Abstract
Background
The C-reactive protein-triglyceride-glucose index (CTI) has emerged as an innovative composite marker for evaluating metabolic-inflammatory dysregulation, integrating markers of insulin resistance and systemic inflammation. However, the association between CTI and cardiovascular disease (CVD) or its mortality has rarely been studied. This study sought to examine CTI's associations with CVD mortality, CVD incidence, and all-cause mortality.
Methods
This study included 8,679 adults from the National Health and Nutrition Examination Survey (NHANES) 2001–2010, 2015–2018. The CTI was derived as: 0.412* Ln (CRP [mg/L]) + Ln (TG [mg/dl] × FPG [mg/dl])/2, with participants categorized into quartiles. We employed Kaplan–Meier curves, cox proportional hazards model, logistic regression analyses, and restricted cubic spline (RCS) to evaluate CTI's associations with CVD mortality, total CVD incidence, and all-cause mortality across sex-stratified, age-specific, and glycemic subgroups.
Results
In this study, CTI was significantly and positively associated with CVD mortality, total CVD incidence, and all-cause mortality. CTI significantly predicted both CVD mortality (HR 2.28 [1.69–3.24]) and all-cause mortality (HR 2.14 [1.76–2.55]). Additionally, the CTI index correlated with the risk of total CVD (OR 2.85, 95% CI 2.32–3.52), congestive heart failure (OR 3.66, 95% CI 2.46–5.35), coronary heart disease (OR 2.82, 95% CI 1.95–3.97), angina pectoris (OR 2.85, 95% CI 1.89–4.22), heart attack (OR 2.59, 95% CI 1.89–3.52), and stroke (OR 2.86, 95% CI 2.00–3.85). Specifically, the association was similar between male and female, and similar in young participants and elderly participants. In different glycemic status, high levels of CTI were found to be linked to an increased risk of CVD in individuals without diabetes mellitus (DM). However, this association was not observed in individuals with DM.
Conclusions
Our analysis revealed that elevated CTI levels were significantly associated with CVD incidence and mortality. CTI may emerge as a unique predictive marker for CVD risk.
Graphical abstract
Supplementary Information
The online version contains supplementary material available at 10.1186/s12933-025-02835-0.
Keywords: C-reactive protein-triglyceride glucose index, NHANES, Cardiovascular disease mortality, Cardiovascular disease, All-cause mortality
What is currently known about this topic?
CTI has emerged as an innovative composite biomarker capable of simultaneously assessing both insulin resistance and inflammatory status.
What is the key research question?
The relationship between CTI and CVD mortality, and various CVD outcomes including congestive heart failure, coronary heart disease, angina pectoris, heart attack, and stroke, across different demographic groups.
What is new?
As the first large-scale investigation of its kind, this study reveals significant associations between CTI and CVD mortality, while evaluating these relationships across, age, gender, and glycemic-status.
How might this study influence clinical practice?
The study demonstrates a significant positive relationship between CTI values and CVD mortality. Consequently, personalized CTI modulation strategies—accounting for gender, age, and diabetes status—should be implemented for CVD prevention.
Introduction
Cardiovascular disease (CVD) remains the primary driver of premature mortality and population health deterioration globally [1–4]. The rapid aging of populations has precipitated a concerning escalation in CVD prevalence and fatality rates, elevating this condition to a critical global health challenge [5, 6]. Early identification of CVD risk factors is crucial for implementing preventive strategies. Notably, type 2 diabetes mellitus (T2DM) frequently coexists with manifest CVD and correlates with adverse clinical outcomes [7–10]. Identifying risk factors of CVD patients with different glucose metabolism statuses is critical for reducing mortality, particularly the risk of cardiovascular death.
C-reactive protein-triglyceride-glucose index (CTI) was first proposed by Ruan et al. [11], is an innovative marker combining C-reactive protein (CRP) with the triglyceride-glucose (TyG) index that simultaneously evaluates both inflammatory status and insulin resistance severity [12, 13]. CTI has demonstrated significant predictive value for cancer mortality [14]. A previous study showed a significant association between CTI and nonalcoholic fatty liver disease (NAFLD) [15]. Previous studies have shown that some inflammatory markers such as IL-6, TNF-α, NLRP3 inflammasome, calprotectin, neopterin, lactoferrin, and neutrophil extracellular traps (NETs) are associated with CVD risk [16–22]. Recently, a study including data from 10,443 participants in the CHARLS database showed that elevated CTI levels were significantly associated with increased stroke prevalence [23]. Another study has shown that elevated CTI levels are associated with an increased risk of stroke in hypertensive patients [24]. However, the relationship between CTI and CVD risk remains unclear. IR serves as a central pathological driver underlying numerous metabolic disorders and critically contributes to CVD pathogenesis through multiple mechanisms [25, 26]. Since its development in 2008, the TyG index has become a clinically established biomarker for assessing insulin resistance [27, 28]. Emerging evidence has established robust associations between the TyG index and CVD risk across diverse populations [29–32]. Previous studies have assessed the prognostic value of the TyG index for adverse outcomes in patients with various CVD phenotypes [33–35]. However, these studies primarily focused on the detrimental role of IR in CVD progression, overlooking the potential synergistic or mediating influence of systemic inflammation. Moreover, CRP, a nonspecific inflammatory marker, is strongly associated with CVD risk [36]. Among individuals with pre-existing CVD, elevated CRP concentrations independently predict subsequent heart failure development [37]. Moreover, one study showed that elevated plasma CRP and blood glucose concentrations were predictors of CVD mortality [38]. Consequently, developing an integrated biomarker that captures both insulin resistance and inflammatory status represents a critical advancement for CVD risk prediction. The CTI index has already established prognostic utility across multiple clinical contexts, including cancer-associated wasting syndromes, population-level cancer mortality, and coronary heart disease development [12, 14, 39]. Nevertheless, the relationship between CTI and CVD risk—including both incidence and mortality—remains incompletely characterized across demographic subgroups stratified by age, gender, and glycemic status.
To address these critical research gaps, we conducted an in-depth analysis of the NHANES dataset, with the aim of investigating the complex relationships between CTI and CVD mortality, total CVD events, and all-cause mortality across various subgroups including age, gender, and glycemic status. Furthermore, our study examined the associations of the CTI index with congestive heart failure, coronary heart disease, angina pectoris, heart attack, and stroke.
Materials and methods
Data source and study population
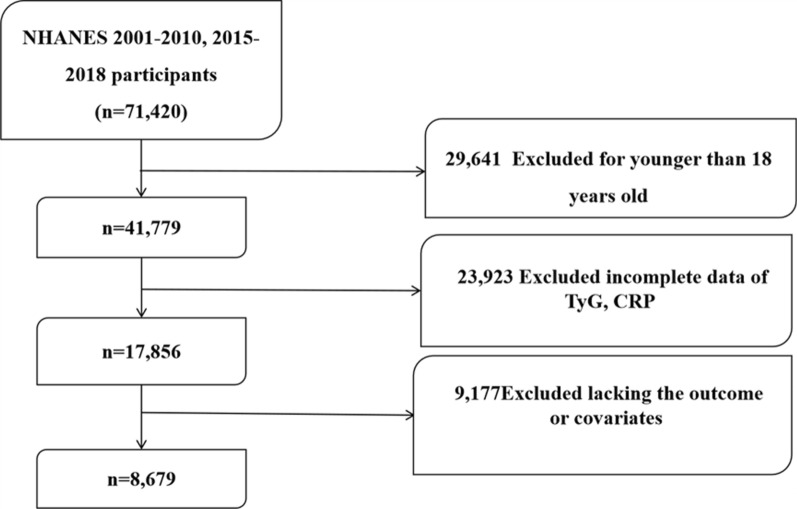
This study included 8,679 adult participants from the NHANES 2001–2010, 2015–2018. Figure 1 outlines the study design flowchart. Exclusion criteria included: (1) individuals aged ≤ 18 years (n = 29,641); (2) those with unavailable TyG index or CRP data (n = 23,923); and (3) cases missing outcome variables or covariates (n = 9,177). The NCHS Research Ethics Review Board granted ethical approval for the NHANES protocol, with written informed consent obtained from all study participants.
Fig. 1.
Flowchart depicting the participants’ selection
Calculation of CTI
CRP and TyG were used to reflect patients’ inflammation and IR status, respectively. The CRP-TyG index (CTI) was composed of CRP and TyG [11]. The CTI index was calculated according to the established equation: CTI = 0.412 × Ln (CRP [mg/L]) + Ln (TG [mg/dl] × FPG [mg/dl])/2 [11, 23].
Assessments of covariates
Trained interviewers systematically collected baseline data through standardized questionnaires, capturing: (1) Demographic and lifestyle characteristics: Gender, age, racial/ethnic background, educational level, smoking status, and alcohol use. (2) Body measurements: Body mass index (BMI) as the primary adiposity indicator, and systolic blood pressure (SBP). (3) Clinical histories: Documented hypertension and diabetes diagnoses, along with current medications. (4) Laboratory test data: high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), and glycated hemoglobin (HbA1c) levels.
