ABSTRACT
Pulmonary Fibrosis (PF) is a life‐threatening illness that is characterized by progressive scarring in the lung interstitium. There is an urgent need for new PF therapies because current treatments only slow down the progression of fibrosis, and the median life expectancy post‐diagnosis is only 4–6 years. Since PF patients frequently exhibit telomere attrition, overexpressing telomerase, the enzyme responsible for synthesizing telomeres, represents a compelling therapeutic option. In this study, we in vitro transcribed human telomerase reverse transcriptase (hTERT) mRNA using modified nucleosides (modRNA). ModRNA hTERT treatment led to transient activation of telomerase activity in a dose‐dependent manner in MRC‐5 cells and, importantly, in primary human alveolar type II pneumocytes. Consequently, the proliferative capacity was increased, concomitant with reduced DNA damage and elongated telomere length. Notably, the induction of cellular immune response was only detectable at the highest modRNA concentration and returned to normal levels within 48 h. Next, we demonstrated that circularized, exonuclease‐resistant modRNA hTERT extended the transient expression profile, which may be clinically advantageous. Finally, we provided therapeutic proof of concept in organotypic 3D ex vivo human precision‐cut lung slices derived from end‐stage PF patients. Intriguingly, a single modRNA hTERT treatment inhibited senescence, as indicated by significantly lower levels of senescence‐associated β‐galactosidase. Pro‐inflammatory markers (IL6 and IL8) and, concurrently, the key fibrosis mediators TGFβ and COL1A1 were markedly reduced after modRNA and circular RNA hTERT treatment. In conclusion, the data presented herein provide initial evidence for the potential of RNA‐based hTERT therapy for treating human lung fibrosis.
Keywords: lung ATII cell proliferation, modRNA, pulmonary fibrosis, senescence, telomerase reverse transcriptase (TERT), telomere elongation, TERT therapy
Recombinant linear and circular hTERT mRNA restore telomerase activity in primary ATII cells and ex vivo lung tissue from patients with pulmonary fibrosis, leading to telomere elongation, reduced DNA damage, and decreased senescence and fibrosis markers. These findings support RNA‐based hTERT therapy as a promising strategy for treating pulmonary fibrosis.
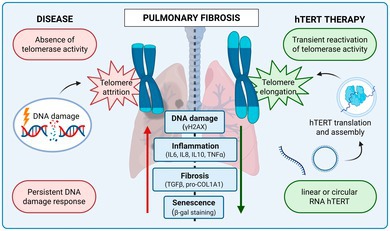
1. Introduction
Lung diseases arise due to a variety of factors such as viral or bacterial infection, asthma, bronchitis, fibrosis, air pollution, or occupational risks affecting specific cell types of the lung. Interstitial lung diseases (ILD) comprise several heterogeneous conditions which ultimately lead to inflammation and scarring of the lung tissue. Out of these, pulmonary fibrosis (PF) is a chronic fatal subtype of ILD, marked by remodeling of the alveolar compartment, progressive fibrosis in the interstitium, and hindrance of proper gas exchange, ultimately resulting in respiratory failure (Cottin et al. 2018; Fan et al. 2024). In addition, a worse survival prognosis of only 4–6 years post‐diagnosis (Cottin et al. 2018; Khor et al. 2023; Strand et al. 2014; Yamazaki et al. 2022) puts these lung patients at high risk. There are currently two FDA‐approved drugs, pirfenidone and nintedanib, which show potential to slow down the fibrotic progression but fail to reverse or cure the disease, highlighting an urgent need to discover novel therapeutic strategies against PF (Cilli and Uzer 2023; Wijsenbeek and Cottin 2020).
Telomere dysfunction, a hallmark of aging, has also been recognized as a major risk factor for both PF and its subtype idiopathic pulmonary fibrosis (IPF) pathology caused by chronic alveolar epithelial injury, senescence, and tissue remodeling (Alder and Armanios 2022; McDonough et al. 2018; Newton et al. 2016; Piñeiro‐Hermida et al. 2022; Van Batenburg et al. 2020). The aging population is most affected by IPF, with an overall prevalence of 10–60 cases per 100,000 individuals, which drastically increased to 400 cases per 100,000 in individuals over 65 years of age (Esposito et al. 2015; Raghu et al. 2014). There is a high correlation between telomere length and IPF disease severity (Adegunsoye et al. 2023; Duckworth et al. 2021). Around 25% of individuals with familial pulmonary fibrosis (FPF) exhibit mutations in telomere maintenance genes, such as TERT, TERC, DKC1, TIN2, and RTEL1, predisposing these patients to senescence and subsequently early alveolar epithelial cell failure (Peljto et al. 2023; Podolanczuk et al. 2023). Notably, those with sporadic IPF without any known mutations in the telomere machinery possess elevated levels of telomere shortening too; indicating that senescence induced by shortened telomeres is a unifying factor of both the sporadic and FPF forms of IPF (Cronkhite et al. 2008). Mechanistically, telomere shortening and dysfunction in alveolar type II pneumocytes (ATII cells) have been shown to promote DNA damage and cellular senescence (Alder et al. 2015; Van Batenburg et al. 2021; Yazicioglu et al. 2020). In addition to producing surfactant proteins (SP), the ATII cells also serve as progenitors for alveolar type I pneumocytes (ATI cells). Hence, ATII cell senescence results in limited regeneration capacity after lung injury (Hirsch et al. 2024; Povedano et al. 2015). Moreover, senescent ATII cells secrete pro‐fibrotic factors which promote the development of PF. Ultimately, the progressive expansion of fibroblast and myofibroblasts leads to extracellular matrix remodeling (Spagnolo et al. 2021).
Considering that telomere shortening is a key driver and strongly associated with PF, reactivating the enzyme telomerase which synthesizes telomere repeats would evidently offer a powerful therapeutic approach to replenish telomeres and to mitigate cellular senescence in PF (Povedano et al. 2018; Bär and Blasco 2016). Promising results after reactivation of telomerase reverse transcriptase (TERT), the catalytic subunit of telomerase, have been shown in a mouse model of bleomycin induced PF. AAV9‐Tert‐mediated gene therapy showed improvement in lung volume and mechanics as well as lower inflammation concomitant with improved regression of the fibrosis (Povedano et al. 2018). Despite successful TERT reactivation using gene therapy tools in mouse models, AAV vectors present some concerns regarding clinical application due to the slow kinetics of transgene expression and the long‐term and constitutive TERT overexpression which imposes risks of tumorigenesis (Penaud‐Budloo et al. 2008). Alternatively, the use of an mRNA encoding human telomerase reverse transcriptase (hTERT) enables a much faster and transient reactivation of telomerase activity which might be sufficient to achieve telomere elongation, thus offering a safer and efficient therapeutic approach. If needed, the transient RNA‐mediated hTERT reactivation may be extended by utilizing circularized versions of the mRNA or self‐amplifying mRNAs which offer higher stability and longer expression (Wesselhoeft et al. 2018).
In this study, we utilized modRNA hTERT to reactivate telomerase activity in primary human ATII cells, which led to improved cellular fitness and telomere elongation. We demonstrated that circularized RNA hTERT is functional and extends the window of transient telomerase activity. Finally, we utilized precision‐cut lung slices (PCLS) from end‐stage lung fibrosis patients to provide the first proof‐of‐principle for the anti‐senescent and anti‐fibrotic effectiveness of hTERT modRNA therapy in human lung fibrosis.
2. Methods
2.1. Human Lung Material
Human primary ATII cells (EP71AL Epithelix) were obtained from non‐smoking donors with no reported pathology (Epithelix Sarl, Geneva, Switzerland).
Human Precision‐cut lung slices (PCLS) of end‐stage PF patients were diagnosed with usual interstitial pneumonia (UIP) pattern and were provided by the Hanover Medical School (MHH, Hanover, Germany). Patients' demographic and diagnosis are summarized in Table 1. The experiments with human lung tissue were approved by the ethics committee of the Hannover Medical School (Hannover, Germany) in compliance with “The Code of Ethics of the World Medical Association” (renewed on 2015/04/22, number 2701–2015). All patients or their next of kin, gave written informed consent for using their lung tissue for research.
TABLE 1.
Patients' demographic and diagnosis.
| Gender, age (years) | Diagnosis | Corresponding figure(s) |
|---|---|---|
| Female, 64 | Tumor | Figure 4A, Figure S4B |
| Female, 58 | UIP, EAA fibrosis | Figure 4A, Figure S4B |
| Male, 49 | UIP; secondary fibrosis | Figure 4B–D |
| Male, 53 | UIP; secondary fibrosis | Figure 4A–D |
| Male, 61 | UIP; IPF | Figure 4B–D |
| Male, 56 | UIP; IPF | Figure 4B–D |
| Female, 64 | UIP; secondary fibrosis | Figure 4E–G, Figure S4C,D |
| Male, 48 | UIP; secondary fibrosis | Figure 4E–G, Figure S4C,D |
| Female, 63 | UIP; IPF | Figure 4E–G, Figure S4C,D |
Abbreviations: EAA, extrinsic allergic alveolitis; UIP, usual interstitial pneumonia.
2.2. Cell Culture, Quantitative Growth Curve and Transfection
Human primary fetal lung cells (MRC‐5 cell line) and HEK293 cells were cultured in high glucose DMEM (41965062 GibcoTM) with 10% fetal bovine serum and 1% penicillin/streptomycin. Human primary ATII cells were cultured with ready‐to‐use Epithelix Culture Media. Cells were counted with the Countess II from Invitrogen. To generate the growth curve, population doubling level (PDL) was calculated by using the equation PDL = 3.32 · (log n2—log n1) + x, where n1 and n2 are the respective population sizes and x is the PDL from the previous passage. For transfection, Lipofectamine RNAiMAX (133778075 Thermo Fisher Scientific) was mixed with 500 ng modRNA in Opti‐MEM in a 2:1 vol:vol ratio and added to the cells in a 0.5 mL transfection volume to achieve a final treatment concentration of 1 μg/mL. For dose‐dependent concentration, the modRNA amounts were adjusted accordingly. After 4 h, culture media (1.5× of from the total transfection volume) was added onto the transfected cells. All the transfections were performed at the cell confluence of 80%.
2.3. Precision‐Cut Lung Slices
Precision‐cut lung slices were generated based on previously described protocols (Hesse et al. 2022; Switalla et al. 2010). After an overnight recovery period, PCLS were treated with Lipofectamine RNAiMAX (133778075 Thermo Fisher Scientific) and 500 ng modRNA in Opti‐MEM in a 2:1 vol:vol ratio for 4 h. Afterwards, 1.5× culture media (of the total transfection volume) was added onto the transfected cells for subsequent culture at 37°C and 5% CO2, with a medium exchange 48 h post‐transfection. Culture media for PCLS comprised of DMEM/F12 (11039021 Gibco) and 1% penicillin/streptomycin.
2.4. Lactate Dehydrogenase (LDH) Assay
Tissue viability was assessed using the lactate dehydrogenase (LDH) based Cytotoxicity Detection Kit (11644793001 Roche) according to the manufacturer's recommendations.
2.5. Caspase 3/7 Assay
Apoptosis was assessed by measuring caspase 3/7 activity with the Caspase Glo 3/7 Assay system (G8090 Promega) according to the manufacturer's recommendations.
