiv.gov/en/guidelines/hiv-clinical-guidelines-adult-and-adolescent-arv/what-start-advantages-and-disadvantages [Accessed Apr 15, 2024].
- 169.Orkin C, Arasteh K, Górgolas Hernández-Mora M, et al. Long-acting cabotegravir and rilpivirine after oral induction for HIV-1 infection. N Engl J Med. 2020;382:1124–1135. 10.1056/NEJMoa1909512. [DOI] [PubMed] [Google Scholar]
- 170.Swindells S, Andrade-Villanueva J, Richmond GJ, et al. Long-acting cabotegravir and rilpivirine for maintenance of HIV-1 suppression. N Engl J Med. 2020;382:1112–1123. 10.1056/NEJMoa1904398. [DOI] [PubMed] [Google Scholar]
- 171.Clinical Info HIV.gov. What's New: Adult and Adolescent ARV Guidelines | NIH. 2024. https://clinicalinfo.hiv.gov/en/guidelines/hiv-clinical-guidelines-adult-and-adolescent-arv/whats-new [Accessed Apr 15, 2024].
- 172.Lara AN, Sartori AM, Fonseca MO, Lopes MH. Long-term protection after hepatitis B vaccination in people living with HIV. Vaccine. 2017;35:4155–4161. 10.1016/j.vaccine.2017.06.040. [DOI] [PubMed] [Google Scholar]
- 173.Congly SE, Doucette KE, Coffin CS. Outcomes and management of viral hepatitis and human immunodeficiency virus co-infection in liver transplantation. World J Gastroenterol. 2014;20:414–424. 10.3748/wjg.v20.i2.414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Cooper C, Kanters S, Klein M, et al. Liver transplant outcomes in HIV-infected patients: A systematic review and meta-analysis with synthetic cohort. AIDS. 2011;25:777–786. 10.1097/QAD.0b013e328344febb. [DOI] [PubMed] [Google Scholar]
- 175.Indolfi G, Easterbrook P, Dusheiko G, et al. Hepatitis B virus infection in children and adolescents. Lancet Gastroenterol Hepatol. 2019;4:466–476. 10.1016/S2468-1253(19)30042-1. [DOI] [PubMed] [Google Scholar]
- 176.Marx G, Martin SR, Chicoine J, Alvarez F. Long-term follow-up of chronic hepatitis B virus infection in children of different ethnic origins. J Infect Dis. 2002;186:295–301. 10.1086/341508. [DOI] [PubMed] [Google Scholar]
- 177.Wu J, Tsai W, Hsu H, et al. Effect of puberty onset on spontaneous hepatitis B virus e antigen seroconversion in men. Gastroenterology. 2010;138:942.e1–948.e1. 10.1053/j.gastro.2009.11.051. [DOI] [PubMed] [Google Scholar]
- 178.Yandza T, Alvarez F, Laurent J, Gauthier F, Dubousset AM, Valayer J. Pediatric liver transplantation for primary hepatocellular carcinoma associated with hepatitis virus infection. Transpl Int. 1993;6:95–98. 10.1007/BF00336652. [DOI] [PubMed] [Google Scholar]
- 179.Bortolotti F, Guido M, Bartolacci S, et al. Chronic hepatitis B in children after e antigen seroclearance: Final report of a 29-year longitudinal study. Hepatology. 2006;43:556–562. 10.1002/hep.21077. [DOI] [PubMed] [Google Scholar]
- 180.Xue MM, Glenn JS, Leung DH. Hepatitis D in children. J Pediatr Gastroenterol Nutr. 2015;61:271–281. 10.1097/MPG.0000000000000859. [DOI] [PubMed] [Google Scholar]
- 181.Rao DP, Kropac E, Do MT, Roberts KC, Jayaraman GC. Childhood overweight and obesity trends in Canada. Health Promot Chronic Dis Prev Can. 2016;36:194-198. 10.24095/hpcdp.36.9.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Jonas MM, Chang M, Sokal E, et al. Randomized, controlled trial of entecavir versus placebo in children with hepatitis B envelope antigen-positive chronic hepatitis B. Hepatology. 2016;63:377–387. 10.1002/hep.28015. [DOI] [PubMed] [Google Scholar]
- 183.Murray KF, Szenborn L, Wysocki J, et al. Randomized, placebo-controlled trial of tenofovir disoproxil fumarate in adolescents with chronic hepatitis B. Hepatology. 2012;56:2018–2026. 10.1002/hep.25818. [DOI] [PubMed] [Google Scholar]
- 184.Cathcart AL, Chan HL, Bhardwaj N, et al. No resistance to tenofovir alafenamide detected through 96 weeks of treatment in patients with chronic hepatitis B infection. Antimicrob Agents Chemother. 2018;62:1064. 10.1128/AAC.01064-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Waalewijn H, Szubert AJ, Wasmann RE, et al. First pharmacokinetic data of tenofovir alafenamide fumarate and tenofovir with dolutegravir or boosted protease inhibitors in African children: A substudy of the CHAPAS-4 trial. Clin Infect Dis. 2023;77:875–882. 10.1093/cid/ciad267. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
N/A