Total CVD, congestive heart failure, coronary heart disease, angina pectoris, heart attack, and stroke ascertainment
Physician-diagnosed cardiovascular disease was ascertained through standardized interviews, wherein participants self-reported existing CVD conditions using a validated medical history questionnaire. Participants self-reported physician-diagnosed CVD conditions by responding to the question: "Has a healthcare provider ever diagnosed you with congestive heart failure, coronary heart disease, angina pectoris, heart attack, or stroke?" [40] Affirmative responses to any condition qualified participants as having CVD. These CVD conditions—including congestive heart failure, coronary heart disease, angina pectoris, heart attack and stroke—were similarly classified using standardized diagnostic criteria consistent with their respective clinical presentations[40–42].
The primary outcome was CVD mortality, classified using ICD-10 codes for heart diseases (I00-I09, I11, I13, and I20-I51) and cerebrovascular diseases (I60-I69). Mortality status was ascertained through linkage between the NHANES Public-Use Linked Mortality File and National Death Index, with follow-up data available until December 31, 2019. Follow-up duration spanned from the initial interview date to either the occurrence of death, loss to follow-up, or the study's censoring date (December 31, 2019) [40, 43].
Definitions
Hypertension was classified according to four criteria: (1) systolic blood pressure ≥ 140 mmHg, (2) diastolic blood pressure ≥ 90 mmHg, (3) physician-diagnosed hypertension based on self-report, or (4) active use of antihypertensive drugs [44]. Diabetes mellitus (DM) diagnosis required fulfillment of any one condition: (1) fasting plasma glucose ≥ 126 mg/dL, (2) hemoglobin A1c ≥ 6.5%, or (3) ongoing antidiabetic medication use [45].
Statistical analysis
All analyses followed CDC analytical protocols for complex survey data. To account for NHANES's multi-stage, stratified probability sampling design, we applied appropriate sample weights and adjusted for clustering and stratification effects in our statistical modeling. Continuous variables were presented as weighted means with standard errors and were compared using one-way analysis of variance; categorical variables were presented as unweighted frequencies but weighted proportions and were compared using chi-square tests.
Participant characteristics were stratified and analyzed by CTI quartiles. Following variance homogeneity assessment, between-group comparisons were performed using Bonferroni-adjusted post hoc tests. Cox proportional hazards regression analysis was employed to quantify associations between CTI and mortality outcomes, generating hazard ratios (HRs) with 95% confidence intervals (CIs) for both CVD-specific and all-cause mortality. Logistic regression analysis evaluated relationships between CTI and CVD. These associations were quantified across model 3, with outcomes reported as odds ratios (ORs) and 95% confidence intervals (CIs). Model 1 was unadjusted. Model 2 adjusted for age, sex. Model 3 adjusted for the same variables as Model 2 and for race, education, smoking status, alcohol use, systolic blood pressure (SBP), BMI, LDL, HDL, glycated hemoglobin, diabetes, and hypertension. The selection of covariates including age, sex, race, education, smoking status, alcohol use, systolic blood pressure (SBP), BMI, LDL-C, HDL-C, glycosylated hemoglobin (HbA1c), diagnosis of diabetes and hypertension were based on clinically meaningful indicators and those demonstrating significance in univariate analysis, with collinearity controlled (VIF < 5). The results of the Variance Inflation Factors (GVIF) Analysis were shown in Tables S7-S9. Potential confounding factors were controlled through multivariate adjustment in both Cox proportional hazards and logistic regression models, where these factors were included as covariates to isolate the association between CTI and CVD. Stratified analyses by sex, age, and glycemic status were additionally performed to verify the consistency of associations while controlling for potential confounding by these stratification factors.
Receiver operating characteristic (ROC) curve analysis was carried out to compare the predictive value of the CTI index for total CVD, congestive heart failure, coronary heart disease, angina pectoris, heart attack, and stroke. Restricted cubic spline (RCS) regression, incorporating the same covariates as Model 3, was employed to characterize potential linear or nonlinear dose–response patterns between CTI and three critical outcomes: CVD mortality, all-cause mortality, and total CVD incidence. To evaluate result consistency, we conducted stratified analyses examining CTI's association with CVD and all-cause mortality across key demographic and clinical subgroups. Complementing this, Kaplan–Meier survival curves compared mortality risks among participants categorized by CTI quartiles.
Results
Baseline characteristics of participants across CTI quartiles.
Table 1 presents participant characteristics stratified by CTI quartiles. Individuals in upper CTI quartiles exhibited distinct demographic and clinical profiles: they were typically older, predominantly male, less educated, and more frequently current smokers. These participants also demonstrated significantly higher prevalence rates of hypertension and diabetes mellitus compared to lower quartile groups. Furthermore, elevated BMI values, LDL cholesterol concentrations, and HbA1c levels were consistently observed in higher CTI.
Table 1.
Baseline characteristics according to CTI quartiles
| CTI quartiles (N = 8679) | P | ||||
|---|---|---|---|---|---|
| Q1 (n = 2170) | Q2 (n = 2170) | Q3 (n = 2170) | Q4 (n = 2169) | ||
| Age, years | 43.85 [20,85] | 50.61 [20,85] | 52.31 [20,85] | 52.86 [20,85] | < 0.001a |
| Sex (n, %) | < 0.001b | ||||
| Male | 1016 (46.82%) | 1129 (52.03%) | 1055 (48.62%) | 1016 (46.84%) | |
| Female | 1154 (53.18%) | 1041 (47.97%) | 1115 (51.38%) | 1153 (53.16%) | |
| First_HbA1c% (mean) | 5.37 [3.8,11] | 5.53 [3.9,11.9] | 5.70 [4.0,12.2] | 6.32 [3.9,17.0] | < 0.001a |
| Race (n, %) | < 0.001b | ||||
| Mexican American | 288 (13.27%) | 371 (17.10%) | 444 (20.46%) | 470 (21.67%) | |
| Other Hispanic | 169 (7.79%) | 209 (9.63%) | 221 (10.18%) | 283 (13.05%) | |
| Non-Hispanic White | 1051 (48.43%) | 1017 (46.87%) | 999 (46.04%) | 942 (43.43%) | |
| Non-Hispanic Black | 510 (23.50%) | 440 (20.28%) | 371 (17.10%) | 340 (15.68%) | |
| Other races | 152 (7.00%) | 133 (6.13%) | 135 (6.22%) | 134 (6.18%) | |
| Hypertension (n, %) | < 0.001b | ||||
| Yes | 485 (22.35%) | 726 (33.46%) | 854 (39.35%) | 1046 (48.22%) | |
| No | 1685 (77.65%) | 1444 (66.54%) | 1316 (60.65%) | 1123 (51.78%) | |
| Diabetes (n, %) | < 0.001b | ||||
| Yes | 135 (6.22%) | 277 (12.76%) | 420 (19.35%) | 800 (36.88%) | |
| No | 2035 (93.78%) | 1893 (87.24%) | 1750 (80.65%) | 1369 (63.12%) | |
| Education (n, %) | < 0.001b | ||||
| Less than 9th grade | 144 (6.64%) | 258 (11.89%) | 317 (14.61%) | 318 (14.66%) | |
| 9-11th grade | 294 (13.55%) | 334 (15.39%) | 311 (14.33%) | 376 (17.34%) | |
| High school graduate or equivalent | 453 (20.88%) | 524 (24.15%) | 534 (24.61%) | 541 (24.94%) | |
| Some college or AA degree | 635 (29.26%) | 577 (26.59%) | 612 (28.20%) | 568 (26.19%) | |
| College graduate or above | 644 (29.68%) | 477 (21.98%) | 396 (18.25%) | 366 (16.87%) | |
| Smoking (n, %) | < 0.001b | ||||
| Yes | 884 (40.74%) | 998 (45.99%) | 1055 (48.62%) | 1114 (51.36%) | |
| No | 1286 (59.26%) | 1172 (54.01%) | 1115 (51.38%) | 1055 (48.64%) | |
| Alcohol use (n, %) | < 0.001b | ||||
| Yes | 1596 (73.55%) | 1540 (70.97%) | 1533 (70.65%) | 1433 (66.07%) | |
| No | 574(26.45%) | 630 (29.03%) | 637 (29.35%) | 736 (33.93%) | |
| First_HDL (mg/dl) (mean) | 104.57 [22,341] | 56.44 [26,188] | 52.27 [21,226] | 47.62 [6,129] | < 0.001a |
| First_LDL (mg/dl) (mean) | 63.41 [26,164] | 115.72 [26,289] | 118.95 [24,328] | 119.92 [22,344] | < 0.001a |
| First_BMI (mean) | 25.11 [14.59,52.48] | 28.03 [15.25,71.30] | 30.40 [16.70,67.71] | 32.87 [16.60,130.21] | < 0.001a |
| First_SBP (mean) | 118.82 [80,216] | 124.6 [82,226] | 126.56 [78,236] | 128.01 [72,208] | < 0.001a |
a: Represents the use of the Kruskal–Wallis test
b: Represents the use of the Chi-square test
Relationship between CTI and specific CVD conditions and Mortality outcomes
During a follow-up period, 1,152 (13.27%) of the 8,679 participants died, with 360 (7.26%) deaths attributed to cardiovascular disease. Figure 2 demonstrates the association of CTI with CVD mortality, total CVD, all-cause mortality, congestive heart failure, coronary heart disease, angina pectoris, heart attack, and stroke from model 3. Table 2 provide detailed information on all associations. Model 1 revealed no significant relationship between CTI elevation and CVD mortality risk. However, after covariate adjustment in Model 2, each unit increase in CTI corresponded to a 63% greater mortality hazard (HR = 1.63, 95% CI:1.18–2.24, p < 0.001). After adjustment for covariates, the results showed CTI was positively associated with CVD mortality. A clear dose–response relationship emerged between CTI elevation and CVD mortality risk. Relative to Q1, progressively higher quartiles demonstrated increasing hazard ratios: Q2 (HR 1.45, 95% CI:1.16–2.23), Q3 (HR 1.92, 1.52–2.81), and Q4 (HR 2.28, 1.69–3.24).