2.6. Enzyme‐Linked Immunosorbent Assay (ELISA) Assay
PCLS culture supernatants were collected after 96 h, supplemented with 0.2% protease inhibitor cocktail (87,785 Thermo Fisher Scientific), and stored at −80°C. Human IL6, IL8, TGFβ and pro‐COL1A1 were measured using DuoSet ELISA kits from R&D Systems (DY206, DY208, DY6220 and DY240) according to the manufacturer's recommendations.
Broad panel of immune cytokines was measured using V‐PLEX Pro‐inflammatory Panel 1 Human Kit (K15049D, Meso Scale Diagnostics) according to manufacturer's recommendations.
2.7. Senescence‐Associated β‐Galactosidase Staining
Senescence‐associated β‐galactosidase (SA‐β‐gal) levels were assessed within the tissue after 96 h using a β‐Gal Staining Kit (9860 Cell Signaling) and performed according to the manufacturer's recommendations, adjusting staining/fixation volumes to 500 μL per PCLS. Overview images of the stained PCLS were recorded using a stereomicroscope (Discovery V8; Zeiss, Germany) controlled by the Axio Vision 4.8.2 software program (Zeiss, Germany). Afterwards, the tissue was embedded in OCT (6478.2 Roth), frozen to −20°C, and sectioned into 10 μm‐thick slices. Cryo sections were fixed on SuperFrost microscope slides (J1800AMNZ Epredia) with DPX mounting medium (10021203 Sigma Aldrich). Embedded β‐gal stained PCLS slides were imaged with a 20× objective (PlanApo 20 × 0.75/0.60 mm) at a fluorescence microscope (BZ‐X810 Keyence). Images were stitched together and quantified using the BZ‐X800 Analyzer Software.
2.8. mRNA Template Design, Synthesis and Validation
2.8.1. Linear Construct
To generate the mRNA construct, the open reading frame (ORF) of the respective gene (hTERT, GFP) was cloned into the multiple cloning site of a pMA‐RQ plasmid containing a CleanCap‐adjusted T7 promoter (TAATACGACTCACTATAAG), 5′ untranslated region (UTR) human α‐globin, 3′ UTR human β‐globin, segmented poly‐A tail (60A‐G‐60A) and a BspQI linearization site. Before modRNA production, the plasmid was sequenced, linearized, and then in vitro transcription (IVT) was performed to produce modRNA. For IVT production, a final concentration of 5 mM for each rATP, rCTP, rGTP, and m1Ψ, 4 mM CleanCap AG (N‐7113‐1 TriLink BioTechnologies), transcription buffer containing 40 mM Tris–HCl (pH 8), 10 mM dithiothreitol (DTT), 2 mM spermidine, 0.002% Triton X‐100, 16.5 mM magnesium acetate, 1 U/μL RNA inhibitor, 0.002 U/μL inorganic pyrophosphatase, and 8 U/μL T7 RNA polymerase were used. After 4 h incubation at 37°C, the IVT product was treated with 0.04 U/μL TURBO DNase (AM2239 Thermo Fisher Scientific) for 15 min at 37°C, filtered with Amicon Ultra‐4 (UFC801024 Merck), treated with 5 U/μL Antarctic Phosphatase (M0289L New England Biolabs) for 1 h at 37°C, and purified again with the Monarch RNA Cleanup Kit (T2050L New England Biolabs). The final modRNA product was verified with a 1% denaturing agarose gel electrophoresis.
2.8.2. Circular Construct
The hTERT ORF was cloned into the multiple cloning site, containing T7 promoter, group I self‐splicing intron, and IRES (internal ribosomal entry site) sequence. The splicing strategy was adapted as previously described (Wesselhoeft et al. 2018). To generate circular RNA, 400 ng of the sequenced plasmid was used for a PCR using a master mix consisting of 1× Phusion High Fidelity PCR Master Mix with GC Buffer (M0532 New England Biolabs), 1 μM primer pair (fw: TAATACGACTCACTATAGGGGGAGA; rev: GTTTAAACGGGCCCTCTAGACTCGAG) and 10% DMSO. The thermocycler protocol included the following steps: 98°C for 30 s; 30 cycles of 98°C for 5 s, 64°C for 10 s, 72°C for 2 min; 72°C for 5 min. After purification with QIAquick PCR Purification Kit (28,106 QIAGEN) the IVT protocol, as described above, was performed with an overnight (12–16 h) incubation. CleanCap AG was not used, and m1Ψ was substituted by rUTP. Purification was performed, followed by 20 U/μL RNase R (172010 Biozym) treatment for 30 min at 37°C and repeated purification with Monarch RNA Cleanup Kit (T2050L New England Biolabs). The final product was verified with sequencing, 1% denaturing agarose gel electrophoresis, and additionally reverse transcription followed by PCR with divergent primer pairs (primer list provided in Table 2).
TABLE 2.
List of genes and their corresponding primers in the qPCR and PCR.
| Gene name | Primer sequence (5′ ➔ 3′) |
|---|---|
| TBP |
Fw: CCACTCACAGACTCTCACAAC Rev.: CTGCGGTACAATCCCAGAACT |
| IFNA |
Fw: ACTCATACACCAGGTCACGC Rev.: CAGTGTAAAGGTGCACATGACG |
| IFNB |
Fw: AGTAGGCGACACTGTTCGTG Rev.: GCCTCCCATTCAATTGCCAC |
| IFIH1 |
Fw: GACTTGCCCTCTCCATCGTT Rev.: TCTGCAGCAGCAATCCGGT |
| RIGI |
Fw: AGAGCACTTGTGGACGCTTT Rev.: CTTCCTCTGCCTGTGTTCTGA |
| GFP |
Fw: CACAACGTCTATATCATGGC Rev.: TGTGATCGCGCTTCTC |
| hTERT |
Fw: TTCCTACGCTTCATGTGCCA Rev.: TACTCAGGGACACCTCGGAC |
| CDKN2A |
Fw: GGGAGCAGCATGGAGCCTT Rev.: TGCCCATCATCATGACCTGGATC |
| CDKN1A |
Fw: GCAGACCAGCATGACAGATTTC Rev.: GGATTAGGGCTTCCTCTTGGA |
| TP53 |
Fw: TGACACGCTTCCCTGGATTG Rev.: GCTCGACGCTAGGATCTGAC |
| GFP (divergent) |
Fw: CCGCGTGAGCAGTCTATTGA Rev.: AGAGGGCAGTGTGTCGTAAC |
| hTERT (divergent) |
Fw: CAGCAGGTGAACCAGCAC Rev.: TGTTTCTGGATTTGCAGGTG |
2.9. RNA Isolation and Quantitative PCR
PCLS RNA isolation was performed based on a previously described protocol (Niehof et al. 2017). For all the other samples in this study, RNA was isolated using QIAzol Lysis Reagent (79,306, QIAGEN), precipitated in 75% ethanol, and eluted in DNase/RNase‐free H2O. The RNA concentration was measured with Take3 Plates on a BioTek plate reader (Synergy HT). To remove all DNA contamination, 500–1000 ng RNA was treated with 0.2 U/μL RNase‐Free DNase (79,254 QIAGEN) for 30 min at 37°C, followed by reverse transcription using the Biozym cDNA synthesis kit (331470 Biozym). The real‐time PCR was performed on QuantStudio 7 Flex System (Thermo Fisher Scientific) with specific primer pairs (Table 2).
2.10. BrdU Incorporation Assay
Cell Proliferation ELISA, BrdU (11647229001, Merck) was used according to the manufacturer's instructions.
2.11. Immunostaining
Cultured cells were shortly washed with PBS and fixed with 4% paraformaldehyde (PFA) by incubating for 10 min, followed by three washes with PBS. Afterwards, cells were permeabilized with 0.1% Triton X‐100 for 10 min, followed by three washes with PBS. A blocking buffer consisting of 5% donkey serum in PBS (blocking buffer) was added for 30 min, followed by the primary antibodies (rabbit‐anti‐Ki67 1:250 ab16667 abcam, mouse‐anti‐pH 3 1:400 #9706 Cell Signaling Technology, rabbit‐anti‐γH2AX 1:2000 ab11174 abcam). Primary antibodies were diluted in blocking buffer and incubated at 4°C overnight. Cells were washed three times with PBS and then incubated with the 1:1000 diluted secondary antibodies (donkey‐anti‐rabbit Alexa Fluor 488 A21206 Invitrogen, donkey‐anti‐mouse Alexa Fluor 488 A21202 Invitrogen) and 1:1000 diluted Hoechst for 30 min. Next, the cells were washed three times with PBS. Images were acquired with Cytation 1 (Biotek) and analyzed with the BioTek Gen5 software.
2.12. Telomerase Repeat Amplification Protocol (TRAP)
The protocol was adapted from a previously described protocol (Herbert et al. 2006). 200,000 cells (based on live cell count) were harvested, washed with PBS, and centrifuged at 5000 g for 5 min at 4°C. Then the supernatant was removed, the cell pellet was snap frozen in liquid nitrogen, and stored at −70°C until further processing. To lyse the cells, ice‐cold NP‐40 lysis buffer was added to a final concentration of 1000 cells/μL and incubated for 30 min on ice. 2 μL cell lysate was added to 48 μL master mix. The master mix contained 50× primer mix (100 ng/mL each of ACX and NT primers along with 0.001 attomol/mL TSNT primers) and DY‐682 labeled TS primer (Table 3). Additionally, each sample had a respective heat‐inactivated (10 min at 85°C) cell lysate as a quality control for the TRAP assay. The TRAP PCR was run on a thermocycler with the following protocol: 25°C for 30 min; 95°C for 5 min; 24 cycles of 95°C for 30 s, 52°C for 30 s, 72°C for 40 s. Loading buffer was added to each sample before loading on a 10% acrylamide gel (19:1 Acryl: Bis acrylamide in Tris‐borate‐EDTA buffer) and the gel was run for around 3.5 h at 250 V. The gel was fixed for 15 min in fixative solution (0.5 M NaCl, 50% EtOH, 40 mM Sodium Acetate at pH 4.2) and then transferred to H2O right before scanning with the LICOR Odyssey Imaging System 9120.
TABLE 3.
List of primers used in TRAP assay.
| Primer | Sequence (5′➔ 3′) |
|---|---|
| TSNT | AATCCGTCGAGCAGAGTTAAAAGGCCGAGAAGCGAT |
| NT | ATCGCTTCTCGGCCTTTT |
| ACX | GCGCGGCTAACCCTAACCCTAACC |
| DY‐682 labeled TS | AATCCGTCGAGCAGAGTT |
2.13. Telomere Length Measurement
We employed an adapted version of Quantitative fluorescence in situ hybridization (qFISH) (O'Sullivan et al. 2005) and qPCR to measure telomere length as described previously (Jahn et al. 2025). The detailed method is available in the Supplementary file.
