Abstract
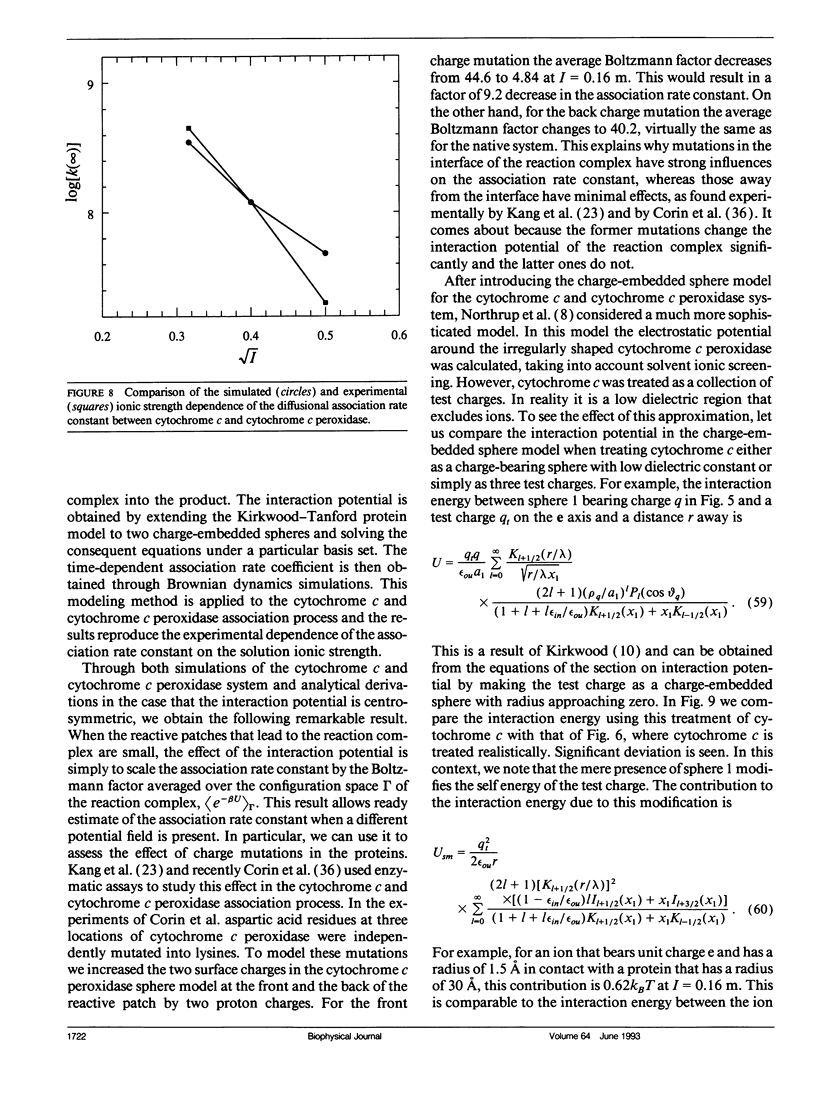
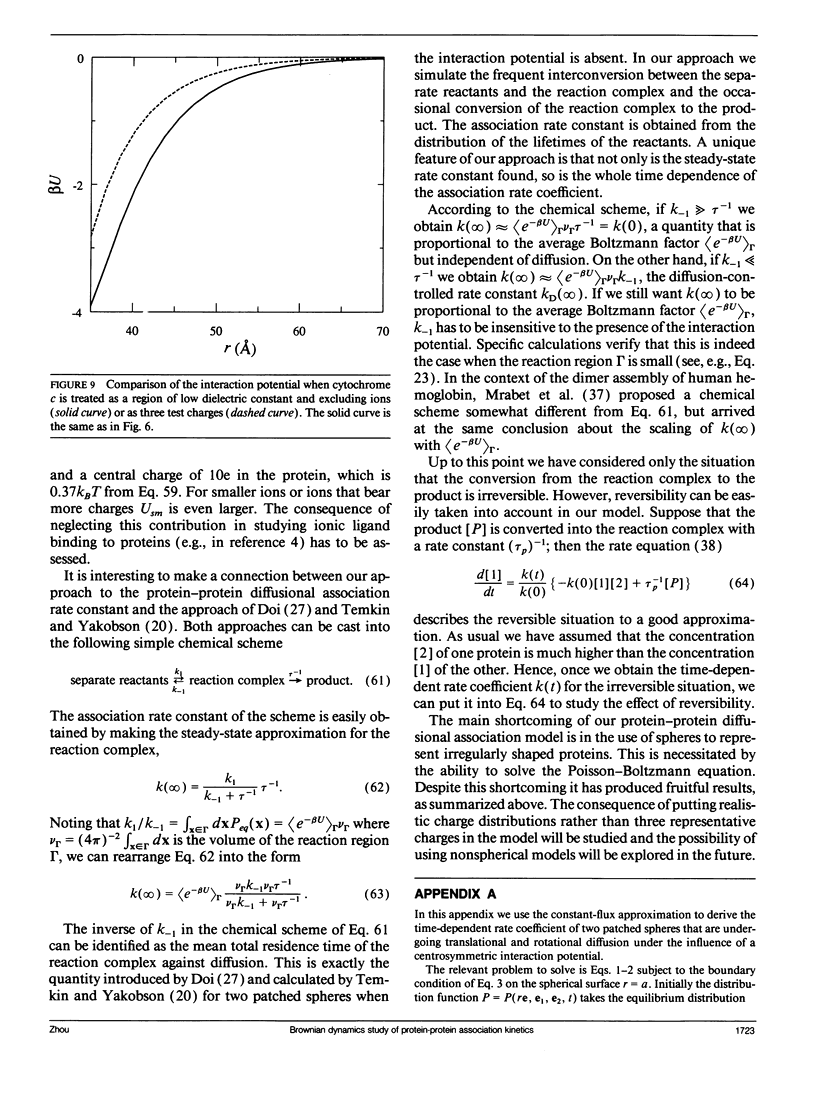
A unified model is presented for protein-protein association processes that are under the influences of electrostatic interaction and diffusion (e.g., protein oligomerization, enzyme catalysis, electron and energy transfer). The proteins are modeled as spheres that bear point charges and undergo translational and rotational Brownian motion. Before association can occur the two spheres have to be aligned properly to form a reaction complex via diffusion. The reaction complex can either go on to form the product or it can dissociate into the separate reactants through diffusion. The electrostatic interaction, like diffusion, influences every step except the one that brings the reaction complex into the product. The interaction potential is obtained by extending the Kirkwood-Tanford protein model (Tanford, C., and J. G. Kirkwood. 1957. J. Am. Chem. Soc. 79:5333-5339) to two charge-embedded spheres and solving the consequent equations under a particular basis set. The time-dependent association rate coefficient is then obtained through Brownian dynamics simulations according an algorithm developed earlier (Zhou, H.-X. 1990. J. Phys. Chem. 94:8794-8800). This method is applied to a model system of the cytochrome c and cytochrome c peroxidase association process and the results confirm the experimental dependence of the association rate constant on the solution ionic strength. An important conclusion drawn from this study is that when the product is formed by very specific alignment of the reactants, as is often the case, the effect of the interaction potential is simply to scale the association rate constant by a Boltzmann factor. This explains why mutations in the interface of the reaction complex have strong influences on the association rate constant whereas those away from the interface have minimal effects. It comes about because the former mutations change the interaction potential of the reaction complex significantly and the latter ones do not.
Full text
PDF









Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berg O. G. Orientation constraints in diffusion-limited macromolecular association. The role of surface diffusion as a rate-enhancing mechanism. Biophys J. 1985 Jan;47(1):1–14. doi: 10.1016/S0006-3495(85)83870-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corin A. F., McLendon G., Zhang Q., Hake R. A., Falvo J., Lu K. S., Ciccarelli R. B., Holzschu D. Effects of surface amino acid replacements in cytochrome c peroxidase on complex formation with cytochrome c. Biochemistry. 1991 Dec 10;30(49):11585–11595. doi: 10.1021/bi00113a014. [DOI] [PubMed] [Google Scholar]
- Kang C. H., Brautigan D. L., Osheroff N., Margoliash E. Definitaion of cytochrome c binding domains by chemical modification. Reaction of carboxydinitrophenyl- and trinitrophenyl-cytochromes c with baker's yeast cytochrome c peroxidase. J Biol Chem. 1978 Sep 25;253(18):6502–6510. [PubMed] [Google Scholar]
- Matthew J. B. Electrostatic effects in proteins. Annu Rev Biophys Biophys Chem. 1985;14:387–417. doi: 10.1146/annurev.bb.14.060185.002131. [DOI] [PubMed] [Google Scholar]
- Mrabet N. T., McDonald M. J., Turci S., Sarkar R., Szabo A., Bunn H. F. Electrostatic attraction governs the dimer assembly of human hemoglobin. J Biol Chem. 1986 Apr 15;261(11):5222–5228. [PubMed] [Google Scholar]
- Northrup S. H., Erickson H. P. Kinetics of protein-protein association explained by Brownian dynamics computer simulation. Proc Natl Acad Sci U S A. 1992 Apr 15;89(8):3338–3342. doi: 10.1073/pnas.89.8.3338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shire S. J., Hanania G. I., Gurd F. R. Electrostatic effects in myoglobin. Hydrogen ion equilibria in sperm whale ferrimyoglobin. Biochemistry. 1974 Jul 2;13(14):2967–2974. doi: 10.1021/bi00711a028. [DOI] [PubMed] [Google Scholar]
- Shoup D., Lipari G., Szabo A. Diffusion-controlled bimolecular reaction rates. The effect of rotational diffusion and orientation constraints. Biophys J. 1981 Dec;36(3):697–714. doi: 10.1016/S0006-3495(81)84759-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warwicker J., Watson H. C. Calculation of the electric potential in the active site cleft due to alpha-helix dipoles. J Mol Biol. 1982 Jun 5;157(4):671–679. doi: 10.1016/0022-2836(82)90505-8. [DOI] [PubMed] [Google Scholar]