Fig. 2.
Forest plot of the CTI association with CVD mortality, total CVD, and all-cause mortality using logistic regression model and Cox proportional hazards model
Table 2.
Association between the CTI and CVD incidence
| Died/participants | Model 1 | Model 2 | Model 3 | |
|---|---|---|---|---|
| CVD mortality | 1152/8679 | |||
| Q1 | 205/2170 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 313/2170 | 0.99 (0.71–1.39) | 1.01 (0.74–1.40) | 1.45 (1.16–2.23)** |
| Q3 | 335/2170 | 1.22 (0.87–1.71) | 1.24 (0.91–1.69) | 1.92 (1.52–2.81)** |
| Q4 | 299/2169 | 1.43 (0.98–2.08) | 1.63 (1.18–2.24)** | 2.28 (1.69–3.24)** |
| All-cause mortality | 360/8679 | |||
| Q1 | 63/2170 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 91/2170 | 1.10 (0.92–1.32) | 1.08 (0.91–1.29) | 1.62 (1.34–1.92)** |
| Q3 | 108/2170 | 1.23 (1.02–1.48)* | 1.19 (0.99–1.42) | 1.87 (1.49–2.23)** |
| Q4 | 98/2169 | 1.44 (1.17–1.78)** | 1.50 (1.26–1.80)** | 2.14 (1.76–2.55)** |
| Total-CVD | 972/8679 | |||
| Q1 | 140/2170 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 230/2170 | 1.01 (0.79–1.30) | 1.18 (0.94–1.49) | 1.81 (1.42–2.21)** |
| Q2 | 247/2170 | 0.88 (0.68–1.13) | 1.18 (0.94–1.49) | 1.91 (1.49–2.39)** |
| Q4 | 355/2169 | 1.12 (0.86–1.46) | 2.00 (1.61–2.49)** | 2.85 (2.32–3.52)** |
| Congestive heart failure | 283/8679 | |||
| Q1 | 35/2170 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 64/2170 | 1.07 (0.69–1.66) | 1.29 (0.84–1.96) | 1.87 (1.20–2.79)** |
| Q3 | 62/2170 | 0.85 (0.54–1.35) | 1.14 (0.74–1.75) | 1.77 (1.16–2.69)** |
| Q4 | 122/2169 | 1.41 (0.90–2.21) | 2.51 (1.70–3.70)** | 3.66 (2.46–5.35)** |
| Coronary heart disease | 363/8679 | |||
| Q1 | 45/2170 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 99/2170 | 1.49 (1.02–2.20)* | 1.56 (1.08–2.26)* | 2.28 (1.61–3.25)** |
| Q3 | 98/2170 | 1.21 (0.81–1.80) | 1.41 (0.97–2.04) | 2.24 (1.59–3.21)** |
| Q4 | 121/2169 | 1.28 (0.84–1.94) | 1.94 (1.35–2.78)** | 2.82 (1.95–3.97)** |
| Angina pectoris | 235/8679 | |||
| Q1 | 32/2170 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 50/2170 | 0.94 (0.58–1.49) | 1.11 (0.71–1.75) | 1.61 (1.03–2.50)* |
| Q3 | 65/2170 | 0.97 (0.61–1.55) | 1.36 (0.88–2.11) | 2.08 (1.37–3.19)** |
| Q4 | 88/2169 | 1.02 (0.63–1.65) | 1.97 (1.30–2.98)** | 2.85 (1.89–4.22)** |
| Heart attack | 399/8679 | |||
| Q1 | 56/2170 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 101/2170 | 1.16 (0.81–1.65)** | 1.29 (0.92–1.82) | 1.87 (1.32–2.55)** |
| Q3 | 103/2170 | 0.99 (0.68–1.42) | 1.22 (0.87–1.72) | 1.91 (1.36–2.61)** |
| Q4 | 139/2169 | 1.15 (0.79–1.68) | 1.81 (1.31–2.50)** | 2.59 (1.89–3.52)** |
| Stroke | 325/8679 | |||
| Q1 | 46/2170 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 76/2170 | 0.98 (0.66–1.45) | 1.17 (0.80–1.71) | 1.54 (1.04–2.22)** |
| Q3 | 78/2170 | 0.83 (0.55–1.25) | 1.11 (0.76–1.62) | 1.50 (1.08–2.27)** |
| Q4 | 125/2169 | 1.18 (0.78–1.77) | 1.94 (1.37–2.75)** | 2.86 (2.00–3.85)** |
Model 1 did not adjust for any variables. Model 2 adjusted for sex and age. Model 3 adjusted for age, sex, education, race, smoking, alcohol use, self-reported diabetes, self-reported hypertension, systolic blood pressure (SBP), BMI, HDL, LDL, and HbA1c% levels
*P < 0.05, **P < 0.01
Cox proportional hazards regression analysis revealed a consistent positive association between CTI elevation and all-cause mortality. The unadjusted model (Model 1) demonstrated a 44% increased mortality hazard per CTI unit increase (HR = 1.44, 95% CI:1.17–1.78), with this relationship strengthening to 50% higher risk after covariate adjustment in Model 2 (HR = 1.50, 95% CI:1.26–1.80, p < 0.001). After adjust the potential confounding variables (Model 3), compared to Q1, all-cause mortality risk increased with higher CTI levels, with HRs of 1.62 (95% CI 1.34–1.92), 1.87 (95% CI 1.49–2.23), 2.14 (95% CI 1.76–2.55) for Q2, Q3, and Q4.
After adjustment for covariates, the results showed CTI was positively associated with total CVD (OR 2.85, 95% CI 2.32–3.52). In addition, the results showed CTI was positively associated with congestive heart failure, coronary heart disease, angina pectoris, heart attack, and stroke. Logistic regression analysis demonstrated a substantial increase in congestive heart failure in the higher CTI group, from Q2 (OR 1.87, 95% CI (1.20–2.79) to Q3 (OR 1.77, 95% CI (1.16–2.69) and Q4 (OR 3.66, 95% CI 2.46–5.35), compared with lower CTI group. For heart attack, CTI had a high association from model 3(OR 2.59, 95% CI 1.89–3.52). For angina pectoris, CTI had a high association from model 3 (OR 2.85, 95% CI 1.89–4.22). For coronary heart disease, CTI had a high association from model 3 (OR 2.82, 95% CI 1.95–3.97). For stroke, CTI had a high association from model 3 (OR 2.86, 95% CI 2.00–3.85).
RCS analysis and Kaplain-Meier survival curves investigating the relationship between CTI and specific CVD conditions and Mortality outcomes
Figure 3 presents restricted cubic spline (RCS) analyses examining CTI's associations with multiple cardiovascular outcomes: CVD mortality, total CVD prevalence, all-cause mortality, congestive heart failure, heart attack, angina pectoris, stroke, and coronary heart disease. The RCS modeling demonstrated a significant positive linear relationship between CTI and both CVD-specific and all-cause mortality in the general population (Fig. 3). After full covariate adjustment (Model 3), CTI exhibited significant linear associations with total CVD as well as specific manifestations including congestive heart failure, heart attack, angina pectoris, stroke, and coronary heart disease.
Fig. 3.
Associations between CTI with CVD mortality, total CVD, congestive heart failure, coronary heart disease, angina pectoris, heart attack, and stroke were evaluated by RCS after adjustment for the covariables. The solid lines correspond to the central estimates, and the shaded regions indicate the 95% confidence intervals. A CVD mortality; B Total CVD; C All-cause mortality; D Congestive heart failure; E Coronary heart disease; F Angina pectoris; G Heart attack; H Stroke
Figure 4 displays Kaplan–Meier survival curves stratified by CTI quartiles. Significant differences in CVD mortality risk emerged across the CTI quartile groups (log-rank p < 0.001), demonstrating the index's prognostic value. Furthermore, the association between the CTI index and all-cause mortality was also significant. The test for trend reached statistical significance. Kaplan–Meier analysis demonstrated progressively higher cumulative incidence rates for both cardiovascular and all-cause mortality across ascending CTI quartiles (Q1-Q4), with interquartile differences reaching statistical significance (P < 0.001).