2.14. Transmission Electron Microscopy
Cell culture inserts (10482885 Thermo Fisher Scientific) with ATII cells were treated with Lipofectamine RNAiMAX (133778075 Thermo Fisher Scientific) and 500 ng modRNA in Opti‐MEM in a 2:1 vol:vol ratio. After 4 h, culture media (1.5× of the total transfection volume) was added onto the transfected cells. Cells were fixed using a mixture of 1.5% paraformaldehyde and 1.5% glutaraldehyde in 0.15 M HEPES buffer for at least 24 h. The samples were then embedded in epoxy resin as described previously (Brandenberger et al. 2018; Kling et al. 2017) and cut into ultrathin sections for transmission electron microscopic (TEM) analysis. Imaging was done with a Morgagni 268 microscope (FEI).
2.15. Statistics
Batch correction was performed for all experiments, besides growth curve, immunostaining, qFISH, and SA‐β‐gal analysis before statistical analysis. All data were analyzed with GraphPad Prism 8 software and are depicted as mean ± SD. Significant differences between two groups were calculated using an unpaired two‐tailed t‐test. A one‐way ANOVA was performed to assess differences among multiple treatment groups unless mentioned otherwise. A two‐way repeated measures ANOVA was performed to evaluate the effects of two factors (multiple treatment groups and time points). Dunnett's or Games‐Howell's multiple comparisons test was applied in all ANOVA tests; besides qFISH analysis, Sidak's multiple comparisons test was used. Homogeneity of variances was confirmed using the Brown‐Forsythe test in one‐way ANOVA and sphericity (Geisser–Greenhouse's epsilon) in two‐way repeated measures ANOVA. All data were tested positive for normality distribution using the Kolmogorov–Smirnov test. Normality in qFISH data was assumed based on a sample size of n ≥ 84, following the Central Limit Theorem.
3. Results
3.1. ModRNA hTERT Reactivates Telomerase Activity in MRC‐5 Cells
We first set out to produce modified hTERT mRNA (modRNA hTERT) that consists of CleanCap Reagent AG as a 5′ cap, 5′ and 3′ UTRs from the α‐globin and β‐globin genes, respectively, a segmented 120 bp poly (A) tail, and the hTERT ORF (Figure 1A,B). The modRNA consists of the modified nucleoside m1Ψ to reduce innate immune reaction and enhance stability and translational efficiency (Karikó et al. 2005; Nance and Meier 2021). In parallel to hTERT, modRNA GFP was produced as a control. Purified modRNAs for hTERT and GFP were loaded onto a denaturing gel to ensure the correct size of 3708 bp for modRNA hTERT and 1034 bp for modRNA GFP (Figure S1A). To validate the modRNA functionality, we chose MRC‐5, a fetal lung fibroblast cell line. 1 μg/mL modRNA GFP revealed a high transfection efficiency as GFP expression was detectable from 8 to 120 h post‐transfection (Figure S1B). Since the GFP expression peaked between 24 and 48 h, these time points were chosen for further experimental analysis (Figure 1C). Next, we demonstrated a dose‐dependent hTERT overexpression upon modRNA hTERT transfection (0.25–2 μg/mL) in the MRC‐5 cells, which possess negligible telomerase expression at baseline (Figure S1C). To prove that hTERT is translated into a catalytically active telomerase protein, the telomerase repeat amplification protocol (TRAP) was performed. Strikingly, even the lowest concentration of modRNA hTERT, 0.25 μg/mL, was sufficient to induce robust telomerase activity in MRC‐5 cells (Figure S1D,E). Although the hTERT modRNA expression levels started to decrease at 48 h (Figure S1C), the telomerase activity at 48 h was comparable to the 24 h time point.
FIGURE 1.
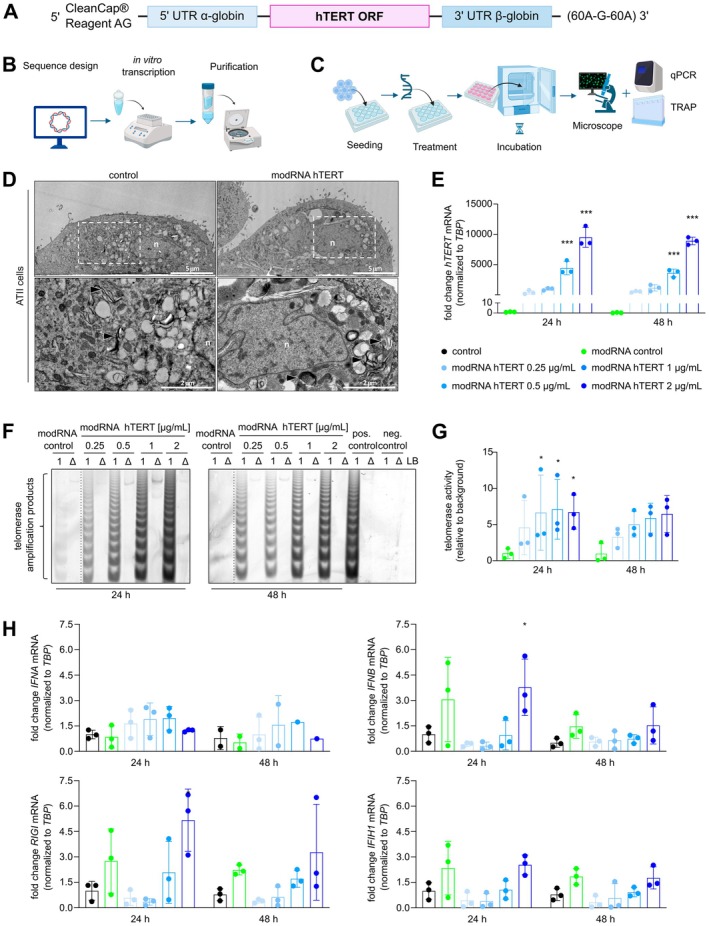
Dose‐dependent increase of hTERT mRNA and telomerase activity in vitro after modRNA hTERT transfection. (A) Schematic presentation of the modRNA hTERT template. (B) The modRNA production is divided into three main steps: the sequence design, then in vitro transcription (IVT) where the modRNA is generated and lastly the purification step. (C) Schematic describing experimental setup used to validate transfection efficiency of modRNA GFP and functionality of modRNA hTERT. (D) Transmission electron microscopy of ATII cells. Arrowheads indicate lamellar body like structures. Nucleus is depicted as n. (E) Fold change of hTERT mRNA expression in ATII cells transfected with modRNA hTERT in a concentration range of 0.25–2 μg/mL compared to modRNA GFP 2 μg/mL control (modRNA control) after 24 and 48 h (n = 3). (F) TRAP assay revealed reactivation of telomerase activity in ATII cells after 24 and 48 h in dose‐dependent effect. Positive (pos.) control = human induced pluripotent stem cells, negative (neg.) control = HUVEC cells, LB = lysis buffer, 1 = cell lysate, Δ = heat‐inactivated lysate. (G) Quantification of TRAP assays indicated a dose‐dependent increase of telomerase activity compared to modRNA control (n = 3). (H) No prolonged immune reaction was detected after 48 h modRNA treatment in ATII cells compared to untreated control (n = 3). *p < 0.05; ***p < 0.001; Two‐way ANOVA, Dunnett's multiple comparisons test.
In summary, we successfully validated our optimized modRNA hTERT design and observed a dose‐dependent overexpression on transcriptional and functional levels in MRC‐5. Thus, transient mRNA expression of modRNA hTERT, even at a minimal dosage, is capable of inducing telomerase activity.
3.2. ModRNA hTERT Treatment Robustly Induces Telomerase Activity in Primary Human Alveolar Type II Pneumocytes
To validate and assess the therapeutic potential of modRNA hTERT in a more relevant model for lung disease, we utilized human primary ATII cells. First, we confirmed the cellular identity by assessing the ultrastructure through transmission electron microscopy, highlighting the presence of lamellar bodies, specialized organelles that store and secrete SPs. Notably, transfection of ATII cells with modRNA hTERT did not influence cell morphology (Figure 1D).
The efficiency of modRNA GFP transfection in ATII cells was consistent with those observed in MRC‐5 cells (Figure S2A). Moreover, transfecting ATII cells with the modRNA hTERT again showed a dose‐dependent increase in hTERT expression and telomerase activity (Figure 1E–G). Since it is well established that “foreign” RNA can activate the innate immune system and induce an inflammatory response, we additionally evaluated the expression of several innate immune marker genes. Interferon α (IFNA) and interferon β (IFNB) are general markers for the activation of innate immunity, while retinoic acid‐inducible gene I (RIGI) and interferon induced with helicase C domain 1 (IFH1) are cytoplasmic sensors that detect foreign RNA and trigger the interferon signaling pathways. Only a high dose of modRNA GFP (modRNA control) and modRNA hTERT (2 μg/mL) compared to untransfected cells (control) led to a significant increase in IFNB and RIGI at 24 h, which quickly diminished at 48 h, indicating a transient and very brief immune reaction. IFNA and IFH1 expression remained unchanged (Figure 1H). Taken together, we validated that modRNA hTERT treatment can significantly induce hTERT expression and reactivate telomerase activity in primary human ATII cells without provoking a prolonged immune response.
3.3. ModRNA hTERT Enhances the Proliferation Capacity and Improves Cellular Health
Next, we investigated whether the modRNA‐mediated reactivation of hTERT expression and activity in ATII cells could lead to telomere elongation. To do so, we treated ATII cells with modRNA hTERT for 48 and 96 h, followed by telomere qFISH analysis to quantify the telomere lengths. Telomere length at these time points did not increase, suggesting that the given times were too short or repetitive treatment would be necessary to achieve telomere elongation (Figure S2B).
Therefore, modRNA hTERT was transfected in ATII cells at day 0 of the experiment (1× modRNA hTERT) and a subset of the 1× modRNA hTERT treated cells was transfected again for a second time at day 11 of the experiment (2× modRNA hTERT) followed by a quantitative growth curve analysis. This repetitive modRNA hTERT treatment resulted in a marked increase in population doubling level (PDL) by day 11, which continued to diverge significantly from the modRNA GFP treated control group until day 29 (fourth passage), indicating enhanced proliferative capacity after consecutive modRNA hTERT treatment (Figure 2A; Figure S2C). Increased PDL was accompanied by a decreasing trend in mRNA levels of senescence markers such as cyclin‐dependent kinase inhibitor 2A (CDKN2A), cyclin‐dependent kinase inhibitor 1A (CDKN1A) and tumor protein (TP53) (Figure S2D). To further substantiate these findings, we conducted a BrdU incorporation assay, which confirmed an increase in DNA synthesis, thereby validating the sustained proliferative activity driven by telomerase reactivation (Figure 2B). In line, immunofluorescence analysis of cell proliferation markers Ki67 and phospho‐histone H3 (pH3) showed a significant increase already after a single treatment of modRNA hTERT (Figure 2C). Conversely, γH2AX, a marker of DNA damage, was reduced after modRNA hTERT treatment. In contrast to the initial acute treatment, the long‐term and repetitive treatment led to a substantial elongation in telomere length, reinforcing the observed pro‐survival effects (Figure 2D,E). Collectively, our findings demonstrate that a double modRNA hTERT treatment is capable of enhancing proliferative capacity and reducing DNA damage, likely through telomere elongation, and ultimately improves the overall cellular fitness of ATII cells.
FIGURE 2.