Fig. 4.
Forest plot of the CTI association with CVD mortality, total CVD, and all-cause mortality using logistic regression model and Cox proportional hazards model in subgroups. A Gender; B Age; C Glycemic status
Stratification of CTI in relation to specific CVD conditions and Mortality outcomes
After controlling for variables, segmented analyses according to age, gender, glycemic status, identified that significant correlation between CTI with total CVD, CVD mortality, and all-cause mortality. Figure 5 demonstrates these associations according to age, gender, glycemic status. All association analyses are detailed in Tables 3, 4, 5. Following multivariable adjustment, the results showed that CTI were positively associated with CVD mortality, total CVD, and all-cause mortality.
Fig. 5.
The Kaplan–Meier curves analysis depicts the cumulative incidence of CVD mortality and all-cause mortality across the CTI index quartiles. A All-cause mortality; B CVD mortality
Table 3.
Association between the CTI and CVD mortality according to gender
| Died/participants | Model 1 | Model 2 | Model 3 | |
|---|---|---|---|---|
| Male | ||||
| CVD mortality | 210/4216 | |||
| Q1 | 36/1016 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 59/1129 | 1.20 (0.78–1.84) | 1.21 (0.80–1.83) | 1.68 (1.06–2.39)* |
| Q3 | 63/1055 | 1.31 (0.84–2.03) | 1.39 (0.92–2.09) | 2.13 (1.37–3.06)** |
| Q4 | 52/1016 | 1.42 (0.86–2.34) | 1.63 (1.06–2.50)* | 2.28 (1.47–3.46)** |
| All-cause mortality | 656/4216 | |||
| Q1 | 116/1016 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 178/1129 | 1.18 (0.93–1.50) | 1.13 (0.90–1.43) | 1.47 (1.15–2.81)** |
| Q3 | 194/1055 | 1.38 (1.07–1.77)* | 1.32 (1.05–1.67)* | 1.79 (1.08–2.38)** |
| Q4 | 168/1016 | 1.60 (1.21–2.12)** | 1.61 (1.27–2.05)** | 2.21 (2.76–4.89)** |
| Total-CVD | 555/4216 | |||
| Q1 | 90/1016 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 140/1129 | 1.03 (0.74–1.43) | 1.11 (0.82–1.51) | 1.43 (1.06–1.91)** |
| Q3 | 138/1055 | 0.80 (0.57–1.12) | 1.01 (0.74–1.37) | 1.65 (1.15–2.01)** |
| Q4 | 187/1016 | 1.01 (0.71–1.45) | 1.71 (1.27–2.29)** | 2.41 (1.80–3.05)** |
| Female | ||||
| CVD mortality | 150/4463 | |||
| Q1 | 27/1154 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 32/1041 | 0.71 (0.41–1.20) | 0.76 (0.46–1.27) | 1.58 (0.87–2.45) |
| Q3 | 45/1115 | 1.01 (0.60–1.72) | 1.03 (0.64–1.66) | 1.96 (1.21–3.12)** |
| Q4 | 46/1153 | 1.30 (0.73–2.33) | 1.56 (0.97–2.52) | 2.44 (1.53–3.94)** |
| All-cause mortality | 496/4463 | |||
| Q1 | 89/1154 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 135/1041 | 1.02 (0.77–1.34) | 1.02 (0.78–1.34) | 1.79 (1.43–2.45)** |
| Q3 | 141/1115 | 1.03 (0.77–1.38) | 1.02 (0.78–1.33) | 1.82 (1.46–2.47)** |
| Q4 | 131/1153 | 1.25 (0.90–1.73) | 1.35 (1.03–1.77)* | 2.11 (1.68–2.84)** |
| Total-CVD | 417/4463 | |||
| Q1 | 50/1154 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 90/1041 | 1.00 (0.67–1.49) | 1.34 (0.93–1.95) | 2.02 (1.42–2.93)** |
| Q3 | 109/1115 | 0.95 (0.64–1.42) | 1.49 (1.04–2.14)* | 2.36 (1.65–3.36)** |
| Q4 | 168/1153 | 1.24 (0.82–1.88) | 2.52 (1.79–3.54)** | 3.79 (2.68–5.29)** |
Model 1 did not adjust for any variables. Model 2 adjusted for sex and age. Model 3 adjusted for age, sex, education, race, smoking, alcohol use,self-reported diabetes, self-reported hypertension, systolic blood pressure (SBP), BMI, HDL, LDL, and HbA1c% levels
*P < 0.05, **P < 0.01
Table 4.
Association between the CTI and CVD mortality according to age
| Died/participants | Model 1 | Model 2 | Model 3 | |
|---|---|---|---|---|
| Age < 60 | ||||
| CVD mortality | 48/5694 | |||
| Q1 | 5/1690 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 9/1392 | 1.58 (0.52–4.84) | 1.78 (0.59–5.35) | 2.35 (0.77–3.81) |
| Q3 | 16/12977 | 3.06 (1.02–9.13)* | 3.64 (1.33–9.96)* | 2.64 (1.69–4.56)** |
| Q4 | 18/1315 | 3.12 (0.96–10.11) | 4.77 (1.75–12.97)** | 3.49 (2.37–5.51)** |
| All-cause mortality | 201/5694 | |||
| Q1 | 38/1690 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 53/1392 | 1.31 (0.85–2.03) | 1.38 (0.91–2.10) | 1.81 (1.19–2.80)** |
| Q3 | 43/1297 | 1.21 (0.74–1.96) | 1.28 (0.83–1.99) | 1.66 (1.10–2.60)* |
| Q4 | 67/1315 | 1.62 (0.97–2.70) | 2.36 (1.58–3.53)** | 3.31 (2.23–4.92)** |
| Total-CVD | 245/5694 | |||
| Q1 | 41/1690 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 49/1392 | 1.03 (0.65–1.61) | 1.17 (0.77–1.80) | 1.49 (0.96–2.27) |
| Q3 | 53/1297 | 0.99 (0.62–1.60) | 1.34 (0.88–2.04) | 1.72 (1.14–2.61)* |
| Q4 | 102/1315 | 1.40 (0.87–2.25) | 2.34 (1.60–3.42) | 3.39 (2.34–4.93)** |
| Age > = 60 | 2985 | |||
| CVD mortality | 312/2985 | |||
| Q1 | 58/480 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 82/778 | 0.92 (0.66–1.29) | 0.97 (0.70–1.36) | 0.94 (0.69–1.32) |
| Q3 | 92/873 | 1.01 (0.73–1.40) | 1.11 (0.80–1.55) | 1.13 (0.76–1.59) |
| Q4 | 80/954 | 1.17 (0.83–1.64) | 1.47 (1.05–2.07)* | 1.32 (0.85–1.99) |
| All-cause mortality | 951/2985 | |||
| Q1 | 167/480 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 260/778 | 1.01 (0.83–1.23) | 1.06 (0.87–1.28) | 1.08 (0.89–1.35) |
| Q3 | 292/873 | 1.11 (0.92–1.34) | 1.20 (0.99–1.45) | 1.27 (1.06–1.55)* |
| Q4 | 232/854 | 1.17 (0.95–1.42) | 1.41 (1.16–1.73)** | 1.46 (1.15–1.82)** |
| Total-CVD | 727/2985 | |||
| Q1 | 99/480 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 181/778 | 1.02 (0.75–1.38) | 1.18 (0.89–1.57) | 1.16 (0.85–1.57) |
| Q3 | 194/873 | 0.85 (0.63–1.16) | 1.11 (0.84–1.47) | 1.12 (0.86–1.47) |
| Q4 | 253/854 | 1.03 (0.74–1.42) | 1.79 (1.37–2.35)** | 1.60 (1.24–2.12)** |
Model 1 did not adjust for any variables. Model 2 adjusted for sex and age. Model 3 adjusted for age, sex, education, race, smoking, alcohol use, self-reported diabetes, self-reported hypertension, systolic blood pressure (SBP), BMI, HDL, LDL, and HbA1c% levels
*P < 0.05, **P < 0.01
Table 5.
Association between the CTI and CVD mortality according to Diabetes
| Died/participants | Model 1 | Model 2 | Model 3 | |
|---|---|---|---|---|
| Diabetes | ||||
| CVD mortality | 132/1632 | |||
| Q1 | 15/135 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 29/277 | 1.01 (0.54–1.87) | 0.99 (0.53–1.85) | 1.06 (0.57–1.96) |
| Q3 | 38/420 | 0.93 (0.51–1.69) | 1.04 (0.57–1.90) | 1.15 (0.63–2.12) |
| Q4 | 50/800 | 0.78 (0.44–1.39) | 1.08 (0.60–1.92) | 1.19 (0.60–2.23) |
| All-cause mortality | 407/1632 | |||
| Q1 | 34/135 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 86/277 | 1.31 (0.88–1.95) | 1.29 (0.87–1.93) | 1.29 (0.86–1.97) |
| Q3 | 122/420 | 1.31 (0.89–1.91) | 1.45 (0.99–2.12) | 1.57 (1.04–2.24)* |
| Q4 | 165/800 | 1.12 (0.77–1.62) | 1.48 (1.02–2.14)* | 1.61 (1.07–2.34)* |
| Total-CVD | 418/1632 | |||
| Q1 | 37/135 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 79/277 | 1.06 (0.67–1.67) | 0.96 (0.59–1.55) | 0.86 (0.49–1.45) |
| Q3 | 101/420 | 0.84 (0.54–1.30) | 0.79 (0.50–1.26) | 0.63 (0.38–1.05) |
| Q4 | 201/800 | 0.89 (0.59–1.34) | 1.03 (0.67–1.59) | 0.76 (0.41–1.17) |
| Non-diabetes | ||||
| CVD mortality | 228/7047 | |||
| Q1 | 48/2035 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 62/1893 | 0.94 (0.64–1.40) | 0.97 (0.67–1.42) | 1.52 (1.06–2.21)* |
| Q3 | 70/1750 | 1.21 (0.80–1.81) | 1.21 (0.84–1.75) | 1.89 (1.33–2.91)** |
| Q4 | 48/1369 | 1.72 (1.08–2.74)* | 1.76 (1.17–2.63)** | 2.21 (1.49–3.28)** |
| All-cause mortality | 745/7047 | |||
| Q1 | 171/2035 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 227/1893 | 1.04 (0.84–1.27) | 1.01 (0.83–1.23) | 1.55 (1.23–1.81)** |
| Q3 | 213/1750 | 1.11 (0.89–1.39) | 1.05 (0.86–1.28) | 1.63 (1.39–2.03)** |
| Q4 | 134/1369 | 1.48 (1.14–1.92)** | 1.36 (1.08–1.70)** | 1.75 (1.41–2.18)** |
| Total-CVD | 554/7047 | |||
| Q1 | 103/2035 | 1 (referent) | 1 (referent) | 1 (referent) |
| Q2 | 151/1893 | 1.08 (0.81–1.45) | 1.14 (0.87–1.50) | 1.65 (1.31–2.11)** |
| Q3 | 146/1750 | 1.03 (0.76–1.41) | 1.12 (0.85–1.48) | 1.72 (1.36–2.25)** |
| Q4 | 154/1369 | 1.45 (1.04–2.02)** | 1.84 (1.40–2.42)** | 2.44 (1.83–3.06)** |
Model 1 did not adjust for any variables. Model 2 adjusted for sex and age. Model 3 adjusted for age, sex, education, race, smoking, alcohol use, self-reported diabetes, self-reported hypertension, systolic blood pressure (SBP), BMI, HDL, LDL, and HbA1c% levels
*P < 0.05, **P < 0.01
As shown in Table 3, after controlling for variables, there was a significant association between CTI and CVD mortality in male individuals (HR 2.28, 95% CI 1.47–3.46) and female (HR 2.44, 95% CI 1.53–3.94). Our results also indicated that male individuals in the Q4 group with the highest CTI had a 2.41 times higher odds ratio for total CVD (95% CI: 1.80–3.05) compared to those in the Q1 group with the lowest CTI. Moreover, the ORs estimates show that the risks of total CVD in female with CTI in high concentrations groups were higher than the low group (OR, 3.79 [95% CI, 2.68–5.29]).
As shown in Table 4, in elderly participants, this research uncovered that individual categorized in the highest CTI (Q4) group exhibited a 1.60 odds ratio for total CVD (95% CI: 1.24–2.12) when compared to those in the lowest CTI (Q1) group. Our results also indicated that CTI had no association with CVD mortality in elderly participants. However, among younger individuals, CTI showed stronger CVD mortality prediction (HR 3.49, 2.37–5.51).
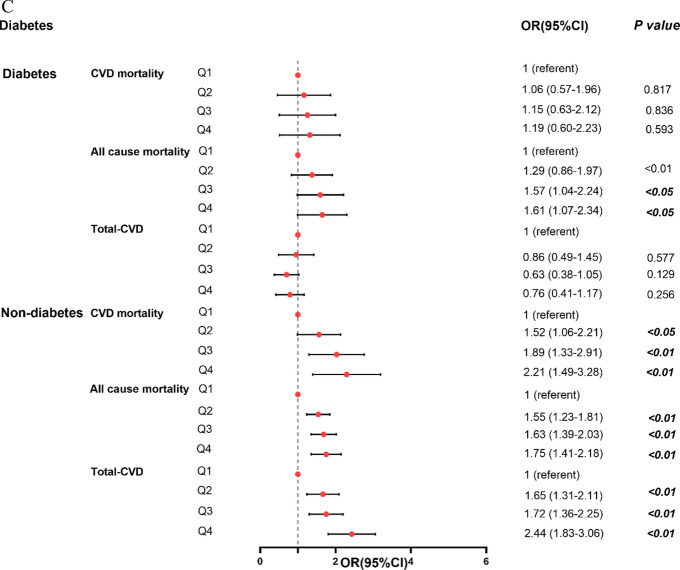
As shown in Table 5, elevated CTI levels were significantly associated with increased all-cause mortality among diabetic participants (HR = 1.61, 95% CI:1.07–2.34). For CVD mortality and total-CVD, CTI had no association in diabetes group. However, CTI had the highest association with CVD mortality (HR 2.21, 95% CI 1.49–3.28), total CVD (OR 2.44, 95% CI 1.83–3.06), and all-cause mortality (HR 1.75, 95% CI 1.41–2.18) in participants without DM.
As shown in Tables S1 and S2, in male participants, there were significant associations between CTI and congestive heart failure (OR 2.41, 95% CI 1.47–3.95), coronary heart disease (OR 1.97, 95% CI 1.30–2.97), angina pectoris (OR 2.02, 95% CI 1.39–4.02), heart attack (OR 2.29, 95% CI 1.53–3.42), and stroke (OR 2.24, 95% CI 1.39–3.61). As for female participants, similar associations were observed. As shown in Tables S3 and S4, in young (< 60 years old) participants, there were significant associations between CTI and coronary heart disease (OR 2.90, 95% CI 1.26–6.64), angina pectoris (OR 5.48, 95% CI 2.37–8.42), heart attack (OR 3.48, 95% CI 1.88–6.45), and stroke (OR 2.31, 95% CI 1.23–4.32). However, among older adults, CTI levels showed no statistically significant association with either angina pectoris or heart attack (all P > 0.05).
In addition, as shown in Tables S5 and S6, there were significant associations between CTI and congestive heart failure (OR 3.32, 95% CI 1.88–5.87), coronary heart disease (OR 2.37, 95% CI 1.50–3.75), angina pectoris (OR 2.82, 95% CI 1.54–5.17), heart attack (OR 2.17, 95% CI 1.44–3.26), and stroke (OR 2.06, 95% CI 1.31–3.24) in participants without diabetes. However, as for participants with DM, the risk of angina pectoris was significantly lower in Q4 (OR 0.53, 95% CI 0.29–0.98) compared to Q1, but insignificantly in Q2, and Q3. Furthermore, the risk of congestive heart failure was significantly lower in Q3 (OR 0.56, 95% CI 0.29–0.95) compared to Q1 in participants with DM.
The predictive value of CTI for specific CVD conditions and Mortality outcomes
In the ROC curve analysis, the predictive value of CTI for total-CVD, congestive heart failure, coronary heart disease, angina pectoris, heart attack, and stroke were shown in Fig. 6. The AUC of CTI for predicting coronary heart disease risk was 0.884 (95%CI = 0.835–0.933), followed by total-CVD (AUC = 0.850, 95%CI = 0.818–0.882), heart attack (AUC = 0.849, 95%CI = 0.799–0.898), congestive heart failure (AUC = 0.840, 95%CI = 0.782–0.899), angina pectoris (AUC = 0.814, 95%CI = 0.748–0.880) and stroke (AUC = 0.808, 95%CI = 0.747–0.868). The optimal CTI cutoff value, AUC values and DeLong’s test for CVDs were shown in Table S10.
Fig. 6.
Comparison of the predictive value of CTI index for total CVD, congestive heart failure, coronary heart disease, angina pectoris, heart attack, and stroke. a congestive heart failure; b coronary heart disease; c angina pectoris; d heart attack; e stroke; f Total CVD
Discussion
This investigation represents the first large-scale analysis to demonstrate significant associations between the CTI index and multiple CVD outcomes, including CVD-specific mortality, total CVD prevalence, and all-cause mortality, while evaluating these relationships across sex-specific, age-stratified, and glycemic subgroups. Our results indicate a robust positive correlation between elevated CTI levels and CVD incidence. Notably, these associations remained consistent regardless of sex or age, showing comparable effect magnitudes between male and female participants as well as across younger and older age groups. Stratified by glycemic status, elevated CTI levels demonstrated significant association with heightened CVD risk exclusively in non-diabetic individuals, with no such correlation detected in diabetic populations.