Repetitive modRNA hTERT treatment increased proliferation capacity and enhanced cell viability in ATII cells. (A) Population doubling level (PDL) demonstrated an increase after single modRNA hTERT treatment (1×; at day 0), which was further increased upon double modRNA hTERT treatment (2×; at day 0 and 11) Arrowheads indicate the time point of modRNA hTERT treatment performed in ATII cells (n = 3). Geisser–Greenhouse's epsilon; Two‐way ANOVA; Dunnett's multiple comparison test. (B) BrdU assay indicated toward an increase in proliferation in ATII cells after modRNA hTERT treatment (n = 5). (C) Immunofluorescence staining of Ki67, pH3 and γH2AX, (green) revealed an increase in proliferation and a decrease in DNA damage after modRNA hTERT transfection in ATII cells (n = 3). (D) Representative images of qFISH staining in ATII cells after modRNA hTERT treatment. Brighter telomere spots (yellow, Tel Cy3), representing longer telomeres, were visible after 2× treatment with modRNA hTERT compared to 1× treatment and modRNA control group. Nucleus is depicted in blue (Hoechst). (E) Telomeric spots analysis revealed an increase of telomere length compared to modRNA control group (n ≥ 95 nuclei per group out of 3 biological replicates were imaged). Welch ANOVA; Games‐Howell's multiple comparisons test. *p < 0.05; **p < 0.01; ***p < 0.001; One‐way ANOVA or Two‐way ANOVA, Dunnett's multiple comparisons test.
3.4. Self‐Spliced Circular RNA hTERT as an Alternative to Prolong Telomerase Activity
We next sought to explore possibilities to extend the transient expression of modRNA hTERT by circularizing the in vitro transcription (IVT) product to increase stability, thereby prolonging its therapeutic effects.
To produce circular RNA hTERT, the hTERT ORF was cloned into a vector adjacent to an internal ribosomal entry site (IRES). IRES‐hTERT was then flanked by intron‐exon sequences that serve as group I introns which induce circularization through self‐splicing in the presence of the cofactors GTP and Mg2+ (Figure 3A) (Wesselhoeft et al. 2018). In order to obtain a pure circular construct and to prove superior stability, IVT products were treated with exonuclease RNase R, which cleaved linear RNAs thereby enriching circular RNAs (Figure S3A). Reverse transcription and PCR amplification with divergent primers was performed to validate the circular RNA hTERT. The PCR amplicon was sequenced to confirm the expected splice site and circularized sequence (Figure 3B; Figure S3B). Circular RNA GFP (circular RNA control) was transfected into HEK293 cells to assess transfection efficiency and protein translation potential (Figure S3C). After validating successful transfection and GFP expression in HEK293, the modRNA hTERT (linear RNA) was transfected in parallel with circular RNA hTERT in MRC‐5 cells. At 24 h post‐transfection, the levels of linear and circular hTERT transcripts were comparable. However, at 48 h post transfection the level of linear RNA hTERT transcript started to decline, while the circular RNA transcript levels remained stable (Figure 3C). These observations were reinforced by the TRAP assay (Figure 3D) where telomerase activity at 24 h was similar, but at 48 h the circular RNA hTERT treated group presented a significantly higher telomerase activity compared to the linear RNA hTERT (Figure 3E). Strikingly, in contrast to modRNA hTERT, circular RNA hTERT resulted in significant telomere elongation after 96 h of treatment (Figure 3F,G). To exclude potential cytotoxic effects of the linear and circular constructs, caspase 3/7 activity was measured at 24 and 48 h. By the 48 h time point, neither of the constructs induced apoptosis indicating no additive cellular stress 48 h post transfection (Figure S3D). To conclude, we provide pioneering results for the testing and validation of circularized hTERT RNA as an alternative to linear modRNA hTERT constructs to reactivate telomerase expression and activity.
FIGURE 3.
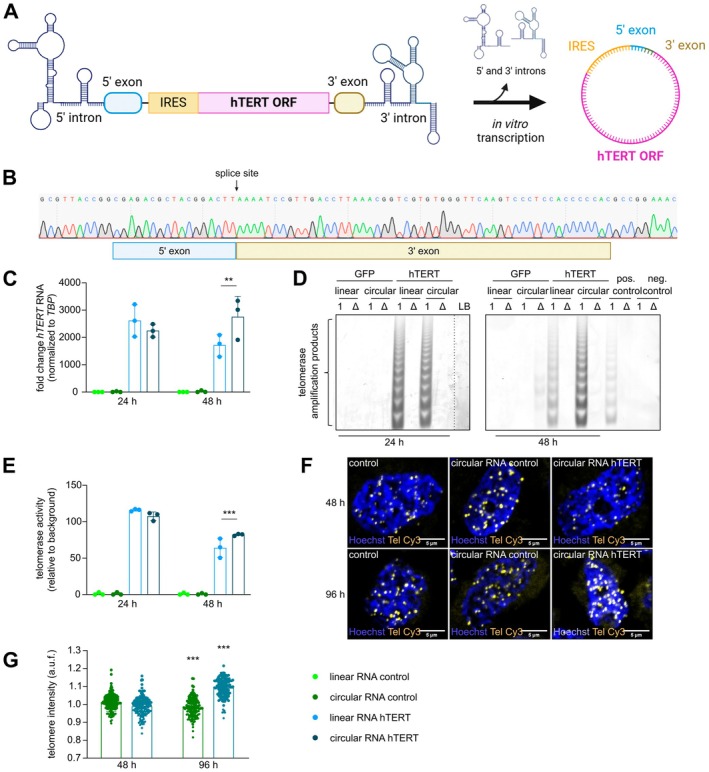
Circular RNA hTERT is more stable, resulting in a prolonged telomerase activity compared to linear RNA hTERT. (A) Schematic presentation of the circular RNA hTERT construct design. (B) Sanger sequencing of the hTERT PCR product, obtained after reverse transcription of circular RNA using divergent primers, revealed the circularization of the exons with expected splice site. (C) Transcript levels of hTERT comparing linear and circular RNA hTERT treatments after 24 and 48 h in MRC‐5 cells. (n = 3). (D) TRAP assay confirmed the telomerase activity level after transfection of both linear and circular hTERT RNA in MRC‐5 cells. Positive (pos.) control = HEK293 cells, negative (neg.) control = HUVEC cells, LB = lysis buffer, 1 = lysate, Δ = heat‐inactivated lysate. (E) Quantification of TRAP assays indicating an increase of telomerase activity after circular RNA in comparison to linear RNA at 48 h time point (n = 3). (F) Representative images of qFISH staining in MRC‐5 cells after circular RNA hTERT transfection. Brighter telomere spots (yellow, Tel Cy3), representing longer telomeres, were visible after 96 h treatment with circular RNA hTERT. Nucleus is depicted in blue (Hoechst). (G) Analysis of telomeric spots revealed an increase of telomere length in circular RNA hTERT treatment group at 96 h compared to circular RNA control group (n ≥ 132 nuclei per group out of 3 biological replicates were imaged). **p < 0.01; ***p < 0.001; One‐way ANOVA or Two‐way ANOVA, Dunnett's multiple comparisons test.
3.5. ModRNA hTERT Reduces Senescence in Human Precision‐Cut Lung Slices Derived From Patients With Idiopathic Pulmonary Fibrosis
Following up on the pro‐proliferative and pro‐survival effects conferred by the modRNA hTERT treatment on cells in vitro, we aimed to test its potential in a state‐of‐the‐art, patho‐physiologically and clinically relevant platform. We employed human PCLS derived from explanted lungs, which exhibited end‐stage PF (Table 1, Figure S4A). First, we assessed the expression timeline after modRNA hTERT transfection from a non‐fibrotic patient (Table 1) and two PF patients (Table 1), where a strong induction of hTERT mRNA was observed after 24 h (Figure 4A) and 48 h (Figure S4B). To evaluate the efficacy of modRNA hTERT therapy, PCLS from four PF patients diagnosed with either secondary fibrosis or IPF were employed. The presence of senescent alveolar epithelial cells in PF patients contributes to impaired regeneration, chronic inflammation, and fibrosis (Parimon et al. 2023). Strikingly, levels of senescence‐associated β‐galactosidase (SA‐β‐gal) were significantly lower after modRNA hTERT compared to modRNA control, which exhibited strong SA‐β‐gal staining at 96 h after transfection (Figure 4B,C). Subsequently, supernatants were collected to evaluate SASP‐related inflammatory responses and to analyze fibrosis markers in these PF‐derived PCLS after modRNA hTERT treatment. Notably, 96 h after modRNA hTERT treatment, the inflammatory response of IL6 and IL8, as well as the fibrotic TGFβ signaling and the fibrosis marker pro‐COL1A1 were significantly reduced (Figure 4D).
FIGURE 4.

Therapeutic treatment of linear and ciruclar RNA hTERT in PF patient‐derived PCLS leads to reduced inflammation and fibrosis. (A) Validation of overexpression of hTERT transcript level at 24 h after modRNA hTERT treatment in PCLS (n = 4). (B) Representative images of SA‐β‐gal stained PCLS (n = 4). SA‐β‐gal positive region in the PCLS tissues are in blue color. (C) Quantification of area occupied by SA‐β‐gal positive staining within the PCLS (N = 4). (D) Downregulation of the IL6, IL8, TGFβ and pro‐COL1A1 protein level relative to total expression after modRNA hTERT treatment (n = 4). (E) ModRNA hTERT (linear RNA hTERT) revealed a higher decrease of hTERT transcript compared to circular RNA hTERT from 24 to 48 h after transfection (n = 3). (F) Relative telomere length was increased in circular RNA hTERT treated PCLS at 96 h compared to circular RNA control. (G) LDH release is given as % of triton‐lysed control (set to 100%). No increase in LDH release was detected after linear RNA hTERT as well as circular RNA hTERT compared to modRNA GFP (linear RNA control) (n = 3). *p < 0.05; **p < 0.01; ***p < 0.001; unpaired t test; Two‐way ANOVA, Dunnett's or Sidak's multiple comparisons test.
PCLS from three individuals with PF, one with IPF and two with secondary fibrosis, were employed to compare the effects of linear versus circular RNA hTERT (Table 1). An approximately 50% higher degradation in the hTERT transcript levels was observed in the linear RNA hTERT from 24 to 48 h when compared to the circular RNA hTERT (Figure 4E). Telomere length measured via qPCR confirmed a superior telomere elongation efficacy in the circular RNA hTERT treated PCLS group at 96 h (Figure 4F). Notably, LDH levels released between the linear and circular RNA did not show any statistical difference, indicating optimal viability throughout the entire experimental period of 96 h (Figure 4G). Moreover, a broad panel of inflammatory cytokines was assessed (Figure S4C). Intriguingly, irrespective of hTERT delivery, immune markers such as IFNγ and TNFα were significantly upregulated at 24 and 48 h following circular RNA treatment but returned to baseline levels by 96 h. At this time point, circular RNA hTERT even led to a significant reduction in IL2, IL4, IL10, IL12p70, and IL13 levels. Additionally, circular RNA hTERT treatment resulted in a further decrease in TGFβ and pro‐COL1A1 compared to linear RNA hTERT (Figure S4D). Taken together, these findings confirmed that modRNA hTERT treatment in human end‐stage PF‐PCLS conferred anti‐senescence effects. Although linear modRNA hTERT treatment did not significantly increase telomere length in PF‐PCLS, even a single dose was sufficient to induce measurable therapeutic effects, underlining its therapeutic relevance. Circular RNA hTERT not only promoted telomere elongation but also further decreased pro‐fibrotic markers without inducing long‐term cytotoxicity. Our data suggest the general therapeutic potential of linear and circular hTERT against fibrotic interstitial lung diseases, including but not limited to IPF, as it effectively reduced inflammatory and fibrotic responses.