The CTI represents a novel biomarker, integrating two established physiological indicators: CRP for inflammatory status and the TyG index for IR [11]. The TyG index, calculated from fasting glucose and triglyceride measurements, has emerged as a widely adopted surrogate for IR due to its favorable combination of computational simplicity, sensitivity, and diagnostic specificity [46–48]. Recently, many new markers of metabolic inflammation have been shown to be associated with CVD risk. A novelty study demonstrates that the SEX-SHOCK score—a newly developed metric incorporating CRP—exhibits strong predictive accuracy for acute coronary syndromes (ACS) in both sexes across the ACS spectrum. These findings support its potential utility in guiding future intervention trials and refining contemporary ACS management strategies [49, 50]. Research conducted by Cao et al. demonstrates that elevated Systemic Inflammatory Response Index (SIRI) are associated with increased risks of all-cause and CVD mortality. This finding highlights the significance of targeted interventions to address systemic inflammation [51]. Additionally, a study has identified that a high hs-CRP/HDL-C ratio is a significant risk factor for CVD, stroke, and heart problems [52].
Previous studies have consistently demonstrated that higher TyG index values correlate with greater CVD risk. A U.S. cohort study of diabetic and prediabetic CVD patients identified a U-shaped relationship between baseline TyG levels and both CVD and all-cause mortality [6]. Beyond the conventional TyG index, its body composition-adjusted variants (TyG-WC and TyG-WHtR) show superior predictive capacity for CVD mortality and diagnostic performance for CVD outcomes compared to standard TyG measurements [40]. Complementary research by Wang et al. established CRP as an independent predictor of poor prognosis in non-infected stroke survivors during 90-day follow-up [53], while Cui et al. elucidated the synergistic interaction and bidirectional mediation between TyG and CRP in CVD pathogenesis [54]. Studies have shown that IBI levels, a conforming indicator of CRP and NLR, are associated with CVD prevalence in US adults [55]. Other studies have shown that CRP is associated with an increased risk of CVD in middle-aged Chinese population [56]. Moreover, the research of Burger et al. revealed that greater CRP is independently associated with an increased risk of recurrent CVD and mortality, irrespective of previous CVD location [57].
The enhanced clinical utility of the CTI index compared to isolated TyG or CRP biomarkers arises from three synergistic mechanisms. Firstly, CTI integrates complementary biological insights—the TyG index's reflection of insulin resistance risk and CRP's inflammatory status—providing a holistic metabolic-inflammatory profile. Additionally, isolated use of TyG or CRP captures singular pathological dimensions (glucose-lipid dysregulation or inflammation, respectively), while CTI's dual-marker framework reveals cross-system interactions through simultaneous metric evaluation. Notably, interindividual variability in biomarker expression exists, where the TyG index and CRP provide complementary pathophysiological perspectives—quantifying metabolic dysfunction and systemic inflammation, respectively [36, 58]. This integrative strategy improves precision in identifying high-risk subgroups requiring targeted intervention.
Among 8,679 NHANES participants, higher CTI values correlated with increased CVD mortality and incidence. Clinical interventions targeting CTI reduction could mitigate CVD risk in both younger and older adults. RCS modeling revealed a significant dose-dependent association between increasing CTI values and CVD event incidence. Our findings further identified consistent CTI-mortality relationships across sexes, though with marginally elevated HRs in female participants. This observation aligns with established sex-specific CVD pathophysiology—women exhibit distinct biological and psychosocial CVD risk profiles that develop across the life course [59, 60]. Particularly, their heightened susceptibility to autoimmune-mediated conditions (e.g., rheumatic disorders) contributes substantially to CVD pathogenesis [61]. Moreover, conventional risk factors (including smoking, hypertension, diabetes, obesity, sedentary behavior, mental health comorbidities, and genetic predisposition) demonstrate amplified effects in women relative to men [59, 61]. These collective findings underscore the clinical imperative for enhanced CTI surveillance in female populations as part of comprehensive CVD prevention strategies. Notably, younger adults exhibited elevated HRs compared to older populations, underscoring the importance of CTI monitoring in this age group. Our glycemic-stratified analysis revealed differential associations—while CTI showed significant predictive value for CVD mortality in non-diabetic individuals, this relationship was absent in those with established DM. CTI showed no meaningful association with CVD risk in DM patients. This may be due to individual differences in patients and the fact that diabetes-induced metabolic derangement drives CVD risk through other mechanisms (microvascular dysfunction, obesity, hypertension), which may mask the predictive role of CTI [62–64]. These findings indicate that maintaining optimal CTI ranges should be a clinical priority for non-diabetic individuals, while active reduction of elevated CTI may help prevent CVD events and associated mortality in this population. ROC curve showed that CTI had the best predictive ability for coronary heart disease. The diagnostic effects of CTI on total cardiovascular disease, congestive heart failure, angina pectoris, heart attack, and stroke were similar. The RCS analysis confirmed a significant linear relationship between CTI and CVD mortality, total CVD, and all-cause mortality. CTI lowering appears clinically warranted. This pioneering study examines CTI-CVD associations across demographic and metabolic subgroups. Furthermore, we also investigated the association between CTI and congestive heart failure, coronary heart disease, angina pectoris, heart attack, and stroke.
While the precise interaction between CTI and CVD remains incompletely characterized, emerging evidence points to several plausible pathways. CVD pathogenesis involves multifaceted biological processes, among which chronic inflammation acts as a central driver. Elevated levels of inflammatory biomarkers such as CRP correlate strongly with heart failure progression and adverse clinical outcomes [65]. Previous studies show that colchicine was the first drug to show potential benefits for coronary artery disease patients through reduced CRP levels, indicating inflammatory suppression [66]. Acute stroke triggers a rapid rise in CRP levels, reflecting systemic inflammatory activation during this critical phase [67]. Epidemiological evidence further demonstrates that higher CRP concentrations not only predict greater stroke risk in the general population [68], but also correlate with worse post-stroke recovery and increased fatality rates [69]. In conclusion, CRP is positively associated with the development of CVD and is risk factor for CVD [70–74]. Combined consideration of the levels of these inflammatory factors can help in the prediction of future CVD [70]. Recent studies have suggested that TyG index is an independent predictor of CVD [75]. Higher TyG index level was independently associated with a higher incidence of CVD [75]. The TyG index has gained recognition as a novel IR marker [27, 76–78], demonstrating strong correlations with various CVD conditions including hypertension, atherosclerotic disease, cerebrovascular events, and adverse CVD outcomes [29, 79, 80]. Parallel evidence supports the clinical utility of CTI as a dual indicator of inflammatory status and IR severity. This composite index has shown prognostic relevance in diverse clinical contexts, from cancer-associated wasting syndromes [81] to population-level malignancy mortality [14]. While CTI's pathophysiological pathways in CVD remain incompletely understood, our investigation advances this knowledge by evaluating its predictive capacity across sex-stratified, age-specific, and glucose-metabolic subgroups. These findings acquire particular significance given the escalating global CVD burden, especially among individuals exhibiting concurrent inflammatory and metabolic dysfunction-a high-risk population disproportionately vulnerable to CVD events. These results underscore the importance of early identification of high CVD risk individuals. Clinically, maintaining CTI within optimal ranges may serve as an effective strategy for CVD prevention and mortality reduction.
This study also has some limitations. Firstly, despite the inclusion of many potential confounders, there may still be some unconsidered factors such as environmental factors, lifestyle, and the time duration of living with CVD prior to death. This will be explored in greater depth as the database is updated in the future. Secondly, the use of single-timepoint TyG measurements at baseline. This methodological constraint precluded longitudinal assessment of TyG's association with both CVD and all-cause mortality outcomes. Thirdly, we only used NHANES database for the study, which may result in the regional, healthcare setting, and patient population bias, and future studies should consider the use of multicenter data to improve external validity. Further investigation should verify CTI's cardiovascular predictive capacity across diverse populations and clinical settings. Subsequent intervention trials could then evaluate whether CTI modulation effectively mitigates both CVD incidence and associated mortality.
Conclusions
Collectively, our findings demonstrate that elevated CTI levels are robustly associated with increased risks of CVD mortality, total CVD incidence, and all-cause mortality. These associations were similar between male and female, and similar in young and elderly participants. In different glycemic status, the association was significant in individuals without diabetes. However, this association was not observed in individuals with diabetes. These findings carry significant public health relevance, offering improved methods for early CVD risk detection and enhanced stratification approaches applicable to population-level studies.
Supplementary Information
Acknowledgements
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Abbreviations
- TyG
Triglyceride-glucose
- CVD
Cardiovascular disease
- CRP
C-reactive protein
- NHANES
National health and nutrition examination survey
- RCS
Restricted cubic spline
- ROC
Receiver operating characteristic
- BMI
Body mass index
- HDL
High-density lipoprotein cholesterol
- LDL
Low-density lipoprotein cholesterol
- TG
Triglyceride
- HbA1c
Glycated hemoglobin
- Ors
Odds ratios
- Cis
Confidence intervals
Author contributions
All authors contributed to the study conception and design. YS, and YG designed the study and conducted the data analysis. YS drafted the manuscript. SYM, ZM, DGM, KGX, RGL, XDP and XYZ proposed critical revisions to the manuscript. All included authors made contributions to the manuscript and approved the version submitted.