4. Discussion
We report a novel treatment option for PF by employing modRNA technology to transiently reactivate telomerase activity in ATII cells. Accounting for the most aggressive form of ILDs, IPF occurs with the highest frequency in ILD patients (Maher 2024; Rea et al. 2024). One risk factor for IPF is the premature shortening of telomeres in the lung (Van Batenburg et al. 2020). Povedano et al. already demonstrated in rodents that AAV9‐Tert could be utilized as a therapeutic option to ameliorate PF induced by bleomycin treatment in mice with short telomeres (Povedano et al. 2018). Of note, bleomycin induced fibrosis in mice is not progressive and therefore this model does not recapitulate the full complexity of PF. Liu et al. performed a conditional knockdown of TERT in ATII cells combined with a bleomycin stress treatment. Although they observed signs of lung fibrosis, there were no significant effects on telomere lengths in these mice (Liu et al. 2019). This might have occurred since laboratory bred mice have extremely long telomeres and recent efforts have been made to generate mice with telomeres comparable to humans, highlighting that most studies in in vivo mice models have limited translatability (Calado and Dumitriu 2013; Smoom et al. 2023). Moreover, AAV gene therapy strategies have some limitations. Firstly, the existence of neutralizing antibodies against naturally occurring AAV in up to 70% of the population represents a major clinical barrier (Wang, Gessler, et al. 2024). Secondly, due to the constitutive and long‐term overexpression provided by AAVs, its use for overexpressing hTERT in patients might lead to regulatory concerns due to the potential risk for tumorigenesis. By contrast, due to their non‐integrative and transient expression profile, modRNAs appear to be an ideal vector for hTERT delivery in patients (Sahin et al. 2014). Additionally, the modRNA production using IVT method is scalable and cost efficient thus making it an affordable therapy compared to AAV gene therapy (Chaudhary et al. 2021). One groundbreaking example of this modRNA technology is the COVID‐19 vaccine development which highlights the clinical readiness of this drug class (Warne et al. 2023).
In our study, both in vitro (MRC‐5 cells as wells as primary ATII cells) and ex vivo human end‐stage PF‐PCLS tissue were successfully transfected with modRNA hTERT leading to a transient expression of the functionally active telomerase protein. The catalytically active protein increased the proliferative capacity, decreased DNA damage while significantly increasing the telomere length in the treated ATII cells. This observation is in line with a previous study by Ramunas et al., where they applied modRNA hTERT in vitro in a non‐disease setting to increase the proliferation capacity in lung fibroblasts and primary skeletal muscle myoblasts (Ramunas et al. 2015). It is important to acknowledge that these general benefits could be the result of other telomerase‐related functions aside from telomerase‐induced telomere elongation. Apart from its canonical role, hTERT is also known to possess non‐telomeric functions in the mitochondria (Chatterjee et al. 2021). It has been implicated that hTERT localizes within the mitochondria, thereby reducing oxidative stress, improving mitochondrial membrane potential, and protecting mitochondrial DNA. All these beneficial effects lead to an overall enhanced mitochondrial function and cell survival (Denham 2023). Thus, there might be other non‐canonical functions of hTERT due to which primary lung cells can benefit after hTERT treatment. This aspect should be further investigated in the context of transient modRNA hTERT therapy for lung fibrosis in more complex pre‐clinical in vivo models with a specific focus on ATII cell repair and regeneration using functional assays such as the release of SPs or other bronchioloalveolar stem cells (BASCs) which are crucial for lung repair (Wang, Wang, et al. 2024). In this study, telomere elongation was directly associated with modRNA hTERT treatment. Biologically, alternative lengthening of telomeres (ALT) is a telomerase‐independent mechanism of telomere maintenance. However, it is a rare phenomenon restricted to specific telomerase‐negative tumor cells (Cesare and Reddel 2010), and to date, there are no reports that ALT can be pharmacologically targeted to maintain telomere length in healthy cells (Heaphy et al. 2011).
To overcome the various limitations of available mouse models and in vitro cell lines for investigation of PF, we utilized PCLS, a state‐of‐the‐art organotypic model of the human lung. Particularly, we used viable diseased lung tissue from patients suffering from end‐stage PF. These PCLS retain the complex pathogenesis of PF by preserving active cellular and mechanical function of various cell types in their native lung architecture (Hesse et al. 2022; Koziol‐White et al. 2024; Neuhaus et al. 2018, 2017). This model enables ex vivo investigation of integrated cellular response to various treatment options, including modRNA hTERT, to support pre‐clinical tests and toxicity studies. Our data reveals clear downregulation of SA‐β‐gal activity within the tissue as well as SASP‐related inflammatory responses highlighting the therapeutic potential of modRNA hTERT in mitigating inflammation and fibrosis in PF.
It is commonly observed that isolated ATII cells undergo spontaneous differentiation toward ATI cells, which leads to a reduction in their proliferative capacity (Strunz et al. 2020). In elderly patients, the lung ATII cells also exhibit advanced stages of senescence in lung fibrosis (Parimon et al. 2023). In this context, we showed that modRNA hTERT treatment has the ability to rescue ATII cells from the senescent state, since we observed the modest reduction of the senescence markers CDKN2A, CDKN1A, and TP53 after modRNA hTERT treatment at later passages in vitro and a significant reduction in β‐gal staining in human PCLS tissue ex vivo. In line with this finding, we could demonstrate that modRNA hTERT treatment was able to alleviate the secretion of SASP pro‐inflammatory markers IL6 and IL8 along with reduced levels of TGFβ and pro‐COL1A1, which are key mediators for fibrosis progression. This highlights the potential of modRNA hTERT to prevent senescence, leading to a decrease in inflammation as well as mediators of fibrosis.
The human innate immune system has several pattern recognition receptors that detect foreign RNA and activate downstream signaling cascades to trigger an immune reaction, like TLR7, RIGI, or IFH1 (Fang et al. 2022; Nance and Meier 2021). We demonstrated that after treatment with modRNA hTERT in vitro, there was no prolonged upregulation of immune markers, such as RIGI, IFH1, IFNA, and IFNB. In contrast, circular RNA treatment in PF‐PCLS induced a transient increase in certain immune markers at 24 or 48 h, potentially due to the IRES element in the construct. However, consistent with our in vitro results, cytokine levels returned to baseline by 96 h, indicating a self‐limiting immune response. These findings are in alignment with the immune safety analysis of the FDA‐approved modRNA vaccine (BNT162b2) against COVID‐19, where systemic reactions due to the vaccine were resolved within one to two days (Polack et al. 2020). Finally, in an attempt to prolong the transient expression of hTERT, we produced a circular, exonuclease‐resistant form of RNA, which provided higher stability and a longer window of modRNA hTERT therapeutic efficacy. Although the observed fold changes were relatively modest, these levels were sufficient to drive measurable biological effects, as evidenced by significant telomere elongation. Recently, Qin et al. demonstrated the successful application of circular RNA hTERT as a treatment for progeria, where circular RNA hTERT was superior to the linear RNA hTERT in overexpressing telomerase, reducing senescence and inflammation in diseased endothelial cells, further supporting the feasibility of this approach (Qin et al. 2025). Another very recent study presented a strategy to synthesize and stabilize the telomerase RNA component (TERC), termed engineered TERC (eTERC) and further demonstrated how a single eTERC treatment can effectively prevent telomere‐induced senescence in cells exhibiting telomere biology disorders (Nagpal and Agarwal 2025). These findings demonstrate that functional reconstitution of a stabilized synthetic TERC long non‐coding RNA, in addition to the presence of hTERT, can also act as a potential therapy to enhance replicative capacity in human stem cells. Irrespective of how telomere elongation is achieved, tumorigenesis remains a major concern. As a result, AAV‐mediated hTERT therapies, which are associated with persistent and constitutive overexpression, are generally not favored for clinical translation, despite evidence that TERT alone is insufficient to transform normal cells. Moreover, long‐term overexpression of TERT via AAV has been shown to be beneficial without an associated increase in cancer (Bär et al. 2016; Bernardes de Jesus et al. 2012). Therefore, our approach via modRNA hTERT offers a transient and self‐limiting profile. On one hand, it substantially reduces the risk of cancer, but on the other hand, it creates the need for higher and/or repeated dosing, which is accompanied by potential off‐targeting events. A strong immune reaction can be detrimental to translational efficiency and even lead to toxicity (Shi et al. 2024). Taking these into account, we acknowledge that any RNA‐therapy‐based intervention targeting telomere maintenance should be carefully evaluated for oncogenic and immunogenic potential, particularly in long‐term or repeated dosing regimens. Future in vivo studies in pre‐clinical models with extensive follow‐up will be essential to ascertain the safety and immunogenicity profiles of modRNA hTERT therapy.
While these findings underline the feasibility of hTERT delivery for the treatment of PF, it is important to acknowledge that this approach may not be effective in all PF patients. For example, in FPF, mutations can exist in other telomere‐related genes such as TERC, DJC1, TIN2, and RTEK1, aside from hTERT (Peljto et al. 2023). Subsequently, for a certain subset of FPF patients, this underlying mutation might lead to a varied level of effectiveness after the hTERT‐based RNA therapy. However, due to the heterogeneous nature of PF and based on our study, hTERT therapy still holds promise for a diverse group of patients suffering from PF.
This study demonstrates the biological efficacy and feasibility of hTERT in the physiologically relevant PCLS platform. However, the limited ex vivo viability of PCLS restricts long‐term observation and precludes the evaluation of repeated and long‐term modRNA hTERT administration within this model. Additionally, due to the required agarose embedding to preserve tissue architecture, certain functional assays such as direct telomerase activity measurement cannot be performed in PCLS. These technical constraints underscore the need for complementary models, such as 3D lung organoid cultures or preclinical in vivo models, where direct assessment of functional telomerase reactivation is possible in addition to studies that can be conducted for extended time points.
Moreover, we acknowledge the importance of delivery to minimize off‐target effects. One of the most common and investigated methods to deliver RNA relies on lipid nanoparticles (LNPs) (Webb et al. 2022). A key limitation in developing pulmonary therapeutics remains the efficient and specific delivery of cargo to ATII cells. Due to the deep location of ATII cells within the alveolar niche and the presence of mucosal and immune barriers, systemically administered LNPs often exhibit poor localization to this cell type (Bai et al. 2024). Furthermore, conventional LNP formulations tend to accumulate in the liver rather than in the lung. To this end, inhalable LNP formulations offer a more suitable administration route to directly deliver into the lung (Zhang et al. 2023). Recent advances have focused on incorporating optimized LNP formulations (Bai et al. 2024; Cheng et al. 2020) or branched‐tail ionizable lipids (Petersen et al. 2024), which bias biodistribution toward the lung alveoli. Additionally, antibody‐conjugated nanoparticles targeting ATII‐specific markers, such as surfactant protein C (SPC), have demonstrated selective binding and internalization, improving cellular specificity (Kang et al. 2025; Wu et al. 2015). Complementary to these biochemical approaches, localized delivery methods such as bronchoscopic administration or intratracheal instillation provide direct access to the alveolar space, ensuring precise regional deposition and bypassing systemic clearance. However, further validation in this context is necessary to fully realize its translational potential.
In summary, our findings support the therapeutic potential of repeated modRNA‐mediated hTERT therapy for PF, and highlight circular RNA hTERT as a promising alternative approach, by transient yet significant elevation of telomerase expression and activity which is sufficient to elongate telomeres and improve cellular health in primary lung cells and organotypic tissue from end‐stage PF, including IPF.
Author Contributions
J.L.Y. designed and performed the experiments with subsequent analysis. K.G. and J.L.Y. performed the human lung tissue experiments. D.L., M.J., S.T., and L.O. contributed to the modified RNA and circular RNA methods. C.B. (Christina Brandenberger) performed electron microscopy experiments. M.J. (Maria Jordan) contributed to RNA isolation from PCLS. K.G., P.Z., C.W., C.H., and K.S. provided human lung tissues. K.G. and C.H. performed LDH and ELISA assays in PCLS. C.B. conceived the original idea of the study and supported manuscript preparation. T.T., K.S., S.T., C.B. (Christina Brandenberger), S.C., and C.B. received funding. S.C. was involved in training, supervision, and experimental design of this project. J.L.Y., C.B., and S.C. wrote the manuscript. C.B. (Christina Brandenberger), C.H., K.S., S.T., T.T., and C.B. revised the manuscript. All authors reviewed and approved the final version of the manuscript.
Conflicts of Interest
T.T. is founder and CSO/CMO of Cardior Pharmaceuticals GmbH, a wholly‐owned subsidiary of Novo Nordisk A/S Europe (outside of this paper). T.T. and C.B. filed and licensed patents on the therapeutic use of RNAs (outside of this paper). C.B. has filed and licensed patents on the therapeutic use of AAV9‐mediated delivery of telomerase (outside of this paper). The remaining authors declare no conflicts of interest.
Supporting information
Figure S1: Dose‐dependent increase of hTERT mRNA and telomerase activity in vitro after modRNA hTERT transfection in MRC‐5 cells. (A) Validation of the hTERT and GFP modRNA product size after IVT in 1.5% denatured gel. ModRNA GFP is generated by substituting hTERT ORF with the GFP coding sequence. (B) Images showing the successful transfection of modRNA GFP 2 μg/mL and stable GFP expression for until 48 h and a significant decline at 120 h in MRC‐5 cells. (C) Fold change of hTERT mRNA expression after transfection with modRNA hTERT in a concentration range of 0.25–2 μg/mL compared to modRNA control (modRNA GFP 2 μg/mL) after 24 and 48 h in MRC‐5 cells (n = 3). (D) TRAP assay revealed telomerase activity after 24 h modRNA hTERT transfection in MRC‐5 cells. Positive (pos.) control = HEK293 cells, negative (neg.) control = HUVEC cells, LB = lysis buffer, 1 = lysate, Δ = heat‐inactivated lysate. (E) Quantification of TRAP assays in MRC‐5 (n = 3). *p < 0.05; ***p < 0.001; Two‐way ANOVA, Dunnett's multiple comparisons test.
Figure 2: Repetitive treatment of modRNA hTERT increase proliferation capacity and decrease expression of senescence‐related marker in ATII cells. (A) Images showing successful transfection of modRNA GFP 2 μg/mL in ATII cells as observed by GFP expression in 24 an 48 h post transfection. (B) Telomere qFISH analysis demonstrated no telomere elongation after 48 and 96 h modRNA hTERT treatment in ATII cells (n ≥ 84 nuclei per group out of 3 biological replicates were imaged) modRNA GFP is used as the modRNA control. (C) Representative brightfield images on the day of passaging in ATII cells of modRNA control compared to 2× treatment modRNA hTERT. (D) A decreasing trend of senescence‐related markers (CDKN2A, CDKN1A, TP53) was detected after two doses of modRNA hTERT treatment (n = 4). *p < 0.05; One‐way ANOVA or Two‐way ANOVA, Dunnett's or Sidak's multiple comparisons test.
Figure 3: Validation of circular RNA. (A) Validation of the size of produced linear and circular RNA products using IVT encoding for hTERT or GFP. To prove superior stability of circular RNA the linear and circular RNAs were treated with RNase R and loaded on 1.5% denatured gel. Circular GFP is generated by substituting hTERT ORF with the GFP coding sequence. (B) Validation of circularization by performing PCR with divergent primer of reverse transcribed circular RNA. Loaded on 1.5% agarose gel. (C) Representative images confirming successful transfection of circular RNA GFP 2 μg/mL in HEK293 cells. (D) No significant increase observed in caspase 3/7 activity between linear and circular hTERT RNA products at 24 h. Caspase 3/7 activity returned back to baseline at 48 h indicating no adverse effect on cell viability (n = 3). Two‐way ANOVA, Dunnett's multiple comparisons test.
Figure 4: Overexpression of hTERT mRNA after modRNA hTERT transfection in PCLS. (A) Schematic presentation of PCLS (precision‐cut lung slices) preparation. PF: pulmonary fibrosis. (B) Fold change of hTERT mRNA expression after 48 h of transfection with modRNA hTERT (1 μg/mL) compared to modRNA GFP (modRNA control) (1 μg/mL) in PCLS at 24 h (n = 2). (C) Protein expression levels of cytokines (IFNγ, IL1β, IL2, IL4, IL6, IL8, IL10, IL12p70, IL13 and TNFα) relative to total expression after RNA hTERT treatment compared to modRNA GFP (linear RNA control) (n = 3). (D) TGFβ and pro‐COL1A1 protein levels at 96 h relative to total expression after RNA hTERT treatment compared to modRNA GFP (linear RNA control) (n = 3). *p < 0.05; **p < 0.01; ***p < 0.001; One‐way ANOVA or Two‐way ANOVA, Dunnett's multiple comparisons test.
Acknowledgements
We thank A. Gietz, I. Riedel, N. Lehmann, and E. Westerheide from the Institute of Molecular and Translational Therapeutic Strategies (IMTTS), Hannover Medical School (MHH) for excellent technical support. We appreciate the support from the Central Laser Microscopy Unit, MHH. Additionally, we thank Taihua Yang, PhD, from the Department of Liver Surgery, Renji Hospital, School of Medicine, Shanghai Jiao Tong University in China for kindly providing the plasmid for circular RNA generation used in this study. Schematic figures were created with Biorender.com. Open Access funding enabled and organized by Projekt DEAL.
Funding: This work was supported by Deutsche Forschungsgemeinschaft DFG (BR 5347/4‐1 to C.B. (Christina Brandenberger); CH 3292/1‐4 to S.C. and BA 5631/2‐4 and BA 5631/5‐1 to C.B.), the Institute of Biomedical Translation Lower Saxony (Project LuFex, to T.T.) and by the Fraunhofer Cluster of Excellence Immune‐Mediated Diseases (CIMD) RNA Therapeutics Platform (to C.B., K.S., and S.T.) This work was also supported as a Fraunhofer Flagship project «RNAuto» (to C.B. and S.T.).
Contributor Information
Shambhabi Chatterjee, Email: chatterjee.shambhabi@mh-hannover.de.
Christian Bär, Email: baer.christian@mh-hannover.de.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.
References
- Adegunsoye, A. , Newton C. A., Oldham J. M., et al. 2023. “Telomere Length Associates With Chronological Age and Mortality Across Racially Diverse Pulmonary Fibrosis Cohorts.” Nature Communications 14, no. 1: 1–13. 10.1038/s41467-023-37193-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alder, J. K. , and Armanios M.. 2022. “Telomere‐Mediated Lung Disease.” Physiological Reviews 102, no. 4: 1703–1720. 10.1152/PHYSREV.00046.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alder, J. K. , Barkauskas C. E., Limjunyawong N., et al. 2015. “Telomere Dysfunction Causes Alveolar Stem Cell Failure.” Proceedings of the National Academy of Sciences of the United States of America 112, no. 16: 5099–5104. 10.1073/pnas.1504780112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bai, X. , Chen Q., Li F., et al. 2024. “Optimized Inhaled LNP Formulation for Enhanced Treatment of Idiopathic Pulmonary Fibrosis via mRNA‐Mediated Antibody Therapy.” Nature Communications 15, no. 1: 1–18. 10.1038/s41467-024-51056-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bär, C. , and Blasco M. A.. 2016. “Telomeres and Telomerase as Therapeutic Targets to Prevent and Treat Age‐Related Diseases.” F1000Research 5, no. 89. 10.12688/f1000research.7020.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bär, C. , Povedano J. M., Serrano R., et al. 2016. “Telomerase Gene Therapy Rescues Telomere Length, Bone Marrow Aplasia, and Survival in Mice With Aplastic Anemia.” Blood 127, no. 14: 1770–1779. 10.1182/blood-2015-08-667485. [DOI] [PubMed] [Google Scholar]
- Bernardes de Jesus, B. , Vera E., Schneeberger K., et al. 2012. “Telomerase Gene Therapy in Adult and Old Mice Delays Aging and Increases Longevity Without Increasing Cancer.” EMBO Molecular Medicine 4, no. 8: 691–704. 10.1002/EMMM.201200245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brandenberger, C. , Kling K. M., Vital M., and Mühlfeld C.. 2018. “The Role of Pulmonary and Systemic Immunosenescence in Acute Lung Injury.” Aging and Disease 9, no. 4: 553–565. 10.14336/AD.2017.0902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calado, R. T. , and Dumitriu B.. 2013. “Telomere Dynamics in Mice and Humans.” Seminars in Hematology 50, no. 2: 165–174. 10.1053/J.SEMINHEMATOL.2013.03.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cesare, A. J. , and Reddel R. R.. 2010. “Alternative Lengthening of Telomeres: Models, Mechanisms and Implications.” Nature Reviews Genetics 11, no. 5: 319–330. 10.1038/nrg2763. [DOI] [PubMed] [Google Scholar]