Funding
This study was supported by the National Natural Science Foundation of China (grant number: 82471351 and 82271346).
Data availability
Data described in the manuscript, code book, and analytic code will be made publicly and freely available without restriction at https://www.cdc.gov/nchs/nhanes/index.htm.
Declarations
Ethics approval and consent to participate
NHANES is conducted by the Centers for Disease Control and Prevention (CDC) and the National Center for Health Statistics (NCHS). And the NHANES study protocol was reviewed and approved by the NCHS Research Ethics Review Committee. All participants in NHANES provided written informed consent.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yu Sun and Yu Guo have contributed equally to this work.
Contributor Information
Ruogu Lu, Email: ruogulu2023@163.com.
Xudong Pan, Email: drpan022@qdu.edu.cn.
Xiaoyan Zhu, Email: zxysdjm@qdu.edu.cn.
References
- 1.Vaduganathan M, Mensah GA, Turco JV, Fuster V, Roth GA. The global burden of cardiovascular diseases and risk: a compass for future health. J Am Coll Cardiol. 2022;80(25):2361–71 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 2.Roth GA, et al. Global burden of cardiovascular diseases and risk factors, 1990–2019: update from the GBD 2019 study. J Am Coll Cardiol. 2020;76(25):2982–3021 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhao D, Liu J, Wang M, Zhang X, Zhou M. Epidemiology of cardiovascular disease in China: current features and implications. Nat Rev Cardiol. 2019;16(4):203–12 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 4.Woodruff RC, et al. Trends in cardiovascular disease mortality rates and excess deaths, 2010–2022. Am J Prev Med. 2024;66(4):582–9 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Virani SS, et al. Heart disease and stroke statistics-2020 update: a report from the american heart association. Circulation. 2020;141(9):e139–596 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 6.Zhang Q, Xiao S, Jiao X, Shen Y. The triglyceride-glucose index is a predictor for cardiovascular and all-cause mortality in CVD patients with diabetes or pre-diabetes: evidence from NHANES 2001–2018. Cardiovasc Diabetol. 2023;22(1):279 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cosentino F, et al. 2019 ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur Heart J. 2020;41(2):255–323 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 8.Abdul-Ghani MA, Jayyousi A, DeFronzo RA, Asaad N, Al-Suwaidi J. Insulin resistance the link between T2DM and CVD: basic mechanisms and clinical implications. Curr Vasc Pharmacol. 2019;17(2):153–63 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 9.Wong ND, Sattar N. Cardiovascular risk in diabetes mellitus: epidemiology, assessment and prevention. Nat Rev Cardiol. 2023;20(10):685–95 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 10.Einarson TR, Acs A, Ludwig C, Panton UH. Prevalence of cardiovascular disease in type 2 diabetes: a systematic literature review of scientific evidence from across the world in 2007–2017. Cardiovasc Diabetol. 2018;17(1):83 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ruan G-T, et al. A novel inflammation and insulin resistance related indicator to predict the survival of patients with cancer. Front Endocrinol. 2022;13: 905266 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huang C, et al. Association between C-reactive protein-triglyceride glucose index and depressive symptoms in American adults: results from the NHANES 2005 to 2010. BMC Psychiatry. 2024;24(1):890 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mei Y, Li Y, Zhang B, Xu R, Feng X. Association between the C-reactive protein-triglyceride glucose index and erectile dysfunction in US males: results from NHANES 2001–2004. Int J Impot Res. 2024. 10.1038/s41443-024-00945-z. ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhao D-F. "Value of C-reactive protein-triglyceride glucose index in predicting cancer mortality in the general population: results from national health and nutrition examination survey. Nutr Cancer. 2023;75(10):1934–44 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 15.Zhou Y, Lin H, Weng X, Dai H, Xu J. Correlation between hs-CRP-triglyceride glucose index and NAFLD and liver fibrosis. BMC Gastroenterol. 2025;25(1):252 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pedersen BK. Anti-inflammatory effects of exercise: role in diabetes and cardiovascular disease. Eur J Clin Invest. 2017;47(8):600–11 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 17.Giannakopoulou S-P, Antonopoulos A, Panagiotakos D. Serum inflammatory markers used in cardiovascular disease risk prediction models: a systematic review. Angiology. 2024. 10.1177/00033197241239691. ((in eng)). [DOI] [PubMed] [Google Scholar]
- 18.Olsen MB, et al. Targeting the inflammasome in cardiovascular disease. JACC Basic Transl Sci. 2022;7(1):84–98 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kunutsor SK, et al. Plasma calprotectin and risk of cardiovascular disease: findings from the PREVEND prospective cohort study. Atherosclerosis. 2018;275:205–13 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 20.Avci E, Karabulut A, Alp AG, Baba B, Bilgi C. Crucial markers showing the risk of coronary artery disease in obesity: ADMA and neopterin. J Med Biochem. 2020;39(4):452–9 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Løfblad L, Hov GG, Åsberg A, Videm V. Inflammatory markers and risk of cardiovascular mortality in relation to diabetes status in the HUNT study. Sci Rep. 2021;11(1):15644 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Natorska J, Ząbczyk M, Undas A. Neutrophil extracellular traps (NETs) in cardiovascular diseases: from molecular mechanisms to therapeutic interventions. Kardiol Pol. 2023;81(12):1205–16 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 23.Huo G, Tang Y, Liu Z, Cao J, Yao Z, Zhou D. Association between C-reactive protein-triglyceride glucose index and stroke risk in different glycemic status: insights from the China Health and Retirement Longitudinal Study (CHARLS). Cardiovasc Diabetol. 2025;24(1):142 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tang S, et al. C-reactive protein-triglyceride glucose index predicts stroke incidence in a hypertensive population: a national cohort study. Diabetol Metab Syndr. 2024;16(1):277 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Di Pino A, DeFronzo RA. Insulin resistance and atherosclerosis: implications for insulin-sensitizing agents. Endocr Rev. 2019;40(6):1447–67 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Scott DA, Ponir C, Shapiro MD, Chevli PA. Associations between insulin resistance indices and subclinical atherosclerosis: a contemporary review. Am J Prev Cardiol. 2024;18: 100676 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Simental-Mendía LE, Rodríguez-Morán M, Guerrero-Romero F. The product of fasting glucose and triglycerides as surrogate for identifying insulin resistance in apparently healthy subjects. Metab Syndr Relat Disord. 2008;6(4):299–304 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 28.Tao L-C, Xu J-N, Wang T-T, Hua F, Li J-J. Triglyceride-glucose index as a marker in cardiovascular diseases: landscape and limitations. Cardiovasc Diabetol. 2022;21(1):68 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lu Y-W, et al. Gender difference in the association between TyG index and subclinical atherosclerosis: results from the I-Lan Longitudinal Aging Study. Cardiovasc Diabetol. 2021;20(1):206 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wu Y, Yang Y, Zhang J, Liu S, Zhuang W. The change of triglyceride-glucose index may predict incidence of stroke in the general population over 45 years old. Cardiovasc Diabetol. 2023;22(1):132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cui C, et al. Comparison of triglyceride glucose index and modified triglyceride glucose indices in prediction of cardiovascular diseases in middle aged and older Chinese adults. Cardiovasc Diabetol. 2024;23(1):185 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Xia X, et al. Association of triglyceride-glucose index and its related parameters with atherosclerotic cardiovascular disease: evidence from a 15-year follow-up of Kailuan cohort. Cardiovasc Diabetol. 2024;23(1):208 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Xu L, et al. Triglyceride-glucose index associates with incident heart failure: a cohort study. Diabetes Metab. 2022;48(6): 101365 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 34.Sun C, et al. Triglyceride-glucose index’s link to cardiovascular outcomes post-percutaneous coronary intervention in China: a meta-analysis. ESC Heart Failure. 2024;11(3):1317–28 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Liu Y, et al. Triglyceride-glucose index as a marker of adverse cardiovascular prognosis in patients with coronary heart disease and hypertension. Cardiovasc Diabetol. 2023;22(1):133 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nordestgaard BG, Zacho J. Lipids, atherosclerosis and CVD risk: is CRP an innocent bystander? Nutr Metab Cardiovasc Dis. 2009;19(8):521–4 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 37.Burger PM, et al. C-reactive protein and risk of incident heart failure in patients with cardiovascular disease. J Am Coll Cardiol. 2023;82(5):414–26 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 38.Rolver MG, Emanuelsson F, Nordestgaard BG, Benn M. Contributions of elevated CRP, hyperglycaemia, and type 2 diabetes to cardiovascular risk in the general population: observational and Mendelian randomization studies. Cardiovasc Diabetol. 2024;23(1):165 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Xu M, Zhang L, Xu D, Shi W, Zhang W. Usefulness of C-reactive protein-triglyceride glucose index in detecting prevalent coronary heart disease: findings from the national health and nutrition examination survey 1999–2018. Front Cardiovasc Med. 2024;11:1485538 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dang K, et al. The association between triglyceride-glucose index and its combination with obesity indicators and cardiovascular disease: NHANES 2003–2018. Cardiovasc Diabetol. 