- Chatterjee, S. , Hofer T., Costa A., et al. 2021. “Telomerase Therapy Attenuates Cardiotoxic Effects of Doxorubicin.” Molecular Therapy 29, no. 4: 1395–1410. 10.1016/j.ymthe.2020.12.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaudhary, N. , Weissman D., and Whitehead K. A.. 2021. “mRNA Vaccines for Infectious Diseases: Principles, Delivery and Clinical Translation.” Nature Reviews Drug Discovery 20: 817–838. 10.1038/s41573-021-00283-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng, Q. , Wei T., Farbiak L., Johnson L. T., Dilliard S. A., and Siegwart D. J.. 2020. “Selective Organ Targeting (SORT) Nanoparticles for Tissue‐Specific mRNA Delivery and CRISPR–Cas Gene Editing.” Nature Nanotechnology 15, no. 4: 313–320. 10.1038/s41565-020-0669-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cilli, A. , and Uzer F.. 2023. “Disease Progression in Idiopathic Pulmonary Fibrosis Under Anti‐Fibrotic Treatment.” Sarcoidosis, Vasculitis, and Diffuse Lung Diseases 40, no. 3: e2023034. 10.36141/SVDLD.V40I3.14048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cottin, V. , Hirani N. A., Hotchkin D. L., et al. 2018. “Presentation, Diagnosis and Clinical Course of the Spectrum of Progressive‐Fibrosing Interstitial Lung Diseases.” European Respiratory Review 27, no. 150: 180076. 10.1183/16000617.0076-2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cronkhite, J. T. , Xing C., Raghu G., et al. 2008. “Telomere Shortening in Familial and Sporadic Pulmonary Fibrosis.” American Journal of Respiratory and Critical Care Medicine 178, no. 7: 729–737. 10.1164/rccm.200804-550oc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denham, J. 2023. “Canonical and Extra‐Telomeric Functions of Telomerase: Implications for Healthy Ageing Conferred by Endurance Training.” Aging Cell 22, no. 6: e13836. 10.1111/ACEL.13836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duckworth, A. , Gibbons M. A., Allen R. J., et al. 2021. “Telomere Length and Risk of Idiopathic Pulmonary Fibrosis and Chronic Obstructive Pulmonary Disease: A Mendelian Randomisation Study.” Lancet Respiratory Medicine 9, no. 3: 285–294. 10.1016/S2213-2600(20)30364-7. [DOI] [PubMed] [Google Scholar]
- Esposito, D. B. , Lanes S., Donneyong M., et al. 2015. “Idiopathic Pulmonary Fibrosis in United States Automated Claims. Incidence, Prevalence, and Algorithm Validation.” American Journal of Respiratory and Critical Care Medicine 192, no. 10: 1200–1207. 10.1164/RCCM.201504-0818OC. [DOI] [PubMed] [Google Scholar]
- Fan, W. , Gui B., Zhou X., Li L., and Chen H.. 2024. “A Narrative Review on Lung Injury: Mechanisms, Biomarkers, and Monitoring.” Critical Care 28, no. 1: 352. 10.1186/S13054-024-05149-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fang, E. , Liu X., Li M., et al. 2022. “Advances in COVID‐19 mRNA Vaccine Development.” Signal Transduction and Targeted Therapy 7, no. 1: 1–31. 10.1038/s41392-022-00950-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heaphy, C. M. , Subhawong A. P., Hong S. M., et al. 2011. “Prevalence of the Alternative Lengthening of Telomeres Telomere Maintenance Mechanism in Human Cancer Subtypes.” American Journal of Pathology 179, no. 4: 1608–1615. 10.1016/J.AJPATH.2011.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herbert, B.‐S. , Hochreiter A. E., Wright W. E., and Shay J. W.. 2006. “Nonradioactive Detection of Telomerase Activity Using the Telomeric Repeat Amplification Protocol.” Nature Protocols 1: 1583–1590. 10.1038/nprot.2006.239. [DOI] [PubMed] [Google Scholar]
- Hesse, C. , Beneke V., Konzok S., et al. 2022. “Nintedanib Modulates Type III Collagen Turnover in Viable Precision‐Cut Lung Slices From Bleomycin‐Treated Rats and Patients With Pulmonary Fibrosis.” Respiratory Research 23, no. 1: 201. 10.1186/s12931-022-02116-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirsch, M. S. , Hildebrand C. B., Geltinger F., et al. 2024. “Senescence in Alveolar Epithelial Type II Cells Promotes Acute Lung Injury and Impairs Regeneration.” American Journal of Respiratory Cell and Molecular Biology 71, no. 6: 688–701. 10.1165/RCMB.2024-0054OC. [DOI] [PubMed] [Google Scholar]
- Jahn, K. , Chatterjee S., Sinke C., et al. 2025. “An Incidental Finding During a Brain Plasticity Study: Substantial Telomere Length Shortening After COVID‐19 Lockdown in the Older Population.” GeroScience 47, no. 3: 5225–5234. 10.1007/S11357-025-01602-Z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang, Z. Y. , Xuan N. X., Zhou Q. C., et al. 2025. “Targeting Alveolar Epithelial Cells With Lipid Micelle‐Encapsulated Necroptosis Inhibitors to Alleviate Acute Lung Injury.” Communications Biology 8, no. 1: 1–16. 10.1038/S42003-025-08010-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karikó, K. , Buckstein M., Ni H., and Weissman D.. 2005. “Suppression of RNA Recognition by Toll‐Like Receptors: The Impact of Nucleoside Modification and the Evolutionary Origin of RNA.” Immunity 23, no. 2: 165–175. 10.1016/J.IMMUNI.2005.06.008. [DOI] [PubMed] [Google Scholar]
- Khor, Y. H. , Farooqi M., Hambly N., et al. 2023. “Patient Characteristics and Survival for Progressive Pulmonary Fibrosis Using Different Definitions.” American Journal of Respiratory and Critical Care Medicine 207, no. 1: 102–105. 10.1164/RCCM.202205-0910LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kling, K. M. , Lopez‐Rodriguez E., Pfarrer C., Mühlfeld C., and Brandenberger C.. 2017. “Aging Exacerbates Acute Lung Injury‐Induced Changes of the Air‐Blood Barrier, Lung Function, and Inflammation in the Mouse.” American Journal of Physiology. Lung Cellular and Molecular Physiology 312, no. 1: L1–L12. 10.1152/AJPLUNG.00347.2016. [DOI] [PubMed] [Google Scholar]
- Koziol‐White, C. , Gebski E., Cao G., and Panettieri R. A.. 2024. “Precision Cut Lung Slices: An Integrated Ex Vivo Model for Studying Lung Physiology, Pharmacology, Disease Pathogenesis and Drug Discovery.” Respiratory Research 25, no. 1: 231. 10.1186/s12931-024-02855-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu, T. , De Los G., Santos F., et al. 2019. “Telomerase Reverse Transcriptase Ameliorates Lung Fibrosis by Protecting Alveolar Epithelial Cells Against Senescence.” Journal of Biological Chemistry 294, no. 22: 8861–8871. 10.1074/jbc.RA118.006615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maher, T. M. 2024. “Interstitial Lung Disease: A Review.” JAMA 331, no. 19: 1655–1665. 10.1001/JAMA.2024.3669. [DOI] [PubMed] [Google Scholar]
- McDonough, J. E. , Martens D. S., Tanabe N., et al. 2018. “A Role for Telomere Length and Chromosomal Damage in Idiopathic Pulmonary Fibrosis.” Respiratory Research 19, no. 1: 1–8. 10.1186/S12931-018-0838-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagpal, N. , and Agarwal S.. 2025. “Extension of Replicative Lifespan by Synthetic Engineered Telomerase RNA in Patient Induced Pluripotent Stem Cells.” Nature Biomedical Engineering: 1–15. 10.1038/S41551-025-01429-1. [DOI] [PubMed] [Google Scholar]
- Nance, K. D. , and Meier J. L.. 2021. “Modifications in an Emergency: The Role of N1‐Methylpseudouridine in COVID‐19 Vaccines.” ACS Central Science 7, no. 5: 748–756. 10.1021/acscentsci.1c00197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neuhaus, V. , Danov O., Konzok S., et al. 2018. “Assessment of the Cytotoxic and Immunomodulatory Effects of Substances in Human Precision‐Cut Lung Slices.” Journal of Visualized Experiments 2018, no. 135: 57042. 10.3791/57042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neuhaus, V. , Schaudien D., Golovina T., et al. 2017. “Assessment of Long‐Term Cultivated Human Precision‐Cut Lung Slices as an Ex Vivo System for Evaluation of Chronic Cytotoxicity and Functionality.” Journal of Occupational Medicine and Toxicology 12, no. 1: 1–8. 10.1186/S12995-017-0158-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newton, C. A. , Batra K., Torrealba J., et al. 2016. “Telomere‐Related Lung Fibrosis Is Diagnostically Heterogeneous but Uniformly Progressive.” European Respiratory Journal 48, no. 6: 1710–1720. 10.1183/13993003.00308-2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niehof, M. , Hildebrandt T., Danov O., et al. 2017. “RNA Isolation From Precision‐Cut Lung Slices (PCLS) From Different Species.” BMC Research Notes 10, no. 1: 121. 10.1186/S13104-017-2447-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Sullivan, J. N. , Finley J. C., Risques R. A., Shen W. T., Gollahon K. A., and Rabinovitch P. S.. 2005. “Quantitative Fluorescence In Situ Hybridization (QFISH) of Telomere Lengths in Tissue and Cells.” Current Protocols in Cytometry 33, no. 1: 12.6.1–12.6.15. 10.1002/0471142956.CY1206S33. [DOI] [PubMed] [Google Scholar]
- Parimon, T. , Chen P., Stripp B. R., et al. 2023. “Senescence of Alveolar Epithelial Progenitor Cells: A Critical Driver of Lung Fibrosis.” American Journal of Physiology‐Cell Physiology 325, no. 2: 483–495. 10.1152/AJPCELL.00239.2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peljto, A. L. , Blumhagen R. Z., Walts A. D., et al. 2023. “Idiopathic Pulmonary Fibrosis Is Associated With Common Genetic Variants and Limited Rare Variants.” American Journal of Respiratory and Critical Care Medicine 207, no. 9: 1194–1202. 10.1164/RCCM.202207-1331OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penaud‐Budloo, M. , Le Guiner C., Nowrouzi A., et al. 2008. “Adeno‐Associated Virus Vector Genomes Persist as Episomal Chromatin in Primate Muscle.” Journal of Virology 82, no. 16: 7875–7885. 10.1128/JVI.00649-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petersen, D. M. S. , Weiss R. M., Hajj K. A., et al. 2024. “Branched‐Tail Lipid Nanoparticles for Intravenous mRNA Delivery to Lung Immune, Endothelial, and Alveolar Cells in Mice.” Advanced Healthcare Materials 13, no. 22: 2400225. 10.1002/ADHM.202400225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piñeiro‐Hermida, S. , Martínez P., Bosso G., et al. 2022. “Consequences of Telomere Dysfunction in Fibroblasts, Club and Basal Cells for Lung Fibrosis Development.” Nature Communications 13, no. 1: 5656. 10.1038/S41467-022-32771-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Podolanczuk, A. J. , Thomson C. C., Remy‐Jardin M., et al. 2023. “Idiopathic Pulmonary Fibrosis: State of the Art for 2023.” European Respiratory Journal 61, no. 4: 2200957. 10.1183/13993003.00957-2022. [DOI] [PubMed] [Google Scholar]
- Polack, F. P. , Thomas S. J., Kitchin N., et al. 2020. “Safety and Efficacy of the BNT162b2 mRNA Covid‐19 Vaccine.” New England Journal of Medicine 383, no. 27: 2603–2615. 10.1056/NEJMOA2034577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Povedano, J. M. , Martinez P., Flores J. M., Mulero F., and Blasco M. A.. 2015. “Mice With Pulmonary Fibrosis Driven by Telomere Dysfunction.” Cell Reports 12, no. 2: 286–299. 10.1016/j.celrep.2015.06.028. [DOI] [PubMed] [Google Scholar]