2024;23(1):8 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Li H, et al. Association of depressive symptoms with incident cardiovascular diseases in middle-aged and older chinese adults. JAMA Netw Open. 2019;2(12): e1916591 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Xie W, Zheng F, Yan L, Zhong B. Cognitive decline before and after incident coronary events. J Am Coll Cardiol. 2019;73(24):3041–50 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 43.He H-M, et al. The synergistic effect of the triglyceride-glucose index and a body shape index on cardiovascular mortality: the construction of a novel cardiovascular risk marker. Cardiovasc Diabetol. 2025;24(1):69 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Yin B, Wu Z, Xia Y, Xiao S, Chen L, Li Y. Non-linear association of atherogenic index of plasma with insulin resistance and type 2 diabetes: a cross-sectional study. Cardiovasc Diabetol. 2023;22(1):157 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Qiu Z, et al. Associations of serum carotenoids with risk of cardiovascular mortality among individuals with type 2 diabetes: results from NHANES. Diabetes Care. 2022;45(6):1453–61 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 46.Khan SH, Sobia F, Niazi NK, Manzoor SM, Fazal N, Ahmad F. Metabolic clustering of risk factors: evaluation of Triglyceride-glucose index (TyG index) for evaluation of insulin resistance. Diabetol Metab Syndr. 2018;10:74 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Du T, Yuan G, Zhang M, Zhou X, Sun X, Yu X. Clinical usefulness of lipid ratios, visceral adiposity indicators, and the triglycerides and glucose index as risk markers of insulin resistance. Cardiovasc Diabetol. 2014;13:146 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Guerrero-Romero F, et al. The product of triglycerides and glucose, a simple measure of insulin sensitivity. Comparison with the euglycemic-hyperinsulinemic clamp. J Clin Endocrinol MetabIsm. 2010;95(7):3347–51 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 49.Wang Y, et al. Sex-specific prediction of cardiogenic shock after acute coronary syndromes: the SEX-SHOCK score. Eur Heart J. 2024;45(43):4564–78 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kraler S, Mueller C, Libby P, Bhatt DL. Acute coronary syndromes: mechanisms, challenges, and new opportunities. Eur Hear J. 2025. 10.1093/eurheartj/ehaf28. ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cao Y, Wang W, Xie S, Xu Y, Lin Z. Joint association of the inflammatory marker and cardiovascular-kidney-metabolic syndrome stages with all-cause and cardiovascular disease mortality: a national prospective study. BMC Public Health. 2025;25(1):10 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gao Y, et al. The predictive value of the hs-CRP/HDL-C ratio, an inflammation-lipid composite marker, for cardiovascular disease in middle-aged and elderly people: evidence from a large national cohort study. Lipids Health Dis. 2024;23(1):66 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wang L, Li Y, Wang C, Guo W, Liu M. C-reactive protein, infection, and outcome after acute ischemic stroke: a registry and systematic review. Curr Neurovasc Res. 2019;16(5):405–15. [DOI] [PubMed] [Google Scholar]
- 54.Cui C, et al. Joint association of TyG index and high sensitivity C-reactive protein with cardiovascular disease: a national cohort study. Cardiovasc Diabetol. 2024;23(1):156 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Yu F, Peng J. Association between Inflammatory Burden Index and cardiovascular disease in adult Americans: Evidence from NHANES 2005–2010. Heliyon. 2024;10(18): e38273 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Dong Y, et al. High-sensitivity C reactive protein and risk of cardiovascular disease in China-CVD study. J Epidemiol Community Health. 2019;73(2):188–92 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 57.Burger PM, et al. C-reactive protein and risk of cardiovascular events and mortality in patients with various cardiovascular disease locations. Am J Cardiol. 2023;197:13–23 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 58.Li J, Ma H. Associations of the hs-CRP/HDL-C ratio with cardiovascular disease among US adults: evidence from NHANES 2015–2018. Nutr Metab Cardiovasc Dis. 2025;35(4): 103814 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 59.Mulvagh SL, et al. The canadian women’s heart health alliance atlas on the epidemiology, diagnosis, and management of cardiovascular disease in women - chapter 4: sex- and gender-unique disparities: CVD across the lifespan of a woman. CJC Open. 2022;4(2):115–32 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Norris CM, et al. State of the science in women’s cardiovascular disease: a canadian perspective on the influence of sex and gender. J Am Heart Assoc. 2020;9(4): e015634 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Jaffer S, et al. The canadian womens heart health alliance ATLAS on the epidemiology diagnosis and management of cardiovascular disease in women-chapter 2 scope of the problem. CJC Open. 2021;3(1):1–11 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.van Sloten TT, Sedaghat S, Carnethon MR, Launer LJ, Stehouwer CDA. Cerebral microvascular complications of type 2 diabetes: stroke, cognitive dysfunction, and depression. Lancet Diabetes Endocrinol. 2020;8(4):325–36 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Bhupathiraju SN, Hu FB. Epidemiology of Obesity and Diabetes and Their Cardiovascular Complications. Circ Res. 2016;118(11):1723–35 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Bruno A. Pre-diabetes, diabetes, hyperglycemia, and stroke: bittersweet therapeutic opportunities. Curr Neurol Neurosci Rep. 2022;22(11):781–7 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 65.Zhang Y, Bauersachs J, Langer HF. Immune mechanisms in heart failure. Eur J Heart Fail. 2017;19(11):1379–89 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 66.Nidorf M, Thompson PL. Effect of colchicine (0.5 mg twice daily) on high-sensitivity C-reactive protein independent of aspirin and atorvastatin in patients with stable coronary artery disease. Am J Cardiol. 2007;99(6):805–7 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 67.Emsley HCA, et al. An early and sustained peripheral inflammatory response in acute ischaemic stroke: relationships with infection and atherosclerosis. Journal of Neuroimmunology. 2003;139:1–2 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 68.Ballantyne CM, et al. Lipoprotein-associated phospholipase A2, high-sensitivity C-reactive protein, and risk for incident ischemic stroke in middle-aged men and women in the Atherosclerosis Risk in Communities (ARIC) study. Arch Intern Med. 2005;165(21):2479–84 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 69.Elkind MSV, Tai W, Coates K, Paik MC, Sacco RL. High-sensitivity C-reactive protein, lipoprotein-associated phospholipase A2, and outcome after ischemic stroke. Arch Intern Med. 2006;166(19):2073–80 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 70.Liu Y, Guan S, Xu H, Zhang N, Huang M, Liu Z. Inflammation biomarkers are associated with the incidence of cardiovascular disease: a meta-analysis. Front Cardiovasc Med. 2023;10:1175174 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Bafei SEC, et al. Interactive effect of increased high sensitive C-reactive protein and dyslipidemia on cardiovascular diseases: a 12-year prospective cohort study. Lipids Health Dis. 2023;22(1):95 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.de Ferranti S, Rifai N. C-reactive protein and cardiovascular disease: a review of risk prediction and interventions. Clin Chim Acta Int J Clin Chem. 2002;317:1–2 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 73.Chia JE, Ang SP. Elevated C-reactive protein and cardiovascular risk. Curr Opin Cardiol. 2025. 10.1097/HCO.0000000000001215. ((in eng)). [DOI] [PubMed] [Google Scholar]
- 74.Mazer SP, Rabbani LE. Evidence for C-reactive protein’s role in (CRP) vascular disease: atherothrombosis, immuno-regulation and CRP. J Thromb Thrombolysis. 2004;17(2):95–105 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 75.Li H, et al. Triglyceride-glucose index variability and incident cardiovascular disease: a prospective cohort study. Cardiovasc Diabetol. 2022;21(1):105 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Tahapary DL, et al. Challenges in the diagnosis of insulin resistance: Focusing on the role of HOMA-IR and Tryglyceride/glucose index. Diabetes & MetabIc Syndr. 2022;16(8): 102581 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 77.Ramdas Nayak VK, Satheesh P, Shenoy MT, Kalra S. Triglyceride Glucose (TyG) Index: a surrogate biomarker of insulin resistance, JPMA. J Pak Med Assoc. 2022;72(5):986–8 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 78.Sergi D, et al. HOMA-IR and TyG index differ for their relationship with dietary, anthropometric, inflammatory factors and capacity to predict cardiovascular risk. Diabetes Res Clin Pract. 2025;222: 112103 ((in eng)). [DOI] [PubMed] [Google Scholar]
- 79.Gao Q, et al. Positive association of triglyceride-glucose index with new-onset hypertension among adults: a national cohort study in China. Cardiovasc Diabetol. 2023;22(1):58 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Liu C, Liang D. The association between the triglyceride-glucose index and the risk of cardiovascular disease in US population aged ≤ 65 years with prediabetes or diabetes: a population-based study. Cardiovasc Diabetol. 2024;23(1):168 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ruan G-T, et al. Systemic inflammation and insulin resistance-related indicator predicts poor outcome in patients with cancer cachexia. Cancer & Metabolism. 2024;12(1):3 ((in eng)). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data described in the manuscript, code book, and analytic code will be made publicly and freely available without restriction at https://www.cdc.gov/nchs/nhanes/index.htm.