- Povedano, J. M. , Martinez P., Serrano R., et al. 2018. “Therapeutic Effects of Telomerase in Mice With Pulmonary Fibrosis Induced by Damage to the Lungs and Short Telomeres.” eLife 7: e31299. 10.7554/eLife.31299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qin, W. , Castillo K. D., Li H., et al. 2025. “Circular RNA Telomerase Reverses Endothelial Senescence in Progeria.” Aging Cell 24, no. 6: e70021. 10.1111/ACEL.70021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raghu, G. , Chen S. Y., Yeh W. S., et al. 2014. “Idiopathic Pulmonary Fibrosis in US Medicare Beneficiaries Aged 65 Years and Older: Incidence, Prevalence, and Survival, 2001‐11.” Lancet. Respiratory Medicine 2, no. 7: 566–572. 10.1016/S2213-2600(14)70101-8. [DOI] [PubMed] [Google Scholar]
- Ramunas, J. , Yakubov E., Brady J. J., et al. 2015. “Transient Delivery of Modified mRNA Encoding TERT Rapidly Extends Telomeres in Human Cells.” FASEB Journal 29, no. 5: 1930–1939. 10.1096/FJ.14-259531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rea, G. , Lieto R., Bruzzese D., et al. 2024. “The Density Histograms‐Derived Computerized Integrated Index (CII) Predicts Mortality in Idiopathic Pulmonary Fibrosis.” Scientific Reports 14, no. 1: 1–9. 10.1038/s41598-024-77328-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahin, U. , Karikó K., and Türeci Ö.. 2014. “mRNA‐Based Therapeutics — Developing a New Class of Drugs.” Nature Reviews Drug Discovery 13, no. 10: 759–780. 10.1038/nrd4278. [DOI] [PubMed] [Google Scholar]
- Shi, Y. , Shi M., Wang Y., and You J.. 2024. “Progress and Prospects of mRNA‐Based Drugs in Pre‐Clinical and Clinical Applications.” Signal Transduction and Targeted Therapy 9, no. 1: 1–43. 10.1038/s41392-024-02002-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smoom, R. , May C. L., Ortiz V., et al. 2023. “Telomouse—A Mouse Model With Human‐Length Telomeres Generated by a Single Amino Acid Change in RTEL1.” Nature Communications 14, no. 1: 1–18. 10.1038/s41467-023-42534-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spagnolo, P. , Kropski J. A., Jones M. G., et al. 2021. “Idiopathic Pulmonary Fibrosis: Disease Mechanisms and Drug Development.” Pharmacology & Therapeutics 222: 107798. 10.1016/J.PHARMTHERA.2020.107798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strand, M. J. , Sprunger D., Cosgrove G. P., et al. 2014. “Pulmonary Function and Survival in Idiopathic vs Secondary Usual Interstitial Pneumonia.” Chest 146, no. 3: 775–785. 10.1378/CHEST.13-2388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strunz, M. , Simon L. M., Ansari M., et al. 2020. “Alveolar Regeneration Through a Krt8+ Transitional Stem Cell State That Persists in Human Lung Fibrosis.” Nature Communications 11, no. 1: 1–20. 10.1038/s41467-020-17358-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Switalla, S. , Lauenstein L., Prenzler F., et al. 2010. “Natural Innate Cytokine Response to Immunomodulators and Adjuvants in Human Precision‐Cut Lung Slices.” Toxicology and Applied Pharmacology 246, no. 3: 107–115. 10.1016/J.TAAP.2010.04.010. [DOI] [PubMed] [Google Scholar]
- Van Batenburg, A. A. , Kazemier K. M., Van Oosterhout M. F. M., et al. 2020. “From Organ to Cell: Multi‐Level Telomere Length Assessment in Patients With Idiopathic Pulmonary Fibrosis.” PLoS One 15, no. 1: e0226785. 10.1371/JOURNAL.PONE.0226785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Batenburg, A. A. , Kazemier K. M., Van Oosterhout M. F. M., et al. 2021. “Telomere Shortening and DNA Damage in Culprit Cells of Different Types of Progressive Fibrosing Interstitial Lung Disease.” ERJ Open Research 7, no. 2: 691–2020. 10.1183/23120541.00691-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, J. H. , Gessler D. J., Zhan W., Gallagher T. L., and Gao G.. 2024. “Adeno‐Associated Virus as a Delivery Vector for Gene Therapy of Human Diseases.” Signal Transduction and Targeted Therapy 9, no. 1: 1–33. 10.1038/s41392-024-01780-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, Y. , Wang L., Ma S., Cheng L., and Yu G.. 2024. “Repair and Regeneration of the Alveolar Epithelium in Lung Injury.” FASEB Journal 38, no. 8: e23612. 10.1096/FJ.202400088R. [DOI] [PubMed] [Google Scholar]
- Warne, N. , Ruesch M., Siwik P., et al. 2023. “Delivering 3 Billion Doses of Comirnaty in 2021.” Nature Biotechnology 41, no. 2: 183–188. 10.1038/s41587-022-01643-1. [DOI] [PubMed] [Google Scholar]
- Webb, C. , Ip S., Bathula N. V., et al. 2022. “Current Status and Future Perspectives on MRNA Drug Manufacturing.” Molecular Pharmaceutics 19, no. 4: 1047–1058. 10.1021/ACS.MOLPHARMACEUT.2C00010. [DOI] [PubMed] [Google Scholar]
- Wesselhoeft, R. A. , Kowalski P. S., and Anderson D. G.. 2018. “Engineering Circular RNA for Potent and Stable Translation in Eukaryotic Cells.” Nature Communications 9: 2629. 10.1038/s41467-018-05096-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wijsenbeek, M. , and Cottin V.. 2020. “Spectrum of Fibrotic Lung Diseases.” New England Journal of Medicine 383, no. 10: 958–968. 10.1056/NEJMRA2005230. [DOI] [PubMed] [Google Scholar]
- Wu, Y. , Ma J., Woods P. S., et al. 2015. “Selective Targeting of Alveolar Type II Respiratory Epithelial Cells by Anti‐Surfactant Protein‐C Antibody‐Conjugated Lipoplexes.” Journal of Controlled Release 203: 140–149. 10.1016/J.JCONREL.2015.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamazaki, R. , Nishiyama O., Yoshikawa K., Tohda Y., and Matsumoto H.. 2022. “Outcome of Patients Who Were Incidentally Diagnosed With Idiopathic Pulmonary Fibrosis: How Early in the Disease Should We Identify Patients?” Respiratory Medicine 201: 106933. 10.1016/J.RMED.2022.106933. [DOI] [PubMed] [Google Scholar]
- Yazicioglu, T. , Mühlfeld C., Autilio C., et al. 2020. “Aging Impairs Alveolar Epithelial Type II Cell Function in Acute Lung Injury.” American Journal of Physiology. Lung Cellular and Molecular Physiology 319, no. 5: L755–L769. 10.1152/AJPLUNG.00093.2020. [DOI] [PubMed] [Google Scholar]
- Zhang, M. , Lu H., Xie L., Liu X., Cun D., and Yang M.. 2023. “Inhaled RNA Drugs to Treat Lung Diseases: Disease‐Related Cells and Nano–Bio Interactions.” Advanced Drug Delivery Reviews 203: 115144. 10.1016/J.ADDR.2023.115144. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1: Dose‐dependent increase of hTERT mRNA and telomerase activity in vitro after modRNA hTERT transfection in MRC‐5 cells. (A) Validation of the hTERT and GFP modRNA product size after IVT in 1.5% denatured gel. ModRNA GFP is generated by substituting hTERT ORF with the GFP coding sequence. (B) Images showing the successful transfection of modRNA GFP 2 μg/mL and stable GFP expression for until 48 h and a significant decline at 120 h in MRC‐5 cells. (C) Fold change of hTERT mRNA expression after transfection with modRNA hTERT in a concentration range of 0.25–2 μg/mL compared to modRNA control (modRNA GFP 2 μg/mL) after 24 and 48 h in MRC‐5 cells (n = 3). (D) TRAP assay revealed telomerase activity after 24 h modRNA hTERT transfection in MRC‐5 cells. Positive (pos.) control = HEK293 cells, negative (neg.) control = HUVEC cells, LB = lysis buffer, 1 = lysate, Δ = heat‐inactivated lysate. (E) Quantification of TRAP assays in MRC‐5 (n = 3). *p < 0.05; ***p < 0.001; Two‐way ANOVA, Dunnett's multiple comparisons test.
Figure 2: Repetitive treatment of modRNA hTERT increase proliferation capacity and decrease expression of senescence‐related marker in ATII cells. (A) Images showing successful transfection of modRNA GFP 2 μg/mL in ATII cells as observed by GFP expression in 24 an 48 h post transfection. (B) Telomere qFISH analysis demonstrated no telomere elongation after 48 and 96 h modRNA hTERT treatment in ATII cells (n ≥ 84 nuclei per group out of 3 biological replicates were imaged) modRNA GFP is used as the modRNA control. (C) Representative brightfield images on the day of passaging in ATII cells of modRNA control compared to 2× treatment modRNA hTERT. (D) A decreasing trend of senescence‐related markers (CDKN2A, CDKN1A, TP53) was detected after two doses of modRNA hTERT treatment (n = 4). *p < 0.05; One‐way ANOVA or Two‐way ANOVA, Dunnett's or Sidak's multiple comparisons test.
Figure 3: Validation of circular RNA. (A) Validation of the size of produced linear and circular RNA products using IVT encoding for hTERT or GFP. To prove superior stability of circular RNA the linear and circular RNAs were treated with RNase R and loaded on 1.5% denatured gel. Circular GFP is generated by substituting hTERT ORF with the GFP coding sequence. (B) Validation of circularization by performing PCR with divergent primer of reverse transcribed circular RNA. Loaded on 1.5% agarose gel. (C) Representative images confirming successful transfection of circular RNA GFP 2 μg/mL in HEK293 cells. (D) No significant increase observed in caspase 3/7 activity between linear and circular hTERT RNA products at 24 h. Caspase 3/7 activity returned back to baseline at 48 h indicating no adverse effect on cell viability (n = 3). Two‐way ANOVA, Dunnett's multiple comparisons test.
Figure 4: Overexpression of hTERT mRNA after modRNA hTERT transfection in PCLS. (A) Schematic presentation of PCLS (precision‐cut lung slices) preparation. PF: pulmonary fibrosis. (B) Fold change of hTERT mRNA expression after 48 h of transfection with modRNA hTERT (1 μg/mL) compared to modRNA GFP (modRNA control) (1 μg/mL) in PCLS at 24 h (n = 2). (C) Protein expression levels of cytokines (IFNγ, IL1β, IL2, IL4, IL6, IL8, IL10, IL12p70, IL13 and TNFα) relative to total expression after RNA hTERT treatment compared to modRNA GFP (linear RNA control) (n = 3). (D) TGFβ and pro‐COL1A1 protein levels at 96 h relative to total expression after RNA hTERT treatment compared to modRNA GFP (linear RNA control) (n = 3). *p < 0.05; **p < 0.01; ***p < 0.001; One‐way ANOVA or Two‐way ANOVA, Dunnett's multiple comparisons test.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.


